A Novel Missense Variant Associated with A Splicing Defect in A Myopathic Form of PGK1 Deficiency in The Spanish Population
Abstract
:1. Introduction
1.1. PGK Enzyme
1.2. PGK1 Deficiency
2. Materials and Methods
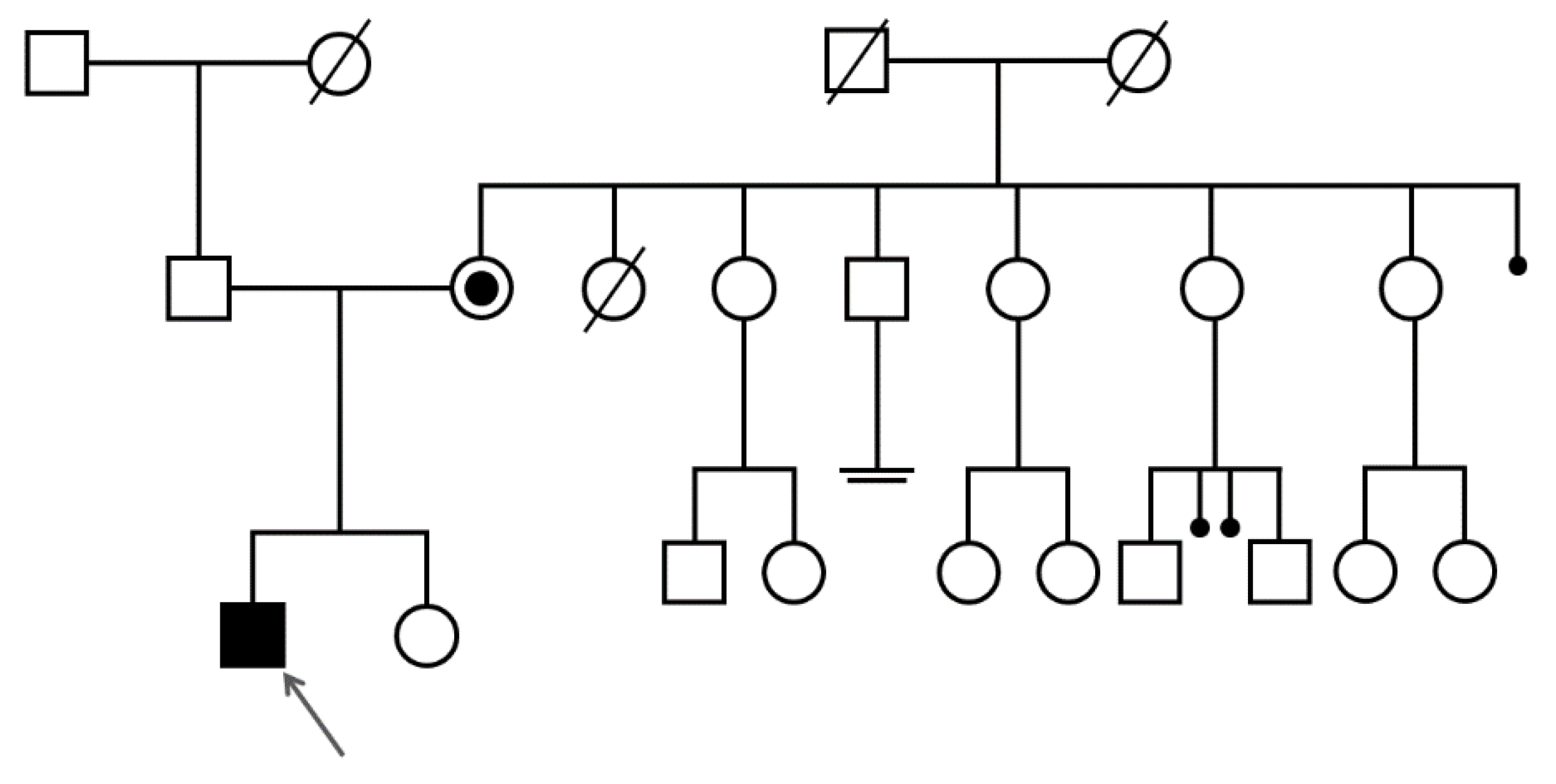
2.1. Case Report
2.2. Molecular Genetic Studies
2.2.1. Metabolic Myopathies Panel
2.2.2. Clinical and Methodological Validation
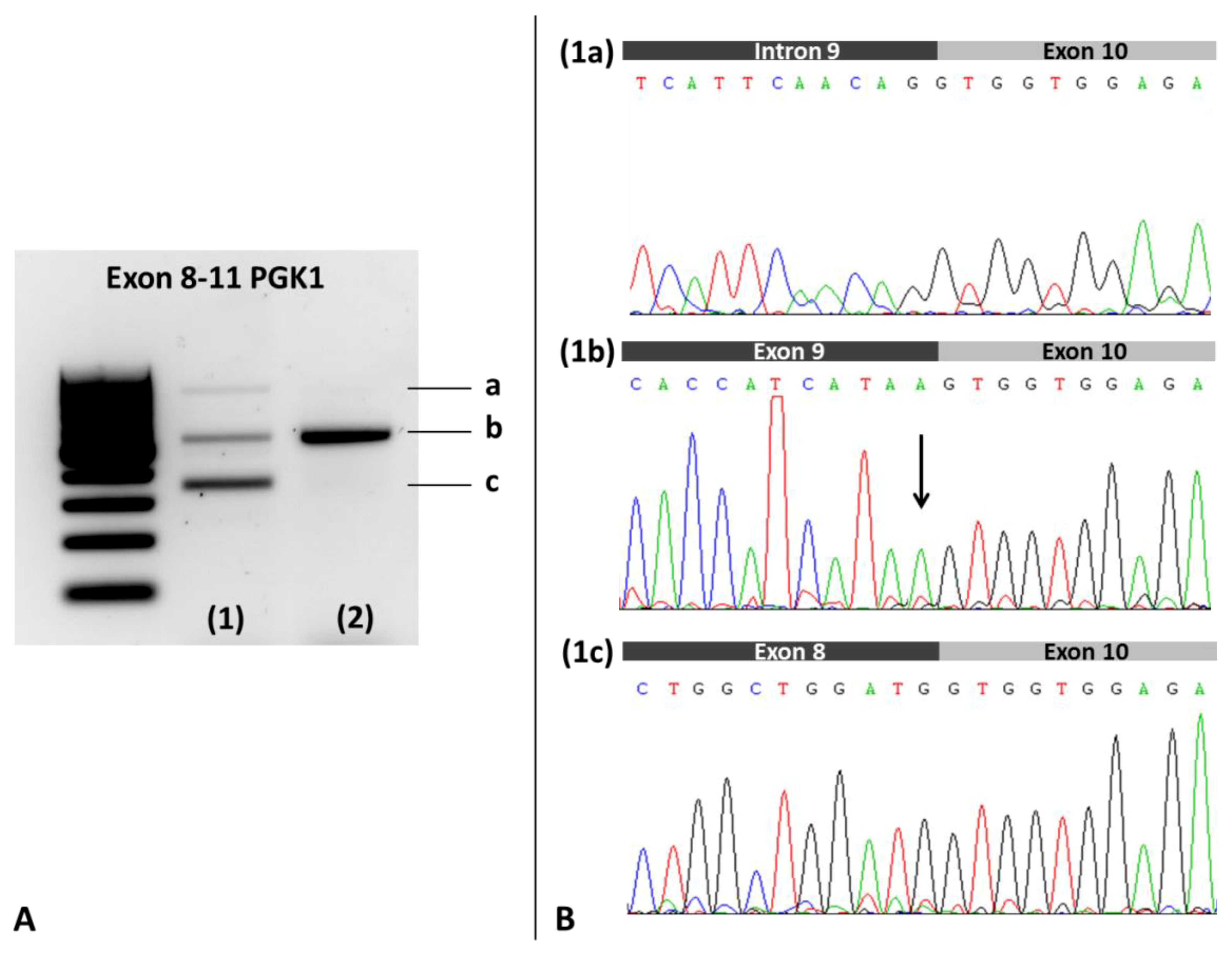
2.2.3. cDNA Analysis of PGK1
2.3. Enzyme Activity of PGK1
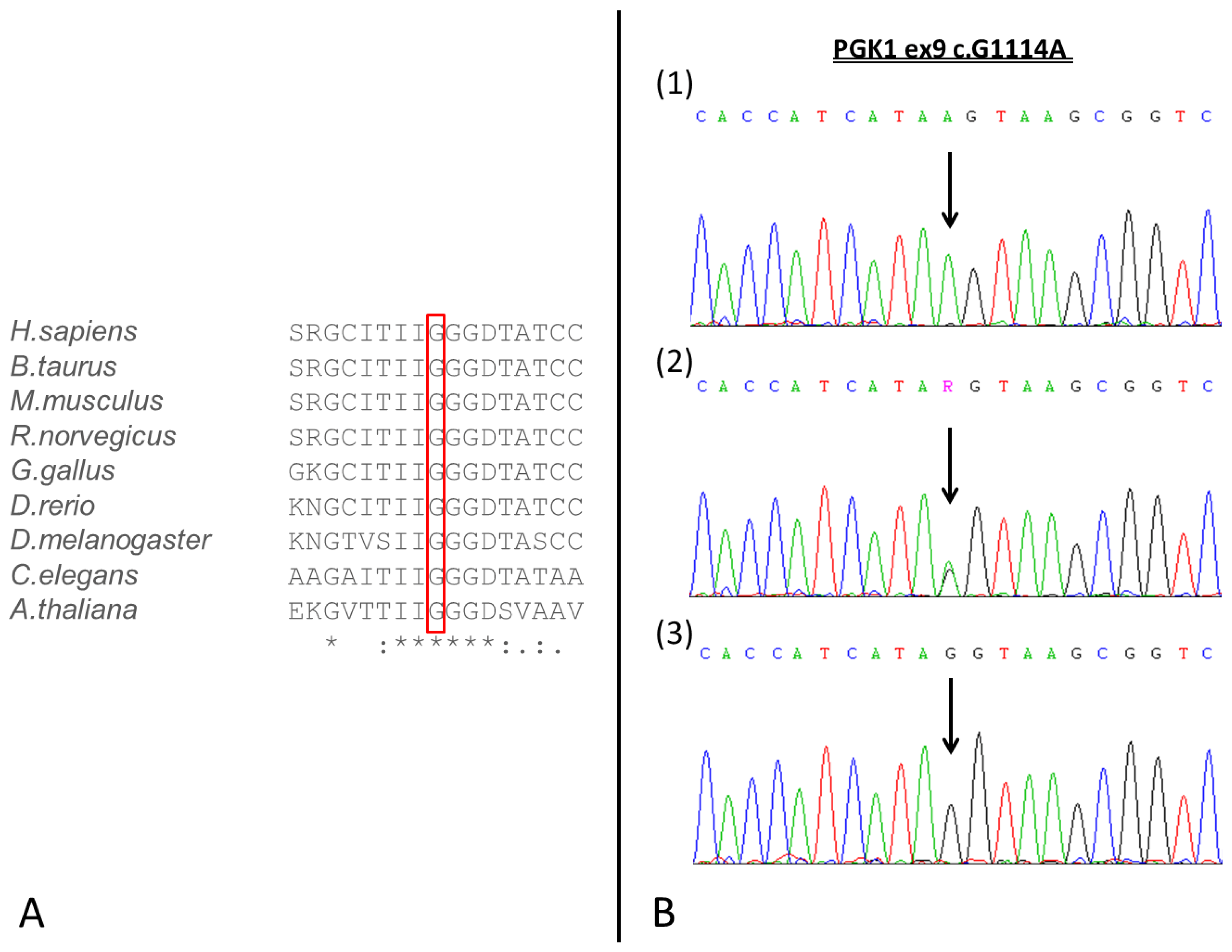
3. Results
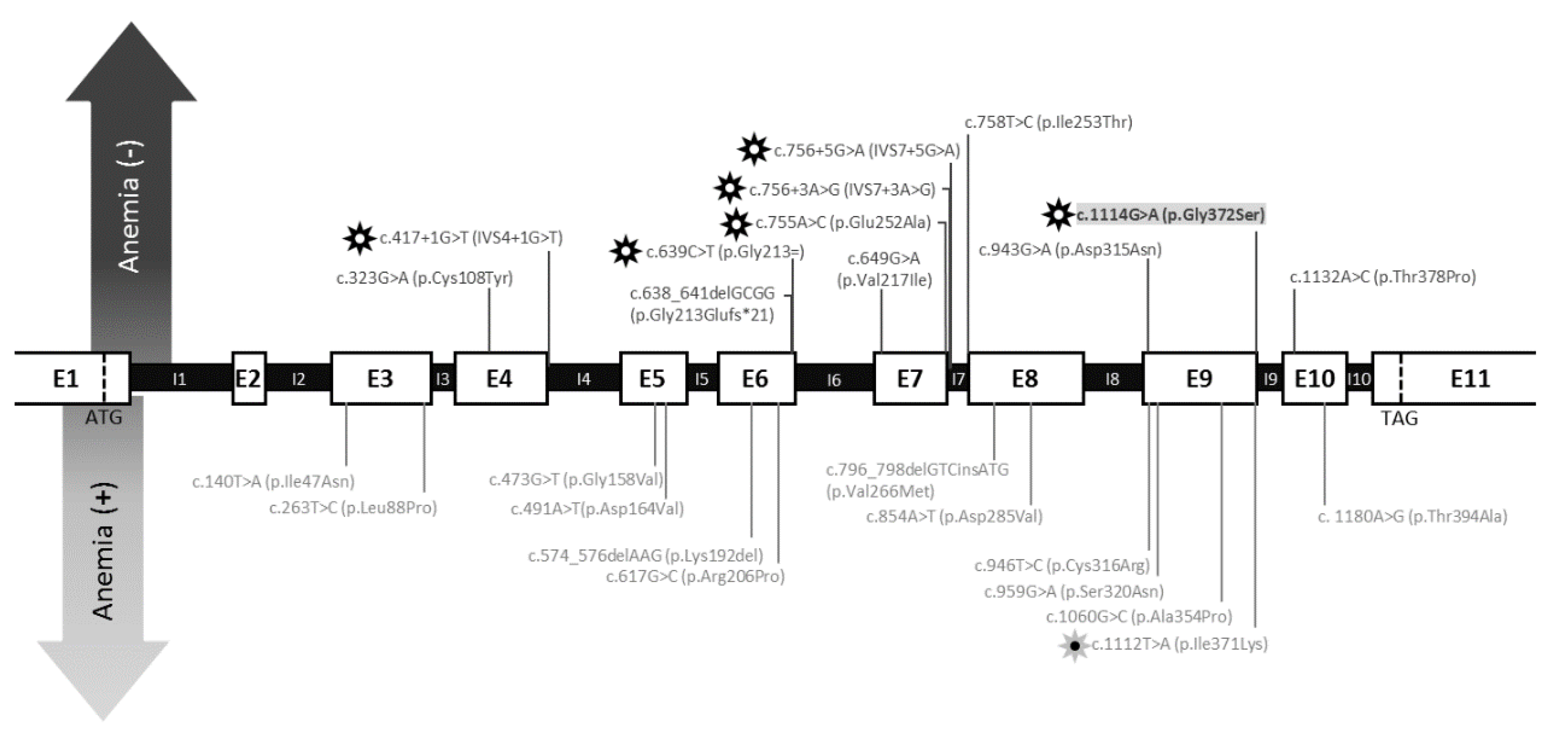
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beutler, E. PGK deficiency. Br. J. Haematol. 2007, 136, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Keitt, A.S. Pyruvate kinase deficiency and related disorders of red cell glycolysis. Am. J. Med. 1966, 41, 762–785. [Google Scholar] [CrossRef]
- Jindal, H.K.; Vishwanatha, J.K. Functional identity of a primer recognition protein as phosphoglycerate kinase. J. Biol. Chem. 1990, 265, 6540–6543. [Google Scholar] [PubMed]
- Lay, A.J.; Jiang, X.M.; Kisker, O.; Flynn, E.; Underwood, A.; Condron, R.; Hogg, P.J. Phosphoglycerate kinase acts in tumour angiogenesis as a disulphide reductase. Nature 2000, 408, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Shichijo, S.; Azuma, K.; Komatsu, N.; Ito, M.; Maeda, Y.; Ishihara, Y.; Itoh, K. Two proliferation-related proteins, TYMS and PGK1, could be new cytotoxic T lymphocyte-directed tumor-associated antigens of HLA-A2+ colon cancer. Clin. Cancer Res. 2004, 10, 5828–5836. [Google Scholar] [CrossRef]
- Krishnan, P.; Liou, J.Y.; Cheng, Y.C. Phosphorylation of pyrimidine L-deoxynucleoside analog diphosphates. Kinetics of phosphorylation and dephosphorylation of nucleoside analog diphosphates and triphosphates by 3-phosphoglycerate kinase. J. Biol. Chem. 2002, 277, 31593–31600. [Google Scholar] [CrossRef] [PubMed]
- Mathe, C.; Gosselin, G. L-nucleoside enantiomers as antivirals drugs: A mini-review. Antivir. Res. 2006, 71, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Chiarelli, L.R.; Morera, S.M.; Bianchi, P.; Fermo, E.; Zanella, A.; Galizzi, A.; Valentini, G. Molecular insights on pathogenic effects of mutations causing phosphoglycerate kinase deficiency. PLoS ONE 2012, 7, e32065. [Google Scholar] [CrossRef]
- Ramirez-Bajo, M.J.; Repiso, A.; la Ossa, P.P.; Banon-Maneus, E.; de Atauri, P.; Climent, F.; Corrons, J.L.; Cascante, M.; Carreras, J. Enzymatic and metabolic characterization of the phosphoglycerate kinase deficiency associated with chronic hemolytic anemia caused by the PGK-Barcelona mutation. Blood Cells Mol. Dis. 2011, 46, 206–211. [Google Scholar] [CrossRef]
- DiMauro, S.; Spiegel, R. Progress and problems in muscle glycogenoses. Acta Myol. 2011, 30, 96–102. [Google Scholar]
- Fermo, E.; Bianchi, P.; Chiarelli, L.R.; Maggi, M.; Mandara, G.M.; Vercellati, C.; Marcello, A.P.; Barcellini, W.; Cortelezzi, A.; Valentini, G.; et al. A new variant of phosphoglycerate kinase deficiency (p.I371K) with multiple tissue involvement: Molecular and functional characterization. Mol. Genet. Metab. 2012, 106, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Kraus, A.P.; Langston, M.F., Jr.; Lynch, B.L. Red cell phosphoglycerate kinase deficiency. A new cause of non-spherocytic hemolytic anemia. Biochem. Biophys. Res. Commun. 1968, 30, 173–177. [Google Scholar] [CrossRef]
- Tonin, P.; Shanske, S.; Miranda, A.F.; Brownell, A.K.; Wyse, J.P.; Tsujino, S.; DiMauro, S. Phosphoglycerate kinase deficiency: Biochemical and molecular genetic studies in a new myopathic variant (PGK Alberta). Neurology 1993, 43, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Spanu, C.; Oltean, S. Familial phosphoglycerate kinase deficiency associated with rhabdomyolysis and acute renal failure: Abnormality in mRNA splicing? Nephrol. Dial. Transplant. 2003, 18, 445–446. [Google Scholar] [CrossRef] [PubMed]
- Krietsch, W.K.; Krietsch, H.; Kaiser, W.; Dunnwald, M.; Kuntz, G.W.; Duhm, J.; Bucher, T. Hereditary deficiency of phosphoglycerate kinase: A new variant in erythrocytes and leucocytes, not associated with haemolytic anaemia. Eur. J. Clin. Investig. 1977, 7, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Krietsch, W.K.; Yoshida, A. A single amino acid substitution (Asp leads to Asn) in a phosphoglycerate kinase variant (PGK Munchen) associated with enzyme deficiency. J. Biol. Chem. 1980, 255, 6421–6423. [Google Scholar] [PubMed]
- Chen, S.H.; Malcolm, L.A.; Yoshida, A.; Giblett, E.R. Phosphoglycerate kinase: An X-linked polymorphism in man. Am. J. Hum. Genet. 1971, 23, 87–91. [Google Scholar] [PubMed]
- Fujii, H.; Chen, S.H.; Akatsuka, J.; Miwa, S.; Yoshida, A. Use of cultured lymphoblastoid cells for the study of abnormal enzymes: Molecular abnormality of a phosphoglycerate kinase variant associated with hemolytic anemia. Proc. Natl. Acad. Sci. USA 1981, 78, 2587–2590. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, R.; Gomez, E.A.; Akman, H.O.; Krishna, S.; Horovitz, Y.; DiMauro, S. Myopathic form of phosphoglycerate kinase (PGK) deficiency: A new case and pathogenic considerations. Neuromuscul. Disord. 2009, 19, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Noel, N.; Flanagan, J.M.; Ramirez Bajo, M.J.; Kalko, S.G.; Manu Mdel, M.; Garcia Fuster, J.L.; Perez de la Ossa, P.; Carreras, J.; Beutler, E.; Vives Corrons, J.L. Two new phosphoglycerate kinase mutations associated with chronic haemolytic anaemia and neurological dysfunction in two patients from Spain. Br. J. Haematol. 2006, 132, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Miwa, S.; Nakashima, K.; Oda, S.; Ogawa, H.; Nagafuji, H. Phosphoglycerate kinase (PKG) deficiency hereditary nonspherocytic hemolytic anemia: Report of a case found in a Japanese family. Nihon Ketsueki Gakkai Zasshi 1972, 35, 511–514. [Google Scholar] [PubMed]
- Tani, K.; Takizawa, T.; Yoshida, A. Normal mRNA content in a phosphoglycerate kinase variant with severe enzyme deficiency. Am. J. Hum. Genet. 1985, 37, 931–937. [Google Scholar] [PubMed]
- Maeda, M.; Yoshida, A. Molecular defect of a phosphoglycerate kinase variant (PGK-Matsue) associated with hemolytic anemia: Leu----Pro substitution caused by T/A----C/G transition in exon 3. Blood 1991, 77, 1348–1352. [Google Scholar] [PubMed]
- Echaniz-Laguna, A.; Nadjar, Y.; Behin, A.; Biancalana, V.; Piraud, M.; Malfatti, E.; Laforet, P. Phosphoglycerate kinase deficiency: A nationwide multicenter retrospective study. J. Inherit. Metab. Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Tsujino, S.; Tonin, P.; Shanske, S.; Nohria, V.; Boustany, R.M.; Lewis, D.; Chen, Y.T.; DiMauro, S. A splice junction mutation in a new myopathic variant of phosphoglycerate kinase deficiency (PGK North Carolina). Ann. Neurol. 1994, 35, 349–353. [Google Scholar] [CrossRef]
- Fujii, H.; Kanno, H.; Hirono, A.; Shiomura, T.; Miwa, S. A single amino acid substitution (157 Gly----Val) in a phosphoglycerate kinase variant (PGK Shizuoka) associated with chronic hemolysis and myoglobinuria. Blood 1992, 79, 1582–1585. [Google Scholar] [Green Version]
- Valentine, W.N.; Hsieh, H.S.; Paglia, D.E.; Anderson, H.M.; Baughan, M.A.; Jaffe, E.R.; Garson, O.M. Hereditary hemolytic anemia associated with phosphoglycerate kinase deficiency in erythrocytes and leukocytes. A probable X-chromosome-linked syndrome. N. Engl. J. Med. 1969, 280, 528–534. [Google Scholar] [CrossRef]
- Cohen-Solal, M.; Valentin, C.; Plassa, F.; Guillemin, G.; Danze, F.; Jaisson, F.; Rosa, R. Identification of new mutations in two phosphoglycerate kinase (PGK) variants expressing different clinical syndromes: PGK Creteil and PGK Amiens. Blood 1994, 84, 898–903. [Google Scholar] [Green Version]
- Turner, G.; Fletcher, J.; Elber, J.; Yanagawa, Y.; Dave, V.; Yoshida, A. Molecular defect of a phosphoglycerate kinase variant associated with haemolytic anaemia and neurological disorders in a large kindred. Br. J. Haematol. 1995, 91, 60–65. [Google Scholar] [CrossRef]
- Flanagan, J.M.; Rhodes, M.; Wilson, M.; Beutler, E. The identification of a recurrent phosphoglycerate kinase mutation associated with chronic haemolytic anaemia and neurological dysfunction in a family from USA. Br. J. Haematol. 2006, 134, 233–237. [Google Scholar] [CrossRef]
- Mohd Khialdin, S.; Grigg, J.; Rowe, N.; Crofts, S.; Wilson, M.; Troedson, C. A negative waveform in the scotopic response in a patient with phosphoglycerate kinase deficiency: A visual electrophysiology report. Doc. Ophthalmol. 2015, 131, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Twele, T.W.; Dave, V.; Beutler, E. Molecular abnormality of a phosphoglycerate kinase variant (PGK-Alabama). Blood Cells Mol. Dis. 1995, 21, 179–181. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Yoshida, A. Molecular abnormality of phosphoglycerate kinase-Uppsala associated with chronic nonspherocytic hemolytic anemia. Proc. Natl. Acad. Sci. USA 1980, 77, 5461–5465. [Google Scholar] [CrossRef] [PubMed]
- Hjelm, M.; Wadam, B.; Yoshida, A. A phosphoglycerate kinase variant, PGK Uppsala, associated with hemolytic anemia. J. Lab. Clin. Med. 1980, 96, 1015–1021. [Google Scholar] [PubMed]
- Hamano, T.; Mutoh, T.; Sugie, H.; Koga, H.; Kuriyama, M. Phosphoglycerate kinase deficiency: An adult myopathic form with a novel mutation. Neurology 2000, 54, 1188–1190. [Google Scholar] [CrossRef] [PubMed]
- Aasly, J.; van Diggelen, O.P.; Boer, A.M.; Bronstad, G. Phosphoglycerate kinase deficiency in two brothers with McArdle-like clinical symptoms. Eur. J. Neurol. 2000, 7, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Svaasand, E.K.; Aasly, J.; Landsem, V.M.; Klungland, H. Altered expression of PGK1 in a family with phosphoglycerate kinase deficiency. Muscle Nerve 2007, 36, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Matsumaru, S.; Oguni, H.; Ogura, H.; Shimojima, K.; Nagata, S.; Kanno, H.; Yamamoto, T. A novel PGK1 mutation associated with neurological dysfunction and the absence of episodes of hemolytic anemia or myoglobinuria. Intractable Rare Dis. Res. 2017, 6, 132–136. [Google Scholar] [CrossRef] [Green Version]
- Ookawara, T.; Dave, V.; Willems, P.; Martin, J.J.; de Barsy, T.; Matthys, E.; Yoshida, A. Retarded and aberrant splicings caused by single exon mutation in a phosphoglycerate kinase variant. Arch. Biochem. Biophys. 1996, 327, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Coppens, S.; Koralkova, P.; Aeby, A.; Mojzikova, R.; Deconinck, N.; Kadhim, H.; van Wijk, R. Recurrent episodes of myoglobinuria, mental retardation and seizures but no hemolysis in two brothers with phosphoglycerate kinase deficiency. Neuromuscul. Disord. 2016, 26, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, K.; Takahashi, Y.; Miyajima, H. Intronic mutation in the PGK1 gene may cause recurrent myoglobinuria by aberrant splicing. Neurology 2006, 66, 925–927. [Google Scholar] [CrossRef] [PubMed]
- Sugie, H.; Sugie, Y.; Ito, M.; Fukuda, T. A novel missense mutation (837T-->C) in the phosphoglycerate kinase gene of a patient with a myopathic form of phosphoglycerate kinase deficiency. J. Child Neurol. 1998, 13, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Valentin, C.; Birgens, H.; Craescu, C.T.; Brodum-Nielsen, K.; Cohen-Solal, M. A phosphoglycerate kinase mutant (PGK Herlev; D285V) in a Danish patient with isolated chronic hemolytic anemia: Mechanism of mutation and structure-function relationships. Hum. Mutat. 1998, 12, 280–287. [Google Scholar] [CrossRef]
- Maeda, M.; Bawle, E.V.; Kulkarni, R.; Beutler, E.; Yoshida, A. Molecular abnormalities of a phosphoglycerate kinase variant generated by spontaneous mutation. Blood 1992, 79, 2759–2762. [Google Scholar] [PubMed]
- Morimoto, A.; Ueda, I.; Hirashima, Y.; Sawai, Y.; Usuku, T.; Kano, G.; Kuriyama, K.; Todo, S.; Sugimoto, T.; Kanno, H.; et al. A novel missense mutation (1060G --> C) in the phosphoglycerate kinase gene in a Japanese boy with chronic haemolytic anaemia, developmental delay and rhabdomyolysis. Br. J. Haematol. 2003, 122, 1009–1013. [Google Scholar] [CrossRef]
- Tamai, M.; Kawano, T.; Saito, R.; Sakurai, K.; Saito, Y.; Yamada, H.; Ida, H.; Akiyama, M. Phosphoglycerate kinase deficiency due to a novel mutation (c. 1180A>G) manifesting as chronic hemolytic anemia in a Japanese boy. Int. J. Hematol. 2014, 100, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef]
- Beutler, E.; Blume, K.G.; Kaplan, J.C.; Lohr, G.W.; Ramot, B.; Valentine, W.N. International Committee for Standardization in Haematology: Recommended methods for red-cell enzyme analysis. Br. J. Haematol. 1977, 35, 331–340. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. ClinVar; Variation ID 167467. Available online: https://preview.ncbi.nlm.nih.gov/clinvar/variation/167467 (accessed on 9 August 2019).
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef]
- Ward, A.J.; Cooper, T.A. The pathobiology of splicing. J. Pathol. 2010, 220, 152–163. [Google Scholar] [CrossRef]
- Wang, E.T.; Sandberg, R.; Luo, S.; Khrebtukova, I.; Zhang, L.; Mayr, C.; Kingsmore, S.F.; Schroth, G.P.; Burge, C.B. Alternative isoform regulation in human tissue transcriptomes. Nature 2008, 456, 470–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pey, A.L.; Maggi, M.; Valentini, G. Insights into human phosphoglycerate kinase 1 deficiency as a conformational disease from biochemical, biophysical, and in vitro expression analyses. J. Inherit. Metab. Dis. 2014, 37, 909–916. [Google Scholar] [CrossRef] [PubMed]
| Variant | Age of Diagnosis | Age of Last Review | PGK Residual Activity (%) | Nucleotide Change | Amino Acid Change | Symptoms | Comments on The Studies: Phenotype or Methodology | |||
|---|---|---|---|---|---|---|---|---|---|---|
| RBC | Muscle | H | M | C N S | ||||||
| Barcelona [9,20] | 3 | 7 | 10.4/20 | NA | c.140T > A | p.Ile47Asn | + | - | + | Neonatal anemia (Hb: 7.3g/dL) and progressive neurological impairment leading to mental retardation (7 years). The decrease in PGK activity is more closely related to a loss of enzyme stability than to a decrease in catalytic function. |
| Matsue [21,22,23] | 9 | 9 † | 5/10 | NA | c.263T > C | p.Leu88Pro | + | - | + | Higher Michaelis-Menten constant (Km) for all substrates, particularly for ATP and 1,3-BPG |
| [24] | 32 | NA | 1.9 | 2.6 | c.323G > A | p.Cys108Tyr | - | + | + | Chronic axonal sensorimotor polyneuropathy and mental retardation, microcephaly and ophthalmoplegia. |
| North Carolina [25] | 12 | NA | 2.7 | 2 | c.417 + 1G > T | IVS4 + 1G > T | - | + | + | Mild intellectual delay with attention-deficit disorder. |
| Shizuoka [26] | 27 | NA | 0.7 | NA | c.473G > T | p.Gly158Val | -/+ | + | - | No neonatal hemolytic anemia (Hb: 12.8 g/dL from diagnosis). |
| Amiens/New York [27,28,29,30,31] | 0/3 | 23 | <5 | c.491A > T | p.Asp164Val | + | - | + | Seizures associated with hemolytic anemia. Retinal dystrophy. The most deleterious variant was at the protein level. | |
| Alabama [32] | 37 | NA | 4 | NA | c.574_576delAAG | p.Lys192del | -/+ | - | - | School teacher who has been in excellent general health. At age 20, he had a self-limited febrile illness associated with a high bilirubin level. |
| Uppsala [33,34] | 26 | 31 | 10 | NA | c.617G > C | p.Arg206Pro | + | - | + | Anemia and jaundice since 4 months after birth. Lower affinity for substrates, ATP and 1,3-BPG. Significant accumulation of 2,3-BPG and 2-phosphoglycerate (2-PG). |
| Fukui [35] | 36 | NA | 5.6 | 2.9 | c.638_641delGCGG | p.Gly213Glufs* 21 | - | + | - | Complementary DNA sequence of a reverse transcriptase PCR product of exon 6 in the PGK1 gene from leukocytes. No more information about methodology. |
| [36,37] | 16/21 | NA | 4–5 | 2–3 | c.639C > T | p.Gly213 = | - | + | - | Two brothers with normal intelligence. |
| [38] | 16 | NA | 78–91 | NA | c.649G > A | p.Val217Ile | - | + | + | Polymorphism: Male with intellectual disability, epileptic seizures, mild cerebral and cerebellar atrophy and peculiar episodes of muscle weakness of unknown etiology. |
| Antwerp [39] | 25 | NA | 5.6 | 8 | c.755A > C | p.Glu252Ala | - | + | - | Slightly decreased hemoglobin (13.2 g/100 mL) (normal: 13.3). |
| [40] | 14/16 | NA | <15 | NA | c.756 + 3A > G | IVS7 + 3A > G | - | + | + | Mild intellectual deficiency. At 8 years old, after 20 min in a swimming pool, one of them had myoglobinuria. |
| Fukuroi [41] | 33 | NA | 13.6 | 8.9 | c.756 + 5G > A | IVS7 + 5G > A | - | + | + | A 33-year-old man first had severe muscle pain and myoglobinuria after a short run at age 20 years. No history of epileptic attacks despite small spike waves on electroencephalogram (EEG). |
| Hamamatsu [42] | 11 | NA | 8.2 | 4.4 | c.758T > C | p.Ile253Thr | - | + | + | PGK1 mRNA was reverse transcribed and amplified in three fragments and subcloned and sequenced. |
| Tokyo [18] | NA | NA | 10 | NA | c.796_798delGTCinsATG | p.Val266Met | + | - | + | Lower specific activity and increased thermal instability. |
| München [15,16] | polymorphism | c.802G > A | p.Asp268Val | - | - | - | Associated with enzyme deficiency and heat instability | |||
| Herlev [43] | 69 | 72 † | 49 | NA | c.854A > T | p.Asp285Val | -/+ | - | - | Pronounced reticulocytosis (10–45%). Approximately 90% of the mutated nucleotide T, approximately 10% of normal A nucleotide. Mosaicism? Somatic mutation? |
| Creteil [28] | 31 | NA | 3 | 25 | c.943G > A | p.Asp315Asn | - | + | - | Since his childhood, he presented several symptoms during physical exercises, notably rhabdomyolysis crises, |
| Michigan [44] | 9 | 14 | 10 | NA | c.946T > C | p.Cys316Arg | -/+ | - | + | Compensated hemolytic anemia with occasional hemolysis crises (infections). The variant was more labile than the normal enzyme. De novo variant. |
| Murcia [20] | 6 | 7† | 49.2 | NA | c.959G > A | p.Ser320Asn | + | - | + | Required transfusions from birth every 3–4 weeks. Cortical and subcortical atrophy. |
| PGKII/Samoa [17,18] | polymorphism | c.1055C > A | p.Thr352Asn | - | - | - | Electrophoretic variant not associated with enzyme deficiency. | |||
| Kyoto [45] | 3 | 3.2 | 6.3 | NA | c.1060G > C | p.Ala354Pro | + | + | + | Anemia and jaundice at birth. Respiratory infection-associated hemolytic crisis and rhabdomyolysis during early infancy. |
| [46] | 4 | 25 | 14.6 | NA | c.1112T > A | p.Ile371Lys | + | + | + | At the age of 25 years, he shows generalized myopathy, intelligence quotient (IQ)52 and cerebellar atrophy. |
| Present study | 38 | 40 | 19.7 | NA | c.1114G > A | p.Gly372Ser | - | + | + | Mild intellectual deficiency. Progressive exercise intolerance, cramps and sporadic episodes of rhabdomyolysis. |
| Afula [19] | 18/25 | NA | 2 | 0.9/1.1 | c.1132A > C | p.Thr378Pro | - | + | - | (18) For 7 years, he has experienced recurrent episodes of muscle cramps, myalgia and pigmenturia after intense exercise.(25) He had severe parkinsonism that was responsive to levodopa. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Solaesa, V.; Serrano-Lorenzo, P.; Ramos-Arroyo, M.A.; Blázquez, A.; Pagola-Lorz, I.; Artigas-López, M.; Arenas, J.; Martín, M.A.; Jericó-Pascual, I. A Novel Missense Variant Associated with A Splicing Defect in A Myopathic Form of PGK1 Deficiency in The Spanish Population. Genes 2019, 10, 785. https://doi.org/10.3390/genes10100785
Garcia-Solaesa V, Serrano-Lorenzo P, Ramos-Arroyo MA, Blázquez A, Pagola-Lorz I, Artigas-López M, Arenas J, Martín MA, Jericó-Pascual I. A Novel Missense Variant Associated with A Splicing Defect in A Myopathic Form of PGK1 Deficiency in The Spanish Population. Genes. 2019; 10(10):785. https://doi.org/10.3390/genes10100785
Chicago/Turabian StyleGarcia-Solaesa, Virginia, Pablo Serrano-Lorenzo, Maria Antonia Ramos-Arroyo, Alberto Blázquez, Inmaculada Pagola-Lorz, Mercè Artigas-López, Joaquín Arenas, Miguel A. Martín, and Ivonne Jericó-Pascual. 2019. "A Novel Missense Variant Associated with A Splicing Defect in A Myopathic Form of PGK1 Deficiency in The Spanish Population" Genes 10, no. 10: 785. https://doi.org/10.3390/genes10100785
APA StyleGarcia-Solaesa, V., Serrano-Lorenzo, P., Ramos-Arroyo, M. A., Blázquez, A., Pagola-Lorz, I., Artigas-López, M., Arenas, J., Martín, M. A., & Jericó-Pascual, I. (2019). A Novel Missense Variant Associated with A Splicing Defect in A Myopathic Form of PGK1 Deficiency in The Spanish Population. Genes, 10(10), 785. https://doi.org/10.3390/genes10100785







