Genetic Association between Swine Leukocyte antigen Class II Haplotypes and Reproduction Traits in Microminipigs
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. SLA Class II Typing
2.3. Measurement of Reproductive Performances
2.4. Statistical Analyses
3. Results
3.1. Reproductive Performance in MMPs
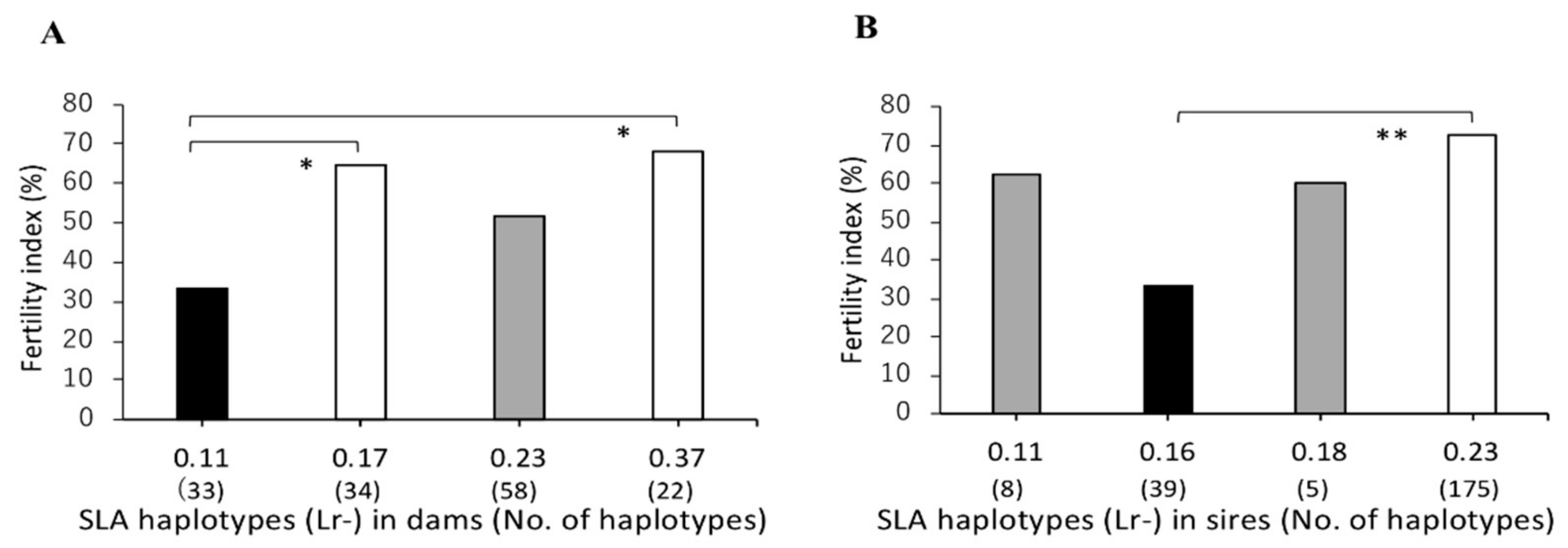
3.2. Association between SLA Class II Haplotypes and Fertility Index
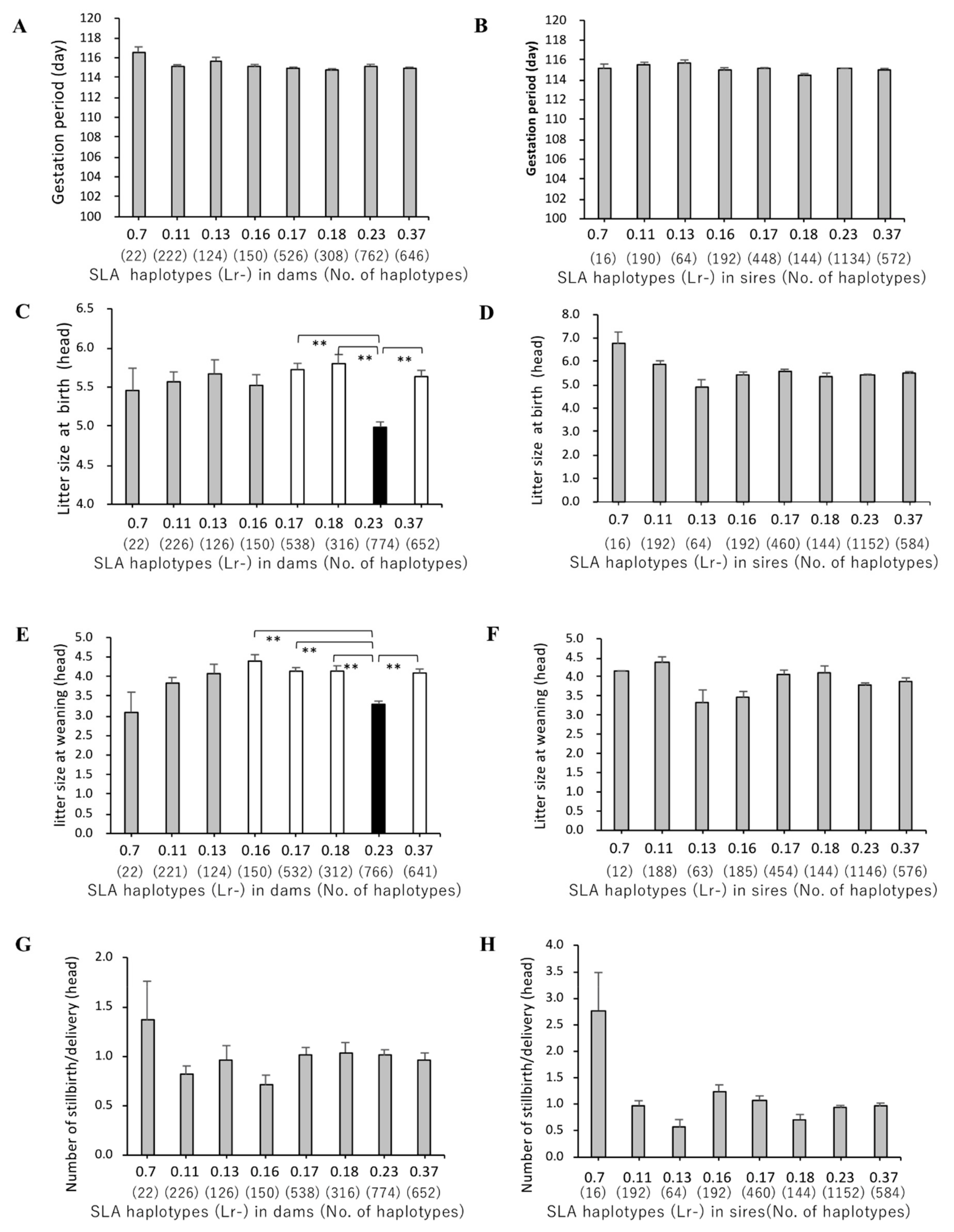
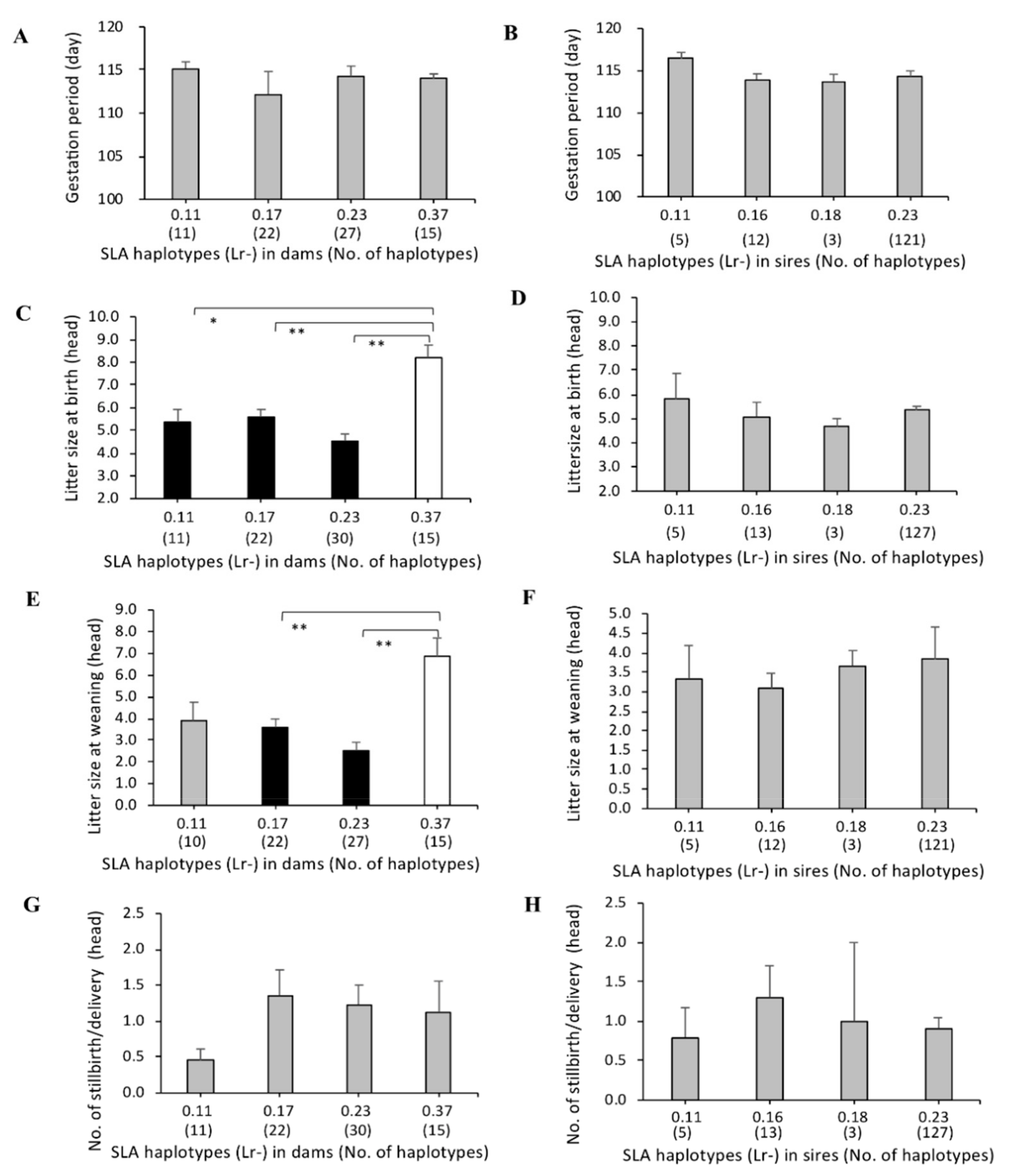
3.3. Association of SLA Class II Haplotypes and Gestation Periods
3.4. Association between SLA Class II Haplotypes and Litter Sizes at Birth and Weaning
3.5. Association of SLA Class II Haplotypes and Number of Stillbirths per Delivery
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kaneko, N.; Itoh, K.; Sugiyama, A.; Izumi, Y. Microminipig, a non-rodent experimental animal optimized for life science research: Preface. J. Pharmacl. Sci. 2011, 115, 112–114. [Google Scholar] [CrossRef]
- Takasu, M.; Tsuji, E.; Imaeda, N.; Kaneko, N.; Matsubara, T.; Maeda, M.; Ito, Y.; Shibata, S.; Ando, A.; Nishii, N.; et al. Body and, major organ sizes of young mature Microminipigs determined by computed tomography. Lab. Anim. 2015, 49, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Takasu, M.; Imaeda, N.; Nishii, N.; Takashima, S.; Nishimura, T.; Nishimura, T.; Shiina, T.; Ando, A.; Kitagawa, H. Genetic association of swine leukocyte antigen class II haplotypes and body weight in Microminipigs. Asian Aust. J. Animal Sci. 2018, 31, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Ando, A.; Imaeda, N.; Ohshima, S.; Miyamoto, A.; Kaneko, N.; Takasu, M.; Shiina, T.; Kulski, J.K.; Inoko, H.; Kitagawa, H. Characterization of swine leucocyte antigen alleles and haplotypes on a novel miniature pig line, Microminipig. Anim. Gen. 2015, 45, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Renard, C.; Hart, E.; Sehra, H.; Beasley, H.; Coggill, P.; Howe, K.; Harrow, J.; Gilbert, J.; Sims, S.; Rogers, J.; et al. The genomic sequence and analysis of the swine major histocompatibility complex. Genomics 2006, 88, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.S.; Lunney, J.K.; Franzo-Romain, M.H.; Martens, G.W.; Lee, Y.J.; Lee, J.H.; Wysocki, M.; Rowland, R.R.; Smith, D.M. Molecular characterization of swine leucocyte antigen class I genes in outbred pig populations. Anim. Genet. 2009, 40, 468–478. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.S.; Lunney, J.K.; Franzo-Romain, M.H.; Martens, G.W.; Rowland, R.R.; Smith, D.M. Molecular characterization of swine leucocyte antigen class II genes in outbred pig populations. Anim. Genet. 2010, 41, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Ando, A.; Shigenari, A.; Ota, M.; Sada, M.; Kawata, H.; Azuma, F.; Kojima-Shibata, C.; Nakajoh, M.; Suzuki, K.; Uenishi, H.; et al. SLA-DRB1 and -DQB1 genotyping by the PCR-SSOP-Luminex method. Tissue Antigens 2011, 78, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Kita, Y.F.; Ando, A.; Tanaka, K.; Suzuki, S.; Ozaki, Y.; Uenishi, H.; Inoko, H.; Kulski, J.K.; Shiina, T. Application of high-resolution, massively parallel pyrosequencing for estimation of haplotypes and gene expression levels of swine leukocyte antigen (SLA) class I genes. Immunogenetics 2012, 64, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Le, M.; Choi, H.; Choi, M.K.; Cho, H.; Kim, J.H.; Seo, H.G.; Cha, S.Y.; Seo, K.; Dadi, H.; Park, C. Development of a simultaneous high resolution typing method for three SLA class II genes, SLA-DQA, SLA-DQB1, and SLA-DRB1 and the analysis of SLA class II haplotypes. Gene 2015, 564, 228–232. [Google Scholar] [CrossRef]
- Sørensen, M.R.; Ilsøe, M.; Strube, M.L.; Bishop, R.; Erbs, G.; Hartmann, S.B.; Jungersen, G. Sequence-Based Genotyping of Expressed Swine Leukocyte Antigen Class I Alleles by Next-Generation Sequencing Reveal Novel Swine Leukocyte Antigen Class I Haplotypes and Alleles in Belgian, Danish, and Kenyan Fattening Pigs and Göttingen Minipigs. Front. Immunol. 2017, 8, 701. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Moroldo, M.; Perdomo-Sabogal, A.; Mach, N.; Marthey, S.; Lecardonnel, J.; Wahlberg, P.; Chong, A.Y.; Estellé, J.; Ho, S.Y.W.; et al. Inferring the evolution of the major histocompatibility complex of wild pigs and peccaries using hybridisation DNA capture-based sequencing. Immunogenetics 2018, 70, 401–417. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.S.; Lunney, J.K.; Ando, A.; Rogel-Gaillard, C.; Lee, J.H.; Schook, L.B.; Smith, D.M. Nomenclature for factors of the SLA system, update 2008. Tissue Antigens 2009, 73, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, J.M.; Jamieson, S.E.; Burgner, D. HLA and infectious diseases. Clin. Microbiol. Rev. 2009, 22, 370–385. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, S.E.; Esteban, E.N.; Lützelschwab, C.M.; Juliarena, M.A. Chapter 6, Major Histocompatibility Complex-associated resistance to infectious diseases: The case of Bovine leukemia virus infection. In Trends and Advances in Veterinary Genetics; Abubakar, M., Ed.; IntechOpen Limited: London, UK, 2017; pp. 101–126. [Google Scholar] [CrossRef]
- Sommer, S. The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front. Zool. 2005, 2, 16. [Google Scholar] [CrossRef] [PubMed]
- Renard, C.; Vaiman, M. Possible relationships between SLA and porcine reproduction. Reprod. Nutr. Dev. 1989, 29, 569–576. [Google Scholar] [CrossRef][Green Version]
- Gautschi, C.; Gaillard, C. Influence of major histocompatibility complex on reproduction and production traits in swine. Anim. Genet. 1989, 21, 161–170. [Google Scholar] [CrossRef]
- Conley, A.J.; Jung, Y.C.; Schwartz, N.K.; Warner, C.M.; Rothschild, M.F.; Ford, S.P. Influence of SLA haplotype on ovulation rate and litter size in miniature pigs. J. Reprod. Fertil. 1988, 82, 595–601. [Google Scholar] [CrossRef]
- Imaeda, N.; Ando, A.; Takasu, M.; Matsubara, T.; Nishii, N.; Takashima, S.; Shigenari, A.; Shiina, T.; Kitagawa, H. Influences of swine leukocyte antigen haplotypes on serum antigen titers against swine erysipelas vaccine and traits of reproductive ability and meat production in a SLA-defined Duroc pigs. J. Vet. Med. Sci. 2018, 80, 1662–1668. [Google Scholar] [CrossRef]
- Vaiman, M.; Chardon, P.; Rothschild, M.F. Porcine major histocompatibility complex. Rev. Sci. Tech. Off. Int. Epiz. 1998, 17, 95–107. [Google Scholar] [CrossRef]
- Ando, A.; Shigenari, A.; Kojima-Shibata, C.; Nakajoh, M.; Suzuki, K.; Kitagawa, H.; Shiina, T.; Inoko, H.; Uenishi, H. Association of swine leukocyte antigen class II haplotypes and immune-related traits in a swine line selected for resistance to mycoplasmal pneumonia. Comp. Immunol. Microbiol. Infect. Dis. 2016, 48, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, J.; Wang, L.; Li, Z.; Pang, P.; Li, F. SLA-11 mutations are associated with litter size traits in Large White and Chinese DIV pigs. Anim. Biotech. 2018. [Google Scholar] [CrossRef] [PubMed]
- Glusman, G.; Cox, H.C.; Roach, J.C. Whole-genome haplotyping approaches and genomic medicine. Genome Med. 2014, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Molano, E.; Tsiokos, D.; Chatziplis, D.; Jorjani, H.; Degano, L.; Diaz, C.; Rossoni, A.; Schwarzenbacher, H.; Seefried, F.; Varona, L.; et al. A practical approach to detect ancestral haplotypes in livestock populations. BMC Genet. 2016, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Tu, J.; Lu, Z. Recent Advances in Experimental Whole Genome Haplotyping Methods. Int. J. Mol. Sci. 2017, 18, 1944. [Google Scholar] [CrossRef] [PubMed]
- Murphy, N.M.; Burton, M.; Powell, D.R.; Rossello, F.J.; Cooper, D.; Chopra, A.; Hsieh, M.J.; Sayer, D.C.; Gordon, L.; Pertile, M.D.; et al. Haplotyping the human leukocyte antigen system from single chromosomes. Sci. Rep. 2016, 6, 30381. [Google Scholar] [CrossRef] [PubMed]
- Min, F.; Pan, J.; Wang, X.; Chen, R.; Wang, F.; Luo, S.; Ye, J. Biological characteristics of captive Chinese Wuzhishan minipigs (Sus scrofa). Int. Sch. Res. Notices 2014, 761257. [Google Scholar] [CrossRef]
- Agenor, A.; Bhattacharya, S. Infertility and miscarriage: Common pathways in manifestation and management. Womens Health 2015, 11, 527–541. [Google Scholar] [CrossRef]
- Rapacz-Leonard, A.; Dąbrowska, M.; Janowski, T. Major histocompatibility complex I mediates immunological tolerance of the trophoblast during pregnancy and may mediate rejection during parturition. Mediat. Inflamm. 2014, 2014, 579279. [Google Scholar] [CrossRef]
- Suzuki, K.; Somei, H. Present condition and opinions of artificial insemination in pig farming. Bull. Chiba Prefect. Livestock Exptl. Station 1982, 6, 39–43. (In Japanese) [Google Scholar]
- Rothkötter, H.J.; Sowa, E.; Pabst, R. The pig as a model of developmental immunology. Hum. Exp. Toxicol. 2002, 21, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Tsumagari, S. Textbook of Theriogenology, 2nd ed.; Mori, J., Kanagawa, H., Hamana, K., Eds.; Buneido Co., Ltd.: Tokyo, Japan, 2002; pp. 159–160. ISBN 4-8300-3184-0. (In Japanese) [Google Scholar]
- Koketsu, Y. Reproductive productivity measurements in Japanese swine breeding herds. J. Vet. Med. Sci. 2002, 64, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Yamane, I.; Ishizaki, S.; Yamazaki, H. Parameters Contributing to Improved Reproductive Performance at Farrow-to-Finish Swine Farms in Japan. J. Jpn. Vet. Med. Assoc. 2014, 67, 177–182. (In Japanese) [Google Scholar] [CrossRef][Green Version]
- Casellas, J.; Ibáñez-Escriche, N.; Varona, L.; Rosas, J.P.; Noguera, J.I. Inbreeding depression load for litter size in Entrepelado and Retinto Iberian pig varieties. J. Anim. Sci. 2019, skz084. [Google Scholar] [CrossRef]
- Schuleri, K.H.; Boyle, A.J.; Centola, M.; Amado, L.C.; Evers, R.; Zimmet, J.M.; Evers, K.S.; Ostbye, K.M.; Scorpio, D.G.; Hare, J.M.; et al. The adult Göttingen Minipig as a model for chronic heart failure after myocardial infarction: Focus on cardiovascular imaging and regenerative therapies. Comp. Med. 2008, 58, 568–579. [Google Scholar] [PubMed]
- Saitoh, T. Utilization of a swine other than food. All About Swine 2009, 35, 14–20. (In Japanese) [Google Scholar]
- Lawlor, P.G.; Lynch, P.B. A review of factors influencing litter size in Irish sows. Ir. Vet. J. 2007, 60, 359–366. [Google Scholar] [CrossRef]
- Rutherford, K.M.D.; Baxter, E.M.; D’Eath, R.B.; Turner, S.P.; Arnott, G.; Roehe, R.; Ask, B.; Sandoe, P.; Moustsen, V.A.; Thorup, F.; et al. The welfare implications of large litter size in the domestic pig I: Biological factors. Anim. Welfare 2013, 22, 199–218. [Google Scholar] [CrossRef]
- Chen, P.; Baas, T.J.; Mabry, J.W.; Koehler, K.J.; Dekkers, J.C. Genetic parameters and trends for litter traits in U.S.; Yorkshire, Duroc, Hampshire, and Landrace pigs. J. Anim. Sci. 2003, 81, 46–53. [Google Scholar] [CrossRef]
- Kwon, S.G.; Hwang, J.H.; Park, D.H.; Kim, T.W.; Kang, D.G.; Kang, K.H.; Kim, I.S.; Park, H.C.; Na, C.S.; Ha, J.; et al. Identification of differentially expressed genes associated with litter size in Berkshire pig placenta. PLoS ONE 2016, 11, e0153311. [Google Scholar] [CrossRef]
- Athanassakis-Vassiliadis, I.; Thanos, D.; Papamatheakis, J. Induction of class II major histocompatibility complex antigens in murine placenta by 5-azacytidine and interferon-γ involves different cell populations. Eur. J. Immunol. 1989, 19, 2341–2348. [Google Scholar] [CrossRef]
- Kieckbusch, J.; Balmas, E.; Hawkes, D.A.; Colucci, F. Disrupted PI3K p110δ signaling dysregulates maternal immune cells and increases fetal mortality in mice. Cell Rep. 2015, 13, 2817–2828. [Google Scholar] [CrossRef] [PubMed]
- Stefferl, A.; Schubart, A.; Storch, M.; Amini, A.; Mather, I.; Lassmann, H.; Linington, C. Butyrophilin, a milk protein, modulates the encephalitogenic T cell response to myelin oligodendrocyte glycoprotein in experimental autoimmune encephalomyelitis. J. Immunol. 2000, 165, 2859–2865. [Google Scholar] [CrossRef] [PubMed]
- Posfai, E.; Kunzmann, R.; Brochard, V.; Salvaing, J.; Cabuy, E.; Roloff, T.C.; Liu, Z.; Tardat, M.; van Lohuizen, M.; Vidal, M.; et al. Polycomb function during oogenesis is required for mouse embryonic development. Genes Dev. 2012, 26, 920–932. [Google Scholar] [CrossRef] [PubMed]
- Camlin, N.J.; McLaughlin, E.A.; Holt, J.E. Motoring through: The role of kinesin superfamily proteins in female meiosis. Hum. Reprod. Update 2017, 23, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Schöler, H.R. Role of Oct4 in the early embryo development. Cell Regen. 2014, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ding, X.; Tan, Z.; Xing, K.; Yang, T.; Wang, Y.; Sun, D.; Wang, C. Genome-wide association study for reproductive traits in a Large White pig population. Anim. Genet. 2018, 49, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Demeure, O.; Sanchez, M.P.; Riquet, J.; Iannuccelli, N.; Demars, J.; Fève, K.; Kernaleguen, L.; Gogué, J.; Billon, Y.; Caritez, J.C.; et al. Exclusion of the swine leukocyte antigens as candidate region and reduction of the position interval for the Sus scrofa chromosome 7 QTL affecting growth and fatness. J. Anim. Sci. 2005, 83, 1979–1987. [Google Scholar] [CrossRef][Green Version]
- Wei, W.H.; Skinner, T.M.; Anderson, J.A.; Southwood, O.I.; Plastow, G.; Archibald, A.L.; Haley, C.S. Mapping QTL in the porcine MHC region affecting fatness and growth traits in a Meishan/Large White composite population. Anim. Genet. 2011, 42, 83–85. [Google Scholar] [CrossRef]
- Lunney, J.K.; Ho, C.S.; Wysocki, M.; Smith, D.M. Molecular genetics of the swine major histocompatibility complex, the SLA complex. Dev. Comp. Immunol. 2009, 33, 362–374. [Google Scholar] [CrossRef]

| SLA Class II | Allele Specificity by Low Resolution Typing | * Expected Allele Specificity by High Resolution Typing | Number of Haplotypes (Frequency (%)) | |||||
|---|---|---|---|---|---|---|---|---|
| haplotype | DRB1 | DQB1 | DRB1 | DQB1 | Dams | Sires | ||
| Lr-0.7 | 06:XX | 06:XX | 06:01 | 06:01 | 2 | (0.8) | 1 | (0.9) |
| Lr-0.11 | 09:XX | 04:XX | 09:01 | 04:01:02/04:02 | 19 | (7.4) | 10 | (8.6) |
| Lr-0.13 | 04:XX | 03:XX | 04:03 | 03:03 | 13 | (5.0) | 2 | (1.7) |
| Lr-0.16 | 11:XX | 06:XX | 11:03 | 06:01 | 16 | (6.2) | 7 | (6.0) |
| Lr-0.17 | 08:XX | 05:XX | 08:01 | 05:01/05:02 | 44 | (17.1) | 18 | (15.5) |
| Lr-0.18 | 14:XX | 04:XX | 14:01 | 04:01:02/04:02 | 24 | (9.3) | 8 | (6.9) |
| Lr-0.23 | 10:XX | 06:XX | 10:01 | 06:01 | 79 | (30.6) | 42 | (36.2) |
| Lr-0.37 | 07:XX | 05:XX | 07:01 | 05:01/05:02 | 61 | (23.6) | 28 | (24.1) |
| Trait | Mean * | SE * | Number of Haplotypes ** |
|---|---|---|---|
| Gestation period (day) | 115.1 | 0.06 | 2760 |
| Litter size at birth (No. of piglets) | 5.48 | 0.04 | 2804 |
| Litter size at weaning (No. of piglets) | 3.88 | 0.04 | 2804 |
| No. of stillbirth/delivery (No. of piglets) | 0.97 | 0.03 | 2768 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ando, A.; Imaeda, N.; Matsubara, T.; Takasu, M.; Miyamoto, A.; Oshima, S.; Nishii, N.; Kametani, Y.; Shiina, T.; Kulski, J.K.; et al. Genetic Association between Swine Leukocyte antigen Class II Haplotypes and Reproduction Traits in Microminipigs. Cells 2019, 8, 783. https://doi.org/10.3390/cells8080783
Ando A, Imaeda N, Matsubara T, Takasu M, Miyamoto A, Oshima S, Nishii N, Kametani Y, Shiina T, Kulski JK, et al. Genetic Association between Swine Leukocyte antigen Class II Haplotypes and Reproduction Traits in Microminipigs. Cells. 2019; 8(8):783. https://doi.org/10.3390/cells8080783
Chicago/Turabian StyleAndo, Asako, Noriaki Imaeda, Tatsuya Matsubara, Masaki Takasu, Asuka Miyamoto, Shino Oshima, Naohito Nishii, Yoshie Kametani, Takashi Shiina, Jerzy K. Kulski, and et al. 2019. "Genetic Association between Swine Leukocyte antigen Class II Haplotypes and Reproduction Traits in Microminipigs" Cells 8, no. 8: 783. https://doi.org/10.3390/cells8080783
APA StyleAndo, A., Imaeda, N., Matsubara, T., Takasu, M., Miyamoto, A., Oshima, S., Nishii, N., Kametani, Y., Shiina, T., Kulski, J. K., & Kitagawa, H. (2019). Genetic Association between Swine Leukocyte antigen Class II Haplotypes and Reproduction Traits in Microminipigs. Cells, 8(8), 783. https://doi.org/10.3390/cells8080783







