Artificial Aquatic Ecosystems
Abstract
1. Introduction
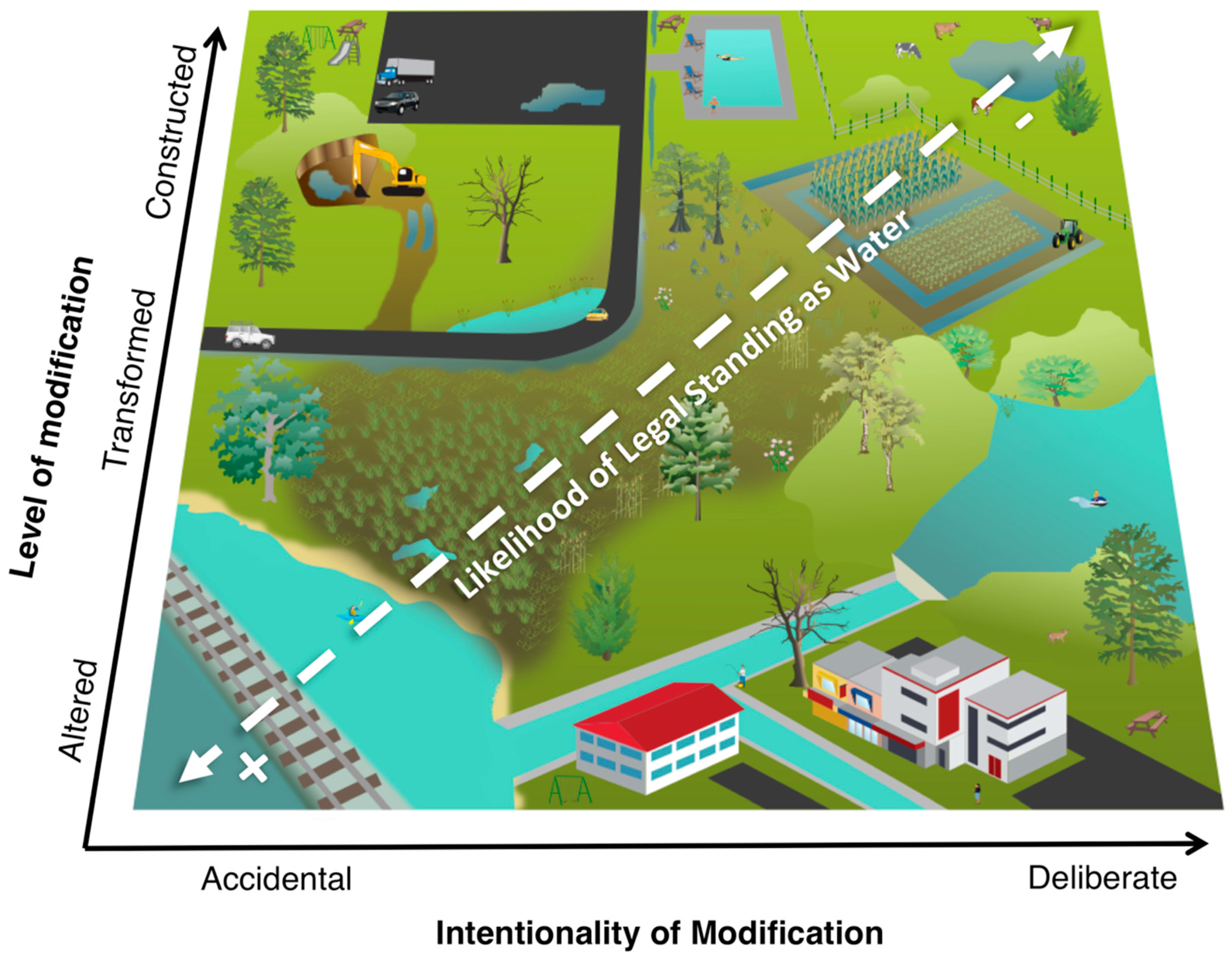
2. Degrees and Axes of Artificiality
Construction, Transformation, and Alteration
3. The Ecological Significance of Artificial Aquatic Systems
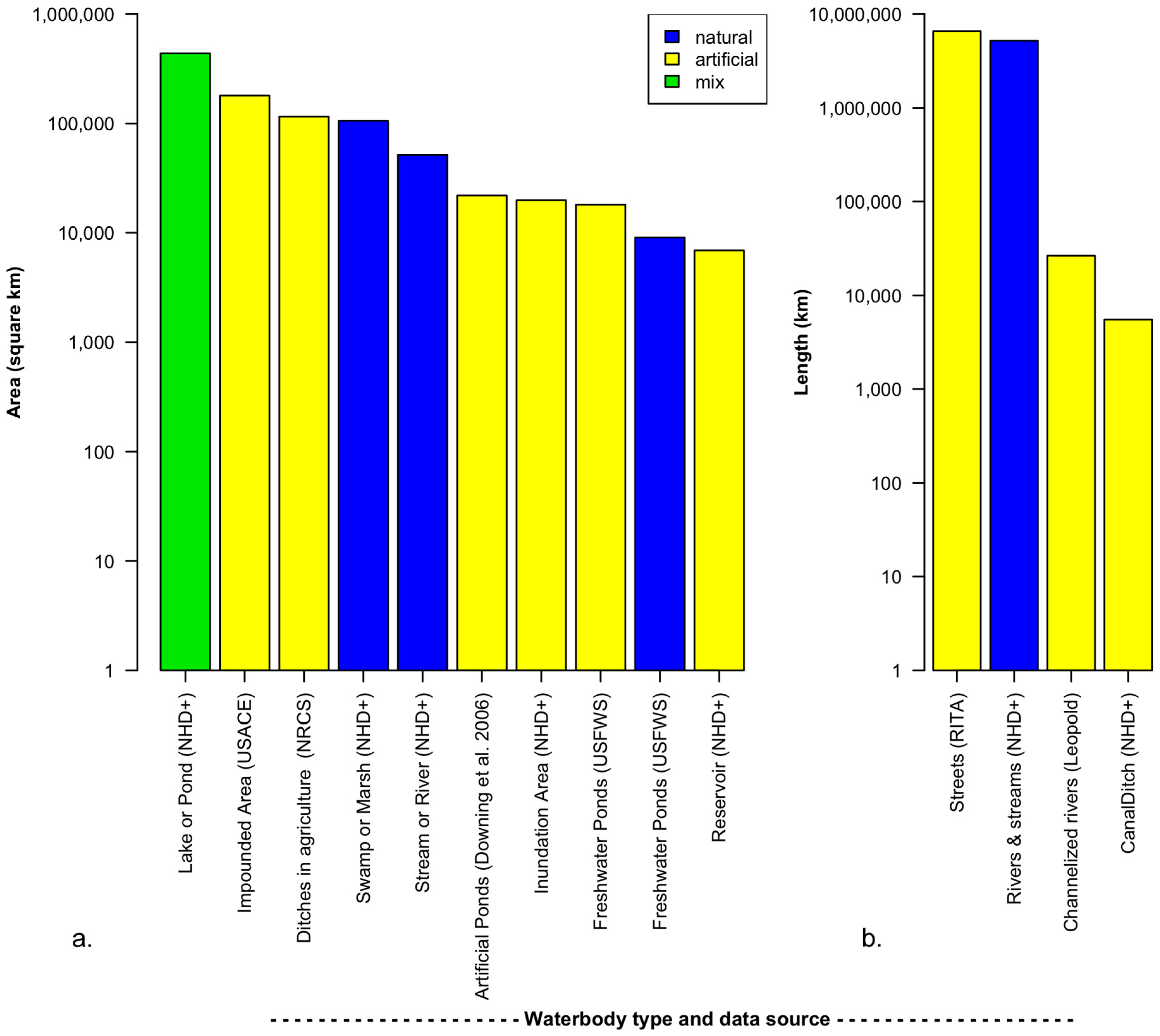
3.1. The Extent and Dynamics of Artificial Aquatic Systems
3.1.1. Deliberately Modified Waterbodies
3.1.2. Accidental Waterbodies
3.1.3. Change
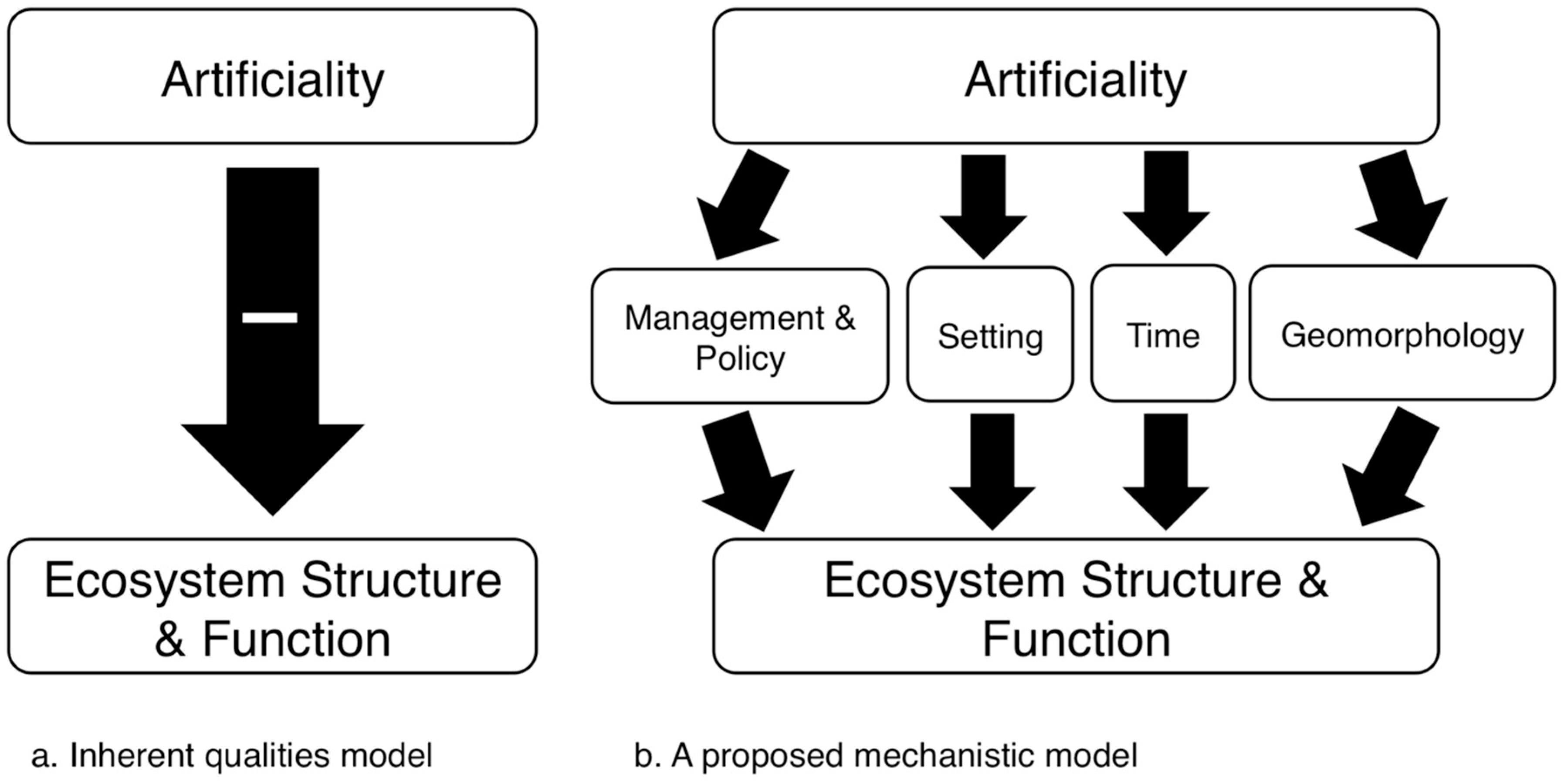
4. The Condition of Artificial Aquatic Systems and Its Drivers
4.1. Setting
4.2. Time
4.3. Design
4.3.1. Design Goals
4.3.2. Planning and Construction
4.3.3. Management and Policy
4.3.4. Monitoring, Learning, and Iteration
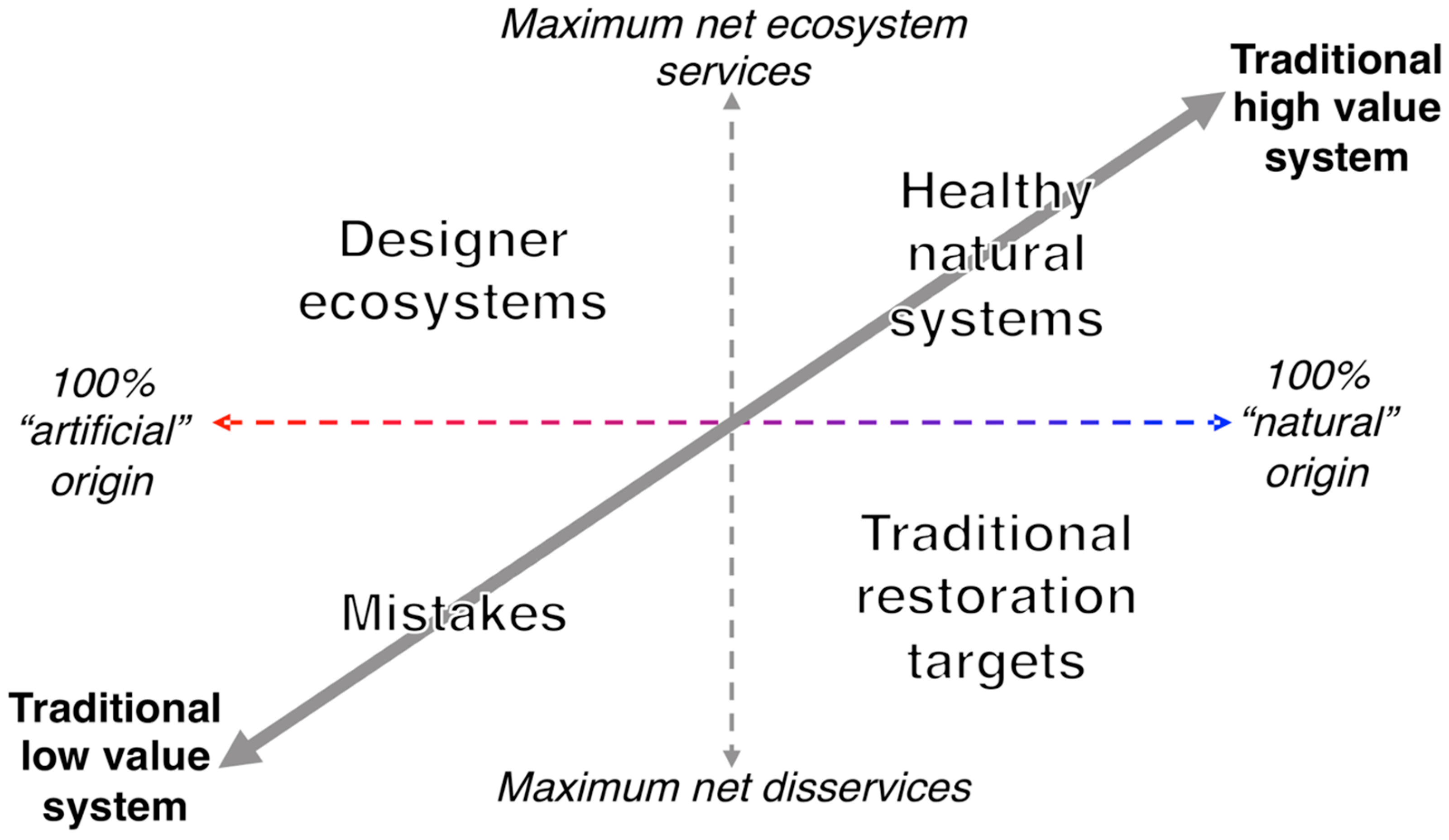
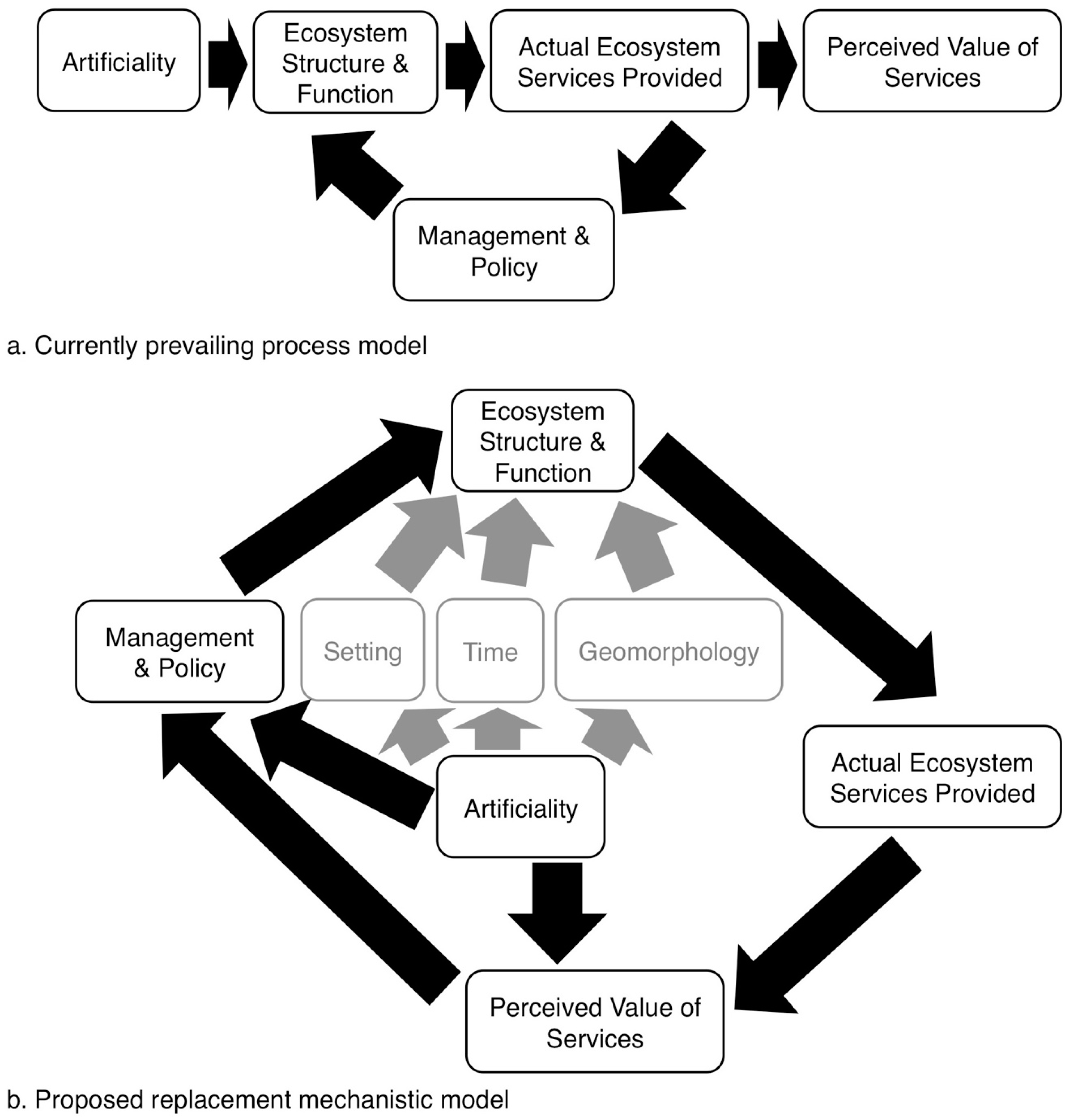
5. Artificiality and Perception of Ecosystem Value
Interactions between Perception and Condition in Artificial Aquatic Systems
6. Conclusions—Artificial Aquatic Ecosystems in Hybrid Hydroscapes
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Ecosystem Service/Disservice | Waterbody Type | Location |
|---|---|---|
| Supporting | ||
| Biodiversity retention | ||
| Rare damselfly habitat | Agricultural ditches | Czech Republic [144] |
| Mite diversity | Agricultural ditch and depression | Slovakia [184] |
| Diverse macroinvertebrate habitat | Agricultural ditches and peat lakes | The Netherlands [185] |
| Stickleback genetic diversity | Agricultural ditches | Japan [186] |
| Reduced fish diversity | Drainage ditches and dredged streams | Estonia [60] |
| Endangered turtle habitat | Mined peat bogs and drainage ditches | Canada [187] |
| Amphibian habitat and breeding area | Anthropogenic small isolated wetlands | USA [188] |
| Rice paddies | Japan [67], Brazil [189] | |
| Carp aquaculture ponds | Poland [190] | |
| Barriers to amphibian dispersal | Roadside ditches | Japan [67] |
| River fragmentation | Reservoirs & dammed rivers | Global [45] |
| Bird habitat | Open water salt marsh management and mosquito ditches | USA [191] |
| Integrated marsh management | USA [192] | |
| Rice fields | Philippines [193], Brazil [189], China, Japan [194], France [195] | |
| Bomb craters | Hungary [25] | |
| Carp aquaculture ponds | Poland [190] | |
| Macroinvertebrate/ zooplankton habitat | Bomb craters | Hungary [25] |
| Rice fields | Brazil [189] | |
| Urban and agricultural ponds | UK [196] | |
| Wetland plant dispersal | Agricultural ditches | Netherlands [197,198] |
| Wetland/aquatic plant habitat | Paddies and ditches | China [199,200], Brazil [189] |
| Drainage ditches | China [201] | |
| Fen restoration and ditch | UK [202] | |
| Open water salt marsh management | USA [191] | |
| Bomb craters | Hungary [25] | |
| Reduced plant diversity | Fish ponds and managed fens | Czech Republic [203] |
| Wetland habitat loss | Forestry drainage ditches | Southeast Asia [68] |
| Shrimp aquaculture ponds | Mexico, Central America, Indonesia [204] | |
| Agricultural drainage | USA [205], Global [206] | |
| Rice fields | Brazil [189] | |
| Fish ponds and managed fens | Czech Republic [203] | |
| Instream habitat loss | River channelization | Global [31,95] |
| Nutrient cycling | ||
| Habitat for common collector-gatherers | Channelized agricultural headwater streams | USA [207] |
| Habitat for common aquatic vegetation | Agricultural ditches and peat lakes | Netherlands [185] |
| Soil erosion | Roadside ditches and culverts | USA [208] |
| Peatland forestry ditches | Finland [209] | |
| Reduced soil bulk density and mineral content | Salt marsh ditches | USA [116] |
| Groundwater recharge | Agricultural drainage ditches | Netherlands [210], China [115] |
| Lowered groundwater table | Forestry drainage ditches | Southeast Asia [68] |
| Open water salt marsh management (sometimes) | USA [191] | |
| Hot springs swimming pools and baths | Turkey [211] | |
| Water overuse | Rice fields | USA [212] |
| Impoundments and abstractions | Global [95] | |
| Mining and industrial diversion | Global [45] | |
| Swimming pools and golf courses | Turkey [211], Global [213] | |
| Provisioning | ||
| Fisheries | ||
| Dispersal corridors for fish and shrimp | Paddy irrigation ditches | Taiwan [214] |
| Fish and mussel habitat and nursery | Irrigation ditch | Japan [215] |
| Nekton habitat (prey fish and shrimp) | Integrated marsh management | USA [192] |
| Open water salt marsh management | USA [191] | |
| Fish habitat | Agricultural ditches | Japan [216] |
| Constructed wetlands, recycle pits, and ditches | ||
| Hunting | Rice fields | USA [212] |
| Abandoned ditches | USA [personal observation] | |
| Animal aquaculture | ||
| Catfish and prawns | Embankment ponds | USA [217] |
| Shrimp | Shrimp aquaculture ponds | Mexico, Central America, Indonesia [204] |
| Carp | Aquaculture ponds | Poland [190] |
| Duck | Integrated Rice-Duck Farms | China, Japan [194] |
| Crops | ||
| Rice | Paddies / fields | China [194,199], USA [212], Philippines [193], Brazil [189], Japan [194], France [195] |
| Vegetables | Rice fields with High Diversity Vegetation Patches | Philippines [193] |
| Biofuel | Cutaway peatland, reed canary grass field and ditches | Finland [218] |
| Timber | Forestry drainage | Southeast Asia [68] |
| Regulating | ||
| Pest control | ||
| Dispersal corridors for diverse, mostly predaceous spiders and ground beetles | Agricultural drainage ditches | Belgium [219] |
| Habitat for frogs, spiders, dragonfly larvae | Paddy ditches | China [199] |
| Mosquito reduction | Salt marsh mosquito ditches and managed ponds | USA [191] |
| Reduced invasive plants | Integrated marsh management | USA [192] |
| Insectivorous birds | Rice fields | Philippines [193] |
| Weed/invertebrate control/spreading by ducks | Integrated Rice-Duck Farms | China, Japan [194] |
| Habitat for pest fish | Irrigation ditches | Japan [220] |
| Movement of invasive predator fish | Irrigation canals | USA [221] |
| Disease vector | ||
| Fecal bacteria export | Roadside ditches | USA [222] |
| Urban ditches and pond | USA [65] | |
| Intestinal parasites | Open sewage | USA [223] |
| Liver flukes | Irrigation ditches | Southeast Asia [62] |
| Schistosomiasis | Paddies, ditches, ponds | China [224] |
| Malaria (mosquitoes) | Puddles, urban farms, construction sites, drains, ditches | Ghana [61] |
| Pollination | Rice fields with High Diversity Vegetation Patches | Philippines [193] |
| Pollutant removal | Paddy fields, ditches, and reservoirs | China [225] |
| Denitrification | Paddy ditches | China [199] |
| Traditional and ecological agricultural drainage ditches | China [226] | |
| Agricultural drainage ditches | USA [227] | |
| Restored wetlands and two-stage ditches | USA [90] | |
| Soil sorption of P | Traditional and ecological agricultural drainage ditches | China [226], USA [228] |
| P efflux | Agricultural drainage ditches | UK [202], Germany [229] |
| Tile drains and ditches | USA, Canada, Sweden, New Zealand [230] | |
| Plant uptake of nutrients | Aquaculture drainage ditches | USA [217] |
| Traditional and ecological agricultural drainage ditches | China [226] | |
| Restored wetlands and two-stage ditches | USA [90] | |
| Vegetated agricultural drainage ditches | USA [231,232] | |
| Nutrient export | Agricultural drainage ditches | Germany [229], China [233] |
| Rice paddies | China [234] | |
| Roadside ditches | USA [235] | |
| Urban ditches and pond | USA [65] | |
| Algal blooms and hypoxia | Urban ditches and pond | USA [65] |
| Organic matter and C retention | Agricultural drainage ditches | USA [228] |
| Greenhouse gas emissions | Rice paddies and drained peat | Southeast Asia [68] |
| Reed canary grass field and ditches in drained peat | Finland [218] | |
| Drained peatlands | UK [236] | |
| Shrimp aquaculture ponds | Mexico, Central America, Indonesia [204] | |
| DOC efflux | Drained peatlands | UK [236] |
| Sediment/solids retention | Ecological drainage system (wetlands, ditches, ponds) | China [237] |
| Agricultural drainage ditches | USA [228] | |
| Sediment/solids export | Roadside ditches | USA [222] |
| Peatland forestry ditches | Finland [209] | |
| Salt export | Agricultural drainage ditches | China [233] |
| Organic pollutant attenuation | Vegetated agricultural ditch | Mexico [238] |
| Pesticide degradation | Stagnant ditches | Netherlands [239] |
| Antibiotic export | Agricultural ditches | Germany [66] |
| Hormone export | Tile drains and ditches | USA [240] |
| Bad smell | Industrial ditch | Taiwan [69] |
| Flood control | Drainage ditches | Netherlands [210] |
| Increased hydrologic flashiness | Roadside ditches | USA [235], Greece [241] |
| Flooding | Flooding irrigation | Mexico [242] |
| Cultural | ||
| Scientific model system | Irrigation canal | USA [152] |
| Agricultural drainage ditches | Netherlands [154] | |
| Bird-watching, photography | Road borrow pit, reservoir | USA [243] |
| Sport | Canals | Netherlands [244] |
| Source of conflict | Impounded rivers | Global [95] |
References
- Hooke, R.L. On the history of humans as geomorphic agents. Geology 2000, 28, 843–846. [Google Scholar] [CrossRef]
- Haff, P.K. Hillslopes, rivers, plows, and trucks: Mass transport on Earth’s surface by natural and technological processes. Earth Surf. Process. Landf. 2010, 35, 1157–1166. [Google Scholar] [CrossRef]
- Wilkinson, B.H. Humans as geologic agents: A deep-time perspective. Geology 2005, 33, 161–164. [Google Scholar] [CrossRef]
- Ellis, E.C.; Ramankutty, N. Putting people in the map: Anthropogenic biomes of the world. Front. Ecol. Environ. 2008, 6, 439–447. [Google Scholar] [CrossRef]
- Steele, M.K.; Heffernan, J.B.; Bettez, N.; Cavender-Bares, J.; Groffman, P.M.; Grove, J.M.; Hall, S.; Hobbie, S.E.; Larson, K.; Morse, J.L.; et al. Convergent surface water distributions in U.S. Cities. Ecosystems 2014, 17, 685–697. [Google Scholar] [CrossRef]
- Downing, J.A.; Prairie, Y.T.; Cole, J.J.; Duarte, C.M.; Tranvik, L.J.; Striegl, R.G.; McDowell, W.H.; Kortelainen, P.; Caraco, N.F.; Melack, J.M.; et al. The global abundance and size distribution of lakes, ponds, and impoundments. Limnol. Oceanogr. 2006, 51, 2388–2397. [Google Scholar] [CrossRef]
- Sayer, C.D. Conservation of aquatic landscapes: Ponds, lakes, and rivers as integrated systems. Wiley Interdiscip. Rev. Water 2014, 1, 573–585. [Google Scholar] [CrossRef]
- Biggs, J.; von Fumetti, S.; Kelly-Quinn, M. The importance of small waterbodies for biodiversity and ecosystem services: Implications for policy makers. Hydrobiologia 2017, 793, 3–39. [Google Scholar] [CrossRef]
- Anderson, B.S.; Phillips, B.M.; Voorhees, J.P.; Cahn, M. Vegetated treatment systems for removing contaminants associated with surface water toxicity in agriculture and urban runoff. J. Vis. Exp. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Higgs, E. Novel and designed ecosystems. Restor. Ecol. 2017, 25, 8–13. [Google Scholar] [CrossRef]
- Crail, T.D.; Gottgens, J.F.; Krause, A.E. Fish community response to evolving channel complexity in an agricultural headwater system. J. Soil Water Conserv. 2011, 66, 295–302. [Google Scholar] [CrossRef]
- Palmer, M.A.; Bernhardt, E.S. Hydroecology and river restoration: Ripe for research and synthesis. Water Resour. Res. 2006, 42. [Google Scholar] [CrossRef]
- Palmer, M.A.; Filoso, S. Restoration of ecosystem services for environmental markets. Science 2009, 325, 575–576. [Google Scholar] [CrossRef] [PubMed]
- Ross, M.R.V.; Bernhardt, E.S.; Doyle, M.; Heffernan, J.B. Designer ecosystems: Incorporating design approaches into applied ecology. Annu. Rev. Environ. Resour. 2015, 40, 419–443. [Google Scholar] [CrossRef]
- Bateman, H.L.; Stromberg, J.C.; Banville, M.J.; Makings, E.; Scott, B.D.; Suchy, A.; Wolkis, D. Novel water sources restore plant and animal communities along an urban river. Ecohydrology 2015, 8, 792–811. [Google Scholar] [CrossRef]
- Palta, M.; du Bray, M.V.; Stotts, R.; Wolf, A.; Wutich, A. Ecosystem services and disservices for a vulnerable population: Findings from urban waterways and wetlands in an American desert city. Hum. Ecol. 2016, 44, 463–478. [Google Scholar] [CrossRef]
- Sueltenfuss, J.P.; Cooper, D.J.; Knight, R.L.; Waskom, R.M. The creation and maintenance of wetland ecosystems from irrigation canal and reservoir seepage in a semi-arid landscape. Wetlands 2013, 33, 799–810. [Google Scholar] [CrossRef]
- Chester, E.T.; Robson, B.J. Anthropogenic refuges for freshwater biodiversity: Their ecological characteristics and management. Biol. Conserv. 2013, 166, 64–75. [Google Scholar] [CrossRef]
- Kueffer, C.; Kaiser-Bunbury, C.N. Reconciling conflicting perspectives for biodiversity conservation in the Anthropocene. Front. Ecol. Environ. 2013, 12, 131–137. [Google Scholar] [CrossRef]
- Hupy, J.P.; Koehler, T. Modern warfare as a significant form of zoogeomorphic disturbance upon the landscape. Geomorphology 2012, 157, 169–182. [Google Scholar] [CrossRef]
- Meade, R.; Wheeler, B.D. Illtyd Pools, Brecon: Description and conservation. New Phytol. 1990, 115, 187–199. [Google Scholar] [CrossRef]
- Landscape Architecture Foundation (LAF). Landscape Performance Series; LAF: Washington, DC, USA, 2016; Available online: http://landscapeperformance.org/ (accessed on 14 December 2016).
- Zollitsch, B.; Christie, J. Report on State Definitions, Jurisdiction and Mitigation Requirements in State Programs for Ephemeral, Intermittent and Perennial Streams in the United States; Association of State Wetland Managers: Windham, ME, USA, 2014. [Google Scholar]
- U.S. Ecological Protection Agency; U.S. Army Corps of Engineers. Definition of ‘Waters of the United States’ under the Clean Water Act; U.S. Army Corps of Engineers and U.S. Environmental Protection Agency: Washington, DC, USA, 2015.
- Vad, C.F.; Pentek, A.L.; Cozma, N.J.; Foldi, A.; Toth, A.; Toth, B.; Bode, N.A.; Mora, A.; Ptacnik, R.; Acs, E.; et al. Wartime scars or reservoirs of biodiversity? The value of bomb crater ponds in aquatic conservation. Biol. Conserv. 2017, 209, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J. Isolated and Ephemeral Wetlands of Southern Appalachia: Biotic Communities and Environmental Drivers across Multiple Temporal and Spatial Scales. Ph.D. Thesis, Clemson University, Clemson, SC, USA, 2014. [Google Scholar]
- Palta, M.M.; Grimm, N.B.; Groffman, P.M. “Accidental” urban wetlands: Ecosystem functions in unexpected places. Front. Ecol. Environ. 2017, 15, 248–256. [Google Scholar] [CrossRef]
- Boler, R.B.; Sikkema, D.A. Tamiami Trail Modifications: Next Steps Project Summary of Findings and Draft Environmental Impact Statement; National Park Service: Homestead, FL, USA, 2010. [Google Scholar]
- Zollitsch, B.; Christie, J. Status and Trends Report on State Wetland Programs in the United States; Association of State Wetland Managers: Windham, ME, USA, 2016. [Google Scholar]
- Hough, P.; Robertson, M. Mitigation under Section 404 of the Clean Water Act: Where it comes from, what it means. Wetl. Ecol. Manag. 2009, 17, 15–33. [Google Scholar] [CrossRef]
- Gregory, K.J. The human role in changing river channels. Geomorphology 2006, 79, 172–191. [Google Scholar] [CrossRef]
- Walter, R.C.; Merritts, D.J. Natural streams and the legacy of water-powered mills. Science 2008, 319, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.J.; Roy, A.H.; Feminella, J.W.; Cottingham, P.D.; Groffman, P.M.; Morgan, R.P. The urban stream syndrome: Current knowledge and the search for a cure. J. N. Am. Benthol. Soc. 2009, 24, 706–723. [Google Scholar] [CrossRef]
- Naftz, D.; Angeroth, C.; Kenney, T.; Waddell, B.; Darnall, N.; Silva, S.; Perschon, C.; Whitehead, J. Anthropogenic influences on the input and biogeochemical cycling of nutrients and mercury in Great Salt Lake, Utah, USA. Appl. Geochem. 2008, 23, 1731–1744. [Google Scholar] [CrossRef]
- Wohl, E.; Lane, S.N.; Wilcox, A.C. The science and practice of river restoration. Water Resour. Res. 2015, 51, 5974–5997. [Google Scholar] [CrossRef]
- Hallett, L.M.; Standish, R.J.; Hulvey, K.B.; Gardener, M.R.; Suding, K.N.; Starzomski, B.M.; Murphy, S.D.; Harris, J.A. Towards a conceptual framework for novel ecosystems. In Novel Ecosystems; John Wiley & Sons: Hoboken, NJ, USA, 2013; pp. 16–28. [Google Scholar]
- Hobbs, R.J. Degraded or just different? Perceptions and value judgements in restoration decisions. Restor. Ecol. 2016, 24, 153–158. [Google Scholar] [CrossRef]
- Crifasi, R.R. Reflections in a stock pond: Are anthropogenically derived freshwater ecosystems natural, artificial, or something else? Environ. Manag. 2005, 36, 625–639. [Google Scholar] [CrossRef] [PubMed]
- Downing, J.A.; Cole, J.J.; Duarte, C.M.; Middelburg, J.J.; Melack, J.M.; Prairie, Y.T.; Kortelainen, P.; Striegl, R.G.; McDowell, W.H.; Tranvik, L.J. Global abundance and size distribution of streams and rivers. Inland Waters 2012, 2, 229–236. [Google Scholar] [CrossRef]
- Rabkin, J.A. Against the EPA, absurdity is no defense symposium: The federal leviathan: Is there any area of modern life to which federal government does not extend: The thirty-second annual Federalist Society national student symposium on law and public policy-2013 section II: Environmental and property laws. Harv. J. L. Pub. Policy 2014, 37, 41–50. [Google Scholar]
- The United States Department of Agriculture (USDA). Summary Report: 2012 National Resources Inventory; Natural Resources Conservation Service and Center for Survey Statistics and Methodology, Iowa State University: Washington, DC, USA; Ames, IA, USA, 2015.
- Leopold, L.B. A reverence for rivers. Geology 1977, 5, 429–430. [Google Scholar] [CrossRef]
- Naiman, R.J.; Magnuson, J.J.; Stanford, J.A.; McKnight, D.M. The Freshwater Imperative: A Research Agenda; Island Press: Washington, DC, USA, 1995. [Google Scholar]
- Gleick, P.H.; Singh, A.; Shi, H. Threats to the World’s Freshwater Resources; Pacific Institute for Studies in Development, Environment, and Security: Oakland, CA, USA, 2001. [Google Scholar]
- Meybeck, M. Global analysis of river systems: From Earth system controls to Anthropocene syndromes. Philos. Trans. R. Soc. Lond. Biol. Sci. 2003, 358, 1935–1955. [Google Scholar] [CrossRef] [PubMed]
- Research and Innovative Technology Administration (RITA). Public Road and Street Mileage in the United States by Type of Surface; U.S. Department of Transportation, Research and Innovative Technology Administration, Bureau of Transportation Statistics: Washington, DC, USA, 2014. Available online: http://www.rita.dot.gov/bts/sites/rita.dot.gov.bts/files/publications/national_transportation_statistics/html/table_01_04.html (accessed on 18 June 2018).
- Paulsen, S.G.; Stoddard, J.L.; Holdsworth, S.; Mayio, A.; Tarquinio, E. Wadeable Streams Assessment: A Colllaborative Survey of the Nation’s Streams; U.S. Environmental Protection Agency: Washington, DC, USA, 2004. [Google Scholar]
- National Hydrography Dataset; U.S. Geological Survey: Washington, DC, USA, 2015.
- National Inventory of Dams; U.S. Army Corps of Engineers: Washington, DC, USA, 2013.
- Dahl, T.E. Status and Trends of Wetlands in the Conterminous United States 2004 to 2009; U.S. Department of the Interior; Fish and Wildlife Service: Washington, DC, USA, 2011.
- Wohl, E.; Lininger, K.B.; Baron, J. Land before water: The relative temporal sequence of human alteration of freshwater ecosystems in the conterminous United States. Anthropocene 2017, 18, 27–46. [Google Scholar] [CrossRef]
- Pekel, J.F.; Cottam, A.; Gorelick, N.; Belward, A.S. High-resolution mapping of global surface water and its long-term changes. Nature 2016, 540, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Berg, M.D.; Popescu, S.C.; Wilcox, B.P.; Angerer, J.P.; Rhodes, E.C.; McAlister, J.; Fox, W.E. Small farm ponds: Overlooked features with important impacts on watershed sediment transport. J. Am. Water Resour. Assoc. 2016, 52, 67–76. [Google Scholar] [CrossRef]
- Oertli, B. Editorial: Freshwater biodiversity conservation: The role of artificial ponds in the 21st century. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 28, 264–269. [Google Scholar] [CrossRef]
- Rapinel, S.; Hubert-Moy, L.; Clément, B.; Nabucet, J.; Cudennec, C. Ditch network extraction and hydrogeomorphological characterization using lidar-derived DTM in wetlands. Hydrol. Res. 2015, 46, 276–290. [Google Scholar] [CrossRef]
- Bailly, J.S.; Lagacherie, P.; Millier, C.; Puech, C.; Kosuth, P. Agrarian landscapes linear features detection from lidar: Application to artificial drainage networks. Int. J. Remote Sens. 2008, 29, 3489–3508. [Google Scholar] [CrossRef]
- Cazorzi, F.; Dalla Fontana, G.; De Luca, A.; Sofia, G.; Tarolli, P. Drainage network detection and assessment of network storage capacity in agrarian landscape. Hydrol. Process. 2013, 27, 541–553. [Google Scholar] [CrossRef]
- Herzon, I.; Helenius, J. Agricultural drainage ditches, their biological importance and functioning. Biol. Conserv. 2008, 141, 1171–1183. [Google Scholar] [CrossRef]
- National Lakes Assessment: A Collaborative Survey of the Nation’s Lakes; U.S. Environmental Protection Agency: Washington, DC, USA, 2009.
- Rosenvald, R.; Jarvekulg, R.; Lohmus, A. Fish assemblages in forest drainage ditches: Degraded small streams or novel habitats? Limnologica 2014, 46, 37–44. [Google Scholar] [CrossRef]
- Mattah, P.A.D.; Futagbi, G.; Amekudzi, L.K.; Mattah, M.M.; de Souza, D.K.; Kartey-Attipoe, W.D.; Bimi, L.; Wilson, M.D. Diversity in breeding sites and distribution of Anopheles mosquitoes in selected urban areas of southern Ghana. Parasit. Vectors 2017, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C. Examining landscape determinants of Opisthorchis viverrini transmission. EcoHealth 2012, 9, 328–341. [Google Scholar] [CrossRef] [PubMed]
- Maheu-Giroux, M.; de Blois, S. Landscape ecology of Phragmites australis invasion in networks of linear wetlands. Landsc. Ecol. 2007, 22, 285–301. [Google Scholar] [CrossRef]
- O’Connor, J.H.; Rothermel, B.B. Distribution and population characteristics of African Jewelfish and Brown Hoplo in modified wetlands in south Florida. Am. Midl. Nat. 2013, 170, 52–65. [Google Scholar] [CrossRef]
- Mallin, M.A.; McIver, M.R. Pollutant impacts to Cape Hatteras National Seashore from urban runoff and septic leachate. Mar. Pollut. Bull. 2012, 64, 1356–1366. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.; Spielmeyer, A.; Frings, R.M.; Hamscher, G.; Schuttrumpf, H. From agricultural fields to surface water systems: The overland transport of veterinary antibiotics. J. Soils Sedim. 2015, 15, 1630–1634. [Google Scholar] [CrossRef]
- Naito, R.; Sakai, M.; Morimoto, Y. Negative effects of deep roadside ditches on Pelophylax porosa brevipoda dispersal and migration in comparison with Hyla japonica in a rice paddy area in Japan. Zool. Sci. 2012, 29, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Hergoualc’h, K.A.; Verchot, L.V. Changes in soil CH4 fluxes from the conversion of tropical peat swamp forests: A meta-analysis. J. Integr. Environ. Sci. 2012, 9, 31–39. [Google Scholar] [CrossRef]
- Mao, I.F.; Chen, M.R.; Wang, L.; Chen, M.L.; Lai, S.C.; Tsai, C.J. Method development for determining the malodor source and pollution in industrial park. Sci. Total Environ. 2012, 437, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Perring, M.P.; Manning, P.; Hobbs, R.J.; Lugo, A.E.; Ramalho, C.E.; Standish, R.J. Novel urban ecosystems and ecosystem services. In Novel Ecosystems; John Wiley & Sons: Hoboken, NJ, USA, 2013; pp. 310–325. [Google Scholar]
- Salomon Cavin, J. Beyond prejudice: Conservation in the city. A case study from Switzerland. Biol. Conserv. 2013, 166, 84–89. [Google Scholar] [CrossRef]
- Robert, A.; Fontaine, C.; Veron, S.; Monnet, A.C.; Legrand, M.; Clavel, J.; Chantepie, S.; Couvet, D.; Ducarme, F.; Fontaine, B.; et al. Fixism and conservation science. Conserv. Biol. 2017, 31, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.; Soule, M.E.; Terborgh, J. ’New conservation’ or surrender to development? Anim. Conserv. 2014, 17, 509–515. [Google Scholar] [CrossRef]
- Ridder, B. An exploration of the value of naturalness and wild nature. J. Agric. Environ. Ethics 2007, 20, 195–213. [Google Scholar] [CrossRef]
- Reid, N.; Dingerkus, S.K.; Stone, R.E.; Buckley, J.; Beebee, T.J.C.; Marnell, F.; Wilkinson, J.W. Assessing historical and current threats to common frog (Rana temporaria) populations in Ireland. J. Herpetol. 2014, 48, 13–19. [Google Scholar] [CrossRef]
- Sebastian-Gonzalez, E.; Sanchez-Zapata, J.A.; Botella, F. Agricultural ponds as alternative habitat for waterbirds: Spatial and temporal patterns of abundance and management strategies. Eur. J. Wildl. Res. 2010, 56, 11–20. [Google Scholar] [CrossRef]
- Ruggiero, A.; Cereghino, R.; Figuerola, J.; Marty, P.; Angelibert, S. Farm ponds make a contribution to the biodiversity of aquatic insects in a French agricultural landscape. C. R. Biol. 2008, 331, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Cereghino, R.; Ruggiero, A.; Marty, P.; Angelibert, S. Biodiversity and distribution patterns of freshwater invertebrates in farm ponds of a south-western French agricultural landscape. Hydrobiologia 2008, 597, 43–51. [Google Scholar] [CrossRef]
- Abellan, P.; Sanchez-Fernandez, D.; Millan, A.; Botella, F.; Sanchez-Zapata, J.A.; Gimenez, A. Irrigation pools as macroinvertebrate habitat in a semi-arid agricultural landscape (SE Spain). J. Arid. Environ. 2006, 67, 255–269. [Google Scholar] [CrossRef]
- Davies, B.; Biggs, J.; Williams, P.; Whitfield, M.; Nicolet, P.; Sear, D.; Bray, S.; Maund, S. Comparative biodiversity of aquatic habitats in the European agricultural landscape. Agric. Ecosyst. Environ. 2008, 125, 1–8. [Google Scholar] [CrossRef]
- Biggs, J.; Williams, P.; Whitfield, M.; Nicolet, P.; Weatherby, A. 15 years of pond assessment in Britain: Results and lessons learned from the work of pond conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 693–714. [Google Scholar] [CrossRef]
- Cereghino, R.; Boix, D.; Cauchie, H.M.; Martens, K.; Oertli, B. The ecological role of ponds in a changing world. Hydrobiologia 2014, 723, 1–6. [Google Scholar] [CrossRef]
- Kadoya, T.; Suda, S.; Washitani, I. Dragonfly crisis in Japan: A likely consequence of recent agricultural habitat degradation. Biol. Conserv. 2009, 142, 1899–1905. [Google Scholar] [CrossRef]
- Canessa, S.; Oneto, F.; Ottonello, D.; Arillo, A.; Salvidio, S. Land abandonment may reduce disturbance and affect the breeding sites of an endangered amphibian in northern Italy. Oryx 2013, 47, 280–287. [Google Scholar] [CrossRef]
- Casas, J.J.; Sanchez-Oliver, J.S.; Sanz, A.; Furne, M.; Trenzado, C.; Juan, M.; Paracuellos, M.; Suarez, M.D.; Fuentes, F.; Gallego, I.; et al. The paradox of the conservation of an endangered fish species in a Mediterranean region under agricultural intensification. Biol. Conserv. 2011, 144, 253–262. [Google Scholar] [CrossRef]
- Taylor, C.A.; Robison, H.W. A new burrowing crayfish of the genus Fallicambarus Hobbs, 1969 (decapoda: Cambaridae) from the red river drainage of the southcentral united states. Zootaxa 2016, 4144, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Zang, D.K. Cerasus Laoshanensis (Rosaceae), a new species from Shandong, China. Ann. Bot. Fenn. 2017, 54, 135–137. [Google Scholar] [CrossRef]
- Gan, Q.L.; Li, X.W. Impatiens baokangensis (Balsaminaceae), a new species from Hubei, China. Ann. Bot. Fenn. 2016, 53, 145–148. [Google Scholar] [CrossRef]
- Brand, A.B.; Snodgrass, J.W. Value of artificial habitats for amphibian reproduction in altered landscapes. Conserv. Biol. 2010, 24, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Roley, S.S.; Tank, J.L.; Tyndall, J.C.; Witter, J.D. How cost-effective are cover crops, wetlands, and two-stage ditches for nitrogen removal in the Mississippi river basin? Water Resour. Econ. 2016, 15, 43–56. [Google Scholar] [CrossRef]
- Perez-Gutierrez, J.D.; Paz, J.O.; Tagert, M.L.M. Seasonal water quality changes in on-farm water storage systems in a south-central US agricultural watershed. Agric. Water Manag. 2017, 187, 131–139. [Google Scholar] [CrossRef]
- Cooper, C.M.; Moore, M.T.; Bennett, E.R.; Sammie Smith, J.; Farris, J.L. Alternative environmental benefits of agricultural drainage ditches. Verh. Int. Verein Limnol. 2002, 28, 1678–1682. [Google Scholar] [CrossRef]
- Palmer, M.A. Reforming watershed restoration: Science in need of application and applications in need of science. Estuar. Coasts 2009, 32, 1–17. [Google Scholar] [CrossRef]
- Stewart, J.S.; Wang, L.Z.; Lyons, J.; Horwatich, J.A.; Bannerman, R. Influences of watershed, riparian-corridor, and reach-scale characteristics on aquatic biota in agricultural watersheds. J. Am. Water Resour. Assoc. 2001, 37, 1475–1487. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.; Knowler, D.J.; Leveque, C.; Naiman, R.J.; Prieur-Richard, A.H.; Soto, D.; Stiassny, M.L.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. Camb. Philos. Soc. 2006, 81, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Burcher, C.L.; Valett, H.M.; Benfield, E.F. The land-cover cascade: Relationships coupling land and water. Ecology 2007, 88, 228–242. [Google Scholar] [CrossRef]
- Suding, K.N. Toward an era of restoration in ecology: Successes, failures, and opportunities ahead. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 465–487. [Google Scholar] [CrossRef]
- Veraart, A.J.; Dimitrov, M.R.; Schrier-Uijl, A.P.; Smidt, H.; de Klein, J.J.M. Abundance, activity and community structure of denitrifiers in drainage ditches in relation to sediment characteristics, vegetation and land-use. Ecosystems 2017, 20, 928–943. [Google Scholar] [CrossRef]
- Bremigan, M.T.; Soranno, P.A.; Gonzalez, M.J.; Bunnell, D.B.; Arend, K.K.; Renwick, W.H.; Stein, R.A.; Vanni, M.J. Hydrogeomorphic features mediate the effects of land use/cover on reservoir productivity and food webs. Limnol. Oceanogr. 2008, 53, 1420–1433. [Google Scholar] [CrossRef]
- Simon, T.N.; Travis, J. The contribution of man-made ditches to the regional stream biodiversity of the new river watershed in the Florida panhandle. Hydrobiologia 2011, 661, 163–177. [Google Scholar] [CrossRef]
- Francesiaz, C.; Guilbault, E.; Lebreton, J.D.; Trouvilliez, J.; Besnard, A. Colony persistence in waterbirds is constrained by pond quality and land use. Freshw. Biol. 2017, 62, 119–132. [Google Scholar] [CrossRef]
- Mehaffey, M.H.; Nash, M.S.; Wade, T.G.; Ebert, D.W.; Jones, K.B.; Rager, A. Linking land cover and water quality in New York City’s water supply watersheds. Environ. Monit. Assess. 2005, 107, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, E.S.; Palmer, M.A. Restoring streams in an urbanizing world. Freshw. Biol. 2007, 52, 738–751. [Google Scholar] [CrossRef]
- Brown, L.E.; Milner, A.M. Rapid loss of glacial ice reveals stream community assembly processes. Glob. Chang. Biol. 2012, 18, 2195–2204. [Google Scholar] [CrossRef]
- Sayer, C.; Andrews, K.; Shilland, E.; Edmonds, N.; Edmonds-Brown, R.; Patmore, I.; Emson, D.; Axmacher, J. The role of pond management for biodiversity conservation in an agricultural landscape. Aquat. Conserv. Mar. Freshw. Ecosyst. 2012, 22, 626–638. [Google Scholar] [CrossRef]
- Arthaud, F.; Vallod, D.; Robin, J.; Wezel, A.; Bornette, G. Short-term succession of aquatic plant species richness along ecosystem productivity and dispersal gradients in shallow lakes. J. Veg. Sci. 2013, 24, 148–156. [Google Scholar] [CrossRef]
- Drake, C.M.; Stewart, N.F.; Palmer, M.A.; Kindemba, V.L. The Ecological Status of Ditch Systems: An Investigation into the Current Status of the Aquatic Invertebrate and Plant communities of Grazing Marsh Ditch Systems in England and Wales; Buglife-The Invertebrate Conservation Trust: Peterborough, UK, 2010. [Google Scholar]
- Ballantine, K.; Schneider, R. Fifty-five years of soil development in restored freshwater depressional wetlands. Ecol. Appl. 2009, 19, 1467–1480. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, J.L.; Ward, A.D.; Witter, J.D. Evaluating geomorphic change in constructed two-stage ditches. J. Am. Water Resour. Assoc. 2015, 51, 910–922. [Google Scholar] [CrossRef]
- Palmer, M.A.; Drake, M.; Stewart, N. A Manual for the Survey and Evaluation of the Aquatic Plant and Invertebrate Assemblages of Grazing Marsh Ditch Systems; Buglife-The Invertebrate Conservation Trust: Peterborough, UK, 2013. [Google Scholar]
- Rehounkova, K.; Prach, K. Spontaneous vegetation succession in gravel-sand pits: A potential for restoration. Restor. Ecol. 2008, 16, 305–312. [Google Scholar] [CrossRef]
- Oosting, H.J. An ecological analysis of the plant communities of Piedmont, North Carolina. Am. Midl. Nat. 1942, 28, 1–126. [Google Scholar] [CrossRef]
- Zhang, Z.D.; Brown, R.; Bauer, J.; Bedard-Haughn, A. Nutrient dynamics within drainage ditches under recent, medium, and long-term drainage in the black soil zone of southeastern Saskatchewan. Geoderma 2017, 289, 66–71. [Google Scholar] [CrossRef]
- Kaushal, S.; McDowell, W.; Wollheim, W.; Johnson, T.; Mayer, P.; Belt, K.; Pennino, M. Urban evolution: The role of water. Water 2015, 7, 4063–4087. [Google Scholar] [CrossRef]
- Li, M.; Li, L.X.; Liao, H.S.; Huang, Z.W.; Huang, W.D.A. The influence of drainage on wetland degradation in Zoige plateau. Disaster Adv. 2012, 5, 659–666. [Google Scholar]
- Vincent, R.E.; Burdick, D.M.; Dionne, M. Ditching and ditch-plugging in New England salt marshes: Effects on hydrology, elevation, and soil characteristics. Estuar. Coasts 2013, 36, 610–625. [Google Scholar] [CrossRef]
- Bennett, E.M.; Peterson, G.D.; Gordon, L.J. Understanding relationships among multiple ecosystem services. Ecol. Lett. 2009, 12, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Power, A.G. Ecosystem services and agriculture: Tradeoffs and synergies. Philos. Trans. R. Soc. Lond. Biol. Sci. 2010, 365, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Hale, R.; Baxter, C.; Burnham, M.; Marsh, K. Co-Producing Urban River Imaginaries over Time: An Idaho River and Its community; Society for Freshwater Science: Raleigh, NC, USA, 2017. [Google Scholar]
- Landers, J. Environmental engineering-Los Angeles aims to combine river restoration, urban revitalization. Civ. Eng. 2007, 77, 11–13. [Google Scholar]
- Polat, A.T.; Akay, A. Relationships between the visual preferences of urban recreation area users and various landscape design elements. Urban For. Urban Green. 2015, 14, 573–582. [Google Scholar] [CrossRef]
- Smith, R.F.; Hawley, R.J.; Neale, M.W.; Vietz, G.J.; Diaz-Pascacio, E.; Herrmann, J.; Lovell, A.C.; Prescott, C.; Rios-Touma, B.; Smith, B.; et al. Urban stream renovation: Incorporating societal objectives to achieve ecological improvements. Freshw. Sci. 2016, 35, 364–379. [Google Scholar] [CrossRef]
- Lave, R. Fields and Streams: Stream Restoration, Neoliberalism, and the Future of Environmental Science; University of Georgia Press: Athens, GA, USA, 2012. [Google Scholar]
- Doyle, M.W.; Singh, J.; Lave, R.; Robertson, M.M. The morphology of streams restored for market and nonmarket purposes: Insights from a mixed natural-social science approach. Water Resour. Res. 2015, 51, 5603–5622. [Google Scholar] [CrossRef]
- Podolak, K.; Kondolf, G.M. The line of beauty in river designs: Hogarth’s aesthetic theory on capability brown’s eighteenth-century river design and twentieth-century river restoration design. Landsc. Res. 2016, 41, 149–167. [Google Scholar] [CrossRef]
- Dollinger, J.; Dages, C.; Bailly, J.S.; Lagacherie, P.; Voltz, M. Managing ditches for agroecological engineering of landscape. A review. Agron. Sustain. Dev. 2015, 35, 999–1020. [Google Scholar] [CrossRef]
- Palmer, M.A.; Menninger, H.L.; Bernhardt, E. River restoration, habitat heterogeneity and biodiversity: A failure of theory or practice? Freshw. Biol. 2010, 55, 205–222. [Google Scholar] [CrossRef]
- Bernhardt, E.S.; Palmer, M.A. River restoration: The fuzzy logic of repairing reaches to reverse catchment scale degradation. Ecol. Appl. 2011, 21, 1926–1931. [Google Scholar] [CrossRef] [PubMed]
- Kumwimba, M.N.; Zhu, B. Effectiveness of vegetated drainage ditches for domestic sewage effluent mitigation. Bull. Environ. Contam. Toxicol. 2017, 98, 682–689. [Google Scholar] [CrossRef] [PubMed]
- Verlicchi, P.; Zambello, E. How efficient are constructed wetlands in removing pharmaceuticals from untreated and treated urban wastewaters? A review. Sci. Total Environ. 2014, 470, 1281–1306. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Zhu, G.B.; Ng, W.J.; Tan, S.K. A review on removing pharmaceutical contaminants from wastewater by constructed wetlands: Design, performance and mechanism. Sci. Total Environ. 2014, 468, 908–932. [Google Scholar] [CrossRef] [PubMed]
- Powell, G.E.; Ward, A.D.; Mecklenburg, D.E.; Jayakaran, A.D. Two-stage channel systems: Part 1, a practical approach for sizing agricultural ditches. J. Soil Water Conserv. 2007, 62, 277–286. [Google Scholar]
- Davis, R.T.; Tank, J.L.; Mahl, U.H.; Winikoff, S.G.; Roley, S.S. The influence of two-stage ditches with constructed floodplains on water column nutrients and sediments in agricultural streams. J. Am. Water Resour. Assoc. 2015, 51, 941–955. [Google Scholar] [CrossRef]
- Mahl, U.H.; Tank, J.L.; Roley, S.S.; Davis, R.T. Two-stage ditch floodplains enhance n-removal capacity and reduce turbidity and dissolved P in agricultural streams. J. Am. Water Resour. Assoc. 2015, 51, 923–940. [Google Scholar] [CrossRef]
- Cordell, H.K.; Bergstrom, J.C. Comparison of recreation use values among alternative reservoir water level management scenarios. Water Resour. Res. 1993, 29, 247–258. [Google Scholar] [CrossRef]
- Branche, E. The multipurpose water uses of hydropower reservoir: The share concept. C. R. Phys. 2017, 18, 469–478. [Google Scholar] [CrossRef]
- Twisk, W.; Noordervliet, M.A.W.; ter Keurs, W.J. The nature value of the ditch vegetation in peat areas in relation to farm management. Aquat. Ecol. 2003, 37, 191–209. [Google Scholar] [CrossRef]
- Needelman, B.A.; Kleinman, P.J.A.; Strock, J.S.; Allen, A.L. Drainage ditches: Improved management of agricultural drainage ditches for water quality protection: An overview. J. Soil Water Conserv. 2007, 62, 171–178. [Google Scholar]
- Smith, C.; Boughton, E.H.; Pierre, S. Pomacea maculata (island apple snail) invasion in seasonal wetlands on Florida ranchland: Association with plant-community structure and aquatic-predator abundance. Southeast. Nat. 2015, 14, 561–576. [Google Scholar] [CrossRef]
- Fluck, R.C.; Fonyo, C.; Flaig, E. Land-use-based phosphorus balances for lake Okeechobee, Florida, drainage basins. Appl. Eng. Agric 1992, 8, 813–820. [Google Scholar] [CrossRef]
- Final Total Maximum Daily Load for Biochemical Oxygen Demand, Dissolved Oxygen, and Nutrients in the Lake Okeechobee tributaries Osceola, Polk, Okeechobee, Highlands, Glade, and Martin, Florida; U.S. Environmental Protection Association: Atlanta, GA, USA, 2008.
- Compilation of Benefits and Costs of STA and Reservoir Projects in the South Florida Water Management District. Available online: http://www.fresp.org/pdfs/Compilation%20of%20STA%20and%20REZ%20Benefits%20Costs%20HandS%2011_2011.pdf (accessed on 27 September 2014).
- Bohlen, P.J.; Boughton, E.; Fauth, J.E.; Jenkins, D.; Kiker, G.; Quintana-Ascencio, P.F.; Shukla, S.; Swain, H.M. Assessing Trade-Offs Among Ecosystem Services in a Payment-for-Water Services Program on Florida Ranchlands; Archbold Biological Station: Lake Placid, FL, USA, 2014. [Google Scholar]
- Harabis, F.; Dolny, A. Necessity for the conservation of drainage systems as last refugia for threatened damselfly species, Coenagrion ornatum. Insect. Conserv. Divers. 2015, 8, 143–151. [Google Scholar] [CrossRef]
- Baker, S.; Eckerberg, K. Ecological restoration success: A policy analysis understanding. Restor. Ecol. 2016, 24, 284–290. [Google Scholar] [CrossRef]
- Palmer, M.; Allan, J.D.; Meyer, J.; Bernhardt, E.S. River restoration in the twenty-first century: Data and experiential future efforts. Restor. Ecol. 2007, 15, 472–481. [Google Scholar] [CrossRef]
- Jeffries, M.J. Ponds and the importance of their history: An audit of pond numbers, turnover and the relationship between the origins of ponds and their contemporary plant communities in south-east Northumberland, UK. Hydrobiologia 2012, 689, 11–21. [Google Scholar] [CrossRef]
- Felson, A.J.; Pickett, S.T.A. Designed experiments: New approaches to studying urban ecosystems. Front. Ecol. Environ. 2005, 3, 549–556. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Win-Win Ecology: How the Earth’s Species Can Survive in the Midst of Human Enterprise; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Hobbs, R.J.; Higgs, E.; Harris, J.A. Novel ecosystems: Implications for conservation and restoration. Trends Ecol. Evol. 2009, 24, 599–605. [Google Scholar] [CrossRef] [PubMed]
- De Meester, L.; Declerck, S.; Stoks, R.; Louette, G.; Van de Meutter, F.; De Bie, T.; Michels, E.; Brendonck, L. Ponds and pools as model systems in conservation biology, ecology and evolutionary biology. Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 715–725. [Google Scholar] [CrossRef]
- Koetsier, P.; McCauley, L.M.M. An irrigation canal as a lotic mesocosm: Examining the relationship between macroinvertebrate benthos and drift. West. N. Am. Nat. 2015, 75, 259–270. [Google Scholar] [CrossRef]
- Diamond, J.M. Ecology: Laboratory, field and natural experiments. Nature 1983, 304, 586–587. [Google Scholar] [CrossRef]
- Van Gerven, L.P.A.; Kuiper, J.J.; Janse, J.H.; Janssen, A.B.G.; Jeuken, M.; Mooij, W.M.; de Klein, J.J.M. How regime shifts in connected aquatic ecosystems are affected by the typical downstream increase of water flow. Ecosystems 2017, 20, 733–744. [Google Scholar] [CrossRef]
- Petrovich, O. Preschool children’s understanding of the dichotomy between the natural and the artificial. Psychol. Rep. 1999, 84, 3–27. [Google Scholar] [CrossRef]
- Chakrabarty, D. The climate of history: Four theses. Crit. Inq. 2009, 35, 197–222. [Google Scholar] [CrossRef]
- Bensaude-Vincent, B.; Newman, W.R. Introduction: The artificial and the natural: State of the problem. In The Artificial and the Natural; Bensaude-Vincent, B., Newman, W.R., Eds.; The MIT Press: Cambridge, UK, 2007; pp. 1–19. [Google Scholar]
- Purdy, J. After Nature: A Politics for the Anthropocene; Harvard University Press: Cambridge, MA, USA, 2015. [Google Scholar]
- McMahan, E.A.; Cloud, J.M.; Josh, P.; Scott, M. Nature with a human touch: Human-induced alteration negatively impacts perceived naturalness and preferences for natural environments. Ecopsychology 2016, 8, 54–63. [Google Scholar] [CrossRef]
- Rozin, P.; Spranca, M.; Krieger, Z.; Neuhaus, R.; Surillo, D.; Swerdlin, A.; Wood, K. Preference for natural: Instrumental and ideational/moral motivations, and the contrast between foods and medicines. Appetite 2004, 43, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Elliot, R. Faking nature. In Environmental Ethics; Blackwell Publishing: Malden, MA, USA, 1982; pp. 381–389. [Google Scholar]
- Katz, E. The big lie: Human restoration of nature. In Environmental Ethics; Blackwell Publishing: Malden, MA, USA, 1983; pp. 390–397. [Google Scholar]
- Callicott, J.B. A critique of an alternative to the wilderness idea. In Environmental Ethics; Blackwell Publishing: Malden, MA, USA, 1995; pp. 437–443. [Google Scholar]
- Light, A.; Thompson, A.; Higgs, E.S. Valuing novel ecosystems. In Novel ecosystems; John Wiley & Sons: Hoboken, NJ, USA, 2013; pp. 257–268. [Google Scholar]
- Higgs, E. Nature by Design; Massachusetts Institute of Technology: Boston, MA, USA, 2003. [Google Scholar]
- Soule, M. The “New Conservation”. Conserv. Biol. 2013, 27, 895–897. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, R.J.; Arico, S.; Aronson, J.; Baron, J.S.; Bridgewater, P.; Cramer, V.A.; Epstein, P.R.; Ewel, J.J.; Klink, C.A.; Lugo, A.E.; et al. Novel ecosystems: Theoretical and management aspects of the new ecological world order. Glob. Ecol. Biogeogr. 2006, 15, 1–7. [Google Scholar] [CrossRef]
- Tallis, H.; Lubchenco, J. A call for inclusive conservation. Nature 2014, 515, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Matulis, B.S.; Moyer, J.R. Beyond inclusive conservation: The value of pluralism, the need for agonism, and the case for social instrumentalism. Conserv. Lett. 2017, 10, 279–287. [Google Scholar] [CrossRef]
- Gelcich, S.; O’Keeffe, J. Emerging frontiers in perceptions research for aquatic conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 986–994. [Google Scholar] [CrossRef]
- Francis, R.A.; Hoggart, S.P.G. Waste not, want not: The need to utilize existing artificial structures for habitat improvement along urban rivers. Restor. Ecol. 2008, 16, 373–381. [Google Scholar] [CrossRef]
- Introduction to the New EU Water Framework Directive. Available online: http://ec.europa.eu/environment/water/water-framework/info/intro_en.htm (accessed on 19 August 2014).
- Declerck, S.; De Bie, T.; Ercken, D.; Hampel, H.; Schrijvers, S.; Van Wichelen, J.; Gillard, V.; Mandiki, R.; Losson, B.; Bauwens, D.; et al. Ecological characteristic’s of small farmland ponds: Associations with land use practices at multiple spatial scales. Biol. Conserv. 2006, 131, 523–532. [Google Scholar] [CrossRef]
- European Pond Conservation Network. Available online: https://freshwaterhabitats.org.uk/research/epcn/ (accessed on 12 August 2018).
- Million Ponds Project. Available online: http://www.freshwaterhabitats.org.uk/projects/million-ponds/ (accessed on 19 August 2014).
- Canals, R.M.; Ferrer, V.; Iriarte, A.; Carcamo, S.; San Emeterio, L.; Villanueva, E. Emerging conflicts for the environmental use of water in high-valuable rangelands. Can livestock water ponds be managed as artificial wetlands for amphibians? Ecol. Eng. 2011, 37, 1443–1452. [Google Scholar] [CrossRef]
- Clarke, S.J. Conserving freshwater biodiversity: The value, status and management of high quality ditch systems. J. Nat. Conserv. 2015, 24, 93–100. [Google Scholar] [CrossRef]
- Shaw, R.F.; Johnson, P.J.; Macdonald, D.W.; Feber, R.E. Enhancing the biodiversity of ditches in intensively managed UK farmland. PLoS ONE 2015, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Spears, E.G. Baptized in PCBS: Race, Pollution, and Justice in an All-American Town; UNC Press Books: Chapel Hill, NC, USA, 2014. [Google Scholar]
- Checker, M. Polluted Promises: Environmental Racism and the Search for Justice in a Southern Town; NYU Press: New York, NY, USA, 2005. [Google Scholar]
- Setzer, C.; Domino, M.E. Medicaid outpatient utilization for waterborne pathogenic illness following hurricane Floyd. Public Health Rep. 2004, 119, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Goldsmith, P.; Thomas, M.H. Economic impact and public costs of confined animal feeding operations at the parcel level of Craven County, North Carolina. Agric. Hum. Values 2010, 27, 29–42. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Reconciliation ecology and the future of species diversity. Oryx 2003, 37, 194–205. [Google Scholar] [CrossRef]
- Luptacik, P.; Miklisova, D.; Kovac, L. Diversity and community structure of soil Oribatida (Acari) in an arable field with alluvial soils. Eur. J. Soil Biol. 2012, 50, 97–105. [Google Scholar] [CrossRef]
- Verdonschot, R.C.M.; Keizer-vlek, H.E.; Verdonschot, P.F.M. Biodiversity value of agricultural drainage ditches: A comparative analysis of the aquatic invertebrate fauna of ditches and small lakes. Aquat. Conserv Mar. Freshw. Ecosyst. 2011, 21, 715–727. [Google Scholar] [CrossRef]
- Ishiyama, N.; Koizumi, I.; Yuta, T.; Nakamura, F. Differential effects of spatial network structure and scale on population size and genetic diversity of the ninespine stickleback in a remnant wetland system. Freshw. Biol. 2015, 60, 733–744. [Google Scholar] [CrossRef]
- Yagi, K.T.; Litzgus, J.D. The effects of flooding on the spatial ecology of spotted turtles (Clemmys guttata) in a partially mined peatland. Copeia 2012, 179–190. [Google Scholar] [CrossRef]
- Pitt, A.L.; Baldwin, R.F.; Lipscomb, D.J.; Brown, B.L.; Hawley, J.E.; Allard-Keese, C.M.; Leonard, P.B. The missing wetlands: Using local ecological knowledge to find cryptic ecosystems. Biodivers. Conserv. 2012, 21, 51–63. [Google Scholar] [CrossRef]
- Maltchik, L.; Stenert, C.; Batzer, D.P. Can rice field management practices contribute to the conservation of species from natural wetlands? Lessons from Brazil. Basic Appl. Ecol. 2017, 18, 50–56. [Google Scholar] [CrossRef]
- Kloskowski, J.; Nieoczym, M. Management practices to enhance wildlife diversity of man-made fish ponds: The importance of the hydroperiod. Knowl. Manag. Aquat. Ecosyst. 2015, 15. [Google Scholar] [CrossRef]
- James-Pirri, M.J.; Erwin, R.M.; Prosser, D.J.; Taylor, J.D. Responses of salt marsh ecosystems to mosquito control management practices along the Atlantic coast (USA). Restor. Ecol. 2012, 20, 395–404. [Google Scholar] [CrossRef]
- Rochlin, I.; James-Pirri, M.J.; Adamowicz, S.C.; Dempsey, M.E.; Iwanejko, T.; Ninivaggi, D.V. The effects of integrated marsh management (IMM) on salt marsh vegetation, nekton, and birds. Estuar. Coasts 2012, 35, 727–742. [Google Scholar] [CrossRef]
- Horgan, F.G.; Ramal, A.F.; Villegas, J.M.; Almazan, M.L.P.; Bernal, C.C.; Jamoralin, A.; Pasang, J.M.; Orboc, G.; Agreda, V.; Arroyo, C. Ecological engineering with high diversity vegetation patches enhances bird activity and ecosystem services in Philippine rice fields. Reg. Environ. Chang. 2017, 17, 1355–1367. [Google Scholar] [CrossRef]
- Pernollet, C.A.; Simpson, D.; Gauthier-Clerc, M.; Guillemain, M. Rice and duck, a good combination? Identifying the incentives and triggers for joint rice farming and wild duck conservation. Agric. Ecosyst. Environ. 2015, 214, 118–132. [Google Scholar] [CrossRef]
- Pernollet, C.A.; Cavallo, F.; Simpson, D.; Gauthier-Clerc, M.; Guillemain, M. Seed density and waterfowl use of rice fields in Camargue, France. J. Wildl. Manag. 2017, 81, 96–111. [Google Scholar] [CrossRef]
- Hill, M.J.; Ryves, D.B.; White, J.C.; Wood, P.J. Macroinvertebrate diversity in urban and rural ponds: Implications for freshwater biodiversity conservation. Biol. Conserv. 2016, 201, 50–59. [Google Scholar] [CrossRef]
- Soomers, H.; Karssenberg, D.; Soons, M.B.; Verweij, P.A.; Verhoeven, J.T.A.; Wassen, M.J. Wind and water dispersal of wetland plants across fragmented landscapes. Ecosystems 2013, 16, 434–451. [Google Scholar] [CrossRef]
- Van Dijk, W.F.A.; van Ruijven, J.; Berendse, F.; de Snoo, G.R. The effectiveness of ditch banks as dispersal corridor for plants in agricultural landscapes depends on species’ dispersal traits. Biol. Conserv. 2014, 171, 91–98. [Google Scholar] [CrossRef]
- Zhu, L.; Jiang, C.; Xie, X.; Xie, M.; Hu, X.; Cheng, J. Ecological and landscape effects of ditches on farmland ecosystem. In Proceedings of the Bioinformatics and Biomedical Engineering (ICBBE), Beijing, China, 11–13 June 2009. [Google Scholar]
- Zhang, Y.-T.; Zhang, G.-F.; Li, Y.; Li, L.; Yu, L.-P. Diversity of aquatic vascular plants in the Yangtze delta. Plant Sci. J. 2012, 30, 238–249. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, X.R.; Zhang, Z.S.; Hu, Y.G.; Wu, P. Species composition and species richness in the Hetao irrigation region drainage ditches, Northern China. Arid Land Res. Manag. 2013, 27, 167–177. [Google Scholar] [CrossRef]
- Hill, C.R.; Robinson, J.S. Phosphorus flux from wetland ditch sediments. Sci. Total Environ. 2012, 437, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Navrátilová, J.; Hájek, M.; Navrátil, J.; Hájková, P.; Frazier, R.J. Convergence and impoverishment of fen communities in a eutrophicated agricultural landscape of the Czech Republic. Appl. Veg. Sci. 2017, 20, 225–235. [Google Scholar] [CrossRef]
- Henriksson, P.J.G.; Jarvio, N.; Jonell, M.; Guinée, J.B.; Troell, M. The devil is in the details—The carbon footprint of a shrimp. Front. Ecol. Environ. 2018, 16, 10–11. [Google Scholar] [CrossRef]
- Wiebusch, R.K.; Lant, C.L. Policy drivers of US wetland conversion rates, 1955–2009. Soc. Nat. Resour. 2017, 30, 16–30. [Google Scholar] [CrossRef]
- Zedler, J.B.; Kercher, S. Wetland resources: Status, trends, ecosystem services, and restorability. Annu. Rev. Environ. Resour. 2005, 30, 39–74. [Google Scholar] [CrossRef]
- Stone, M.L.; Whiles, M.R.; Webber, J.A.; Williard, K.W.J.; Reeve, J.D. Macroinvertebrate communities in agriculturally impacted Southern Illinois streams: Patterns with riparian vegetation, water quality, and in-stream habitat quality. J. Environ. Qual. 2005, 34, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Katz, H.A.; Daniels, J.M.; Ryan, S. Slope- area thresholds of road- induced gully erosion and consequent hillslope- channel interactions. Int. J. Circuit Theory Appl. 2013, 41, 285–295. [Google Scholar] [CrossRef]
- Stenberg, L.; Tuukkanen, T.; Finer, L.; Marttila, H.; Piirainen, S.; Klove, B.; Koivusalo, H. Ditch erosion processes and sediment transport in a drained peatland forest. Ecol. Eng. 2015, 75, 421–433. [Google Scholar] [CrossRef]
- Bohne, B.; Storchenegger, I.J.; Widmoser, P. An easy to use calculation method for weir operations in controlled drainage systems. Agric. Water Manag. 2012, 109, 46–53. [Google Scholar] [CrossRef]
- Egresi, I. Tourism and sustainability in Turkey: Negative impact of mass tourism development. In Alternative Tourism in Turkey: Role, Potential Development and Sustainability; Egresi, I., Ed.; Springer International Publishing Ag: Cham, Switzerland, 2016; Volume 121, pp. 35–53. [Google Scholar]
- Elphick, C.S.; Taft, O.; Lourenco, P.M. Management of rice fields for birds during the non-growing season. Waterbirds 2010, 33, 181–192. [Google Scholar] [CrossRef]
- Gossling, S.; Peeters, P.; Hall, C.M.; Ceron, J.P.; Dubois, G.; Lehmann, L.; Scott, D. Tourism and water use: Supply, demand, and security. An international review. Tourism Manag. 2012, 33, 1–15. [Google Scholar] [CrossRef]
- Chen, R.S.; Wang, K.L.; Wu, C.Y. A preliminary study on the improvement of the ecological corridor in paddy fields. Irrig. Drain. 2015, 64, 115–123. [Google Scholar] [CrossRef]
- Sato, M.; Kumagai, M.; Azuma, N. Life history of Tanakia lanceolata in irrigation channel network of the iwaki river basin in northern japan. Trans. Jpn. Soc. Irrig. Drain. Rural Eng. 2012, 80, 1–11. [Google Scholar]
- Ishiyama, N.; Sueyoshi, M.; Watanabe, N.; Nakamura, F. Biodiversity and rarity distributions of native freshwater fish in an agricultural landscape: The importance of beta diversity between and within water-body types. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 416–428. [Google Scholar] [CrossRef]
- Flora, C.; Kroger, R. Use of vegetated drainage ditches and low-grade weirs for aquaculture effluent mitigation: I. Nutrients. Aquac. Eng. 2014, 60, 56–62. [Google Scholar] [CrossRef]
- Hyvonen, N.P.; Huttunen, J.T.; Shurpali, N.J.; Lind, S.E.; Marushchak, M.E.; Heitto, L.; Martikainen, P.J. The role of drainage ditches in greenhouse gas emissions and surface leaching losses from a cutaway peatland cultivated with a perennial bioenergy crop. Boreal Environ. Res. 2013, 18, 109–126. [Google Scholar]
- Decleer, K.; Maes, D.; Van Calster, H.; Jansen, I.; Pollet, M.; Dekoninck, W.; Baert, L.; Grootaert, P.; Van Diggelen, R.; Bonte, D. Importance of core and linear marsh elements for wetland arthropod diversity in an agricultural landscape. Insect. Conserv. Divers. 2015, 8, 289–301. [Google Scholar] [CrossRef]
- Onikura, N.; Nakajima, J. Age, growth and habitat use of the topmouth gudgeon, Pseudorasbora parva in irrigation ditches on northwestern Kyushu Island, Japan. J. Appl. Ichthyol. 2013, 29, 186–192. [Google Scholar] [CrossRef]
- Cowley, D.E.; Wissmar, R.C.; Sallenave, R. Fish assemblages and seasonal movements of fish in irrigation canals and river reaches of the Middle Rio Grande, New Mexico (USA). Ecol. Freshw. Fish 2007, 16, 548–558. [Google Scholar] [CrossRef]
- Falbo, K.; Schneider, R.L.; Buckley, D.H.; Walter, M.T.; Bergholz, P.W.; Buchanan, B.P. Roadside ditches as conduits of fecal indicator organisms and sediment: Implications for water quality management. J. Environ. Manag. 2013, 128, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.L.; McAtee, S.; Bryan, P.E.; Jeun, R.; Ward, T.; Kraus, J.; Bottazzi, M.E.; Hotez, P.J.; Flowers, C.C.; Mejia, R. Human intestinal parasite burden and poor sanitation in rural Alabama. Am. J. Trop. Med. Hyg. 2017, 97, 1623–1628. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Wan, X.X.; Liu, Q.; Cao, C.L.; Bao, Z.P.; Zhu, H.Q.; Zhong, B.; Guo, J.G. Cost-effectiveness evaluation and investigation of control measure changes in areas of schistosomiasis transmission control in hilly regions of mountain areas I: Epidemiological investigation and analysis of prevalence factors of schistosomiasis. Chin. J. Schistosomiasis Control 2012, 24, 250–254. [Google Scholar]
- Fan, X.Y.; Cui, B.S.; Zhao, H.; Zhang, Z.M. The changes of wetland network pattern associated with water quality in the Pearl River Delta, China. Clean-Soil Air Water 2012, 40, 1064–1075. [Google Scholar] [CrossRef]
- Fu, D.F.; Gong, W.J.; Xu, Y.; Singh, R.P.; Surampalli, R.Y.; Zhang, T.C. Nutrient mitigation capacity of agricultural drainage ditches in tai lake basin. Ecol. Eng. 2014, 71, 101–107. [Google Scholar] [CrossRef]
- Powell, K.L.; Bouchard, V. Is denitrification enhanced by the development of natural fluvial morphology in agricultural headwater ditches? J. N. Am. Benthol. Soc. 2010, 29, 761–772. [Google Scholar] [CrossRef]
- Usborne, E.L.; Kroger, R.; Pierce, S.C.; Brandt, J.; Goetz, D. Preliminary evidence of sediment and phosphorus dynamics behind newly installed low-grade weirs in agricultural drainage ditches. Water Air Soil Pollut. 2013, 224, 11. [Google Scholar] [CrossRef]
- Kahle, P.; Schonemann, S.; Lennartz, B. Effect of vegetated buffer strips on nitrate inputs into surface waters in drained lowland catchments. Hydrol. Wasserbewirtsch. 2013, 57, 60–68. [Google Scholar]
- Kleinman, P.J.A.; Smith, D.R.; Bolster, C.H.; Easton, Z.M. Phosphorus fate, management, and modeling in artificially drained systems. J. Environ. Qual. 2015, 44, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Tyler, H.L.; Moore, M.T.; Locke, M.A. Potential for phosphate mitigation from agricultural runoff by three aquatic macrophytes. Water Air Soil Pollut. 2012, 223, 4557–4564. [Google Scholar] [CrossRef]
- Tyler, H.L.; Moore, M.T.; Locke, M.A. Influence of three aquatic macrophytes on mitigation of nitrogen species from agricultural runoff. Water Air Soil Pollut. 2012, 223, 3227–3236. [Google Scholar] [CrossRef]
- Li, Q.K.; Hu, Y.W.; Luo, L.G. Source characteristics analysis of discharge and pollutants in typical drainage ditch of Qingtongxia irrigation district. Huanjing Kexue 2012, 33, 1579–1586. [Google Scholar]
- Zhang, Z.Y.; Kong, L.L.; Zhu, L.; Xia, J.H.; Patricia, X. Fate characteristics of nitrogen in runoff from a small agricultural watershed on the south of Huaihe river in China. Environ. Earth Sci. 2012, 66, 835–848. [Google Scholar] [CrossRef]
- Buchanan, B.P.; Falbo, K.; Schneider, R.L.; Easton, Z.M.; Walter, M.T. Hydrological impact of roadside ditches in an agricultural watershed in central New York: Implications for non-point source pollutant transport. Hydrol. Process. 2013, 27, 2422–2437. [Google Scholar] [CrossRef]
- Peacock, M.; Jones, T.G.; Airey, B.; Johncock, A.; Evans, C.D.; Lebron, I.; Fenner, N.; Freeman, C. The effect of peatland drainage and rewetting (ditch blocking) on extracellular enzyme activities and water chemistry. Soil Use Manage. 2015, 31, 67–76. [Google Scholar] [CrossRef]
- Shan, B.Q.; Li, N.; Tang, W.Z. Construct of Yangtze-Huai River rural areas ecological drainage system and its retention effect on pollutants. Chin. J. Environ. Sci. 2012, 33, 3797–3803. [Google Scholar]
- Moeder, M.; Carranza-Diaz, O.; Lopez-Angulo, G.; Vega-Avina, R.; Chavez-Duran, F.A.; Jomaa, S.; Winkler, U.; Schrader, S.; Reemtsma, T.; Delgado-Vargas, F. Potential of vegetated ditches to manage organic pollutants derived from agricultural runoff and domestic sewage: A case study in Sinaloa (Mexico). Sci. Total Environ. 2017, 598, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Adriaanse, P.I.; Boesten, J.; Crum, S.J.H. Estimating degradation rates in outdoor stagnant water by inverse modelling with TOXSWA: A case study with prosulfocarb. Pest Manag. Sci. 2013, 69, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Gall, H.E.; Sassman, S.A.; Jenkinson, B.; Lee, L.S.; Jafvert, C.T. Comparison of export dynamics of nutrients and animal-borne estrogens from a tile-drained midwestern agroecosystem. Water Res. 2015, 72, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Soulis, K.X.; Dercas, N.; Papadaki, C.H. Effects of forest roads on the hydrological response of a small-scale mountain watershed in Greece. Hydrol. Process. 2015, 29, 1772–1782. [Google Scholar] [CrossRef]
- Fernández Tejedo, I. Fragilidad de un espacio productivo: Cambio climático e inundaciones en el bajío, siglo xviii. Tzintzun 2012, 55, 107–156. [Google Scholar]
- Henderson, M. Venice area Audubon rookery. Outdoor Photographer, 10 May 2017. [Google Scholar]
- Milbank, D. Leapin ljeppers get on the stick or wind up all wet—For Frisians in The Netherlands, vaulting murky canals is a hot spectator sport. Wall Street Journal, 6 July 1994; A1. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clifford, C.C.; Heffernan, J.B. Artificial Aquatic Ecosystems. Water 2018, 10, 1096. https://doi.org/10.3390/w10081096
Clifford CC, Heffernan JB. Artificial Aquatic Ecosystems. Water. 2018; 10(8):1096. https://doi.org/10.3390/w10081096
Chicago/Turabian StyleClifford, Chelsea C., and James B. Heffernan. 2018. "Artificial Aquatic Ecosystems" Water 10, no. 8: 1096. https://doi.org/10.3390/w10081096
APA StyleClifford, C. C., & Heffernan, J. B. (2018). Artificial Aquatic Ecosystems. Water, 10(8), 1096. https://doi.org/10.3390/w10081096





