mTOR-Dependent Stimulation of IL20RA Orchestrates Immune Cell Trafficking through Lymphatic Endothelium in Patients with Crohn’s Disease
Abstract
1. Introduction
2. Material and Methods
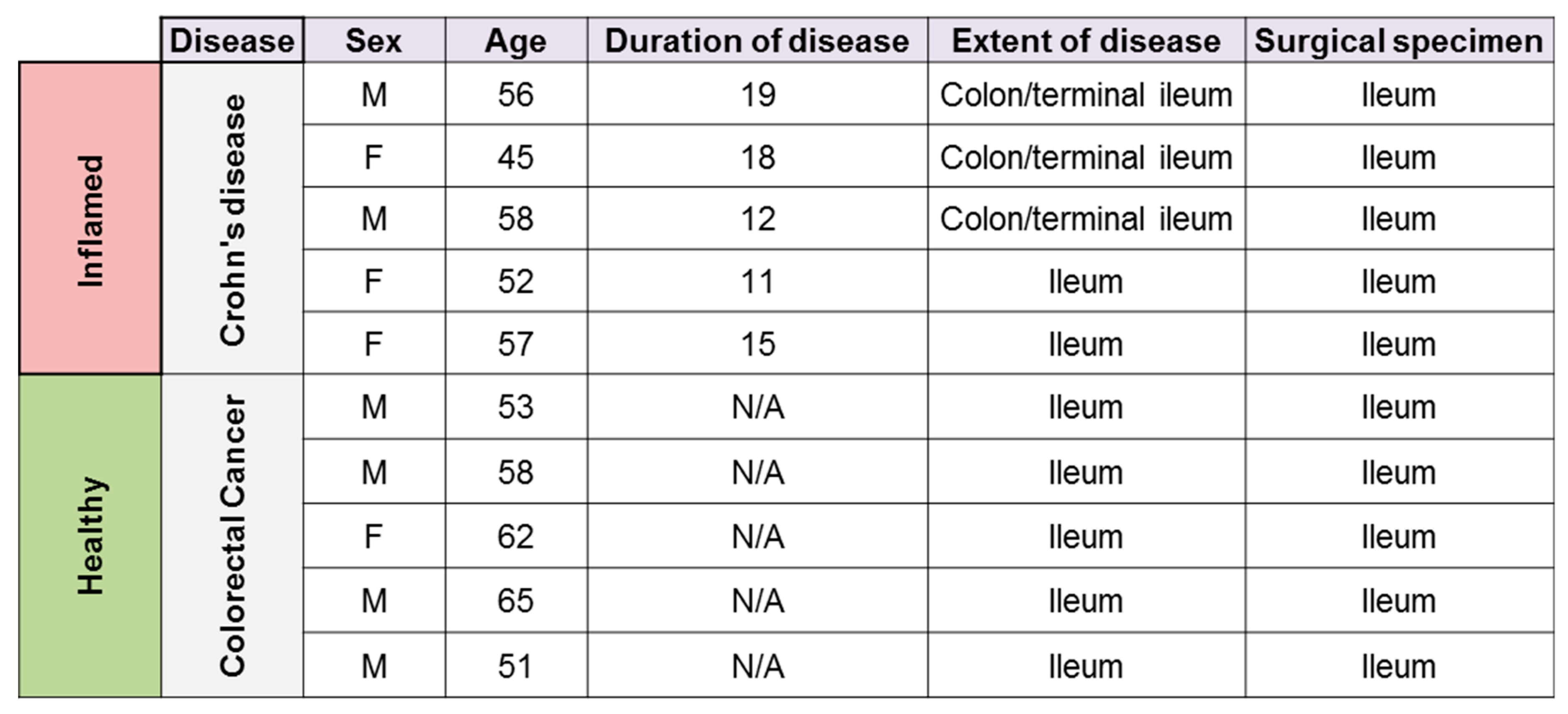
2.1. Human Intestinal Lymphatic Endothelial Cell (HILEC) Isolation and Culture and Lentiviral Transduction
2.2. RNA Extraction and Sequencing Analysis
2.3. Data Visualization
2.4. Lamina Propria Mononuclear Cell (LPMC) Isolation
2.5. Transwell-Based Transmigration Assay
2.6. Statistical Analysis and Data Visualization
3. Results
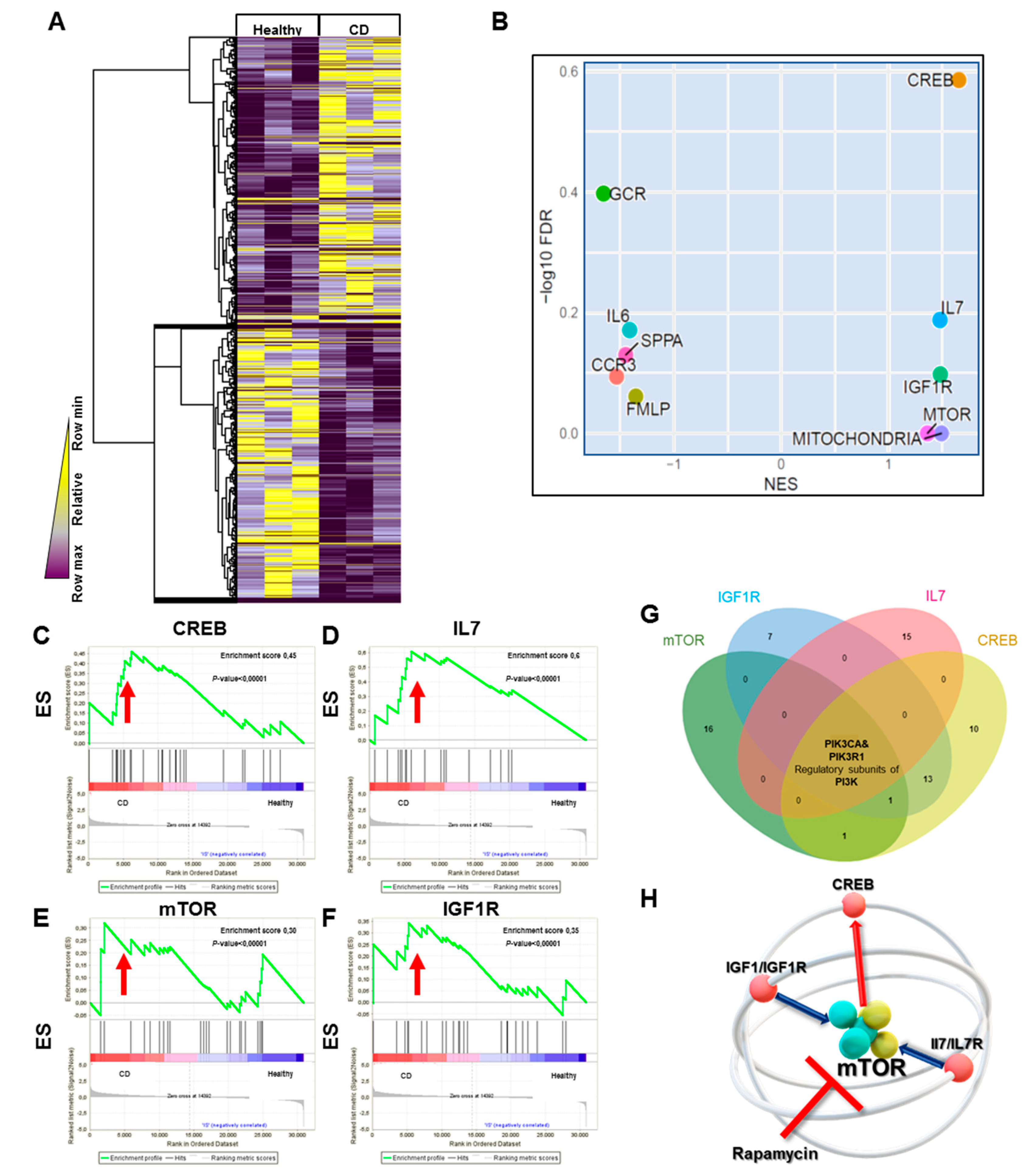
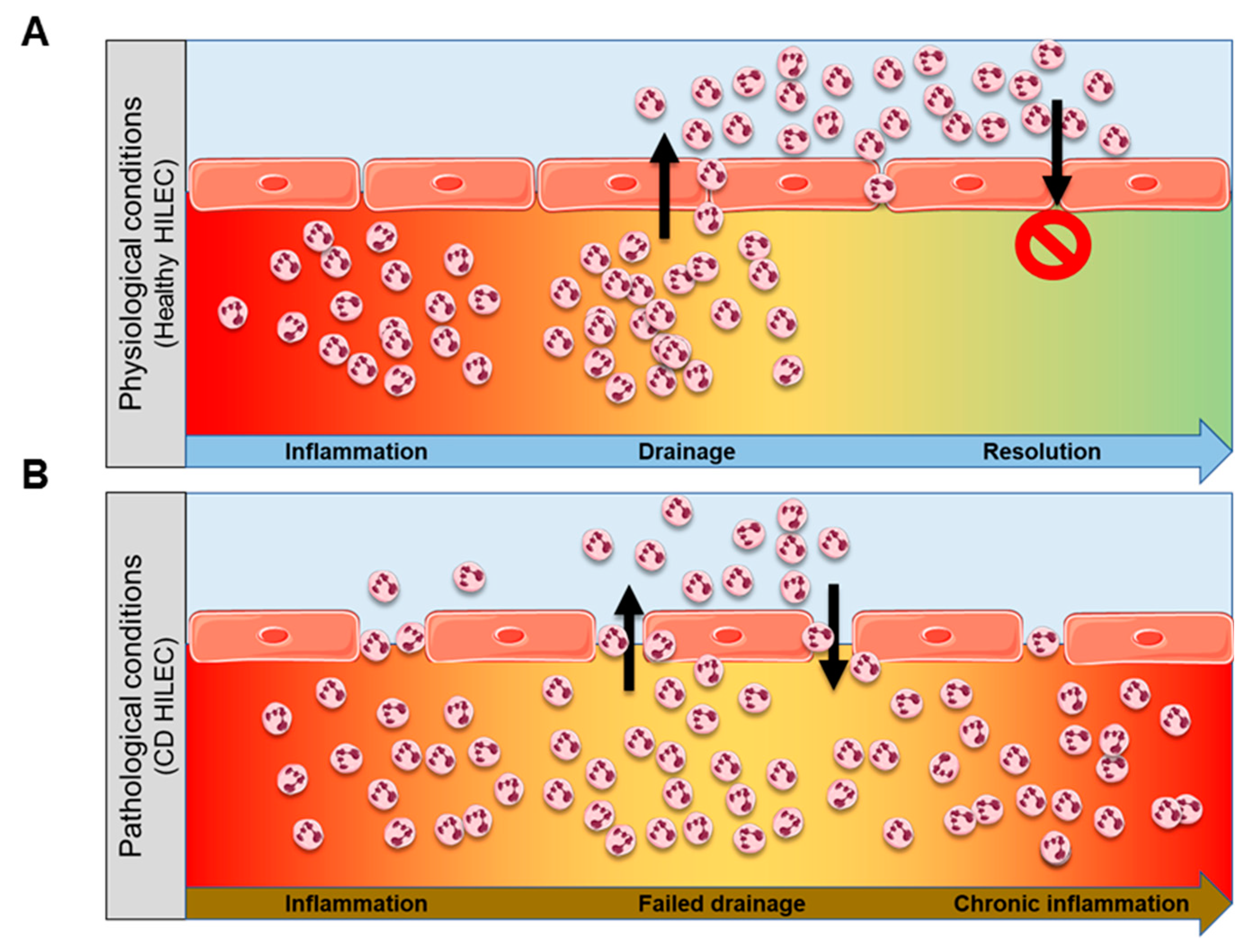
3.1. Crohn’s Disease (CD) HILECs Display Peculiar Transcriptomic Signatures
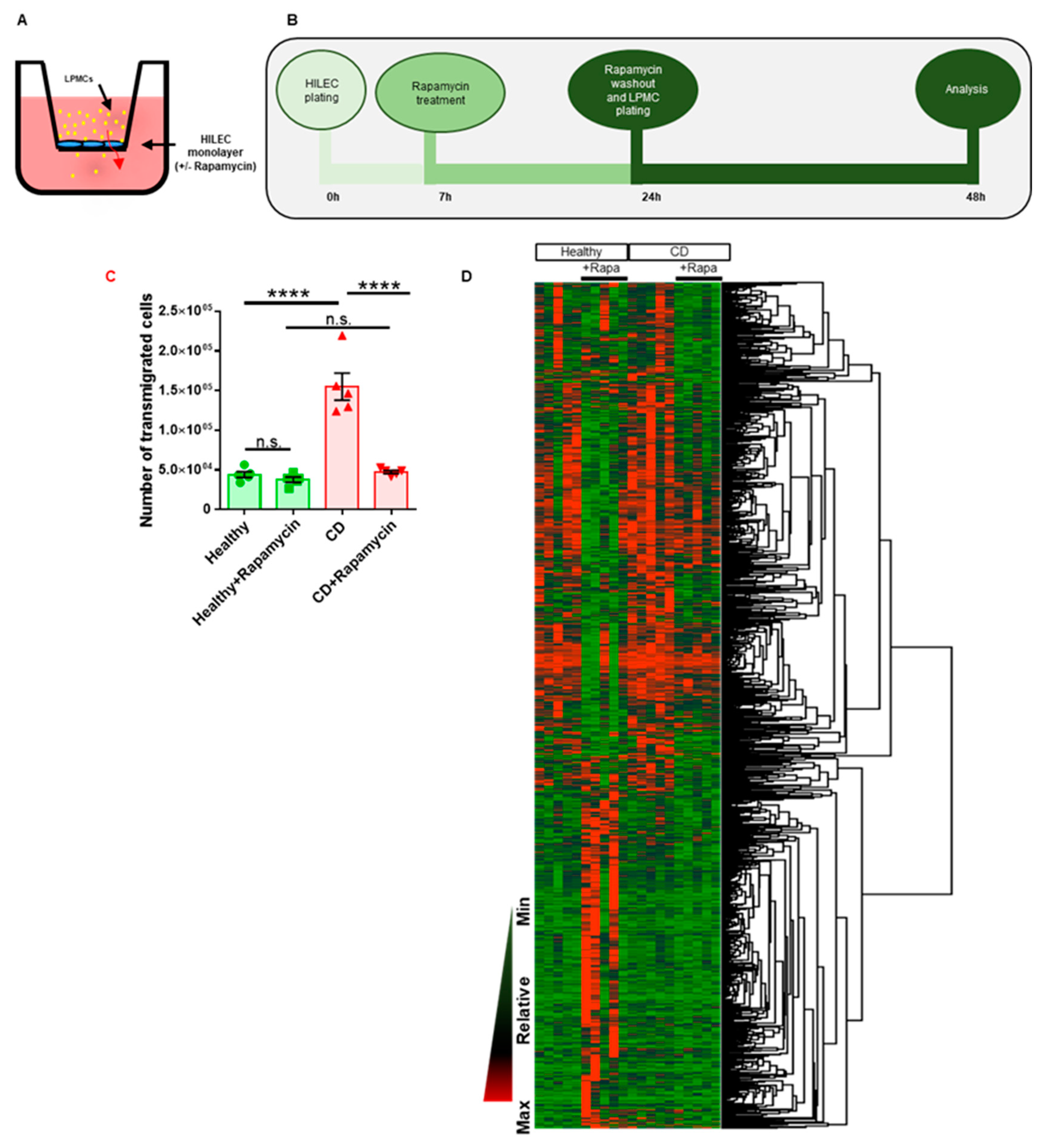
3.2. mTOR Pathway Oversees LPMC Translocation Across CD HILEC
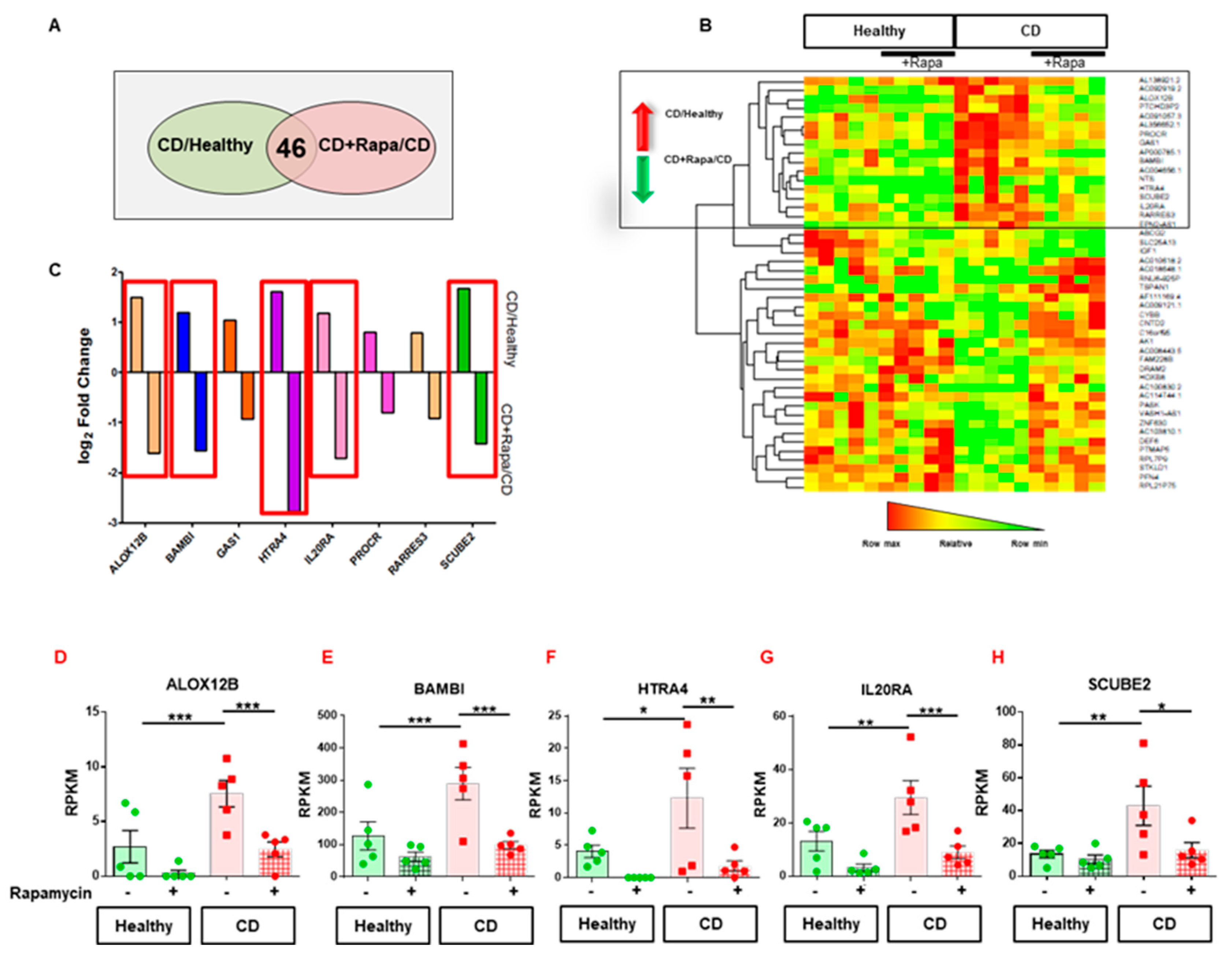
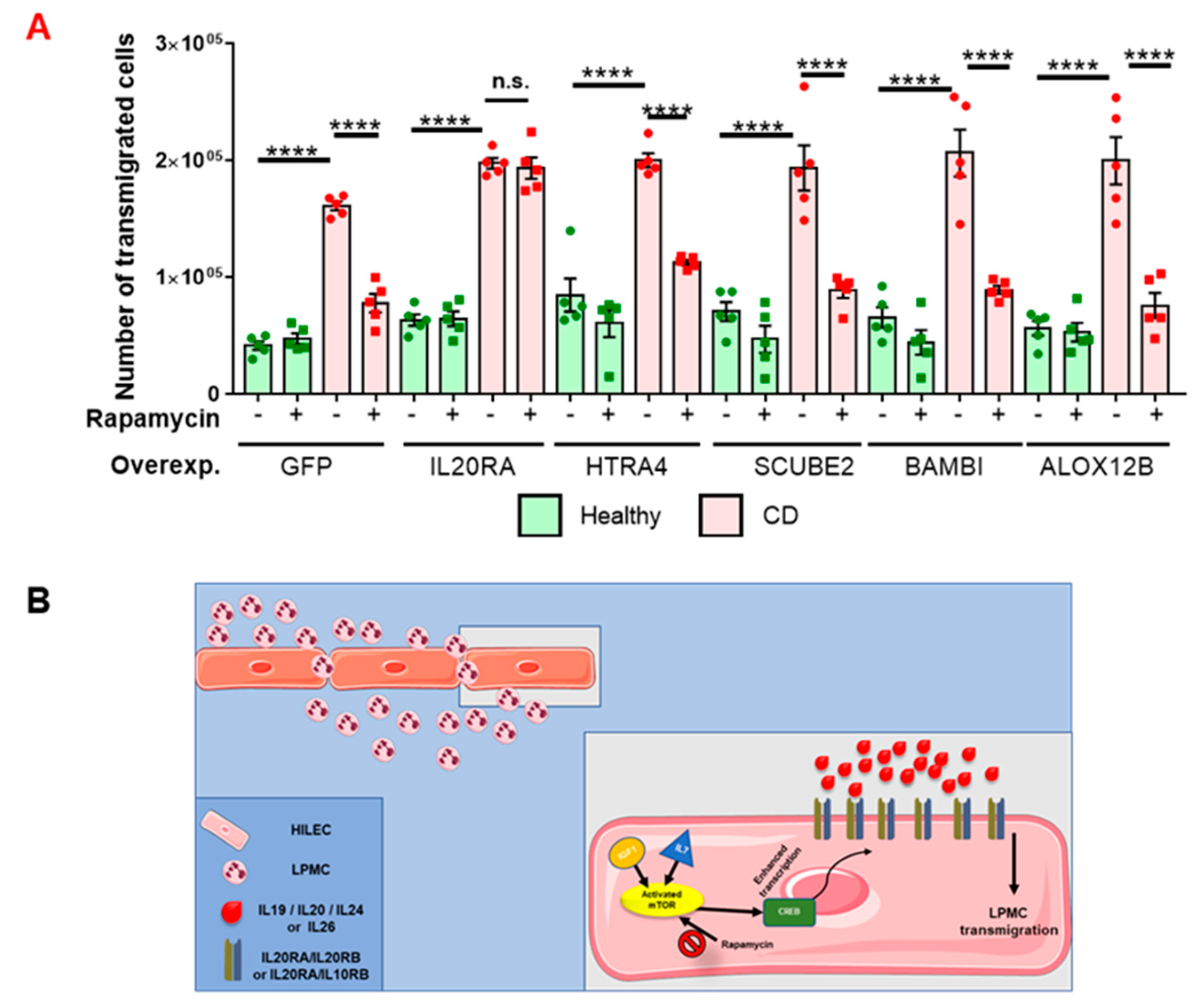
3.3. mTOR-Dependent IL20RA Expression Regulates LPMC Transmigration Across CD HILECs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Torres, J.; Mehandru, S.; Colombel, J.-F.; Peyrin-Biroulet, L. Crohn’s disease. Lancet 2017, 389, 1741–1755. [Google Scholar] [CrossRef]
- D’Alessio, S.; Correale, C.; Tacconi, C.; Gandelli, A.; Pietrogrande, G.; Vetrano, S.; Genua, M.; Arena, V.; Spinelli, A.; Peyrin-Biroulet, L.; et al. VEGF-C-dependent stimulation of lymphatic function ameliorates experimental inflammatory bowel disease. J. Clin. Invest. 2014, 124, 3863–3888. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-W.; Song, J.-H. Emerging Roles of Lymphatic Vasculature in Immunity. Immune Netw. 2017, 17, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Randolph, G.J.; Bala, S.; Rahier, J.-F.; Johnson, M.W.; Wang, P.L.; Nalbantoglu, I.; Dubuquoy, L.; Chau, A.; Pariente, B.; Kartheuser, A.; et al. Lymphoid Aggregates Remodel Lymphatic Collecting Vessels that Serve Mesenteric Lymph Nodes in Crohn Disease. Am. J. Pathol. 2016, 186, 3066–3073. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, F.; Giudici, F.; Liscia, G. Is lymphatic status related to regression of inflammation in Crohn’s disease? World J. Gastrointest. Surg. 2012, 4, 228–233. [Google Scholar] [CrossRef][Green Version]
- von der Weid, P.-Y.; Rehal, S.; Ferraz, J.G. Role of the lymphatic system in the pathogenesis of Crohn’s disease. Curr. Opin. Gastroenterol. 2011, 27, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Rahier, J.-F.; Dubuquoy, L.; Colombel, J.-F.; Jouret-Mourin, A.; Delos, M.; Ferrante, M.; Sokol, H.; De Hertogh, G.; Salleron, J.; Geboes, K.; et al. Decreased lymphatic vessel density is associated with postoperative endoscopic recurrence in Crohn’s disease. Inflamm. Bowel Dis. 2013, 19, 2084–2090. [Google Scholar] [CrossRef]
- Hunter, M.C.; Teijeira, A.; Halin, C. T Cell Trafficking through Lymphatic Vessels. Front. Immunol. 2016. [Google Scholar] [CrossRef]
- Johnson, L.A.; Jackson, D.G. Control of dendritic cell trafficking in lymphatics by chemokines. Angiogenesis 2014, 17, 335–345. [Google Scholar] [CrossRef]
- Platt, A.M.; Randolph, G.J. Dendritic cell migration through the lymphatic vasculature to lymph nodes. Adv. Immunol. 2013, 120, 51–68. [Google Scholar]
- Maisel, K.; Sasso, M.S.; Potin, L.; Swartz, M.A. Exploiting lymphatic vessels for immunomodulation: Rationale, opportunities, and challenges. Adv. Drug Deliv. Rev. 2017, 114, 43–59. [Google Scholar] [CrossRef]
- Ungaro, F.; Colombo, P.; Massimino, L.; Ugolini, G.S.; Correale, C.; Rasponi, M.; Garlatti, V.; Rubbino, F.; Tacconi, C.; Spaggiari, P.; et al. Lymphatic endothelium contributes to colorectal cancer growth via the soluble matrisome component GDF11. Int. J. Cancer 2019, 145, 1913–1920. [Google Scholar] [CrossRef]
- Ungaro, F.; Tacconi, C.; Massimino, L.; Corsetto, P.A.; Correale, C.; Fonteyne, P.; Piontini, A.; Garzarelli, V.; Calcaterra, F.; Della Bella, S.; et al. MFSD2A Promotes Endothelial Generation of Inflammation-resolving Lipid Mediators and Reduces Colitis in Mice. Gastroenterology 2017. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Yoon, M.-S. The Role of Mammalian Target of rapamycin (mTOR) in Insulin Signaling. Nutrients 2017, 9, 1176. [Google Scholar] [CrossRef]
- Venet, F.; Demaret, J.; Blaise, B.J.; Rouget, C.; Girardot, T.; Idealisoa, E.; Rimmelé, T.; Mallet, F.; Lepape, A.; Textoris, J.; et al. IL-7 Restores T Lymphocyte Immunometabolic Failure in Septic Shock Patients through mTOR Activation. J. Immunol. 2017, 199, 1606–1615. [Google Scholar] [CrossRef]
- Singh, A.K.; Kashyap, M.P.; Tripathi, V.K.; Singh, S.; Garg, G.; Rizvi, S.I. Neuroprotection Through rapamycin-Induced Activation of Autophagy and PI3K/Akt1/mTOR/CREB Signaling Against Amyloid-β-Induced Oxidative Stress, Synaptic/Neurotransmission Dysfunction, and Neurodegeneration in Adult Rats. Mol. Neurobiol. 2017, 54, 5815–5828. [Google Scholar] [CrossRef]
- Jones, R.G.; Pearce, E.J. MenTORing Immunity: mTOR Signaling in the Development and Function of Tissue-Resident Immune Cells. Immunity 2017, 46, 730–742. [Google Scholar] [CrossRef]
- Powell, J.D.; Pollizzi, K.N.; Heikamp, E.B.; Horton, M.R. Regulation of Immune Responses by mTOR. Annu. Rev. Immunol. 2012, 30, 39–68. [Google Scholar] [CrossRef]
- Bürger, C.; Shirsath, N.; Lang, V.; Diehl, S.; Kaufmann, R.; Weigert, A.; Han, Y.; Ringel, C.; Wolf, P. Blocking mTOR Signalling with rapamycin Ameliorates Imiquimod-induced Psoriasis in Mice. Acta Derm. Venereol. 2017, 97, 1087–1094. [Google Scholar] [CrossRef]
- Hu, S.; Chen, M.; Wang, Y.; Wang, Z.; Pei, Y.; Fan, R.; Liu, X.; Wang, L.; Zhou, J.; Zheng, S.; et al. mTOR Inhibition Attenuates Dextran Sulfate Sodium-Induced Colitis by Suppressing T Cell Proliferation and Balancing TH1/TH17/Treg Profile. PLoS ONE 2016, 11, e0154564. [Google Scholar] [CrossRef]
- Lyons, J.; Ghazi, P.C.; Starchenko, A.; Tovaglieri, A.; Baldwin, K.R.; Poulin, E.J.; Gierut, J.J.; Genetti, C.; Yajnik, V.; Breault, D.T.; et al. The colonic epithelium plays an active role in promoting colitis by shaping the tissue cytokine profile. PLoS Biol. 2018, 16, e2002417. [Google Scholar] [CrossRef]
- García-Mauriño, S.; Alcaide, A.; Domínguez, C. Pharmacological control of autophagy: Therapeutic perspectives in inflammatory bowel disease and colorectal cancer. Curr. Pharm. Des. 2012, 18, 3853–3873. [Google Scholar] [CrossRef]
- Matsuda, C.; Ito, T.; Song, J.; Mizushima, T.; Tamagawa, H.; Kai, Y.; Hamanaka, Y.; Inoue, M.; Nishida, T.; Matsuda, H.; et al. Therapeutic effect of a new immunosuppressive agent, everolimus, on interleukin-10 gene-deficient mice with colitis. Clin. Exp. Immunol. 2007, 148, 348–359. [Google Scholar] [CrossRef]
- Reinisch, W.; Panés, J.; Lémann, M.; Schreiber, S.; Feagan, B.; Schmidt, S.; Sturniolo, G.C.; Mikhailova, T.; Alexeeva, O.; Sanna, L.; et al. A multicenter, randomized, double-blind trial of everolimus versus azathioprine and placebo to maintain steroid-induced remission in patients with moderate-to-severe active Crohn’s disease. Am. J. Gastroenterol. 2008, 103, 2284–2292. [Google Scholar] [CrossRef]
- Massey, D.C.O.; Bredin, F.; Parkes, M. Use of sirolimus (rapamycin) to treat refractory Crohn’s disease. Gut 2008, 57, 1294–1296. [Google Scholar] [CrossRef]
- Mutalib, M.; Borrelli, O.; Blackstock, S.; Kiparissi, F.; Elawad, M.; Shah, N.; Lindley, K. The use of sirolimus (rapamycin) in the management of refractory inflammatory bowel disease in children. J. Crohns. Colitis 2014, 8, 1730–1734. [Google Scholar] [CrossRef]
- Parrish-Novak, J.; Xu, W.; Brender, T.; Yao, L.; Jones, C.; West, J.; Brandt, C.; Jelinek, L.; Madden, K.; McKernan, P.A.; et al. Interleukins 19, 20, and 24 Signal through Two Distinct Receptor Complexes. J. Biol. Chem. 2002, 277, 47517–47523. [Google Scholar] [CrossRef]
- Sabat, R.; Wallace, E.; Endesfelder, S.; Wolk, K. IL-19 and IL-20: Two novel cytokines with importance in inflammatory diseases. Expert Opin. Ther. Targets 2007, 11, 601–612. [Google Scholar] [CrossRef]
- Blumberg, H.; Conklin, D.; Xu, W.; Grossmann, A.; Brender, T.; Carollo, S.; Eagan, M.; Foster, D.; Haldeman, B.A.; Hammond, A.; et al. Interleukin 20. Cell 2001, 104, 9–19. [Google Scholar] [CrossRef]
- Hammer, T.; Tritsaris, K.; Hübschmann, M.V.; Gibson, J.; Nisato, R.E.; Pepper, M.S.; Dissing, S. IL-20 activates human lymphatic endothelial cells causing cell signalling and tube formation. Microvasc. Res. 2009, 78, 25–32. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ungaro, F.; Garlatti, V.; Massimino, L.; Spinelli, A.; Carvello, M.; Sacchi, M.; Spanò, S.; Colasante, G.; Valassina, N.; Vetrano, S.; et al. mTOR-Dependent Stimulation of IL20RA Orchestrates Immune Cell Trafficking through Lymphatic Endothelium in Patients with Crohn’s Disease. Cells 2019, 8, 924. https://doi.org/10.3390/cells8080924
Ungaro F, Garlatti V, Massimino L, Spinelli A, Carvello M, Sacchi M, Spanò S, Colasante G, Valassina N, Vetrano S, et al. mTOR-Dependent Stimulation of IL20RA Orchestrates Immune Cell Trafficking through Lymphatic Endothelium in Patients with Crohn’s Disease. Cells. 2019; 8(8):924. https://doi.org/10.3390/cells8080924
Chicago/Turabian StyleUngaro, Federica, Valentina Garlatti, Luca Massimino, Antonino Spinelli, Michele Carvello, Matteo Sacchi, Salvatore Spanò, Gaia Colasante, Nicholas Valassina, Stefania Vetrano, and et al. 2019. "mTOR-Dependent Stimulation of IL20RA Orchestrates Immune Cell Trafficking through Lymphatic Endothelium in Patients with Crohn’s Disease" Cells 8, no. 8: 924. https://doi.org/10.3390/cells8080924
APA StyleUngaro, F., Garlatti, V., Massimino, L., Spinelli, A., Carvello, M., Sacchi, M., Spanò, S., Colasante, G., Valassina, N., Vetrano, S., Malesci, A., Peyrin-Biroulet, L., Danese, S., & D’Alessio, S. (2019). mTOR-Dependent Stimulation of IL20RA Orchestrates Immune Cell Trafficking through Lymphatic Endothelium in Patients with Crohn’s Disease. Cells, 8(8), 924. https://doi.org/10.3390/cells8080924






