Abstract
Capuchins are platyrrhines (monkeys found in the Americas) within the Cebidae family. For most of their taxonomic history, the two main morphological types of capuchins, gracile (untufted) and robust (tufted), were assigned to a single genus, Cebus. Further, all tufted capuchins were assigned to a single species, Cebus apella, despite broad geographic ranges spanning Central and northern South America. In 2012, tufted capuchins were assigned to their genus, Sapajus, with eight currently recognized species and five Cebus species, although these numbers are still under debate. Alu retrotransposons are a class of mobile element insertion (MEI) widely used to study primate phylogenetics. However, Alu elements have rarely been used to study capuchins. Recent genome-level assemblies for capuchins (Cebus imitator; [Cebus_imitator_1.0] and Sapajus apella [GSC_monkey_1.0]) facilitated large scale ascertainment of young lineage-specific Alu insertions. Reported here are 1607 capuchin specific and 678 Sapajus specific Alu insertions along with candidate oligonucleotides for locus-specific PCR assays for many elements. PCR analyses identified 104 genus level and 51 species level Alu insertion polymorphisms. The Alu datasets reported in this study provide a valuable resource that will assist in the classification of archival samples lacking phenotypic data and for the study of capuchin phylogenetic relationships.
1. Introduction
Capuchins constitute a monophyletic clade of platyrrhines (monkeys found in the Americas) within the Cebidae family, having an estimated origin of about 6.8 million years ago (mya) [1,2]. Capuchins diverged from their most closely related sister taxon within Cebidae, the squirrel monkeys (genus Saimiri) approximately 13.8 mya [1]. Recognized in popular folklore as the ‘organ grinder’ monkey or Cebus monkey, capuchins comprise a diverse group of morphologically and phenotypically distinct members, distributed across a broad geographic range of Central and northern South America. The current consensus of capuchin systematics is that they are represented by two genera, the Cebus genus, with at least five species, Cebus albifrons, Cebus capucinus, Cebus imitator, Cebus olivaceus and Cebus kaapori [2,3,4] and the Sapajus genus, with eight recognized species, Sapajus xanthosternos, Sapajus nigritus, Sapajus robustus, Sapajus flavius, Sapajus libidinosus, Sapajus cay, Sapajus apella, and Sapajus macrocephalus [2,5]. The geographic distribution of capuchins across maps of South and Central America is nicely illustrated in Lynch Alfaro et al. [6] and Martins-Junior et al. [2].
However, the taxonomy of capuchin monkeys has traditionally not been this refined. Historically, it has been confusing due to changing nomenclature as well as periodic reassessments of the number of species and subspecies [3,7,8]. Early taxonomists grouped all capuchins into a single genus, Cebus, even though morphological differences suggested two distinct types [5] that were later categorized as tufted and untufted, or robust and gracile, respectively [1]. Further, the name Cebus apella was used to encompass all tufted (robust) capuchins into a single species, a nomenclature that remained widely used until the recent decade. Alfaro et al. 2012 [1] proposed the use of Sapajus for tufted (brown; robust) capuchins and suggested that the genus Cebus be restricted to only the untufted (gracile) capuchins. A roundtable of capuchin researchers held at the International Primatological Society Congress in 2012 adopted the use of the term Sapajus as an urgent research priority, calling for the immediate end of the name Cebus apella [9].
Photographic images of tufted and untufted capuchins are shown in Figure 1. Additional illustrations of variations in facial phenotypes among several capuchin species are available in Alfaro et al. [1]. Despite obvious phenotypic distinction between the two general forms, and widespread diversity among various species, the confusing historical nomenclature has created a situation in which most archival tissue and DNA samples for the brown tufted capuchins are labeled as Cebus apella or just Cebus monkey. Those for untufted gracile capuchins are also labeled Cebus monkey. This makes it difficult to determine the actual taxonomic origin when phenotypic descriptions are unavailable. In addition, some species in both genera are considered Endangered such as S. robustus [10], or Critically Endangered such as C. kaapori [11], making it essential to have reliable taxonomic, genetic markers for effective conservation efforts. A recent study conducted on populations of S. libidinosus, a species considered “Near Threatened” [12], emphasized that there is currently a lack of developed genetic systems available to study capuchin population and conservation genetics [13].

Figure 1.
Photographs representative of untufted and tufted capuchins. (A) C. imitator, an untufted, adult male Panamanian white-faced capuchin. Photograph by Amanda Melin. (B) S. apella, a tufted, brown capuchin [14].
Primate specific Alu retrotransposons are well-established diagnostic genetic markers for the study of population genetic and phylogenetic relationships [15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31]. Non-autonomous Alu elements mobilize via a “copy and paste” mechanism through an RNA intermediate, utilizing the enzymatic machinery of autonomous LINE (L1) elements. This mode of mobilization is termed “target-primed reverse transcription” (TPRT) [32]. The TPRT integration process produces 5′ and 3′ flanking target site duplications (TSDs) that can be used to identify each insertion. TPRT is considered a unidirectional mode of integration such that the ancestral state is considered the absence of an insertion, and shared insertions with matching TSDs are accepted as being inherited from a common ancestor. Alu subfamilies evolve by the stepwise accumulation of diagnostic nucleotide arrangements such that each primate lineage derives a unique group of Alu subfamilies [33,34,35,36,37,38]. The oldest subfamily, AluJ, is found in all primates, whereas AluS was active after the separation of Strepsirrhini and Tarsiiformes from Platyrrhini and Catarrhini [36,37], and subfamily AluY is found only in catarrhines [33]. Platyrrhine-specific Alu element subfamilies include AluTa7, AluTa10, and AluTa15, with Ta15 thought to be limited to the Cebidae family [21]. More recently, other platyrrhine Alu subfamilies have been characterized in marmoset [39], squirrel monkey [40], capuchin, and owl monkeys [41,42]. Previous studies have utilized Alu elements in platyrrhine phylogeny [15,31,43,44,45], and although capuchin monkey studies have been conducted using Alu insertions, they involved a relatively small number of elements [44,46]. Recently, genome-level assemblies have become available for capuchins (C. imitator; [Cebus_imitator-1.0] and S. apella [GSC_monkey_1.0]). This study aimed to computationally ascertain a dataset of the youngest Alu insertions specific to these capuchin lineages and to perform locus-specific PCR on a subset of each to identify Alu insertion polymorphisms. The Alu datasets reported in this study provide a valuable resource that will assist in the classification of archival samples and facilitate future studies of capuchin phylogeny, populations genetics, and conservation strategies.
2. Materials and Methods
2.1. Lineage-Specific Alu Elements
Ascertainment of lineage-specific Alu insertions from the C. imitator genome [Cebus_imitator-1.0] [47] was performed as previously described [41,42]. Briefly, the [Cebus_imitator-1.0] genome was obtained from the National Center for Biotechnology Information (NCBI) and analyzed for full-length Alu elements with RepeatMasker [48] (RepeatMasker-Open-4.0). Full-length Alu elements are described as having a start site within 4 bp of its consensus sequence and being 267 bp or longer. A sequential BLAT [49] of the full-length Alu elements extracted from RepeatMasker was conducted with human (Homo sapiens; GRCh38.p13), common marmoset (Callithrix jacchus; caljac3), owl monkey (Aotus nancymaae; Anan_2.0), and squirrel monkey (Saimiri boliviensis; SaiBol1.0) genomes in that order. A sequential BLAT involved analyzing the output after each BLAT for capuchin-specific Alu elements compared to the other four genomes. The genome assembly for S. apella, [GSC_monkey_1.0] was not available at that time. Capuchin lineage-specific Alu insertions were aligned using Crossmatch (www.phrap.org/phredphrapconsed.html; accessed on 4 March 2022), and Alu subfamily structure was determined using COSEG (www.repeatmasker.org/COSEGDownload.html; accessed on 4 March 2022).
The percent divergence of the lineage-specific Alu elements compared to their respective consensus sequences was determined using an in-house installation of RepeatMasker. Young elements, defined as having less than or equal to two percent sequence divergence, were retained. A custom Python script extracted the FASTA files of the young, lineage-specific elements and sorted them into subfamilies. Five elements for each identified Alu subfamily were randomly selected for wet-bench experimental validation. If an Alu subfamily had less than five elements, then all lineage-specific Alu elements for that subfamily were selected for experimental validation. The orthologous sequence and 600 bp of flanking sequences for human, marmoset, squirrel monkey, and owl monkey genomes were obtained for each Alu element using BLAT. BioEdit was used to align the four sequences for each locus [50].
The genome assembly for S. apella, [GSC_monkey_1.0], became available more recently, and therefore, ascertainment of the lineage-specific Alu insertions from this genome was conducted under consideration of the existing C. imitator genome. Full-length Alu elements from the Sapajus genome were first filtered against the C. imitator genome using BLAT, greatly reducing the number of candidates. The output was then filtered using a custom Python script (available on link https://github.com/t-beck; accessed on 4 March 2022) to BLAT the genomes of human, C. imitator, marmoset, owl monkey, and squirrel monkey in a single program. These putatively Sapajus lineage-specific Alu elements, plus 600 bp flanking sequence, were then analyzed using an in-house RepeatMasker library and filtered by position near the center of the FASTA sequence (i.e., 600 bp) to represent the target insertion. Young insertions (≤2% divergence) were retained.
2.2. Oligonucleotide Primer Design
Oligonucleotide primers for the polymerase chain reaction (PCR) for Cebus Alu elements were designed using Primer3 software [51] with the following modifications: Tm range = 57–63, Max Tm difference = 2, max poly x = 3, min GC content = 40. NCBI Primer Blast [52] was used to analyze the primers for Cebus specificity and verify the predicted PCR fragment lengths for Cebus and outgroup genomes. For Sapajus lineage-specific Alu elements, oligonucleotide primers for PCR were designed using an in-house primer design pipeline consisting of a series of custom Python scripts in conjunction with Primer3 (available on link https://github.com/t-beck; accessed on 4 March 2022), followed by screening using NCBI Primer Blast [47]. The oligonucleotide primers were obtained from Sigma Aldrich (Woodlands, TX, USA).
2.3. DNA Samples
DNA samples and their origins are described in Supplementary Files S1 and S2. DNA from tissue samples was prepared as described previously [15]. There were two DNA sample panels used in this study, the capuchin monkey panel and the Sapajus panel. The capuchin monkey panel included DNA from 14 different capuchin monkeys, six individuals from genus Cebus, including the C. imitator (sample Cc_AM_T3) used in the reference genome, and eight S. apella individuals, all obtained with the original designation ‘Cebus apella’ (Supplementary File S1). The Sapajus DNA panel included three additional S. apella individuals (Supplementary File S2), who were originally labeled as ‘Cebus monkeys’ when acquired initially and were only recently determined to be brown tufted capuchins [53,54].
2.4. PCR Amplification
Each DNA sample panel for PCR included a negative control (TLE: 10 mM Tris/0.1 mM EDTA) and four outgroup controls, human (HeLa), the common marmoset (Callithrix jacchus), squirrel monkey (Saimiri sciureus), and owl monkey (Aotus trivirgatus) representing the pre-integration site, or Alu absent PCR amplicon. PCR amplifications were performed in 25 μL reactions containing 25 ng of template DNA, 200 nM of each oligonucleotide primer, 1.5 mM MgCl2, 10 × PCR buffer (1×:50 mM KCl; 10 mM TrisHCl, pH 8.4), 0.2 mM dNTPs, and 1–2 U Taq DNA polymerase. PCR reactions were performed under the following conditions: initial denaturation at 94 °C for 60 s, followed by 32 cycles of denaturation at 94 °C for 30 s, 30 s at 57 °C annealing temperature, and extension at 72 °C for 30 s. PCRs were completed with a final extension at 72 °C for 2 min. 20 μL of each PCR product were fractionated by size in a horizontal gel chamber on a 2% agarose gel containing 0.2 μg/mL ethidium bromide for 60 minutes at 175 V. UV-fluorescence was used to visualize the DNA fragments, and images were saved using a BioRad ChemiDoc XRS imaging system (Hercules, CA, USA). If PCR results were weak or unresolved, the PCR reaction was repeated using a hot-start with the JumpStart Taq DNA polymerase kit (Sigma Aldrich, Woodlands, TX, USA). Following gel electrophoresis, genotypes were recorded in an Excel spreadsheet as (1, 1) for homozygous present, (0, 0) homozygous absent, or (1, 0) heterozygous. “Missing data” was coded as (−9, −9) (Supplementary Files S1 and S2; “Genotypes” worksheet).
2.5. Sanger Chain Termination DNA Sequencing
We used traditional Sanger DNA sequencing [55] to resolve certain cases of ambiguity that arose during PCR analyses. Sequence analysis is the best way to avoid erroneous interpretation of PCR patterns. In one Cebus locus, some Alu present PCR amplicons occurred among S. apella samples for an Alu absent from the non-reference genome S. apella, [GSC_monkey_1.0]. This was sequenced to confirm a shared Alu insertion. In four Sapajus loci, an Alu present PCR amplicon was obtained in a Cebus sample, although the Alu was absent from that corresponding non-reference genome [Cebus_imitator-1.0]. These were analyzed by Sanger DNA sequencing to confirm a shared insertion or to identify a different Alu element in the same region (a near-parallel insertion). If an ambiguous PCR amplicon involved a C. albifrons DNA sample, then the Cebus albifrons (white-fronted capuchin) (GCA_004027755.1) genome assembly [CebAlb_V1_BIUU] was analyzed for comparison. Sanger DNA sequencing was performed as described previously [56]. Briefly, four PCR fragments per locus were gel purified using a Wizard SV gel purification kit (Promega Corporation, Madison, WI, catalog A9282), 4 µL was used for chain termination cycle sequencing using BigDye Terminator v3.1. Four separate reactions were conducted for each locus using forward or reverse PCR primer; internal-Alu primer IntF1: 5′ GGTGGCTCACGCCTGTAATC 3′ [56] and SIntR1: 5′ TCTCGGCTCACCGCAACCTCC 3′ [15]. Following capillary electrophoresis, sequence quality was evaluated using ABI software Sequence Scanner v2.0. Sequence alignments were constructed in BioEdit [50].
3. Results
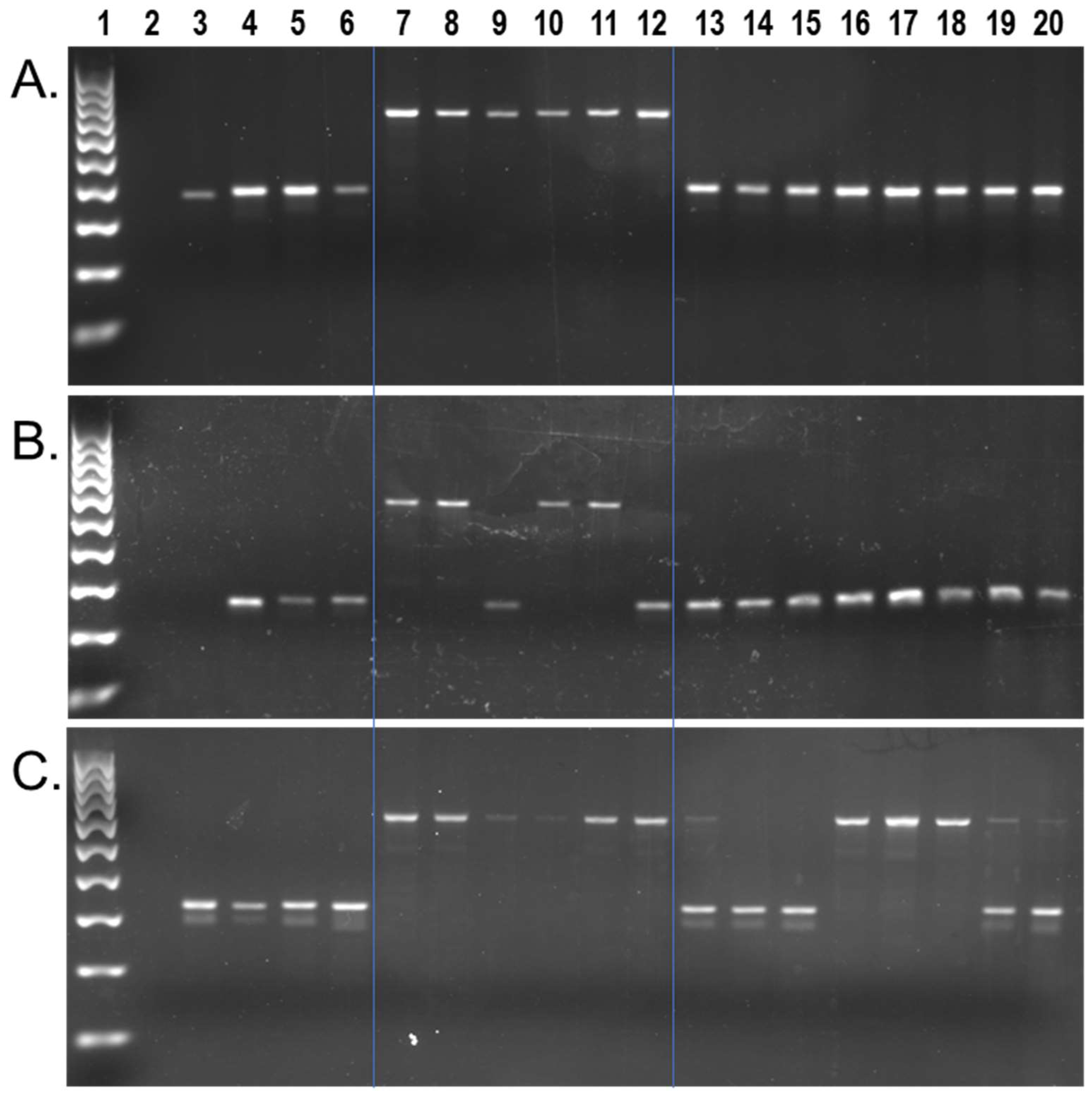
We found approximately 9602 capuchin lineage-specific Alu insertions in the [Cebus_imitator-1.0] genome, from a total of 617,132 full-length insertions [42]. We identified 1607 of these as young (≤2% sequence divergence from their consensus). Local RepeatMasker [48] (RepeatMasker-Open-4.0) output for these loci, along with their genome coordinates and Alu subfamily designation are shown in Supplementary File S1. Wet bench locus-specific PCR analyses for a subset of n = 132 young insertions, representing 29 different Alu subfamilies, identified n = 84 Alu elements that are polymorphic among capuchins on our DNA sample panel. There were n = 54 that were homozygous present in all Cebus species individuals while being absent from Sapajus samples (Figure 2a), n = 29 that were polymorphic for insertion presence/absence within Cebus species samples from C. imitator, C. capucinus and C. albifrons, while absent from Sapajus samples (Figure 2b), and n = 1 insertion found to be polymorphic among eight Sapajus individuals while being homozygous present in all Cebus species individuals (Figure 2c). PCR primer sequences, their predicted amplicon sizes, and the resulting genotypes are available in Supplementary File S1.
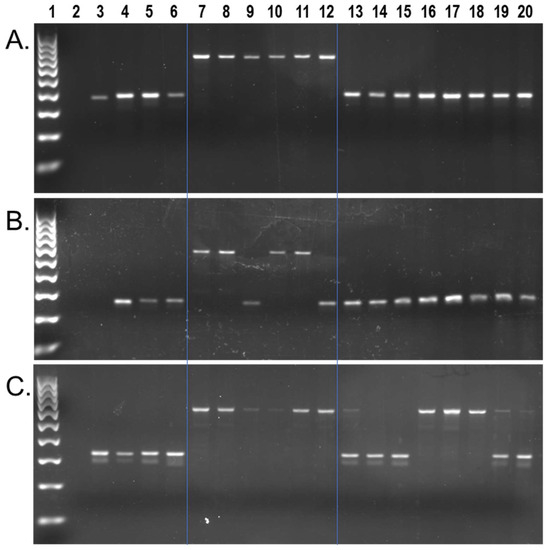
Figure 2.
C. imitator genomic Alu insertion polymorphisms. Lanes: 1 100 bp DNA ladder, 2 TLE (negative control), 3 Human (HeLa), 4 Callithrix jacchus (Common marmoset), 5 Saimiri s. sciureus (Common squirrel monkey), 6 Aotus trivirgatus (Three striped owl monkey), 7 C. imitator, 8–9 C. capucinus, 10–11 C. albifrons, 12 C. a. albifrons, 13–20 S. apella. (A) Locus #69, Alu is present in all Cebus species individuals (~693 bp DNA fragment lanes 7–12) and absent in all Sapajus samples (~381 bp DNA fragment lanes 13–20); (B) Locus #49, Alu is polymorphic among Cebus individuals (~580 bp fragment present, ~275 bp fragment absent) and absent in all Sapajus samples; (C) Locus #9, Alu is present in Cebus individuals (~653 bp fragment) and polymorphic among Sapajus individuals (~653 bp, and ~340 bp DNA fragments). Blue lines superimposed on gel images visually separate Cebus, Sapajus, and outgroups.
To ensure that the locus shown in Figure 2c represented the same Cebus-ascertained locus and not a near-parallel insertion, we first constructed a sequence alignment using homologous regions from the S. apella genome [GSC_monkey_1.0] and the squirrel monkey genome, genus Saimiri, [SaiBol1.0], both of which aligned with the pre-integration site, absent the Alu. Therefore, we performed Sanger DNA sequencing on the Alu present PCR amplicon from S. apella (UAM-46596) and aligned these results to confirm the presence of a shared insertion (Supplementary File S3). The finding of a relatively young Alu insertion from genus Cebus that is polymorphic within genus Sapajus is unexpected but not impossible given that the Cebus dataset was not filtered against [GSC_monkey_1.0] and the integration of this locus occurred prior to the Cebus/Sapajus divergence.
If these experimental results are extrapolated, 30 insertion polymorphisms identified among 132 analyzed by PCR correspond to a Cebus species level insertion polymorphism rate of 22.7%, suggesting that there are potentially over 350 insertion polymorphisms available within this dataset of 1607 young Alu elements. However, our relatively small DNA sample size containing only a few species limits the utility of further PCR for this study. Alternatively, we provide oligonucleotide PCR primer sequences for an additional 632 young candidate loci employed by other research groups with access to a larger number of species and individuals. These oligonucleotides for PCR and their predicted amplicon sizes for Cebus and closely related outgroups are available in Supplementary File S1.
The S. apella [GSC_monkey_1.0] genome assembly (first available in 2019) was analyzed for full-length Alu elements, and the output was then filtered against the [Cebus_imitator-1.0] genome using BLAT [49], resulting in 29,554 putatively Sapajus lineage-specific Alu elements. This output was subsequently filtered using a custom Python script (available on link https://github.com/t-beck; accessed on 4 March 2022) to BLAT the genomes of human, C. imitator, marmoset, owl monkey, and squirrel monkey in a single program, resulting in 8135 Alu elements lineage-specific to the S. apella genome. We identified 1170 of these as young (≤2% sequence divergence from their consensus). The local RepeatMasker output was further screened to ensure the position of the Alu was near the center (i.e., 600 bp) of the target sequence to represent the intended insertion, producing our reported dataset of 678 young Alu insertions, representing 20 different Alu subfamilies, for PCR. Genomic coordinates for these loci are shown in Supplementary File S2. We obtained oligonucleotides for PCR for 214 of these candidate loci. Following assessment by NCBI Primer Blast [52], we report oligonucleotide primer pairs along with their predicted amplicon sizes for 110 Sapajus Alu insertions (Supplementary File S2).
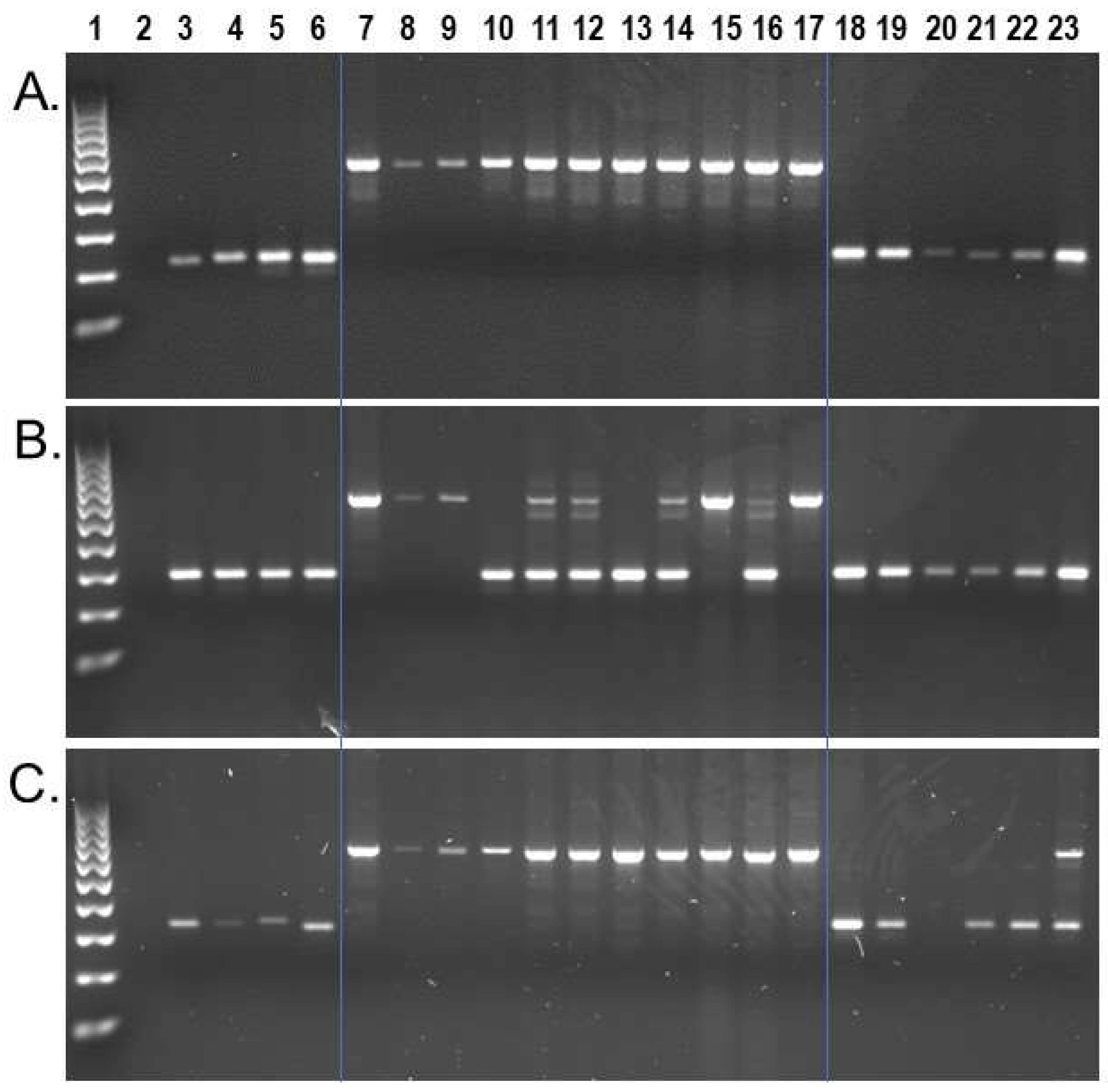
Wet bench locus-specific PCR analyses for a subset of n = 74 identified n = 50 as homozygous present in all Sapajus samples, while homozygous absent in all Cebus samples on our DNA sample panel (Figure 3a). An additional n = 19 displayed polymorphic patterns for insertion presence/absence among the eleven S. apella samples while remaining homozygous absent in all Cebus samples (Figure 3b). Interestingly, we also identified two Alu insertions that, while being homozygous present in all eleven S. apella samples, were also confirmed to be shared by C. albifrons sample KB-4207. Locus #944.49 (Figure 3c) was confirmed to be the same as the target Alu by Sanger chain-termination DNA sequencing, and locus #961.24 was confirmed to have the same Alu insertion present in the C. albifrons genome assembly, [CebAlb_V1_BIUU] (Supplementary File S3). Both insertions are absent in [Cebus_imitator-1.0] and other Cebus species analyzed. A third locus, #993.63, displayed a similar gel electrophoresis pattern to locus #944.49; however, in this case, Sanger DNA sequencing revealed that the Alu present PCR amplicon in sample KB-4207, C. albifrons, represented a different Alu element ~70 bp away from the target insertion. We identified a second incidence of a ‘near-parallel insertion’ in locus #954.16, in which PCR results implied that the Sapajus target Alu insertion was also homozygous present in two Cebus samples, Cc_AM_T3 used for the C. imitator reference genome and C. capucinus sample UF-31995. In this case, Sanger DNA sequencing revealed that the two Cebus samples contained a different Alu element ~100 bp away and in the opposite orientation from the target insertion (Supplementary File S3). The homologous region of the C. imitator genome for this locus is located on an unplaced genomic scaffold and displays the Alu absent pre-integration sequence. Sequence alignments are shown in Supplementary File S3. Only one locus of the n = 74 analyzed by PCR, locus #942.22, also located on an unplaced genomic scaffold in [Cebus_imitator-1.0], appears shared by all capuchins on our DNA sample panel, indicating that our strategy to select Sapajus specific insertions was generally effective.
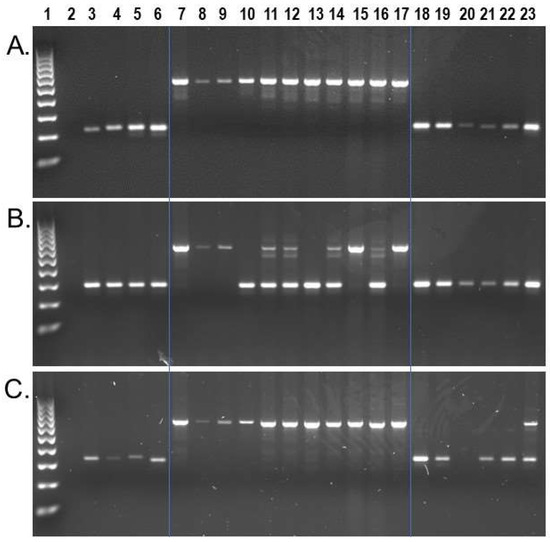
Figure 3.
S. apella genomic Alu insertion polymorphisms. Lanes: 1 100 bp DNA ladder, 2 TLE (negative control), 3 Human (HeLa), 4 C. jacchus (Common marmoset), 5 S. s. sciureus (Common squirrel monkey), 6 A. trivirgatus (Three striped owl monkey), 7–17 S. apella, 18 C. imitator, 19–20 C. capucinus, 21–22 C. albifrons, 23 C. a. albifrons. (A) Locus # 984.99, the Alu element is present in all Sapajus individuals (~528 bp) and absent in all Cebus samples (~227 bp fragment); (B) Locus #978.25, the Alu element is polymorphic among Sapajus individuals (~604 bp and ~292 bp DNA fragments) and absent in Cebus samples; (C) Locus #944.49, the Alu element is present in all Sapajus individuals (~671 bp fragment), and heterozygous present (~671 bp and ~341 bp DNA fragments) in individual KB-4207, C. a. albifrons. Blue lines superimposed on gel images visually separate Cebus, Sapajus and outgroups.
The combined experimental PCR data for these capuchin Alu datasets provides 104 insertion polymorphisms informative at the genus level as either Cebus-indicative (n = 54) or Sapajus-indicative (n = 50) genetic markers. Because of their high allele frequency for the respective genus, these genus indicative Alu elements could assist in clarifying the identity of archival specimens that may have been labeled simply as ‘Cebus monkey’ prior to the use of more refined taxonomic designations. They also provide strong support for Cebus and Sapajus being genetically separate genera, given that ~40% of the capuchin lineage-specific Alu insertions ascertained from the C. imitator genome were completely absent from Sapajus samples. PCR validation experiments provide an additional 51 Alu insertion polymorphisms potentially informative for capuchin phylogeny, with intermediate allele frequency among Cebus species (n = 29 loci), among Sapajus individuals (n = 19 loci), or perhaps both (n = 3). Cebus_locus #9 and Sapajus loci #944.49 and #961.24 integrated into the capuchin genome prior to the divergence of the two genera but remained variable for insertion presence/absence across taxa. The polymorphism rate within the Alu datasets reported here is ~25%, providing potentially hundreds of additional insertion polymorphisms to study capuchin phylogeny and conservation genetics.
4. Discussion
This study provides an extensive dataset of recently integrated Alu mobile elements in the capuchin lineage and demonstrates their phylogenetically diagnostic utility. Even with the large number of Alu insertion polymorphism reported here, these data are still subject to some ascertainment bias towards the two genome assemblies analyzed. The Alu elements ascertained from the [Cebus_imitator-1.0] genome were not computationally filtered against the genome assembly for S. apella, [GSC_monkey_1.0] and therefore were expected to be more broadly represented among capuchins. However, roughly 40% of those analyzed by PCR were absent in all S. apella samples. This provides strong support for the division of capuchins into the two genera as proposed by Lynch Alfaro et al. [1] and bolstered by Martins et al. [46]. It also implies some degree of reproductive restriction during their history, leading to genus level monophyly despite large areas of sympatry [6].
The diversification of capuchins into two separate lineages is believed to have started with Sapajus capuchins first, about 2.7 mya, and then slightly later for Cebus at around 2.1 mya [1], while the radiation of extant capuchin species has occurred relatively recently in the last 1–2 mya [2]. Studies of capuchin phylogenetic relationships using mitochondrial DNA [2,3] generally support these divergence estimates but find only limited support for any single topology of phylogenetic relationships within each genus. A study of Sapajus phylogeny [5] using ultraconserved elements (UCEs) [57] showed strong support for S. xanthosternos, S. nigritus, and S. robustus as defined branches within the clade, but all other species of robust capuchins grouped together. These presently unresolved polytomies within capuchin phylogeny are largely attributed to an extremely rapid speciation and dispersal process that occurred 2–3 mya [2,6]. Such events can lead to incomplete lineage sorting (ILS) of phylogenomic markers that remained unfixed in the population during speciation and later become randomly fixed or extinct in emerging species [58,59].
Contrasting evidence to genus level monophyly and a surprising finding of this study was the discovery of two independent Alu insertions shared by members of S. apella and C. albifrons, to the exclusion of [Cebus_imitator-1.0] and C. capucinus. One of these was confirmed to be shared in the C. albifrons genome assembly [CebAlb_V1_BIUU], eliminating the possibility that this result was due to sample mix-up. The other was validated by DNA sequencing. These data suggest that S. apella and C. albifrons are phylogenetically more closely related to each other than S. apella is to the other Cebus species. This conclusion is unlikely to be true, given the morphological differences and that no studies to date provide substantiating evidence in support of this relationship. This situation could result from ILS as described above or possibly due to introgression. A possible hybrid zone has been reported for S. robustus and S. nigritus [60], and others are suspected to exist in many contact zones between multiple species [2]. Known populations of C. albifrons and S. apella are sympatric in the North Amazon [2], contributing to this possibility. Data for these two Alu insertion polymorphisms from a much larger sample size comprised of representatives of all capuchin species are required to resolve this issue.
This example of a confounding topology highlights the primary limitation of this study. The DNA sample panels used in this study were relatively small, both in the number of individuals and the number of species available. They did not include any samples for two Cebus species, C. kaapori and C. olivaceus. In addition, the only robust capuchin species represented was S. apella, and DNA sample panels lacked the other seven recognized Sapajus species. However, given that all Sapajus samples were acquired having the label ‘Cebus apella’ and the name Cebus apella was universally used for all robust capuchins until recently, it is possible that some samples could be from other Sapajus species that lacked independent designations at the time of collection. This scenario would create an over-estimate of the genetic variance within S. apella from these data. Furthermore, only one DNA sample (Cc_AM_T3) was originally from a wild population. All the other samples were derived from captive subjects, which could result in an under-estimate of the actual variation among individuals and populations in their natural environment. Another potential confounding factor of captive capuchins is the possibility of having cross-species hybrids due to their breeding history while in captivity [61].
Another interesting finding of this study was the identification of two confirmed cases of a ‘near parallel insertion’ among capuchin genomes. This situation occurs when an independent retrotransposon inserts within the sequence span of the PCR amplicon, possibly confounding interpretation of the results. Near parallel insertions have previously been reported to be rare in human genomes [59] and easily resolved by DNA sequencing as they were here. However, the number of capuchin lineage-specific Alu insertions (~9000) is considerably higher than the number of reported human-specific Alu elements (~5000) [62] over roughly the same 6 my evolutionary time frame. A higher Alu mobilization rate increases the likelihood of near-parallel insertions occurring. Future researchers utilizing the datasets reported in this study will need to be cognizant when interpreting PCR results.
The future availability of more genome assemblies for additional capuchin species will help alleviate many confounding issues and make it easier to check for near parallel insertions. The set of young Alu elements ascertained from the S. apella genome [GSC_monkey_1.0] was filtered against the C. imitator genome and may be more specific for targeting allele variance among Sapajus species. Still, one individual from a single species within each genus may not represent the entire genus. Having an adequate number of DNA samples for all species would help to refine capuchin phylogenetic relationships and expose if multiple alternative topologies remain likely. Unresolved polytomies, while using unidirectional Alu insertions as phylogenetic markers, would be indicative of ancient and/or ongoing gene flow and reticulation among Sapajus species or perhaps more broadly with Cebus as well.
5. Conclusions
This study provides the most comprehensive dataset of phylogenetically diagnostic Alu insertion polymorphisms for the capuchin lineage reported to date. It shows that multiple Alu subfamilies have evidence of recent mobilization within capuchin genomes. These datasets of autosomal-based Alu elements that have a unidirectional mode of evolution will provide researchers with a much-needed additional resource for the study of capuchin phylogenetic relationships and for conservation strategies.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13040572/s1. Supplementary File S1 is an Excel file containing the RepeatMasker output for the Alu elements ascertained from [Cebus_imitator-1.0] and separate worksheets for PCR primers, DNA samples and genotypes; Supplementary File S2 is an Excel file containing the RepeatMasker output for the Alu elements ascertained from [GSC_monkey_1.0] and separate worksheets for PCR primers, DNA samples and genotypes; Supplementary File S3 is a *.gb file constructed in BioEdit (Hall 1999) showing DNA sequence alignments for five Alu insertion candidates with ambiguous PCR results; also available as a *.pdf PDF document file.
Author Contributions
J.M.S., J.A.W. and M.A.B. designed the research and wrote the paper; J.M.S., J.A.W., C.E.R. and G.M. conducted the experiments and analyzed the results; J.M.S. performed the Alu repeat analysis of the [Cebus_imitiator-1.0] genome assembly; J.M.S. and J.A.W. performed the Alu repeat analysis of the [GSC_monkey_1.0] genome assembly. T.O.B. and J.M.S. designed custom python scripts for data analysis and filtering; A.D.M., J.D.O. and K.A.P. contributed research samples and edited the article. A.D.M. provided the photograph of an adult male C. imitator. C.R. edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Institute of Health R01 GM59290 (M.A.B.) and by the President’s Future Leaders in Research Scholarship (C.E.R. and G.M.).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The algorithms used in this study are available on GitHub (https://github.com/t-beck; accessed on 4 March 2022). The Supplementary data files are available on the online version of this paper and through the Batzer Lab website under publications, https://biosci-batzerlab.biology.lsu.edu/; accessed on 4 March 2022.
Acknowledgments
The capuchin monkey genome assembly (C. imitator) [Cebus_imitiator-1.0] is provided with the following acknowledgements: Amanda Melin at the University of Calgary, Shoji Kawamura at University of Tokyo, and Wesley Warren, McDonnell Genome Institute, Washington University School of Medicine. The genome assembly of the Tufted Capuchin (S. apella) [GSC_monkey_1.0] is provided with the following acknowledgements: Drs. Luka Culibrk, Sreeja Leelakumari, Kane Tse, Dean Cheng, Eric Chuah, Heather Kirk, Pawan Pandoh, A. Troussard, Yongjun Zhao, Andrew J. Mungall, Richard A. Moore, Marco A. Marra, T. Sinclair-Smith and Steven J. M. Jones at Canada’s Michael Smith Genome Sciences Centre, BC Cancer, 570 7th Ave, Vancouver, British Columbia V5Z 4S6, Canada. Special thanks to the following people and institutions for their generous donation of samples: Frederick H. Sheldon, Curator, and Donna Dittmann of the Louisiana State University Museum of Natural Science Collection of Genetic Resources; The San Diego Zoo Global Biomaterials Review Group, San Diego Zoo Institute for Conservation Research; Link Olson, Curator, and Kyndall Hildebrandt, Genomic Resources Collection Manager, at the University of Alaska Museum of the North Mammal Collection; Verity Mathis, Mammal Collections Manager, Pamela Soltis, and Terry Lott at The University of Florida Board of Trustees—Florida Museum of Natural History—Genetic Resources Repository; Amanda Melin at the University of Calgary, Wesley Warren, Assistant Director McDonnell Genome Institute, Washington University School of Medicine and Michael Schmidt, McDonnell Genome Institute, Washington University School of Medicine; Christopher C. Conroy, Curator, Mammals Collection at the Museum of Vertebrate Zoology, University of California-Berkeley.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Alfaro, J.W.; Silva, J.D., Jr.; Rylands, A.B. How different are robust and gracile capuchin monkeys? An argument for the use of sapajus and cebus. Am. J. Primatol. 2012, 74, 273–286. [Google Scholar] [CrossRef]
- Martins-Junior, A.M.G.; Carneiro, J.; Sampaio, I.; Ferrari, S.F.; Schneider, H. Phylogenetic relationships among Capuchin (Cebidae, Platyrrhini) lineages: An old event of sympatry explains the current distribution of Cebus and Sapajus. Genet. Mol. Biol. 2018, 41, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Boubli, J.P.; Rylands, A.B.; Farias, I.P.; Alfaro, M.E.; Alfaro, J.L. Cebus phylogenetic relationships: A preliminary reassessment of the diversity of the untufted capuchin monkeys. Am. J. Primatol. 2012, 74, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Ciufo, S.; Domrachev, M.; Hotton, C.L.; Kannan, S.; Khovanskaya, R.; Leipe, D.; McVeigh, R.; O’Neill, K.; Robbertse, B.; et al. NCBI Taxonomy: A comprehensive update on curation, resources and tools. Database J. Biol. Databases Curation 2020, baaa062. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.G.M.; Silva-Júnior, J.S.E.; Černý, D.; Buckner, J.C.; Aleixo, A.; Chang, J.; Zheng, J.; Alfaro, M.E.; Martins, A.; Di Fiore, A.; et al. A phylogenomic perspective on the robust capuchin monkey (Sapajus) radiation: First evidence for extensive population admixture across South America. Mol. Phylogenet. Evol. 2018, 124, 137–150. [Google Scholar] [CrossRef]
- Lynch Alfaro, J.W.; Boubli, J.P.; Olson, L.E.; Di Fiore, A.; Wilson, B.; Gutiérrez-Espeleta, G.A.; Chiou, K.L.; Schulte, M.; Neitzel, S.; Ross, V.; et al. Explosive Pleistocene range expansion leads to widespread Amazonian sympatry between robust and gracile capuchin monkeys. J. Biogeogr. 2012, 39, 272–288. [Google Scholar] [CrossRef]
- Ruiz-Garcia, M.; Castillo, M.I.; Ledezma, A.; Leguizamon, N.; Sánchez, R.; Chinchilla, M.; Gutierrez-Espeleta, G.A. Molecular systematics and phylogeography of Cebus capucinus (Cebidae, Primates) in Colombia and Costa Rica by means of the mitochondrial COII gene. Am. J. Primatol. 2012, 74, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-García, M.; Castillo, M.I.; Lichilín-Ortiz, N.; Pinedo-Castro, M. Molecular relationships and classification of several tufted capuchin lineages (Cebus apella, Cebus xanthosternos and Cebus nigritus, Cebidae), by means of mitochondrial cytochrome oxidase II gene sequences. Folia Primatol. Int. J. Primatol. 2012, 83, 100–125. [Google Scholar] [CrossRef] [PubMed]
- Lynch Alfaro, J.W.; Izar, P.; Ferreira, R.G. Capuchin monkey research priorities and urgent issues. Am. J. Primatol. 2014, 76, 705–720. [Google Scholar] [CrossRef]
- Martins, W.P.; de Melo, F.R.; Kierulff, M.C.M.; Mittermeier, R.A.; Lynch Alfaro, J.W.; Jerusalinsky, L. Sapajus robustus (amended version of 2019 assessment). IUCN Red List Threat. Species 2021, e.T42697A192592444. [Google Scholar] [CrossRef]
- Fialho, M.S.; Jerusalinsky, L.; Moura, E.F.; Ravetta, A.L.; Laroque, P.O.; de Queiroz, H.L.; Boubli, J.P.; Lynch Alfaro, J.W. Cebus kaapori (amended version of 2020 assessment). IUCN Red List Threat. Species 2021, e.T40019A191704766. [Google Scholar] [CrossRef]
- Martins, A.B.; Fialho, M.S.; Jerusalinsky, L.; Valença-Montenegro, M.M.; Bezerra, B.M.; Laroque, P.O.; de Melo, F.R.; Lynch Alfaro, J.W. Sapajus libidinosus (amended version of 2019 assessment). IUCN Red List Threat. Species 2021, e.T136346A192593226. [Google Scholar] [CrossRef]
- De Moraes, B.L.C.; Borges, D.B.; Souza-Alves, J.P.; Boubli, J.P.; Bezerra, B. Microsatellite Markers for Bearded Capuchins (Sapajus libidinosus): Transferability and Characterization. An. Acad. Bras. Cienc. 2021, 93, e20190802. [Google Scholar] [CrossRef]
- Gron, K.J. April 17. Primate Factsheets: Tufted capuchin (Cebus apella) Taxonomy, Morphology, & Ecology. 2009. Available online: https://primate.wisc.edu/primate-info-net/pin-factsheets/pin-factsheet-tufted-capuchin/Link (accessed on 2 March 2022).
- Baker, J.N.; Walker, J.A.; Denham, M.W.; Loupe, C.D., 3rd; Batzer, M.A. Recently integrated Alu insertions in the squirrel monkey (Saimiri) lineage and application for population analyses. Mob DNA 2018, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Batzer, M.A.; Deininger, P.L. Alu repeats and human genomic diversity. Nat. Rev. Genet. 2002, 3, 370–379. [Google Scholar] [CrossRef]
- Li, J.; Han, K.; Xing, J.; Kim, H.S.; Rogers, J.; Ryder, O.A.; Disotell, T.; Yue, B.; Batzer, M.A. Phylogeny of the macaques (Cercopithecidae: Macaca) based on Alu elements. Gene 2009, 448, 242–249. [Google Scholar] [CrossRef] [PubMed]
- McLain, A.T.; Meyer, T.J.; Faulk, C.; Herke, S.W.; Oldenburg, J.M.; Bourgeois, M.G.; Abshire, C.F.; Roos, C.; Batzer, M.A. An alu-based phylogeny of lemurs (infraorder: Lemuriformes). PLoS ONE 2012, 7, e44035. [Google Scholar] [CrossRef]
- Meyer, T.J.; McLain, A.T.; Oldenburg, J.M.; Faulk, C.; Bourgeois, M.G.; Conlin, E.M.; Mootnick, A.R.; de Jong, P.J.; Roos, C.; Carbone, L.; et al. An Alu-based phylogeny of gibbons (hylobatidae). Mol. Biol. Evol. 2012, 29, 3441–3450. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Perna, N.T.; Batzer, M.A.; Deininger, P.L.; Stoneking, M. Alu insertion polymorphism: A new type of marker for human population studies. Hum. Biol. 1992, 64, 641–648. [Google Scholar]
- Ray, D.A.; Batzer, M.A. Tracking Alu evolution in New World primates. BMC Evol. Biol. 2005, 5, 51. [Google Scholar] [CrossRef]
- Salem, A.H.; Ray, D.A.; Xing, J.; Callinan, P.A.; Myers, J.S.; Hedges, D.J.; Garber, R.K.; Witherspoon, D.J.; Jorde, L.B.; Batzer, M.A. Alu elements and hominid phylogenetics. Proc. Natl. Acad. Sci. USA 2003, 100, 12787–12791. [Google Scholar] [CrossRef]
- Steely, C.J.; Walker, J.A.; Jordan, V.E.; Beckstrom, T.O.; McDaniel, C.L.; St Romain, C.P.; Bennett, E.C.; Robichaux, A.; Clement, B.N.; Raveendran, M.; et al. Alu Insertion Polymorphisms as Evidence for Population Structure in Baboons. Genome Biol. Evol. 2017, 9, 2418–2427. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.; Kural, D.; Stromberg, M.P.; Walker, J.A.; Konkel, M.K.; Stutz, A.M.; Urban, A.E.; Grubert, F.; Lam, H.Y.; Lee, W.P.; et al. A comprehensive map of mobile element insertion polymorphisms in humans. PLoS Genet. 2011, 7, e1002236. [Google Scholar] [CrossRef]
- Xing, J.; Wang, H.; Han, K.; Ray, D.A.; Huang, C.H.; Chemnick, L.G.; Stewart, C.B.; Disotell, T.R.; Ryder, O.A.; Batzer, M.A. A mobile element based phylogeny of Old World monkeys. Mol. Phylogenet. Evol. 2005, 37, 872–880. [Google Scholar] [CrossRef]
- Xing, J.; Wang, H.; Zhang, Y.; Ray, D.A.; Tosi, A.J.; Disotell, T.R.; Batzer, M.A. A mobile element-based evolutionary history of guenons (tribe Cercopithecini). BMC Biol. 2007, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Roos, C.; Zinner, D.; Kubatko, L.S.; Schwarz, C.; Yang, M.; Meyer, D.; Nash, S.D.; Xing, J.; Batzer, M.A.; Brameier, M.; et al. Nuclear versus mitochondrial DNA: Evidence for hybridization in colobine monkeys. BMC Evol. Biol. 2011, 11, 77. [Google Scholar] [CrossRef] [PubMed]
- Hartig, G.; Churakov, G.; Warren, W.C.; Brosius, J.; Makałowski, W.; Schmitz, J. Retrophylogenomics place tarsiers on the evolutionary branch of anthropoids. Sci. Rep. 2013, 3, 1756. [Google Scholar] [CrossRef]
- Liu, G.E.; Alkan, C.; Jiang, L.; Zhao, S.; Eichler, E.E. Comparative analysis of Alu repeats in primate genomes. Genome Res. 2009, 19, 876–885. [Google Scholar] [CrossRef]
- Schmitz, J.; Ohme, M.; Zischler, H. SINE insertions in cladistic analyses and the phylogenetic affiliations of Tarsius bancanus to other primates. Genetics 2001, 157, 777–784. [Google Scholar] [CrossRef]
- Singer, S.S.; Schmitz, J.; Schwiegk, C.; Zischler, H. Molecular cladistic markers in New World monkey phylogeny (Platyrrhini, Primates). Mol. Phylogenet. Evol. 2003, 26, 490–501. [Google Scholar] [CrossRef]
- Luan, D.D.; Korman, M.H.; Jakubczak, J.L.; Eickbush, T.H. Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: A mechanism for non-LTR retrotransposition. Cell 1993, 72, 595–605. [Google Scholar] [CrossRef]
- Batzer, M.A.; Deininger, P.L. A human-specific subfamily of Alu sequences. Genomics 1991, 9, 481–487. [Google Scholar] [CrossRef]
- Batzer, M.A.; Deininger, P.L.; Hellmann-Blumberg, U.; Jurka, J.; Labuda, D.; Rubin, C.M.; Schmid, C.W.; Zietkiewicz, E.; Zuckerkandl, E. Standardized nomenclature for Alu repeats. J. Mol. Evol. 1996, 42, 3–6. [Google Scholar] [CrossRef]
- Jurka, J. Sequence patterns indicate an enzymatic involvement in integration of mammalian retroposons. Proc. Natl. Acad. Sci. USA 1997, 94, 1872–1877. [Google Scholar] [CrossRef] [PubMed]
- Kapitonov, V.; Jurka, J. The age of Alu subfamilies. J. Mol. Evol. 1996, 42, 59–65. [Google Scholar] [CrossRef]
- Konkel, M.K.; Walker, J.A.; Batzer, M.A. LINEs and SINEs of Primate Evolution. Evol. Anthropol. 2010, 19, 236–249. [Google Scholar] [CrossRef]
- Churakov, G.; Grundmann, N.; Kuritzin, A.; Brosius, J.; Makałowski, W.; Schmitz, J. A novel web-based TinT application and the chronology of the Primate Alu retroposon activity. BMC Evol. Biol. 2010, 10, 376. [Google Scholar] [CrossRef]
- Worley, K.C.; Warren, W.C.; Rogers, J.; Locke, D.; Muzny, D.M.; Mardis, E.R.; Weinstock, G.M.; Tardif, S.D. The common marmoset genome provides insight into primate biology and evolution. Nat. Genet. 2014, 46, 850–857. [Google Scholar] [CrossRef]
- Baker, J.N.; Walker, J.A.; Vanchiere, J.A.; Phillippe, K.R.; St Romain, C.P.; Gonzalez-Quiroga, P.; Denham, M.W.; Mierl, J.R.; Konkel, M.K.; Batzer, M.A. Evolution of Alu Subfamily Structure in the Saimiri Lineage of New World Monkeys. Genome Biol. Evol. 2017, 9, 2365–2376. [Google Scholar] [CrossRef] [PubMed]
- Storer, J. Characterization and Amplification of Retrotransposable Elements Platy-1 and Alu in the Cebidae Lineage of Platyrrhine Primates. LSU Doctoral Dissertations. 5053. 2019. Available online: https://digitalcommons.lsu.edu/gradschool_dissertations/5053 (accessed on 2 March 2022).
- Storer, J.M.; Walker, J.A.; Jordan, V.E.; Batzer, M.A. Sensitivity of the polyDetect computational pipeline for phylogenetic analyses. Anal. Biochem. 2020, 593, 113516. [Google Scholar] [CrossRef] [PubMed]
- Osterholz, M.; Vermeer, J.; Walter, L.; Roos, C. A PCR-based marker to simply identify Saimiri sciureus and S. boliviensis boliviensis. Am. J. Primatol. 2008, 70, 1177–1180. [Google Scholar] [CrossRef] [PubMed]
- Osterholz, M.; Walter, L.; Roos, C. Retropositional events consolidate the branching order among New World monkey genera. Mol. Phylogenet. Evol. 2009, 50, 507–513. [Google Scholar] [CrossRef]
- Ray, D.A.; Xing, J.; Hedges, D.J.; Hall, M.A.; Laborde, M.E.; Anders, B.A.; White, B.R.; Stoilova, N.; Fowlkes, J.D.; Landry, K.E.; et al. Alu insertion loci and platyrrhine primate phylogeny. Mol. Phylogenet. Evol. 2005, 35, 117–126. [Google Scholar] [CrossRef]
- Martins, A.M., Jr.; Amorim, N.; Carneiro, J.C.; de Mello Affonso, P.R.; Sampaio, I.; Schneider, H. Alu elements and the phylogeny of capuchin (Cebus and Sapajus) monkeys. Am. J. Primatol. 2015, 77, 368–375. [Google Scholar] [CrossRef]
- Orkin, J.D.; Montague, M.J.; Tejada-Martinez, D.; de Manuel, M.; Del Campo, J.; Cheves Hernandez, S.; Di Fiore, A.; Fontsere, C.; Hodgson, J.A.; Janiak, M.C.; et al. The genomics of ecological flexibility, large brains, and long lives in capuchin monkeys revealed with fecalFACS. Proc. Natl. Acad. Sci. USA 2021, 118, e2010632118. [Google Scholar] [CrossRef]
- Smit, A.F.A.; Hubley, R.; Green, P. 2013–2015, RepeatMasker Open-4.0. 2015. Available online: http://www.repeatmasker.org (accessed on 4 March 2022).
- Kent, W.J. BLAT—the BLAST-like alignment tool. Genome Res. 2002, 12, 656–664. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3--new capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 2012, 13, 134. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.A.; Thompson, C.R. Hand preference for tool-use in capuchin monkeys (Cebus apella) is associated with asymmetry of the primary motor cortex. Am. J. Primatol. 2013, 75, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Renner, E.; Abramo, A.M.; Karen Hambright, M.; Phillips, K.A. Insightful problem solving and emulation in brown capuchin monkeys. Anim. Cogn. 2017, 20, 531–536. [Google Scholar] [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Konkel, M.K.; Walker, J.A.; Hotard, A.B.; Ranck, M.C.; Fontenot, C.C.; Storer, J.; Stewart, C.; Marth, G.T.; Batzer, M.A. Sequence Analysis and Characterization of Active Human Alu Subfamilies Based on the 1000 Genomes Pilot Project. Genome Biol. Evol. 2015, 7, 2608–2622. [Google Scholar] [CrossRef] [PubMed]
- Faircloth, B.C.; McCormack, J.E.; Crawford, N.G.; Harvey, M.G.; Brumfield, R.T.; Glenn, T.C. Ultraconserved elements anchor thousands of genetic markers spanning multiple evolutionary timescales. Syst. Biol. 2012, 61, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Kuritzin, A.; Kischka, T.; Schmitz, J.; Churakov, G. Incomplete Lineage Sorting and Hybridization Statistics for Large-Scale Retroposon Insertion Data. PLoS Comput. Biol. 2016, 12, e1004812. [Google Scholar] [CrossRef] [PubMed]
- Ray, D.A.; Xing, J.; Salem, A.H.; Batzer, M.A. SINEs of a nearly perfect character. Syst. Biol. 2006, 55, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Martins, W.P.; Lynch Alfaro, J.; Rylands, A.B. Reduced range of the endangered crested capuchin monkey (Sapajus robustus) and a possible hybrid zone with Sapajus nigritus. Am. J. Primatol. 2017, 79, e22696. [Google Scholar] [CrossRef]
- Nieves, M.; Mendez, G.; Ortiz, A.; Mühlmann, M.; Mudry, M.D. Karyological diagnosis of Cebus (Primates, Platyrrhini) in captivity: Detection of hybrids and management program applications. Anim. Reprod. Sci. 2008, 108, 66–78. [Google Scholar] [CrossRef]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).