A Systematic Review of Global Desert Dust and Associated Human Health Effects
Abstract
:1. Introduction
2. Data Sources and Methods
3. Preliminary Statements
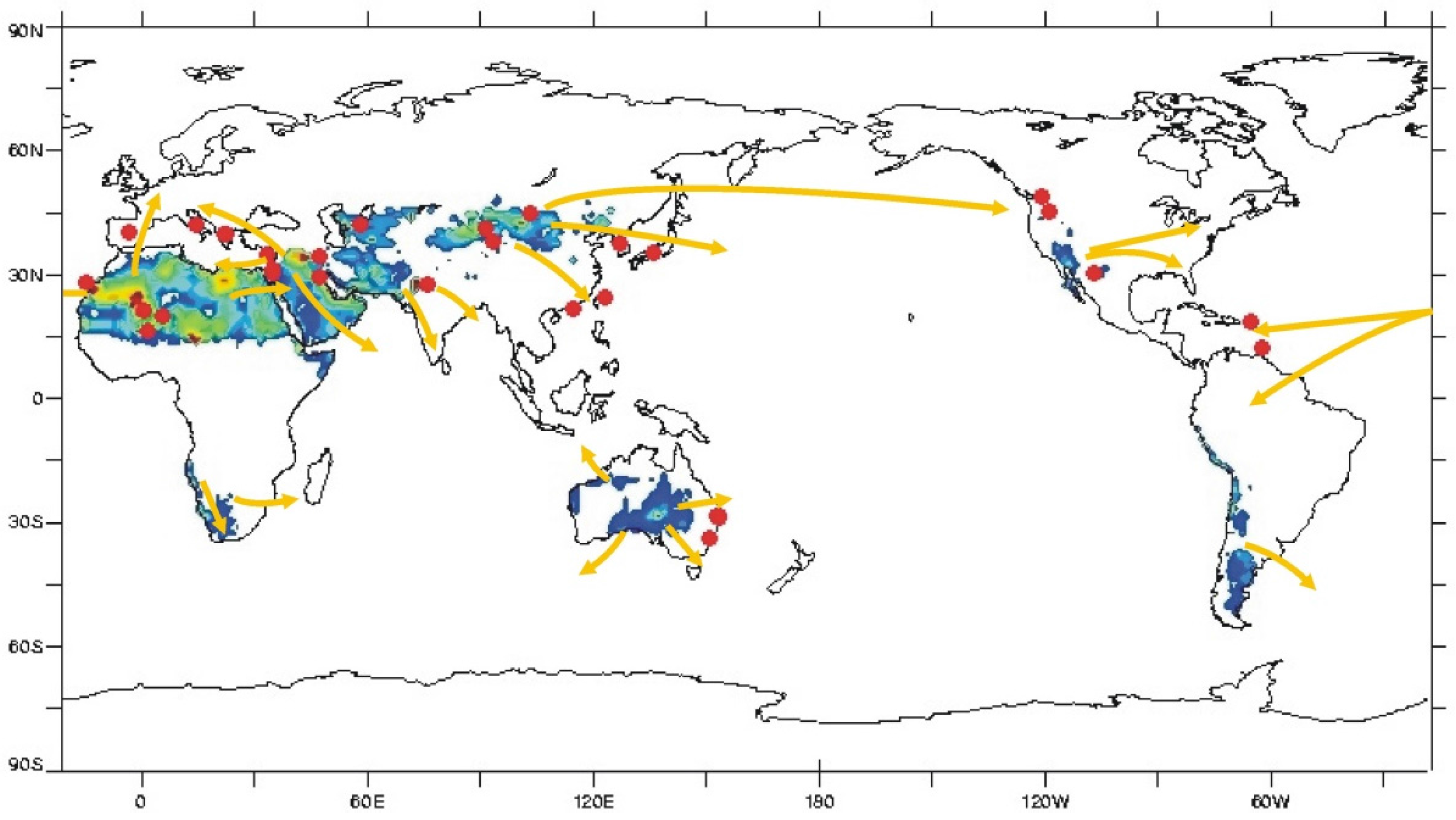
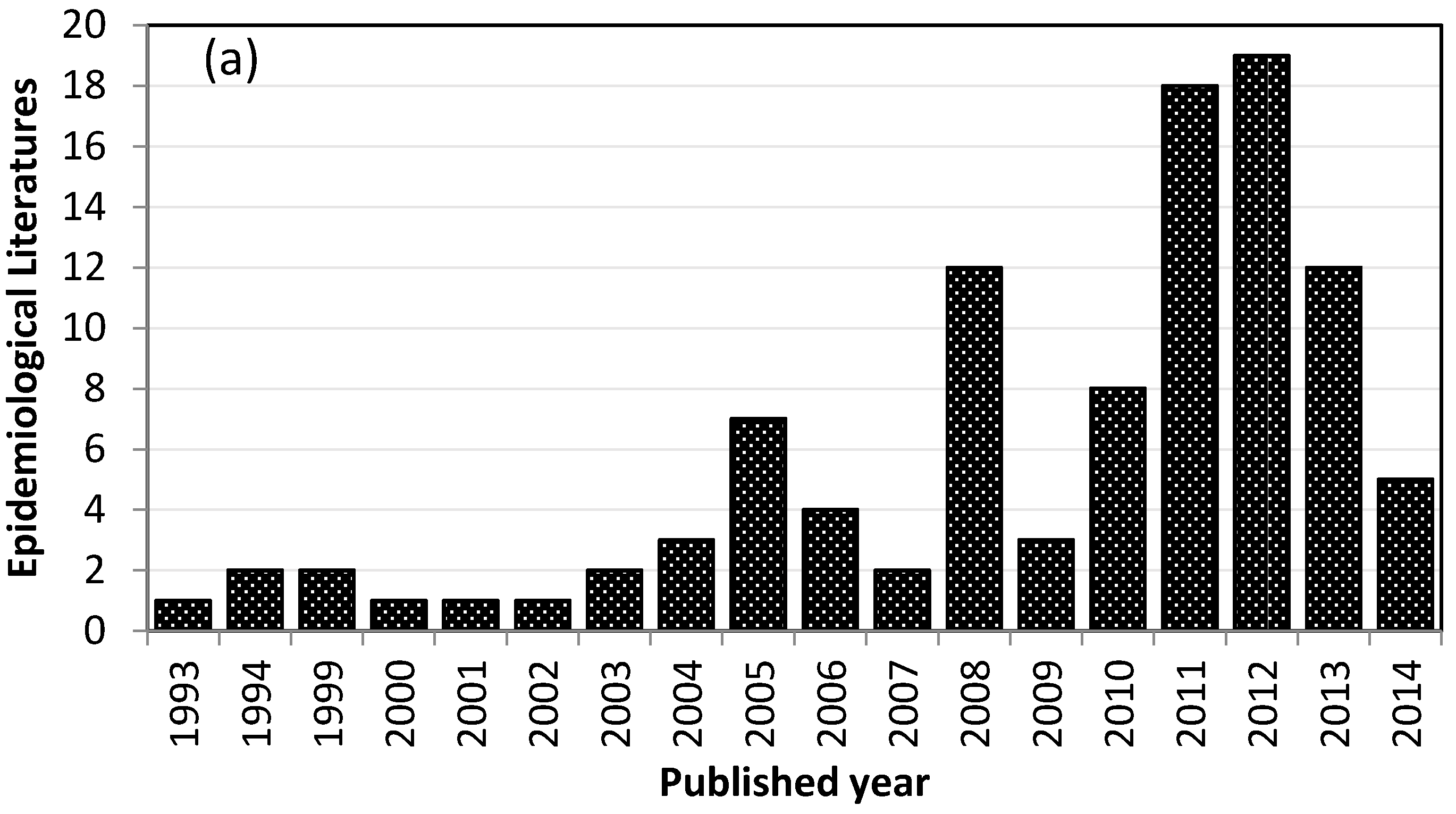
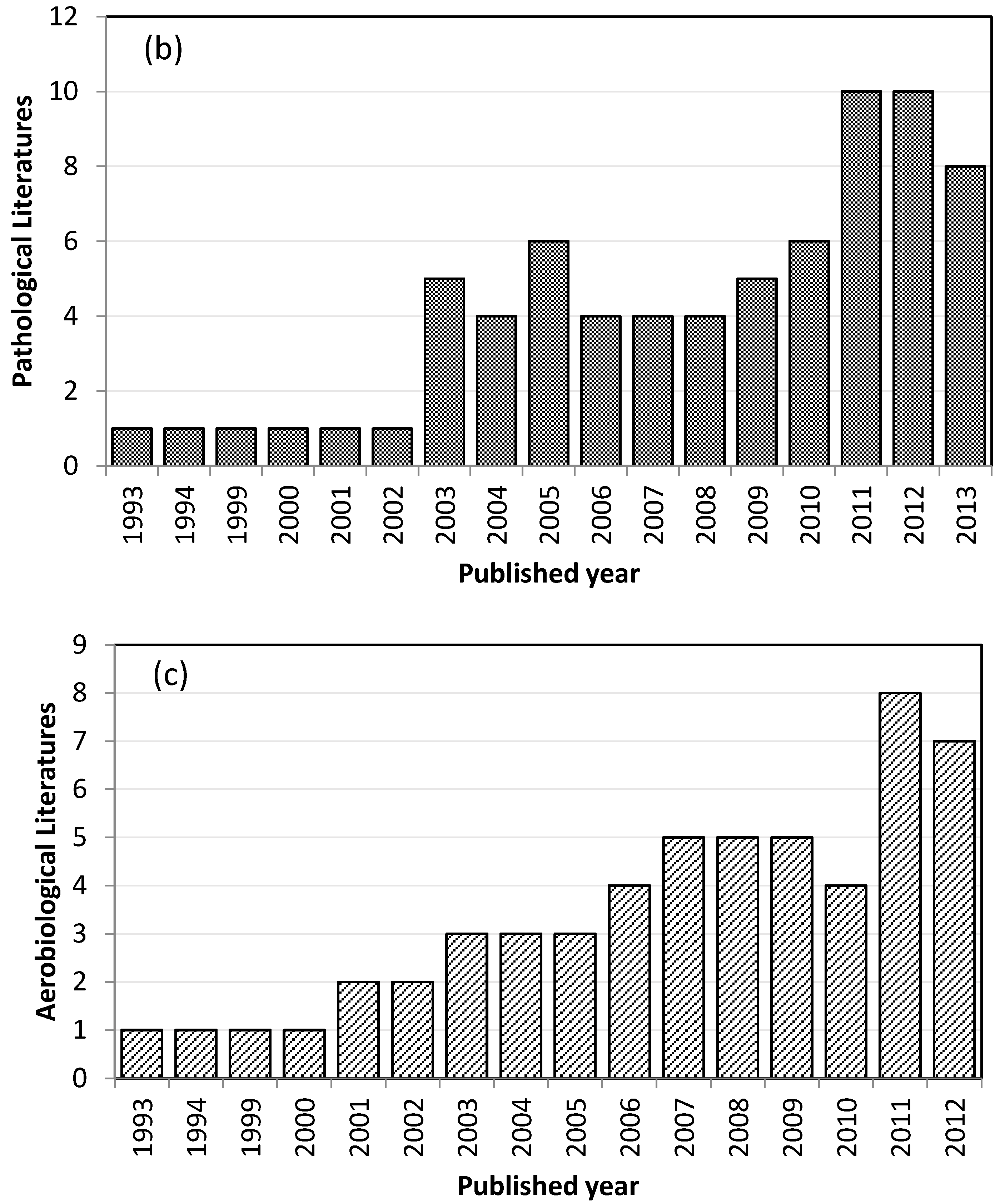
3.1. Geographical Distribution of Studies Addressing Health Effects of Desert Dust
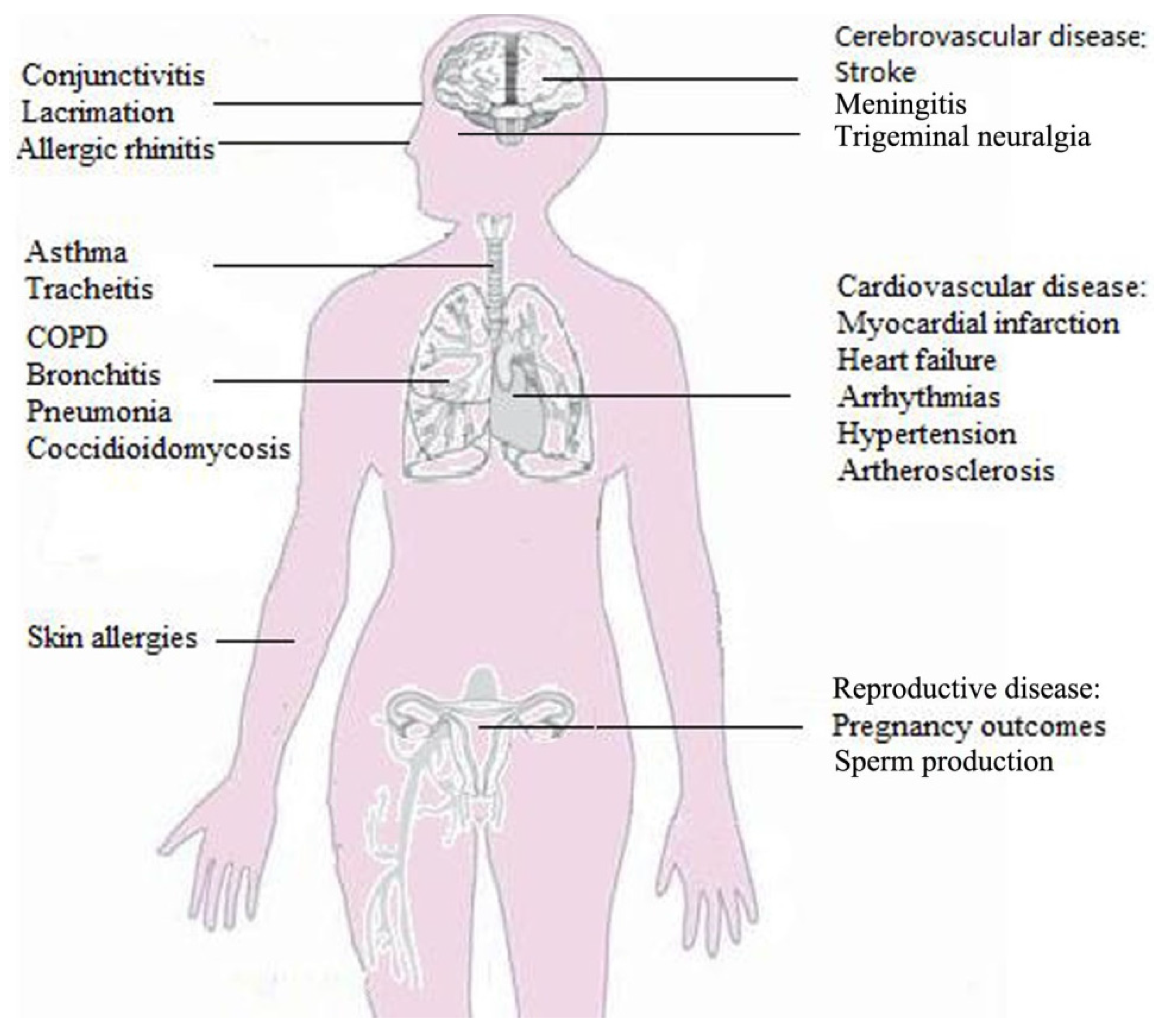
3.2. Desert Dust-Related Health Diseases
4. Desert Dust and Human Mortality
4.1. Mortality Effects from Desert Dust Coarse Particles, PM10 and PM2.5–10
4.2. Mortality Effects of Desert Dust Fine/Ultrafine Particles, PM2.5, PM2.5–1, and PM1.0
5. Desert Dust and Human Morbidity
5.1. Desert Dust and Respiratory Disease
5.1.1. Desert Dust Associated with Asthma and Rhinitis
5.1.2. Desert Dust Associated with Pneumonia and Chronic Obstructive Pulmonary Disease
5.1.3. Desert Dust and Other Respiratory Diseases
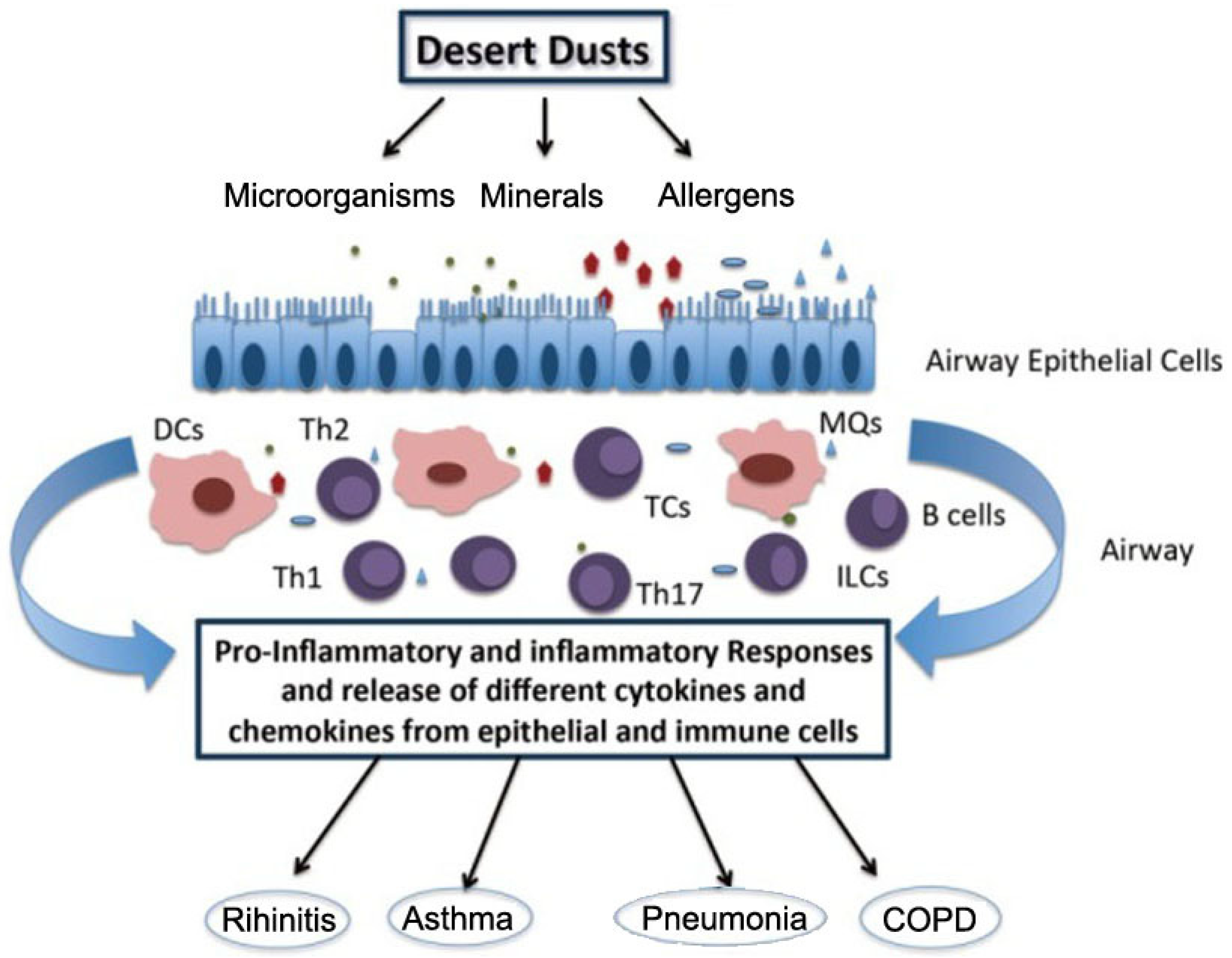
5.1.4. Pathogeny of Desert Dust on Respiratory Diseases
5.2. Desert Dust and Cardiovascular Diseases
5.3. Desert Dust and Maternity and Reproduction
5.4. Desert Dust and Other Infectious Diseases
6. Conclusions and Perspective
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shao, Y.; Wyrwoll, K.H.; Chappell, A.; Huang, J.; Lin, Z.; McTainsh, G.H.; Mikami, M.; Tanaka, T.Y.; Wang, X.; Yoon, S. Dust cycle: An emerging core theme in Earth system science. Aeolian Res. 2011, 2, 181–204. [Google Scholar] [CrossRef]
- Mahowald, N.M.; Kloster, S.; Engelstaedter, S.; Moore, J.K.; Mukhopadhyay, S.; McConnell, J.R.; Albani, S.; Doney, S.C.; Bhattacharya, A.; Curran, M.A.J.; et al. Observed 20th century desert dust variability: Impact on climate and biogeochemistry. Atmos. Chem. Phys. 2010, 10, 10875–10893. [Google Scholar] [CrossRef]
- De Longueville, F.; Hountondji, Y.C.; Henry, S.; Ozer, P. What do we know about effects of desert dust on air quality and human health in West Africa compared to other regions? Sci. Total Environ. 2010, 409, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Giannadaki, D.; Pozzer, A.; Lelieveld, J. Modeled global effects of airborne desert dust on air quality and premature mortality. Atmos. Chem. Phys. 2014, 14, 957–968. [Google Scholar] [CrossRef]
- Gonzalez-Martin, C.; Teigell-Perez, N.; Valladares, B.; Griffin, D.W. The Global dispersion of pathogenic microorganisms by dust storms and its relevance to agriculture. Adv. Argonomy 2014, 127, 1–41. [Google Scholar]
- Kwon, H.J.; Cho, S.H.; Chun, Y.; Lagarde, F.; Pershagen, G. Effects of the Asian dust events on daily mortality in Seoul, Korea. Environ. Res. 2002, 90, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.S.; Sheen, P.C.; Chen, E.R.; Liu, Y.K.; Wu, T.N.; Yang, C.Y. Effects of Asian dust storm events on daily mortality in Taipei, Taiwan. Environ. Res. 2004, 95, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.M.; McKendry, I.G.; Kelly, S.; Denike, K.; Koch, T. Impact of the 1998 Gobi dust event on hospital admissions in the Lower Fraser Valley, British Columbia. Sci. Total Environ. 2006, 366, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Middleton, N.; Yiallouros, P.; Kleanthous, S.; Kolokotroni, O.; Schwartz, J.; Dockery, D.W.; Demokritou, P.; Koutrakis, P. A 10-year time-series analysis of respiratory and cardiovascular morbidity in Nicosia, Cyprus: The effect of short-term changes in air pollution and dust storms. Environ. Health 2008, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pérez, L.L.; Tobias, A.; Querol, X.; Künzli, N.; Pey, J.; Alastuey, A.; Viana, M.; Valero, N.; González-Cabré, M.; Sunyer, J. Coarse particles from Saharan dust and daily mortality. Epidemiology 2008, 19, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Mallone, S.; Stafoggia, M.; Faustini, A.; Gobbi, G.P.; Marconi, A.; Forastiere, F. Saharan dust and associations between particulate matter and daily mortality in Rome, Italy. Environ. Health Perspect. 2011, 119, 1409. [Google Scholar] [CrossRef] [PubMed]
- Tam, W.W.S.; Wong, T.W.; Wong, A.H.S.; Hui, D.S. Effect of dust storm events on daily emergency admissions for respiratory diseases. Respirology 2012, 17, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Neophytou, A.M.; Yiallouros, P.; Coull, B.A.; Kleanthous, S.; Pavlou, P.; Pashiardis, S.; Dockery, D.W.; Koutrakis, P.; Laden, F. Particulate matter concentrations during desert dust outbreaks and daily mortality in Nicosia, Cyprus. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, M.; Ueda, K.; Nishiwaki, Y.; Michikawa, T.; Onozuka, D. Health effects of Asian dust events: A review of the literature. Nihon Eiseigaku Zasshi 2010, 65, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.J. Toxicity and health effects of Asian dust: A literature review. J. Korean Med. Assoc. 2012, 55, 234–242. [Google Scholar] [CrossRef]
- Karanasiou, A.; Moreno, N.; Moreno, T.; Viana, M.; De Leeuw, F.; Querol, X. Health effects from Sahara dust episodes in Europe: Literature review and research gaps. Environ. Int. 2012, 47, 107–114. [Google Scholar] [CrossRef] [PubMed]
- De Longueville, F.; Ozer, P.; Doumbia, S.; Henry, S. Desert dust impacts on human health: An alarming worldwide reality and a need for studies in West Africa. Int. J. Biometeorol. 2013, 57, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Goudie, A.S. Desert dust and human health disorders. Environ. Int. 2014, 63, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Honda, Y.; Lim, Y.H.; Guo, Y.L.; Hashizume, M.; Kim, H. Effect of Asian dust storms on mortality in three Asian cities. Atmos. Environ. 2014, 89, 309–317. [Google Scholar] [CrossRef]
- Al-Taiar, A.; Thalib, L. Short-term effect of dust storms on the risk of mortality due to respiratory, cardiovascular and all-causes in Kuwait. Int. J. Biometeorol. 2014, 58, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Samoli, E.; Kougea, E.; Kassomenos, P.; Analitis, A.; Katsouyanni, K. Does the presence of desert dust modify the effect of PM10 on mortality in Athens, Greece? Sci. Total Environ. 2011, 409, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
- Sajani, S.Z.; Miglio, R.; Bonasoni, P.; Cristofanelli, P.; Marinoni, A.; Sartini, C.; Goldoni, C.A.; Girolamo, G.D.; Lauriola, P. Saharan dust and daily mortality in Emilia-Romagna (Italy). Occup. Environ. Med. 2010. [Google Scholar] [CrossRef]
- Hwang, S.S.; Cho, S.H.; Kwon, H.J. Effects of the severe Asian dust events on daily mortality during the spring of 2002, in Seoul, Korea. J. Prev. Med.Public Health 2005, 38, 197–202. [Google Scholar] [PubMed]
- Lee, H.; Kim, H.; Honda, Y.; Lim, Y.H.; Yi, S. Effect of Asian dust storms on daily mortality in seven metropolitan cities of Korea. Atmos. Environ. 2013, 79, 510–517. [Google Scholar] [CrossRef]
- Lee, J.T.; Son, J.Y.; Cho, Y.S. A comparison of mortality related to urban air particles between periods with Asian dust days and without Asian dust days in Seoul, Korea, 2000–2004. Environ. Res. 2007, 105, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.; Ng, H.C. A case-crossover analysis of Asian dust storms and mortality in the downwind areas using 14-year data in Taipei. Sci. Total Environ. 2011, 410, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Díaz, J.; Tobías, A.; Linares, C. Saharan dust and association between particulate matter and case-specific mortality: A case-crossover analysis in Madrid (Spain). Environ. Health 2012, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Johnston, F.; Hanigan, I.; Henderson, S.; Morgan, G.; Bowman, D. Extreme air pollution events from bushfires and dust storms and their association with mortality in Sydney, Australia 1994–2007. Environ. Res. 2011, 111, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, E.; Linares, C.; Martínez, D.; Díaz, J. Role of Saharan dust in the relationship between particulate matter and short-term daily mortality among the elderly in Madrid (Spain). Sci. Total Environ. 2010, 408, 5729–5736. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, D.S.; Kim, H.; Yi, S.M. Relationship between mortality and fine particles during Asian dust, smog–Asian dust, and smog days in Korea. Int. J. Environ. Health Res. 2012, 22, 518–530. [Google Scholar] [CrossRef] [PubMed]
- Tobías, A.; Pérez, L.; Díaz, J.; Linares, C.; Pey, J.; Alastruey, A.; Querol, X. Short-term effects of particulate matter on total mortality during Saharan dust outbreaks: A case-crossover analysis in Madrid (Spain). Sci. Total Environ. 2011, 412, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Delangizan, S.; Jafari Motlagh, Z. Dust phenomenon effects on cardiovascular and respiratory hospitalizations and mortality—A case study in Kermanshah, during March-September 2010–2011. Iran. J. Health Environ. 2013, 6, 65–76. [Google Scholar]
- Kashima, S.; Yorifuji, T.; Tsuda, T.; Eboshida, A. Asian dust and daily all-cause or cause-specific mortality in western Japan. Occup. Environ. Med. 2012, 69, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Maté, T.; Guaita, R.; Pichiule, M.; Linares, C.; Díaz, J. Short-term effect of fine particulate matter (PM2.5) on daily mortality due to diseases of the circulatory system in Madrid (Spain). Sci. Total Environ. 2010, 408, 5750–5757. [Google Scholar] [CrossRef] [PubMed]
- Ginoux, P.; Chin, M.; Tegen, I.; Prospero, J.M.; Holben, B.; Dubovik, O.; Lin, S.J. Sources and distributions of dust aerosols simulated with the GOCART model. J. Geophys. Res.: Atmos. (1984–2012) 2001, 106, 20255–20273. [Google Scholar] [CrossRef]
- Prospero, J.M.; Ginoux, P.; Torres, O.; Nicholson, S.E.; Gill, T.E. Environmental characterization of global sources of atmospheric soil dust identified with the Nimbus 7 Total Ozone Mapping Spectrometer (TOMS) absorbing aerosol product. Rev. Geophys. 2002, 40. [Google Scholar] [CrossRef]
- Tanaka, T.Y.; Chiba, M. A numerical study of the contributions of dust source regions to the global dust budget. Glob. Planet. Chang. 2006, 52, 88–104. [Google Scholar] [CrossRef]
- Huneeus, N.; Schulz, M.; Balkanski, Y.; Griesfeller, J.; Kinne, S.; Prospero, J.; Bauer, S.; Boucher, O.; Chin, M.; Dentener, F.; et al. Global dust model intercomparison in AeroCom phase I. Atmos. Chem. Phys. 2011, 11, 7781–7816. [Google Scholar] [CrossRef]
- Varga, G. Spatio-temporal distribution of dust storms—A global coverage using NASA TOMS aerosol measurements. Hung Geogr. Bull. 2012, 61, 275–298. [Google Scholar]
- Ginoux, P.; Prospero, J.M.; Gill, T.E.; Hsu, N.C.; Zhao, M. Global-scale attribution of anthropogenic and natural dust sources and their emission rates based on MODIS deep blue aerosol products. Rev. Geophys. 2012, 50, RG3005. [Google Scholar] [CrossRef]
- Tegen, K.; Schepanski, K. The global distribution of mineral dust. In IOP Conference Series: Earth and Environmental Science; IOP: Barcelona, Spain, 2009; Volume 7. [Google Scholar] [CrossRef]
- Zhang, X.; Tong, D.Q.; Wu, G.; Wang, X.; Xiu, A.; Han, Y.; Xu, T.; Zhang, S.; Zhao, H. Identification of dust sources and hotspots in East Asia during 2000–2015: Implications for numerical modeling and forecasting. Atmos. Chem. Phys. Discuss. 2016. [Google Scholar] [CrossRef]
- Uno, I.; Eguchi, K.; Yumimoto, K.; Takemura, T.; Shimizu, A.; Uematsu, M.; Liu, Z.; Wang, Z.; Hara, Y.; Sugimoto, N. Asian dust transported one full circuit around the globe. Nat. Geosci. 2009, 2, 557–560. [Google Scholar] [CrossRef]
- Escudero, M.; Stein, A.F.; Draxler, R.R.; Querol, X.; Alastuey, A.; Castillo, S.; Avila, A. Source apportionment for African dust outbreaks over the Western Mediterranean using the HYSPLIT model. Atmos. Res. 2011, 99, 518–527. [Google Scholar] [CrossRef]
- Israelevich, P.; Ganor, E.; Alpert, P.; Kishcha, P.; Stupp, A. Predominant transport paths of Saharan dust over the Mediterranean Sea to Europe. J. Geophys. Res.: Atmos. (1984–2012) 2012, 117, D02205. [Google Scholar] [CrossRef]
- Varga, G.; Kovács, J.; Újvári, G. Analysis of Saharan dust intrusions into the Carpathian Basin (Central Europe) over the period of 1979–2011. Glob. Planet. Chang. 2013, 100, 333–342. [Google Scholar] [CrossRef]
- Tanaka, T.Y.; Kurosaki, Y.; Chiba, M.; Matsumura, T.; Nagai, T.; Yamazaki, A.; Uchiyama, A.; Tsunematsu, N.; Kai, K. Possible transcontinental dust transport from North Africa and the Middle East to East Asia. Atmos. Environ. 2005, 39, 3901–3909. [Google Scholar] [CrossRef]
- Ben-Ami, Y.; Koren, I.; Rudich, Y.; Artaxo, P.; Martin, S.T.; Andreae, M.O. Transport of North African dust from the Bodélé depression to the Amazon Basin: A case study. Atmos. Chem. Phys. 2010, 10, 7533–7544. [Google Scholar] [CrossRef]
- Barkan, J.; Alpert, P. Synoptic analysis of a rare event of Saharan dust reaching the Arctic region. Weather 2010, 65, 208–211. [Google Scholar] [CrossRef]
- Sunnu, A.; Resch, F.; Afeti, G. Back-trajectory model of the Saharan dust flux and particle mass distribution in West Africa. Aeolian Res. 2013, 9, 125–132. [Google Scholar] [CrossRef]
- McTainsh, G.H.; Nickling, W.G.; Lynch, A.W. Dust deposition and particle size in Mali, West Africa. Catena 1997, 29, 307–322. [Google Scholar] [CrossRef]
- Breuning-Madsen, H.; Awadzi, T.W. Harmattan dust deposition and particle size in Ghana. Catena 2005, 63, 23–38. [Google Scholar] [CrossRef]
- Anuforom, A.C. Spatial distribution and temporal variability of Harmattan dust haze in sub-Sahel West Africa. Atmos. Environ. 2007, 41, 9079–9090. [Google Scholar] [CrossRef]
- Resch, F.; Sunnu, A.; Afeti, G. Saharan dust flux and deposition rate near the Gulf of Guinea. Tellus B 2008, 60, 98–105. [Google Scholar] [CrossRef]
- Ridley, D.A.; Heald, C.L.; Prospero, J.M. What controls the recent changes in African mineral dust aerosol across the Atlantic? Atmos. Chem. Phys. Discuss. 2014, 14, 3583–3627. [Google Scholar] [CrossRef]
- Prospero, J.M.; Mayol-Bracero, O.L. Uderstanding the transport and imapct of Afican dust on the Caribbean Basin. Bull. Am. Meteorol. Soc. 2013, 94, 1329–1337. [Google Scholar] [CrossRef]
- Kallos, G.; Astitha, M.; Katsafados, P.; Spyrou, C. Long-range transport of anthropogenically and naturally produced particulate matter in the Mediterranean and North Atlantic: Current state of knowledge. J. Appl. Meteorol. Climatol. 2007, 46, 1230–1251. [Google Scholar] [CrossRef]
- Pey, J.; Querol, X.; Alastuey, A.; Forastiere, F.; Stafoggia, M. African dust outbreaks over the Mediterranean Basin during 2001–2011: PM10 concentrations, phenomenology and trends, and its relation with synoptic and mesoscale meteorology. Atmos. Chem. Phys. 2013, 13, 1395–1410. [Google Scholar] [CrossRef]
- International Classification of Diseases, Version 9. Available online: http://www.cdc.gov/nchs/icd/icd9cm.htm (accessed on 25 November 2016).
- International Classification of Diseases, Version 10. Available online: http://apps.who.int/classifications/icd10/browse/2016/en (accessed on 25 November 2016).
- Dominici, F.; McDermott, A.; Zeger, S.L.; Samet, J.M. On the use of generalized additive models in time-series studies of air pollution and health. Am. J. Epidemiol. 2002, 156, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Richardson, D.B.; Loomis, D. The impact of exposure categorisation for grouped analyses of cohort data. Occup. Environ. Med. 2004, 61, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.W. Atmospheric movement of microorganisms in clouds of desert dust and implications for human health. Clin. Microbiol. Rev. 2007, 20, 459–477. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Tobias, A.; Querol, X.; Alastuey, A.; Amato, F.; Pey, J.; Pérez, N.; Sunyer, J. The effects of particulate matter sources on daily mortality: A case crossover study of Barcelona Spain. Environ. Health Perspect. 2011, 119, 1781. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Villarrubia, E.; Iñiguez, C.; Ballester, F.; Peral, N.; García, D. Association of fine and coarse particles with mortality in Canary Island cities affected by Saharan dust intrusion. Epidemiology 2009, 20, S199–S200. [Google Scholar] [CrossRef]
- Hashizume, M.; Nishiwaki, Y.; Michikawa, T.; Ueda, K.; Onozuka, D.; Yokota, K.; Mine, M.; Mori, A.; Shimizu, A.; Sugimoto, N.; et al. Effects of Asian dust events on daily mortality in Nagasaki, Japan. Epidemiology 2011, 22, S130. [Google Scholar] [CrossRef]
- Pérez, L.; Tobías, A.; Querol, X.; Pey, J.; Alastuey, A.; Díaz, J.; Sunyer, J. Saharan dust, particulate matter and cause-specific mortality: A case–crossover study in Barcelona (Spain). Environ. Int. 2012, 48, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.S.; Yang, C.Y. Effects of Asian dust storm events on daily hospital admissions for cardiovascular disease in Taipei, Taiwan. J. Toxicol. Environ. Health Part A 2005, 68, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, S.; Alastuey, A.; Alonso-Pérez, S.; Querol, X.; Cuevas, E.; Abreu-Afonso, J.; Viana, M.; Pérez, N.; Pandolfi, M.; de la Rosa, J. Transport of desert dust mixed with North African industrial pollutants in the subtropical Saharan Air Layer. Atmos. Chem. Phys. 2011, 11, 6663–6685. [Google Scholar] [CrossRef]
- Hsieh, N.H.; Liao, C.M. Assessing exposure risk for dust storm events-associated lung function decrement in asthmatics and implications for control. Atmos. Environ. 2013, 68, 6e264. [Google Scholar] [CrossRef]
- Roy, C.J.; Milton, D.K. Airborne transmission of communicable infection--the elusive pathway. N. Engl. J. Med. 2004, 350, 1710–1712. [Google Scholar] [CrossRef] [PubMed]
- Prospero, J.M.; Blades, E.; Naidu, R.; Mathison, G.; Thani, H.; Lavoie, M.C. Relationship between African dust carried in the Atlantic trade winds and surges in pediatric asthma attendances in the Caribbean. Int. J. Biometeorol. 2008, 52, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Gyan, K.; Henry, W.; Lacaille, S.; Laloo, A.; Lamsee-Ebanks, C.; McKay, S.; Antoine, R.M.; Monteil, M.A. African dust clouds are associated with increased paediatric asthma accident and emergency admissions on the Caribbean island of Trinidad. Int. J. Biometeorol. 2005, 49, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Cadelis, G.; Tourres, R.; Molinie, J. Short-term effects of the particulate pollutants contained in Saharan dust on the visits of children to the emergency department due to asthmatic conditions in Guadeloupe (French Archipelago of the Caribbean). PLoS ONE 2014, 9, e91136. [Google Scholar] [CrossRef] [PubMed]
- Meo, S.A.; Al-Kheraiji, M.F.A.; AlFaraj, Z.F.; abdulaziz Alwehaibi, N.; Aldereihim, A.A. Respiratory and general health complaints in subjects exposed to sandstorm at Riyadh, Saudi Arabia. Pak. J. Med. Sci. 2013, 29, 642. [Google Scholar] [CrossRef] [PubMed]
- Thalib, L.; Al-Taiar, A. Dust storms and the risk of asthma admissions to hospitals in Kuwait. Sci. Total Environ. 2012, 433, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, S.; Clark, E.; McTainsh, G.; Simpson, R.; Mitchell, C. Characteristics of rural dust events shown to impact on asthma severity in Brisbane, Australia. Int. J. Biometeorol. 1999, 42, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Merrifield, A.; Schindeler, S.; Jalaludin, B.; Smith, W. Health effects of the September 2009 dust storm in Sydney, Australia: Did emergency department visits and hospital admissions increase? Environ. Health 2013, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Samoli, E.; Nastos, P.T.; Paliatsos, A.G.; Katsouyanni, K.; Priftis, K.N. Acute effects of air pollution on pediatric asthma exacerbation: Evidence of association and effect modification. Environ. Res. 2011, 111, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Grineski, S.E.; Staniswalis, J.G.; Bulathsinhala, P.; Peng, Y.; Gill, T.E. Hospital admissions for asthma and acute bronchitis in El Paso, Texas: Do age, sex, and insurance status modify the effects of dust and low wind events? Environ. Res. 2011, 111, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.L.; Levy, J.K.; Lin, Z. The effect of sandstorms and air pollution on cause-specific hospital admissions in Taipei, Taiwan. Occup. Environ. Med. 2008, 65, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.C.; Leem, J.; Hong, Y.C.; Kim, H.; Kim, H.C. Effects of Asian dust storm events on daily admissions for asthma and stroke in seven metropolitans of Korea. Epidemiology 2008, 19, S145. [Google Scholar]
- Lee, J.W.; Lee, K.K. Effects of Asian dust events on daily asthma patients in Seoul, Korea. Meteorol. Appl. 2014, 21, 202–209. [Google Scholar] [CrossRef]
- Kanatani, K.T.; Ito, I.; Al-Delaimy, W.K.; Adachi, Y.; Mathews, W.C.; Ramsdell, J.W. Desert dust exposure is associated with increased risk of asthma hospitalization in children. Am. J. Respir. Crit. Care Med. 2010, 182, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M. Influence of Asian dust storms on asthma in western Japan. Genes Environ. 2014, 36, 137–144. [Google Scholar] [CrossRef]
- AI-Dowaisan, A.; Fakim, N.; Khan, M.R.; Arifhodzic, N.; Panicker, R.; Hanoon, A.; Khan, I. Salsola pollen as a predominant cause of respiratory allergies in Kuwait. Ann. Allergy Asthma Immunol. 2004, 92, 262–267. [Google Scholar] [CrossRef]
- Ichinose, T.; Hiyoshi, K.; Yoshida, S.; Takano, H.; Inoue, K.; Nishikawa, M.; Mori, I.; Kawazato, H.; Yasuda, A.; Shibamoto, T. Asian sand dust aggravates allergic rhinitis in guinea pigs induced by Japanese cedar pollen. Inhal. Toxicol. 2009, 21, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Igishi, T.; Burioka, N.; Yamaski, A.; Kurai, J.; Takeuchi, H.; Sako, T.; Yoshida, A.; Yoneda, K.; Fukuoka, Y.; et al. Pollen augments the influence of desert dust on symptoms of adult asthma patients. Allergol. Int. 2011, 60, 517. [Google Scholar] [CrossRef] [PubMed]
- Ueda, K.; Nitta, H.; Odajima, H. The effects of weather, air pollutants, and Asian dust on hospitalization for asthma in Fukuoka. Environ. Health Prev. Med. 2010, 15, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Tsai, S.S.; Chang, C.C.; Ho, S.C. Effects of Asian dust storm events on daily admissions for asthma in Taipei, Taiwan. Inhal. Toxicol. 2005, 17, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Min, P.K.; Kim, C.W.; Yun, Y.J.; Chang, J.H.; Chu, J.K.; Lee, K.E.; Han, J.Y.; Park, J.W.; Hong, C.S. Effect of yellow sand on respiratory symptoms and diurnal variation of peak expiratory flow in patients with bronchial asthma. J. Asthma, Allergy Clin. Immunol. 2001, 21, 1179–1186. [Google Scholar]
- Meltzer, E.O.; Blaiss, M.S.; Naclerio, R.M.; Stoloff, S.W.; Derebery, M.J.; Nelson, H.S.; Boyle, J.M.; Wingertzahn, M.A. Burden of allergic rhinitis: Allergies in America, Latin America, and Asia-Pacific adult surveys. Allergy Asthma Proc. 2012, 33, S113–S141. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, S.; Dales, R.E.; Burnett, R.T.; Judek, S.; Coates, F.; Brook, J.R. Effect of airborne allergens on emergency visits by children for conjunctivitis and rhinitis. Lancet 2002, 16, 947–948. [Google Scholar] [CrossRef]
- Sato, T. The study on the change of symptoms in allergic rhinitis during Asian sand dust phenomenon. Practica Oto-Rhino-Laryngologica 2009, 102, 831–839. [Google Scholar] [CrossRef]
- Yang, Z.H.; Meng, Z.Q.; Pan, J.J.; Yan, X.Z. Epidemiological survey on the diseases of the respiratory and circulatory system among the people in a region where dust events occurred frequently. J. Environ. Occup. Med. 2008, 1, 004. [Google Scholar]
- Chang, C.C.; Lee, I.M.; Tsai, S.S.; Yang, C.Y. Correlation of Asian dust storm events with daily clinic visits for allergic rhinitis in Taipei, Taiwan. J. Toxicol. Environ. Health Part A 2006, 69, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Bjorksten, B.; Dumitrascu, D.; Foucard, T.; Khetsuriani, N.; Khaitov, R.; Leja, M.; Lis, G.; Pekkanen, J.; Priftanji, A.; Riikjarv, M.A. Prevalence of childhood asthma, rhinitis and eczema in Scandinavia and Eastern Europe. Eur. Respir. J. 1998, 12, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Ezeamuzie, C.I.; Thomson, M.S.; Al-Ali, S.; Dowaisan, A.; Khan, M.; Hijazi, Z. Asthma in the desert: Spectrum of the sensitizing aeroallergens. Allergy 2000, 55, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Shamssain, M.H.; Shamsian, N. Prevalence and severity of asthma, rhinitis, and atopic eczema in 13- to 14-year-old schoolchildren from the northeast of England. Ann. Allergy Asthma Immunol. 2001, 86, 428–432. [Google Scholar] [CrossRef]
- Uduma, A.U.; Jimoh, W.L.O. High incidence of Asthma, Bronchitis, Pneumonia and Sinusitis in Kano State, North West Nigeria during Saharan dust events. Am. J. Environ. Energy Power Res. 2013, 1, 174–185. [Google Scholar]
- Korenyi-Both, A.L.; Molnar, A.C.; Fidelus-Gort, R. Al Eskan disease: Desert storm pneumonitis. Mil. Med. 1992, 157, 452–462. [Google Scholar] [PubMed]
- Ozer, P. Dust in the wind and public health: Example from Mauritania. In Desertification: Migration, Health, Remediation and Local Governance; Royal Academy for Overseas Sciences: Brussels, Belgium, 2008; pp. 55–74. [Google Scholar]
- Meng, Z.; Lu, B. Dust events as a risk factor for daily hospitalization for respiratory and cardiovascular diseases in Minqin, China. Atmos. Environ. 2007, 41, 7048–7058. [Google Scholar] [CrossRef]
- Cheng, M.F.; Ho, S.C.; Chiu, H.F.; Wu, T.N.; Chen, P.S.; Yang, C.Y. Consequences of exposure to Asian dust storm events on daily pneumonia hospital admissions in Taipei, Taiwan. J. Toxicol. Environ. Health Part A 2008, 71, 1295–1299. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Keller, J.J.; Chen, C.S.; Lin, H.C. Asian dust storm events are associated with an acute increase in pneumonia hospitalization. Ann. Epidemiol. 2012, 22, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Gudavalli, R.; Garcha, P.; Farver, C.; Yun, J.; Pettersson, G.B.; Mason, D.P.; Murthy, S.P.; Budev, M. Interstitial lung disease associated with Asian dust storm exposure. CHEST J. 2009, 136, 33S–34S. [Google Scholar] [CrossRef]
- Chiu, H.F.; Tiao, M.M.; Ho, S.C.; Kuo, H.W.; Wu, T.N.; Yang, C.Y. Effects of Asian dust storm events on hospital admissions for chronic obstructive pulmonary disease in Taipei, Taiwan. Inhal. Toxicol. 2008, 20, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.; Chuang, K.J.; Chen, W.J.; Chang, W.T.; Lee, C.T.; Peng, C.M. Increasing cardiopulmonary emergency visits by long-range transported Asian dust storms in Taiwan. Environ. Res. 2008, 106, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Vodonos, A.; Friger, M.; Katra, I.; Avnon, L.; Krasnov, H.; Koutrakis, P.; Schwartz, J.; Lior, O.; Novack, V. The impact of desert dust exposures on hospitalizations due to exacerbation of chronic obstructive pulmonary disease. Air Qual. Atmos. Health 2014, 7, 433–439. [Google Scholar] [CrossRef]
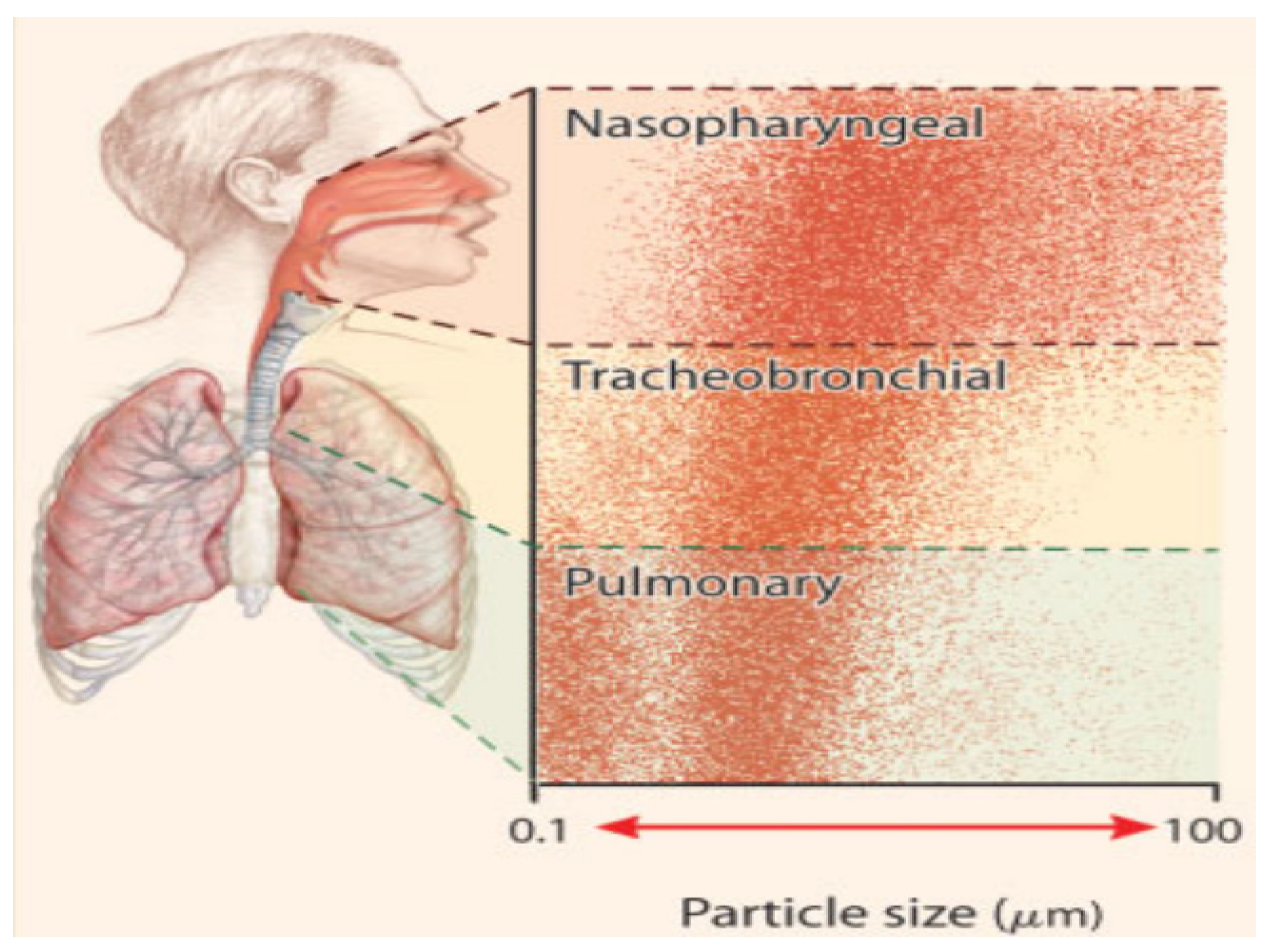
- Kim, C.S.; Hu, S.C. Regional deposition of inhaled particles in human lungs: Comparison between men and women. J. Appl. Physiol. 1998, 84, 1834–1844. [Google Scholar] [PubMed]
- Seaton, A.; Godden, D.; MacNee, W.; Donaldson, K. Particulate air pollution and acute health effects. Lancet 1995, 345, 176–178. [Google Scholar] [CrossRef]
- Gwack, J.; Hwang, S.S.; Kwon, H.J.; Hong, Y.C.; Cho, S.H.; Yoo, Y.; Yoo, J.H.; Ko, Y.R. Effects of Asian dust events on diurnal variation of peak expiratory flow rate in children with bronchial asthma and healthy children. Epidemiology 2005, 16, S109–S110. [Google Scholar] [CrossRef]
- Park, J.W.; Lim, Y.H.; Kyung, S.Y.; An, C.H.; Lee, S.P.; Jeong, S.H.; Ju, Y.S. Effects of ambient particulate matter on peak expiratory flow rates and respiratory symptoms of asthmatics during Asian dust periods in Korea. Respirology 2005, 10, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.; Choung J, T.; Yu, J.; Kim, D.K.; Koh, Y.Y. Acute effects of Asian dust events on respiratory symptoms and peak expiratory flow in children with mild asthma. J. Korean Med. Sci. 2008, 23, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.C.; Pan, X.C.; Kim, S.Y.; Park, K.; Park, E.J.; Jin, X.; Yi, S.M.; Kim, Y.H.; Park, C.H.; Song, S.; et al. Asian dust storm and pulmonary function of school children in Seoul. Sci. Total Environ. 2010, 408, 754–759. [Google Scholar] [CrossRef] [PubMed]
- Kelsall, H.L.; Sim, M.R.; Forbes, A.B.; McKenzie, D.P.; Glass, D.C.; Ikin, J.F.; Ittak, P.; Abramson, M.J. Respiratory health status of Australian veterans of the 1991 Gulf War and the effects of exposure to oil fire smoke and dust storms. Thorax 2004, 59, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Derbyshire, E. Natural minerogenic dust and human health. AMBIO: J. Hum. Environ. 2007, 36, 73–77. [Google Scholar] [CrossRef]
- Richards, A.L.; Hyams, K.C.; Watts, D.M.; Rozmajzl, P.J.; Woody, J.N.; Merrell, B.R. Respiratory disease among military personnel in Saudi Arabia during Operation Desert Shield. Am. J. Public Health 1993, 83, 1326–1329. [Google Scholar] [CrossRef] [PubMed]
- Crum, N.F.; Potter, M.; Pappagianis, D. Seroincidence of coccidioidomycosis during military desert training exercises. J. Clin. Microbiol. 2004, 42, 4552–4555. [Google Scholar] [CrossRef] [PubMed]
- Korzeniewski, K.; Nitsch-Osuch, A.; Chciałowski, A.; Korsak, J. Environmental factors, immune changes and respiratory diseases in troops during military activities. Respir. Physiol. Neurobiol. 2013, 187, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Engelbrecht, J.P.; McDonald, E.V.; Gillies, J.A.; Gertler, A.W. Department of Defense Enhanced Particulate Matter Surveillance Program (EPMSP). Available online: http://oai.dtic.mil/oai/oai?verb=getRecord&metadataPrefix=html&identifier=ADA605600 (accessed on 25 November 2016).
- Szema, A.M.; Peters, M.C.; Weissinger, K.M.; Gagliano, C.A.; Chen, J.J. New-onset asthma among soldiers serving in Iraq and Afghanistan. In Allergy and Asthma Proceedings; OceanSide Publications: Rhode Island, RI, USA, 2010; pp. 67–71. [Google Scholar]
- King, M.S.; Eisenberg, R.; Newman, J.H.; Tolle, J.J.; Harrell, F.E.; Nian, H.; Ninan, M.; Lambright, E.S.; Scheller, J.R.; Johnson, J.E.; et al. Constrictive bronchiolitis in soldiers returning from Iraq and Afghanistan. N. Engl. J. Med. 2011, 365, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.J.; Dodson, D.W.; Lucero, P.F.; Haislip, G.D.; Gallup, R.A.; Nicholson, K.L.; Zacher, L.L. Study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE). Am. J. Respir. Crit. Care Med. 2014, 190, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Tabuena, R.P.; Niimi, A.; Inoue, H.; Ito, I.; Yamaguchi, M.; Otsuka, K.; Takeda, T.; Oguma, T.; Nakaji, H.; et al. Cough triggers and their pathophysiology in patients with prolonged or chronic cough. Allergol. Int. 2012, 61, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Higashi, T.; Kambayashi, Y.; Ohkura, N.; Fujimura, M.; Nakanishi, S.; Yoshizaki, T.; Saijoh, K.; Hayakawa, K.; Kobayashi, F.; Michigami, Y.; et al. Exacerbation of daily cough and allergic symptoms in adult patients with chronic cough by Asian dust: A hospital-based study in Kanazawa. Atmos. Environ. 2014, 97, 537–543. [Google Scholar] [CrossRef]
- Higashi, T.; Kambayashi, Y.; Ohkura, N.; Fujimura, M.; Nakai, S.; Honda, Y.; Saijoh, K.; Hayakawa, K.; Kobayashi, F.; Michigami, Y.; et al. Effects of Asian dust on daily cough occurrence in patients with chronic cough: A panel study. Atmos. Environ. 2014, 92, 506–513. [Google Scholar] [CrossRef]
- Yanagisawa, R.; Takano, H.; Ichinose, T.; Mizushima, K.; Nishikawa, M.; Mori, I.; Inoue, K.I.; Sadakane, K.; Yoshikawa, T. Gene expression analysis of murine lungs following pulmonary exposure to Asian sand dust particles. Exp. Biol. Med. 2007, 232, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.; Foster, M.L.; Law, J.M.H.; Centeno, J.A.; Fornero, E.; Henderson, M.S.; Trager, S.A.; Stockelman, M.G.; Dorman, D.C. Assessment of geographical variation in the respiratory toxicity of desert dust particles. Inhal. Toxicol. 2013, 25, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Feng, L.; Huang, C.; Yan, X.; Zhang, X. Potential hazardous elements (PHEs) in atmospheric particulate matter (APM) in the south of Xi’an during the dust episodes of 2001–2012 (NW China): Chemical fractionation, ecological and health risk assessment. Environ. Earth Sci. 2014, 71, 4115–4126. [Google Scholar] [CrossRef]
- Naota, M.; Mukaiyama, T.; Shimada, A.; Kohara, Y.; Morita, T.; Inoue, K.; Takano, H. Pathological study of acute pulmonary toxicity induced by intratracheally instilled Asian sand dust (Kosa). Toxicol. Pathol. 2010, 38, 1099–1110. [Google Scholar] [CrossRef] [PubMed]
- Honda, A.; Matsuda, Y.; Murayama, R.; Tsuji, K.; Nishikawa, M.; Koike, E.; Yoshida, S.; Ichinose, T.; Takano, H. Effects of Asian sand dust particles on the respiratory and immune system. J. Appl. Toxicol. 2014, 34, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kurai, J.; Tomita, K.; Sano, H.; Abe, S.; Saito, R.; Minato, S.; Igishi, T.; Burioka, N.; Sako, T.; et al. Effects on asthma and induction of interleukin-8 caused by Asian dust particles collected in western Japan. J. Asthma 2014, 51, 595–602. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Ichinose, T.; Yoshida, S.; Nishikawa, M.; Mori, I.; Yanagisawa, R.; Takano, H.; Inoue, K.; Sun, G.; Shibamoto, T. Airborne Asian sand dust enhances murine lung eosinophilia. Inhal. Toxicol. 2010, 22, 1012–1025. [Google Scholar] [CrossRef] [PubMed]
- Kang, I.G.; Jung, J.H.; Kim, S.T. Asian sand dust enhances allergen-induced Th2 allergic inflammatory changes and mucin production in BALB/c mouse lungs. Allergy Asthma Immunol. 2012, 4, 206–213. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Ichinose, T.; Yoshida, S.; Takano, H.; Nishikawa, M.; Sun, G.; Shibamoto, T. Induction of immune tolerance and reduction of aggravated lung eosinophilia by co-exposure to Asian sand dust and ovalbumin for 14 weeks in mice. Allerg. Asthma Clin. Immunol. 2013, 9, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Gheybi, M.K.; Movahed, A.M.; Dehdari, R.; Amiri, S.; Khazaei, H.A.; Gooya, M.; Dehbashi, F.; Fatemi, A.; Sovid, N.; Hajiani, G.; et al. Dusty air pollution is associated with an increased risk of allergic diseases in southwestern part of Iran. Iran. J. Allergy Asthma Immunol. 2014, 13, 404. [Google Scholar] [PubMed]
- Ichinose, T.; Yoshida, S.; Sadakane, K.; Takano, H.; Yanagisawa, R.; Inoue, K.; Nishikawa, M.; Mori, I.; Kawazato, H.; Yasuda, A.; et al. Effects of asian sand dust, Arizona sand dust, amorphous silica and aluminum oxide on allergic inflammation in the murine lung. Inhal. Toxicol. 2008, 20, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Ichinose, T.; He, M.; Arashidani, K.; Yoshida, Y.; Yoshida, S.; Nishikawa, M.; Takano, H.; Sun, G.; Shibamoto, T. Aggravation of ovalbumin-induced murine asthma by co-exposure to desert-dust and organic chemicals: An animal model study. Environ. Health 2014, 13, 83. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Wang, Z.; Wang, S. Toxic effect of desert dust of northwest areas of China on alveolar macrophages of rats. J. Environ. Health 2008, 25, 1056–1058. [Google Scholar]
- Kyung, S.Y.; Yoon, J.Y.; Kim, Y.J.; Lee, S.P.; Park, J.W.; Jeong, S.H. Asian dust particles induce TGF-β1 via reactive oxygen species in bronchial epithelial cells. Tuberc. Respir. Dis. 2012, 73, 84–92. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Ichinose, T.; Song, Y.; Yoshida, Y.; Arashidani, K.; Yoshida, S.; Liu, B.; Nishikawa, M.; Takano, H.; Sun, G. Effects of two Asian sand dusts transported from the dust source regions of Inner Mongolia and northeast China on murine lung eosinophilia. Toxicol. Appl. Pharmacol. 2013, 272, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Alcorn, J.F.; Crowe, C.R.; Kolls, J.K. TH17 cells in asthma and COPD. Annu. Rev. Physiol. 2010, 72, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Aujla, S.J.; Alcorn, J.F. TH17 cells in asthma and inflammation. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2011, 1810, 1066–1079. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.C.; Chan, C.C.; Wang, P.Y.; Lee, C.T.; Cheng, T.J. Effects of Asian dust event particles on inflammation markers in peripheral blood and bronchoalveolar lavage in pulmonary hypertensive rats. Environ. Res. 2004, 95, 71–76. [Google Scholar] [CrossRef]
- Yeo, N.K.; Hwang, Y.J.; Kim, S.T.; Kwon, H.J.; Jang, Y.J. Asian sand dust enhances rhinovirus-induced cytokine secretion and viral replication in human nasal epithelial cells. Inhal. Toxicol. 2010, 22, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.H.; Ye, M.K.; Hwang, Y.J.; Kim, S.T. The effect of Asian sand dust-activated respiratory epithelial cells on activation and migration of eosinophils. Inhal. Toxicol. 2013, 25, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Higashisaka, K.; Fujimura, M.; Taira, M.; Yoshida, T.; Tsunoda, S.; Baba, T.; Yamaguchi, N.; Nabeshi, H.; Yoshikawa, T.; Nasu, M.; et al. Asian Dust Particles Induce Macrophage Inflammatory Responses via Mitogen-Activated Protein Kinase Activation and Reactive Oxygen Species Production. Available online: http://dx.doi.org/10.1155/2014/856154 (accessed on 25 November 2016).
- He, M.; Ichinose, T.; Yoshida, S.; Yamamoto, S.; Inoue, K.; Takano, H.; Yanagisawa, R.; Nishikawa, M.; Mori, I.; Sun, G.; et al. Asian sand dust enhances murine lung inflammation caused by Klebsiella pneumoniae. Toxicol. Appl. Pharmacol. 2012, 258, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Dorman, D.C.; Mokashi, V.; Wagner, D.J.; Wagner, D.J.; Olabisi, A.O.; Wong, B.A.; Moss, O.R.; Centeno, J.A.; Guandalini, G.; Jackson, D.A.; et al. Biological responses in rats exposed to cigarette smoke and Middle East sand (dust). Inhal. Toxicol. 2012, 24, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Ghio, A.J.; Kummarapurugu, S.T.; Tong, H.; Soukup, J.M.; Dailey, L.A.; Boykin, E.; Ian Gilmour, M.; Ingram, P.; Roggli, V.L.; Goldstein, H.L.; et al. Biological effects of desert dust in respiratory epithelial cells and a murine model. Inhal. Toxicol. 2014, 26, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Krewski, D.; Yokel, R.A.; Nieboer, E.; Borchelt, D.; Cohen, J.; Harry, J.; Kacew, S.; Lindsay, J.; Mahfouz, A.M.; Rondeau, V. Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J. Toxicol. Environ. Health Part B 2007, 10, 1–269. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J.; Celli, B.R. Systemic manifestations and comorbidities of COPD. Eur. Respir. J. 2009, 33, 1165–1185. [Google Scholar] [CrossRef] [PubMed]
- Ezzie, M.E.; Crawford, M.; Cho, J.H.; Orellana, R.; Zhang, S.; Gelinas, R.; Batte, K.; Yu, L.; Nuovo, G.; Galas, D.; et al. Gene expression networks in COPD: MicroRNA and mRNA regulation. Thorax 2011. [Google Scholar] [CrossRef] [PubMed]
- Marjanović, N.; Bosnar, M.; Michielin, F.; Willé, D.R.; Anić-Milić, T.; Culić, O.; Popović-Grle, S.; Bogdan, M.; Parnham, M.J.; Eraković Haber, V. Macrolide antibiotics broadly and distinctively inhibit cytokine and chemokine production by COPD sputum cells in vitro. Pharmacol. Res. 2011, 63, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Brusselle, G.G.; Joos, G.F.; Bracke, K.R. New insights into the immunology of chronic obstructive pulmonary disease. Lancet 2011, 378, 1015–1026. [Google Scholar] [CrossRef]
- Meng, Z.; Zhang, Q. Damage effects of dust storm PM2.5 on DNA in alveolar macrophages and lung cells of rats. Food Chem. Toxicol. 2007, 45, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Alessandrini, E.R.; Stafoggia, M.; Faustini, A.; Gobbi, G.P.; Forastiere, F. Saharan dust and the association between particulate matter and daily hospitalisations in Rome, Italy. Occup. Environ. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, J.; Yang, Z.; Zhang, Y.; Meng, Z. Impact of PM2.5 derived from dust events on daily outpatient numbers for respiratory and cardiovascular diseases in Wuwei, China. Procedia Environ. Sci. 2013, 18, 290–298. [Google Scholar] [CrossRef]
- Tam, W.W.; Wong, T.W.; Wong, A.H. Effect of dust storm events on daily emergency admissions for cardiovascular diseases. Circ. J. 2012, 76, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Cheng, M.H.; Chen, C.C. Effects of Asian dust storm events on hospital admissions for congestive heart failure in Taipei, Taiwan. J. Toxicol. Environ. Health Part A 2009, 72, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, R.; Michikawa, T.; Ueda, K.; Nitta, H.; Kawasaki, T.; Tashiro, H.; Mohri, M.; Yamamoto, Y. Desert dust is a risk factor for the incidence of acute myocardial infarction in Western Japan. Circ.: Cardiovasc. Qual. Outcomes 2014, 7, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Chen, Y.S.; Chiu, H.F.; Goggins, W.B. Effects of Asian dust storm events on daily stroke admissions in Taipei, Taiwan. Environ. Res. 2005, 99, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Liu, T.C.; Keller, J.; Lin, H.C. Asian dust storm events are associated with an acute increase in stroke hospitalisation. J. Epidemiol. Community Health 2012. [Google Scholar] [CrossRef] [PubMed]
- Kamouchi, M.; Ueda, K.; Ago, T.; Nitta, H.; Kitazono, T.; Fukuoka stroke registry investigators. Relationship between Asian dust and ischemic stroke a time-stratified case-crossover study. Stroke 2012, 43, 3085–3087. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.; Meng, Z. Evaluation of micronucleus induction of sand dust storm fine particles (PM2.5) in human blood lymphocytes. Environ. Toxicol. Pharmacol. 2006, 22, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Lipsett, M.J.; Tsai, F.C.; Roger, L.; Woo, M.; Ostro, B.D. Coarse particles and heart rate variability among older adults with coronary artery disease in the Coachella Valley, California. Environ. Health Perspect. 2006, 114, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Hwang, J.S.; Chan, C.C.; Wang, P.Y.; Cheng, T.J. Effects of concentrated ambient particles on heart rate, blood pressure, and cardiac contractility in spontaneously hypertensive rats during a dust storm event. Inhal. Toxicol. 2007, 19, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Brook, R. Cardiovascular effects of air pollution. Clin. Sci. 2008, 115, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Šrám, R.J.; Binková, B.; Dejmek, J.; Bobak, M. Ambient air pollution and pregnancy outcomes: A review of the literature. Environ. Health Perspect. 2005, 113, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.S.; Balkhair, T. Air pollution and birth outcomes: A systematic review. Environ. Int. 2011, 37, 498–516. [Google Scholar] [CrossRef] [PubMed]
- Stieb, D.M.; Chen, L.; Eshoul, M.; Judek, S. Ambient air pollution, birth weight and preterm birth: A systematic review and meta-analysis. Environ. Res. 2012, 117, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Hyder, A.; Lee, H.J.; Ebisu, K.; Koutrakis, P.; Belanger, K.; Bell, M.L. PM2.5 exposure and birth outcomes: Use of satellite-and monitor-based data. Epidemiology 2014, 25, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Dadvand, P.; Basagaña, X.; Figueras, F.; Amoly, E.; Tobias, A.; de Nazelle, A.; Querol, X.; Sunyer, J.; Nieuwenhuijsen, M.J. Saharan dust episodes and pregnancy. J. Environ. Monit. 2011, 13, 3222–3228. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Hiyoshi, K.; Ichinose, T.; Nishikawa, M.; Takano, H.; Sugawara, I.; Takeda, K. Aggravating effect of natural sand dust on male reproductive function in mice. Reprod. Med. Biol. 2009, 8, 151–156. [Google Scholar] [CrossRef]
- Yang, C.Y. Effects of Asian dust storm events on daily clinical visits for conjunctivitis in Taipei, Taiwan. J. Toxicol. Environ. Health Part A 2006, 69, 1673–1680. [Google Scholar] [CrossRef] [PubMed]
- Mimura, T.; Yamagami, S.; Fujishima, H.; Noma, H.; Kamei, Y.; Goto, M.; Kondo, A.; Matsubara, M. Sensitization to Asian dust and allergic rhinoconjunctivitis. Environ. Res. 2014, 132, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Al-Hurban, A.E.; Al-Ostad, A.N. Textural characteristics of dust fallout and potential effect on public health in Kuwait City and suburbs. Environ. Earth Sci. 2010, 60, 169–181. [Google Scholar] [CrossRef]
- Formenti, P.; Schütz, L.; Balkanski, Y.; Desboeufs, K.; Ebert, M.; Kandler, K.; Petzold, A.; Scheuvens, D.; Weinbruch, S.; Zhang, D. Recent progress in understanding physical and chemical properties of African and Asian mineral dust. Atmos. Chem. Phys. 2011, 11, 8231–8256. [Google Scholar] [CrossRef]
- Formenti, P.; Caquineau, S.; Desboeufs, K.; Klaver, A.; Chevaillier, S.; Journet, E.; Rajot, J.L. Mapping the physico-chemical properties of mineral dust in western Africa: Mineralogical composition. Atmos. Chem. Phys. 2014, 14, 10663–10686. [Google Scholar] [CrossRef]
- Zhang, X.L.; Wu, G.J.; Zhang, C.L.; Xu, T.L.; Zhou, Q.Q. What is the real role of iron oxides in the optical properties of dust aerosols? Atmos. Chem. Phys. 2015, 15, 12159–12177. [Google Scholar] [CrossRef]
- Otani, S.; Onishi, K.; Mu, H.; Kurozawa, Y. The effect of Asian dust events on the daily symptoms in Yonago, Japan: A pilot study on healthy subjects. Arch. Environ. Occup. Health 2011, 66, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Otani, S.; Onishi, K.; Mu, H.; Yokoyama, Y.; Hosoda, T.; Okamoto, M.; Kurozawa, Y. The relationship between skin symptoms and allergic reactions to Asian dust. Int. J. Environ. Res. Public Health 2012, 9, 4606–4614. [Google Scholar] [CrossRef] [PubMed]
- Onishi, K.; Otani, S.; Yoshida, A.; Mu, H.; Kurozawa, Y. Adverse health effects of Asian dust particles and heavy metals in Japan. Asia-Pac. J. Public Health 2012, 27, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Onishi, K.; Kurosaki, Y.; Otani, S.; Yoshida, A.; Sugimoto, N.; Kurozawa, Y. Atmospheric transport route determines components of Asian dust and health effects in Japan. Atmos. Environ. 2012, 49, 94–102. [Google Scholar] [CrossRef]
- Choi, H.; Shin, D.W.; Kim, W.; Doh, S.J.; Lee, S.H.; Noh, M. Asian dust storm particles induce a broad toxicological transcriptional program in human epidermal keratinocytes. Toxicol. Lett. 2011, 200, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhao, X.G.; Wang, Q. Effects of dust event on the upper respiratory tract and eyes uncomfortable symptoms of primary school students in Southern Xinjiang. J. Environ. Health 2010, 27, 767–771. [Google Scholar]
- Mu, H.; Battsetseg, B.; Ito, T.Y.; Otani, S.; Onishi, K.; Kurozawa, Y. Health effects of dust storms: Subjective eye and respiratory system symptoms in inhabitants in Mongolia. J. Environ. Health 2011, 73, 18–20. [Google Scholar] [PubMed]
- Doganay, H.; Akcali, D.; Goktaş, T.; Caglar, K.; Erbas, D.; Saydam, C.; Bolay, H. African dust-laden atmospheric conditions activate the trigeminovascular system. Cephalalgia 2009, 29, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, C.A.; Griffin, D.W. Aerobiology and the global transport of desert dust. Trends Ecol. Evol. 2006, 21, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.W.; Kellogg, C.A. Dust storms and their impact on ocean and human health: Dust in Earth’s atmosphere. EcoHealth 2004, 1, 284–295. [Google Scholar] [CrossRef]
- Leski, T.A.; Malanoski, A.P.; Gregory, M.J.; Lin, B.; Stenger, D.A. Application of broad-range resequencing array RPM-TEI for detection of pathogens in desert dust samples from Kuwait and Iraq. Appl. Environ. Microbiol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Malo, J.; Luraschi-Monjagatta, C.; Wolk, D.M.; Thompson, R.; Hage, C.A.; Knox, K.S. Update on the diagnosis of pulmonary coccidioidomycosis. Ann. Am. Thorac. Soc. 2014, 11, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Kolivras, K.N.; Johnson, P.S.; Comrie, A.C.; Yool, S.R. Environmental variability and coccidioidomycosis (valley fever). Aerobiologia 2001, 17, 31–42. [Google Scholar] [CrossRef]
- Nguyen, C.; Barker, B.M.; Hoover, S.; Nix, DE.; Ampel, N.M.; Frelinger, J.A.; Orbach, M.J.; Galgiani, J.N. Recent advances in our understanding of the environmental, epidemiological, immunological, and clinical dimensions of coccidioidomycosis. Clin. Microbiol. Rev. 2013, 26, 505–525. [Google Scholar] [CrossRef] [PubMed]
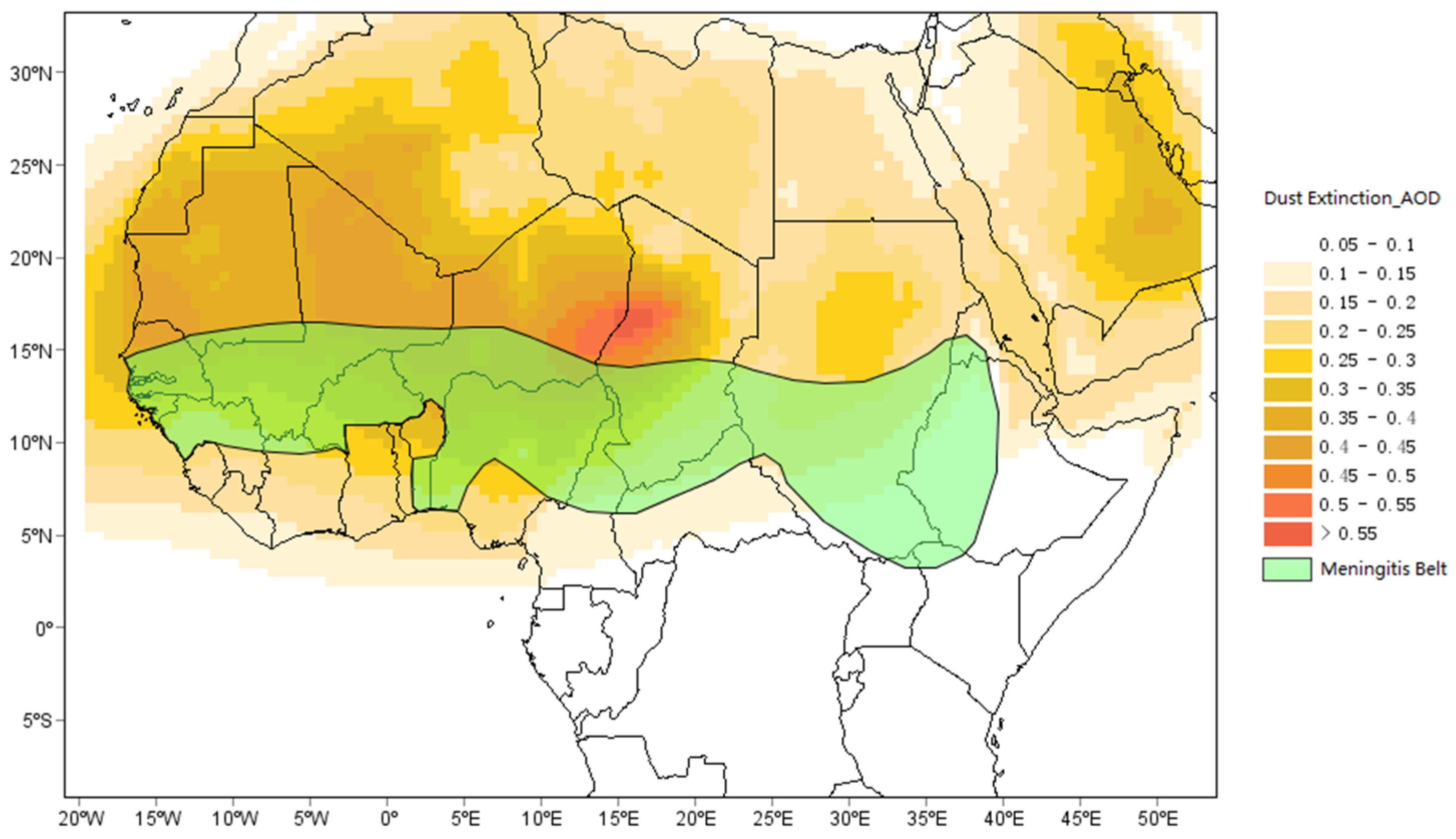
- Thomson, M.C.; Connor, S.J.; Ward, N.; Molyneux, D. Impact of climate variability on infectious disease in West Africa. EcoHealth 2004, 1, 138–150. [Google Scholar] [CrossRef]
- Sultan, B.; Labadi, K.; Guégan, J.F.; Janicot, S. Climate drives the meningitis epidemics onset in West Africa. PLoS Med 2005, 2, e6. [Google Scholar] [CrossRef] [PubMed]
- Yaka, P.; Sultan, B.; Broutin, H.; Janicot, S.; Philippon, S.; Fourquet, N. Relationships between climate and year-to-year variability in meningitis outbreaks: A case study in Burkina Faso and Niger. Int. J. Health Geogr. 2008, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Thomson, M.C.; Jeanne, I.; Djingarey, M. Dust and epidemic meningitis in the Sahel: A public health and operational research perspective. In IOP Conference Series: Earth and Environmental Science; IOP: Barcelona, Spain, 2009; Volume 7. [Google Scholar] [CrossRef]
- Dukić, V.; Hayden, M.; Forgor, A.A.; Hopson, T.; Akweongo, P.; Hodgson, A.; Monaghan, A.; Wiedinmyer, C.; Yoksas, T.; Thomson, M.C.; et al. The role of weather in meningitis outbreaks in Navrongo, Ghana: A generalized additive modeling approach. J. Agric. Biol. Environ. Stat. 2012, 17, 442–460. [Google Scholar] [CrossRef]
- Molesworth, A.M.; Cuevas, L.E.; Connor, S.J.; Morse, A.P.; Thomson, M.C. Environmental risk and meningitis epidemics in Africa. Emerg. Infect. Dis. 2003, 9, 1287. [Google Scholar] [CrossRef] [PubMed]
- Tobías, A.; Caylà J, A.; Pey, J.; Alastuey, A.; Querol, X. Are Saharan dust intrusions increasing the risk of meningococcal meningitis? Int. J. Infectious Dis. 2011, 15, e503. [Google Scholar] [CrossRef] [PubMed]
- Agier, L.; Deroubaix, A.; Martiny, N.; Yaka, P.; Djibo, A.; Broutin, H. Seasonality of meningitis in Africa and climate forcing: Aerosols stand out. J. R. Soc. Interface 2013, 10, 20120814. [Google Scholar] [CrossRef] [PubMed]
- Martiny, N.; Chiapello, I. Assessments for the impact of mineral dust on the meningitis incidence in West Africa. Atmos. Environ. 2013, 70, 245–253. [Google Scholar] [CrossRef]
- Deroubaix, A.; Martiny, N.; Chiapello, I.; Marticoréna, B. Suitability of OMI aerosol index to reflect mineral dust surface conditions: Preliminary application for studying the link with meningitis epidemics in the Sahel. Remote Sens. Environ. 2013, 133, 116–127. [Google Scholar] [CrossRef]
- Pérez, G.P.C.; Stanton, M.C.; Diggle, P.J.; Trzaska, S.; Miller, R.L.; Perlwitz, J.P.; Baldasano, J.M.; Cuevas, E.; Ceccato, P.; Yaka, P.; et al. Soil dust aerosols and wind as predictors of seasonal meningitis incidence in Niger. Environ. Health Perspect. 2014, 122, 679–696. [Google Scholar]
- Jordan, P.W.; Saunders, N.J. Host iron binding proteins acting as niche indicators for Neisseria meningitidis. PLoS ONE 2009, 4, e5198. [Google Scholar] [CrossRef] [PubMed]
- Meningitis Belt over North Africa, the Meningitis Vaccine Project. Available online: http://www.meningvax.org (accessed on 25 November 2016).
| Mortality Causes | Population | PM Fraction | % Risk per 10 μg/m3 (95% CI) | Location | Reference |
|---|---|---|---|---|---|
| TotM | All ages | PM10 | −1.62(−2.76,−0.415) | Taipei, Taiwan, China | [19] |
| TotM | All ages | PM10 | −1.5(−3.0,0.0) | Kuwait | [20] |
| TotM | All ages | PM10 | −0.1(−0.6,0.4) | Athens, Greece | [21] |
| TotM | All ages | PM10 | 0.0(−3.5,3.6) | Emilia-Romagna, Italy | [22] |
| TotM | All ages | PM10 | 0.102(−0.20,0.403) | Seoul, Korea | [23]] |
| TotM | PM10 | 0.13(−1.03,1.30) | Cyprus | [13] | |
| TotM | All ages | PM10 | 0.15(0.00,0.30) | 7 cities of Korea | [24] |
| TotM | All ages | PM10 | 0.17(−0.16,0.53) | Seoul, Korea | [6] |
| TotM | All ages | PM10 | 0.17(0.05,0.31) | Seoul, Korea | [25] |
| TotM | PM10 | 0.190(−0.028,0.412) | Seoul, Korea | [19] | |
| TotM | All ages | PM10 | 0.722 | Taipei, Taiwan, China | [7] |
| TotM | All ages | PM10 | 0.79(0.12,1.45) | Taipei, Taiwan, China | [26] |
| TotM | All ages | PM10 | 1.16(−1.09,3.63) | Kitakyushu, Japan | [19] |
| TotM | PM10 | 1.70(0.79,2.62) | Madrid, Spain | [27] | |
| TotM | All ages | PM10 | 1.96(0.38,3.78) | Sydney, Australia | [28] |
| TotM | Age > 75 | PM10 | 2.7(1.4,4.1) | Madrid, Spain | [29] |
| TotM | Age > 35 | PM10 | 3.0(0.0,6.0) | Rome, Italy | [11] |
| TotM | All ages | PM2.5 | 0.3(0.1,0.5) | Seoul, Korea | [30] |
| TotM | Age > 35 | PM2.5 | 2.5(−0.9,6.1) | Rome, Italy | [11] |
| TotM | All ages | PM2.5 | 2.9(−1.1,6.9) | Madrid, Spain | [31] |
| TotM | All ages | PM2.5 | 5.0(0.5,9.7) | Barcelona, Spain | [10] |
| TotM | Age > 35 | PM2.5–10 | 1.1(−0.6,2.7) | Rome, Italy | [11] |
| TotM | All ages | PM2.5–10 | 2.8(0.1,5.8) | Madrid, Spain | [31] |
| TotM | All ages | PM2.5–10 | 8.4(1.5,15.8) | Barcelona, Spain | [10] |
| RES | PM10 | −5.31(−8.64,−1.71) | Taipei, Taiwan, China | [19] | |
| RES | PM10 | −4.0(−1.2,4.0) | Kuwait | [20] | |
| RES | All ages | PM10 | −0.2(−10.8,11.5) | Emilia-Romagna, Italy | [22] |
| RES | All ages | PM10 | 0.2(−0.5,2.7) | Athens, Greece | [21] |
| RES | All ages | PM10 | 0.25(−0.33,0.86) | 7 cities of Korea | [24] |
| RES | PM10 | 0.79(−4.69,3.28) | Cyprus | [13] | |
| RES | All ages | PM10 | 1.49(0.03,3.47) | Seoul, Korea | [23] |
| RES | Age > 35 | PM10 | 2.5(−11.9,19.3) | Rome, Italy | [11] |
| RES | All ages | PM10 | 2.91(−4.18,3.04) | Sydney, Australia | [28] |
| RES | PM10 | 3.48(1.22,5.79) | Madrid, Spain | [27] | |
| RES | Age > 75 | PM10 | 4.0(1.7,6.3) | Madrid, Spain | [29] |
| RES | PM10 | 0.637(−0.222,1.572) | Seoul, Korea | [19] | |
| RES | All ages | PM10 | 7.66 | Taipei, Taiwan, China | [7] |
| RES | PM10 | 6.27(0.50,13.03) | Kitakyushu, Japan | [19] | |
| RES | All ages | PM2.5–10 | 3.51(−8.19,16.60) | Barcelona, Spain | [10] |
| RES | Age > 35 | PM2.5–10 | 9.8(0.2,21.3) | Rome, Italy | [11] |
| RES | Age > 35 | PM2.5 | 6.6(−10,27) | Rome, Italy | [11] |
| RES | All ages | PM2.5–1 | 15.71(−2.43,36.29) | Barcelona, Spain | [10] |
| RES | All ages | PM1 | 1.95(−9.20,14.37) | Barcelona, Spain | [10] |
| CIRC | PM10 | 0.30 | Kermanshah, Iran | [32] | |
| CIRC | Age > 65 | PM10 | 0.6(0.1,1.1) | Western Japan | [33] |
| CIRC | PM10 | 1.95(0.29,3.64) | Madrid, Spain | [27] | |
| CIRC | All ages | PM10 | 2.59% | Taipei, Taiwan, China | [7] |
| CIRC | Age > 75 | PM10 | 3.5(0.9,6.1) | Madrid, Spain | [29] |
| CIRC | Age > 35 | PM10 | 5.5(0.9,10.2) | Rome, Italy | [11] |
| CIRC | Age > 35 | PM2.5 | −0.7(−5.5,4.4) | Rome, Italy | [11] |
| CIRC | PM2.5 | 2.5(0.7,4.5) | Madrid, Spain | [34] | |
| CIRC | Age > 35 | PM2.5–10 | 4.0(1.6,6.5) | Rome, Italy | [11] |
| CAR | PM10 | −1.83(−4.09,0.55) | Taipei, Taiwan, China | [19] | |
| CAR | PM10 | −1.4(−4.0,1.2) | Kuwait | [20] | |
| CAR | All ages | PM10 | −0.8(−5.9,4.6) | Emilia-Romagna, Italy | [22] |
| CAR | All ages | PM10 | −0.25(−0.80,0.40) | Seoul, Korea | [23] |
| CAR | All ages | PM10 | 0.2(−0.4,0.9) | Athens, Greece | [21] |
| CAR | PM10 | 0.30 | Kermanshah, Iran | [32] | |
| CAR | All ages | PM10 | 0.30(0.01,0.59) | 7 cities of Korea | [24] |
| CAR | PM10 | 0.408(−0.003,0.837) | Seoul, Korea | [19] | |
| CAR & RES | All ages | PM10 | 0.41(−0.38,1.26) | Seoul, Korea | [6] |
| CAR | All ages | PM10 | 1.52(−1.65,5.57) | Sydney, Australia | [28] |
| CAR | PM10 | 2.43 (0.53, 4.37) | Cyprus | [13] | |
| CAR | PM10 | 4.09(0.006,8.73) | Kitakyushu, Japan | [19] | |
| CAR | Age > 35 | PM10 | 8.9(3.5,14.5) | Rome, Italy | [11] |
| CAR | Age > 35 | PM2.5–10 | 4.9(2.2,7.8) | Rome, Italy | [11] |
| CAR | All ages | PM2.5–10 | 10.32(3.09,17.98) | Barcelona, Spain | [10] |
| CAR | All ages | PM2.5 | 0.2(−0.1,0.5) | Seoul, Korea | [30] |
| CAR | Age > 35 | PM2.5 | 1.1(−4.6,7.2) | Rome, Italy | [11] |
| CAR | All ages | PM2.5–1 | 13.29(2.57,24.71) | Barcelona, Spain | [10] |
| CAR | All ages | PM1 | 4.14(−3.22,12.07) | Barcelona, Spain | [10] |
| CER | Age > 35 | PM10 | 1.6(−4.7,8.4) | Rome, Italy | [11] |
| CER | PM10 | 0.46(−2.93,3.96) | Madrid, Spain | [27] | |
| CER | Age > 35 | PM2.5 | −2.5(−9.1,4.8) | Rome, Italy | [11] |
| CER | All ages | PM2.5–10 | 8.62(4.36,23.30) | Barcelona, Spain | [10] |
| CER | Age > 35 | PM2.5–10 | 3.5(0.6,6.7) | Barcelona, Spain | [10] |
| CER | All ages | PM2.5–1 | 0.29(18.43,21.86) | Barcelona, Spain | [10] |
| CER | All ages | PM1 | 5.29(−8.16,20.46) | Barcelona, Spain | [10] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Zhao, L.; Tong, D.Q.; Wu, G.; Dan, M.; Teng, B. A Systematic Review of Global Desert Dust and Associated Human Health Effects. Atmosphere 2016, 7, 158. https://doi.org/10.3390/atmos7120158
Zhang X, Zhao L, Tong DQ, Wu G, Dan M, Teng B. A Systematic Review of Global Desert Dust and Associated Human Health Effects. Atmosphere. 2016; 7(12):158. https://doi.org/10.3390/atmos7120158
Chicago/Turabian StyleZhang, Xuelei, Lijing Zhao, Daniel Q. Tong, Guangjian Wu, Mo Dan, and Bo Teng. 2016. "A Systematic Review of Global Desert Dust and Associated Human Health Effects" Atmosphere 7, no. 12: 158. https://doi.org/10.3390/atmos7120158
APA StyleZhang, X., Zhao, L., Tong, D. Q., Wu, G., Dan, M., & Teng, B. (2016). A Systematic Review of Global Desert Dust and Associated Human Health Effects. Atmosphere, 7(12), 158. https://doi.org/10.3390/atmos7120158





