Recent Applications of Three Dimensional Printing in Cardiovascular Medicine
Abstract
1. Introduction
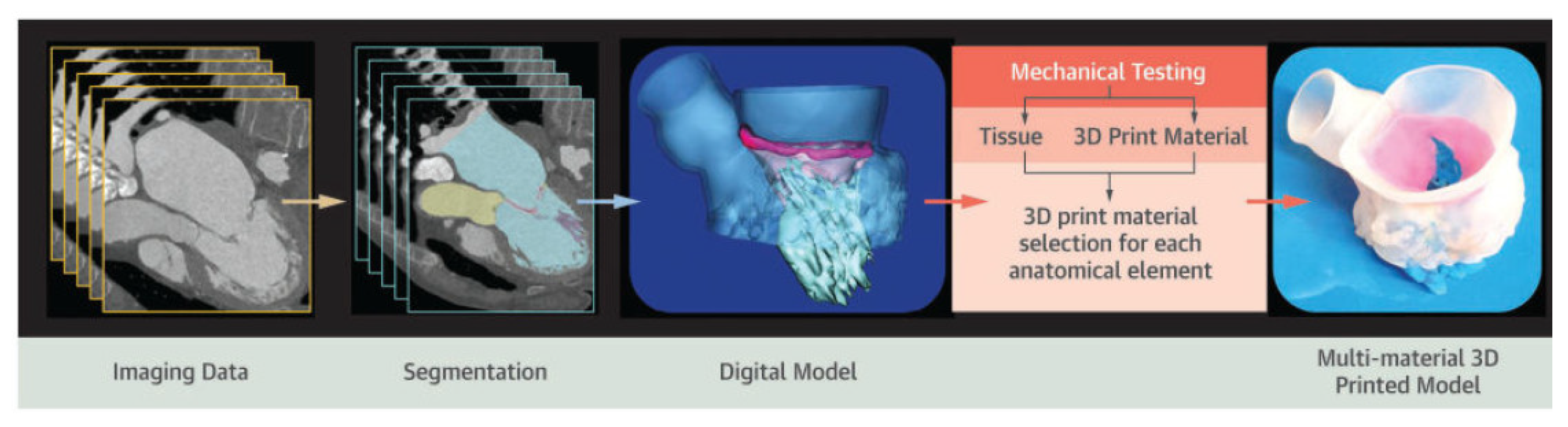
2. Process and Technologies of Cardiovascular 3D Printing
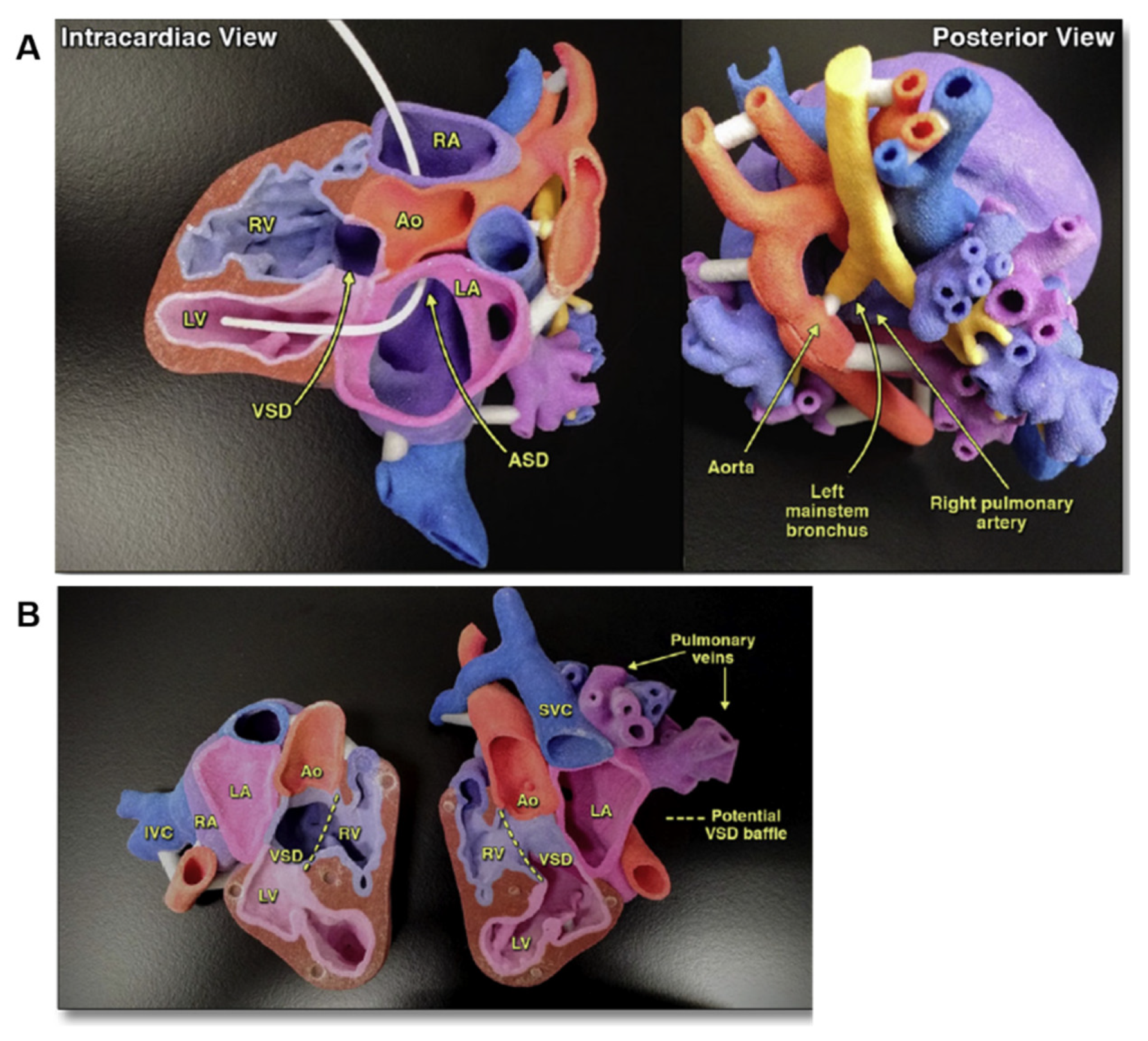
3. 3D Printing for Teaching and Surgical Training in Cardiovascular Medicine
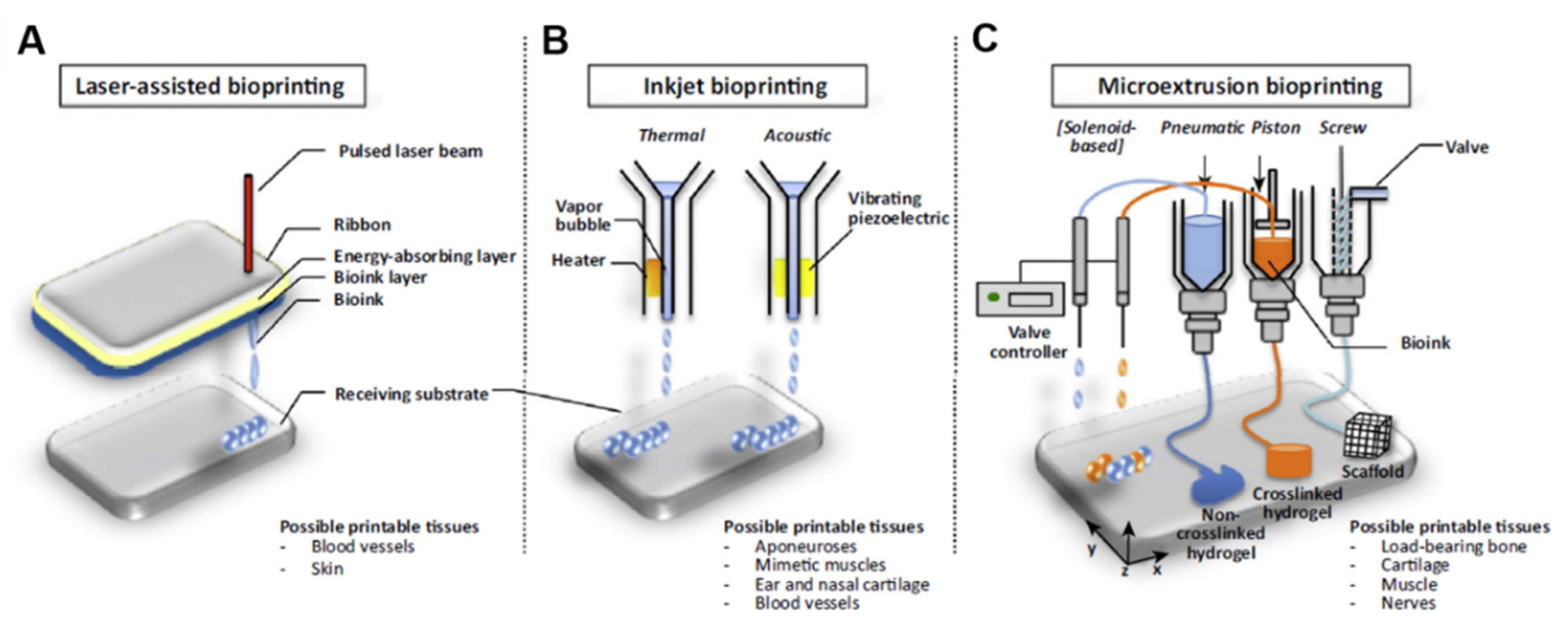
4. 3D Bioprinting for Cardiovascular Applications
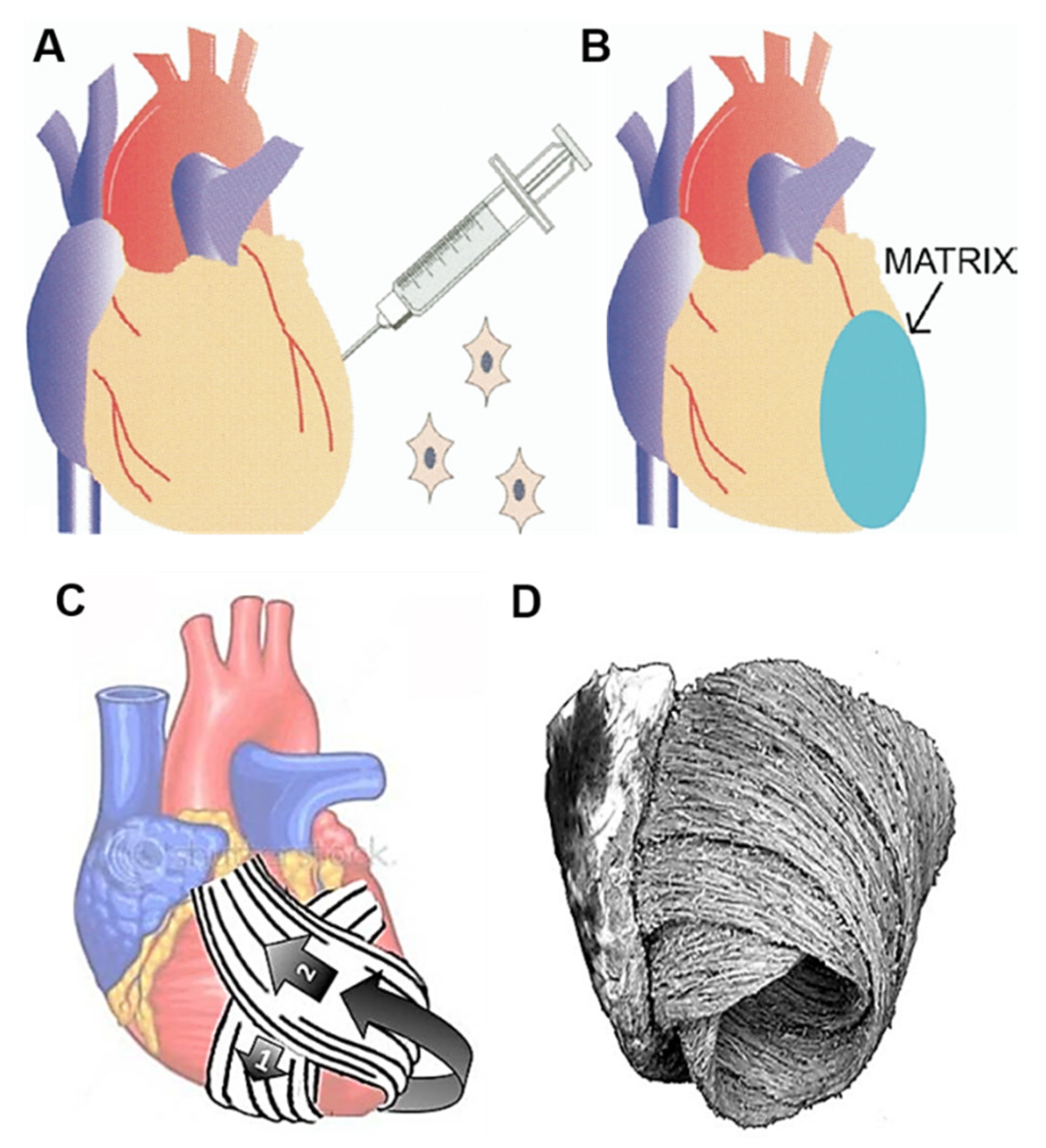
4.1. 3D Bioprinting of Functional Myocardium
4.2. 3D Bioprinting of Heart Valves
4.3. 3D Bioprinted Heart Tissue Patches for Drug Screening
5. 3D Printing for Testing and Realizing New Heart Devices
6. Regulatory Considerations and Commercialization of 3D Printed and 3D Bioprinted Products
7. Current Limitations, Future Perspectives, and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paul, G.M.; Rezaienia, A.; Wen, P.; Condoor, S.; Parkar, N.; King, W.; Korakianitis, T. Medical Applications for 3D Printing: Recent Developments. Mo Med. 2018, 115, 75–81. [Google Scholar] [PubMed]
- Ventola, C.L. Medical Applications for 3D Printing: Current and Projected Uses. Pharm. Ther. 2014, 39, 704–711. [Google Scholar]
- Hull, C.W. Apparatus for Production of Three-Dimensional Objects by Stereolithography. U.S. Patent Application No. 4,575,330, 1986, 11 March 1986. [Google Scholar]
- Zuniga, J.; Katsavelis, D.; Peck, J.; Stollberg, J.; Petrykowski, M.; Carson, A.; Fernandez, C. Cyborg beast: A low-cost 3d-printed prosthetic hand for children with upper-limb differences. BMC Res. Notes 2015, 8, 10. [Google Scholar] [CrossRef]
- Vaccarezza, M.; Papa, V. 3D printing: A valuable resource in human anatomy education. Anat. Sci. Int. 2015, 90, 64–65. [Google Scholar] [CrossRef]
- Lau, I.; Wong, Y.H.; Yeong, C.H.; Abdul Aziz, Y.F.; Md Sari, N.A.; Hashim, S.A.; Sun, Z. Quantitative and qualitative comparison of low- and high-cost 3D-printed heart models. Quant. Imaging Med. Surg. 2019, 9, 107–114. [Google Scholar] [CrossRef]
- Lau, I.; Sun, Z. Three-dimensional printing in congenital heart disease: A systematic review. J. Med. Radiat. Sci. 2018, 65, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Garner, K.H.; Singla, D.K. 3D modeling: A future of cardiovascular medicine. Can. J. Physiol. Pharmacol. 2019, 97, 277–286. [Google Scholar] [CrossRef]
- Valverde, I. Three-dimensional Printed Cardiac Models: Applications in the Field of Medical Education, Cardiovascular Surgery, and Structural Heart Interventions. Rev. Esp. Cardiol. (Engl. Ed.) 2017, 70, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Giannopoulos, A.A.; Mitsouras, D.; Yoo, S.J.; Liu, P.P.; Chatzizisis, Y.S.; Rybicki, F.J. Applications of 3D printing in cardiovascular diseases. Nat. Rev. Cardiol. 2016, 13, 701–718. [Google Scholar] [CrossRef]
- Byrne, N.; Velasco Forte, M.; Tandon, A.; Valverde, I.; Hussain, T. A systematic review of image segmentation methodology, used in the additive manufacture of patient-specific 3D printed models of the cardiovascular system. JRSM Cardiovasc. Dis. 2016, 5, 2048004016645467. [Google Scholar] [CrossRef]
- Vukicevic, M.; Mosadegh, B.; Min, J.K.; Little, S.H. Cardiac 3D Printing and its Future Directions. JACC Cardiovasc. Imaging 2017, 10, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.J.; Hall, E.J. Computed tomography—An increasing source of radiation exposure. N. Engl. J. Med. 2007, 357, 2277–2284. [Google Scholar] [CrossRef] [PubMed]
- Bateman, M.G.; Durfee, W.K.; Iles, T.L.; Martin, C.M.; Liao, K.; Erdman, A.G.; Iaizzo, P.A. Cardiac patient-specific three-dimensional models as surgical planning tools. Surgery 2019, 167, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.N. Echocardiography in the era of multimodality cardiovascular imaging. BioMed Res. Int. 2013, 2013, 310483. [Google Scholar] [CrossRef]
- Wang, D.D.; Gheewala, N.; Shah, R.; Levin, D.; Myers, E.; Rollet, M.; O’Neill, W.W. Three-Dimensional Printing for Planning of Structural Heart Interventions. Interv. Cardiol. Clin. 2018, 7, 415–423. [Google Scholar] [CrossRef]
- Otton, J.M.; Birbara, N.S.; Hussain, T.; Greil, G.; Foley, T.A.; Pather, N. 3D printing from cardiovascular CT: A practical guide and review. Cardiovasc. Diagn. Ther. 2017, 7, 507–526. [Google Scholar] [CrossRef]
- Yoo, S.J.; Thabit, O.; Kim, E.K.; Ide, H.; Yim, D.; Dragulescu, A.; Seed, M.; Grosse-Wortmann, L.; van Arsdell, G. 3D printing in medicine of congenital heart diseases. 3D Print. Med. 2015, 2, 3. [Google Scholar] [CrossRef]
- Bücking, T.M.; Hill, E.R.; Robertson, J.L.; Maneas, E.; Plumb, A.A.; Nikitichev, D.I. From medical imaging data to 3D printed anatomical models. PLoS ONE 2017, 12, e0178540. [Google Scholar] [CrossRef]
- Kerby, J.; Shukur, Z.N.; Shalhoub, J. The relationships between learning outcomes and methods of teaching anatomy as perceived by medical students. Clin. Anat. 2011, 24, 489–497. [Google Scholar] [CrossRef]
- Li, K.H.C.; Kui, C.; Lee, E.K.M.; Ho, C.S.; Wong, S.H.; Wu, W.; Wong, W.T.; Voll, J.; Li, G.; Liu, T.; et al. The Role of 3D Printing in Anatomy Education and Surgical Training: A Narrative Review; MedEdPublisher: Dundee, UK, 2017. [Google Scholar]
- Fasel, J.H.; Aguiar, D.; Kiss-Bodolay, D.; Montet, X.; Kalangos, A.; Stimec, B.V.; Ratib, O. Adapting anatomy teaching to surgical trends: A combination of classical dissection, medical imaging, and 3D-printing technologies. Surg. Radiol. Anat. 2016, 38, 361–367. [Google Scholar] [CrossRef]
- Al-Ramahi, J.; Luo, H.; Fang, R.; Chou, A.; Jiang, J.; Kille, T. Development of an Innovative 3D Printed Rigid Bronchoscopy Training Model. Ann. Otol. Rhinol. Laryngol. 2016, 125, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Jonas, R.A. Training fellows in paediatric cardiac surgery. Cardiol. Young 2016, 26, 1474–1483. [Google Scholar] [CrossRef] [PubMed]
- Shui, W.; Zhou, M.; Chen, S.; Pan, Z.; Deng, Q.; Yao, Y.; Pan, H.; He, T.; Wang, X. The production of digital and printed resources from multiple modalities using visualization and three-dimensional printing techniques. Int. J. Comput. Assist. Radiol. Surg. 2017, 12, 13–23. [Google Scholar] [CrossRef]
- Ong, C.S.; Hibino, N. The use of 3D printing in cardiac surgery. J. Thorac. Dis. 2017, 9, 2301–2302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, M.; Moharem-Elgamal, S.; Beckingham, R.; Hamilton, M.; Manghat, N.; Milano, E.G.; Bucciarelli-Ducci, C.; Caputo, M.; Biglino, G. Evaluating 3D-printed models of coronary anomalies: A survey among clinicians and researchers at a university hospital in the UK. BMJ Open 2019, 9, e025227. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, M. Three-dimensional imaging and printing in cardiology. Eur. Heart J. 2017, 38, 230–231. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fan, Y.; Yang, F.; Cheung, G.S.; Chan, A.K.; Wang, D.D.; Lam, Y.Y.; Chow, M.C.; Leong, M.C.; Kam, K.K.; So, K.C.; et al. Device Sizing Guided by Echocardiography-Based Three-Dimensional Printing Is Associated with Superior Outcome after Percutaneous Left Atrial Appendage Occlusion. J. Am. Soc. Echocardiogr. 2019, 32, 708–719.e701. [Google Scholar] [CrossRef]
- Motwani, M.; Burley, O.; Luckie, M.; Cunnington, C.; Pisaniello, A.D.; Hasan, R.; Malik, I.; Fraser, D.G. 3D-printing assisted closure of paravalvular leak. J. Cardiovasc. Comput. Tomogr. 2019. [Google Scholar] [CrossRef]
- Vukicevic, M.; Puperi, D.S.; Jane Grande-Allen, K.; Little, S.H. 3D Printed Modeling of the Mitral Valve for Catheter-Based Structural Interventions. Ann. Biomed. Eng. 2017, 45, 508–519. [Google Scholar] [CrossRef]
- Premyodhin, N.; Mandair, D.; Ferng, A.S.; Leach, T.S.; Palsma, R.P.; Albanna, M.Z.; Khalpey, Z.I. 3D printed mitral valve models: Affordable simulation for robotic mitral valve repair. Interact. Cardiovasc. Thorac. Surg. 2018, 26, 71–76. [Google Scholar] [CrossRef]
- Engelhardt, S.; Sauerzapf, S.; Preim, B.; Karck, M.; Wolf, I.; De Simone, R. Flexible and comprehensive patient-specific mitral valve silicone models with chordae tendineae made from 3D-printable molds. Int. J. Comput. Assist. Radiol. Surg. 2019, 14, 1177–1186. [Google Scholar] [CrossRef]
- Holzhey, D.M.; Seeburger, J.; Misfeld, M.; Borger, M.A.; Mohr, F.W. Learning minimally invasive mitral valve surgery: A cumulative sum sequential probability analysis of 3895 operations from a single high-volume center. Circulation 2013, 128, 483–491. [Google Scholar] [CrossRef]
- Van Praet, K.M.; Stamm, C.; Sündermann, S.H.; Meyer, A.; Unbehaun, A.; Montagner, M.; Nazari Shafti, T.Z.; Jacobs, S.; Falk, V.; Kempfert, J. Minimally Invasive Surgical Mitral Valve Repair: State of the Art Review. Interv. Cardiol. 2018, 13, 14–19. [Google Scholar] [CrossRef]
- Olivieri, L.J.; Krieger, A.; Loke, Y.H.; Nath, D.S.; Kim, P.C.; Sable, C.A. Three-dimensional printing of intracardiac defects from three-dimensional echocardiographic images: Feasibility and relative accuracy. J. Am. Soc. Echocardiogr. 2015, 28, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, K.M.; Nielsen, J.C.; Uppu, S.C.; Srivastava, S.; Parness, I.A.; Sanz, J.; Nguyen, K. Use of 3-dimensional printing to demonstrate complex intracardiac relationships in double-outlet right ventricle for surgical planning. Circ. Cardiovasc. Imaging 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Garekar, S.; Bharati, A.; Chokhandre, M.; Mali, S.; Trivedi, B.; Changela, V.P.; Solanki, N.; Gaikwad, S.; Agarwal, V. Clinical Application and Multidisciplinary Assessment of Three Dimensional Printing in Double Outlet Right Ventricle With Remote Ventricular Septal Defect. World J. Pediatr. Congenit. Heart Surg. 2016, 7, 344–350. [Google Scholar] [CrossRef]
- Anwar, S.; Singh, G.K.; Varughese, J.; Nguyen, H.; Billadello, J.J.; Sheybani, E.F.; Woodard, P.K.; Manning, P.; Eghtesady, P. 3D Printing in Complex Congenital Heart Disease: Across a Spectrum of Age, Pathology, and Imaging Techniques. JACC Cardiovasc. Imaging 2017, 10, 953–956. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Boland, T.; D’Lima, D.D.; Lotz, M.K. Thermal inkjet printing in tissue engineering and regenerative medicine. Recent Pat. Drug Deliv. Formul. 2012, 6, 149–155. [Google Scholar] [CrossRef]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Giraud, M.N.; Guex, A.G.; Tevaearai, H.T. Cell therapies for heart function recovery: Focus on myocardial tissue engineering and nanotechnologies. Cardiol. Res. Pract. 2012, 2012, 971614. [Google Scholar] [CrossRef]
- Akhyari, P.; Kamiya, H.; Haverich, A.; Karck, M.; Lichtenberg, A. Myocardial tissue engineering: The extracellular matrix. Eur. J. Cardiothorac. Surg. 2008, 34, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Steinhoff, G.; Stock, U.; Karim, N.; Mertsching, H.; Timke, A.; Meliss, R.R.; Pethig, K.; Haverich, A.; Bader, A. Tissue engineering of pulmonary heart valves on allogenic acellular matrix conduits: In Vivo restoration of valve tissue. Circulation 2000, 102, iii-50–iii-55. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, N.; Bullock, A.J.; Macneil, S.; Janes, S.M.; Birchall, M. Tissue engineering airway mucosa: A systematic review. Laryngoscope 2014, 124, 961–968. [Google Scholar] [CrossRef]
- Klebe, R.J. Cytoscribing: A method for micropositioning cells and the construction of two- and three-dimensional synthetic tissues. Exp. Cell Res. 1988, 179, 362–373. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Miao, S.; Esworthy, T.; Zhou, X.; Lee, S.J.; Liu, C.; Yu, Z.X.; Fisher, J.P.; Mohiuddin, M.; Zhang, L.G. 3D bioprinting for cardiovascular regeneration and pharmacology. Adv. Drug Deliv. Rev. 2018, 132, 252–269. [Google Scholar] [CrossRef]
- Ozbolat, I.T.; Yu, Y. Bioprinting toward organ fabrication: Challenges and future trends. IEEE Trans. Biomed. Eng. 2013, 60, 691–699. [Google Scholar] [CrossRef]
- Visscher, D.O.; Farré-Guasch, E.; Helder, M.N.; Gibbs, S.; Forouzanfar, T.; van Zuijlen, P.P.; Wolff, J. Advances in Bioprinting Technologies for Craniofacial Reconstruction. Trends Biotechnol. 2016, 34, 700–710. [Google Scholar] [CrossRef]
- Chung, J.H.Y.; Naficy, S.; Yue, Z.; Kapsa, R.; Quigley, A.; Moulton, S.E.; Wallace, G.G. Bio-ink properties and printability for extrusion printing living cells. Biomater. Sci. 2013, 1, 763–773. [Google Scholar] [CrossRef]
- Gopinathan, J.; Noh, I. Recent trends in bioinks for 3D printing. Biomater. Res. 2018, 22, 11. [Google Scholar] [CrossRef]
- Ji, S.; Guvendiren, M. Recent Advances in Bioink Design for 3D Bioprinting of Tissues and Organs. Front. Bioeng. Biotechnol. 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Donderwinkel, I.; van Hest, J.C.M.; Cameron, N.R. Bio-inks for 3D bioprinting: Recent advances and future prospects. Polym. Chem. 2017, 8, 4451–4471. [Google Scholar] [CrossRef]
- Jang, J.; Park, H.J.; Kim, S.W.; Kim, H.; Park, J.Y.; Na, S.J.; Kim, H.J.; Park, M.N.; Choi, S.H.; Park, S.H.; et al. 3D printed complex tissue construct using stem cell-laden decellularized extracellular matrix bioinks for cardiac repair. Biomaterials 2017, 112, 264–274. [Google Scholar] [CrossRef]
- Jung, J.P.; Bhuiyan, D.B.; Ogle, B.M. Solid organ fabrication: Comparison of decellularization to 3D bioprinting. Biomater. Res. 2016, 20, 27. [Google Scholar] [CrossRef]
- Song, J.J.; Ott, H.C. Organ engineering based on decellularized matrix scaffolds. Trends Mol. Med. 2011, 17, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Moroni, F.; Mirabella, T. Decellularized matrices for cardiovascular tissue engineering. Am. J. Stem Cells 2014, 3, 1–20. [Google Scholar] [PubMed]
- Moldovan, N.I.; Hibino, N.; Nakayama, K. Principles of the Kenzan Method for Robotic Cell Spheroid-Based Three-Dimensional Bioprinting. Tissue Eng. Part B Rev. 2017, 23, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Moldovan, N.I. Progress in scaffold-free bioprinting for cardiovascular medicine. J. Cell. Mol. Med. 2018, 22, 2964–2969. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Unutmaz, D.; Ozbolat, I.T. Bioprinting towards Physiologically Relevant Tissue Models for Pharmaceutics. Trends Biotechnol. 2016, 34, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.S.; Nam, L.; Ong, K.; Krishnan, A.; Huang, C.Y.; Fukunishi, T.; Hibino, N. 3D and 4D Bioprinting of the Myocardium: Current Approaches, Challenges, and Future Prospects. BioMed Res. Int. 2018, 2018, 6497242. [Google Scholar] [CrossRef] [PubMed]
- Laflamme, M.A.; Murry, C.E. Heart regeneration. Nature 2011, 473, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Mathur, A.; Ma, Z.; Loskill, P.; Jeeawoody, S.; Healy, K.E. In Vitro cardiac tissue models: Current status and future prospects. Adv. Drug Deliv. Rev. 2016, 96, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Olivetti, G.; Melissari, M.; Capasso, J.M.; Anversa, P. Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circ. Res. 1991, 68, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Gaetani, R.; Doevendans, P.A.; Metz, C.H.; Alblas, J.; Messina, E.; Giacomello, A.; Sluijter, J.P. Cardiac tissue engineering using tissue printing technology and human cardiac progenitor cells. Biomaterials 2012, 33, 1782–1790. [Google Scholar] [CrossRef]
- Eschenhagen, T.; Fink, C.; Remmers, U.; Scholz, H.; Wattchow, J.; Weil, J.; Zimmermann, W.; Dohmen, H.H.; Schäfer, H.; Bishopric, N.; et al. Three-dimensional reconstitution of embryonic cardiomyocytes in a collagen matrix: A new heart muscle model system. FASEB J. 1997, 11, 683–694. [Google Scholar] [CrossRef]
- Chachques, J.C.; Trainini, J.C.; Lago, N.; Cortes-Morichetti, M.; Schussler, O.; Carpentier, A. Myocardial Assistance by Grafting a New Bioartificial Upgraded Myocardium (MAGNUM trial): Clinical feasibility study. Ann. Thorac. Surg. 2008, 85, 901–908. [Google Scholar] [CrossRef]
- Chachques, J.C.; Pradas, M.M.; Bayes-Genis, A.; Semino, C. Creating the bioartificial myocardium for cardiac repair: Challenges and clinical targets. Expert Rev. Cardiovasc. Ther. 2013, 11, 1701–1711. [Google Scholar] [CrossRef]
- Madonna, R.; Van Laake, L.W.; Botker, H.E.; Davidson, S.M.; De Caterina, R.; Engel, F.B.; Eschenhagen, T.; Fernandez-Aviles, F.; Hausenloy, D.J.; Hulot, J.S.; et al. ESC Working Group on Cellular Biology of the Heart: Position paper for Cardiovascular Research: Tissue engineering strategies combined with cell therapies for cardiac repair in ischaemic heart disease and heart failure. Cardiovasc. Res. 2019, 115, 488–500. [Google Scholar] [CrossRef]
- Chachques, J.C.; Lila, N.; Soler-Botija, C.; Martinez-Ramos, C.; Valles, A.; Autret, G.; Perier, M.C.; Mirochnik, N.; Monleon-Pradas, M.; Bayes-Genis, A.; et al. Elastomeric cardiopatch scaffold for myocardial repair and ventricular support. Eur. J. Cardiothorac. Surg. 2019. [Google Scholar] [CrossRef]
- Gaetani, R.; Barile, L.; Forte, E.; Chimenti, I.; Ionta, V.; Di Consiglio, A.; Miraldi, F.; Frati, G.; Messina, E.; Giacomello, A. New perspectives to repair a broken heart. Cardiovasc. Hematol. Agents Med. Chem. 2009, 7, 91–107. [Google Scholar] [CrossRef]
- Gaetani, R.; Feyen, D.A.; Verhage, V.; Slaats, R.; Messina, E.; Christman, K.L.; Giacomello, A.; Doevendans, P.A.; Sluijter, J.P. Epicardial application of cardiac progenitor cells in a 3D-printed gelatin/hyaluronic acid patch preserves cardiac function after myocardial infarction. Biomaterials 2015, 61, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Shin, S.R.; van Kempen, T.; Li, Y.C.; Ponraj, V.; Nasajpour, A.; Mandla, S.; Hu, N.; Liu, X.; Leijten, J.; et al. Gold Nanocomposite Bioink for Printing 3D Cardiac Constructs. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef] [PubMed]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef] [PubMed]
- Colosi, C.; Shin, S.R.; Manoharan, V.; Massa, S.; Costantini, M.; Barbetta, A.; Dokmeci, M.R.; Dentini, M.; Khademhosseini, A. Microfluidic Bioprinting of Heterogeneous 3D Tissue Constructs Using Low-Viscosity Bioink. Adv. Mater. 2016, 28, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Ashammakhi, N.; Ahadian, S.; Xu, C.; Montazerian, H.; Ko, H.; Nasiri, R.; Barros, N.; Khademhosseini, A. Bioinks and bioprinting technologies to make heterogeneous and biomimetic tissue constructs. Mater. Today Bio 2019, 1, 100008. [Google Scholar] [CrossRef] [PubMed]
- Gaebel, R.; Ma, N.; Liu, J.; Guan, J.; Koch, L.; Klopsch, C.; Gruene, M.; Toelk, A.; Wang, W.; Mark, P.; et al. Patterning human stem cells and endothelial cells with laser printing for cardiac regeneration. Biomaterials 2011, 32, 9218–9230. [Google Scholar] [CrossRef]
- Koike, N.; Fukumura, D.; Gralla, O.; Au, P.; Schechner, J.S.; Jain, R.K. Tissue engineering: Creation of long-lasting blood vessels. Nature 2004, 428, 138–139. [Google Scholar] [CrossRef]
- Pati, F.; Jang, J.; Ha, D.H.; Won Kim, S.; Rhie, J.W.; Shim, J.H.; Kim, D.H.; Cho, D.W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 2014, 5, 3935. [Google Scholar] [CrossRef]
- Pati, F.; Cho, D.W. Bioprinting of 3D Tissue Models Using Decellularized Extracellular Matrix Bioink. Methods Mol. Biol. 2017, 1612, 381–390. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, R.Y.; Park, B.W.; Lee, S.; Choi, S.W.; Park, J.H.; Choi, J.J.; Kim, S.W.; Jang, J.; Cho, D.W.; et al. Dual stem cell therapy synergistically improves cardiac function and vascular regeneration following myocardial infarction. Nat. Commun. 2019, 10, 3123. [Google Scholar] [CrossRef] [PubMed]
- Bejleri, D.; Streeter, B.W.; Nachlas, A.L.Y.; Brown, M.E.; Gaetani, R.; Christman, K.L.; Davis, M.E. A Bioprinted Cardiac Patch Composed of Cardiac-Specific Extracellular Matrix and Progenitor Cells for Heart Repair. Adv. Healthc. Mater. 2018, 7, e1800672. [Google Scholar] [CrossRef]
- Das, S.; Kim, S.W.; Choi, Y.J.; Lee, S.; Lee, S.H.; Kong, J.S.; Park, H.J.; Cho, D.W.; Jang, J. Decellularized extracellular matrix bioinks and the external stimuli to enhance cardiac tissue development in vitro. Acta Biomater. 2019, 95, 188–200. [Google Scholar] [CrossRef]
- Noor, N.; Shapira, A.; Edri, R.; Gal, I.; Wertheim, L.; Dvir, T. 3D Printing of Personalized Thick and Perfusable Cardiac Patches and Hearts. Adv. Sci. (Weinh) 2019, 6, 1900344. [Google Scholar] [CrossRef] [PubMed]
- Atmanli, A.; Domian, I.J. Generation of aligned functional myocardial tissue through microcontact printing. J. Vis. Exp. 2013, e50288. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.S.; Fukunishi, T.; Nashed, A.; Blazeski, A.; Zhang, H.; Hardy, S.; DiSilvestre, D.; Vricella, L.; Conte, J.; Tung, L.; et al. Creation of Cardiac Tissue Exhibiting Mechanical Integration of Spheroids Using 3D Bioprinting. J. Vis. Exp. 2017. [Google Scholar] [CrossRef]
- Ong, C.S.; Fukunishi, T.; Zhang, H.; Huang, C.Y.; Nashed, A.; Blazeski, A.; DiSilvestre, D.; Vricella, L.; Conte, J.; Tung, L.; et al. Biomaterial-Free Three-Dimensional Bioprinting of Cardiac Tissue using Human Induced Pluripotent Stem Cell Derived Cardiomyocytes. Sci. Rep. 2017, 7, 4566. [Google Scholar] [CrossRef] [PubMed]
- Farrar, E.J.; Butcher, J.T. Valvular heart diseases in the developing world: Developmental biology takes center stage. J. Heart Valve Dis. 2012, 21, 234–240. [Google Scholar]
- Vashistha, R.; Kumar, P.; Dangi, A.K.; Sharma, N.; Chhabra, D.; Shukla, P. Quest for cardiovascular interventions: Precise modeling and 3D printing of heart valves. J. Biol. Eng. 2019, 13, 12. [Google Scholar] [CrossRef]
- Aikawa, E.; Libby, P. A Rock and a Hard Place: Chiseling Away at the Multiple Mechanisms of Aortic Stenosis. Circulation 2017, 135, 1951–1955. [Google Scholar] [CrossRef]
- Butcher, J.T.; Mahler, G.J.; Hockaday, L.A. Aortic valve disease and treatment: The need for naturally engineered solutions. Adv. Drug Deliv. Rev. 2011, 63, 242–268. [Google Scholar] [CrossRef]
- Harris, C.; Croce, B.; Cao, C. Tissue and mechanical heart valves. Ann. Cardiothorac. Surg. 2015, 4, 399. [Google Scholar] [CrossRef] [PubMed]
- Vijayavenkataraman, S.; Yan, W.C.; Lu, W.F.; Wang, C.H.; Fuh, J.Y.H. 3D bioprinting of tissues and organs for regenerative medicine. Adv. Drug Deliv. Rev. 2018, 132, 296–332. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, T.C.; Cornelissen, C.; Koch, S.; Tschoeke, B.; Sachweh, J.S.; Schmitz-Rode, T.; Jockenhoevel, S. The In Vitro development of autologous fibrin-based tissue-engineered heart valves through optimised dynamic conditioning. Biomaterials 2007, 28, 3388–3397. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Kiick, K.L. Hybrid multicomponent hydrogels for tissue engineering. Macromol. Biosci. 2009, 9, 140–156. [Google Scholar] [CrossRef]
- Duan, B.; Hockaday, L.A.; Kang, K.H.; Butcher, J.T. 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. J. Biomed. Mater. Res. Part A 2013, 101, 1255–1264. [Google Scholar] [CrossRef]
- Mendez, M.G.; Kojima, S.; Goldman, R.D. Vimentin induces changes in cell shape, motility, and adhesion during the epithelial to mesenchymal transition. FASEB J. 2010, 24, 1838–1851. [Google Scholar] [CrossRef]
- Lu, S.H.; Lin, A.T.; Chen, K.K.; Chiang, H.S.; Chang, L.S. Characterization of smooth muscle differentiation of purified human skeletal muscle-derived cells. J. Cell. Mol. Med. 2011, 15, 587–592. [Google Scholar] [CrossRef][Green Version]
- Duan, B.; Kapetanovic, E.; Hockaday, L.A.; Butcher, J.T. Three-dimensional printed trileaflet valve conduits using biological hydrogels and human valve interstitial cells. Acta Biomater. 2014, 10, 1836–1846. [Google Scholar] [CrossRef] [PubMed]
- Hockaday, L.A.; Kang, K.H.; Colangelo, N.W.; Cheung, P.Y.; Duan, B.; Malone, E.; Wu, J.; Girardi, L.N.; Bonassar, L.J.; Lipson, H.; et al. Rapid 3D printing of anatomically accurate and mechanically heterogeneous aortic valve hydrogel scaffolds. Biofabrication 2012, 4, 035005. [Google Scholar] [CrossRef]
- Kang, L.H.; Armstrong, P.A.; Lee, L.J.; Duan, B.; Kang, K.H.; Butcher, J.T. Optimizing Photo-Encapsulation Viability of Heart Valve Cell Types in 3D Printable Composite Hydrogels. Ann. Biomed. Eng. 2017, 45, 360–377. [Google Scholar] [CrossRef]
- Fairbanks, B.D.; Schwartz, M.P.; Bowman, C.N.; Anseth, K.S. Photoinitiated polymerization of PEG-diacrylate with lithium phenyl-2,4,6-trimethylbenzoylphosphinate: Polymerization rate and cytocompatibility. Biomaterials 2009, 30, 6702–6707. [Google Scholar] [CrossRef] [PubMed]
- Duan, B.; Hockaday, L.A.; Kapetanovic, E.; Kang, K.H.; Butcher, J.T. Stiffness and adhesivity control aortic valve interstitial cell behavior within hyaluronic acid based hydrogels. Acta Biomater. 2013, 9, 7640–7650. [Google Scholar] [CrossRef] [PubMed]
- Rouillard, A.D.; Berglund, C.M.; Lee, J.Y.; Polacheck, W.J.; Tsui, Y.; Bonassar, L.J.; Kirby, B.J. Methods for photocrosslinking alginate hydrogel scaffolds with high cell viability. Tissue Eng. Part C Methods 2011, 17, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Alić, I.; Kosi, N.; Kapuralin, K.; Gorup, D.; Gajović, S.; Pochet, R.; Mitrečić, D. Neural stem cells from mouse strain Thy1 YFP-16 are a valuable tool to monitor and evaluate neuronal differentiation and morphology. Neurosci. Lett. 2016, 634, 32–41. [Google Scholar] [CrossRef]
- Konig, N.; Trolle, C.; Kapuralin, K.; Adameyko, I.; Mitrecic, D.; Aldskogius, H.; Shortland, P.J.; Kozlova, E.N. Murine neural crest stem cells and embryonic stem cell-derived neuron precursors survive and differentiate after transplantation in a model of dorsal root avulsion. J. Tissue Eng. Regen Med. 2017, 11, 129–137. [Google Scholar] [CrossRef]
- Mitrecic, D.; Nicaise, C.; Klimaschewski, L.; Gajovic, S.; Bohl, D.; Pochet, R. Genetically modified stem cells for the treatment of neurological diseases. Front. Biosci. (Elite Ed.) 2012, 4, 1170–1181. [Google Scholar] [CrossRef]
- Van der Valk, D.C.; van der Ven, C.F.T.; Blaser, M.C.; Grolman, J.M.; Wu, P.J.; Fenton, O.S.; Lee, L.H.; Tibbitt, M.W.; Andresen, J.L.; Wen, J.R.; et al. Engineering a 3D-Bioprinted Model of Human Heart Valve Disease Using Nanoindentation-Based Biomechanics. Nanomaterials 2018, 8, 296. [Google Scholar] [CrossRef]
- Peng, W.; Datta, P.; Ayan, B.; Ozbolat, V.; Sosnoski, D.; Ozbolat, I.T. 3D bioprinting for drug discovery and development in pharmaceutics. Acta Biomater. 2017, 57, 26–46. [Google Scholar] [CrossRef]
- Knowlton, S.; Tasoglu, S. A Bioprinted Liver-on-a-Chip for Drug Screening Applications. Trends Biotechnol. 2016, 34, 681–682. [Google Scholar] [CrossRef]
- Wrzesinski, K.; Fey, S.J. From 2D to 3D—A New Dimension for Modelling the Effect of Natural Products on Human Tissue. Curr. Pharm. Des. 2015, 21, 5605–5616. [Google Scholar] [CrossRef]
- Vanderburgh, J.; Sterling, J.A.; Guelcher, S.A. 3D Printing of Tissue Engineered Constructs for In Vitro Modeling of Disease Progression and Drug Screening. Ann. Biomed. Eng. 2017, 45, 164–179. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranti, S.; Després, J.P.; Fullerton, H.J.; Howard, V.J.; et al. Heart disease and stroke statistics—2015 update: A report from the American Heart Association. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Liu, J.; Zhu, W.; Tang, M.; Lawrence, N.; Yu, C.; Gou, M.; Chen, S. 3D bioprinting of functional tissue models for personalized drug screening and in vitro disease modeling. Adv. Drug Deliv. Rev. 2018, 132, 235–251. [Google Scholar] [CrossRef]
- Zhang, Y.S.; Arneri, A.; Bersini, S.; Shin, S.R.; Zhu, K.; Goli-Malekabadi, Z.; Aleman, J.; Colosi, C.; Busignani, F.; Dell’Erba, V.; et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials 2016, 110, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Lind, J.U.; Busbee, T.A.; Valentine, A.D.; Pasqualini, F.S.; Yuan, H.; Yadid, M.; Park, S.J.; Kotikian, A.; Nesmith, A.P.; Campbell, P.H.; et al. Instrumented cardiac microphysiological devices via multimaterial three-dimensional printing. Nat. Mater. 2017, 16, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lee, S.J.; Cheng, H.J.; Yoo, J.J.; Atala, A. 3D bioprinted functional and contractile cardiac tissue constructs. Acta Biomater. 2018, 70, 48–56. [Google Scholar] [CrossRef]
- Gu, Y.; Chen, X.; Lee, J.H.; Monteiro, D.A.; Wang, H.; Lee, W.Y. Inkjet printed antibiotic- and calcium-eluting bioresorbable nanocomposite micropatterns for orthopedic implants. Acta Biomater. 2012, 8, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Tarcha, P.J.; Verlee, D.; Hui, H.W.; Setesak, J.; Antohe, B.; Radulescu, D.; Wallace, D. The application of ink-jet technology for the coating and loading of drug-eluting stents. Ann. Biomed. Eng. 2007, 35, 1791–1799. [Google Scholar] [CrossRef]
- Sandler, N.; Salmela, I.; Fallarero, A.; Rosling, A.; Khajeheian, M.; Kolakovic, R.; Genina, N.; Nyman, J.; Vuorela, P. Towards fabrication of 3D printed medical devices to prevent biofilm formation. Int. J. Pharm. 2014, 459, 62–64. [Google Scholar] [CrossRef]
- Goyanes, A.; Wang, J.; Buanz, A.; Martínez-Pacheco, R.; Telford, R.; Gaisford, S.; Basit, A.W. 3D Printing of Medicines: Engineering Novel Oral Devices with Unique Design and Drug Release Characteristics. Mol. Pharm. 2015, 12, 4077–4084. [Google Scholar] [CrossRef]
- Kalejs, M.; von Segesser, L.K. Rapid prototyping of compliant human aortic roots for assessment of valved stents. Interact. Cardiovasc. Thorac. Surg. 2009, 8, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Biglino, G.; Verschueren, P.; Zegels, R.; Taylor, A.M.; Schievano, S. Rapid prototyping compliant arterial phantoms for in-vitro studies and device testing. J. Cardiovasc. Magn. Reson. 2013, 15, 2. [Google Scholar] [CrossRef] [PubMed]
- Mashari, A.; Knio, Z.; Jeganathan, J.; Montealegre-Gallegos, M.; Yeh, L.; Amador, Y.; Matyal, R.; Saraf, R.; Khabbaz, K.; Mahmood, F. Hemodynamic Testing of Patient-Specific Mitral Valves Using a Pulse Duplicator: A Clinical Application of Three-Dimensional Printing. J. Cardiothorac. Vasc. Anesth. 2016, 30, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, M.J.; Kasinpila, P.; Imbrie-Moore, A.M.; Wang, H.; Hironaka, C.E.; Koyano, T.K.; Fong, R.; Chiu, P.; Goldstone, A.B.; Steele, A.N.; et al. Modeling conduit choice for valve-sparing aortic root replacement on biomechanics with a 3-dimensional-printed heart simulator. J. Thorac. Cardiovasc. Surg. 2019, 158, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, X.; Li, W.; Di, Y.; Xing, Q.; Cao, Q. 3D printing and biocompatibility study of a new biodegradable occluder for cardiac defect. J. Cardiol. 2019, 74, 182–188. [Google Scholar] [CrossRef] [PubMed]
- 3D Printing Market by Offering (Printer, Material, Software, Service), Process (Binder Jetting, Direct Energy Deposition, Material Extrusion, Material Jetting, Powder Bed Fusion), Application, Vertical, Technology, and Geography—Global Forecast to 2024. Available online: https://www.marketsandmarkets.com/Market-Reports/3d-printing-market-1276.html (accessed on 25 February 2020).
- 3D Bioprinting Market by Component (3D Bioprinters (Microextrusion, Inkjet, Laser), Bioink (Natural, Synthetic, Hybrid)), Material (Hydrogel, Living, Cells), Application (Skin, Drug Research), End User (Biopharma, Academia)—Global Forecast to 2024. Available online: https://www.marketsandmarkets.com/Market-Reports/3d-bioprinting-market-170201787 (accessed on 21 February 2020).
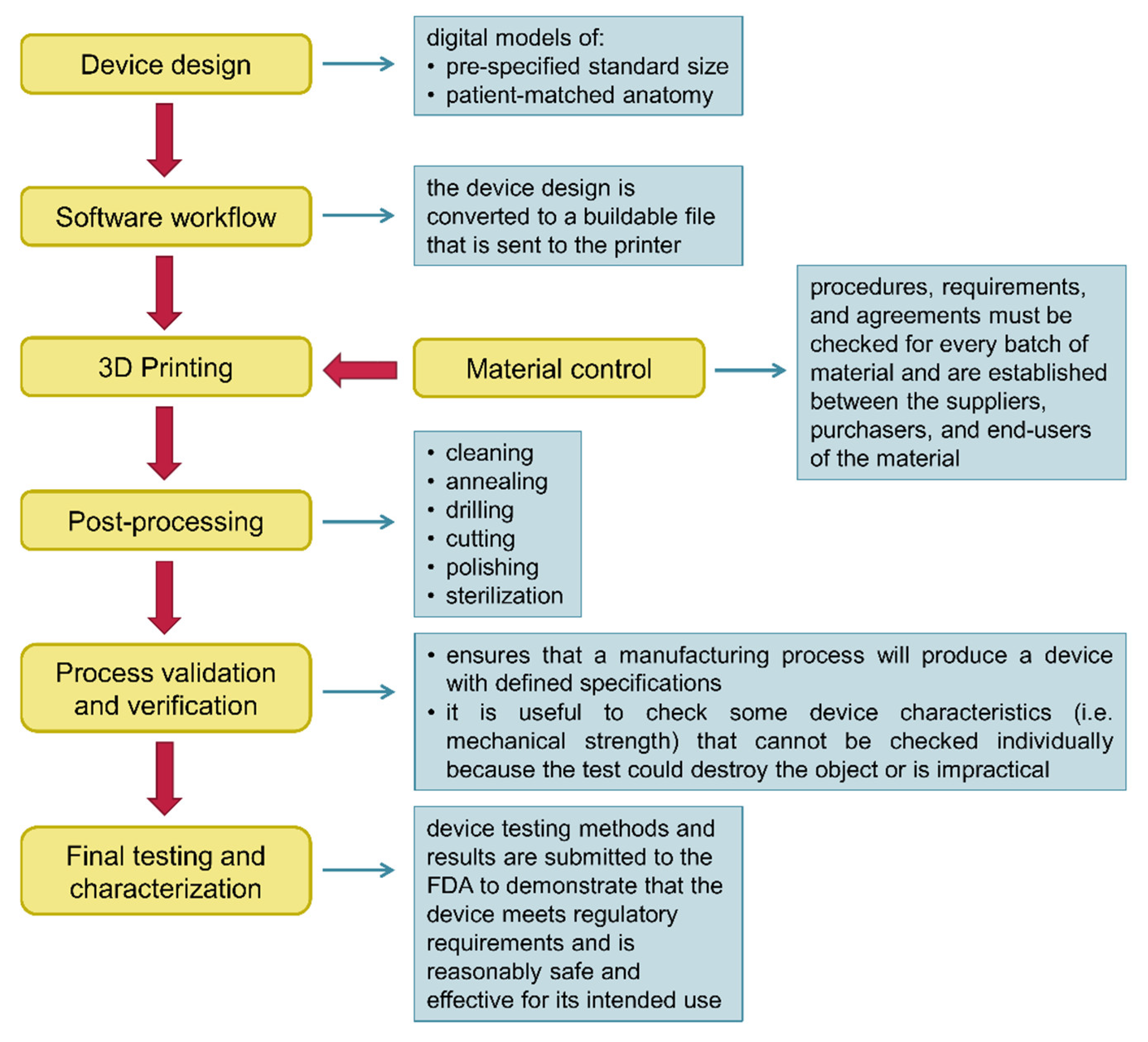
- Sweet, B.V.; Schwemm, A.K.; Parsons, D.M. Review of the processes for FDA oversight of drugs, medical devices, and combination products. J. Manag. Care Pharm. 2011, 17, 40–50. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Available online: https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device (accessed on 20 February 2020).
- Van Norman, G.A. Drugs, Devices, and the FDA: Part 2: An Overview of Approval Processes: FDA Approval of Medical Devices. JACC Basic Transl. Sci. 2016, 1, 277–287. [Google Scholar] [CrossRef]
- Hourd, P.; Medcalf, N.; Segal, J.; Williams, D.J. A 3D bioprinting exemplar of the consequences of the regulatory requirements on customized processes. Regen. Med. 2015, 10, 863–883. [Google Scholar] [CrossRef] [PubMed]
- Parvizi, N.; Woods, K. Regulation of medicines and medical devices: Contrasts and similarities. Clin. Med. 2014, 14, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Van Norman, G.A. Drugs and Devices: Comparison of European and U.S. Approval Processes. JACC Basic Transl. Sci. 2016, 1, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Classification of Medical Devices and Their Route to CE Markin. Available online: https://support.ce-check.eu/hc/en-us/articles/360008712879-Classification-Of-Medical-Devices-And-Their-Routes-To-CE-Marking (accessed on 25 February 2020).
- Technical Considerations for Additive Manufactured Medical Devices—Guidance for Industry and Food and Drug Administration Staff. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/technical-considerations-additive-manufactured-medical-devices-guidance-industry-and-food-and-drug (accessed on 20 February 2020).
- Ricles, L.M.; Coburn, J.C.; Di Prima, M.; Oh, S.S. Regulating 3D-printed medical products. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Rahman, Z.; Barakh Ali, S.F.; Ozkan, T.; Charoo, N.A.; Reddy, I.K.; Khan, M.A. Additive Manufacturing with 3D Printing: Progress from Bench to Bedside. AAPS J. 2018, 20, 101. [Google Scholar] [CrossRef] [PubMed]
- Horst, A.; McDonald, F.; Hutmacher, D.W. A clarion call for understanding regulatory processes for additive manufacturing in the health sector. Expert Rev. Med. Devices 2019, 16, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-López, C.; Agustí, A.; Obach, M.; Vallano, A. Regulatory Framework for Advanced Therapy Medicinal Products in Europe and United States. Front. Pharmacol. 2019, 10, 921. [Google Scholar] [CrossRef]
- Early Development Considerations for Innovative Combination Products. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/early-development-considerations-innovative-combination-products (accessed on 25 February 2020).
- Gilbert, F.; O’Connell, C.D.; Mladenovska, T.; Dodds, S. Print Me an Organ? Ethical and Regulatory Issues Emerging from 3D Bioprinting in Medicine. Sci. Eng. Ethics 2018, 24, 73–91. [Google Scholar] [CrossRef]
- Kritikos, M. 3D Bio-Printing for Medical and Enhancement Purposes: Legal and Ethical Aspects; EPRS, Ed.; EPRS: Brussels, Belgium, 2018. [Google Scholar]
- Meier, L.M.; Meineri, M.; Qua Hiansen, J.; Horlick, E.M. Structural and congenital heart disease interventions: The role of three-dimensional printing. Neth. Heart J. 2017, 25, 65–75. [Google Scholar] [CrossRef]
- Duan, B. State-of-the-Art Review of 3D Bioprinting for Cardiovascular Tissue Engineering. Ann. Biomed. Eng. 2017, 45, 195–209. [Google Scholar] [CrossRef]
- Hospodiuk, M.; Dey, M.; Sosnoski, D.; Ozbolat, I.T. The bioink: A comprehensive review on bioprintable materials. Biotechnol. Adv. 2017, 35, 217–239. [Google Scholar] [CrossRef]
- Liaw, C.Y.; Guvendiren, M. Current and emerging applications of 3D printing in medicine. Biofabrication 2017, 9, 024102. [Google Scholar] [CrossRef]
- Qasim, M.; Haq, F.; Kang, M.H.; Kim, J.H. 3D printing approaches for cardiac tissue engineering and role of immune modulation in tissue regeneration. Int. J. Nanomed. 2019, 14, 1311–1333. [Google Scholar] [CrossRef]
- Yang, G.H.; Yeo, M.; Koo, Y.W.; Kim, G.H. 4D Bioprinting: Technological Advances in Biofabrication. Macromol. Biosci. 2019, 19, e1800441. [Google Scholar] [CrossRef] [PubMed]
- Kirillova, A.; Maxson, R.; Stoychev, G.; Gomillion, C.T.; Ionov, L. 4D Biofabrication Using Shape-Morphing Hydrogels. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]


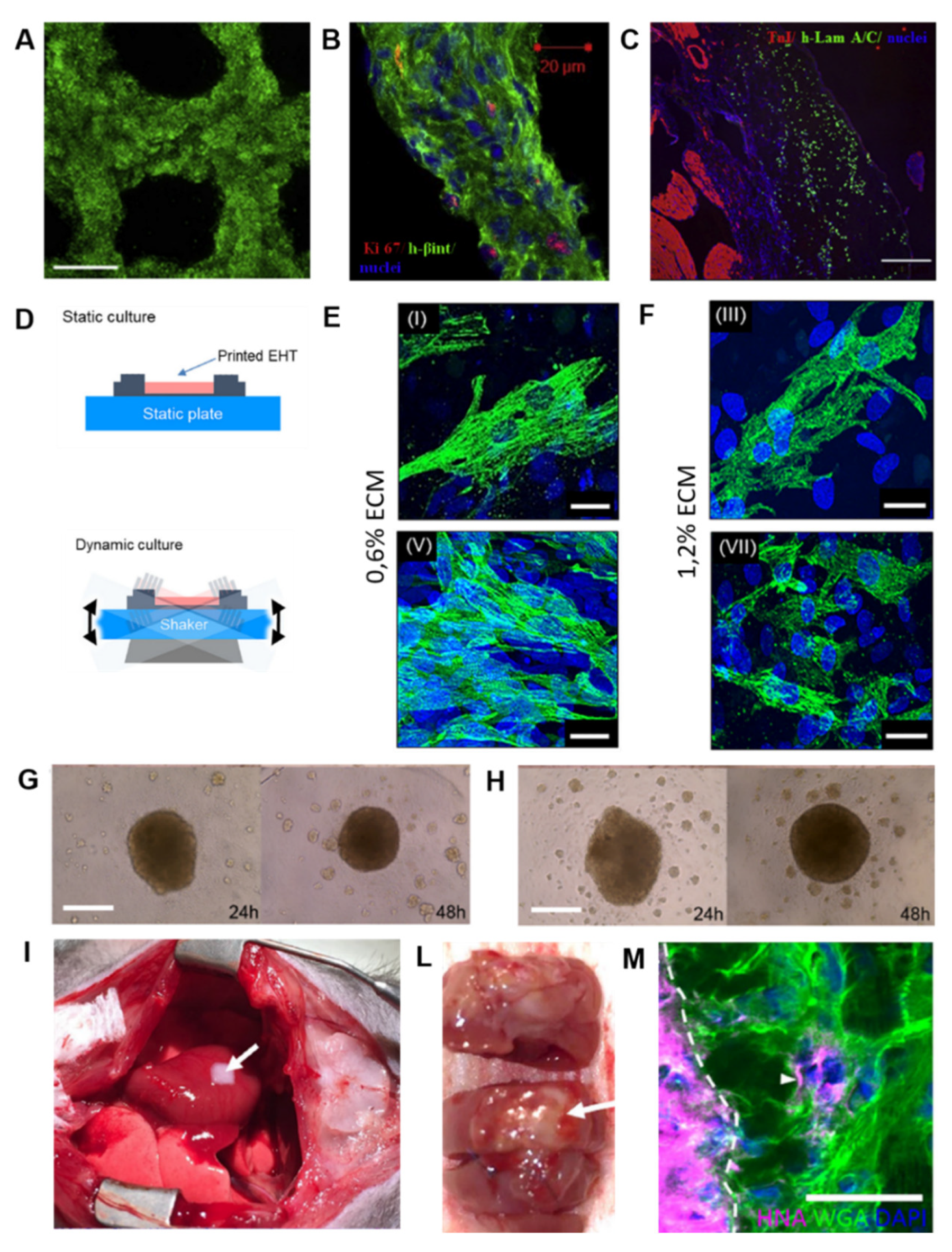
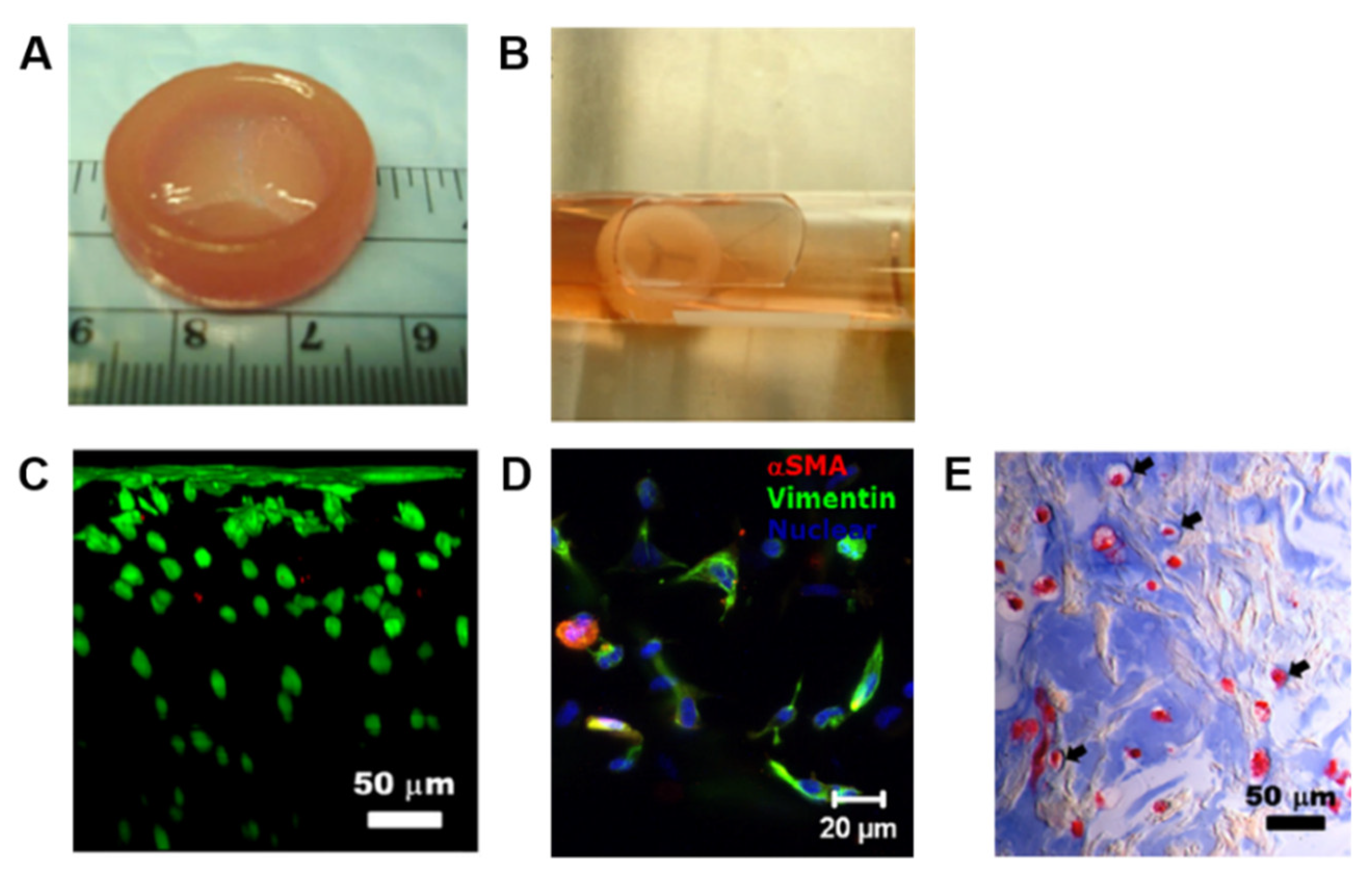
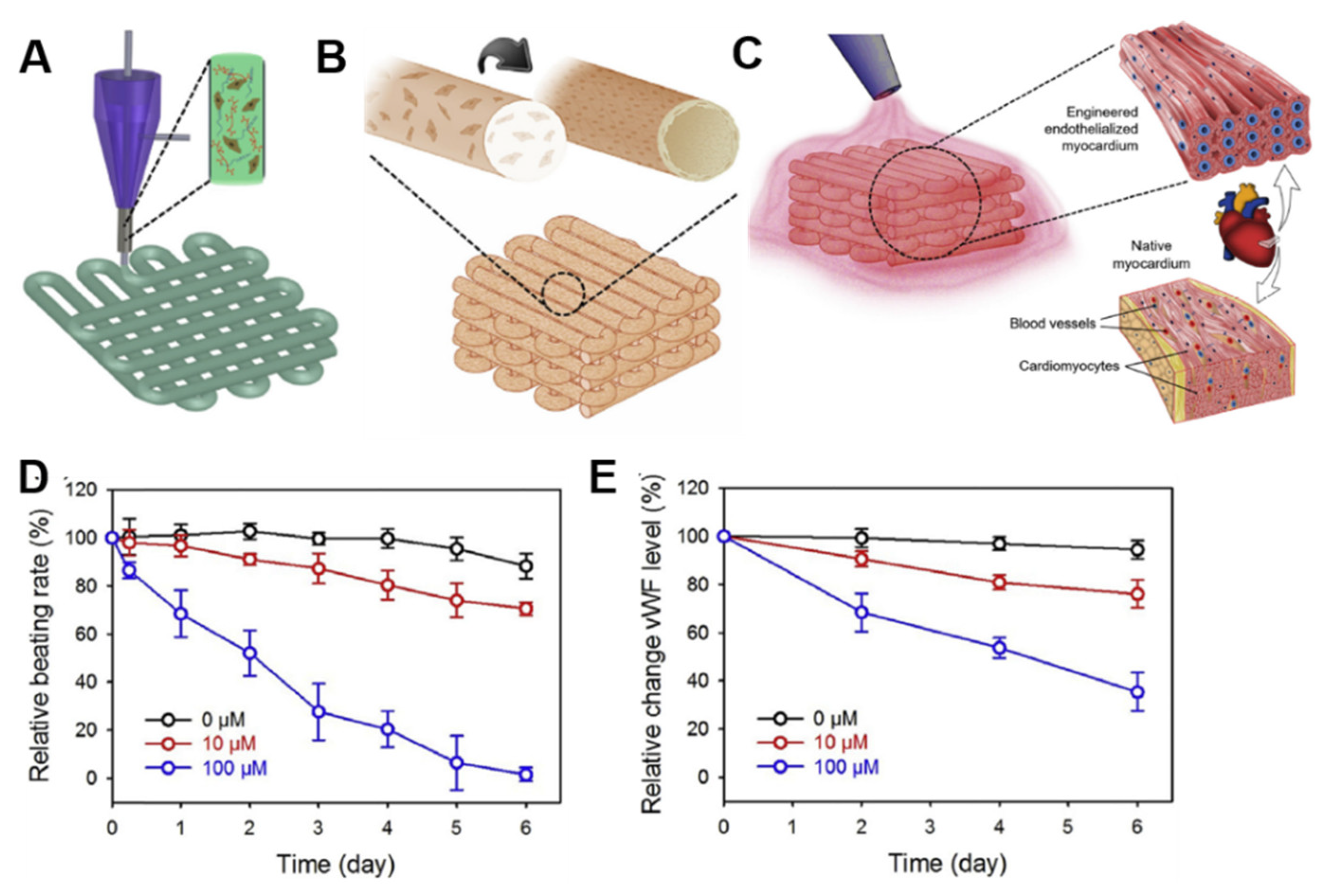


| Bioink | Cell (Concentration) | 3D Bioprinting Technique | Significance | Ref. |
|---|---|---|---|---|
| alginate | human CMPCs 1 (30 × 106/mL) | extrusion-based bioprinting | cells were able to migrate out of the alginate matrix and fully colonize a matrigel layer, forming tubular-like structures in vitro | [66] |
| HA 2 and gelatin | human CMPCs (30 × 106/mL) | extrusion-based bioprinting | the scaffold was able to support cell survival, engraftment, and differentiation; in addition, it improved cardiac function after epicardial transplantation in a mouse model of myocardial infarction | [73] |
| GelMA 3 and alginate incorporating GNRs 4 | CMs 5 (1 × 106/mL) and cardiac FBs 6 (1.5 × 106/mL) | extrusion-based bioprinting | the presence of a conductive nanomaterial (GNRs) into the hydrogel improved the electrical propagation between adjacent CMs, that finally resulted in a synchronized contraction of the bioprinted construct in vitro | [74] |
| PEUU 7 | human ECs 8 (4 × 106) and human MSCs 9 (2 × 106) | laser-based bioprinting | co-implantation of ECs and MSCs in a defined printed pattern enhanced the vascularization of the construct and improved cardiac function after acute myocardial infarction in rats | [78] |
| decellularized ECM 10 from the LV 11 of porcine heart | rat myoblast cells (from 1 to 5 × 106/mL) | extrusion-based bioprinting | the construct possessed a microarchitecture having a native-like organization | [80,81] |
| decellularized ECM from the LV of 6-month-old Korea domestic pig | human CPCs 13 (5 × 106/mL) and human MSCs (5 × 106/mL) | extrusion-based bioprinting | the use of two different bioink formulations, one containing CPCs and the other made of MSCs supplemented with VEGF 14, allowed for the development of pre-vascularized cardiac patch | [55] |
| decellularized ECM from the LV of porcine heart | CMs derived from human iPSCs 15 (1 × 106/rat) and human MSCs (1 × 106/mL) | extrusion-based bioprinting | the strategy of intramyocardially applying CMs derived from human iPSCs-CM and epicardially implanting a cardiac patch containing human MSCs significantly improved cardiac function and vessel formation in a rat model of myocardial infarction | [82] |
| decellularized ECM from porcine ventricular tissue combined with GelMA | pediatric human CPCs (3 × 106/mL) | extrusion-based bioprinting | possibility of using the cardiac patch in pediatric patients suffering from RV 16 failure, or for treating adult myocardial dysfunction | [83] |
| decellularized ECM from the LV of porcine heart or collagen | neonatal rat CMs (2 × 107/mL) | extrusion-based bioprinting | the culture conditions (dynamic versus static) are decisive factors for the structural arrangement of CMs, and affect gene expression and the related signaling pathways | [84] |
| decellularized human omental tissue | human iPSCs-CMs 17 (2 × 107/mL) and ECs (2 × 107/mL) | extrusion-based bioprinting | possibility of generating vascularized patches that fully match the immunological, biochemical and anatomical properties of any individual | [85] |
| scaffold-free | cardiospheres (33 × 105 cells/cardiosphere) composed of human iPSCs-CMs, FBs and ECs at different ratios | 3D bioprinting on a needle array | the biomaterial-free 3D printed cardiac patch produced from human iPSCs showed spontaneous beating, electrical integration of the cardiospheres, and in vivo engraftment and vascularization | [87,88] |
| Bioink | Cell (Concentration) | 3D Bioprinting Technique | Significance | Ref. |
|---|---|---|---|---|
| alginate/gelatin | human aortic root smooth muscle cells (SMCs) 1 (1 × 107/mL) and porcine aortic valve interstitial cells (VICs) 2 (1 × 107/mL) | extrusion-based bioprinting | the use of a dual syringe system, each containing a defined cell population (SMCs or VICs), allowed for the creation of a 3D printed aortic valve conduit complete of valve root and leaflet | [97] |
| Me-HA 3 and Me-Gel 4 | HAVICs 5 (5 × 106/mL) | extrusion-based bioprinting | a heart valve conduit was bioprinted with acellular root and three leaflets encapsulating HAVICs; by varying the concentration of the hydrogel formulations it was possible to modulate the behavior of the encapsulated cells | [100] |
| MEGEL/PEGDA/alginate 6 | HAVICs, human aortic valve sinus smooth muscle cells (HASSMCs) 7, and human adipose derived mesenchymal stem cells (HADMSCs) 8 (2.5 × 106/mL) | extrusion-based bioprinting | variable combinations of photoinitiator type (Irgagure 2959 versus VA086) and concentration, and light intensity (2–136 mW/cm2) can be used to optimize cell viability during 3D printing for multiple cell types | [102] |
| GelMA/HAMA 9 | human VICs (10 × 106/mL) | extrusion-based bioprinting | a 3D model of calcific aortic valve disease (CAVD) 10 was created recapitulating leaflet layer-specific mechanical properties which is useful for studying the valvular mechanobiology and for high-throughput drug screening | [109] |
| Bioink | Cell (Concentration) | Drug Tested | Significance | Ref. |
|---|---|---|---|---|
| alginate/GelMA 1 | human ECs 2 (1 × 107/mL) and neonatal rat CMs 3 (1 × 106/mL) | doxorubicin (anti-cancer drug) | the doxorubicin dose-concentration response was evaluated in the endothelialized-myocardium-on-a-chip both as beating rate in CMs and as relative expression levels of vWF 4 in ECs | [116] |
| dextran, TPU 5, CB 6:TPU, Ag:PA 7, soft PDMS 8, rigid PDMS | iPSCs-CMs 9 (220 k/cm2) | verapamil (cardiac drug), isoproterenol (cardiac drug) | the engineered microtissues displayed inotropic responses to verapamil and isoproterenol comparable to data obtained from engineered 3D neonatal rat ventricular myocardial tissues and isolated postnatal whole rat hearts | [117] |
| fibrin-based composite hydrogel (20 mg/mL fibrinogen, 30 mg/mL gelatin, 20 μg/mL aprotinin, 10% glycerol, and 3 mg/mL HA 10) | rat CMs (10 × 106/mL) | epinephrine (cardiac drug) and carbachol (cardiac drug) | the bioprinted cardiac tissues physiologically responded to the tested cardiac drugs by modulating the CMs beating frequency; reversible effects of the drugs were observed once these were removed from the bioprinted tissues, thus confirming the effectiveness of these constructs as in vitro 3D tissue models | [118] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gardin, C.; Ferroni, L.; Latremouille, C.; Chachques, J.C.; Mitrečić, D.; Zavan, B. Recent Applications of Three Dimensional Printing in Cardiovascular Medicine. Cells 2020, 9, 742. https://doi.org/10.3390/cells9030742
Gardin C, Ferroni L, Latremouille C, Chachques JC, Mitrečić D, Zavan B. Recent Applications of Three Dimensional Printing in Cardiovascular Medicine. Cells. 2020; 9(3):742. https://doi.org/10.3390/cells9030742
Chicago/Turabian StyleGardin, Chiara, Letizia Ferroni, Christian Latremouille, Juan Carlos Chachques, Dinko Mitrečić, and Barbara Zavan. 2020. "Recent Applications of Three Dimensional Printing in Cardiovascular Medicine" Cells 9, no. 3: 742. https://doi.org/10.3390/cells9030742
APA StyleGardin, C., Ferroni, L., Latremouille, C., Chachques, J. C., Mitrečić, D., & Zavan, B. (2020). Recent Applications of Three Dimensional Printing in Cardiovascular Medicine. Cells, 9(3), 742. https://doi.org/10.3390/cells9030742







