1. Introduction
Fibrous particles can induce long-term inflammation and disease if they are respired into the alveolar region of the lung and are neither dissolved in lung fluids nor removed by lung clearance mechanisms due to their fiber morphology. This epidemiologically substantiated observation has been formulated as the so-called fiber pathogenicity paradigm [
1,
2]. It states that sufficiently long and biodurable respirable fibrous objects exhibit carcinogenic potential in lung tissue.
In light of the history of asbestos and asbestos-related diseases [
3], it is of utmost importance for human health to detect, assess and control excessive concentrations of respirable biopersistent airborne fibers. For such an assessment, several measurement strategies, categorization concepts and counting rules have been developed internationally, mainly focusing on the availability of analytical techniques and on the industrial relevance of fibers of toxicological concern.
An important fiber counting method was developed by the World Health Organization (WHO) [
4]. Fibers matching the dimensions specified in this rule are colloquially called WHO-fibers. They exhibit—motivated by respirability—diameters smaller than
, their length must exceed
and their aspect ratio (length to diameter)
. A detail sometimes neglected is the so-called visibility criterion,
, of the WHO rules, which corresponds to the visibility limit of asbestos and other mineral fibers in optical phase-contrast microscopy (PCM) and sets a lower limit on the diameter of fibers to be counted.
However, asbestos-like toxicity has also been reported for biodurable fibers thinner than
[
5,
6,
7,
8], including nanofibers below
[
9,
10]. For comparable exposure concentrations, it is mandatory to explicitly specify the minimum diameter of the fibers that were actually included in a count result [
11]. Using the term WHO-analogue fibers for analyses including sub-200-nm-fibers can help in emphasizing such ensemble differences.
To indicate concentrations of fiber fractions properly, the symbol will in the following denote the number of WHO-fibers with diameters between and . Standard -fibers are thus denoted as -fibers, whereas, e.g., denotes WHO-analogue fibers in the range of 20–3000 nm.
1.1. Established Limits for WHO-Fibers
Many countries have established legally binding concentration limits like occupational exposure limits (OEL) for selected types of
-fibers. The GESTIS database gives an international overview on workplace shift-averaged (eight hours) and short-term OELs, e.g., for asbestos or mineral fibers (
https://www.dguv.de/ifa/gestis/gestis-internationale-grenzwerte-fuer-chemische-substanzen-limit-values-for-chemical-agents). The eight-hour limits are generally set between 10,000–100,000
for asbestos, with limits sometimes higher for chrysotile than amphibole asbestos, and around 200,000–1,000,000
for minerals and vitreous wools and fibers.
The Netherlands implemented two different asbestos OEL values, depending on analysis technique [
12]. For the same exposure risk level, the associated fiber concentrations are set a factor of two higher for transmission electron microscopy (TEM) than for PCM results, since electron microscopic techniques enable visualizing fibers around the PCM visibility limit of
with higher reliability than PCM.
In Germany, only fibers thicker than
are counted, and the implemented concentration levels vary between
and
, depending on risk level and protection objectives. The German Technical Rule for Hazardous Substances TRGS 910 [
13] for carcinogenic fibers distinguishes acceptance and tolerance concentrations of
and
, which are related to
and
fiber-induced cancer cases per worker life, respectively, using asbestos exposure-risk relations. A concentration below the clearance level of
is mandatory before lifting protective measures on asbestos-contaminated buildings. These concentration ranges may be considered relevant also for pathogenic WHO-analogue fibers.
1.2. Established Limits for WHO-Analogue Fibers
Various toxicological studies suggest that assessing and limiting exposure concentrations of WHO-analogue fiber fractions is necessary. The authors who first introduced the fiber pathogenicity paradigm, Stanton et al., had already reported pathogenic effects of thin fibers, now sometimes called Stanton fibers, that were longer than 8
and thinner than
and found a strong correlation between tumor incidence and fiber number dose [
14,
15]. The French Agency for Environmental and Occupational Health Safety (ANSES) recommended “given the carcinogenic potential” to include thin asbestos fibers when determining dust levels in workplaces with TEM [
16]. Toxicological concerns on biodurable nanofibers, especially carbon nanotubes (CNTs), have also motivated the inclusion of fiber fractions thinner than
into concentration measurements [
5,
7].
Meanwhile, several research projects and authors, as well as national standards and government advisory bodies, have recommended limits for nanofiber concentrations [
13,
17,
18,
19,
20].
The National Institute for Occupational Safety and Health (NIOSH) of the United States, for instance, has published a (recommended) exposure level (REL) for the specific class of carbon nanotubes and carbon nanofibers (CNFs). At workplaces with potential exposures to CNTs and CNFs, the eight-hour average concentration of respirable elemental carbon is recommended to be kept below
[
21]. Since other sources of elemental carbon in a workplace can interfere with the determination of CNTs and CNFs exposures, other analytical techniques such as TEM could assist in characterizing exposures by visualizing particle and fibrous morphologies [
22]. The REL of
corresponds to the current analytical limit of elemental carbon quantification [
21]. However, since this REL is not a fiber number but a mass concentration limit, it circumvents the problem of categorizing complex fiber morphologies as countable fibers or uncountable agglomerates and has the drawback of not allowing to identify correlations between toxicological effects and fiber number dose. In addition, even an analytical limit of
is about two to three orders of magnitude too insensitive to allow the detection of
individual toxicologically relevant nanofibers per cubic meter. Even if just
of the nanofiber mass was aerosolized as individual WHO-analogue fibers, the sensitivity would be insufficient since
graphitic fibers of 15
length,
diameter and a density of 1.8–2.2
would exhibit about
. Currently, MWCNTs with diameters of
and above are considered carcinogenic [
7].
The British Standards Institution [
20] proposed a legally non-binding benchmark concentration for insoluble fibrous nanomaterials of
to be assessed by scanning EM (SEM) or TEM.
The German Committee on Hazardous Substances (AGS) prepared the German Technical Rule TRGS 527 “Activities with nanomaterials”, which was published recently by the German government [
13]. The rule implements a benchmark concentration for the exposure to fibrous nanomaterials at workplaces
“For biopersistent fibrous nanomaterials with a length exceeding
, a diameter below
and a length/diameter ratio exceeding 3:1, a concentration of below
should be ensured in the air at the workplace [
13].”
The term nanomaterial here refers to the proposed EU definition [
10], which includes nanofibers down to
and also thinner single-walled CNTs (SWCNTs). Since the definition comprises materials with a certain number fraction of nanoscale particles and fibers, consequently, microscale fibers may be present in an EU nanomaterial. The German value can thus be interpreted to correspond to
including SWCNTs. In contrast to the German and other asbestos OELs, this benchmark value is not health-based, since epidemiology-based exposure–risk relations are missing for nanofibers.
1.3. Guidelines for Determining WHO-Fiber Concentrations
Reproducible visualization, recognition, categorization, identification and counting of fibrous morphologies require detailed technical guidelines that specify how to achieve reliable imaging, identification of material composition, categorization of complex objects etc.
WHO-fiber measurement guidelines established on national levels aim at determining airborne fiber concentrations for consumer and occupational health and safety regulations. The majority were designed for asbestos and other mineral and vitreous fibers, including NIOSH Method 7400 and 7402 [
23,
24], VDI Guideline 3492 [
25] and DGUV Information 213–546 [
26]. They are based on the
counting rule and, generally, the
PCM visibility criterion to maintain epidemiological comparability of contemporary data to historic reports on fiber concentrations independent of the achievable microscopic resolution.
In Germany, for monitoring asbestos concentrations at workplaces, the technical rules for hazardous substances TRGS 402 [
27], 519 [
28] and 517 [
29] apply the measurement strategy of the DGUV Information 213–546 [
26]. The latter requires the collection of respirable fibers on filters and the analysis of filter areas of up to
to detect up to
-fibers, whichever occurs first. The DGUV protocol aims at an analytical sensitivity, defined in
Appendix A.2, of
to quantify concentrations above
. The German asbestos clearance level below
is tested by following the VDI Guideline 3492 [
25].
1.4. Development of WHO-Analogue Fiber Guidelines
For
-fibers, reliable visualization and recognition of their morphology is achievable at microscopic resolutions of about 100–200 nm, see
Section 2.4.1. At such low resolutions, the analytical effort required for mapping and analyzing a filter area of
is well manageable. For thinner WHO-analogue fibers, higher microscopic resolutions than
are required. However, the attempt to quantify thinner fibers with methods that were implemented for
-fibers can, especially for nanofibers, result in excessive analytical workload, if only the image resolution is increased, see
Section 2.5. Therefore, nanofiber-adapted assessment concepts are necessary.
In the following, we propose such a strategy for assessing airborne concentrations of micro- and nanoscale fibers. This approach to reducing the workload is schematized in
Table 1. Similar to established methods for
-fibers, we aim at detecting fibers after flow-controlled sampling on filters by microscopic analysis of finite filter areas. A first workload reduction is achieved by implementing a lower limit on fiber diameter. Such a cut-off is already in use in established counting rules for
-fibers: Fibers thinner than
are ignored.
For nanoscale fibers, careful selection of this cut-off diameter is essential to reduce the analytical workload without ignoring potentially pathogenic fiber fractions. In
Section 2.5.1, our choice of cut-off diameter of
is motivated in more detail. A second workload reduction can be achieved by waiving the aim to absolutely quantify fiber concentrations. Instead, we propose to check compliance of a workplace concentration with an exposure limit by estimating an upper limit on the true concentration, see
Section 2.7.3. This is achieved by so-called null hypothesis testing.
For comparison, a first, rough estimate for a concentration from a Poisson-statistical interpretation of a fiber counting experiment requires observing about 15 fibers, whereas an upper limit on the true concentration can be estimated with a confidence level of
from observing zero fibers, when
was expected at the exposure limit concentration. A threshold of 15 fibers is suggested here, since the relative size of the probability interval
exceeds
for less than
fibers cf.
Table A2 in
Appendix D.4.
2. Methodology
This section describes in detail the methodology underlying the proposed measurement strategy, including experimental aspects of sampling, imaging, categorization and quantification, as well as the statistical null hypothesis concept for compliance testing.
2.1. Measurement Outline
Table 2 outlines the workflow of the proposed measurement strategy. It also refers to the sections of this paper that describe further methodical details.
2.2. Analytical Requirements and Experimental Constraints
Determining the concentration of a specific fraction of airborne fibers is a demanding task. It requires a series of experimental and analytical steps that depend on several on-site conditions and experimental parameters.
On-site conditions
- ○
Characteristics of emission events: Continuous or transient, localized or long-term
- ○
Monitoring conditions: Source distribution, site ventilation and ambient dustiness
- ○
Fiber-to-dust ratio: Probability of fiber concealment by overlaying dust particles
Some of these analytical requirements are universal in the sense that they are independent of fiber dimension. Universal analytical details require no nano-specific adaptation. For instance, the task of recognizing fibers on sharp microscopic images via their morphology is independent of fiber diameter, whereas the requirements for sharp imaging, individual fiber material analysis, as well as aerosol distribution and collection dynamics, depend both on fiber dimensions and composition. Only visualized and identified product fiber fractions will affect the resulting fiber concentration.
2.3. Airborne Fiber Sampling
The proposed measurement strategy applies sampling of airborne dust by filtration on membrane filters. The requirements on filters and sampling conditions are discussed below.
2.3.1. Filtration Materials
Imaging and counting of sampled fibers requires filter substrates that allow reliable recognition of fiber morphologies by means of microscopic imaging. Unless a filter is dissolved or dry-etched prior to analysis, it must not be composed of fibrous structures that might prevent recognition of the fibers under study, but rather of a porous membrane. Established asbestos and mineral fiber concentration methods use foamy cellulose ester membranes or porous polymer films of typically less than porosity. A smooth surface between pores can help in discriminating fibers from the filter. Membrane porosities around should be preferred, since the larger contiguous area between pores facilitates tracking a fiber’s path.
Nuclear track-etched pore filters made from polycarbonate (PC) exhibit smooth surfaces and generally round pores. Using track-etched membranes of poor surface quality and with many oblique-angle pores should be avoided. Sputter coating the polymer with noble metals like gold, palladium or iridium provides electrically conductive surfaces of small grain-size that allow high-resolution electron scanning microscopic analysis of nanoscale objects.
For -fibers, typically track-etched pore filters of pore diameter are used. Filtering airborne nanoscale fibers may suggest the use of significantly smaller pore sizes to increase the filtration efficiency, as PC membranes with pores down to diameter are commercially available. However, unless a very thin and mechanically sensitive membrane is used, at the same porosity, smaller pores can drastically enhance the pressure drop over the filter and limit the achievable airflow. For example, an airflow of through a PC membrane of diameter, porosity, with pores of diameter and a film thickness of causes a pressure drop of . In our experience, many standard air sampling pumps for asbestos measurement do not exhibit the necessary flow-versus-pressure performance to maintain desired sampling flows in the multiple-liters-per-minute range with the high pressure drops caused by small filter diameter, low porosity, small pores and/or high film thickness.
The choice of pore size is therefore a trade-off between fiber deposition efficiency and achievable airflow. The task of counting WHO-analogue nanofibers, i.e., fibers longer than , should admit using larger pores since inertial filter impaction and filter pore-edge interception probability tend to increase with fiber length and airflow rate.
Another lower constraint on pore diameter of track-etched membranes results from the requirement of unambiguously localizing a fiber’s position. This is possible by recognizing constellations of random pore patterns in the vicinity of the fiber. To also enable filter orientation in optical microscopes, we recommend using filters with pores of at least
diameter. As a benefit of such post-imaging localizability of fibers, fiber material analysis becomes possible by correlative microscopic techniques using electron microscopy, energy-dispersive and Raman spectroscopy, see
Section 3.6.
Typical filter areas to be analyzed for the presence of fibers amount to, e.g.,
for quantifying occupational exposures in the order of
[
26]. Since gravimetric dust quantification, which requires high filter loading and larger filter area, is not combinable with fiber quantification, the filter diameter can be reduced from the
of the German PGP-FAP sampling head to, e.g.,
of the standard asbestos filter heads or even
, still providing more than
times the required area to be analyzed, see
https://www.gsa-messgeraete.de/PGP-FAP-adapter-universal-sampler-fibre-plastics-cylinder-supporting-sieve.
2.3.2. Filter Sampling Constraints
The achievable fiber deposition densities on a filter are subject to several constraints. The higher the density, the smaller the area to analyze for an exposure limit compliance check, the lower the visualization workload at a given microscopic resolution. It is, therefore, desirable to increase the specific air volume , i.e., the total sampled volume per open filter area . The total collected volume depends on the sampling duration and airflow ; for constant flow . For a clamped filter membrane of diameter and clamping of width , the open filter area is with effective diameter .
The practically achievable specific air volume is constraint by the following aspects:
- 1.
The sampling duration
must be adapted to characterize brief or transient emission processes and short-term or longer work shifts. If durations are enforced to be short, higher flows will be favored to keep the filter area to analyze small.
- 2.
The acceptable sampling flow
range is generally constraint by the required filter face velocity range that ensures effective fiber filtration by interception and inertial impaction. Additionally, the filter pressure drop and the pump performance profile, as well as dust pre-separation requirements by means of vertical elutriation may limit the accessible flow range.
- 3.
Especially in dusty sampling environments, the specific air volume
may need to be adapted to the ratio of the ambient dust concentration
to the targeted fiber concentration
Overloading filters with particles that may conceal sampled fibers and prevent recognizing a fiber or tracking its complete length must be avoided, since this would reduce the recognition probability of (long) fibers. Practical experience, test measurements and online-detection techniques like aerosol spectrometers or condensation particle counters may help to estimate airborne dust concentrations and to adapt sampling parameters accordingly. For completely unknown ambient dust levels, different specific air volumes should be collected in parallel to obtain analyzable samples.
- 4.
Fibers traversing or being trapped inside pores are lost for analysis. The fiber deposition efficiency
of the filter at the chosen flow rate must therefore be known or estimated since only fibers that can be visualized on the filter can be categorized and counted, cf.
Section 2.3.1.
As
Appendix B shows in detail, it is important to avoid any
overestimation of the filter deposition efficiency
and the flow
as well as
underestimation of the open filter area by filter deposition studies, applying careful flow calibration and mechanically reliable filter clamping.
2.4. Quantification of Sampled Fibers
Quantification of airborne fiber concentration requires collection, imaging, recognition, morphology characterization, categorization, identification, counting and, finally, normalization to the analyzed air volume. Besides methodological flaws in selecting a suitable sampling strategy and sampling position at a worksite, all these steps are possible sources of systematic and statistical error.
2.4.1. Digital Imaging Resolution and Pixel Size
Contemporary optical, Raman and electron transmission or scanning microscopes use digital cameras or scanning techniques to provide pixelated image data. The microscopic visibility of particles and fibers in pixelated microscope images is primarily constraint by the achieved physical microscopic resolution and, secondarily, by the lateral extension of the acquired image pixels , called pixel size. If the pixel size is chosen to be larger than the physical resolution, , the effectively usable digital image information will be reduced to a resolution of by the spatial averaging of a sharp image. Meanwhile, a pixel size smaller than the resolution, , corresponds to the digitization of an unsharp image and is generally data inefficient, since no significant additional information is acquired.
For the subsequent discussion on the required digital image information content, we therefore assume the pixel size to be equal to or exceed the achieved microscopic resolution
In this way, pixel size becomes a very convenient parameter to control image acquisition at resolutions necessary for recognition of relevant structures. It is better adapted to our problem than the traditionally used image magnification factor
, which is meaningless without specifying the total image size in number of pixels. If not specified by the microscope control software, the size
of square pixels can be calculated in nanometers for a microscopy image of
pixels with
from the
-inch-Polaroid-image-related magnification factor
2.4.2. Digitized Fiber Recognition Requirements
For morphology assessment and categorization, the shape of individual fibers must be reliably recognizable. In digital, pixelated images, this recognition step relies on identifying a chain of grey-level-correlated pixels that must exhibit significant contrast to filter (or particle) background pixels. The image contrast results from a combination of the microscopic resolution and the material contrast, which depends on the microscopic technique used.
Any image resolution worse than a fiber’s diameter reduces the perceivable contrast by blurring. For visualizing a fiber, the image resolution and pixel size must not exceed the fiber diameter
For reliable visualization, a resolution of
, whereas for diameter determination, a resolution of
is recommended. Thus, the thinner a fiber, the higher the required imaging resolution and the more pixel data are to be generated per analyzed filter area
. As a consequence, the number of image pixels
to be acquired increases quadratically with decreasing pixel size
2.4.3. Measurability of Fiber Diameter, Length and Aspect Ratio
For categorizing a fiber according to the WHO or WHO-analogue fiber criterion, its relevant morphological fiber features length
and diameter
must be determined pairwise to be able to calculate the aspect ratio
. Such pairwise measuring, in principle, requires the fiber to be fully contained in a microscope image. Since the criteria only set upper or lower limits on diameter, length and aspect ratio, some fibers can be categorized as WHO-analogue fibers even if they are not fully contained in a microscope image, provided
with
being the cut-off diameter for nanofibers, e.g.,
, as introduced below.
Diameter determination requires calculating the average of the measured diameters at at least three different positions along the fiber. For WHO categorization purposes, the diameter requires to be accurate only to a certain level. Firstly, to decide whether a fiber is thinner than
is easily achievable at the microscopic level. Secondly, whether an aspect ratio exceeds
must be decided only for fibers with
. Otherwise, the length criterion dominates. Thirdly, analytically more demanding is to decide whether a fiber is thicker than the lower visibility or the cut-off diameter
of
Section 2.5.2. However, for fiber counting alone, high-accuracy diameter measurement is generally not required, cf. Equation (3). It is even counter-productive for fiber categorization, since high diameter accuracy limits the offline measureable aspect ratio, see below.
For length determination of bent or curled fibers, the so-called rectified length along the fiber’s path must be determined. It requires to be able to reliably recognize the course of a fiber, even across fiber crossings. Image analysis software may help to obtain reliable results.
For a fiber in a microscope image that is shorter than , direct WHO categorization is not possible if that fiber is not fully contained in the frame. During online filter analysis, the image frame will either be interactively repositioned to follow the course of the fiber and determine its length, or, if the fiber’s visibility can be maintained, the image magnification will be reduced to image a larger filter area.
In the case of offline image analysis, cf.
Section 2.8, the same position-calibrated filter area must be imaged a second time to include neighboring image frames to measure the full length of the fiber of interest. It is, therefore, instructive to estimate the fiber containment probability. The expectation value for the length of randomly placed and randomly oriented straight fibers that are fully contained in an image frame of
pixels at a pixel size
is
and their aspect ratio
is
For different image resolutions and pixel sizes,
Table 3 lists calculated expectation values for aspect ratio and length. Images large enough to fully contain fibers longer than
with fair probability should be larger than
, i.e.,
for
. For the fiber toxicology-related cut-off diameter
of our approach, see
Section 2.5.2, and the associated pixel resolution of about
required for reliable visualization of such thin fibers, it is recommended to acquire images with significantly more than
each.
Flexible fibers may be deposited on filters in a bent or curled shape. This will enhance the fraction of contained fibers longer than the WHO limit.
2.5. Fiber Diameter-Related Workload
Controlling an exposure limit of
for
-fibers thicker than
according to the German “Method for determining concentrations of respirable inorganic fibers in working areas” [
26] requires complete analysis of a filter area
or counting of at least 50
-fibers. The quadratic scaling law of Equation (4) is exemplified in
Table 4 for exposure limit control on a such an area of
. With a pixel size of
required to reliably visualize fibers of diameter
and above, imaging of such an area is achievable with little effort.
However, for nanoscale fibers of about
in diameter, on a filter area of
, reliable visualization becomes a gargantuan task, requiring the acquisition and analysis of
at a pixel resolution of
. For nanofibers of
, at least
are required to image and evaluate the area
, which appears practically impossible with contemporary technology. A motivation for limiting the fiber diameter to image will be given in
Section 2.5.1, whereas a desirable reduction of filter area to evaluate motivates waiving the determination of absolute fiber numbers in
Section 2.7.1.
2.5.1. Workload Reduction by Introducing a Cut-Off Diameter
With the aim of reducing the analysis effort to a practicable amount, we propose using the so-called fiber rigidity hypothesis [
6,
8,
30,
31]. It toxicologically motivates ignoring a specific fraction of thin fibers and thus permits reducing the resolution of image acquisition. The rigidity hypothesis extends the fiber pathogenicity paradigm by imposing that only fibers that exceed a specific threshold of flexural rigidity show fiber-like toxicity. Fibers of lower rigidity, i.e., higher flexibility, are assumed to spontaneously curl or bend under forces applied by cells or tissue and to not behave in a fiber-like manner, but rather a more granular material-like manner [
32]. Analogous to the fiber pathogenicity paradigm, the extended paradigm also ignores the specific material composition and considers only properties assumed to be relevant for inhalative toxicity. Physically, flexural rigidity
is a product of flexural, or bending modulus
and second moment of a fiber’s axial cross-section area
For isotropic and homogeneous materials, the flexural and tensile, or Young’s, modulus are identical. For a fiber of diameter
, the second moment of axial area
is
Flexural rigidity thus scales proportionally with the modulus and to the fourth power of diameter. As a consequence, the effects of fiber diameter on rigidity easily dominate over small modulus differences. For materials of similar flexural modulus, a “critical rigidity” value , which separates flexible from rigid, carcinogenic fibers, can be simplified to a “critical diameter” . Thus, for materials of identical composition and microstructure, the introduction of a toxicologically motivated cut-off value on fiber diameter appears justified.
The analytical consequences of imposing such a rigidity-motivated cut-off diameter are similar to that of the PCM visibility-motivated diameter limit of implemented in international and national counting rules: Both allow a reduction in the number of pixels to image and search for toxicologically relevant or PCM-visible fibers, respectively.
2.5.2. Critical Flexural Rigidity to Derive a Cut-Off Diameter
Any toxicologically motivated cut-off diameter must—in principle—be chosen according to the alveolar response to fibers of known flexural rigidity. Currently, however, the level of critical rigidity causing fiber-like toxicity of biodurable fibers has not been determined exactly. Since no epidemiological data are available on nanoscale fiber toxicology, the data most relevant for human toxicology have been obtained by
intraperitoneal (
i.p.) testing. Most
i.p. test results on nanofibers of known diameter and composition are available for multi-walled carbon nanotubes (MWCNTs) Figure 7.2 in Ref. [
8]. Rittinghausen et al. reported a positive, fiber-like
i.p. test result for the—so far—thinnest MWCNT material of
mean diameter [
7].
Carbon nanotubes (CNTs) became famous for being very high modulus materials compared to other nano- and microscale fibers Table 7.3 in Ref. [
8]. However, depending on the synthesis method and the measurement technique, MWCNT can show a rather broad variation of elastic modulus values of 1200–20 GPa
Table 1 in [
30]. The higher values resulting from single-walled CNTs and (early) arc synthesis, the lower ones from (later) mass-produced MWCNTs by catalytical chemical vapor deposition (cCVD).
For MWCNTs, Broßell et al. proposed a first estimate of the critical rigidity of about
, based on 37–44 nm (mean
) diameter and 36–1000 GPa (mean
) elastic modulus [
8]. Even if the
i.p.-tested MWCNTs with a mean diameter of
had a very low modulus of
, the resulting rigidity of about
would correspond to that of nanofibers of just
and a modulus of about
, cf. Equation (9).
It therefore appears justified to propose a toxicologically motivated critical diameter of that separates rigid from flexible high-modulus fibers. More flexible, low-modulus fibers should show larger critical diameters. With respect to a critical rigidity estimated from i.p. test data on fibers, a includes a safety margin of about for inaccurately known modulus values and/or missing i.p. test data on thinner fibers. Deriving a more reliable or value will also require careful characterization of thinner i.p. test batches using—currently missing—validated rigidity measurement techniques.
Reliable imaging of fibers with requires microscopic resolutions of at least 10–20 nm, which are also still achievable for most low-end scanning electron microscopes. For reasons of the broad applicability of the proposed nanofiber measurement strategy, a technical argument can thus also motivate a diameter cut in this range.
The proposed critical fiber diameter of
is one order of magnitude below the current visibility criterion of
-fiber counting rules of
. If it is applied with the aim of nanofiber-including fiber concentration determination, the analytical workload will increase by a factor of
, cf.
Table 1. Smaller cut-off values tend to result in unmanageably large pixel numbers, cf.
Table 4.
2.6. Fiber Number Determination
To check compliance with a limit for WHO-fiber number concentration, the EM images of the filter samples must be evaluated visually or with the help of image analysis software. Recognized particles are to be dimensionally measured, morphologically categorized, substance identified to arise at a count of potentially biopersistent WHO-fibers. By analogy with the WHO asbestos counting rules, a distinction is made between respirable and not-respirable objects. Moreover, only fiber-containing objects will be analyzed in detail.
2.6.1. Morphological Categorization of Respirable Fiber-Containing Objects
Air and liquid suspensions of fibers generally not only contain individual and sometimes self-crossing fibers, but also loosely tangled or tightly bundled fibers, and, last but not least, agglomerates comprising many entangled fibers. The measured suspension state therefore partially depends on preparation and handling processes, partially on mechanical and surface-chemical properties of the fibers [
33]. Any fiber number concentration that is determined by analyzing sampled aerosols or suspensions therefore only reflects the state of a specific fiber ensemble at the time of sampling. In addition, determined concentrations will be highly dependent on the details of categorizing these various morphologies. Therefore, detailed decision rules and well-trained personnel are necessary to arrive at comparable analysis results.
The case of bundled fibers illustrates the requirements for a categorization guideline: Both chrysotile asbestos and SWCNTs are nanoscale fibers that form multi-fibril bundles already during synthesis. Any resulting fiber concentration will thus depend on whether the fibrillary structure of bundles can be resolved microscopically, what fiber and bundle diameters are to be included in the analysis, and whether bundles are counted as one object or as individual fibrils.
The measurement strategy defines five morphological classes for the categorization of fiber-containing objects that are considered respirable
, cf.
Table 5. Only fiber-shaped
, and not the not fiber-shaped
, objects contribute to the counting result. The fiber-shaped objects are individual fibers or are composed of individual fibers to form bundled, tangled or agglomerated fibers. All countable fiber-shaped objects with length
contribute to the number of WHO-analogue fibers in the sample, see
Section 2.6.2.
2.6.2. Fiber Counting Rules
All objects that can be assigned to one of the categories listed in
Table 5 must be analyzed further using a set of counting rules that are defined in the following and are illustrated in
Table 6. For an assignment of recognized fiber-containing objects to one to the categories in
Table 5, it is necessary to pairwise determine their length and diameter as described in
Section 2.4.3.
In case individual fibers are visible within a fiber agglomerate or fiber bundle, they are to be individually measured, categorized and counted, provided they can be traced from tail to tail. If this is not feasible, the object is considered an agglomerate and its total length and mean diameter are used for its categorization. A bundle with no individually recognizable constituent fibers is to be treated as an individual fiber.
A fiber that has no tail contained in the EM image frame is not counted, since its tails would be further analyzed when appearing in neighboring images. For a fiber that shows one end while the other protrudes the image frame border, its visible length is measured. If the visible length exceeds , the fiber is weighted as a half WHO-analogue fiber. If the protruding fiber is shorter than 5 µm, the offline evaluation approach requires the filter sample to be re-inserted into the SEM to determine the true total length of this fiber in a second imaging run to distinguish “half” WHO-analogue fibers from short, not counted fibers. For this purpose, the positions of image frame and the fibers of interest must be documented during the first run and be retrievable during the second run for accurate SEM stage positioning. During an online EM evaluation, the image frame can be adjusted interactively along a fiber’s path to determine its full length.
Table 6 schematically exemplifies some important counting cases.
2.6.3. Identification of Product Fibers
Although the fiber pathogenicity paradigm is formally independent of a fiber’s bulk and surface chemistry, as long as a fiber is biodurable, composition analysis is practically required to assess material-related biodurability aspects. Additionally, toxicological relevance according to the extended fiber pathogenicity paradigm [
8,
30] requires material information to assess the composition- and diameter-dependent rigidity aspects of fibers. In addition, last but not least, the origin and potential source of a fiber must be clarified to distinguish background fibers of, e.g., natural origin, from fibers that were released by a work process, subsequently called product fibers. Exposure to product fibers evokes occupational responsibilities. Therefore, the identity of all countable WHO-analogue fibers must be determined to correct a fiber count for non-product fibers.
Ambient dust at assessed workplaces increases the overall occupancy of a filter sample and sometimes causes difficulties in recognizing a fiber or distinguishing a product from a background fiber. For such distinctions, identification of fibers should attempt to recognize characteristic morphological features of product fibers. For our workflow, pristine product fibers that were handled at a studied workplace were dispersed in our laboratories using the vibro-fluidizer dustiness test [
33]. These reference aerosols were then sampled on filters and studied for compositional and morphological characteristics with imaging conditions identical to those applied to workplace samples. An example of a direct comparison of aerosol particles collected during dustiness testing (left) and the workplace aerosol measurement (right) is shown in
Figure 1. The workplace fiber in the right image resembles the product fibers in the left image with respect to length, diameter, surface structure and imaging properties like contrast, charging and edge effect.
Additional analyses were performed whenever fiber identification based on morphological features alone was questionable. Combining morphological and compositional information requires to spatially correlate EM images with elemental and/or chemical analyses at exactly the filter position of the fiber of interest. For nanoscale objects, the required spatial correlation accuracy can be an experimental challenge.
Information on elemental composition can be gained from energy dispersive X-ray spectroscopy (EDS). Sometimes, elemental composition alone is not sufficient, and Raman, FTIR or Laser direct infrared imaging (LDIR) spectroscopy may be required to identify chemical and structural differences. For this work, focusing on carbonaceous fibers, correlative Raman microscopic analysis was performed at a fiber’s filter positions using a confocal Raman Spectrometer equipped with EMCCD detector (WITec Apyron, WITec GmbH, Ulm, Germany) with laser excitation at 532 nm and 0.5 mW power. Two example Raman analyses are presented in
Figure 2 and
Figure 3. Although the analyzed nanoscale fibers were not reliably visible in the optical microscope, the unique constellation of membrane pores in the fibers’ vicinities enabled positioning the confocal Raman detection volume with an accuracy of about 50 nm. This is better than the lateral confocal resolution of about 300 nm, using a objective with 0.75 NA.
2.7. Estimating Fiber Concentrations
Analyzing a filter area
for fibers of a specific category will result in observing zero or a positive number of
fibers. According to the counting rules in
Section 2.6.1, integer and half integer values of
are possible. The corresponding analyzed air volume
is the product of area
with the specific air volume
, which was defined as total sampled volume
per total filter area
. An estimate
of the true airborne fiber concentration
can be calculated from the ratio of observed fibers per analyzed air volume for known or estimated filter deposition efficiency
The subsequent reasoning will deal with the observed fiber number , the actual measurand. Equation (10) then allows calculating airborne concentrations estimates from observed fiber numbers or true fiber number estimates and their probability interval limits .
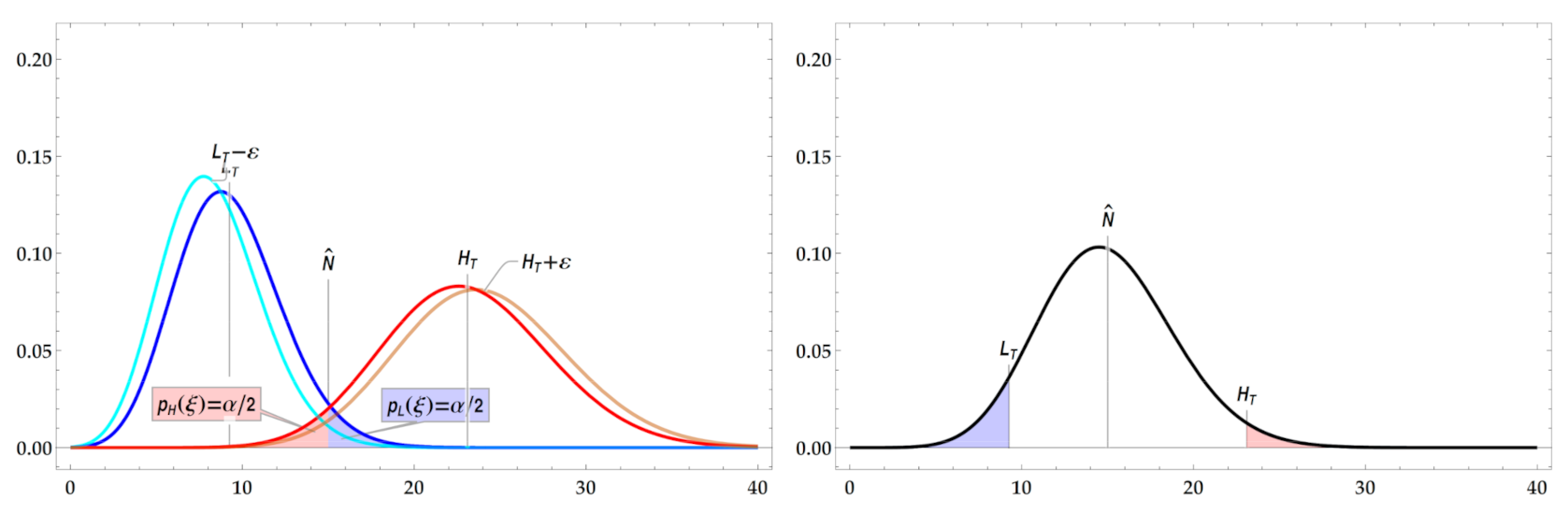
2.7.1. Confidence versus Probability Interval Definition
Due to the high workload required for analyzing a filter area for the presence of deposited micro- and nanoscale fibers, currently only a single or very few of such measurements will be performable at a real-life workplace. Unfortunately, from a single (or very few) experiment(s), the—principally unknown—statistical distribution of the observable “number of fibers ” cannot be determined empirically in a Frequentist’s manner by histogramming the outcome of many repeated experiments on an atmosphere of stationary composition. As a consequence of the unknown shape of the distribution density function, no confidence interval can be specified that would make it possible to estimate a probable value range around the true fiber number .
We therefore impose a statistical model for the distribution of our experimental results. In the case of uncorrelated deposition of airborne fibers on a filter by sampling a homogeneous aerosol and on evenly coated regions of the filter, it appears justified to assume the number of experimentally observed fibers to be a Poisson-distributed random variable with the probability density function with integer-valued and real-valued expectation value .
The expectation value is considered to be the true value that caused the observed experimental outcome . Our model assumption now enables us to estimate an interval of probable values around the true number of fibers from a single experimental observation. Mathematically correctly, such an interval is called a probability interval, not a confidence interval. Nonetheless, applied sciences generally use the term confidence interval together with an associated confidence level (), correctly probability level, that denotes the probability content of the probability interval between a pair of lower and an upper limit values, e.g., or .
In the following, we will concentrate on estimating the upper limit of Poisson probability intervals of the form
, as this upper limit
is required for concentration limit compliance testing.
Appendix C describes how lower and upper limits
can be estimated to specify a probability interval around the true fiber number. Different to the traditionally used limit
, we recommend using the lower limit
since the interval
exhibits the expected probability content
, cf.
Appendix D.3.
2.7.2. Upper Probability Interval Limit for Compliance Testing
A single-test upper limit
for a Poisson probability interval can be derived by performing a so-called left-tailed test of a single null hypothesis
. Our null hypothesis states that the true fiber number
to expect from sampling a specific volume
of a true airborne fiber concentration
exceeds a hypothetical fiber number
that would result from sampling a volume
that contains fibers with exposure limit concentration
, see
Table 7.
The probability of experimentally observing
or less fibers is called the
-value and is given by the cumulated probabilities to observe
fibers. At the so-called critical point, this
-value
starts to exceed a required nominal significance level
. This allows calculating the critical expectation value
that depends on
and
For a Poisson process, we find
More information on
, the
-quantile function, is given in
Appendix A.3. At the critical expectation value
, our null hypothesis
, which assumes the true value for the number of fibers
to exceed the critical value
, becomes insignificant at level
and can be rejected. The reason is that
, the probability to observe
or less fibers, decreases below
for any
that exceeds
, see
Figure 4. The remaining complement hypothesis
is thus accepted and infers that
is an upper limit on the true number of fibers
. Proper selection of such an upper limit by combining Equations (10) and (11) enables us to test compliance with exposure limit concentrations.
However, the insignificance of the null hypotheses on a level of
does mathematically not infer the complement hypotheses to be significant at a level of
. In
Appendix D.1 it is shown that the Poisson probability interval
indeed exhibits a probability level, also called confidence level
in applied sciences, of
. It is, therefore, justified to consider the complement hypothesis
probable with level
.
For the case of zero observed fibers,
Table 8 lists the minimum number of sampled fibers
that must be expected to keep the
-value of the observation below different nominal significance levels
. Whereas
Table 9 lists examples of the upper limit for zero or more observed fibers
at fixed significance
for the single
and twin
hypothesis testing in
Appendix C. Due to the Bonferroni correction, for identical upper limits a value the single test would require half the significance
of the twin test:
.
2.7.3. Limit Compliance Testing for Reducing the Workload
The single-test upper limit
from
Section 2.7.2 is central for reducing the workload of the proposed measurement strategy even further than is possible by imposing a cut-off diameter alone. For our intention of exposure limit control, we should initially not aim at estimating an absolute fiber concentration and its associated probability interval. The reason is that this concentration assessment requires analyzing filter areas which, at the exposure limit concentration
, let expect about 15 or more fibers. This number of observed fibers would allow obtaining a probability interval of about 100% relative size and below, cf.
Table A2 in
Appendix D.4. Instead, we should test whether the workplace concentration
does not exceed our limit-of-detection, as defined in
Appendix A.1, which was adjusted to be just equivalent to the exposure limit
. In this way, already for the case of observing zero fibers, exposure limit compliance can be tested, provided the probability of observing more than zero fibers is insignificant at a level of
.
For a Poisson process, this is the case if the fiber expectation value equals
, as discussed subsequently. Several such
fibers are expected on a filter area
that has sampled an air volume
containing exactly
fibers at the (hypothetically true) exposure limit concentration
. This compliance testing approach can also be generalized to cases in which actually more than zero fibers are observed. The fiber expectation value of the Poisson process must then be increased to
to render the probability of observing more than
fibers insignificant at a level of
. Consequently, the filter area
to analyze must have sampled the air volume
. with a deposition efficiency
at the exposure limit concentration
to be tested
Such an approach, using solely an estimate of the upper probability interval limit, is justified and sufficient, since the lower limit on the concentration estimate is obsolete when testing whether a concentration surmounts a specific exposure limit. However, it must be noted that the single-test upper limit
is actually lower than the twin-test upper limit,
, which was derived from a two-hypotheses test in
Appendix A, due to the different significance levels
and
. resulting from the Bonferroni correction, cf.
Appendix C. For exposure level compliance testing, it is, therefore, not correct to take
as the upper limit, since it corresponds to a significance of
for single hypothesis testing.
The algorithm to follow for our limit value compliance test strategy is schematized in
Section 2.9.
2.8. Online versus Offline Analysis
Traditionally, filter samples are inspected in electron microscopes by a workflow that will be called online in the following. A selected area of a filter is first imaged and then directly analyzed by the operator who recognizes fiber morphologies, and traces their full length and performs composition analysis by EDS before moving to the next filter area. That person must therefore be experienced in EM microscopy, EDS analysis and fiber categorization. The online approach is efficient if the fiber categorization expertise is frequently required directly at the microscope.
However, as
Table 4 shows, the number of images to acquire and analyze to identify presumably very few submicroscale fibers can become very large. Therefore, offline image inspection may be a more efficient approach if many images must be acquired before a fiber is finally encountered and requires categorization expertise. While the online approach saves images only for documentation purposes, the offline approach is solely based on stored images that are searched for fibers only after completion of a filter mapping run.
Table 10 illustrates different strategies for delegating different analysis steps to personnel of differing expertise and for distributing the workload to a team. It is a benefit of offline analysis that the microscope does not need to be operated by a fiber expert, but filter imaging can be performed by standard EM operators, or can even be automatized. Automated imaging can make the best use of (generally limited) microscope operation hours, e.g., by overnight acquisition, and can guarantee image acquisition at algorithmically or truly randomly chosen filter positions.
Likewise, the task of recognizing and localizing rare fibers in a huge amount of image pixel data can be performed by non-experts or even software. Only after localization of “something resembling a fiber” on a filter is its imaged morphology reviewed by a fiber expert who decides whether additional imaging or material analysis is required prior to final categorization and counting. This may be necessary if the fiber material needs to be identified and/or if a fiber’s length cannot be categorized since it is not fully contained in the image. This probability of a fiber to not be contained can be reduced by acquiring images extending over several tens of micrometers. For some fiber materials, chemical identification may not be necessary or possible, e.g., due to a very characteristic morphology, EDS-insensitive composition or a fiber volume insufficient for EDS analysis. Re-measurement may therefore become a rare necessity, which would increase the efficiency of the offline analysis workflow.
If additional EM-, EDS- or, e.g., Raman-based analyses of fiber candidates should be necessary, the filter sample must be re-loaded into the microscope and be re-aligned to the orientation that was used during the initial imaging run. In our experience, a high-accuracy EM stage together with three or more needlepoint marks in the outer rim of a filter can achieve an alignment reproducibility of a few micrometers [
34]. This is generally sufficient to re-address and recognize those previously stored, in order to be able to detect fibers with image frames several tens of micrometers in size.
To be able to visually recognize fibers with diameters down to the fiber cut-off value on a computer monitor, not only the pixel resolution during EM imaging must be chosen appropriately but also the display conditions. It is essential to perform the fiber search at pixel-accurate scaling, so that one image pixel corresponds to at least one monitor screen pixel. This requirement, however, restricts the online image size to the EM’s monitor screen size, currently to about 1600 × 1200 pixels. Images acquired for offline analysis can, which is a benefit, be significantly larger. However, such multi-megapixel images must be inspected at pixel-accurate zoom levels or higher to exploit the full information content and to be able to detect fibers just one or two pixels in diameter. Dedicated image visualization and annotation software can help to streamline the pixelwise inspection of filter areas, document the overall progress and store discovered fiber locations and snapshots of interesting objects. The object recognition reliability of algorithm- or machine learning-based image analysis techniques critically depends on the separability of fiber morphologies and filter surface structures. Especially recognition of complex, tangled fiber morphology is subject of on-going research [
35,
36].
2.9. Detailed Analysis Workflow
In
Section 2.1, the general analysis workflow was introduced. Here, in
Table 11, the filter evaluation part and result calculation is further detailed.
Single null hypothesis testing according to
Section 2.7.2 requires evaluation of a filter area
that expects to find
fibers at the tested limit concentration
. If the number
of observed fibers is less than or equal to the expected number
, the true fiber concentration
is in compliance with the limit at the probability (confidence) level
. The limit concentration
and confidence level
may be fixed by an implemented regulation or measurement guideline. The workload can then only be minimized by selecting the expected number
as zero.
Otherwise, the estimated upper limit
on the true fiber concentration
exceeds the compliance limit
. However, if only a few more fibers are observed (
Case B of
Table 11) than were expected, e.g.,
, additional work may be invested in
Loop 4 of
Table 11 for re-checking compliance with
by expecting no additional fibers to be found during evaluation of an extended filter area, which expects to find
fibers at concentration
. If really no additional fibers are found on the extended filter area, compliance with
can be stated based on a larger evaluated air volume.
5. Discussion
The proposed strategy for compliance checking concentrations of airborne nano- and microscale fibers was studied with a focus on practicability, a property that is mandatory for future implementation and routine application of new measurement concepts. At this stage, we did not aim at reporting shift-related, statistically sound exposure assessments, which would have required repeating, sampling and evaluating work tasks three or more times [
27,
41]. Testing compliance of workplace atmospheres with the benchmark concentration value of
that was published in the German TRGS 527 [
13] for WHO-analogue fibers down to
diameter is still a challenge due to the impracticably high analytical effort stated above. Even with our new approach, this limit is currently only routinely testable if very thin nanofibers below 10–20 nm diameter are excluded from filter imaging and evaluation. In
Section 2.5.2, toxicological and practical justifications were given for ignoring the fraction thinner than
. This enabled us to test compliance with exposure levels of
here. Please note that our approach has no limitations, in principle, with respect to the included diameter range. The applied cut-off diameter can and certainly will be lowered with progress in further automating SEM image acquisition and evaluation. Currently, however, the
cut-off is crucial for rendering our testing strategy practicable and makes it the starting point for routinely monitoring compliance with fiber number-based concentration limits for airborne submicroscale fibers.
The most common significance level used in occupational health and safety is
, i.e.,
[
23,
24,
25,
26]. With the aim of estimating a probability interval around the true fiber number
from an observation, different to the traditionally used lower limit
, we recommend using the somewhat larger limit
, since for
only the interval
with
, and not the interval
, exhibits the assumed probability content
, cf.
Appendix D.3. It should also be noted that when deriving both upper and lower limits
, the significance level
is divided by two according to the Bonferroni correction, cf.
Appendix C. This is not the case for single hypothesis compliance testing with the upper limit
. Thus, at the same significance level
, the values of the upper limits derived from single
and twin hypothesis testing
will differ. To avoid this possible source of confusion, we recommend performing compliance testing with
at a significance level of
and upper and lower limit estimation
at a level of
, i.e., to effectively undo the Bonferroni correction. In this way, identical values for the upper limits
are obtained that, however, exhibit different significance levels
and
. Considering the efficiency gain of our compliance testing approach, an increased significance level of
appears acceptable. The required increase of the filter area to evaluate for zero expected fibers is given in
Table 8.
Our measurement strategy was successfully conducted at 14 workplaces by sampling and evaluating a total of 93 air samples.
Table 14 compiles information on the workplaces and sampling parameters and reports fiber counting results for the nearfield filter samples and the corresponding upper limit concentration estimates. Since microscale fibers were already included in the image evaluation rules, the analysis of the workplace samples WP13 and WP14 performed with our nanofiber-sensitive strategy revealed that a purely microscale fiber fraction caused the observed violation of the exposure limit value for
-fibers [
37]. For workplaces in which no
product fibers were observed in the nearfield, no product fibers were found on personal or farfield samples, either. Whereas at workplaces WP02 and WP13, where the fiber concentration exceeded
, the personal and farfield samples also showed an excess in fibers, see
Section 4.3. This supports the assumed validity of our sampling and evaluation approach.
Please note that our measurements determined process-related, not shift-related, concentrations by sampling only during specific work tasks involving the processing of fiber-containing materials, whereas established exposure limits are generally defined as shift-related (eight hours) averaged values. Thus, extrapolation to shift values may be necessary. More results of this measurement campaign have been or will be reported in dedicated workplace- and material-related publications [
37,
40].
The decision as to whether fibers observed on a filter were released from materials handled or machined at the workplace is crucial for deriving airborne concentrations of product fiber and for assigning occupational safety responsibilities. For the present study, both pristine product fiber and background aerosol samples were analyzed with SEM to identify characteristic morphological features that allow distinguishing release of product from background fibers. However, since morphology alone does not always allow unambiguous identification, additional information on elemental composition should be gained from, e.g., EDS. In our case of carbon-based materials, EDS was not appropriate to distinguish carbon fibers from polymer or cellulose background fibers. Raman or FTIR spectroscopy should therefore be applied to identify chemical and structural differences of fibers located on a filter. By spatial correlation of SEM and Raman microscopic analyses, we were able to identify different CNT materials and carbon fiber fragments and to reliably distinguish them from background fibers.
Not only the identification of fibers, but also their categorization, can be difficult. An inter-laboratory comparison that applied the counting rules proposed here to highly occupied filter samples revealed differences in the categorization of (1) individual self-crossing fibers, (2) pair- or group-wise crossing fibers, or (3) loosely agglomerated fibers. Especially the classification decision for fiber-containing objects as (A) individually countable fibers or (B) non-countable fiber agglomerates performed by different human evaluators led to significant fiber counting results [
42]. The workplace samples examined here showed very low occupancy rates compared to previous samples of the inter-laboratory comparison. Therefore, the subjectivity of human evaluators in the assessment of single fibers only becomes relevant if fibrous objects are observed at all. For compliance checking an exposure limit of potentially hazardous fibers at the limit-of-detection, any ambiguity in categorizing tangled or crossed fibers as countable should be resolved in favor of countability. This increases, and may even overestimate, fiber counts, which is preferable to underestimation, as it promotes protection of exposed workers.
A single compliance check of an aerosol sample for an concentration benchmark value of
according to the proposed method requires the analysis of 40–180 SEM images at
pixels with 0.8–3.5 gigapixels, typically at
pixel resolution, to reach or surpass the required limit-of-detection sensitivity that, as defined in
Appendix A.1, is necessary for null hypothesis testing. Due to the offline analysis approach using SEM images that were generally acquired automatically overnight, the subsequent workload could be distributed to several human evaluators. The workflow currently consumes 1.5–6 h of SEM time plus 2–9 man hours of image evaluation time per filter sample. For offline analysis, archiving position-calibrated filter samples is mandatory to be able to re-address product fiber candidates in both the SEM and Raman microscope for a second in-depth material analysis or for tracking long fibers that were not fully contained in the initially taken SEM image.
6. Conclusions
Despite compelling reports on asbestos-like pathogenicity, regulatory bodies have thus far been hesitant to implement fiber number-based exposure limits for biodurable nanoscale fibers. One reason has been a lack of a practicable strategy for assessing airborne fiber number concentrations, since inclusion of nanofibers in the range of 200–1 nm causes a -fold increase in the analytical workload compared to that for -fiber number concentration determination.
The goal of the present work was to lay out a proposed compliance test strategy in detail and to study its practicability. Confirming practicability is the mandatory first step in implementing and routinely applying a new measurement concept. The 14 studied workplaces and the 93 evaluated filter samples allowed us to optimize the analysis workflow with respect to imaging and evaluation conditions and to help colleagues who study MWCNT-containing materials, mostly in research institutes, to obtain a first preliminary assessment of the effectiveness of their safety measures. At this early stage, we did not aim at reporting shift-related, statistically sound exposure assessments that would have required our partners to repeat their work tasks at least three times on consecutive days.
The large number of processed samples demonstrates that the proposed measurement strategy can indeed be employed for compliance testing concentrations of airborne fibers of possible toxicological concern with a manageable workload. Analogous to established methods for -fiber fractions, our strategy includes micro- and submicroscale airborne fibers only down to a so-called critical diameter in the nanoscale. The cut-off diameter of about proposed here was motivated by toxicological data on MWCNTs and should, when adapted to other fiber materials, be adapted considering the extended fiber pathogenicity paradigm. The extended paradigm introduced a critical fiber rigidity which is considered responsible for asbestos-like effects of respirable biodurable WHO-analogue fibers in lung tissue.
For aerosols containing nanoscale fibers, this cut-off diameter is currently the most relevant step for arriving at practically manageable workloads. Thus, ignoring nanofibers of potentially lower toxicological relevance in the diameter range of 1–20 nm allows the reduction of the workload by a factor of . Additional workload reduction results from not aspiring to absolutely quantify fiber concentrations but to work at the limit-of-detection right from the start. For such fiber non-appearance testing, the fiber sampling and detection process was assumed to be governed by Poisson statistics. The fiber counting result on a filter area, which was carefully adjusted to a required confidence level and a specific exposure limit value, either confirms or rejects the hypothesis of compliance to an upper limit on the true fiber concentration.
As set out in the Appendix, in the case of sparse fiber observations, as is typical for testing low concentration limits, testing compliance with an upper limit requires the analysis of filter areas considerably smaller than those required for estimating true fiber concentrations, cf.
Appendix D.4. We therefore recommend starting with performing upper limit compliance testing and to only report an estimate for the true concentration
if the observed number of fibers
is large enough to result in a small probability interval that actually constrains the estimate in a practically valuable manner. To avoid different values for the upper limit from compliance testing
and confidence interval limits estimation
, we recommend performing compliance testing at a significance level of
, and confidence interval limit estimation at a level of
. This effectively undoes the typically applied Bonferroni correction, cf.
Appendix C, and results in identical values of differing statistical significance.
We conclude that the application of the proposed testing strategy for nano- and microscale fibers to 14 very different workplaces showed the resulting workload to be high but well manageable. Our approach thus enables the occupational health community to routinely control fiber number-based concentration limits at a level of for nano- and microscale fibers with diameters of and above. Automated microscopes, re-addressable, position-calibrated filter samples allow performing the image evaluation in so-called offline mode. This makes it possible to distribute the task of visually searching for fibers on EM images with a total of about pixels per sampling to trained personnel, thus keeping the EM operator free to perform the imaging. Our workflow currently requires 1.5–6 h of SEM time plus 2–9 man hours of image evaluation time per filter sample. Progress in the degree of automation of image acquisition and object recognition is already underway and will not only further lower the costs of analysis and render the proposed strategy a standard service for workplace exposure assessment but will also allow including fibers thinner than in an evaluation in the near future.