Mesenchymal Stem Cell Migration and Tissue Repair
Abstract
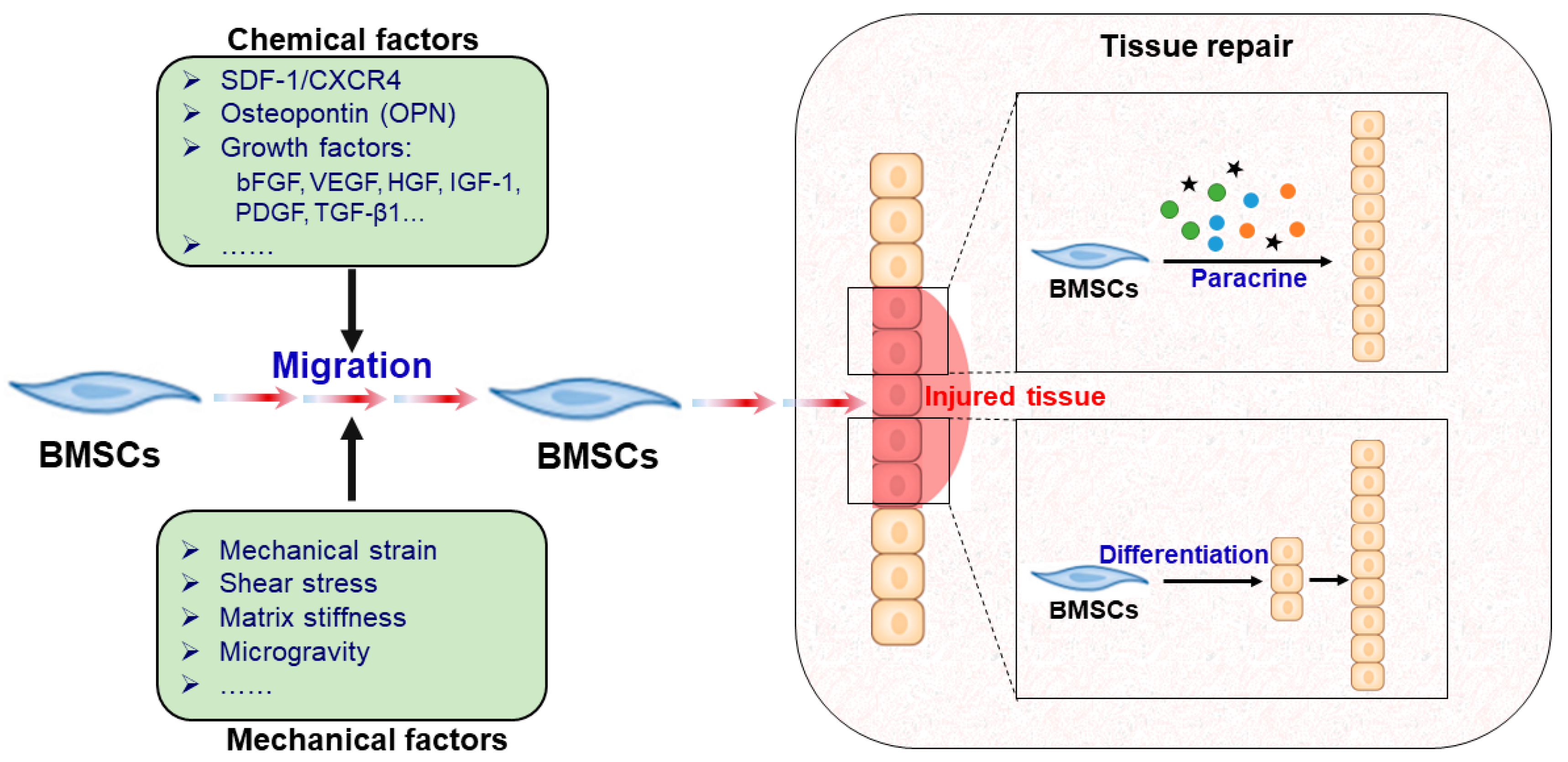
:1. Introduction
2. Chemical Factors Regulating BMSC Migration
2.1. SDF-1/CXCR4 Axis
2.2. Osteopontin (OPN)
2.3. Growth Factors
2.3.1. bFGF
2.3.2. VEGF
2.3.3. HGF
2.3.4. IGF-1
2.3.5. PDGF
2.3.6. TGF-β1
3. Mechanical Factors Regulating BMSC Migration
3.1. Mechanical Strain
3.2. Shear Stress
3.3. Matrix Stiffness
3.4. Microgravity
| Mechanical Regime | Cell Migration | Outcomes | References |
|---|---|---|---|
| Mechanical stretch (5%, 6 h) | ↑ | Enhanced homing and transdifferentiation of BMSCs under mechanical stretch in the expanded skin, and BMSCs were recruited to sites where SDF-1αwas most highly expressed. | [67] |
| Mechanical stretch (10%, 8 h) | ↑ | Promoted BMSC migration via FAK and ERK1/2 signals. | [68] |
| Mechanical stretch (10%, 12 h) | ↑ | In vivo and in vitro results showed that mechanical stretch can upregulate SDF-1α in skin and recruit circulating BMSCs through the SDF-1α/CXCR4 pathway. | [70] |
| Shear stress (0.2 Pa/>2 Pa) | ↓↑ | High shear stress (>2 Pa) hindered human BMSC migration, whereas lower shear stress (0.2 Pa) induced cell migration. | [71] |
| Shear stress (0.2 Pa) | ↑ | The SDF-1/CXCR4 axis mediated low-shear-stress-induced human BMSC migration through the JNK and p38 MAPK pathways. | [72] |
| Matrix stiffness (1 kPa, 2.3 h; 34 kPa, 6.3 h) | ↑ | BMSCs migrated from the soft matrix to the stiff matrix by polarizing the cytoskeleton function and the phosphorylated myosin-II heavy chain. | [74] |
| Matrix stiffness (≥5-6 kPa, 2 h) | ↑ | Extracellular matrix (ECM) stiffness influenced the position of the microtubule organizing center (MTOC) in MSCs by polarizing it in front of the nucleus only when the matrix was sufficiently stiff, which increased MSC migration. | [75] |
| Matrix stiffness (1 to 12 kPa, 3 days) | ↑ | Human MSCs migrated to stiffer portions of the substrates by increasing the assembled microtubule network. | [73] |
| Matrix stiffness (2 kPa, 4 h) | ↑ | AFSCs cultured on softer substrates secreted more autocrine cytokines, which increased AFSC migration. | [76] |
| Microgravity (rotated at 10 rpm, approximately 1 × 10−3 g; 24 h) | ↓ | The migration of BMSCs was inhibited by simulated microgravity via reorganizing F-actin and increasing cell stiffness. | [82] |
| Microgravity (rotated at 10 to 12 rpm, approximately 1 × 10–3 g to 1.2×10–3 g; 2 to 3 days) | ↓ | The culture of HSCs in a microgravity environment inhibited the migration of HSCs by a significant reduction of SDF-1α-directed migration, which correlated with a decreased expression of F-actin. | [83] |
4. Mechanisms of BMSCs in Tissue Repair
4.1. BMSC Paracrine Factors and Tissue Repair
4.1.1. Paracrine Factors of Transplanting BMSCs
4.1.2. Conditioned Medium from BMSCs for Tissue Repair
4.2. Directed Differentiation of BMSCs and Tissue Repair
5. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AFSCs | Amniotic fluid-derived stem cells |
| bFGF | Basic fibroblast growth factor |
| BMSCs | Bone marrow-derived mesenchymal stem cells |
| BMSC-CM | Conditioned medium from BMSCs |
| CCL2 | CC chemokine ligand 2 |
| CCR2 | CC chemokine receptor 2 |
| CM | Conditioned medium |
| CRE | Cockroach allergen extract |
| CXCL7 | Chemokine (C-X-C motif) ligand 7 |
| CXCR4 | CXC chemokine receptor 4 |
| ECM | Extracellular matrix |
| EGF | Epithelial growth factor |
| ERK1/2 | Extracellular signal-regulated kinase 1/2 |
| ESCs | Embryonic stem cells |
| FAK | Focal adhesion kinase |
| Flt3 | Fms-like tyrosine kinase 3 |
| GDF-11 | Growth differentiation factor-11 |
| GFP | Green fluorescent protein |
| HGF | Hepatocyte growth factor |
| HSCs | Hematopoietic stem cells |
| HMGB1 | High mobility group box 1 |
| IGF-1 | Insulin-like growth factor-1 |
| IL1-RA | Interleukin-1 receptor antagonist |
| IL-6 | Interleukin 6 |
| IL-8 | Interleukin-8 |
| IL-10 | Interleukin-10 |
| JNK | c-Jun N-terminal kinase |
| LL-37 | Leu-Leu-37 |
| MAPK | Mitogen-activated protein kinase |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MSCs | Mesenchymal stem cells |
| MTOC | Microtubule organizing center |
| NF-κB | Nuclear factor kappa B |
| NGF | Nerve growth factor |
| NOD/SCID | Non-obese diabetic/severe combined immunodeficiency |
| OPN | Osteopontin |
| PDGF | Platelet derived growth factor |
| PDGF-AB | Platelet derived growth factor isoforms AB |
| PDGF-BB | Platelet derived growth factor isoforms BB |
| PDGF-CC | Platelet derived growth factor isoforms CC |
| PDGFRs | Platelet-derived growth factor receptors |
| PDGFR-α | Platelet-derived growth factor receptor isoform α |
| PDGFR-β | Platelet-derived growth factor receptor isoform β |
| PI3K | Phosphatidylinositol 3-kinase |
| RAGE | Receptor for advanced glycation end |
| Rho A | Ras homolog gene family member A |
| SCF | Stem cell factor |
| SDF-1 | Stromal derived factor-1 |
| SUN1 | Sad-1/UNC-84 1 |
| TGF-β1 | Transforming growth factor β1 |
| TNF-α | Tumor necrosis factor |
| UC-MSC-CM | Umbilical cord blood-derived mesenchymal stem cells conditioned media |
| VEGF | Vascular endothelial growth factor |
| VEGFR | Vascular endothelial growth factor receptor |
| WJ-MSCs | Wharton’s jelly-derived mesenchymal stem cells |
References
- Selvasandran, K.; Makhoul, G.; Jaiswal, P.K.; Jurakhan, R.; Li, L.; Ridwan, K.; Cecere, R.A. Tumor necrosis factor-α and hypoxia-induced secretome therapy for myocardial repair. Ann. Thorac. Surg. 2017, 105, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.J.; Lee, H.W.; Kalimuthu, S.; Kim, T.J.; Kim, H.M.; Baek, S.H.; Zhu, L.; Ji, M.O.; Son, S.H.; Chung, H.Y. In vivo migration of mesenchymal stem cells to burn injury sites and their therapeutic effects in a living mouse model. J. Control. Release 2018, 279, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Katagiri, W.; Osugi, M.; Sugimura, Y.; Hibi, H.; Ueda, M. Secretomes from bone marrow–derived mesenchymal stromal cells enhance periodontal tissue regeneration. Cytotherapy 2015, 17, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Lee, S.G.; Lee, S.W.; Oh, B.J.; Kim, J.H.; Kim, J.A.; Lee, G.; Jang, J.D.; Joe, Y.A. A Subset of paracrine factors as efficient biomarkers for predicting vascular regenerative efficacy of mesenchymal stromal/stem cells. Stem Cells 2018, 37, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Ishikawa, H.; Kawai, K.; Tabata, Y.; Suzuki, S. Enhanced wound healing by topical administration of mesenchymal stem cells transfected with stromal cell-derived factor-1. Biomaterials 2013, 34, 9393–9400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, T.; Lv, Z.; Chen, Q.; Guo, M.; Wang, X.; Huang, F. Vascular endothelial growth factor over-expressed mesenchymal stem cells-conditioned media ameliorate palmitate-induced diabetic endothelial dysfunction through PI-3K/AKT/m-TOR/eNOS and p38/MAPK signaling pathway. Biomed. Pharmacother. 2018, 106, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Qian, J.; Ma, J.; Huang, Z.; Shen, Y.; Chen, X.; Sun, A.; Ge, J.; Chen, H. Myocardial transfection of hypoxia-inducible factor-1α and co-transplantation of mesenchymal stem cells enhance cardiac repair in rats with experimental myocardial infarction. Stem Cell Res. Ther. 2014, 5, 22. [Google Scholar] [CrossRef]
- Folestad, E.; Kunath, A.; Wagsäter, D. PDGF-C and PDGF-D signaling in vascular diseases and animal models. Mol. Asp. Med. 2018, 62, 1–11. [Google Scholar] [CrossRef]
- Zhang, S.J.; Song, X.Y.; He, M.; Yu, S.B. Effect of TGF-β1/SDF-1/CXCR4 signal on BM-MSCs homing in rat heart of ischemia/perfusion injury. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 899–905. [Google Scholar]
- Gnecchi, M.; Danieli, P.; Malpasso, G.; Ciuffreda, M.C. Paracrine Mechanisms of Mesenchymal Stem Cells in Tissue Repair. Methods Mol. Biol. 2016, 1416, 123–146. [Google Scholar]
- Liu, C.; Tsai, A.L.; Li, P.C.; Huang, C.W.; Wu, C.C. Endothelial differentiation of bone marrow mesenchyme stem cells applicable to hypoxia and increased migration through Akt and NFκB signals. Stem Cell Res. Ther. 2017, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Song, G.; Ju, Y.; Li, X.; Song, Y.; Watanabe, S. RhoA/ROCK, cytoskeletal dynamics, and focal adhesion kinase are required for mechanical stretch-induced tenogenic differentiation of human mesenchymal stem cells. J. Cell. Physiol. 2012, 227, 2722–2729. [Google Scholar] [CrossRef] [PubMed]
- Sacchetti, B.; Funari, A.; Michienzi, S.; Di Cesare, S.; Piersanti, S.; Saggio, I.; Tagliafico, E.; Ferrari, S.; Robey, P.G.; Riminucci, M.; et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 2007, 131, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Funari, A.; Alimandi, M.; Pierelli, L.; Pino, V.; Gentileschi, S.; Sacchetti, B. Human sinusoidal subendothelial cells regulate homing and invasion of circulating metastatic prostate cancer cells to bone marrow. Cancers 2019, 11, 763. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Xu, L.; Zwingenberger, S.; Gibon, E.; Goodman, S.B.; Li, G. Mesenchymal stem cells homing to improve bone healing. J. Orthop. Transl. 2017, 9, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Kuai, X.L.; Li, P.; Zhang, J.F.; Cao, W.; Yuan, W.Y.; Shao, N.; Qi, G.C.; Wang, Z.W. Stromal derived factor-1/CXCR4 sxis involved in bone marrow mesenchymal stem cells recruitment to injured liver. Stem Cells Int. 2016, 2016, 1–10. [Google Scholar]
- Deng, Q.J.; Xu, X.F.; Ren, J. Effects of SDF-1/CXCR4 on the repair of traumatic brain injury in rats by mediating bone marrow derived mesenchymal stem cells. Cell. Mol. Neurobiol. 2017, 38, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, K.; Kołodziejczyk, A.; Sikorska, M.; Płaczkiewicz, J.; Cichosz, P.; Kowalewska, M.; Stremińska, W.; Jańczykilach, K.; Koblowska, M.; Fogtman, A. Stem cells migration during skeletal muscle regeneration - the role of SDF-1/Cxcr4 and Sdf-1/Cxcr7 axis. Cell Adhes. Migr. 2016, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Song, G.; Luo, Q.; Yuan, L.; Yang, L. Mesenchymal stem cells require integrin β1 for directed migration induced by osteopontin in vitro. Vitr. Cell. Dev. Biol. Anim. 2011, 47, 241–250. [Google Scholar] [CrossRef]
- Leah, F.R.; Genetos, D.C.; Yellowley, C.E. Hypoxic osteocytes recruit human MSCs through an OPN/CD44-mediated pathway. Biochem. Biophys. Res. Commun. 2008, 366, 1061–1066. [Google Scholar]
- Zou, C.; Luo, Q.; Qin, J.; Shi, Y.; Yang, L.; Ju, B.; Song, G. Osteopontin romotes mesenchymal stem cell migration and lessens cell stiffness via integrin beta 1, FAK, and ERK pathways. Cell Biochem. Biophys. 2013, 65, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Luo, Q.; Sun, J.; Wang, A.; Shi, Y.; Ju, Y.; Morita, Y.; Song, G. Decreased nuclear stiffness via FAK-ERK1/2 signaling is necessary for osteopontin-promoted migration of bone marrow-derived mesenchymal stem cells. Exp. Cell Res. 2017, 355, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Luo, Q.; Sun, J.; Song, G. Cytoskeletal control of nuclear morphology and stiffness are required for OPN-induced bone marrow-derived mesenchymal stem cell migration. Biochem. Cell Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Luo, Q.; Sun, J.; Ju, Y.; Morita, Y.; Song, G. Chromatin organization regulated by EZH2-mediated H3K27me3 is required for OPN-induced migration of bone marrow-derived mesenchymal stem cells. Int. J. Biochem. Cell Biol. 2018, 96, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhen, L.; Miao, H.; Sun, Q.; Yang, Y.; Que, B.; Lopes Lao, E.P.; Wu, X.; Ren, H.; Shi, S.; et al. Concomitant retrograde coronary venous infusion of basic fibroblast growth factor enhances engraftment and differentiation of bone marrow mesenchymal stem cells for cardiac repair after myocardial infarction. Theranostics 2015, 5, 995–1006. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Ladage, D.; Schinköthe, T.; Klausmann, U.; Ulrichs, C.; Klinz, F.J.; Brixius, K.; Arnhold, S.; Desai, B.; Mehlhorn, U. Basic fibroblast growth factor controls migration in human mesenchymal stem cells. Stem Cells 2010, 24, 1750–1758. [Google Scholar] [CrossRef] [PubMed]
- Ball, S.G.; Shuttleworth, C.A.; Kielty, C.M. Vascular endothelial growth factor can signal through platelet-derived growth factor receptors. J. Cell Biol. 2007, 177, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Forte, G.; Minieri, M.; Cossa, P.; Antenucci, D.; Sala, M.; Gnocchi, V.; Fiaccavento, R.; Carotenuto, F.; De Vito, P.; Baldini, P.M.; et al. Hepatocyte growth factor effects on mesenchymal stem cells: Proliferation, migration, and differentiation. Stem Cells 2006, 24, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yu, X.; Lin, S.; Li, X.; Zhang, S.; Song, Y.H. Insulin-like growth factor 1 enhances the migratory capacity of mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2007, 356, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Xinaris, C.; Morigi, M.; Benedetti, V.; Imberti, B.; Fabricio, A.S.; Squarcina, E.; Benigni, A.; Gagliardini, E.; Remuzzi, G. A novel strategy to enhance mesenchymal stem cell migration capacity and promote tissue repair in an injury specific fashion. Cell Transplant 2013, 22, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Mishima, Y.; Lotz, M. A14 chemotaxis of human articular chondrocytes and mesenchymal stem cells. Osteoarthr. Cartil. 2008, 16, S19. [Google Scholar] [CrossRef]
- Nedeau, A.E.; Bauer, R.J.; Gallagher, K.; Chen, H.; Liu, Z.J.; Velazquez, O.C. A CXCL5- and bFGF-dependent effect of PDGF-B-activated fibroblasts in promoting trafficking and differentiation of bone marrow-derived mesenchymal stem cells. Exp. Cell Res. 2008, 314, 2176–2186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, D.; Mcgrail, D.J.; Dawson, M.R. TGF-β1 pretreatment improves the function of mesenchymal stem cells in the wound bed. Front. Cell Dev. Biol. 2017, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Dubon, M.J.; Yu, J.; Choi, S.; Park, K. Transforming growth factor β induces bone marrow mesenchymal stem cell migration via noncanonical signals and N-cadherin. J. Cell. Physiol. 2018, 233, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Duan, B.; Cheng, Z.; Jia, X.; Mao, L.; Fu, H.; Kong, D. SDF-1/CXCR4 axis modulates bone marrow mesenchymal stem cell apoptosis, migration and cytokine secretion. Protein Cell 2011, 2, 845–854. [Google Scholar] [CrossRef] [Green Version]
- Pillarisetti, K.; Gupta, S.K. Cloning and relative expression analysis of rat stromal cell derived factor-1 (SDF-1)1: SDF-1 alpha mRNA is selectively induced in rat model of myocardial infarction. Inflammation 2001, 25, 293–300. [Google Scholar] [CrossRef]
- Askari, A.T.; Samuel, U.; Popovic, Z.B.; Goldman, C.K.; Farhad, F.; Matthew, K.; Aleksandr, R.; Ellis, S.G.; Thomas, J.D.; Dicorleto, P.E. Effect of stromal-cell-derived factor 1 on stem-cell homing and tissue regeneration in ischaemic cardiomyopathy. Lancet 2003, 362, 697–703. [Google Scholar] [CrossRef]
- Sipkins, D.A.; Wei, X.B.; Wu, J.W.; Runnels, J.M.; Daniel, C.; Means, T.K.; Luster, A.D.; Scadden, D.T.; Lin, C.P. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature 2005, 435, 969–973. [Google Scholar] [CrossRef]
- Lien, C.Y.; Kevin, C.Y.H.; Lee, O.K.; Blun, B.W.; Yeu, S. Restoration of bone mass and strength in glucocorticoid-treated mice by systemic transplantation of CXCR4 and cbfa-1 co-expressing mesenchymal stem cells. J. Bone Miner. Res. 2010, 24, 837–848. [Google Scholar] [CrossRef]
- Cheng, Z.K.; Ou, L.L.; Zhou, X.; Li, F.; Jia, X.H.; Melo, L.G. Directed migration of mesenchymal stem cells modified with CXCR4 gene to infarcted myocardium improves cardiac performance. Mol. Ther. J. Am. Soc. Gene Ther. 2008, 16, 571–579. [Google Scholar] [CrossRef]
- Hirano, Y.; Aziz, M.; Yang, W.L.; Wang, Z.; Zhou, M.; Ochani, M.; Khader, A.; Wang, P. Neutralization of osteopontin attenuates neutrophil migration in sepsis-induced acute lung injury. Crit. Care 2015, 19, 53. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.T.; Zou, F.Z.; Cai, L.N.; Xu, W.L. The downregulation of OPN inhibits proliferation and migration and regulate activation of Erk1/2 in ECA-109 cells. Int. J. Clin. Exp. Med. 2015, 8, 5361–5369. [Google Scholar] [PubMed]
- Bear, J.E.; Haugh, J.M. Directed migration of mesenchymal cells: Where signaling and the cytoskeleton meet. Curr. Opin. Cell Biol. 2014, 30, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lovett, D.; Zhang, Q.; Neelam, S.; Kuchibhotla, R.A.; Zhu, R.; Gundersen, G.G.; Lele, T.P.; Dickinson, R.B. Moving cell boundaries drive nuclear shaping during cell spreading. Biophys. J. 2015, 109, 670–686. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.; Gu, S.; Cheng, Y.; Ding, L. bFGF promotes Sca-1+ cardiac stem cell migration through activation of the PI3K/Akt pathway. Mol. Med. Rep. 2018, 17, 2349–2356. [Google Scholar] [CrossRef]
- Xuan, Y.; Chi, L.; Tian, H.; Cai, W.; Sun, C.; Wang, T.; Zhou, X.; Shao, M.; Zhu, Y.; Niu, C.; et al. The activation of the NF-κB-JNK pathway is independent of the PI3K-Rac1-JNK pathway involved in the bFGF-regulated human fibroblast cell migration. J. Dermatol. Sci. 2016, 82, 28–37. [Google Scholar] [CrossRef]
- Tang, J.M.; Wang, J.N.; Zhang, L.; Zheng, F.; Yang, J.Y.; Kong, X.; Chen, S.Y. VEGF/SDF-1 promotes cardiac stem cell mobilization and myocardial repair in the infarcted heart. Cardiovasc. Res. 2011, 91, 402–411. [Google Scholar] [CrossRef] [Green Version]
- Langer, H.F.; Stellos, K.; Steingen, C.; Froihofer, A.; Bigalke, B.; May, A.E.; Seizer, P.; Müller, I. Platelet derived bFGF mediates vascular integrative mechanisms of mesenchymal stem cells in vitro. J. Mol. Cell. Cardiol. 2009, 47, 315–325. [Google Scholar] [CrossRef]
- Holmes, D.I.; Zachary, I. The vascular endothelial growth factor (VEGF) family: Angiogenic factors in health and disease. Genome Biol. 2005, 6, 209. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Morita, T. Molecular and functional diversity of vascular endothelial growth factors. Mol. Divers. 2006, 10, 515–527. [Google Scholar] [CrossRef]
- Trusolino, L.; Comoglio, P.M. Boswellia frereana suppresses HGF-mediated breast cancer cell invasion and migration through inhibition of c-Met signalling. J. Transl. Med. 2018, 16, 281. [Google Scholar] [Green Version]
- Zhu, A.; Kang, N.; He, L.; Li, X.; Xu, X.; Zhang, H. MiR-221 and miR-26b regulate chemotactic migration of MSCs toward HGF through activation of Akt and FAK. J. Cell. Biochem. 2016, 117, 1370–1383. [Google Scholar] [CrossRef] [PubMed]
- Mytilinaiou, M.; Nikitovic, D.; Berdiaki, A.; Papoutsidakis, A.; Papachristou, D.J.; Tsatsakis, A.; Tzanakakis, G.N. IGF-I regulates HT1080 fibrosarcoma cell migration through a syndecan-2/Erk/ezrin signaling axis. Exp. Cell Res. 2017, 36, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Manzat Saplacan, R.M.; Balacescu, L.; Gherman, C.; Chira, R.I.; Craiu, A.; Mircea, P.A.; Lisencu, C.; Balacescu, O. The Role of PDGFs and PDGFRs in Colorectal Cancer. Mediat. Inflamm. 2017, 2017, 4708076. [Google Scholar]
- Hart, C.E.; Forstrom, J.W.; Kelly, J.D.; Seifert, R.A.; Smith, R.A.; Ross, R.; Murray, M.J.; Bowen-Pope, D.F. Two classes of PDGF receptor recognize different isoforms of PDGF. Science 1988, 240, 1529–1531. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Yan, Y.; Komuves, L.G.; Yonkovich, S.; Sullivan, C.M.; Stringer, B.; Galbraith, S.; Lokker, N.A.; Hwang, S.S.; Nurden, P. The PDGF system and its antagonists in liver fibrosis. Cytokine Growth Factor Rev. 2016, 28, 53–61. [Google Scholar]
- Wang, S.; Mo, M.; Wang, J.; Sadia, S.; Shi, B.; Fu, X.; Yu, L.; Tredget, E.E.; Wu, Y. Platelet-derived growth factor receptor beta identifies mesenchymal stem cells with enhanced engraftment to tissue injury and pro-angiogenic property. Cell. Mol. Life Sci. 2018, 75, 547–561. [Google Scholar] [CrossRef]
- Kei, S.; Yasuhiko, K.; Miwako, T.; Tomonori, S.; Mieko, O.; Mayu, O.; Eiji, O.; Yukihito, H.; Shinji, T.; Wataru, Y. Stroma-directed imatinib therapy impairs the tumor-promoting effect of bone marrow-derived mesenchymal stem cells in an orthotopic transplantation model of colon cancer. Int. J. Cancer 2012, 132, 813–823. [Google Scholar]
- Popova, A.P.; Kelley, J.B.; Cui, T.X.; Richardson, M.N.; Linn, M.J.; Jing, L.; Qiang, C.; Goldsmith, A.M.; Pryhuber, G.S.; Hershenson, M.B. Reduced platelet-derived growth factor receptor expression is a primary feature of human bronchopulmonary dysplasia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 307, 231–239. [Google Scholar] [CrossRef]
- Petrova, V.V.; Vermeesch, J.R.; Van Duppen, V.J.; Vekemans, K.; Fagard, R.H.; Lijnen, P.J. TGF-β-induced cardiac myofibroblasts are nonproliferating functional cells carrying DNA damages. Exp. Cell Res. 2008, 314, 1480–1494. [Google Scholar] [CrossRef]
- Gao, P.; Zhou, Y.; Xian, L.; Li, C.; Xu, T.; Plunkett, B.; Cao, X. Functional effects of TGF-β1 on mesenchymal stem cell mobilization in cockroach allergen-induced asthma. J. Immunol. 2014, 192, 4560–4570. [Google Scholar] [CrossRef] [PubMed]
- Almeida, C.R.; Caires, H.R.; Vasconcelos, D.P.; Barbosa, M.A. NAP-2 secreted by human NK cells can stimulate mesenchymal stem/stromal cell recruitment. Stem Cell Rep. 2016, 6, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Choi, H.; Seon, M.; Cho, D.; Bang, S.I. LL-37 stimulates the functions of adipose-derived stromal/stem cells via early growth response 1 and the MAPK pathway. Stem Cell Res. Ther. 2016, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Oubari, F.; Amirizade, N.; Mohammadpour, H.; Nakhlestani, M.; Zarif, M.N. The important role of FLT3-L in ex vivo expansion of hematopoietic stem cells following co-culture with mesenchymal stem cells. Cell J. 2015, 17, 201–210. [Google Scholar] [PubMed]
- Enciso, N.; Ostronoff, L.L.K.; Mejías, G.; León, L.G.; Fermín, M.L.; Merino, E.; Fragio, C.; Avedillo, L.; Tejero, C. Stem cell factor supports migration in canine mesenchymal stem cells. Vet. Res. Commun. 2018, 42, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Tian, Y.; Yang, C.; Ma, X.; Wang, X.; Pei, J.; Qian, A. Mesenchymal stem cell migration during bone formation and bone diseases therapy. Int. J. Mol. Sci. 2018, 19, 2343. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Huang, X.; Zhou, Y.; Jin, R.; Li, Q. Mechanical stretching promotes skin tissue regeneration via enhancing mesenchymal stem cell homing and transdifferentiation. Stem Cells Transl. Med. 2016, 5, 960–969. [Google Scholar] [CrossRef]
- Zhang, B.; Luo, Q.; Chen, Z.; Sun, J.; Xu, B.; Ju, Y.; Song, G. Cyclic mechanical stretching promotes migration but inhibits invasion of rat bone marrow stromal cells. Stem Cell Res. 2015, 14, 155–164. [Google Scholar] [CrossRef] [Green Version]
- Fu, X.; Halim, A.; Tian, B.; Luo, Q.; Song, G. MT1-MMP downregulation via the PI3K/Akt signaling pathway is required for the mechanical stretching-inhibited invasion of bone marrow-derived mesenchymal stem cells. J. Cell. Physiol. 2019, 234, 14133–14144. [Google Scholar] [CrossRef]
- Zhou, S.B.; Wang, J.; Chiang, C.A.; Sheng, L.L.; Li, Q.F. Mechanical stretch upregulates SDF-1α in skin tissue and induces migration of circulating bone marrow-derived stem cells into the expanded skin. Stem Cells 2013, 31, 2703–2713. [Google Scholar] [CrossRef]
- Yuan, L.; Sakamoto, N.; Song, G.; Sato, M. Migration of human mesenchymal stem cells under low shear stress mediated by mitogen-activated protein kinase signaling. Stem Cells Dev. 2012, 21, 2520–2530. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Sakamoto, N.; Song, G.; Sato, M. Low-level shear stress induces human mesenchymal stem cell migration through the SDF-1/CXCR4 axis via MAPK signaling pathways. Stem Cells Dev. 2013, 22, 2384–2393. [Google Scholar] [CrossRef] [PubMed]
- Vincent, L.G.; Choi, Y.S.; Alonso-Latorre, B.; Álamo, J.C.; Engler, A.J. Mesenchymal stem cell durotaxis depends on substrate stiffness gradient strength. Biotech. J. 2013, 8, 472–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raab, M.; Swift, J.; Dingal, P.C.D.; Shah, P.; Shin, J.W.; Discher, D.E. Crawling from soft to stiff matrix polarizes the cytoskeleton and phosphoregulates myosin-II heavy chain. J. Cell Biol. 2012, 199, 669–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raab, M.; Discher, D.E. Matrix rigidity regulates microtubule network polarization in migration. Cytoskeleton 2017, 74, 114–124. [Google Scholar] [CrossRef]
- Skardal, A.; Mack, D.; Atala, A.; Soker, S. Substrate elasticity controls cell proliferation, surface marker expression and motile phenotype in amniotic fluid-derived stem cells. J. Mech. Behav. Biome. 2013, 17, 307–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saxena, N.; Mogha, P.; Dash, S.; Majumder, A.; Jadhav, S.; Sen, S. Matrix elasticity regulates mesenchymal stem cell chemotaxis. J. Cell Sci. 2018, 131, 211391. [Google Scholar] [CrossRef]
- Grimm, D.; Grosse, J.; Wehland, M.; Mann, V.; Reseland, J.E.; Sundaresan, A.; Corydon, T.J. Microgravity: The immune response and bone. Bone 2016, 87, 44–56. [Google Scholar] [CrossRef]
- Berg-Johansen, B.; Liebenberg, E.C.; Li, A.; Macias, B.R.; Hargens, A.R.; Lotz, J.C. Spaceflight-induced bone loss alters failure mode and reduces bending strength in murine spinal segments. J. Orthop. Res. 2015, 34, 48–57. [Google Scholar] [CrossRef]
- Rea, G.; Cristofaro, F.; Pani, G.; Pascucci, B.; Ghuge, S.A.; Corsetto, P.A.; Rizzo, A.M. Microgravity-driven remodeling of the proteome reveals insights into molecular mechanisms and signal networks involved in response to the space flight environment. J. Proteomics. 2016, 137, 3–18. [Google Scholar] [CrossRef]
- Chen, Z.; Luo, Q.; Lin, C.; Kuang, D.; Song, G. Simulated microgravity inhibits osteogenic differentiation of mesenchymal stem cells via depolymerizing F-actin to impede TAZ nuclear translocation. Sci. Rep. 2016, 6, 30322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, X.; Chen, Z.; Luo, Q.; Zhang, B.; Song, G. Simulated microgravity inhibits the migration of mesenchymal stem cells by remodeling actin cytoskeleton and increasing cell stiffness. Cytotechnology 2016, 68, 2235–2243. [Google Scholar] [CrossRef] [PubMed]
- Plett, P.A.; Abonour, R.; Frankovitz, S.M.; Orschell, C.M. Impact of modeled microgravity on migration, differentiation, and cell cycle control of primitive human hematopoietic progenitor cells. Exp. Hematol. 2004, 32, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Francois, S.; Mouiseddine, M.; Allenet, L.B.; Voswinkel, J.; Douay, L.; Benderitter, M.; Chapel, A. Human mesenchymal stem cells provide protection against radiation-induced liver injury by antioxidative process, vasculature protection, hepatocyte differentiation and trophic effects. Biomed. Res. Int. 2013, 2013, 151679. [Google Scholar] [CrossRef] [PubMed]
- Barbara, I.; Marina, M.; Susanna, T.; Cinzia, R.; Daniela, C.; Lorena, L.; Daniela, R.; Federica, V.; Ariela, B.; Jun, W. Insulin-like growth factor-1 sustains stem cell mediated renal repair. J. Am. Soc. Nephrol. JASN 2007, 18, 2921–2928. [Google Scholar]
- Wakabayashi, K.; Nagai, A.; Sheikh, A.M.; Shiota, Y.; Narantuya, D.; Watanabe, T.; Masuda, J.; Kobayashi, S.; Kim, S.U.; Yamaguchi, S. Transplantation of human mesenchymal stem cells promotes functional improvement and increased expression of neurotrophic factors in a rat focal cerebral ischemia model. J. Neurosci. Res. 2010, 88, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.W.; Chang, D.Y.; Lee, H.S.; Kim, G.H.; Park, J.S.; Ryu, B.Y.; Joe, E.H.; Lee, Y.D.; Kim, S.S.; Suh, K.H. Immune following suppression mesenchymal stem cell transplantation in the ischemic brain is mediated by TGF-β. Neurobiol. Dis. 2013, 58, 249–257. [Google Scholar] [CrossRef]
- Yang, W.K.; Heo, S.C.; Jeong, G.O.; Yoon, J.W.; Mo, W.M.; Mi, J.L.; Jang, I.H.; Sang, M.K.; Lee, J.S.; Kim, J.H. Tumor necrosis factor-α-activated mesenchymal stem cells promote endothelial progenitor cell homing and angiogenesis. Biochim. Biophys. Acta 2013, 1832, 2136–2144. [Google Scholar]
- Kim, Y.J.; Seo, D.H.; Lee, S.H.; Lee, S.H.; An, G.H.; Ahn, H.J.; Kwon, D.; Seo, K.W.; Kang, K.S. Conditioned media from human umbilical cord blood-derived mesenchymal stem cells stimulate rejuvenation function in human skin. Biochem. Biophys. Rep. 2018, 16, 96–102. [Google Scholar] [CrossRef]
- Li, M.; Luan, F.; Zhao, Y.; Hao, H.; Liu, J.; Dong, L.; Fu, X.; Han, W. Mesenchymal stem cell-conditioned medium accelerates wound healing with fewer scars. Int. Wound J. 2017, 14, 64–73. [Google Scholar] [CrossRef]
- Zheng, B.; Wang, C.; He, L.; Xu, X.; Qu, J.; Hu, J.; Zhang, H. Neural differentiation of mesenchymal stem cells influences chemotactic responses to HGF. J. Cell. Physiol. 2013, 228, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.B.; Chiang, C.A.; Liu, K.; Li, Q.F. Intravenous transplantation of bone marrow mesenchymal stem cells could effectively promote vascularization and skin regeneration in mechanically stretched skin. Br. J. Dermatol. 2014, 172, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Chen, M.; Su, W.; Tao, X.; Sun, M.; Zou, X.; Ying, R.; Wei, W.; Wang, B. The differentiation of mesenchymal stem cells to vascular cells regulated by the HMGB1/RAGE axis: Its application in cell therapy for transplant arteriosclerosis. Stem Cell Res. Ther. 2018, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Cucarián, J.D.; Berrío, J.P.; Rodrigues, C.; Zancan, M.; Wink, M.R.; de Oliveira, A. Physical exercise and human adipose-derived mesenchymal stem cells ameliorate motor disturbances in a male rat model of Parkinson’s disease. J. Neurosci. Res. 2019, 97, 1095–1109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Luo, Q.; Deng, B.; Morita, Y.; Ju, Y.; Song, G. Construction of tendon replacement tissue based on collagen sponge and mesenchymal stem cells by coupled mechano-chemical induction and evaluation of its tendon repair abilities. Acta Biomater. 2018, 74, 247–259. [Google Scholar] [CrossRef] [PubMed]
| Chemical Factor | Concentration | Cell Migration | Outcomes | References |
|---|---|---|---|---|
| Stromal derived factor-1(SDF-1) | 50 ng/mL, 100 ng/mL | ↑ | SDF-1 increased BMSC recruitment to injured liver and promoted the repair of injured liver. | [16] |
| SDF-1 | 100 ng/mL | ↑ | SDF-1 increased BMSCs with CXCR4 expression and promoted the repair of traumatic brain injury. | [17] |
| SDF-1 | 10 ng/mL | ↑ | SDF-1 increased stem cell recruitment, and the pretreatment of stem cells (Wharton’s jelly-derived mesenchymal stem cells (WJ-MSCs), embryonic stem cells (ESCs)) enhanced skeletal muscle regeneration. | [18] |
| Osteopontin (OPN) | 1 μg/mL | ↑ | Increased integrin β1 expression in BMSCs and promoted BMSC migration through the ligation to integrin β1. | [19] |
| OPN | 10 μg/mL, 20 μg/mL | ↑ | Increased mesenchymal stem cell (MSC) migration in a dose-dependent manner. | [20] |
| OPN | 1 μg/mL | ↑ | OPN reduced the number of organized actin cytoskeletons through the FAK and ERK pathways to increase BMSC migration. | [21] |
| OPN | 1 μg/mL | ↑ | Reduced the number of organized actin cytoskeletons through the FAK and ERK pathways to increase BMSC migration. | [22] |
| OPN | 1 μg/mL | ↑ | Cytoskeletal control of nuclear morphology and stiffness through the SUN1 proteins plays an important role in OPN-promoted BMSC migration. | [23] |
| OPN | 1 μg/mL | ↑ | Chromatin organization was altered by the application of OPN via the ERK1/2 signaling pathway, which also contributed to BMSC migration. | [24] |
| Basic fibroblast growth factor (bFGF) | 200 ng/mL | ↑ | Augmented the engraftment and differentiation capacity of transplanted BMSCs, recovering cardiac function. | [25] |
| bFGF | 1 ng/mL up to 400 ng/mL | ↓↑ | Low concentrations led to an attraction of BMSCs, whereas higher concentrations resulted in repulsion. | [26] |
| Vascular endothelial growth factor (VEGF)-A | 10 ng/mL | ↑ | Increased BMSC migration and proliferation. | [27] |
| Hepatocyte growth factor (HGF) | 20 ng/mL | ↑ | Increased BMSC migration via PI3K pathways. | [28] |
| Insulin-like growth factor (IGF)-1 | 10 ng/mL | ↑ | Increased BMSC migratory responses via CXCR4 chemokine receptor signaling, which is PI3/Akt-dependent. | [29] |
| IGF-1 | 20 ng/mL | ↑ | Preconditioning of BMSCs with IGF-1 before infusion improved cell migration capacity and restored normal renal function after acute kidney injury. | [30] |
| Platelet-derived growth factor (PDGF) | 50 ng/mL | ↑ | Increased BMSC migration significantly. | [31] |
| PDGF-B | 40 ng/mL | ↑ | Increased recruitment/migration and differentiation of BMSCs. | [32] |
| Transforming growth factor (TGF)-β 1 | 100 pM | ↑ | Promoted the homing of BMSCs in myocardial ischemia/reperfusion injury and improved myocardial function. | [9] |
| TGF-β1 | 5 ng/mL | ↑ | Improved BMSC recruitment and wound closure in a syngeneic murine wound model. | [33] |
| TGF-β | 1 ng/mL~100 ng/mL | ↑ | Activated noncanonical signaling molecules, such as Akt, ERK1/2, FAK, and p38, via TGF-β type I receptor to increase stem cell (BMSCs, BM-MSC-like ST2 cells) migration. | [34] |
| Paracrine Factors | Animal Models | Outcomes | References |
|---|---|---|---|
| TGF-β, FGF-2, angiopoietin-2, VEGF-1 | Rat myocardial infarction model | Triggered angiogenic and migratory effects at the site of the infarct to promote myocardial healing and improve the cardiac function. | [1] |
| NGF, HGF, IL-10, IL1-RA | NOD/SCID mouse model | Contributed to the prevention of apoptosis, increasing cell proliferation in the damaged liver. | [86] |
| TGF-β1, VEGF | Mouse burn injury model | Assisted in burn wound healing. | [2] |
| IGF-1 | Mouse acute kidney injury model | Exerted beneficial effects on tubular cell repair in acute kidney injury. | [87] |
| Angiogenin, IL-8, MCP-1, and VEGF | Mouse hind limb ischemia model | Represented efficient biomarkers for predicting vascular regenerative efficacy of MSCs. | [4] |
| IGF-1, VEGF, EGF, and bFGF | Rat middle cerebral artery occlusion ischemia model | Induced functional improvement, reduced infarct volume, and showed neuroprotection in ischemic rats. | [88] |
| TGF-β | Rat stroke model | Suppressed immune propagation in the ischemic rat brain. | [89] |
| SDF-1, VEGF, HGF, and IL-6 | Rat skin wound model | Enhanced the activity of dermal fibroblasts and keratinocytes to promote re-epithelialization and angiogenesis and, consequently, promoted wound healing. | [5] |
| Conditioned Medium | Animal Models | Outcomes | References |
|---|---|---|---|
| IGF-1, VEGF, TGF-β1 and HGF | Rat periodontal defect model | Contributed to many processes of complicated periodontal tissue regeneration. | [3] |
| IL-6, IL-8 | Rat hind limb ischemia model | Stimulated angiogenesis and tissue repair through an increase in homing of human cord blood-derived endothelial progenitor. | [90] |
| EGF, bFGF, PDGF, HGF, collagen type 1, and GDF-11 | In vivo human test | Stimulated skin rejuvenation by increasing growth and ECM production. | [91] |
| Collagen types III and I and a high MMPs/TIMPs ratio | Mouse skin excisional wound model | Accelerated healing, with fewer scars compared with control groups. | [92] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, X.; Liu, G.; Halim, A.; Ju, Y.; Luo, Q.; Song, G. Mesenchymal Stem Cell Migration and Tissue Repair. Cells 2019, 8, 784. https://doi.org/10.3390/cells8080784
Fu X, Liu G, Halim A, Ju Y, Luo Q, Song G. Mesenchymal Stem Cell Migration and Tissue Repair. Cells. 2019; 8(8):784. https://doi.org/10.3390/cells8080784
Chicago/Turabian StyleFu, Xiaorong, Ge Liu, Alexander Halim, Yang Ju, Qing Luo, and Guanbin Song. 2019. "Mesenchymal Stem Cell Migration and Tissue Repair" Cells 8, no. 8: 784. https://doi.org/10.3390/cells8080784
APA StyleFu, X., Liu, G., Halim, A., Ju, Y., Luo, Q., & Song, G. (2019). Mesenchymal Stem Cell Migration and Tissue Repair. Cells, 8(8), 784. https://doi.org/10.3390/cells8080784





