The Characterization of Biodiversity and Soil Emission Activity of the “Ladoga” Carbon-Monitoring Site
Abstract
1. Introduction
2. Materials and Methods
2.1. Geography, Climate, Topography, and Parent Material
2.2. Field and Laboratory Studies
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abakumov, E.V.; Polyakov, V.I.; Chukov, S.N. Approaches and Methods for Studying Soil Organic Matter in the Carbon Polygons of Russia (Review). Eurasian Soil Sci. 2022, 55, 849–860. [Google Scholar] [CrossRef]
- Carbon Supersites. Russian Federation. Available online: https://carbon-polygons.ru/en/ (accessed on 10 January 2024).
- Bossio, D.A.; Cook-Patton, S.C.; Ellis, P.W.; Fargione, J.; Sanderman, J.; Smith, P.; Wood, S.; Zomer, R.J.; von Unger, M.; Emmer, I.M.; et al. The role of soil carbon in natural climate solutions. Nat. Sustain. 2020, 3, 391–398. [Google Scholar] [CrossRef]
- Polyakov, V.; Abakumov, E.; Nizamutdinov, T.; Shevchenko, E.; Makarova, M. Estimation of Carbon Stocks and Stabilization Rates of Organic Matter in Soils of the «Ladoga» Carbon Monitoring Site. Agronomy 2023, 13, 807. [Google Scholar] [CrossRef]
- Makarova, M.V.; Abakumov, E.V.; Shevchenko, E.V.; Paramonova, N.N.; Pakhomova, N.V.; Lvova, N.A.; Vetrova, M.A.; Foka, S.C.; Guzov, Y.N.; Ivakhov, V.M.; et al. From carbon polygon to carbon farm: The potential and ways of developing the sequestration carbon industry in the Leningrad Region and St. Petersburg. Earth Sci. 2023, 68, 82–102. [Google Scholar] [CrossRef]
- Suleymanov, A.; Abakumov, E.; Nizamutdinov, T.; Polyakov, V.; Shevchenko, E.; Makarova, M. Soil organic carbon stock retrieval from Sentinel-2A using a hybrid approach. Environ. Monit. Assess. 2024, 196, 23. [Google Scholar] [CrossRef] [PubMed]
- Batjes, N.H. Total carbon and nitrogen in the soils of the world. Eur. J. Soil Sci. 1996, 47, 151–163. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Junaid, M.F.; Ijaz, N.; Khalid, U.; Ijaz, Z. Remediation methods of heavy metal contaminated soils from environmental and geotechnical standpoints. Sci. Total Environ. 2023, 867, 161468. [Google Scholar] [CrossRef] [PubMed]
- Zomer, R.J.; Bossio, D.A.; Sommer, R.; Verchot, L.V. Global Sequestration Potential of Increased Organic Carbon in Cropland Soils. Sci. Rep. 2017, 7, 15554. [Google Scholar] [CrossRef] [PubMed]
- Kudeyarov, V.N. Current state of the carbon budget and the capacity of Russian soils for carbon sequestration. Eurasian Soil Sci. 2015, 48, 923–933. [Google Scholar] [CrossRef]
- Kurganova, I.N.; de Gerenyu, V.O.L.; Ipp, S.L.; Kaganov, V.V.; Khoroshaev, D.A.; Rukhovich, D.I.; Sumin, Y.V.; Durmanov, N.D.; Kuzyakov, Y.V. Pilot carbon polygon in Russia: Analysis of carbon stocks in soils and vegetation. J. Soils Environ. 2022, 5, e169. [Google Scholar] [CrossRef]
- Chernova, O.V.; Golozubov, O.M.; Alyabina, I.O.; Schepaschenko, D.G. Integrated Approach to Spatial Assessment of Soil Organic Carbon in the Russian Federation. Eurasian Soil Sci. 2021, 54, 325–336. [Google Scholar] [CrossRef]
- Zhao, F.; Wu, Y.; Hui, J.; Sivakumar, B.; Meng, X.; Liu, S. Projected soil organic carbon loss in response to climate warming and soil water content in a loess watershed. Carbon Balance Manag. 2021, 16, 24. [Google Scholar] [CrossRef] [PubMed]
- Beillouin, D.; Corbeels, M.; Demenois, J.; Berre, D.; Boyer, A.; Fallot, A.; Feder, F.; Cardinael, R. A global meta-analysis of soil organic carbon in the Anthropocene. Nat. Commun. 2023, 14, 3700. [Google Scholar] [CrossRef] [PubMed]
- Winkler, K.; Fuchs, R.; Rounsevell, M.; Herold, M. Global land use changes are four times greater than previously estimated. Nat. Commun. 2021, 12, 2501. [Google Scholar] [CrossRef] [PubMed]
- Bullock, E.L.; Woodcock, C.E. Carbon loss and removal due to forest disturbance and regeneration in the Amazon. Sci. Total Environ. 2021, 764, 142839. [Google Scholar] [CrossRef]
- Fokich, Y.V. (Ed.) Red Data Book of Nature of the Leningrad Region. Volume 1: Protected Areas; Biological Research Institute of the St. Petersburg State University: St. Petersburg, Russia, 1999; 352p. [Google Scholar]
- Glushkovskaya, N.B.; Semenova, I.S. Major periods in the Koltushi heights background, their current state and outlooks for development. Proc. Russ. State Hydrometeorol. Univ. 2009, 11, 123–131. [Google Scholar]
- Malakhovsky, D.B.; Markov, K.K. (Eds.) Geomorphology and Quaternary Deposits of the North-West of the European Part of the USSR; Nauka: Leningrad, Russia, 1969; 256p. [Google Scholar]
- Gagarina, E.I.; Matinyan, N.N.; Schastnaya, L.S.; Kasatkina, G.A. Soils and Soil Cover in Northwest Russia; Saint-Petersburg State University: Saint-Petersburg, Russia, 1995; 236p. [Google Scholar]
- Gagarina, E.I. Lithological Factor of Soil Formation (on Example of North-West of Russian Plain); Saint-Petersburg State University: Saint-Petersburg, Russia, 2004; 260p. [Google Scholar]
- IUSS Working Group WRB. World Reference Base for Soil Resources. In International Soil Classification System for Naming Soils and Creating Legends for Soil Maps, 4th ed.; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022. [Google Scholar]
- V.V. Dokuchaep Soil Institute (VASKhNIL). Classification and Diagnostics of Soils in Russia; Oykumena: Smolensk, Russia, 2004; 342p. [Google Scholar]
- Khitrov, N.B.; Gerasimova, M.I. Diagnostic Properties and Soil Forming Materials in the Classification System of Russian Soils: Version of 2021. Eurasian Soil Sci. 2022, 55, 1–10. [Google Scholar] [CrossRef]
- Khitrov, N.B.; Gerasimova, M.I. Diagnostic Horizons in the Classification System of Russian Soils: Version 2021. Eurasian Soil Sci. 2021, 54, 1131–1140. [Google Scholar] [CrossRef]
- Ipatov, V.S.; Mirin, D.M. Description of Phytocenosis. Methodical Recommendations. Educational and Methodical Manual; Saint Petersburg State University: Saint Petersburg, Russia, 2008; 71p. [Google Scholar]
- Vorobyeva, L.N. (Ed.) Theory and Practice of Chemical Analysis of Soils; GEOS: Moscow, Russia, 2006; 400p. [Google Scholar]
- Shein, E.V. (Ed.) Field and Laboratory Methods of Research of Physical Properties and Soil Regimes; Moscow State University: Moscow, Russia, 2001; 200p. [Google Scholar]
- Semenov, V.M.; Lebedeva, T.N.; de Gerenyu, V.O.L.; Ovsepyan, L.A.; Semenov, M.V.; Kurganova, I.N. Pools and fractions of organic carbon in soil: Structure, functions and methods of determination. J. Soils Environ. 2023, 6, e199. [Google Scholar] [CrossRef]
- Semenov, V.M.; Ivannikova, L.A.; Kuznetsova, T.V.; Semenova, N.A.; Tulina, A.S. Mineralization of organic matter and the carbon sequestration capacity of zonal soils. Eurasian Soil Sci. 2008, 41, 717–730. [Google Scholar] [CrossRef]
- Pinaev, A.G.; Kichko, A.A.; Aksenova, T.S.; Safronova, V.I.; Kozhenkova, E.V.; Andronov, E.E. RIAM: A Universal Accessible Protocol for the Isolation of High Purity DNA from Various Soils and Other Humic Substances. Methods Protoc. 2022, 5, 99. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Callahan, B.; Mcmurdie, P.; Rosen, M.; Han, A.; Johnson, A.J.; Holmes, S. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Mcmurdie, P.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Parte, A.C.; Carbasse, J.S.; Meier-Kolthoff, J.P.; Reimer, L.C.; Göker, M. List of Prokaryotic Names with Standing in Nomenclature (LPSN) Moves to the DSMZ. Int. J. Syst. Evol. Microbiol. 2020, 70, 5607–5612. [Google Scholar] [CrossRef] [PubMed]
- Isachenko, A.G.; Dashkevich, Z.V.; Karnaukhova, E.V. Physical-Geographical Zoning of the North-West of Russia; Nauka: Leningrad, Russia, 1965. [Google Scholar]
- Lukina, E.V.; Noskov, G.A. To the avifauna of Koltushsky heights (Leningrad region). Russ. Ornithol. J. 2015, 24, 3476–3477. [Google Scholar]
- Sukhacheva, E.U.; Aparin, B.F. Soil Cover Patterns in Anthropogenically Transformed Landscapes of Leningrad Oblast. Eurasian Soil Sci. 2019, 52, 1146–1158. [Google Scholar] [CrossRef]
- Zakharova, M.K.; Morgach, Y.R. Specific Features of the Soil Cover of Recreation Areas in Leningrad Oblast. Eurasian Soil Sci. 2023, 56 (Suppl. S2), S202–S213. [Google Scholar] [CrossRef]
- Subetto, D.A. (Ed.) The Atlas of the Leningrad Region; Publishing House A. I. Herzen Russian State Pedagogical University: Saint Petersburg, Russia, 2022; 112p. [Google Scholar]
- Gorbatenko, S. World Heritage—The Historical Landscape of the Saint Petersburg Agglomeration; Documentation; ICOMOS St. Petersburg Regional Branch: Saint Petersburg, Russia, 2011; 115p. [Google Scholar]
- Isachenko, G.A. “Window to Europe”: History and Landscapes; Saint Petersburg State University: St. Petersburg, Russia, 1998; 476p. [Google Scholar]
- Pestryakov, V.K. (Ed.) Soils of Leningrad Oblast; Leningraddat: Leningrad, Russia, 1973; 344p. [Google Scholar]
- Zmitrovich, I.V. Middle taiga of Karelian isthmus: Zonal, intrazonal and extrazonal phenomena. Vestn. Ekol. Lesoved. Landshaftoved. 2011, 12, 54–76. [Google Scholar]
- Kovalev, D.N.; Noskov, G.A.; Noskova, M.G.; Popov, I.Y.; Rymkevich, T.A. A developmental concept for regional networks of protected areas as applied to Saint Petersburg and Leningrad oblast. Part I: Ecological aspects. Biosfera 2012, 4, 427–462. [Google Scholar]
- Chertov, O.G. Ecology of Forest Lands; Botanical Institute Named after V.L. Komarov: Leningrad, Russia, 1981; 192p. [Google Scholar]
- Aparin, B.F.; Kasatkina, G.A.; Matinan, N.N.; Sukhacheva, E.Y. Red Soil Data Book of Leningrad Region; Aeroplan: Saint-Petersburg, Russia, 2007; 320p. [Google Scholar]
- Rehman, Z.U.; Khalid, U.; Ijaz, N.; Mujtaba, H.; Haider, A.; Farooq, K.; Ijaz, Z. Machine learning-based intelligent modeling of hydraulic conductivity of sandy soils considering a wide range of grain sizes. Eng. Geol. 2022, 311, 106899. [Google Scholar] [CrossRef]
- Kuznetsova, A.I.; Geraskina, A.P.; Lukina, N.V.; Smirnov, V.E.; Tikhonova, E.V.; Shevchenko, N.E.; Gornov, A.V.; Ruchinskaya, E.V.; Tebenkova, D.N. Linking Vegetation, Soil Carbon Stocks, and Earthworms in Upland Coniferous–Broadleaf Forests. Forests 2021, 12, 1179. [Google Scholar] [CrossRef]
- Lukina, N.V. (Ed.) Carbon Accumulation in Forest Soils and Successional Status of Forests; Partnership of Scientific Publications KMK: Moscow, Russia, 2018; 232p. [Google Scholar]
- Kurganova, I.N.; Semenov, V.M.; Kudeyarov, V.N. Climate and land use as key factors of the stability of organic matter in soils. Dokl. Biol. Sci. 2019, 489, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Semenov, V.M.; Kogut, B.M.; Zinyakova, N.B.; Masyutenko, N.P.; Malyukova, L.S.; Lebedeva, T.N.; Tulina, A.S. Biologically Active Organic Matter in Soils of European Russia. Eurasian Soil Sci. 2018, 51, 434–447. [Google Scholar] [CrossRef]
- Semenov, V.M.; Tulina, A.S. Comparative characteristics of mineralizable organic matter pool in soils of natural and agricultural ecosystems. Agrochimia 2011, 12, 53–63. [Google Scholar]
- Mau, R.L.; Liu, C.M.; Aziz, M.; Schwartz, E.; Dijkstra, P.; Marks, J.C.; Price, L.B.; Keim, P.; Hungate, B.A. Linking soil bacterial biodiversity and soil carbon stability. ISME J. 2014, 9, 1477–1480. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Cui, H.; Fu, C.; Li, R.; Qi, F.; Liu, Z.; Yang, G.; Xiao, K.; Qiao, M. Unveiling the crucial role of soil microorganisms in carbon cycling: A review. Sci. Total Environ. 2024, 909, 168627. [Google Scholar] [CrossRef] [PubMed]
- Xue, P.; Minasny, B.; McBratney, A.; Pino, V.; Fajardo, M.; Luo, Y. Distribution of soil bacteria involved in C cycling across extensive environmental and pedogenic gradients. Eur. J. Soil Sci. 2023, 74, e13337. [Google Scholar] [CrossRef]
- Trifonova, T.; Kosmacheva, A.; Sprygin, A.; Chesnokova, S.; Byadovskaya, O. Enzymatic Activity and Microbial Diversity of Sod-Podzolic Soil Microbiota Using 16S rRNA Amplicon Sequencing following Antibiotic Exposure. Antibiotics 2021, 10, 970. [Google Scholar] [CrossRef]
- Manucharova, N.A.; Pozdnyakov, L.A.; Vlasova, A.P.; Yanovich, A.S.; Ksenofontova, N.A.; Kovalenko, M.A.; Stepanov, P.Y.; Gennadiev, A.N.; Golovchenko, A.V.; Stepanov, A.L. Metabolically Active Prokaryotic Complex in Grassland and Forests’ Sod-Podzol under Polycyclic Aromatic Hydrocarbon Influence. Forests 2021, 12, 1103. [Google Scholar] [CrossRef]
- Evdokimova, E.V.; Gladkov, G.V.; Kuzina, N.I.; Ivanova, E.A.; Kimeklis, A.K.; Zverev, A.O.; Kichko, A.A.; Aksenova, T.S.; Pinaev, A.G.; Andronov, E.E. The difference between cellulolytic ‘culturomes’ and microbiomes inhabiting two contrasting soil types. PLoS ONE 2020, 15, e0242060. [Google Scholar] [CrossRef]
- Bünger, W.; Jiang, X.; Müller, J.; Hurek, T.; Reinhold-Hurek, B. Novel cultivated endophytic Verrucomicrobia reveal deep-rooting traits of bacteria to associate with plants. Sci. Rep. 2020, 10, 8692. [Google Scholar] [CrossRef] [PubMed]
- Tsitko, I.; Lusa, M.; Lehto, J.; Parviainen, L.; Ikonen, A.T.K.; Lahdenperä, A.-M.; Bomberg, M. The Variation of Microbial Communities in a Depth Profile of an Acidic, Nutrient-Poor Boreal Bog in Southwestern Finland. Open J. Ecol. 2014, 4, 832–859. [Google Scholar] [CrossRef]
- Christiansen, N.A.; Green, T.J.; Fryirs, K.A.; Hose, G.C. Bacterial communities in peat swamps reflect changes associated with catchment urbanisation. Urban Ecosyst. 2022, 25, 1455–1468. [Google Scholar] [CrossRef]
- Rakitin, A.L.; Begmatov, S.; Beletsky, A.V.; Philippov, D.A.; Kadnikov, V.V.; Mardanov, A.V.; Dedysh, S.N.; Ravin, N.V. Highly Distinct Microbial Communities in Elevated Strings and Submerged Flarks in the Boreal Aapa-Type Mire. Microorganisms 2022, 10, 170. [Google Scholar] [CrossRef]
- Aksenov, A.S.; Shirokova, L.S.; Kisil, O.Y.; Kolesova, S.N.; Lim, A.G.; Kuzmina, D.; Pouillé, S.; Alexis, M.A.; Castrec-Rouelle, M.; Loiko, S.V.; et al. Bacterial Number and Genetic Diversity in a Permafrost Peatland (Western Siberia): Testing a Link with Organic Matter Quality and Elementary Composition of a Peat Soil Profile. Diversity 2021, 13, 328. [Google Scholar] [CrossRef]
- Serkebaeva, Y.M.; Kim, Y.; Liesack, W.; Dedysh, S.N. Pyrosequencing-based assessment of the bacteria diversity in surface and subsurface peat layers of a northern wetland, with focus on poorly studied phyla and candidate divisions. PLoS ONE 2013, 8, e63994. [Google Scholar] [CrossRef]
- Kulichevskaya, I.S.; Pankratov, T.A.; Dedysh, S.N. Detection of representatives of the Planctomycetes in Sphagnum peat bogs by molecular and cultivation approaches. Microbiology 2006, 75, 329–335. [Google Scholar] [CrossRef]
- Feng, H.; Guo, J.; Peng, C.; Ma, X.; Kneeshaw, D.; Chen, H.; Liu, Q.; Liu, M.; Hu, C.; Wang, W. Global estimates of forest soil methane flux identify a temperate and tropical forest methane sink. Geoderma 2023, 429, 116239. [Google Scholar] [CrossRef]
- Bräuer, S.L.; Basiliko, N.; Siljanen, H.M.P.; Zinder, S.H. Methanogenic archaea in peatlands. FEMS Microbiol. Lett. 2020, 367, fnaa172. [Google Scholar] [CrossRef]
- Shcherbakova, V.; Yoshimura, Y.; Ryzhmanova, Y.; Taguchi, Y.; Segawa, T.; Oshurkova, V.; Rivkina, E. Archaeal communities of Arctic methane-containing permafrost. FEMS Microbiol. Ecol. 2016, 92, fiw135. [Google Scholar] [CrossRef] [PubMed]
- Juottonen, H. Archaea, Bacteria and Methane Production along Environmental Gradients in Fens and Bogs. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2008. [Google Scholar]
- Korzhenkov, A.A.; Toshchakov, S.V.; Bargiela, R.; Gibbard, H.; Ferrer, M.; Teplyuk, A.V.; Jones, D.L.; Kublanov, I.V.; Golyshin, P.N.; Golyshina, O.V. Archaea dominate the microbial community in an ecosystem with low-to-moderate temperature and extreme acidity. Microbiome 2019, 7, 11. [Google Scholar] [CrossRef] [PubMed]


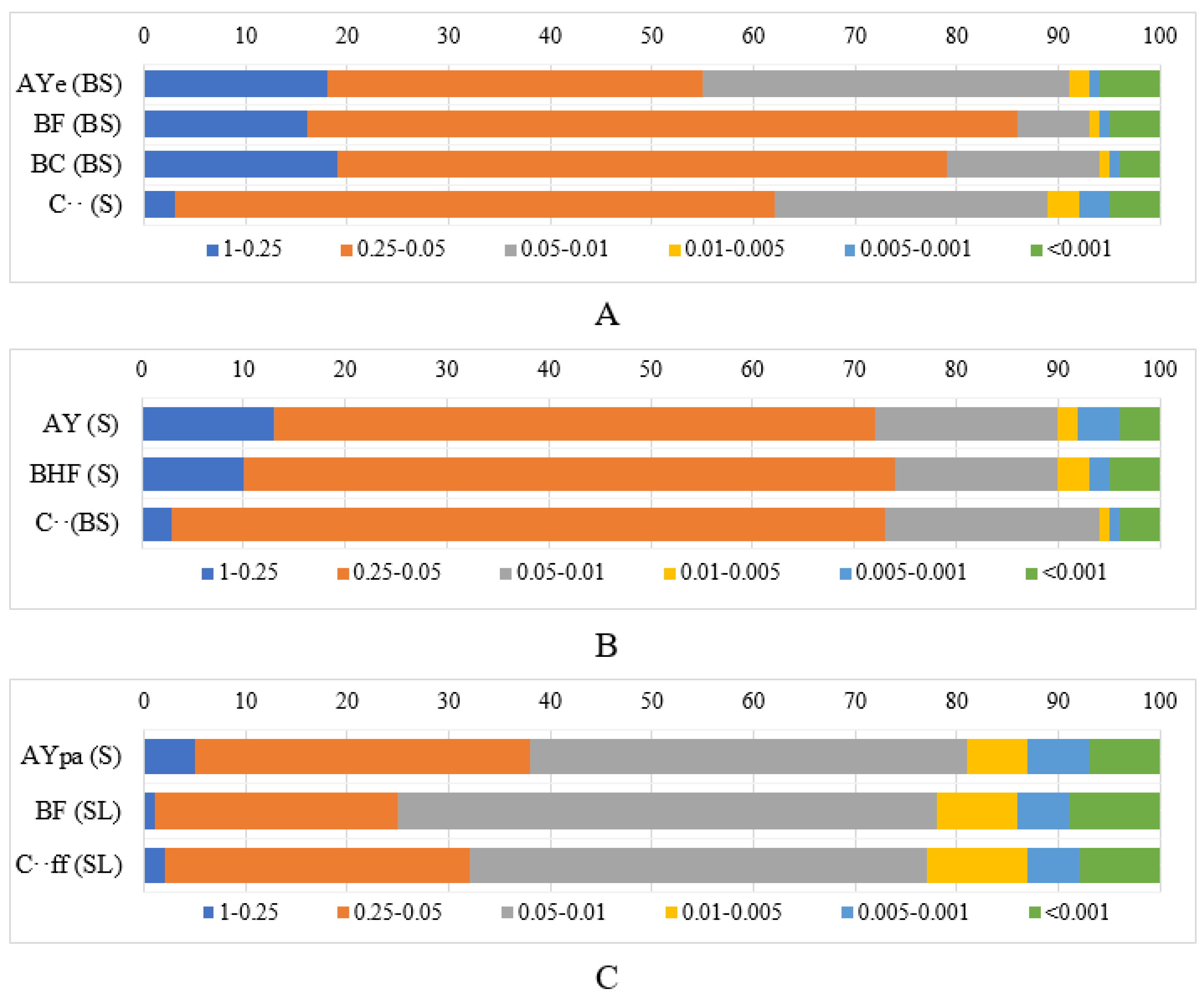

| № | Horizon | Thickness, cm | pHwater | pHsalt | Bulk Density, g × cm−3 | SOC, % | Soil Photo |
|---|---|---|---|---|---|---|---|
| Podzolized sod-podbur on fluvioglacial sediments (Entic Podzol) | |||||||
| CP1 | O | 0–2 | 5.9 | 5.2 | 0.8 | 7.54 |  |
| AYe | 2–13 | 5.6 | 4.5 | 0.9 | 3.33 | ||
| BF | 13–34 | 5.9 | 4.7 | 1.5 | 0.62 | ||
| BC | 34–45 | 5.9 | 5.3 | 1.4 | 0.19 | ||
| C·· | 45–… | 6.0 | 4.9 | 1.6 | 0.17 | ||
| Sod-podbur on fluvioglacial sediments (Entic Podzol) | |||||||
| CP9 | O | 0–2 | - | - | 0.3 | 16.65 |  |
| AY | 2–15 | 4.7 | 4.1 | 1.0 | 2.25 | ||
| BHF | 15–27 | 5.0 | 4.2 | 1.3 | 1.15 | ||
| C·· | 27–… | 4.9 | 4.4 | 1.1 | 0.56 | ||
| № | Horizon | Thickness, cm | pHwater | pHsalt | Bulk Density, g × cm−3 | SOC, % | Soil Photo |
|---|---|---|---|---|---|---|---|
| Medium plowed post-agricultural illuvial-iron agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP3 | O | 0–1 | 5.6 | 5.4 | 0.3 | 16.15 |  |
| O/AO | 1–8 | 5.0 | 4.3 | 0.3 | 5.08 | ||
| AYpa | 8–30 | 4.4 | 4.2 | 1.4 | 1.22 | ||
| BFff | 30–50 | 4.8 | 4.5 | 1.2 | 0.59 | ||
| BCff | 50–80 | 5.3 | 4.5 | 1.2 | 0.45 | ||
| C··g,ff | 80–100 | 4.8 | 4.4 | 1.3 | 0.54 | ||
| Gray humus postpyrogenic soil on compacted sandy loam (Anthrosol) | |||||||
| CP4 | O | 0–2 | 5.2 | 5.0 | 0.4 | 16.63 |  |
| AYpyr | 2–30 | 4.2 | 3.7 | 1.0 | 2.96 | ||
| RYR | 30–70 | 4.4 | 3.8 | 0.9 | 3.95 | ||
| C··ff | 70–100 | 4.4 | 4.1 | 1.0 | 1.46 | ||
| Small plowed postagrogenic illuvial-iron agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP8 | O | 0–2 | - | - | 0.5 | 20.96 |  |
| AYpa | 2–10 | 5.3 | 4.6 | 0.7 | 2.84 | ||
| BF | 10–15 | 5.2 | 4.3 | 1.1 | 0.47 | ||
| BC | 15–35 | 5.2 | 4.5 | 1.2 | 0.36 | ||
| BCff | 35–70 | 5.2 | 4.7 | 1.3 | 1.11 | ||
| C·· | 70–80 | 5.0 | 4.6 | 1.1 | 0.31 | ||
| Medium plowed illuvial-iron postagrogenic agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP12 | AYpa | 0–27 | 4.6 | 4.1 | 0.9 | 1.78 |  |
| BF | 27–50 | 4.9 | 4.2 | 1.1 | 0.72 | ||
| C··ff | 50–85 | 4.7 | 4.3 | 1.4 | 0.60 | ||
| Medium plowed illuvial-iron postagrogenic agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP10 | O | 0–1 | - | - | 0.2 | 41.77 |  |
| AYpa | 1–24 | 3.9 | 3.8 | 1.1 | 4.33 | ||
| BF | 24–45 | 4.8 | 4.3 | 1.2 | 0.84 | ||
| BH | 45–55 | 4.6 | 4.3 | 1.2 | 0.95 | ||
| BCff | 55–70 | 4.5 | 4.3 | 1.3 | 0.47 | ||
| C··ff | 70–… | 4.9 | 4.4 | 1.4 | 0.37 | ||
| № | Horizon | Thickness, cm | pHwater | pHsalt | Bulk Density, g × cm−3 | SOC, % | Soil Photo |
|---|---|---|---|---|---|---|---|
| Deep plowed post-agricultural illuvial-iron agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP5 | O | 0–1 | - | - | 0.15 | 16.82 |  |
| AYpa | 1–40 | 4.9 | 4.3 | 1.2 | 1.93 | ||
| BFff | 40–60 | 5.0 | 4.5 | 1.3 | 0.60 | ||
| C··g,ff | 60–75 | 5.1 | 4.6 | 1.2 | 0.53 | ||
| Medium plowed illuvial-iron postagrogenic agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP13 | AYpa | 0–30 | 4.8 | 4.0 | 1.1 | 1.72 |  |
| BF | 30–60 | 5.0 | 4.5 | 1.1 | 0.64 | ||
| C·· | 60–80 | 5.3 | 4.6 | 1.4 | 0.51 | ||
| Deep plowed post-agricultural illuvial-iron agrozem on fluvioglacial sediments (Plaggic Podzol (Arenic)) | |||||||
| CP14 | AYpa | 0–45 | 4.6 | 3.9 | 1.3 | 2.03 |  |
| BF | 45–65 | 4.9 | 4.3 | 1.0 | 1.04 | ||
| C··ff | 65–85 | 5.1 | 3.8 | 1.3 | 0.51 | ||
| № | Horizon | Thickness, cm | pHwater | pHsalt | Bulk Density, g × cm−3 | SOC, % | Soil Photo |
|---|---|---|---|---|---|---|---|
| Peat-perennial eutrophic soil (Histosol) | |||||||
| CP2 | TE1 | 0–10 | 5.2 | 4.9 | 0.2 | 38.40 |  |
| TE2 | 10–20 | 5.3 | 4.6 | 0.3 | 41.96 | ||
| TE3 | 20–30 | 4.7 | 4.6 | 0.2 | 46.35 | ||
| TE4 | 30–50 | 4.1 | 3.5 | 0.2 | 43.81 | ||
| TT | 50–… | 3.7 | 3.3 | 0.2 | 40.49 | ||
| Peat-perennial eutrophic soil (Histosol) | |||||||
| CP6 | TE1 | 0–15 | 4.4 | 3.9 | 0.1 | 36.54 |  |
| TE2 | 15–30 | 4.1 | 3.8 | 0.15 | 44.51 | ||
| Peat-perennial eutrophic soil (Histosol) | |||||||
| CP7 | TE1 | 0–15 | 4.7 | 4.3 | 0.1 | 41.29 |  |
| TE2 | 15–30 | 4.8 | 4.3 | 0.15 | 46.19 | ||
| CP15 | TE1 | 0–20 | 4.8 | 3.8 | 0.2 | 48.82 |  |
| TE2 | 20–40 | 5.1 | 4.0 | 0.1 | 51.54 | ||
| TE3 | 40–50 | 4.9 | 3.9 | 0.1 | 48.94 | ||
| TT | 50–80 | 5.1 | 4.0 | 0.1 | 51.44 | ||
| № | Horizon | Thickness, cm | pHwater | pHsalt | Bulk Density, g × cm−3 | SOC, % | Soil Photo |
|---|---|---|---|---|---|---|---|
| Dark humus gley soil (Histic Gleysol) | |||||||
| CP11 | AU1 | 0–20 | 4.7 | 4.0 | 0.7 | 5.62 |  |
| AU2 | 20–40 | 4.9 | 4.1 | 0.8 | 6.57 | ||
| AUg1 | 40–60 | 4.7 | 4.4 | 0.8 | 4.66 | ||
| AUg2 | 60–80 | 5.0 | 4.4 | 0.3 | 6.18 | ||
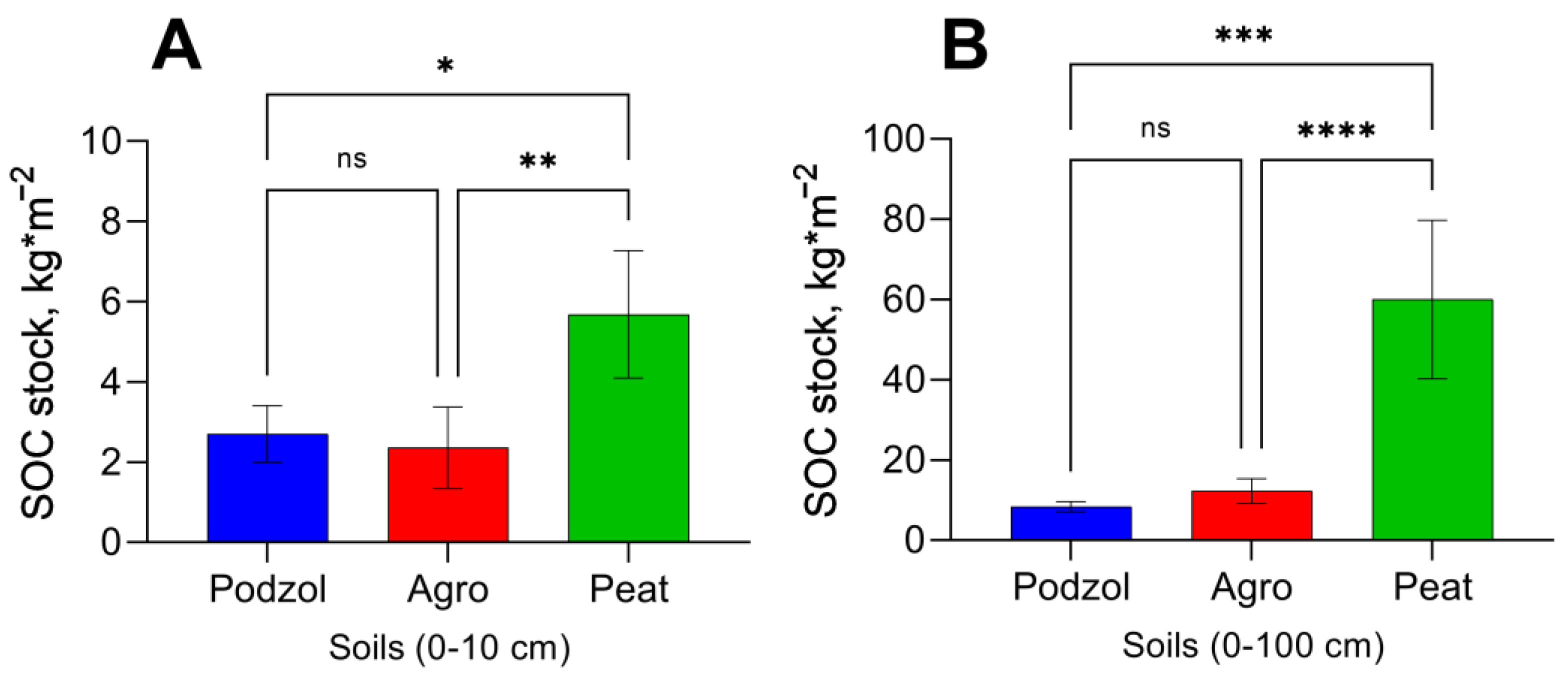
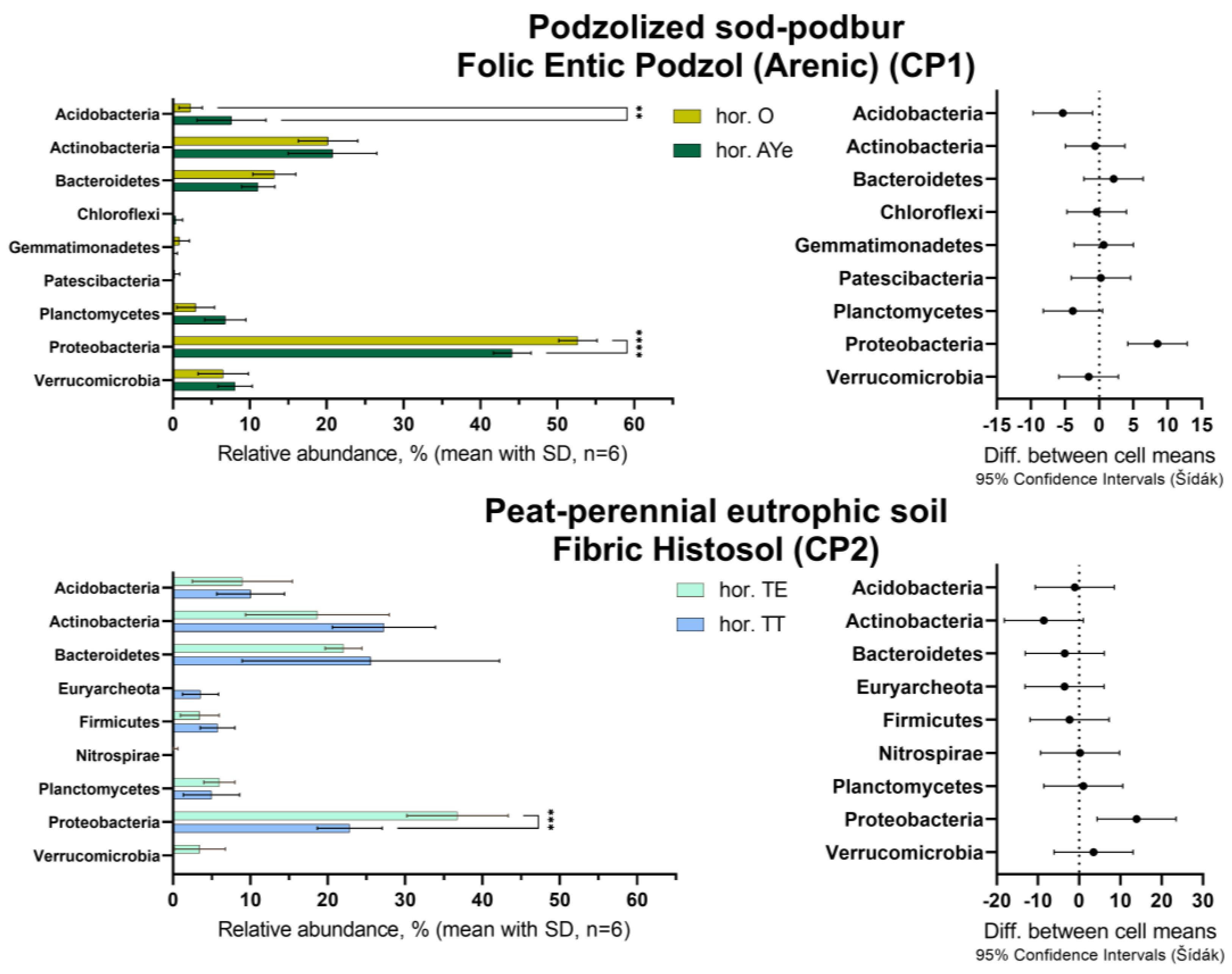
| Parameter | Podzolized Sod-Podbur (n = 3) | Peat-Perennial Eutrophic Soil (n = 3) |
|---|---|---|
| SOC, mg × 100 g−1 | 3330 | 38400 |
| Cpm, mg × 100 g−1 | 502.5 ± 104.6 | 3394.7 ± 380.3 |
| Cpm, % of SOC | 15.1 ± 3.1 | 8.8 ± 0.9 |
| Mineralization constant (k), day−1 | 0.005 ± 0.001 | 0.003 ± 0.001 |
| Mineralization intensity (IM), mg × 100 g−1 × day−1 | 2.5 ± 0.2 | 9.2 ± 0.1 |
| Stability Index (SI) | 5.8 ± 1.3 | 10.4 ± 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abakumov, E.; Nizamutdinov, T.; Zhemchueva, D.; Suleymanov, A.; Shevchenko, E.; Koptseva, E.; Kimeklis, A.; Polyakov, V.; Novikova, E.; Gladkov, G.; et al. The Characterization of Biodiversity and Soil Emission Activity of the “Ladoga” Carbon-Monitoring Site. Atmosphere 2024, 15, 420. https://doi.org/10.3390/atmos15040420
Abakumov E, Nizamutdinov T, Zhemchueva D, Suleymanov A, Shevchenko E, Koptseva E, Kimeklis A, Polyakov V, Novikova E, Gladkov G, et al. The Characterization of Biodiversity and Soil Emission Activity of the “Ladoga” Carbon-Monitoring Site. Atmosphere. 2024; 15(4):420. https://doi.org/10.3390/atmos15040420
Chicago/Turabian StyleAbakumov, Evgeny, Timur Nizamutdinov, Darya Zhemchueva, Azamat Suleymanov, Evgeny Shevchenko, Elena Koptseva, Anastasiia Kimeklis, Vyacheslav Polyakov, Evgenia Novikova, Grigory Gladkov, and et al. 2024. "The Characterization of Biodiversity and Soil Emission Activity of the “Ladoga” Carbon-Monitoring Site" Atmosphere 15, no. 4: 420. https://doi.org/10.3390/atmos15040420
APA StyleAbakumov, E., Nizamutdinov, T., Zhemchueva, D., Suleymanov, A., Shevchenko, E., Koptseva, E., Kimeklis, A., Polyakov, V., Novikova, E., Gladkov, G., & Andronov, E. (2024). The Characterization of Biodiversity and Soil Emission Activity of the “Ladoga” Carbon-Monitoring Site. Atmosphere, 15(4), 420. https://doi.org/10.3390/atmos15040420








