Combining Chemical Flocculation and Disc Filtration with Managed Aquifer Recharge
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation for Jar Tests
2.2. Jar Tests and Disc Filters
- The flocculator was programmed to 30 s rapid mixing (400 rpm) and 20 min slow mixing (75 rpm). The beaker was filled with 1 L of raw water.
- Before the program was started, the pre-calculated amount of HCl was added to the beaker while mixing.
- The program was started, and when 10 s of the rapid mixing remained, FeCl3 was added.
- When the polymer was used, it was added during slow mixing.
- Once the flocculation program had finished, the raw water was transferred to the disc filter. The permeate was collected and measured after 60 s of filtration.
2.3. Coagulants and Additives
2.4. Water Sample Analysis
2.5. Vomb Lake and Drinking Water Quality
3. Results and Discussion
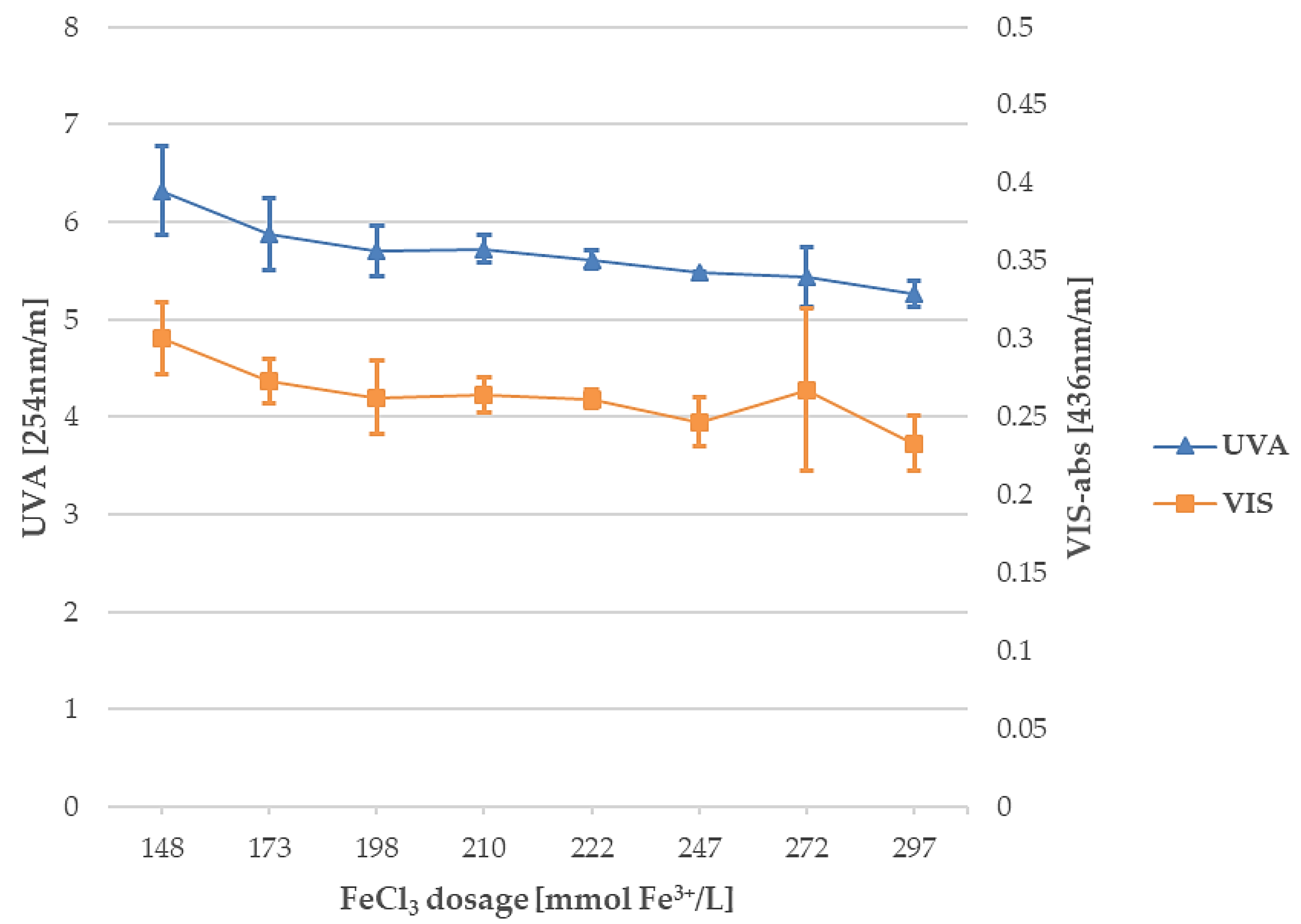
3.1. Fe3+: Coagulant Dosage
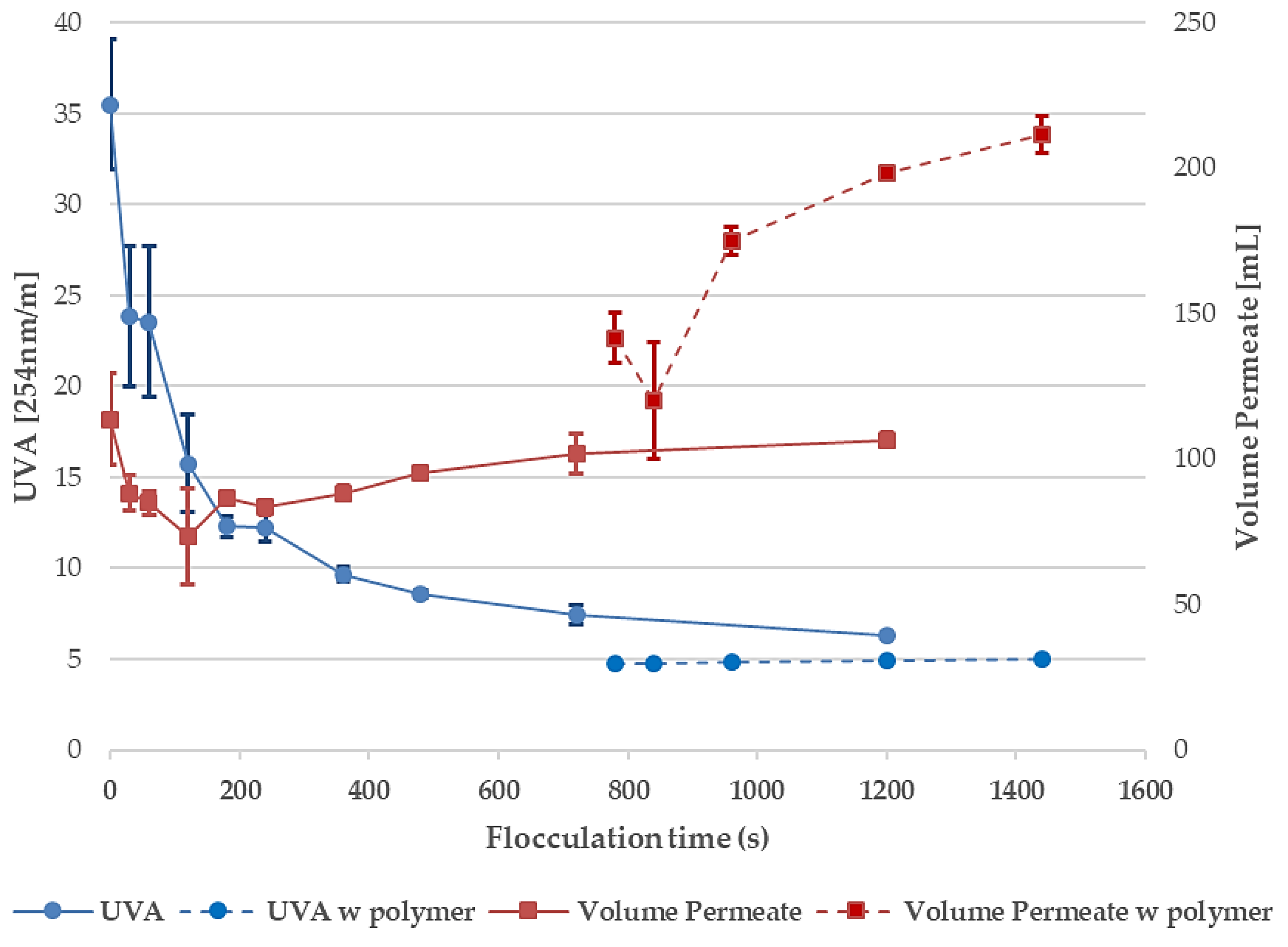
3.2. Effects of Flocculation Time, Polymer Addition, and Disc Filter Filtration
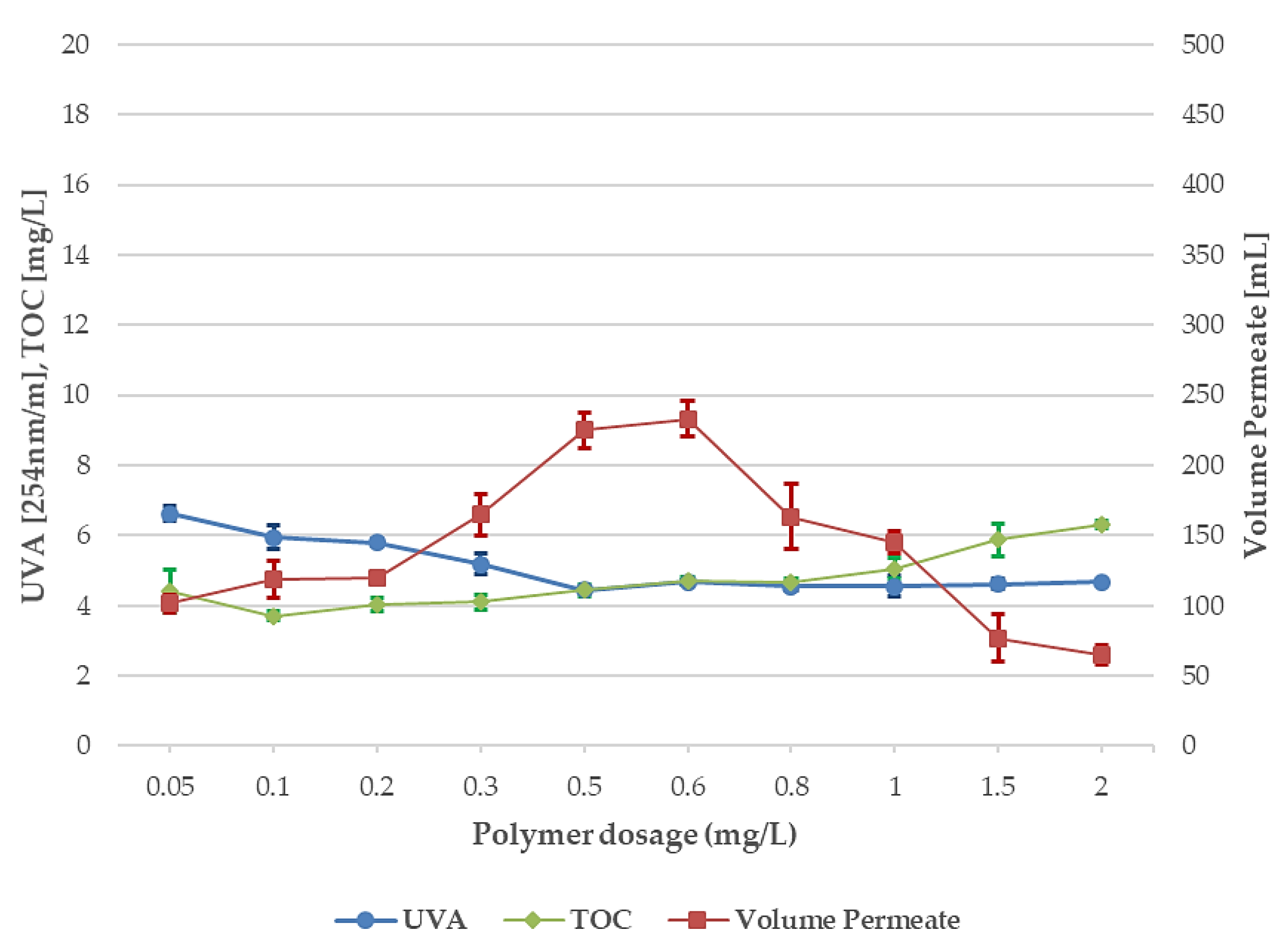
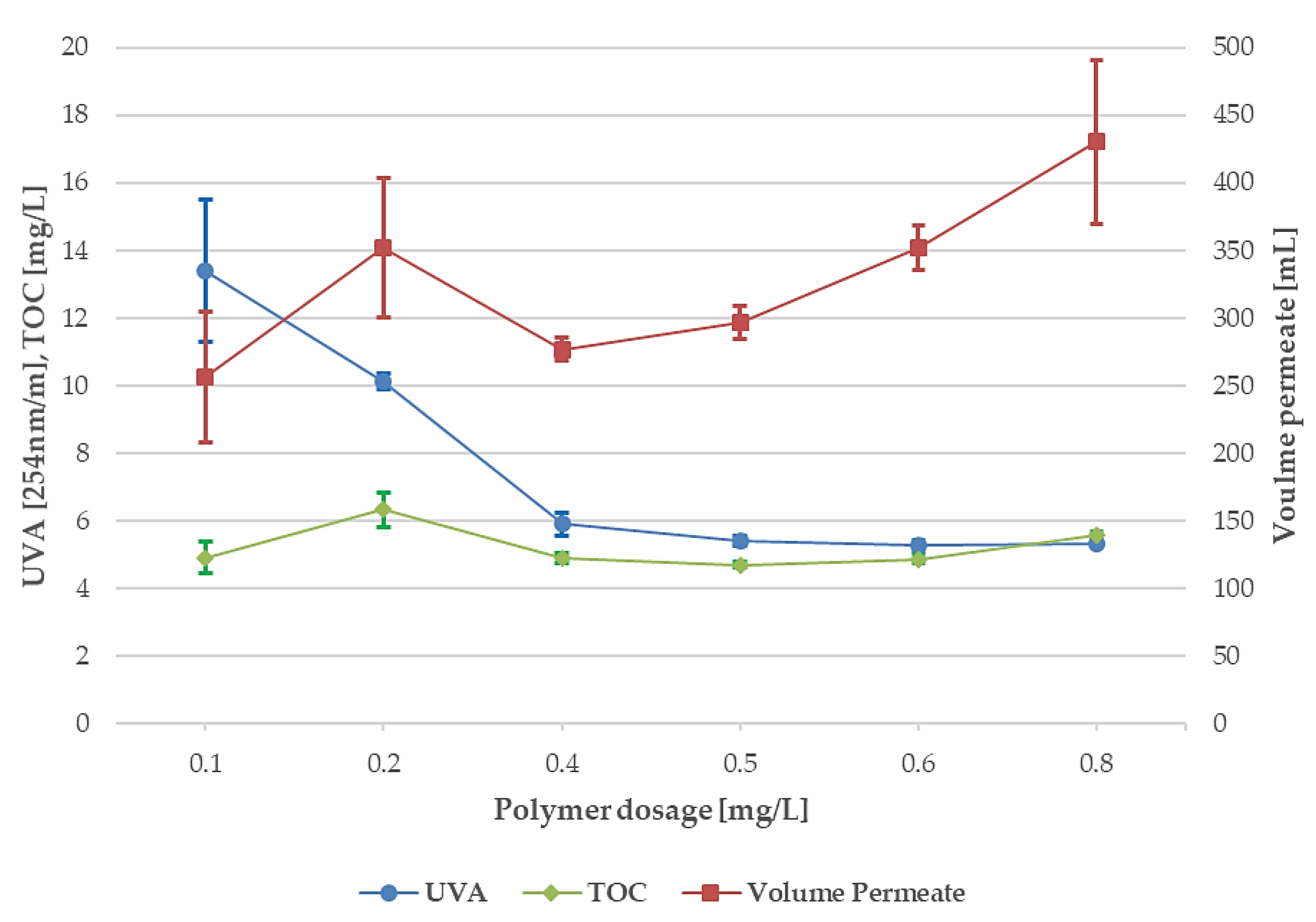
3.3. Polymer Dosage, 10-µm and 40-µm Disc Filters
4. Conclusions
- When combining jar tests with 10- and 40-µm disc filters, it was possible to reduce the UVA and TOC by 74% and 49% and 70% and 50%, respectively.
- The optimum polymer dosage for the TOC and UVA reduction and permeate volume was 0.6 mg/L when using 10-µm disc filters. Good results were also achieved following the Swedish regulatory limits for drinking water, 0.5 mg/L. The polymer dosage exceeding 0.6 mg/L showed signs of clogging and residual polymer in the effluent.
- For 40-µm disc filters, the results varied more at the lower polymer dosage and reached stable results at 0.5 mg/L. Increasing the polymer dosages above 0.5 mg/L did not improve the TOC and UVA reduction, although permeate volume increased.
- Switching the 10-µm disc filters for the 40-µm disc filters using 0.5 mg polymer/L, the permeate volume was increased by approximately 30%.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Svenskt Vatten. 2016. Available online: http://www.svensktvatten.se/Vattentjanster/Dricksvatten/Ravatten/ (accessed on 12 July 2017).
- Hansson, G. Artificial Groundwater Recharge—A Method Used in Swedish Drinking Water Supply; No. 2005-05; Swedish Water Wastewater Assoc. (SWWA): Bromma, Sweden, 2000. [Google Scholar]
- Kolehmainen, R.E.; Langwaldt, J.H.; Puhakka, J.A. Natural organic matter (NOM) removal and structural changes in the bacterial community during artificial groundwater recharge with humic lake water. Water Res. 2007, 41, 2715–2725. [Google Scholar] [CrossRef] [PubMed]
- Hägg, K.; Persson, K.M.; Persson, T.; Zhao, Q. Artificial Recharge Plants for Drinking Water Supply–Groundwork for a Manual for Operation; Swedish Water Wastewater Assoc. (SWWA): Bromma, Sweden, 2018. [Google Scholar]
- Jokela, P.; Eskola, T.; Heinonen, T.; Tanttu, U.; Tyrväinen, J.; Artimo, A. Raw Water Quality and Pretreatment in Managed Aquifer Recharge for Drinking Water Production in Finland. Water 2017, 9, 138. [Google Scholar] [CrossRef]
- Tanttu, U.; Jokela, P. Sustainable drinking water quality improvement by managed aquifer recharge in Tuusula region, Finland. Sustain. Water Resour. Manag. 2018, 4, 225–235. [Google Scholar] [CrossRef]
- Eklund, A.; Axén Mårtensson, J.; Bergström, S.; Björck, E.; Dahné, J.; Lindström, L.; Nordborg, D.; Olsson, J.; Sjökvist, L.S.o.E. Sveriges framtida klimat—Underlag till Dricksvattenutredningen; Swedish Meteorological and Hydrological Institute: Norrköping, Sweden, 2015. [Google Scholar]
- Tranvik, L.; Jansson, M. Terrestrial export of organic carbon. Nature 2002, 415, 861–862. [Google Scholar] [CrossRef]
- Lepistö, A.; Kortelainen, P.; Mattsson, T. Increased organic C and N leaching in a northern boreal river basin in Finland. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Hernebring, C. Operational Experiences at Water Treatment Plants Applying Chemical Coagulation during the Autumn/Spring 1998–99; Swedish Water Wastewater Assoc. (SWWA): Bromma, Sweden, 2003. [Google Scholar]
- Ljunggren, M. Micro screening in wastewater treatment—An overview. Vatten 2006, 62, 171. [Google Scholar]
- Truesdale, G.; Birkbeck, A. Tertiary Treatment of Activaded Sludge Effluents. Water Pollut. Control 1968, 67, 483–492. [Google Scholar]
- Lynam, B.; Ettelt, G.; McAloon, T. Tertiary treatment at Metro Chicago by means of rapid sand filtration and microstrainers. J. (Water Pollut. Control Fed.) 1969, 41, 247–279. [Google Scholar]
- Väänänen, J. Microsieving in Municipal Wastewater Treatment: Chemically Enhanced Primary and Tertiary Treatment; Department of Chemical Engineering, Lund University: Lund, Sweden, 2017. [Google Scholar]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. MWH’s Water Treatment: Principles and Design, 3rd ed.; John Wiley & Sons Incorporated: Hoboken, NJ, USA, 2012. [Google Scholar]
- Lindquist, A.; Gillberg, L.; Hansen, B.; Karlsson, I.; Enkel, A.N.; Pålsson, A. About Water Treatment; Kemira Kemwater: Helsingborg, Sweden, 2003. [Google Scholar]
- Länsstyrelsen i Skåne Län, “Vombsjön,” Redovisning från Sjödatabasen. 2012. Available online: https://www.lansstyrelsen.se/download/18.6ae610001636c9c68e55840/1527064814580/Vombsjön.pdf (accessed on 25 September 2018).
- Löfgren, S.; Andersen, T. The color of Water—Climate induced water color increase in Nordic lakes and streams due to humus. Swed. Univ. Agric. Sci. 2017. Available online: http://info1.ma.slu.se/ima/publikationer/brochure/the_color_of_water.pdf (accessed on 15 May 2017).
- Meili, M. Sources, concentrations and characteristics of organic matter in softwater lakes and streams of the Swedish forest region. Hydrobiologia 1992, 229, 23–41. [Google Scholar] [CrossRef]
- Sydvatten, A.B. Produktionsrapport 2016. 2016. Available online: http://sydvatten.se/wp-content/uploads/2018/04/Produktionsrapport-2016.pdf (accessed on 22 October 2018).
- Livsmedelsverket, Statens livsmedelsverks författningssamling—Statens livsmedelsverks föreskrifter om dricksvatten. 2001. Available online: https://www.livsmedelsverket.se/globalassets/om-oss/lagstiftning/dricksvatten---naturl-mineralv---kallv/slvfs-2001-30-hela_foreskriften.pdf (accessed on 21 September 2018).
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef] [PubMed]


| Sampling Point | pH | TOC [mg/L] | Color [mg Pt/L] | Alkalinity [mg HCO3−/L] | Turbidity [FNU] | U [254 nm/m] | VISVI [436 nm/m] |
|---|---|---|---|---|---|---|---|
| Lake Vomb | 8.3 ± 0.07 1 (n = 7) | 9.1 ± 0.39 1 (n = 4) | 23.8 2 | 152.9 2 | 2.9 2 | 17.7 ± 0.52 1 (n = 8) | 1.54 ± 0.23 1 (n = 8) |
| Limit 3 | >9.0, <7.5 | 4 | 15 | n/a | 0.5 | n/a | n/a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hägg, K.; Cimbritz, M.; Persson, K.M. Combining Chemical Flocculation and Disc Filtration with Managed Aquifer Recharge. Water 2018, 10, 1854. https://doi.org/10.3390/w10121854
Hägg K, Cimbritz M, Persson KM. Combining Chemical Flocculation and Disc Filtration with Managed Aquifer Recharge. Water. 2018; 10(12):1854. https://doi.org/10.3390/w10121854
Chicago/Turabian StyleHägg, Kristofer, Michael Cimbritz, and Kenneth M. Persson. 2018. "Combining Chemical Flocculation and Disc Filtration with Managed Aquifer Recharge" Water 10, no. 12: 1854. https://doi.org/10.3390/w10121854
APA StyleHägg, K., Cimbritz, M., & Persson, K. M. (2018). Combining Chemical Flocculation and Disc Filtration with Managed Aquifer Recharge. Water, 10(12), 1854. https://doi.org/10.3390/w10121854






