Recent Progresses of Forward Osmosis Membranes Formulation and Design for Wastewater Treatment
Abstract
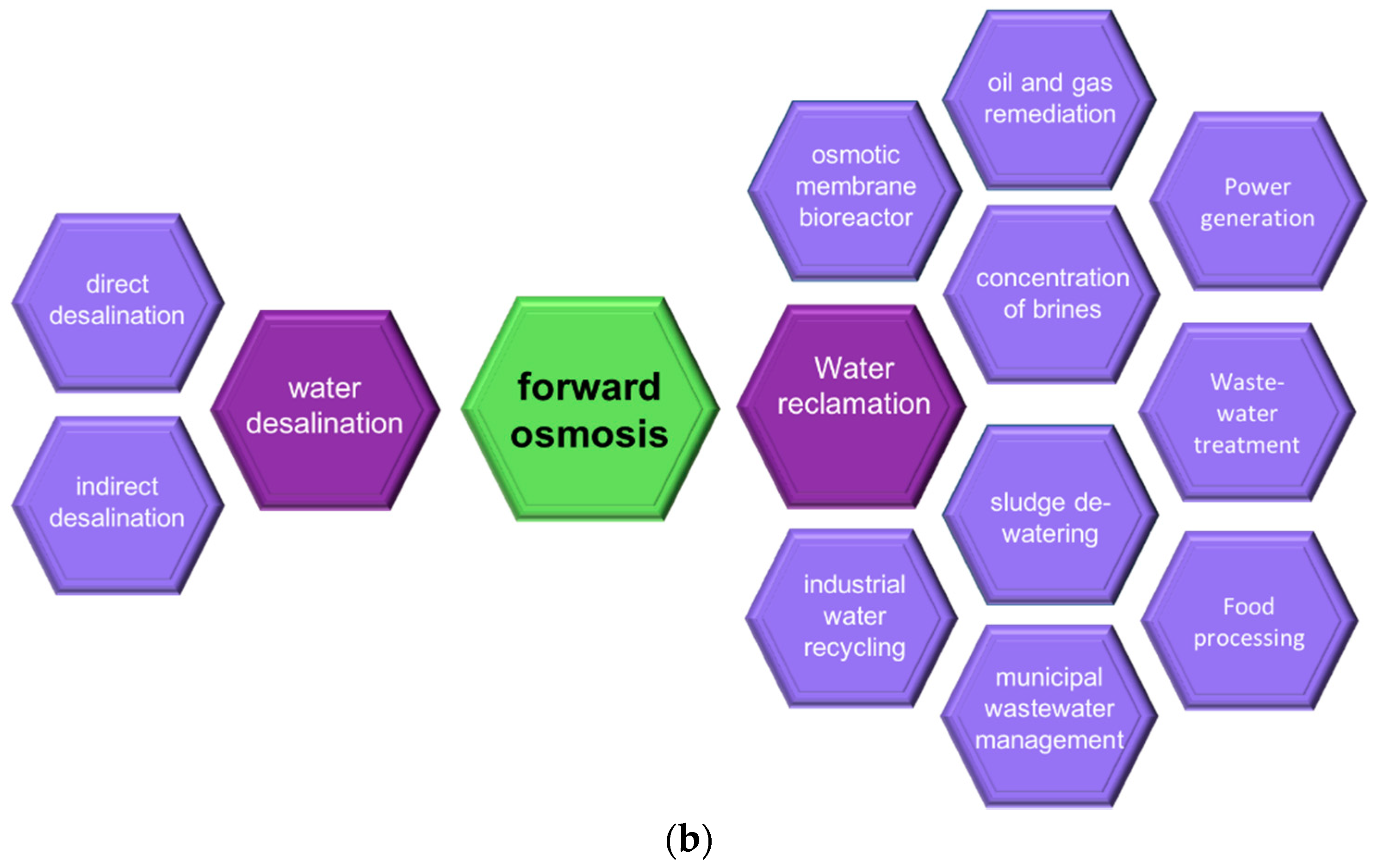
1. Introduction
2. Forward Osmosis—An Overview
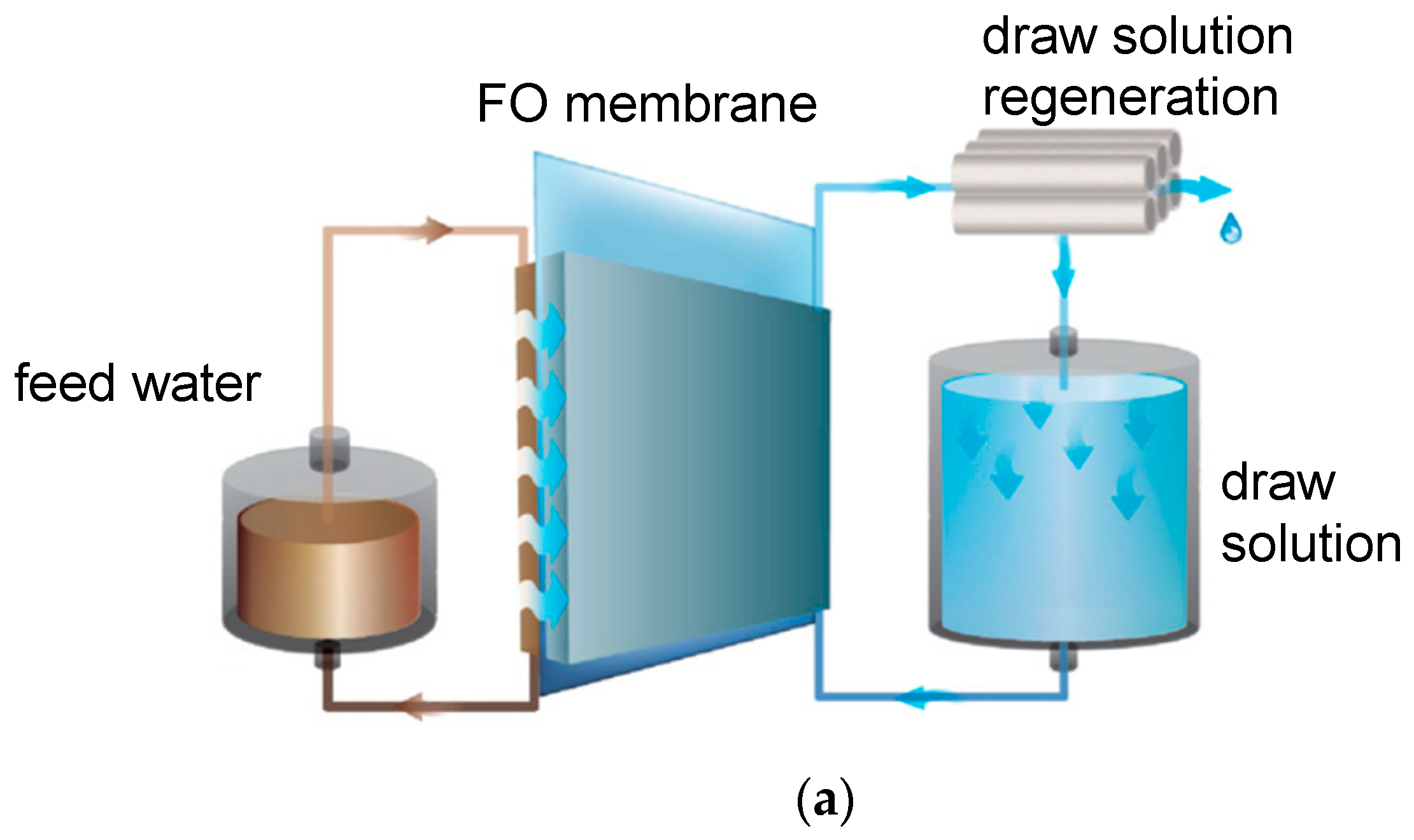
2.1. Principle in Brief
2.2. Forward Osmosis Membranes
2.3. Challenges of Forward Osmosis Processes
3. Approaches in Forward Osmosis Membrane Fabrication and Modifications
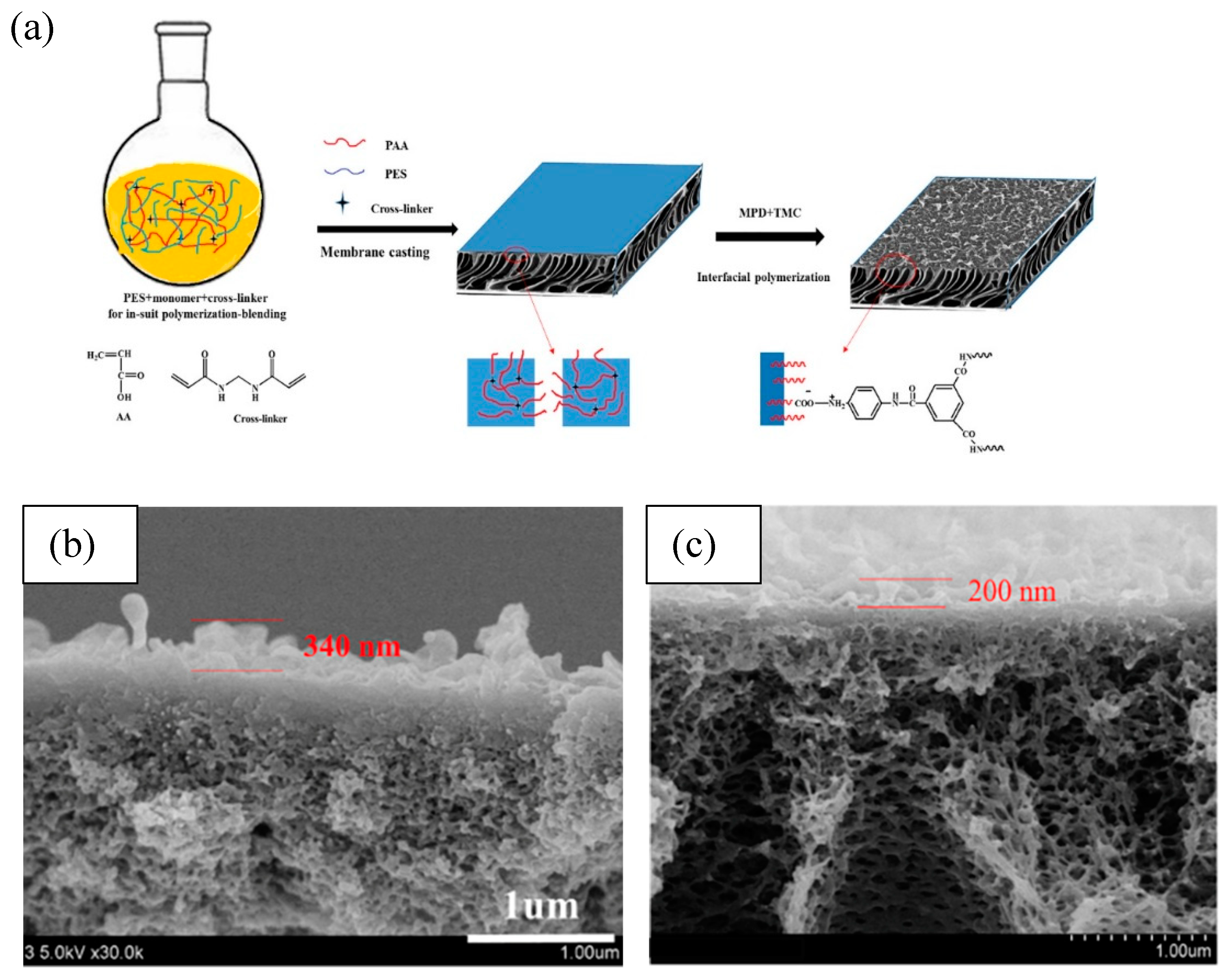
3.1. Modifications of Microporous Substrate
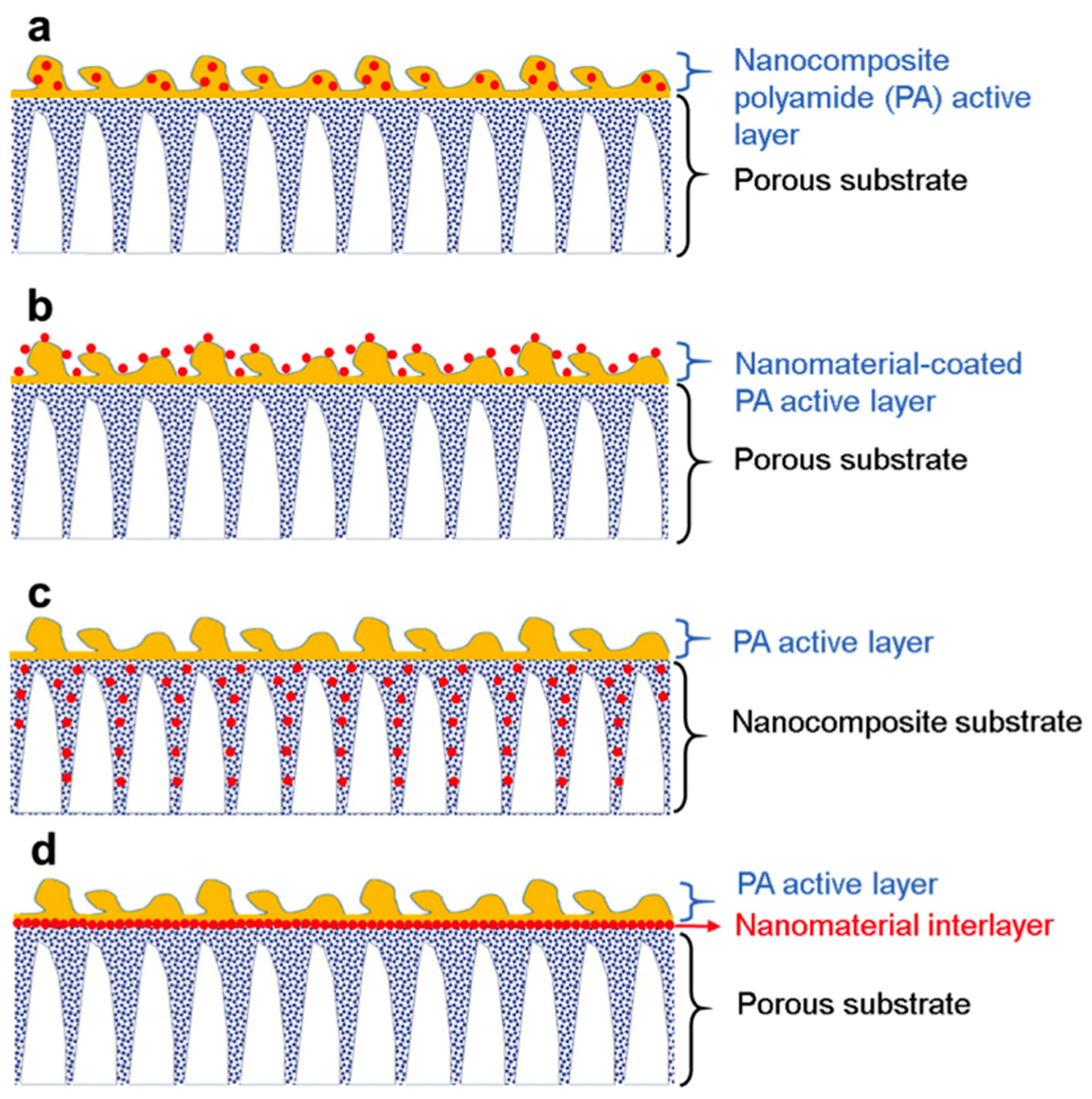
3.2. Incorporation of Nanomaterials
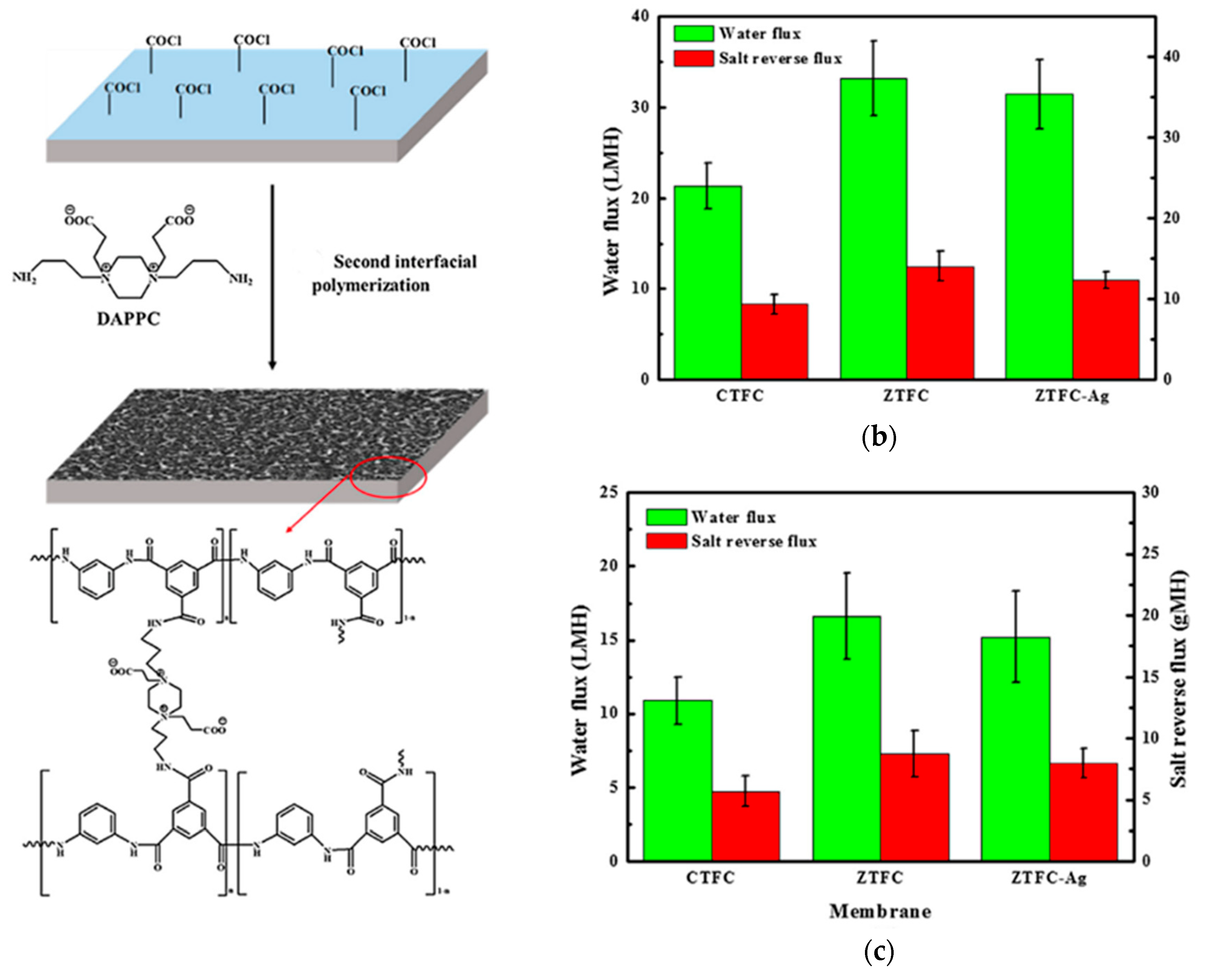
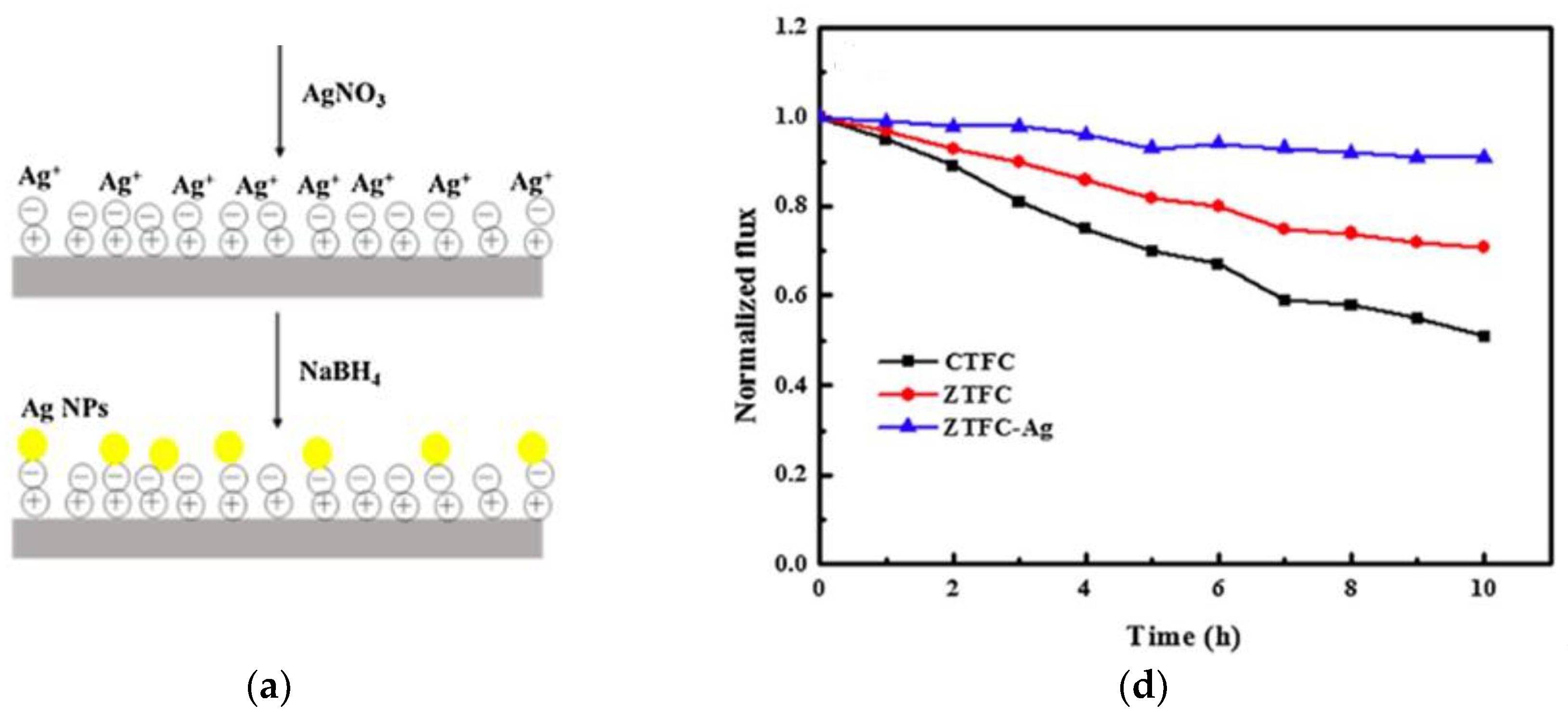
3.3. Double-Skinned Thin Film Composite
3.4. Layer-By-Layer Assembly
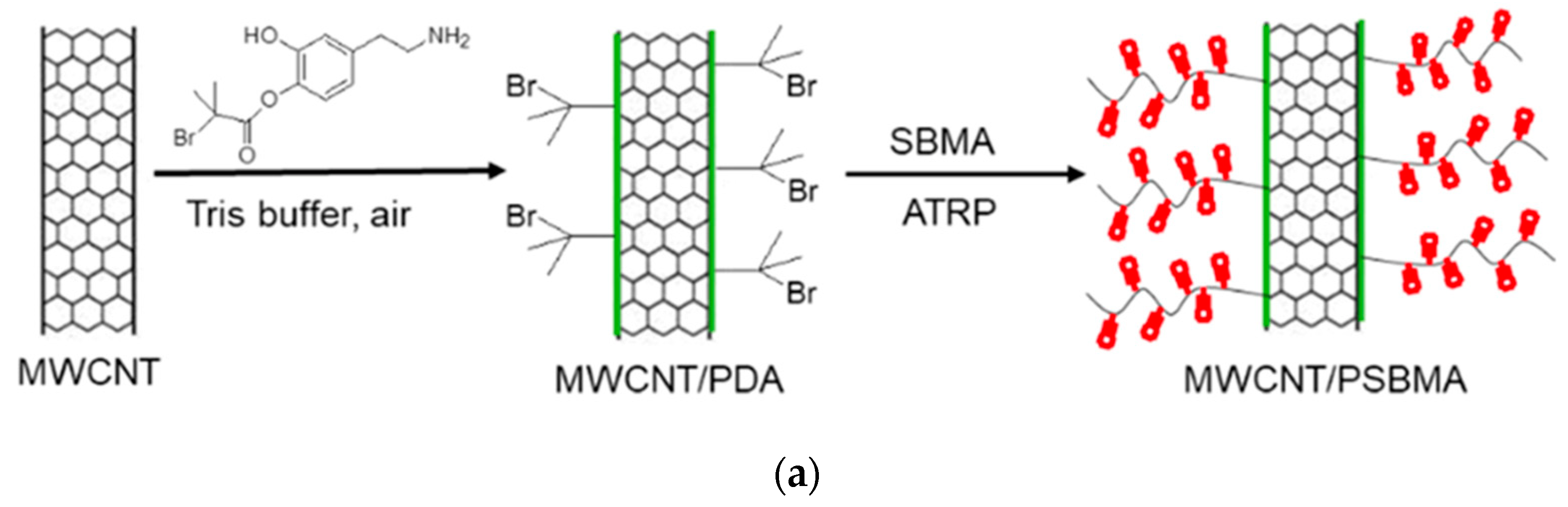
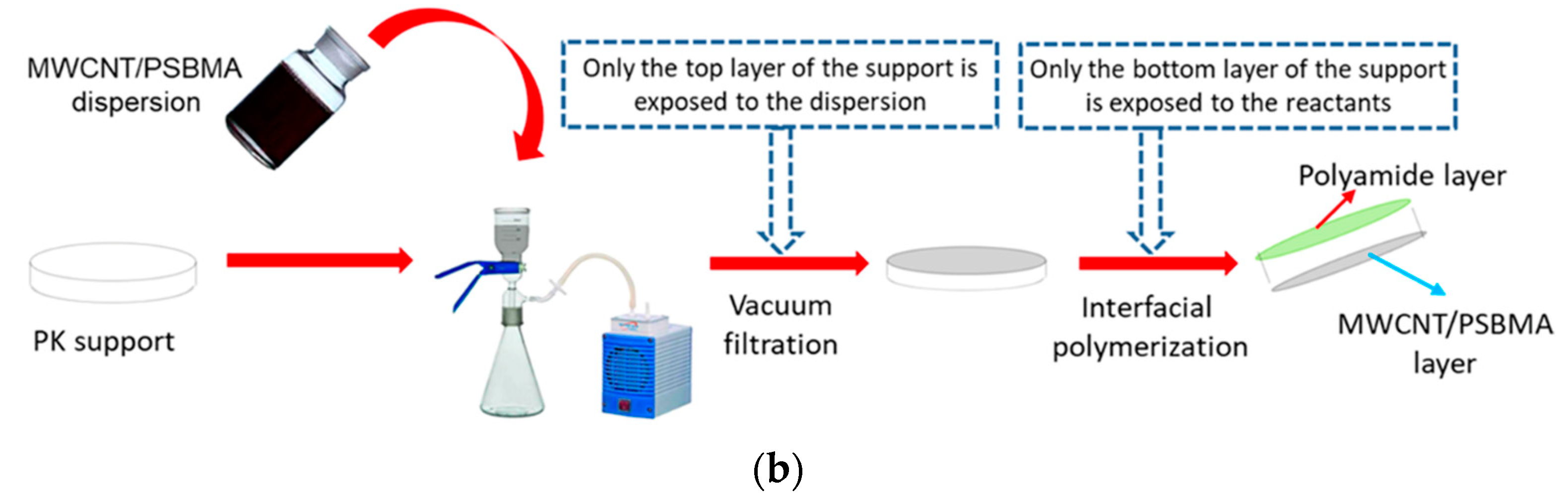
3.5. Surface Grafting
4. Wastewater Treatment with Forward Osmosis
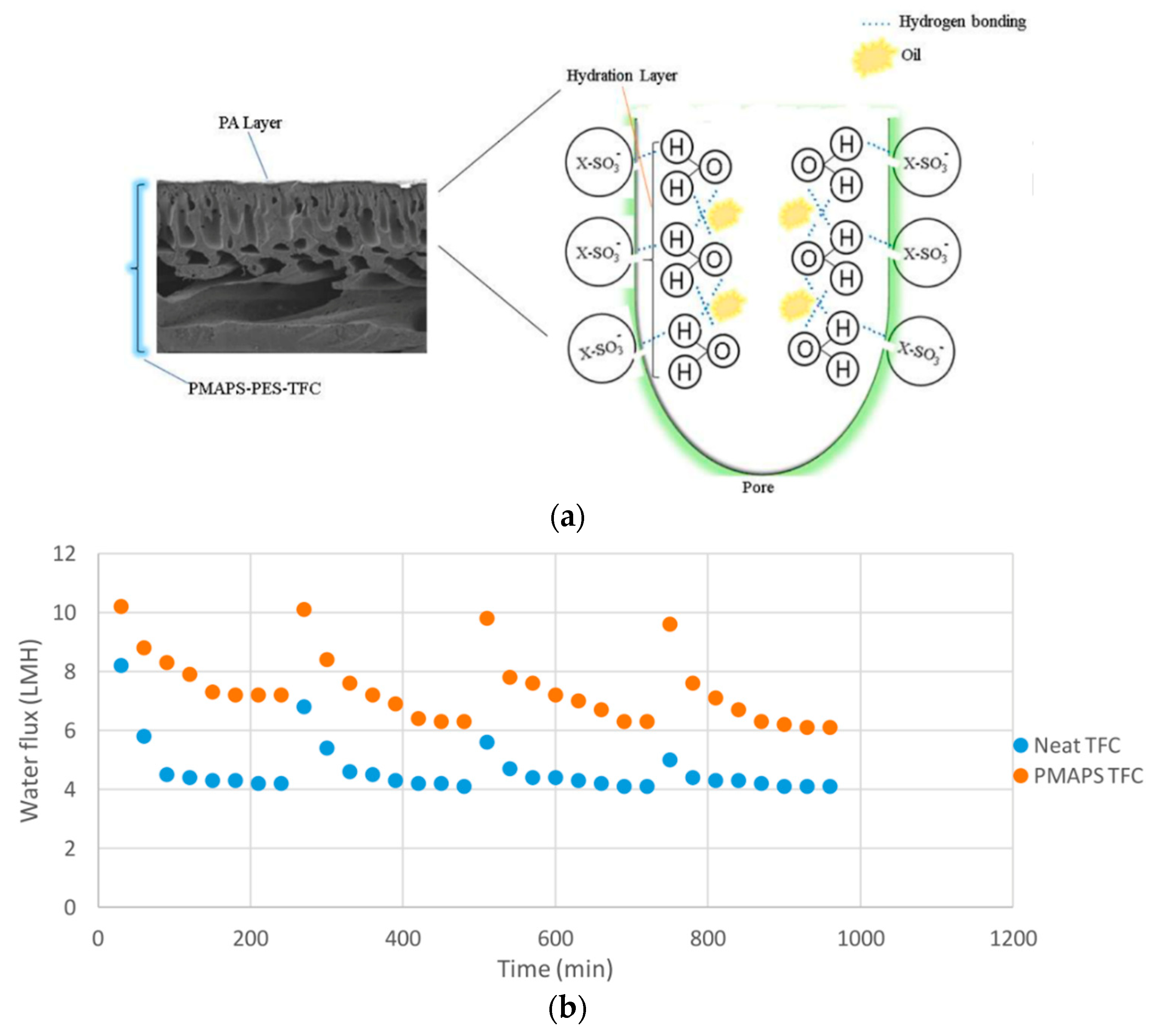
4.1. Oily Wastewater Treatment
4.2. Heavy Metal Ions Removal
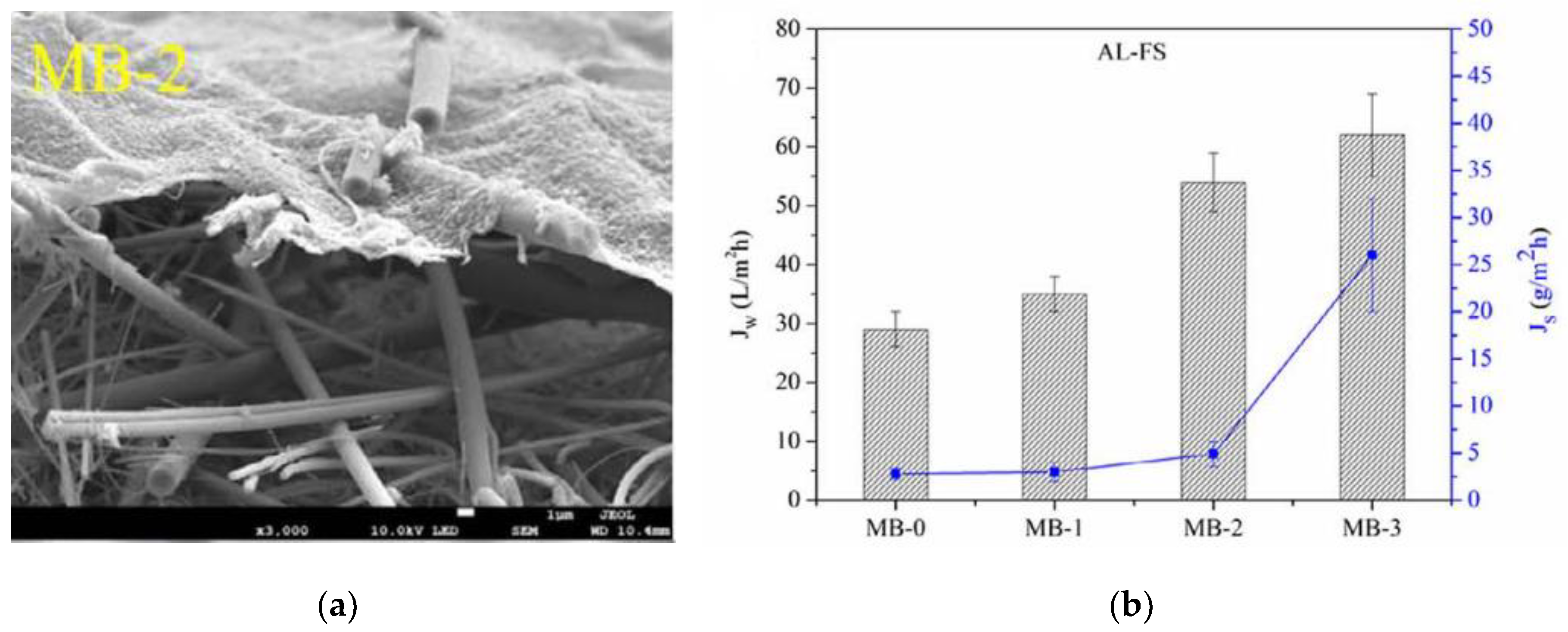
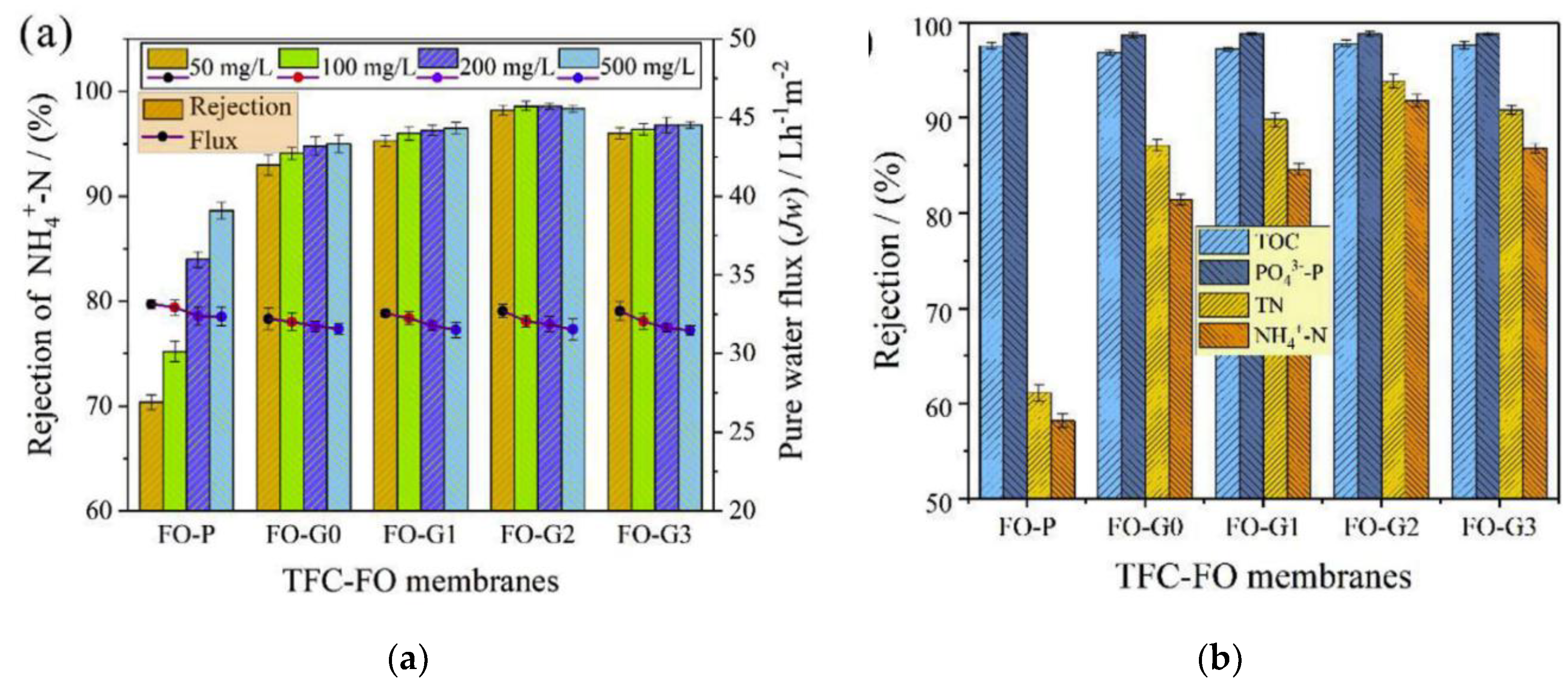
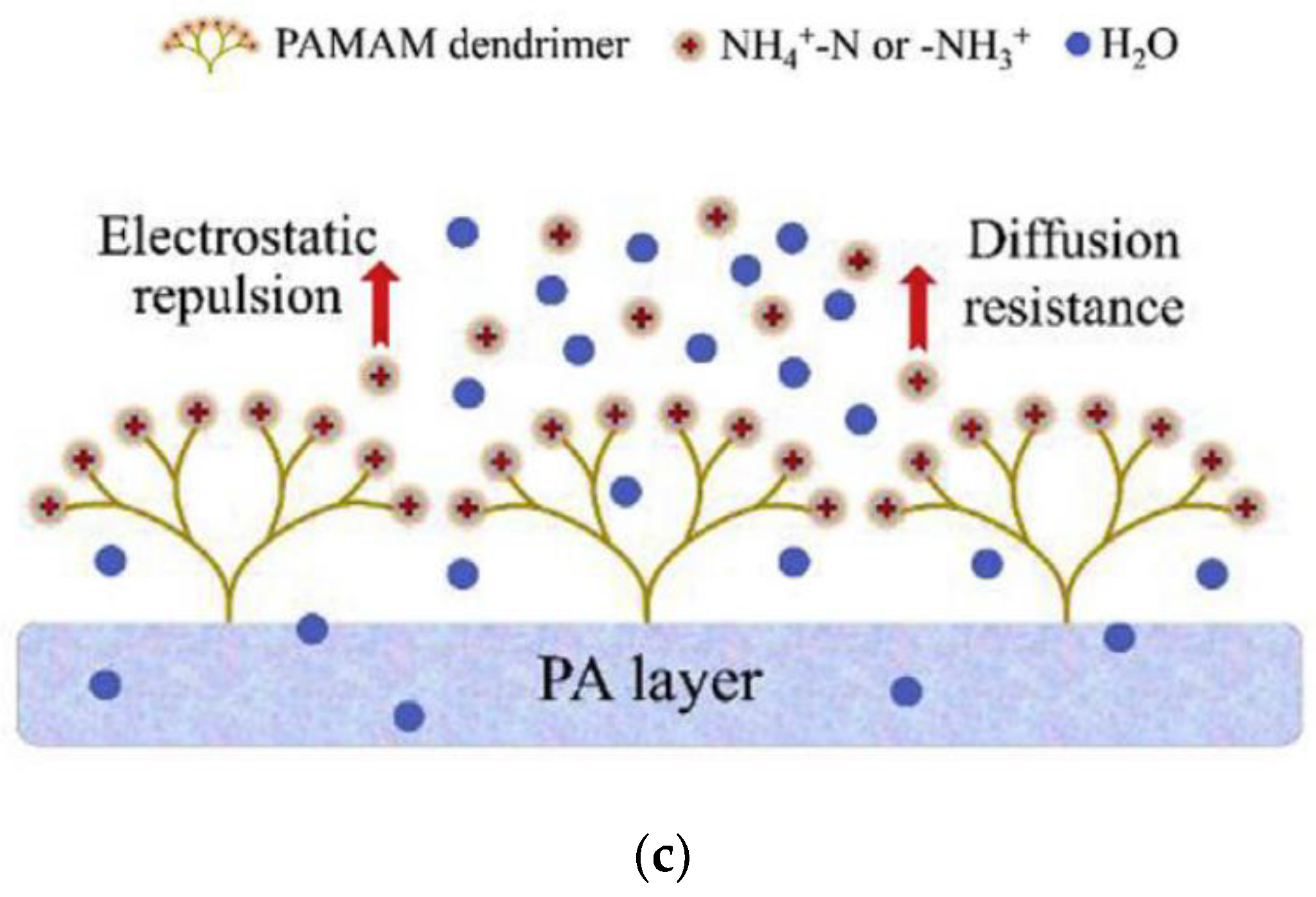
4.3. Municipal Wastewater Treatment
4.4. Contaminants of Emerging Concerns Removal
4.5. Radioactive Wastewater Treatment
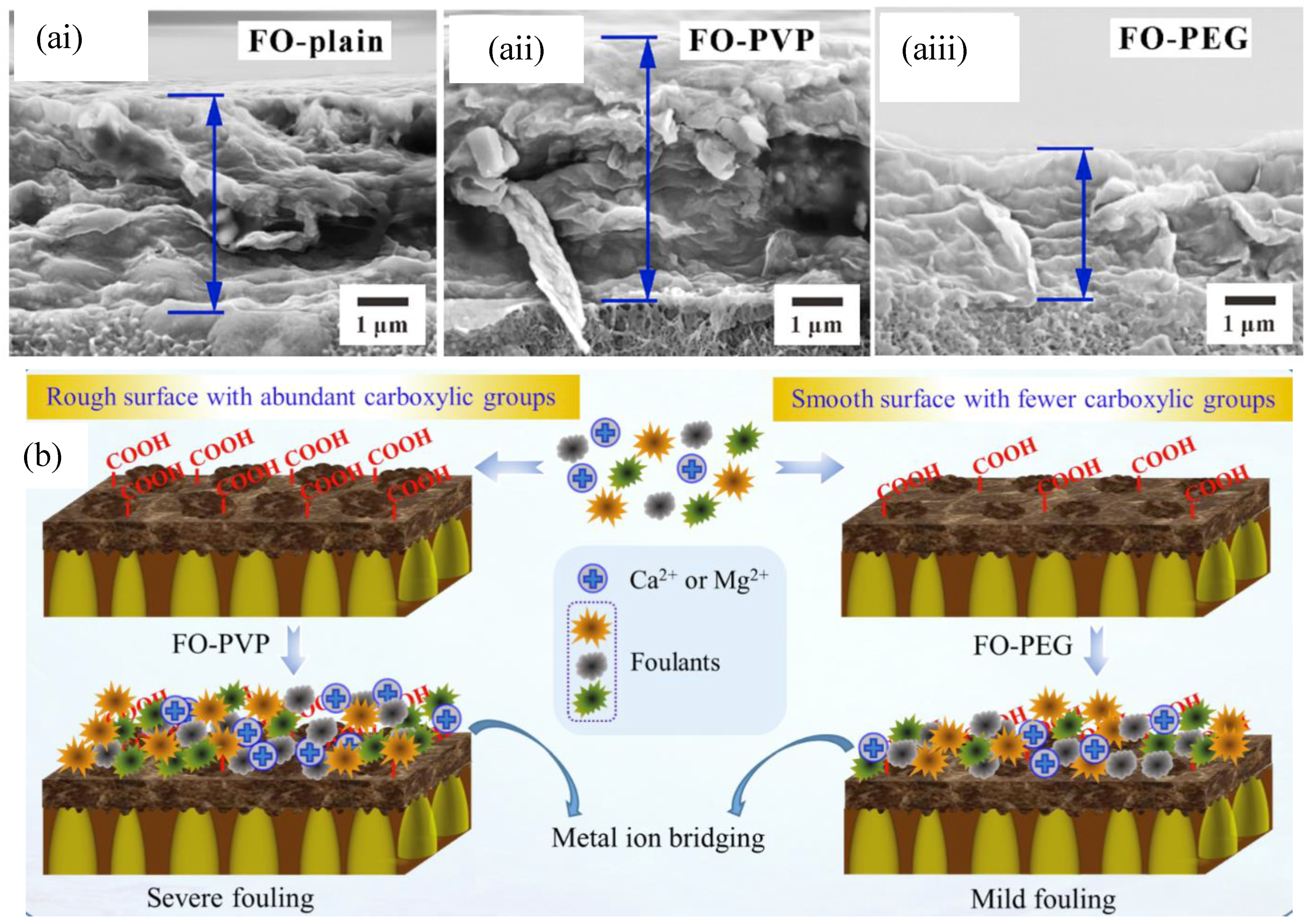
5. Fouling Control Strategies in Forward Osmosis for Wastewater Treatment
6. Current Challenges and Future Outlook
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Goh, P.S.; Matsuura, T.; Ismail, A.F.; Ng, B.C. The Water-Energy Nexus: Solutions towards Energy-Efficient Desalination. Energy Technol. 2017, 2. [Google Scholar] [CrossRef]
- Vareda, J.P.; Valente, A.J.M.; Durães, L. Assessment of heavy metal pollution from anthropogenic activities and remediation strategies: A review. J. Environ. Manag. 2019, 246, 101–118. [Google Scholar] [CrossRef] [PubMed]
- Salgot, M.; Folch, M. Wastewater treatment and water reuse. Curr. Opin. Environ. Sci. Heal. 2018, 2, 64–74. [Google Scholar] [CrossRef]
- Li, W.-W.; Yu, H.; Rittmann, B.E. Chemistry: Reuse water pollutants. Nature 2015, 528, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Yan, X.; Zhu, Q.; Liao, C. The utilization of reclaimed water: Possible risks arising from waterborne contaminants. Environ. Pollut. 2019, 254, 113020. [Google Scholar] [CrossRef] [PubMed]
- Rajasulochana, P.; Preethy, V. Comparison on efficiency of various techniques in treatment of waste and sewage water—A comprehensive review. Resour. Technol. 2016, 2, 175–184. [Google Scholar] [CrossRef]
- Pangarkar, B.L.; Sane, M.G.; Parjane, S.B.; Guddad, M. Status of membrane distillation for water and wastewater treatment—A review. Desalin. Water Treat. 2014, 52, 5199–5218. [Google Scholar] [CrossRef]
- Mohammad, A.W.; Teow, Y.H.; Ang, W.L.; Chung, Y.T.; Oatley-Radcliffe, D.L.; Hilal, N. Nanofiltration membranes review: Recent advances and future prospects. Desalination 2015, 356, 226–254. [Google Scholar] [CrossRef]
- Al-Obaidi, M.A.; Kara-Zaïtri, C.; Mujtaba, I.M. Wastewater treatment by spiral wound reverse osmosis: Development and validation of a two dimensional process model. J. Clean. Prod. 2017, 140, 1429–1443. [Google Scholar] [CrossRef]
- Goh, P.S.; Matsuura, T.; Ismail, A.F.; Hilal, N. Recent trends in membranes and membrane processes for desalination. Desalination 2016, 391. [Google Scholar] [CrossRef]
- Kim, J.; Park, K.; Yang, D.R.; Hong, S. A comprehensive review of energy consumption of seawater reverse osmosis desalination plants. Appl. Energy 2019, 254, 113652. [Google Scholar] [CrossRef]
- Subramani, A.; Jacangelo, J.G. Emerging desalination technologies for water treatment: A critical review. Water Res. 2015, 75, 164–187. [Google Scholar] [CrossRef] [PubMed]
- Coday, B.D.; Xu, P.; Beaudry, E.G.; Herron, J.; Lampi, K.; Hancock, N.T.; Cath, T.Y. The sweet spot of forward osmosis: Treatment of produced water, drilling wastewater, and other complex and difficult liquid streams. Desalination 2014, 333, 23–35. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Li, Z.; Abu-Ghdaib, M.; Wei, C.-H.; Amy, G.; Vrouwenvelder, J.S. Water harvesting from municipal wastewater via osmotic gradient: An evaluation of process performance. J. Membr. Sci. 2013, 447, 50–56. [Google Scholar] [CrossRef]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S.; Kandasamy, J. Fertiliser drawn forward osmosis desalination: The concept, performance and limitations for fertigation. Rev. Environ. Sci. Biotechnol. 2012, 11, 147–168. [Google Scholar] [CrossRef]
- Nicoll, P.G. Forward Osmosis as a Pre-Treatment to Reverse Osmosis. In Proceedings of the International Desalination Association World Congress on Desalination and Water Reuse, Tianjin, China, 20–25 October 2013; Available online: http://www.modernwater.com/assets/downloads/Papers/Forward Osmosis as a Pre-treatment to Reverse Osmosis.pdf (accessed on 29 September 2019).
- Hoover, L.A.; Phillip, W.A.; Tiraferri, A.; Yip, N.Y.; Elimelech, M. Forward with Osmosis: Emerging Applications for Greater Sustainability. Environ. Sci. Technol. 2011, 45, 9824–9830. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Tian, J.; Zhao, Z.; Shi, W.; Liu, D.; Cui, F. Membrane fouling of forward osmosis (FO) membrane for municipal wastewater treatment: A comparison between direct FO and OMBR. Water Res. 2016, 104, 330–339. [Google Scholar] [CrossRef]
- Fern, J.; Alonso-molina, J.L.; Amor, I. The role of salinity on the changes of the biomass characteristics and on the performance of an OMBR treating tannery waste. Water Res. 2018, 142, 129–137. [Google Scholar]
- Blandin, G.; Gautier, C.; Sauchelli Toran, M.; Monclús, H.; Rodriguez-Roda, I.; Comas, J. Retrofitting membrane bioreactor (MBR) into osmotic membrane bioreactor (OMBR): A pilot scale study. Chem. Eng. J. 2018, 339, 268–277. [Google Scholar] [CrossRef]
- Zhao, J.; Li, Y.; Pan, S.; Tu, Q.; Zhu, H. Performance of a forward osmotic membrane bioreactor for anaerobic digestion of waste sludge with increasing solid concentration. J. Environ. Manag. 2019, 246, 239–246. [Google Scholar] [CrossRef]
- Lutchmiah, K.; Verliefde, A.R.D.; Roest, K.; Rietveld, L.C.; Cornelissen, E.R. Forward osmosis for application in wastewater treatment: A review. Water Res. 2014, 58, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-N.; Goh, K.; Li, X.; Setiawan, L.; Wang, R. Membranes and processes for forward osmosis-based desalination: Recent advances and future prospects. Desalination 2018, 434, 81–99. [Google Scholar] [CrossRef]
- She, Q.; Wang, R.; Fane, A.G.; Tang, C.Y. Membrane fouling in osmotically driven membrane processes: A review. J. Membr. Sci. 2016, 499, 201–233. [Google Scholar] [CrossRef]
- Chung, T.S.; Luo, L.; Wan, C.F.; Cui, Y.; Amy, G. What is next for forward osmosis (FO) and pressure retarded osmosis (PRO). Sep. Purif. Technol. 2015, 156, 856–860. [Google Scholar] [CrossRef]
- Chun, Y.; Mulcahy, D.; Zou, L.; Kim, I.S. A short review of membrane fouling in forward osmosis processes. Membranes 2017, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.M.; Jalab, R.; Minier-Matar, J.; Adham, S.; Nasser, M.S.; Judd, S.J. The status of forward osmosis technology implementation. Desalination 2019, 461, 10–21. [Google Scholar] [CrossRef]
- Zhao, S.; Zou, L.; Tang, C.Y.; Mulcahy, D. Recent developments in forward osmosis: Opportunities and challenges. J. Membr. Sci. 2012, 396, 1–21. [Google Scholar] [CrossRef]
- Qi, S.; Qiu, C.Q.; Zhao, Y.; Tang, C.Y. Double-skinned forward osmosis membranes based on layer-by-layer assembly-FO performance and fouling behavior. J. Membr. Sci. 2012, 405–406, 20–29. [Google Scholar] [CrossRef]
- Hancock, N.T.; Cath, T.Y. Solute Coupled Diffusion in Osmotically Driven Membrane Processes. Environ. Sci. Technol. 2009, 43, 6769–6775. [Google Scholar] [CrossRef]
- Shaffer, D.L.; Werber, J.R.; Jaramillo, H.; Lin, S.; Elimelech, M. Forward osmosis: Where are we now? Desalination 2015, 356, 271–284. [Google Scholar] [CrossRef]
- Johnson, D.J.; Suwaileh, W.A.; Mohammed, A.W.; Hilal, N. Osmotic’s potential: An overview of draw solutes for forward osmosis. Desalination 2018, 434, 100–120. [Google Scholar] [CrossRef]
- Gulied, M.; Al Momani, F.; Khraisheh, M.; Bhosale, R.; AlNouss, A. Influence of draw solution type and properties on the performance of forward osmosis process: Energy consumption and sustainable water reuse. Chemosphere 2019, 233, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Holloway, R.W.; Maltos, R.; Vanneste, J.; Cath, T.Y. Mixed draw solutions for improved forward osmosis performance. J. Membr. Sci. 2015, 491, 121–131. [Google Scholar] [CrossRef]
- Islam, M.S.; Sultana, S.; McCutcheon, J.R.; Rahaman, M.S. Treatment of fracking wastewaters via forward osmosis: Evaluation of suitable organic draw solutions. Desalination 2019, 452, 149–158. [Google Scholar] [CrossRef]
- Alejo, T.; Arruebo, M.; Carcelen, V.; Monsalvo, V.M.; Sebastian, V. Advances in draw solutes for forward osmosis: Hybrid organic-inorganic nanoparticles and conventional solutes. Chem. Eng. J. 2017, 309, 738–752. [Google Scholar] [CrossRef]
- Kim, Y.; Li, S.; Chekli, L.; Woo, Y.C.; Wei, C.H.; Phuntsho, S.; Ghaffour, N.; Leiknes, T.O.; Shon, H.K. Assessing the removal of organic micro-pollutants from anaerobic membrane bioreactor effluent by fertilizer-drawn forward osmosis. J. Membr. Sci. 2017, 533, 84–95. [Google Scholar] [CrossRef]
- Dutta, S.; Nath, K. Prospect of ionic liquids and deep eutectic solvents as new generation draw solution in forward osmosis process. J. Water Process. Eng. 2018, 21, 163–176. [Google Scholar] [CrossRef]
- Luo, H.; Wu, K.; Wang, Q.; Zhang, T.C.; Lu, H.; Rong, H.; Fang, Q. Forward osmosis with electro-responsive P(AMPS-co-AM) hydrogels as draw agents for desalination. J. Membr. Sci. 2019, 117406. [Google Scholar] [CrossRef]
- Volpin, F.; Yu, H.; Cho, J.; Lee, C.; Phuntsho, S.; Ghaffour, N.; Vrouwenvelder, J.S.; Shon, H.K. Human urine as a forward osmosis draw solution for the application of microalgae dewatering. J. Hazard. Mater. 2019, 378, 120724. [Google Scholar] [CrossRef]
- Yang, S.; Gao, B.; Jang, A.; Shon, H.K.; Yue, Q. Municipal wastewater treatment by forward osmosis using seawater concentrate as draw solution. Chemosphere 2019, 237, 124485. [Google Scholar] [CrossRef]
- Caicedo, C.; Rosenwinkel, K.H.; Exner, M.; Verstraete, W.; Suchenwirth, R.; Hartemann, P.; Nogueira, R. Legionella occurrence in municipal and industrial wastewater treatment plants and risks of reclaimed wastewater reuse: Review. Water Res. 2019, 149, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, M.; Mishima, S.; Tanaka, Y.; Takahashi, T.; Matsuyama, H. Thin-film composite forward osmosis membrane with high water flux and high pressure resistance using a thicker void-free polyketone porous support. Desalination 2017, 402, 1–9. [Google Scholar] [CrossRef]
- Lutchmiah, K.; Harmsen, D.J.H.; Wols, B.A.; Rietveld, L.C.; Qin, J.; Cornelissen, E.R. Continuous and discontinuous pressure assisted osmosis (PAO). J. Membr. Sci. 2015, 476, 182–193. [Google Scholar] [CrossRef]
- Li, J.Y.; Ni, Z.Y.; Zhou, Z.Y.; Hu, Y.X.; Xu, X.H.; Cheng, L.H. Membrane fouling of forward osmosis in dewatering of soluble algal products: Comparison of TFC and CTA membranes. J. Membr. Sci. 2018, 552, 213–221. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Li, Z.; Sarp, S.; Bucs, S.S.; Amy, G.; Vrouwenvelder, J.S. Forward osmosis niches in seawater desalination and wastewater reuse. Water Res. 2014, 66, 122–139. [Google Scholar] [CrossRef] [PubMed]
- Alshwairekh, A.M.; Alghafis, A.A.; Alwatban, A.M.; Alqsair, U.F.; Oztekin, A. The effects of membrane and channel corrugations in forward osmosis membrane modules—c Numerical analyses. Desalination 2019, 460, 41–55. [Google Scholar] [CrossRef]
- Coday, B.D.; Cath, T.Y. Forward osmosis: Novel desalination of produced water and fracturing flowback. J. Am. Water Works Assoc. 2014, 106, 37–38. [Google Scholar] [CrossRef]
- Blandin, G.; Verliefde, A.R.D.; Comas, J.; Rodriguez-Roda, I.; Le-Clech, P. Efficiently combining water reuse and desalination through forward osmosis-reverse osmosis (FO-RO) hybrids: A critical review. Membranes 2016, 6, 37. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, J.; Gao, S.; Zhang, Z.; Cui, F.; Tang, C.Y. In situ surface modification of thin film composite forward osmosis membranes with sulfonated poly(arylene ether sulfone) for anti-fouling in emulsified oil/water separation. J. Memb. Sci. 2017, 527, 26–34. [Google Scholar] [CrossRef]
- Chiao, Y.H.; Chen, S.T.; Patra, T.; Hsu, C.H.; Sengupta, A.; Hung, W.S.; Huang, S.H.; Qian, X.; Wickramasinghe, R.; Chang, Y.; et al. Zwitterionic forward osmosis membrane modified by fast second interfacial polymerization with enhanced antifouling and antimicrobial properties for produced water pretreatment. Desalination 2019, 469, 114090. [Google Scholar] [CrossRef]
- Ong, C.S.; Al-Anzi, B.; Lau, W.J.; Goh, P.S.; Lai, G.S.; Ismail, A.F.; Ong, Y.S. Anti-Fouling Double-Skinned Forward Osmosis Membrane with Zwitterionic Brush for Oily Wastewater Treatment. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Goh, P.S.; Lau, W.J.; Ong, C.S.; Ismail, A.F. Antifouling zwitterion embedded forward osmosis thin film composite membrane for highly concentrated oily wastewater treatment. Sep. Purif. Technol. 2019, 214, 40–50. [Google Scholar] [CrossRef]
- Hu, B.; Jiang, M.; Zhao, S.; Ji, X.; Shu, Q.; Tian, B.; He, T.; Zhang, L. Biogas slurry as draw solution of forward osmosis process to extract clean water from micro-polluted water for hydroponic cultivation. J. Memb. Sci. 2019, 576, 88–95. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, C. Efficient removal of heavy metal ions based on the optimized dissolution-diffusion-flow forward osmosis process. Chem. Eng. J. 2018, 334, 1128–1134. [Google Scholar] [CrossRef]
- Wu, C.; Mouri, H.; Chen, S.; Zhang, D.; Koga, M.; Kobayashi, J. Removal of trace-amount mercury from wastewater by forward osmosis. J. Water Process Eng. 2016, 14, 108–116. [Google Scholar] [CrossRef]
- Liu, X.; Wu, J.; Liu, C.; Wang, J. Removal of cobalt ions from aqueous solution by forward osmosis. Sep. Purif. Technol. 2017, 177, 8–20. [Google Scholar] [CrossRef]
- Liu, X.; Wu, J.; Wang, J. Removal of Cs(I) from simulated radioactive wastewater by three forward osmosis membranes. Chem. Eng. J. 2018, 344, 353–362. [Google Scholar] [CrossRef]
- Lee, S.; Kim, Y.; Park, J.; Shon, H.K.; Hong, S. Treatment of medical radioactive liquid waste using Forward Osmosis (FO) membrane process. J. Memb. Sci. 2018, 556, 238–247. [Google Scholar] [CrossRef]
- Liu, X.; Wu, J.; Wang, J. Removal of nuclides and boric acid from simulated radioactive wastewater by forward osmosis. Prog. Nucl. Energy 2019, 114, 155–163. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Yu, H.; Zhu, Z.; Zhang, X.; Zhang, Z.; Zhang, R.; Cui, F. Polyamidoamine dendrimer grafted forward osmosis membrane with superior ammonia selectivity and robust antifouling capacity for domestic wastewater concentration. Water Res. 2019, 153, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Wu, Q.; Tian, J.; Shi, W.; Wang, W.; Zhang, Z.; Zhang, R.; Zhang, B.; Guo, Y.; Shu, S.; et al. Fouling mechanism of forward osmosis membrane in domestic wastewater concentration: Role of substrate structures. Chem. Eng. J. 2019, 370, 262–273. [Google Scholar] [CrossRef]
- Cui, Y.; Liu, X.Y.; Chung, T.S.; Weber, M.; Staudt, C.; Maletzko, C. Removal of organic micro-pollutants (phenol, aniline and nitrobenzene) via forward osmosis (FO) process: Evaluation of FO as an alternative method to reverse osmosis (RO). Water Res. 2016, 91, 104–114. [Google Scholar] [CrossRef]
- Engelhardt, S.; Sadek, A.; Duirk, S. Rejection of trace organic water contaminants by an Aquaporin-based biomimetic hollow fiber membrane. Sep. Purif. Technol. 2018, 197, 170–177. [Google Scholar] [CrossRef]
- Phillip, W.; Yong, J.S.; Elimelech, M. Reverse draw solute flux in forward osmosis modules: Modeling and experiments. Environ. Sci. Technol. 2010, 44, 5170–5176. [Google Scholar] [CrossRef] [PubMed]
- Ly, Q.V.; Hu, Y.; Li, J.; Cho, J.; Hur, J. Characteristics and influencing factors of organic fouling in forward osmosis operation for wastewater applications: A comprehensive review. Environ. Int. 2019, 129, 164–184. [Google Scholar] [CrossRef] [PubMed]
- Bogler, A.; Lin, S.; Bar-Zeev, E. Biofouling of membrane distillation, forward osmosis and pressure retarded osmosis: Principles, impacts and future directions. J. Membr. Sci. 2017, 542, 378–398. [Google Scholar] [CrossRef]
- Li, L.; Wang, X.; Xie, M.; Wang, Z.; Li, X.; Ren, Y. In situ extracting organic-bound calcium: A novel approach to mitigating organic fouling in forward osmosis treating wastewater via gradient diffusion thin-films. Water Res. 2019, 156, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Majeed, T.; Phuntsho, S.; Jeong, S.; Zhao, Y.; Gao, B.; Shon, H.K. Understanding the risk of scaling and fouling in hollow fiber forward osmosis membrane application. Process. Saf. Environ. Prot. 2016, 104, 452–464. [Google Scholar] [CrossRef][Green Version]
- Li, S.; Kim, Y.; Chekli, L.; Phuntsho, S.; Shon, H.K.; Leiknes, T.O.; Ghaffour, N. Impact of reverse nutrient diffusion on membrane biofouling in fertilizer-drawn forward osmosis. J. Membr. Sci. 2017, 539, 108–115. [Google Scholar] [CrossRef]
- Suwaileh, W.A.; Johnson, D.J.; Sarp, S.; Hilal, N. Advances in forward osmosis membranes: Altering the sub-layer structure via recent fabrication and chemical modification approaches. Desalination 2018, 436, 176–201. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, X.; Li, Z.; Wang, Q.; Dong, S.; Wang, X.; Ma, Z.; Wang, L.; Wang, X.; Gao, C. Porous membranes in pressure-assisted forward osmosis: Flux behavior and potential applications. J. Ind. Eng. Chem. 2018, 60, 160–168. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, T. Anchoring hydrophilic polymer in substrate: An easy approach for improving the performance of TFC FO membrane. J. Membr. Sci. 2015, 476, 330–339. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; Elimelech, M. Influence of concentrative and dilutive internal concentration polarization on flux behavior in forward osmosis. J. Membr. Sci. 2006, 284, 237–247. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; Elimelech, M. Influence of membrane support layer hydrophobicity on water flux in osmotically driven membrane processes. J. Membr. Sci. 2008, 318, 458–466. [Google Scholar] [CrossRef]
- Huang, L.; Arena, J.T.; McCutcheon, J.R. Surface Modified PVDF Nanofiber Supported Thin Film Composite Membranes for Forward Osmosis. J. Membr. Sci. 2016, 499, 352–360. [Google Scholar] [CrossRef]
- Puguan, J.M.C.; Kim, H.S.; Lee, K.J.; Kim, H. Low internal concentration polarization in forward osmosis membranes with hydrophilic crosslinked PVA nanofibers as porous support layer. Desalination 2014, 336, 24–31. [Google Scholar] [CrossRef]
- Tian, E.; Wang, X.; Zhao, Y.; Ren, Y. Middle support layer formation and structure in relation to performance of three-tier thin film composite forward osmosis membrane. Desalination 2017, 421, 190–201. [Google Scholar] [CrossRef]
- Qiu, M.; Wang, J.; He, C. A stable and hydrophilic substrate for thin-film composite forward osmosis membrane revealed by in-situ cross-linked polymerization. Desalination 2018, 433, 1–9. [Google Scholar] [CrossRef]
- Bassyouni, M.; Abdel-Aziz, M.H.; Zoromba, M.S.; Abdel-Hamid, S.M.S.; Drioli, E. A review of polymeric nanocomposite membranes for water purification. J. Ind. Eng. Chem. 2019, 73, 19–46. [Google Scholar] [CrossRef]
- Camilleri-Rumbau, M.S.; Soler-Cabezas, J.L.; Christensen, K.V.; Norddahl, B.; Mendoza-Roca, J.A.; Vincent-Vela, M.C. Application of aquaporin-based forward osmosis membranes for processing of digestate liquid fractions, Chem. Eng. J. 2019, 371, 583–592. [Google Scholar] [CrossRef]
- Akther, N.; Phuntsho, S.; Chen, Y.; Ghaffour, N.; Shon, H.K. Recent advances in nanomaterial-modified polyamide thin-film composite membranes for forward osmosis processes. J. Membr. Sci. 2019, 584, 20–45. [Google Scholar] [CrossRef]
- Lai, G.S.; Lau, W.J.; Goh, P.S.; Ismail, A.F.; Tan, Y.H.; Chong, C.Y.; Krause-Rehberg, R.; Awad, S. Tailor-made thin film nanocomposite membrane incorporated with graphene oxide using novel interfacial polymerization technique for enhanced water separation. Chem. Eng. J. 2018, 344. [Google Scholar] [CrossRef]
- Sakai, M.; Seshimo, M.; Matsukata, M. Hydrophilic ZSM-5 membrane for forward osmosis operation. J. Water Process. Eng. 2019, 32, 100864. [Google Scholar] [CrossRef]
- Amini, M.; Jahanshahi, M.; Rahimpour, A. Synthesis of novel thin film nanocomposite (TFN) forward osmosis membranes using functionalized multi-walled carbon nanotubes. J. Membr. Sci. 2013, 435, 233–241. [Google Scholar] [CrossRef]
- Azelee, I.W.; Goh, P.S.; Lau, W.J.; Ismail, A.F.; Rezaei-DashtArzhandi, M.; Wong, K.C.; Subramaniam, M.N. Enhanced desalination of polyamide thin film nanocomposite incorporated with acid treated multiwalled carbon nanotube-titania nanotube hybrid. Desalination 2017, 409, 163–170. [Google Scholar] [CrossRef]
- Bi, R.; Zhang, Q.; Zhang, R.; Su, Y.; Jiang, Z. Thin film nanocomposite membranes incorporated with graphene quantum dots for high flux and antifouling property. J. Membr. Sci. 2018, 553, 17–24. [Google Scholar] [CrossRef]
- Zhang, X.; Shen, L.; Guan, C.Y.; Liu, C.X.; Lang, W.Z.; Wang, Y. Construction of SiO2@MWNTs incorporated PVDF substrate for reducing internal concentration polarization in forward osmosis. J. Membr. Sci. 2018, 564, 328–341. [Google Scholar] [CrossRef]
- Le Ouay, B.; Stellacci, F. Antibacterial activity of silver nanoparticles: A surface science insight. Nano Today 2015, 10, 339–354. [Google Scholar] [CrossRef]
- Faria, A.F.; Liu, C.; Xie, M.; Perreault, F.; Nghiem, L.D.; Ma, J.; Elimelech, M. Thin-film composite forward osmosis membranes functionalized with graphene oxide–silver nanocomposites for biofouling control. J. Memb. Sci. 2017, 525, 146–156. [Google Scholar] [CrossRef]
- Rastgar, M.; Bozorg, A.; Shakeri, A.; Sadrzadeh, M. Substantially improved antifouling properties in electro-oxidative graphene laminate forward osmosis membrane. Chem. Eng. Res. Des. 2019, 141, 413–424. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, H.; Yu, M.; Wang, Y.; Gao, T.; Yang, F. Conductive thin film nanocomposite forward osmosis membrane (TFN-FO) blended with carbon nanoparticles for membrane fouling control. Sci. Total Environ. 2019, 134050. [Google Scholar] [CrossRef]
- Jensen, M.Ø.; Mouritsen, O.G. Single-Channel Water Permeabilities of Escherichia coli Aquaporins AqpZ and GlpF. Biophys. J. 2006, 90, 2270–2284. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Linares, R.V.; Bucs, S.; Fortunato, L.; Hélix-Nielsen, C.; Vrouwenvelder, J.S.; Ghaffour, N.; Leiknes, T.O.; Amy, G. Aquaporin based biomimetic membrane in forward osmosis: Chemical cleaning resistance and practical operation. Desalination 2017, 420, 208–215. [Google Scholar] [CrossRef]
- Kuang, W.; Liu, Z.; Yu, H.; Kang, G.; Jie, X.; Jin, Y.; Cao, Y. Investigation of internal concentration polarization reduction in forward osmosis membrane using nano-CaCO 3 particles as sacrificial component. J. Membr. Sci. 2016, 497, 485–493. [Google Scholar] [CrossRef]
- Ahmad, N.; Goh, P.; Karim, Z.A.; Ismail, A. Thin Film Composite Membrane for Oily Waste Water Treatment: Recent Advances and Challenges. Membranes 2018, 8, 86. [Google Scholar] [CrossRef]
- Zhou, Z.; Lee, J.Y. Evaluating the viability of double-skin thin film composite membranes in forward osmosis processes. J. Membr. Sci. 2016, 502, 65–75. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, K.Y.; Chung, T.S.; Chen, H.; Jean, Y.C.; Amy, G. Well-constructed cellulose acetate membranes for forward osmosis: Minimized internal concentration polarization with an ultra-thin selective layer. J. Membr. Sci. 2010, 360, 522–535. [Google Scholar] [CrossRef]
- Wei, R.; Zhang, S.; Cui, Y.; Ong, R.C.; Chung, T.S.; Helmer, B.J.; de Wit, J.S. Highly permeable forward osmosis (FO) membranes for high osmotic pressure but viscous draw solutes. J. Membr. Sci. 2015, 496, 132–141. [Google Scholar] [CrossRef]
- Liu, X.; Ong, S.L.; Ng, H.Y. Fabrication of mesh-embedded double-skinned substrate membrane and enhancement of its surface hydrophilicity to improve anti-fouling performance of resultant thin-film composite forward osmosis membrane. J. Membr. Sci. 2016, 511, 40–53. [Google Scholar] [CrossRef]
- Zhang, X.; Xie, M.; Yang, Z.; Wu, H.C.; Fang, C.; Bai, L.; Fang, L.F.; Yoshioka, T.; Matsuyama, H. Antifouling Double-Skinned Forward Osmosis Membranes by Constructing Zwitterionic Brush-Decorated MWCNT Ultrathin Films. ACS Appl. Mater. Interfaces 2019, 11, 19462–19471. [Google Scholar] [CrossRef] [PubMed]
- Joseph, N.; Ahmadiannamini, P.; Hoogenboom, R.; Vankelecom, I.F.J. Layer-by-layer preparation of polyelectrolyte multilayer membranes for separation. Polym. Chem. 2014, 5, 1817–1831. [Google Scholar] [CrossRef]
- Decher, G. Fuzzy nanoassemblies: Toward layered polymeric multicomposites. Science 1997, 277, 1232–1237. [Google Scholar] [CrossRef]
- Kwon, S.B.; Lee, J.S.; Kwon, S.J.; Yun, S.T.; Lee, S.; Lee, J.H. Molecular layer-by-layer assembled forward osmosis membranes. J. Membr. Sci. 2015, 488, 111–120. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, J.; Song, P.; Wang, Z. Layer-by-Layer Assembly for Preparation of High-Performance Forward Osmosis Membrane. IOP Conf. Ser. Mater. Sci. Eng. 2018, 301. [Google Scholar] [CrossRef]
- Qiu, M.; He, C. Novel zwitterion-silver nanocomposite modified thin-film composite forward osmosis membrane with simultaneous improved water flux and biofouling resistance property. Appl. Surf. Sci. 2018, 455, 492–501. [Google Scholar] [CrossRef]
- Seyedpour, S.F.; Rahimpour, A.; Najafpour, G. Facile in-situ assembly of silver-based MOFs to surface functionalization of TFC membrane: A novel approach toward long-lasting biofouling mitigation. J. Memb. Sci. 2019, 573, 257–269. [Google Scholar] [CrossRef]
- Liden, T.; Hildenbrand, Z.L.; Schug, K.A. Pretreatment Techniques for Produced Water with Subsequent Forward Osmosis Remediation. Water 2019, 11, 1437. [Google Scholar] [CrossRef]
- Yu, L.; Han, M.; He, F. A review of treating oily wastewater. Arab. J. Chem. 2017, 10, S1913–S1922. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Al-Kaabi, M.A.; Ashfaq, M.Y.; Da’na, D.A. Produced water characteristics, treatment and reuse: A review. J. Water Process Eng. 2019, 28, 222–239. [Google Scholar] [CrossRef]
- Demirbas, A.; Bamufleh, H.S.; Edris, G.; Alalayah, W.M. Treatment of contaminated wastewater. Pet. Sci. Technol. 2017, 35, 883–889. [Google Scholar] [CrossRef]
- Hickenbottom, K.L.; Hancock, N.T.; Hutchings, N.R.; Appleton, E.W.; Beaudry, E.G.; Xu, P.; Cath, T.Y. Forward osmosis treatment of drilling mud and fracturing wastewater from oil and gas operations. Desalination 2013, 312, 60–66. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, C. Enhancing the forward osmosis performance via the mesoporous silica hollow spheres assisted fast adsorption-diffusion process. Mater. Lett. 2019, 234, 347–350. [Google Scholar] [CrossRef]
- Zhou, H.; Li, X.; Xu, G.; Yu, H. Overview of strategies for enhanced treatment of municipal/domestic wastewater at low temperature. Sci. Total Environ. 2018, 643, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Ansari, A.J.; Hai, F.I.; Price, W.E.; Drewes, J.E.; Nghiem, L.D. Forward osmosis as a platform for resource recovery from municipal wastewater—A critical assessment of the literature. J. Memb. Sci. 2017, 529, 195–206. [Google Scholar] [CrossRef]
- Yaqub, M.; Lee, W. Zero-liquid discharge (ZLD) technology for resource recovery from wastewater: A review. Sci. Total Environ. 2019, 681, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Chekli, L.; Phuntsho, S.; Kim, J.E.J.J.H.; Kim, J.E.J.J.H.; Choi, J.Y.J.S.; Choi, J.Y.J.S.; Kim, S.; Kim, J.E.J.J.H.; Hong, S.; Sohn, J.; et al. A comprehensive review of hybrid forward osmosis systems: Performance, applications and future prospects. J. Memb. Sci. 2016, 497, 430–449. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Zhu, Z.; Zhang, Z.; Zhang, R.; Zhang, B.; Guo, Y.; Cui, F. Dendritic amine sheltered membrane for simultaneous ammonia selection and fouling mitigation in forward osmosis. J. Memb. Sci. 2019, 584, 9–19. [Google Scholar] [CrossRef]
- Ferrari, F.; Pijuan, M.; Rodriguez-Roda, I.; Blandin, G. Exploring Submerged Forward Osmosis for Water Recovery and Pre-Concentration of Wastewater before Anaerobic Digestion: A Pilot Scale Study. Membranes 2019, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; van Wezel, A.P.; Hollender, J.; Cornelissen, E.; Hofman, R.; van der Hoek, J.P. Development and application of relevance and reliability criteria for water treatment removal efficiencies of chemicals of emerging concern. Water Res. 2019, 161, 274–287. [Google Scholar] [CrossRef] [PubMed]
- López-Serna, R.; Posadas, E.; García-Encina, P.A.; Muñoz, R. Removal of contaminants of emerging concern from urban wastewater in novel algal-bacterial photobioreactors. Sci. Total Environ. 2019, 662, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Zietzschmann, F.; Altmann, J.; Ruhl, A.S.; Dünnbier, U.; Dommisch, I.; Sperlich, A.; Meinel, F.; Jekel, M. Estimating organic micro-pollutant removal potential of activated carbons using UV absorption and carbon characteristics. Water Res. 2014, 56, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Joseph, L.; Jun, B.M.; Jang, M.; Park, C.M.; Muñoz-Senmache, J.C.; Hernández-Maldonado, A.J.; Heyden, A.; Yu, M.; Yoon, Y. Removal of contaminants of emerging concern by metal-organic framework nanoadsorbents: A review. Chem. Eng. J. 2019, 369, 928–946. [Google Scholar] [CrossRef]
- Liu, C.; Takagi, R.; Cheng, L.; Saeki, D.; Matsuyama, H. Enzyme-aided forward osmosis (E-FO) process to enhance removal of micropollutants from water resources. J. Membr. Sci. 2019, 117399. [Google Scholar] [CrossRef]
- Xie, M.; Nghiem, L.D.; Price, W.E.; Elimelech, M. Comparison of the removal of hydrophobic trace organic contaminants by forward osmosis and reverse osmosis. Water Res. 2012, 46, 2683–2692. [Google Scholar] [CrossRef] [PubMed]
- Ruengruehan, K.; Fagkeaw, P.; Jang, D.; Ko, S.; Kang, S. Impact of feed ionic concentration on colloidal and organic fouling of osmotically driven membrane process. Desalin. Water Treat. 2016, 57, 24551–24556. [Google Scholar] [CrossRef]
- Jang, D.; Jeong, S.; Jang, A.; Kang, S. Relating solute properties of contaminants of emerging concern and their rejection by forward osmosis membrane. Sci. Total Environ. 2018, 639, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Alturki, A.A.; McDonald, J.A.; Khan, S.J.; Price, W.E.; Nghiem, L.D.; Elimelech, M. Removal of trace organic contaminants by the forward osmosis process. Sep. Purif. Technol. 2013, 103, 258–266. [Google Scholar] [CrossRef]
- Kim, Y.; Li, S.; Phuntsho, S.; Xie, M.; Shon, H.K.; Ghaffour, N. Understanding the organic micropollutants transport mechanisms in the fertilizer-drawn forward osmosis process. J. Environ. Manag. 2019, 248, 109240. [Google Scholar] [CrossRef]
- Madsen, H.T.; Bajraktari, N.; Hélix-Nielsen, C.; van der Bruggen, B.; Søgaard, E.G. Use of biomimetic forward osmosis membrane for trace organics removal. J. Memb. Sci. 2015, 476, 469–474. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, L.; Tang, C. Investigation on removing recalcitrant toxic organic polluters in coking wastewater by forward osmosis. Chin. J. Chem. Eng. 2019, 101145. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, P.; Liu, Y. Decontamination of radioactive wastewater: State of the art and challenges forward. Chemosphere 2019, 215, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Boo, C.; Elimelech, M.; Hong, S. Comparison of fouling behavior in forward osmosis (FO) and reverse osmosis (RO). J. Memb. Sci. 2010, 365, 34–39. [Google Scholar] [CrossRef]
- Ibrar, I.; Naji, O.; Sharif, A.; Malekizadeh, A.; Alhawari, A.; Alanezi, A.A.; Altaee, A. A review of fouling mechanisms, control strategies and real-time fouling monitoring techniques in forward osmosis. Water 2019, 11, 695. [Google Scholar] [CrossRef]
- Lotfi, F.; Samali, B.; Hagare, D. Cleaning efficiency of the fouled forward osmosis membranes under different experimental conditions. J. Environ. Chem. Eng. 2018, 6, 4555–4563. [Google Scholar] [CrossRef]
- Han, G.; Chan, S.S.; Chung, T.S. Forward Osmosis (FO) for Water Reclamation from Emulsified Oil/Water Solutions: Effects of Membrane and Emulsion Characteristics. ACS Sustain. Chem. Eng. 2016, 4, 5021–5032. [Google Scholar] [CrossRef]
- Liu, X.; Wu, J.; Hou, L.; Wang, J.; Engineering, N.; Wu, J.; Wang, J. Fouling and cleaning protocols for forward osmosis membrane used for radioactive wastewater treatment. Nucl. Eng. Technol. 2019. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Zhu, Z.; Zhang, Z.; Zhang, R.; Zhang, X.; Zhang, B.; Guo, Y.; et al. Insights into simultaneous ammonia-selective and anti-fouling mechanism over forward osmosis membrane for resource recovery from domestic wastewater. J. Membr. Sci. 2019, 573, 135–144. [Google Scholar] [CrossRef]
- Oh, S.H.; Jeong, S.; Kim, I.S.; Shon, H.K.; Jang, A. Removal behaviors and fouling mechanisms of charged antibiotics and nanoparticles on forward osmosis membrane. J. Environ. Manag. 2019, 247, 385–393. [Google Scholar] [CrossRef]
- Morrow, C.P.; McGaughey, A.L.; Hiibel, S.R.; Childress, A.E. Submerged or sidestream? The influence of module configuration on fouling and salinity in osmotic membrane bioreactors. J. Membr. Sci. 2018, 548, 583–592. [Google Scholar] [CrossRef]
- Liu, X.; Wu, J.; Hou, L.; Wang, J. Performance and deterioration of forward osmosis membrane exposed to various dose of gamma-ray irradiation. Ann. Nucl. Energy 2020, 135, 106950. [Google Scholar] [CrossRef]
- Korenak, J.; Hélix-Nielsen, C.; Bukšek, H.; Petrinić, I. Efficiency and economic feasibility of forward osmosis in textile wastewater treatment. J. Clean. Prod. 2019, 210, 1483–1495. [Google Scholar] [CrossRef]
- Zheng, S.; Tu, Q.; Urban, J.J.; Li, S.; Mi, B. Swelling of Graphene Oxide Membranes in Aqueous Solution: Characterization of Interlayer Spacing and Insight into Water Transport Mechanisms. ACS Nano 2017, 11, 6440–6450. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, F.A.; She, Q.; Fane, A.G.; Field, R.W. Exploring the differences between forward osmosis and reverse osmosis fouling. J. Memb. Sci. 2018, 565, 241–253. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Kook, S.; Lee, C.; Field, R.W.; Kim, I.S. Critical flux-based membrane fouling control of forward osmosis: Behavior, sustainability, and reversibility. J. Memb. Sci. 2019, 570, 380–393. [Google Scholar] [CrossRef]
- Han, G.; Liang, C.Z.; Chung, T.S.; Weber, M.; Staudt, C.; Maletzko, C. Combination of forward osmosis (FO) process with coagulation/flocculation (CF) for potential treatment of textile wastewater. Water Res. 2016, 91, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Phuntsho, S.; Chekli, L.; Choi, J.Y.; Shon, H.K. Environmental and economic assessment of hybrid FO-RO/NF system with selected inorganic draw solutes for the treatment of mine impaired water. Desalination 2018, 429, 96–104. [Google Scholar] [CrossRef]
- Chekli, L.; Kim, Y.; Phuntsho, S.; Li, S.; Ghaffour, N.; Leiknes, T.O.; Shon, H.K. Evaluation of fertilizer-drawn forward osmosis for sustainable agriculture and water reuse in arid regions. J. Environ. Manag. 2017, 187, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.C.; Chen, S.S.; Ho, S.T.; Nguyen, H.T.; Ray, S.S.; Nguyen, N.T.; Hsu, H.T.; Le, N.C.; Tran, T.T. Optimising the recovery of EDTA-2Na draw solution in forward osmosis through direct contact membrane distillation. Sep. Purif. Technol. 2018, 198, 108–112. [Google Scholar] [CrossRef]
- Corzo, B.; de la Torre, T.; Sans, C.; Escorihuela, R.; Navea, S.; Malfeito, J.J. Long-term evaluation of a forward osmosis-nanofiltration demonstration plant for wastewater reuse in agriculture. Chem. Eng. J. 2018, 338, 383–391. [Google Scholar] [CrossRef]

| Supplier | Materials/Commercial Name | Configurations |
|---|---|---|
| HTI | CTA-NW | SW |
| HTI | CTA-ES | SW |
| HTI | TFC | SW |
| Aquaporin A/S | AqP | SW, HF |
| Oasys Water | Thin film composite (TFC) | SW |
| Porifera | PFO element | SW |
| Toray | FO membrane | SW |
| Fluid technology solution | Cellulose triacetate (CTA) | SW |
| Modern water | - | SW |
| Toyobo | - | HF |
| Trevi System | - | SW |
| Application | Membrane | Feed Solution | Draw Solution | Rejection | Flux | Ref |
|---|---|---|---|---|---|---|
| Oily wastewater | NH2-BPSH100 grafted PA TFC | Soy bean oil/water emulsion | NaCl | 99.9% | 15 LMH | [50] |
| Oily wastewater | Aminoethyl piperazine propane sulfonate (AEPPS) grafted PA TFC | Produced water | NaCl | - | 15.2 LMH | [51] |
| Oily wastewater | (poly(3-(N-2-methacryloxyethyl-N,N-dimethyl) ammonatopropanesultone) (PMAPS) double skinned TFC | Synthetic cutting oil | NaCl | 99.9% | 13.7 LMH | [52] |
| Oily wastewater | PMAPS modified substrate TFC | Synthetic cutting oil | NaCl | 99% | 15.79 LMH | [53] |
| Heavy metal | Polyethersulfone (PES) hollow fibers | Cd2+ and Pb2+ solution | Biogas slurry | Cd2+ = 98.5% Pb2+ = 97% | 6.8 LMH | [54] |
| Heavy metal | Bovine serum albumin (BSA)- embedded PA TFC | Cd2+ Cu2+ and Pb2+ solution | NaCl | >99% | 45–50 LMH | [55] |
| Heavy metal | HTI TFC | Hg2+ solution | MgCl2 | 99/9% | - | [56] |
| Nuclear waste | CTA-ES | Co2+ solution | NaCl | 99.9% | 15.5 LMH | [57] |
| Nuclear waste | CTA-ES | Cs+ solution | NaCl | 96.24% | 9.7–19.3 LMH | [58] |
| Nuclear waste | Porifera TFC | medical radioactive liquid waste | NaCl | 99 | 21 LMH | [59] |
| Nuclear waste | CTA-ES | Co2+ and Cs+ solution | NaCl | Co2+ = 99% Sr2+ = 97% | 15 LMH | [60] |
| Domestic waste concentration | PAMAM grafted PA TFC | Raw domestic waste | NaCl | TOC, PO43−—P = 99% PO43−-P = 91% | 32 LMH | [61] |
| Domestic waste concentration | PEG modified substrate PA TFC | Raw domestic waste | NaCl | TOC 93% PO43−-P = 98% PO43−-P = 59 | 21.1 LMH | [62] |
| Contaminants of emerging concern (CEC), | Sulfonated polyphenylene sulfone (sPPSU)-PA TFC | aromatic aqueous solution | NaCl | Nitrobenzene = 75.2 Phenol = 72.1 Aniline = 91.5 | 22 LMH | [63] |
| CEC | Aquaporin Inside™ FO hollow fiber module | Trace ognic contaminants | NaCl | BPA = 99% methyl paraben = 95% | - | [64] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goh, P.S.; Ismail, A.F.; Ng, B.C.; Abdullah, M.S. Recent Progresses of Forward Osmosis Membranes Formulation and Design for Wastewater Treatment. Water 2019, 11, 2043. https://doi.org/10.3390/w11102043
Goh PS, Ismail AF, Ng BC, Abdullah MS. Recent Progresses of Forward Osmosis Membranes Formulation and Design for Wastewater Treatment. Water. 2019; 11(10):2043. https://doi.org/10.3390/w11102043
Chicago/Turabian StyleGoh, Pei Sean, Ahmad Fauzi Ismail, Be Cheer Ng, and Mohd Sohaimi Abdullah. 2019. "Recent Progresses of Forward Osmosis Membranes Formulation and Design for Wastewater Treatment" Water 11, no. 10: 2043. https://doi.org/10.3390/w11102043
APA StyleGoh, P. S., Ismail, A. F., Ng, B. C., & Abdullah, M. S. (2019). Recent Progresses of Forward Osmosis Membranes Formulation and Design for Wastewater Treatment. Water, 11(10), 2043. https://doi.org/10.3390/w11102043






