Water Disinfection by Immobilized Photosensitizers
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Thermogravimetric Analysis
2.3. Immobilization of PSs
2.4. Evaluation of PS Inclusion into Polymers
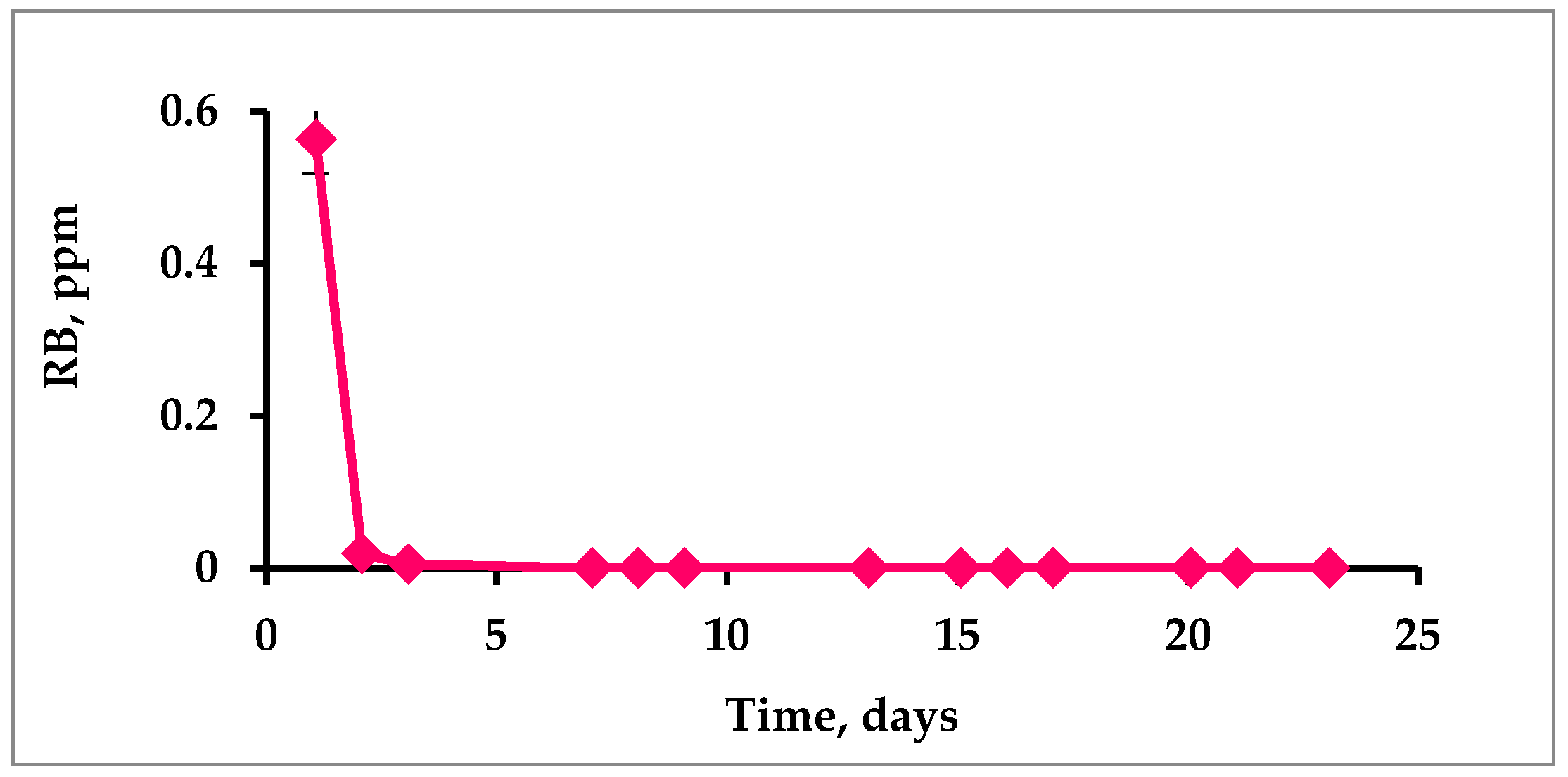
2.5. Testing PS Leakage from the Polymers
2.6. Bacterial Growth
2.7. Antibacterial Activity Assay
2.8. Photostability
2.9. Statistical Data Processing
3. Results
3.1. Thermal Stability of PS
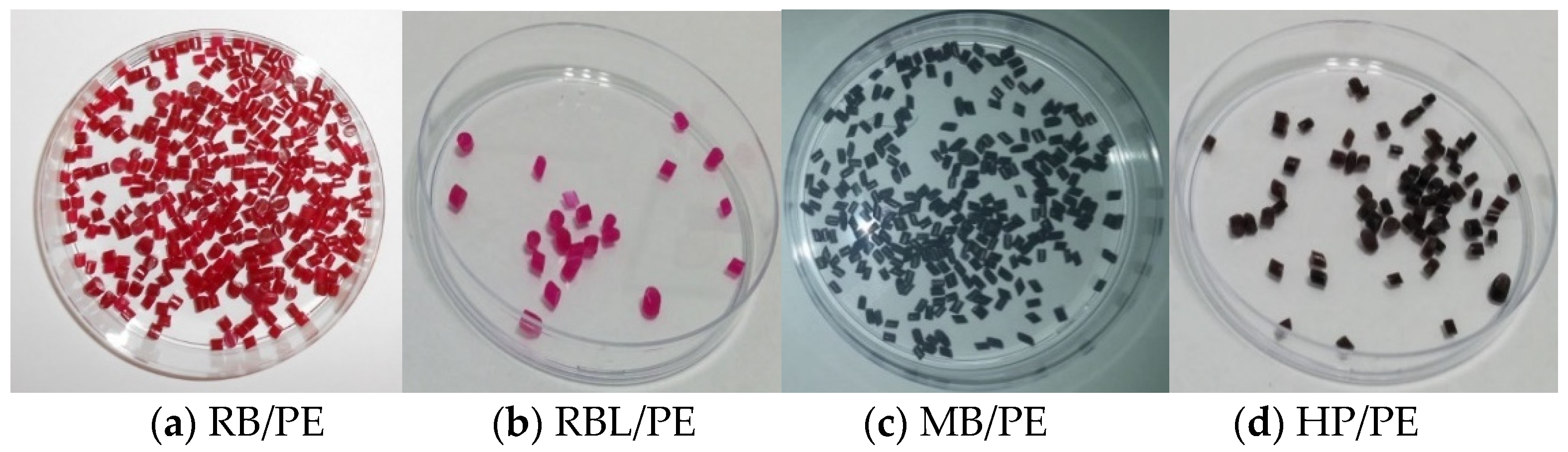
3.2. Immobilization of PS into Polymers
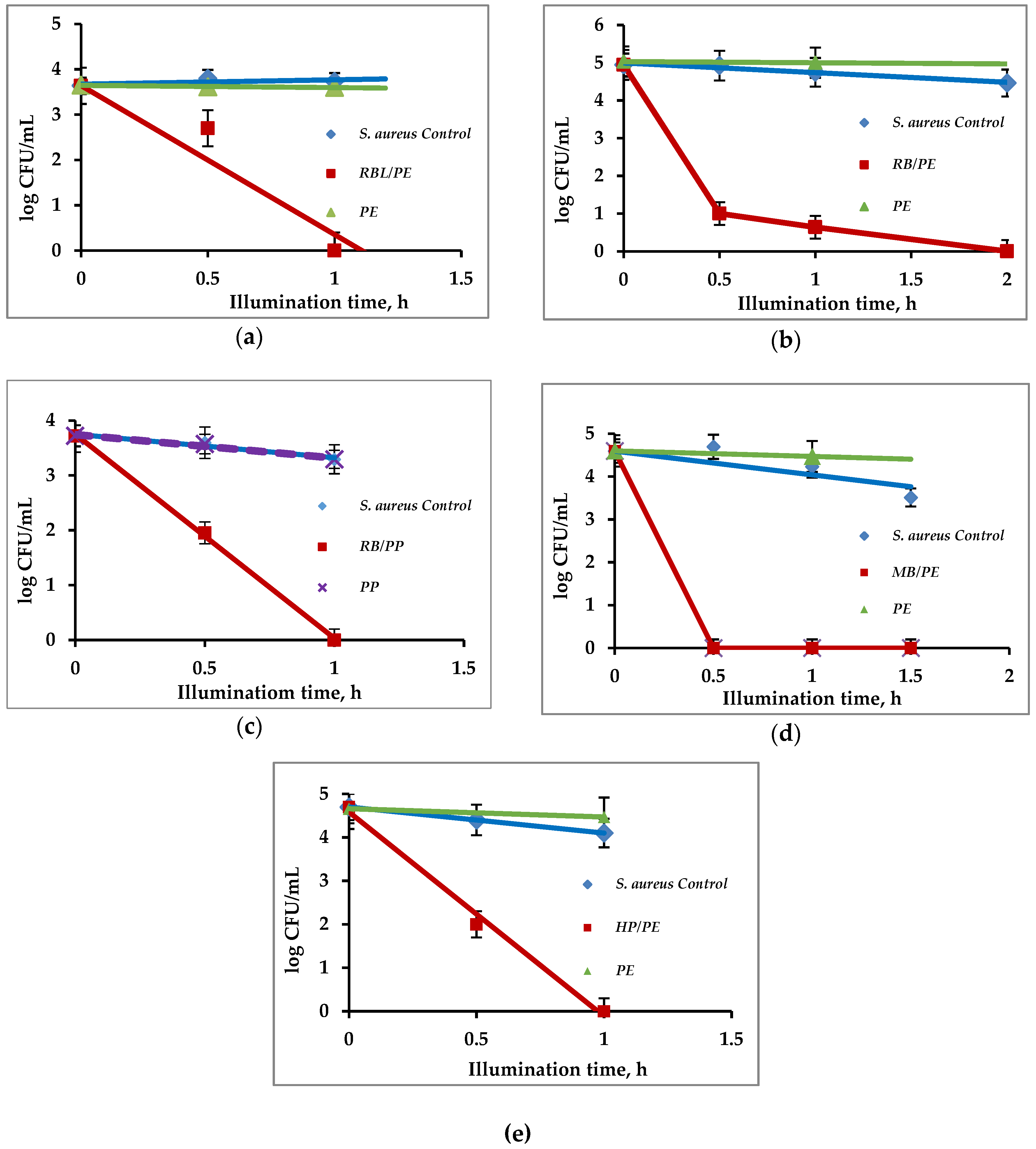
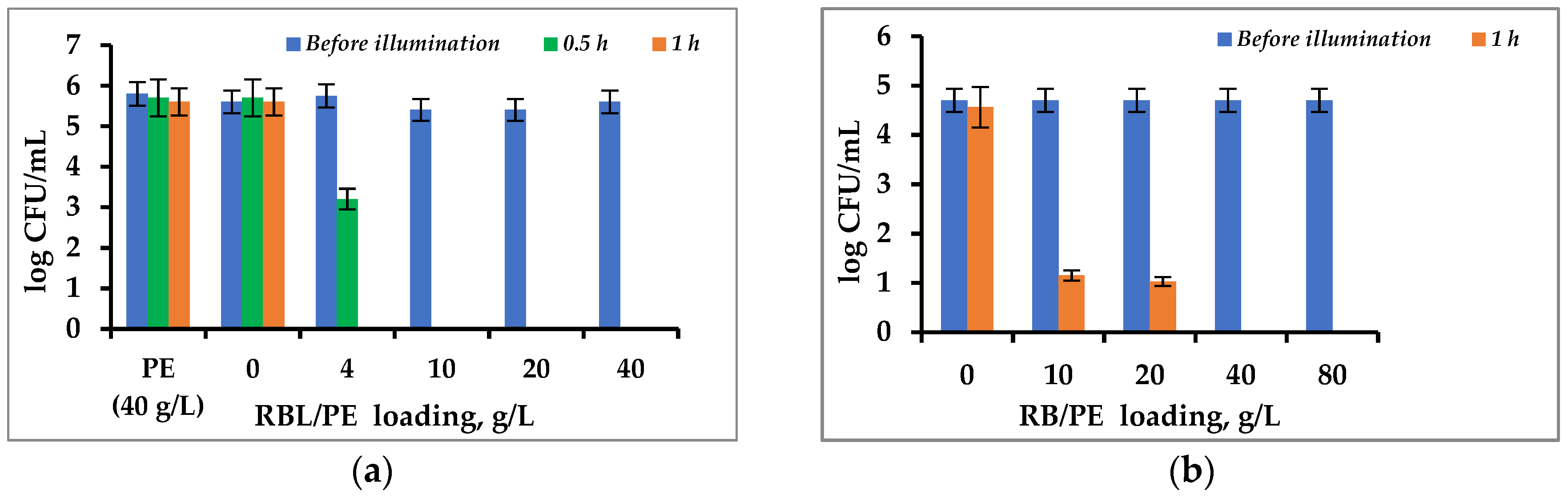
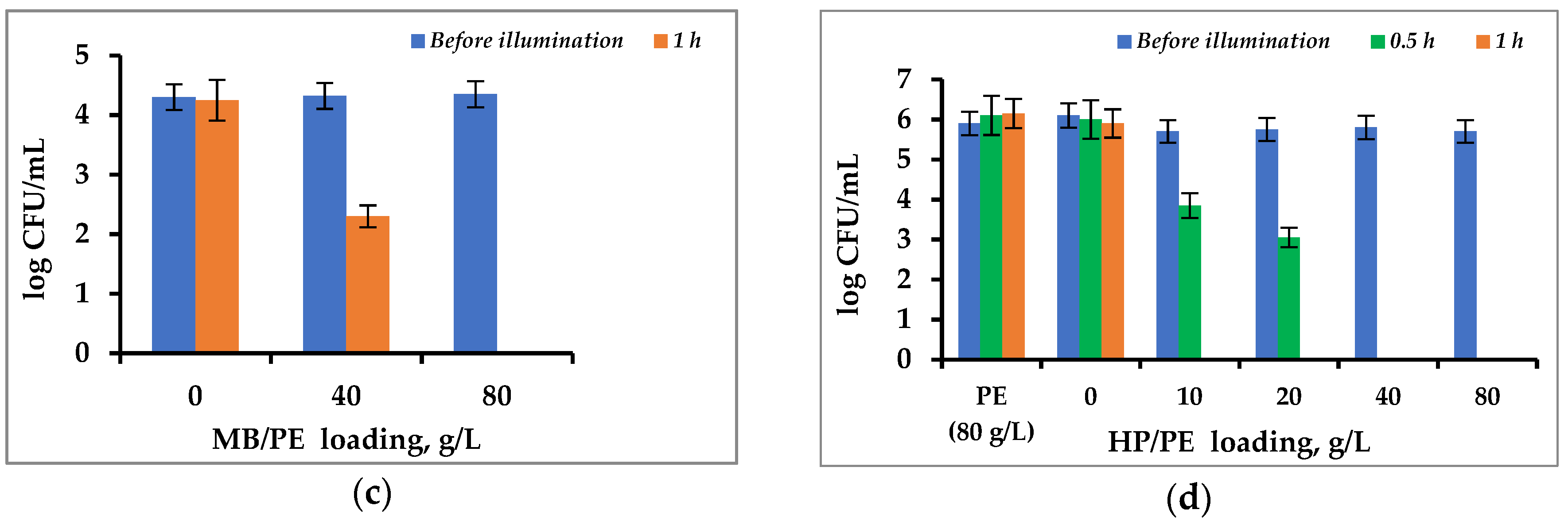
3.3. Antibacterial Activity of Immobilized PS
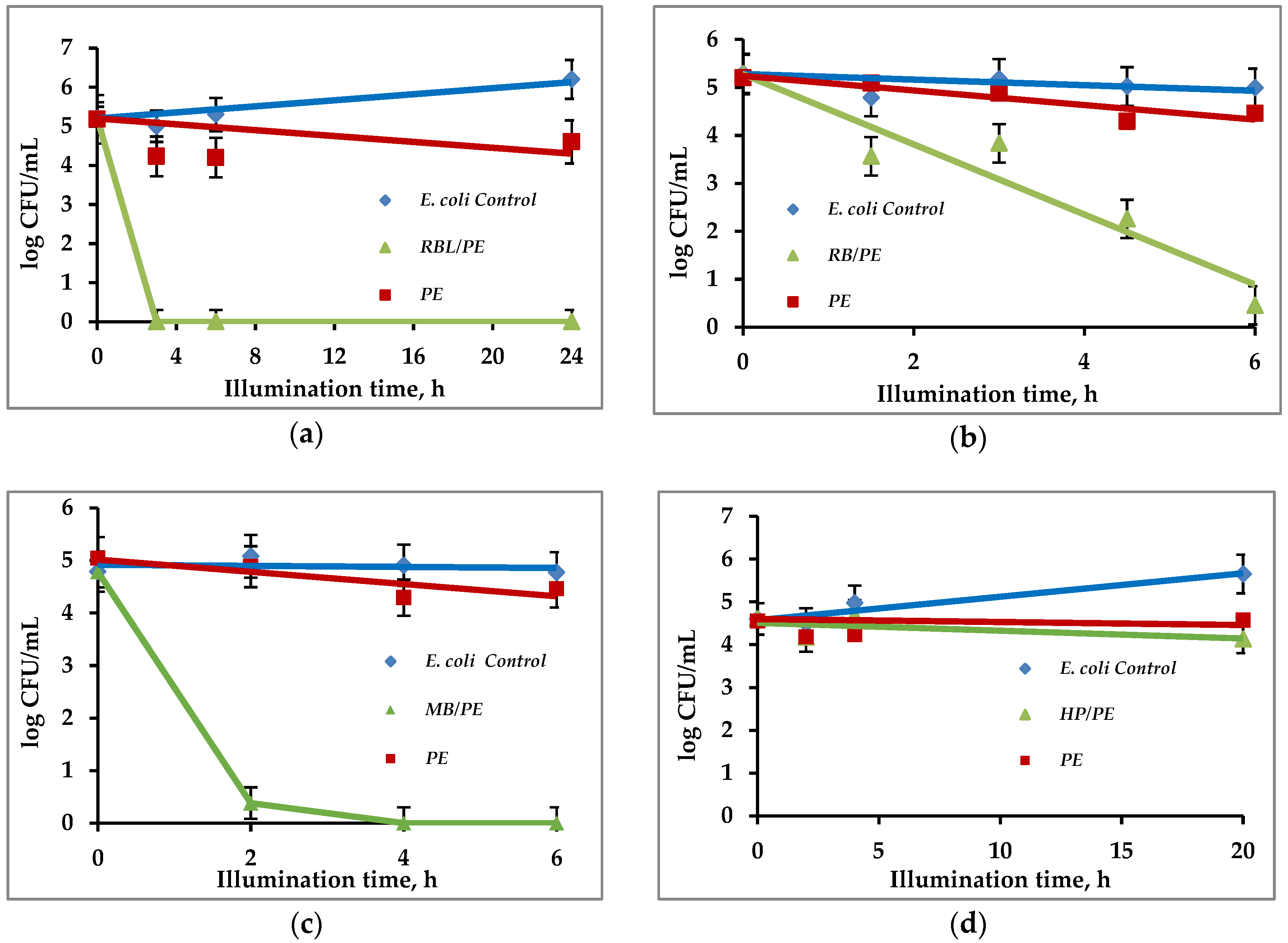
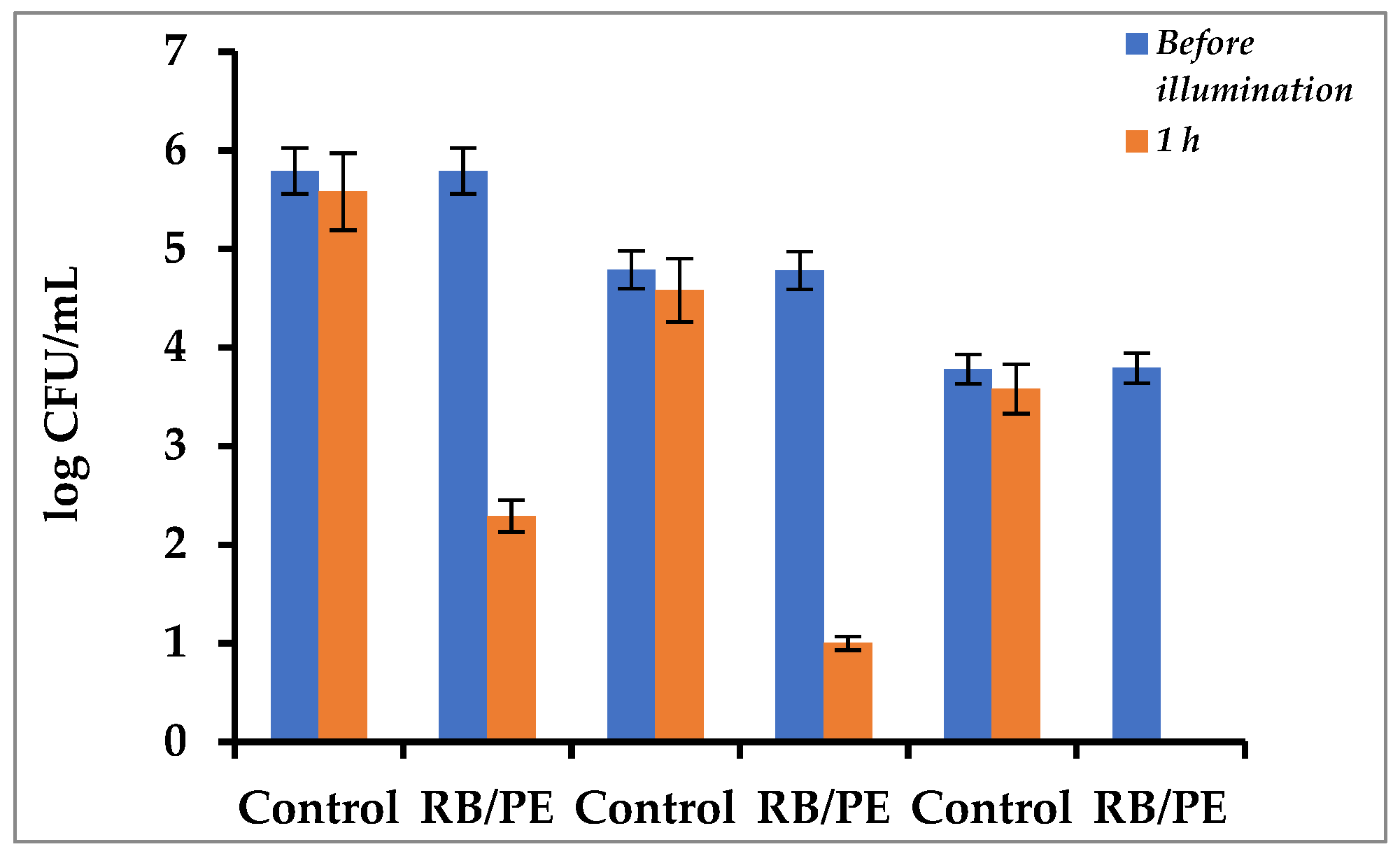
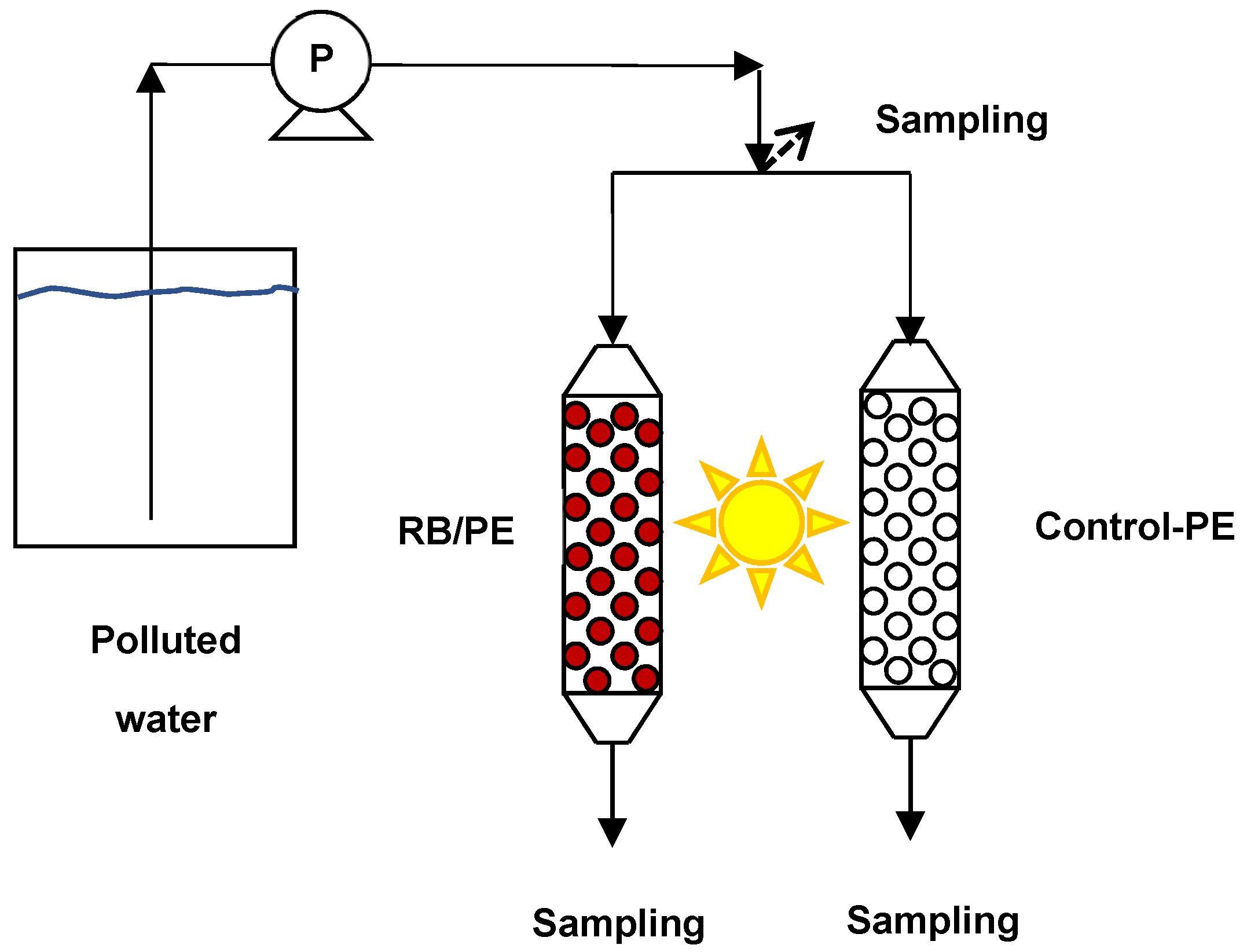
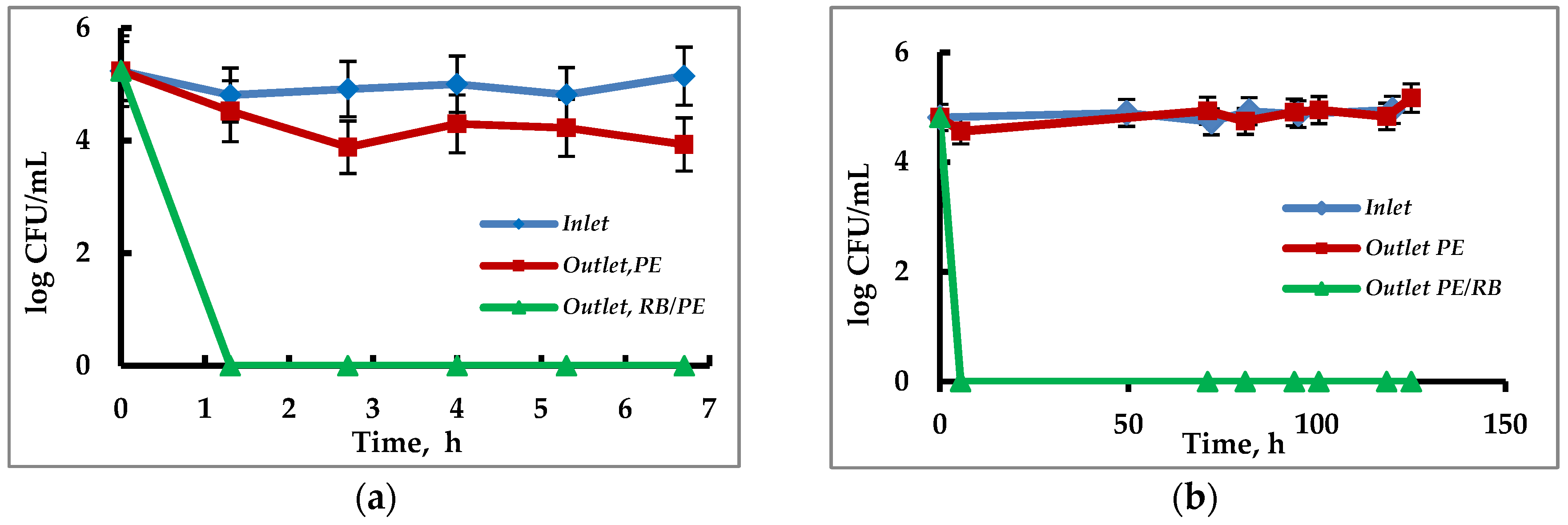
3.4. Continuous Eradication of Waterborne Bacteria by Immobilized PS
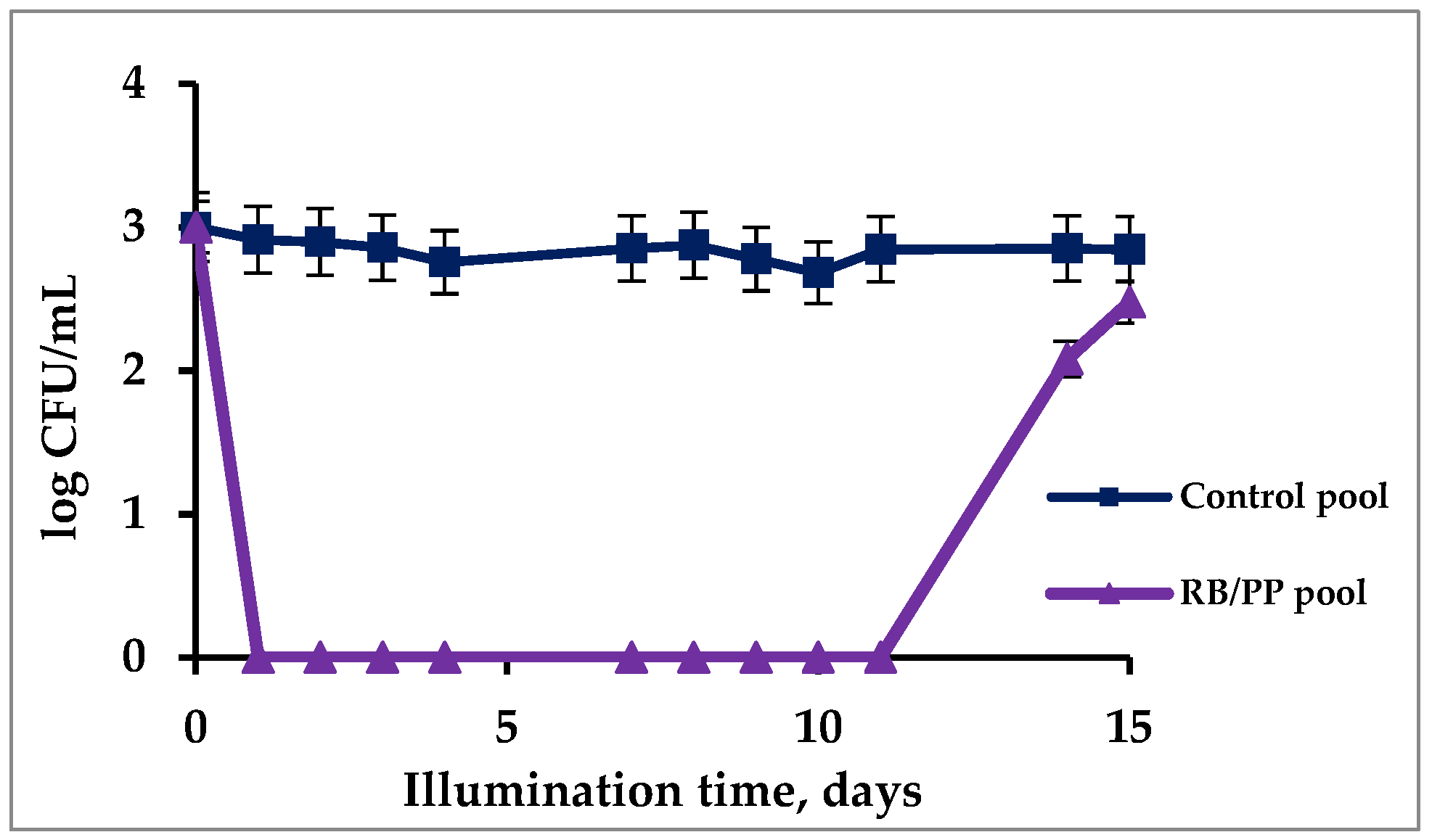
3.5. Photostability of Immobilized PSs
4. Discussion
5. Conclusions
6. Patent
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jemli, M.; Alouini, Z.; Sabbahi, S.; Gueddari, M. Destruction of fecal bacteria in wastewater by three photosensitizers. J. Environ. Monit. 2002, 4, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Thandu, M.; Comuzzi, C.; Goi, D. Phototreatment of water by organic photosensitizers and comparison with inorganic semiconductors. Int. J. Photoenergy 2015. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Abrahamse, H. Inorganic salts and antimicrobial photodynamic therapy: Mechanistic conundrums? Molecules 2018, 23, 3190. [Google Scholar] [CrossRef] [PubMed]
- Lukšienė, Ž. Photodynamic therapy: Mechanism of action and ways to improve the efficiency of treatment. Medicina 2003, 39, 1137–1150. Available online: http://www.sld.cu/galerias/pdf/sitios/rehabilitacion-fis/fotodinamic-mec.pdf (accessed on 25 November 2018). [PubMed]
- Macdonald, I.J.; Dougherty, T.J. Basic principles of photodynamic therapy. J. Porphyr. Phthalocyanines 2001, 5, 105–129. [Google Scholar] [CrossRef]
- Vatansever, F.; de Melo, W.C.; Avci, P.; Vecchio, D.; Sadasivam, M.; Gupta, A.; Chandran, R.; Karimi, M.; Parizotto, N.A.; Yin, R.; et al. Antimicrobial strategies centered around reactive oxygen species—Bactericidal antibiotics, photodynamic therapy and beyond. FEMS Microbiol. Rev. 2013, 37, 955–989. [Google Scholar] [CrossRef]
- García-Fresnadillo, D. Singlet oxygen photosensitizing materials for point-of-use water disinfection with solar reactors. ChemPhotoChem 2018, 2, 512–534. [Google Scholar] [CrossRef]
- Fotinos, N.; Convert, M.; Piffaretti, J.-C.; Gurny, R.; Lange, N. Effects on Gram-negative and Gram-positive bacteria mediated by 5-aminolevulinic acid and 5-aminolevulinic acid derivatives. Antimicrob. Agents Chemother. 2008, 52, 1366–1373. [Google Scholar] [CrossRef]
- Kasimova, K.R.; Sadasivam, M.; Landi, G.; Sarna, T.; Hamblin, M.R. Potentiation of photoinactivation of Gram-positive and Gram-negative bacteria mediated by six phenothiazinium dyes by addition of azide ion. Photochem. Photobiol. Sci. 2014, 13, 1541–1548. [Google Scholar] [CrossRef]
- Käsermann, F.; Kempf, C. Inactivation of enveloped viruses by singlet oxygen thermally generated from a polymeric naphthalene derivative. Antivir. Res. 1998, 38, 55–62. [Google Scholar] [CrossRef]
- Wagner, S.J.; Skripchenko, A.; Robinette, D.; Foley, J.W.; Cincotta, L. Factors affecting virus photoinactivation by a series of phenothiazine dyes. Photochem. Photobiol. 1998, 67, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Sabbahi, S.; Alouini, Z.; Ben Ayed, L.; Jemli, M.; Boudabbous, A. Inactivation of faecal bacteria in wastewater by methylene blue and visible light. Desalin. Water Treat. 2010, 20, 209–219. [Google Scholar] [CrossRef]
- Bonnett, R.; Krysteva, M.; Lalov, I.; Artarsky, S. Water Disinfection Using Photosensitizers Immobilized on Chitosan. Water Res. 2006, 40, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Hernández, M.; Manjón, F.; García-Fresnadillo, D.; Orellana, G. Solar water disinfection by singlet oxygen photogenerated with polymer-supported Ru(II) sensitizers. Sol. Energy 2006, 80, 1382–1387. [Google Scholar] [CrossRef]
- Faust, D.; Funken, K.; Horneck, G.; Milow, B.; Ortner, J.; Sattlegger, M.; Schäfer, M.; Schmitz, C. Immobilized photosensitizers for solar photochemical applications. Sol. Energy 1999, 65, 71–74. [Google Scholar] [CrossRef]
- Nakonechny, F.; Pinkus, A.; Hai, S.; Yehosha, O.; Nitzan, Y.; Nisnevitch, M. Eradication of Gram-positive and Gram-negative bacteria by photosensitizers immobilized in polystyrene. Photochem. Photobiol. 2013, 89, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Valkov, A.; Nakonechny, F.; Nisnevitch, M. Antibacterial properties of Rose Bengal immobilized in polymer supports. Appl. Mech. Mat. 2015, 719–720, 21–24. [Google Scholar] [CrossRef]
- Cahan, R.; Schwartz, R.; Langzam, Y.; Nitzan, Y. Light-activated antibacterial surfaces comprise photosensitizers. Photochem. Photobiol. 2011, 87, 1379–1386. [Google Scholar] [CrossRef]
- Nisnevitch, M.; Lugovskoy, S.; Pinkus, A.; Nakonechny, F.; Nitzan, Y. Antibacterial activity of photosensitizers immobilized onto solid supports via mechanochemical treatment. Recent Res. Devel. Photochem. Photobiol. 2014, 9, 1–23. [Google Scholar]
- Ang, L.Y.; Lim, M.E.; Ong, L.C.; Zhang, Y. Applications of upconversion nanoparticles in imaging, detection and therapy. Nanomedicine 2011, 6, 1273–1288. [Google Scholar] [CrossRef]
- Luz, P.P.; Nobre, T.M.; Serra, O.A.; Zaniquelli, M. Chitosan as a bioadhesive agent between porphyrins and phospholipids in a biomembrane model. J. Nanosci. Nanotechnol. 2011, 11, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Schaap, P.; Thayer, A.L.; Blossey, E.C.; Neckers, D.C. Polymer-based sensitizers for photooxidations. II. J. Am. Chem. Soc. 1975, 97, 3741–3745. [Google Scholar] [CrossRef]
- Nowakowska, M.; Kȩpczyński, M.; Da̧browska, M. Polymeric Photosensitizers, 5. Synthesis and photochemical properties of poly[(N-isopropylacrylamide)-co-(vinylbenzyl chloride)] containing covalently bound rose bengal chromophores. Macromol. Chem. Phys. 2001, 202, 1679–1688. [Google Scholar] [CrossRef]
- Paczkowska, B.; Paczkowski, J.; Neckers, D.C. Heterogeneous and semiheterogeneous photosensitization: Photochemical processes using derivatives of Rose Bengal. Macromolecules 1986, 19, 863–870. [Google Scholar] [CrossRef]
- Nowakowska, M.; Kepczyński, M.; Szczubialka, K. Polymeric photosensitizers, 1. Synthesis and photochemical properties of poly[(sodium p-styrenesulfonate)-co-(4-vinylbenzyl chloride)] containing rose bengal chromophores. Macromol. Chem. Phys. 1995, 196, 2073–2080. [Google Scholar] [CrossRef]
- Manjón, F.; García-Fresnadillo, D.; Orellana, G. Water disinfection with Ru(II) photosensitisers supported on ionic porous silicones. Photochem. Photobiol. Sci. 2009, 8, 926–932. [Google Scholar] [CrossRef]
- Villén, L.; Manjón, F.; García-Fresnadillo, D.; Orellana, G. Solar water disinfection by photocatalytic singlet oxygen production in heterogeneous medium. Appl. Catal. 2006, 69, 1–9. [Google Scholar] [CrossRef]
- Valkov, A.; Nakonechny, F.; Nisnevitch, M. Polymer-immobilized photosensitizers for continuous eradication of bacteria. Int. J. Mol. Sci. 2014, 15, 14984–14996. [Google Scholar] [CrossRef]
- Rivalta, A.E. Synthesis and characterization of group IVB metallocene materials derived from hematoporphyrin IX. In Electronic Thesis; Florida Atlantic University: Boca Raton, FL, USA, 1993; pp. 70–72. Available online: http://fau.digital.flvc.org/islandora/object/fau%3A11720 (accessed on 25 November 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valkov, A.; Raik, K.A.; Mualem-Sinai, Y.; Nakonechny, F.; Nisnevitch, M. Water Disinfection by Immobilized Photosensitizers. Water 2019, 11, 26. https://doi.org/10.3390/w11010026
Valkov A, Raik KA, Mualem-Sinai Y, Nakonechny F, Nisnevitch M. Water Disinfection by Immobilized Photosensitizers. Water. 2019; 11(1):26. https://doi.org/10.3390/w11010026
Chicago/Turabian StyleValkov, Anton, Kate Adar Raik, Yamit Mualem-Sinai, Faina Nakonechny, and Marina Nisnevitch. 2019. "Water Disinfection by Immobilized Photosensitizers" Water 11, no. 1: 26. https://doi.org/10.3390/w11010026
APA StyleValkov, A., Raik, K. A., Mualem-Sinai, Y., Nakonechny, F., & Nisnevitch, M. (2019). Water Disinfection by Immobilized Photosensitizers. Water, 11(1), 26. https://doi.org/10.3390/w11010026





