Gene Expression Profiles Suggest a Better Cold Acclimation of Polyploids in the Alpine Species Ranunculus kuepferi (Ranunculaceae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Design
2.2. RNA Extraction and Sequencing
2.3. Bioinformatics
2.4. qRT-PCR
3. Results
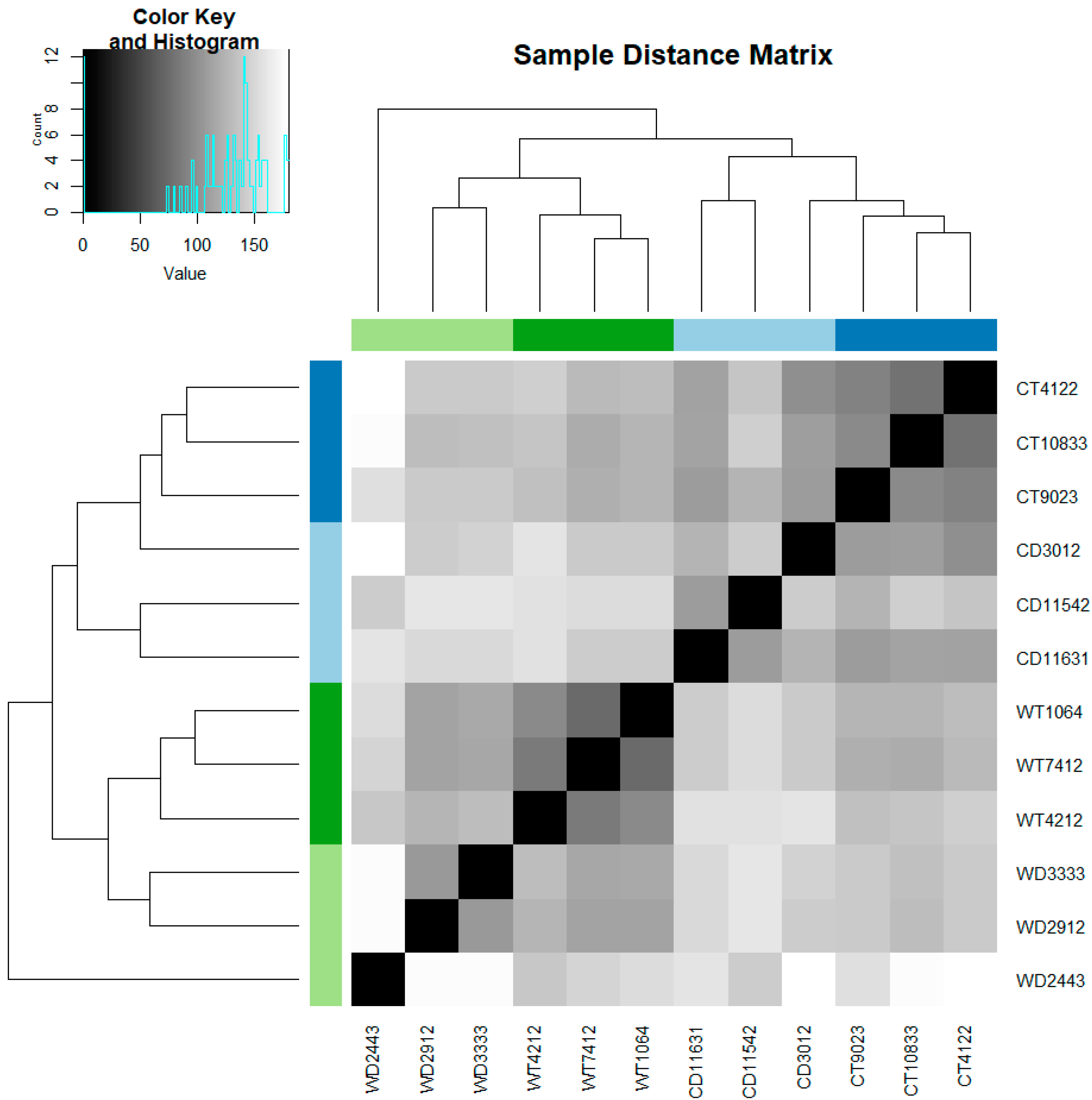
3.1. Pseudoreference and Mapping
3.2. Differential Gene Expression
3.3. Gene Set Enrichment Analyses
3.4. Genes of Interest and qRT-PCR
4. Discussion
4.1. Ploidy Effects on Gene Expression and the Distribution Pattern
4.2. Functional Aspects of Gene Expression Related to Cold Acclimation
4.3. Gene Expression Related to the Epigenetic Mechanism of DNA Methylation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hedhly, A. Sensitivity of flowering plant gametophytes to temperature fluctuations. Environ. Exp. Bot. 2011, 74, 9–16. [Google Scholar] [CrossRef]
- Quint, M.; Delker, C.; Franklin, K.A.; Wigge, P.A.; Halliday, K.J.; van Zanten, M. Molecular and genetic control of plant thermomorphogenesis. Nat. Plants 2016, 2, 15190. [Google Scholar] [CrossRef]
- Gangappa, S.N.; Berriri, S.; Kumar, S.V. PIF4 coordinates thermosensory growth and immunity in Arabidopsis. Curr. Biol. 2017, 27, 243–249. [Google Scholar] [CrossRef]
- Lau, O.S.; Song, Z.; Zhou, Z.; Davies, K.A.; Chang, J.; Yang, X.; Wang, S.; Lucyshyn, D.; Tay, I.H.Z.; Wigge, P.A.; et al. Direct control of SPEECHLESS by PIF4 in the high-temperature response of stomatal development. Curr. Biol. 2018, 28, 1273–1280.e3. [Google Scholar] [CrossRef]
- Casal, J.J.; Balasubramanian, S. Thermomorphogenesis. Annu. Rev. Plant Biol. 2019, 70, 321–346. [Google Scholar] [CrossRef]
- Fiorucci, A.S.; Galvao, V.C.; Ince, Y.C.; Boccaccini, A.; Goyal, A.; Petrolati, L.A.; Trevisan, M.; Fankhauser, C. PHYTOCHROME INTERACTING FACTOR 7 is important for early responses to elevated temperature in Arabidopsis seedlings. New Phytol. 2020, 226, 50–58. [Google Scholar] [CrossRef]
- Chinnusamy, V.; Zhu, J.; Zhu, J.K. Cold stress regulation of gene expression in plants. Trends Plant Sci. 2007, 12, 444–451. [Google Scholar] [CrossRef]
- Liu, T.; Li, Y.; Duan, W.; Huang, F.; Hou, X. Cold acclimation alters DNA methylation patterns and confers tolerance to heat and increases growth rate in Brassica rapa. J. Exp. Bot. 2017, 68, 1213–1224. [Google Scholar] [CrossRef] [PubMed]
- Nagy, L.; Grabherr, G. The Biology of Alpine Habitats; Oxford University Press: New York, NY, USA, 2009. [Google Scholar]
- Wani, S.H.; Singh, N.B.; Haribhushan, A.; Mir, J.I. Compatible solute engineering in plants for abiotic stress tolerance-Role of glycine betaine. Curr. Genet. 2013, 14, 157–165. [Google Scholar] [CrossRef]
- Wani, S.H.; Sah, S.K.; Sanghera, G.; Hussain, W.; Singh, N.B. Genetic engineering for cold stress tolerance in crop plants. In Advances in Genome Science; Atta-ur-Rahman, Ed.; Betham Science: London, UK, 2016; pp. 173–201. [Google Scholar]
- Banerjee, A.; Wani, S.H.; Roychoudhury, A. Epigenetic Control of Plant Cold Responses. Front. Plant Sci. 2017, 8, 1643. [Google Scholar] [CrossRef] [PubMed]
- Körner, C.H. Alpine Plant Life, 3rd ed.; Springer Switzerland AG: Cham, Switzerland, 2021. [Google Scholar]
- Garsed, S.; Davey, H.; Galley, D. The effects of Light and Temperature on the Growth of and Balances of Carbon, Nitrogen and Potassium between Vicia faba L. and Aphis fabae Scop. New Phytol. 1987, 107, 77–102. [Google Scholar] [CrossRef]
- Sanghera, G.S.; Wani, S.H.; Hussain, W.; Singh, N.B. Engineering cold stress tolerance in crop plants. Curr. Genom. 2011, 12, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Theocharis, A.; Clement, C.; AitBarka, E. Physiolgical and molecular changes in plant growth at low temperatures. Planta 2012, 235, 1091–1105. [Google Scholar] [CrossRef]
- Longo, V.; Kamran, R.V.; Michaletti, A.; Toorchi, M.; Zolla, L.; Rinalducci, S. Proteomic and Physiological Response of Spring Barley Leaves to Cold Stress. Int. J. Plant Biol. Res. 2017, 5, 1–10. [Google Scholar]
- Kazemi–Shahandashti, S.S.; Maali-Amiri, R. Global insights of protein responses to cold stress in plants: Signaling, defense and degradation. J. Plant Physiol. 2018, 226, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Megha, S.; Basu, U.; Kav, N.N.V. Regulation of low temperature stress in plants by microRNAs. Plant Cell Dev. 2018, 41, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Yeh, P.J.; Price, T.D. Adaptive phenotypic plasticity and the successful colonization of a novel environment. Am. Nat. 2004, 164, 531–542. [Google Scholar] [CrossRef]
- Sol, D.; Duncan, R.P.; Blackburn, T.M.; Cassey, P.; Lefebvre, L. Big brains, enhanced cognition and response of birds to novel environments. Proc. Natl. Acad. Sci. USA 2005, 102, 5460–6465. [Google Scholar] [CrossRef]
- Schlichting, C.D.; Pigliucci, M. Phenotypic Evolution: A Reaction Norm Perspective; Sinauer: Sunderland, MA, USA, 1998. [Google Scholar]
- West-Eberhard, M.J. Developmental Plasticity and Evolution; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Lande, R. Adaptation to an extraordinary environment by evolution of phenotypic plasticity and genetic assimilation. J. Evolionary Biol. 2009, 22, 1435–1446. [Google Scholar] [CrossRef]
- Whitman, D.W.; Agrawal, A.A. What is phenotypic plasticity and why is it important? In Phenotypic Plasticity of Insects; Ananthakrishnan, T.N., Ed.; Science Publishers: Enfield, NH, USA, 2009; pp. 1–63. [Google Scholar]
- Chevin, L.M.; Lande, R. When do adaptive plasticity and genetic evolution prevent extinction of a density-regulated population? Evolution 2010, 64, 1143–1150. [Google Scholar] [CrossRef]
- Via, S.; Lande, R. Genotype-environment interaction and the evolution of phenotypic plasticity. Evolution 1985, 39, 505–522. [Google Scholar] [CrossRef]
- Schlichting, C.D. The evolution of phenotypic plasticity in plants. Annu. Rev. Ecol. Evol. Syst. 1986, 17, 667–693. [Google Scholar] [CrossRef]
- Sterns, S. The evolutionary significance of phenotypic plasticity. Bioscience 1989, 39, 436–445. [Google Scholar] [CrossRef]
- Pigliucci, M. Phenotypic Plasticity: Beyond Nature and Nurture; John Hopkins Press: Baltimore, MD, USA, 2001. [Google Scholar]
- Franks, S.J.; Weber, J.J.; Aitken, S.N. Evolutionary and plastic responses to climate change in terrestrial plant populations. Evol. Appl. 2013, 7, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Laland, K.N.; Uller, T.; Feldman, M.W.; Sterelny, K.; Müller, G.B.; Moczek, A.; Jablinka, E.; Odling-Smee, J. The extended evolutionary synthesis: Its structure, assumptions and predictions. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151019. [Google Scholar] [CrossRef]
- Kelly, M. Adaptation to climate change through genetic accommodation and assimilation of plastic phenotypes. Philos. Trans. R. Soc. B Biol. Sci. 2019, 374, 20180176. [Google Scholar] [CrossRef]
- Angers, B.; Castonguay, E.; Massicotte, R. Environmentally induced phenotypes and DNA methylation: How to deal with unpredictable conditions until the next generation and after. Mol. Ecol. 2010, 19, 1283–1295. [Google Scholar] [CrossRef]
- Kooke, R.; Johannes, F.; Wardenaar, R.; Becker, F.; Etcheverry, M.; Colot, V.; Vreugdenhil, D.; Keurentjes, J.B. Epigenetic Basis of Morphological Variation and Phenotypic Plasticity in Arabidopsis thaliana. Plant Cell 2015, 27, 337–348. [Google Scholar] [CrossRef]
- Chevin, L.M.; Hoffmann, A.A. Evolution of phenotypic plasticity in extreme environments. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160138. [Google Scholar] [CrossRef]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. BMC Plant Biol. 2011, 11, 163. [Google Scholar] [CrossRef]
- Price, T.D.; Qvarnström, A.; Irwin, D.E. The role of phenotypic plasticity in driving genetic evolution. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2003, 270, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- West-Eberhard, M.J. Phenotypic plasticity. In Encyclopedia of Ecology; Jorgensen, E., Fath, B., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2008; pp. 2701–2707. [Google Scholar]
- Munns, R. Plant adaptations to salt and water stress: Differences and commonalities. Adv. Bot. Res. 2011, 57, 1–32. [Google Scholar]
- Griffith, M.; Yaish, M.W.F. Antifreeze proteins in overwintering plants: A tale of two activities. Trends Plant Sci. 2004, 9, 399–405. [Google Scholar] [CrossRef]
- Nicotra, A.B.; Atkin, O.K.; Bonser, S.P.; Davidson, A.M.; Finnegan, E.J.; Mathesius, U.; Poot, P.; Purugganan, M.D.; Richards, C.L.; Valladares, F.; et al. Plant phenotypic plasticity in a changing climate. Trends Plant Sci. 2010, 15, 684–692. [Google Scholar] [CrossRef]
- Richards, C.L.; Alonso, C.; Becker, C.; Bossdorf, O.; Bucher, E.; Colome-Tatche, M.; Durka, W.; Engelhardt, J.; Gaspar, B.; Gogol-Döring, A.; et al. Ecological plant epigenetics: Evidence from model and non-model species, and the way forward. Ecol. Lett. 2017, 20, 1576–1590. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef]
- Guy, C.L. Cold acclimation and freezing stress tolerance: Role of protein metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1990, 41, 187–223. [Google Scholar] [CrossRef]
- Thomashow, M.F. Plant cold acclimation: Freezing tolerance genes and regulatory mechanisms. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 571–599. [Google Scholar] [CrossRef]
- Ding, Y.; Shi, Y.; Yang, S. Advances and challenges in uncovering cold tolerance regulatory mechanisms in plants. New Phytol. 2019, 222, 1690–1704. [Google Scholar] [CrossRef]
- Jammohammadi, M.; Zolla, L.; Rinalducci, S. Low temperature tolerance in plants: Changes at the protein level. Phytochemistry 2015, 117, 76–89. [Google Scholar] [CrossRef]
- Auld, J.R.; Agrawal, A.A.; Relyea, R.A. Re-evaluating the costs and limits of adaptive phenotypic plasticity. Proc. R. Soc. B Biol. Sci. 2010, 277, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Lu, X.; Gao, Y.; Liu, S.J.; Tao, M.; Xiao, H.; Qiao, Y.Q.; Zhang, Y.P.; Luo, J. Polyploidization and epigenetics. Chin. Sci. Bull. 2011, 3, 245–252. [Google Scholar] [CrossRef]
- te Beest, M.; Le Roux, J.J.; Richardson, D.M.; Brysting, A.K.; Suda, J.; Kubesova, M.; Pysck, P. The more the better? The role of polyploidy in facilitating plant invasions. Ann. Bot. 2012, 109, 19–45. [Google Scholar]
- van de Peer, Y.; Mizrachi, E.; Marchal, K. The evolutionary significance of polyploidy. Nat. Rev. Genet. 2017, 18, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Cronn, R.; Liston, A.; Ashman, T.L. Functional trait divergence and trait plasticity confer polyploidy advantage in heterogeneous environments. New Phytol. 2019, 221, 2286–2297. [Google Scholar] [CrossRef]
- Rice, A.; Smarda, P.; Novosolov, M.; Drori, M.; Glick, L.; Sabath, N.; Meiri, S.; Belmaker, J.; Mayrose, I. The global biogeography of polyploidy plants. Nat. Ecol. Evol. 2019, 3, 265–273. [Google Scholar] [CrossRef]
- Osborn, T.C.; Pires, J.C.; Birchler, J.A.; Auger, D.L.; Chen, Z.J.; Lee, H.-S.; Comai, L.; Madlung, A.; Doerge, R.W.; Colot, V.; et al. Understanding mechanisms of novel gene expression in polyploids. Trends Genet. 2003, 19, 141–147. [Google Scholar] [CrossRef]
- Soltis, D.E.; Visger, C.J.; Soltis, P.S. The polyploidy revolution then... and now: Stebbins revisited. Am. J. Bot. 2014, 101, 1057–1078. [Google Scholar] [CrossRef]
- Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002, 16, 6–21. [Google Scholar] [CrossRef]
- Yan, H.; Kikuchi, S.; Neumann, P.; Zhang, W.; Wu, Y.; Chen, F.; Jiang, J.; Creek, W. Genome-wide mapping of cytosine methylation revealed dynamic DNA methylation patterns associated with genes and centromeres in rice. Plant J. 2010, 63, 353–365. [Google Scholar] [CrossRef]
- Law, J.A.; Jacobsen, S.E. Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 2010, 11, 204–220. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.J.; Scheibe, M.; Wongpalee, S.P.; Liu, W.L.; Cornett, E.M.; Vaughan, R.M.; Li, X.Q.; Chen, W.; Xue, Y.; Zhong, Z.H.; et al. A DNA methylation reader complex that enchances gene transcription. Science 2018, 362, 1182–1186. [Google Scholar] [CrossRef]
- Xiao, X.; Zhang, J.; Li, T.; Fu, X.; Satheesh, V.; Niu, Q.; Lang, Z.; Zhu, J.K.; Lei, M. A group of SUVH methyl-DNA binding proteins regulate expression of the DNA demethylase ROS1 in Arabidopsis. J. Intergrative Plant Biol. 2019, 61, 110–119. [Google Scholar] [CrossRef]
- Bossdorf, O.; Arcuri, D.; Richards, C.L.; Pigliucci, M. Experimental alteration of DNA methylation affects the phenotypic plasticity of ecologically relevant traits in Arabidopsis thaliana. Evol. Ecol. 2010, 24, 541–553. [Google Scholar] [CrossRef]
- Boyko, A.; Blevins, T.; Yao, Y.; Golubov, A.; Bilichak, A.; Ilnytskyy, Y.; Meins, F.; Kovalchuk, I. Transgenerational Adaptation of Arabidopsis to Stress Requires DNA Methylation and the Function of Dicer-Like Proteins. PLoS ONE 2010, 5, e9514. [Google Scholar] [CrossRef]
- Sherman, J.D.; Talbert, L.E. Vernalization-induced changes of the DNA methylation pattern in winter wheat. Genome 2002, 260, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, L.; Zhao, L.; Li, J.; He, S.B.; Zhou, K.; Yang, F.; Huang, M.; Jiang, L.; Li, L.J. Trichostatin A selectively suppresses the cold-induced transcription of ZmDREB1 gene in maize. PLoS ONE 2011, 6, e22132. [Google Scholar] [CrossRef]
- Dowen, R.H.; Pelizzola, M.; Schmitz, R.J.; Lister, R.; Dowen, J.M.; Nery, J.R.; Dixon, J.E.; Ecker, J.R. Widespread dynamic DNA methylation in response to biotic stress. Proc. Natl. Acad. Sci. USA 2012, 109, 2183–2191. [Google Scholar] [CrossRef]
- Vaughn, M.W.; Tanurdzic, M.; Lippman, Z.; Jiang, H.; Carrasquillo, R.; Rabinowicz, P.D.; Dedhia, N.; McCombie, W.R.; Agier, N.; Bulski, A.; et al. Epigenetic Natural Variation in Arabidopsis thaliana. PLoS Biol. 2007, 5, 1617–1629. [Google Scholar] [CrossRef]
- Johannes, F.; Porcher, E.; Teixeira, F.K.; Saliba-Colombani, V.; Simon, M.; Agier, N.; Bulski, A.; Albuisson, J.; Heredia, F.; Audigier, P.; et al. Assessing the Impact of Transgenerational Epigenetic Variation on Complex Traits. PLoS Genet. 2009, 5, e1000530. [Google Scholar] [CrossRef]
- Finnegan, E.J. DNA Methylation a Dynamic Regulator of Genome Organization and Gene Expression in Plants. In Plant Developmental Biology-Biotechnological Perspectives; Pua, E.-C., Davey, M.R., Eds.; Springer: Berlin, Germany, 2010; Volume 2, pp. 295–323. [Google Scholar]
- Hirsch, S.; Baumberger, R.; Grossniklaus, U. Epigenetic variation, inheritance, and selection in plant populations. Cold Spring Harb. Symp. Quant. Biol. 2013, 77, 97–104. [Google Scholar] [CrossRef]
- Adams, K.L.; Wendel, J.F. Polyploidy and genome evolution in plants. Curr. Opin. Plant Biol. 2005, 8, 135–141. [Google Scholar] [CrossRef]
- Grant-Downton, R.T.; Dickinson, H.G. Epigenetics and its implications for Plant Biology: 1. The epigenetic network in plants. Ann. Bot. 2005, 96, 1143–1164. [Google Scholar] [CrossRef]
- Dong, Z.Y.; Wang, Y.M.; Zhang, Z.J.; Shen, Y.; Lin, X.Y.; Ou, X.F.; Han, F.P.; Liu, B. Extent and pattern of DNA methylation alteration in rice lines derived from introgressive hybridization of rice and Zizania latifolia Griseb. Theor. Appl. Genet. 2006, 113, 196–205. [Google Scholar] [CrossRef]
- Baubec, T.; Dinh, H.Q.; Pecinka, A.; Rakic, B.; Rozhon, W.; Wohlrab, B.; von Haeseler, A.; Scheid, O.M. Cooperation of Multiple Chromatin Modifications Can Generate Unanticipated Stability of Epigenetic States in Arabidopsis. Plant Cell 2010, 22, 34–47. [Google Scholar] [CrossRef]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Soltis, D.E.; Buggs, R.J.A.; Doyle, J.J.; Soltis, P.S. What we still don’t know about polyploidy. Taxon 2010, 59, 1387–1403. [Google Scholar] [CrossRef]
- Hegarty, M.J.; Batstone, T.; Barker, G.L.; Edwards, K.J.; Abbott, R.J.; Hiscock, S.J. Nonadditive changes to cytosine methylation as a consequence of hybridization and genome duplication in Senecio (Asteraceae). Mol. Ecol. 2011, 20, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Madlung, A.; Wendel, J.F. Genetic and Epigenetic Aspects of Polyploid Evolution in Plants. Cytogenet. Genome Res. 2013, 140, 270–285. [Google Scholar] [CrossRef]
- Alonso, C.; Balao, F.; Bazaga, P.; Perez, R. Epigenetic contribution to successful polyploidizations: Variation in glabal cytosine methylation along an extensive ploidy series in Dianthus broteri (Caryophyllaceae). New Phytol. 2016, 212, 571–576. [Google Scholar] [CrossRef]
- Brandshaw, A.D. Evolutionary significance of phenotypic plasticity in plants. Adv. Genet. 1965, 13, 115–155. [Google Scholar]
- Donelson, J.M.; Salinas, S.; Munday, P.L.; Shama, L.N.S. Transgenerational plasticity and climate change experiments: Where do we go from here? Glob. Chang. Biol. 2017, 24, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Furumoto, T. Cold Signaling and Cold Response in Plants. Int. J. Mol. Sci. 2013, 14, 5312–5337. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Li, X.M.; Lin, H.X.; Chong, K.; Merchant, S.S. Crop Improvement through Temperature Resilience. Annu. Rev. Plant Biol. 2019, 70, 753–780. [Google Scholar] [CrossRef] [PubMed]
- Burnier, J.; Buerki, S.; Arrigo, N.; Küpfer, P.; Alvarez, N. Genetic structure and evolution of Alpine polyploid complexes: Ranunculus kuepferi (Ranunculaceae) as a case study. Mol. Ecol. 2009, 18, 3730–3744. [Google Scholar] [CrossRef] [PubMed]
- Cosendai, A.C.; Hörandl, E. Cytotype stability, facultative apomixis and geographical parthenogenesis in Ranunculus kuepferi (Ranunculaceae). Ann. Bot. 2010, 105, 457–470. [Google Scholar] [CrossRef][Green Version]
- Cosendai, A.C.; Rodewald, J.; Hörandl, E. Origin and distribution of autopolyploids via apomixis in the alpine species Ranunculus kuepferi (Ranunculaceae). Taxon 2011, 60, 355–364. [Google Scholar] [CrossRef]
- Kirchheimer, B.; Schinkel, C.C.-F.; Dellinger, A.S.; Klatt, S.; Moser, D.; Winkler, M.; Lenoir, J.; Caccianiga, M.; Guisan, A.; Nieto-lugilde, D.; et al. A matter of scale: Apparent niche differentiation of diploid and tetraploid plants may depend on extent and grain of analysis. J. Biogeogr. 2016, 43, 716–726. [Google Scholar] [CrossRef] [PubMed]
- Schinkel, C.C.-F.; Kirchheimer, B.; Dellinger, A.S.; Klatt, S.; Winkler, M.; Dullinger, S.; Hörandl, E. Correlations of polyploidy and apomixis with elevation and associated environmental gradients in an alpine plant. AoB Plants 2016, 8, plw064. [Google Scholar] [CrossRef]
- Cosendai, A.C.; Wagner, J.; Ladinig, U.; Rosche, C.; Hörandl, E. Geographical parthenogenesis and population genetic structure in the alpine species Ranunculus kuepferi (Ranunculaceae). Heredity 2013, 110, 560–569. [Google Scholar] [CrossRef]
- Hörandl, E. The complex causality of geographical parthenogenesis. New Phytol. 2006, 171, 525–538. [Google Scholar] [CrossRef]
- Küpfer, P. Recherches surlesliens de parente entre la floreorophile des Alpesetcelle des Pyrenees. Boissiera 1974, 23, 1–322. [Google Scholar]
- Kirchheimer, B.; Wessely, J.; Gattringer, A.; Hülber, K.; Moser, D.; Schinkel, C.C.-F.; Appelhans, M.; Klatt, S.; Caccianiga, M.; Dellinger, A.; et al. Reconstructing geographical parthenogenesis: Effects of niche differentiation and reproductive mode on Holocene range expansion of an alpine plant. Ecol. Lett. 2018, 21, 392–401. [Google Scholar] [CrossRef]
- Schinkel, C.C.-F.; Kirchheimer, B.; Dullinger, S.; Geelen, D.; De Storme, N.; Hörandl, E. Pathways to polyploidy: Indications of a female triploid bridge in the alpine species Ranunculus kuepferi (Ranunculaceae). Plant Syst. Evol. 2017, 303, 1093–1108. [Google Scholar] [CrossRef] [PubMed]
- Schinkel, C.C.-F.; Syngelaki, E.; Kirchheimer, B.; Dullinger, S.; Klatt, S.; Hörandl, E. Epigenetic patterns and geographical parthenogenesis in the alpine plant species Ranunculus kuepferi (Ranunculaceae). Int. J. Mol. Sci. 2020, 21, 3318. [Google Scholar] [CrossRef] [PubMed]
- Syngelaki, E.; Schinkel, C.C.-F.; Klatt, S.; Hörandl, E. Effects of temperature treatments on cytosine-methylation profiles of diploid and autotetraploid plants of the alpine species Ranunculus kuepferi (Ranunculaceae). Front. Plant Sci. 2020, 11, 435. [Google Scholar] [CrossRef]
- Syngelaki, E.; Daubert, M.; Klatt, S.; Hörandl, E. Phenotypic responses, reproduction mode and epigenetic patterns under temperature treatments in the alpine plant species Ranunculus kuepferi (Ranunculaceae). Biology 2020, 9, 315. [Google Scholar] [CrossRef]
- Klatt, S.; Schinkel, C.C.-F.; Kirchheimer, B.; Dullinger, S.; Hörandl, E. Effects of cold treatments on fitness and mode of reproduction in the diploid and polyploid alpine plant Ranunculus kuepferi (Ranunculaceae). Ann. Bot. 2018, 121, 1287–1298. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S. FastQC: A Quality Control Yool for High Throughput Eequence Data. Available online: http://www.bioinformatics.bbsrc.ac.uk/projects/fastqc (accessed on 10 February 2020).
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Trinity: Reconstructing a full-length transcriptome without a genome from RNA-Seq data. Nat. Biotechnol. 2013, 29, 644–652. [Google Scholar]
- Manni, M.; Berkeley, M.R.; Seppey, M.; Simao, F.A.; Zdobnov, E.M. BUSCO Update: Novel and Streamlined Workflows along with Broader and Deeper Phylogenetic Coverage for Scoring of Eukaryotic, Prokaryotic, and Viral Genomes. Available online: http://arxiv.org/abs/2106.11799 (accessed on 23 March 2020).
- Haas, B.J.; Delcher, A.L.; Mount, S.M.; Wortman, J.R.; Smith, R.K.; Hannick, L.I.; Maiti, R.; Ronning, C.M.; Rusch, D.B.; Town, C.D.; et al. Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nucleic Acids Res. 2013, 31, 5654–5666. [Google Scholar] [CrossRef]
- Johnson, M.; Zaretskaya, I.; Raytselis, Y.; Merezhuk, Y.; McGinnis, S.; Madden, T.L. NCBI BLAST: A better web interface. Nucleic Acids Res. 2008, 36, W5–W9. [Google Scholar] [CrossRef] [PubMed]
- Bryant, D.M.; Johnson, K.; DiTommaso, T.; Tickle, T.; Couger, M.B.; Payzin-Dogru, D.; Lee, T.J.; Leigh, N.D.; Kuo, T.H.; Davis, F.G.; et al. A Tissue-Mapped Axolotl De Novo Transcriptome Enables Identification of Limb Regeneration Factors. Cell Rep. 2017, 18, 762–776. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Smyth, G.K.; Shi, W. The R package Rsubread is easier, faster, cheaper and better for alignment and quantification of RNA sequencing reads. Nucleic Acids Res. 2019, 47, e47. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 7 May 2021).
- RStudio Team. RStudio: Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2016; Available online: http://www.rstudio.com/ (accessed on 7 May 2021).
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Jafari, M.; Ansari-Pour, N. Why, When and How to Adjust your P Values? Cell J. 2019, 20, 604–607. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009. [Google Scholar]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Pages, H.; Carlson, M.; Falcon, S.; Li, N. AnnotationDbi: Manipulation of SQLite-Based Annotations in Bioconductor. R Package Version 1.55.1. Available online: https://bioconductor.org/packages/AnnotationDbi (accessed on 2 May 2021).
- Bell, C.; Soltis, D.; Soltis, P. The age and diversification of the angiosperms re-visited. Am. J. Bot. 2010, 97, 1296–1303. [Google Scholar] [CrossRef]
- Yu, G. Enrichplot: Visualization of Functional Enrichment Result. R Package Version 1.13.1. Available online: https://yulab-smu.top/biomedical-knowledge-mining-book/ (accessed on 7 May 2021).
- Ritonga, F.N.; Chen, S. Physiological and Molecular Mechanism Involved in Cold Stress Tolerance in Plants. Plants 2020, 9, 560. [Google Scholar] [CrossRef] [PubMed]
- Finnegan, E.J.; Kovac, K.A. Plant DNA methyltransferases. Plant Mol. Biol. 2000, 43, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Matzke, M.A.; Mosher, R.A. RNA-directed DNA methylation: An epigenetic pathway of increasing complexity. Nat. Rev. Genet. 2014, 15, 394–408. [Google Scholar] [CrossRef]
- Bewick, A.J.; Niederhuth, C.E.; Ji, L.; Rohr, N.A.; Griffin, P.T.; Leebens-Mack, J.; Schmitz, R.J. The evolution of CHROMOMETHYLASES and gene body DNA methylation in plants. Genome Biol. 2017, 18, 65. [Google Scholar] [CrossRef]
- Lanciano, S.; Mirouze, M. DNA Methylation in Rice and Relevance for Breeding. Epigenomes 2017, 1, 10. [Google Scholar] [CrossRef]
- Bräutigam, K.; Cronk, Q. DNA Methylation and the Evolution of Developmental Complexity in Plants. Front. Plant Sci. 2018, 9, 1447. [Google Scholar] [CrossRef]
- Chang, Y.N.; Chen, Z.; Jiang, J.; Zhang, H.; Zhu, J.K.; Duan, C.G. Epigenetic regulation in plant abiotic stress responses. J. Intergrative Plant Biol. 2020, 62, 563–580. [Google Scholar] [CrossRef]
- Joseph, J.T.; Poolakkalody, N.J.; Shah, J.M. Plant reference gene for development and stress response studies. J. Biosci. 2018, 43, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhang, G.; Chen, Y.; Bai, Q.; Gao, C.; Zeng, L.; Li, Z.; Cheng, Y.; Chen, J.; Sun, X.; et al. Selection of Reference Genes for qPCR Analyses of Gene Expression in Ramie Leaves and Roots across Eleven Abiotic/Biotic Treatment. Sci. Rep. 2019, 9, 20004. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Smallwood, M.; Bowles, D.J. Plants in a cold climate. Philos. Trans. R. Soc. B Biol. Sci. 2002, 357, 831–846. [Google Scholar] [CrossRef]
- Zhu, J.; Jeong, J.; Zhu, Y.; Sokolchik, I.; Miyazaki, S.; Zhu, J.K.; Hasegawa, P.M.; Bohnert, H.J.; Shi, H.; Yun, D.J.; et al. Involvement of Arabidopsis HOS15 in histone deacetylation and cold tolerance. Proc. Natl. Acad. Sci. USA 2007, 105, 4945–4950. [Google Scholar] [CrossRef] [PubMed]
- Hannah, M.A.; Heyer, A.G.; Hincha, D.K. A global survey of gene regulation during cold acclimation in Arabidopsis thaliana. PLoS Genet. 2005, 1, 179–196. [Google Scholar]
- Knight, M.R.; Knight, H. Low-temperature perception leading to gene expression and cold tolerance in higher plants. New Phytol. 2012, 195, 737–751. [Google Scholar] [CrossRef]
- Fowler, S.; Thomashow, M.F. Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 2002, 14, 1675–1690. [Google Scholar] [CrossRef]
- Robinson, S.J.; Parkin, I.A. Differential SAGE analysis in Arabidopsis uncovers increased transcriptome complexity in response to low temperature. BMC Genom. 2008, 9, 434. [Google Scholar] [CrossRef] [PubMed]
- Nohales, M.A.; Kay, S.A. Molecular mechanisms at the core of the plant circadian oscillator. Nat. Struct. Mol. Biol. 2016, 23, 1061–1069. [Google Scholar] [CrossRef]
- Pellino, M.; Hojsgaard, D.; Hörandl, E.; Sharbel, T. Chasing the apomictic factors in the Ranunculus auricomus complex: Exploring gene expression patterns in microdissected sexual and apomictic ovules. Genes 2020, 11, 728. [Google Scholar] [CrossRef]
- Wagner, A. Energy constrains on the evolution of gene expression. Mol. Biol. Evol. 2005, 22, 1365–1374. [Google Scholar] [CrossRef]
- Drummond, D.A.; Wilke, C.O. Mistranslation-induced protein misfolding as a dominant constraint on coding-sequence evolution. Cell 2008, 134, 341–352. [Google Scholar] [CrossRef]
- Qian, W.; Liao, B.Y.; Chang, A.Y.F.; Zhang, J. Maintenance of duplicate genes and their functional redundancy by reduced expression. Trends Genet. 2010, 26, 425–430. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Y.; Xia, E.H.; Yao, Q.Y.; Liu, X.D.; Gao, L.Z. Autotetraploid rice methylome analysis reveals methylation variation of transposable elements and their effects on gene expression. Proc. Natl. Acad. Sci. USA 2015. [Google Scholar] [CrossRef] [PubMed]
- Bateson, P. Why are individuals so different from each other? Heredity 2015, 115, 285–292. [Google Scholar] [CrossRef]
- Takahashi, S.; Seki, M.; Ishida, J.; Satou, M.; Sakurai, T.; Narusaka, M.; Kamiya, A.; Nakajima, M.; Enju, A.; Akiyama, K.; et al. Monitoring the expression profiles of genes induced by hyperosmotic, high salinity, and oxidative stress and abscisic acid treatment in Arabidopsis cell culture using full-length cDNA microarray. Plant Mol. Biol. 2004, 56, 29–55. [Google Scholar] [CrossRef]
- Puijalon, S.; Bouma, T.J.; Douady, C.J.; Groenendael, J.V.; Anten, N.P.R.; Martel, E.; Bornette, G. Plant resistanse to mechanical stress: Evidence of an avoidance-Tolerance trade-off. New Phytol. 2011, 191, 1141–1149. [Google Scholar] [CrossRef]
- Jutsz, A.M.; Gnida, A. Mechanisms of stress avoidance and tolerance by plants used in phytoremediation of heavy metals. Arch. Environ. Prot. 2015, 41, 104–114. [Google Scholar] [CrossRef]
- Vrijenhoek, R.C.; Parker, E.D. Geographical parthenogenesis: General purpose genotypes and frozen niche variation BT. In Lost Sex: The Evolutionary Biology of Parthenogenesis; Schön, I., Martens, K., Dijk, P., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 99–131. [Google Scholar]
- Chen, L.; Zhao, Y.; Xu, S.; Zhang, Z.; Xu, Y.; Zhang, J.; Chong, K. Os MADS 57 together with Os TB 1 coordinates transcription of its target Os WRKY 94 and D14 to switch its organogenesis to defense for cold tolerance in rice. J. Exp. Bot. 2018, 63, 6467–6480. [Google Scholar]
- de Palma, M.; Grillo, S.; Massarelli, I.; Costa, A.; Balong, G.; Vigh, L.; Leone, A. Regulation of desaturase gene expression, changes in membrane lipid composition and freezing tolerance in potato plants. Mol. Breed. 2008, 21, 138–149. [Google Scholar] [CrossRef]
- Janska, A.; Marsik, P.; Zelenkova, S.; Ovesna, J. Cold stress and acclimation–what is important for metabolic adjustment? Plant Biol. 2010, 12, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Dutilleul, C.; Benhaissaine-Kesri, G.; Demandre, C.; Reze, N.; Launay, A.; Pelletier, S.; Renou, J.P.; Zachowski, A.; Baudouin, E.; Guillas, I. Phytosphingosine-phosphate is a signal for AtMPK6 action and Arabidopsis response to chilling. New Phytol. 2012, 194, 282–292. [Google Scholar] [CrossRef]
- Murata, N.; Los, D.A. Membrane fluidity and temperature perception. Plant Physiol. 1997, 115, 875–879. [Google Scholar] [CrossRef]
- Martiniere, A.; Shvedunova, M.; Thomshon, A.J.; Evans, N.H.; Penfield, S.; Runions, J.; McWatters, H.G. Homeostasis of plasma membrane viscosity in fluctuating temperatures. New Phytol. 2011, 192, 328–337. [Google Scholar] [CrossRef]
- Ma, H.; Liu, M. The microtubule cytoskeleton acts as a sensor for stress response signaling in plants. Mol. Biol. Rep. 2019, 46, 5603–5608. [Google Scholar] [CrossRef] [PubMed]
- Orvar, B.L.; Sangwan, V.; Omann, F.; Dhindsa, R. Early steps in cold sensing by plant cells: The role of actin cytoskeleton and membrane fluidity. Plant J. 2000, 23, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, V.; Örvar, B.L.; Dhindsa, R.S. Early events during low temperature signaling. In Plant Cold Hardiness; Li, C., Palva, E.T., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 43–53. [Google Scholar]
- Zhu, J.K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–314. [Google Scholar] [CrossRef]
- Tenhaken, R. Cell wall remodeling under abiotic stress. Front. Plant Sci. 2014, 5, 771. [Google Scholar] [CrossRef]
- Sulova, Z.; Baran, R.; Farkas, V. Divergent modes of action on xyloglycan of two isoenzymes of xyloglycan endo-transglycosylase from Tropaelum Majus. Plant Physiol. Biochem. 2003, 41, 431–437. [Google Scholar] [CrossRef]
- Eklof, J.M.; Brumer, H. The XTH gene family: An update on enzyme structure, function, and phylogeny in xyloglycan remodeling. Plant Physiol. 2010, 153, 456–466. [Google Scholar] [CrossRef]
- Pokorna, J.; Schwarzerova, K.; Zelenkova, S.; Petrasek, J.; Janotova, I.; Capkova, V.; Opatrny, Z. Sites of actin filament initiation and reorganization in cold-treated tobacco cells. Plant Cell Dev. 2004, 27, 641–653. [Google Scholar] [CrossRef]
- Abdrakhamanova, A.; Wang, Q.Y.; Khokhlova, L.; Nick, P. Is microtubule disassembly a trigger for cold acclimation? Plant Cell Physiol. 2003, 44, 676–686. [Google Scholar] [CrossRef]
- Monroy, A.F.; Sarhan, F.; Dhindsa, R.S. Cold-induced changes in freezing tolerance, protein phosphorylation and gene expression (evidence for a role of calcium). Plant Physiol. 1993, 102, 1227–1235. [Google Scholar] [CrossRef]
- Lewis, B.D.; Karlin-Neumann, G.; Davis, R.W.; Spalding, E.P. Ca2+-activated anion channels and membrane depolarizations induced by blue light and cold in Arabidopsis seedlings. Plant Physiol. 1997, 114, 1327–1334. [Google Scholar] [CrossRef]
- Sanders, D.; Pelloux, J.; Brownlee, C.; Harper, J.F. Calcium at the crossroads of signaling. Plant Cell 2002, 14, 401–417. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Ding, Y.; Yang, S. Cold signal transduction and its interplay with phytohormones during cold acclimation. Plant Cell Physiol. 2015, 56, 7–15. [Google Scholar] [CrossRef]
- Zhu, X.; Feng, Y.; Liang, G.; Liu, N.; Zhu, J.K. Aequorin-based luminescence imaging reveals stimulus- and tissue-specific Ca2+ dynamics in Arabidopsis plants. Mol. Plant 2013, 6, 444–455. [Google Scholar] [CrossRef] [PubMed]
- Plieth, C. Temperature sensing by plants: Calcium-permeable channels as primary sensors- a model. J. Membr. Biol. 1999, 172, 121–127. [Google Scholar] [CrossRef]
- White, P.J. Depolarization-activated calcium channels shape the calcium signatures induced by low-temperature stress. New Phytol. 2009, 183, 6–8. [Google Scholar] [CrossRef]
- Allen, G.J.; Chu, S.P.; Schumacher, K.; Shimazaki, C.T.; Vafeados, D.; Kemper, A.; Hawke, S.D.; Tallman, G.; Tsien, R.Y.; Harper, J.F.; et al. Alteration of stimulus-specific guard cell calcium oscillations and stomatal closing in Arabidopsis det3 mutant. Science 2000, 289, 2338–2342. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Yan, W.; Guo, J.; Du, B.; Chen, R.; Zhu, L.; He, G. A rice lectin receptor-like kinase that is involved in innate immune responses also contributes to seed germination. Plant J. 2013, 76, 687–698. [Google Scholar] [CrossRef]
- Ye, Y.; Ding, Y.; Jiang, Q.; Wang, F.; Sun, J. The role of receptor-like protein kinases (RLKs) in abiotic stress response in plants. Plant Cell Rep. 2017, 36, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, C.; Zhu, Y.; Zhang, L.; Chen, T.; Zhou, F.; Chen, H.; Lin, Y. The calcium-dependent kinase OsCPK24 functions in cold stress responses in rice. J. Intergrative Plant Biol. 2018, 60, 173–188. [Google Scholar] [CrossRef] [PubMed]
- Knight, H.; Trewavas, A.J.; Knight, M.R. Cold calcium signaling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell 1996, 8, 489–503. [Google Scholar] [PubMed]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed.; Sinauer Associates, Inc.: Sunderland, UK, 2010. [Google Scholar]
- Zhu, J.K. Epigenetic sequencing comes of age. Cell 2008, 133, 395–397. [Google Scholar] [CrossRef] [PubMed]
- Chinnusamy, V.; Zhu, J.K. Epigenetic regulation of stress responses in plants. Curr. Opin. Plant Biol. 2009, 12, 133–139. [Google Scholar] [CrossRef]
- Kim, J.M.; Sasaki, T.; Ueda, M.; Sako, K.; Seki, M. Chromatin changes in response to drought, salinity, heat and cold stresses in plants. Front. Plant Sci. 2015, 6, 114. [Google Scholar] [CrossRef]
- Strahl, B.D.; Allis, C.D. The language of covalent histone modifications. Nature 2000, 403, 41–45. [Google Scholar] [CrossRef]
- Lang-Mladek, C.; Popova, O.; Kiok, K.; Berlinger, M.; Rakic, B.; Aufsatz, W.; Jonak, C.; Hauser, M.T.; Lusching, C. Transgenerational inheritance and resetting of stress-induced loss of epigenetic gene silencing in Arabidopsis. Mol. Plant 2010, 3, 594–602. [Google Scholar] [CrossRef]
- Kumar, S.V.; Wigge, P.A. H2A.Z-containing nucleosomes mediate the thermosensory response of Arabidopsis. Cell 2010, 140, 136–147. [Google Scholar]
- Kumar, S.V.; Lucyshyn, D.; Jaeger, K.E.; Alos, E.; Harberd, N.P.; Wigge, P.A. Transcription factor PIF4 controls the thermosensory activation of flowering. Nature 2012, 484, 242–245. [Google Scholar] [CrossRef]


| No. Plants | Cold Treatment | Warm Treatment | ||
|---|---|---|---|---|
| 164 | 189 | |||
| Diploid | Tetraploid | Diploid | Tetraploid | |
| 74 | 90 | 92 | 97 | |
| Light regime (μmol m−2 s−1, SAR) | ~700 | |||
| Photoperiod (h) | 16; 10 of full light and 3 + 3 of twilight | |||
| Temperature during the light/dark period (°C) | ||||
| Daytime | 7 | 15 | ||
| Night | 2 −1 (cold shocks for three nights per week) | 10 | ||
| Group † | Sample ID | Untrimmed Reads | Total Trimmed Reads | Mapped Reads | Mapped Reads (%) |
|---|---|---|---|---|---|
| CD | 30_1_2 | 1,335,657 | 1,326,485 | 1,214,557 | 91.56 |
| CD | 115_4_2 | 39,260,122 | 39,044,345 | 37,240,848 | 95.38 |
| CD | 116_3_1 | 46,805,434 | 46,545,107 | 43,938,918 | 94.4 |
| CT | 41_2_2 | 37,813,775 | 37,625,107 | 34,877,792 | 92.7 |
| CT | 90_2_3 | 35,989,875 | 35,824,317 | 33,353,051 | 93.1 |
| CT | 108_3_3 | 36,888,834 | 36,687,214 | 33,980,212 | 92.62 |
| WD | 24_4_3 | 29,197,541 | 29,058,835 | 27,467,724 | 94.52 |
| WD | 29_1_2 | 34,122,764 | 33,708,095 | 31,629,139 | 93.83 |
| WD | 33_3_3 | 29,530,425 | 29,378,666 | 27,521,885 | 93.68 |
| WT | 42_1_2 | 29,277,225 | 29,108,345 | 26,919,488 | 92.48 |
| WT | 74_1_2 | 31,858,591 | 31,626,671 | 28,839,855 | 91.19 |
| WT | 106_4_1 | 30,939,580 | 30,551,281 | 27,988,255 | 91.61 |
| Gene Count | Percentage (%) | |
|---|---|---|
| Expressed genes | 19,033 | |
| Differentially expressed genes | 2617 | 13.75 |
| Up-regulated | 1055 | 5.5 |
| Down-regulated | 1562 | 8.2 |
| Outliers | 469 | 2.5 |
| GeneID | Organism ‡ | Function | Regulation |
|---|---|---|---|
| AGL15 | BRANA | Agamous-like MADS-box protein | Down |
| AGL62 | ARATH | Agamous-like MADS-box protein | Down |
| AGO1A | ORYSJ | Protein argonaute 1A | Down |
| AGO4B | ORYSJ | Protein argonaute 4B | Down |
| ALKB2 | ARATH | DNA oxidative demethylase | Down |
| AP1 | VITVI | Agamous-like MADS-box protein | Down |
| ATX4 | ARATH | Histone-lysine N-methyltransferase | Up |
| CAMT3 | PETHY | Caffeoyl-CoA O-methyltransferase 3 | Down |
| CMTA3 | ARATH | Calmodulin-binding transcription activator | Down |
| COMT1 | POPKI | Caffeic acid 3-O-methyltransferase | Down |
| DRM1L | ARATH | DNA (cytosine-5)-methyltransferase | Down |
| EPFL2 | ARATH | EPIDERMAL PATTERNING FACTOR-like protein 2 | Down |
| EPFL6 | ARATH | EPIDERMAL PATTERNING FACTOR-like protein 6 | Down |
| EPFL9 | ARATH | EPIDERMAL PATTERNING FACTOR-like protein 9 | Down |
| FDM1 | ARATH | Factor of DNA methylation 1 | Up |
| JM706 | ORYSJ | Lysine-specific demethylase | Down |
| JMJ25 | ARATH | Lysine-specific demethylase | Down |
| LAMT | CATRO | Loganic acid O-methyltransferase | Down |
| MADS1 | VITVI | Agamous-like MADS-box protein | Up |
| MBD2 | ARATH | Methyl-CpG-binding domain-containing protein 2 | Up |
| MBD6 | ARATH | Methyl-CpG-binding domain-containing protein 6 | Down |
| METE | CATRO | 5-methyltetrahydropteroyltriglutamate--homocysteine methyltransferase | Down |
| METE2 | ORYSJ | 5-methyltetrahydropteroyltriglutamate--homocysteine methyltransferase 2 | Down |
| PEAM1 | ARATH | Phosphoethanolamine N-methyltransferase 1 | Down |
| PMT1 | ARATH | Probable methyltransferase PMT1 | Down |
| PMT2 | ARATH | Probable methyltransferase | Down |
| PMT4 | ARATH | Probable methyltransferase | Down |
| PMT7 | ARATH | Probable methyltransferase | Down |
| PMT8 | ARATH | Probable methyltransferase | Down |
| PMTB | ARATH | Probable methyltransferase | Down |
| PMTD | ARATH | Probable pectin methyltransferase | Down |
| PMTI | ARATH | Probable methyltransferase | Down |
| PMTQ | ARATH | Probable methyltransferase | Down |
| PMTT | ARATH | Probable pectin methyltransferase | Down |
| RP6L1 | ARATH | Protein RRP6-like 1 | Up |
| RP6L2 | ARATH | Protein RRP6-like 2 | Up |
| RRP8 | ARATH | Ribosomal RNA-processing protein 8 | Up |
| SUVR1 | ARATH | Probable inactive histone-lysine N-methyltransferase | Down |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Syngelaki, E.; Paetzold, C.; Hörandl, E. Gene Expression Profiles Suggest a Better Cold Acclimation of Polyploids in the Alpine Species Ranunculus kuepferi (Ranunculaceae). Genes 2021, 12, 1818. https://doi.org/10.3390/genes12111818
Syngelaki E, Paetzold C, Hörandl E. Gene Expression Profiles Suggest a Better Cold Acclimation of Polyploids in the Alpine Species Ranunculus kuepferi (Ranunculaceae). Genes. 2021; 12(11):1818. https://doi.org/10.3390/genes12111818
Chicago/Turabian StyleSyngelaki, Eleni, Claudia Paetzold, and Elvira Hörandl. 2021. "Gene Expression Profiles Suggest a Better Cold Acclimation of Polyploids in the Alpine Species Ranunculus kuepferi (Ranunculaceae)" Genes 12, no. 11: 1818. https://doi.org/10.3390/genes12111818
APA StyleSyngelaki, E., Paetzold, C., & Hörandl, E. (2021). Gene Expression Profiles Suggest a Better Cold Acclimation of Polyploids in the Alpine Species Ranunculus kuepferi (Ranunculaceae). Genes, 12(11), 1818. https://doi.org/10.3390/genes12111818






