Compositional and Mineralogical Effects on Ice Nucleation Activity of Volcanic Ash
Abstract
1. Introduction
2. Experiments
3. Results
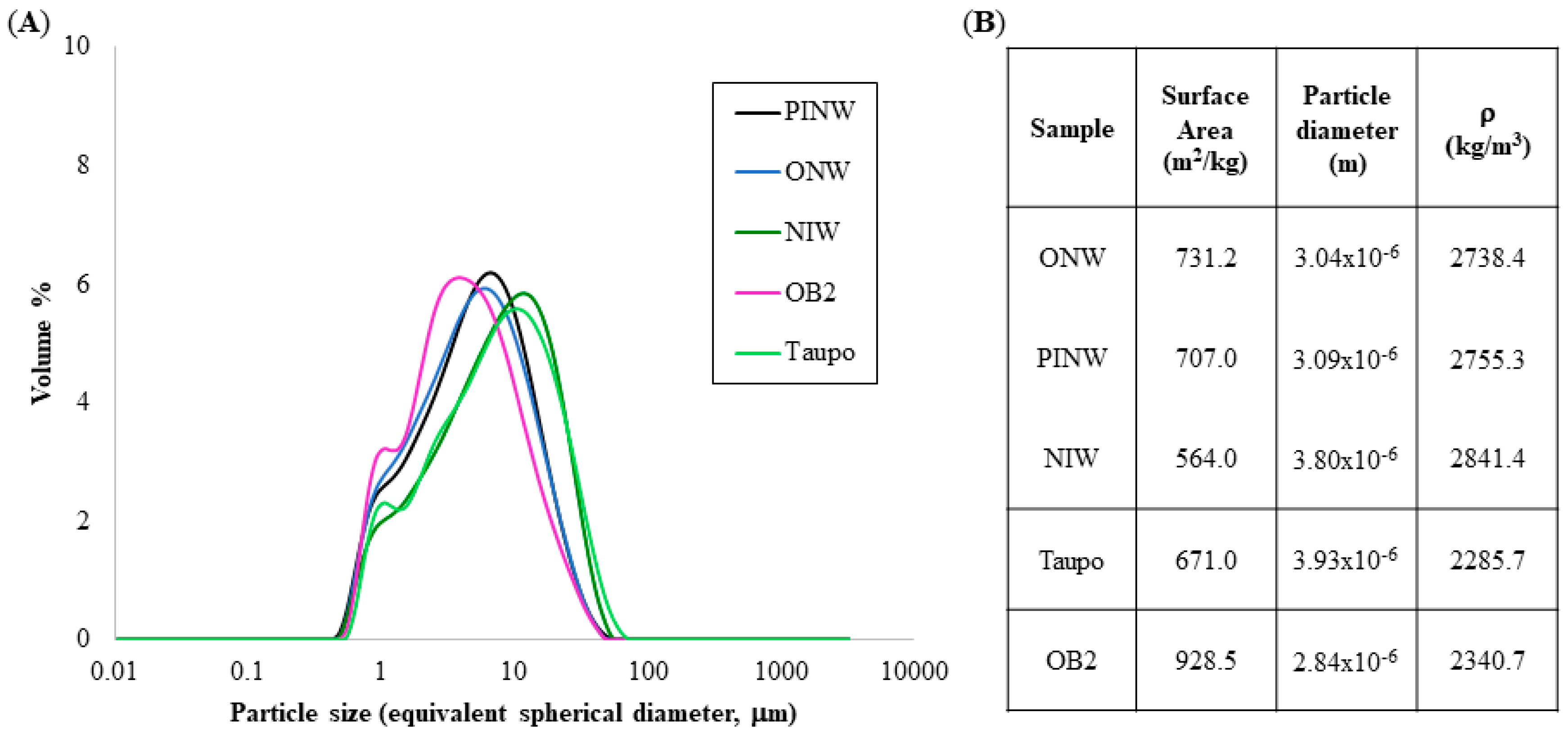
3.1. Sample Characteristics
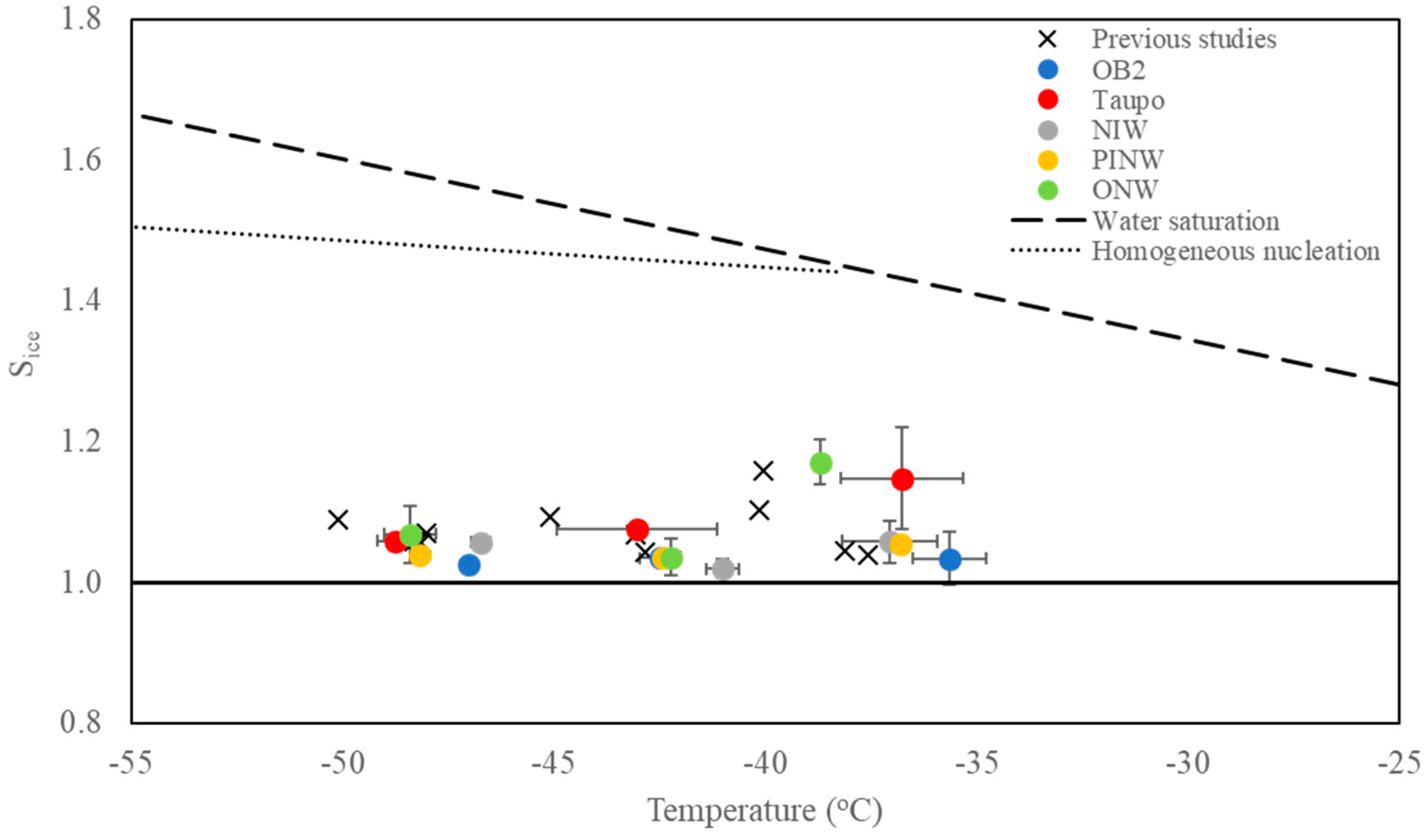
3.2. Deposition-Mode Ice Nucleation Activity
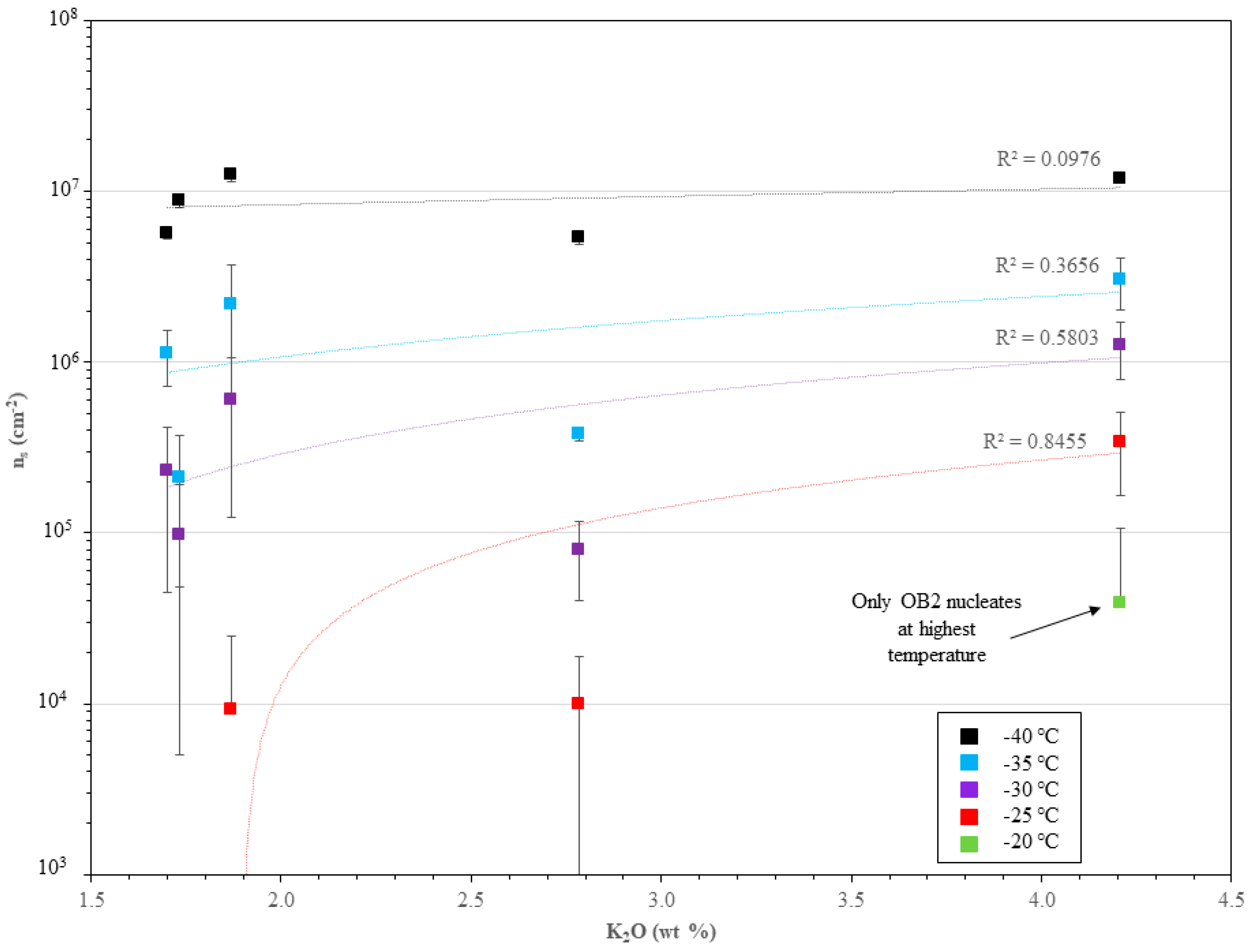
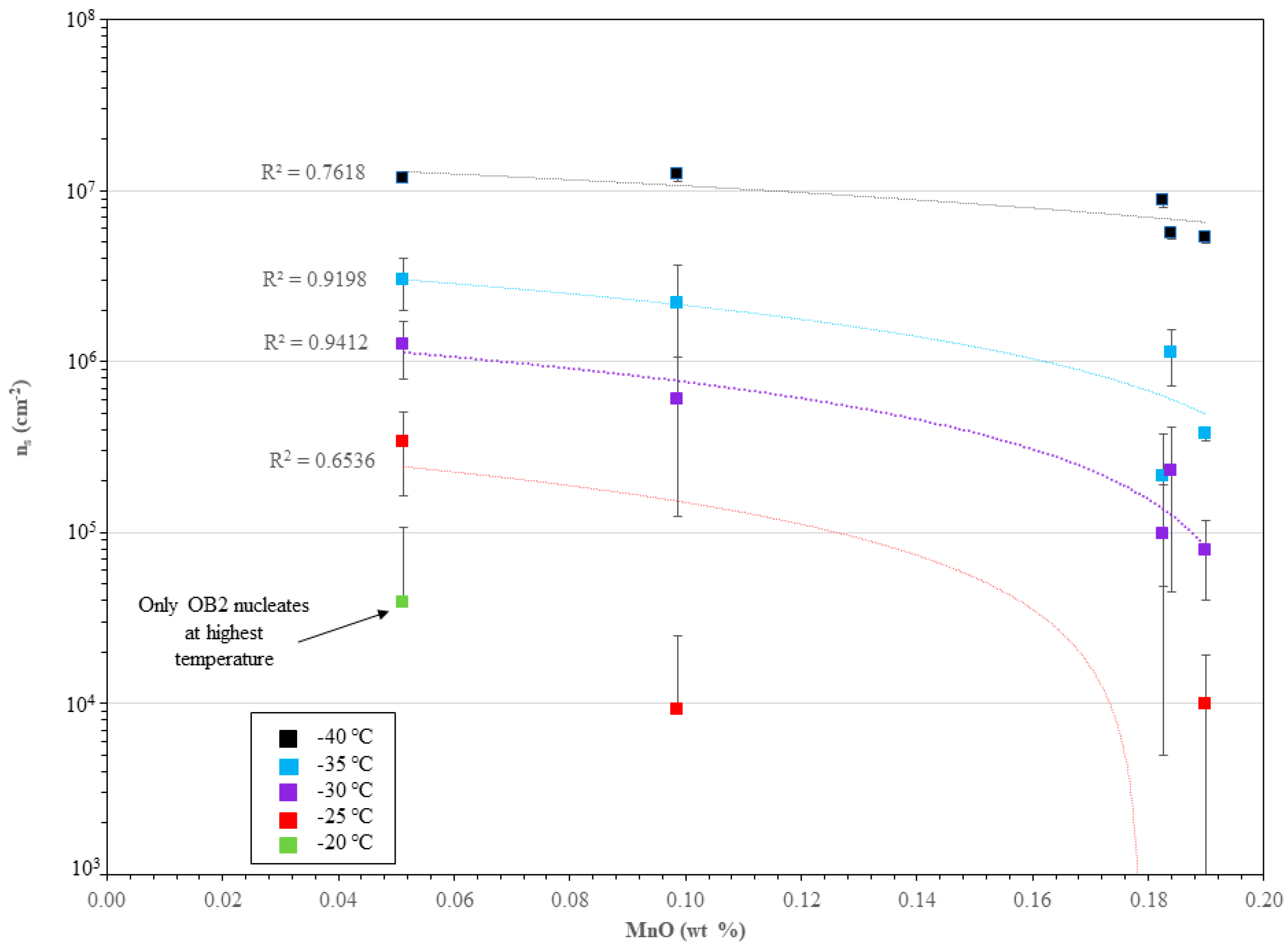
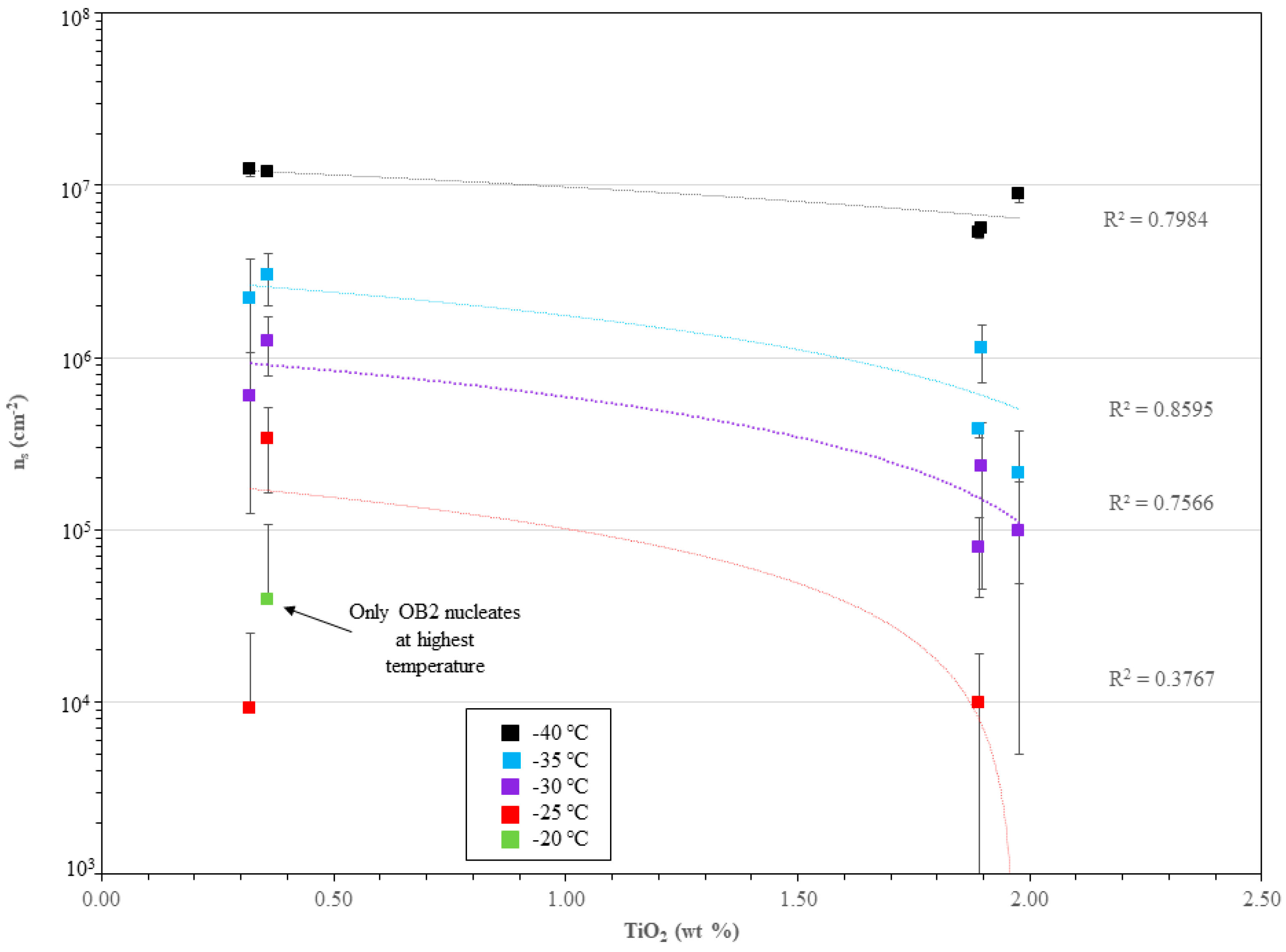
3.3. Immersion-Mode Ice Nucleation Activity
4. Discussion
4.1. Deposition-Mode Ice Nucleation Activity
4.2. Immersion-Mode Ice Nucleation Activity
4.3. Compositional Effects
4.4. Lightning Generation in Volcanic Plumes
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Thomas, R.J.; Krehbiel, P.R.; Rison, W.; Edens, H.E.; Aulich, G.D.; Winn, W.P.; McNutt, S.R.; Tytgat, G.; Clark, E. Electrical activity during the 2006 Mount St. Augustine volcanic eruptions. Science 2007, 315, 1097. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.J.; McNutt, S.R.; Krehbiel, P.R.; Rison, W.; Aulich, G.; Edens, H.E.; Tytgat, G.; Clark, E. Lightning and Electrical Activity during the 2006 Eruption of Augustine Volcano. Available online: https://pubs.usgs.gov/pp/1769/chapters/p1769_chapter25.pdf (accessed on 17 June 2018).
- Gilbert, J.S.; Lane, S.J.; Sparks, R.S.J.; Koyaguchi, T. Charge measurements on particle fallout from a volcanic plume. Nature 1991, 349, 598–600. [Google Scholar] [CrossRef]
- James, M.R.; Wilson, L.; Lane, S.J.; Gilbert, J.S.; Mather, T.A.; Harrison, R.G.; Martin, R.S. Electrical charging of volcanic plumes. Space Sci. Rev. 2008, 137, 399–418. [Google Scholar] [CrossRef]
- McNutt, S.R.; Williams, E.R. Volcanic lightning: Global observations and constraints on source mechanisms. Bull. Volcanol. 2010, 72, 1153–1167. [Google Scholar] [CrossRef]
- Arason, P.; Petersen, G.N.; Bjornsson, H. Observations of the altitude of the volcanic plume during the eruption of Eyjafjallajokull, April–May 2010. Earth Syst. Sci. Data 2011, 3, 9–17. [Google Scholar] [CrossRef]
- Behnke, S.A.; Thomas, R.J.; McNutt, S.R.; Schneider, D.J.; Krehbiel, P.R.; Rison, W.; Edens, H.E. Observations of volcanic lightning during the 2009 eruption of Redoubt Volcano. J. Volcanol. Geotherm. Res. 2013, 259, 214–234. [Google Scholar] [CrossRef]
- Durant, A.J.; Shaw, R.A.; Rose, W.I.; Mi, Y.; Ernst, G.G.J. Ice nucleation and overseeding of ice in volcanic clouds. J. Geophys. Res. Atmos. 2008, 113. [Google Scholar] [CrossRef]
- Van Eaton, A.R.; Muirhead, J.D.; Wilson, C.J.; Cimarelli, C. Growth of volcanic ash aggregates in the presence of liquid water and ice: An experimental approach. Bull. Volcanol. 2012, 74, 1963–1984. [Google Scholar] [CrossRef]
- Behnke, S.A.; Thomas, R.J.; Edens, H.E.; Krehbiel, P.R.; Rison, W. The 2010 eruption of Eyjafjallajökull: Lightning and plume charge structure. J. Geophys. Res. Atmos. 2014, 119, 833–859. [Google Scholar] [CrossRef]
- Cimarelli, C.; Alatorre-Ibargüengoitia, M.A.; Aizawa, K.; Yokoo, A.; Díaz-Marina, A.; Iguchi, M.; Dingwell, D.B. Multiparametric observation of volcanic lightning: Sakurajima Volcano, Japan. Geophys. Res. Lett. 2016, 43, 4221–4228. [Google Scholar] [CrossRef]
- Van Eaton, A.R.; Amigo, Á.; Bertin, D.; Mastin, L.G.; Giacosa, R.E.; González, J.; Valderrama, O.; Fontijn, K.; Behnke, S.A. Volcanic lightning and plume behavior reveal evolving hazards during the April 2015 eruption of Calbuco volcano, Chile. Geophys. Res. Lett. 2016, 43, 3563–3571. [Google Scholar] [CrossRef]
- Firstov, P.P.; Akbashev, R.R.; Holzworth, R.; Cherneva, N.V.; Shevtsov, B.M. Atmospheric electric effects during the explosion of Shiveluch volcano on November 16, 2014. Izv. Atmos. Ocean. Phys. 2017, 53, 24–31. [Google Scholar] [CrossRef]
- Isono, K.; Komabayasi, M.; Ono, A. Volcanoes as a source of atmospheric ice nuclei. Nature 1959, 183, 317–318. [Google Scholar] [CrossRef]
- Hobbs, P.V.; Fullerton, C.M.; Bluhm, G.C. Ice nucleus storms in Hawaii. Nat. Phys. Sci. 1971, 230, 90–91. [Google Scholar] [CrossRef]
- Fornea, A.P.; Brooks, S.D.; Dooley, J.B.; Saha, A. Heterogeneous freezing of ice on atmospheric aerosols containing ash, soot, and soil. J. Geophys. Res. Atmos. 2009, 114. [Google Scholar] [CrossRef]
- Seifert, P.; Ansmann, A.; Gross, S.; Freudenthaler, V.; Heinold, B.; Hiebsch, A.; Mattis, I.; Schmidt, J.; Schnell, F.; Tesche, M.; et al. Ice formation in ash-influenced clouds after the eruption of the Eyjafjallajökull volcano in April 2010. J. Geophys. Res. Atmos. 2011, 116. [Google Scholar] [CrossRef]
- Vali, G. Nucleation terminology. Bull. Am. Meteorol. Soc. 1985, 66, 1426–1427. [Google Scholar]
- Pruppacher, H.R.; Klett, J.D. Microphysics of Clouds and Precipitation, 2nd ed.; Kluwer Academic Publishing: Dordrecht, The Netherlands, 1997. [Google Scholar]
- Vali, G.; DeMott, P.J.; Möhler, O.; Whale, T.F. A proposal for ice nucleation terminology. Atmos. Chem. Phys. 2015, 15, 10263–10270. [Google Scholar] [CrossRef]
- Mason, B.J.; Maybank, J. Ice-nucleating properties of some natural mineral dusts. Q. J. R. Meteorol. Soc. 1958, 84, 235–241. [Google Scholar] [CrossRef]
- Roberts, P.; Hallett, J. A laboratory study of the ice nucleating properties of some mineral particulates. Q. J. R. Meteorol. Soc. 1968, 94, 25–34. [Google Scholar] [CrossRef]
- Knopf, D.A.; Koop, T. Heterogeneous nucleation of ice on surrogates of mineral dust. J. Geophys. Res. Atmos. 2006, 111. [Google Scholar] [CrossRef]
- Möhler, O.; Field, P.R.; Connolly, P.; Benz, S.; Saathoff, H.; Schnaiter, M.; Wagner, R.; Cotton, R.; Krämer, M.; Mangold, A.; et al. Efficiency of the deposition mode ice nucleation on mineral dust particles. Atmos. Chem. Phys. 2006, 6, 3007–3021. [Google Scholar] [CrossRef]
- Marcolli, C.; Gedamke, S.; Peter, T.; Zobrist, B. Efficiency of immersion mode ice nucleation on surrogates of mineral dust. Atmos. Chem. Phys. 2007, 7, 5081–5091. [Google Scholar] [CrossRef]
- Broadley, S.L.; Murray, B.J.; Herbert, R.J.; Atkinson, J.D.; Dobbie, S.; Malkin, T.L.; Condliffe, E.; Neve, L. Immersion mode heterogeneous ice nucleation by an illite rich powder representative of atmospheric mineral dust. Atmos. Chem. Phys. 2012, 12, 287–307. [Google Scholar] [CrossRef]
- Zimmermann, F.; Weinbruch, S.; Schütz, L.; Hofmann, H.; Ebert, M.; Kandler, K.; Worringen, A. Ice nucleation properties of the most abundant mineral dust phases. J. Geophys. Res. Atmos. 2008, 113. [Google Scholar] [CrossRef]
- Claquin, T.; Schulz, M.; Balkanski, Y.J. Modeling the mineralogy of atmospheric dust sources. J. Geophys. Res. Atmos. 1999, 104, 22243–22256. [Google Scholar] [CrossRef]
- Atkinson, J.D.; Murray, B.J.; Woodhouse, M.T.; Whale, T.F.; Baustian, K.J.; Carslaw, K.S.; Dobbie, S.; O’Sullivan, D.; Malkin, T.L. The importance of feldspar for ice nucleation by mineral dust in mixed-phase clouds. Nature 2013, 498, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Yakobi-Hancock, J.D.; Ladino, L.A.; Abbatt, J.P.D. Feldspar minerals as efficient deposition ice nuclei. Atmos. Chem. Phys. 2013, 13, 11175–11185. [Google Scholar] [CrossRef]
- Zolles, T.; Burkart, J.; Häusler, T.; Pummer, B.; Hitzenberger, R.; Grothe, H. Identification of ice nucleation active sites on feldspar dust particles. J. Phys. Chem. A 2015, 119, 2692–2700. [Google Scholar] [CrossRef] [PubMed]
- Peckhaus, A.; Kiselev, A.; Hiron, T.; Ebert, M.; Leisner, T. A comparative study of K-rich and Na/Ca-rich feldspar ice-nucleating particles in a nanoliter droplet freezing assay. Atmos. Chem. Phys. 2016, 16, 11477–11496. [Google Scholar] [CrossRef]
- Kiselev, A.; Bachmann, F.; Pedevilla, P.; Cox, S.J.; Michaelides, A.; Gerthsen, D.; Leisner, T. Active sites in heterogeneous ice nucleation—The example of K-rich feldspars. Science 2017, 355, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, G.; Nandasiri, M.; Zelenyuk, A.; Beranek, J.; Madaan, N.; Devaraj, A.; Shutthanandan, V.; Thevuthasan, S.; Varga, T. Effects of crystallographic properties on the ice nucleation properties of volcanic ash particles. Geophys. Res. Lett. 2015, 42, 3048–3055. [Google Scholar] [CrossRef]
- Hoyle, C.R.; Pinti, V.; Welti, A.; Zobrist, B.; Marcolli, C.; Luo, B.; Höskuldsson, Á.; Mattsson, H.B.; Stetzer, O.; Thorsteinsson, T.; et al. Ice nucleation properties of volcanic ash from Eyjafjallajökull. Atmos. Chem. Phys. 2011, 11, 9911–9926. [Google Scholar] [CrossRef]
- Gibbs, A.; Charman, M.; Schwarzacher, W.; Rust, A.C. Immersion freezing of supercooled water drops containing glassy volcanic ash particles. GeoResJ 2015, 7, 66–69. [Google Scholar] [CrossRef]
- Schill, G.P.; Genareau, K.; Tolbert, M.A. Deposition and immersion-mode nucleation of ice by three distinct samples of volcanic ash. Atmos. Chem. Phys. 2015, 15, 7523–7536. [Google Scholar] [CrossRef]
- Mangan, T.P.; Atkinson, J.D.; Neuberg, J.W.; O’Sullivan, D.; Wilson, T.W.; Whale, T.F.; Neve, L.; Umo, N.S.; Malkin, T.L.; Murray, B.J. Heterogeneous ice nucleation by soufriere hills volcanic ash immersed in water droplets. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, S.; Goff, F.; Ikeda, S.; Ikeya, M. ESR dating of quartz phenocrysts in the El Cajete and Battleship Rock Members of Valles Rhyolite, Valles Caldera, New Mexico. J. Volcanol. Geotherm. Res. 1995, 67, 29–40. [Google Scholar] [CrossRef]
- Reneau, S.L.; Gardner, J.N.; Forman, S.L. New evidence for the age of the youngest eruptions in the Valles caldera, New Mexico. Geology 1996, 24, 7–10. [Google Scholar] [CrossRef]
- Walker, G.P. The Taupo pumice: Product of the most powerful known (ultraplinian) eruption? J. Volcanol. Geotherm. Res. 1980, 8, 69–94. [Google Scholar] [CrossRef]
- Wilson, C.J.N. The Taupo eruption, New Zealand I. General aspects. Philos. Trans. R. Soc. Lond. A 1985, 314, 199–228. [Google Scholar] [CrossRef]
- Wilson, C.J.N. Stratigraphy, chronology, styles and dynamics of late quaternary eruptions from Taupo volcano, New Zealand. Philos. Trans. R. Soc. Lond. A 1993, 343, 205–306. [Google Scholar] [CrossRef]
- Houghton, B.F.; Carey, R.J.; Cashman, K.V.; Wilson, C.J.; Hobden, B.J.; Hammer, J.E. Diverse patterns of ascent, degassing, and eruption of rhyolite magma during the 1.8 ka Taupo eruption, New Zealand: Evidence from clast vesicularity. J. Volcanol. Geotherm. Res. 2010, 195, 31–47. [Google Scholar] [CrossRef]
- Dunbar, N.W.; Kyle, P.R. Lack of volatile gradient in the Taupo plinian-ignimbrite transition: Evidence from melt inclusion analysis. Am. Mineral. 1993, 78, 612–618. [Google Scholar]
- Sutton, A.N.; Blake, S.; Wilson, C.J. An outline geochemistry of rhyolite eruptives from Taupo volcanic centre, New Zealand. J. Volcanol. Geotherm. Res. 1995, 68, 153–175. [Google Scholar] [CrossRef]
- Fleck, R.J.; Turrin, B.D.; Sawyer, D.A.; Warren, R.G.; Champion, D.E.; Hudson, M.R.; Minor, S.A. Age and character of basaltic rocks of the Yucca Mountain region, southern Nevada. J. Geophys. Res. Solid Earth 1996, 101, 8205–8227. [Google Scholar] [CrossRef]
- Heizler, M.T.; Perry, F.V.; Crowe, B.M.; Peters, L.; Appelt, R. The age of Lathrop Wells volcanic center: An 40Ar/39Ar dating investigation. J. Geophys. Res. Solid Earth 1999, 104, 767–804. [Google Scholar] [CrossRef]
- Valentine, G.A.; Krier, D.; Perry, F.V.; Heiken, G. Scoria cone construction mechanisms, Lathrop Wells Volcano, Southern Nevada, USA. Geology 2005, 33, 629–632. [Google Scholar] [CrossRef]
- Valentine, G.A.; Perry, F.V. Decreasing magmatic footprints of individual volcanoes in a waning basaltic field. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Genareau, K.; Valentine, G.A.; Moore, G.; Hervig, R.L. Mechanisms for transition in eruptive style at a monogenetic scoria cone revealed by microtextural analyses (Lathrop Wells Volcano, Nevada, USA). Bull. Volcanol. 2010, 72, 593–607. [Google Scholar] [CrossRef]
- Broto, F.; Clausse, D. A study of the freezing of supercooled water dispersed within emulsions by differential scanning calorimetry. J. Phys. C 1976, 9. [Google Scholar] [CrossRef]
- Koop, T.; Luo, B.; Tsias, A.; Peter, T. Water activity as the determinant for homogeneous ice nucleation in aqueous solutions. Nature 2000, 406, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Steinke, I.; Möhler, O.; Kiselev, A.; Niemand, M.; Saathoff, H.; Schnaiter, M.; Skrotzki, J.; Hoose, C.; Leisner, T. Ice nucleation properties of fine ash particles from the Eyjafjallajökull eruption in April 2010. Atmos. Chem. Phys. 2011, 11, 12945–12958. [Google Scholar] [CrossRef]
- Murray, B.J.; Broadley, S.L.; Wilson, T.W.; Atkinson, J.D.; Wills, R.H. Heterogeneous freezing of water droplets containing kaolinite particles. Atmos. Chem. Phys. 2011, 11, 4191–4207. [Google Scholar] [CrossRef]
- Pinti, V.; Marcolli, C.; Zobrist, B.; Hoyle, C.R.; Peter, T. Ice nucleation efficiency of clay minerals in the immersion mode. Atmos. Chem. Phys. 2012, 12, 5859–5878. [Google Scholar] [CrossRef]
- Harrison, A.D.; Whale, T.F.; Carpenter, M.A.; Holden, M.A.; Neve, L.; O’Sullivan, D.; Vergara Temprado, J.; Murray, B.J. Not all feldspars are equal: A survey of ice nucleating properties across the feldspar group of minerals. Atmos. Chem. Phys. 2016, 16, 10927–10940. [Google Scholar] [CrossRef]
- Amis, E.; Hinton, J.F. Solvent Effects on Chemical Phenomena; Academic Press: New York, NY, USA, 1973. [Google Scholar]
- Takahashi, T. Riming electrification as a charge generation mechanism. J. Atmos. Sci. 1978, 35, 1536–1548. [Google Scholar] [CrossRef]
- Jayaratne, E.; Saunders, C.P.R.; Hallett, J. Laboratory studies of the charging of soft hail during ice crystal interactions. Q. J. R. Meteorol. Soc. 1983, 109, 609–630. [Google Scholar] [CrossRef]
- Saunders, C.P.R.; Bax-Norman, H.; Emersic, C.; Avila, E.E.; Castellano, N.E. Laboratory studies of the effect of cloud conditions on graupel/crystal charge transfer in thunderstorm electrification. Q. J. R. Meteorol. Soc. 2006, 132, 2653–2673. [Google Scholar] [CrossRef]



| Major Elements | OB2 | Taupo | NIW | ONW | PINW |
|---|---|---|---|---|---|
| SiO2 | 73.05 | 73.48 | 48.43 | 49.12 | 49.29 |
| TiO2 | 0.36 | 0.32 | 1.89 | 1.90 | 1.98 |
| Al2O3 | 13.74 | 13.81 | 16.22 | 17.14 | 17.09 |
| Fe2O3 | 2.23 | 2.58 | 11.92 | 11.36 | 11.51 |
| MnO | 0.05 | 0.10 | 0.19 | 0.18 | 0.18 |
| MgO | 0.70 | 0.37 | 7.04 | 5.73 | 5.68 |
| CaO | 1.64 | 1.77 | 8.07 | 8.46 | 7.96 |
| Na2O | 3.94 | 4.72 | 3.38 | 3.22 | 3.40 |
| K2O | 4.21 | 2.78 | 1.70 | 1.73 | 1.87 |
| P2O5 | 0.08 | 0.06 | 1.16 | 1.16 | 1.05 |
| Total | 100 | 100 | 100 | 100 | 100 |
| Sample | Fds (%) | Glass + Fds (%) | Pyx + Olv (%) | Fe-oxides (%) | Total Minerals (%) |
|---|---|---|---|---|---|
| Taupo | 99.43 ± 1.74 | 0.57 ± 1.74 | 0.57 ± 1.74 | ||
| OB2 | 99.59 ± 0.45 | 0.41 ± 0.45 | 0.41 ± 0.45 | ||
| NIW | 76.64 ± 4.20 | 16.67 ± 4.89 | 5.82 ± 2.72 | 0.86 ± 0.73 | 83.33 ± 4.89 |
| ONW | 67.93 ± 4.61 | 19.20 ± 5.07 | 9.02 ± 4.39 | 3.85 ± 2.70 | 80.80 ± 5.07 |
| PINW | 70.50 ± 6.99 | 22.06 ± 7.06 | 6.17 ± 2.71 | 1.28 ± 0.71 | 77.94 ± 7.06 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Genareau, K.; Cloer, S.M.; Primm, K.; Tolbert, M.A.; Woods, T.W. Compositional and Mineralogical Effects on Ice Nucleation Activity of Volcanic Ash. Atmosphere 2018, 9, 238. https://doi.org/10.3390/atmos9070238
Genareau K, Cloer SM, Primm K, Tolbert MA, Woods TW. Compositional and Mineralogical Effects on Ice Nucleation Activity of Volcanic Ash. Atmosphere. 2018; 9(7):238. https://doi.org/10.3390/atmos9070238
Chicago/Turabian StyleGenareau, Kimberly, Shelby M. Cloer, Katherine Primm, Margaret A. Tolbert, and Taylor W. Woods. 2018. "Compositional and Mineralogical Effects on Ice Nucleation Activity of Volcanic Ash" Atmosphere 9, no. 7: 238. https://doi.org/10.3390/atmos9070238
APA StyleGenareau, K., Cloer, S. M., Primm, K., Tolbert, M. A., & Woods, T. W. (2018). Compositional and Mineralogical Effects on Ice Nucleation Activity of Volcanic Ash. Atmosphere, 9(7), 238. https://doi.org/10.3390/atmos9070238





