Immunophenotyping of a Stromal Vascular Fraction from Microfragmented Lipoaspirate Used in Osteoarthritis Cartilage Treatment and Its Lipoaspirate Counterpart
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Stromal Vascular Fraction Isolation
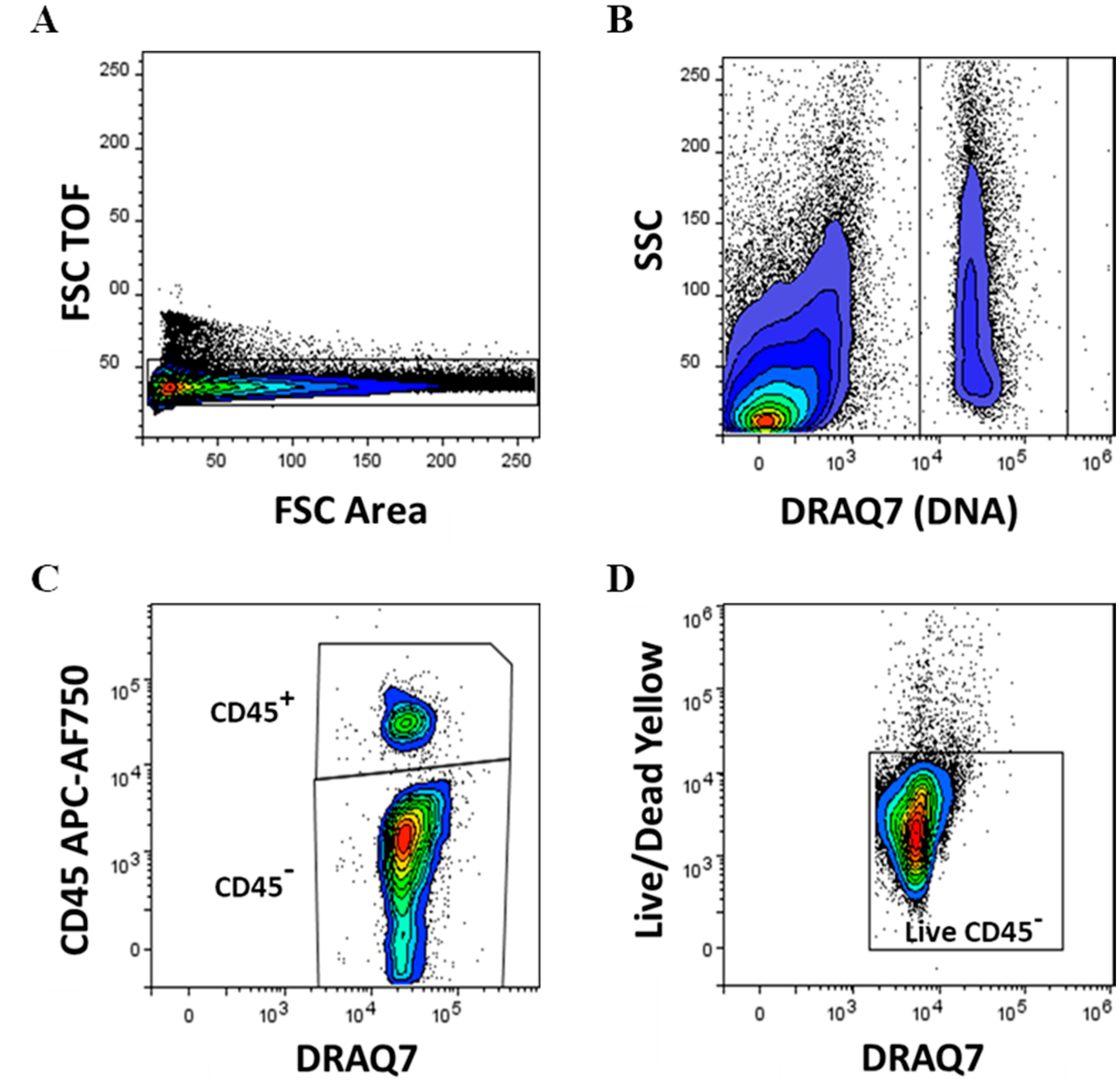
2.3. Flow Cytometry
2.4. Statistical Analysis
3. Results
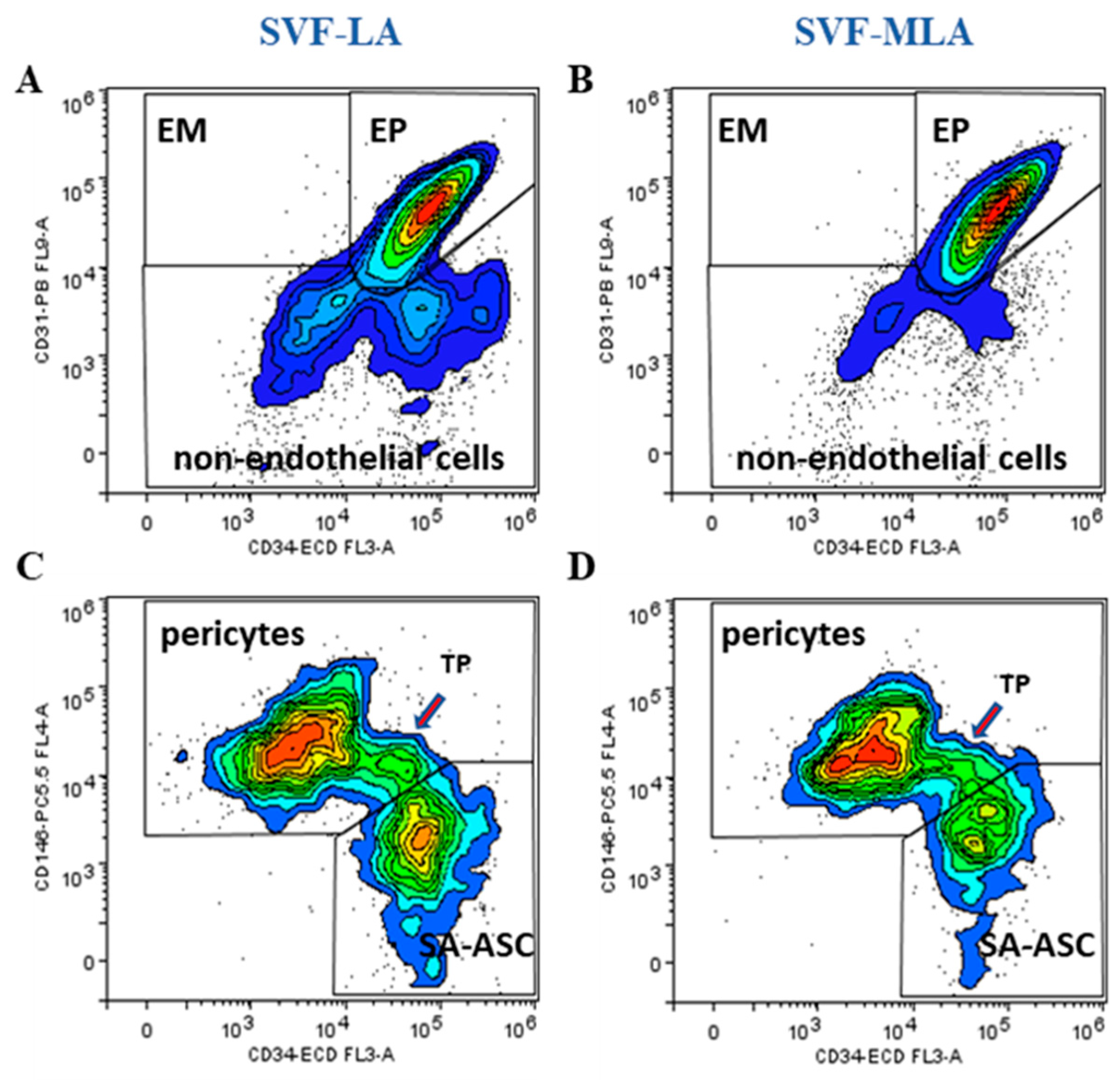
3.1. Immunophenotyping Analysis of Stromal Vascular Fraction from Lipoasirate and Microfragmented Lipoasirate Samples by Polychromatic Flow Cytometry
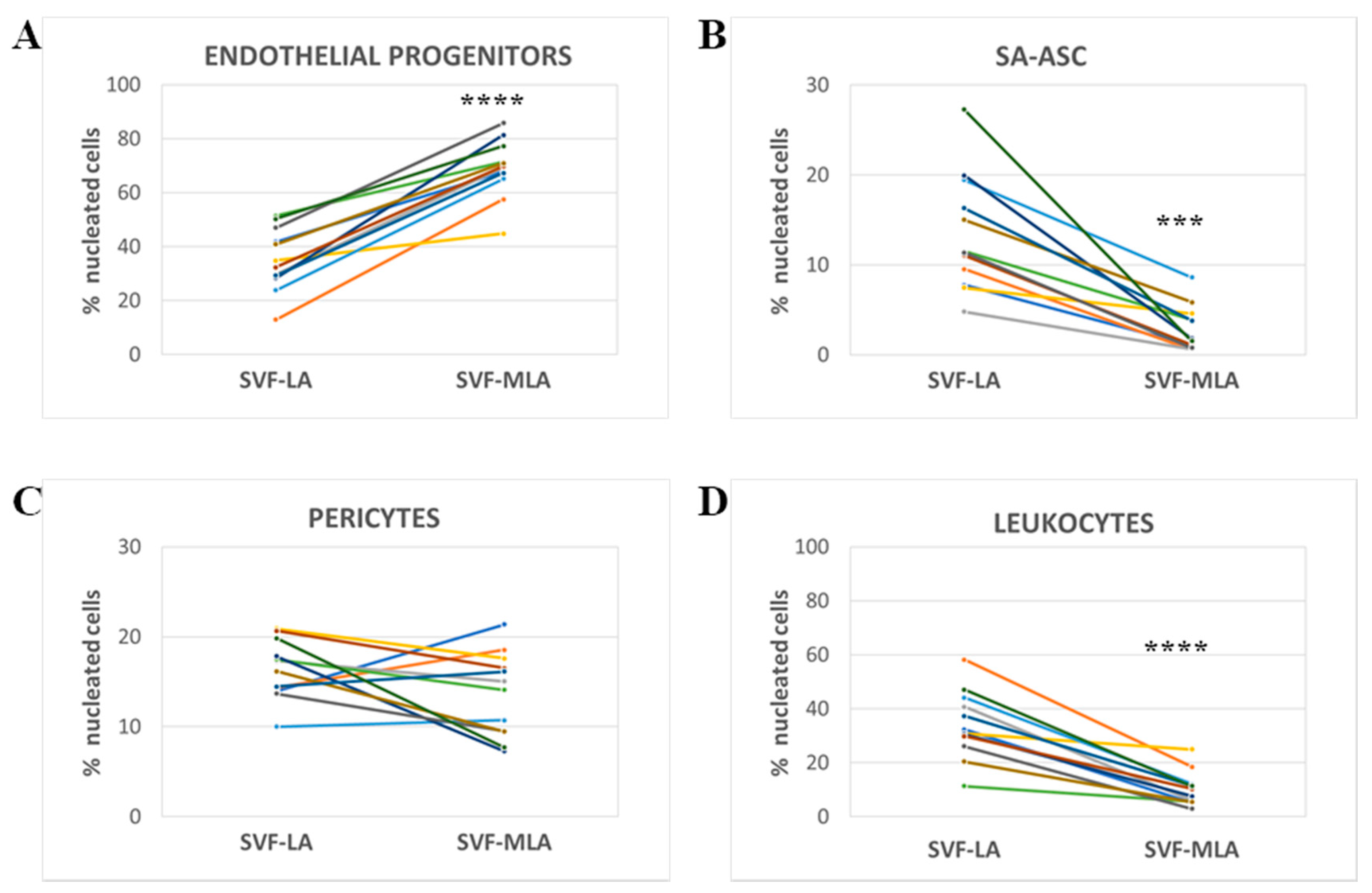
3.2. Stromal Vascular Fraction from Microfragmented Lipoasirate Significantly Differs in Cell Content from Stromal Vascular Fraction from Lipoasirate
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Cao, Y.; Sun, Z.; Liao, L.M.; Meng, Y.; Han, Q.; Zhao, R.C.H. Human adipose tissue-derived stem cells differentiate into endothelial cells in vitro and improve postnatal neovascularization in vivo. Biochem. Biophys. Res. Commun. 2005, 332, 370–379. [Google Scholar] [CrossRef]
- Murphy, M.B.; Moncivais, K.; Caplan, A.I. Mesenchymal stem cells: Environmentally responsive therapeutics for regenerative medicine. Exp. Mol. Med. 2013, 45, e54. [Google Scholar] [CrossRef]
- Caplan, A.I. Mesenchymal stem cells: Time to change the name! Stem Cells Transl. Med. 2017, 6, 1445–1451. [Google Scholar] [CrossRef]
- Tremolada, C.; Colombo, V.; Ventura, C. Adipose tissue and mesenchymal stem cells: State of the art and lipogems® technology development. Curr. Stem Cell Rep. 2016, 2, 304–312. [Google Scholar] [CrossRef]
- Bianchi, F.; Maioli, M.; Leonardi, E.; Olivi, E.; Pasquinelli, G.; Valente, S.; Mendez, A.J.; Ricordi, C.; Raffaini, M.; Tremolada, C.; et al. A new nonenzymatic method and device to obtain a fat tissue derivative highly enriched in pericyte-like elements by mild mechanical forces from human lipoaspirates. Cell Transplant. 2013, 22, 2063–2077. [Google Scholar] [CrossRef]
- Boshuizen, H.C.; Poos, M.J.J.C.; van den Akker, M.; van Boven, K.; Korevaar, J.C.; de Waal, M.W.M.; Biermans, M.C.J.; Hoeymans, N. Estimating incidence and prevalence rates of chronic diseases using disease modeling. Popul. Health Metr. 2017, 15, 13. [Google Scholar] [CrossRef]
- Hudetz, D.; Boric, I.; Rod, E.; Jelec, Z.; Radic, A.; Vrdoljak, T.; Skelin, A.; Lauc, G.; Trbojevic-Akmacic, I.; Plecko, M.; et al. The effect of intra-articular injection of autologous microfragmented fat tissue on proteoglycan synthesis in patients with knee osteoarthritis. Genes 2017, 8, 270. [Google Scholar] [CrossRef]
- Vezzani, B.; Shaw, I.; Lesme, H.; Yong, L.; Khan, N.; Tremolada, C.; Peault, B. Higher pericyte content and secretory activity of microfragmented human adipose tissue compared to enzymatically derived stromal vascular fraction. Stem Cells Transl. Med. 2018, 7, 876–886. [Google Scholar] [CrossRef]
- Zimmerlin, L.; Donnenberg, V.S.; Rubin, J.P.; Donnenberg, A.D. Mesenchymal markers on human adipose stem/progenitor cells. Cytom. Part A J. Int. Soc. Anal. Cytol. 2013, 83, 134–140. [Google Scholar] [CrossRef]
- Zimmerlin, L.; Donnenberg, V.S.; Pfeifer, M.E.; Meyer, E.M.; Peault, B.; Rubin, J.P.; Donnenberg, A.D. Stromal vascular progenitors in adult human adipose tissue. Cytom. Part A J. Int. Soc. Anal. Cytol. 2010, 77, 22–30. [Google Scholar] [CrossRef]
- Collas Lab. Protocol: Isolation of Stromal Stem Cells from Human Adipose Tissue. Available online: https://www.med.uio.no/imb/english/research/groups/chromatin-regulation-stem-cells/documents/adipose-stem-cell-isolation.pdf (accessed on 19 April 2019).
- Carvalho, P.P.; Wu, X.; Yu, G.; Dias, I.R.; Gomes, M.E.; Reis, R.L.; Gimble, J.M. The effect of storage time on adipose-derived stem cell recovery from human lipoaspirates. Cells Tissues Organs 2011, 194, 494–500. [Google Scholar] [CrossRef]
- Hudetz, D.; Boric, I.; Rod, E.; Jelec, Z.; Kunovac, B.; Polasek, O.; Vrdoljak, T.; Plecko, M.; Skelin, A.; Polancec, D.; et al. Early results of intra-articular micro-fragmented lipoaspirate treatment in patients with late stages knee osteoarthritis: A prospective study. Croat. Med. J. 2019, 60, 227–236. [Google Scholar] [CrossRef]
- Zimmerlin, L.; Park, T.S.; Donnenberg, V.S.; Zambidis, E.T.; Donnenberg, A.D. Pericytes: A Ubiquitous Source of Multipotent Adult Tissue Stem Cells. In Stem Cells in Aesthetic Procedures: Art, Science, and Clinical Techniques; Shiffman, M.A., Di Giuseppe, A., Bassetto, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 135–148. [Google Scholar]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef]
- da Silva Meirelles, L.; Malta, T.M.; Panepucci, R.A.; da Silva, W.A., Jr. Transcriptomic comparisons between cultured human adipose tissue-derived pericytes and mesenchymal stromal cells. Genome Data 2016, 7, 20–25. [Google Scholar] [CrossRef]
- Peplow, P.V. Growth factor- and cytokine-stimulated endothelial progenitor cells in post-ischemic cerebral neovascularization. Neural Regen Res. 2014, 9, 1425–1429. [Google Scholar] [CrossRef]
- Yang, Z.J.; von Ballmoos, M.W.; Faessler, D.; Voelzmann, J.; Ortmann, J.; Diehm, N.; Kalka-Moll, W.; Baumgartner, I.; Di Santo, S.; Kalka, C. Paracrine factors secreted by endothelial progenitor cells prevent oxidative stress-induced apoptosis of mature endothelial cells. Atherosclerosis 2010, 211, 103–109. [Google Scholar] [CrossRef]
- Seebach, C.; Henrich, D.; Wilhelm, K.; Barker, J.H.; Marzi, I. Endothelial progenitor cells improve directly and indirectly early vascularization of mesenchymal stem cell-driven bone regeneration in a critical bone defect in rats. Cell Transplant. 2012, 21, 1667–1677. [Google Scholar] [CrossRef]
- Liang, T.Z.; Zhu, L.; Gao, W.L.; Gong, M.; Ren, J.H.; Yao, H.; Wang, K.; Shi, D.H. Coculture of endothelial progenitor cells and mesenchymal stem cells enhanced their proliferation and angiogenesis through PDGF and Notch signaling. FEBS Open Bio 2017, 7, 1722–1736. [Google Scholar] [CrossRef]
- Kolbe, M.; Xiang, Z.; Dohle, E.; Tonak, M.; Kirkpatrick, C.J.; Fuchs, S. Paracrine effects influenced by cell culture medium and consequences on microvessel-like structures in cocultures of mesenchymal stem cells and outgrowth endothelial cells. Tissue Eng. Part A 2011, 17, 2199–2212. [Google Scholar] [CrossRef]
- Duttenhoefer, F.; de Freitas, R.L.; Meury, T.; Loibl, M.; Benneker, L.M.; Richards, R.G.; Alini, M.; Verrier, S. 3d scaffolds co-seeded with human endothelial progenitor and mesenchymal stem cells: Evidence of prevascularisation within 7 days. Eur. Cells Mater. 2013, 26, 49–65. [Google Scholar] [CrossRef]
- Loibl, M.; Binder, A.; Herrmann, M.; Duttenhoefer, F.; Richards, R.G.; Nerlich, M.; Alini, M.; Verrier, S. Direct cell-cell contact between mesenchymal stem cells and endothelial progenitor cells induces a pericyte-like phenotype in vitro. Biomed. Res. Int. 2014, 395781. [Google Scholar] [CrossRef]
- Ge, Q.; Zhang, H.; Hou, J.; Wan, L.; Cheng, W.; Wang, X.; Dong, D.; Chen, C.; Xia, J.; Guo, J.; et al. VEGF secreted by mesenchymal stem cells mediates the differentiation of endothelial progenitor cells into endothelial cells via paracrine mechanisms. Mol. Med. Rep. 2018, 17, 1667–1675. [Google Scholar] [CrossRef]
- Aguirre, A.; Planell, J.A.; Engel, E. Dynamics of bone marrow-derived endothelial progenitor cell/mesenchymal stem cell interaction in co-culture and its implications in angiogenesis. Biochem. Biophys. Res. Commun. 2010, 400, 284–291. [Google Scholar] [CrossRef]
- Melchiorri, A.J.; Nguyen, B.N.; Fisher, J.P. Mesenchymal stem cells: Roles and relationships in vascularization. Tissue Eng. Part B Rev. 2014, 20, 218–228. [Google Scholar] [CrossRef]
- Heo, H.R.; Chen, L.; An, B.; Kim, K.S.; Ji, J.; Hong, S.H. Hormonal regulation of hematopoietic stem cells and their niche: A focus on estrogen. Int. J. Stem Cells 2015, 8, 18–23. [Google Scholar] [CrossRef]
- Ray, R.; Novotny, N.M.; Crisostomo, P.R.; Lahm, T.; Abarbanell, A.; Meldrum, D.R. Sex steroids and stem cell function. Mol. Med. 2008, 14, 493–501. [Google Scholar] [CrossRef]
- Li, J.S.; Peng, X.C.; Zeng, X.Q.; Liu, B.X.; Hao, Q.; Yu, X.Y.; Zhu, L.P.; Hu, Q.H. Estrogen secreted by mesenchymal stem cells necessarily determines their feasibility of therapeutical application. Sci. Rep. UK 2015, 5, 15286. [Google Scholar] [CrossRef]
- Ceserani, V.; Ferri, A.; Berenzi, A.; Benetti, A.; Ciusani, E.; Pascucci, L.; Bazzucchi, C.; Cocce, V.; Bonomi, A.; Pessina, A.; et al. Angiogenic and anti-inflammatory properties of micro-fragmented fat tissue and its derived mesenchymal stromal cells. Vasc. Cell 2016, 8, 3. [Google Scholar] [CrossRef]
- Harrell, C.R.; Simovic Markovic, B.; Fellabaum, C.; Arsenijevic, A.; Djonov, V.; Volarevic, V. Molecular mechanisms underlying therapeutic potential of pericytes. J. Biomed. Sci. 2018, 25, 21. [Google Scholar] [CrossRef]
- Holmes, D. Closing the gap. Nature 2017, 550, S194–S195. [Google Scholar] [CrossRef]
- Hudetz, D. The Future of Cartilage Repair. In Comprehensive Approach to Personalised Medicine; Pavelic, K., Ed.; Springer: Belin, Germany, in press.
- Polancec, D.; Zenic, L.; Hudetz, D.; Boric, I.; Jelec, Z.; Rod, E.; Vrdoljak, T.; Skelin, A.; Plecko, M.; Turkalj, M.; et al. Imunofenotipizacija stromalne vaskularne frakcije iz mikrofragmentiranog adipoznog tkiva i lipoaspirata pacijenata s osteoartirtisom. Paediatr. Croat. 2018, 62, 171. [Google Scholar]



| Immunophenotype | Lineage Markers | Mesenchymal Stem/Stromal Cell (MSC) Markers | Frequency within Nucleated SVF Cells | |
|---|---|---|---|---|
| LA | MLA | |||
| Endothelial progenitors | CD45−CD31+CD34+CD146± | CD73±CD90±CD105± | 13–51% | 45–89% |
| Pericytes | CD45−CD31−CD34−CD146+ | CD73±CD90+CD105− | 10–21% | 7–21% |
| SA-ASC | CD45−CD31−CD34+CD146− | CD73highCD90±CD105− | 5–27% | 1–9% |
| Leukocytes | CD45+CD31−CD34-CD146− | CD73−CD90−CD105− | 11–58% | 3–25% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polancec, D.; Zenic, L.; Hudetz, D.; Boric, I.; Jelec, Z.; Rod, E.; Vrdoljak, T.; Skelin, A.; Plecko, M.; Turkalj, M.; et al. Immunophenotyping of a Stromal Vascular Fraction from Microfragmented Lipoaspirate Used in Osteoarthritis Cartilage Treatment and Its Lipoaspirate Counterpart. Genes 2019, 10, 474. https://doi.org/10.3390/genes10060474
Polancec D, Zenic L, Hudetz D, Boric I, Jelec Z, Rod E, Vrdoljak T, Skelin A, Plecko M, Turkalj M, et al. Immunophenotyping of a Stromal Vascular Fraction from Microfragmented Lipoaspirate Used in Osteoarthritis Cartilage Treatment and Its Lipoaspirate Counterpart. Genes. 2019; 10(6):474. https://doi.org/10.3390/genes10060474
Chicago/Turabian StylePolancec, Denis, Lucija Zenic, Damir Hudetz, Igor Boric, Zeljko Jelec, Eduard Rod, Trpimir Vrdoljak, Andrea Skelin, Mihovil Plecko, Mirjana Turkalj, and et al. 2019. "Immunophenotyping of a Stromal Vascular Fraction from Microfragmented Lipoaspirate Used in Osteoarthritis Cartilage Treatment and Its Lipoaspirate Counterpart" Genes 10, no. 6: 474. https://doi.org/10.3390/genes10060474
APA StylePolancec, D., Zenic, L., Hudetz, D., Boric, I., Jelec, Z., Rod, E., Vrdoljak, T., Skelin, A., Plecko, M., Turkalj, M., Nogalo, B., & Primorac, D. (2019). Immunophenotyping of a Stromal Vascular Fraction from Microfragmented Lipoaspirate Used in Osteoarthritis Cartilage Treatment and Its Lipoaspirate Counterpart. Genes, 10(6), 474. https://doi.org/10.3390/genes10060474






