Cerebellar Abiotrophy in Australian Working Kelpies Is Associated with Two Major Risk Loci
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Phenotyping
2.2. DNA Extraction and Array-Based Genotyping
2.3. Genome-Wide Association Analysis
2.4. Whole Genome Sequencing for Mutation Detection
2.5. Histopathology
3. Results
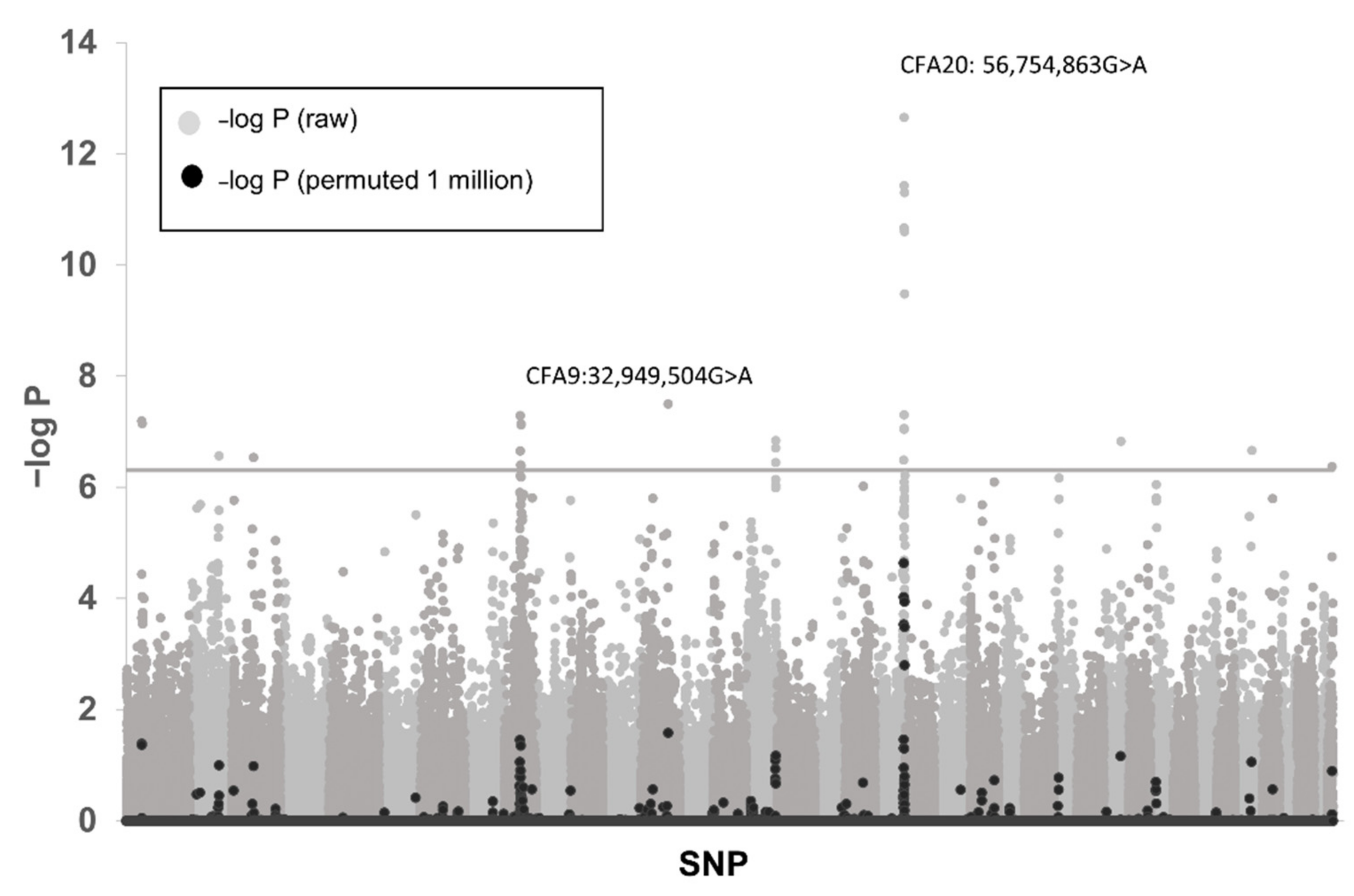
3.1. Genome-Wide Association
3.2. Clinical Findings and Histopathology
3.3. Sequence Alignment and Variant Calling
3.4. Functional Annotation of Variants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sandy, J.R.; Slocombe, R.E.; Mitten, R.W.; Jedwab, D. Cerebellar abiotrophy in a family of Border Collie dogs. Vet. Pathol. 2002, 39, 736–738. [Google Scholar] [CrossRef]
- Jokinen, T.S.; Rusbridge, C.; Steffen, F.; Viitmaa, R.; Syrja, P.; De Lahunta, A.; Snellman, M.; Cizinauskas, S. Cerebellar cortical abiotrophy in Lagotto Romagnolo dogs. J. Small Anim. Pract. 2007, 48, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Bildfell, R.J.; Mitchell, S.K.; de Lahunta, A. Cerebellar cortical degeneration in a Labrador retriever. Can. Vet. J. 1995, 36, 570–572. [Google Scholar] [PubMed]
- Tatalick, L.M.; Marks, S.L.; Baszler, T.V. Cerebellar abiotrophy characterized by granular cell loss in a Brittany. Vet. Pathol. 1993, 30, 385–388. [Google Scholar] [CrossRef]
- Gandini, G.; Botteron, C.; Brini, E.; Fatzer, R.; Diana, A.; Jaggy, A. Cerebellar cortical degeneration in three English bulldogs: Clinical and neuropathological findings. J. Small Anim. Pract. 2005, 46, 291–294. [Google Scholar] [CrossRef]
- Agler, C.; Nielsen, D.M.; Urkasemsin, G.; Singleton, A.; Tonomura, N.; Sigurdsson, S.; Tang, R.; Linder, K.; Arepalli, S.; Hernandez, D.; et al. Canine Hereditary Ataxia in Old English Sheepdogs and Gordon Setters Is Associated with a Defect in the Autophagy Gene Encoding RAB24. PLoS Genet. 2014, 10, e1003991. [Google Scholar] [CrossRef]
- Kyöstilä, K.; Cizinauskas, S.; Seppälä, E.H.; Suhonen, E.; Jeserevics, J.; Sukura, A.; Syrjä, P.; Lohi, H. A SEL1L mutation links a canine progressive early-onset cerebellar ataxia to the endoplasmic reticulum–associated protein degradation (ERAD) machinery. PLoS Genet. 2012, 8, e1002759. [Google Scholar] [CrossRef]
- Gumber, S.; Cho, D.Y.; Morgan, T.W. Late onset of cerebellar abiotrophy in a boxer dog. Vet. Med. Int. 2010, 2010, 406275. [Google Scholar] [CrossRef]
- Thomas, J.B.; Robertson, D. Hereditary cerebellar abiotrophy in Australian kelpie dogs. Aust. Vet. J. 1989, 66, 301–302. [Google Scholar] [CrossRef]
- Shearman, J.R.; Cook, R.W.; McCowan, C.; Fletcher, J.L.; Taylor, R.M.; Wilton, A.N. Mapping cerebellar abiotrophy in Australian Kelpies. Anim. Genet. 2011, 42, 675–678. [Google Scholar] [CrossRef]
- Coates, J.R.; O’Brien, D.P.; Kline, K.L.; Storts, R.W.; Johnson, G.C.; Shelton, G.D.; Patterson, E.E.; Abbott, L.C. Neonatal cerebellar ataxia in Coton de Tulear dogs. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2002, 16, 680–689. [Google Scholar] [CrossRef]
- Yasuba, M.; Okimoto, K.; Iida, M.; Itakura, C. Cerebellar cortical degeneration in beagle dogs. Vet. Pathol. 1988, 25, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Forman, O.P.; De Risio, L.; Matiasek, K.; Platt, S.; Mellersh, C. Spinocerebellar ataxia in the Italian Spinone dog is associated with an intronic GAA repeat expansion in ITPR1. Mamm. Genome 2015, 26, 108–117. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Urkasemsin, G. Hereditary cerebellar degeneration in Scottish terriers. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2010, 24, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.R.; LeCouteur, A.R.; Kornegay, N.J.; Coates, R.J. Late-onset progressive spinocerebellar degeneration in Brittany Spaniel dogs. Acta Neuropathol. 1998, 96, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, H.S. Cerebellar degeneration in Old English Sheepdogs. J. Am. Vet. Med. Assoc. 2000, 217, 1162–1165. [Google Scholar] [CrossRef]
- Sisó, S.; Hanzlíček, D.; Fluehmann, G.; Kathmann, I.; Tomek, A.; Papa, V.; Vandevelde, M. Neurodegenerative diseases in domestic animals: A comparative review. Vet. J. 2006, 171, 20–38. [Google Scholar] [CrossRef]
- Forman, O.P.; De Risio, L.; Mellersh, C.S. Missense Mutation in CAPN1 Is Associated with Spinocerebellar Ataxia in the Parson Russell Terrier Dog Breed. PLoS ONE 2013, 8, e64627. [Google Scholar]
- Forman, O.P.; De Risio, L.; Stewart, J.; Mellersh, C.S.; Beltran, E. Genome-wide mRNA sequencing of a single canine cerebellar cortical degeneration case leads to the identification of a disease associated SPTBN2 mutation. BMC Genet. 2012, 13, 55. [Google Scholar] [CrossRef]
- Gilliam, D.; O’Brien, D.P.; Coates, J.R.; Johnson, G.S.; Johnson, G.C.; Mhlanga-Mutangadura, T.; Hansen, L.; Taylor, J.F.; Schnabel, R.D. A Homozygous KCNJ10 Mutation in Jack Russell Terriers and Related Breeds with Spinocerebellar Ataxia with Myokymia, Seizures, or Both. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2014, 28, 871–877. [Google Scholar] [CrossRef]
- Speciale, J.; de Lahunta, A. Cerebellar degeneration in a mature Staffordshire terrier. J. Am. Anim. Hosp. Assoc. 2003, 39, 459–462. [Google Scholar] [CrossRef]
- Zeng, R.; Farias, F.H.G.; Johnson, G.S.; McKay, S.D.; Schnabel, R.D.; Decker, J.E.; Taylor, J.F.; Mann, C.S.; Katz, M.L.; Johnson, G.C.; et al. A Truncated Retrotransposon Disrupts the GRM1 Coding Sequence in Coton de Tulear Dogs with Bandera’s Neonatal Ataxia. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2011, 25, 267–272. [Google Scholar] [CrossRef]
- Bernardino, F.; Rentmeister, K.; Schmidt, M.J.; Bruehschwein, A.; Matiasek, K.; Matiasek, L.A.; Lauda, A.; Schoon, H.A.; Fischer, A. Inferior Cerebellar Hypoplasia Resembling a Dandy-Walker-Like Malformation in Purebred Eurasier Dogs with Familial Non-Progressive Ataxia: A Retrospective and Prospective Clinical Cohort Study. PLoS ONE 2015, 10, e0117670. [Google Scholar] [CrossRef]
- Kyöstilä, K.; Syrjä, P.; Jagannathan, V.; Chandrasekar, G.; Jokinen, T.S.; Seppälä, E.H.; Becker, D.; Drögemüller, M.; Dietschi, E.; Drögemüller, C.; et al. A Missense Change in the ATG4D Gene Links Aberrant Autophagy to a Neurodegenerative Vacuolar Storage Disease. PLoS Genet. 2015, 11, e1005169. [Google Scholar] [CrossRef]
- Mauri, N.; Kleiter, M.; Leschnik, M.; Högler, S.; Dietschi, E.; Wiedmer, M.; Dietrich, J.; Henke, D.; Steffen, F.; Schuller, S.; et al. A Missense Variant in KCNJ10 in Belgian Shepherd Dogs Affected by Spongy Degeneration with Cerebellar Ataxia (SDCA1). G3 Genes Genomes Genet. 2017, 7, 663–669. [Google Scholar] [CrossRef]
- Wang, Y.; Hersheson, J.; Lopez, D.; Hamad, M.B.; Liu, Y.; Lee, K.-H.; Pinto, V.; Seinfeld, J.; Wiethoff, S.; Sun, J.; et al. Defects in the CAPN1 gene result in alterations in cerebellar development and in cerebellar ataxia in mice and humans. Cell Rep. 2016, 16, 79–91. [Google Scholar] [CrossRef]
- Fenn, J.; Boursnell, M.; Hitti, R.J.; Jenkins, C.A.; Terry, R.L.; Priestnall, S.L.; Kenny, P.J.; Mellersh, C.S.; Forman, O.P. Genome sequencing reveals a splice donor site mutation in the SNX14 gene associated with a novel cerebellar cortical degeneration in the Hungarian Vizsla dog breed. BMC Genet. 2016, 17, 123. [Google Scholar] [CrossRef]
- Pan, A.Y.H.; Wade, C.M.; Taylor, R.M.; Williamson, P. Exclusion of known gene loci for cerebellar abiotrophy in the Australian Working Kelpie. Anim. Genet. 2017, 48, 730–732. [Google Scholar] [CrossRef]
- Arnott, E.R.; Peek, L.; Early, J.B.; Pan, A.Y.; Haase, B.; Chew, T.; McGreevy, P.D.; Wade, C.M. Strong selection for behavioural resilience in Australian stock working dogs identified by selective sweep analysis. Canine Genet. Epidemiol. 2015, 2, 6. [Google Scholar] [CrossRef]
- Velie, B.D.; Wilson, B.J.; Arnott, E.R.; Early, J.B.; McGreevy, P.D.; Wade, C.M. Inbreeding levels in an open-registry pedigreed dog breed: The Australian working kelpie. Vet. J. 2021, 269, 105609. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- McKenna, A. The Genome Analysis Toolkit: A MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010, 20, 1297–1303. [Google Scholar] [CrossRef]
- Wang, C.; Wallerman, O.; Arendt, M.L.; Sundstrom, E.; Karlsson, A.; Nordin, J.; Makelainen, S.; Pielberg, G.R.; Hanson, J.; Ohlsson, A.; et al. A novel canine reference genome resolves genomic architecture and uncovers transcript complexity. Commun. Biol. 2021, 4, 185. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
- DePristo, M.A.; Banks, E.; Poplin, R.; Garimella, K.V.; Maguire, J.R.; Hartl, C.; Philippakis, A.A.; del Angel, G.; Rivas, M.A.; Hanna, M.; et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 2011, 43, 491–498. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef]
- McLaren, W.; Pritchard, B.; Rios, D.; Chen, Y.; Flicek, P.; Cunningham, F. Deriving the consequences of genomic variants with the Ensembl API and SNP Effect Predictor. Bioinformatics 2010, 26, 2069–2070. [Google Scholar] [CrossRef]
- Hinrichs, A.S.; Raney, B.J.; Speir, M.L.; Rhead, B.; Casper, J.; Karolchik, D.; Kuhn, R.M.; Rosenbloom, K.R.; Zweig, A.S.; Haussler, D.; et al. UCSC Data Integrator and Variant Annotation Integrator. Bioinformatics 2016, 32, 1430–1432. [Google Scholar] [CrossRef]
- Jagannathan, V.; Drogemuller, C.; Leeb, T.; Dog Biomedical Variant Database Consortium (DBVDC). A comprehensive biomedical variant catalogue based on whole genome sequences of 582 dogs and eight wolves. Anim. Genet. 2019, 50, 695–704. [Google Scholar]
- Plassais, J.; Kim, J.; Davis, B.W.; Karyadi, D.M.; Hogan, A.N.; Harris, A.C.; Decker, B.; Parker, H.G.; Ostrander, E.A. Whole genome sequencing of canids reveals genomic regions under selection and variants influencing morphology. Nat. Commun. 2019, 10, 1489. [Google Scholar] [CrossRef]
- Chew, T.; Willet, C.E.; Haase, B.; Wade, C.M. Genomic Characterization of External Morphology Traits in Kelpies Does Not Support Common Ancestry with the Australian Dingo. Genes 2019, 10, 337. [Google Scholar] [CrossRef]
- Adzhubei, I.; Jordan, D.M.; Sunyaev, S.R. Predicting functional effect of human missense mutations using PolyPhen-2. Curr. Protoc. Hum. Genet. 2013, 76, 7.20.1–7.20.41. [Google Scholar] [CrossRef]
- Bovio, P.P.; Franz, H.; Heidrich, S.; Rauleac, T.; Kilpert, F.; Manke, T.; Vogel, T. Differential Methylation of H3K79 Reveals DOT1L Target Genes and Function in the Cerebellum In Vivo. Mol. Neurobiol. 2019, 56, 4273–4287. [Google Scholar] [CrossRef]
- Molejon, M.I.; Ropolo, A.; Re, A.L.; Boggio, V.; Vaccaro, M.I. The VMP1-Beclin 1 interaction regulates autophagy induction. Sci. Rep. 2013, 3, 1055. [Google Scholar] [CrossRef]
- He, C.; Bassik, M.C.; Moresi, V.; Sun, K.; Wei, Y.; Zou, Z.; An, Z.; Loh, J.; Fisher, J.; Sun, Q.; et al. Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature 2012, 481, 511–515. [Google Scholar] [CrossRef]
- Komatsu, M.; Waguri, S.; Chiba, T.; Murata, S.; Iwata, J.; Tanida, I.; Ueno, T.; Koike, M.; Uchiyama, Y.; Kominami, E.; et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 2006, 441, 880–884. [Google Scholar]
| ID | Sex | Onset Age | Onset-Type | Country | Video (Table S5) | Neurological Exam (Table S4) | Cerebellar Abiotrophy Diagnosis Supported | Homozygous Risk Allele | Genotype CFA20 BICF2P1109624 (AA Risk) | Genotype CFA9 BICF2G630835610 (AA Risk) | Genotype CFA1 TIGRP2P904 (AA Risk) | Genotype CFA13 BICF2P336113 (AA Risk) | Clinical Assessment Source |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1827 | F | 4 weeks | early | Australia | Y | Y | Y | A A | G G | G G | G G | Owner-checklist | |
| 3596 | M | 6 weeks | early | Australia | Y | A A | A G | G G | A G | Owner | |||
| 6273 | M | 6 weeks | early | Australia | Y | Y | Y | A A | G G | A G | G G | Veterinarian | |
| 6274 | F | 6 weeks | early | Australia | Y | Y | Y | A A | G G | A G | A A | Veterinarian | |
| 6317 | M | 4 weeks | early | Australia | Y | Y | Y | A A | G G | G G | A G | Neurologist | |
| 6343 | F | 6 weeks | early | Germany | Y | Y | G G | G G | G G | A G | Checklist | ||
| 6344 | M | 6 weeks | early | Germany | Y | Y | G G | G G | G G | A G | Checklist | ||
| 6345 | M | 7 weeks | early | Germany | Y | Y | G G | A G | G G | G G | Checklist | ||
| 6346 | M | 7 weeks | early | Germany | Y | Y | G G | G G | G G | A G | Checklist | ||
| 6359 | F | 6 weeks | early | Australia | Y | Y | Y | A A | A G | G G | G G | Neurologist | |
| 6360 | F | 6 weeks | early | Australia | Y | Y | Y | A A | A A | A G | G G | Owner | |
| 6369 | M | 6 weeks | early | Australia | Y | Y | Y | A A | G G | G G | G G | Owner-checklist | |
| 6370 | M | 6 weeks | early | Australia | Y | Y | Y | A A | G G | G G | G G | Owner-checklist | |
| 6376 | M | 7 weeks | early | Australia | Y | A A | G G | G G | A G | Owner | |||
| 6386 | F | 7 weeks | early | Australia | Y | Y | Y | A A | A G | G G | A G | Owner-checklist | |
| 6387 | F | 5 weeks | early | Australia | Y | A A | A G | A G | A G | Owner | |||
| 6390 | F | 6 weeks | early | Australia | Y | A A | G G | G G | G G | Owner | |||
| 6394 | M | 8 weeks | early | Australia | Y | Y | Y | G G | A A | G G | G G | Owner-checklist | |
| 6395 | M | 8 weeks | early | Australia | Y | A A | G G | A G | G G | Owner | |||
| NH3 | F | 4 weeks | early | Australia | Y | Y | Y | A A | G G | A G | A G | Owner-checklist | |
| NH4 | F | 5 weeks | early | Australia | Y | Y | Y | A A | G G | A G | A G | Owner-checklist | |
| 6205 | M | 8 months | late | Australia | Y | Y | Y | Y | G G | A A | G G | G G | Veterinarian |
| 6263 | M | 5 months | late | Australia | Y | Y | Y | Y | G G | A A | A G | A G | Checklist |
| 6363 | F | 16 weeks | late | Australia | Y | Y | Y | G G | A A | A G | A G | Checklist | |
| 6364 | M | 16 weeks | late | Australia | Y | Y | Y | G G | A A | G G | G G | Checklist | |
| 6365 | F | 6 months | late | Australia | Y | Y | Y | G G | A A | G G | A G | Checklist | |
| 6368 | F | 12 months | late | Australia | Y | Y | Y | G G | A A | G G | A G | Checklist | |
| 6374 | F | 18 months | late | Australia | Y | Y | Y | A G | A A | G G | A G | Checklist | |
| 6375 | F | 16 weeks | late | Australia | Y | Y | Y | A G | A A | G G | A G | Checklist | |
| 6389 | F | 15 weeks | late | Australia | Y | A G | A A | G G | G G | Owner | |||
| 6275 | F | 24 weeks | late | Australia | Y | N | G G | G G | G G | G G | Veterinarian | ||
| 6266 | M | 10 months | late | Canada | Y | N | 0 0 | A G | A G | A G | Veterinarian | ||
| 6025 | F | unknown | unknown | Australia | G G | G G | G G | G G | Previous | ||||
| 6050 | M | unknown | unknown | Australia | Y | Y | Y | A A | G G | A G | G G | Veterinarian | |
| 6054 | F | unknown | unknown | Australia | G G | G G | A G | A G | Previous | ||||
| 6065 | M | unknown | unknown | Australia | Y | A A | G G | A G | A A | Previous | |||
| 6066 | F | unknown | unknown | Australia | Y | A A | G G | A G | A A | Previous | |||
| 6067 | F | unknown | unknown | Australia | A G | A G | A G | G G | Previous | ||||
| 6110 | F | unknown | unknown | Australia | Y | A A | A G | A A | A G | Previous | |||
| 6111 | F | unknown | unknown | Australia | Y | A A | A A | A A | A G | Previous | |||
| 6149 | F | unknown | unknown | Australia | A G | A G | G G | G G | Previous | ||||
| 6181 | F | unknown | unknown | Australia | G G | A G | A G | A G | Previous | ||||
| 6182 | F | unknown | unknown | Australia | A G | G G | A A | G G | Previous | ||||
| 6183 | F | unknown | unknown | Australia | A G | G G | A A | G G | Previous | ||||
| 6184 | F | unknown | unknown | Australia | A G | G G | A G | A G | Previous | ||||
| 6195 | F | unknown | unknown | Australia | A G | G G | G G | A G | Previous | ||||
| 6203 | F | unknown | unknown | Australia | A G | G G | G G | A G | Previous | ||||
| 6206 | F | unknown | unknown | Australia | Y | Y | Y | Y | A A | G G | G G | G G | Veterinarian |
| 6256 | M | unknown | unknown | Australia | Y | A A | G G | G G | G G | Previous |
| CHR | SNP | Base (UU GSD1.0) | Allele 1 (A1) | Frequency of A1 in Affected | Frequency of A1 in Unaffected | Allele 2 (A2) | CHISQ | P | P Bonferroni | Ppermuted(1,000,000) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TIGRP2P904 | 27279310 | A | 0.28 | 0.08 | G | 29.24 | 6.40 × 10−8 | 0.006974 | 0.04 |
| 1 | BICF2S2379011 | 27300794 | A | 0.28 | 0.09 | C | 29.06 | 7.03 × 10−8 | 0.007665 | 0.04 |
| 2 | BICF2P394061 | 63856960 | A | 0.31 | 0.11 | G | 26.45 | 2.71 × 10−7 | 0.02951 | 0.10 |
| 3 | BICF2S23915658 | 50921021 | A | 0.29 | 0.10 | C | 26.31 | 2.91 × 10−7 | 0.03167 | 0.10 |
| 9 | BICF2G630835610 | 32949504 | A | 0.33 | 0.12 | G | 29.67 | 5.13 × 10−8 | 0.005588 | 0.04 |
| 9 | BICF2G630835611 | 32954974 | A | 0.38 | 0.15 | G | 26.87 | 2.18 × 10−7 | 0.02372 | 0.09 |
| 9 | BICF2G630835989 | 33963736 | C | 0.26 | 0.08 | A | 25.71 | 3.97 × 10−7 | 0.04326 | 0.12 |
| 9 | BICF2G630836291 | 34689620 | A | 0.41 | 0.17 | G | 29.03 | 7.14 × 10−8 | 0.007777 | 0.04 |
| 9 | BICF2G630836293 | 34700358 | G | 0.41 | 0.17 | A | 28.95 | 7.42 × 10−8 | 0.008085 | 0.04 |
| 9 | BICF2G630836318 | 34777634 | A | 0.13 | 0.41 | G | 25.65 | 4.10 × 10−7 | 0.04465 | 0.13 |
| 13 | BICF2P336113 | 43940212 | A | 0.30 | 0.09 | G | 30.61 | 3.16 × 10−8 | 0.003439 | 0.03 |
| 16 | BICF2P228482 | 57096165 | A | 0.40 | 0.17 | G | 25.91 | 3.58 × 10−7 | 0.03903 | 0.12 |
| 16 | BICF2P1097507 | 57109139 | A | 0.40 | 0.16 | C | 27.09 | 1.94 × 10−7 | 0.02112 | 0.08 |
| 16 | BICF2P957000 | 57402220 | A | 0.48 | 0.22 | C | 27.68 | 1.43 × 10−7 | 0.01563 | 0.07 |
| 20 | BICF2P204406 | 56017675 | A | 0.72 | 0.43 | G | 26.12 | 3.21 × 10−7 | 0.03504 | 0.11 |
| 20 | BICF2P113598 | 56247267 | A | 0.16 | 0.46 | G | 28.64 | 8.70 × 10−8 | 0.009478 | 0.05 |
| 20 | BICF2P1109624 | 56283529 | A | 0.64 | 0.25 | G | 53.81 | 2.21 × 10−13 | 2.41 × 10−8 | 2.30 × 10−5 |
| 20 | BICF2P393253 | 56293416 | T | 0.63 | 0.27 | A | 44.85 | 2.13 × 10−11 | 2.32 × 10−6 | 0.0003 |
| 20 | TIGRP2P278295 | 56309785 | A | 0.63 | 0.26 | C | 48.27 | 3.71 × 10−12 | 4.05 × 10−7 | 9.40 × 10−5 |
| 20 | BICF2P1309051 | 56352043 | G | 0.69 | 0.38 | A | 29.75 | 4.91 × 10−8 | 0.005357 | 0.03 |
| 20 | BICF2P331357 | 56704521 | A | 0.07 | 0.35 | G | 28.58 | 8.98 × 10−8 | 0.009784 | 0.05 |
| 20 | BICF2P696292 | 57458652 | A | 0.36 | 0.11 | G | 39.49 | 3.29 × 10−10 | 3.59 × 10−5 | 0.0016 |
| 20 | BICF2P287740 | 57465507 | A | 0.36 | 0.10 | G | 44.53 | 2.50 × 10−11 | 2.72 × 10−6 | 0.0003 |
| 20 | TIGRP2P278607 | 57512779 | A | 0.35 | 0.09 | G | 47.7 | 4.97 × 10−12 | 5.42 × 10−7 | 0.0001 |
| 28 | BICF2S22962329 | 30582750 | G | 0.37 | 0.14 | A | 27.63 | 1.47 × 10−7 | 0.016 | 0.07 |
| 34 | BICF2G630459982 | 25388891 | C | 0.32 | 0.12 | A | 26.88 | 2.16 × 10−7 | 0.02359 | 0.09 |
| 39 | BICF2G630539070 | 6537056 | A | 0.19 | 0.52 | C | 25.59 | 4.22 × 10−7 | 0.04599 | 0.13 |
| Chromosome | ID | Base (UU GSD1.0) | Reference | 6359 | 6350 | 6348 | 634 | 635 | 636 | 639 | 640 | Gene | NCBI | Ensembl ID | Predicted Change |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 9 | rs24562265 | 33714212 | A A | A A | A A | A T | T T | A T | T T | T T | T T | PRR11 | NM_018304 | ENSCAFT00000027880.5 | N|Y (9) |
| 9 | rs851657894 | 33725826 | C C | T C | C C | C C | C C | C C | C C | C C | C C | SMG8 | NM_018149 | ENSCAFG00000017590 | T|I (112) |
| 9 | rs24562703 | 33780316 | G G | G G | G G | A G | G G | G G | G G | G G | G G | GDPD1 | - | ENSCAFG00000017594 | splice region |
| 9 | 34218228 | C C | C C | A C | C C | C C | A A | C C | C C | C C | VMP1 | NM_030938 | ENSCAFT00000078851.1 | P|Q (160) | |
| 9 | 34322737 | G G | G G | G G | G G | G G | G G | G G | G G | T G | TUBD1 | - | ENSCAFT00000062569.1 | splice region | |
| 20 | 56731656–56744321 | INS/INS | DEL/DEL | DEL/DEL | INS/DEL | INS/INS | INS/INS | INS/INS | DEL/DEL | INS/INS | intronic ZNF77.5 | NM_001080404 | intronic deletion | ||
| 20 | rs8708774 | 56749717 | A A | G G | G G | G A | A A | A A | A A | G G | A A | ZNF778 | NM_001201407 | - | N|S (11) |
| 20 | rs8708768 | 56750586 | G G | A A | A A | A G | G G | G G | A G | A A | G G | ZNF77.5 | NM_001080404 | - | G|E (26) |
| 20 | 57228029 | C C | T T | T T | T C | 0 0 | C C | 0 0 | T T | C C | LINGO3 | NM_001101391 | ENSCAFT00000030859.4 | R|C (359) | |
| 20 | 57306977 | C C | T T | 0 0 | 0 0 | C C | C C | T C | C C | C C | DOT1L | NM_0324823 | ENSCAFT00000075488.1 | R|Q (200) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wade, C.M.; Pan, A.Y.H.; Taylor, R.M.; Williamson, P. Cerebellar Abiotrophy in Australian Working Kelpies Is Associated with Two Major Risk Loci. Genes 2022, 13, 1709. https://doi.org/10.3390/genes13101709
Wade CM, Pan AYH, Taylor RM, Williamson P. Cerebellar Abiotrophy in Australian Working Kelpies Is Associated with Two Major Risk Loci. Genes. 2022; 13(10):1709. https://doi.org/10.3390/genes13101709
Chicago/Turabian StyleWade, Claire M., Annie Y. H. Pan, Rosanne M. Taylor, and Peter Williamson. 2022. "Cerebellar Abiotrophy in Australian Working Kelpies Is Associated with Two Major Risk Loci" Genes 13, no. 10: 1709. https://doi.org/10.3390/genes13101709
APA StyleWade, C. M., Pan, A. Y. H., Taylor, R. M., & Williamson, P. (2022). Cerebellar Abiotrophy in Australian Working Kelpies Is Associated with Two Major Risk Loci. Genes, 13(10), 1709. https://doi.org/10.3390/genes13101709





