Quantifying Water Quality Improvements through Use of Precision Herbicide Application Technologies in a Dry-Tropical, Furrow-Irrigated Cropping System
Abstract
:1. Introduction
2. Materials and Methods
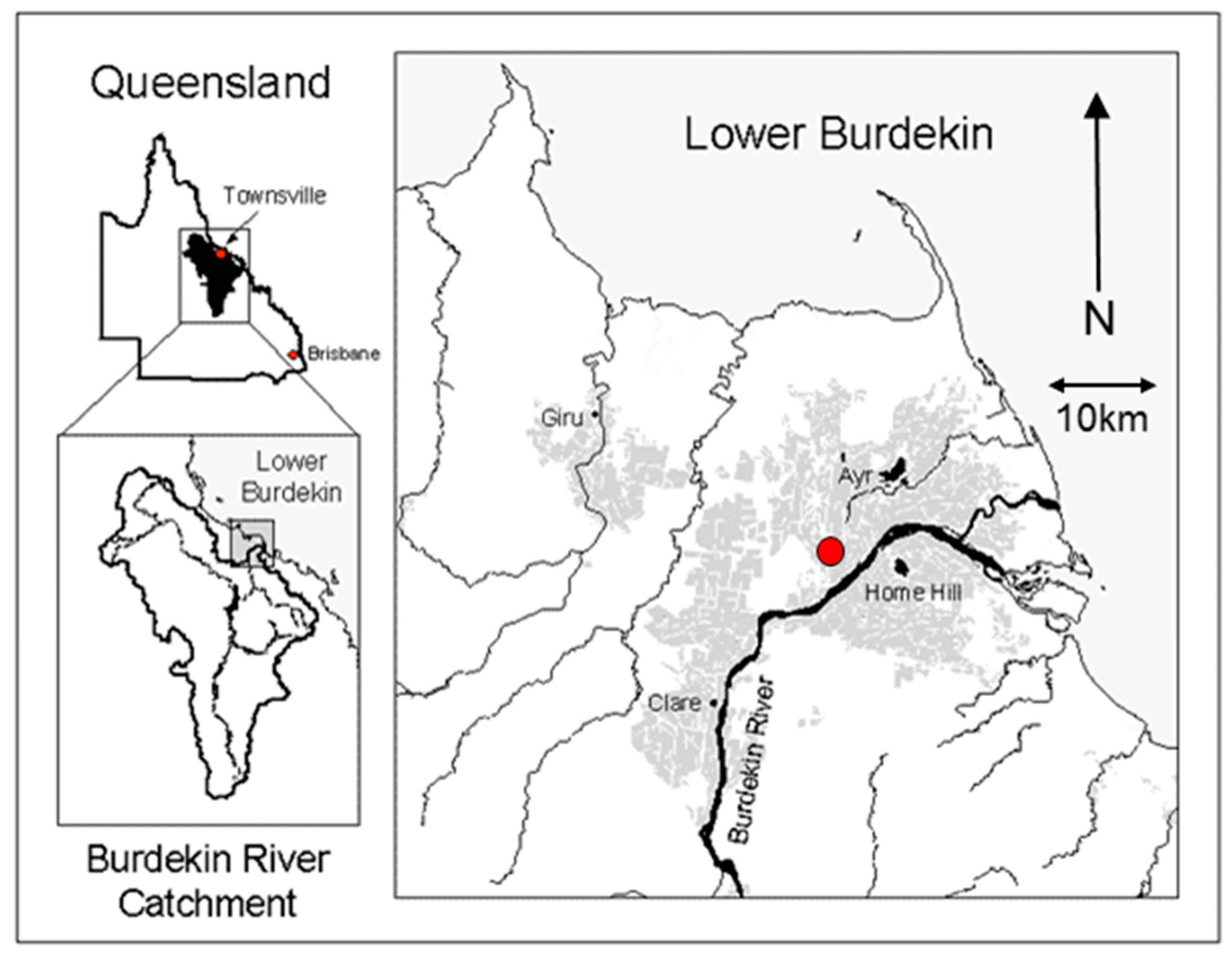
2.1. Study Area
2.2. Trial and Experimental Design
2.3. Irrigation Measurements
2.4. Herbicide Analyses
2.5. Statistical Analyses
3. Results
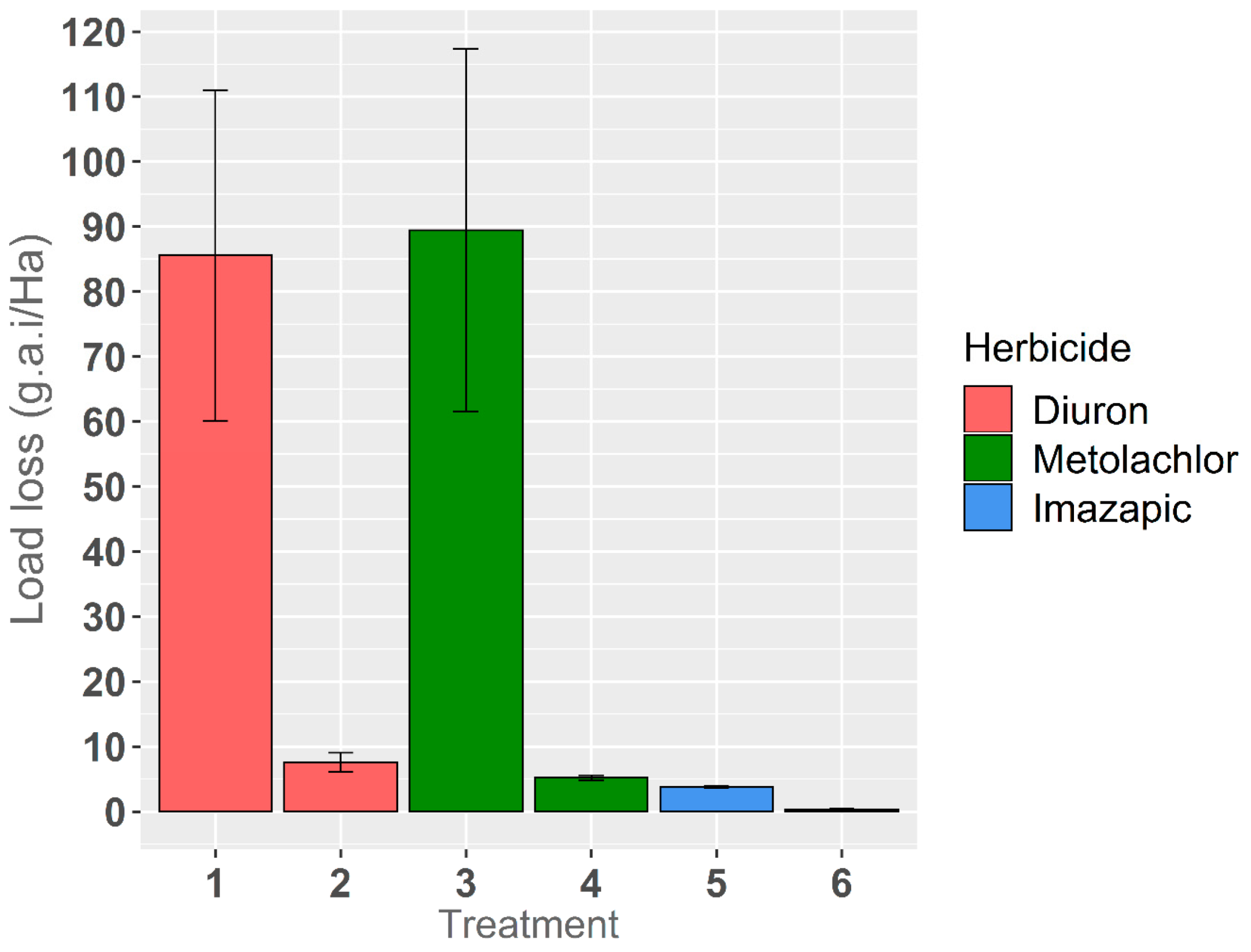
3.1. Irrigation Runoff and Losses of Applied Herbicides
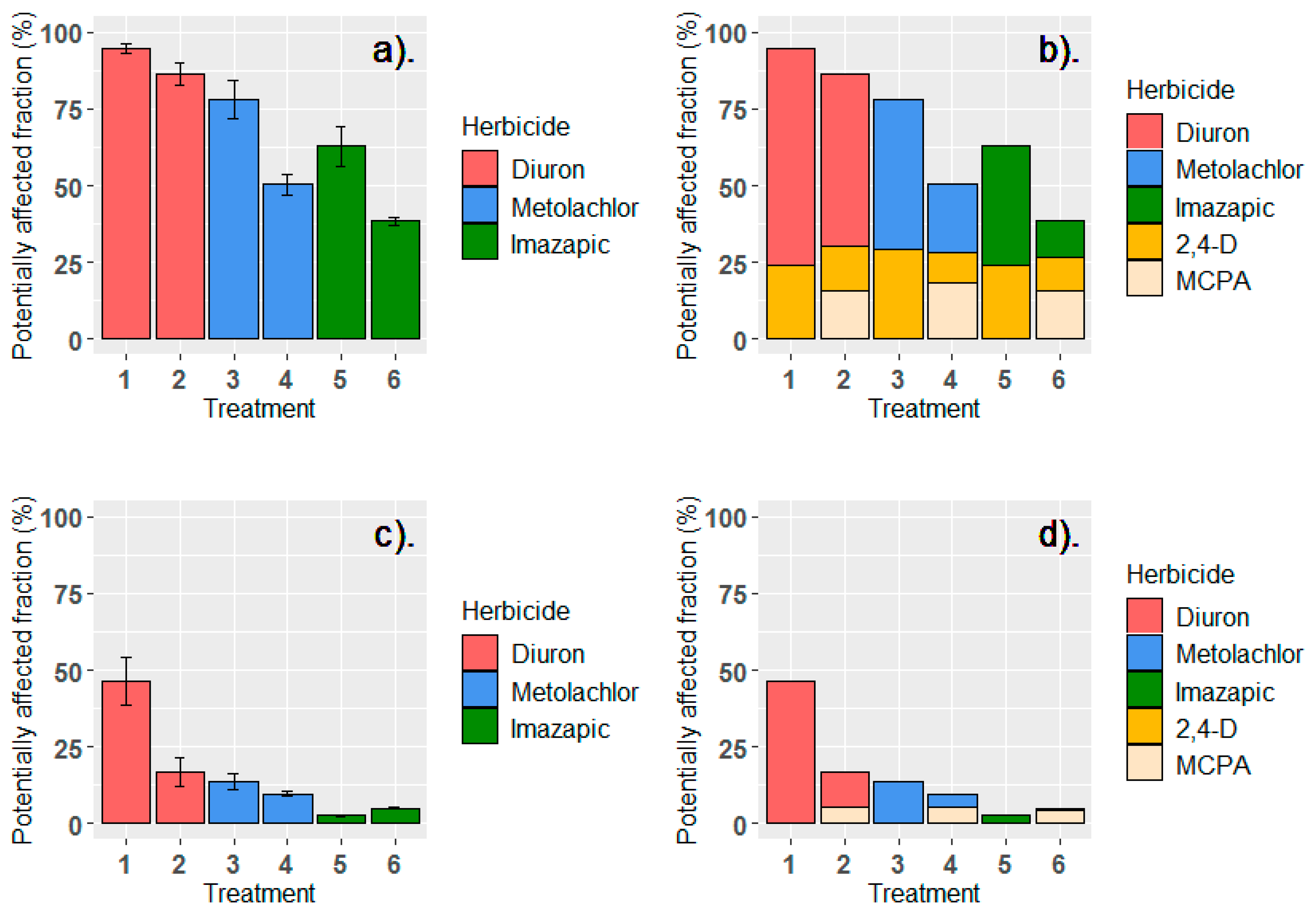
3.2. Toxic Load Losses of Applied Herbicide Mixture
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, S.E.; Brodie, J.E.; Bainbridge, Z.T.; Rohde, K.; Davis, A.; Masters, B.; Maughan, M.; Devlin, M.; Mueller, J.; Schaffelke, B. Herbicides: A new threat to the Great Barrier Reef. Environ. Pollut. 2009, 157, 2470–2484. [Google Scholar] [CrossRef]
- Brodie, J.E.; Kroon, F.J.; Schaffelke, B.; Wolanski, E.; Lewis, S.E.; Devlin, M.J.; Bainbridge, Z.T.; Waterhouse, J.; Davis, A.M. Terrestrial pollutant runoff to the Great Barrier Reef: An update of issues, priorities and management responses. Mar. Pollut. Bull. 2012, 65, 81–100. [Google Scholar] [CrossRef]
- Waterhouse, J.; Brodie, J.; Tracey, D.; Smith, R.; Vandergragt, M.; Collier, C.; Petus, C.; Baird, M.; Kroon, F.; Mann, R.; et al. Scientific Consensus Statement 2017: A Synthesis of the Science of Land-Based Water Quality Impacts on the Great Barrier Reef, Chapter 3: The Risk from Anthropogenic Pollutants to Great Barrier Reef Coastal and Marine Ecosystems; State of Queensland: Brisbane, Australia, 2017.
- Müller, J.F.; Duquesne, S.; Ng, J.; Shaw, G.R.; Krrishnamohan, K.; Manonmanii, K.; Hodge, M.; Eaglesham, G.K. Pesticides in sediments from Queensland irrigation channels and drains. Mar. Pollut. Bull. 2000, 41, 294–301. [Google Scholar] [CrossRef]
- Stork, P.R.; Bennett, F.R.; Bell, M.J. The environmental fate of diuron under a conventional production regime in a sugarcane farm during the plant cane phase. Pest Manag. Sci. 2008, 64, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.; Lewis, S.E.; Bainbridge, Z.T.; Brodie, J.; Shannon, E. Pesticide residues in waterways of the lower Burdekin region: Challenges in ecotoxicological interpretation of monitoring data. Aust. J. Ecotoxicol. 2008, 14, 89–108. [Google Scholar]
- Davis, A.M.; Thorburn, P.J.; Lewis, S.E.; Bainbridge, Z.T.; Attard, S.J.; Milla, R. Environmental impacts of fully irrigated sugarcane production: Herbicide run-off dynamics from farms and associated drainage systems. Agric. Ecosyst. Environ. 2013, 180, 123–135. [Google Scholar] [CrossRef]
- O’Brien, D.S.; Lewis, S.; Davis, A.; Gallen, C.; Smith, R.; Turner, R.; Warne, M.; Turner, S.; Caswell, S.; Mueller, J.F.; et al. Spatial and Temporal Variability in Pesticide Exposure Downstream of a Heavily Irrigated Cropping Area: Application of Different Monitoring Techniques. J. Agric. Food Chem. 2016, 64, 3975–3989. [Google Scholar] [CrossRef] [PubMed]
- Haynes, D.; Müller, J.; Carter, S. Pesticide and herbicide residues in sediments and seagrasses from the Great Barrier Reef World Heritage Area and Queensland coast. Mar. Pollut. Bull. 2000, 41, 279–287. [Google Scholar] [CrossRef]
- Kennedy, K.; Schroeder, T.; Shaw, M.; Haynes, D.; Lewis, S.; Bentley, C.; Paxman, C.; Carter, S.; Brando, V.; Bartkow, M.; et al. Photosystem-II herbicides on the Great Barrier Reef- results from up to five years of monitoring and a preliminary comparison with remote sensing derived water quality parameters. Mar. Pollut. Bull. 2012, 65, 292–305. [Google Scholar] [CrossRef]
- Furnas, M.J. Catchments and Corals: Terrestrial Runoff to the Great Barrier Reef; Australian Institute Marine Science: Townsville, Australia, 2003.
- Johnson, A.K.L.; Ebert, S.P. Quantifying inputs of pesticides to the Great Barrier Reef Marine Park—A case study in the Herbert River catchment of north-east Queensland. Mar. Pollut. Bull. 2000, 41, 302–309. [Google Scholar] [CrossRef]
- Davis, A.M.; Lewis, S.E.; Brodie, J.E.; Benson, A. The potential benefits of herbicide regulation: A cautionary note for the Great Barrier Reef catchment area. Sci. Total Environ. 2014, 490, 81–92. [Google Scholar] [CrossRef] [PubMed]
- State of Queensland. Reef Water Quality Protection Plan 2009; Queensland Department of Premier and Cabinet, Reef Water Quality Plan Secretariat: Brisbane, Australia, 2009.
- State of Queensland. Reef Water Quality Protection Plan 2013; Reef Water Quality Protection Plan Secretariat: Brisbane, Australia, 2013.
- Australian Pesticides and Veterinary Medicines Authority. The Reconsideration of the Registrations of Selected Products Containing Diuron and Their Associated Labels; Diuron Review. Final Review Report and Regulatory Decision; Australian Pesticides and Veterinary Medicines Authority: Canberra, Australia, 2012; ISBN 978-0-9873591-7-9. Available online: http://apvma.gov.au/sites/default/files/publication/15396-diuron-review-report.pdf (accessed on 21 October 2019).
- State of Queensland. Reef 2050 Water Quality Improvement Plan; Reef Water Quality Protection Plan Secretariat: Brisbane, Australia, 2018.
- Smathers, R.L. Economics of Surface Irrigation Systems; University of Idaho Cooperative Extension System: Moscow, Idaho, 1995. [Google Scholar]
- Tiercelin, J.R.; Vidal, A. Traité d’Irrigation, 2nd ed.; Lavoisier: Paris, France, 2006. [Google Scholar]
- Dauda, T.O.; Asiribo, O.E.; Akinbode, S.O.; Saka, J.O.; Salahu, B.F. An assessment of the roles of irrigation farming in the millennium development goals. Afr. J. Agric. Res. 2009, 4, 445–450. [Google Scholar]
- Holden, J.R. Irrigation of Sugarcane; Bureau of Sugar Experiment Stations: Brisbane, Australia, 1998.
- Tilley, L.; Chapman, L. Benchmarking Crop Water Index for the Queensland Sugar Industry; Bureau of Sugar Experiment Stations: Brisbane, Australia, 1999.
- Oliver, D.P.; Anderson, J.S.; Davis, A.; Lewis, S.; Brodie, J.; Kookana, R. Banded applications are highly effective in minimising herbicide migration from furrow-irrigated sugar cane. Sci. Total Environ. 2014, 466–467, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.M.; Pradolin, J. Precision herbicide application technologies to decrease herbicide losses in furrow irrigation outflows in a northeastern Australian cropping system. J. Agric. Food Chem. 2016, 64, 4021–4028. [Google Scholar] [CrossRef]
- Thorburn, P.J.; Biggs, J.S.; Attard, S.J.; Kemei, J. Environmental impacts of irrigated sugarcane production: Nitrogen lost through runoff and leaching. Agric. Ecosyst. Environ. 2011, 144, 1–12. [Google Scholar] [CrossRef]
- Huggins, R.; Wallace, R.; Orr, D.N.; Smith, R.A.; Taylor, O.; King, O.C.; Gardiner, R.; Wallace, S.; Ferguson, B.; Preston, S.; et al. Total Suspended Solids, Nutrient and Pesticide Loads (2015–2016) for Rivers That Discharge to the Great Barrier Reef—Great Barrier Reef Catchment Loads Monitoring Program; Queensland Department of Environment and Science: Brisbane, Australia, 2017.
- Clemmens, A.; Wahl, T.; Bos, M.; Replogle, J. Water Measurement with Flumes and Weirs, Publication No. 58; International Institute for Land Reclamation and Improvement: Wageningen, The Netherlands, 2001. [Google Scholar]
- Wahl, T.; Clemmens, A.; Replogle, J.; Bos, M. Simplified Design of Flumes and Weirs. Irrig. Drain. 2005, 54, 231–247. [Google Scholar] [CrossRef]
- Winfield, T.W.; Bashe, W.J.; Baker, T.V. Method 547: Determination of Glyphosate in Drinking Water by Direct-Aqueous Injection HPLC, Post-Column Derivatization, and Fluorescence Detection; U.S. Environmental Protection Agency: Cincinatti, OH, USA, 1990.
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and dispersive solid-phase extraction for the determination of pesticide residues in produce. J. Assoc. Off. Agric. Chem. Int. 2003, 86, 412–431. [Google Scholar]
- Bassett, M.V.; Wendelken, S.C.; Dattilio, T.A.; Pepich, B.V. Munch, D.J. Method 532 Revision 1.0: Determination of Phenylurea Compounds in Drinking Water by Solid Phase Extraction and High Performance Liquid Chromotography with UV Detection; U.S. Environmental Protection Agency: Cincinatti, OH, USA, 2000; pp. 1–47.
- Shoemaker, J.A.; Bassett, M.V. Method 535 Version 1.1: Measurement of Chloroacetanilide and Other Acetamide Herbicide Degradates in Drinking Water by Solid Phase Extraction and Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS); U.S. Environmental Protection Agency: Cincinatti, OH, USA, 2005; pp. 1–61.
- Munch, J.W.; Bashe, W.J. Method 549.2 Revision 1.0: Determination of Diquat and Paraquat in Drinking Water by Liquid-Solid Extraction and High Performance Liquid Chromatography and Ultraviolet Detection; U.S. Environmental Protection Agency: Cincinatti, OH, USA, 1997; pp. 1–61.
- eWater Cooperative Research Centre. Water Quality Analyser v2.0.0 User Guide; eWater Cooperative Research Centre: Canberra, Australia, 2011; ISBN 978-1-921543-47-0. [Google Scholar]
- Warne, M.S.J.; Neelamraju, C.; Strauss, J.; Smith, R.A.; Turner, R.D.R.; Mann, R. Development of a Pesticide Risk Baseline for the Reef 2050 Water Quality Improvement Plan; Queensland Department of Environment and Science: Queensland, Australia; Department of Science, Information Technology and Innovation: Brisbane, Australia; p. 228. (in press)
- Smith, R.A.; Warne, M.S.J.; Mengersen, K.; Turner, R.D.R. An improved method for calculating toxicity-based pollutant loads: Part 1. Method development. Integr. Environ. Manag. Assess. 2017, 13, 746–753. [Google Scholar] [CrossRef]
- Smith, R.A.; Warne, M.S.J.; Mengersen, K.; Turner, R.D.R. An improved method for calculating toxicity based pollutant loads: Part 2. Application to contaminants discharged to the Great Barrier Reef, Queensland, Australia. Integr. Environ. Manag. Assess. 2017, 13, 754–764. [Google Scholar] [CrossRef]
- King, O.C.; Smith, R.A.; Mann, R.; Warne, M.S.J. Proposed Aquatic Ecosystem Protection Guideline Values for Pesticides Commonly Used in the Great Barrier Reef Catchment Area: Part 1—2,4-D, Ametryn, Diuron, Glyphosate, Hexazinone, Imazapic, Imidacloprid, Isoxaflutole, Metolachlor, Metribuzin, Metsulfuron-Methyl, Simazine, Tebuthiuron; Department of Science, Information Technology and Innovation: Brisbane, Australia, 2017; p. 294.
- King, O.C.; Smith, R.A.; Warne, M.S.J.; Frangos, J.S.; Mann, R. Proposed Aquatic Ecosystem Protection Guideline Values for Pesticides Commonly Used in the Great Barrier Reef Catchment Area: Part 2—Bromacil, Chlorothalonil, Fipronil, Fluometuron, Fluroxypyr, Haloxyfop, MCPA, Pendimethalin, Prometryn, Propazine, Propiconazole, Terbutryn, Triclopyr and Terbuthylazine; Department of Science, Information Technology and Innovation: Brisbane, Australia, 2017; p. 209.
- Davis, A.M.; Lewis, S.E.; Bainbridge, Z.T.; Glendenning, L.; Turner, R.D.R.; Brodie, J.E. Dynamics of herbicide transport and partitioning under event flow conditions in the lower Burdekin region, Australia. Mar. Pollut. Bull. 2012, 65, 182–193. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 12 October 2019).
- Masters, B.; Rohde, K.; Gurner, N.; Reid, D. Reducing the risk of herbicide runoff in sugarcane farming through controlled traffic and early-banded application. Agric. Ecosyst. Environ. 2013, 180, 29–39. [Google Scholar] [CrossRef]
- Silburn, D.M.; Foley, J.L.; deVoil, R.C. Managing runoff of herbicides under rainfall and furrow irrigation with wheel traffic and banded spraying. Agric. Ecosyst. Environ. 2013, 180, 40–53. [Google Scholar] [CrossRef]
- State of Queensland. User Manual. Dual Herbicide Sprayer. Version 2, November 2017; Department of Agriculture and Fisheries: Brisbane, Australia, 2017; p. 28.
- Zemolin, C.R.; Avila, L.A.; Cassol, G.V.; Massey, J.H.; Camargo, E.R. Environmental fate of S-metolachlor: A review. Planta Daninha 2014, 32, 655–664. [Google Scholar] [CrossRef]
- Little, D.L.; Shaner, D.L. Absorption and translocation of the imidazolinone herbicide. In The Imidazolinone Herbicide; Shaner, D.L., O’Connor, S.L., Eds.; CRC: Boca Raton, FL, USA, 1991; pp. 53–69. [Google Scholar]
- Roberts, T.R.; Dyson, J.S.; Lane, M.C.G. Deactivation of the biological activity of paraquat in the soil environment: A review of long-term environmental fate. J. Agric. Food Chem. 2002, 50, 3623–3631. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.M.; Tink, M.; Rohde, K.; Brodie, J.E. Urea contributions to dissolved ‘organic’ nitrogen losses from intensive, fertilised agriculture. Agric. Ecosyst. Environ. 2016, 223, 190–196. [Google Scholar] [CrossRef]
- Australian and Queensland Queensland. Pesticide Risk Baseline Methods, Reef Water Quality Report Card 2017 and 2018, Reef 2050 Water Quality Improvement Plan; Queensland Government: Brisbane, Australia, 2019. Available online: https://www.reefplan.qld.gov.au/__data/assets/pdf_file/0026/82925/report-card-2017-2018-methods-pesticide-risk-baseline.pdf (accessed on 22 October 2019).

| Soil Physico-Chemical Property | Value |
|---|---|
| Type-texture | grey-brown clay |
| Sample depth | 0–0.2 m |
| Soil pH | 7.3–8.3 |
| Cation exchange capacity | 18–24 meq/100 g |
| Electrical conductivity (1:5 extract, dS/m) | 0.17–0.2 |
| Soil organic carbon (%) | 0.94–1.00 |
| Product (Active) * | a.i. g/L | Application Rate to Paddock (L-Kg/ha) | Irvin Leg Rate (a.i. g/ha) | Shielded Sprayer Rate (a.i. g/ha) |
|---|---|---|---|---|
| Agritone 750 (MCPA) | 750 | 1 | 450 | |
| Wipeout 540 (Glyphosate) | 540 | 1 | 324 | |
| Zulu Evo (2,4-D) | 720 | 1.8 | 1296 | 518.4 |
| Gramoxone (Paraquat) | 250 | 1.6 | 400 | 160 |
| Diurex (Diuron) | 900 | 1.9 | 1710 | 684 |
| Dual Gold (Metolachlor) | 960 | 1.8 | 1728 | 691.2 |
| Spark (Imazapic) | 240 | 0.4 | 96 | 38.4 |
| Flume: Treatment | Runoff (mm) |
|---|---|
| Flume 1-Treatment 1: Diuron broadcast | 22.0 |
| Flume 2-Treatment 1: Diuron broadcast | 16.5 |
| Flume 3-Treatment 2: Diuron banded | 19.5 |
| Flume 4-Treatment 2: Diuron banded | 14.6 |
| Flume 5-Treatment 3: Metolachlor broadcast | 20.6 |
| Flume 6-Treatment 3: Metolachlor broadcast | 15.7 |
| Flume 7-Treatment 4: Metolachlor banded | 19.5 |
| Flume 8-Treatment 4: Metolachlor banded | 17.2 |
| Flume 9-Treatment 5: Imazapic broadcast | 18.5 |
| Flume 10-Treatment 5: Imazapic broadcast | 21.1 |
| Flume 11-Treatment 6: Imazapic banded | 15.9 |
| Flume 12-Treatment 6: Imazapic banded | 20.4 |
| Mean (C.V.) | 18.46 (0.13) |
| Treatment (T) | Metolachlor | Imazapic | Diuron | GlyphosateA | MCPA A | 2,4-D | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Load Loss (g.a.i./ha) | % of Applied Lost | Load Loss (g.a.i./ha) | % of Applied Lost | Load Loss (g.a.i./ha) | % of Applied Lost | Load Loss (g.a.i./ha) | % of Applied Lost | Load Loss (g.a.i./ha) | % of Applied Lost | Load Loss (g.a.i./ha) | % of Applied Lost | |
| T1-Diuron broadcast | 60.1 | 3.5 | 16.1 | 1.2 | ||||||||
| T1-Diuron broadcast | 111.0 | 6.5 | 28.4 | 2.2 | ||||||||
| T2-Diuron banded | 6.1 | 0.9 | 12.2 | 3.8 | 26.7 | 5.9 | 7.9 | 1.5 | ||||
| T2-Diuron banded | 9.1 | 1.3 | 14.1 | 4.3 | 36.1 | 8.0 | 10.0 | 1.9 | ||||
| T3-Metolachlor broadcast | 61.5 | 3.6 | 23.2 | 1.8 | ||||||||
| T3-Metolachlor broadcast | 117.4 | 6.8 | 34.5 | 2.7 | ||||||||
| T4-Metolachlor banded | 4.8 | 0.7 | 19.7 | 6.1 | 32.0 | 7.1 | 5.3 | 1.0 | ||||
| T4-Metolachlor banded | 5.6 | 0.8 | 21.0 | 6.5 | 45.8 | 10.2 | 6.7 | 1.3 | ||||
| T5-Imazapic broadcast | 3.9 | 4.1 | 31.0 | 2.4 | ||||||||
| T5-Imazapic broadcast | 3.6 | 3.7 | 12.3 | 1.0 | ||||||||
| T6-Imazapic banded | 0.3 | 0.7 | 14.0 | 4.3 | 20.4 | 4.5 | 5.9 | 1.1 | ||||
| T6-Imazapic banded | 0.4 | 1.1 | 11.8 | 3.6 | 17.6 | 3.9 | 6.6 | 1.3 | ||||
| Herbicide | CAS Registry Number | DT50 (Field) | Solubility in Water (mg/L) (20 °C, pH 7) | KOC | Proposed 99% Freshwater Ecosystem Protection Guideline (µg/L) |
|---|---|---|---|---|---|
| Diuron | 330-54-1 | 229 | 35.6 | 680 | 0.08 |
| S-Metolachlor | 87392-12-9 | 21 | 480 | 120 | 0.016 |
| Imazapic | 104098-48-8 | 232 | 2230 | 137 | 0.036 |
| 2,4-D | 94-75-7 | 28.8 | 24,300 | 39.3 | 1000 ^ |
| MCPA | 94-74-6 | 25 | 29,390 | 46.0 | 1.0 ^ |
| Paraquat | 1910-42-5 | 2800 | 620,000 | 1,000,000 | NA |
| Glyphosate | 1071-83-6 | 23.79 | 10,500 | 1424 | 140 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davis, A.M.; Neelamraju, C. Quantifying Water Quality Improvements through Use of Precision Herbicide Application Technologies in a Dry-Tropical, Furrow-Irrigated Cropping System. Water 2019, 11, 2326. https://doi.org/10.3390/w11112326
Davis AM, Neelamraju C. Quantifying Water Quality Improvements through Use of Precision Herbicide Application Technologies in a Dry-Tropical, Furrow-Irrigated Cropping System. Water. 2019; 11(11):2326. https://doi.org/10.3390/w11112326
Chicago/Turabian StyleDavis, Aaron M., and Catherine Neelamraju. 2019. "Quantifying Water Quality Improvements through Use of Precision Herbicide Application Technologies in a Dry-Tropical, Furrow-Irrigated Cropping System" Water 11, no. 11: 2326. https://doi.org/10.3390/w11112326
APA StyleDavis, A. M., & Neelamraju, C. (2019). Quantifying Water Quality Improvements through Use of Precision Herbicide Application Technologies in a Dry-Tropical, Furrow-Irrigated Cropping System. Water, 11(11), 2326. https://doi.org/10.3390/w11112326




