In Silico Evaluation of Putative S100B Interacting Proteins in Healthy and IBD Gut Microbiota
Abstract
1. Introduction
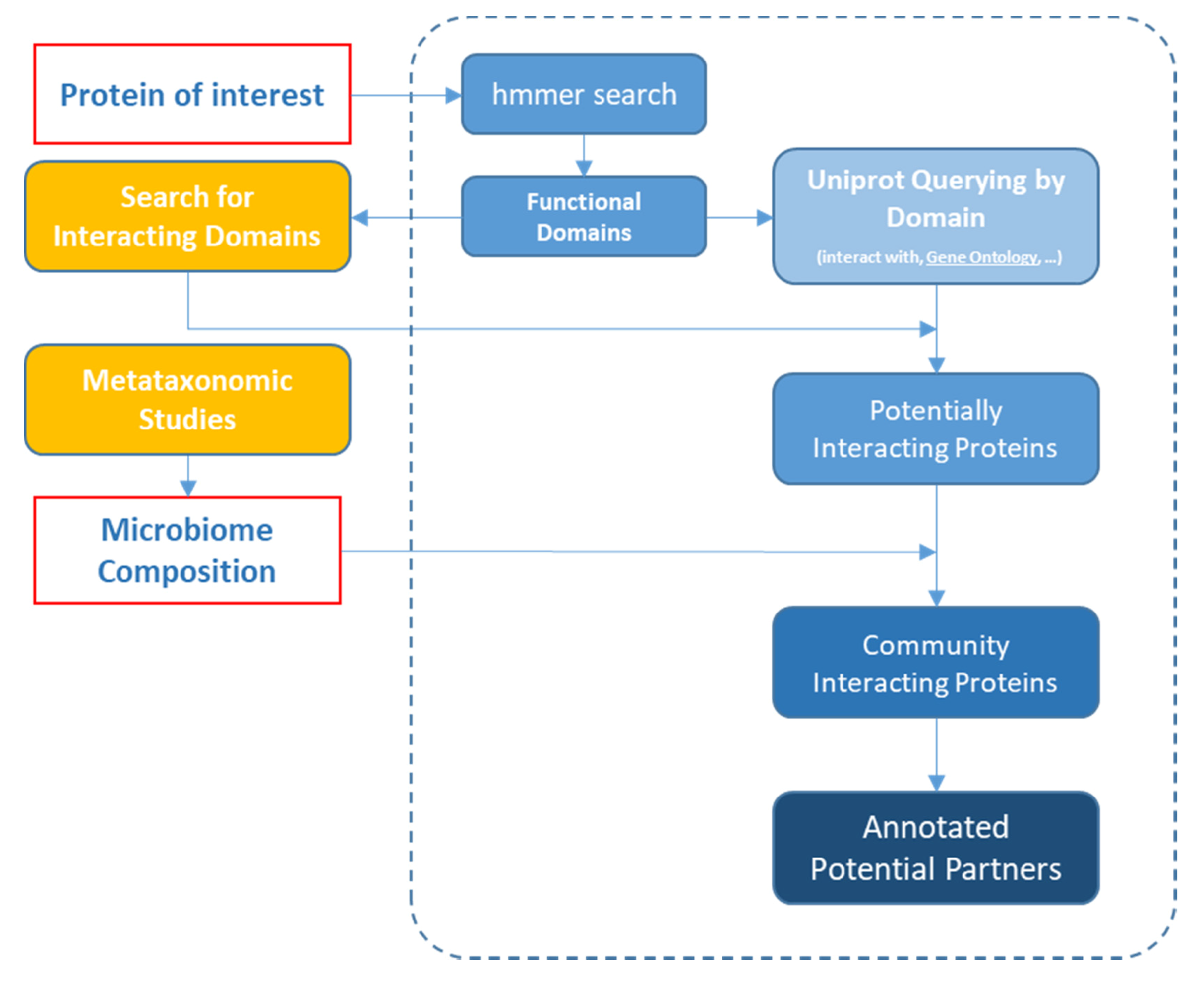
2. Materials and Methods
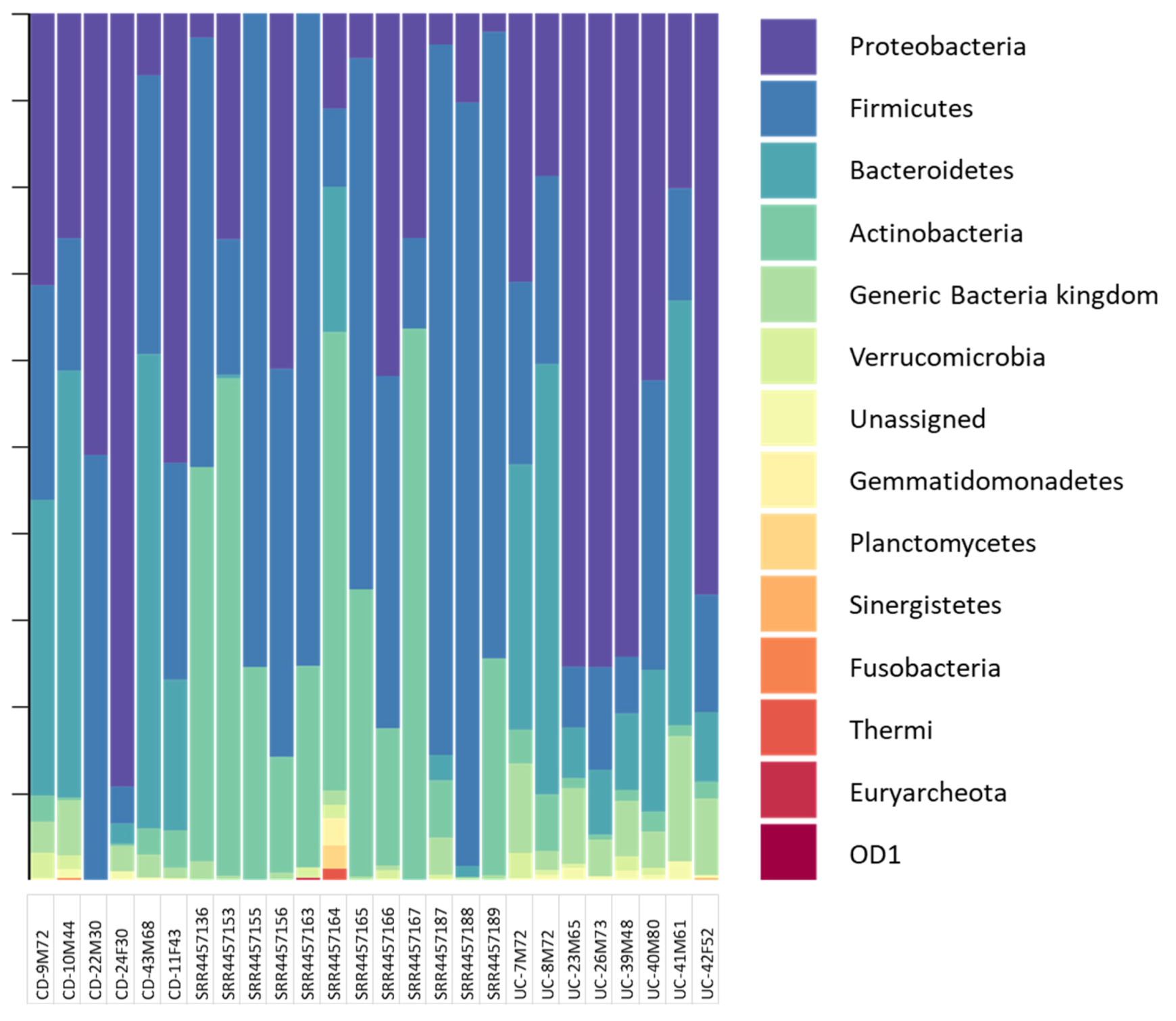
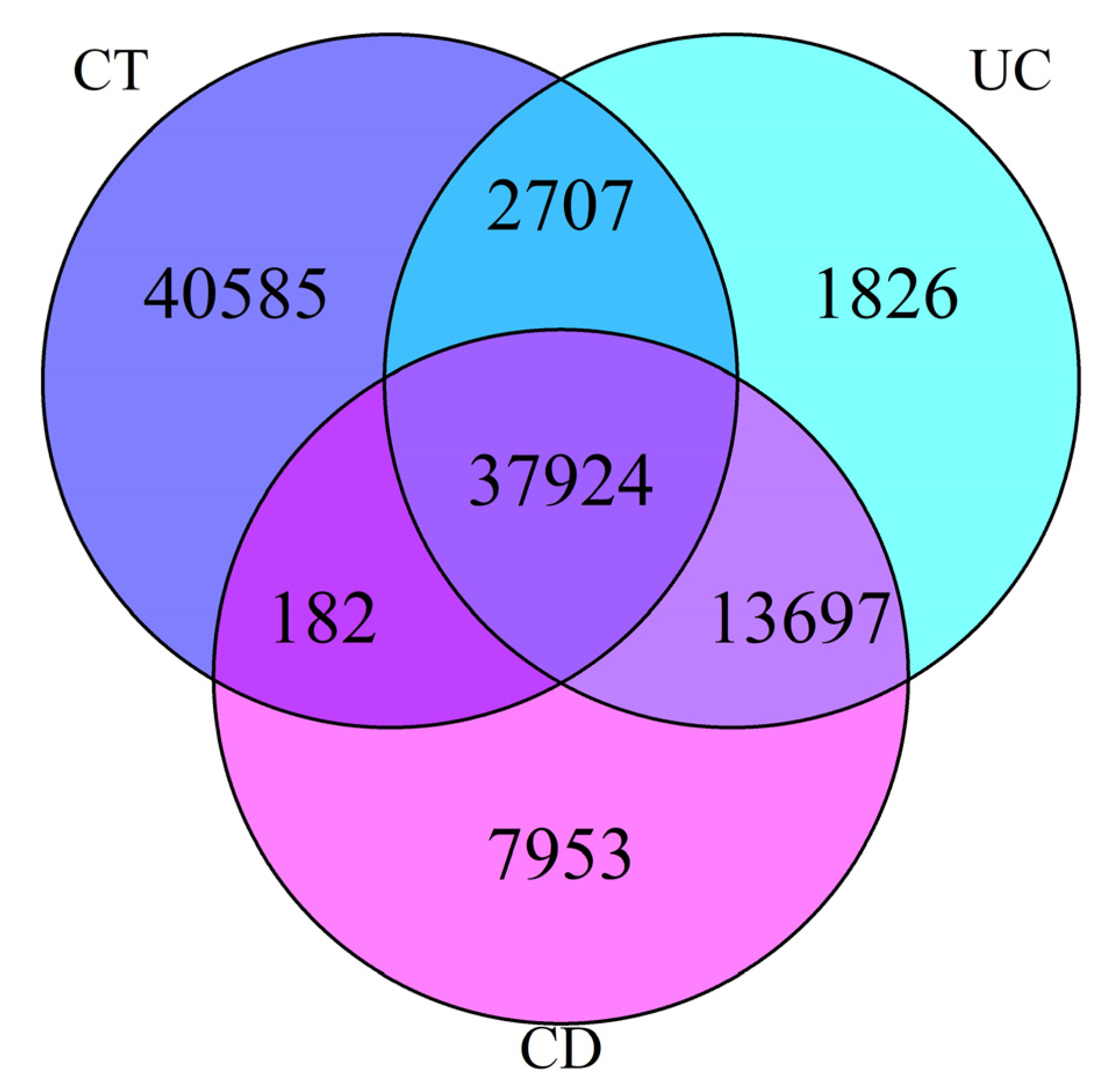
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Michetti, F.; D’Ambrosi, N.; Toesca, A.; Puglisi, M.A.; Serrano, A.; Marchese, E.; Corvino, V.; Geloso, M.C. The S100B story: From biomarker to active factor in neural injury. J. Neurochem. 2019, 148, 168–187. [Google Scholar] [CrossRef]
- Ludwin, S.K.; Kosek, J.C.; Eng, L.F. The topographical distribution of S-100 and GFA proteins in the adult rat brain: An immunohistochemical study using horseradish peroxidase-labelled antibodies. J. Comp. Neurol. 1976, 165, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ferri, G.L.; Probert, L.; Cocchia, D.; Michetti, F.; Marangos, P.J.; Polak, J.M. Evidence for the presence of S-100 protein in the glial component of the human enteric nervous system. Nature 1982, 297, 409–410. [Google Scholar] [CrossRef] [PubMed]
- Brockes, J.P.; Fields, K.L.; Raff, M.C. Studies on cultured rat Schwann cells. I. Establishment of purified populations from cultures of peripheral nerve. Brain Res. 1979, 165, 105–118. [Google Scholar] [CrossRef]
- Didier, M.; Harandi, M.; Aguera, M.; Bancel, B.; Tardy, M.; Fages, C.; Calas, A.; Stagaard, M.; Møllgård, K.; Belin, M.F. Differential immunocytochemical staining for glial fibrillary acidic (GFA) protein, S-100 protein and glutamine synthetase in the rat subcommissural organ, nonspecialized ventricular ependyma and adjacent neuropil. Cell Tissue Res. 1986, 245, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Rickmann, M.; Wolff, J.R. S100 protein expression in subpopulations of neurons of rat brain. Neuroscience 1995, 67, 977–991. [Google Scholar] [CrossRef]
- Yang, Q.; Hamberger, A.; Hyden, H.; Wang, S.; Stigbrand, T.; Haglid, K.G. S-100 beta has a neuronal localisation in the rat hindbrain revealed by an antigen retrieval method. Brain Res. 1995, 696, 49–61. [Google Scholar] [CrossRef]
- Cocchia, D.; Michetti, F.; Donato, R. Immunochemical and immuno-cytochemical localization of S-100 antigen in normal human skin. Nature 1981, 294, 85–87. [Google Scholar] [CrossRef]
- Cocchia, D.; Tiberio, G.; Santarelli, R.; Michetti, F. S-100 protein in “follicular dendritic” cells or rat lymphoid organs. An immunochemical and immunocytochemical study. Cell Tissue Res. 1983, 230, 95–103. [Google Scholar] [CrossRef]
- Stefansson, K.; Wollmann, R.L.; Moore, B.W.; Arnason, B.G. S-100 protein in human chondrocytes. Nature 1982, 295, 63–64. [Google Scholar] [CrossRef]
- Michetti, F.; Dell’Anna, E.; Tiberio, G.; Cocchia, D. Immunochemical and immunocytochemical study of S-100 protein in rat adipocytes. Brain Res. 1983, 262, 352–356. [Google Scholar] [CrossRef]
- Lauriola, L.; Maggiano, N.; Sentinelli, S.; Michetti, F.; Cocchia, D. Satellite cells in the normal human adrenal gland and in pheochromocytomas. An immunohistochemical study. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1985, 49, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Tubaro, C.; Arcuri, C.; Giambanco, I.; Donato, R. S100B protein in myoblasts modulates myogenic differentiation via NF-kappaB-dependent inhibition of MyoD expression. J. Cell Physiol. 2010, 223, 270–282. [Google Scholar]
- Michetti, F.; Rende, M.; Calogero, G.; Dell’Anna, E.; Cocchia, D. Immunochemical detection of S-100 protein in non-nervous structures of the rabbit eye. Brain Res. 1985, 332, 358–360. [Google Scholar] [CrossRef]
- Moore, B.W. A soluble protein characteristic of the nervous system. Biochem. Biophys. Res. Commun. 1965, 19, 739–744. [Google Scholar] [CrossRef]
- Donato, R.; Cannon, B.R.; Sorci, G.; Riuzzi, F.; Hsu, K.; Weber, D.J.; Geczy, C.L. Functions of S100 proteins. Curr. Mol. Med. 2013, 13, 24–57. [Google Scholar] [CrossRef] [PubMed]
- Gong, T.; Liu, L.; Jiang, W.; Zhou, R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 2020, 20, 95–112. [Google Scholar] [CrossRef]
- Cirillo, C.; Sarnelli, G.; Esposito, G.; Grosso, M.; Petruzzelli, R.; Izzo, P.; Calì, G.; D’Armiento, F.P.; Rocco, A.; Nardone, G.; et al. Increased mucosal nitric oxide production in ulcerative colitis is mediated in part by the enteroglial-derived S100B protein. Neurogastroenterol. Motil. 2009, 21, 1209–e112. [Google Scholar] [CrossRef]
- Cirillo, C.; Sarnelli, G.; Turco, F.; Mango, A.; Grosso, M.; Aprea, G.; Masone, S.; Cuomo, R. Proinflammatory stimuli activates human-derived enteroglial cells and induces autocrine nitric oxide production. Neurogastroenterol. Motil. 2011, 23, e372–e382. [Google Scholar] [CrossRef]
- Esposito, G.; Cirillo, C.; Sarnelli, G.; De Filippis, D.; D’Armiento, F.P.; Rocco, A.; Nardone, G.; Petruzzelli, R.; Grosso, M.; Izzo, P.; et al. Enteric glial-derived S100B protein stimulates nitric oxide production in celiac disease. Gastroenterology 2007, 133, 918–925. [Google Scholar] [CrossRef]
- Esposito, G.; Capoccia, E.; Sarnelli, G.; Scuderi, C.; Cirillo, C.; Cuomo, R.; Steardo, L. The antiprotozoal drug pentamidine ameliorates experimentally induced acute colitis in mice. J. Neuroinflammation 2012, 9, 277. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Microbiota-Brain-Gut Axis and Neurodegenerative Diseases. Curr. Neurol. Neurosci. Rep. 2017, 17, 94. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Jiang, Y.; Xu, K.; Cui, M.; Ye, W.; Zhao, G.; Jin, L.; Chen, X. The progress of gut microbiome research related to brain disorders. J. Neuroinflammation 2020, 17, 25. [Google Scholar] [CrossRef]
- Di Liddo, R.; Piccione, M.; Schrenk, S.; Dal Magro, C.; Cosma, C.; Padoan, A.; Contran, N.; Scapellato, M.L.; Pagetta, A.; Romano Spica, V.; et al. S100B as a new fecal biomarker of inflammatory bowel diseases. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 323–332. [Google Scholar] [PubMed]
- Ostendorp, T.; Heizmann, C.W.; Kroneck, P.M.H.; Fritz, G. Purification, crystallization and preliminary X-ray diffraction studies on human Ca2+-binding protein S100B. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2005, 61, 673–675. [Google Scholar] [CrossRef] [PubMed]
- Prez, K.D.; Fan, L. Structural Basis for S100B Interaction with its Target Proteins. J. Mol. Genet. Med. 2018, 12, 366. [Google Scholar] [PubMed]
- El-Gebali, S.; Mistry, J.; Bateman, A.; Eddy, S.R.; Luciani, A.; Potter, S.C.; Qureshi, M.; Richardson, L.J.; Salazar, G.A.; Smart, A.; et al. The Pfam protein families database in 2019. Nucleic Acids Res. 2019, 47, D427–D432. [Google Scholar] [CrossRef]
- Wheeler, T.J.; Eddy, S.R. nhmmer: DNA homology search with profile HMMs. Bioinformatics 2013, 29, 2487–2489. [Google Scholar] [CrossRef]
- Finn, R.D.; Miller, B.L.; Clements, J.; Bateman, A. iPfam: A database of protein family and domain interactions found in the Protein Data Bank. Nucleic Acids Res. 2014, 42, D364–D373. [Google Scholar] [CrossRef]
- Berman, H.M.; Battistuz, T.; Bhat, T.N.; Bluhm, W.F.; Bourne, P.E.; Burkhardt, K.; Feng, Z.; Gilliland, G.L.; Iype, L.; Jain, S.; et al. The Protein Data Bank. Acta Crystallogr. D Biol. Crystallogr. 2002, 58, 899–907. [Google Scholar] [CrossRef]
- Alexa, A.; Rahnenfuhrer, J. TopGO: Enrichment Analysis for Gene Ontology. R package version 2.40.0. 2020. Available online: https://bioconductor.org/packages/release/bioc/html/topGO.html (accessed on 15 February 2020).
- Hansen, K.D.; Gentry, J.; Long, L.; Gentleman, R.; Falcon, S.; Hahne, F.; Sarkar, D. Rgraphviz: Provides Plotting Capabilities for R Graph Objects. Bioconductor version: Release (3.11). 2020. Available online: https://bioconductor.org/packages/release/bioc/html/Rgraphviz.html (accessed on 30 June 2020).
- Valeriani, F.; Protano, C.; Gianfranceschi, G.; Leoni, E.; Galasso, V.; Mucci, N.; Vitali, M.; Romano Spica, V. Microflora Thermarum Atlas project: Biodiversity in thermal spring waters and natural SPA pools. Water Sci. Tech-W. Supply 2018, 18, 1472–1483. [Google Scholar] [CrossRef]
- Kodama, Y.; Shumway, M.; Leinonen, R. International Nucleotide Sequence Database Collaboration. The Sequence Read Archive: Explosive growth of sequencing data. Nucleic Acids Res. 2012, 40, D54–D56. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Capoccia, E.; Cirillo, C.; Gigli, S.; Pesce, M.; D’Alessandro, A.; Cuomo, R.; Sarnelli, G.; Steardo, L.; Esposito, G. Enteric glia: A new player in inflammatory bowel diseases. Int. J. Immunopathol. Pharmacol. 2015, 28, 443–451. [Google Scholar] [CrossRef] [PubMed]
- de Souza, H.S.P.; Fiocchi, C. Immunopathogenesis of IBD: Current state of the art. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Turco, F.; Sarnelli, G.; Cirillo, C.; Palumbo, I.; De Giorgi, F.; D’Alessandro, A.; Cammarota, M.; Giuliano, M.; Cuomo, R. Enteroglial-derived S100B protein integrates bacteria-induced Toll-like receptor signalling in human enteric glial cells. Gut 2014, 63, 105–115. [Google Scholar] [CrossRef]
- Franzosa, E.A.; Sirota-Madi, A.; Avila-Pacheco, J.; Fornelos, N.; Haiser, H.J.; Reinker, S.; Vatanen, T.; Brantley Hall, A.; Mallick, H.; McIver, L.J.; et al. Gut microbiome structure and metabolic activity in inflammatory bowel disease. Nat. Microbiol. 2019, 4, 293–305. [Google Scholar] [CrossRef]
- Halfvarson, J.; Brislawn, C.J.; Lamendella, R.; Vázquez-Baeza, Y.; Walters, W.A.; Bramer, L.M.; D’Amato, M.; Bonfiglio, F.; McDonald, D.; Gonzalez, A.; et al. Dynamics of the human gut microbiome in inflammatory bowel disease. Nat. Microbiol. 2017, 2, 17004. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Lu, Y.; Li, X.; Liu, S.; Zhang, Y.; Zhang, D. Toll-like Receptors and Inflammatory Bowel Disease. Front. Immunol. 2018, 9, 72. [Google Scholar] [CrossRef]
- Ferguson, P.L.; Shaw, G.S. Human S100B protein interacts with the Escherichia coli division protein FtsZ in a calcium-sensitive manner. J. Biol. Chem. 2004, 279, 18806–18813. [Google Scholar] [CrossRef] [PubMed]
- Baudier, J.; Gentil, B.J. The S100B Protein and Partners in Adipocyte Response to Cold Stress and Adaptive Thermogenesis: Facts, Hypotheses, and Perspectives. Biomolecules 2020, 10, E843. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, M.; Franzosa, E.A.; Lloyd-Price, J.; McIver, L.J.; Schwager, R.; Poon, T.W.; Ananthakrishnan, A.N.; Andrews, E.; Barron, G.; Lake, K.; et al. Dynamics of metatranscription in the inflammatory bowel disease gut microbiome. Nat. Microbiol. 2018, 3, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, T.; Schallert, K.; Vilchez-Vargas, R.; Benndorf, D.; Püttker, S.; Sydor, S.; Schulz, C.; Bechmann, L.; Canbay, A.; Heidrich, B.; et al. Metaproteomics of fecal samples of Crohn’s disease and Ulcerative Colitis. J. Proteomics 2019, 201, 93–103. [Google Scholar] [CrossRef] [PubMed]
| Domain | Partner | Pfam ID | Description |
|---|---|---|---|
| S100B | C2 | PF00168 | Structural domain involved in targeting proteins to membrane |
| EF-hand_1 | PF00036 | Helix-loop-helix domain or motif found in a large family of Ca-binding proteins. | |
| F-actin_cap_A | PF01267 | Domain binding in a Ca independent manner the fast-growing ends of actin filaments | |
| FGF | PF00167 | Fibroblast growth factors, Family of cell signalling proteins | |
| Ig_2 | PF13895 | Immunoglobulin domain | |
| IL1 | PF00340 | Interleukins | |
| Myosin_tail_1 | PF01576 | Myosin domain | |
| S_100 | PF01023 | S100 type calcium-binding domain | |
| SGS | PF05002 | Structural domain found in calcyclin binding proteins | |
| EF-hand | ATP_Ca_trans_C | PF12424 | Plasma membrane calcium transporter ATPase C terminal |
| CA_chan_IQ | PF08763 | Voltage-gated calcium channel IQ domain | |
| CaATP_NAI | PF12515 | Ca2+-ATPase N terminal autoinhibitory domain | |
| CaMBD | PF02888 | Calmodulin binding domain | |
| CH | PF00307 | Calponin homology (CH) domain | |
| Death | PF00531 | Death domain | |
| EF-hand_1 | PF00036 | Helix-loop-helix domain or motif found in a large family of Ca-binding proteins. | |
| EF-hand_7 | PF13499 | EF-hand domain pair | |
| EF-hand_5 | PF13202 | EF hand | |
| EF-hand_6 | PF13405 | EF-hand domain | |
| EF-hand_8 | PF13833 | EF-hand domain pair | |
| EnY2 | PF10163 | Transcription factor e(y)2 | |
| F-actin_cap_A | PF01267 | F-actin capping protein alpha subunit | |
| FGF | PF00167 | Fibroblast growth factor | |
| IQ | PF00612 | IQ calmodulin-binding motif | |
| KCNQ_channel | PF03520 | KCNQ voltage-gated potassium channel | |
| Melittin | PF01372 | Melittin | |
| Metallophos | PF00149 | Calcineurin-like phosphoesterase | |
| Myosin_head | PF00063 | Myosin head | |
| PI3KA | PF00613 | Phosphoinositide 3-kinase family, accessory domain | |
| Pik1 | PF11522 | Yeast phosphatidylinositol-4-OH kinase Pik1 | |
| Pkinase | PF00069 | Protein kinase domain | |
| S_100 | PF01023 | S100 type calcium-binding domain | |
| SAC3 | PF12209 | Leucine permease transcriptional regulator helical domain | |
| Sfi1 | PF08457 | Sfi1 spindle body protein | |
| Synuclein | PF01387 | Synuclein | |
| Troponin | PF00992 | Troponin |
| Category | Total Proteins | From Genus | S100B Interacting |
|---|---|---|---|
| CD | 22,066,981 | 51 | 61,086 |
| UC | 21,402,684 | 50 | 57,344 |
| CT | 24,306,740 | 32 | 83,008 |
| Domain | CT | Exp. | CD | Exp. | UC | Exp. | Description |
|---|---|---|---|---|---|---|---|
| PF00036 | 35 | 110 | 16 | 100 | 17 | 97 | Helix-loop-helix structural domain or motif found in a large family of calcium-binding proteins. |
| PF00069 | 35255 | 33790 | 16544 | 30657 | 13444 | 29731 | Protein kinase domain |
| PF13895 | 149 | 76 | 12 | 69 | 10 | 67 | Immunoglobulin domain |
| PF00612 | 8 | 20 | 9 | 18 | 9 | 17 | IQ calmodulin-binding motif |
| PF00149 | 44022 | 43137 | 41838 | 39136 | 41095 | 37955 | Calcineurin-like phosphoesterase |
| PF13202 | 1962 | 3421 | 2420 | 3104 | 2524 | 3010 | EF hand |
| PF13499 | 2192 | 1226 | 696 | 1113 | 724 | 1079 | EF-hand domain pair |
| PF13405 | 60 | 55 | 56 | 50 | 58 | 49 | EF-hand domain |
| PF00063 | 2 | 3 | 2 | 3 | 2 | 3 | Myosin head (motor domain) |
| Category | GO Term | CD | UC | CT | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GOID | Description | Annotated | Observed | Expect. | Fisher | Observed | Observed | Fisher | Observed | Expect. | Fisher | |
| MF | GO:0016787 | hydrolase activity | 24136 | 20345 | 16818 | <1e-30 | 18513 | 15394 | <1e-30 | 15954 | 13142 | <1e-30 |
| GO:0003824 | catalytic activity | 28253 | 20731 | 19687 | <1e-30 | 18808 | 18020 | <1e-30 | 16191 | 15384 | <1e-30 | |
| GO:0016462 | pyrophosphatase activity | 2508 | 2363 | 1748 | <1e-30 | 2290 | 1600 | <1e-30 | 1762 | 1366 | <1e-30 | |
| GO:0008796 | bis(5′-nucleosyl)-tetraphosphatase activity | 2155 | 2062 | 1502 | <1e-30 | 1990 | 1374 | <1e-30 | 1478 | 1173 | <1e-30 | |
| GO:0008803 | bis(5′-nucleosyl)-tetraphosphatase (symmetrical) activity | 2099 | 2014 | 1463 | <1e-30 | 1950 | 1339 | <1e-30 | 1436 | 1143 | <1e-30 | |
| GO:0004551 | nucleotide diphosphatase activity | 2156 | 2062 | 1502 | <1e-30 | 1990 | 1375 | <1e-30 | 1478 | 1174 | <1e-30 | |
| GO:0016818 | hydrolase activity, acting on acid anhydrides, in phosphorus containing anhydrides | 2517 | 2363 | 1754 | <1e-30 | 2290 | 1605 | <1e-30 | 1762 | 1371 | <1e-30 | |
| GO:0016817 | hydrolase activity, acting on acid anhydrides | 2519 | 2363 | 1755 | <1e-30 | 2290 | 1607 | <1e-30 | 1762 | 1372 | <1e-30 | |
| GO:0004527 | exonuclease activity | 911 | 848 | 635 | <1e-30 | 663 | 496 | <1e-30 | ||||
| GO:0004518 | nuclease activity | 1004 | 856 | 700 | <1e-30 | |||||||
| GO:0016791 | phosphatase activity | 869 | 645 | 606 | 0.0016 | |||||||
| GO:0004081 | bis(5′-nucleosyl)-tetraphosphatase (asymmetrical) activity | 55 | 48 | 38 | 0.0020 | 42 | 30 | 0.00065 | ||||
| GO:0004721 | phosphoprotein phosphatase activity | 512 | 355 | 327 | 0.0044 | 452 | 279 | <1e-30 | ||||
| GO:0004115 | 3′,5′-cyclic-AMP phosphodiesterase activity | 12 | 12 | 8 | 0.0045 | |||||||
| CC | GO:0016021 | integral component of membrane | 5163 | 161 | 155 | 0.0065 | ||||||
| GO:0031224 | intrinsic component of membrane | 5163 | 161 | 155 | 0.0065 | |||||||
| GO:0044425 | membrane part | 5165 | 161 | 155 | 0.0069 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orsini, M.; Di Liddo, R.; Valeriani, F.; Mancin, M.; D’Incà, R.; Castagnetti, A.; Aceti, A.; Parnigotto, P.P.; Romano Spica, V.; Michetti, F. In Silico Evaluation of Putative S100B Interacting Proteins in Healthy and IBD Gut Microbiota. Cells 2020, 9, 1697. https://doi.org/10.3390/cells9071697
Orsini M, Di Liddo R, Valeriani F, Mancin M, D’Incà R, Castagnetti A, Aceti A, Parnigotto PP, Romano Spica V, Michetti F. In Silico Evaluation of Putative S100B Interacting Proteins in Healthy and IBD Gut Microbiota. Cells. 2020; 9(7):1697. https://doi.org/10.3390/cells9071697
Chicago/Turabian StyleOrsini, Massimiliano, Rosa Di Liddo, Federica Valeriani, Marzia Mancin, Renata D’Incà, Andrea Castagnetti, Antonio Aceti, Pier Paolo Parnigotto, Vincenzo Romano Spica, and Fabrizio Michetti. 2020. "In Silico Evaluation of Putative S100B Interacting Proteins in Healthy and IBD Gut Microbiota" Cells 9, no. 7: 1697. https://doi.org/10.3390/cells9071697
APA StyleOrsini, M., Di Liddo, R., Valeriani, F., Mancin, M., D’Incà, R., Castagnetti, A., Aceti, A., Parnigotto, P. P., Romano Spica, V., & Michetti, F. (2020). In Silico Evaluation of Putative S100B Interacting Proteins in Healthy and IBD Gut Microbiota. Cells, 9(7), 1697. https://doi.org/10.3390/cells9071697










