Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methodes
2.2.1. Powder Characterization Techniques
2.2.2. Adsorption Isotherm Models
3. Results and Discussion
3.1. Characterization of Products Used
3.1.1. CBZ Characterization
3.1.2. Mineralogy, Surface Properties and Texture of Adsorbents
3.2. Retention of Carbamazepine
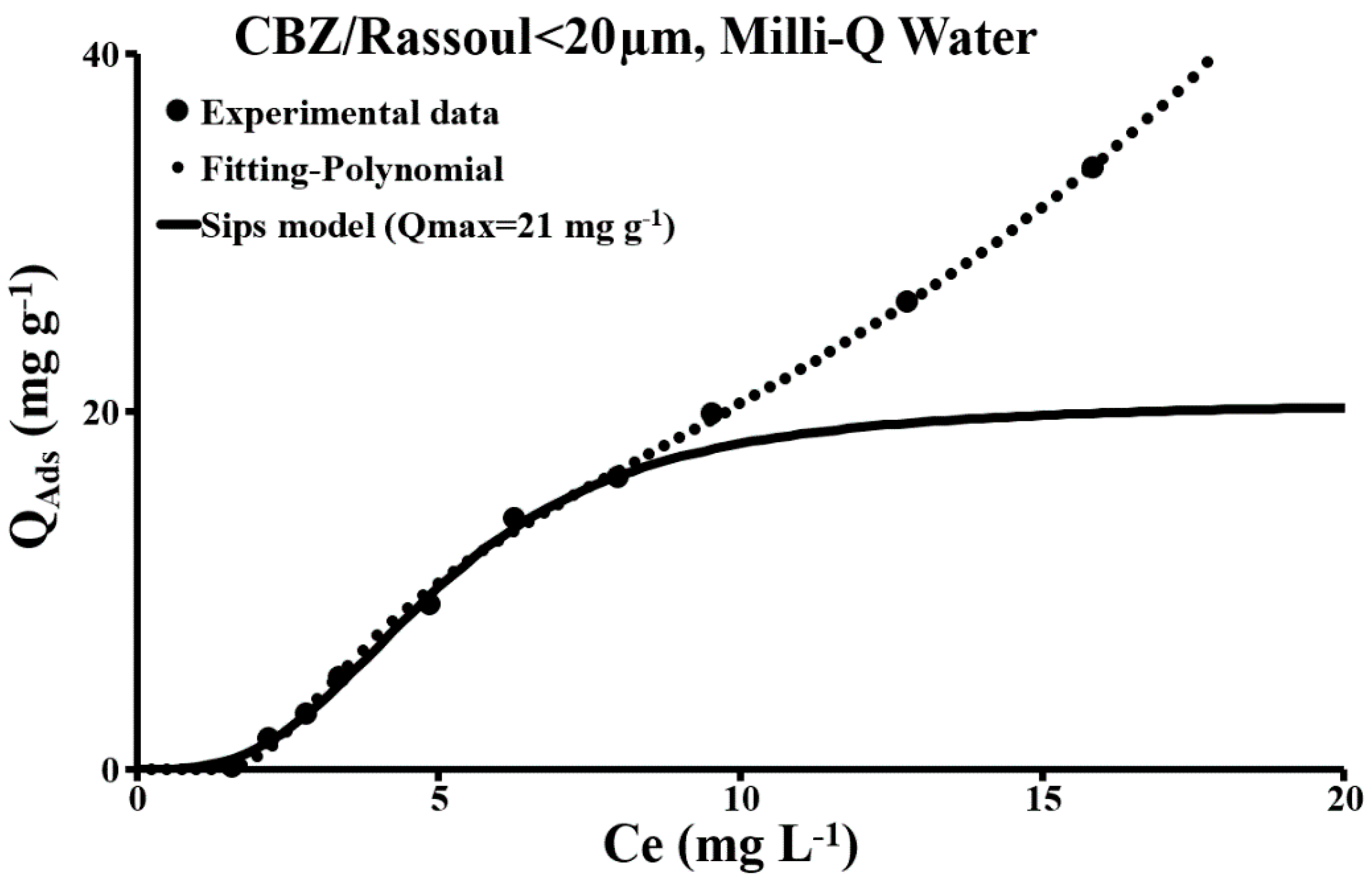
3.2.1. The Principal Mineral Constituents of a Naturel Soil in the Presence of Carbamazepine
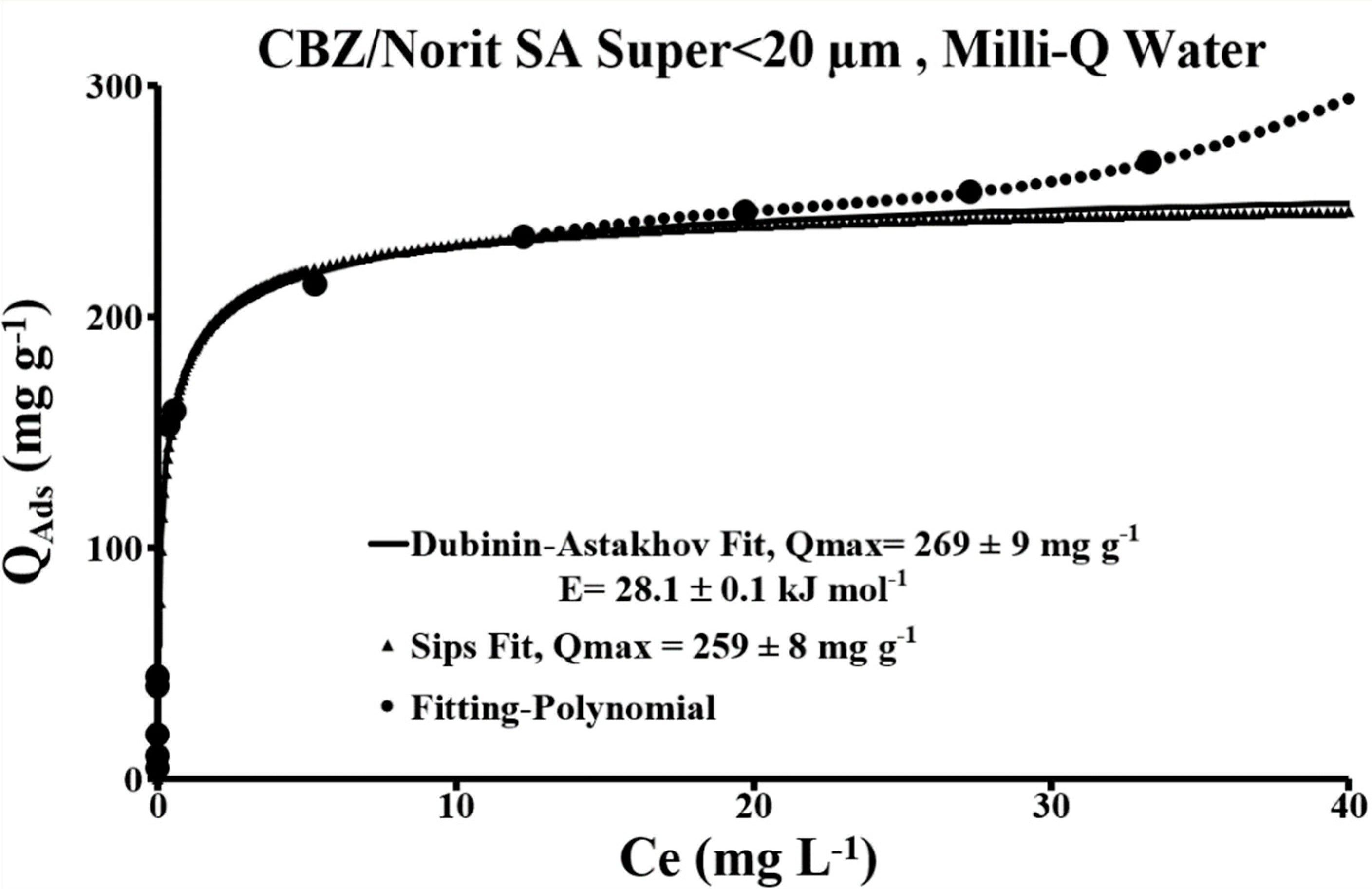
3.2.2. Activated Carbon in Contact with Carbamazepine
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Hai, F.I.; Yang, S.; Asif, M.B.; Sencadas, V.; Shawkat, S.; Sanderson-Smith, M.; Gorman, J.; Xu, Z.Q.; Yamamoto, K. Carbamazepine as a possible anthropogenic marker in water: Occurrences, toxicological effects, regulations and removal by wastewater treatment technologies. Water 2014, 10, 107. [Google Scholar] [CrossRef]
- Björlenius, B.; Ripszám, M.; Haglund, P.; Lindberg, R.H.; Tysklind, M.; Fick, J. Pharmaceutical residues arewidespread in Baltic Sea coastal and offshore waters—Screening for pharmaceuticals and modeling of environmental concentrations of carbamazepine. Sci. Total Environ. 2018, 633, 1496–1509. [Google Scholar] [CrossRef] [PubMed]
- Paz, A.; Tadmor, G.; Malchi, T.; Blotevogel, J.; Borch, T.; Polubesova, T.; Chefetz, B. Fate of carbamazepine, its metabolites, and lamotrigine in soils irrigated with reclaimed wastewater: Sorption, leaching and plant uptake. Chemosphere 2016, 160, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Paíga, P.; Correia, M.; Fernandes, M.J.; Silva, A.; Carvalho, M.; Vieira, J.; Jorge, S.; Silva, J.G.; Freire, C.; Delerue-Matos, C. Assessment of 83 pharmaceuticals in WWTP influent and effluent samples by UHPLC-MS/MS: Hourly variation. Sci. Total Environ. 2019, 648, 582–600. [Google Scholar] [CrossRef] [PubMed]
- Kostich, M.S.; Batt, A.L.; Lazorchak, J.M. Concentrations of prioritized pharmaceuticals in effluents from 50 large wastewater treatment plants in the US and implications for risk estimation. Environ. Pollut. 2014, 184, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Simazaki, D.; Kubota, R.; Suzuki, T.; Akiba, M.; Nishimura, T.; Kunikane, S. Occurrence of selected pharmaceuticals at drinking water purification plants in Japan and implications for human health. Water Res. 2015, 76, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Tauxe Würsch, A. Wastewaters: Occurrence of Pharmaceutical Substances and Genotoxicity. Ph.D. Thesis, Federal Polytechnic School of Lausanne, Lausanne, Switzerland, 2005. [Google Scholar]
- Verlicchi, P.; Al Aukidy, M.; Zambello, E. Occurrence of pharmaceutical compounds in urban wastewater: Removal, mass load and environmental risk after a secondary treatment–A review. Sci. Total Environ. 2012, 429, 123–155. [Google Scholar] [CrossRef]
- Freitas, R.; Almeida, Â.; Calisto, V.; Velez, C.; Moreira, A.; Schneider, R.J.; Esteves, V.I.; Wrona, F.J.; Figueira, E.; Soares, A.M.V.M. The impacts of pharmaceutical drugs under ocean acidification: Newdata on single and combined long-term effects of carbamazepine on Scrobicularia plana. Sci. Total Environ. 2016, 541, 977–985. [Google Scholar] [CrossRef]
- Ferrari, B.; Paxéus, N.; Lo, G.R.; Pollio, A.; Garric, J. Ecotoxicological impact of pharmaceuticals found in treated wastewaters: Study of carbamazepine, clofibric acid, and diclofenac. Ecotoxicol. Environ. Saf. 2003, 55, 359–370. [Google Scholar] [CrossRef]
- Andreozzi, R.; Marotta, R.; Pinto, G.; Pollio, A. Carbamazepine in water: Persistence in the environment, ozonation treatment and preliminary assessment on algal toxicity. Water Res. 2002, 36, 2869–2877. [Google Scholar] [CrossRef]
- Riemenschneider, C.; Seiwert, B.; Moeder, M.; Schwarz, D.; Reemtsma, T. Extensive transformation of the pharmaceutical carbamazepine following uptake into intact tomato plants. Environ. Sci. Technol. 2017, 51, 6100–6109. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Li, L.; Sun, L.; Zhang, S.; Shan, X.Q.; Yang, S.; Wen, B. Adsorption characteristics of 1,2,4-trichlorobenzene, 2,4,6-trichlorophenol, 2-naphthol and naphthalene on graphene and graphene oxide. Carbon 2013, 51, 156–163. [Google Scholar] [CrossRef]
- Al-Khateeb, L.A.; Almotiry, S.; Abdel Salam, M. Adsorption of pharmaceutical pollutants onto graphene nanoplatelets. Chem. Eng. J. 2014, 248, 191–199. [Google Scholar] [CrossRef]
- Yu, Z.; Peldszus, S.; Huck, P.M. Adsorption characteristics of selected pharmaceuticals and an endocrine disrupting compound-naproxen, carbamazepine and nonylphenol-on activated carbon. Water Res. 2008, 42, 2873–2882. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Zeng, G.; Tang, L.; Fan, C.; Zhang, C.; He, X.; He, Y. An overview on limitations of TiO2-based particles for photocatalytic degradation of organic pollutants and the corresponding countermeasures. Water Res. 2015, 79, 128–146. [Google Scholar] [CrossRef]
- Oturan, M.A.; Aaron, J.J. Advanced oxidation processes in water/wastewater treatment: Principles and applications—A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Takagi, K.; Kataoka, R.; Kamei, I.; Kiyota, H.; Sato, Y. Biodegradation of dieldrin by a soil fungus isolated from a soil with annual endosulfan applications. Environ. Sci. Technol. 2010, 44, 6343–6349. [Google Scholar]
- Wanga, J.; Bi, Y.; Pfister, G.; Henkelmann, B.; Zhu, K.; Schramm, K.W. Determination of PAH, PCB, and OCP in water from the three gorges reservoir accumulated by semipermeable membrane devices (SPMD). Chemosphere 2009, 75, 1119–1127. [Google Scholar] [CrossRef]
- Cox, M.; Négré, P.; Yurramendi, L. A Guide Book on the Treatment of Effluents from the Mining/Metallurgy, Paper, Plating and Textile Industries; Inasment-Tecnalia on behalf of the European Commission; INASMENT-Tecnalia: San Sebastian, Spain, 2006. [Google Scholar]
- Laurent, A.; Ettrup, K.; Kounina, A.; Hansen, S.F.; Meesters, J.A.J.; Vea, E.B. Development of comparative toxicity potentials of TiO2 nanoparticles for use in life cycle assessment. Environ. Sci. Technol. 2017, 51, 4027–4037. [Google Scholar]
- Ambrozini, B.; Pinto, M.A.L.; Ferreira, A.P.G.; Cavalheiro, É.T.G. Thermoanalytical studies of carbamazepine: Hydration/dehydration, thermal decomposition, and solid phase transitions. Braz. J. Pharm. Sci. 2014, 50, 877–884. [Google Scholar]
- A-Razzak, M.; Glen, R.C. Application of rule-induction in the derivation of quantitative structure-activity relationships. J. Comput. Aided Mol. Des. 1992, 6, 349–383. [Google Scholar] [CrossRef] [PubMed]
- Oppel, J.; Broll, G.; Löffler, D.; Meller, M.; Römbke, J.; Ternes, T. Leaching behaviour of pharmaceuticals in soil-testing-systems: A part of an environmental risk assessment for groundwater protection. Sci. Total Environ. 2005, 336, 285. [Google Scholar] [CrossRef]
- Mular, A.L.; Roberts, R.B. A simplified method to determine isoelectric points of oxides. Trans. Can. Inst. Min. Metall. 1966, 69, 438–439. [Google Scholar]
- Sips, R. On the structure of a catalyst surface. J. Chem. Phys. 1948, 16, 490–495. [Google Scholar] [CrossRef]
- Inglezakis, V.L. Solubility-normalized dubinin–astakhov adsorption isotherm for ion-exchange systems. Micropor. Mesopor. Mater. 2007, 103, 72–81. [Google Scholar] [CrossRef]
- Ouadjenia, F.; Marouf, R.; Schott, J.; Yahiaoui, A. Removal of Cu(II), Cd(II) and Cr(III) ions from aqueous solution by dam silt. Arab. J. Chem. 2013, 6, 401–406. [Google Scholar] [CrossRef]
- Moreno-Castilla, C. Micropore structure of activated carbons prepared from a Spanish subbituminous coal studied by CO2, benzene, and cyclohexane adsorption. Langmuir 1995, 11, 247–252. [Google Scholar] [CrossRef]
- Nghiem, L.; Schäfer, A.L.; Elimelech, M. Pharmaceutical retention mechanisms by nanofiltration membranes. Environ. Sci. Technol. 2005, 39, 7698–7705. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquérol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special refrence to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Stoeckli, H.F. Microporous carbons and their characterization: The present state of the art. Carbon 1990, 28, 1–6. [Google Scholar] [CrossRef]
- Himes, V.L.; Mighell, A.D.; De Camp, W.H. Structure of carbamazepine: 5H-Dibenz[b,f]azepine-5-carboxamide. Acta Cryst. 1981, B37, 2242–2245. [Google Scholar] [CrossRef]
- Kareru, P.G.; Keriko, J.M.; Gachanja, A.N.; Kenji, G.M. Direct detection of triterpenoid saponins in medicinal plants. Afr. J. Trad. Complement. Altern. Med. 2008, 5, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Dinda, B.; Debnath, S.; Mohanta, B.C.; Harigaya, Y. Naturally occurring triterpenoid saponins. Chem. Biodivers. 2010, 7, 2327–2580. [Google Scholar] [CrossRef] [PubMed]
- Czernicki, W.; Baranska, M. Carbamazepine polymorphs: Theoretical and experimental vibrational spectroscopy studies. Vib. Spectrosc. 2013, 65, 12–23. [Google Scholar] [CrossRef]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry, 5th ed.; W H Freeman: New York, NY, USA, 2002. [Google Scholar]
- Koopal, L.K.; Van Riemsdijk, W.H.; De Wit, J.C.M.; Benedetti, M.F. Analytical isotherm equations for multicomponent adsorption to heterogeneous surfaces. J. Colloid Interface Sci. 1994, 166, 51–60. [Google Scholar] [CrossRef]
- Brandenburg, J.G.; Potticary, J.; Sparkes, H.A.; Price, S.L.; Hall, S.R. Thermal expansion of carbamazepine: Systematic crystallographic measurements challenge quantum chemical calculations. J. Phys. Chem. Lett. 2017, 8, 4319–4324. [Google Scholar] [CrossRef]
- Giles, C.H.; Smith, D.; Huitson, A. A general treatment and classification of the solute adsorption isotherm. I. Theoretical. J. Colloid Interface Sci. 1974, 47, 755–765. [Google Scholar] [CrossRef]
- Moreno-Castilla, C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 2004, 42, 83–94. [Google Scholar] [CrossRef]
- Coughlin, R.W.; Ezra, F.S. Role of surface acidity in the adsorption of organic pollutants on the surface of carbon. Environ. Sci. Technol. 1968, 2, 291–297. [Google Scholar] [CrossRef]
- Hunter, C.A.; Singh, J.; Thornton, J.M. π-π interactions: The geometry and energetics of phenylalanine-phenylalanine interactions in proteins. J. Mol. Biol. 1991, 218, 837–846. [Google Scholar] [CrossRef]
- Haydar, S.; Ferro-García, M.A.; Rivera-Utrilla, J.; Joly, J.P. Adsorption of p-nitrophenol on an activated carbon with different oxidations. Carbon 2003, 41, 387–395. [Google Scholar] [CrossRef]
- Nishio, M.; Hirota, M.; Umezawa, Y. The CH/π Interaction: Evidence, Nature, and Consequences; Wiley-VCH: New York, NY, USA, 1998. [Google Scholar]
- Shafeeyan, M.S.; Daud, W.M.A.W.; Houshmand, A.; Shamiri, A. A review on surface modification of activated carbon for carbon dioxide adsorption. J. Anal. Appl. Pyrol. 2010, 89, 143–151. [Google Scholar] [CrossRef]









| Adsorbent | da µm | ZPC | SSBET m²/g | SSµP m²/g | Lµ-t-plot nm | VµP cm3/g | VmP cm3/g |
|---|---|---|---|---|---|---|---|
| Alumine-a | 0.3 | 8.2 | 6.7 | 0.0 | n.c | 0.00 | 0.01 |
| Quartz-a | 1.5 | 2.0 | 5.4 | 0.0 | n.c | 0.00 | 0.01 |
| Goethite | 1.2 | 7.9 | 14.2 | 0.0 | n.c | 0.00 | 0.03 |
| Hematite | 0.8 | 8.2 | 9.9 | 0.0 | n.c | 0.00 | 0.02 |
| K7A | 1.4 | 4.0 | 14.5 | 5.5 | 0.93 | 0.00 | 0.07 |
| SWy3 <20 µm | 3.4 | 7.0 | 41.9 | 12.5 | 0.98 | 0.01 | 0.08 |
| Rassoul <20 µm | 8.0 | 8.3 | 188.2 | 107.0 | 0.90 | 0.05 | 0.05 |
| Norit <20 µm | 9.0 | 6.9 | 989.3 | 693.5 | 1.74 | 0.30 | 0.32 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
BIZI, M. Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine. Water 2019, 11, 2290. https://doi.org/10.3390/w11112290
BIZI M. Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine. Water. 2019; 11(11):2290. https://doi.org/10.3390/w11112290
Chicago/Turabian StyleBIZI, Mohamed. 2019. "Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine" Water 11, no. 11: 2290. https://doi.org/10.3390/w11112290
APA StyleBIZI, M. (2019). Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine. Water, 11(11), 2290. https://doi.org/10.3390/w11112290





