The Path towards Predicting Evolution as Illustrated in Yeast Cell Polarity
Abstract
1. Introduction
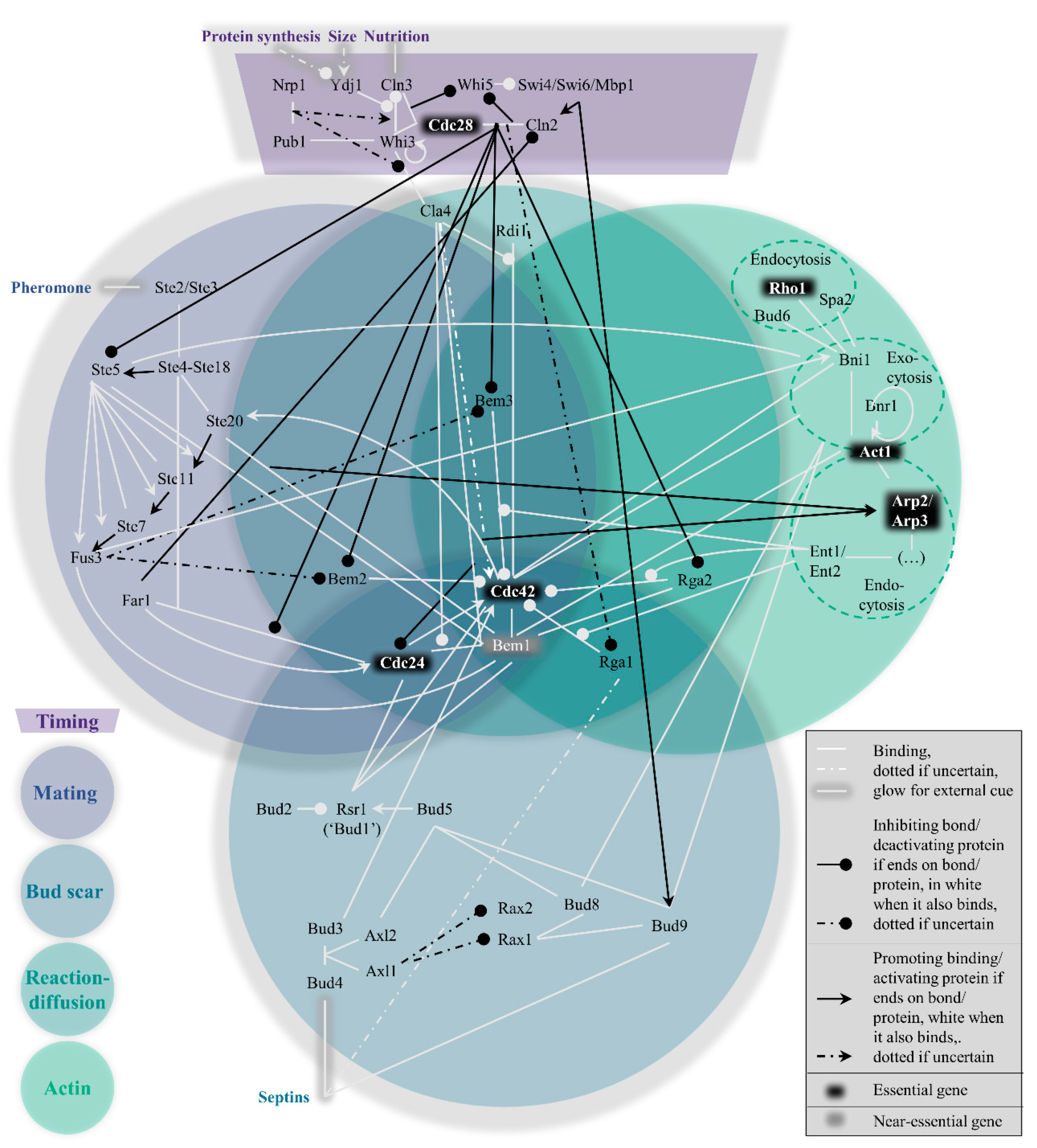
2. Polarity Overview
2.1. Timing: The Control Knob
2.2. Mating: Heavily Cross-Linked
2.3. Bud Scar: Mostly Modular and Ordered
2.4. Reaction−Diffusion: Ample Redundancy
2.5. Actin: The Mysterious Auxiliary Layer
3. Case Studies
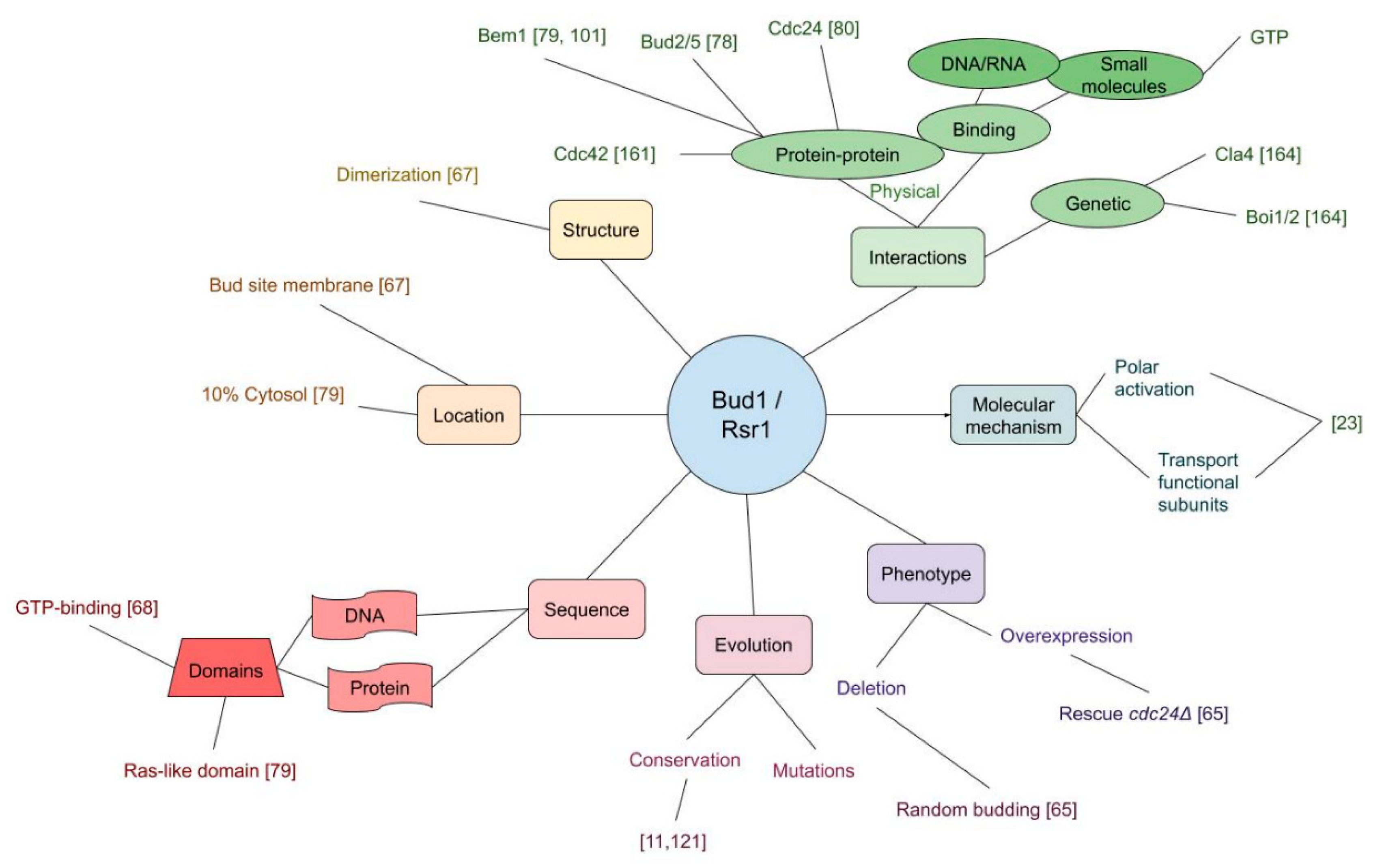
3.1. Bud Scar Proteins
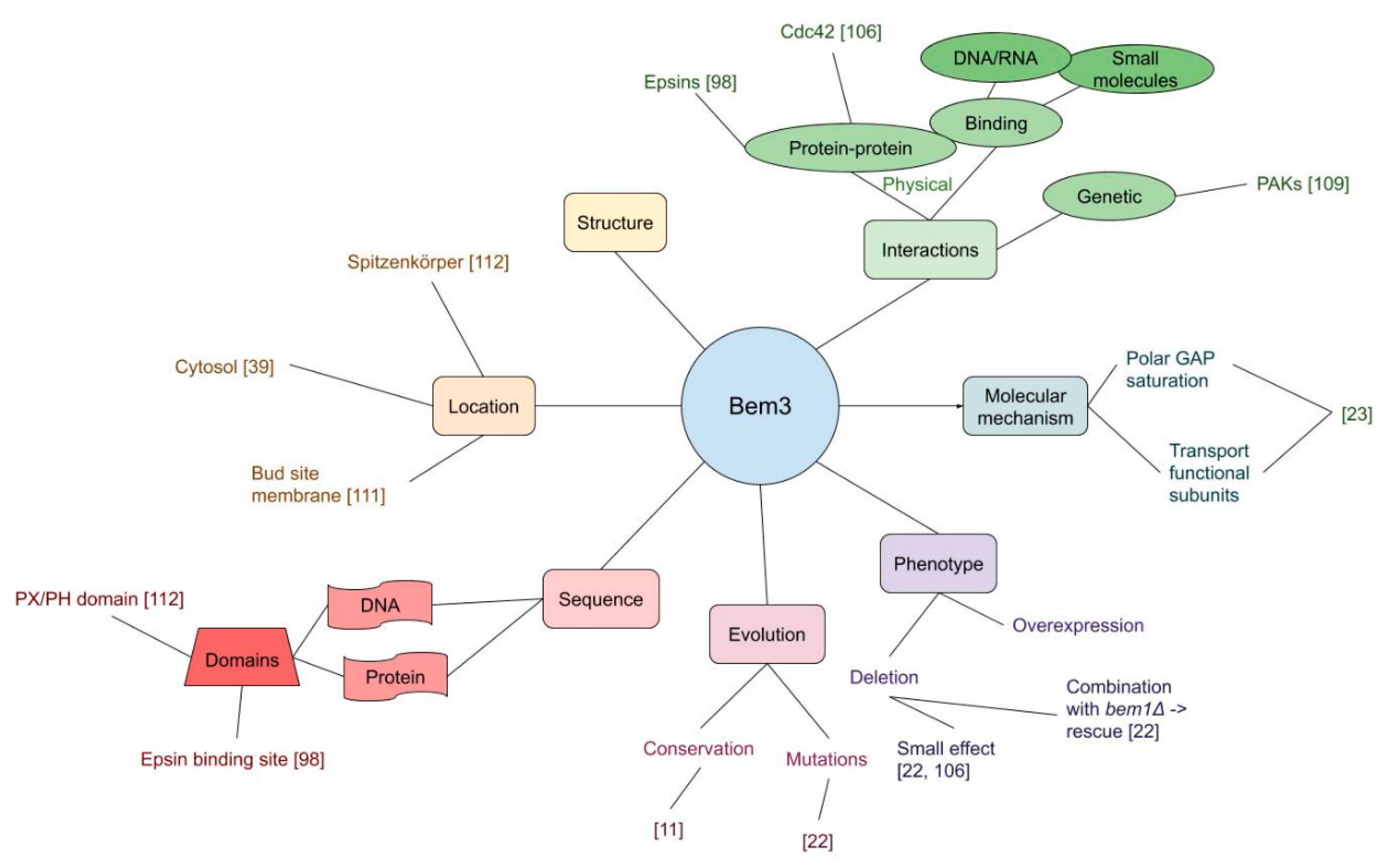
3.2. GAPs
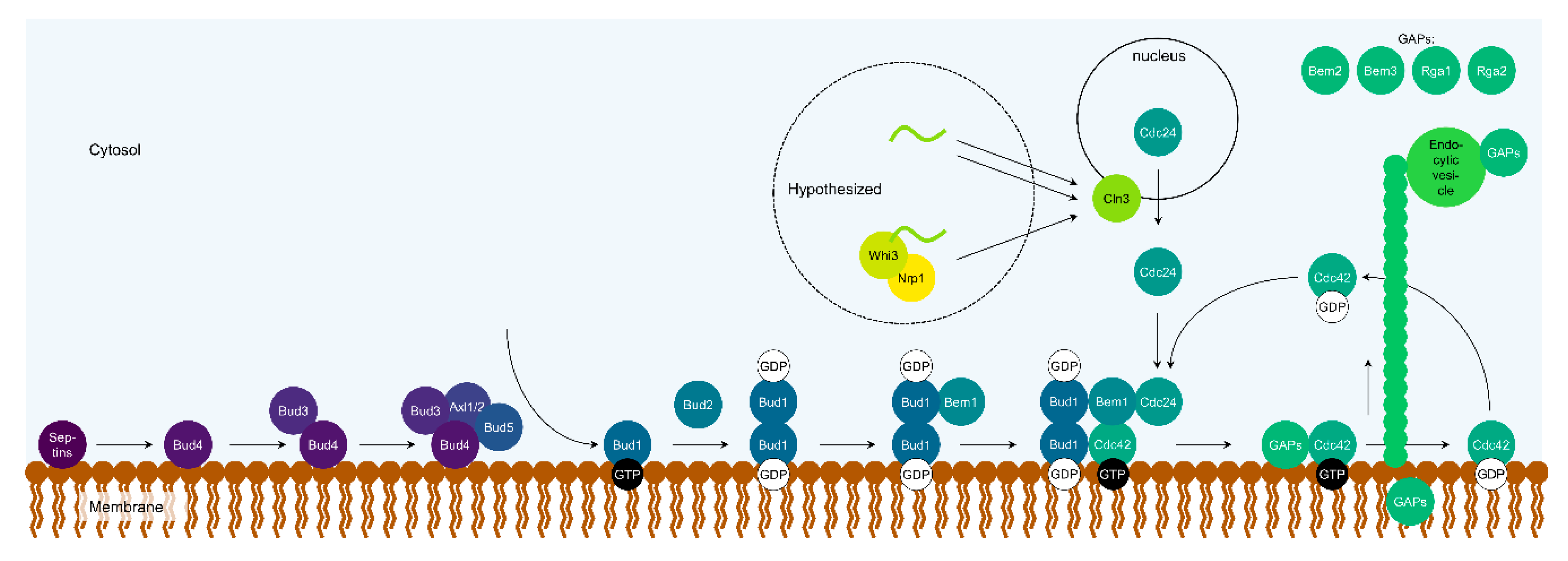
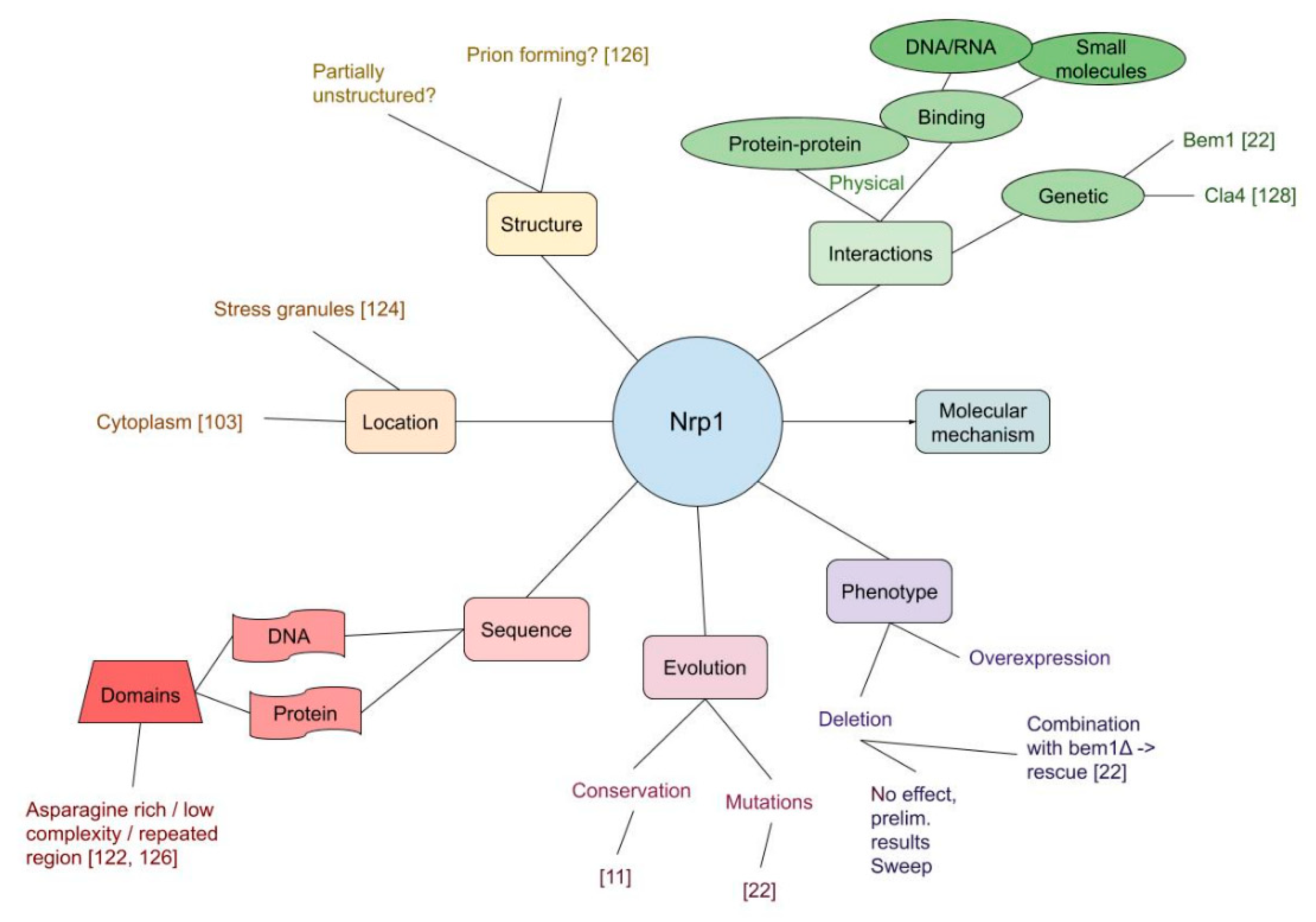
3.3. Nrp1
4. Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Description | Reference |
|---|---|
| Nrp1 binds Pub1 | [153] |
| Nrp1-Pub1 promotes Whi3-Cln3 bond (putative) | |
| Pub1 binds Whi3 | [154] |
| Nrp1-Pub1 inhibits Whi3-Cla4 (mRNA) binding (putative) | |
| Whi3 binds and inhibits Cln3 | [33,155] |
| Whi3 binds Cdc28 | [34] |
| Cln3 binds Cdc28 | [34] |
| Cdc28-Cln3 inhibits activation of Whi5 | [36] |
| Whi5 binds and inhibits activation of Swi4/Swi6/Mbp1 | [36] |
| Cdc28-Cln2 inhibits activation of Whi5 as well | [36,44] |
| Swi4/Swi6/Mbp1 promotes activity of Cln2 | [35] |
| Whi3 binds Cla4 (mRNA) | [156] |
| Whi3 can aggregate in stress granules | [33] |
| Cdc28 binds Cln2 | [157] |
| Cdc28-Cln2 inhibits Ste5 | [43] |
| Cdc28-Cln2 inhibits Far1-Cdc24 binding | [42] |
| Cdc28-Cln2 inhibits Bem2 | [39] |
| Cdc28-Cln2 inhibits Rga1 | [38] |
| Cdc28-Cln2 inhibits Bem3 | [39] |
| Cdc28-Cln2 inhibits Rga2 | [40,41] |
| Far1 inhibits Cln2 | [44,45] |
| Swi4/Swi6/Mbp1 promotes activity of Bud9 | [82] |
| Ydj1 inhibits Whi3 | [32] |
| Protein synthesis inhibits Ydj1 | [32] |
| Size promotes Ydj1 | [32] |
| Nutrition promotes Cln3 | [31] |
| Pheromone activates Ste2/Ste3 | [46,47] |
| Ste2/Ste3 binds Ste4-Ste18 | [48] |
| Ste4-Ste18 binds Ste20 | [158] |
| Ste4-Ste18 binds Cdc24-Far1 | [59] |
| Ste20 activates Ste11 | [51] |
| Ste5 binds Ste11 | [50] |
| Ste11 activates Ste7 | [51] |
| Ste5 binds Ste7 | [50] |
| Ste7 activates Fus3 | [52] |
| Ste5 binds Fus3 | [50] |
| Ste5 binds Bem1 | [57] |
| Fus3 inhibits Bem3 activity (putative) | [39] |
| Fus3 inhibits Bem2 activity (putative) | [39] |
| Far1 binds Cdc24 | [59] |
| Far1 binds and promotes Cdc24 activity | [159] |
| Cdc42 binds and promotes Ste20 activity | [54] |
| Bem1 binds Far1 | [58] |
| Bem1 binds Ste20 | [57] |
| Fus3 binds and promotes Bni1 | [61] |
| Ste5 binds the exocytosis network | [56] |
| Ste4 binds Ste5 | [51] |
| Ste5 promotes Ste20 activating Ste11 | [51] |
| Ste5 promotes Ste11 activating Ste7 | [51] |
| Ste5 promotes Ste7 activating Fus3 | [51,52] |
| Septins bind Bud4 | [73] |
| Bud3 binds Bud4 | [72] |
| Bud3-Bud4 binds Axl1 | [72] |
| Bud3-Bud4 binds Axl2 | [72] |
| Axl2 binds Bud5 | [72] |
| Septins bind Rga1 (putative) | [81] |
| Septins bind Bud9 | [82] |
| Axl1 inhibits Rax2 (putative) | [74] |
| Axl1 inhibits Rax1 (putative) | [74] |
| Rax1 binds Bud8 | [75,76] |
| Rax1 binds Bud9 | [75,76] |
| Bud8 binds exocytosis network | [82] |
| Bud2 binds and inhibits Bud1 | [78] |
| Bud5 binds and activates Bud1 | [78,160] |
| Bud1 binds Cdc24 | [80] |
| Bud1 binds Bem1 | [79,101] |
| Bud9 binds exocytosis network | [82] |
| Bud1 binds Cdc42 | [67,161] |
| Bud3 binds Cdc42 | [105] |
| Bud5 bind Bud8 | [76] |
| Bud5 binds Bud9 | [76] |
| Rdi1 binds Cdc42 | [162] |
| Rga2 binds and deactivates Cdc42 | [109] |
| Rga2 binds Bem1 | [40] |
| Rga1 binds and deactivates Cdc42 | [109] |
| Cdc42 binds Bem1 | [163] |
| Cla4 binds Bem1 | [164] |
| Bem1 binds Cdc24, activates it | [60,84,164] |
| Cla4 inhibits binding Bem1-Cdc24 | [89,163,165] |
| Cdc24 binds and activates Cdc42 | [106,164] |
| Bem2 binds and deactivates Cdc42 | [110] |
| Cla4 binds and activates Cdc42 | [88] |
| Bem3 binds and deactivates Cdc42 | [106] |
| Cla4 inhibits Cdc42-Rdi1 bond | [88] |
| Cdc24 binds Bem1-Cla4 | [164] |
| Bni1 binds Act1 | [166] |
| Act1 self-organizes | [167] |
| Bnr1 binds Act1 | [166] |
| Exocytosis network binds Cdc42 | [94] |
| Act1 binds Arp2/Arp3 | [115] |
| Arp2/Arp3 interacts with intermediate | [168,169] |
| Intermediates interact with Ent1/Ent2 | [168,169] |
| Ent2 inhibits Bem3 deactivating Cdc42 | [113] |
| Ent1/Ent2 inhibits Rga2 deactivating Cdc42 | [98] |
| Cdc42-Cla4 activates Arp2/3 | [115] |
| Cdc42-Ste20 activates Arp2/3 | [115] |
| Ent1/Ent2 inhibits Rga1 deactivating Cdc42 | [98] |
| Bni1 binds Cdc42 | [55] |
| Act1 binds Bem1 | [57] |
| Bud6 binds Bni1 | [93] |
| Rho1 binds Bni1 | [93] |
| Spa2 binds Bni1 | [93] |
References
- Greaves, M.; Maley, C.C. Clonal evolution in cancer. Nature 2012, 481, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, M.; VanderSluis, B.; Koch, E.N.; Baryshnikova, A.; Pons, C.; Tan, G.; Wang, W.; Usaj, M.; Hanchard, J.; Lee, S.D. A global genetic interaction network maps a wiring diagram of cellular function. Science 2016, 353. [Google Scholar] [CrossRef] [PubMed]
- Fraser, H.B.; Wall, D.P.; Hirsh, A.E. A simple dependence between protein evolution rate and the number of protein-protein interactions. BMC Evol. Biol. 2003, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ratmann, O.; Wiuf, C.; Pinney, J.W. From evidence to inference: Probing the evolution of protein interaction networks. HFSP J. 2009, 3, 290–306. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J. Evolution by gene duplication: An update. Trends Ecol. Evol. 2003, 18, 292–298. [Google Scholar] [CrossRef]
- Näsvall, J.; Sun, L.; Roth, J.R.; Andersson, D.I. Real-time evolution of new genes by innovation, amplification, and divergence. Science 2012, 338, 384–387. [Google Scholar] [CrossRef]
- Magadum, S.; Banerjee, U.; Murugan, P.; Gangapur, D.; Ravikesavan, R. Gene duplication as a major force in evolution. J. Genet. 2013, 92, 155–161. [Google Scholar] [CrossRef]
- Eyre-Walker, A.; Keightley, P.D. The distribution of fitness effects of new mutations. Nat. Rev. Genet. 2007, 8, 610–618. [Google Scholar] [CrossRef]
- Thoday, J.M. Non-Darwinian “evolution” and biological progress. Nature 1975, 255, 675–677. [Google Scholar] [CrossRef]
- Khan, A.I.; Dinh, D.M.; Schneider, D.; Lenski, R.E.; Cooper, T.F. Negative epistasis between beneficial mutations in an evolving bacterial population. Science 2011, 332, 1193–1196. [Google Scholar] [CrossRef]
- Diepeveen, E.T.; Gehrmann, T.; Pourquié, V.; Abeel, T.; Laan, L. Patterns of Conservation and Diversification in the Fungal Polarization Network. Genome Biol. Evol. 2018, 10, 1765–1782. [Google Scholar] [CrossRef] [PubMed]
- Winzeler, E.A. Functional Characterization of the S. cerevisiae Genome by Gene Deletion and Parallel Analysis. Science 1999, 285, 901–906. [Google Scholar] [CrossRef]
- Deutschbauer, A.M.; Jaramillo, D.F.; Proctor, M.; Kumm, J.; Hillenmeyer, M.E.; Davis, R.W.; Nislow, C.; Giaever, G. Mechanisms of Haploinsufficiency Revealed by Genome-Wide Profiling in Yeast. Genetics 2005, 169, 1915–1925. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.G.; Arkowitz, R.A. Cell polarization in budding and fission yeasts. FEMS Microbiol. Rev. 2014, 38, 228–253. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, M.; Baryshnikova, A.; Bellay, J.; Kim, Y.; Spear, E.D.; Sevier, C.S.; Ding, H.; Koh, J.L.; Toufighi, K.; Mostafavi, S.; et al. The genetic landscape of a cell. Science 2010, 327, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.T.; Guimera, R.; Li, H.; Pinto, I.M.; Sales-Pardo, M.; Wai, S.C.; Rubinstein, B.; Li, R. Modular coherence of protein dynamics in yeast cell polarity system. Proc. Natl. Acad. Sci. USA 2011, 108, 7647–7652. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Bork, P. Evolution of biomolecular networks—Lessons from metabolic and protein interactions. Nat. Rev. Mol. Cell Biol. 2009, 10, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Tuch, B.B.; Galgoczy, D.J.; Hernday, A.D.; Li, H.; Johnson, A.D. The evolution of combinatorial gene regulation in fungi. PLoS Biol. 2008, 6, e38. [Google Scholar] [CrossRef]
- Tanay, A.; Regev, A.; Shamir, R. Conservation and evolvability in regulatory networks: The evolution of ribosomal regulation in yeast. Proc. Natl. Acad. Sci. USA 2005, 102, 7203–7208. [Google Scholar] [CrossRef]
- Dhar, R.; Sagesser, R.; Weikert, C.; Wagner, A. Yeast Adapts to a Changing Stressful Environment by Evolving Cross-Protection and Anticipatory Gene Regulation. Mol. Biol. Evol. 2013, 30, 573–588. [Google Scholar] [CrossRef]
- Slot, J.C.; Rokas, A. Multiple GAL pathway gene clusters evolved independently and by different mechanisms in fungi. Proc. Natl. Acad. Sci. USA 2010, 107, 10136–10141. [Google Scholar] [CrossRef] [PubMed]
- Laan, L.; Koschwanez, J.H.; Murray, A.W. Evolutionary adaptation after crippling cell polarization follows reproducible trajectories. eLife 2015, 4, e09638. [Google Scholar] [CrossRef] [PubMed]
- Brauns, F.; De la Cruz, L.M.I.; Daalman, W.K.-G.; De Bruin, I.; Halatek, J.; Laan, L.; Frey, E. Adaptability and evolution of the cell polarization machinery in budding yeast. bioRxiv 2020. [Google Scholar] [CrossRef]
- Freisinger, T.; Klünder, B.; Johnson, J.; Müller, N.; Pichler, G.; Beck, G.; Costanzo, M.; Boone, C.; Cerione, R.A.; Frey, E.; et al. Establishment of a robust single axis of cell polarity by coupling multiple positive feedback loops. Nat. Commun. 2013, 4, 1807. [Google Scholar] [CrossRef] [PubMed]
- Howell, A.S.; Jin, M.; Wu, C.-F.; Zyla, T.R.; Elston, T.C.; Lew, D.J. Negative Feedback Enhances Robustness in the Yeast Polarity Establishment Circuit. Cell 2012, 149, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Daalman, W.K.-G.; Laan, L. Predicting an epistasis-rich genotype-phenotype map with a coarse-grained bottom-up model of budding yeast polarity. bioRxiv 2020. [Google Scholar] [CrossRef]
- Breen, M.S.; Kemena, C.; Vlasov, P.K.; Notredame, C.; Kondrashov, F.A. Epistasis as the primary factor in molecular evolution. Nature 2012, 490, 535–538. [Google Scholar] [CrossRef]
- Yu, M.K.; Kramer, M.; Dutkowski, J.; Srivas, R.; Licon, K.; Kreisberg, J.F.; Ng, C.T.; Krogan, N.; Sharan, R.; Ideker, T. Translation of Genotype to Phenotype by a Hierarchy of Cell Subsystems. Cell Syst. 2016, 2, 77–88. [Google Scholar] [CrossRef]
- Park, H.-O.; Bi, E. Central Roles of Small GTPases in the Development of Cell Polarity in Yeast and Beyond. Microbiol. Mol. Biol. Rev. 2007, 71, 48–96. [Google Scholar] [CrossRef]
- Hall, A. The cellular functions of small GTP-binding proteins. Science 1990, 249, 635–640. [Google Scholar] [CrossRef]
- Gallego, C.; Garí, E.; Colomina, N.; Herrero, E.; Aldea, M. The Cln3 cyclin is down-regulated by translational repression and degradation during the G1 arrest caused by nitrogen deprivation in budding yeast. EMBO J. 1997, 16, 7196–7206. [Google Scholar] [CrossRef] [PubMed]
- Vergés, E.; Colomina, N.; Garí, E.; Gallego, C.; Aldea, M. Cyclin Cln3 Is Retained at the ER and Released by the J Chaperone Ydj1 in Late G1 to Trigger Cell Cycle Entry. Mol. Cell 2007, 26, 649–662. [Google Scholar] [CrossRef]
- Cai, Y.; Futcher, B. Effects of the Yeast RNA-Binding Protein Whi3 on the Half-Life and Abundance of CLN3 mRNA and Other Targets. PLoS ONE 2013, 8, e84630. [Google Scholar] [CrossRef]
- Wang, H.; Garí, E.; Vergés, E.; Gallego, C.; Aldea, M. Recruitment of Cdc28 by Whi3 restricts nuclear accumulation of the G1 cyclin–Cdk complex to late G1. EMBO J. 2004, 23, 180–190. [Google Scholar] [CrossRef]
- Koch, C.; Schleiffer, A.; Ammerer, G.; Nasmyth, K. Switching transcription on and off during the yeast cell cycle: Cln/Cdc28 kinases activate bound transcription factor SBF (Swi4/Swi6) at start, whereas Clb/Cdc28 kinases displace it from the promoter in G2. Genes Dev. 1996, 10, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, M.; Nishikawa, J.L.; Tang, X.; Millman, J.S.; Schub, O.; Breitkreuz, K.; Dewar, D.; Rupes, I.; Andrews, B.; Tyers, M. CDK activity antagonizes Whi5, an inhibitor of G1/S transcription in yeast. Cell 2004, 117, 899–913. [Google Scholar] [CrossRef] [PubMed]
- Stuart, D.; Wittenberg, C. CLN3, not positive feedback, determines the timing of CLN2 transcription in cycling cells. Genes Dev. 1995, 9, 2780–2794. [Google Scholar] [CrossRef] [PubMed]
- Archambault, V.; Chang, E.J.; Drapkin, B.J.; Cross, F.R.; Chait, B.T.; Rout, M.P. Targeted proteomic study of the cyclin-Cdk module. Mol. Cell 2004, 14, 699–711. [Google Scholar] [CrossRef]
- Knaus, M.; Pelli-Gulli, M.-P.; Van Drogen, F.; Springer, S.; Jaquenoud, M.; Peter, M. Phosphorylation of Bem2p and Bem3p may contribute to local activation of Cdc42p at bud emergence. EMBO J. 2007, 26, 4501–4513. [Google Scholar] [CrossRef]
- McCusker, D.; Denison, C.; Anderson, S.; Egelhofer, T.A.; Yates, J.R.; Gygi, S.P.; Kellogg, D.R. Cdk1 coordinates cell-surface growth with the cell cycle. Nat. Cell Biol. 2007, 9, 506–515. [Google Scholar] [CrossRef]
- Sopko, R.; Huang, D.; Smith, J.C.; Figeys, D.; Andrews, B.J. Activation of the Cdc42p GTPase by cyclin-dependent protein kinases in budding yeast. EMBO J. 2007, 26, 4487–4500. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Gulli, M.-P.; Peter, M. Nuclear sequestration of the exchange factor Cdc24 by Far1 regulates cell polarity during yeast mating. Nat. Cell Biol. 2000, 2, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Garrenton, L.S.; Braunwarth, A.; Irniger, S.; Hurt, E.; Kunzler, M.; Thorner, J. Nucleus-Specific and Cell Cycle-Regulated Degradation of Mitogen-Activated Protein Kinase Scaffold Protein Ste5 Contributes to the Control of Signaling Competence. Mol. Cell. Biol. 2009, 29, 582–601. [Google Scholar] [CrossRef] [PubMed]
- Doncic, A.; Falleur-Fettig, M.; Skotheim, J.M. Distinct Interactions Select and Maintain a Specific Cell Fate. Mol. Cell 2011, 43, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Doncic, A.; Skotheim, J.M. Feedforward Regulation Ensures Stability and Rapid Reversibility of a Cellular State. Mol. Cell 2013, 50, 856–868. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Blumer, K.J.; Reneke, J.E.; Thorner, J. The STE2 gene product is the ligand-binding component of the alpha-factor receptor of Saccharomyces cerevisiae. J. Biol. Chem. 1988, 263, 10836–10842. [Google Scholar] [PubMed]
- Hagen, D.C.; McCaffrey, G.; Sprague, G.F. Evidence the yeast STE3 gene encodes a receptor for the peptide pheromone a factor: Gene sequence and implications for the structure of the presumed receptor. Proc. Natl. Acad. Sci. USA 1986, 83, 1418–1422. [Google Scholar] [CrossRef]
- Whiteway, M.; Hougan, L.; Dignard, D.; Thomas, D.Y.; Bell, L.; Saari, G.C.; Grant, F.J.; O’Hara, P.; MacKay, V.L. The STE4 and STE18 genes of yeast encode potential β and γ subunits of the mating factor receptor-coupled G protein. Cell 1989, 56, 467–477. [Google Scholar] [CrossRef]
- Hirschman, J.E.; De Zutter, G.S.; Simonds, W.F.; Jenness, D.D. The Gβγ Complex of the Yeast Pheromone Response Pathway SUBCELLULAR FRACTIONATION AND PROTEIN-PROTEIN INTERACTIONS. J. Biol. Chem. 1997, 272, 240–248. [Google Scholar] [CrossRef]
- Chol, K.-Y.; Satterberg, B.; Lyons, D.M.; Elion, E.A. Ste5 tethers multiple protein kinases in the MAP kinase cascade required for mating in S. cerevisiae. Cell 1994, 78, 499–512. [Google Scholar] [CrossRef]
- Feng, Y.; Song, L.Y.; Kincaid, E.; Mahanty, S.K.; Elion, E.A. Functional binding between Gβ and the LIM domain of Ste5 is required to activate the MEKK Ste11. Curr. Biol. 1998, 8, 267–282. [Google Scholar] [CrossRef]
- Good, M.; Tang, G.; Singleton, J.; Reményi, A.; Lim, W.A. The Ste5 Scaffold Directs Mating Signaling by Catalytically Unlocking the Fus3 MAP Kinase for Activation. Cell 2009, 136, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, W.; Simpson, D.M.; Elion, E.A. Cdc24 Regulates Nuclear Shuttling and Recruitment of the Ste5 Scaffold to a Heterotrimeric G Protein in Saccharomyces cerevisiae. J. Biol. Chem. 2005, 280, 13084–13096. [Google Scholar] [CrossRef] [PubMed]
- Lamson, R.E.; Winters, M.J.; Pryciak, P.M. Cdc42 Regulation of Kinase Activity and Signaling by the Yeast p21-Activated Kinase Ste20. Mol. Cell. Biol. 2002, 22, 2939–2951. [Google Scholar] [CrossRef]
- Evangelista, M.; Blundell, K.; Longtine, M.S.; Chow, C.J.; Adames, N.; Pringle, J.R.; Peter, M.; Boone, C. Bni1p, a yeast formin linking cdc42p and the actin cytoskeleton during polarized morphogenesis. Science 1997, 276, 118–122. [Google Scholar] [CrossRef]
- Qi, M.; Elion, E.A. Formin-induced actin cables are required for polarized recruitment of the Ste5 scaffold and high level activation of MAPK Fus3. J. Cell Sci. 2005, 118, 2837–2848. [Google Scholar] [CrossRef][Green Version]
- Leeuw, T.; Fourest-Lieuvin, A.; Wu, C.; Chenevert, J.; Clark, K.; Whiteway, M.; Thomas, D.Y.; Leberer, E. Pheromone Response in Yeast: Association of Bem1p with Proteins of the MAP Kinase Cascade and Actin. Science 1995, 1210–1213. [Google Scholar] [CrossRef]
- Lyons, D.M.; Mahanty, S.K.; Choi, K.-Y.; Manandhar, M.; Elion, E.A. The SH3-domain protein Bem1 coordinates mitogen-activated protein kinase cascade activation with cell cycle control in Saccharomyces cerevisiae. Mol. Cell. Biol. 1996, 16, 4095–4106. [Google Scholar] [CrossRef]
- Butty, A.-C.; Pryciak, P.M.; Huang, L.S.; Herskowitz, I.; Peter, M. The role of Far1p in linking the heterotrimeric G protein to polarity establishment proteins during yeast mating. Science 1998, 282, 1511–1516. [Google Scholar] [CrossRef]
- Peterson, J. Interactions between the bud emergence proteins Bem1p and Bem2p and Rho- type GTPases in yeast. J. Cell Biol. 1994, 127, 1395–1406. [Google Scholar] [CrossRef]
- Matheos, D.; Metodiev, M.; Muller, E.; Stone, D.; Rose, M.D. Pheromone-induced polarization is dependent on the Fus3p MAPK acting through the formin Bni1p. J. Cell Biol. 2004, 165, 99–109. [Google Scholar] [CrossRef]
- Kim, H.B. Cellular morphogenesis in the Saccharomyces cerevisiae cell cycle: Localization of the CDC3 gene product and the timing of events at the budding site. J. Cell Biol. 1991, 112, 535–544. [Google Scholar] [CrossRef]
- Flescher, E.G. Components required for cytokinesis are important for bud site selection in yeast. J. Cell Biol. 1993, 122, 373–386. [Google Scholar] [CrossRef]
- Freifelder, D. Bud position in Saccharomyces cerevisiae. J. Bacteriol. 1960, 80, 567. [Google Scholar] [CrossRef]
- Chant, J.; Herskowitz, I. Genetic control of bud site selection in yeast by a set of gene products that constitute a morphogenetic pathway. Cell 1991, 65, 1203–1212. [Google Scholar] [CrossRef]
- Chant, J.; Corrado, K.; Pringle, J.R.; Herskowitz, I. Yeast BUD5, encoding a putative GDP-GTP exchange factor, is necessary for bud site selection and interacts with bud formation gene BEM1. Cell 1991, 65, 1213–1224. [Google Scholar] [CrossRef][Green Version]
- Kang, P.J.; Béven, L.; Hariharan, S.; Park, H.-O. The Rsr1/Bud1 GTPase interacts with itself and the Cdc42 GTPase during bud-site selection and polarity establishment in budding yeast. Mol. Biol. Cell 2010, 21, 3007–3016. [Google Scholar] [CrossRef]
- Bender, A.; Pringle, J.R. Multicopy suppression of the cdc24 budding defect in yeast by CDC42 and three newly identified genes including the ras-related gene RSR1. Proc. Natl. Acad. Sci. USA 1989, 86, 9976–9980. [Google Scholar] [CrossRef]
- Fujita, A.; Oka, C.; Arikawa, Y.; Katagai, T.; Tonouchi, A.; Kuhara, S.; Misumi, Y. A yeast gene necessary for bud-site selection encodes a protein similar to insulin-degrading enzymes. Nature 1994, 372, 567. [Google Scholar] [CrossRef]
- Chant, J.; Mischke, M.; Mitchell, E.; Herskowitz, I.; Pringle, J.R. Role of Bud3p in producing the axial budding pattern of yeast. J. Cell Biol. 1995, 129, 767–778. [Google Scholar] [CrossRef]
- Sanders, S.L.; Herskowitz, I. The BUD4 protein of yeast, required for axial budding, is localized to the mother/BUD neck in a cell cycle-dependent manner. J. Cell Biol. 1996, 134, 413–427. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.J.; Angerman, E.; Jung, C.-H.; Park, H.-O. Bud4 mediates the cell-type-specific assembly of the axial landmark in budding yeast. J. Cell Sci. 2012, 125, 3840–3849. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.J.; Hood-DeGrenier, J.K.; Park, H.-O. Coupling of septins to the axial landmark by Bud4 in budding yeast. J. Cell Sci. 2013, 126, 1218–1226. [Google Scholar] [CrossRef] [PubMed]
- Bi, E.; Park, H.-O. Cell Polarization and Cytokinesis in Budding Yeast. Genetics 2012, 191, 347–387. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.J.; Angerman, E.; Nakashima, K.; Pringle, J.R.; Park, H.-O. Interactions among Rax1p, Rax2p, Bud8p, and Bud9p in marking cortical sites for bipolar bud-site selection in yeast. Mol. Biol. Cell 2004, 15, 5145–5157. [Google Scholar] [CrossRef]
- Krappmann, A.-B.; Taheri, N.; Heinrich, M.; Mösch, H.-U. Distinct domains of yeast cortical tag proteins Bud8p and Bud9p confer polar localization and functionality. Mol. Biol. Cell 2007, 18, 3323–3339. [Google Scholar] [CrossRef][Green Version]
- Marston, A.L.; Chen, T.; Yang, M.C.; Belhumeur, P.; Chant, J. A localized GTPase exchange factor, Bud5, determines the orientation of division axes in yeast. Curr. Biol. 2001, 11, 803–807. [Google Scholar] [CrossRef]
- Bender, A. Genetic evidence for the roles of the bud-site-selection genes BUD5 and BUD2 in control of the Rsr1p (Bud1p) GTPase in yeast. Proc. Natl. Acad. Sci. USA 1993, 90, 9926–9929. [Google Scholar] [CrossRef]
- Park, H.-O.; Bi, E.; Pringle, J.R.; Herskowitz, I. Two active states of the Ras-related Bud1/Rsr1 protein bind to different effectors to determine yeast cell polarity. Proc. Natl. Acad. Sci. USA 1997, 94, 4463–4468. [Google Scholar] [CrossRef]
- Zheng, Y.; Bender, A.; Cerione, R.A. Interactions among proteins involved in bud-site selection and bud-site assembly in Saccharomyces cerevisiae. J. Biol. Chem. 1995, 270, 626–630. [Google Scholar] [CrossRef]
- Tong, Z.; Gao, X.-D.; Howell, A.S.; Bose, I.; Lew, D.J.; Bi, E. Adjacent positioning of cellular structures enabled by a Cdc42 GTPase-activating protein mediated zone of inhibition. J. Cell Biol. 2007, 179, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Schenkman, L.R.; Caruso, C.; Pagé, N.; Pringle, J.R. The role of cell cycle–regulated expression in the localization of spatial landmark proteins in yeast. J. Cell Biol. 2002, 156, 829–841. [Google Scholar] [CrossRef] [PubMed]
- Irazoqui, J.E.; Gladfelter, A.S.; Lew, D.J. Scaffold-mediated symmetry breaking by Cdc42p. Nat. Cell Biol. 2003, 5, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Rubinstein, B.; Mendes Pinto, I.; Slaughter, B.D.; Unruh, J.R.; Li, R. Independence of symmetry breaking on Bem1-mediated autocatalytic activation of Cdc42. J. Cell Biol. 2013, 202, 1091–1106. [Google Scholar] [CrossRef]
- Ozbudak, E.M.; Becskei, A.; Van Oudenaarden, A. A System of Counteracting Feedback Loops Regulates Cdc42p Activity during Spontaneous Cell Polarization. Dev. Cell 2005, 9, 565–571. [Google Scholar] [CrossRef]
- Goryachev, A.B.; Pokhilko, A.V. Dynamics of Cdc42 network embodies a Turing-type mechanism of yeast cell polarity. FEBS Lett. 2008, 582, 1437–1443. [Google Scholar] [CrossRef]
- Klünder, B.; Freisinger, T.; Wedlich-Söldner, R.; Frey, E. GDI-Mediated Cell Polarization in Yeast Provides Precise Spatial and Temporal Control of Cdc42 Signaling. PLoS Comput. Biol. 2013, 9, e1003396. [Google Scholar] [CrossRef]
- Tiedje, C.; Sakwa, I.; Just, U.; Höfken, T. The rho gdi rdi1 regulates rho gtpases by distinct mechanisms. Mol. Biol. Cell 2008, 19, 2885–2896. [Google Scholar] [CrossRef]
- Kuo, C.-C.; Savage, N.S.; Chen, H.; Wu, C.-F.; Zyla, T.R.; Lew, D.J. Inhibitory GEF Phosphorylation Provides Negative Feedback in the Yeast Polarity Circuit. Curr. Biol. 2014, 24, 753–759. [Google Scholar] [CrossRef]
- Cvrckova, F.; De Virgilio, C.; Manser, E.; Pringle, J.R.; Nasmyth, K. Ste20-like protein kinases are required for normal localization of cell growth and for cytokinesis in budding yeast. Genes Dev. 1995, 9, 1817–1830. [Google Scholar] [CrossRef]
- Daalman, W.K.-G. Coupling of Genotype-Phenotype Maps to Noise-Driven Adaptation, Showcased in Yeast Polarity; TU Delft: Delft, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Pruyne, D.; Gao, L.; Bi, E.; Bretscher, A. Stable and dynamic axes of polarity use distinct formin isoforms in budding yeast. Mol. Biol. Cell 2004, 15, 4971–4989. [Google Scholar] [CrossRef] [PubMed]
- Prosser, D.C.; Drivas, T.G.; Maldonado-Báez, L.; Wendland, B. Existence of a novel clathrin-independent endocytic pathway in yeast that depends on Rho1 and formin. J. Cell Biol. 2011, 195, 657–671. [Google Scholar] [CrossRef] [PubMed]
- Wedlich-Soldner, R.; Altschuler, S.; Wu, L.; Li, R. Spontaneous cell polarization through actomyosin-based delivery of the Cdc42 GTPase. Science 2003, 299, 1231–1235. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, B.D.; Unruh, J.R.; Das, A.; Smith, S.E.; Rubinstein, B.; Li, R. Non-uniform membrane diffusion enables steady-state cell polarization via vesicular trafficking. Nat. Commun. 2013, 4, 1380. [Google Scholar] [CrossRef] [PubMed]
- Layton, A.T.; Savage, N.S.; Howell, A.S.; Carroll, S.Y.; Drubin, D.G.; Lew, D.J. Modeling Vesicle Traffic Reveals Unexpected Consequences for Cdc42p-Mediated Polarity Establishment. Curr. Biol. 2011, 21, 184–194. [Google Scholar] [CrossRef]
- Watson, L.J.; Rossi, G.; Brennwald, P. Quantitative Analysis of Membrane Trafficking in Regulation of Cdc42 Polarity. Traffic 2014, 15, 1330–1343. [Google Scholar] [CrossRef]
- Aguilar, R.C.; Longhi, S.A.; Shaw, J.D.; Yeh, L.-Y.; Kim, S.; Schön, A.; Freire, E.; Hsu, A.; McCormick, W.K.; Watson, H.A.; et al. Epsin N-terminal homology domains perform an essential function regulating Cdc42 through binding Cdc42 GTPase-activating proteins. Proc. Natl. Acad. Sci. USA 2006, 103, 4116–4121. [Google Scholar] [CrossRef]
- Gerhart, J.; Kirschner, M. The theory of facilitated variation. Proc. Natl. Acad. Sci. USA 2007, 104, 8582–8589. [Google Scholar] [CrossRef]
- Wedlich-Soldner, R.; Li, R. Yeast and fungal morphogenesis from an evolutionary perspective. Semin. Cell Dev. Biol. 2008, 19, 224–233. [Google Scholar] [CrossRef][Green Version]
- Miller, K.E.; Lo, W.-C.; Chou, C.-S.; Park, H.-O. Temporal regulation of cell polarity via the interaction of the Ras GTPase Rsr1 and the scaffold protein Bem1. Mol. Biol. Cell 2019, 30, 2543–2557. [Google Scholar] [CrossRef]
- Niedenthal, R.K.; Riles, L.; Johnston, M.; Hegemann, J.H. Green fluorescent protein as a marker for gene expression and subcellular localization in budding yeast. Yeast 1996, 12, 773–786. [Google Scholar] [CrossRef]
- Huh, W.-K.; Falvo, J.V.; Gerke, L.C.; Carroll, A.S.; Howson, R.W.; Weissman, J.S.; O’Shea, E.K. Global analysis of protein localization in budding yeast. Nature 2003, 425, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.J.; Lee, B.; Park, H.-O. Specific Residues of the GDP/GTP Exchange Factor Bud5p Are Involved in Establishment of the Cell Type-specific Budding Pattern in Yeast. J. Biol. Chem. 2004, 279, 27980–27985. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.J.; Lee, M.E.; Park, H.-O. Bud3 activates Cdc42 to establish a proper growth site in budding yeast. J. Cell Biol. 2014, 206, 19–28. [Google Scholar] [CrossRef]
- Zheng, Y.; Cerione, R.; Bender, A. Control of the yeast bud-site assembly GTPase Cdc42. Catalysis of guanine nucleotide exchange by Cdc24 and stimulation of GTPase activity by Bem3. J. Biol. Chem. 1994, 269, 2369–2372. [Google Scholar]
- Stevenson, B.J.; Ferguson, B.; De Virgilio, C.; Bi, E.; Pringle, J.R.; Ammerer, G.; Sprague, G.F. Mutation of RGA1, which encodes a putative GTPase-activating protein for the polarity-establishment protein Cdc42p, activates the pheromone-response pathway in the yeast Saccharomyces cerevisiae. Genes Dev. 1995, 9, 2949–2963. [Google Scholar] [CrossRef]
- Gladfelter, A.S.; Bose, I.; Zyla, T.R.; Bardes, E.S.G.; Lew, D.J. Septin ring assembly involves cycles of GTP loading and hydrolysis by Cdc42p. J. Cell Biol. 2002, 156, 315–326. [Google Scholar] [CrossRef]
- Smith, G.R.; Givan, S.A.; Cullen, P.; Sprague, G.F. GTPase-Activating Proteins for Cdc42. Eukaryot. Cell 2002, 1, 469–480. [Google Scholar] [CrossRef]
- Marquitz, A.R.; Harrison, J.C.; Bose, I.; Zyla, T.R.; McMillan, J.N.; Lew, D.J. The Rho-GAP Bem2p plays a GAP-independent role in the morphogenesis checkpoint. EMBO J. 2002, 21, 4012–4025. [Google Scholar] [CrossRef][Green Version]
- Caviston, J.P.; Longtine, M.; Pringle, J.R.; Bi, E. The role of Cdc42p GTPase-activating proteins in assembly of the septin ring in yeast. Mol. Biol. Cell 2003, 14, 4051–4066. [Google Scholar] [CrossRef]
- Mukherjee, D.; Sen, A.; Boettner, D.R.; Fairn, G.D.; Schlam, D.; Bonilla Valentin, F.J.; McCaffery, J.M.; Hazbun, T.; Staiger, C.J.; Grinstein, S.; et al. Bem3, a Cdc42 GTPase-activating protein, traffics to an intracellular compartment and recruits the secretory Rab GTPase Sec4 to endomembranes. J. Cell Sci. 2013, 126, 4560–4571. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Coon, B.G.; Edwards, D.F.; Hanna, C.B.; Longhi, S.A.; McCaffery, J.M.; Wendland, B.; Retegui, L.A.; Bi, E.; Aguilar, R.C. The yeast endocytic protein Epsin 2 functions in a cell-division signaling pathway. J. Cell Sci. 2009, 122, 2453–2463. [Google Scholar] [CrossRef]
- Wild, A.C.; Yu, J.W.; Lemmon, M.A.; Blumer, K.J. The p21-activated Protein Kinase-related Kinase Cla4 Is a Coincidence Detector of Signaling by Cdc42 and Phosphatidylinositol 4-Phosphate. J. Biol. Chem. 2004, 279, 17101–17110. [Google Scholar] [CrossRef] [PubMed]
- Lechler, T.; Shevchenko, A.; Shevchenko, A.; Li, R. Direct Involvement of Yeast Type I Myosins in Cdc42-Dependent Actin Polymerization. J. Cell Biol. 2000, 148, 363–374. [Google Scholar] [CrossRef]
- Goryachev, A.B.; Leda, M. Many roads to symmetry breaking: Molecular mechanisms and theoretical models of yeast cell polarity. Mol. Biol. Cell 2017, 28, 370–380. [Google Scholar] [CrossRef]
- Howell, A.S.; Savage, N.S.; Johnson, S.A.; Bose, I.; Wagner, A.W.; Zyla, T.R.; Nijhout, H.F.; Reed, M.C.; Goryachev, A.B.; Lew, D.J. Singularity in Polarization: Rewiring Yeast Cells to Make Two Buds. Cell 2009, 139, 731–743. [Google Scholar] [CrossRef]
- Kamada, Y.; Qadota, H.; Python, C.P.; Anraku, Y.; Ohya, Y.; Levin, D.E. Activation of yeast protein kinase C by Rho1 GTPase. J. Biol. Chem. 1996, 271, 9193–9196. [Google Scholar] [CrossRef]
- Qadota, H.; Python, C.P.; Inoue, S.B.; Arisawa, M.; Anraku, Y.; Zheng, Y.; Watanabe, T.; Levin, D.E.; Ohya, Y. Identification of yeast Rho1p GTPase as a regulatory subunit of 1, 3-β-glucan synthase. Science 1996, 272, 279–281. [Google Scholar] [CrossRef]
- Saka, A.; Abe, M.; Okano, H.; Minemura, M.; Qadota, H.; Utsugi, T.; Mino, A.; Tanaka, K.; Takai, Y.; Ohya, Y. Complementing Yeast rho1 Mutation Groups with Distinct Functional Defects. J. Biol. Chem. 2001, 276, 46165–46171. [Google Scholar] [CrossRef]
- Diepeveen, E.T.; De la Cruz, L.I.; Laan, L. Evolutionary dynamics in the fungal polarization network, a mechanistic perspective. Biophys. Rev. 2017, 9, 375–387. [Google Scholar] [CrossRef]
- Reynaud, A.; Facca, C.; Sor, F.; Faye, G. Disruption and functional analysis of six ORFs of chromosome IV: YDL103c (QRI1), YDL105w (QRI2), YDL112w (TRM3), YDL113c, YDL116w (NUP84) and YDL167c (NRP1). Yeast 2001, 18, 273–282. [Google Scholar] [CrossRef]
- Outten, C.E.; Falk, R.L.; Culotta, V.C. Cellular factors required for protection from hyperoxia toxicity in Saccharomyces cerevisiae. Biochem. J. 2005, 388, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Buchan, J.R.; Muhlrad, D.; Parker, R. P bodies promote stress granule assembly in Saccharomyces cerevisiae. J. Cell Biol. 2008, 183, 441–455. [Google Scholar] [CrossRef] [PubMed]
- Hoepfner, D.; Helliwell, S.B.; Sadlish, H.; Schuierer, S.; Filipuzzi, I.; Brachat, S.; Bhullar, B.; Plikat, U.; Abraham, Y.; Altorfer, M.; et al. High-resolution chemical dissection of a model eukaryote reveals targets, pathways and gene functions. Microbiol. Res. 2014, 169, 107–120. [Google Scholar] [CrossRef]
- Alberti, S.; Halfmann, R.; King, O.; Kapila, A.; Lindquist, S. A Systematic Survey Identifies Prions and Illuminates Sequence Features of Prionogenic Proteins. Cell 2009, 137, 146–158. [Google Scholar] [CrossRef]
- Hogan, D.J.; Riordan, D.P.; Gerber, A.P.; Herschlag, D.; Brown, P.O. Diverse RNA-Binding Proteins Interact with Functionally Related Sets of RNAs, Suggesting an Extensive Regulatory System. PLoS Biol. 2008, 6, e255. [Google Scholar] [CrossRef]
- Sharifpoor, S.; Van Dyk, D.; Costanzo, M.; Baryshnikova, A.; Friesen, H.; Douglas, A.C.; Youn, J.-Y.; VanderSluis, B.; Myers, C.L.; Papp, B.; et al. Functional wiring of the yeast kinome revealed by global analysis of genetic network motifs. Genome Res. 2012, 22, 791–801. [Google Scholar] [CrossRef]
- Etienne-Manneville, S. Cdc42—The centre of polarity. J. Cell Sci. 2004, 117, 1291–1300. [Google Scholar] [CrossRef]
- Johnson, D.I. Cdc42: An Essential Rho-Type GTPase Controlling Eukaryotic Cell Polarity. Microbiol. Mol. Biol. Rev. 1999, 63, 54–105. [Google Scholar] [CrossRef]
- Lee, C.; Occhipinti, P.; Gladfelter, A.S. PolyQ-dependent RNA–protein assemblies control symmetry breaking. J. Cell Biol. 2015, 208, 533–544. [Google Scholar] [CrossRef]
- Kato, M.; McKnight, S.L. Cross-β Polymerization of Low Complexity Sequence Domains. Cold Spring Harb. Perspect. Biol. 2017, 9, a023598. [Google Scholar] [CrossRef]
- Wuthrich, K. Protein structure determination in solution by nuclear magnetic resonance spectroscopy. Science 1989, 243, 45–50. [Google Scholar] [CrossRef]
- Hrdlickova, R.; Toloue, M.; Tian, B. RNA-Seq methods for transcriptome analysis: RNA-Seq. Wiley Interdiscip. Rev. RNA 2017, 8, e1364. [Google Scholar] [CrossRef] [PubMed]
- Keren, L.; Hausser, J.; Lotan-Pompan, M.; Vainberg Slutskin, I.; Alisar, H.; Kaminski, S.; Weinberger, A.; Alon, U.; Milo, R.; Segal, E. Massively Parallel Interrogation of the Effects of Gene Expression Levels on Fitness. Cell 2016, 166, 1282–1294.e18. [Google Scholar] [CrossRef]
- Vendel, K.J.A.; Tschirpke, S.; Shamsi, F.; Dogterom, M.; Laan, L. Minimal in vitro systems shed light on cell polarity. J. Cell Sci. 2019, 132, jcs217554. [Google Scholar] [CrossRef] [PubMed]
- Heym, R.G.; Zimmermann, D.; Edelmann, F.T.; Israel, L.; Ökten, Z.; Kovar, D.R.; Niessing, D. In vitro reconstitution of an mRNA-transport complex reveals mechanisms of assembly and motor activation. J. Cell Biol. 2013, 203, 971–984. [Google Scholar] [CrossRef] [PubMed]
- Cerulus, B.; New, A.M.; Pougach, K.; Verstrepen, K.J. Noise and Epigenetic Inheritance of Single-Cell Division Times Influence Population Fitness. Curr. Biol. 2016, 26, 1138–1147. [Google Scholar] [CrossRef]
- Xue, Y.; Acar, M. Mechanisms for the epigenetic inheritance of stress response in single cells. Curr. Genet. 2018, 64, 1221–1228. [Google Scholar] [CrossRef]
- Stark, C. BioGRID: A general repository for interaction datasets. Nucleic Acids Res. 2006, 34, D535–D539. [Google Scholar] [CrossRef]
- Michel, A.H.; Hatakeyama, R.; Kimmig, P.; Arter, M.; Peter, M.; Matos, J.; De Virgilio, C.; Kornmann, B. Functional mapping of yeast genomes by saturated transposition. Elife 2017, 6, e23570. [Google Scholar] [CrossRef]
- Kryazhimskiy, S.; Rice, D.P.; Jerison, E.R.; Desai, M.M. Global epistasis makes adaptation predictable despite sequence-level stochasticity. Science 2014, 344, 1519–1522. [Google Scholar] [CrossRef] [PubMed]
- Fields, S.; Song, O. A novel genetic system to detect protein–protein interactions. Nature 1989, 340, 245–246. [Google Scholar] [CrossRef] [PubMed]
- Jares-Erijman, E.A.; Jovin, T.M. FRET imaging. Nat. Biotechnol. 2003, 21, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Phizicky, E.M.; Fields, S. Protein-protein interactions: Methods for detection and analysis. Microbiol. Rev. 1995, 59, 94–123. [Google Scholar] [CrossRef] [PubMed]
- Foltman, M.; Sanchez-Diaz, A. Studying Protein–Protein Interactions in Budding Yeast Using Co-immunoprecipitation. In Yeast Cytokinesis: Methods and Protocols; Sanchez-Diaz, A., Perez, P., Eds.; Springer: New York, NY, USA, 2016; pp. 239–256. ISBN 978-1-4939-3145-3. [Google Scholar]
- Nishimura, K.; Fukagawa, T.; Takisawa, H.; Kakimoto, T.; Kanemaki, M. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nat. Methods 2009, 6, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Papagiannakis, A.; De Jonge, J.J.; Zhang, Z.; Heinemann, M. Quantitative characterization of the auxin-inducible degron: A guide for dynamic protein depletion in single yeast cells. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jost, A.P.-T.; Weiner, O.D. Probing Yeast Polarity with Acute, Reversible, Optogenetic Inhibition of Protein Function. ACS Synth. Biol. 2015, 4, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E. Technical review: In situ hybridization. Anat. Rec. 2014, 297, 1349–1353. [Google Scholar] [CrossRef]
- Pelillo, M.; Poli, I.; Roli, A.; Serra, R.; Slanzi, D.; Villani, M. (Eds.) Artificial Life and Evolutionary Computation; Communications in Computer and Information Science; Springer International Publishing: Cham, Switzerland, 2018; Volume 830, ISBN 978-3-319-78657-5. [Google Scholar]
- Giaever, G.; Chu, A.M.; Ni, L.; Connelly, C.; Riles, L.; Véronneau, S.; Dow, S.; Lucau-Danila, A.; Anderson, K.; Andre, B. Functional profiling of the Saccharomyces cerevisiae genome. Nature 2002, 418, 387–391. [Google Scholar] [CrossRef]
- Gavin, A.-C.; Bösche, M.; Krause, R.; Grandi, P.; Marzioch, M.; Bauer, A.; Schultz, J.; Rick, J.M.; Michon, A.-M.; Cruciat, C.-M. Functional organization of the yeast proteome by systematic analysis of protein complexes. Nature 2002, 415, 141. [Google Scholar] [CrossRef]
- Tarassov, K.; Messier, V.; Landry, C.R.; Radinovic, S.; Molina, M.M.S.; Shames, I.; Malitskaya, Y.; Vogel, J.; Bussey, H.; Michnick, S.W. An in vivo map of the yeast protein interactome. Science 2008, 320, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Garí Marsol, E.; Volpe, T.; Wang, H.; Gallego, C.; Futcher, B.; Aldea, M. Whi3 binds the mRNA of the G (1) cyclin CLN3 to modulate cell fate in budding yeast. Genes Dev. 2001, 15, 2803–2808. [Google Scholar]
- Colomina, N.; Ferrezuelo, F.; Wang, H.; Aldea, M.; Garí, E. Whi3, a Developmental Regulator of Budding Yeast, Binds a Large Set of mRNAs Functionally Related to the Endoplasmic Reticulum. J. Biol. Chem. 2008, 283, 28670–28679. [Google Scholar] [CrossRef] [PubMed]
- Wittenberg, C.; Sugimoto, K.; Reed, S.I. G1-specific cyclins of S. cerevisiae: Cell cycle periodicity, regulation by mating pheromone, and association with the p34CDC28 protein kinase. Cell 1990, 62, 225–237. [Google Scholar] [CrossRef]
- Leeuw, T.; Wu, C.; Schrag, J.D.; Whiteway, M.; Thomas, D.Y.; Leberer, E. Interaction of a G-protein β-subunit with a conserved sequence in Ste20/PAK family protein kinases. Nature 1998, 391, 191. [Google Scholar] [CrossRef]
- Wiget, P.; Shimada, Y.; Butty, A.-C.; Bi, E.; Peter, M. Site-specific regulation of the GEF Cdc24p by the scaffold protein Far1p during yeast mating. EMBO J. 2004, 23, 1063–1074. [Google Scholar] [CrossRef]
- Camus, C.; Boy-Marcotte, E.; Jacquet, M. Two subclasses of guanine exchange factor (GEF) domains revealed by comparison of activities of chimeric genes constructed from CDC25, SDC25 and BUD5 in Saccharomyces cerevisiae. Mol. Gen. Genet. MGG 1994, 245, 167–176. [Google Scholar] [CrossRef]
- Kozminski, K.G.; Beven, L.; Angerman, E.; Tong, A.H.Y.; Boone, C.; Park, H.-O. Interaction between a Ras and a Rho GTPase couples selection of a growth site to the development of cell polarity in yeast. Mol. Biol. Cell 2003, 14, 4958–4970. [Google Scholar] [CrossRef]
- Koch, G.; Tanaka, K.; Masuda, T.; Yamochi, W.; Nonaka, H.; Takai, Y. Association of the Rho family small GTP-binding proteins with Rho GDP dissociation inhibitor (Rho GDI) in Saccharomyces cerevisiae. Oncogene 1997, 15, 417. [Google Scholar] [CrossRef]
- Bose, I.; Irazoqui, J.E.; Moskow, J.J.; Bardes, E.S.G.; Zyla, T.R.; Lew, D.J. Assembly of Scaffold-mediated Complexes Containing Cdc42p, the Exchange Factor Cdc24p, and the Effector Cla4p Required for Cell Cycle-regulated Phosphorylation of Cdc24p. J. Biol. Chem. 2001, 276, 7176–7186. [Google Scholar] [CrossRef]
- Kozubowski, L.; Saito, K.; Johnson, J.M.; Howell, A.S.; Zyla, T.R.; Lew, D.J. Symmetry-Breaking Polarization Driven by a Cdc42p GEF-PAK Complex. Curr. Biol. 2008, 18, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Gulli, M.-P.; Jaquenoud, M.; Shimada, Y.; Niederhäuser, G.; Wiget, P.; Peter, M. Phosphorylation of the Cdc42 exchange factor Cdc24 by the PAK-like kinase Cla4 may regulate polarized growth in yeast. Mol. Cell 2000, 6, 1155–1167. [Google Scholar] [CrossRef]
- Sagot, I.; Klee, S.K.; Pellman, D. Yeast formins regulate cell polarity by controlling the assembly of actin cables. Nat. Cell Biol. 2002, 4, 42. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.E. Relationship of actin and tubulin distribution to bud growth in wild- type and morphogenetic-mutant Saccharomyces cerevisiae. J. Cell Biol. 1984, 98, 934–945. [Google Scholar] [CrossRef]
- Wendland, B. Epsins: Adaptors in endocytosis? Nat. Rev. Mol. Cell Biol. 2002, 3, 971. [Google Scholar] [CrossRef]
- Goode, B.L.; Eskin, J.A.; Wendland, B. Actin and Endocytosis in Budding Yeast. Genetics 2015, 199, 315–358. [Google Scholar] [CrossRef]
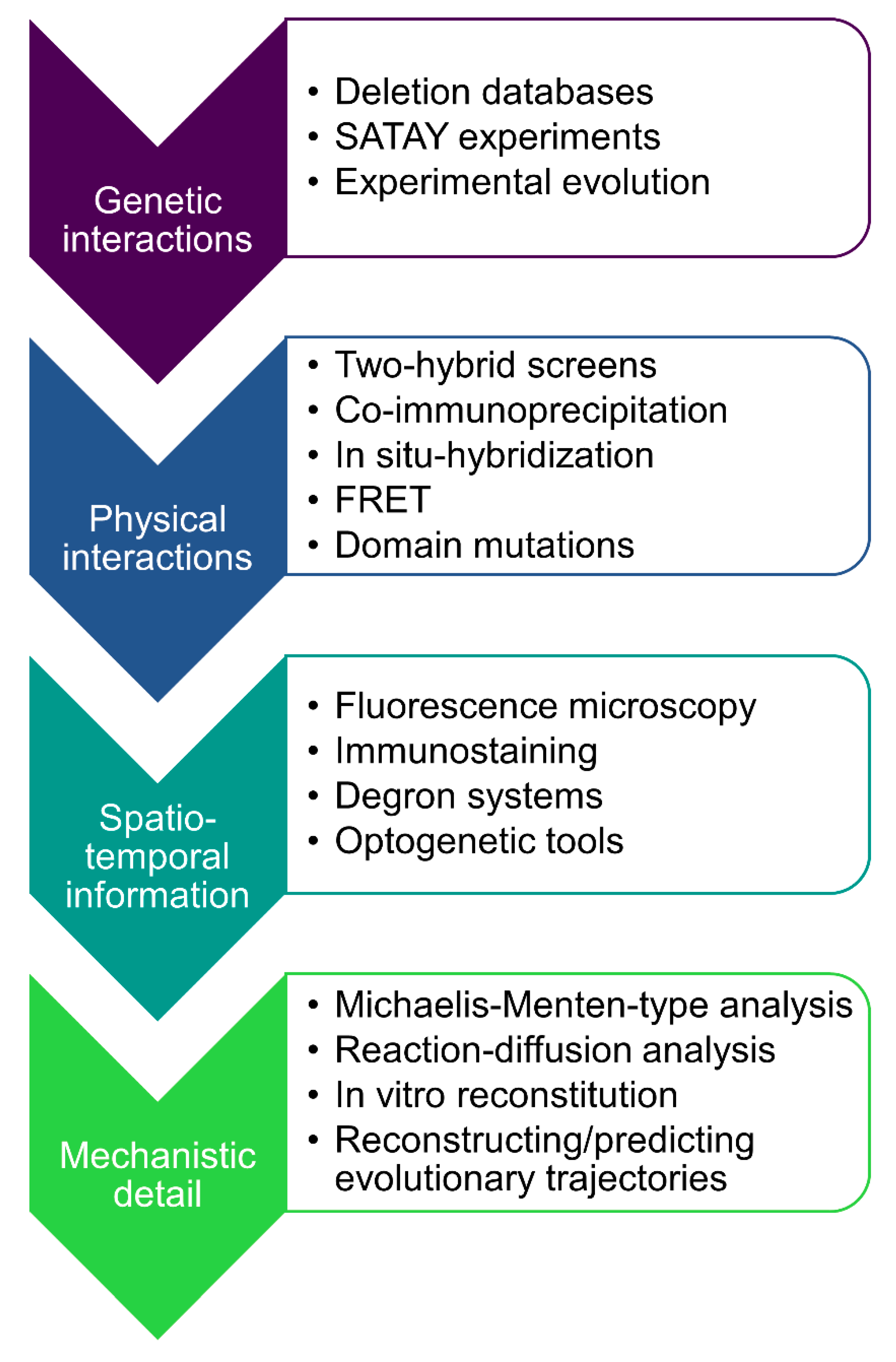
| Term | Definition |
|---|---|
| Protein network | Group of proteins with physical interactions together performing a function |
| Connectivity | Degree to which parts of the network are embedded with other parts in the network. In this sense, it can be received as the reciprocal of modularity. |
| Modularity | Potential to group parts of a protein network given a certain representation of the protein network (e.g., in terms of mechanisms, genetic or physical interactions) |
| Hub protein | Highly connected protein in a network (often essential) |
| Neutrality | No consequence of a mutation to phenotype (in current environment) |
| Hierarchy | Clear layering of pathways inside a protein network |
| Redundancy | Multiple mechanisms that can to some extent interchangeably contribute to the same function |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daalman, W.K.-G.; Sweep, E.; Laan, L. The Path towards Predicting Evolution as Illustrated in Yeast Cell Polarity. Cells 2020, 9, 2534. https://doi.org/10.3390/cells9122534
Daalman WK-G, Sweep E, Laan L. The Path towards Predicting Evolution as Illustrated in Yeast Cell Polarity. Cells. 2020; 9(12):2534. https://doi.org/10.3390/cells9122534
Chicago/Turabian StyleDaalman, Werner Karl-Gustav, Els Sweep, and Liedewij Laan. 2020. "The Path towards Predicting Evolution as Illustrated in Yeast Cell Polarity" Cells 9, no. 12: 2534. https://doi.org/10.3390/cells9122534
APA StyleDaalman, W. K.-G., Sweep, E., & Laan, L. (2020). The Path towards Predicting Evolution as Illustrated in Yeast Cell Polarity. Cells, 9(12), 2534. https://doi.org/10.3390/cells9122534






