Molecular Therapies for Choroideremia
Abstract
1. Introduction
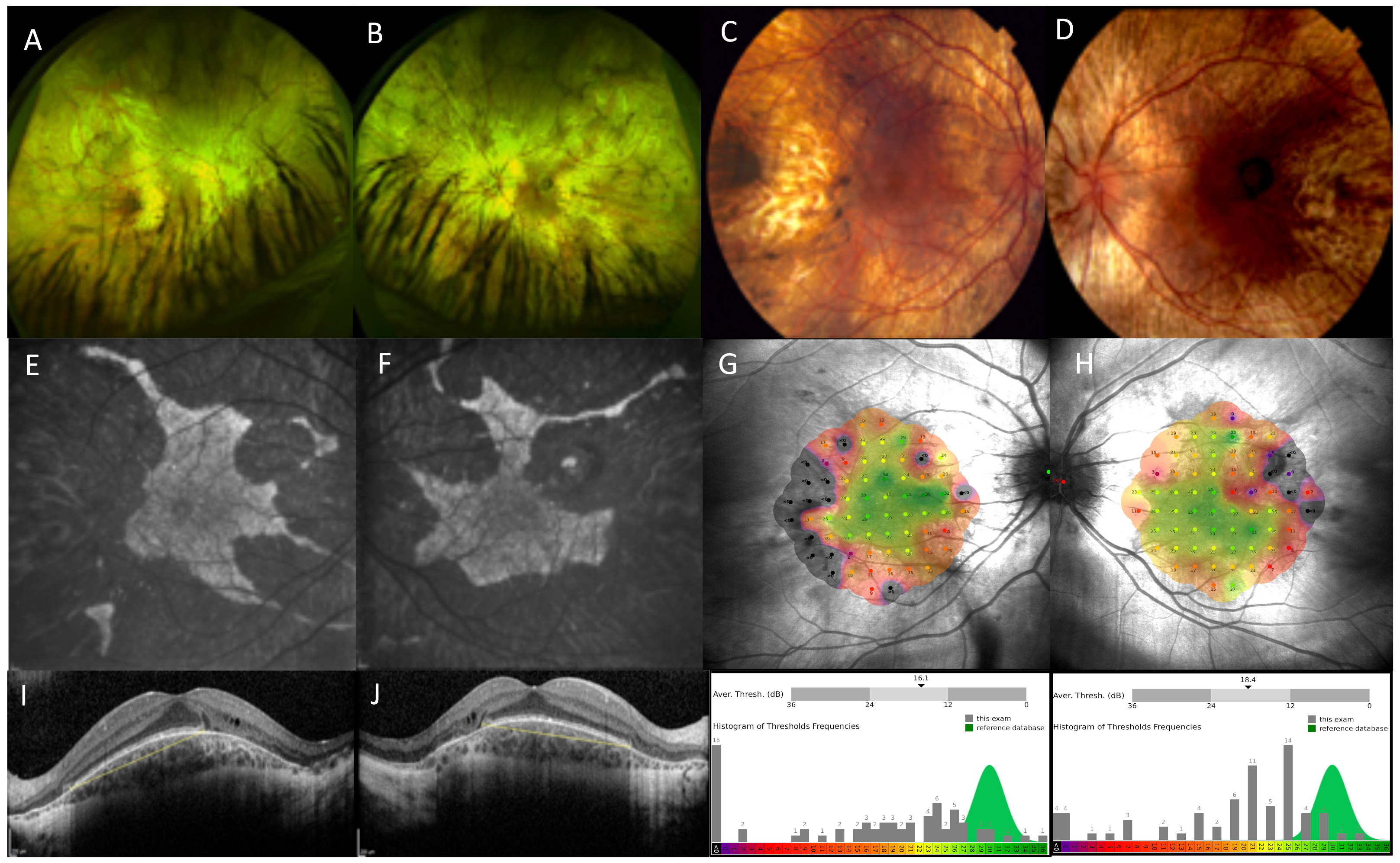
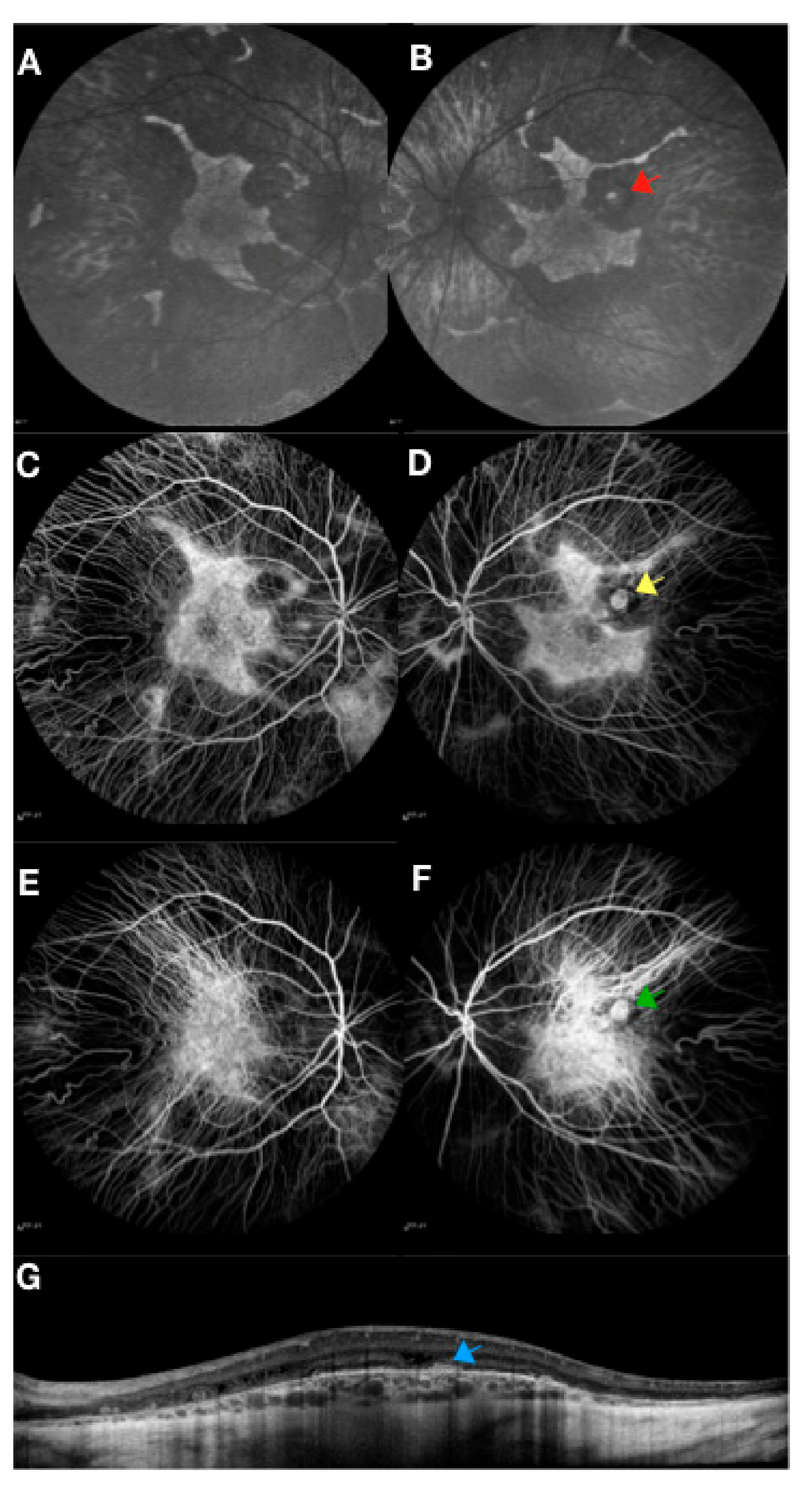
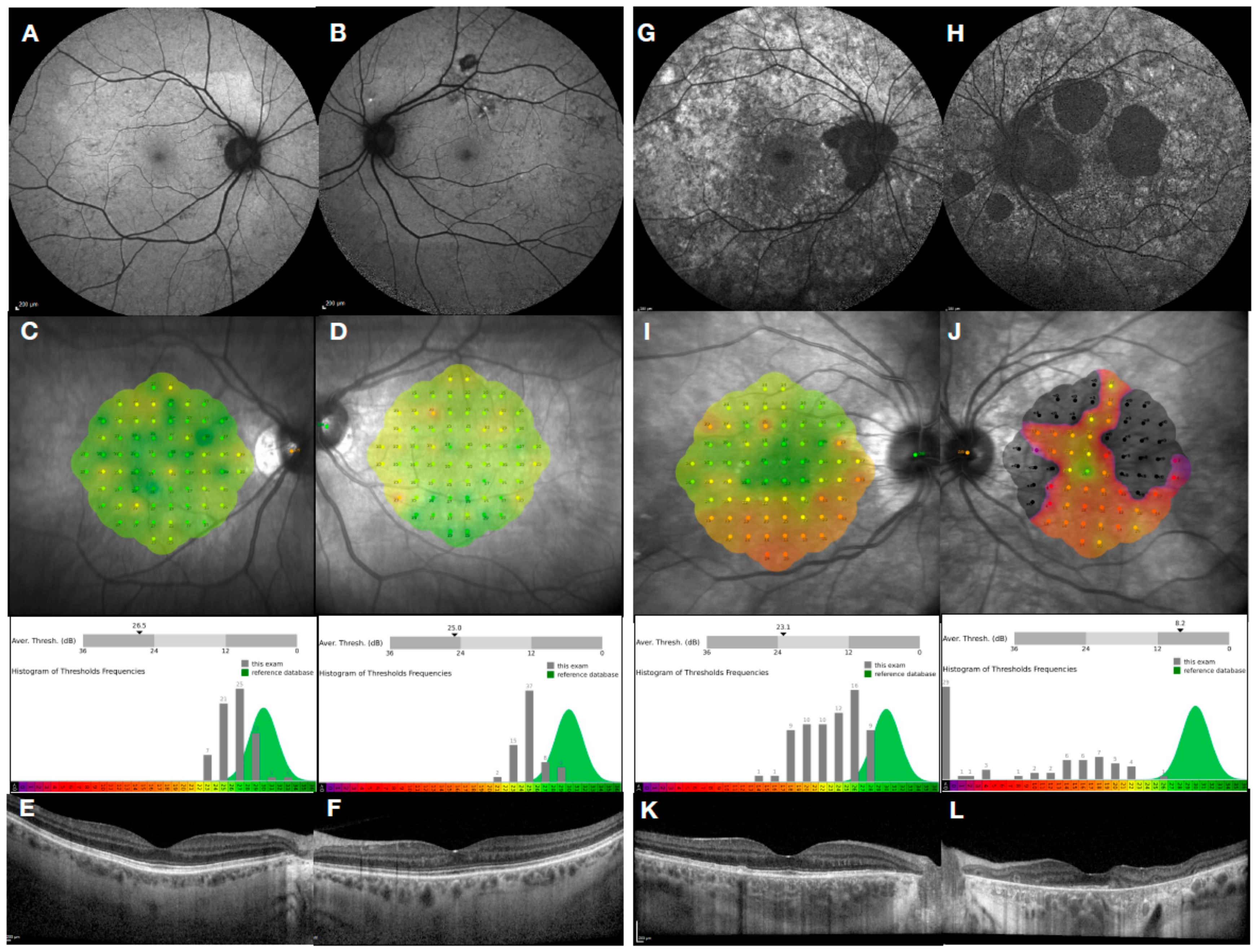
2. Choroideremia Phenotype
3. Choroideremia Genotype
3.1. Genotype–Phenotype Correlation in Choroideremia
3.2. Molecular Mechanisms of Choroideremia
3.3. Gene Therapy for Choroideremia
4. Should We Treat Female Carriers in the Future?
5. Alternative Therapies
6. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Cremers, F.P.; van de Pol, D.J.; van Kerkhoff, L.P.; Wieringa, B.; Ropers, H.H. Cloning of a gene that is rearranged in patients with choroideraemia. Nature 1990, 347, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Seabra, M.C.; Brown, M.S.; Goldstein, J.L. Retinal degeneration in choroideremia: Deficiency of rab geranylgeranyl transferase. Science 1993, 259, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, J.A.; Schwartz, M.; van Bokhoven, H.; Van de Pol, T.J.R.; Bogerd, L.; Pinckers, A.J.L.G.; Bleeker-Wagemakers, E.M.; Pawlowitzki, I.H.; Rüther, K.; Ropers, H.H.; et al. Molecular basis of choroideremia (CHM): Mutations involving the Rab escort protein-1 (REP-1) gene. Hum. Mutat. 1997, 9, 110–117. [Google Scholar] [CrossRef]
- Tolmachova, T.; Anders, R.; Abrink, M.; Bugeon, L.; Dallman, M.J.; Futter, C.E.; Ramalho, J.S.; Tonagel, F.; Tanimoto, N.; Seeliger, M.W.; et al. Independent degeneration of photoreceptors and retinal pigment epithelium in conditional knockout mouse models of choroideremia. J. Clin. Investig. 2006, 116, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Patrício, M.I.; Barnard, A.R.; Xue, K.; MacLaren, R.E. Choroideremia: Molecular mechanisms and development of AAV gene therapy. Expert Opin. Biol. Ther. 2018, 18, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Aleman, T.S.; Han, G.; Serrano, L.W.; Fuerst, N.M.; Charlson, E.S.; Pearson, D.J.; Chung, D.C.; Traband, A.; Pan, W.; Ying, G.S.; et al. Natural history of the central structural abnormalities in choroideremia: A Prospective Cross-Sectional Study. Ophthalmology 2017, 124, 359–373. [Google Scholar] [CrossRef] [PubMed]
- Hariri, A.H.; Velaga, S.B.; Girach, A.; Ip, M.S.; Le, P.V.; Lam, B.L.; Fischer, M.D.; Sankila, E.M.; Pennesi, M.E.; Holz, F.G.; et al. Measurement and reproducibility of preserved ellipsoid zone area and preserved retinal pigment epithelium area in eyes with choroideremia. Am. J. Ophthalmol. 2017, 179, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.W.; Johnson, R.D.; Williams, V.; Summerfelt, P.; Dubra, A.; Weinberg, D.V.; Stepien, K.E.; Fishman, G.A.; Carroll, J. Multimodal imaging of photoreceptor structure in choroideremia. PLoS ONE 2016, 11, e0167526. [Google Scholar] [CrossRef] [PubMed]
- MacLaren, R.E.; Groppe, M.; Barnard, A.R.; Cottriall, C.L.; Tolmachova, T.; Seymour, L.; Clark, K.R.; During, M.J.; Cremers, F.P.; Black, G.C.; et al. Retinal gene therapy in patients with choroideremia: Initial findings from a phase 1/2 clinical trial. Lancet 2014, 383, 1129–1137. [Google Scholar] [CrossRef]
- Edwards, T.L.; Jolly, J.K.; Groppe, M.; Barnard, A.R.; Cottriall, C.L.; Tolmachova, T.; Black, G.C.; Webster, A.R.; Lotery, A.J.; Holder, G.E.; et al. Visual Acuity after Retinal Gene Therapy for Choroideremia. N. Engl. J. Med. 2016, 374, 1996–1998. [Google Scholar] [CrossRef]
- Xue, K.; Jolly, J.K.; Barnard, A.R.; Rudenko, A.; Salvetti, A.P.; Patrício, M.I.; Edwards, T.L.; Groppe, M.; Orlans, H.O.; Tolmachova, T.; et al. Beneficial effects on vision in patients undergoing retinal gene therapy for choroideremia. Nat. Med. 2018, 24, 1507–1512. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, I.S.; Hoang, S.C.; Radziwon, A.; Binczyk, N.M.; Seabra, M.C.; MacLaren, R.E.; Somani, R.; Tennant, M.T.; MacDonald, I.M. Two-year results after aav2-mediated gene therapy for choroideremia: The Alberta experience. Am. J. Ophthalmol. 2018, 193, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Lam, B.L.; Davis, J.L.; Gregori, N.Z.; MacLaren, R.E.; Girach, A.; Verriotto, J.D.; Rodriguez, B.; Rosa, P.R.; Zhang, X.; Feuer, W.J. Choroideremia Gene Therapy Phase 2 Clinical Trial: 24-Month Results. Am. J. Ophthalmol. 2019, 197, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.D.; Ochakovski, G.A.; Beier, B.; Seitz, I.P.; Vaheb, Y.; Kortuem, C.; Reichel, F.F.; Kuehlewein, L.; Kahle, N.A.; Peters, T.; et al. Changes in retinal sensitivity after gene therapy in choroideremia. Retina 2018. [Google Scholar] [CrossRef] [PubMed]
- Jolly, J.K.; Xue, K.; Edwards, T.L.; Groppe, M.; MacLaren, R.E. Characterizing the natural history of visual function in choroideremia using microperimetry and multimodal retinal imaging. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5575–5583. [Google Scholar] [CrossRef] [PubMed]
- Hariri, A.H.; Ip, M.S.; Girach, A.; Lam, B.L.; Fischer, M.D.; Sankila, E.M.; Pennesi, M.E.; Holz, F.G.; Maclaren, R.E.; Birch, D.G.; et al. Macular spatial distribution of preserved autofluorescence in patients with choroideremia. For Natural History of the Progression of Choroideremia (NIGHT) Study Group. Br. J. Ophthalmol. 2019, 103, 933–937. [Google Scholar] [CrossRef] [PubMed]
- Patrício, M.I.; Barnard, A.R.; Cox, C.I.; Blue, C.; MacLaren, R.E. The Biological Activity of AAV Vectors for Choroideremia Gene Therapy Can Be Measured by In Vitro Prenylation of RAB6A. Mol. Ther. Methods Clin. Dev. 2018, 9, 288–295. [Google Scholar] [CrossRef]
- Syed, N.; Smith, J.E.; John, S.K.; Seabra, M.C.; Aguirre, G.D.; Milam, A.H. Evaluation of retinal photoreceptors and pigment epithelium in a female carrier of choroideremia. Ophthalmology 2001, 108, 711–720. [Google Scholar] [CrossRef]
- Morgan, J.I.; Han, G.; Klinman, E.; Maguire, W.M.; Chung, D.C.; Maguire, A.M.; Bennett, J. High-resolution adaptive optics retinal imaging of cellular structure in choroideremia. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6381–6397. [Google Scholar] [CrossRef]
- Xue, K.; Oldani, M.; Jolly, J.K.; Edwards, T.L.; Groppe, M.; Downes, S.M.; MacLaren, R.E. Correlation of Optical Coherence Tomography and Autofluorescence in the Outer Retina and Choroid of Patients with Choroideremia. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3674–3684. [Google Scholar] [CrossRef]
- Aylward, J.W.; Xue, K.; Patrício, M.I.; Jolly, J.K.; Wood, J.C.; Brett, J.; Jasani, K.M.; MacLaren, R.E. Retinal Degeneration in Choroideremia follows an Exponential Decay Function. Ophthalmology 2018, 125, 1122–1124. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, S.C.; O’Grady, A.; Fletcher, T.; O’Sullivan, J.; Hart-Holden, N.; Barton, S.J.; Hall, G.; Moore, A.T.; Webster, A.R.; Black, G.C. A clinical molecular genetic service for United Kingdom families with choroideraemia. Eur. J. Med. Genet. 2013, 56, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Freund, P.; Furgoch, M.; MacDonald, I. Genotype—Phenotype analysis of male subjects affected by choroideremia. Investig. Ophthalmol. Vis. Sci. 2013, 54, 1567. [Google Scholar]
- Freund, P.R.; Sergeev, Y.V.; MacDonald, I.M. Analysis of a large choroideremia dataset does not suggest a preference for inclusion of certain genotypes in future trials of gene therapy. Mol. Genet. Genomic Med. 2016, 4, 344–358. [Google Scholar] [CrossRef] [PubMed]
- Simunovic, M.P.; Jolly, J.K.; Xue, K.; Edwards, T.L.; Groppe, M.; Downes, S.M.; MacLaren, R.E. The Spectrum of CHM Gene Mutations in Choroideremia and Their Relationship to Clinical Phenotype. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6033–6039. [Google Scholar] [CrossRef] [PubMed]
- Carss, K.; Arno, G.; Erwood, M.; Stephens, J.; Sanchis-Juan, A.; Hull, S.; Megy, K.; Grozeva, D.; Dewhurst, E.; Malka, S.; et al. Comprehensive rare variant analysis via whole-genome sequencing to determine the molecular pathology of inherited retinal disease. Am. J. Hum. Genet. 2017, 100, 75–90. [Google Scholar] [CrossRef]
- Radziwon, A.; Arno, G.K.; Wheaton, D.; McDonagh, E.M.; Baple, E.L.; Webb-Jones, K.G.; Birch, D.; Webster, A.R.; MacDonald, I.M. Single-base substitutions in the CHM promoter as a cause of choroideremia. Hum. Mutat. 2017, 38, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.L.; Casey, P.J. Protein prenylation: Molecular mechanisms and functional consequences. Ann. Rev. Biochem. 1996, 65, 241–269. [Google Scholar] [CrossRef]
- Seabra, M.C.; Ho, Y.K.; Anant, J.S. Deficient geranylgeranylation of Ram/Rab27 in choroideremia. J. Biol. Chem. 1995, 270, 24420–24427. [Google Scholar] [CrossRef]
- Gordiyenko, N.V.; Fariss, R.N.; Zhi, C.; MacDonald, I.M. Silencing of the CHM gene alters phagocytic and secretory pathways in the retinal pigment epithelium. Investig. Ophthalmol. Vis. Sci. 2010, 51, 1143–1150. [Google Scholar] [CrossRef]
- Tolmachova, T.; Wavre-Shapton, S.T.; Barnard, A.R.; MacLaren, R.E.; Futter, C.E.; Seabra, M.C. Retinal pigment epithelium defects accelerate photoreceptor degeneration in cell type-specific knock-out mouse models of Choroideremia. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4913–4920. [Google Scholar] [CrossRef] [PubMed]
- Wavre-Shapton, S.T.; Tolmachova, T.; da Silva, M.L.; Futter, C.E.; Seabra, M.C. Conditional ablation of the choroideremia gene causes age-related changes in mouse retinal pigment epithelium. PLoS ONE 2013, 8, e57769. [Google Scholar] [CrossRef]
- Flannery, J.G.; Bird, A.C.; Farber, D.B.; Weleber, R.G.; Bok, D. A histopathologic study of a choroideremia carrier. Investig. Ophthalmol. Vis. Sci. 1990, 31, 229–236. [Google Scholar]
- Cremers, F.P.; Molloy, C.M.; van de Pol, D.J.; van den Hurk, J.A.; Bach, I.; Geurts van Kessel, A.H.; Ropers, H.H. An autosomal homologue of the choroideremia gene colocalizes with the Usher syndrome type II locus on the distal part of chromosome 1q. Hum. Mol. Genet. 1992, 1, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Cremers, F.P.M.; Armstrong, S.A.; Seabra, M.C.; Brown, M.S.; Goldstein, J.L. REP-2, a Rab escort protein encoded by the choroideremia-like gene. J. Biol. Chem. 1994, 269, 2111–2117. [Google Scholar] [PubMed]
- Anand, V.; Barral, D.C.; Zeng, Y.; Brunsmann, F.; Maguire, A.M.; Seabra, M.C.; Bennett, J. Gene therapy for choroideremia: In vitro rescue mediated by recombinant adenovirus. Vis. Res. 2003, 43, 919–926. [Google Scholar] [CrossRef]
- Tolmachova, T.; Tolmachov, O.E.; Wavre-Shapton, S.T.; Tracey-White, D.; Futter, C.E.; Seabra, M.C. CHM/REP1 cDNA delivery by lentiviral vectors provides functional expression of the transgene in the retinal pigment epithelium of choroideremia mice. J. Gene Med. 2012, 14, 158–168. [Google Scholar] [CrossRef]
- Zhang, A.Y.; Mysore, N.; Vali, H.; Koenekoop, J.; Cao, S.N.; Li, S.; Ren, H.; Keser, V.; Lopez-Solache, I.; Siddiqui, S.N.; et al. Choroideremia Is a Systemic Disease with Lymphocyte Crystals and Plasma Lipid and RBC Membrane Abnormalities. Investig. Ophthalmol. Vis. Sci. 2015, 56, 8158–8165. [Google Scholar] [CrossRef]
- Radziwon, A.; Cho, W.J.; Szkotak, A.; Suh, M.; MacDonald, I.M. Crystals and Fatty AcidAbnormalities Are Not Present in Circulating Cells from Choroideremia Patients. Investig. Ophthalmol. Vis. Sci. 2018, 59, 4464–4470. [Google Scholar] [CrossRef]
- Futter, C.E.; Ramalho, J.S.; Jaissle, G.B.; Seeliger, M.W.; Seabra, M.C. The role of Rab27a in the regulation of melanosome distribution within retinal pigment epithelial cells. Mol. Biol. Cell 2004, 15, 2264–2275. [Google Scholar] [CrossRef]
- Kwok, M.C.M.; Holopainen, J.M.; Molday, L.L.; Foster, L.J.; Molday, R.S. Proteomics of photoreceptor outer segments identifies a subset of SNARE and Rab proteins implicated in membrane vesicle trafficking and fusion. Mol. Cell Proteom. 2008, 7, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Deretic, D. Molecular complexes that direct rhodopsin transport to primary cilia. Prog. Retin. Eye Res. 2014, 38, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Larijani, B.; Hume, A.N.; Tarafder, A.K.; Seabra, M.C. Multiple factors contribute to inefficient prenylation of Rab27a in Rab Prenylation diseases. J. Biol. Chem. 2003, 278, 46798–46804. [Google Scholar] [CrossRef] [PubMed]
- Rak, A.; Pylypenko, O.; Niculae, A.; Pyatkov, K.; Goody, R.S.; Alexandrov, K. Structure of the Rab7: REP-1 Complex. Cell 2004, 117, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Köhnke, M.; Delon, C.; Hastie, M.L.; Nguyen, U.T.; Wu, Y.W.; Waldmann, H.; Goody, R.S.; Gorman, J.J.; Alexandrov, K. Rab GTPase prenylation hierarchy and its potential role in choroideremia disease. PLoS ONE 2013, 8, e81758. [Google Scholar] [CrossRef] [PubMed]
- Barnard, A.R.; Groppe, M.; MacLaren, R.E. Gene therapy for choroideremia using an adeno-associated viral (AAV) vector. Cold Spring Harb. Perspect. Med. 2014, 5, a017293. [Google Scholar] [CrossRef] [PubMed]
- Edwards, T.L.; Groppe, M.; Jolly, J.K.; Downes, S.M.; MacLaren, R.E. Correlation of retinal structure and function in choroideremia carriers. Ophthalmology 2015, 122, 1274–1276. [Google Scholar] [CrossRef]
- Diakatou, M.; Manes, G.; Bocquet, B.; Meunier, I.; Kalatzis, V. Genome Editing as a Treatment for the Most Prevalent Causative Genes of Autosomal Dominant Retinitis Pigmentosa. Int. J. Mol. Sci. 2019, 20, 2542. [Google Scholar] [CrossRef]
- Cideciyan, A.V.; Jacobson, S.G.; Beltran, W.A.; Sumaroka, A.; Swider, M.; Iwabe, S.; Roman, A.J.; Olivares, M.B.; Schwartz, S.B.; Komaromy, A.M.; et al. Human retinal gene therapy for Leber congenital amaurosis shows advancing retinal degeneration despite enduring visual improvement. Proc. Natl. Acad. Sci. USA 2013, 110, E517–E525. [Google Scholar] [CrossRef]
- Tolmachova, T.; Tolmachov, O.E.; Barnard, A.R.; de Silva, S.R.; Lipinski, D.M.; Walker, N.J.; Maclaren, R.E.; Seabra, M.C. Functional expression of Rab escort protein 1 following AAV2-mediated gene delivery in the retina of choroideremia mice and human cells ex vivo. J. Mol. Med. 2013, 91, 825–837. [Google Scholar] [CrossRef]
- Nagel-Wolfrum, K.; Möller, F.; Penner, I.; Baasov, T.; Wolfrum, U. Targeting NonsenseMutations in Diseases with Translational Read-Through-Inducing Drugs (TRIDs). BioDrugs 2016, 30, 49–74. [Google Scholar] [CrossRef]
- Moosajee, M.; Ramsden, S.C.; Black, G.C.; Seabra, M.C.; Webster, A.R. Clinical utility gene card for: Choroideremia. Eur. J. Hum. Genet. 2014, 22, 572. [Google Scholar] [CrossRef]
- McDonald, C.M.; Campbell, C.; Torricelli, R.E.; Finkel, R.S.; Flanigan, K.M.; Goemans, N.; Heydemann, P.; Kaminska, A.; Kirschner, J.; Muntoni, F.; et al. Clinical Evaluator Training Group; ACT DMD Study Group. Ataluren in patients with nonsense mutation Duchenne muscular dystrophy (ACT DMD): A multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 390, 1489–1498. [Google Scholar] [CrossRef]
- Moosajee, M.; Gregory-Evans, K.; Ellis, C.D.; Seabra, M.C.; Gregory-Evans, C.Y. Translational bypass of nonsense mutations in zebrafish rep1, pax2.1 and lamb1 highlights a viable therapeutic option for untreatable genetic eye disease. Hum. Mol. Genet. 2008, 17, 3987–4000. [Google Scholar] [CrossRef]
- Moosajee, M.; Tracey-White, D.; Smart, M.; Weetall, M.; Torriano, S.; Kalatzis, V.; da Cruz, L.; Coffey, P.; Webster, A.R.; Welch, E. Functional rescue of REP1 following treatment with PTC124 and novel derivative PTC-414 in human choroideremia fibroblasts and the nonsense-mediated zebrafish model. Hum. Mol. Genet. 2016, 25, 3416–3431. [Google Scholar] [CrossRef]
- Torriano, S.; Erkilic, N.; Baux, D.; Cereso, N.; De Luca, V.; Meunier, I.; Moosajee, M.; Roux, A.F.; Hamel, C.P.; Kalatzis, V. The effect of PTC124 on choroideremia fibroblasts and iPSC-derived RPE raises considerations for therapy. Sci. Rep. 2018, 8, 8234. [Google Scholar] [CrossRef]
- Dabrowski, M.; Bukowy-Bieryllo, Z.; Zietkiewicz, E. Advances in therapeutic use of a drug-stimulated translational readthrough of premature termination codons. Mol. Med. 2018, 24, 25. [Google Scholar] [CrossRef]
- Garanto, A.; van der Velde-Visser, S.D.; Cremers, F.P.M.; Collin, R.W.J. Antisense Oligonucleotide-Based Splice Correction of a Deep-Intronic Mutation in CHM Underlying Choroideremia. Adv. Exp. Med. Biol. 2018, 1074, 83–89. [Google Scholar]
- Collin, R.W.; den Hollander, A.I.; van der Velde-Visser, S.D.; Bennicelli, J.; Bennett, J.; Cremers, F.P. Antisense Oligonucleotide (AON)-based Therapy for Leber Congenital Amaurosis Caused by a Frequent Mutation in CEP290. Mol. Ther. Nucleic Acids. 2012, 1, e14. [Google Scholar] [CrossRef]
- Gerard, X.; Perrault, I.; Hanein, S.; Silva, E.; Bigot, K.; Defoort-Delhemmes, S.; Rio, M.; Munnich, A.; Scherman, D.; Kaplan, J.; et al. AON-mediated Exon Skipping Restores Ciliation in Fibroblasts Harboring the Common Leber Congenital Amaurosis CEP290 Mutation. Mol. Ther. Nucleic Acids. 2012, 1, e29. [Google Scholar] [CrossRef]
- Parmeggiani, F.; De Nadai, K.; Piovan, A.; Binotto, A.; Zamengo, S.; Chizzolini, M. Optical coherence tomography imaging in the management of the Argus II retinal prosthesis system. Eur. J. Ophthalmol. 2017, 27, e16–e21. [Google Scholar] [CrossRef]
- Luo, Y.H.; da Cruz, L. The Argus® II Retinal Prosthesis System. Prog. Retin. Eye Res. 2016, 50, 89–107. [Google Scholar] [CrossRef]
- Simunovic, M.P.; Shen, W.; Lin, J.Y.; Protti, D.A.; Lisowski, L.; Gillies, M.C. Optogenetic approaches to vision restoration. Exp. Eye Res. 2019, 178, 15–26. [Google Scholar] [CrossRef]
- Cehajic-Kapetanovic, J.; Eleftheriou, C.; Allen, A.E.; Milosavljevic, N.; Pienaar, A.; Bedford, R.; Davis, K.E.; Bishop, P.N.; Lucas, R.J. Restoration of Vision with Ectopic Expression of Human Rod Opsin. Curr. Biol. 2015, 25, 2111–2122. [Google Scholar] [CrossRef]
- Eleftheriou, C.G.; Cehajic-Kapetanovic, J.; Martial, F.P.; Milosavljevic, N.; Bedford, R.A.; Lucas, R.J. Meclofenamic acid improves the signal to noise ratio for visual responses produced by ectopic expression of human rod opsin. Mol. Vis. 2017, 23, 334–345. [Google Scholar]
| Clinical trial | Intervention | Clinical centre | References |
|---|---|---|---|
| Phase I/II NCT01461213 Start date: 2011 Completed 2018 | Gene therapy involving subretinal delivery of AAV2-REP1 | University of Oxford, UK | Lancet, 2014 [9] NEJM, 2016 [10] Nat Med, 2018 [11] |
| Phase I/II NCT02341807 Start date: 2015 Ongoing | Gene therapy involving subretinal delivery of AAV2-REP1 | Philadelphia, USA Spark Therapeutics | No reports to date |
| Phase I/II NCT02077361 Start date: 2015 Completed 2018 | Gene therapy involving subretinal delivery of AAV2-REP1 | University of Alberta, Canada | Am J Ophthalmol, 2018 [12] |
| Phase II NCT02553135 Start date: 2015 Completed 2019 | Gene therapy involving subretinal delivery of AAV2-REP1 | University of Miami, USA | Am J Ophthalmol, 2019 [13] |
| Phase IINCT02671539 THOR TRIAL Start date: 2016 Completed 2018 | Gene therapy involving subretinal delivery of AAV2-REP1 | University of Tubingen, Germany | Retina, 2018 [14] |
| Phase II NCT02407678 REGENERATE TRIAL Start date: 2016 Ongoing | Gene therapy involving subretinal delivery of AAV2-REP1 | University of Oxford and Moorfields Eye Hospital, UK | No reports to date |
| Phase II NCT03507686 GEMINI TRIAL Start date: 2017 Ongoing | Gene therapy involving bilateral subretinal delivery of AAV2-REP1 | Nightstar Therapeutics (now Biogen) International, Multi-centre | No reports to date |
| Phase III NCT03496012 STAR TRIAL Start date: 2017 Ongoing | Gene therapy involving subretinal delivery of AAV2-REP1 | Nightstar Therapeutics (now Biogen), International, Multi-centre | No reports to date |
| Observational NCT03584165 SOLSTICE TRIAL Start date: 2018 Ongoing | Long-term follow up study evaluating the safety and efficacy of AAV2-REP1 used in antecedent interventional choroideremia studies, 100 participants | Nightstar Therapeutics (now Biogen), International, Multi-centre | No reports to date |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cehajic Kapetanovic, J.; Barnard, A.R.; MacLaren, R.E. Molecular Therapies for Choroideremia. Genes 2019, 10, 738. https://doi.org/10.3390/genes10100738
Cehajic Kapetanovic J, Barnard AR, MacLaren RE. Molecular Therapies for Choroideremia. Genes. 2019; 10(10):738. https://doi.org/10.3390/genes10100738
Chicago/Turabian StyleCehajic Kapetanovic, Jasmina, Alun R. Barnard, and Robert E. MacLaren. 2019. "Molecular Therapies for Choroideremia" Genes 10, no. 10: 738. https://doi.org/10.3390/genes10100738
APA StyleCehajic Kapetanovic, J., Barnard, A. R., & MacLaren, R. E. (2019). Molecular Therapies for Choroideremia. Genes, 10(10), 738. https://doi.org/10.3390/genes10100738





