Mapping Dynamics of Bacterial Communities in a Full-Scale Drinking Water Distribution System Using Flow Cytometry
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Sampling
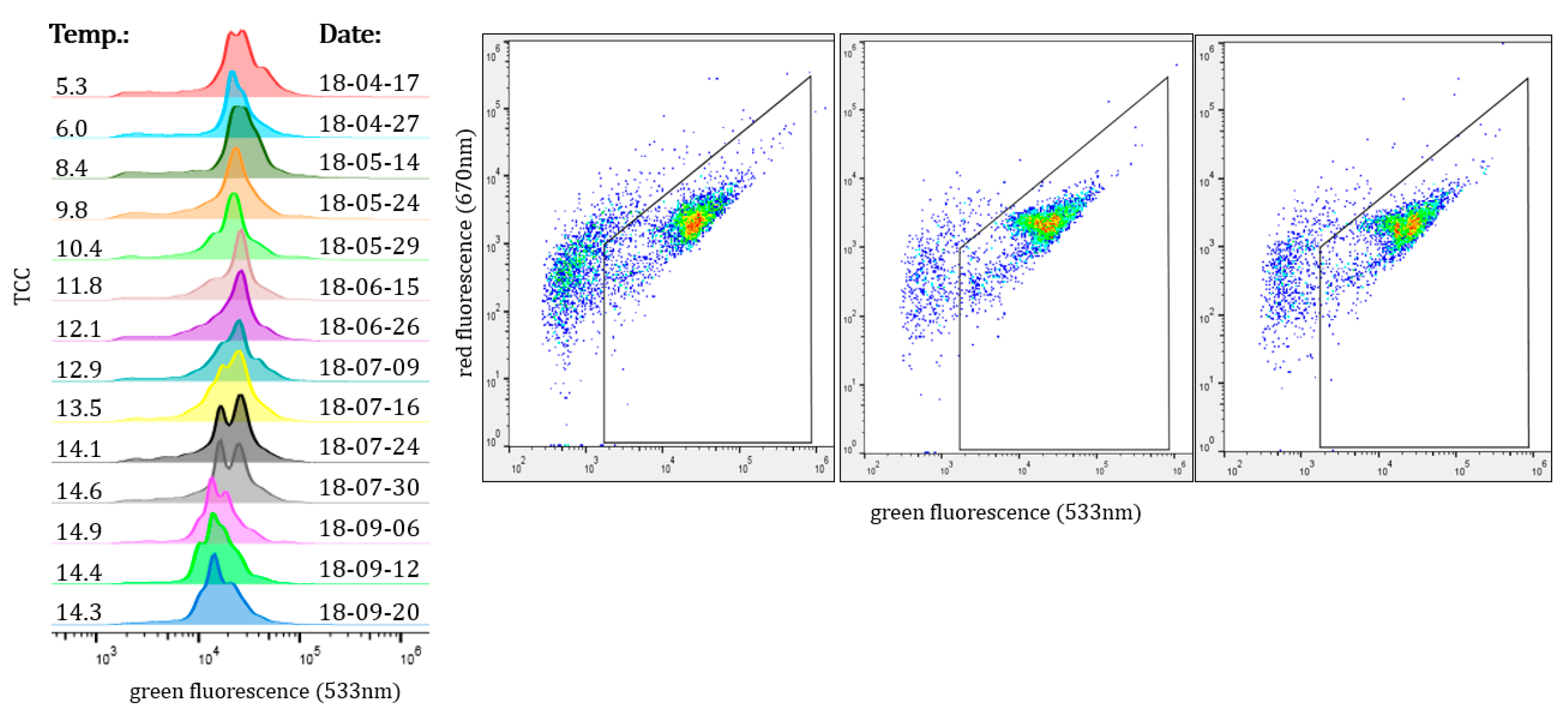
2.2. FCM Analysis
2.3. Other Water Quality Parameters
2.4. Data Analysis
3. Results and Discussion
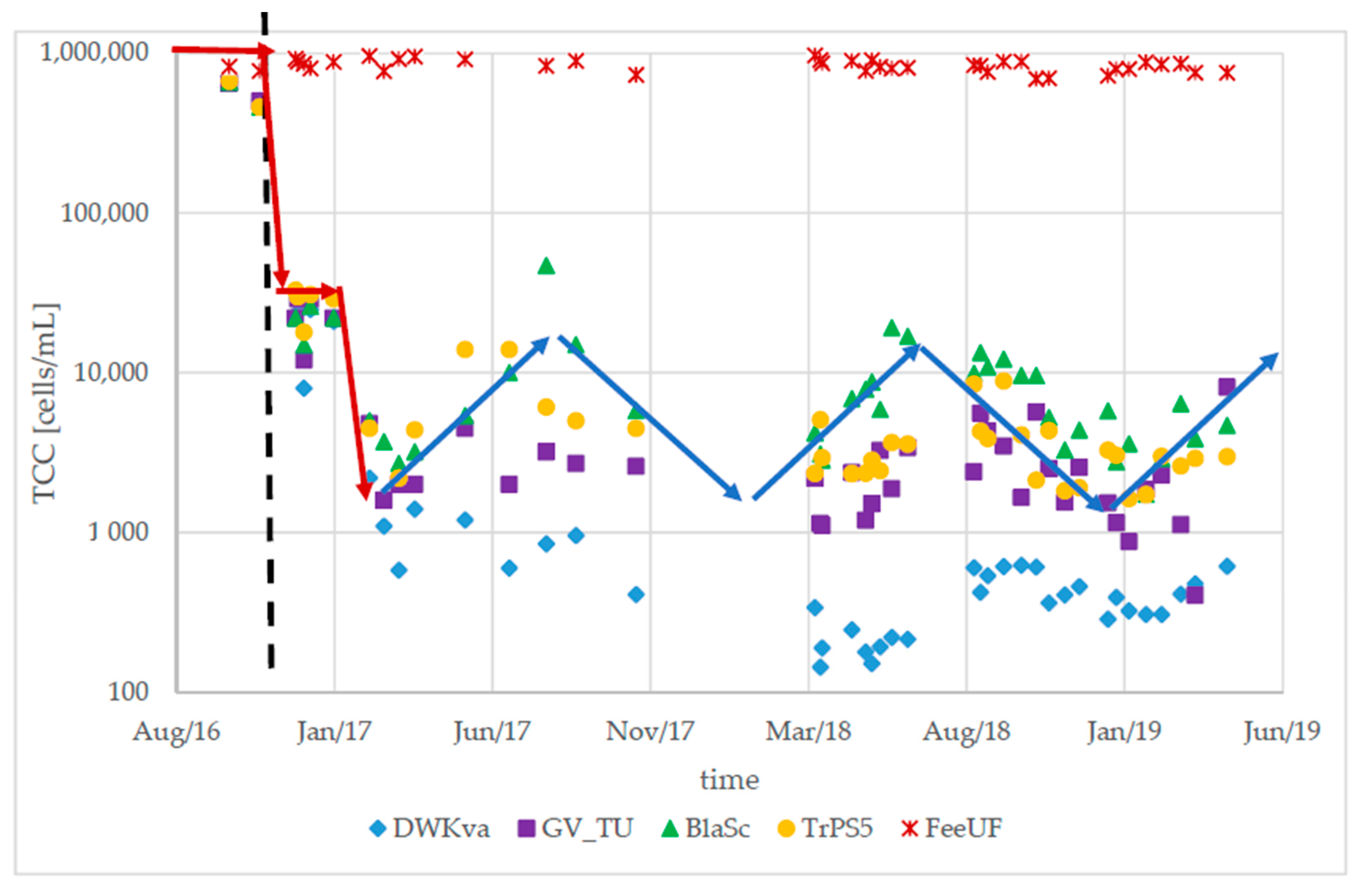
3.1. Long-Term Stability After an Upgrade of the Treatment Process
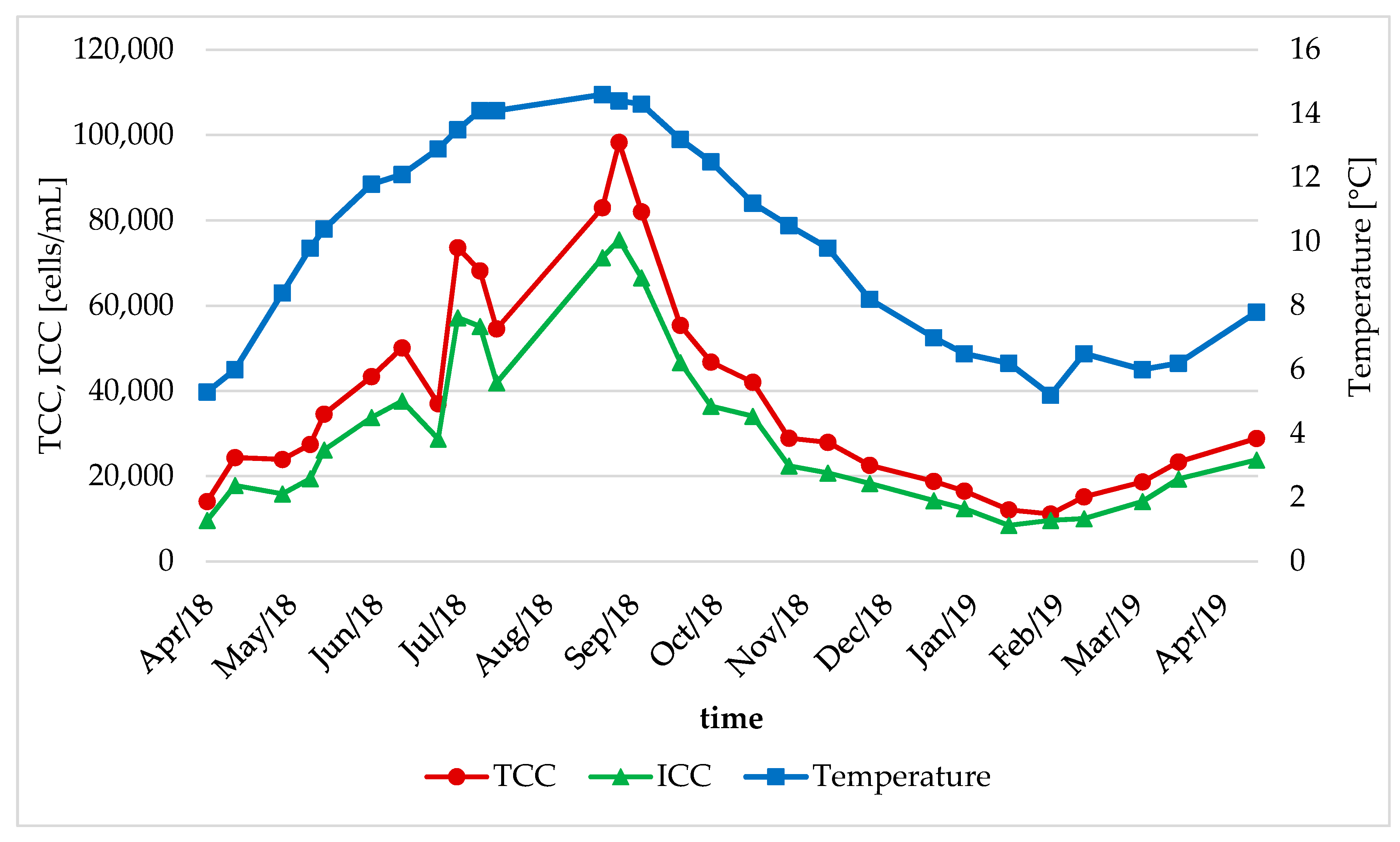
3.2. Seasonal Changes in the Bacteral Community
3.3. Detection of Abnormalities Within the DWDS Using TCC Baselines
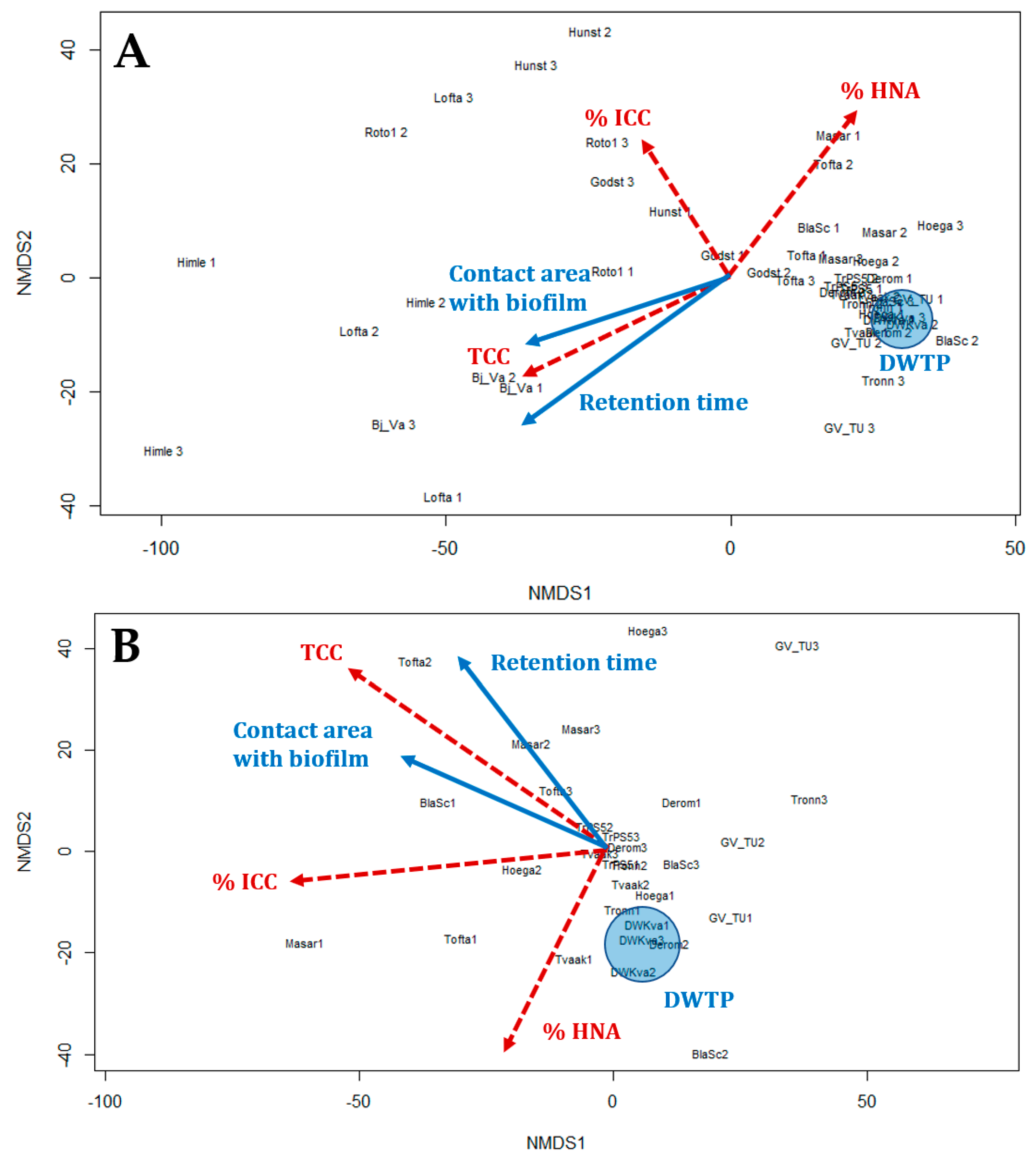
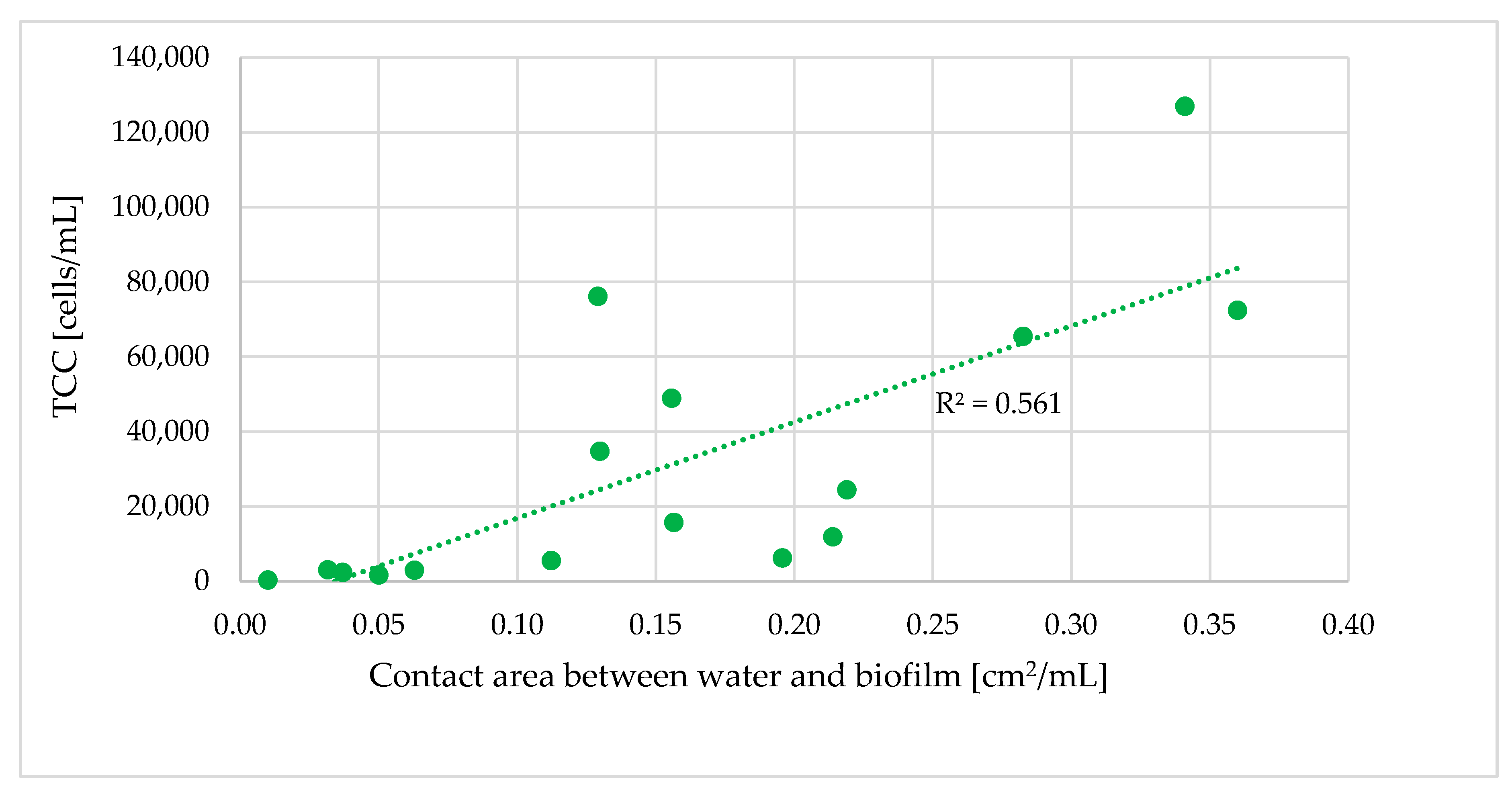
3.4. Relationship of the Bacterial Population to Contact Time with Biofilm
4. Conclusions
- 1.
- The bacterial community in the DWDS experienced clear seasonal changes with similar patterns for different areas of the DWDS. An increase in water temperature led to a significant increase in TCC during the summer period (range: 1.51–5.24-fold increase) at some locations.
- 2.
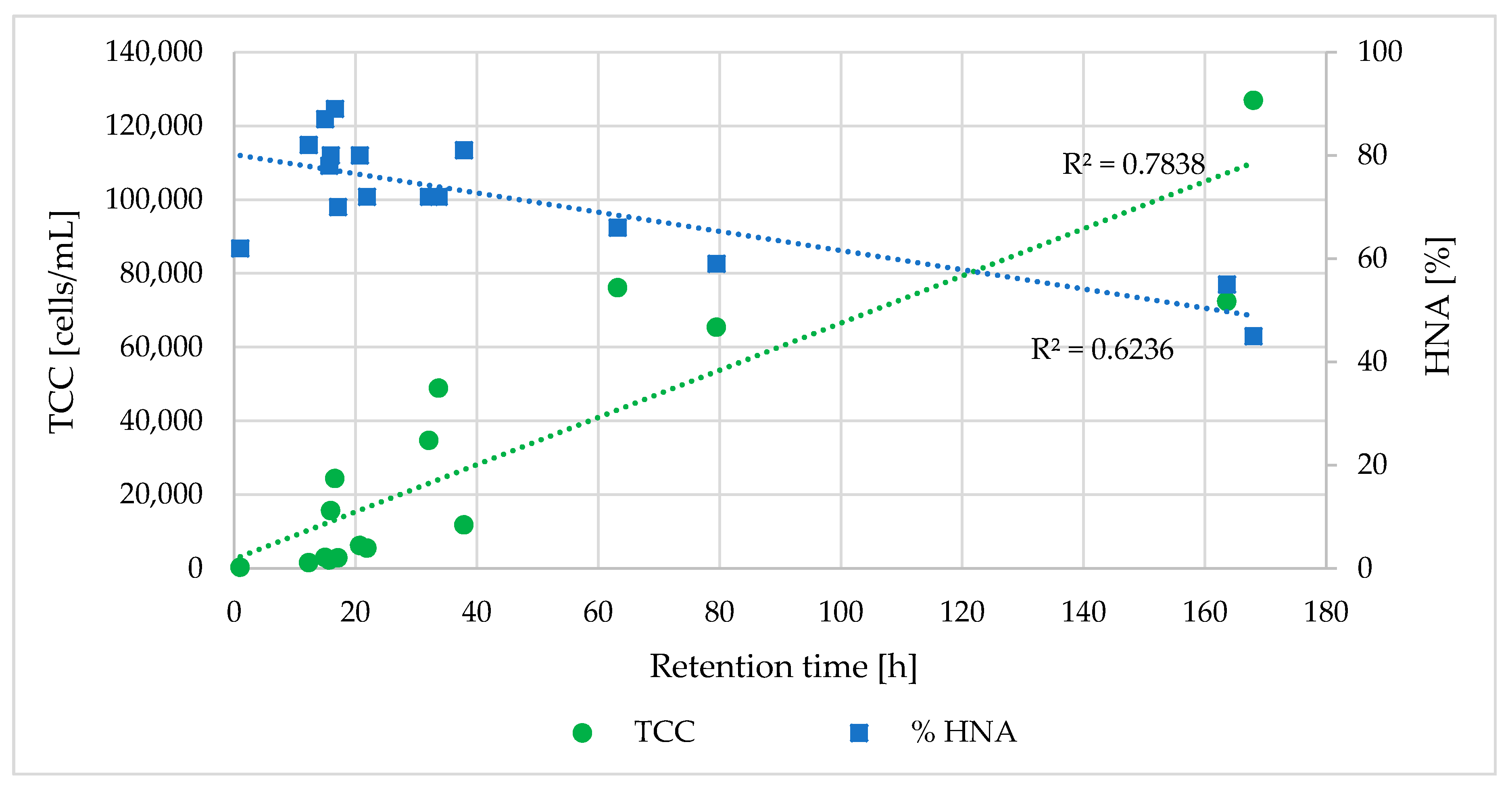
- Hydraulic and specific pipe conditions influence the bacterial community in the water. FCM results indicated that an increase in retention time led to a decrease in the %HNA bacteria in the drinking water (correlation coefficient R = −0.79).
- 3.
- Longer retention times and increased contact between the water and pipe biofilm led to an increase in TCC. Significant differences in the TCC could be seen in different areas of the DWDS depending on distance from the DWTP and retention time.
5. Limitations and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, S. Processes Governing the Drinking Water Microbiome. Ph.D. Thesis, Faculty of Engineering (LTH), Lund, Sweden, 2018. [Google Scholar]
- Chan, S.; Pullerits, K.; Keucken, A.; Persson, K.M.; Paul, C.J.; Rådström, P. Bacterial release from pipe biofilm in a full-scale drinking water distribution system. NPJ Biofilms Microbiomes 2019, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Zhang, Y.; van der Mark, E.; Magic-Knezev, A.; Pinto, A.; van den Bogert, B.; Liu, W.; van der Meer, W.; Medema, G. Assessing the origin of bacteria in tap water and distribution system in an unchlorinated drinking water system by SourceTracker using microbial community fingerprints. Water Res. 2018, 138, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Waak, M.B.; Hozalski, R.M.; Hallé, C.; Lapara, T.M. Comparison of the microbiomes of two drinking water distribution systems-With and without residual chloramine disinfection. Microbiome 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Feron, D.; Neumann, E. “Biocorrosion 2012”—From advanced technics towards scientific perspectives. Bioelectrochemistry 2014, 97, 1. [Google Scholar] [CrossRef]
- Piriou, P.; Malleret, L.; Bruchet, A.; Kiéné, L. Trichloroanisole kinetics and musty tastes in drinking water distribution systems. Water Supply 2001, 1, 11–18. [Google Scholar] [CrossRef]
- Van der Kooij, D.; van der Wielen, P.W.J.J. Microbial Growth in Drinking-Water Supplies: Problems, Causes, Control and Research Needs, 1st ed.; IWA Publishing: London, UK, 2013; pp. 1–32. [Google Scholar]
- Baskerville, A.; Broster, M.; Fitzgeorge, R.B.; Hambleton, P.; Dennis, P.J. Experimental Transmission of Legionnaires’ Disease by Exposure to Aerosols of Legionella Pneumophila. Lancet 1981, 318, 1389–1390. [Google Scholar] [CrossRef]
- Bartram, J.; Cotruvo, J.A.; Exner, M.; Fricker, C.; Glasmacher, A. Heterotrophic Plate Counts and Drinking-Water Safety-The Significance of HPCs for Water Quality and Human Health, 1st ed.; IWA Publishing: London, UK, 2003; pp. 80–118. [Google Scholar]
- Pinto, A.J.; Xi, C.; Raskin, L. Bacterial community structure in the drinking water microbiome is governed by filtration processes. Environ. Sci. Technol. 2012, 46, 8851–8859. [Google Scholar] [CrossRef]
- Hijnen, W.A.M.; Beerendonk, E.F.; Medema, G.J. Inactivation credit of UV radiation for viruses, bacteria and protozoan (oo)cysts in water: A review. Water Res. 2006, 40, 3–22. [Google Scholar] [CrossRef]
- Douterelo, I.; Boxall, J.B.; Deines, P.; Sekar, R.; Fish, K.E.; Biggs, C.A. Methodological approaches for studying the microbial ecology of drinking water distribution systems. Water Res. 2014, 65, 134–156. [Google Scholar] [CrossRef]
- Hammes, F.; Berney, M.; Wang, Y.; Vital, M.; Köster, O.; Egli, T. Flow-cytometric total bacterial cell counts as a descriptive microbiological parameter for drinking water treatment processes. Water Res. 2008, 42, 269–277. [Google Scholar] [CrossRef]
- Lautenschlager, K.; Boon, N.; Wang, Y.; Egli, T.; Hammes, F. Overnight stagnation of drinking water in household taps induces microbial growth and changes in community composition. Water Res. 2010, 44, 4868–4877. [Google Scholar] [CrossRef] [PubMed]
- Payment, P.; Trudel, M.; Plante, R. Elimination of viruses and indicator bacteria at each step of treatment during preparation of drinking water at seven water treatment plants. Appl. Environ. Microbiol. 1985, 49, 1418–1428. [Google Scholar] [PubMed]
- Van Nevel, S.; Buysschaert, B.; De Gusseme, B.; Boon, N. Flow cytometric examination of bacterial growth in a local drinking water network. Water Environ. J. 2016, 30, 167–176. [Google Scholar] [CrossRef]
- Lautenschlager, K.; Hwang, C.; Liu, W.T.; Boon, N.; Köster, O.; Vrouwenvelder, H.; Egli, T.; Hammes, F. A microbiology-based multi-parametric approach towards assessing biological stability in drinking water distribution networks. Water Res. 2013, 47, 3015–3025. [Google Scholar] [CrossRef]
- Van Nevel, S.; Koetzsch, S.; Proctor, C.R.; Besmer, M.D.; Prest, E.I.; Vrouwenvelder, J.S.; Knezev, A.; Boon, N.; Hammes, F. Flow cytometric bacterial cell counts challenge conventional heterotrophic plate counts for routine microbiological drinking water monitoring. Water Res. 2017, 113, 191–206. [Google Scholar] [CrossRef]
- Gatza, E.; Hammes, F.A.; Prest, E.I. White Paper: Assessing Water Quality with the BD Accuri TM C 6 Flow Cytometer. 2013. Available online: https://www.semanticscholar.org/paper/White-Paper-Assessing-Water-Quality-with-the-BD-TM-Gatza-Hammes/8e96101b49bfcb09e28425cf40c0cce2dec1afca (accessed on 18 July 2019).
- Besmer, M.D.; Sigrist, J.A.; Props, R.; Buysschaert, B.; Mao, G.; Boon, N.; Hammes, F. Laboratory-scale simulation and real-time tracking of a microbial contamination event and subsequent shock-chlorination in drinking water. Front. Microbiol. 2017, 8, 1900. [Google Scholar] [CrossRef]
- Keucken, A.; Heinicke, G.; Persson, K.M.; Köhler, S.J. Combined coagulation and ultrafiltration process to counteract increasing NOM in brown surface water. Water 2017, 9, 697. [Google Scholar] [CrossRef]
- Prest, E.I.; Hammes, F.; Kötzsch, S.; van Loosdrecht, M.C.M.; Vrouwenvelder, J.S. Monitoring microbiological changes in drinking water systems using a fast and reproducible flow cytometric method. Water Res. 2013, 47, 7131–7142. [Google Scholar] [CrossRef]
- Chan, S.; Pullerits, K.; Riechelmann, J.; Persson, K.M.; Rådström, P.; Paul, C.J. Monitoring biofilm function in new and matured full-scale slow sand filters using flow cytometric histogram image comparison (CHIC). Water Res. 2018, 138, 27–36. [Google Scholar] [CrossRef]
- Koch, C.; Fetzer, I.; Harms, H.; Müller, S. CHIC-an automated approach for the detection of dynamic variations in complex microbial communities. Cytom. Part A 2013, 83, 561–567. [Google Scholar] [CrossRef]
- Ellis, B.; Haaland, P.; Hahne, F.; Meur, N.L.; Gopalakrishnan, N.; Spidlen, J.; Jiang, M. FlowCore: Basic Structures for Flow Cytometry Data, Bioconductor R. Package Version 1.40.0. 2016. Available online: https://rdrr.io/bioc/flowCore/ (accessed on 7 August 2019).
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.B.; Simpson, G.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.2-1. 2015. Available online: http://CRAN.R-project.org/package=vegan (accessed on 14 August 2019).
- Keucken, A. Climate Change Adaption of Waterworks for Browning Surface Waters: Nano- and Ultrafiltration Membrane Applications for Drinking Water Treatment. Ph.D. Thesis, Faculty of Engineering (LTH), Lund, Sweden, 2017. [Google Scholar]
- Pinto, A.J.; Schroeder, J.; Lunn, M.; Sloan, W.; Raskin, L. Spatial-temporal survey and occupancy-abundance modeling to predict bacterial community dynamics in the drinking water microbiome. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Gunawan, C.; Barraud, N.; Rice, S.A.; Harry, E.J.; Amal, R. Understanding, monitoring, and controlling biofilm growth in drinking water distribution systems. Environ. Sci. Technol. 2016, 50, 8954–8976. [Google Scholar] [CrossRef] [PubMed]
- Prest, E.I.; Weissbrodt, D.G.; Hammes, F.; Van Loosdrecht, M.C.M.; Vrouwenvelder, J.S. Long-term bacterial dynamics in a full-scale drinking water distribution system. PLoS ONE 2016, 11, e0164445. [Google Scholar] [CrossRef] [PubMed]
- Douterelo, I.; Sharpe, R.L.; Boxall, J.B. Influence of hydraulic regimes on bacterial community structure and composition in an experimental drinking water distribution system. Water Res. 2013, 47, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Lipphaus, P.; Hammes, F.; Kötzsch, S.; Green, J.; Gillespie, S.; Nocker, A. Microbiological tap water profile of a medium-sized building and effect of water stagnation. Environ. Technol. 2014, 35, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Chiao, T.H.; Clancy, T.M.; Pinto, A.; Xi, C.; Raskin, L. Differential resistance of drinking water bacterial populations to monochloramine disinfection. Environ. Sci. Technol. 2014, 48, 4038–4047. [Google Scholar] [CrossRef]
- Boe-Hansen, R.; Albrechtsen, H.J.; Arvin, E.; Jørgensen, C. Bulk water phase and biofilm growth in drinking water at low nutrient conditions. Water Res. 2002, 36, 4477–4486. [Google Scholar] [CrossRef]
- Universität Göttingen. Available online: https://lp.uni-goettingen.de/get/text/4908 (accessed on 17 June 2019).
- Liu, J.; Zhao, Z.; Chen, C.; Cao, P.; Wang, Y. In-situ features of LNA and HNA bacteria in branch ends of drinking water distribution systems. J. Water Supply Res. Technol. AQUA 2017, 66, 300–307. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Hammes, F.; Boon, N.; Chami, M.; Egli, T. Isolation and characterization of low nucleic acid (LNA)-content bacteria. ISME J. 2009, 3, 889–902. [Google Scholar] [CrossRef]
- Ren, H.; Wang, W.; Liu, Y.; Liu, S.; Lou, L.; Cheng, D.; He, X.; Zhou, X.; Qiu, S.; Fu, L.; et al. Pyrosequencing analysis of bacterial communities in biofilms from different pipe materials in a city drinking water distribution system of East China. Appl. Microbiol. Biotechnol. 2015, 99, 10713–10724. [Google Scholar] [CrossRef]
- Henne, K.; Kahlisch, L.; Brettar, I.; Höfle, M.G. Analysis of structure and composition of bacterial core communities in mature drinking water biofilms and bulk water of a citywide network in Germany. Appl. Environ. Microbiol. 2012, 78, 3530–3538. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Harrington, G.W.; Xagoraraki, I.; Goel, R. Factors affecting bulk to total bacteria ratio in drinking water distribution systems. Water Res. 2008, 42, 3393–3404. [Google Scholar] [CrossRef] [PubMed]
- Medema, G.J.; Smeets, P.W.; Blokker, E.J.; van Lieverloo, J.H. Safe distribution without a disinfectant residual. In Microbial Growth in Drinking-Water Supplies: Problems, Causes, Control and Research Needs, 1st ed.; van der Kooij, D., van der Wielen, P.W.J.J., Eds.; IWA Publishing: London, UK, 2013; pp. 95–125. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schleich, C.; Chan, S.; Pullerits, K.; Besmer, M.D.; Paul, C.J.; Rådström, P.; Keucken, A. Mapping Dynamics of Bacterial Communities in a Full-Scale Drinking Water Distribution System Using Flow Cytometry. Water 2019, 11, 2137. https://doi.org/10.3390/w11102137
Schleich C, Chan S, Pullerits K, Besmer MD, Paul CJ, Rådström P, Keucken A. Mapping Dynamics of Bacterial Communities in a Full-Scale Drinking Water Distribution System Using Flow Cytometry. Water. 2019; 11(10):2137. https://doi.org/10.3390/w11102137
Chicago/Turabian StyleSchleich, Caroline, Sandy Chan, Kristjan Pullerits, Michael D. Besmer, Catherine J. Paul, Peter Rådström, and Alexander Keucken. 2019. "Mapping Dynamics of Bacterial Communities in a Full-Scale Drinking Water Distribution System Using Flow Cytometry" Water 11, no. 10: 2137. https://doi.org/10.3390/w11102137
APA StyleSchleich, C., Chan, S., Pullerits, K., Besmer, M. D., Paul, C. J., Rådström, P., & Keucken, A. (2019). Mapping Dynamics of Bacterial Communities in a Full-Scale Drinking Water Distribution System Using Flow Cytometry. Water, 11(10), 2137. https://doi.org/10.3390/w11102137




