Expression of Melatonin and Dopamine D3 Receptor Heteromers in Eye Ciliary Body Epithelial Cells and Negative Correlation with Ocular Hypertension
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drugs
2.2. Cell Culture and Transient Transfection
2.3. Fusion Proteins and Expression Vectors
2.4. Bioluminescence Resonance Energy Transfer (BRET) Assays
2.5. cAMP Level Determination
2.6. Extracellular Signal-Regulated Kinase Phosphorylation Assays
2.7. Label-Free Dynamic Mass Redistribution (DMR) Assay
2.8. Human Eye Postmortem Samples
2.9. Immunofluorescence Studies
2.10. In Situ and In Vitro Proximity Ligation Assay (PLA)
2.11. Data Analysis
3. Results
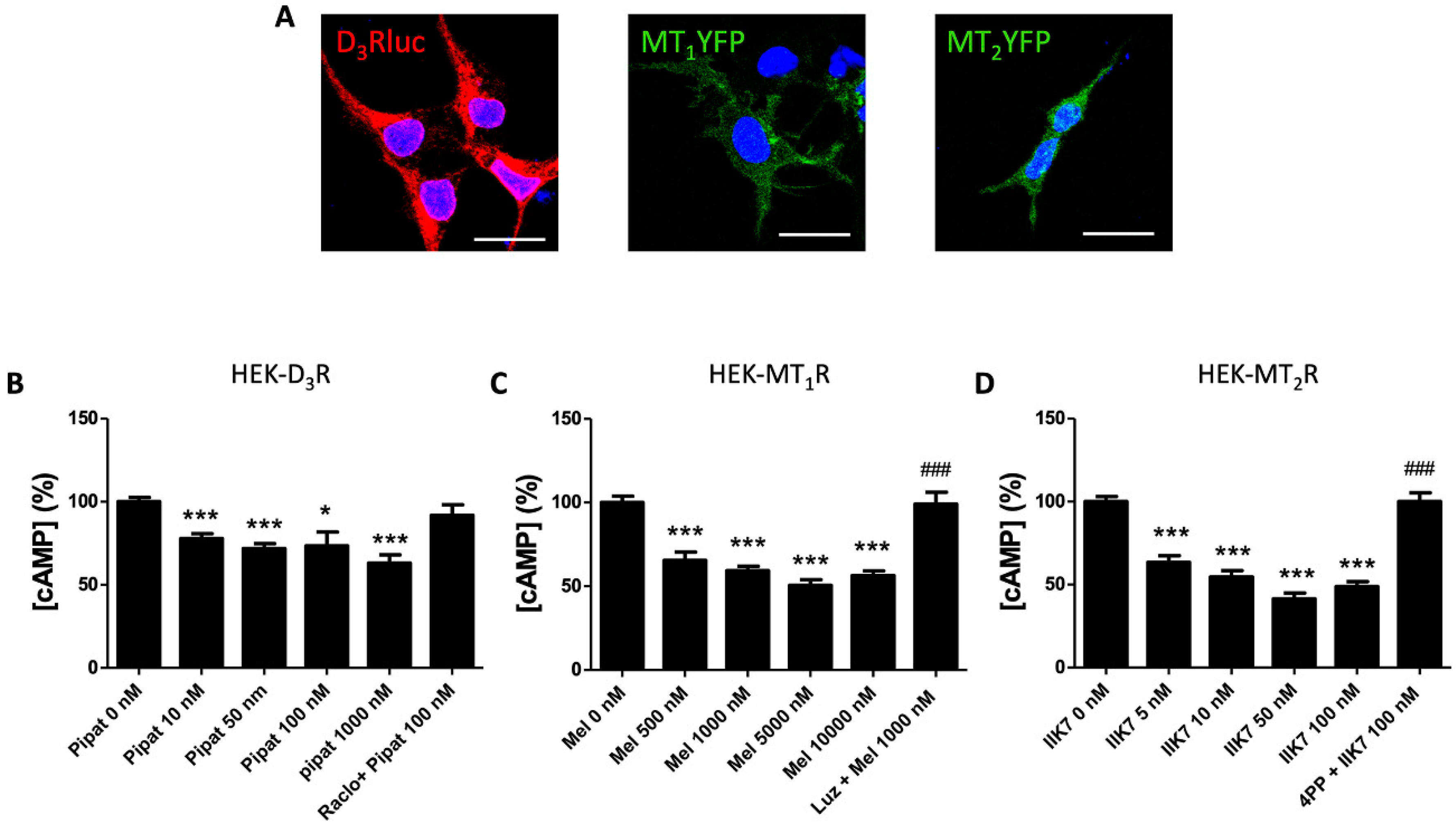
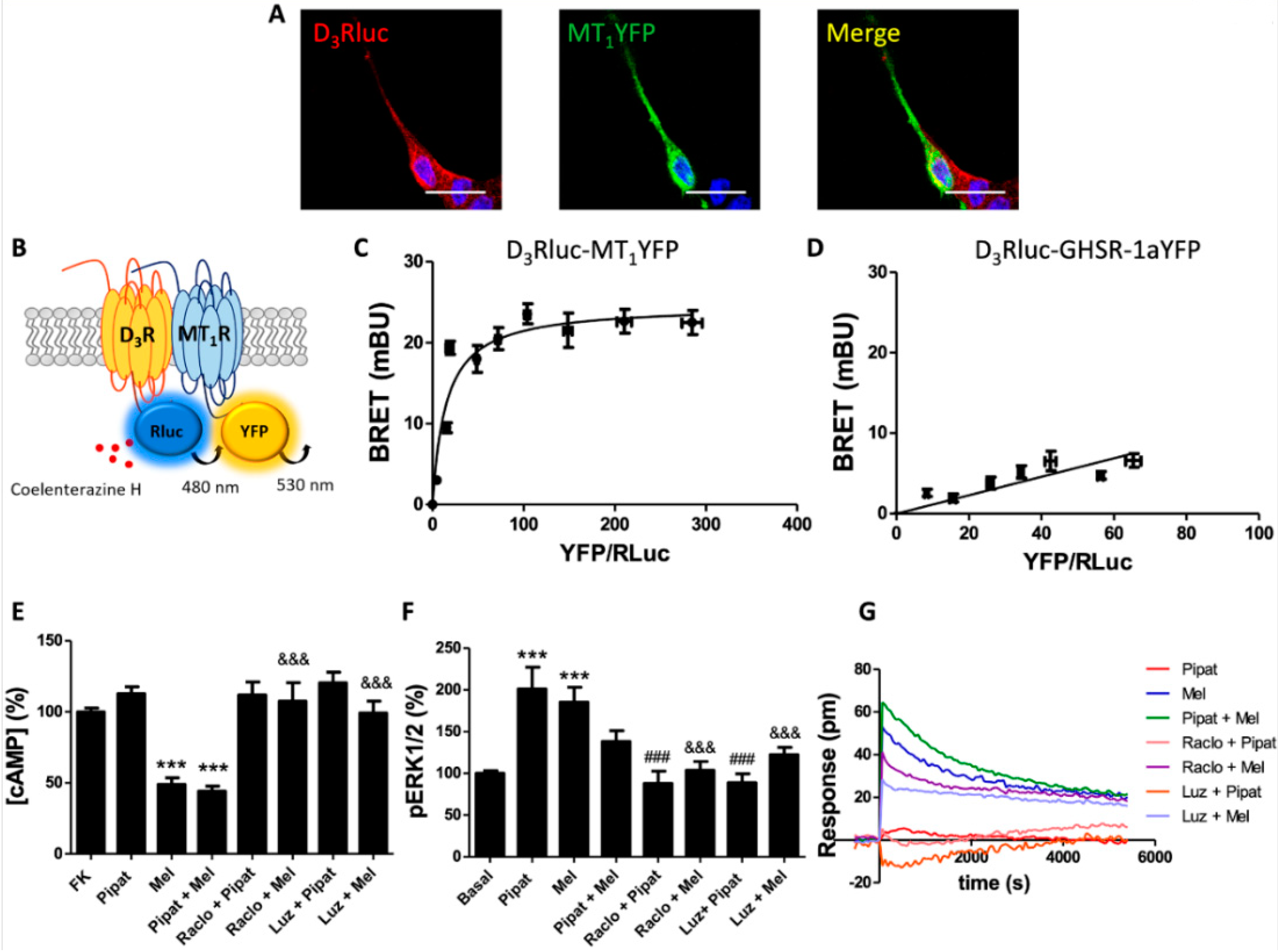
3.1. Dopamine D3 Receptors Interact with Melatonin MT1 Receptors in the HEK-293T Cells
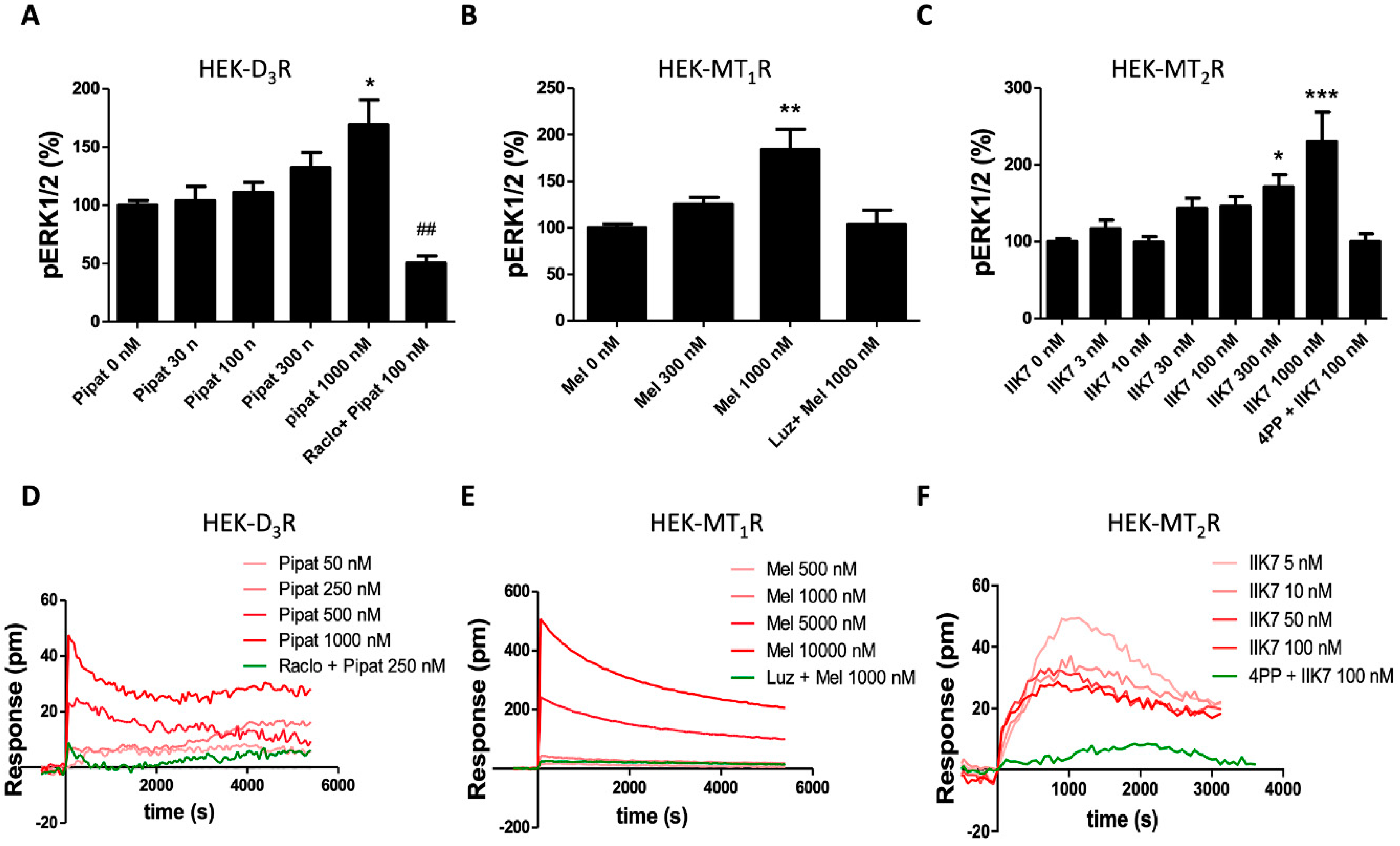
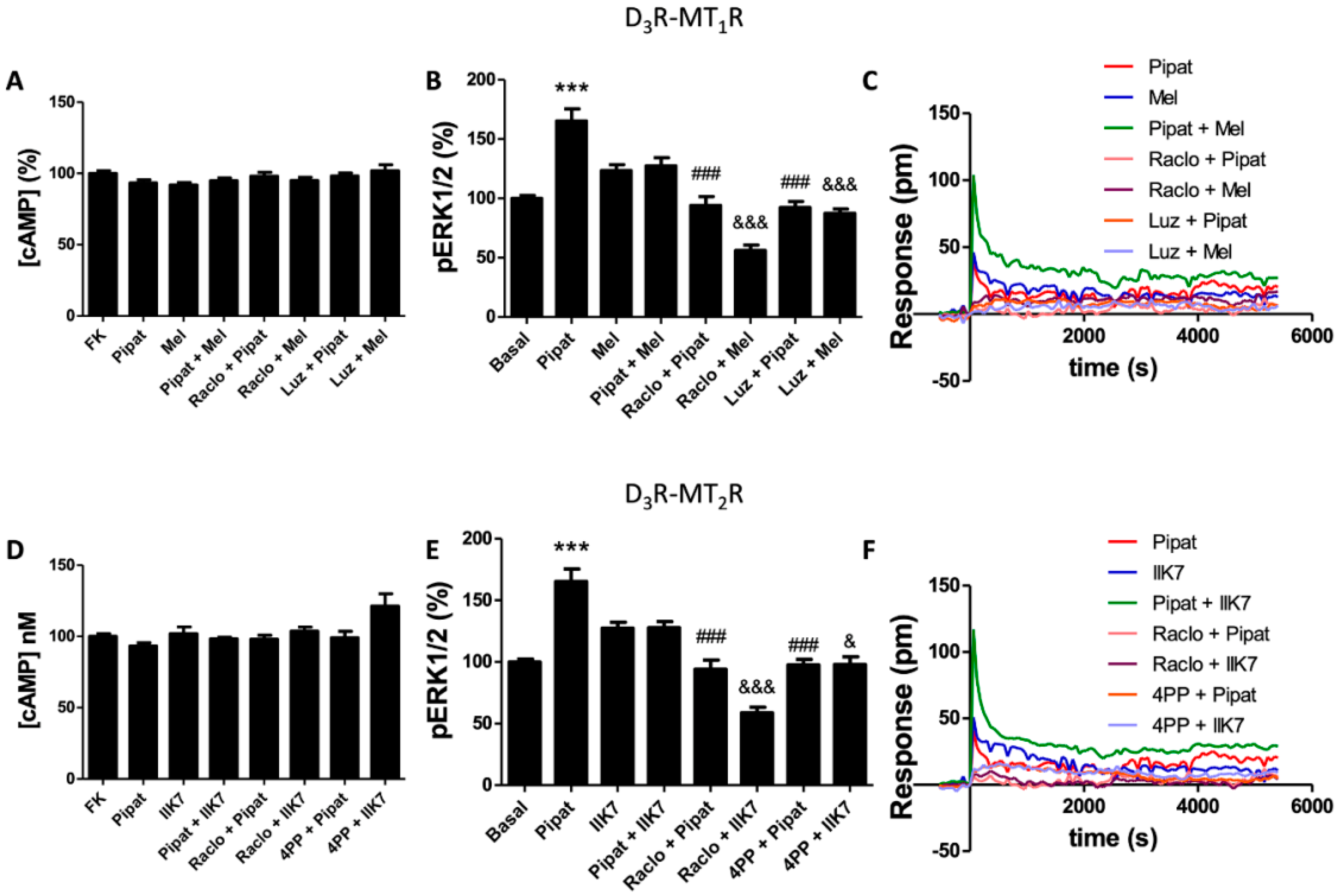
3.2. Functional Characterization of D3-MT1 Heteroreceptor Complexes in HEK-293T Cells
3.3. Dopamine D3 Receptors Interact with Melatonin MT2 Receptors in HEK-293T Cells
3.4. Functional Characterization of the D3-MT2 Heteroreceptor Complexes in HEK-293T Cells
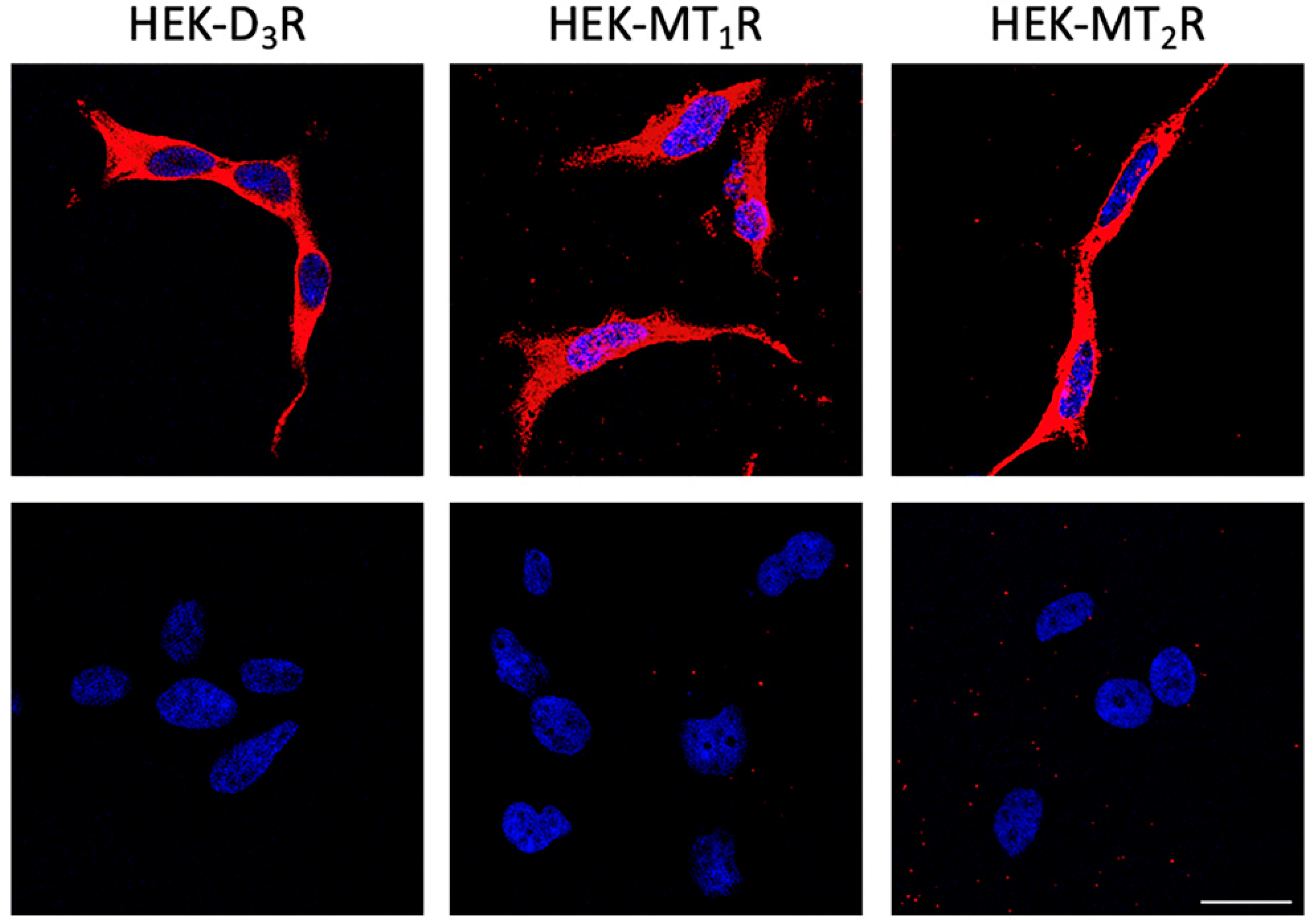
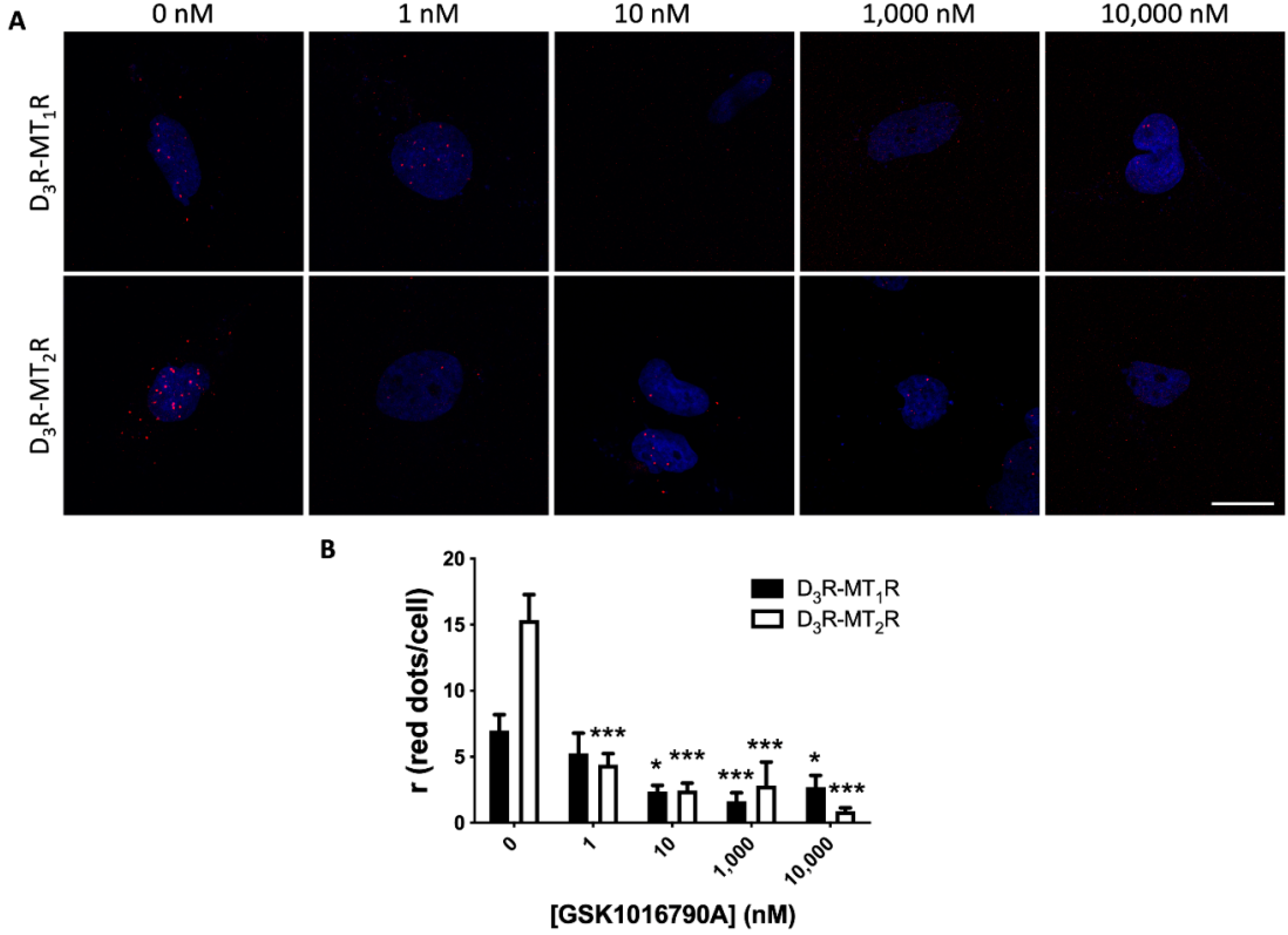
3.5. Detection and Functional Characterization of D3-MT1 and D3-MT2 Heteroreceptor Complexes in Human Non-Pigmented Ciliary Body Epithelial Cells
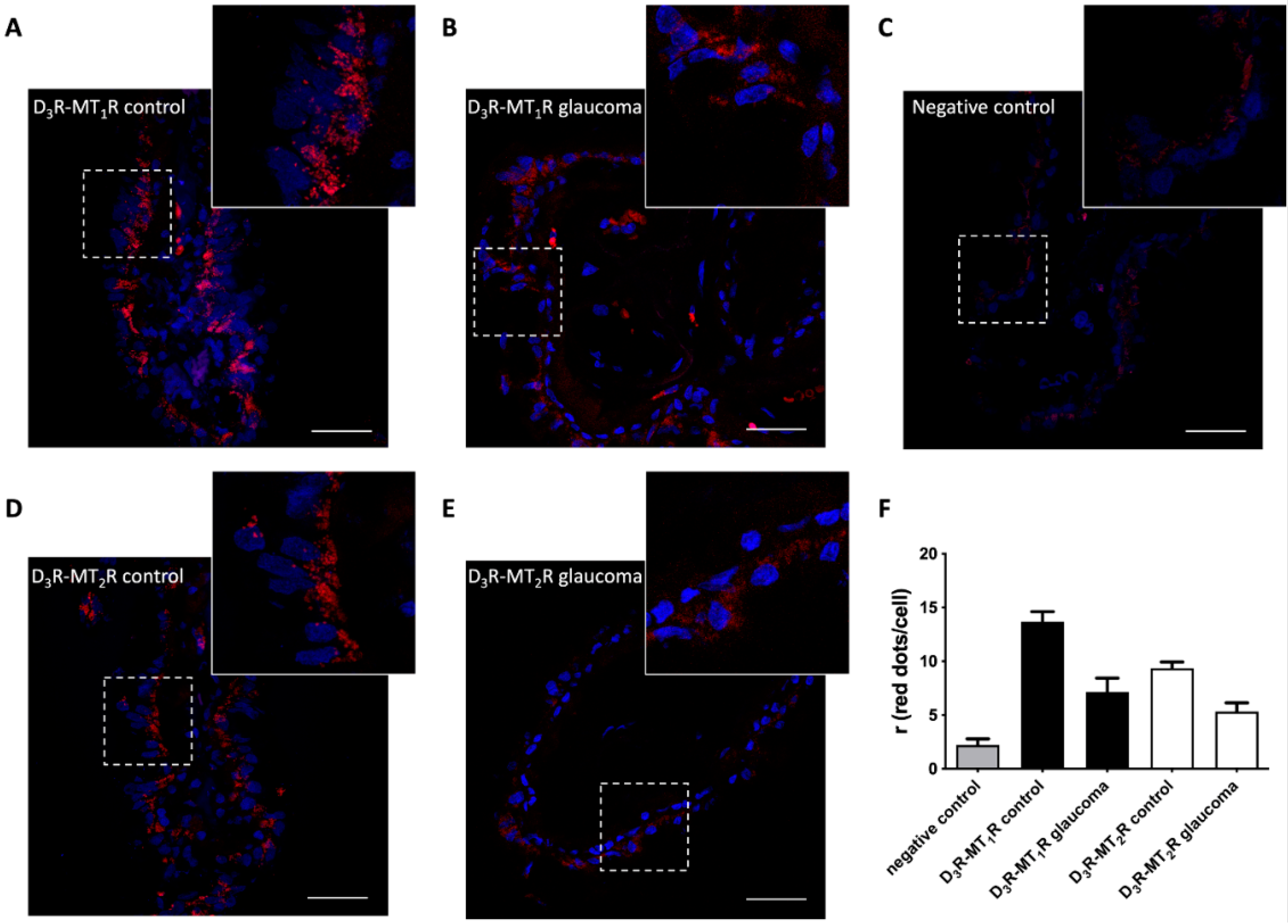
3.6. Differential Expression of D3-MT1 and D3-MT2 Heteroreceptor Complexes in the Glaucomatous Eye
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Delamere, N.A. Ciliary Body and Ciliary Epithelium. Adv. Organ. Biol. 2005, 10, 127–148. [Google Scholar] [PubMed] [Green Version]
- Tamm, E.R.; Lütjen-Drecoll, E. Ciliary body. Microsc. Res. Tech. 1996, 33, 390–439. [Google Scholar] [CrossRef]
- Civan, M.M.; Macknight, A.D.C. The ins and outs of aqueous humour secretion. Exp. Eye Res. 2004, 78, 625–631. [Google Scholar] [CrossRef]
- Adler, F.H. Is the Aqueous Humor a Dialysate? Trans. Am. Ophthalmol. Soc. 1933, 31, 131–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davson, H.; Duke-Elder, W.S.; Benham, G.H. The ionic equilibrium between the aqueous humour and blood plasma of cats. Biochem. J. 1936, 30, 773–775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krupin, T.; Wax, M.; Moolchandani, J. Aqueous production. Trans. Ophthalmol. Soc. U. K. 1986, 105, 156–161. [Google Scholar] [PubMed]
- Casson, R.J.; Chidlow, G.; Wood, J.P.M.; Crowston, J.G.; Goldberg, I. Definition of glaucoma: Clinical and experimental concepts. Clin. Experiment. Ophthalmol. 2012, 40, 341–349. [Google Scholar] [CrossRef] [Green Version]
- McCannel, C.A.; Heinrich, S.R.; Brubaker, R.F. Acetazolamide but not timolol lowers aqueous humor flow in sleeping humans. Graefe’s Arch. Clin. Exp. Ophthalmol. 1992, 230, 518–520. [Google Scholar] [CrossRef]
- Asrani, S.; Zeimer, R.; Wilensky, J.; Gieser, D.; Vitale, S.; Lindenmuth, K. Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma. J. Glaucoma 2000, 9, 134–142. [Google Scholar] [CrossRef]
- Lerner, A.B.; Case, J.D.; Takahashi, Y. Isolation of melatonin and 5-methoxyindole-3-acetic acid from bovine pineal glands. J. Biol. Chem. 1960, 235, 1992–1997. [Google Scholar]
- Arendt, J. Jet-lag and shift work: (2) Therapeutic use of melatonin. J. R. Soc. Med. 1999, 92, 402–405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Barceló, E.J.; Mediavilla, M.D.; Tan, D.X.; Reiter, R.J. Clinical uses of melatonin: Evaluation of human trials. Curr. Med. Chem. 2010, 17, 2070–2095. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.F.; Yu, H.S.; Suen, H.C.; Brown, G.M. Melatonin in the retina of rats: A diurnal rhythm. J. Endocrinol. 1980, 87, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Aimoto, T.; Rohde, B.H.; Chiou, G.C.; Lauber, J.K. N-acetyltransferase activity and melatonin level in the eyes of glaucomatous chickens. J. Ocul. Pharmacol. 1985, 1, 149–160. [Google Scholar] [CrossRef]
- Abe, M.; Itoh, M.T.; Miyata, M.; Shimizu, K.; Sumi, Y. Circadian rhythm of serotonin N-acetyltransferase activity in rat lens. Exp. Eye Res. 2000, 70, 805–808. [Google Scholar] [CrossRef]
- Quay, W.B. Increases in Volume, Fluid Content, and Lens Weight of Eyes Following Systemic Administration of Melatonin. J. Pineal Res. 1984, 1, 3–13. [Google Scholar] [CrossRef]
- Djeridane, Y.; Vivien-Roels, B.; Simonneaux, V.; Miguez, J.M.; Pévet, P. Evidence for melatonin synthesis in rodent Harderian gland: A dynamic in vitro study. J. Pineal Res. 1998, 25, 54–64. [Google Scholar] [CrossRef]
- Mhatre, M.C.; van Jaarsveld, A.S.; Reiter, R.J. Melatonin in the lacrimal gland: First demonstration and experimental manipulation. Biochem. Biophys. Res. Commun. 1988, 153, 1186–1192. [Google Scholar] [CrossRef]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T. Cloning and characterization of a mammalian melatonin receptor that mediates reproductive and circadian responses. Neuron 1994, 13, 1177–1185. [Google Scholar] [CrossRef]
- Reppert, S.M.; Godson, C.; Mahle, C.D.; Weaver, D.R.; Slaugenhaupt, S.A.; Gusella, J.F. Molecular characterization of a second melatonin receptor expressed in human retina and brain: The Mel1b melatonin receptor. Proc. Natl. Acad. Sci. USA 1995, 92, 8734–8738. [Google Scholar] [CrossRef] [Green Version]
- Li, D.Y.; Smith, D.G.; Hardeland, R.; Yang, M.Y.; Xu, H.L.; Zhang, L.; Yin, H.D.; Zhu, Q. Melatonin receptor genes in vertebrates. Int. J. Mol. Sci. 2013, 14, 11208–11223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crooke, A.; Guzman-Aranguez, A.; Mediero, A.; Alarma-Estrany, P.; Carracedo, G.; Pelaez, T.; Peral, A.; Pintor, J. Effect of Melatonin and Analogues on Corneal Wound Healing: Involvement of Mt2 Melatonin Receptor. Curr. Eye Res. 2015, 40, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.-C.; Hu, D.-D.; Zhao, L.; Li, N.; Shen, H.-Y. Correlation between presence of primary iris- and cilliary body cysts and intraocular pressure. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 3985–3989. [Google Scholar] [PubMed]
- Bietti, G. Surgical intervention on the ciliary body: New Trends for the Relief of Glaucoma. J. Am. Med. Assoc. 1950, 142, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, D.P.; Delagrange, P.; Dubocovich, M.L.; Jockers, R.; Krause, D.N.; Markus, R.P.; Olcese, J.; Pintor, J.; Renault, N.; Sugden, D.; et al. Melatonin receptors (version 2019.4) in the IUPHAR/BPS Guide to Pharmacology Database. IUPHAR/BPS Guid. to Pharmacol. CITE 2019, 2019. [Google Scholar] [CrossRef] [Green Version]
- Alexander, S.P.; Christopoulos, A.; Davenport, A.P.; Kelly, E.; Marrion, N.V.; Peters, J.A.; Faccenda, E.; Harding, S.D.; Pawson, A.J.; Sharman, J.L.; et al. CGTP Collaborators The concise guide to Pharmacology 2017/18: G protein-coupled receptors. Br. J. Pharmacol. 2017, 174, S17–S129. [Google Scholar] [CrossRef]
- Von Gall, C.; Weaver, D.R.; Kock, M.; Korf, H.W.; Stehle, J.H. Melatonin limits transcriptional impact of phosphoCREB in the mouse SCN via the Mel1a receptor. Neuroreport 2000, 11, 1803–1807. [Google Scholar] [CrossRef]
- Brydon, L.; Roka, F.; Petit, L.; De Coppet, P.; Tissot, M.; Barrett, P.; Morgan, P.J.; Nanoff, C.; Strosberg, A.D.; Jockers, R. Dual signaling of human Mel1a melatonin receptors via Gi2, Gi3, and Gq/11 proteins. Mol. Endocrinol. 1999, 13, 2025–2038. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Trakht, I.; Srinivasan, V.; Spence, D.W.; Maestroni, G.J.M.; Zisapel, N.; Cardinali, D.P. Physiological effects of melatonin: Role of melatonin receptors and signal transduction pathways. Prog. Neurobiol. 2008, 85, 335–353. [Google Scholar] [CrossRef]
- Huete-Toral, F.; Crooke, A.; Martínez-Águila, A.; Pintor, J.; Martinez-Aguila, A.; Pintor, J.; Martínez-Águila, A.; Pintor, J. Melatonin receptors trigger cAMP production and inhibit chloride movements in nonpigmented ciliary epithelial cells. J. Pharmacol. Exp. Ther. 2015, 352, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Cecon, E.; Oishi, A.; Jockers, R. Melatonin receptors: Molecular pharmacology and signalling in the context of system bias. Br. J. Pharmacol. 2017, 175, 3263–3280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levoye, A.; Dam, J.; Ayoub, M.A.; Guillaume, J.L.; Couturier, C.; Delagrange, P.; Jockers, R. The orphan GPR50 receptor specifically inhibits MT1 melatonin receptor function through heterodimerization. EMBO J. 2006, 25, 3012–3023. [Google Scholar] [CrossRef] [PubMed]
- Kamal, M.; Gbahou, F.; Guillaume, J.-L.; Daulat, A.M.; Benleulmi-Chaachoua, A.; Luka, M.; Chen, P.; Kalbasi Anaraki, D.; Baroncini, M.; Mannoury la Cour, C.; et al. Convergence of melatonin and serotonin (5-HT) signaling at MT2/5-HT2C receptor heteromers. J. Biol. Chem. 2015, 290, 11537–11546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alkozi, H.A.; Navarro, G.; Aguinaga, D.; Reyes-Resina, I.; Sanchez-Naves, J.; de Lara, M.J.P.; Franco, R.; Pintor, J. Adrenergic-melatonin heteroreceptor complexes are key in controlling ion homeostasis and intraocular eye pressure and their disruption contributes to hypertensive glaucoma. bioRxiv 2019, 636688. [Google Scholar] [CrossRef]
- Cahill, G.M.; Besharse, J.C. Resetting the circadian clock in cultured Xenopus eyecups: Regulation of retinal melatonin rhythms by light and D2 dopamine receptors. J. Neurosci. 1991, 11, 2959–2971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iuvone, P.M.; Besharse, J.C. Dopamine receptor-mediated inhibition of serotonin N-acetyltransferase activity in retina. Brain Res. 1986, 369, 168–176. [Google Scholar] [CrossRef]
- Iuvone, P.M.; Besharse, J.C. Cyclic AMP stimulates serotonin N-acetyltransferase activity in Xenopus retina in vitro. J. Neurochem. 1986, 46, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, T.N.; Alonso-Gomez, A.L.; Iuvone, P.M. Dopamine D4 receptors regulate intracellular calcium concentration in cultured chicken cone photoreceptor cells: Relationship to dopamine receptor-mediated inhibition of cAMP formation. Brain Res. 2008, 1207, 111–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adachi, A.; Nogi, T.; Ebihara, S. Phase-relationship and mutual effects between circadian rhythms of ocular melatonin and dopamine in the pigeon. Brain Res. 1998, 792, 361–369. [Google Scholar] [CrossRef]
- Adachi, A.; Suzuki, Y.; Nogi, T.; Ebihara, S. The relationship between ocular melatonin and dopamine rhythms in the pigeon: Effects of melatonin inhibition on dopamine release. Brain Res. 1999, 815, 435–440. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Nowak, J.Z. Dopamine D4-like receptors in vertebrate retina: Does the retina offer a model for the D4-receptor analysis? Pol. J. Pharmacol. 1997, 49, 201–211. [Google Scholar]
- Caravaggio, F.; Scifo, E.; Sibille, E.L.; Hernandez-Da Mota, S.E.; Gerretsen, P.; Remington, G.; Graff-Guerrero, A. Expression of dopamine D2 and D3 receptors in the human retina revealed by positron emission tomography and targeted mass spectrometry. Exp. Eye Res. 2018, 175, 32–41. [Google Scholar] [CrossRef]
- Pescosolido, N.; Parisi, F.; Russo, P.; Buomprisco, G.; Nebbioso, M. Role of dopaminergic receptors in glaucomatous disease modulation. Biomed. Res. Int. 2013, 2013, 193048. [Google Scholar] [CrossRef] [PubMed]
- Chu, E.; Socci, R.; Chu, T.-C. PD128,907 induces ocular hypotension in rabbits: Involvement of D2/D3 dopamine receptors and brain natriuretic peptide. J. Ocul. Pharmacol. Ther. 2004, 20, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Chu, E.; Chu, T.C.; Potter, D.E. Mechanisms and sites of ocular action of 7-hydroxy-2-dipropylaminotetralin: A dopamine3 receptor agonist. J. Pharmacol. Exp. Ther. 2000, 293, 710–716. [Google Scholar] [PubMed]
- Curtis, M.J.; Alexander, S.; Cirino, G.; Docherty, J.R.; George, C.H.; Giembycz, M.A.; Hoyer, D.; Insel, P.A.; Izzo, A.A.; Ji, Y.; et al. Experimental design and analysis and their reporting II: Updated and simplified guidance for authors and peer reviewers. Br. J. Pharmacol. 2018, 175, 987–993. [Google Scholar] [CrossRef] [Green Version]
- Alexander, S.P.H.; Roberts, R.E.; Broughton, B.R.S.; Sobey, C.G.; George, C.H.; Stanford, S.C.; Cirino, G.; Docherty, J.R.; Giembycz, M.A.; Hoyer, D.; et al. Goals and practicalities of immunoblotting and immunohistochemistry: A guide for submission to the British Journal of Pharmacology. Br. J. Pharmacol. 2018, 175, 407–411. [Google Scholar] [CrossRef] [Green Version]
- Sugden, D.; Yeh, L.K.; Teh, M.T. Design of subtype selective melatonin receptor agonists and antagonists. In Reproduction Nutrition Development; Elsevier Masson SAS: Paris, France, 1999; Volume 39, pp. 335–344. [Google Scholar]
- Martínez-Pinilla, E.; Varani, K.; Reyes-Resina, I.; Angelats, E.; Vincenzi, F.; Ferreiro-Vera, C.; Oyarzabal, J.; Canela, E.I.; Lanciego, J.L.; Nadal, X.; et al. Binding and signaling studies disclose a potential allosteric site for cannabidiol in cannabinoid CB2receptors. Front. Pharmacol. 2017, 8, 744. [Google Scholar] [CrossRef]
- Navarro, G.; Borroto-Escuela, D.; Angelats, E.; Etayo, I.; Reyes-Resina, I.; Pulido-Salgado, M.; Rodríguez-Pérez, A.; Canela, E.; Saura, J.; Lanciego, J.L.; et al. Receptor-heteromer mediated regulation of endocannabinoid signaling in activated microglia. Role of CB1 and CB2 receptors and relevance for Alzheimer’s disease and levodopa-induced dyskinesia. Brain Behav. Immun. 2018, 67, 139–151. [Google Scholar] [CrossRef]
- Hinz, S.; Navarro, G.; Borroto-Escuela, D.; Seibt, B.F.; Ammon, C.; De Filippo, E.; Danish, A.; Lacher, S.K.; Červinková, B.; Rafehi, M.; et al. Adenosine A2A receptor ligand recognition and signaling is blocked by A2B receptors. Oncotarget 2018, 9, 13593–13611. [Google Scholar] [CrossRef] [Green Version]
- Rivas-Santisteban, R.; Reyes-Resina, I.; Raich, I.; Pintor, J.J.; Alkozi, H.A.; Navarro, G.; Franco, R. Specificity and nanomolar potency of melatonin on G-protein coupled melatonin MT1 and MT2 receptors expressed in HEK-293T human embryo kidney cells. Melatonin Res. 2019, 2, 121–131. [Google Scholar] [CrossRef]
- Marcellino, D.; Ferré, S.; Casadó, V.; Cortés, A.; Le Foll, B.; Mazzola, C.; Drago, F.; Saur, O.; Stark, H.; Soriano, A.; et al. Identification of dopamine D1-D3 receptor heteromers: Indications for a role of synergistic D1-D3 receptor interactions in the striatum. J. Biol. Chem. 2008, 283, 26016–26025. [Google Scholar] [CrossRef] [Green Version]
- Alkozi, H.A.; Perez de Lara, M.J.; Sánchez-Naves, J.; Pintor, J. TRPV4 Stimulation Induced Melatonin Secretion by Increasing Arylalkymine N-acetyltransferase (AANAT) Protein Level. Int. J. Mol. Sci. 2017, 18, 746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ng, G.Y.K.; O’Dowd, B.F.; Lee, S.P.; Chung, H.T.; Brann, M.R.; Seeman, P.; George, S.R. Dopamine D2 receptor dimers and receptor-blocking peptides. Biochem. Biophys. Res. Commun. 1996, 227, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.P.; So, C.H.; Rashid, A.J.; Varghese, G.; Cheng, R.; Lança, A.J.; O’Dowd, B.F.; George, S.R. Dopamine D1 and D2 receptor Co-activation generates a novel phospholipase C-mediated calcium signal. J. Biol. Chem. 2004, 279, 35671–35678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perreault, M.L.; Hasbi, A.; Alijaniaram, M.; Fan, T.; Varghese, G.; Fletcher, P.J.; Seeman, P.; O’Dowd, B.F.; George, S.R. The dopamine D1-D2 receptor heteromer localizes in dynorphin/enkephalin neurons: Increased high affinity state following amphetamine and in schizophrenia. J. Biol. Chem. 2010, 285, 36625–36634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, H.; Wang, Z.; Weng, S.J.; Sun, X.H.; Yang, X.L. Neuromodulatory role of melatonin in retinal information processing. Prog. Retin. Eye Res. 2013, 32, 64–87. [Google Scholar] [CrossRef] [PubMed]
- Nagle, C.A.; Cardinali, D.P.; Rosner, J.M. Retinal and pineal hydroxyindole-O-methyl transferases in the rat: Changes following cervical sympathectomy, pinealectomy or blinding. Endocrinology 1973, 92, 1560–1564. [Google Scholar] [CrossRef] [PubMed]
- Franco, R.; Aguinaga, D.; Jiménez, J.; Lillo, J.; Martínez-Pinilla, E.; Navarro, G. Biased receptor functionality versus biased agonism in G-protein-coupled receptors. Biomol. Concepts 2018, 9, 143–154. [Google Scholar] [CrossRef]
- Pérez de Lara, M.J.; Santano, C.; Guzmán-Aránguez, A.; Valiente-Soriano, F.J.; Avilés-Trigueros, M.; Vidal-Sanz, M.; de la Villa, P.; Pintor, J. Assessment of inner retina dysfunction and progressive ganglion cell loss in a mouse model of glaucoma. Exp. Eye Res. 2014, 122, 40–49. [Google Scholar] [CrossRef]
- John, S.W.M.; Smith, R.S.; Savinova, O.V.; Hawes, N.L.; Chang, B.; Turnbull, D.; Davisson, M.; Roderick, T.H.; Heckenlively, J.R. Essential iris atrophy, pigment dispersion, and glaucoma in DBA/2J mice. Investig. Ophthalmol. Vis. Sci. 1998, 39, 951–962. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Resina, I.; Awad Alkozi, H.; del Ser-Badia, A.; Sánchez-Naves, J.; Lillo, J.; Jiménez, J.; Pintor, J.; Navarro, G.; Franco, R. Expression of Melatonin and Dopamine D3 Receptor Heteromers in Eye Ciliary Body Epithelial Cells and Negative Correlation with Ocular Hypertension. Cells 2020, 9, 152. https://doi.org/10.3390/cells9010152
Reyes-Resina I, Awad Alkozi H, del Ser-Badia A, Sánchez-Naves J, Lillo J, Jiménez J, Pintor J, Navarro G, Franco R. Expression of Melatonin and Dopamine D3 Receptor Heteromers in Eye Ciliary Body Epithelial Cells and Negative Correlation with Ocular Hypertension. Cells. 2020; 9(1):152. https://doi.org/10.3390/cells9010152
Chicago/Turabian StyleReyes-Resina, Irene, Hanan Awad Alkozi, Anna del Ser-Badia, Juan Sánchez-Naves, Jaume Lillo, Jasmina Jiménez, Jesús Pintor, Gemma Navarro, and Rafael Franco. 2020. "Expression of Melatonin and Dopamine D3 Receptor Heteromers in Eye Ciliary Body Epithelial Cells and Negative Correlation with Ocular Hypertension" Cells 9, no. 1: 152. https://doi.org/10.3390/cells9010152
APA StyleReyes-Resina, I., Awad Alkozi, H., del Ser-Badia, A., Sánchez-Naves, J., Lillo, J., Jiménez, J., Pintor, J., Navarro, G., & Franco, R. (2020). Expression of Melatonin and Dopamine D3 Receptor Heteromers in Eye Ciliary Body Epithelial Cells and Negative Correlation with Ocular Hypertension. Cells, 9(1), 152. https://doi.org/10.3390/cells9010152






