First Long-Time Airborne Fungal Spores Study in Dublin, Ireland (1978–1980)
Abstract
1. Introduction
2. Materials and Methods
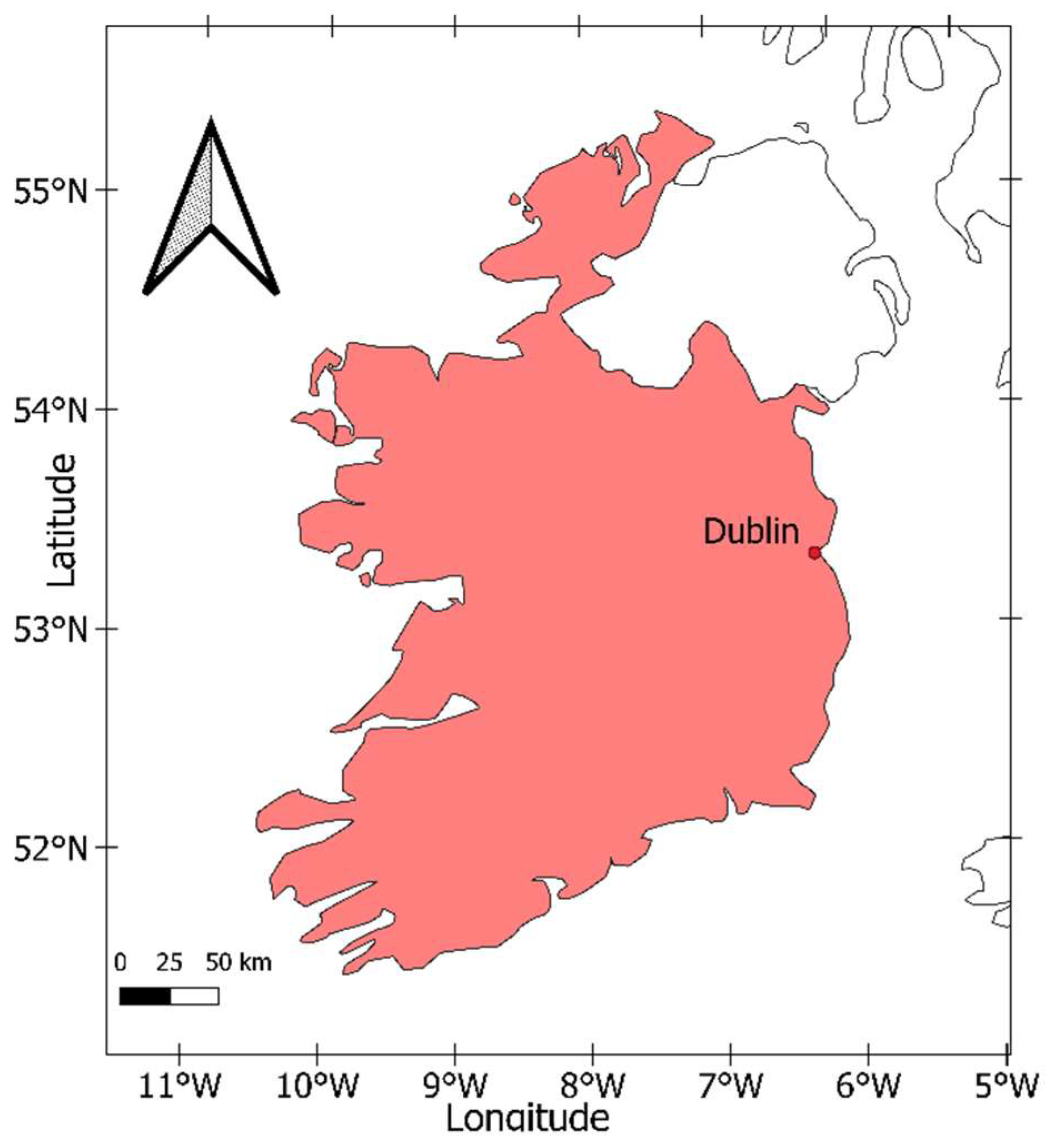
2.1. Study Location
2.2. Spore Monitoring Periods and Spore Identification
2.3. Meteorological Data
2.4. Statistical Analyses
2.4.1. Meteorological Analysis
2.4.2. Fungal Spore Fructification Period Chart
2.4.3. Geographical Origin of Airborne Spores
3. Results
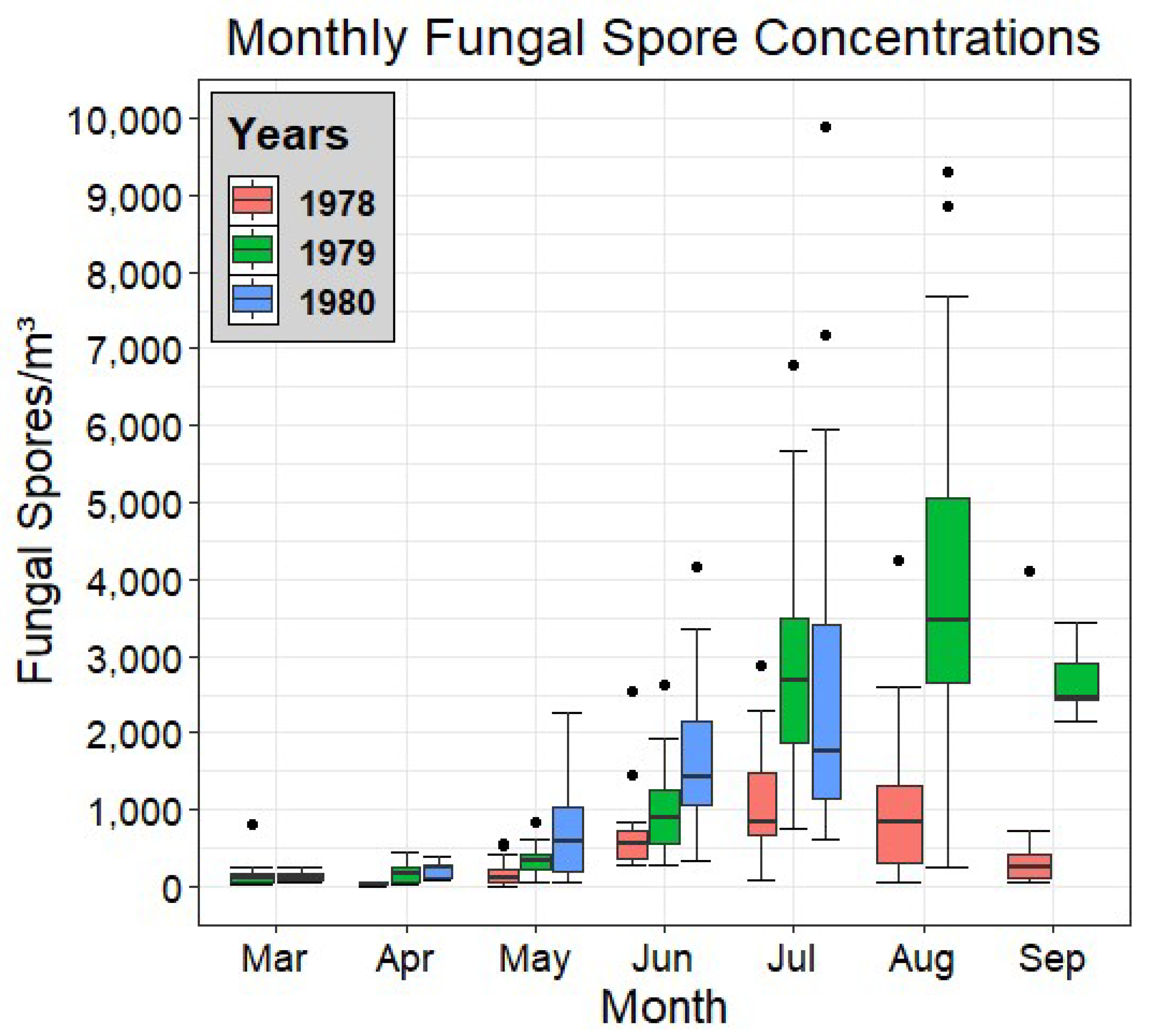
3.1. Distribution and Analysis of Airborne Fungal Spore Content
3.2. Meteorological Analysis
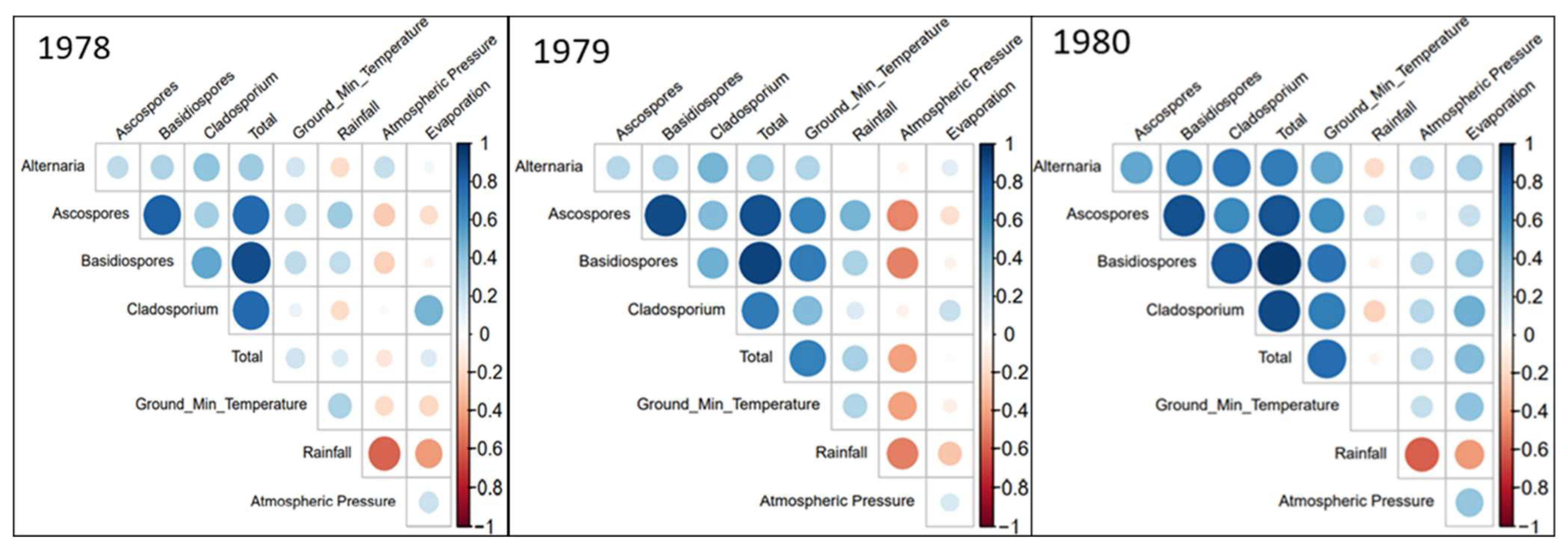
3.2.1. Correlation Coefficient Analysis
3.2.2. Multivariate Regression Tree (MRT) Analysis
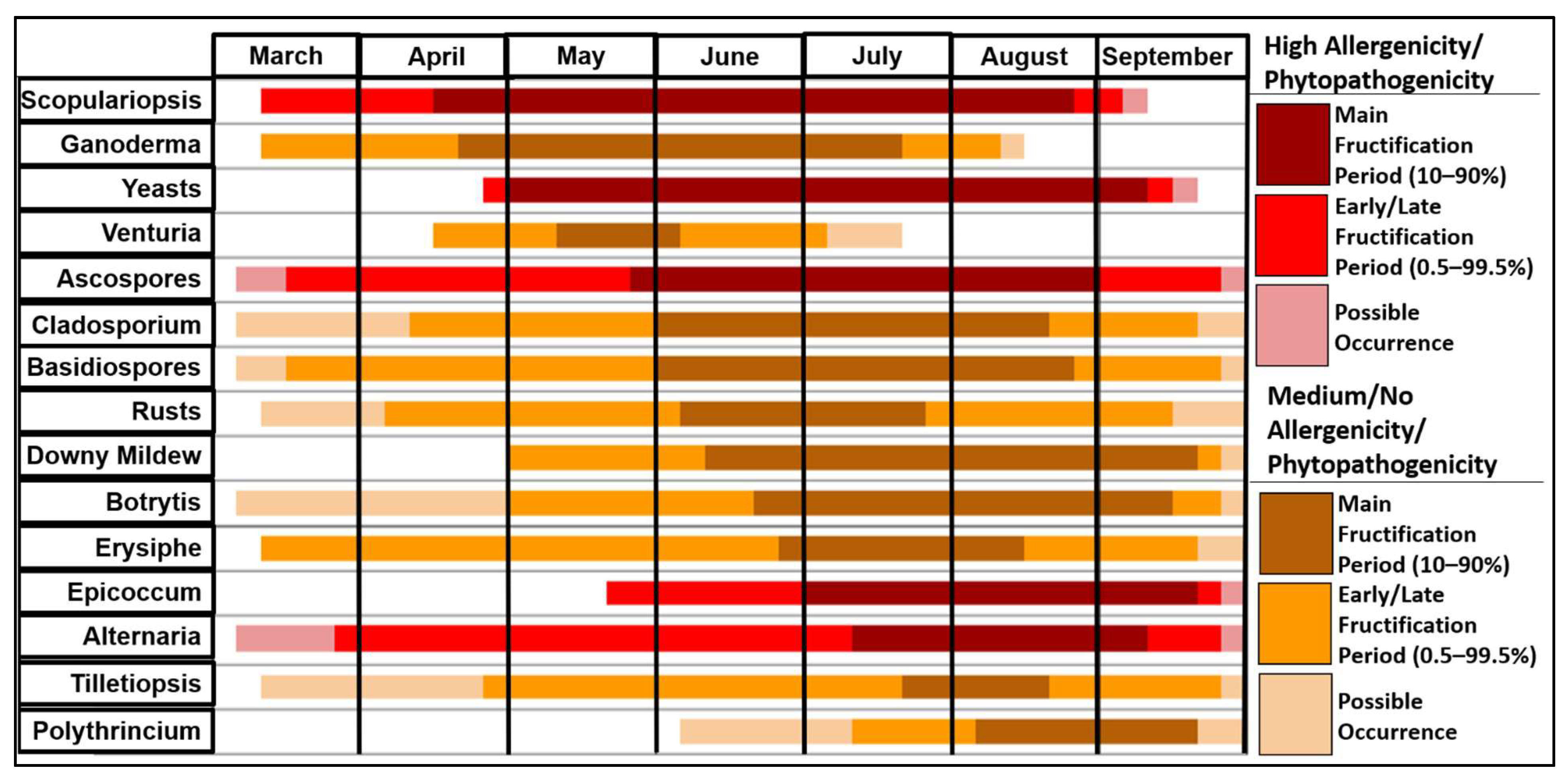
3.3. Fungal Spore Fructification Period Chart
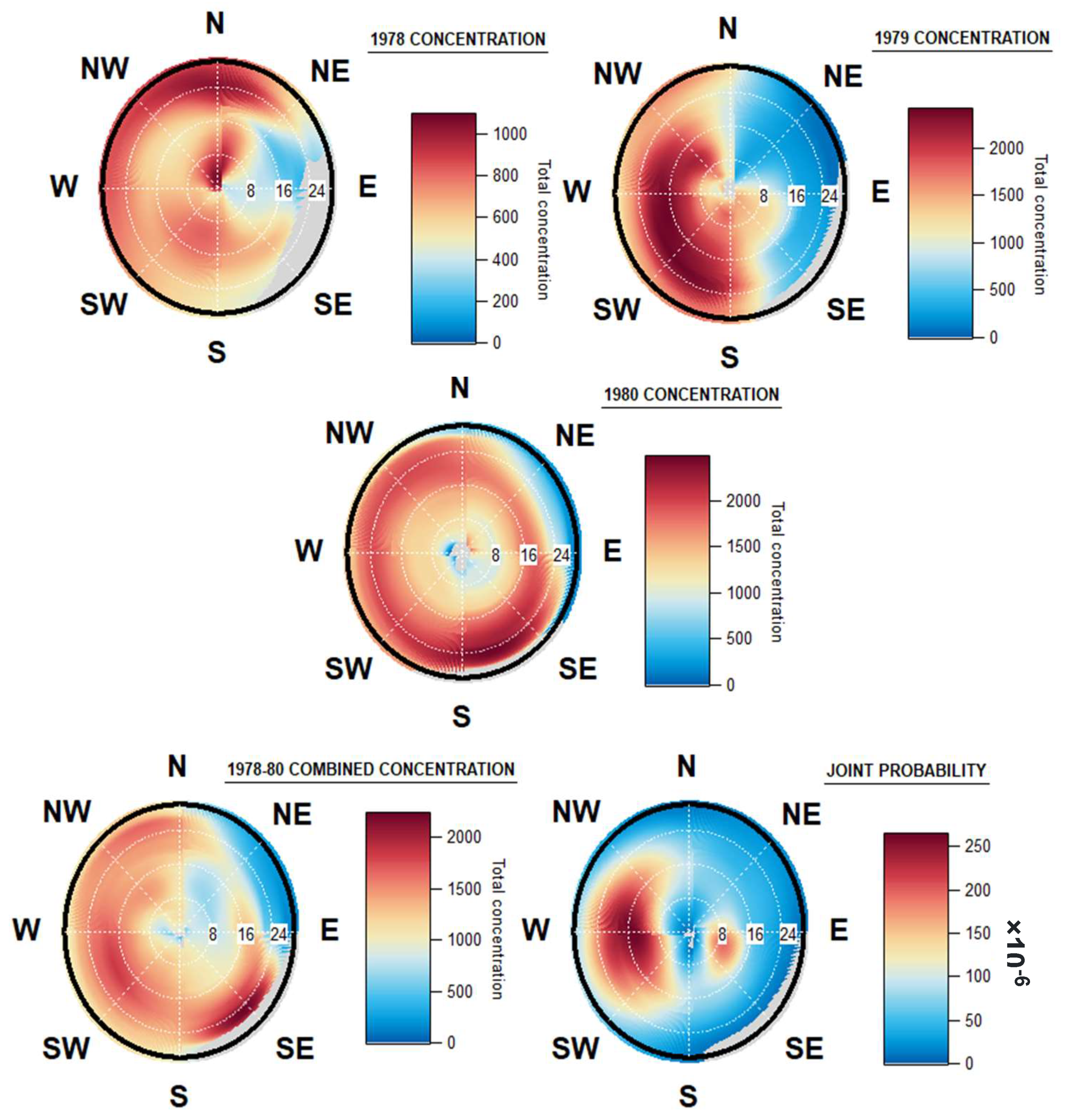
3.4. Geographical Origin of Airborne Spores
4. Discussion
4.1. Airborne Fungal Spore Monitoring
4.2. Meteorololy and Fungal Spore Concentrations
4.3. Geographical Origin of Airborne Spores
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Troutt, C.; Levetin, E. Correlation of Spring Spore Concentrations and Meteorological Conditions in Tulsa, Oklahoma. Int. J. Biometeorol. 2001, 45, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Burch, M.; Levetin, E. Effects of Meteorological Conditions on Spore Plumes. Int. J. Biometeorol. 2002, 46, 107–117. [Google Scholar] [PubMed]
- D’amato, G.; Chatzigeorgiou, G.; Corsico, R.; Gioulekas, D.; Jäger, L.; Jäger, S.; Kontou-Fili, K.; Kouridakis, S.; Liccardi, G.; Meriggi, A. Evaluation of the Prevalence of Skin Prick Test Positivity to Alternaria and Cladosporium in Patients with Suspected Respiratory Allergy: A European Multicenter Study Promoted by the Subcommittee on Aerobiology and Environmental Aspects of Inhalant Allergens of the European Academy of Allergology and Clinical Immunology. Allergy 1997, 52, 711–716. [Google Scholar] [PubMed]
- Rapiejko, P.; Lipiec, A.; Wojdas, A.; Jurkiewicz, D. Threshold Pollen Concentration Necessary to Evoke Allergic Symptoms. Int. Rev. Allergol. Clin. Immunol. 2004, 10, 91–94. [Google Scholar]
- Gostic, K.; Gomez, A.C.; Mummah, R.O.; Kucharski, A.J.; Lloyd-Smith, J.O. Estimated Effectiveness of Symptom and Risk Screening to Prevent the Spread of COVID-19. Elife 2020, 9, e55570. [Google Scholar] [CrossRef]
- Reich, J.; Chatterton, S.; Johnson, D. Temporal Dynamics of Botrytis Cinerea and Sclerotinia Sclerotiorum in Seed Alfalfa Fields of Southern Alberta, Canada. Plant Dis. 2017, 101, 331–343. [Google Scholar] [CrossRef]
- Coker, T.L.; Rozsypálek, J.; Edwards, A.; Harwood, T.P.; Butfoy, L.; Buggs, R.J. Estimating Mortality Rates of European Ash (Fraxinus Excelsior) under the Ash Dieback (Hymenoscyphus Fraxineus) Epidemic. Plants People Planet 2019, 1, 48–58. [Google Scholar] [CrossRef]
- O’Connor, D.J.; Sadyś, M.; Skjøth, C.A.; Healy, D.A.; Kennedy, R.; Sodeau, J.R. Atmospheric Concentrations of Alternaria, Cladosporium, Ganoderma and Didymella Spores Monitored in Cork (Ireland) and Worcester (England) during the Summer of 2010. Aerobiologia 2014, 30, 397–411. [Google Scholar] [CrossRef]
- Sadyś, M.; Strzelczak, A.; Grinn-Gofroń, A.; Kennedy, R. Application of Redundancy Analysis for Aerobiological Data. Int. J. Biometeorol. 2015, 59, 25–36. [Google Scholar] [CrossRef]
- Corden, J.M.; Millington, W.M. The Long-Term Trends and Seasonal Variation of the Aeroallergen Alternaria in Derby, UK. Aerobiologia 2001, 17, 127–136. [Google Scholar] [CrossRef]
- Skjøth, C.A.; Sommer, J.; Frederiksen, L.; Karlson, U.G. Crop Harvest in Denmark and Central Europe Contributes to the Local Load of Airborne Alternaria Spore Concentrations in Copenhagen. Atmos. Chem. Phys. 2012, 12, 11107–11123. [Google Scholar] [CrossRef]
- Stepalska, D.; Wolek, J. The Estimation of Fungal Spore Concentrations Using Two Counting Methods. Acta Agrobot. 2009, 62, 117–123. [Google Scholar] [CrossRef]
- Oliveira, M.; Guerner-Moreira, J.; Mesquita, M.M.; Abreu, I. Important Phytopathogenic Airborne Fungal Spores in a Rural Area: Incidence of Botrytis Cinerea and Oidium Spp. Ann. Agric. Environ. Med. 2009, 16, 197–204. [Google Scholar] [PubMed]
- Rodríguez-Rajo, F.J.; Iglesias, I.; Jato, V. Variation Assessment of Airborne Alternaria and Cladosporium Spores at Different Bioclimatical Conditions. Mycol. Res. 2005, 109, 497–507. [Google Scholar] [CrossRef]
- Antón, S.F.; de la Cruz, D.R.; Sánchez, J.S.; Sánchez Reyes, E. Analysis of the Airborne Fungal Spores Present in the Atmosphere of Salamanca (MW Spain): A Preliminary Survey. Aerobiologia 2019, 35, 447–462. [Google Scholar] [CrossRef]
- Jesús Aira, M.; Rodríguez-Rajo, F.-J.; Fernández-González, M.; Seijo, C.; Elvira-Rendueles, B.; Gutiérrez-Bustillo, M.; Abreu, I.; Pérez-Sánchez, E.; Oliveira, M.; Recio, M. Cladosporium Airborne Spore Incidence in the Environmental Quality of the Iberian Peninsula. Grana 2012, 51, 293–304. [Google Scholar] [CrossRef]
- Ricci, S.; Bruni, M.; Meriggi, A.; Corsico, R. Aerobiological Monitoring OfAlternaría Fungal Spores: A Comparison between Surveys in 1992 and 1993 and Local Meteorological Conditions. Aerobiologia 1995, 11, 195–199. [Google Scholar] [CrossRef]
- Şakiyan, N.; Inceoğlu, Ö. Atmospheric Concentrations of Cladosporium Link and Alternaria Nées Spores in Ankara and the Effects of Meteorological Factors. Turk. J. Bot. 2003, 27, 77–81. [Google Scholar]
- Grinn-Gofroń, A.; Çeter, T.; Pinar, N.M.; Bosiacka, B.; Çeter, S.; Keçeli, T.; Myśliwy, M.; Şahin, A.A.; Bogawski, P. Airborne Fungal Spore Load and Season Timing in the Central and Eastern Black Sea Region of Turkey Explained by Climate Conditions and Land Use. Agric. For. Meteorol. 2020, 295, 108191. [Google Scholar] [CrossRef]
- McDonald, M.; O’driscoll, B. Aerobiological Studies Based in Galway. A Comparison of Pollen and Spore Counts over Two Seasons of Widely Differing Weather Conditions. Clin. Exp. Allergy 1980, 10, 211–215. [Google Scholar] [CrossRef]
- Abu-Dieyeh, M.H.; Barham, R.; Abu-Elteen, K.; Al-Rashidi, R.; Shaheen, I. Seasonal Variation of Fungal Spore Populations in the Atmosphere of Zarqa Area, Jordan. Aerobiologia 2010, 26, 263–276. [Google Scholar] [CrossRef]
- Mitakakis, T.Z.; Guest, D.I. A Fungal Spore Calendar for the Atmosphere of Melbourne, Australia, for the Year 1993. Aerobiologia 2001, 17, 171–176. [Google Scholar] [CrossRef]
- Seasonal Fluctuations of Airborne Fungal Allergens. Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S0953756289800085?token=EE4F79D91959E292735FF077411133F8DB14A9F6A1B1350246C69BC2A518D76A870ADD72BD1564FC26F160E97C6FA0C4&originRegion=eu-west-1&originCreation=20220116152956 (accessed on 16 January 2022).
- Bednarz, A.; Pawlowska, S. A Fungal Spore Calendar for the Atmosphere of Szczecin, Poland. Acta Agrobot. 2016, 69, 1669. [Google Scholar] [CrossRef]
- Kasprzyk, I.; Rzepowska, B.; Wasylów, M. Fungal Spores in the Atmosphere of Rzeszów (South-East Poland). Ann. Agric. Environ. Med. 2004, 11, 6. [Google Scholar]
- Ščevková, J.; Kováč, J. First Fungal Spore Calendar for the Atmosphere of Bratislava, Slovakia. Aerobiologia 2019, 35, 343–356. [Google Scholar] [CrossRef]
- Reyes, E.S.; de la Cruz, D.R.; Sánchez, J.S. First Fungal Spore Calendar of the Middle-West of the Iberian Peninsula. Aerobiologia 2016, 32, 529–539. [Google Scholar] [CrossRef]
- Galan, C.; Ariatti, A.; Bonini, M.; Clot, B.; Crouzy, B.; Dahl, A.; Fernandez-González, D.; Frenguelli, G.; Gehrig, R.; Isard, S.; et al. Recommended Terminology for Aerobiological Studies. Aerobiologia 2017, 33, 293–295. [Google Scholar] [CrossRef]
- Grinn-Gofroń, A.; Bosiacka, B.; Bednarz, A.; Wolski, T. A Comparative Study of Hourly and Daily Relationships between Selected Meteorological Parameters and Airborne Fungal Spore Composition. Aerobiologia 2018, 34, 45–54. [Google Scholar] [CrossRef]
- Lacey, J. Reproduction: Patterns of Spore Production, Liberation and Dispersal; Cambridge University Press: Cambridge, UK, 1986; p. 65. [Google Scholar]
- Edmonds, R.L. Aerobiology: The Ecological Systems Approach; Dowden, Hutchinson and Ross Inc.: Stroudsburg, PA, USA, 1979. [Google Scholar]
- Kasprzyk, I. Aeromycology—Main Research Fields of Interest during the Last 25 Years. Ann. Agric. Environ. Med. 2008, 15, 1–7. [Google Scholar]
- Hirst, J.M. An Automatic Volumetric Spore Trap. Ann. Appl. Biol. 1952, 39, 257–265. [Google Scholar] [CrossRef]
- Past Weather Statements—Met Éireann—The Irish Meteorological Service. Available online: https://www.met.ie/climate/past-weather-statements (accessed on 25 November 2021).
- The Irish National Meteorological Service. Available online: https://www.met.ie/climate/available-data/daily-data (accessed on 31 December 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2021. [Google Scholar]
- Grinn-Gofroń, A.; Strzelczak, A. Changes in Concentration of Alternaria and Cladosporium Spores during Summer Storms. Int. J. Biometeorol. 2013, 57, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Zeileis, A.; Hothorn, T.; Hornik, K. Party with the Mob: Model-Based Recursive Partitioning in R (Tech. Rep.). Retrieved Novemb. 2012, 19. [Google Scholar]
- Rasmussen, A. The Effects of Climate Change on the Birch Pollen Season in Denmark. Aerobiologia 2002, 18, 253–265. [Google Scholar] [CrossRef]
- Adams-Groom, B.; Skjøth, C.; Selby, K.; Pashley, C.; Satchwell, J.; Head, K.; Ramsay, G. Regional Calendars and Seasonal Statistics for the United Kingdom’s Main Pollen Allergens. Allergy Eur. J. Allergy Clin. Immunol. 2020, 75, 1492–1494. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Rendueles, B.; Moreno, J.M.; Costa, I.; Bañón, D.; Martínez-García, M.J.; Moreno-Grau, S. Pollen Calendars of Cartagena, Lorca, and Murcia (Region of Murcia), Southeastern Iberian Peninsula: 2010–2017. Aerobiologia 2019, 35, 477–496. [Google Scholar] [CrossRef]
- Lo, F.; Bitz, C.M.; Battisti, D.S.; Hess, J.J. Pollen Calendars and Maps of Allergenic Pollen in North America. Aerobiologia 2019, 35, 613–633. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Bracero, M.; Alcázar, P.; Díaz de la Guardia, C.; González-Minero, F.J.; Ruiz, L.; Trigo Pérez, M.M.; Galán, C. Pollen Calendars: A Guide to Common Airborne Pollen in Andalusia. Aerobiologia 2015, 31, 549–557. [Google Scholar] [CrossRef]
- Petit, J.E.; Favez, O.; Albinet, A.; Canonaco, F. A User-Friendly Tool for Comprehensive Evaluation of the Geographical Origins of Atmospheric Pollution: Wind and Trajectory Analyses. Environ. Model. Softw. 2017, 88, 183–187. [Google Scholar] [CrossRef]
- Donnelly, A.; Misstear, B.; Broderick, B. Real Time Air Quality Forecasting Using Integrated Parametric and Non-Parametric Regression Techniques. Atmos. Environ. 2015, 103, 53–65. [Google Scholar] [CrossRef]
- Han, Q.; Meng, F.; Hu, T.; Chu, F. Non-Parametric Hybrid Models for Wind Speed Forecasting. Energy Convers. Manag. 2017, 148, 554–568. [Google Scholar] [CrossRef]
- Wang, H.; Reponen, T.; Adhikari, A.; Grinshpun, S.A. Contribution of Fungal Spores to Organic Carbon Aerosol in Indoor and Outdoor Environments in the Greater Cincinnati Area. Aerosol Air Qual. Res. 2013, 13, 1348–1355. [Google Scholar] [CrossRef]
- Henry, R.; Norris, G.A.; Vedantham, R.; Turner, J.R. Source Region Identification Using Kernel Smoothing. Environ. Sci. Technol. 2009, 43, 4090–4097. [Google Scholar] [CrossRef] [PubMed]
- Ajouray, N.; Bouziane, H.; Trigo Pérez, M.M.; Kadiri, M. Variation interannuelle des spores fongiques de Tétouan (Nord-Ouest du Maroc) et calendrier sporal. J. Mycol. Méd. 2016, 26, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Sarda-Estève, R.; Baisnée, D.; Guinot, B.; Sodeau, J.; O’Connor, D.; Belmonte, J.; Besancenot, J.-P.; Petit, J.-E.; Thibaudon, M.; Oliver, G.; et al. Variability and Geographical Origin of Five Years Airborne Fungal Spore Concentrations Measured at Saclay, France from 2014 to 2018. Remote Sens. 2019, 11, 1671. [Google Scholar] [CrossRef]
- Genera of Phytopathogenic Fungi: GOPHY 1—Science Direct. Available online: https://www.sciencedirect.com/science/article/pii/S0166061617300209 (accessed on 29 October 2021).
- Simon-Nobbe, B.; Denk, U.; Pöll, V.; Rid, R.; Breitenbach, M. The Spectrum of Fungal Allergy. Int. Arch. Allergy Immunol. 2008, 145, 58–86. [Google Scholar] [CrossRef]
- Martínez-Bracero, M.; González-Fernández, E.; Wójcik, M.; Alcázar, P.; Fernández-González, M.; Kasprzyk, I.; Rodríguez-Rajo, F.J.; Galán, C. Airborne Fungal Phytopathological Spore Assessment in Three European Vineyards from Different Bioclimatic Areas. Aerobiologia 2020, 36, 715–729. [Google Scholar] [CrossRef]
- Pecero-Casimiro, R.; Maya-Manzano, J.M.; Fernández-Rodríguez, S.; Tormo-Molina, R.; Silva-Palacios, I.; Monroy-Colín, A.; Gonzalo-Garijo, Á. Pollen Calendars and Regional Gradients as Information Tools in the Extremadura Pollen Monitoring Network (SW Spain). Aerobiologia 2020, 36, 731–748. [Google Scholar] [CrossRef]
- Olsen, Y.; Skjøth, C.A.; Hertel, O.; Rasmussen, K.; Sigsgaard, T.; Gosewinkel, U. Airborne Cladosporium and Alternaria Spore Concentrations through 26 Years in Copenhagen, Denmark. Aerobiologia 2020, 36, 141–157. [Google Scholar] [CrossRef]
- Apangu, G.P.; Frisk, C.A.; Adams-Groom, B.; Satchwell, J.; Pashley, C.H.; Skjøth, C.A. Air Mass Trajectories and Land Cover Map Reveal Cereals and Oilseed Rape as Major Local Sources of Alternaria Spores in the Midlands, UK. Atmos. Pollut. Res. 2020, 11, 1668–1679. [Google Scholar] [CrossRef]
- Reyes, E.S.; de la Cruz, D.R.; Merino, E.; Sánchez, J.S. Meteorological and Agricultural Effects on Airborne Alternaria and Cladosporium Spores and Clinical Aspects in Valladolid [Spain]. Ann. Agric. Environ. Med. 2009, 16, 53–61. [Google Scholar]
- Saari, S.; Mensah-Attipoe, J.; Reponen, T.; Veijalainen, A.M.; Salmela, A.; Pasanen, P.; Keskinen, J. Effects of Fungal Species, Cultivation Time, Growth Substrate, and Air Exposure Velocity on the Fluorescence Properties of Airborne Fungal Spores. Indoor Air 2015, 25, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Vélez-Pereira, A.M.; De Linares, C.; Canela, M.-A.; Belmonte, J. Logistic Regression Models for Predicting Daily Airborne Alternaria and Cladosporium Concentration Levels in Catalonia (NE Spain). Int. J. Biometeorol. 2019, 63, 1541–1553. [Google Scholar] [CrossRef] [PubMed]
- Grinn-Gofroń, A.; Nowosad, J.; Bosiacka, B.; Camacho, I.; Pashley, C.; Belmonte, J.; De Linares, C.; Ianovici, N.; Manzano, J.M.M.; Sadyś, M. Airborne Alternaria and Cladosporium Fungal Spores in Europe: Forecasting Possibilities and Relationships with Meteorological Parameters. Sci. Total Environ. 2019, 653, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Mallo, A.C.; Nitiu, D.S.; Gardella Sambeth, M.C. Airborne Fungal Spore Content in the Atmosphere of the City of La Plata, Argentina. Aerobiologia 2011, 27, 77–84. [Google Scholar] [CrossRef]
- Ingold, C. T Fungal Spores. Their Liberation and Dispersal; Oxford, Clarendon Press: Oxford, UK, 1971. [Google Scholar]
- Ianovici, N. Atmospheric Concentrations of Selected Allergenic Fungal Spores in Relation to Some Meteorological Factors, in Timişoara (Romania). Aerobiologia 2016, 32, 139–156. [Google Scholar] [CrossRef]
- Jarvis, W. Epidemiology. In The Biology of Botrytis; Coley-Smith, J.R., Verhoeff, K., Jarvis, W.R., Eds.; Academic Press Inc.: London, UK, 1980. [Google Scholar]
- Latorre, B.; Rioja, M. The Effect of Temperature and Relative Humidity on Conidial Germination of Botrytis Cinerea. Int. J. Agric. Nat. Resour. 2002, 29, 66–72. [Google Scholar] [CrossRef]
- Oteros, J.; García-Mozo, H.; Alcázar, P.; Belmonte, J.; Bermejo, D.; Boi, M.; Cariñanos, P.; de la Guardia, C.D.; Fernández-González, D.; González-Minero, F. A New Method for Determining the Sources of Airborne Particles. J. Environ. Manag. 2015, 155, 212–218. [Google Scholar] [CrossRef]
- Sarda-Estève, R.; Baisnée, D.; Guinot, B.; Petit, J.E.; Sodeau, J.; O’Connor, D.; Besancenot, J.P.; Thibaudon, M.; Gros, V. Temporal Variability and Geographical Origins of Airborne Pollen Grains Concentrations from 2015 to 2018 at Saclay, France. Remote Sens. 2018, 10, 1932. [Google Scholar] [CrossRef]
- Rojo, J.; Oteros, J.; Picornell, A.; Ruëff, F.; Werchan, B.; Werchan, M.; Bergmann, K.-C.; Schmidt-Weber, C.B.; Buters, J. Land-Use and Height of Pollen Sampling Affect Pollen Exposure in Munich, Germany. Atmosphere 2020, 11, 145. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, S.; Sadyś, M.; Smith, M.; Tormo-Molina, R.; Skjøth, C.A.; Maya-Manzano, J.M.; Silva-Palacios, I.; Gonzalo-Garijo, Á. Potential Sources of Airborne Alternaria spp. Spores in South-West Spain. Sci. Total Environ. 2015, 533, 165–176. [Google Scholar] [CrossRef]

| Year | Selected Parameter | Threshold | p-Value | N1 | N2 | |
|---|---|---|---|---|---|---|
| Alternaria | 1978 | |||||
| 1979 | Maximum temperature (°C) | 17.9 | 0.001 | 52 | 129 | |
| Maximum temperature (°C) | 15.4 | <0.001 | 44 | 85 | ||
| 1980 | Mean 10 cm soil temperature (°C) | 17.275 | 0.002 | 7 | 118 | |
| Maximum temperature (°C) | 15.3 | 0.006 | 79 | 39 | ||
| Ascospores | 1978 | Precipitation Amount (mm) | 0.9 | <0.001 | 35 | 105 |
| 1979 | 09utc Grass Minimum Temperature (°C) | 5.2 | <0.001 | 102 | 79 | |
| Mean 10 cm soil temperature (°C) | 10.550 | <0.001 | 21 | 58 | ||
| Precipitation Amount (mm) | 4 | <0.001 | 9 | 49 | ||
| Mean 10 cm soil temperature (°C) | 7.150 | 0.032 | 18 | 31 | ||
| Mean CBL pressure (hpa) | 1001.2 | <0.001 | 30 | 72 | ||
| 1980 | 09utc Grass Minimum Temperature (°C) | 4.5 | <0.001 | 62 | 33 | |
| Mean CBL pressure (hpa) | 999.2 | 0.041 | 26 | 7 | ||
| Basidiospores | 1978 | 09utc Grass Minimum Temperature (°C) | 10.7 | 0.002 | 31 | 109 |
| 1979 | Maximum temperature (°C) | 17 | <0.001 | 68 | 113 | |
| Mean 10 cm soil temperature (°C) | 17.775 | 0.029 | 12 | 101 | ||
| 1980 | 09utc Grass Minimum Temperature (°C) | 8.5 | <0.001 | 35 | 60 | |
| 09utc Grass Minimum Temperature (°C) | 3.9 | 0.033 | 33 | 27 | ||
| Botrytis | 1978 | |||||
| 1979 | Maximum temperature (°C) | 21.5 | <0.001 | 9 | 172 | |
| Mean 10 cm soil temperature (°C) | 15.700 | <0.001 | 59 | 113 | ||
| Mean 10 cm soil temperature (°C) | 13.625 | <0.001 | 32 | 81 | ||
| 1980 | Precipitation Amount (mm) | 5.6 | 0.04 | 8 | 87 | |
| wind direction (deg) | 260 | 0.005 | 35 | 52 | ||
| 09utc Grass Minimum Temperature (°C) | 4.4 | 0.01 | 30 | 22 | ||
| Cladosporium | 1978 | Potential evapotranspiration (mm) | 3.4 | <0.001 | 12 | 128 |
| Mean 10 cm soil temperature (°C) | 17.975 | 0.012 | 10 | 118 | ||
| 1979 | Maximum temperature (°C) | 21.5 | <0.001 | 9 | 172 | |
| Mean 10 cm soil temperature (°C) | 15.700 | <0.001 | 59 | 113 | ||
| Mean 10 cm soil temperature (°C) | 13.625 | <0.001 | 32 | 81 | ||
| 1980 | Mean 10 cm soil temperature (°C) | 17.275 | <0.001 | 7 | 88 | |
| Mean 10 cm soil temperature (°C) | 13.075 | 0.001 | 69 | 19 | ||
| Erysiphe | 1978 | Mean 10 cm soil temperature (°C) | ||||
| 1979 | 09utc Grass Minimum Temperature (°C) | 10.06 | 0.005 | 33 | 148 | |
| 1980 | Mean 10 cm soil temperature (°C) | 14.2 | <0.001 | 66 | 29 | |
| Tilletiopsis | 1978 | Mean CBL pressure (hpa) | 1012.2 | 0.012 | 30 | 110 |
| 1979 | Maximum temperature (°C) | 16.2 | 0.007 | 83 | 98 | |
| 1980 | ||||||
| Uredospores | 1978 | Mean CBL pressure (hpa) | 994.1 | 0.002 | 132 | 8 |
| 1979 | Mean 10 cm soil temperature (°C) | 19.750 | <0.001 | 7 | 174 | |
| Mean 10 cm soil temperature (°C) | 16.700 | <0.001 | 36 | 138 | ||
| Mean 10 cm soil temperature (°C) | 15.050 | <0.001 | 45 | 93 | ||
| Mean 10 cm soil temperature (°C) | 13.925 | 0.005 | 9 | 84 | ||
| 1980 | wind direction (deg) | 310 | <0.001 | 16 | ||
| Highest ten-minute mean wind speed (kt) | 21 | 0.003 | 7 | 72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Bracero, M.; Markey, E.; Clancy, J.H.; Sodeau, J.; O’Connor, D.J. First Long-Time Airborne Fungal Spores Study in Dublin, Ireland (1978–1980). Atmosphere 2022, 13, 313. https://doi.org/10.3390/atmos13020313
Martínez-Bracero M, Markey E, Clancy JH, Sodeau J, O’Connor DJ. First Long-Time Airborne Fungal Spores Study in Dublin, Ireland (1978–1980). Atmosphere. 2022; 13(2):313. https://doi.org/10.3390/atmos13020313
Chicago/Turabian StyleMartínez-Bracero, Moisés, Emma Markey, Jerry Hourihane Clancy, John Sodeau, and David J. O’Connor. 2022. "First Long-Time Airborne Fungal Spores Study in Dublin, Ireland (1978–1980)" Atmosphere 13, no. 2: 313. https://doi.org/10.3390/atmos13020313
APA StyleMartínez-Bracero, M., Markey, E., Clancy, J. H., Sodeau, J., & O’Connor, D. J. (2022). First Long-Time Airborne Fungal Spores Study in Dublin, Ireland (1978–1980). Atmosphere, 13(2), 313. https://doi.org/10.3390/atmos13020313







