Landscape of Germline Mutations in DNA Repair Genes for Breast Cancer in Latin America: Opportunities for PARP-Like Inhibitors and Immunotherapy
Abstract
1. Introduction
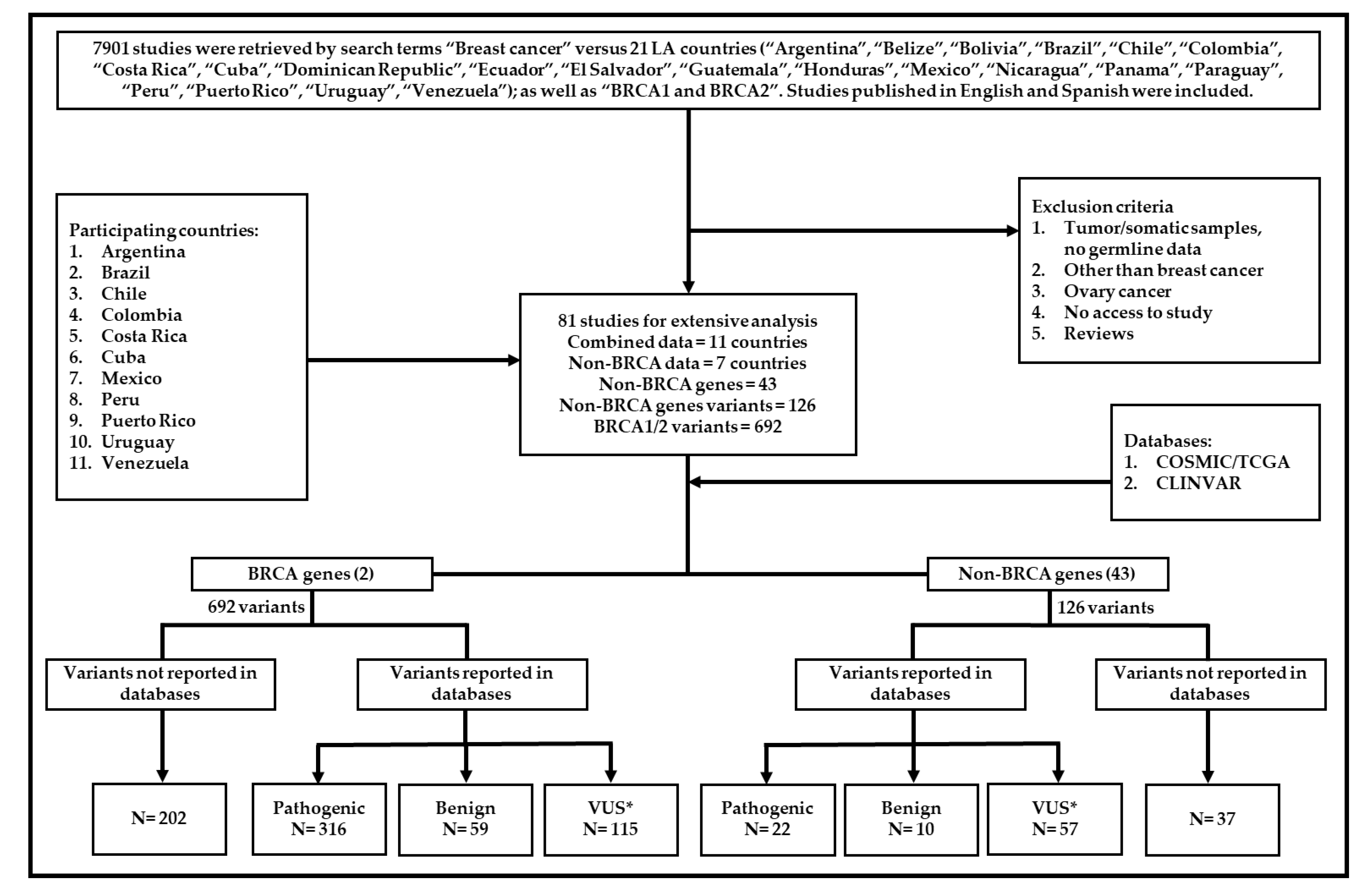
2. Materials and Methods
3. Results
3.1. The Scope of Germline Mutations in Breast Cancer in LA Countries
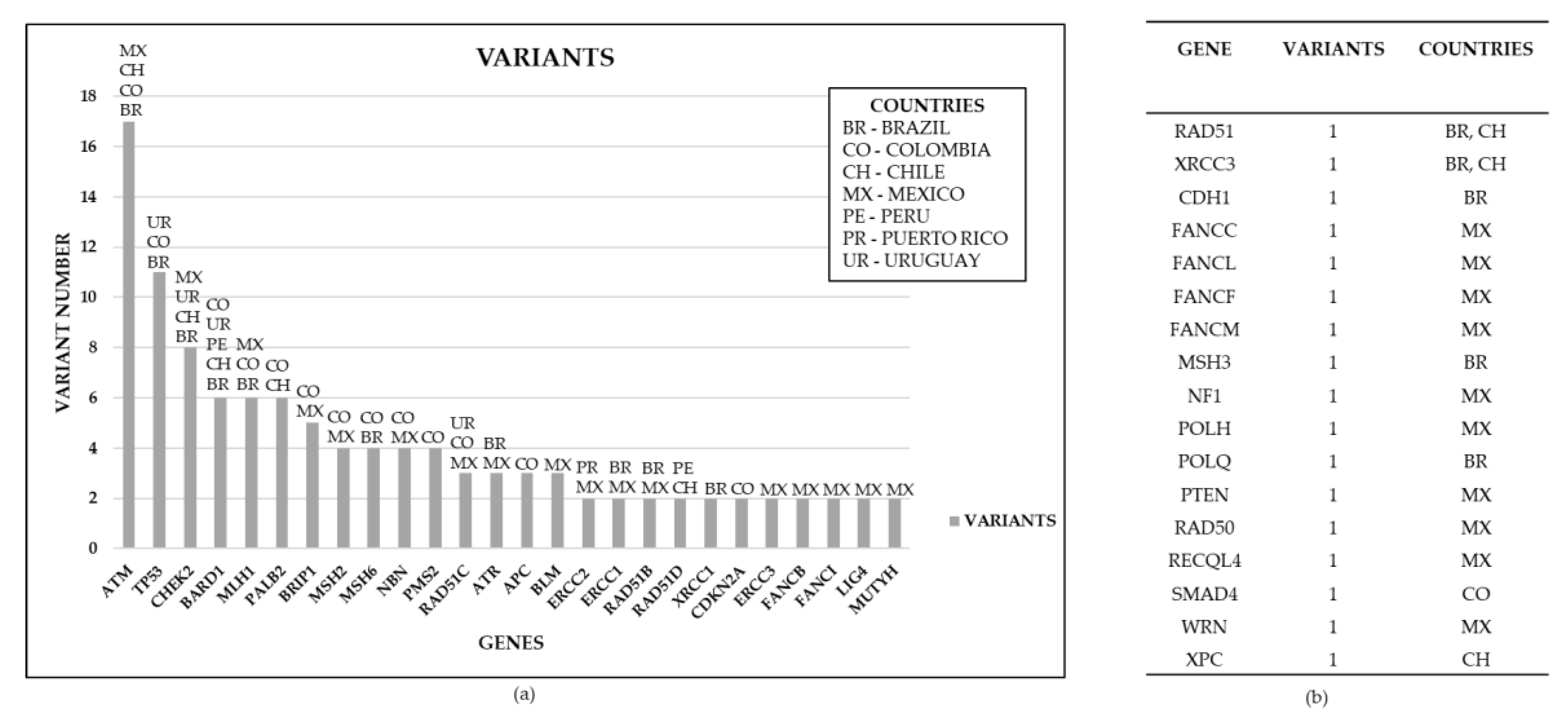
3.2. Genes reported for HBC in LA countries
3.3. BRCA1 and BRCA2 Genes in LA countries
3.4. Non-BRCA Genes Reported in Breast Cancer in LA Countries
4. Biallelic Cases, Double Heterozygosity and Young Women in LA Countries
5. Therapy Recommendation for DNA Repair Related Genes
6. Conclusions
7. Take Home Messages
- BRCA1/2 are the most analyzed and studied genes in LA countries, few studies report non-BRCA gene status in breast cancer.
- In addition to BRCA1/2, non-BRCA genes provide information about the DNA repair capacity status.
- Targeted therapy such as immunotherapy and mainly PARP inhibitors are focused on DNA repair gene status for better response.
- Studies focusing on non-BRCA genes are needed in LA countries.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Global cancer statistics 2018. CA Cancer J. Clin. 2018. [Google Scholar] [CrossRef]
- El Tannouri, R.; Albuisson, E.; Jonveaux, P.; Luporsi, E. Is there a genetic anticipation in breast and/or ovarian cancer families with the germline c.3481_3491del11 mutation? Fam. Cancer 2018, 17, 5–14. [Google Scholar] [CrossRef]
- Cragun, D.; Weidner, A.; Kechik, J.; Pal, T. Genetic testing across young hispanic and non-hispanic white breast cancer survivors: Facilitators, barriers, and awareness of the genetic information nondiscrimination act. Genet. Test. Mol. Biomark. 2019, 23, 75–83. [Google Scholar] [CrossRef]
- Kwong, A.; Chen, J.; Shin, V.Y. A new paradigm of genetic testing for hereditary breast/ovarian cancers. Hong Kong Med. J. 2016. [Google Scholar] [CrossRef]
- Miki, Y.; Swensen, J.; Shattuck-Eidens, D.; Futreal, P.; Harshman, K.; Tavtigian, S.; Liu, Q.; Cochran, C.; Bennett, L.; Ding, W.; et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 1994, 266, 66–71. [Google Scholar] [CrossRef]
- Wooster, R.; Bignell, G.; Lancaster, J.; Swift, S.; Seal, S.; Mangion, J.; Collins, N.; Gregory, S.; Gumbs, C.; Micklem, G. Identification of the breast cancer susceptibility gene BRCA2. Nature 1995, 378, 789–792. [Google Scholar] [CrossRef]
- Friedman, L.S.; Ostermeyer, E.A.; Szabo, C.I.; Dowd, P.; Lynch, E.D.; Rowell, S.E.; King, M.C. Confirmation of BRCA1 by analysis of germline mutations linked to breast and ovarian cancer in ten families. Nat. Genet. 1994, 8, 399–404. [Google Scholar] [CrossRef]
- Grynberg, M.; Dagher Hayeck, B.; Papanikolaou, E.G.; Sifer, C.; Sermondade, N.; Sonigo, C. BRCA1/2 gene mutations do not affect the capacity of oocytes from breast cancer candidates for fertility preservation to mature in vitro. Hum. Reprod. Oxf. Engl. 2019, 34, 374–379. [Google Scholar] [CrossRef]
- Walsh, C.S. Two decades beyond BRCA1/2: Homologous recombination, hereditary cancer risk and a target for ovarian cancer therapy. Gynecol. Oncol. 2015, 137, 343–350. [Google Scholar] [CrossRef]
- Foulkes, W.D. Inherited susceptibility to common cancers. N. Engl. J. Med. 2008, 359, 2143–2153. [Google Scholar] [CrossRef]
- Gómez-Flores-Ramos, L.; Castro-Sanchez, A.; Peña-Curiel, O.; Mohar-Betancourt, A. Molecular biology in young women with breast cancer: From tumor gene expression to DNA mutations. Rev. Investig. Clínica 2017, 69. [Google Scholar]
- Economopoulou, P.; Dimitriadis, G.; Psyrri, A. Beyond BRCA: New hereditary breast cancer susceptibility genes. Cancer Treat. Rev. 2015, 41, 1–8. [Google Scholar] [CrossRef]
- Daza-Martin, M.; Starowicz, K.; Jamshad, M.; Tye, S.; Ronson, G.E.; MacKay, H.L.; Chauhan, A.S.; Walker, A.K.; Stone, H.R.; Beesley, J.F.J.; et al. Isomerization of BRCA1–BARD1 promotes replication fork protection. Nature 2019, 1. [Google Scholar] [CrossRef]
- Postel-Vinay, S.; Vanhecke, E.; Olaussen, K.A.; Lord, C.J.; Ashworth, A.; Soria, J.-C. The potential of exploiting DNA-repair defects for optimizing lung cancer treatment. Nat. Rev. Clin. Oncol. 2012, 9, 144–155. [Google Scholar] [CrossRef]
- Dietlein, F.; Thelen, L.; Reinhardt, H.C. Cancer-specific defects in DNA repair pathways as targets for personalized therapeutic approaches. Trends Genet. 2014, 30, 326–339. [Google Scholar] [CrossRef]
- Daly, M.B.; Pilarski, R.; Berry, M.; Buys, S.S.; Farmer, M.; Friedman, S.; Garber, J.E.; Kauff, N.D.; Khan, S.; Klein, C.; et al. NCCN guidelines insights: Genetic/familial high-risk assessment: Breast and ovarian, version 2.2017. J. Natl. Compr. Cancer Netw. 2017, 15, 9–20. [Google Scholar] [CrossRef]
- Liu, Q.; Yao, S.; Zhao, H.; Hu, Q.; Kwan, M.L.; Roh, J.M.; Ambrosone, C.B.; Kushi, L.H.; Liu, S.; Zhu, Q. Early-onset triple-negative breast cancer in multiracial/ethnic populations: Distinct trends of prevalence of truncation mutations. Cancer Med. 2019. [Google Scholar] [CrossRef]
- Chavarri-Guerra, Y.; Blazer, K.R.; Weitzel, J.N. Genetic cancer risk assessment for breast cancer in Latin America. Rev. Investig. Clín. 2017, 69. [Google Scholar] [CrossRef]
- Moreno-Estrada, A.; Gravel, S.; Zakharia, F.; McCauley, J.L.; Byrnes, J.K.; Gignoux, C.R.; Ortiz-Tello, P.A.; Martínez, R.J.; Hedges, D.J.; Morris, R.W.; et al. Reconstructing the population genetic history of the Caribbean. PLoS Genet. 2013, 9, e1003925. [Google Scholar] [CrossRef]
- Zavala, V.A.; Serrano-Gomez, S.J.; Dutil, J.; Fejerman, L. Genetic epidemiology of breast cancer in Latin America. Genes 2019, 10, 153. [Google Scholar] [CrossRef]
- Homburger, J.R.; Moreno-Estrada, A.; Gignoux, C.R.; Nelson, D.; Sanchez, E.; Ortiz-Tello, P.; Pons-Estel, B.A.; Acevedo-Vasquez, E.; Miranda, P.; Langefeld, C.D.; et al. Genomic insights into the ancestry and demographic history of South America. PLoS Genet. 2015, 11, e1005602. [Google Scholar] [CrossRef]
- Asakawa, H.; Koizumi, H.; Koike, A.; Takahashi, M.; Wu, W.; Iwase, H.; Fukuda, M.; Ohta, T. Prediction of breast cancer sensitivity to neoadjuvant chemotherapy based on status of DNA damage repair proteins. Breast Cancer Res. 2010, 12. [Google Scholar] [CrossRef]
- Meindl, A.; Ditsch, N.; Kast, K.; Rhiem, K.; Schmutzler, R.K. Hereditary breast and ovarian cancer. Dtsch. Aerzteblatt Online 2011, 275, 1885–1892. [Google Scholar] [CrossRef]
- Yin, L.; Liu, Y.; Peng, Y.; Peng, Y.; Yu, X.; Gao, Y.; Yuan, B.; Zhu, Q.; Cao, T.; He, L.; et al. PARP inhibitor veliparib and HDAC inhibitor SAHA synergistically co-target the UHRF1/BRCA1 DNA damage repair complex in prostate cancer cells. J. Exp. Clin. Cancer Res. 2018, 37. [Google Scholar] [CrossRef]
- Annunziata, C.M.; Bates, S.E. PARP inhibitors in BRCA1/BRCA2 germline mutation carriers with ovarian and breast cancer. F1000 Biol. Rep. 2010, 2. [Google Scholar] [CrossRef]
- Sullivan-Reed, K.; Bolton-Gillespie, E.; Dasgupta, Y.; Langer, S.; Siciliano, M.; Nieborowska-Skorska, M.; Hanamshet, K.; Belyaeva, E.A.; Bernhardy, A.J.; Lee, J.; et al. Simultaneous targeting of PARP1 and RAD52 triggers dual synthetic lethality in BRCA-deficient tumor cells. Cell Rep. 2018, 23, 3127–3136. [Google Scholar] [CrossRef]
- Brown, J.S.; O’Carrigan, B.; Jackson, S.P.; Yap, T.A. Targeting DNA repair in cancer: Beyond PARP inhibitors. Cancer Discov. 2017, 7, 20–37. [Google Scholar] [CrossRef]
- Chae, Y.K.; Anker, J.F.; Bais, P.; Namburi, S.; Giles, F.J.; Chuang, J.H. Mutations in DNA repair genes are associated with increased neo-antigen load and activated T cell infiltration in lung adenocarcinoma. Oncotarget 2018, 9. [Google Scholar] [CrossRef]
- Strickland, K.C.; Howitt, B.E.; Shukla, S.A.; Rodig, S.; Ritterhouse, L.L.; Liu, J.F.; Garber, J.E.; Chowdhury, D.; Wu, C.J.; D’Andrea, A.D.; et al. Association and prognostic significance of BRCA1/2-mutation status with neoantigen load, number of tumor-infiltrating lymphocytes and expression of PD-1/PD-L1 in high grade serous ovarian cancer. Oncotarget 2016, 7. [Google Scholar] [CrossRef]
- Gong, J.; Chehrazi-Raffle, A.; Reddi, S.; Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J. Immunother. Cancer 2018, 6. [Google Scholar] [CrossRef]
- Overman, M.J.; Lonardi, S.; Wong, K.Y.M.; Lenz, H.-J.; Gelsomino, F.; Aglietta, M.; Morse, M.A.; Van Cutsem, E.; McDermott, R.; Hill, A.; et al. Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair–deficient/microsatellite instability–high metastatic colorectal cancer. J. Clin. Oncol. 2018, 36, 773–779. [Google Scholar] [CrossRef]
- Yi, D.; Xu, L.; Luo, J.; You, X.; Huang, T.; Zi, Y.; Li, X.; Wang, R.; Zhong, Z.; Tang, X.; et al. Germline TP53 and MSH6 mutations implicated in sporadic triple-negative breast cancer (TNBC): A preliminary study. Hum. Genom. 2019, 13. [Google Scholar] [CrossRef]
- Hodges, T.R.; Ott, M.; Xiu, J.; Gatalica, Z.; Swensen, J.; Zhou, S.; Huse, J.T.; de Groot, J.; Li, S.; Overwijk, W.W.; et al. Mutational burden, immune checkpoint expression, and mismatch repair in glioma: Implications for immune checkpoint immunotherapy. Neuro Oncol. 2017, 19, 1047–1057. [Google Scholar] [CrossRef]
- Brandalize, A.P.C.; Schüler-Faccini, L.; Hoffmann, J.-S.; Caleffi, M.; Cazaux, C.; Ashton-Prolla, P. A DNA repair variant in POLQ (c.-1060A > G) is associated to hereditary breast cancer patients: A case–control study. BMC Cancer 2014, 14. [Google Scholar] [CrossRef]
- de Souza Timoteo, A.R.; Gonçalves, A.É.M.M.; Sales, L.A.P.; Albuquerque, B.M.; de Souza, J.E.S.; de Moura, P.C.P.; de Aquino, M.A.A.; Agnez-Lima, L.F.; Lajus, T.B.P. A portrait of germline mutation in Brazilian at-risk for hereditary breast cancer. Breast Cancer Res. Treat. 2018, 172, 637–646. [Google Scholar] [CrossRef]
- Assumpção, J.G.; Seidinger, A.L.; Mastellaro, M.J.; Ribeiro, R.C.; Zambetti, G.P.; Ganti, R.; Srivastava, K.; Shurtleff, S.; Pei, D.; Zeferino, L.C.; et al. Association of the germline TP53R337H mutation with breast cancer in southern Brazil. BMC Cancer 2008, 8. [Google Scholar] [CrossRef]
- Lourenço, J.J.; Vargas, F.R.; Bines, J.; Santos, E.M.; Lasmar, C.A.P.; Costa, C.H.; Teixeira, E.M.B.; Maia, M.C.M.; Coura, F.; Silva, C.H.D.; et al. BRCA1 mutations in Brazilian patients. Genet. Mol. Biol. 2004, 27, 500–504. [Google Scholar] [CrossRef][Green Version]
- Leistner-Segal. Analysis of the R72P polymorphism of the TP53 gene in patients with invasive ductal breast carcinoma. Mol. Med. Rep. 2009, 2. [Google Scholar]
- Abud, J.; Prolla, J.C.; Koehler-Santos, P.; Ashton-Prolla, P. CHEK2 1100DELC germline mutation: A frequency study in hereditary breast and colon cancer Brazilian families. Arq. Gastroenterol. 2012, 49, 273–278. [Google Scholar] [CrossRef]
- Torrezan, G.T.; de Almeida, F.G.D.S.R.; de Figueiredo Barros, B.D.; de Paula, C.A.; Valieris, R.; de Souza, J.E.S.; Ramalho, R.F.; da Silva, F.C.C.; Ferreira, E.N.; et al. Complex landscape of germline variants in Brazilian patients with hereditary and early onset breast cancer. Front. Genet. 2018, 9. [Google Scholar] [CrossRef]
- Ewald, I.P.; Cossio, S.L.; Palmero, E.I.; Pinheiro, M.; de Oliveira Nascimento, I.L.; Machado, T.M.B.; Sandes, K.A.; Toralles, B.; Garicochea, B.; Izetti, P.; et al. BRCA1 and BRCA2 rearrangements in Brazilian individuals with hereditary breast and ovarian cancer syndrome. Genet. Mol. Biol. 2016, 39, 223–231. [Google Scholar] [CrossRef]
- Carraro, D.M.; Koike Folgueira, M.A.A.; Garcia Lisboa, B.C.; Ribeiro Olivieri, E.H.; Vitorino Krepischi, A.C.; de Carvalho, A.F.; de Carvalho Mota, L.D.; Puga, R.D.; do Socorro Maciel, M.; Michelli, R.A.D.; et al. Comprehensive analysis of BRCA1, BRCA2 and TP53 germline mutation and tumor characterization: a portrait of early-onset breast cancer in Brazil. PLoS ONE 2013, 8, e57581. [Google Scholar] [CrossRef]
- Dufloth, R.M.; Costa, S.; Schmitt, F.; Zeferino, L.C. DNA repair gene polymorphisms and susceptibility to familial breast cancer in a group of patients from Campinas, Brazil. Genet. Mol. Res. 2005, 13. [Google Scholar]
- Cipriano, N.M.; de Brito, A.M.; de Oliveira, E.S.; de Faria, F.C.; Lemos, S.; Rodrigues, A.N.; de Oliveira Lopes, D.; dos Santos, L.L. Mutation screening of TP53, CHEK2 and BRCA genes in patients at high risk for hereditary breast and ovarian cancer (HBOC) in Brazil. Breast Cancer 2018. [Google Scholar] [CrossRef]
- Brianese, R.C.; de Mello Nakamura, K.D.; Ramalho, R.F.; de Figueiredo Barros, B.D.; e Ferreira, E.N.; da Cruz Formiga, M.N.; de Andrade, V.P.; de Lima, V.C.C.; Carraro, D.M. BRCA1 deficiency is a recurrent event in early-onset triple-negative breast cancer: A comprehensive analysis of germline mutations and somatic promoter methylation. Breast Cancer Res. Treat. 2018, 167, 803–814. [Google Scholar] [CrossRef]
- Felix, G.E.; Abe-Sandes, C.; Machado-Lopes, T.M.; Bomfim, T.F.; Guindalini, R.S.C.; Santos, V.C.S.; Meyer, L.; Oliveira, P.C.; Cláudio Neiva, J.; Meyer, R.; et al. Germline mutations in BRCA1, BRCA2, CHEK2 and TP53 in patients at high-risk for HBOC: Characterizing a northeast Brazilian population. Hum. Genome Var. 2014, 1. [Google Scholar] [CrossRef]
- Gomes, M.C.; Kotsopoulos, J.; de Almeida, G.L.; Costa, M.M.; Vieira, R.; de AG Filho, F.; Pitombo, M.B.; Leal, P.R.; Royer, R.; Zhang, P.; et al. The R337H mutation in TP53 and breast cancer in Brazil. Hered. Cancer Clin. Practi. 2012, 10. [Google Scholar] [CrossRef]
- Cury, N.M.; Ferraz, V.E.; Silva, W.A. TP53 p.R337H prevalence in a series of Brazilian hereditary breast cancer families. Hered. Cancer Clin. Practi. 2014, 12, 8. [Google Scholar] [CrossRef]
- Hahn, E.C.; Bittar, C.M.; Vianna, F.S.L.; Netto, C.B.O.; Biazús, J.V.; Cericatto, R.; Cavalheiro, J.A.; de Melo, M.P.; Menke, C.H.; Rabin, E.; et al. TP53 p.Arg337His germline mutation prevalence in Southern Brazil: Further evidence for mutation testing in young breast cancer patients. PLoS ONE 2018, 13, e0209934. [Google Scholar] [CrossRef]
- Dillenburg, C.V.; Bandeira, I.C.; Tubino, T.V.; Rossato, L.G.; Dias, E.S.; Bittelbrunn, A.C.; Leistner-Segal, S. Prevalence of 185delAG and 5382insC mutations in BRCA1, and 6174delT in BRCA2 in women of Ashkenazi Jewish origin in southern Brazil. Genet. Mol. Biol. 2012, 35, 599–602. [Google Scholar] [CrossRef]
- Esteves, V.F.; Thuler, L.C.S.; Amêndola, L.C.; Koifman, R.J.; Koifman, S.; Frankel, P.P.; Vieira, R.J.S. Prevalence of BRCA1 and BRCA2 gene mutations in families with medium and high risk of breast and ovarian cancer in Brazil. Braz. J. Med. Biol. Res. 2009, 42, 453–457. [Google Scholar] [CrossRef]
- Gomes, M.C.B.; Costa, M.M.; Borojevic, R.; Monteiro, A.N.A.; Vieira, R.; Koifman, S.; Koifman, R.J.; Li, S.; Royer, R.; Zhang, S.; et al. Prevalence of BRCA1 and BRCA2 mutations in breast cancer patients from Brazil. Breast Cancer Res. Treat. 2007, 103, 349–353. [Google Scholar] [CrossRef]
- Fernandes, G.C.; Michelli, R.A.D.; Galvão, H.C.R.; Paula, A.E.; Pereira, R.; Andrade, C.E.; Felicio, P.S.; Souza, C.P.; Mendes, D.R.P.; Volc, S.; et al. Prevalence of BRCA1/BRCA2 mutations in a Brazilian population sample at-risk for hereditary breast cancer and characterization of its genetic ancestry. Oncotarget 2016, 7. [Google Scholar] [CrossRef]
- Ewald, I.P.; Izetti, P.; Vargas, F.R.; Moreira, M.A.; Moreira, A.S.; Moreira-Filho, C.A.; Cunha, D.R.; Hamaguchi, S.; Camey, S.A.; Schmidt, A.; et al. Prevalence of the BRCA1 founder mutation c.5266dupin Brazilian individuals at-risk for the hereditary breast and ovarian cancer syndrome. Hered. Cancer Clin. Practi. 2011, 9. [Google Scholar] [CrossRef]
- Giacomazzi, J.; Graudenz, M.S.; Osorio, C.A.B.T.; Koehler-Santos, P.; Palmero, E.I.; Zagonel-Oliveira, M.; Michelli, R.A.D.; Neto, C.S.; Fernandes, G.C.; Achatz, M.I.W.S.; et al. Prevalence of the TP53 p.R337H mutation in breast cancer patients in Brazil. PLoS ONE 2014, 9, e99893. [Google Scholar] [CrossRef]
- Palmero, E.I.; Alemar, B.; Schüler-Faccini, L.; Hainaut, P.; Moreira-Filho, C.A.; Ewald, I.P.; dos Santos, P.K.; Ribeiro, P.L.I.; de Oliveira Netto, C.B.; Calvez-Kelm, F.L.; et al. Screening for germline BRCA1, BRCA2, TP53 and CHEK2 mutations in families at-risk for hereditary breast cancer identified in a population-based study from Southern Brazil. Genet. Mol. Biol. 2016, 39, 210–222. [Google Scholar] [CrossRef]
- Andrade, K.C.; Santiago, K.M.; Fortes, F.P.; Mambelli, L.I.; Nóbrega, A.F.; Achatz, M.I. Early-onset breast cancer patients in the South and Southeast of Brazil should be tested for the TP53 p.R337H mutation. Genet. Mol. Biol. 2016, 39, 199–202. [Google Scholar] [CrossRef]
- da Costa, E.C.B.; Vargas, F.R.; Moreira, A.S.; Lourenço, J.J.; Caleffi, M.; Ashton-Prolla, P.; Martins Moreira, M.A.M. Founder effect of the BRCA1 5382insC mutation in Brazilian patients with hereditary breast ovary cancer syndrome. Cancer Genet. Cytogenet. 2008, 184, 62–66. [Google Scholar] [CrossRef]
- de Oliveira, E.S.; Soares, B.L.; Lemos, S.; Rosa, R.C.A.; Rodrigues, A.N.; Barbosa, L.A.; de Oliveira Lopes, D.; dos Santos, L.L. Screening of the BRCA1 gene in Brazilian patients with breast and/or ovarian cancer via high-resolution melting reaction analysis. Fam. Cancer 2016, 15, 173–181. [Google Scholar] [CrossRef]
- Palmero, E.I.; Carraro, D.M.; Alemar, B.; Moreira, M.A.M.; Ribeiro-dos-Santos, Â.; Abe-Sandes, K.; Galvão, H.C.R.; Reis, R.M.; de Pádua Souza, C.; Campacci, N.; et al. The germline mutational landscape of BRCA1 and BRCA2 in Brazil. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Almeida, B.C.; Kleine, J.P.F.O.; Camargo-Kosugi, C.M.; Lisboa, M.R.; França, C.N.; França, J.P.; Silva, I.D.C.G. Analysis of polymorphisms in codons 11, 72 and 248 of TP53 in Brazilian women with breast cancer. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Rodrigues, M.S.; Machado, C.A.; Pagnoncelli, D.; Avvad, E.; da Paixão, J.C.; Gallo, C.V.D.M. TP53 and XRCC1 polymorphisms and breast cancer prognosis: A case-case study. Clinics 2011, 66, 1097–1100. [Google Scholar] [CrossRef]
- Alemar, B.; Gregório, C.; Herzog, J.; Matzenbacher Bittar, C.; Brinckmann Oliveira Netto, C.; Artigalas, O.; Schwartz, I.V.D.; Coffa, J.; Alves Camey, S.; Weitzel, J.; et al. BRCA1 and BRCA2 mutational profile and prevalence in hereditary breast and ovarian cancer (HBOC) probands from Southern Brazil: Are international testing criteria appropriate for this specific population? PLoS ONE 2017, 12, e0187630. [Google Scholar] [CrossRef]
- de Souza Timoteo, A.R.; Albuquerque, B.M.; Moura, P.P.P.; de Oliveira Ramos, C.C.; Agnez-Lima, L.F.; Walsh, T.; King, M.-C.; Lajus, T.B.P. Identification of a new BRCA2 large genomic deletion associated with high risk male breast cancer. Hered. Cancer Clin. Practi. 2015, 13. [Google Scholar]
- Silva, F.C.; Lisboa, B.C.; Figueiredo, M.C.; Torrezan, G.T.; Santos, É.M.; Krepischi, A.C.; Rossi, B.M.; Achatz, M.I.; Carraro, D.M. Hereditary breast and ovarian cancer: Assessment of point mutations and copy number variations in Brazilian patients. BMC Med. Genet. 2014, 15, 55. [Google Scholar] [CrossRef]
- Tapia, T.; Sanchez, A.; Vallejos, M.; Alvarez, C.; Moraga, M.; Smalley, S.; Camus, M.; Alvarez, M.; Carvallo, P. ATM allelic variants associated to hereditary breast cancer in 94 Chilean women: Susceptibility or ethnic influences? Breast Cancer Res. Treat. 2008, 107, 281–288. [Google Scholar] [CrossRef]
- Alvarez, C.; Tapia, T.; Perez-Moreno, E.; Gajardo-Meneses, P.; Ruiz, C.; Rios, M.; Missarelli, C.; Silva, M.; Cruz, A.; Matamala, L.; et al. BRCA1 and BRCA2 founder mutations account for 78% of germline carriers among hereditary breast cancer families in Chile. Oncotarget 2017, 8. [Google Scholar] [CrossRef]
- Jara, L.; Ampuero, S.; Santibáñez, E.; Seccia, L.; Rodríguez, J.; Bustamante, M.; Martínez, V.; Catenaccio, A.; Lay-Son, G.; Blanco, R.; et al. BRCA1 and BRCA2 mutations in a South American population. Cancer Genet. Cytogenet. 2006, 166, 36–45. [Google Scholar] [CrossRef]
- González-Hormazábal, P.; Bravo, T.; Blanco, R.; Valenzuela, C.Y.; Gómez, F.; Waugh, E.; Peralta, O.; Ortuzar, W.; Reyes, J.M.; Jara, L. Association of common ATM variants with familial breast cancer in a South American population. BMC Cancer 2008, 8, 117. [Google Scholar] [CrossRef]
- Gallardo, M.; Silva, A.; Rubio, L.; Alvarez, C.; Torrealba, C.; Salinas, M.; Tapia, T.; Faundez, P.; Palma, L.; Riccio, M.E.; et al. Incidence of BRCA1 and BRCA2 mutations in 54 Chilean families with breast/ovarian cancer, genotype–phenotype correlations. Breast Cancer Res. Treat. 2006, 95, 81–87. [Google Scholar] [CrossRef]
- Leyton, Y.; Gonzalez-Hormazabal, P.; Blanco, R.; Bravo, T.; Fernandez-Ramires, R.; Morales, S.; Landeros, N.; Reyes, J.M.; Peralta, O.; Tapia, J.C.; et al. Association of PALB2 sequence variants with the risk of familial and early-onset breast cancer in a South-American population. BMC Cancer 2015, 15. [Google Scholar] [CrossRef]
- Gonzalez-Hormazabal, P.; Gutierrez-Enriquez, S.; Gaete, D.; Reyes, J.M.; Peralta, O.; Waugh, E.; Gomez, F.; Margarit, S.; Bravo, T.; Blanco, R.; et al. Spectrum of BRCA1/2 point mutations and genomic rearrangements in high-risk breast/ovarian cancer Chilean families. Breast Cancer Res. Treat. 2011, 126, 705–716. [Google Scholar] [CrossRef]
- Jara, L.; Ampuero, S.; Santibáñez, E.; Seccia, L.; Rodríguez, J.; Bustamante, M.; Lay-Son, G.; Ojeda, J.M.; Reyes, J.M.; Blanco, R. Molecular analysis of the eighteen most frequent mutations in the BRCA1 gene in 63 Chilean breast cancer families. Biol. Res. 2004, 37. [Google Scholar] [CrossRef]
- Jara, L.; Acevedo, M.L.; Blanco, R.; Castro, V.G.; Bravo, T.; Gómez, F.; Waugh, E.; Peralta, O.; Cabrera, E.; Reyes, J.M.; et al. RAD51 135G>C polymorphism and risk of familial breast cancer in a South American population. Cancer Genet. Cytogenet. 2007, 178, 65–69. [Google Scholar] [CrossRef]
- Gonzalez-Hormazabal, P.; Reyes, J.M.; Blanco, R.; Bravo, T.; Carrera, I.; Peralta, O.; Gomez, F.; Waugh, E.; Margarit, S.; Ibañez, G.; et al. The BARD1 Cys557Ser variant and risk of familial breast cancer in a South-American population. Mol. Biol. Rep. 2012, 39, 8091–8098. [Google Scholar] [CrossRef]
- Jara, L.; Dubois, K.; Gaete, D.; de Mayo, T.; Ratkevicius, N.; Bravo, T.; Margarit, S.; Blanco, R.; Gómez, F.; Waugh, E.; et al. Variants in DNA double-strand break repair genes and risk of familial breast cancer in a South American population. Breast Cancer Res. Treat. 2010, 122, 813–822. [Google Scholar] [CrossRef]
- Jara, L.; Ampuero Ll, S.; Seccia, L.; Bustamante, M.; Blanco, R.; Santibáñez, E.; Reyes, J.M.; Ojeda, J.M. Frecuencia de la mutación 185delAG en el gen BRCA1 en mujeres chilenas sanas con antecedentes familiares de cáncer de mama. Rev. Médica Chile 2002, 130, 1113–1123. [Google Scholar] [CrossRef]
- Gallardo, C.M.; Faúndez, J.P.; Cruz, A.; Rodríguez, M.; Alvarez, Z.M.; Carvallo SQ, P. Determinación de una mutación en el gen BRCA1 en una familia que presenta cáncer de mama hereditario. Rev. Médica Chile 2004, 132. [Google Scholar] [CrossRef]
- González-Hormazábal, P.; Castro, V.G.; Blanco, R.; Gómez, F.; Peralta, O.; Waugh, E.; Bravo, T.; Reyes, J.M.; Jara, L. Absence of CHEK2 1100delC mutation in familial breast cancer cases from a South American population. Breast Cancer Res. Treat. 2008, 110, 543–545. [Google Scholar] [CrossRef]
- Fernández-Lopez, J.C.; Romero-Córdoba, S.; Rebollar-Vega, R.; Alfaro-Ruiz, L.A.; Jiménez-Morales, S.; Beltrán-Anaya, F.; Arellano-Llamas, R.; Cedro-Tanda, A.; Rios-Romero, M.; Ramirez-Florencio, M.; et al. Population and breast cancer patients’ analysis reveals the diversity of genomic variation of the BRCA genes in the Mexican population. Hum. Genom. 2019, 13. [Google Scholar] [CrossRef]
- Quezada Urban, R.; Díaz Velásquez, C.; Gitler, R.; Rojo Castillo, M.; Sirota Toporek, M.; Figueroa Morales, A.; Moreno García, O.; García Esquivel, L.; Torres Mejía, G.; Dean, M.; et al. Comprehensive analysis of germline variants in mexican patients with hereditary breast and ovarian cancer susceptibility. Cancers 2018, 10, 361. [Google Scholar] [CrossRef]
- Calderón-Zúñiga, F.D.C.; Ocampo-Gómez, G.; López-Márquez, F.C.; Recio-Vega, R.; Serrano-Gallardo, L.B.; Ruiz-Flores, P. ATM polymorphisms IVS24-9delT, IVS38-8T>C, and 5557G>A in Mexican women with familial and/or early-onset breast cancer. Salud Publica Mexico 2014, 56, 206–212. [Google Scholar] [CrossRef]
- Ruiz-Flores, P.; Sinilnikova, O.M.; Badzioch, M.; Calderon-Garcidueñas, A.L.; Chopin, S.; Fabrice, O.; González-Guerrero, J.F.; Szabo, C.; Lenoir, G.; Goldgar, D.E.; et al. BRCA1 and BRCA2 mutation analysis of early-onset and familial breast cancer cases in Mexico: Mutations in brief. Hum. Mutat. 2002, 20, 474–475. [Google Scholar] [CrossRef]
- Villarreal-Garza, C.; Weitzel, J.N.; Llacuachaqui, M.; Sifuentes, E.; Magallanes-Hoyos, M.C.; Gallardo, L.; Alvarez-Gómez, R.M.; Herzog, J.; Castillo, D.; Royer, R.; et al. The prevalence of BRCA1 and BRCA2 mutations among young Mexican women with triple-negative breast cancer. Breast Cancer Res. Treat. 2015, 150, 389–394. [Google Scholar] [CrossRef]
- Gómez-Díaz, B.; De La Luz Ayala-Madrigal, M.; Gutiérrez-Angulo, M.; Valle-Solis, A.E.; Linares-González, L.M.; González-Guzmán, R.; Cruz-Guillén, D.; Cedeño-Garcidueñas, A.L.; Canto, P.; López-Hernández, L.B. Analysis of ERCC1 and ERCC2 gene variants in osteosarcoma, colorectal and breast cancer. Oncol. Lett. 2015, 9, 1657–1661. [Google Scholar] [CrossRef][Green Version]
- Ziv, E. Genetics of breast cancer: Applications to the Mexican population. Salud Pública México 2011, 53, 5. [Google Scholar]
- Calderón-Garcidueñas, A.L.; Ruiz-Flores, P.; Cerda-Flores, R.M.; Barrera-Saldaña, H.A. Clinical follow up of Mexican women with early onset of breast cancer and mutations in the BRCA1 and BRCA2 genes. Salud Pública México 2005, 47, 110–115. [Google Scholar]
- Villarreal-Garza, C.; Alvarez-Gómez, R.M.; Pérez-Plasencia, C.; Herrera, L.A.; Herzog, J.; Castillo, D.; Mohar, A.; Castro, C.; Gallardo, L.N.; Gallardo, D.; et al. Significant clinical impact of recurrent BRCA1 and BRCA2 mutations in Mexico: Recurrent BRCA mutations in Mexico. Cancer 2015, 121, 372–378. [Google Scholar] [CrossRef]
- Vaca-Paniagua, F.; Alvarez-Gomez, R.M.; Fragoso-Ontiveros, V.; Vidal-Millan, S.; Herrera, L.A.; Cantú, D.; Bargallo-Rocha, E.; Mohar, A.; López-Camarillo, C.; Pérez-Plasencia, C. Full-exon pyrosequencing screening of BRCA germline mutations in Mexican women with inherited breast and ovarian cancer. PLoS ONE 2012, 7, e37432. [Google Scholar] [CrossRef]
- Torres-Mejia, G.; Royer, R.; Llacuachaqui, M.; Akbari, M.R.; Giuliano, A.R.; Martinez-Matsushita, L.; Angeles-Llerenas, A.; Ortega-Olvera, C.; Ziv, E.; Lazcano-Ponce, E.; et al. Recurrent BRCA1 and BRCA2 mutations in Mexican women with breast cancer. Cancer Epidemiol. Prev. Biomark. 2015, 24, 498–505. [Google Scholar] [CrossRef]
- Macías-Gómez, N.M.; Peralta-Leal, V.; Meza-Espinoza, J.P.; Gutiérrez-Angulo, M.; Durán-González, J.; Ramírez-González, J.M.; Gaspar-Del Toro, A.; Norberto-Rodríguez, A.; Leal-Ugarte, E. Polymorphisms of the XRCC1 gene and breast cancer risk in the Mexican population. Fam. Cancer 2015, 14, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Cock-Rada, A.M.; Ossa, C.A.; Garcia, H.I.; Gomez, L.R. A multi-gene panel study in hereditary breast and ovarian cancer in Colombia. Fam. Cancer 2018, 17, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Hernández, J.E.L.; Llacuachaqui, M.; Palacio, G.V.; Figueroa, J.D.; Madrid, J.; Lema, M.; Royer, R.; Li, S.; Larson, G.; Weitzel, J.N.; et al. Prevalence of BRCA1 and BRCA2 mutations in unselected breast cancer patients from Medellín, Colombia. Hered. Cancer Clin. Pract. 2014, 12. [Google Scholar] [CrossRef] [PubMed]
- Torres, D.; Bermejo, J.L.; Rashid, M.U.; Briceño, I.; Gil, F.; Beltran, A.; Ariza, V.; Hamann, U. Prevalence and penetrance of BRCA1 and BRCA2 germline mutations in Colombian breast cancer patients. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Briceño-Balcázar, I.; Gómez-Gutiérrez, A.; Díaz-Dussán, N.A.; Noguera-Santamaría, M.C.; Díaz, D.; Casas-Gómez, M.C. Mutational spectrum in breast cancer associated BRCA1 and BRCA2 genes in Colombia. Colomb. Méd. 2017, 48, 6. [Google Scholar]
- Torres, D.; Rashid, M.U.; Gil, F.; Umana, A.; Ramelli, G.; Robledo, J.F.; Tawil, M.; Torregrosa, L.; Briceno, I.; Hamann, U. High proportion of BRCA1/2 founder mutations in Hispanic breast/ovarian cancer families from Colombia. Breast Cancer Res. Treat. 2007, 103, 225–232. [Google Scholar] [CrossRef]
- Llinás-Quintero, N.; Cabrera-Florez, E.; Mendoza-Fandiño, G.; Matute-Turizo, G.; Vasquez-Trespalacios, E.M.; Gallón-Villegas, L.J. Synchronous ovarian and breast cancers with a novel variant in BRCA2 gene: A case report. Case Rep. Oncol. Med. 2019, 2019, 1–5. [Google Scholar] [CrossRef]
- Solano, A.R.; Aceto, G.M.; Delettieres, D.; Veschi, S.; Neuman, M.I.; Alonso, E.; Chialina, S.; Chacón, R.D.; Renato, M.-C.; Podestá, E.J. BRCA1 And BRCA2 analysis of Argentinean breast/ovarian cancer patients selected for age and family history highlights a role for novel mutations of putative south-American origin. SpringerPlus 2012, 1. [Google Scholar] [CrossRef]
- Solano, A.R.; Liria, N.C.; Jalil, F.S.; Faggionato, D.M.; Mele, P.G.; Mampel, A.; Cardoso, F.C.; Podesta, E.J. BRCA1 and BRCA2 mutations other than the founder alleles among Ashkenazi Jewish in the population of Argentina. Front. Oncol. 2018, 8. [Google Scholar] [CrossRef]
- Solano, A.R.; Cardoso, F.C.; Romano, V.; Perazzo, F.; Bas, C.; Recondo, G.; Santillan, F.B.; Gonzalez, E.; Abalo, E.; Viniegra, M.; et al. Spectrum of BRCA1/2 variants in 940 patients from Argentina including novel, deleterious and recurrent germline mutations: Impact on healthcare and clinical practice. Oncotarget 2017, 8. [Google Scholar] [CrossRef]
- Aceto, G.M.; Solano, A.R.; Neuman, M.I.; Veschi, S.; Morgano, A.; Malatesta, S.; Chacon, R.D.; Pupareli, C.; Lombardi, M.; Battista, P.; et al. High-risk human papilloma virus infection, tumor pathophenotypes, and BRCA1/2 and TP53 status in juvenile breast cancer. Breast Cancer Res. Treat. 2010, 122, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Abugattas, J.; Llacuachaqui, M.; Allende, Y.S.; Velásquez, A.A.; Velarde, R.; Cotrina, J.; Garcés, M.; León, M.; Calderón, G.; de la Cruz, M.; et al. Prevalence of BRCA1 and BRCA2 mutations in unselected breast cancer patients from Peru: Prevalence of BRCA1/2 mutations in unselected breast cancer patients from Peru. Clin. Genet. 2015, 88, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Buleje, J.; Guevara-Fujita, M.; Acosta, O.; Huaman, F.D.P.; Danos, P.; Murillo, A.; Pinto, J.A.; Araujo, J.M.; Aguilar, A.; Ponce, J.; et al. Mutational analysis of BRCA1 and BRCA2 genes in Peruvian families with hereditary breast and ovarian cancer. Mol. Genet. Genom. Med. 2017, 5, 481–494. [Google Scholar] [CrossRef] [PubMed]
- González-Rivera, M.; Lobo, M.; López-Tarruella, S.; Jerez, Y.; del Monte-Millán, M.; Massarrah, T.; Ramos-Medina, R.; Ocaña, I.; Picornell, A.; Garzón, S.S.; et al. Frequency of germline DNA genetic findings in an unselected prospective cohort of triple-negative breast cancer patients participating in a platinum-based neoadjuvant chemotherapy trial. Breast Cancer Res. Treat. 2016, 156, 507–515. [Google Scholar] [CrossRef]
- Diaz-Zabala, H.; Ortiz, A.; Garland, L.; Jones, K.; Perez, C.; Mora, E.; Arroyo, N.; Oleksyk, T.; Echenique, M.; Matta, J.; et al. A Recurrent BRCA2 Mutation explains the majority of hereditary breast and ovarian cancer syndrome cases in Puerto Rico. Cancers 2018, 10, 419. [Google Scholar] [CrossRef]
- Dutil, J.; Colon-Colon, J.L.; Matta, J.L.; Sutphen, R.; Echenique, M. Identification of the prevalent BRCA1 and BRCA2 mutations in the female population of Puerto Rico. Cancer Genet. 2012, 205, 242–248. [Google Scholar] [CrossRef]
- Pérez-Mayoral, J.; Pacheco-Torres, A.L.; Morales, L.; Acosta-Rodríguez, H.; Matta, J.L.; Dutil, J. Genetic polymorphisms in RAD23B and XPC modulate DNA repair capacity and breast cancer risk in Puerto Rican women: RAD23B polymorphisms in breast cancer risk. Mol. Carcinog. 2013, 52, 127–138. [Google Scholar] [CrossRef]
- Delgado, L.; Fernández, G.; Grotiuz, G.; Cataldi, S.; González, A.; Lluveras, N.; Heguaburu, M.; Fresco, R.; Lens, D.; Sabini, G.; et al. BRCA1 and BRCA2 germline mutations in Uruguayan breast and breast–ovarian cancer families. Identification of novel mutations and unclassified variants. Breast Cancer Res. Treat. 2011, 128, 211–218. [Google Scholar] [CrossRef]
- Valle, D.A.D.; Acevedo, C.; Esperón, P.; Neffa, F.; Artagaveytia, N.; Santander, G.; Menini, M.; Vergara, B.C.; Carusso, B.F.; Sapone, L.M. Cáncer de mama y ovario hereditario en Uruguay: Resultados del screening para mutaciones en genes de susceptibilidad por secuenciación de nueva generación. Rev. Med. Urug. 2017, 33, 40–52. [Google Scholar]
- Delgado, L.; Fernández, G.; González, A.; Paillerets, B.B.; Gualco, G.; Bombled, J.; Cataldi, S.; Sabini, G.; Roca, R.; Musé, I.M. Hereditary breast cancer associated with a germline BRCA2 mutation in identical female twins with similar disease expression. Cancer Genet. Cytogenet. 2002, 133, 24–28. [Google Scholar] [CrossRef]
- Gutiérrez Espeleta, G.; Llacuachaqui, M.; García-Jiménez, L.; Aguilar Herrera, M.; Loáiciga Vega, K.; Ortiz, A.; Royer, R.; Li, S.; Narod, S. BRCA1 and BRCA2 mutations among familial breast cancer patients from Costa Rica. Clin. Genet. 2012, 82, 484–488. [Google Scholar] [CrossRef] [PubMed]
- García-Jiménez, L.; Gutiérrez-Espeleta, G.; Narod, S.A. Epidemiología descriptiva y genética molecular del cáncer de mama hereditario en Costa Rica. Rev. Biol. Trop. 2012, 60, 1663–1668. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.C.; Esperon, A.A.; Ropero, R.; Rubio, M.C.; Rodriguez, R.; Ortiz, R.M.; Anta, J.J.L.; de los Rios, M.; Carnesolta, D.; del Olivera, M.C.; et al. Prevalence of BRCA1 and BRCA2 mutations in breast cancer patients from Cuba. Fam. Cancer 2008, 7, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Lara, K.; Consigliere, N.; Pérez, J.; Porco, A. BRCA1 and BRCA2mutations in breast cancer patients from Venezuela. Biol. Res. 2012, 45, 117–130. [Google Scholar] [CrossRef]
- Ashton-Prolla, P.; Vargas, F.R. Prevalence and impact of founder mutations in hereditary breast cancer in Latin America. Genet. Mol. Biol. 2014, 37, 234–240. [Google Scholar] [CrossRef]
- Ossa, C.A.; Torres, D. Founder and recurrent mutations in BRCA1 and BRCA2 genes in Latin American countries: State of the art and literature review. Oncology 2016, 21, 832–839. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 2013, 6, pl1. [Google Scholar] [CrossRef]
- Palmirotta, R.; Lovero, D.; Stucci, L.; Silvestris, E.; Quaresmini, D.; Cardascia, A.; Silvestris, F. Double heterozygosity for BRCA1 pathogenic variant and BRCA2 polymorphic stop codon K3326X: A case report in a southern Italian family. Int. J. Mol. Sci. 2018, 19, 285. [Google Scholar] [CrossRef]
- Riaz, N.; Blecua, P.; Lim, R.S.; Shen, R.; Higginson, D.S.; Weinhold, N.; Norton, L.; Weigelt, B.; Powell, S.N.; Reis-Filho, J.S. Pan-cancer analysis of bi-allelic alterations in homologous recombination DNA repair genes. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef]
- Easton, D.F.; Pharoah, P.D.P.; Antoniou, A.C.; Tischkowitz, M.; Tavtigian, S.V.; Nathanson, K.L.; Devilee, P.; Meindl, A.; Couch, F.J.; Southey, M.; et al. Gene-panel sequencing and the prediction of breast-cancer risk. N. Engl. J. Med. 2015, 372, 2243–2257. [Google Scholar] [CrossRef] [PubMed]
- Vink, G.R.; van Asperen, C.J.; Devilee, P.; Breuning, M.H.; Bakker, E. Unclassified variants in disease-causing genes: Nonuniformity of genetic testing and counselling, a proposal for guidelines. Eur. J. Hum. Genet. 2005, 13, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Lindor, N.M.; Goldgar, D.E.; Tavtigian, S.V.; Plon, S.E.; Couch, F.J. BRCA1/2 sequence variants of uncertain significance: A primer for providers to assist in discussions and in medical management. Oncology 2013, 18, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Caswell-Jin, J.L.; Gupta, T.; Hall, E.; Petrovchich, I.M.; Mills, M.A.; Kingham, K.E.; Koff, R.; Chun, N.M.; Levonian, P.; Lebensohn, A.P.; et al. Racial/ethnic differences in multiple-gene sequencing results for hereditary cancer risk. Genet. Med. Off. J. Am. Coll. Med. Genet. 2018, 20, 234–239. [Google Scholar] [CrossRef]
- Jara, L.; Morales, S.; de Mayo, T.; Gonzalez-Hormazabal, P.; Carrasco, V.; Godoy, R. Mutations in BRCA1, BRCA2 and other breast and ovarian cancer susceptibility genes in Central and South American populations. Biol. Res. 2017, 50. [Google Scholar] [CrossRef]
- Palla, V.-V.; Karaolanis, G.; Katafigiotis, I.; Anastasiou, I.; Patapis, P.; Dimitroulis, D.; Perrea, D. Gamma-H2AX: Can it be established as a classical cancer prognostic factor? Tumor Biol. 2017, 39, 101042831769593. [Google Scholar] [CrossRef]
- Helena, J.; Joubert, A.; Grobbelaar, S.; Nolte, E.; Nel, M.; Pepper, M.; Coetzee, M.; Mercier, A. Deoxyribonucleic acid damage and repair: Capitalizing on our understanding of the mechanisms of maintaining genomic integrity for therapeutic purposes. Int. J. Mol. Sci. 2018, 19, 1148. [Google Scholar] [CrossRef]
- Ranjha, L.; Howard, S.M.; Cejka, P. Main steps in DNA double-strand break repair: An introduction to homologous recombination and related processes. Chromosoma 2018, 127, 187–214. [Google Scholar] [CrossRef]
- Liu, D.; Keijzers, G.; Rasmussen, L.J. DNA mismatch repair and its many roles in eukaryotic cells. Mutat. Res. 2017, 773, 174–187. [Google Scholar] [CrossRef]
- Gallmeier, E.; Kern, S.E. Targeting Fanconi anemia/BRCA2 pathway defects in cancer: The significance of preclinical pharmacogenomic models. Clin. Cancer Res. 2007, 13, 4–10. [Google Scholar] [CrossRef]
- Tulay, P.; Sengupta, S.B. MicroRNA expression and its association with DNA repair in preimplantation embryos. J. Reprod. Dev. 2016, 62, 225–234. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dhawan, M.; Ryan, C.J. BRCAness and prostate cancer: Diagnostic and therapeutic considerations. Prostate Cancer Prostatic Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gavande, N.S.; VanderVere-Carozza, P.S.; Hinshaw, H.D.; Jalal, S.I.; Sears, C.R.; Pawelczak, K.S.; Turchi, J.J. DNA repair targeted therapy: The past or future of cancer treatment? Pharm. Ther. 2016, 160, 65–83. [Google Scholar] [CrossRef] [PubMed]
- Pilié, P.G.; Tang, C.; Mills, G.B.; Yap, T.A. State-of-the-art strategies for targeting the DNA damage response in cancer. Nat. Rev. Clin. Oncol. 2018. [Google Scholar] [CrossRef]
- Dine, J.; Gordon, R.; Shames, Y.; Kasler, M.K.; Barton-Burke, M. Immune Checkpoint inhibitors: An innovation in immunotherapy for the treatment and management of patients with cancer. Asia-Pac. J. Oncol. Nurs. 2017, 4, 127–135. [Google Scholar] [CrossRef]
- Yi, M.; Jiao, D.; Xu, H.; Liu, Q.; Zhao, W.; Han, X.; Wu, K. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol. Cancer 2018, 17. [Google Scholar] [CrossRef]
- Palles, C.; Cazier, J.-B.; Howarth, K.M.; Domingo, E.; Jones, A.M.; Broderick, P.; Kemp, Z.; Spain, S.L.; Almeida, E.G.; Salguero, I.; et al. Germline mutations in the proof-reading domains of POLE and POLD1 predispose to colorectal adenomas and carcinomas. Nat. Genet. 2013, 45, 136–144. [Google Scholar] [CrossRef]
- Marginean, E.C.; Melosky, B. Is there a role for programmed death ligand-1 testing and immunotherapy in colorectal cancer with microsatellite instability? Part I—Colorectal cancer: Microsatellite instability, testing, and clinical implications. Arch. Pathol. Lab. Med. 2018, 142, 17–25. [Google Scholar] [CrossRef]
- Bai, F.; Chan, H.L.; Scott, A.; Smith, M.D.; Fan, C.; Herschkowitz, J.I.; Perou, C.M.; Livingstone, A.S.; Robbins, D.J.; Capobianco, A.J.; et al. BRCA1 suppresses epithelial-to-mesenchymal transition and stem cell dedifferentiation during mammary and tumor development. Cancer Res. 2014, 74, 6161–6172. [Google Scholar] [CrossRef]
- Deniz, M.; Kaufmann, J.; Stahl, A.; Gundelach, T.; Janni, W.; Hoffmann, I.; Keimling, M.; Hampp, S.; Ihle, M.; Wiesmüller, L. In vitro model for DNA double-strand break repair analysis in breast cancer reveals cell type–specific associations with age and prognosis. FASEB J. 2016, 30, 3786–3799. [Google Scholar] [CrossRef]
- Keimling, M.; Deniz, M.; Varga, D.; Stahl, A.; Schrezenmeier, H.; Kreienberg, R.; Hoffmann, I.; König, J.; Wiesmüller, L. The power of DNA double-strand break (DSB) repair testing to predict breast cancer susceptibility. FASEB J. 2012, 26, 2094–2104. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration. FDA Approves Olaparib for Germline BRCA-Mutated Metastatic Breast Cancer. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-olaparib-germline-brca-mutated-metastatic-breast-cancer (accessed on 4 July 2019).
- US Food and Drug Administration. FDA Approves Talazoparib for GBRCAm HER2-Negative Locally Advanced or Metastatic Breast Cancer. Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-talazoparib-gbrcam-her2-negative-locally-advanced-or-metastatic-breast-cancer (accessed on 4 July 2019).
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.-A.; Wright, G.S.; et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N. Engl. J. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Nanda, R.; Liu, M.C.; Yau, C.; Asare, S.; Hylton, N.; Veer, L.V.; Perlmutter, J.; Wallace, A.M.; Chien, A.J.; Forero-Torres, A.; et al. Pembrolizumab plus standard neoadjuvant therapy for high-risk breast cancer (BC): Results from I-SPY 2. J. Clin. Oncol. 2017, 35, 506. [Google Scholar] [CrossRef]
- Gilmore, E.; McCabe, N.; Kennedy, R.D.; Parkes, E.E. DNA Repair Deficiency in Breast Cancer: Opportunities for Immunotherapy. Available online: https://www.hindawi.com/journals/jo/2019/4325105/ (accessed on 4 July 2019).
- Barrios, C.H.; Reinert, T.; Werutsky, G. Access to high-cost drugs for advanced breast cancer in Latin America, particularly trastuzumab. Ecancermedicalscience 2019, 13. [Google Scholar] [CrossRef]
- Ruiz, R.; Strasser-Weippl, K.; Touya, D.; Vincent, C.H.; Hernandez-Blanquisett, A.; Louis, J.S.; Bukowski, A.; Goss, P.E. Improving access to high-cost cancer drugs in Latin America: Much to be done. Cancer 2017, 123, 1313–1323. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.A.; Pinillos, L.; Villarreal-Garza, C.; Morante, Z.; Villarán, M.V.; Mejía, G.; Caglevic, C.; Aguilar, A.; Fajardo, W.; Usuga, F.; et al. Barriers in Latin America for the management of locally advanced breast cancer. Ecancermedicalscience 2019, 13. [Google Scholar] [CrossRef]
- Cazap, E. Breast Cancer in Latin America: A Map of the Disease in the Region. Am. Soc. Clin. Oncol. Educ. Book 2018, 38, 451–456. [Google Scholar] [CrossRef]

| Country | Total Retrieved Papers 1 | Germline Data 2 | BRCA1/2 Papers | Non-BRCA Papers | Total Papers | References | |
|---|---|---|---|---|---|---|---|
| 1 | Brazil | 3290 | ✔ | 13 | 19 | 32 | [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65] |
| 2 | Chile | 455 | ✔ | 7 | 7 | 14 | [66,67,68,69,70,71,72,73,74,75,76,77,78,79] |
| 3 | Mexico | 2014 | ✔ | 8 | 4 | 12 | [80,81,82,83,84,85,86,87,88,89,90,91] |
| 4 | Colombia | 274 | ✔ | 5 | 1 | 6 | [92,93,94,95,96,97] |
| 5 | Argentina | 893 | ✔ | 4 | 4 | [98,99,100,101] | |
| 6 | Peru | 161 | ✔ | 2 | 1 | 3 | [102,103,104] |
| 7 | Puerto Rico | 253 | ✔ | 2 | 1 | 3 | [105,106,107] |
| 8 | Uruguay | 126 | ✔ | 3 | 3 | [108,109,110] | |
| 9 | Costa Rica | 56 | ✔ | 2 | 2 | [111,112] | |
| 10 | Cuba | 142 | ✔ | 1 | 1 | [113] | |
| 11 | Venezuela | 76 | ✔ | 1 | 1 | [114] | |
| 12 | Ecuador | 42 | |||||
| 13 | Paraguay | 31 | |||||
| 14 | Panama | 23 | |||||
| 15 | Dominican Republic | 18 | |||||
| 16 | El Salvador | 16 | |||||
| 17 | Bolivia | 13 | |||||
| 18 | Guatemala | 8 | |||||
| 19 | Nicaragua | 7 | |||||
| 20 | Honduras | 2 | |||||
| 21 | Belize | 1 | |||||
| Total | 7901 | 11 | 48 | 33 | 81 |
| Gene | Number of Variants | Argentina | Brazil | Chile | Colombia | Costa Rica | Cuba | Mexico | Peru | Puerto Rico | Uruguay | Venezuela |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BRCA2 | 363 | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ |
| BRCA1 | 329 | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ |
| ATM | 17 | ✔ | ✔ | ✔ | ✔ | |||||||
| TP53 | 11 | ✔ | ✔ | ✔ | ||||||||
| CHEK2 | 8 | ✔ | ✔ | ✔ | ✔ | |||||||
| BARD1 | 6 | ✔ | ✔ | ✔ | ✔ | ✔ | ||||||
| MLH1 | 6 | ✔ | ✔ | ✔ | ||||||||
| PALB2 | 6 | ✔ | ✔ | |||||||||
| BRIP1 | 5 | ✔ | ✔ | |||||||||
| MSH2 | 4 | ✔ | ✔ | |||||||||
| MSH6 | 4 | ✔ | ✔ | |||||||||
| NBN | 4 | ✔ | ✔ | |||||||||
| PMS2 | 4 | ✔ | ||||||||||
| APC | 3 | ✔ | ||||||||||
| ATR | 3 | ✔ | ✔ | |||||||||
| BLM | 3 | ✔ | ||||||||||
| RAD51C | 3 | ✔ | ✔ | ✔ | ||||||||
| CDKN2A | 2 | ✔ | ||||||||||
| ERCC1 | 2 | ✔ | ✔ | |||||||||
| ERCC2 | 2 | ✔ | ✔ | ✔ | ||||||||
| ERCC3 | 2 | ✔ | ||||||||||
| FANCB | 2 | ✔ | ||||||||||
| FANCI | 2 | ✔ | ||||||||||
| LIG4 | 2 | ✔ | ||||||||||
| MUTYH | 2 | ✔ | ||||||||||
| RAD51B | 2 | ✔ | ✔ | |||||||||
| RAD51D | 2 | ✔ | ✔ | |||||||||
| XRCC1 | 2 | ✔ | ✔ | |||||||||
| CDH1 | 1 | ✔ | ||||||||||
| FANCC | 1 | ✔ | ||||||||||
| FANCF | 1 | ✔ | ||||||||||
| FANCL | 1 | ✔ | ||||||||||
| FANCM | 1 | ✔ | ||||||||||
| MSH3 | 1 | ✔ | ||||||||||
| NF1 | 1 | ✔ | ||||||||||
| POLH | 1 | ✔ | ||||||||||
| POLQ | 1 | ✔ | ||||||||||
| PTEN | 1 | ✔ | ||||||||||
| RAD50 | 1 | ✔ | ||||||||||
| RAD51 | 1 | ✔ | ✔ | |||||||||
| RECQL4 | 1 | ✔ | ||||||||||
| SMAD4 | 1 | ✔ | ||||||||||
| WRN | 1 | ✔ | ||||||||||
| XPC | 1 | ✔ | ||||||||||
| XRCC3 | 1 | ✔ | ✔ |
| Gene | rs | Exon | Argentina | Brazil | Chile | Colombia | Costa Rica | Cuba | Mexico | Peru | Puerto Rico | Uruguay | Venezuela |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BRCA1 VARIANT | |||||||||||||
| c.68_69delAG | rs386833395 | 2 | (4) | (4) | (2) | (1) | (1) | (1) | |||||
| c.181T>G | rs28897672 | 5 | (1) | (4) | (1) | ||||||||
| c.211A>G | rs80357382 | 5 | (2) | (3) | (1) | (3) | (1) | (1) | |||||
| c.3113A>G | rs16941 | 11 | (1) | (3) | (2) | (1) | |||||||
| c.3548A>G | rs16942 | 11 | (1) | (3) | (1) | (1) | |||||||
| c.1067A>G | rs1799950 | 11 | 2 | (3) | (1) | ||||||||
| c.3119G>A | rs4986852 | 11 | (1) | (3) | (1) | (1) | (1) | ||||||
| c.2612C>T | rs799917 | 11 | (1) | (3) | (1) | ||||||||
| c.3331_3334delCAAG | rs80357701 | 11 | (9) | (3) | (6) | ||||||||
| c.4308C>T | rs1060915 | 13 | (1) | (2) | (1) | ||||||||
| c.4837A>G | rs1799966 | 16 | (1) | (3) | (1) | (1) | |||||||
| c.5123C>A | rs28897696 | 18 | (1) | (2) | (8) | (3) | |||||||
| c.5266dupC | rs397507247 | 20 | (4) | (16) | (1) | (1) | |||||||
| BRCA2 VARIANT | |||||||||||||
| c.865A>C | rs766173 | 10 | (1) | (2) | (1) | (1) | |||||||
| c.2971A>G | rs1799944 | 11 | (1) | (2) | (2) | (1) | (1) | ||||||
| c.5744C>T | rs4987117 | 11 | (1) | (2) | (2) | (1) | (1) | ||||||
| c.2808_2811del | rs80359351 | 11 | (3) | (5) | (2) | (1) | (2) | (1) | (1) | ||||
| c.5351dupA | rs80359507 | 11 | (1) | (2) | (2) | ||||||||
| c.5946delT | rs80359550 | 11 | (5) | (4) | (1) | (4) | (1) | ||||||
| c.7469T>C | rs11571707 | 15 | (2) | (4) | (1) | (1) | (1) | (1) | (1) | ||||
| c.10234A>G | rs1801426 | 27 | (1) | (3) | (2) | (1) |
| Gene | Mutation | Cases | Brazil | Colombia | Mexico | Peru | Puerto Rico | Uruguay | References |
|---|---|---|---|---|---|---|---|---|---|
| BRCA1 | exon 1-2 deletion | 3 | ✔ | ✔ | [60,63,106] | ||||
| BRCA1 | exon 3 deletion | 2 | ✔ | [60,63] | |||||
| BRCA1 | exon 4-6 deletion | 2 | ✔ | [60,63] | |||||
| BRCA1 | exon 5-7 deletion | 2 | ✔ | [53,60] | |||||
| BRCA1 | exon 8 deletion | 2 | ✔ | [60,63] | |||||
| BRCA1 | exon 8-13 deletion | 1 | ✔ | [104] | |||||
| BRCA1 | exon 9-11 deletion | 1 | ✔ | [80] | |||||
| BRCA1 | exon 9-12 deletion | 25 | ✔ | [80,81,84,88] | |||||
| BRCA1 | exon 9-19 deletion | 2 | ✔ | [41,60] | |||||
| BRCA1 | exon 12 deletion | 1 | ✔ | [80] | |||||
| BRCA1 | exon 14 deletion | 1 | ✔ | [109] | |||||
| BRCA1 | exon 14-16 deletion | 1 | ✔ | [60] | |||||
| BRCA1 | exon 16-17 deletion | 2 | ✔ | [60,65] | |||||
| BRCA1 | exon 18-19 deletion | 2 | ✔ | ✔ | [60,104] | ||||
| BRCA1 | exon 19 deletion | 1 | ✔ | [60] | |||||
| BRCA1 | exon 21-23 deletion | 1 | ✔ | [60] | |||||
| BRCA1 | exon 24 duplication | 1 | ✔ | [65] | |||||
| BRCA1 | 6-KB DUP EX13 | 7 | ✔ | [51] | |||||
| BRCA2 | exon 1 deletion | 3 | ✔ | [80] | |||||
| BRCA2 | exon 1-14 deletion | 2 | ✔ | [94] | |||||
| BRCA2 | exon 2 deletion | 1 | ✔ | [60] | |||||
| BRCA2 | exon 11 deletion | 1 | ✔ | [80] | |||||
| BRCA2 | exon 13 deletion | 1 | ✔ | [60] | |||||
| BRCA2 | exon 14 deletion | 1 | ✔ | [60] | |||||
| BRCA2 | exon 15-16 deletion | 1 | ✔ | [92] | |||||
| BRCA2 | exon 17 deletion | 1 | ✔ | [80] | |||||
| BRCA2 | exon 22-24 deletion | 2 | ✔ | [80] | |||||
| BRCA2 | exon 23 deletion | 2 | ✔ | [80] | |||||
| BRCA2 | exon 25 deletion | 1 | ✔ | [60] | |||||
| BRCA2 | exon 26 deletion | 1 | ✔ | [80] | |||||
| BRCA2 | g.26826_30318del | 2 | ✔ | [35,64] |
| Gene | rs | Variant | COSMIC | CLINVAR | Brazil | Chile | Colombia | Mexico | Peru | Uruguay |
|---|---|---|---|---|---|---|---|---|---|---|
| ATM | NA | c.634delT | ✔ | x | ||||||
| ATM | NA | c.5648_5655del | x | |||||||
| ATM | rs145119475 | c.4060C>A | ✔ | x | ||||||
| ATM | rs1800056 | c.2572T>C | ✔ | ✔ | x | |||||
| ATM | rs1801516 | c.5557G>A | ✔ | ✔ | x | x | ||||
| ATM | rs1801673 | c.5558A>T | ✔ | ✔ | x | |||||
| ATM | rs200381392 | c.1703G>T | ✔ | x | ||||||
| ATM | rs202173660 | c.1444A>C | ✔ | ✔ | x | |||||
| ATM | rs2234997 | c.378T>A | ✔ | ✔ | x | |||||
| ATM | rs2235006 | c.1744T>C | ✔ | x | ||||||
| ATM | rs4986761 | c.2119T>C | ✔ | ✔ | x | |||||
| ATM | rs587782153 | c.5039C>T | ✔ | ✔ | x | |||||
| ATM | rs758962678 | c.241A>G | ✔ | x | ||||||
| ATM | rs759965045 | c.7702_7703del | ✔ | x | ||||||
| ATM | rs771887195 | c.43del | ✔ | x | ||||||
| ATM | rs786203421 | c.7000_7003delTACA | ✔ | x | ||||||
| ATM | rs786204433 | c.5644C>T | ✔ | ✔ | x | |||||
| TP53 | rs1042522 | c.215C>G | ✔ | ✔ | x | |||||
| TP53 | rs11540652 | c.743G>A | ✔ | ✔ | x | |||||
| TP53 | rs121912664 | c.1010G>A | ✔ | ✔ | x | |||||
| TP53 | rs121913344 | c.916C>T | ✔ | ✔ | x | |||||
| TP53 | rs144386518 | c.173C>G | ✔ | ✔ | x | |||||
| TP53 | rs1800370 | c.108G>A | ✔ | ✔ | x | |||||
| TP53 | rs1800371 | c.139C>T | ✔ | ✔ | x | |||||
| TP53 | rs28934576 | c.818G>A | ✔ | ✔ | x | |||||
| TP53 | rs55863639 | c.375G>A | ✔ | ✔ | x | |||||
| TP53 | rs587782144 | c.473G>A | ✔ | ✔ | x | |||||
| TP53 | rs587782620 | c.427G>A | ✔ | ✔ | x | |||||
| CHEK2 | NA | c.1015C>T | x | |||||||
| CHEK2 | NA | c.1151delT | x | |||||||
| CHEK2 | NA | c.705A>C | x | |||||||
| CHEK2 | NA | c.852G>T | x | |||||||
| CHEK2 | rs1555926890? | c.506T>C | x | |||||||
| CHEK2 | rs555607708 | c.1100delC | ✔ | x | x | |||||
| CHEK2 | rs587781652 | c.485A>G | ✔ | x | ||||||
| CHEK2 | rs864622149 | c.846+1G>C | ✔ | x | ||||||
| BARD1 | NA | c.2215dupT | x | |||||||
| BARD1 | rs143914387 | c.33G>T | x | |||||||
| BARD1 | rs28997576 | c.1670G>C | x | |||||||
| BARD1 | rs587781948 | c.1921C>T | x | |||||||
| BARD1 | rs758972589 | c.334C>T | ✔ | ✔ | x | |||||
| BARD1 | rs777937955 | c.1622C>A | x | |||||||
| MLH1 | NA | c.1966C>T | x | |||||||
| MLH1 | NA | c.413A>G | x | |||||||
| MLH1 | NA | c.791G>A | x | |||||||
| MLH1 | rs148317871 | c.2213G>A | ✔ | x | ||||||
| MLH1 | rs63751615 | c.676C>T | ✔ | x | ||||||
| MLH1 | del_exon8 | NA | NA | x | ||||||
| PALB2 | NA | c.1861C>A | x | |||||||
| PALB2 | NA | c.483C>G | x | |||||||
| PALB2 | rs150390726 | c.23C>T | ✔ | x | ||||||
| PALB2 | rs152451 | c.1676A>G | ✔ | x | ||||||
| PALB2 | rs180177100 | c.1240C>T | ✔ | ✔ | x | |||||
| PALB2 | rs45551636 | c.2993C>T | x | |||||||
| BRIP1 | rs202072866 | c.415T>G | ✔ | ✔ | x | |||||
| BRIP1 | rs28997569 | c.790C>T | ✔ | x | ||||||
| BRIP1 | rs371185409 | c.3079G>A | ✔ | x | ||||||
| BRIP1 | rs45589637 | c.2220G>T | ✔ | x | ||||||
| BRIP1 | rs759031349 | c.689C>T | ✔ | x |
| Patient ID | Gene 1 | Gene 2 | Age Onset | Country | Family History | Subtype | Reference |
|---|---|---|---|---|---|---|---|
| NA | BRCA1: c.1674del (pathogenic) | MSH6: c.2419G>A (uncertain significance) | NA | Colombia | YES | NA | [92] |
| NA | BRCA1: c.1674del (pathogenic) | PMS2: c.2395C>T (uncertain significance); RAD51C: c.492T>G (uncertain significance) | NA | Colombia | YES | NA | [92] |
| 15 | PALB2: c.1240C>T (pathogenic) | PMS2: c.241G>A (uncertain significance) | 36 | Colombia | YES | ER+, HER2-, invasive ductal carcinoma | [92] |
| 8 | BRCA2: c.5616-5620del (not reported) | SMAD4: c.677C>T (conflicting interpretations) | 35 | Colombia | YES | HR+, HER2- invasive ductal carcinoma | [92] |
| NA | BRCA1: c.4357+1G>T (pathogenic) | BRCA2: c.6405_6409delCTTAA (pathogenic) | 38 | Brazil | NA | ipsilateral BC | [63] |
| NA | BRCA1: LGR (deletion of exons 4–6) | BRCA2: c.9004G>A (conflicting interpretation) | 43 | Brazil | NA | NA | [63] |
| NA | BRCA2: c.8878C>T (pathogenic), c.9699_9702delTATG (pathogenic) | 52 | Brazil | NA | NA | [63] | |
| CM001 | BRCA1: c.1129_1135insA (not reported), c.4063_4065delAAT (conflicting interpretations) | 37 | Venezuela | YES | ER+, PR+ | [114] | |
| CM055 | BRCA1: c.1129_1135insA (not reported), c.4063_4065delAAT (conflicting interpretations) | 48 | Venezuela | YES | ER-, PR- | [114] | |
| CM031 | BRCA2: c.1282T>C (not reported), c.3479G>A (conflicting interpretations) | 49 | Venezuela | YES | NA | [114] | |
| 5 | BRCA2: c.865A>C (benign), c.2971A>G(benign), c.8851G>A (benign) | 33 | Mexico | NA | ductal | [87] | |
| 12 | BRCA1: c.2245G>T (uncertain significance) | BRCA2: p.Ile3412Val (benign) | 34 | Mexico | NA | ductal | [87] |
| 7 | BRCA1: c.442-34C>T (benign) | BRCA2: c.865A>C (benign), c.2971A>G (benign) | 34 | Mexico | NA | ductal | [87] |
| 17 | BRCA1: c.3548A>G (benign), c.442-34C>T (benign) | 30 | Mexico | YES | ER+, PR+, HER2- | [89] | |
| A11 | BRCA1: c.4308T>C (benign), c.442-34C (not reported), c.5152+66G>A (benign), c.548-58delT (benign) | BRCA2: c.426+67A>C (not reported), c.426-89T>C (benign), c.7435+53C>T (benign) | 12 | Argentina | YES | TNBC, Secretory carcinoma | [101] |
| A17 | BRCA1: c.4308T>C (benign), c.5152+66G>A (benign), c.548-58delT (benign) | 25 | Argentina | NO | PR+, ER+, HER2+, infiltrating ductal carcinoma | [101] | |
| A18 | BRCA1: c.442-34T>C (not reported) | BRCA2: c.7469T>C (benign), c.681+56C>T (benign), c.7242A>G (benign) | 21 | Argentina | NO | ER+, PR+ HER2-, infiltrating lobular carcinoma | [101] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbina-Jara, L.K.; Rojas-Martinez, A.; Martinez-Ledesma, E.; Aguilar, D.; Villarreal-Garza, C.; Ortiz-Lopez, R. Landscape of Germline Mutations in DNA Repair Genes for Breast Cancer in Latin America: Opportunities for PARP-Like Inhibitors and Immunotherapy. Genes 2019, 10, 786. https://doi.org/10.3390/genes10100786
Urbina-Jara LK, Rojas-Martinez A, Martinez-Ledesma E, Aguilar D, Villarreal-Garza C, Ortiz-Lopez R. Landscape of Germline Mutations in DNA Repair Genes for Breast Cancer in Latin America: Opportunities for PARP-Like Inhibitors and Immunotherapy. Genes. 2019; 10(10):786. https://doi.org/10.3390/genes10100786
Chicago/Turabian StyleUrbina-Jara, Laura Keren, Augusto Rojas-Martinez, Emmanuel Martinez-Ledesma, Dione Aguilar, Cynthia Villarreal-Garza, and Rocio Ortiz-Lopez. 2019. "Landscape of Germline Mutations in DNA Repair Genes for Breast Cancer in Latin America: Opportunities for PARP-Like Inhibitors and Immunotherapy" Genes 10, no. 10: 786. https://doi.org/10.3390/genes10100786
APA StyleUrbina-Jara, L. K., Rojas-Martinez, A., Martinez-Ledesma, E., Aguilar, D., Villarreal-Garza, C., & Ortiz-Lopez, R. (2019). Landscape of Germline Mutations in DNA Repair Genes for Breast Cancer in Latin America: Opportunities for PARP-Like Inhibitors and Immunotherapy. Genes, 10(10), 786. https://doi.org/10.3390/genes10100786






