Bifunctional g-C3N4/WO3 Thin Films for Photocatalytic Water Purification
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of WO3 Precursor Solution
2.2. Preparation of g-C3N4
2.3. Preparation of Bilayered g-C3N4/WO3 Thin Films
2.4. Characterization Techniques
2.5. Evaluation of the Photocatalytic Activity
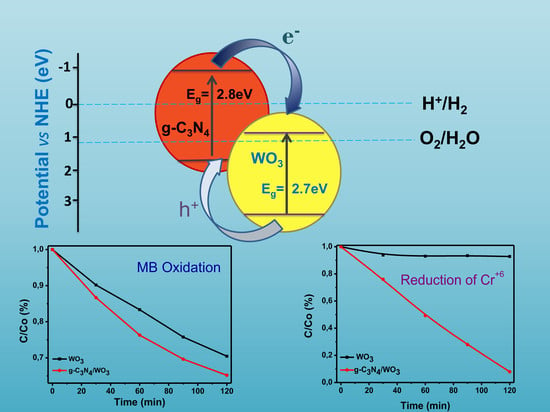
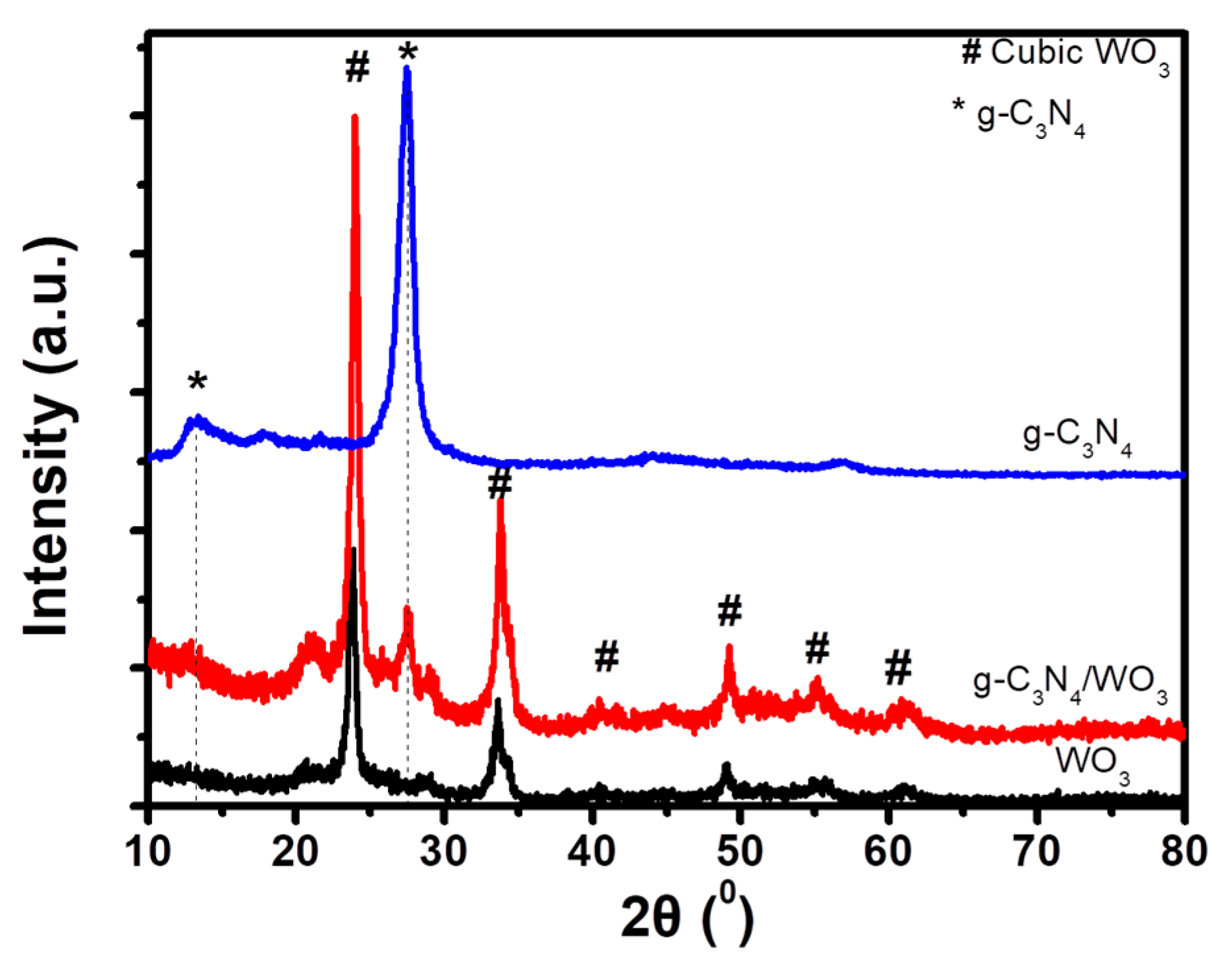
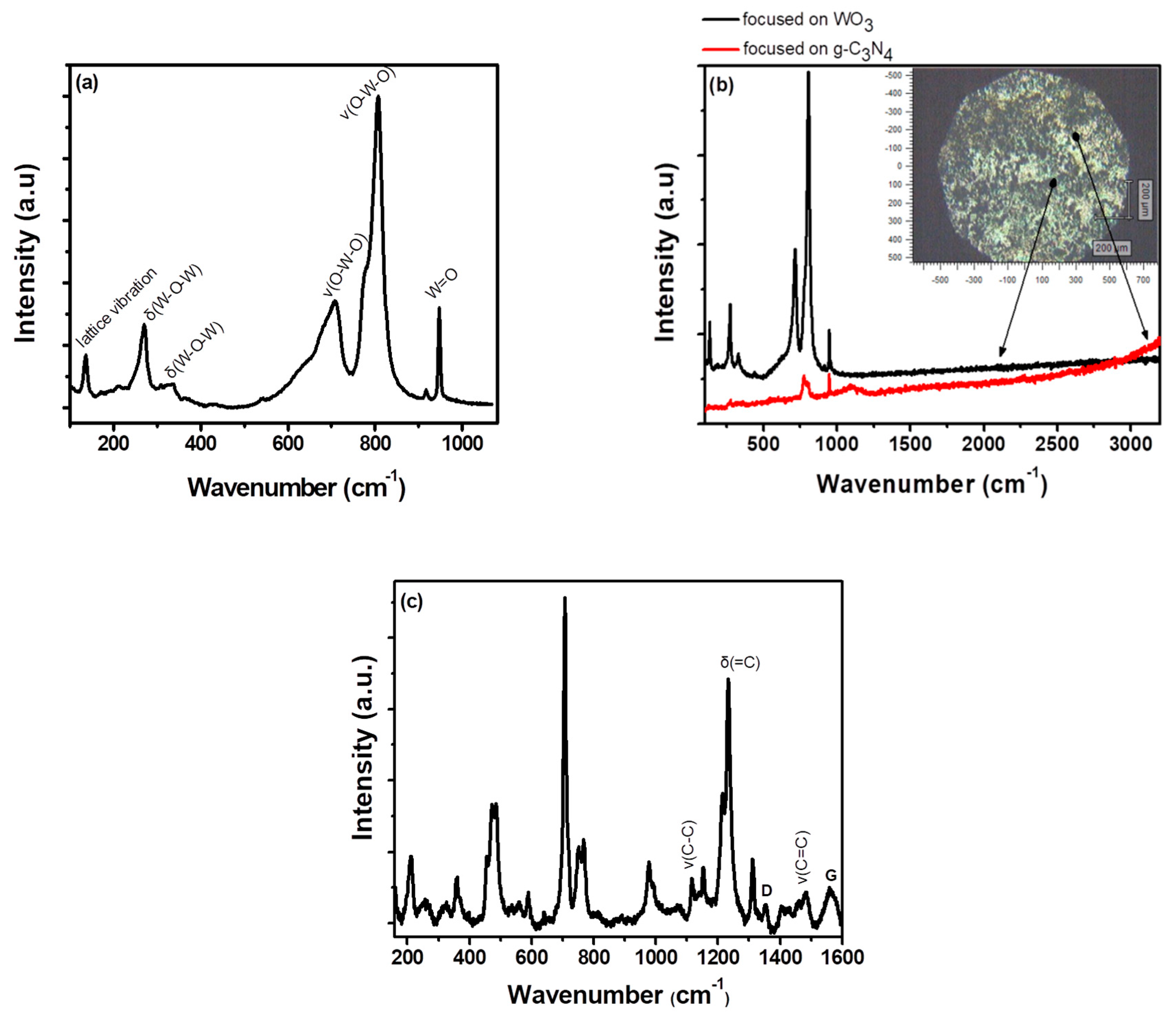
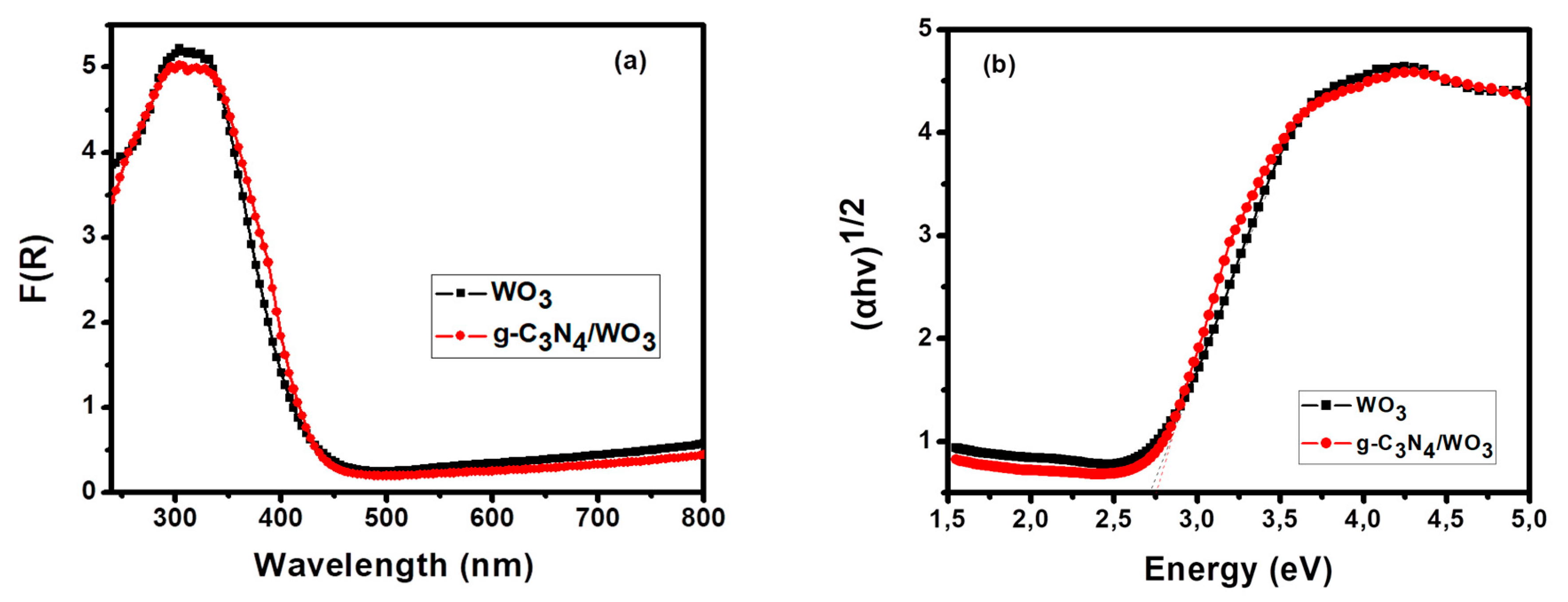
3. Results and Discussion
3.1. Characterization
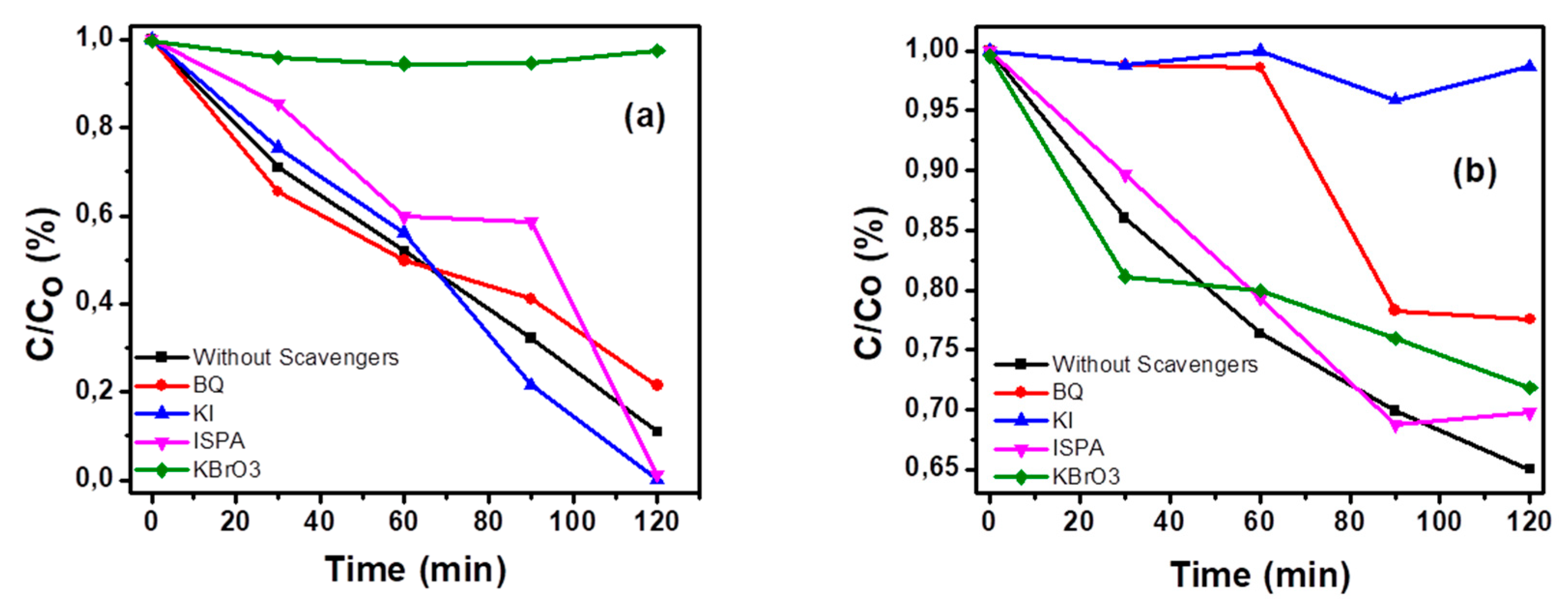
3.2. Photocatalysis and Trapping Experiments
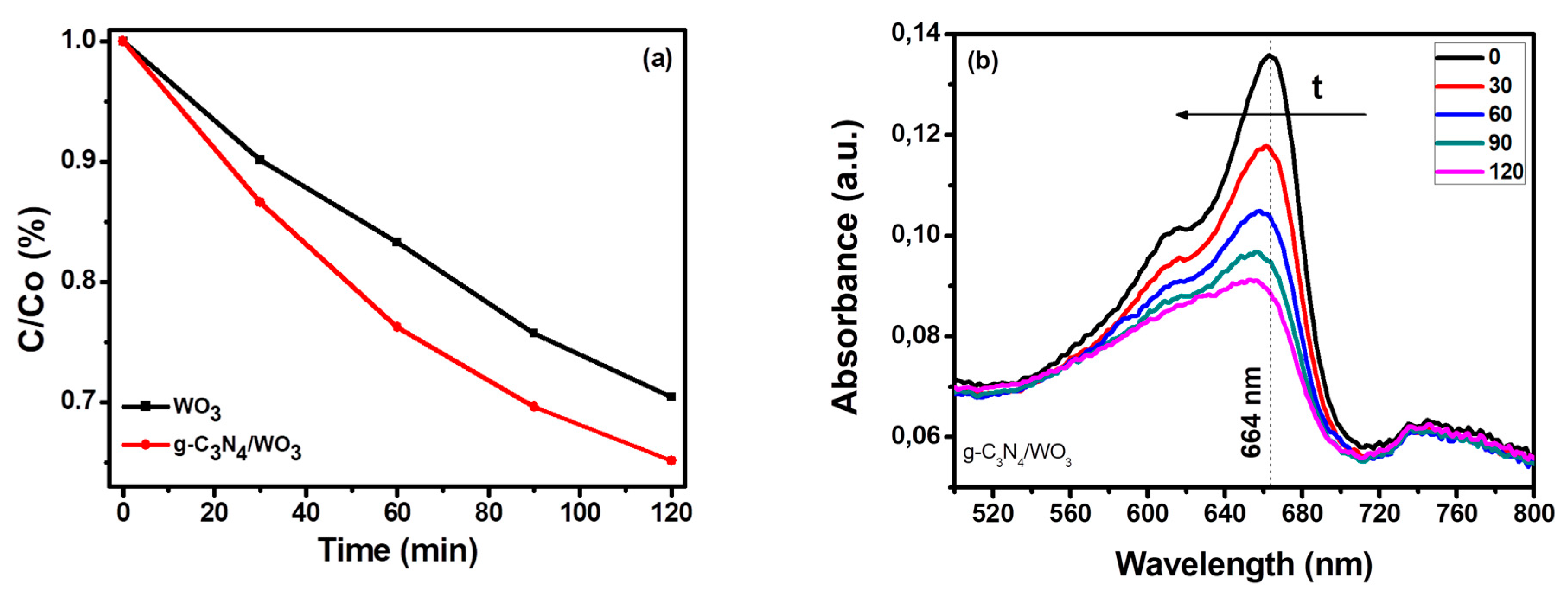
3.2.1. Photocatalytic Oxidation of Methylene Blue
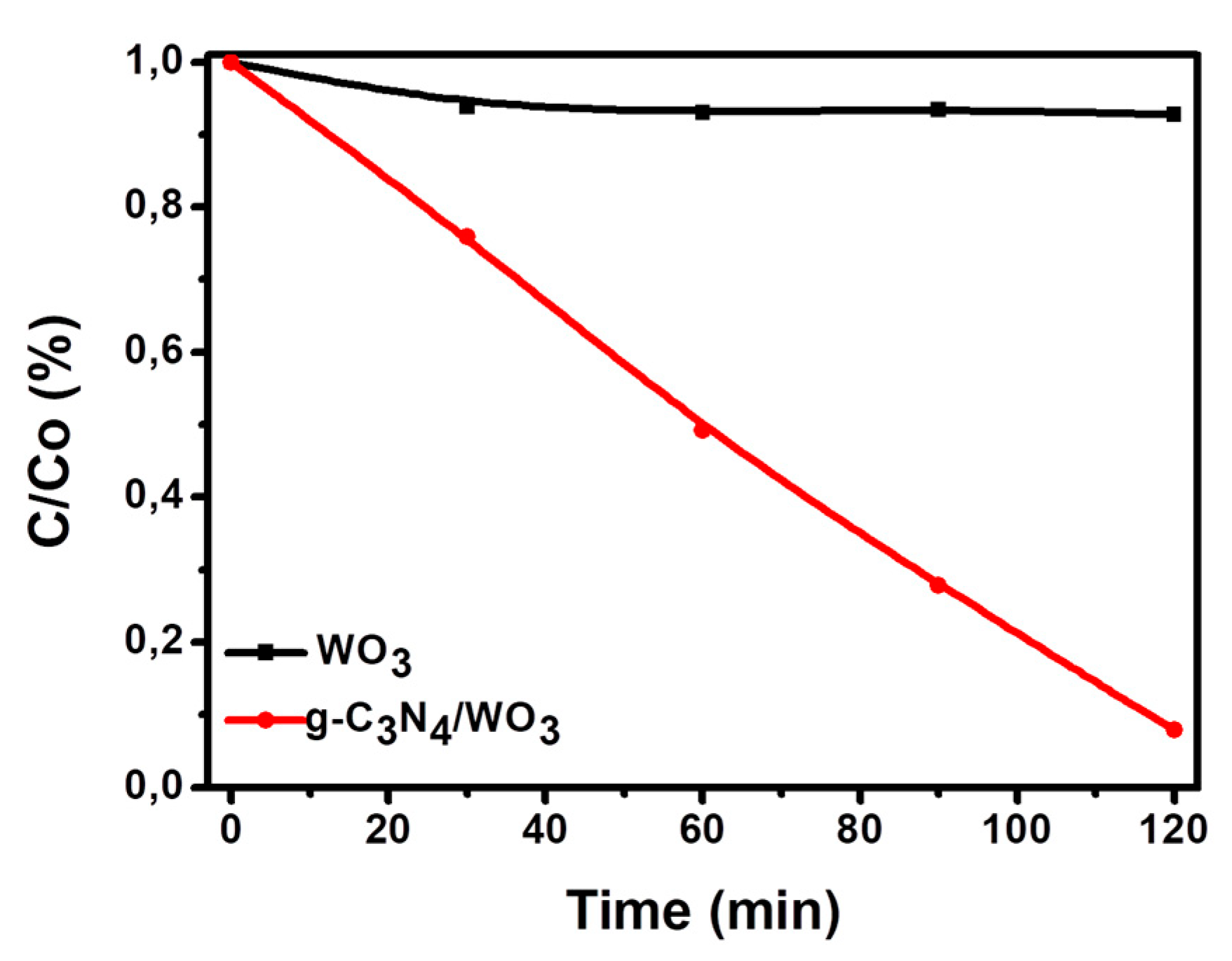
3.2.2. Photocatalytic Reduction of Cr+6
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ibrahim, I.; Ali, I.O.; Salama, T.M.; Bahgat, A.A.; Mohamed, M.M. Synthesis of magnetically recyclable spinel ferrite (MFe2O4, M = Zn, Co, Mn) nanocrystals engineered by sol gel-hydrothermal technology: High catalytic performances for nitroarenes reduction. Appl. Catal. B Environ. 2016, 181, 389–402. [Google Scholar] [CrossRef]
- Yan, M.; Wu, Y.; Zhu, F.; Hua, Y.; Shi, W. The fabrication of a novel Ag3VO4/WO3 heterojunction with enhanced visible light efficiency in the photocatalytic degradation of TC. Phys. Chem. Chem. Phys. 2016, 18, 3308–3315. [Google Scholar] [CrossRef] [PubMed]
- Su, X.T.; Xiao, F.; Li, Y.N.; Jian, Q.J.K.; Sun, J.; Wang, J.D. Synthesis of uniform WO3 square nanoplates via an organic acid-assisted hydrothermal process. Mater. Lett. 2010, 64, 1232–1234. [Google Scholar] [CrossRef]
- Wen, Z.H.; Wu, W.; Liu, Z.; Zhang, H.; Li, J.H.; Chen, J.H. Ultrahigh-efficiency photocatalysts based on mesoporous Pt–WO3 nanohybrids. Phys. Chem. Chem. Phys. 2013, 15, 6773–6778. [Google Scholar] [CrossRef]
- Karimi-Nazarabad, M.; Goharshadi, E.K. Highly efficient photocatalytic and photoelectrocatalytic activity of solar light driven WO3/g-C3N4 nanocomposite. Sol. Energy Mater. Sol. Cells 2017, 160, 484–493. [Google Scholar] [CrossRef]
- Wang, X.C.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef]
- Chen, J.; Xiao, X.; Wang, Y.; Ye, Z. Ag nanoparticles decorated WO3/g-C3N4 2D/2D heterostructure with enhanced photocatalytic activity for organic pollutants degradation. Appl. Surf. Sci. 2019, 467, 1000–1010. [Google Scholar] [CrossRef]
- Liu, L.; Hu, P.; Cui, W.; Li, X.; Zhang, Z. Increased photocatalytic hydrogen evolution and stability over nano-sheet g-C3N4 hybridized CdS core@shell structure. Int. J. Hydrogen Energy 2017, 42, 17435–17445. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, R.; Lin, J.; Zhu, Y. Enhancement of photocurrent and photocatalytic activity of ZnO hybridized with graphite like C3N4. Energy Environ. Sci. 2011, 4, 2922–2929. [Google Scholar] [CrossRef]
- Han, C.; Gao, Y.; Liu, S.; Ge, L.; Xiao, N.; Dai, D.; Xu, B.; Chen, C. Facile synthesis of AuPd/g-C3N4 nanocomposite: An effective strategy to enhance photocatalytic hydrogen evolution activity. Int. J. Hydrogen Energy 2017, 42, 22765–22775. [Google Scholar] [CrossRef]
- Zang, Y.; Li, L.; Zuo, Y.; Lin, H.; Li, G.; Guan, X. Facile synthesis of composite g-C3N4/WO3: A nontoxic photocatalyst with excellent catalytic activity under visible light. RSC Adv. 2013, 3, 13646–13650. [Google Scholar] [CrossRef]
- Katsumata, K.; Motoyoshi, R.; Matsushit, N.; Okada, K. Preparation of graphitic carbon nitride (g-C3N4)/WO3 composites and enhanced visible-light-driven photodegradation of acetaldehyde gas. J. Hazard. Mater. 2013, 260, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Murakami, N.; Tsubota, T.; Ohno, T. Complete oxidation of acetaldehyde over a composite photocatalyst of graphitic carbon nitride and tungsten(VI) oxide under visible-light irradiation. Appl. Catal. B Environ. 2014, 150, 479–485. [Google Scholar] [CrossRef]
- Zhao, J.L.; Jia, Z.; Shen, X.; Zhou, H.; Ma, L. Facile synthesis of WO3 nanorods/g-C3N4 composites with enhanced photocatalytic activity. Ceram. Int. 2015, 141, 5600–5606. [Google Scholar] [CrossRef]
- Athanasekou, C.P.; Likodimos, V.; Falaras, P. Recent developments of TiO2 photocatalysis involving advanced oxidation and reduction reactions in water. J. Environ. Chem. Eng. 2018, 6, 7386–7394. [Google Scholar] [CrossRef]
- Raptis, D.; Dracopoulos, V.; Lianos, P. Renewable energy production by photoelectrochemical oxidation of organic wastes using WO3 photoanodes. J. Hazard. Mater. 2017, 333, 259–264. [Google Scholar] [CrossRef]
- Arfanis, M.K.; Adamou, P.; Moustakas, N.G.; Triantis, T.M.; Kontos, A.G.; Falaras, P. Photocatalytic degradation of salicylic acid and caffeine emerging contaminants using titania nanotubes. Chem. Eng. J. 2017, 130, 525–536. [Google Scholar] [CrossRef]
- Arfanis, M.K.; Athanasekou, C.P.; Sakellis, E.; Boukos, N.; Ioannidis, N.; Likodimos, V.; Sygellou, L.; Bouroushian, M.; Kontos, A.G.; Falaras, P. Photocatalytic properties of copper—Modified core-shell titania Nanocomposites. J. Photochem. Photobiol. A 2019, 370, 145–155. [Google Scholar] [CrossRef]
- Ibrahim, I.; Athanasekou, C.; Manolis, G.; Kaltzoglou, A.; Nasikas, N.K.; Katsaros, F.; Devlin, E.; Kontos, A.G.; Falaras, P. Photocatalysis as an advanced reduction process (ARP): The reduction of 4-nitrophenol using titania nanotubes-ferrite nanocomposites. J. Hazard. Mater. 2019, 372, 37–44. [Google Scholar] [CrossRef]
- Wang, D.; Sun, H.; Luo, Q.; Yang, X.N.; Yin, R. An efficient visible-light photocatalyst prepared from g-C3N4 and polyvinyl chloride. Appl. Catal. B Environ. 2014, 156, 323–330. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, Q.; Chai, G.; Liang, M.; Dong, G.; Zhang, Q.; Qiu, J. Synthesis and luminescence mechanism of multicolor-emitting g-C3N4 nanopowders by low temperature thermal condensation of melamine. Sci. Rep. 2013, 3, 1943. [Google Scholar] [CrossRef]
- Rajagopal, S.; Nataraj, D.; Mangalaraj, D.; Djaoued, Y.; Robichaud, J.; Khyzhun Yu, O. Controlled Growth of WO3 Nanostructures with Three Different Morphologies and Their Structural, Optical, and Photodecomposition Studies. Nanoscale Res. Lett. 2009, 4, 1335–1342. [Google Scholar] [CrossRef]
- Balaji, S.; Djaoued, Y.; Albert, A.S.; Bruning, R.; Beaudoin, N.; Robichaud, J. Porous orthorhombic tungsten oxide thin films: Synthesis, characterization, and application in electrochromic and photochromic devices. J. Mater. Chem. 2011, 21, 3940–3948. [Google Scholar] [CrossRef]
- Daniel, M.F.; Desbat, B.; Lassegues, J.C. Infrared and Raman Study of WO3 Tungsten Trioxides and WO3, xHzO Tungsten Trioxide Hydrates. J. Solid State Chem. 1987, 67, 235–247. [Google Scholar] [CrossRef]
- Santato, C.; Odziemkowski, M.K.; Ulmann, M.; Augustynski, J. Crystallographically Oriented Mesoporous WO3 Films: Synthesis, Characterization, and Applications. J. Am. Chem. Soc. 2001, 123, 10639–10649. [Google Scholar] [CrossRef]
- Delichere, P.; Falaras, P.; Froment, M.; Hugot-Le Goff, A.; Agius, B. Electrochromism in anodic WO3 films: Preparation and physicochemical properties of films in the virgin and coloured states. Thin Solid Films 1988, 161, 35–46. [Google Scholar] [CrossRef]
- Djaoued, Y.; Balaji, S.; Bruning, R. Electrochromic Devices Based on Porous Tungsten Oxide Thin Films. J. Nanomater. 2012, 2012, 7. [Google Scholar] [CrossRef]
- Xu, C.; Wang, X.; Xu, G.; Chen, Y.; Dai, L. Facile construction of leaf-like WO3 nanoflakes decorated on g-C3N4 towards efficient oxidation of alcohols under mild conditions. New J. Chem. 2018, 42, 16523–16532. [Google Scholar] [CrossRef]
- Zinin, P.V.; Ming, L.C.; Sharma, S.K.; Khabashesku, V.N.; Liu, X.; Hong, S.; Endo, S.; Acosta, T. Ultraviolet and near-infrared Raman spectroscopy of graphitic C3N4 phase. Chem. Phys. Lett. 2009, 472, 69–73. [Google Scholar] [CrossRef]
- Giannakopoulou, T.; Papailias, I.; Todorova, N.; Boukos, N.; Liu, Y.; Yu, J.; Trapalis, C. Tailoring the energy band gap and edges’ potentials of g-C3N4/TiO2 composite photocatalysts for NOx removal. Chem. Eng. J. 2017, 310, 571–580. [Google Scholar] [CrossRef]
- Miller, T.S.; Belen Jorge, A.; Suter, T.M.; Sella, A.; Cora, F.; McMillan, P.F. Carbon nitrides: Synthesis and characterization of a new class of functional materials. Phys. Chem. Chem. Phys. 2017, 19, 15613–15638. [Google Scholar] [CrossRef] [PubMed]
- Brady, R.L.; Southmayd, D.; Contescu, C.; Zhang, R.; Schwarz, J.A. Surface Area Determination of Supported Oxides: WO3/AI2O3. J. Catal. 1991, 129, 195–201. [Google Scholar] [CrossRef]
- Moustakas, N.G.; Kontos, A.G.; Likodimos, V.; Katsaros, F.; Boukos, N.; Tsoutsou, D.; Dimoulas, A.; Romanos, G.E.; Dionysiou, D.D.; Falaras, P. Inorganic-organic core-shell titania nanoparticles for efficient visible light activated photocatalysis. Appl. Catal. B Environ. 2013, 130, 14–24. [Google Scholar] [CrossRef]
- Ibrahim, I.; Kaltzoglou, A.; Athanasekou, C.; Katsaros, F.; Devlin, E.; Kontos, A.G.; Ioannidis, N.; Perraki, M.; Tsakiridis, P.; Sygellou, L.; et al. Magnetically separable TiO2/CoFe2O4/Ag nanocomposites for the photocatalytic reduction of hexavalent chromium pollutant under UV and artificial solar light. Chem. Eng. J. 2020, 381, 122730. [Google Scholar] [CrossRef]
- Sharma, S.; Basu, S. Highly reusable visible light active hierarchical porous WO3/SiO2 monolith in centimeter length scale for enhanced photocatalytic degradation of toxic pollutants. Sep. Purif. Technol. 2020, 231, 115916. [Google Scholar] [CrossRef]
- Li, S.T.; Wang, P.F.; Wang, R.D.; Liu, Y.F.; Jing, R.S.; Li, Z.; Meng, Z.L.; Liu, Y.Y.; Zhang, Q. One-step co-precipitation method to construct black phosphorus nanosheets/ZnO nanohybrid for enhanced visible light photocatalytic activity. Appl. Surf. Sci. 2019, 497, 143682. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antoniadou, M.; Arfanis, M.K.; Ibrahim, I.; Falaras, P. Bifunctional g-C3N4/WO3 Thin Films for Photocatalytic Water Purification. Water 2019, 11, 2439. https://doi.org/10.3390/w11122439
Antoniadou M, Arfanis MK, Ibrahim I, Falaras P. Bifunctional g-C3N4/WO3 Thin Films for Photocatalytic Water Purification. Water. 2019; 11(12):2439. https://doi.org/10.3390/w11122439
Chicago/Turabian StyleAntoniadou, Maria, Michalis K. Arfanis, Islam Ibrahim, and Polycarpos Falaras. 2019. "Bifunctional g-C3N4/WO3 Thin Films for Photocatalytic Water Purification" Water 11, no. 12: 2439. https://doi.org/10.3390/w11122439
APA StyleAntoniadou, M., Arfanis, M. K., Ibrahim, I., & Falaras, P. (2019). Bifunctional g-C3N4/WO3 Thin Films for Photocatalytic Water Purification. Water, 11(12), 2439. https://doi.org/10.3390/w11122439