Assessment of Mercury Concentrations and Fluxes Deposited from the Atmosphere on the Territory of the Yamal-Nenets Autonomous Area
Abstract
1. Introduction
2. Materials and Methods
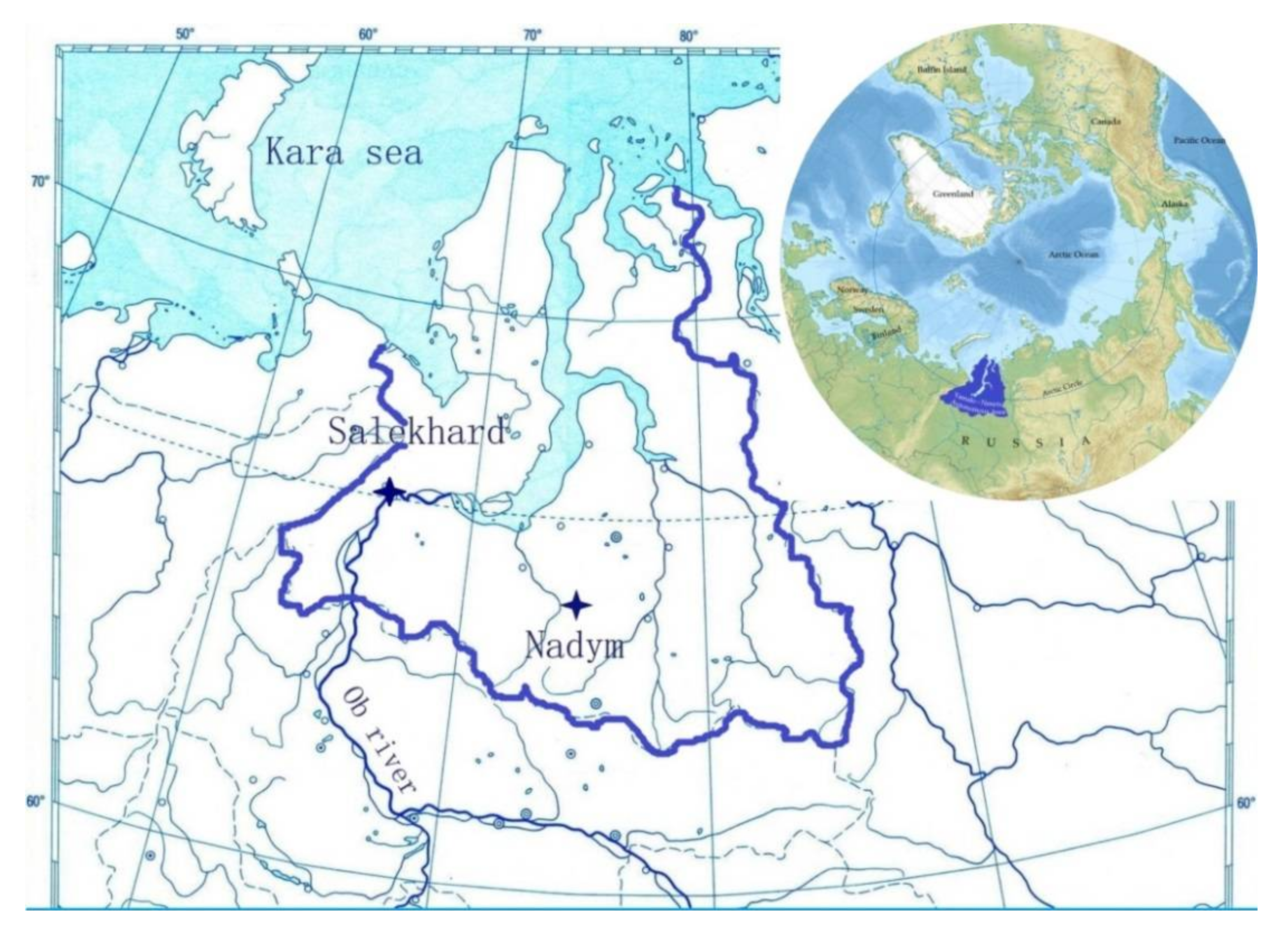
2.1. Study Area
2.2. Sample Collection and Analytical Procedure
2.3. Calculation of Hg Volume-Weighted Concentration and Wet Deposition Flux
3. Results and Discussion
3.1. Hg Concentrations in Wet Atmospheric Precipitation of Yamal-Nenets Autonomous Area
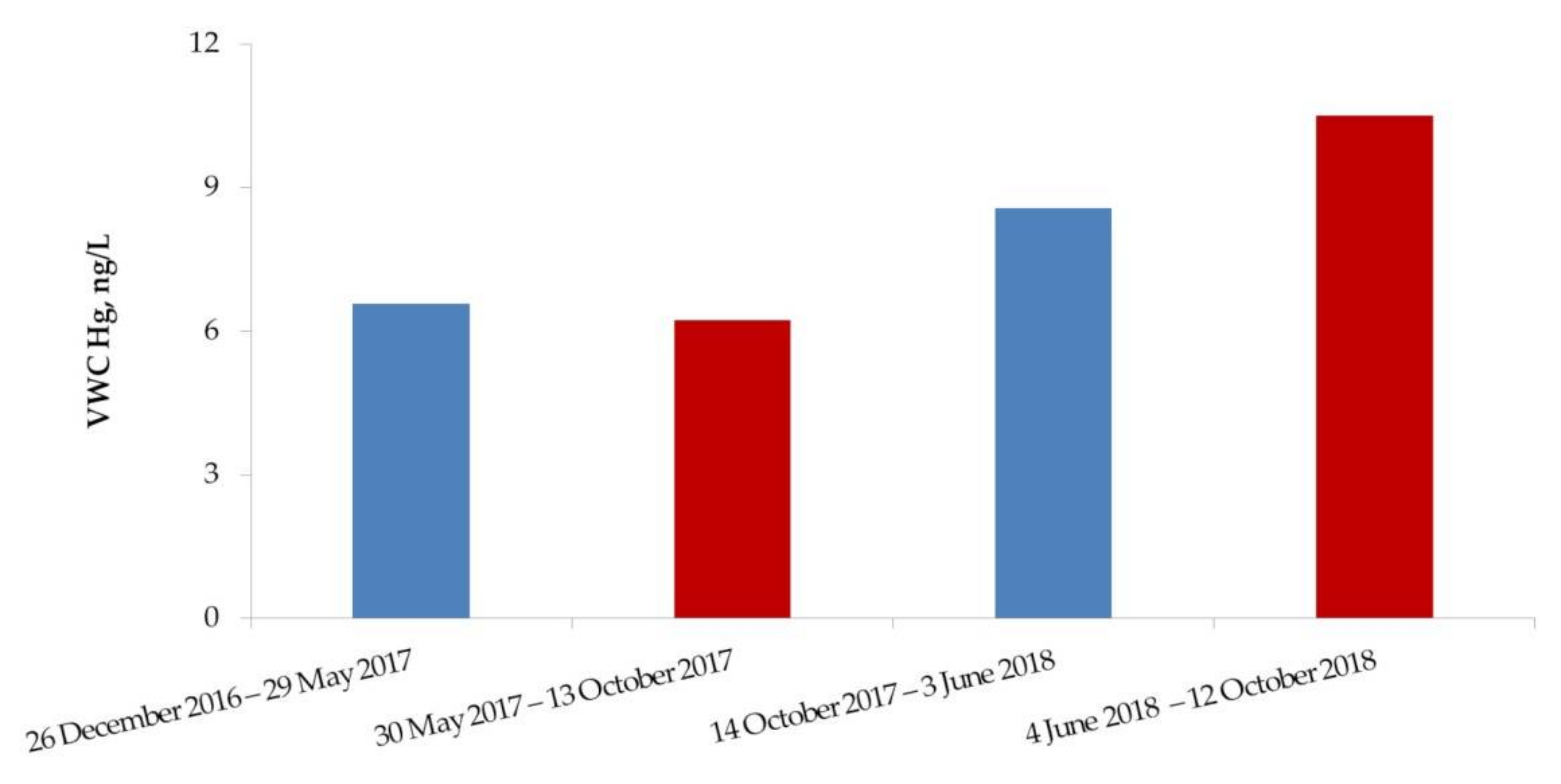
3.2. Seasonal and Interannual Changes of Hg Concentrations in Atmospheric Precipitation in Nadym
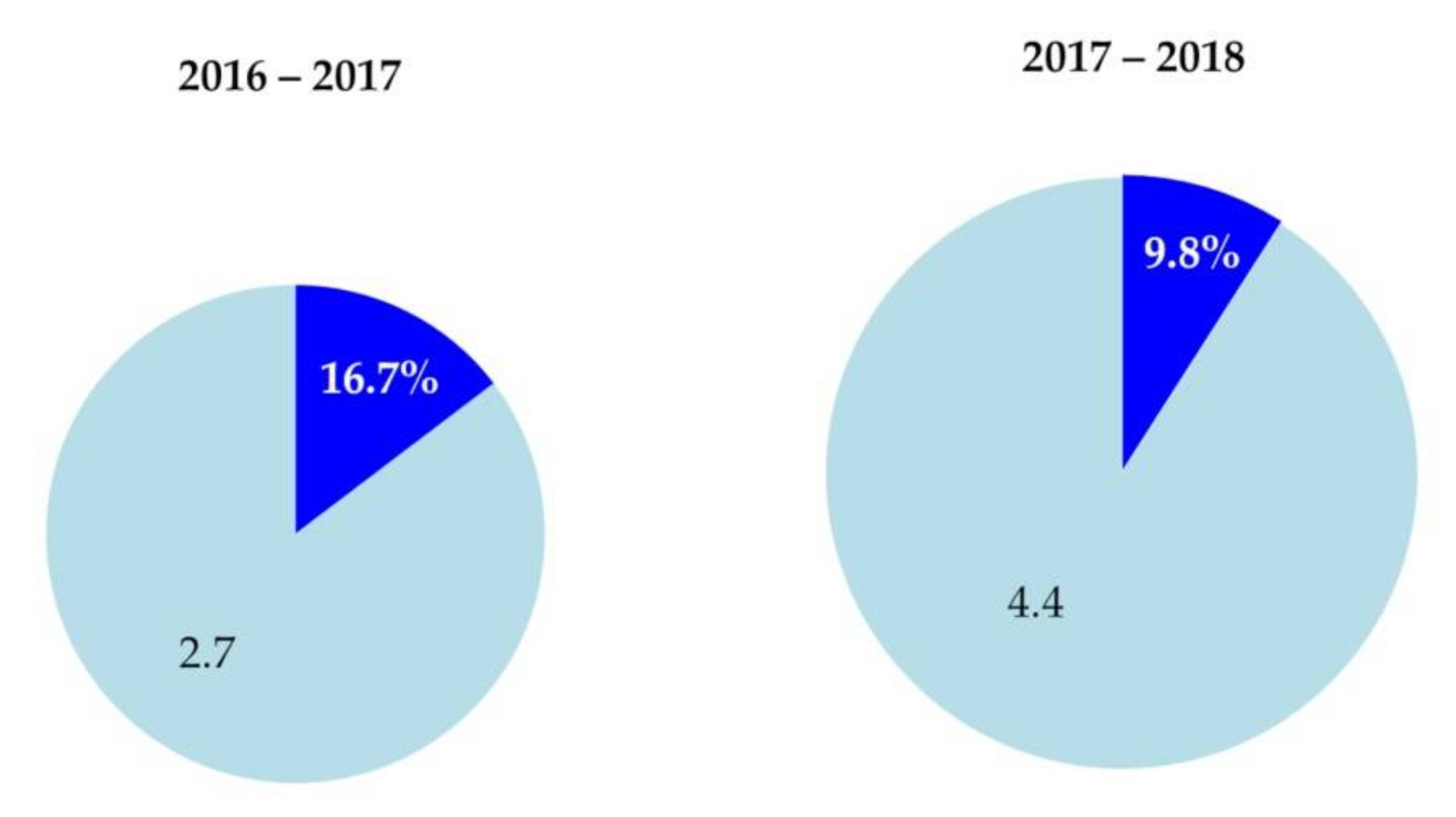
3.3. Wet Deposition Flux
3.4. Spring Increase of Mercury Concentrations in Wet Precipitation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mason, R.P. Mercury Emissions from Natural Processes and Their Importance in the Global Mercury Cycle. In Mercury Fate and Transport in the Global Atmosphere; Masson, R., Pirrone, N., Eds.; Springer: Boston, MA, USA, 2009; pp. 173–191. [Google Scholar] [CrossRef]
- Pirrone, N.; Cinnirella, S.; Feng, X.; Finkelman, R.B.; Friedli, H.R.; Leaner, J.; Mason, R.; Mukherjee, A.B.; Stracher, G.B.; Streets, D.G.; et al. Global mercury emissions to the atmosphere from anthropogenic and natural sources. Atmos. Chem. Phys. Discuss. 2010, 10, 5951–5964. [Google Scholar] [CrossRef]
- AMAP; UN Environment. Technical Background Report for the Global Mercury Assessment 2018; United Nations Environment Programme: Geneva, Switzerland; Arctic Monitoring and Assessment Programme: Oslo, Norway, 2019; p. 426. [Google Scholar]
- Pacyna, J.M.; Travnikov, O.; De Simone, F.; Hedgecock, I.M.; Sundseth, K.; Pacyna, E.G.; Steenhuisen, F.; Pirrone, N.; Munthe, J.; Kindbom, K. Current and future levels of mercury atmospheric pollution on a global scale. Atmos. Chem. Phys. Discuss. 2016, 16, 12495–12511. [Google Scholar] [CrossRef]
- AMAP. Assessment 2011: Mercury in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2011; p. 193. [Google Scholar]
- Ilyin, I.; Rozovskaya, O.; Travnikov, O. Assessment of Heavy Metal. Transboundary Pollution on Global, Regional and National Scales. In EMEP Status Report; Joint MSC-E & CCC; Meteorological Synthesizing Centre—East (MSC-E): Moscow, Russia, 2018; p. 69. [Google Scholar]
- Macdonald, R. Climate Change, Risks and Contaminants: A Perspective from Studying the Arctic. Hum. Ecol. Risk Assess. Int. J. 2005, 11, 1099–1104. [Google Scholar] [CrossRef]
- Douglas, T.A.; Loseto, L.L.; Macdonald, R.W.; Outridge, P.; Dommergue, A.; Poulain, A.; Amyot, M.; Barkay, T.; Berg, T.; Chételat, J.; et al. The fate of mercury in Arctic terrestrial and aquatic ecosystems, a review. Environ. Chem. 2012, 9, 321–355. [Google Scholar] [CrossRef]
- Poissant, L.; Zhang, H.H.; Canário, J.; Constant, P. Critical review of mercury fates and contamination in the arctic tundra ecosystem. Sci. Total Environ. 2008, 400, 173–211. [Google Scholar] [CrossRef]
- Sundseth, K.; Pacyna, J.M.; Banel, A.; Pacyna, E.G.; Rautio, A. Climate Change Impacts on Environmental and Human Exposure to Mercury in the Arctic. Int. J. Environ. Res. Public Health 2015, 12, 3579–3599. [Google Scholar] [CrossRef]
- Atwell, L.; Hobson, K.A.; Welch, H.E. Biomagnification and bioaccumulation of mercury in an arctic marine food web: Insights from stable nitrogen isotope analysis. Can. J. Fish. Aquat. Sci. 1998, 55, 1114–1121. [Google Scholar] [CrossRef]
- Rigét, F.; Braune, B.; Bignert, A.; Wilson, S.; Aars, J.; Born, E.; Dam, M.; Dietz, R.; Evans, M.; Evans, T.; et al. Temporal trends of Hg in Arctic biota, an update. Sci. Total Environ. 2011, 409, 3520–3526. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Mason, R.; Chan, H.M.; Jacob, D.J.; Pirrone, N. Mercury as a Global Pollutant: Sources, Pathways, and Effects. Environ. Sci. Technol. 2013, 47, 4967–4983. [Google Scholar] [CrossRef] [PubMed]
- Travnikov, O. Atmospheric Transport of Mercury. In Environmental Chemistry and Toxicology of Mercury; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 331–365. [Google Scholar]
- Lyman, S.N.; Cheng, I.; Gratz, L.E.; Weiss-Penzias, P.; Zhang, L. An updated review of atmospheric mercury. Sci. Total Environ. 2020, 707, 135575. [Google Scholar] [CrossRef]
- Schroeder, W.H.; Anlauf, K.G.; Barrie, L.A.; Lu, J.Y.; Steffen, A.T.; Schneeberger, D.R.; Berg, T. Arctic springtime depletion of mercury. Nat. Cell Biol. 1998, 394, 331–332. [Google Scholar] [CrossRef]
- Simpson, W.R.; von Glasow, R.; Riedel, K.; Anderson, P.; Ariya, P.; Bottenheim, J.; Burrows, J.; Carpenter, L.J.; Frieß, U.; Goodsite, M.E.; et al. Halogens and their role in polar boundary-layer ozone depletion. Atmos. Chem. Phys. Discuss. 2007, 7, 4375–4418. [Google Scholar] [CrossRef]
- Steffen, A.; Douglas, T.; Amyot, M.; Ariya, P.; Aspmo, K.; Berg, T.; Bottenheim, J.; Brooks, S.; Cobbett, F.; Dastoor, A.; et al. A synthesis of atmospheric mercury depletion event chemistry in the atmosphere and snow. Atmos. Chem. Phys. Discuss. 2008, 8, 1445–1482. [Google Scholar] [CrossRef]
- Skov, H.; Christensen, J.H.; Goodsite, M.E.; Heidam, N.Z.; Jensen, B.; Wåhlin, P.; Geernaert, G. Fate of Elemental Mercury in the Arctic during Atmospheric Mercury Depletion Episodes and the Load of Atmospheric Mercury to the Arctic. Environ. Sci. Technol. 2004, 38, 2373–2382. [Google Scholar] [CrossRef]
- Dommergue, A.; Ferrari, C.P.; Amyot, M.; Brooks, S.; Sprovieri, F.; Steffen, A. Spatial coverage and temporal trends of atmospheric mercury measurements in Polar Regions. In Mercury Fate and Transport in the Global Atmosphere: Emissions, Measurements and Models; Mason, R., Pirrone, N., Eds.; Springer: Boston, MA, USA, 2009; pp. 293–321. [Google Scholar]
- Douglas, T.; Amyot, M.; Barkay, T.; Berg, T.; Chételat, J.N.; Dommergue, A.; Evans, M.; Ferrari, C.; Gantner, K.; Nson, M.J.; et al. What is the Fate of Mercury Entering the Arctic Environment. In AMAP Assessment 2011: Mercury in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2011; pp. 45–65. [Google Scholar]
- Steffen, A.; Lehnherr, I.; Cole, A.; Ariya, P.; Dastoor, A.; Durnford, D.; Kirk, J.; Pilote, M. Atmospheric mercury in the Canadian Arctic. Part I: A review of recent field measurements. Sci. Total Environ. 2015, 509–510, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Weiss-Penzias, P.S.; Gay, D.A.; Brigham, M.E.; Parsons, M.T.; Gustin, M.S.; Ter Schure, A. Trends in mercury wet deposition and mercury air concentrations across the U.S. and Canada. Sci. Total Environ. 2016, 568, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.S.; Steffen, A.; Eckley, C.S.; Narayan, J.; Pilote, M.; Tordon, R.; Graydon, J.A.; Louis, V.L.S.; Xu, X.; Branfireun, B.A. A Survey of Mercury in Air and Precipitation across Canada: Patterns and Trends. Atmosphere 2014, 5, 635–668. [Google Scholar] [CrossRef]
- Steffen, A.; Schroeder, W.; Macdonald, R.W.; Poissant, L.; Konoplev, A. Mercury in the arctic atmosphere: An analysis of eight years of measurements of GEM at Alert (Canada) and a comparison with observations at Amderma (Russia) and Kuujjuarapik (Canada). Sci. Total Environ. 2005, 342, 185–198. [Google Scholar] [CrossRef]
- Danilov, A.A.; Evseev, A.V.; Gordeev, V.V.; Kochemasov, Y.V.; Lukyanov, Y.S.; Lystsov, V.N.; Moiseenko, T.I.; Murashko, O.A.; Nemirovskaya, I.A.; Patin, S.A.; et al. Diagnostic Analysis of the Environmental Status of the Russian Arctic (Advanced Summary); Scientific World: Moscow, Russia, 2011. [Google Scholar]
- Zhulidov, A.V.; Robarts, R.D.; Pavlov, D.F.; Kämäri, J.; Gurtovaya, T.Y.; Meriläinen, J.J.; Pospelov, I.N. Long-term changes of heavy metal and sulphur concentrations in ecosystems of the Taymyr Peninsula (Russian Federation) North of the Norilsk Industrial Complex. Environ. Monit. Assess. 2011, 181, 539–553. [Google Scholar] [CrossRef]
- Vasilevich, M.I.; Vasilevich, R.S.; Gabov, D.N.; Kondratenok, B.M. Evaluation of aerial technogenic pollution near industrial enterprises in the tundra zone (by the example of Vorkuta city). Геоэкология Инженерная геология Гидрогеология. Геокриология 2019, 6, 94–105. [Google Scholar] [CrossRef]
- Golubeva, N.I.; Matishov, G.G.; Burtseva, L.V. Precipitation of heavy metals in the Barents Sea region. Dokl. Earth Sci. 2005, 401, 469–472. [Google Scholar]
- Ji, X.; Abakumov, E.; Xie, X. Atmosphere–ocean exchange of heavy metals and polycyclic aromatic hydrocarbons in the Russian Arctic Ocean. Atmos. Chem. Phys. Discuss. 2019, 19, 13789–13807. [Google Scholar] [CrossRef]
- Ivanov, M. Mercury in the bottom sediments of the Chukotka Sea and adjacent Arctic areas. In Geology and Geoecology of Eurasian Continental Margins. Special Issue. Geology and Mineral Resources of the Eurasian Marginal Seas; GEOS: Moscow, Russia, 2012; pp. 81–87. [Google Scholar]
- Pankratov, F. Dynamics of Atmospheric Mercury in the Russian Arctic. Ph.D. Thesis, Moscow State University, Moscow, Russia, 2015. [Google Scholar]
- Agbalyan, E.V.; Listishenko, A.A. Accumulation of pollutants (mercury and cadmium) in soil, plants and animals. Scientific bulletin of the YNAO. Ecol. Yamalo-Nenets Auton. Okrug 2017, 3, 4–10. [Google Scholar]
- Agbalyan, E. Monitoring of national security: The priorities and some results of studies of the state of the environment in the Yamal-Nenets Autonomous Okrug. Bus. Mag. Neftegaz.RU 2018, 10, 118–125. [Google Scholar]
- Agbalyan, E.V.; Shinkaruk, E. The mercury content in the blood of inhabitants of the yamalo-nenets autonomous district. Hyg. Sanit. 2018, 97, 799–802. [Google Scholar] [CrossRef]
- State Report On the State and Protection of the Environment of the Russian Federation in 2018; Ministry of Natural Resources of Russia: Moscow, Russia; NPP Cadastre: Yaroslavl, Russia, 2019; p. 844.
- The State Report On the State and Environmental Protection of the Russian Federation in 2016; Ministry of Natural Resources of Russia: Moscow, Russia; NIA-Nature: Moscow, Russia, 2017; p. 760.
- Petrov, S.; Mamaeva, N.; Gabdullin, M.; Kraev, A. The Ecological Situation in the Russian Arctic Permafrost Zone. MATEC Web Conf. 2016, 73, 5008. [Google Scholar] [CrossRef]
- Мамаева, Н.Л.; Петрoв, С.А. Natural and anthropogenic dynamics of permafrost soils in the areas of oil and gas production in yamalo-nenets autonomous okrug. Oil Gas Stud. 2015, 6, 99–104. [Google Scholar] [CrossRef][Green Version]
- Bishop, K.; Shanley, J.B.; Riscassi, A.; de Wit, H.A.; Eklöf, K.; Meng, B.; Mitchell, C.; Osterwalder, S.; Schuster, P.F.; Webster, J.; et al. Recent advances in understanding and measurement of mercury in the environment: Terrestrial Hg cycling. Sci. Total Environ. 2020, 721, 137647. [Google Scholar] [CrossRef]
- Schuster, P.F.; Schaefer, K.M.; Aiken, G.R.; Antweiler, R.C.; DeWild, J.F.; Gryziec, J.D.; Gusmeroli, A.; Hugelius, G.; Jafarov, E.E.; Krabbenhoft, D.P.; et al. Permafrost Stores a Globally Significant Amount of Mercury. Geophys. Res. Lett. 2018, 45, 1463–1471. [Google Scholar] [CrossRef]
- Schaefer, K.; Elshorbany, Y.; Jafarov, E.; Schuster, P.F.; Striegl, R.G.; Wickland, K.P.; Sunderland, E.M. Potential impacts of mercury released from thawing permafrost. Nat. Commun. 2020, 11, 1–6. [Google Scholar] [CrossRef]
- Geography of Yamal. Available online: https://www.yanao.ru/region/geography/ (accessed on 15 September 2021).
- Kalinin, V.M.; Kozin, B.V.; Larin, S.I.; Osipov, V.A.; Popova, T.V.; Soromotina, O.V.; Starkov, V.D. Geography of the Yamal-Nenets Autonomous Okrug; Tyumen State University Publishing House: Tyumen, Russia, 2001. [Google Scholar]
- Arnfield, A.J. Köppen climate classification, in Encyclopedia Britannica. 11 November 2020. Available online: https://www.britannica.com/science/Koppen-climate-classification (accessed on 12 December 2021).
- Average Daily Incident Shortwave Solar Energy. Available online: https://weatherspark.com/compare/y/107495~106461/Comparison-of-the-Average-Weather-in-Nadym-and-Salekhard#Figures-SolarEnergy (accessed on 2 December 2021).
- Archive of Weather in Nadym. Available online: https://rp5.ru/Weather_in_Yamalo-Nenets_Autonomous_Okrug (accessed on 4 September 2021).
- Papina, T.S. Assessment of the transboundary transport of pollutants to the territory of the Yamal-Nenets Autonomous Okrug. In Obdoria. Modern Scientific Research in the Arctic; Siberian Branch of the Russian Academy of Sciences: Salekhard, Russia, 2018; p. 38. [Google Scholar]
- Kaulfus, A.S.; Nair, U.S.; Holmes, C.D.; Landing, W.M. Mercury Wet Scavenging and Deposition Differences by Precipitation Type. Environ. Sci. Technol. 2017, 51, 2628–2634. [Google Scholar] [CrossRef] [PubMed]
- Papina, T.S.; Eirikh, A.N.; Malygina, N.S.; Eyrikh, S.S.; Ostanin, O.V.; Yashina, T.V. Microelement and stable isotopic composition of snowpack in the katunsky biosphere reserve (altai republic). Ice Snow 2018, 58, 41–55. [Google Scholar] [CrossRef]
- Malygina, N.S.; Eyrikh, A.N.; Agbalyan, E.V.; Papina, T.S. Isotopic composition and source regions of winter precipitation in the Nadym Lowland. Ice Snow 2020, 60, 98–108. [Google Scholar]
- US EPA. Method 1631, Revision E: Mercury in Water by Oxidation, Purge and Trap, and Cold Vapor Atomic Fluorescence Spectrometry; U.S. Environmental Protection Agency, Office of Water, Office of Science and Technology, Engineering and Analysis Division (4303): Washington, DC, USA, 2002; p. 38. [Google Scholar]
- Pearson, C.; Howard, D.; Moore, C.; Obrist, D. Mercury and trace metal wet deposition across five stations in Alaska: Controlling factors, spatial patterns, and source regions. Atmos. Chem. Phys. Discuss. 2019, 19, 6913–6929. [Google Scholar] [CrossRef]
- Sprovieri, F.; Pirrone, N.; Bencardino, M.; D’Amore, F.; Angot, H.; Barbante, C.; Brunke, E.-G.; Arcega-Cabrera, F.; Cairns, W.; Comero, S.; et al. Five-year records of mercury wet deposition flux at GMOS sites in the Northern and Southern hemispheres. Atmos. Chem. Phys. Discuss. 2017, 17, 2689–2708. [Google Scholar] [CrossRef]
- Zhang, Y.; Jacob, D.J.; Horowitz, H.M.; Chen, L.; Amos, H.M.; Krabbenhoft, D.P.; Slemr, F.; Louis, V.L.S.; Sunderland, E.M. Observed decrease in atmospheric mercury explained by global decline in anthropogenic emissions. Proc. Natl. Acad. Sci. USA 2016, 113, 526–531. [Google Scholar] [CrossRef]
- Muntean, M.; Janssens-Maenhout, G.; Song, S.; Selin, N.E.; Olivier, J.G.; Guizzardi, D.; Maas, R.; Dentener, F. Trend analysis from 1970 to 2008 and model evaluation of EDGARv4 global gridded anthropogenic mercury emissions. Sci. Total Environ. 2014, 494–495, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Obrist, D.; Kirk, J.L.; Zhang, L.; Sunderland, E.M.; Jiskra, M.; Selin, N.E. A review of global environmental mercury processes in response to human and natural perturbations: Changes of emissions, climate, and land use. Ambio 2018, 47, 116–140. [Google Scholar] [CrossRef]
- Olson, C.I.; Fakhraei, H.; Driscoll, C.T. Mercury Emissions, Atmospheric Concentrations, and Wet Deposition across the Conterminous United States: Changes over 20 Years of Monitoring. Environ. Sci. Technol. Lett. 2020, 7, 376–381. [Google Scholar] [CrossRef]
- Munthe, J.; Wängberg, I.; Rognerud, S.; Fjeld, E.; Verta, M.; Porvari, P.; Meili, M. Mercury in Nordic Ecosystems; IVL-B-1761; The Swedish Environmental Research Institute Ltd.: Stockholm, Sweden, 2007. [Google Scholar]
- Sanei, H.; Outridge, P.; Goodarzi, F.; Wang, F.; Armstrong, D.; Warren, K.; Fishback, L. Wet deposition mercury fluxes in the Canadian sub-Arctic and southern Alberta, measured using an automated precipitation collector adapted to cold regions. Atmos. Environ. 2010, 44, 1672–1681. [Google Scholar] [CrossRef]
- Siudek, P.; Falkowska, L.; Brodecka, A.; Kowalski, A.; Frankowski, M.; Siepak, J. Mercury in precipitation over the coastal zone of the southern Baltic Sea, Poland. Environ. Sci. Pollut. Res. 2014, 22, 2546–2557. [Google Scholar] [CrossRef][Green Version]
- Huang, J.; Kang, S.; Wang, S.; Wang, L.; Zhang, Q.; Guo, J.; Wang, K.; Zhang, G.; Tripathee, L. Wet deposition of mercury at Lhasa, the capital city of Tibet. Sci. Total Environ. 2013, 447, 123–132. [Google Scholar] [CrossRef]
- Sakata, M.; Marumoto, K. Wet and dry deposition fluxes of mercury in Japan. Atmos. Environ. 2005, 39, 3139–3146. [Google Scholar] [CrossRef]
- Tripathee, L.; Guo, J.; Kang, S.; Paudyal, R.; Huang, J.; Sharma, C.; Zhang, Q.; Chen, P.; Ghimire, P.S.; Sigdel, M. Spatial and temporal distribution of total mercury in atmospheric wet precipitation at four sites from the Nepal-Himalayas. Sci. Total Environ. 2019, 655, 1207–1217. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhou, C.; Hopke, P.K.; Holsen, T.M. Mercury wet deposition and speciated mercury air concentrations at rural and urban sites across New York state: Temporal patterns, sources and scavenging coefficients. Sci. Total Environ. 2018, 637–638, 943–953. [Google Scholar] [CrossRef]
- Gichuki, S.W.; Mason, R. Mercury and metals in South African precipitation. Atmos. Environ. 2013, 79, 286–298. [Google Scholar] [CrossRef]
- Shi, G.; Chen, Z.; Teng, J.; Li, Y. Spatio-temporal variation of total mercury in precipitation in the largest industrial base in China: Impacts of meteorological factors and anthropogenic activities. Tellus B Chem. Phys. Meteorol. 2015, 67, 25660. [Google Scholar] [CrossRef]
- Travnikov, O.; Batrakova, N.; Gusev, A.; Ilyin, I.; Kleimenov, M.; Rozovskaya, O.; Shatalov, V.; Strijkina, I.; Aas, W.; Breivik, K.; et al. Assessment of Transboundary Pollution by Toxic Substances: Heavy Metals and POPs. In EMEP Status Report; Joint MSC-E & CCC & CEIP Report; Meteorological Synthesizing Centre—East (MSC-E): Moscow, Russia, 2020; p. 149. [Google Scholar]
- Obrist, D.; Agnan, Y.; Jiskra, M.; Olson, C.L.; Colegrove, D.P.; Hueber, J.; Moore, C.W.; Sonke, J.; Helmig, D. Tundra uptake of atmospheric elemental mercury drives Arctic mercury pollution. Nat. Cell Biol. 2017, 547, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.A.; Nelson, P.F. Atmospheric mercury in Australia: Recent findings and future research needs. Elem. Sci. Anthr. 2020, 8, 70. [Google Scholar] [CrossRef]
- Fu, X.; Yang, X.; Lang, X.; Zhou, J.; Zhang, H.; Yu, B.; Yan, H.; Lin, C.-J.; Feng, X. Atmospheric wet and litterfall mercury deposition at urban and rural sites in China. Atmos. Chem. Phys. Discuss. 2016, 16, 11547–11562. [Google Scholar] [CrossRef]
- Prestbo, E.M.; Gay, D.A. Wet deposition of mercury in the U.S. and Canada, 1996–2005: Results and analysis of the NADP mercury deposition network (MDN). Atmos. Environ. 2009, 43, 4223–4233. [Google Scholar] [CrossRef]
- Kiryushin, P.A.; Knizhnikov, A.Y.; Kochi, K.V.; Puzanova, T.A.; Uvarov, S.A. Associated petroleum gas in Russia: “You can’t burn it, recycle it!” In Analytical Report on the Economic and Environmental Costs of Associated Gas Flaring in Russia; World Wildlife Fund (WWF): Moscow, Russia, 2013; p. 88. [Google Scholar]
- Global Wildfire Information System (GWIS). Available online: https://gwis.jrc.ec.europa.eu/apps/country.profile/maps (accessed on 1 December 2021).
- Shcherbov, B.L. The role of forest floor in migration of metals and artificial nuclides during forest fires in Siberia. Contemp. Probl. Ecol. 2012, 5, 191–199. [Google Scholar] [CrossRef]
- Arctic Region | Wildfires Overview 2015–2020—Emergency Response Coordination Centre (ERCC); ECHO. 2021. Available online: https://erccportal.jrc.ec.europa.eu/ercmaps/ECDM_20210308_Arctic_ForestFires_Emissions.pdf (accessed on 10 December 2021).
- A Report on Climate Features on the Territory of the Russian Federation in 2018; Russian Federal Service for Hydrometeorology and Environmental Monitoring (Roshydromet): Moscow, Russia, 2019; p. 79.
- Macsween, K.; Edwards, G.; Howard, D. Up-scaling mercury emissions from terrestrial surfaces as a response to sustained temperature increase. Atmos. Environ. 2019, 223, 117190. [Google Scholar] [CrossRef]
- Yang, Y.; Meng, L.; Yanai, R.D.; Montesdeoca, M.; Templer, P.H.; Asbjornsen, H.; Rustad, L.E.; Driscoll, C.T. Climate change may alter mercury fluxes in northern hardwood forests. Biogeochemistry 2019, 146, 1–16. [Google Scholar] [CrossRef]
- Angot, H.; Dastoor, A.; De Simone, F.; Gårdfeldt, K.; Gencarelli, C.N.; Hedgecock, I.M.; Langer, S.; Magand, O.; Mastromonaco, M.N.; Nordstrøm, C.; et al. Chemical cycling and deposition of atmospheric mercury in polar regions: Review of recent measurements and comparison with models. Atmos. Chem. Phys. Discuss. 2016, 16, 10735–10763. [Google Scholar] [CrossRef]
- Pankratov, F.; Mahura, A.; Popov, V.; Katz, O. Increase of winter atmospheric mercury depletion events during long-term monitoring of mercury in the Russian Arctic. In State of Arctic Seas and Territories under Climate Change; NArFU: Arkhangel’sk, Russia, 2014. [Google Scholar]




| Parameter | Methodological Criteria | Results |
|---|---|---|
| Initial and Ongoing Precision and Recovery (IPR), % | ||
| Residual Standard deviation (RSD) | 21 | <5 |
| Recovery | 79–121 | 96–101 |
| Matrix Spike/Matrix Spike Duplicate (MS and MSD), % | ||
| Relative Percent Difference (RPD) | 24 | <10 |
| Recovery | 71–125 | 87–118 |
| City | Hg Concentration, Ng/L | Period | |||
|---|---|---|---|---|---|
| Min | Max | Average | VWC | ||
| Cold period | |||||
| Salekhard | 1.5 | 26.1 | 6.6 | 5.4 | 5 October 2016–01 May 2017 * |
| 1.2 | 20.4 | 5.5 | 5.6 | 1 November 2017–18 April 2018 * | |
| Nadym | <0.5 | 27.5 | 6.3 | 6.6 | 26 October 2016–29 May 2017 |
| <0.5 | 63.3 | 14.8 | 8.6 | 14 October 2017–03 July 2018 | |
| Warm period | |||||
| Nadym | 3.7 | 22.6 | 8.6 | 6.3 | 30 May 2017–13 October 2017 |
| 1.0 | 29.4 | 12.2 | 10.5 | 4 July 2018–12 October 2018 | |
| Location | Hg Concentration, ng/L | Annual Flux, μg/m2 | Study Period | |
|---|---|---|---|---|
| Median | Annual VWC | |||
| Nadym, Russia [this study] | 6.7 | 8.3 | 3.6 | 2016–2018 |
| Dalniye Zelentsy, Russia [29] | – | 6.0 | 3.0 | 2001–2002 |
| Ny-Ålesund, Norway [54] | – | 4.5 | 1.1 | 2012–2015 |
| Pallas, Finland [54] | – | 6.1 | 2.1 | 2011–2014 |
| Nome, USA [53] | 3.5 | 6.2 | 2.3 | 2013–2015 |
| Glacier Bay National Park, USA [53] | 1.8 | 1.9 | 3.0 | 2010–2013 |
| Dutch Harbor, USA [53] | 2.3 | 2.9 | 4.5 | 2009–2015 |
| Gates of the Arctic National Park, USA [53] | 3.6 | 6.0 | 2.1 | 2008–2015 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eyrikh, S.; Shol, L.; Shinkaruk, E. Assessment of Mercury Concentrations and Fluxes Deposited from the Atmosphere on the Territory of the Yamal-Nenets Autonomous Area. Atmosphere 2022, 13, 37. https://doi.org/10.3390/atmos13010037
Eyrikh S, Shol L, Shinkaruk E. Assessment of Mercury Concentrations and Fluxes Deposited from the Atmosphere on the Territory of the Yamal-Nenets Autonomous Area. Atmosphere. 2022; 13(1):37. https://doi.org/10.3390/atmos13010037
Chicago/Turabian StyleEyrikh, Stella, Liliya Shol, and Elena Shinkaruk. 2022. "Assessment of Mercury Concentrations and Fluxes Deposited from the Atmosphere on the Territory of the Yamal-Nenets Autonomous Area" Atmosphere 13, no. 1: 37. https://doi.org/10.3390/atmos13010037
APA StyleEyrikh, S., Shol, L., & Shinkaruk, E. (2022). Assessment of Mercury Concentrations and Fluxes Deposited from the Atmosphere on the Territory of the Yamal-Nenets Autonomous Area. Atmosphere, 13(1), 37. https://doi.org/10.3390/atmos13010037








