Endocytic Adaptor Proteins in Health and Disease: Lessons from Model Organisms and Human Mutations
Abstract
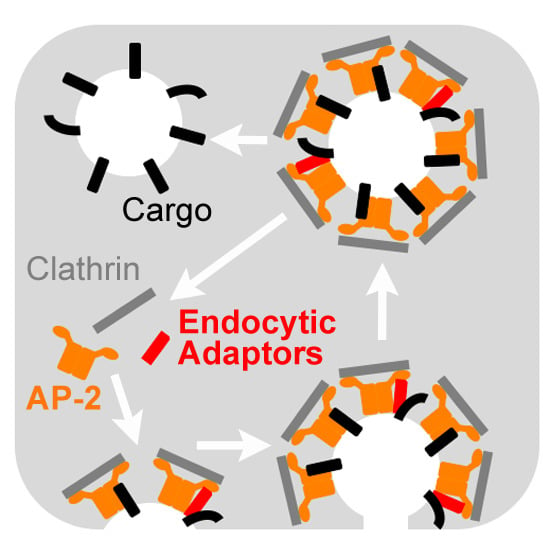
1. Clathrin-Mediated Endocytosis
2. Endocytic Adaptors
2.1. AP-2
2.1.1. Lessons from AP-2-Deficient Mouse Models
2.1.2. Lessons from Human Mutations in AP-2 Subunits
2.1.3. Additional Links of AP-2 to Human Disease
2.2. Stonin1 and Stonin2
2.2.1. Stonin2 as a Sorter for Synaptotagmin1
2.2.2. Lessons from Stonin-Deficient Mouse Models
2.2.3. Links of Stonin Proteins to Human Disease
2.3. FCHO1, FCHO2 and SGIP1
2.3.1. Molecular Functions of Muniscins
2.3.2. Insights from Muniscin-Deficient Model Organisms
2.3.3. Links of Muniscin Proteins to Human Disease
2.4. AP180 and CALM
2.4.1. Studies of AP180 and CALM in Non-Mammalian Model Organisms
2.4.2. Lessons from AP180- and CALM-Deficient Mouse Models
2.4.3. Links of AP180 and CALM to Human Disease
2.4.4. Non-Canonical Roles of CALM
2.5. HIP1 and HIP1R
2.5.1. Lessons from HIP1- and HIP1R-Deficient Animal Models
2.5.2. Links of HIP1 to Human Disease
2.6. Ubiquitin Interacting Motif (UIM)-Containing Adaptors: Eps15 and Eps15R
2.6.1. Studies of Eps15 in Non-Mammalian Organisms
2.6.2. Lessons from Eps15- and Eps15R-Deficient Mouse Models
2.6.3. Involvement of Eps15 and Eps15R in Human Disease
2.7. Epsin N-Terminal Homology (ENTH)- and UIM-Containing Adaptors: Epsins
2.7.1. Lessons from Epsin-Deficient Animal Models
2.7.2. Involvement of Epsins in Human Disease
2.8. PTB-Domain Containing Adaptors: ARH, Dab2, Numb and Numbl
2.8.1. The Role of ARH in Human Disease
2.8.2. Phenotypes of ARH-Deficient and ARH/Dab2 DKO Mice
2.8.3. Functions of Dab2
2.8.4. Insights from Dab2-Deficient Mouse Models
2.8.5. Links of Dab2 to Human Disease
2.8.6. Insights into Numb Function from D. Melanogaster
2.8.7. The Role of Numb im Mammals
2.8.8. Insights from Numb-Deficient Mouse Models
2.8.9. Involvement of Numb in Human Disease
2.9. Arrestins—Adaptors for G Protein-Coupled Receptors
2.9.1. Insights from Arrestin-Deficient Mouse Models
2.9.2. Links of Arrestins to Human Disease
2.10. Hrb and Hrbl
2.10.1. Hrb as a Specific Sorter for VAMP7
2.10.2. Phenotypes of Hrb- and Hrbl-Deficient Mouse Models
2.11. TTP/SH3BP4 and MACC1
2.11.1. Links of TTP/SH3BP4 to Human Disease
2.11.2. MACC1, a Putative Endocytic Adaptor
3. One Cargo—Many Adaptors? Redundancy in Cargo Recognition
4. Endocytic Adaptors as Reticular Adhesion Components
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Aβ | Amyloid β |
| AD | Alzheimer’s disease |
| ALL | Acute lymphoblastic leukaemia |
| AML | Acute myeloid leukaemia |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| AMPAR | AMPA receptor |
| ANTH | AP180 N-terminal homology domain |
| APA | AP-2 activation domain |
| AP-2 | Assembly protein 2 |
| ArfGAP | Arf GTPase activating protein |
| ARH | Autosomal recessive hypercholesterolemia |
| ASD | Autism spectrum disorder |
| BAR | Curved domain named after Bin, Amphiphysin, Rvs |
| BDNF | Brain-derived neurotrophic factor |
| CC | Coiled coil domain |
| CHC | Clathrin heavy chain |
| CMML | Chronic myelomonocytic leukaemia |
| CLC | Clathrin light chain |
| CLL | Chronic lymphocytic leukaemia |
| CT | Carboxy-terminus |
| DD | Death domain |
| DKO | Double knock-out |
| ECG | Electrocardiagram |
| EEG | Electroencephalogram |
| EGF | Epidermal growth factor |
| EGFR | Epidermal growth factor receptor |
| EH | Eps15 homology domain |
| ENTH | Epsin N-terminal homology domain |
| F-BAR | FCH-BAR domain |
| FHH | Familial hypocalciuric hypercalcemia |
| GAP | GTPase activating protein |
| GPCR | G protein-coupled receptor |
| GTP | Guanosine triphosphate |
| HCV | Hepatitis C Virus |
| HD | Huntington’s disease |
| HIV | Human Immunodeficiency Virus |
| IDRs | Intrinsically disordered protein regions |
| IMPC | International mouse phenotyping consortium |
| iPSCs | Induced pluripotent stem cells |
| LDL | Low density lipoprotein |
| LDLR | Low density lipoprotein receptor |
| LTD | Long term depression |
| MP | Membrane lipid binding domain |
| µHD | µ-Homology domain |
| NK cells | Natural killer cells |
| NMDA | N-methyl-d-aspartate |
| NT | Amino-terminus |
| KO | Knock-out |
| LTD | Long term depression |
| SHD | Stonin homology domain |
| PI | Phosphoinositide |
| PI(4,5)P2 | Phosphatidylinositol(4,5)bisphosphate |
| PRR | Prolin-rich region |
| PS | Phosphatidylserine |
| PTB | Phosphotyrosine binding domain |
| PTH | Parathyroid hormone |
| SHFM | Split-hand/split-foot malformation |
| SH3 | Src homology 3 domain |
| SNARE | Soluble N-ethylmaleimide sensitive factor attachment protein receptor |
| SNP | Single nucleotide polymorphism |
| Talin-like | Actin binding domain |
| TfR | Transferrin receptor |
| TGFβ-R | Transforming growth factor b receptor |
| TrkB | Tropomyosin receptor kinase B |
| Ub | Ubiquitin |
| UIM | Ubiquitin interacting motifs |
| UPA | Domain named after Unc5, PIDD and Ankyrins |
| VAMP | Vesicle-associated membrane protein |
| VEGFR | Vascular endothelial growth factor receptor |
| ZU5 | Domain named after tight junction protein ZO-1 and C. elegans Unc5 |
References
- Garcia, C.K.; Wilund, K.; Arca, M.; Zuliani, G.; Fellin, R.; Maioli, M.; Calandra, S.; Bertolini, S.; Cossu, F.; Grishin, N.; et al. Autosomal recessive hypercholesterolemia caused by mutations in a putative LDL receptor adaptor protein. Science 2001, 292, 1394–1398. [Google Scholar] [CrossRef] [PubMed]
- Tessneer, K.L.; Pasula, S.; Cai, X.; Dong, Y.; Liu, X.; Yu, L.; Hahn, S.; McManus, J.; Chen, Y.; Chang, B.; et al. Endocytic adaptor protein epsin is elevated in prostate cancer and required for cancer progression. ISRN Oncol. 2013, 2013, 420597. [Google Scholar] [CrossRef] [PubMed]
- Helbig, I.; Lopez-Hernandez, T.; Shor, O.; Galer, P.; Ganesan, S.; Pendziwiat, M.; Rademacher, A.; Ellis, C.A.; Humpfer, N.; Schwarz, N.; et al. A Recurrent Missense Variant in AP2M1 Impairs Clathrin-Mediated Endocytosis and Causes Developmental and Epileptic Encephalopathy. Am. J. Hum. Genet. 2019, 104, 1060–1072. [Google Scholar] [CrossRef] [PubMed]
- Kaksonen, M.; Roux, A. Mechanisms of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 2018, 19, 313–326. [Google Scholar] [CrossRef]
- Letourneur, F.; Klausner, R.D. A novel di-leucine motif and a tyrosine-based motif independently mediate lysosomal targeting and endocytosis of CD3 chains. Cell 1992, 69, 1143–1157. [Google Scholar] [CrossRef]
- Pond, L.; Kuhn, L.A.; Teyton, L.; Schutze, M.P.; Tainer, J.A.; Jackson, M.R.; Peterson, P.A. A role for acidic residues in di-leucine motif-based targeting to the endocytic pathway. J. Biol. Chem. 1995, 270, 19989–19997. [Google Scholar] [CrossRef]
- Kelly, B.T.; McCoy, A.J.; Spate, K.; Miller, S.E.; Evans, P.R.; Honing, S.; Owen, D.J. A structural explanation for the binding of endocytic dileucine motifs by the AP2 complex. Nature 2008, 456, 976–979. [Google Scholar] [CrossRef]
- Traub, L.M.; Bonifacino, J.S. Cargo recognition in clathrin-mediated endocytosis. Cold Spring Harb. Perspect. Biol. 2013, 5, a016790. [Google Scholar] [CrossRef]
- Ohno, H.; Fournier, M.C.; Poy, G.; Bonifacino, J.S. Structural determinants of interaction of tyrosine-based sorting signals with the adaptor medium chains. J. Biol. Chem. 1996, 271, 29009–29015. [Google Scholar] [CrossRef]
- Ohno, H.; Stewart, J.; Fournier, M.C.; Bosshart, H.; Rhee, I.; Miyatake, S.; Saito, T.; Gallusser, A.; Kirchhausen, T.; Bonifacino, J.S. Interaction of tyrosine-based sorting signals with clathrin-associated proteins. Science 1995, 269, 1872–1875. [Google Scholar] [CrossRef]
- Jung, N.; Wienisch, M.; Gu, M.; Rand, J.B.; Muller, S.L.; Krause, G.; Jorgensen, E.M.; Klingauf, J.; Haucke, V. Molecular basis of synaptic vesicle cargo recognition by the endocytic sorting adaptor stonin 2. J. Cell Biol. 2007, 179, 1497–1510. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Maritzen, T.; Wichmann, C.; Jing, Z.; Neef, A.; Revelo, N.H.; Al-Moyed, H.; Meese, S.; Wojcik, S.M.; Panou, I.; et al. Disruption of adaptor protein 2mu (AP-2mu) in cochlear hair cells impairs vesicle reloading of synaptic release sites and hearing. EMBO J. 2015, 34, 2686–2702. [Google Scholar] [CrossRef] [PubMed]
- Kelly, B.T.; Graham, S.C.; Liska, N.; Dannhauser, P.N.; Honing, S.; Ungewickell, E.J.; Owen, D.J. Clathrin adaptors. AP2 controls clathrin polymerization with a membrane-activated switch. Science 2014, 345, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Idrissi, F.Z.; Blasco, A.; Espinal, A.; Geli, M.I. Ultrastructural dynamics of proteins involved in endocytic budding. Proc. Natl. Acad. Sci. USA 2012, 109, E2587–E2594. [Google Scholar] [CrossRef] [PubMed]
- Kukulski, W.; Schorb, M.; Kaksonen, M.; Briggs, J.A. Plasma membrane reshaping during endocytosis is revealed by time-resolved electron tomography. Cell 2012, 150, 508–520. [Google Scholar] [CrossRef]
- Collins, A.; Warrington, A.; Taylor, K.A.; Svitkina, T. Structural organization of the actin cytoskeleton at sites of clathrin-mediated endocytosis. Curr. Biol. 2011, 21, 1167–1175. [Google Scholar] [CrossRef]
- Almeida-Souza, L.; Frank, R.A.W.; Garcia-Nafria, J.; Colussi, A.; Gunawardana, N.; Johnson, C.M.; Yu, M.; Howard, G.; Andrews, B.; Vallis, Y.; et al. A Flat BAR Protein Promotes Actin Polymerization at the Base of Clathrin-Coated Pits. Cell 2018, 174, 325–337.e14. [Google Scholar] [CrossRef]
- Antonny, B.; Burd, C.; De Camilli, P.; Chen, E.; Daumke, O.; Faelber, K.; Ford, M.; Frolov, V.A.; Frost, A.; Hinshaw, J.E.; et al. Membrane fission by dynamin: What we know and what we need to know. EMBO J. 2016, 35, 2270–2284. [Google Scholar] [CrossRef]
- Bashkirov, P.V.; Akimov, S.A.; Evseev, A.I.; Schmid, S.L.; Zimmerberg, J.; Frolov, V.A. GTPase cycle of dynamin is coupled to membrane squeeze and release, leading to spontaneous fission. Cell 2008, 135, 1276–1286. [Google Scholar] [CrossRef]
- Ungewickell, E. The 70-kd mammalian heat shock proteins are structurally and functionally related to the uncoating protein that releases clathrin triskelia from coated vesicles. EMBO J. 1985, 4, 3385–3391. [Google Scholar] [CrossRef]
- Newmyer, S.L.; Christensen, A.; Sever, S. Auxilin-dynamin interactions link the uncoating ATPase chaperone machinery with vesicle formation. Dev. Cell 2003, 4, 929–940. [Google Scholar] [CrossRef]
- Sousa, R.; Lafer, E.M. The role of molecular chaperones in clathrin mediated vesicular trafficking. Front. Mol. Biosci. 2015, 2, 26. [Google Scholar] [CrossRef] [PubMed]
- Kirchhausen, T.; Owen, D.; Harrison, S.C. Molecular structure, function, and dynamics of clathrin-mediated membrane traffic. Cold Spring Harb. Perspect. Biol. 2014, 6, a016725. [Google Scholar] [CrossRef] [PubMed]
- McMahon, H.T.; Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 2011, 12, 517–533. [Google Scholar] [CrossRef]
- Maritzen, T.; Podufall, J.; Haucke, V. Stonins--specialized adaptors for synaptic vesicle recycling and beyond? Traffic 2010, 11, 8–15. [Google Scholar] [CrossRef]
- Reider, A.; Barker, S.L.; Mishra, S.K.; Im, Y.J.; Maldonado-Baez, L.; Hurley, J.H.; Traub, L.M.; Wendland, B. Syp1 is a conserved endocytic adaptor that contains domains involved in cargo selection and membrane tubulation. EMBO J. 2009, 28, 3103–3116. [Google Scholar] [CrossRef]
- Maritzen, T.; Koo, S.J.; Haucke, V. Turning CALM into excitement: AP180 and CALM in endocytosis and disease. Biol. Cell 2012, 104, 588–602. [Google Scholar] [CrossRef]
- Gottfried, I.; Ehrlich, M.; Ashery, U. The Sla2p/HIP1/HIP1R family: Similar structure, similar function in endocytosis? BioChem. Soc. Trans. 2010, 38, 187–191. [Google Scholar] [CrossRef]
- Itoh, T.; De Camilli, P. BAR, F-BAR (EFC) and ENTH/ANTH domains in the regulation of membrane-cytosol interfaces and membrane curvature. Biochim. Biophys. Acta 2006, 1761, 897–912. [Google Scholar] [CrossRef]
- Traub, L.M. A nanobody-based molecular toolkit provides new mechanistic insight into clathrin-coat initiation. Elife 2019, 8. [Google Scholar] [CrossRef]
- Mishra, S.K.; Jha, A.; Steinhauser, A.L.; Kokoza, V.A.; Washabaugh, C.H.; Raikhel, A.S.; Foster, W.A.; Traub, L.M. Internalization of LDL-receptor superfamily yolk-protein receptors during mosquito oogenesis involves transcriptional regulation of PTB-domain adaptors. J. Cell Sci. 2008, 121, 1264–1274. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maurer, M.E.; Cooper, J.A. Endocytosis of megalin by visceral endoderm cells requires the Dab2 adaptor protein. J. Cell Sci. 2005, 118, 5345–5355. [Google Scholar] [CrossRef] [PubMed]
- Gulino, A.; Di Marcotullio, L.; Screpanti, I. The multiple functions of Numb. Exp. Cell Res. 2010, 316, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Puca, L.; Brou, C. Alpha-arrestins - new players in Notch and GPCR signaling pathways in mammals. J. Cell Sci. 2014, 127, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Pryor, P.R.; Jackson, L.; Gray, S.R.; Edeling, M.A.; Thompson, A.; Sanderson, C.M.; Evans, P.R.; Owen, D.J.; Luzio, J.P. Molecular basis for the sorting of the SNARE VAMP7 into endocytic clathrin-coated vesicles by the ArfGAP Hrb. Cell 2008, 134, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Tosoni, D.; Puri, C.; Confalonieri, S.; Salcini, A.E.; De Camilli, P.; Tacchetti, C.; Di Fiore, P.P. TTP specifically regulates the internalization of the transferrin receptor. Cell 2005, 123, 875–888. [Google Scholar] [CrossRef]
- Mudduluru, G.; Ilm, K.; Dahlman, M.; Stein, U. MACC1, a Novel Player in Solid Cancer Carcinogenesis. In Mechanisms of Molecular Carcinogenesis; Springer: Cham, Switzerland, 2017; Volume 1, pp. 11–38. [Google Scholar]
- Kononenko, N.L.; Classen, G.A.; Kuijpers, M.; Puchkov, D.; Maritzen, T.; Tempes, A.; Malik, A.R.; Skalecka, A.; Bera, S.; Jaworski, J.; et al. Retrograde transport of TrkB-containing autophagosomes via the adaptor AP-2 mediates neuronal complexity and prevents neurodegeneration. Nat. Commun. 2017, 8, 14819. [Google Scholar] [CrossRef]
- Mitsunari, T.; Nakatsu, F.; Shioda, N.; Love, P.E.; Grinberg, A.; Bonifacino, J.S.; Ohno, H. Clathrin adaptor AP-2 is essential for early embryonal development. Mol. Cell Biol. 2005, 25, 9318–9323. [Google Scholar] [CrossRef]
- Li, W.; Puertollano, R.; Bonifacino, J.S.; Overbeek, P.A.; Everett, E.T. Disruption of the murine Ap2beta1 gene causes nonsyndromic cleft palate. Cleft Palate Craniofac. J. 2010, 47, 566–573. [Google Scholar] [CrossRef]
- Gorvin, C.M.; Rogers, A.; Stewart, M.; Paudyal, A.; Hough, T.A.; Teboul, L.; Wells, S.; Brown, S.D.; Cox, R.D.; Thakker, R.V. N-ethyl-N-nitrosourea-Induced Adaptor Protein 2 Sigma Subunit 1 (Ap2s1) Mutations Establish Ap2s1 Loss-of-Function Mice. JBMR Plus 2017, 1, 3–15. [Google Scholar] [CrossRef]
- Li, K.; Li, J.; Zheng, J.; Qin, S. Reactive Astrocytes in Neurodegenerative Diseases. Aging Dis. 2019, 10, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Nesbit, M.A.; Hannan, F.M.; Howles, S.A.; Reed, A.A.; Cranston, T.; Thakker, C.E.; Gregory, L.; Rimmer, A.J.; Rust, N.; Graham, U.; et al. Mutations in AP2S1 cause familial hypocalciuric hypercalcemia type 3. Nat. Genet. 2013, 45, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Shoback, D.M. Familial hypocalciuric hypercalcemia and related disorders. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Hannan, F.M.; Howles, S.A.; Rogers, A.; Cranston, T.; Gorvin, C.M.; Babinsky, V.N.; Reed, A.A.; Thakker, C.E.; Bockenhauer, D.; Brown, R.S.; et al. Adaptor protein-2 sigma subunit mutations causing familial hypocalciuric hypercalcaemia type 3 (FHH3) demonstrate genotype-phenotype correlations, codon bias and dominant-negative effects. Hum. Mol. Genet. 2015, 24, 5079–5092. [Google Scholar] [CrossRef]
- Gorvin, C.M.; Metpally, R.; Stokes, V.J.; Hannan, F.M.; Krishnamurthy, S.B.; Overton, J.D.; Reid, J.G.; Breitwieser, G.E.; Thakker, R.V. Large-scale exome datasets reveal a new class of adaptor-related protein complex 2 sigma subunit (AP2sigma) mutations, located at the interface with the AP2 alpha subunit, that impair calcium-sensing receptor signalling. Hum. Mol. Genet. 2018, 27, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Raj, T.; Li, Y.I.; Wong, G.; Humphrey, J.; Wang, M.; Ramdhani, S.; Wang, Y.C.; Ng, B.; Gupta, I.; Haroutunian, V.; et al. Integrative transcriptome analyses of the aging brain implicate altered splicing in Alzheimer’s disease susceptibility. Nat. Genet. 2018, 50, 1584–1592. [Google Scholar] [CrossRef] [PubMed]
- Buser, D.P.; Ritz, M.F.; Moes, S.; Tostado, C.; Frank, S.; Spiess, M.; Mariani, L.; Jeno, P.; Boulay, J.L.; Hutter, G. Quantitative proteomics reveals reduction of endocytic machinery components in gliomas. EBioMedicine 2019, 46, 32–41. [Google Scholar] [CrossRef]
- Montagnac, G.; Meas-Yedid, V.; Irondelle, M.; Castro-Castro, A.; Franco, M.; Shida, T.; Nachury, M.V.; Benmerah, A.; Olivo-Marin, J.C.; Chavrier, P. alphaTAT1 catalyses microtubule acetylation at clathrin-coated pits. Nature 2013, 502, 567–570. [Google Scholar] [CrossRef]
- Neveu, G.; Ziv-Av, A.; Barouch-Bentov, R.; Berkerman, E.; Mulholland, J.; Einav, S. AP-2-associated protein kinase 1 and cyclin G-associated kinase regulate hepatitis C virus entry and are potential drug targets. J. Virol. 2015, 89, 4387–4404. [Google Scholar] [CrossRef]
- Stoneham, C.A.; Singh, R.; Jia, X.; Xiong, Y.; Guatelli, J. Endocytic activity of HIV-1 Vpu: Phosphoserine-dependent interactions with clathrin adaptors. Traffic 2017, 18, 545–561. [Google Scholar] [CrossRef]
- Rollason, R.; Korolchuk, V.; Hamilton, C.; Schu, P.; Banting, G. Clathrin-mediated endocytosis of a lipid-raft-associated protein is mediated through a dual tyrosine motif. J. Cell Sci. 2007, 120, 3850–3858. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.; Kirchner, M.; Uyar, B.; Cheng, J.Y.; Russo, G.; Hernandez-Miranda, L.R.; Szymborska, A.; Zauber, H.; Rudolph, I.M.; Willnow, T.E.; et al. Mutations in Disordered Regions Can Cause Disease by Creating Dileucine Motifs. Cell 2018, 175, 239–253.e217. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.; Smith, M.; Merakovsky, J.; Coulson, M.; Hannan, F.; Kelly, L.E. The stoned locus of Drosophila melanogaster produces a dicistronic transcript and encodes two distinct polypeptides. Genetics 1996, 143, 1699–1711. [Google Scholar] [PubMed]
- Grigliatti, T.A.; Hall, L.; Rosenbluth, R.; Suzuki, D.T. Temperature-sensitive mutations in Drosophila melanogaster. XIV. A selection of immobile adults. Mol. Gen. Genet. 1973, 120, 107–114. [Google Scholar] [CrossRef]
- Fergestad, T.; Broadie, K. Interaction of stoned and synaptotagmin in synaptic vesicle endocytosis. J. Neurosci. 2001, 21, 1218–1227. [Google Scholar] [CrossRef]
- Fergestad, T.; Davis, W.S.; Broadie, K. The stoned proteins regulate synaptic vesicle recycling in the presynaptic terminal. J. Neurosci. 1999, 19, 5847–5860. [Google Scholar] [CrossRef]
- Fernandez-Chacon, R.; Konigstorfer, A.; Gerber, S.H.; Garcia, J.; Matos, M.F.; Stevens, C.F.; Brose, N.; Rizo, J.; Rosenmund, C.; Sudhof, T.C. Synaptotagmin I functions as a calcium regulator of release probability. Nature 2001, 410, 41–49. [Google Scholar] [CrossRef]
- Grass, I.; Thiel, S.; Honing, S.; Haucke, V. Recognition of a basic AP-2 binding motif within the C2B domain of synaptotagmin is dependent on multimerization. J. Biol. Chem. 2004, 279, 54872–54880. [Google Scholar] [CrossRef]
- Haucke, V.; Wenk, M.R.; Chapman, E.R.; Farsad, K.; De Camilli, P. Dual interaction of synaptotagmin with mu2- and alpha-adaptin facilitates clathrin-coated pit nucleation. Embo J. 2000, 19, 6011–6019. [Google Scholar] [CrossRef]
- Feany, M.B.; Buckley, K.M. The synaptic vesicle protein synaptotagmin promotes formation of filopodia in fibroblasts. Nature 1993, 364, 537–540. [Google Scholar] [CrossRef]
- Jarousse, N.; Kelly, R.B. The AP2 binding site of synaptotagmin 1 is not an internalization signal but a regulator of endocytosis. J. Cell Biol. 2001, 154, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Diril, M.K.; Wienisch, M.; Jung, N.; Klingauf, J.; Haucke, V. Stonin 2 is an AP-2-dependent endocytic sorting adaptor for synaptotagmin internalization and recycling. Dev. Cell 2006, 10, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Martina, J.A.; Bonangelino, C.J.; Aguilar, R.C.; Bonifacino, J.S. Stonin 2: An adaptor-like protein that interacts with components of the endocytic machinery. J. Cell Biol. 2001, 153, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.M.; Smith, M.; Ramaswami, M.; Kelly, L.E. The products of the Drosophila stoned locus interact with synaptic vesicles via synaptotagmin. J. Neurosci. 2000, 20, 8254–8261. [Google Scholar] [CrossRef]
- Walther, K.; Krauss, M.; Diril, M.K.; Lemke, S.; Ricotta, D.; Honing, S.; Kaiser, S.; Haucke, V. Human stoned B interacts with AP-2 and synaptotagmin and facilitates clathrin-coated vesicle uncoating. EMBO Rep. 2001, 2, 634–640. [Google Scholar] [CrossRef]
- Kononenko, N.L.; Diril, M.K.; Puchkov, D.; Kintscher, M.; Koo, S.J.; Pfuhl, G.; Winter, Y.; Wienisch, M.; Klingauf, J.; Breustedt, J.; et al. Compromised fidelity of endocytic synaptic vesicle protein sorting in the absence of stonin 2. Proc. Natl. Acad. Sci. USA 2013, 110, E526–E535. [Google Scholar] [CrossRef]
- Bartholome, O.; Van den Ackerveken, P.; Sanchez Gil, J.; de la Brassinne Bonardeaux, O.; Leprince, P.; Franzen, R.; Rogister, B. Puzzling Out Synaptic Vesicle 2 Family Members Functions. Front. Mol. Neurosci. 2017, 10, 148. [Google Scholar] [CrossRef]
- Lazzell, D.R.; Belizaire, R.; Thakur, P.; Sherry, D.M.; Janz, R. SV2B regulates synaptotagmin 1 by direct interaction. J. Biol. Chem. 2004, 279, 52124–52131. [Google Scholar] [CrossRef]
- Schivell, A.E.; Batchelor, R.H.; Bajjalieh, S.M. Isoform-specific, calcium-regulated interaction of the synaptic vesicle proteins SV2 and synaptotagmin. J. Biol. Chem. 1996, 271, 27770–27775. [Google Scholar] [CrossRef]
- Schivell, A.E.; Mochida, S.; Kensel-Hammes, P.; Custer, K.L.; Bajjalieh, S.M. SV2A and SV2C contain a unique synaptotagmin-binding site. Mol. Cell. Neurosci. 2005, 29, 56–64. [Google Scholar] [CrossRef]
- Yao, J.; Nowack, A.; Kensel-Hammes, P.; Gardner, R.G.; Bajjalieh, S.M. Cotrafficking of SV2 and synaptotagmin at the synapse. J. Neurosci. 2010, 30, 5569–5578. [Google Scholar] [CrossRef]
- Kaempf, N.; Kochlamazashvili, G.; Puchkov, D.; Maritzen, T.; Bajjalieh, S.M.; Kononenko, N.L.; Haucke, V. Overlapping functions of stonin 2 and SV2 in sorting of the calcium sensor synaptotagmin 1 to synaptic vesicles. Proc. Natl. Acad. Sci. USA 2015, 112, 7297–7302. [Google Scholar] [CrossRef]
- Gordon, S.L.; Leube, R.E.; Cousin, M.A. Synaptophysin is required for synaptobrevin retrieval during synaptic vesicle endocytosis. J. Neurosci. 2011, 31, 14032–14036. [Google Scholar] [CrossRef]
- Rajappa, R.; Gauthier-Kemper, A.; Boning, D.; Huve, J.; Klingauf, J. Synaptophysin 1 Clears Synaptobrevin 2 from the Presynaptic Active Zone to Prevent Short-Term Depression. Cell Rep. 2016, 14, 1369–1381. [Google Scholar] [CrossRef]
- Awasthi, A.; Ramachandran, B.; Ahmed, S.; Benito, E.; Shinoda, Y.; Nitzan, N.; Heukamp, A.; Rannio, S.; Martens, H.; Barth, J.; et al. Synaptotagmin-3 drives AMPA receptor endocytosis, depression of synapse strength, and forgetting. Science 2019, 363. [Google Scholar] [CrossRef]
- Feutlinske, F.; Browarski, M.; Ku, M.C.; Trnka, P.; Waiczies, S.; Niendorf, T.; Stallcup, W.B.; Glass, R.; Krause, E.; Maritzen, T. Stonin1 mediates endocytosis of the proteoglycan NG2 and regulates focal adhesion dynamics and cell motility. Nat. Commun. 2015, 6, 8535. [Google Scholar] [CrossRef]
- Breedveld, G.J.; Fabbrini, G.; Oostra, B.A.; Berardelli, A.; Bonifati, V. Tourette disorder spectrum maps to chromosome 14q31.1 in an Italian kindred. Neurogenetics 2010, 11, 417–423. [Google Scholar] [CrossRef]
- Luan, Z.; Zhang, Y.; Lu, T.; Ruan, Y.; Zhang, H.; Yan, J.; Li, L.; Sun, W.; Wang, L.; Yue, W.; et al. Positive association of the human STON2 gene with schizophrenia. Neuroreport 2011, 22, 288–293. [Google Scholar] [CrossRef]
- Shimada, A.; Yamaguchi, A.; Kohda, D. Structural basis for the recognition of two consecutive mutually interacting DPF motifs by the SGIP1 mu homology domain. Sci. Rep. 2016, 6, 19565. [Google Scholar] [CrossRef]
- Mulkearns, E.E.; Cooper, J.A. FCH domain only-2 organizes clathrin-coated structures and interacts with Disabled-2 for low-density lipoprotein receptor endocytosis. Mol. Biol. Cell 2012, 23, 1330–1342. [Google Scholar] [CrossRef]
- Katoh, M.; Katoh, M. Identification and characterization of human FCHO2 and mouse Fcho2 genes in silico. Int. J. Mol. Med. 2004, 14, 327–331. [Google Scholar]
- Henne, W.M.; Kent, H.M.; Ford, M.G.; Hegde, B.G.; Daumke, O.; Butler, P.J.; Mittal, R.; Langen, R.; Evans, P.R.; McMahon, H.T. Structure and analysis of FCHo2 F-BAR domain: A dimerizing and membrane recruitment module that effects membrane curvature. Structure 2007, 15, 839–852. [Google Scholar] [CrossRef]
- Uezu, A.; Horiuchi, A.; Kanda, K.; Kikuchi, N.; Umeda, K.; Tsujita, K.; Suetsugu, S.; Araki, N.; Yamamoto, H.; Takenawa, T.; et al. SGIP1alpha is an endocytic protein that directly interacts with phospholipids and Eps15. J. Biol. Chem. 2007, 282, 26481–26489. [Google Scholar] [CrossRef]
- Henne, W.M.; Boucrot, E.; Meinecke, M.; Evergren, E.; Vallis, Y.; Mittal, R.; McMahon, H.T. FCHo proteins are nucleators of clathrin-mediated endocytosis. Science 2010, 328, 1281–1284. [Google Scholar] [CrossRef]
- Uezu, A.; Umeda, K.; Tsujita, K.; Suetsugu, S.; Takenawa, T.; Nakanishi, H. Characterization of the EFC/F-BAR domain protein, FCHO2. Genes Cells 2011, 16, 868–878. [Google Scholar] [CrossRef]
- Umasankar, P.K.; Sanker, S.; Thieman, J.R.; Chakraborty, S.; Wendland, B.; Tsang, M.; Traub, L.M. Distinct and separable activities of the endocytic clathrin-coat components Fcho1/2 and AP-2 in developmental patterning. Nat. Cell Biol. 2012, 14, 488–501. [Google Scholar] [CrossRef]
- Cocucci, E.; Aguet, F.; Boulant, S.; Kirchhausen, T. The first five seconds in the life of a clathrin-coated pit. Cell 2012, 150, 495–507. [Google Scholar] [CrossRef]
- Hollopeter, G.; Lange, J.J.; Zhang, Y.; Vu, T.N.; Gu, M.; Ailion, M.; Lambie, E.J.; Slaughter, B.D.; Unruh, J.R.; Florens, L.; et al. The membrane-associated proteins FCHo and SGIP are allosteric activators of the AP2 clathrin adaptor complex. Elife 2014, 3. [Google Scholar] [CrossRef]
- Ma, L.; Umasankar, P.K.; Wrobel, A.G.; Lymar, A.; McCoy, A.J.; Holkar, S.S.; Jha, A.; Pradhan-Sundd, T.; Watkins, S.C.; Owen, D.J.; et al. Transient Fcho1/2Eps15/RAP-2 Nanoclusters Prime the AP-2 Clathrin Adaptor for Cargo Binding. Dev. Cell 2016, 37, 428–443. [Google Scholar] [CrossRef]
- Umasankar, P.K.; Ma, L.; Thieman, J.R.; Jha, A.; Doray, B.; Watkins, S.C.; Traub, L.M. A clathrin coat assembly role for the muniscin protein central linker revealed by TALEN-mediated gene editing. Elife 2014, 3. [Google Scholar] [CrossRef]
- Lee, S.E.; Jeong, S.; Lee, U.; Chang, S. SGIP1alpha functions as a selective endocytic adaptor for the internalization of synaptotagmin 1 at synapses. Mol. Brain 2019, 12, 41. [Google Scholar] [CrossRef]
- Hajkova, A.; Techlovska, S.; Dvorakova, M.; Chambers, J.N.; Kumpost, J.; Hubalkova, P.; Prezeau, L.; Blahos, J. SGIP1 alters internalization and modulates signaling of activated cannabinoid receptor 1 in a biased manner. Neuropharmacology 2016, 107, 201–214. [Google Scholar] [CrossRef]
- Calzoni, E.; Platt, C.D.; Keles, S.; Kuehn, H.S.; Beaussant-Cohen, S.; Zhang, Y.; Pazmandi, J.; Lanzi, G.; Pala, F.; Tahiat, A.; et al. F-BAR domain only protein 1 (FCHO1) deficiency is a novel cause of combined immune deficiency in human subjects. J. Allergy Clin. Immunol. 2019, 143, 2317–2321.e2312. [Google Scholar] [CrossRef]
- Trevaskis, J.; Walder, K.; Foletta, V.; Kerr-Bayles, L.; McMillan, J.; Cooper, A.; Lee, S.; Bolton, K.; Prior, M.; Fahey, R.; et al. Src homology 3-domain growth factor receptor-bound 2-like (endophilin) interacting protein 1, a novel neuronal protein that regulates energy balance. Endocrinology 2005, 146, 3757–3764. [Google Scholar] [CrossRef]
- Cummings, N.; Shields, K.A.; Curran, J.E.; Bozaoglu, K.; Trevaskis, J.; Gluschenko, K.; Cai, G.; Comuzzie, A.G.; Dyer, T.D.; Walder, K.R.; et al. Genetic variation in SH3-domain GRB2-like (endophilin)-interacting protein 1 has a major impact on fat mass. Int. J. Obes. (Lond.) 2012, 36, 201–206. [Google Scholar] [CrossRef]
- Yako, Y.Y.; Echouffo-Tcheugui, J.B.; Balti, E.V.; Matsha, T.E.; Sobngwi, E.; Erasmus, R.T.; Kengne, A.P. Genetic association studies of obesity in Africa: A systematic review. Obes. Rev. 2015, 16, 259–272. [Google Scholar] [CrossRef]
- Chwedorowicz, R.; Raszewski, G.; Kapka-Skrzypczak, L.; Sawicki, K.; Studzinski, T. Event-related potentials (ERP) and SGIP1 gene polymorphisms in alcoholics: Relation to family history of alcoholism and drug usage. Ann. Agric. Environ. Med. 2016, 23, 618–624. [Google Scholar] [CrossRef]
- Derringer, J.; Krueger, R.F.; Manz, N.; Porjesz, B.; Almasy, L.; Bookman, E.; Edenberg, H.J.; Kramer, J.R.; Tischfield, J.A.; Bierut, L.J. Nonreplication of an association of SGIP1 SNPs with alcohol dependence and resting theta EEG power. Psychiatr. Genet. 2011, 21, 265–266. [Google Scholar] [CrossRef]
- Hodgkinson, C.A.; Enoch, M.A.; Srivastava, V.; Cummins-Oman, J.S.; Ferrier, C.; Iarikova, P.; Sankararaman, S.; Yamini, G.; Yuan, Q.; Zhou, Z.; et al. Genome-wide association identifies candidate genes that influence the human electroencephalogram. Proc. Natl. Acad. Sci. USA 2010, 107, 8695–8700. [Google Scholar] [CrossRef]
- van Setten, J.; Verweij, N.; Mbarek, H.; Niemeijer, M.N.; Trompet, S.; Arking, D.E.; Brody, J.A.; Gandin, I.; Grarup, N.; Hall, L.M.; et al. Genome-wide association meta-analysis of 30,000 samples identifies seven novel loci for quantitative ECG traits. Eur. J. Hum. Genet. 2019, 27, 952–962. [Google Scholar] [CrossRef]
- Ahle, S.; Ungewickell, E. Purification and properties of a new clathrin assembly protein. EMBO J. 1986, 5, 3143–3149. [Google Scholar] [CrossRef]
- Kohtz, D.S.; Puszkin, S. A neuronal protein (NP185) associated with clathrin-coated vesicles. Characterization of NP185 with monoclonal antibodies. J. Biol. Chem. 1988, 263, 7418–7425. [Google Scholar]
- Dreyling, M.H.; Martinez-Climent, J.A.; Zheng, M.; Mao, J.; Rowley, J.D.; Bohlander, S.K. The t(10;11)(p13;q14) in the U937 cell line results in the fusion of the AF10 gene and CALM, encoding a new member of the AP-3 clathrin assembly protein family. Proc. Natl. Acad. Sci. USA 1996, 93, 4804–4809. [Google Scholar] [CrossRef]
- Koo, S.J.; Kochlamazashvili, G.; Rost, B.; Puchkov, D.; Gimber, N.; Lehmann, M.; Tadeus, G.; Schmoranzer, J.; Rosenmund, C.; Haucke, V.; et al. Vesicular Synaptobrevin/VAMP2 Levels Guarded by AP180 Control Efficient Neurotransmission. Neuron 2015, 88, 330–344. [Google Scholar] [CrossRef]
- Tebar, F.; Bohlander, S.K.; Sorkin, A. Clathrin assembly lymphoid myeloid leukemia (CALM) protein: Localization in endocytic-coated pits, interactions with clathrin, and the impact of overexpression on clathrin-mediated traffic. Mol. Biol. Cell 1999, 10, 2687–2702. [Google Scholar] [CrossRef]
- Yao, P.J.; Petralia, R.S.; Bushlin, I.; Wang, Y.; Furukawa, K. Synaptic distribution of the endocytic accessory proteins AP180 and CALM. J. Comp. Neurol. 2005, 481, 58–69. [Google Scholar] [CrossRef]
- Ford, M.G.; Pearse, B.M.; Higgins, M.K.; Vallis, Y.; Owen, D.J.; Gibson, A.; Hopkins, C.R.; Evans, P.R.; McMahon, H.T. Simultaneous binding of PtdIns(4,5)P2 and clathrin by AP180 in the nucleation of clathrin lattices on membranes. Science 2001, 291, 1051–1055. [Google Scholar] [CrossRef]
- Ye, W.; Lafer, E.M. Bacterially expressed F1-20/AP-3 assembles clathrin into cages with a narrow size distribution: Implications for the regulation of quantal size during neurotransmission. J. Neurosci. Res. 1995, 41, 15–26. [Google Scholar] [CrossRef]
- Miller, S.E.; Mathiasen, S.; Bright, N.A.; Pierre, F.; Kelly, B.T.; Kladt, N.; Schauss, A.; Merrifield, C.J.; Stamou, D.; Honing, S.; et al. CALM regulates clathrin-coated vesicle size and maturation by directly sensing and driving membrane curvature. Dev. Cell 2015, 33, 163–175. [Google Scholar] [CrossRef]
- Meyerholz, A.; Hinrichsen, L.; Groos, S.; Esk, P.C.; Brandes, G.; Ungewickell, E.J. Effect of clathrin assembly lymphoid myeloid leukemia protein depletion on clathrin coat formation. Traffic 2005, 6, 1225–1234. [Google Scholar] [CrossRef]
- Sahlender, D.A.; Kozik, P.; Miller, S.E.; Peden, A.A.; Robinson, M.S. Uncoupling the functions of CALM in VAMP sorting and clathrin-coated pit formation. PLoS ONE 2013, 8, e64514. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Maeda, M.; Pasham, M.; Aguet, F.; Tacheva-Grigorova, S.K.; Masuda, T.; Yi, H.; Lee, S.U.; Xu, J.; Teruya-Feldstein, J.; et al. Role of the clathrin adaptor PICALM in normal hematopoiesis and polycythemia vera pathophysiology. Haematologica 2015, 100, 439–451. [Google Scholar] [CrossRef]
- Burston, H.E.; Maldonado-Baez, L.; Davey, M.; Montpetit, B.; Schluter, C.; Wendland, B.; Conibear, E. Regulators of yeast endocytosis identified by systematic quantitative analysis. J. Cell Biol. 2009, 185, 1097–1110. [Google Scholar] [CrossRef]
- Wen, Y.; Stavrou, I.; Bersuker, K.; Brady, R.J.; De Lozanne, A.; O’Halloran, T.J. AP180-mediated trafficking of Vamp7B limits homotypic fusion of Dictyostelium contractile vacuoles. Mol. Biol. Cell 2009, 20, 4278–4288. [Google Scholar] [CrossRef]
- Dittman, J.S.; Kaplan, J.M. Factors regulating the abundance and localization of synaptobrevin in the plasma membrane. Proc. Natl. Acad. Sci. USA 2006, 103, 11399–11404. [Google Scholar] [CrossRef]
- Nonet, M.L.; Holgado, A.M.; Brewer, F.; Serpe, C.J.; Norbeck, B.A.; Holleran, J.; Wei, L.; Hartwieg, E.; Jorgensen, E.M.; Alfonso, A. UNC-11, a Caenorhabditis elegans AP180 homologue, regulates the size and protein composition of synaptic vesicles. Mol. Biol. Cell 1999, 10, 2343–2360. [Google Scholar] [CrossRef]
- Burbea, M.; Dreier, L.; Dittman, J.S.; Grunwald, M.E.; Kaplan, J.M. Ubiquitin and AP180 regulate the abundance of GLR-1 glutamate receptors at postsynaptic elements in C. elegans. Neuron 2002, 35, 107–120. [Google Scholar] [CrossRef]
- Bao, H.; Daniels, R.W.; MacLeod, G.T.; Charlton, M.P.; Atwood, H.L.; Zhang, B. AP180 maintains the distribution of synaptic and vesicle proteins in the nerve terminal and indirectly regulates the efficacy of Ca2+-triggered exocytosis. J. Neurophysiol. 2005, 94, 1888–1903. [Google Scholar] [CrossRef]
- Zhang, B.; Koh, Y.H.; Beckstead, R.B.; Budnik, V.; Ganetzky, B.; Bellen, H.J. Synaptic vesicle size and number are regulated by a clathrin adaptor protein required for endocytosis. Neuron 1998, 21, 1465–1475. [Google Scholar] [CrossRef]
- Koo, S.J.; Markovic, S.; Puchkov, D.; Mahrenholz, C.C.; Beceren-Braun, F.; Maritzen, T.; Dernedde, J.; Volkmer, R.; Oschkinat, H.; Haucke, V. SNARE motif-mediated sorting of synaptobrevin by the endocytic adaptors clathrin assembly lymphoid myeloid leukemia (CALM) and AP180 at synapses. Proc. Natl. Acad. Sci. USA 2011, 108, 13540–13545. [Google Scholar] [CrossRef]
- Miller, S.E.; Sahlender, D.A.; Graham, S.C.; Honing, S.; Robinson, M.S.; Peden, A.A.; Owen, D.J. The molecular basis for the endocytosis of small R-SNAREs by the clathrin adaptor CALM. Cell 2011, 147, 1118–1131. [Google Scholar] [CrossRef]
- Moreau, K.; Fleming, A.; Imarisio, S.; Lopez Ramirez, A.; Mercer, J.L.; Jimenez-Sanchez, M.; Bento, C.F.; Puri, C.; Zavodszky, E.; Siddiqi, F.; et al. PICALM modulates autophagy activity and tau accumulation. Nat. Commun. 2014, 5, 4998. [Google Scholar] [CrossRef]
- Suzuki, M.; Tanaka, H.; Tanimura, A.; Tanabe, K.; Oe, N.; Rai, S.; Kon, S.; Fukumoto, M.; Takei, K.; Abe, T.; et al. The Clathrin Assembly Protein PICALM Is Required for Erythroid Maturation and Transferrin Internalization in Mice. PLoS ONE 2012, 7, e31854. [Google Scholar] [CrossRef]
- Goes, F.S.; Hamshere, M.L.; Seifuddin, F.; Pirooznia, M.; Belmonte-Mahon, P.; Breuer, R.; Schulze, T.; Nothen, M.; Cichon, S.; Rietschel, M.; et al. Genome-wide association of mood-incongruent psychotic bipolar disorder. Trans. Psychiatry 2012, 2, e180. [Google Scholar] [CrossRef]
- Ben-David, E.; Shifman, S. Networks of neuronal genes affected by common and rare variants in autism spectrum disorders. PLoS Genet. 2012, 8, e1002556. [Google Scholar] [CrossRef]
- Harold, D.; Abraham, R.; Hollingworth, P.; Sims, R.; Gerrish, A.; Hamshere, M.L.; Pahwa, J.S.; Moskvina, V.; Dowzell, K.; Williams, A.; et al. Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease. Nat. Genet. 2009, 41, 1088–1093. [Google Scholar] [CrossRef]
- Zhao, Z.; Sagare, A.P.; Ma, Q.; Halliday, M.R.; Kong, P.; Kisler, K.; Winkler, E.A.; Ramanathan, A.; Kanekiyo, T.; Bu, G.; et al. Central role for PICALM in amyloid-beta blood-brain barrier transcytosis and clearance. Nat. Neurosci. 2015, 18, 978–987. [Google Scholar] [CrossRef]
- Merthan, L.; Haller, A.; Thal, D.R.; von Einem, B.; von Arnim, C.A.F. The role of PTB domain containing adaptor proteins on PICALM-mediated APP endocytosis and localization. BioChem. J. 2019, 476, 2093–2109. [Google Scholar] [CrossRef]
- Xiao, Q.; Gil, S.C.; Yan, P.; Wang, Y.; Han, S.; Gonzales, E.; Perez, R.; Cirrito, J.R.; Lee, J.M. Role of phosphatidylinositol clathrin assembly lymphoid-myeloid leukemia (PICALM) in intracellular amyloid precursor protein (APP) processing and amyloid plaque pathogenesis. J. Biol. Chem. 2012, 287, 21279–21289. [Google Scholar] [CrossRef]
- Kanatsu, K.; Morohashi, Y.; Suzuki, M.; Kuroda, H.; Watanabe, T.; Tomita, T.; Iwatsubo, T. Decreased CALM expression reduces Abeta42 to total Abeta ratio through clathrin-mediated endocytosis of gamma-secretase. Nat. Commun. 2014, 5, 3386. [Google Scholar] [CrossRef]
- Park, S.K.; Ratia, K.; Ba, M.; Valencik, M.; Liebman, S.W. Inhibition of Abeta42 oligomerization in yeast by a PICALM ortholog and certain FDA approved drugs. Microb. Cell 2016, 3, 53–64. [Google Scholar] [CrossRef]
- Treusch, S.; Hamamichi, S.; Goodman, J.L.; Matlack, K.E.; Chung, C.Y.; Baru, V.; Shulman, J.M.; Parrado, A.; Bevis, B.J.; Valastyan, J.S.; et al. Functional links between Abeta toxicity, endocytic trafficking, and Alzheimer’s disease risk factors in yeast. Science 2011, 334, 1241–1245. [Google Scholar] [CrossRef]
- Kalchman, M.A.; Koide, H.B.; McCutcheon, K.; Graham, R.K.; Nichol, K.; Nishiyama, K.; Kazemi-Esfarjani, P.; Lynn, F.C.; Wellington, C.; Metzler, M.; et al. HIP1, a human homologue of S. cerevisiae Sla2p, interacts with membrane-associated huntingtin in the brain. Nat. Genet. 1997, 16, 44–53. [Google Scholar] [CrossRef]
- Chopra, V.S.; Metzler, M.; Rasper, D.M.; Engqvist-Goldstein, A.E.; Singaraja, R.; Gan, L.; Fichter, K.M.; McCutcheon, K.; Drubin, D.; Nicholson, D.W.; et al. HIP12 is a non-proapoptotic member of a gene family including HIP1, an interacting protein with huntingtin. Mamm Genome 2000, 11, 1006–1015. [Google Scholar] [CrossRef]
- Seki, N.; Muramatsu, M.; Sugano, S.; Suzuki, Y.; Nakagawara, A.; Ohhira, M.; Hayashi, A.; Hori, T.; Saito, T. Cloning, expression analysis, and chromosomal localization of HIP1R, an isolog of huntingtin interacting protein (HIP1). J. Hum. Genet. 1998, 43, 268–271. [Google Scholar] [CrossRef]
- Legendre-Guillemin, V.; Metzler, M.; Charbonneau, M.; Gan, L.; Chopra, V.; Philie, J.; Hayden, M.R.; McPherson, P.S. HIP1 and HIP12 display differential binding to F-actin, AP2, and clathrin. Identification of a novel interaction with clathrin light chain. J. Biol. Chem. 2002, 277, 19897–19904. [Google Scholar] [CrossRef]
- Metzler, M.; Li, B.; Gan, L.; Georgiou, J.; Gutekunst, C.A.; Wang, Y.; Torre, E.; Devon, R.S.; Oh, R.; Legendre-Guillemin, V.; et al. Disruption of the endocytic protein HIP1 results in neurological deficits and decreased AMPA receptor trafficking. EMBO J. 2003, 22, 3254–3266. [Google Scholar] [CrossRef]
- Hyun, T.S.; Li, L.; Oravecz-Wilson, K.I.; Bradley, S.V.; Provot, M.M.; Munaco, A.J.; Mizukami, I.F.; Sun, H.; Ross, T.S. Hip1-related mutant mice grow and develop normally but have accelerated spinal abnormalities and dwarfism in the absence of HIP1. Mol. Cell Biol. 2004, 24, 4329–4340. [Google Scholar] [CrossRef]
- Engqvist-Goldstein, A.E.; Kessels, M.M.; Chopra, V.S.; Hayden, M.R.; Drubin, D.G. An actin-binding protein of the Sla2/Huntingtin interacting protein 1 family is a novel component of clathrin-coated pits and vesicles. J. Cell Biol. 1999, 147, 1503–1518. [Google Scholar] [CrossRef]
- Metzler, M.; Legendre-Guillemin, V.; Gan, L.; Chopra, V.; Kwok, A.; McPherson, P.S.; Hayden, M.R. HIP1 functions in clathrin-mediated endocytosis through binding to clathrin and adaptor protein 2. J. Biol. Chem. 2001, 276, 39271–39276. [Google Scholar] [CrossRef]
- Waelter, S.; Scherzinger, E.; Hasenbank, R.; Nordhoff, E.; Lurz, R.; Goehler, H.; Gauss, C.; Sathasivam, K.; Bates, G.P.; Lehrach, H.; et al. The huntingtin interacting protein HIP1 is a clathrin and alpha-adaptin-binding protein involved in receptor-mediated endocytosis. Hum. Mol. Genet. 2001, 10, 1807–1817. [Google Scholar] [CrossRef]
- Metzler, M.; Gan, L.; Wong, T.P.; Liu, L.; Helm, J.; Liu, L.; Georgiou, J.; Wang, Y.; Bissada, N.; Cheng, K.; et al. NMDA receptor function and NMDA receptor-dependent phosphorylation of huntingtin is altered by the endocytic protein HIP1. J. Neurosci. 2007, 27, 2298–2308. [Google Scholar] [CrossRef]
- Mishra, S.K.; Agostinelli, N.R.; Brett, T.J.; Mizukami, I.; Ross, T.S.; Traub, L.M. Clathrin- and AP-2-binding sites in HIP1 uncover a general assembly role for endocytic accessory proteins. J. Biol. Chem. 2001, 276, 46230–46236. [Google Scholar] [CrossRef]
- Legendre-Guillemin, V.; Metzler, M.; Lemaire, J.F.; Philie, J.; Gan, L.; Hayden, M.R.; McPherson, P.S. Huntingtin interacting protein 1 (HIP1) regulates clathrin assembly through direct binding to the regulatory region of the clathrin light chain. J. Biol. Chem. 2005, 280, 6101–6108. [Google Scholar] [CrossRef]
- Senetar, M.A.; Foster, S.J.; McCann, R.O. Intrasteric inhibition mediates the interaction of the I/LWEQ module proteins Talin1, Talin2, Hip1, and Hip12 with actin. Biochemistry 2004, 43, 15418–15428. [Google Scholar] [CrossRef]
- Le Clainche, C.; Pauly, B.S.; Zhang, C.X.; Engqvist-Goldstein, A.E.; Cunningham, K.; Drubin, D.G. A Hip1R-cortactin complex negatively regulates actin assembly associated with endocytosis. EMBO J. 2007, 26, 1199–1210. [Google Scholar] [CrossRef]
- Engqvist-Goldstein, A.E.; Zhang, C.X.; Carreno, S.; Barroso, C.; Heuser, J.E.; Drubin, D.G. RNAi-mediated Hip1R silencing results in stable association between the endocytic machinery and the actin assembly machinery. Mol. Biol. Cell 2004, 15, 1666–1679. [Google Scholar] [CrossRef]
- Bradley, S.V.; Hyun, T.S.; Oravecz-Wilson, K.I.; Li, L.; Waldorff, E.I.; Ermilov, A.N.; Goldstein, S.A.; Zhang, C.X.; Drubin, D.G.; Varela, K.; et al. Degenerative phenotypes caused by the combined deficiency of murine HIP1 and HIP1r are rescued by human HIP1. Hum. Mol. Genet. 2007, 16, 1279–1292. [Google Scholar] [CrossRef]
- Parker, J.A.; Metzler, M.; Georgiou, J.; Mage, M.; Roder, J.C.; Rose, A.M.; Hayden, M.R.; Neri, C. Huntingtin-interacting protein 1 influences worm and mouse presynaptic function and protects Caenorhabditis elegans neurons against mutant polyglutamine toxicity. J. Neurosci. 2007, 27, 11056–11064. [Google Scholar] [CrossRef]
- Rao, D.S.; Chang, J.C.; Kumar, P.D.; Mizukami, I.; Smithson, G.M.; Bradley, S.V.; Parlow, A.F.; Ross, T.S. Huntingtin interacting protein 1 Is a clathrin coat binding protein required for differentiation of late spermatogenic progenitors. Mol. Cell Biol. 2001, 21, 7796–7806. [Google Scholar] [CrossRef]
- Khatchadourian, K.; Smith, C.E.; Metzler, M.; Gregory, M.; Hayden, M.R.; Cyr, D.G.; Hermo, L. Structural abnormalities in spermatids together with reduced sperm counts and motility underlie the reproductive defect in HIP1-/- mice. Mol. Reprod Dev. 2007, 74, 341–359. [Google Scholar] [CrossRef]
- Oravecz-Wilson, K.I.; Kiel, M.J.; Li, L.; Rao, D.S.; Saint-Dic, D.; Kumar, P.D.; Provot, M.M.; Hankenson, K.D.; Reddy, V.N.; Lieberman, A.P.; et al. Huntingtin Interacting Protein 1 mutations lead to abnormal hematopoiesis, spinal defects and cataracts. Hum. Mol. Genet. 2004, 13, 851–867. [Google Scholar] [CrossRef]
- Liu, Z.; Demitrack, E.S.; Keeley, T.M.; Eaton, K.A.; El-Zaatari, M.; Merchant, J.L.; Samuelson, L.C. IFNgamma contributes to the development of gastric epithelial cell metaplasia in Huntingtin interacting protein 1 related (Hip1r)-deficient mice. Lab. Investig. 2012, 92, 1045–1057. [Google Scholar] [CrossRef]
- Wijayatunge, R.; Holmstrom, S.R.; Foley, S.B.; Mgbemena, V.E.; Bhargava, V.; Perez, G.L.; McCrum, K.; Ross, T.S. Deficiency of the Endocytic Protein Hip1 Leads to Decreased Gdpd3 Expression, Low Phosphocholine, and Kypholordosis. Mol. Cell Biol. 2018, 38. [Google Scholar] [CrossRef]
- Laragione, T.; Brenner, M.; Lahiri, A.; Gao, E.; Harris, C.; Gulko, P.S. Huntingtin-interacting protein 1 (HIP1) regulates arthritis severity and synovial fibroblast invasiveness by altering PDGFR and Rac1 signalling. Ann. Rheum Dis. 2018, 77, 1627–1635. [Google Scholar] [CrossRef]
- Bradley, S.V.; Oravecz-Wilson, K.I.; Bougeard, G.; Mizukami, I.; Li, L.; Munaco, A.J.; Sreekumar, A.; Corradetti, M.N.; Chinnaiyan, A.M.; Sanda, M.G.; et al. Serum antibodies to huntingtin interacting protein-1: A new blood test for prostate cancer. Cancer Res. 2005, 65, 4126–4133. [Google Scholar] [CrossRef]
- Bradley, S.V.; Smith, M.R.; Hyun, T.S.; Lucas, P.C.; Li, L.; Antonuk, D.; Joshi, I.; Jin, F.; Ross, T.S. Aberrant Huntingtin interacting protein 1 in lymphoid malignancies. Cancer Res. 2007, 67, 8923–8931. [Google Scholar] [CrossRef]
- Hyun, T.S.; Ross, T.S. HIP1: Trafficking roles and regulation of tumorigenesis. Trends Mol. Med. 2004, 10, 194–199. [Google Scholar] [CrossRef]
- Rao, D.S.; Hyun, T.S.; Kumar, P.D.; Mizukami, I.F.; Rubin, M.A.; Lucas, P.C.; Sanda, M.G.; Ross, T.S. Huntingtin-interacting protein 1 is overexpressed in prostate and colon cancer and is critical for cellular survival. J. Clin. Investig. 2002, 110, 351–360. [Google Scholar] [CrossRef]
- Bradley, S.V.; Holland, E.C.; Liu, G.Y.; Thomas, D.; Hyun, T.S.; Ross, T.S. Huntingtin interacting protein 1 is a novel brain tumor marker that associates with epidermal growth factor receptor. Cancer Res. 2007, 67, 3609–3615. [Google Scholar] [CrossRef]
- Choi, S.A.; Kim, S.J.; Chung, K.C. Huntingtin-interacting protein 1-mediated neuronal cell death occurs through intrinsic apoptotic pathways and mitochondrial alterations. FEBS Lett. 2006, 580, 5275–5282. [Google Scholar] [CrossRef]
- Mills, I.G.; Gaughan, L.; Robson, C.; Ross, T.; McCracken, S.; Kelly, J.; Neal, D.E. Huntingtin interacting protein 1 modulates the transcriptional activity of nuclear hormone receptors. J. Cell Biol. 2005, 170, 191–200. [Google Scholar] [CrossRef]
- Hong, M.; Kim, R.N.; Song, J.Y.; Choi, S.J.; Oh, E.; Lira, M.E.; Mao, M.; Takeuchi, K.; Han, J.; Kim, J.; et al. HIP1-ALK, a novel fusion protein identified in lung adenocarcinoma. J. Thorac. Oncol. 2014, 9, 419–422. [Google Scholar] [CrossRef]
- Ou, S.H.; Klempner, S.J.; Greenbowe, J.R.; Azada, M.; Schrock, A.B.; Ali, S.M.; Ross, J.S.; Stephens, P.J.; Miller, V.A. Identification of a novel HIP1-ALK fusion variant in Non-Small-Cell Lung Cancer (NSCLC) and discovery of ALK I1171 (I1171N/S) mutations in two ALK-rearranged NSCLC patients with resistance to Alectinib. J. Thorac. Oncol. 2014, 9, 1821–1825. [Google Scholar] [CrossRef]
- Ross, T.S.; Bernard, O.A.; Berger, R.; Gilliland, D.G. Fusion of Huntingtin interacting protein 1 to platelet-derived growth factor beta receptor (PDGFbetaR) in chronic myelomonocytic leukemia with t(5;7)(q33;q11.2). Blood 1998, 91, 4419–4426. [Google Scholar] [CrossRef]
- Ramocki, M.B.; Bartnik, M.; Szafranski, P.; Kolodziejska, K.E.; Xia, Z.; Bravo, J.; Miller, G.S.; Rodriguez, D.L.; Williams, C.A.; Bader, P.I.; et al. Recurrent distal 7q11.23 deletion including HIP1 and YWHAG identified in patients with intellectual disabilities, epilepsy, and neurobehavioral problems. Am. J. Hum. Genet. 2010, 87, 857–865. [Google Scholar] [CrossRef]
- Wanker, E.E.; Rovira, C.; Scherzinger, E.; Hasenbank, R.; Walter, S.; Tait, D.; Colicelli, J.; Lehrach, H. HIP-I: A huntingtin interacting protein isolated by the yeast two-hybrid system. Hum. Mol. Genet. 1997, 6, 487–495. [Google Scholar] [CrossRef]
- Fazioli, F.; Minichiello, L.; Matoskova, B.; Wong, W.T.; Di Fiore, P.P. eps15, a novel tyrosine kinase substrate, exhibits transforming activity. Mol. Cell Biol. 1993, 13, 5814–5828. [Google Scholar] [CrossRef]
- Milesi, C.; Alberici, P.; Pozzi, B.; Oldani, A.; Beznoussenko, G.V.; Raimondi, A.; Soppo, B.E.; Amodio, S.; Caldieri, G.; Malabarba, M.G.; et al. Redundant and nonredundant organismal functions of EPS15 and EPS15L1. Life Sci. Alliance 2019, 2. [Google Scholar] [CrossRef]
- Polo, S.; Sigismund, S.; Faretta, M.; Guidi, M.; Capua, M.R.; Bossi, G.; Chen, H.; De Camilli, P.; Di Fiore, P.P. A single motif responsible for ubiquitin recognition and monoubiquitination in endocytic proteins. Nature 2002, 416, 451–455. [Google Scholar] [CrossRef]
- Confalonieri, S.; Di Fiore, P.P. The Eps15 homology (EH) domain. FEBS Lett. 2002, 513, 24–29. [Google Scholar] [CrossRef]
- Wong, W.T.; Schumacher, C.; Salcini, A.E.; Romano, A.; Castagnino, P.; Pelicci, P.G.; Di Fiore, P.P. A protein-binding domain, EH, identified in the receptor tyrosine kinase substrate Eps15 and conserved in evolution. Proc. Natl. Acad. Sci. USA 1995, 92, 9530–9534. [Google Scholar] [CrossRef] [PubMed]
- Mayer, B.J. Endocytosis: EH domains lend a hand. Curr. Biol. 1999, 9, R70–R73. [Google Scholar] [CrossRef]
- Polo, S.; Confalonieri, S.; Salcini, A.E.; Di Fiore, P.P. EH and UIM: Endocytosis and more. Sci. STKE 2003, 2003, re17. [Google Scholar] [CrossRef]
- Salcini, A.E.; Confalonieri, S.; Doria, M.; Santolini, E.; Tassi, E.; Minenkova, O.; Cesareni, G.; Pelicci, P.G.; Di Fiore, P.P. Binding specificity and in vivo targets of the EH domain, a novel protein-protein interaction module. Genes Dev. 1997, 11, 2239–2249. [Google Scholar] [CrossRef]
- Tebar, F.; Confalonieri, S.; Carter, R.E.; Di Fiore, P.P.; Sorkin, A. Eps15 is constitutively oligomerized due to homophilic interaction of its coiled-coil region. J. Biol. Chem. 1997, 272, 15413–15418. [Google Scholar] [CrossRef]
- Sengar, A.S.; Wang, W.; Bishay, J.; Cohen, S.; Egan, S.E. The EH and SH3 domain Ese proteins regulate endocytosis by linking to dynamin and Eps15. EMBO J. 1999, 18, 1159–1171. [Google Scholar] [CrossRef]
- Iannolo, G.; Salcini, A.E.; Gaidarov, I.; Goodman, O.B., Jr.; Baulida, J.; Carpenter, G.; Pelicci, P.G.; Di Fiore, P.P.; Keen, J.H. Mapping of the molecular determinants involved in the interaction between eps15 and AP-2. Cancer Res. 1997, 57, 240–245. [Google Scholar]
- Evergren, E.; Cobbe, N.; McMahon, H.T. Eps15R and clathrin regulate EphB2-mediated cell repulsion. Traffic 2018, 19, 44–57. [Google Scholar] [CrossRef]
- Salcini, A.E.; Chen, H.; Iannolo, G.; De Camilli, P.; Di Fiore, P.P. Epidermal growth factor pathway substrate 15, Eps15. Int. J. BioChem. Cell Biol. 1999, 31, 805–809. [Google Scholar] [CrossRef]
- Torrisi, M.R.; Lotti, L.V.; Belleudi, F.; Gradini, R.; Salcini, A.E.; Confalonieri, S.; Pelicci, P.G.; Di Fiore, P.P. Eps15 is recruited to the plasma membrane upon epidermal growth factor receptor activation and localizes to components of the endocytic pathway during receptor internalization. Mol. Biol. Cell 1999, 10, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Carbone, R.; Fre, S.; Iannolo, G.; Belleudi, F.; Mancini, P.; Pelicci, P.G.; Torrisi, M.R.; Di Fiore, P.P. eps15 and eps15R are essential components of the endocytic pathway. Cancer Res. 1997, 57, 5498–5504. [Google Scholar] [PubMed]
- de Melker, A.A.; van der Horst, G.; Borst, J. c-Cbl directs EGF receptors into an endocytic pathway that involves the ubiquitin-interacting motif of Eps15. J. Cell Sci. 2004, 117, 5001–5012. [Google Scholar] [CrossRef] [PubMed]
- Girao, H.; Catarino, S.; Pereira, P. Eps15 interacts with ubiquitinated Cx43 and mediates its internalization. Exp. Cell Res. 2009, 315, 3587–3597. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Man, H.Y. Endocytic adaptor epidermal growth factor receptor substrate 15 (Eps15) is involved in the trafficking of ubiquitinated alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors. J. Biol. Chem. 2014, 289, 24652–24664. [Google Scholar] [CrossRef]
- Parachoniak, C.A.; Park, M. Distinct recruitment of Eps15 via Its coiled-coil domain is required for efficient down-regulation of the met receptor tyrosine kinase. J. Biol. Chem. 2009, 284, 8382–8394. [Google Scholar] [CrossRef]
- Chi, S.; Cao, H.; Chen, J.; McNiven, M.A. Eps15 mediates vesicle trafficking from the trans-Golgi network via an interaction with the clathrin adaptor AP-1. Mol. Biol. Cell 2008, 19, 3564–3575. [Google Scholar] [CrossRef]
- Kent, H.M.; McMahon, H.T.; Evans, P.R.; Benmerah, A.; Owen, D.J. Gamma-adaptin appendage domain: Structure and binding site for Eps15 and gamma-synergin. Structure 2002, 10, 1139–1148. [Google Scholar] [CrossRef]
- Poupon, V.; Polo, S.; Vecchi, M.; Martin, G.; Dautry-Varsat, A.; Cerf-Bensussan, N.; Di Fiore, P.P.; Benmerah, A. Differential nucleocytoplasmic trafficking between the related endocytic proteins Eps15 and Eps15R. J. Biol. Chem. 2002, 277, 8941–8948. [Google Scholar] [CrossRef]
- Salcini, A.E.; Hilliard, M.A.; Croce, A.; Arbucci, S.; Luzzi, P.; Tacchetti, C.; Daniell, L.; De Camilli, P.; Pelicci, P.G.; Di Fiore, P.P.; et al. The Eps15 C. elegans homologue EHS-1 is implicated in synaptic vesicle recycling. Nat. Cell Biol. 2001, 3, 755–760. [Google Scholar] [CrossRef]
- Koh, T.W.; Korolchuk, V.I.; Wairkar, Y.P.; Jiao, W.; Evergren, E.; Pan, H.; Zhou, Y.; Venken, K.J.; Shupliakov, O.; Robinson, I.M.; et al. Eps15 and Dap160 control synaptic vesicle membrane retrieval and synapse development. J. Cell Biol. 2007, 178, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, A.; Ramagiri, S.; Rikhy, R. Drosophila homologue of Eps15 is essential for synaptic vesicle recycling. Exp. Cell Res. 2006, 312, 2288–2298. [Google Scholar] [CrossRef]
- Seiler, C.; Gebhart, N.; Zhang, Y.; Shinton, S.A.; Li, Y.S.; Ross, N.L.; Liu, X.; Li, Q.; Bilbee, A.N.; Varshney, G.K.; et al. Mutagenesis Screen Identifies agtpbp1 and eps15L1 as Essential for T lymphocyte Development in Zebrafish. PLoS ONE 2015, 10, e0131908. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, B.; Amodio, S.; Lucano, C.; Sciullo, A.; Ronzoni, S.; Castelletti, D.; Adler, T.; Treise, I.; Betsholtz, I.H.; Rathkolb, B.; et al. The endocytic adaptor Eps15 controls marginal zone B cell numbers. PLoS ONE 2012, 7, e50818. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Liu, Z.; Zhang, S. Over-expression of EPS15 is a favorable prognostic factor in breast cancer. Mol. Biosyst. 2015, 11, 2978–2985. [Google Scholar] [CrossRef]
- Bens, S.; Haake, A.; Tonnies, H.; Vater, I.; Stephani, U.; Holterhus, P.M.; Siebert, R.; Caliebe, A. A de novo 1.1Mb microdeletion of chromosome 19p13.11 provides indirect evidence for EPS15L1 to be a strong candidate for split hand split foot malformation. Eur. J. Med. Genet. 2011, 54, e501–e504. [Google Scholar] [CrossRef]
- Umair, M.; Ullah, A.; Abbas, S.; Ahmad, F.; Basit, S.; Ahmad, W. First direct evidence of involvement of a homozygous loss-of-function variant in the EPS15L1 gene underlying split-hand/split-foot malformation. Clin. Genet. 2018, 93, 699–702. [Google Scholar] [CrossRef]
- Spradling, K.D.; Burke, T.J.; Lohi, J.; Pilcher, B.K. Cloning and initial characterization of human epsin 3, a novel matrix-induced keratinocyte specific transcript. J. Investig. Dermatol. 2000, 115, 332. [Google Scholar] [CrossRef]
- Ko, G.; Paradise, S.; Chen, H.; Graham, M.; Vecchi, M.; Bianchi, F.; Cremona, O.; Di Fiore, P.P.; De Camilli, P. Selective high-level expression of epsin 3 in gastric parietal cells, where it is localized at endocytic sites of apical canaliculi. Proc. Natl. Acad. Sci. USA 2010, 107, 21511–21516. [Google Scholar] [CrossRef]
- Chen, H.; Fre, S.; Slepnev, V.I.; Capua, M.R.; Takei, K.; Butler, M.H.; Di Fiore, P.P.; De Camilli, P. Epsin is an EH-domain-binding protein implicated in clathrin-mediated endocytosis. Nature 1998, 394, 793–797. [Google Scholar] [CrossRef]
- Drake, M.T.; Downs, M.A.; Traub, L.M. Epsin binds to clathrin by associating directly with the clathrin-terminal domain. Evidence for cooperative binding through two discrete sites. J. Biol. Chem. 2000, 275, 6479–6489. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.C.; Katzmann, D.J.; Schnell, J.D.; Sutanto, M.; Emr, S.D.; Hicke, L. Epsins and Vps27p/Hrs contain ubiquitin-binding domains that function in receptor endocytosis. Nat. Cell Biol. 2002, 4, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Kalthoff, C.; Alves, J.; Urbanke, C.; Knorr, R.; Ungewickell, E.J. Unusual structural organization of the endocytic proteins AP180 and epsin 1. J. Biol. Chem. 2002, 277, 8209–8216. [Google Scholar] [CrossRef] [PubMed]
- Ford, M.G.; Mills, I.G.; Peter, B.J.; Vallis, Y.; Praefcke, G.J.; Evans, P.R.; McMahon, H.T. Curvature of clathrin-coated pits driven by epsin. Nature 2002, 419, 361–366. [Google Scholar] [CrossRef]
- Chen, B.; Dores, M.R.; Grimsey, N.; Canto, I.; Barker, B.L.; Trejo, J. Adaptor protein complex-2 (AP-2) and epsin-1 mediate protease-activated receptor-1 internalization via phosphorylation- and ubiquitination-dependent sorting signals. J. Biol. Chem. 2011, 286, 40760–40770. [Google Scholar] [CrossRef]
- Kazazic, M.; Bertelsen, V.; Pedersen, K.W.; Vuong, T.T.; Grandal, M.V.; Rodland, M.S.; Traub, L.M.; Stang, E.; Madshus, I.H. Epsin 1 is involved in recruitment of ubiquitinated EGF receptors into clathrin-coated pits. Traffic 2009, 10, 235–245. [Google Scholar] [CrossRef]
- Boucrot, E.; Pick, A.; Camdere, G.; Liska, N.; Evergren, E.; McMahon, H.T.; Kozlov, M.M. Membrane fission is promoted by insertion of amphipathic helices and is restricted by crescent BAR domains. Cell 2012, 149, 124–136. [Google Scholar] [CrossRef]
- Messa, M.; Fernandez-Busnadiego, R.; Sun, E.W.; Chen, H.; Czapla, H.; Wrasman, K.; Wu, Y.; Ko, G.; Ross, T.; Wendland, B.; et al. Epsin deficiency impairs endocytosis by stalling the actin-dependent invagination of endocytic clathrin-coated pits. Elife 2014, 3, e03311. [Google Scholar] [CrossRef]
- Aguilar, R.C.; Longhi, S.A.; Shaw, J.D.; Yeh, L.Y.; Kim, S.; Schon, A.; Freire, E.; Hsu, A.; McCormick, W.K.; Watson, H.A.; et al. Epsin N-terminal homology domains perform an essential function regulating Cdc42 through binding Cdc42 GTPase-activating proteins. Proc. Natl. Acad. Sci. USA 2006, 103, 4116–4121. [Google Scholar] [CrossRef]
- Szymanska, M.; Fosdahl, A.M.; Raiborg, C.; Dietrich, M.; Liestol, K.; Stang, E.; Bertelsen, V. Interaction with epsin 1 regulates the constitutive clathrin-dependent internalization of ErbB3. Biochim. Biophys. Acta 2016, 1863, 1179–1188. [Google Scholar] [CrossRef]
- Cardano, M.; Zasso, J.; Ruggiero, L.; Di Giacomo, G.; Marcatili, M.; Cremona, O.; Conti, L. Epsins Regulate Mouse Embryonic Stem Cell Exit from Pluripotency and Neural Commitment by Controlling Notch Activation. Stem Cells Int. 2019, 2019, 4084351. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ko, G.; Zatti, A.; Di Giacomo, G.; Liu, L.; Raiteri, E.; Perucco, E.; Collesi, C.; Min, W.; Zeiss, C.; et al. Embryonic arrest at midgestation and disruption of Notch signaling produced by the absence of both epsin 1 and epsin 2 in mice. Proc. Natl. Acad. Sci. USA 2009, 106, 13838–13843. [Google Scholar] [CrossRef] [PubMed]
- Langridge, P.D.; Struhl, G. Epsin-Dependent Ligand Endocytosis Activates Notch by Force. Cell 2017, 171, 1383–1396.e1312. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Hansen, D.; Schedl, T.; Skeath, J.B. Epsin potentiates Notch pathway activity in Drosophila and C. elegans. Development 2004, 131, 5807–5815. [Google Scholar] [CrossRef]
- Wang, W.; Struhl, G. Drosophila Epsin mediates a select endocytic pathway that DSL ligands must enter to activate Notch. Development 2004, 131, 5367–5380. [Google Scholar] [CrossRef]
- Pasula, S.; Cai, X.; Dong, Y.; Messa, M.; McManus, J.; Chang, B.; Liu, X.; Zhu, H.; Mansat, R.S.; Yoon, S.J.; et al. Endothelial epsin deficiency decreases tumor growth by enhancing VEGF signaling. J. Clin. Investig. 2012, 122, 4424–4438. [Google Scholar] [CrossRef]
- Tessneer, K.L.; Pasula, S.; Cai, X.; Dong, Y.; McManus, J.; Liu, X.; Yu, L.; Hahn, S.; Chang, B.; Chen, Y.; et al. Genetic reduction of vascular endothelial growth factor receptor 2 rescues aberrant angiogenesis caused by epsin deficiency. Arterioscler Thromb Vasc. Biol. 2014, 34, 331–337. [Google Scholar] [CrossRef]
- Brophy, M.L.; Dong, Y.; Tao, H.; Yancey, P.G.; Song, K.; Zhang, K.; Wen, A.; Wu, H.; Lee, Y.; Malovichko, M.V.; et al. Myeloid-Specific Deletion of Epsins 1 and 2 Reduces Atherosclerosis by Preventing LRP-1 Downregulation. Circ. Res. 2019, 124, e6–e19. [Google Scholar] [CrossRef]
- Overstreet, E.; Chen, X.; Wendland, B.; Fischer, J.A. Either part of a Drosophila epsin protein, divided after the ENTH domain, functions in endocytosis of delta in the developing eye. Curr. Biol. 2003, 13, 854–860. [Google Scholar] [CrossRef]
- Wang, W.; Struhl, G. Distinct roles for Mind bomb, Neuralized and Epsin in mediating DSL endocytosis and signaling in Drosophila. Development 2005, 132, 2883–2894. [Google Scholar] [CrossRef]
- Dong, Y.; Wu, H.; Rahman, H.N.; Liu, Y.; Pasula, S.; Tessneer, K.L.; Cai, X.; Liu, X.; Chang, B.; McManus, J.; et al. Motif mimetic of epsin perturbs tumor growth and metastasis. J. Clin. Investig. 2016, 126, 1607. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Pasula, S.; Song, H.; Tessneer, K.L.; Dong, Y.; Hahn, S.; Yago, T.; Brophy, M.L.; Chang, B.; Cai, X.; et al. Temporal and spatial regulation of epsin abundance and VEGFR3 signaling are required for lymphatic valve formation and function. Sci. Signal. 2014, 7, ra97. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.; Tessneer, K.L.; McManus, J.; Liu, X.; Hahn, S.; Pasula, S.; Wu, H.; Song, H.; Chen, Y.; Cai, X.; et al. Epsin is required for Dishevelled stability and Wnt signalling activation in colon cancer development. Nat. Commun. 2015, 6, 6380. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zheng, Y. A requirement for epsin in mitotic membrane and spindle organization. J. Cell Biol. 2009, 186, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Han, L.; Zhang, J.; Liu, X.; Ma, R.; Hou, X.; Ge, J.; Wang, Q. Epsin2 promotes polarity establishment and meiotic division through activating Cdc42 in mouse oocyte. Oncotarget 2016, 7, 50927–50936. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mishra, S.K.; Watkins, S.C.; Traub, L.M. The autosomal recessive hypercholesterolemia (ARH) protein interfaces directly with the clathrin-coat machinery. Proc. Natl. Acad. Sci. USA 2002, 99, 16099–16104. [Google Scholar] [CrossRef]
- He, G.; Gupta, S.; Yi, M.; Michaely, P.; Hobbs, H.H.; Cohen, J.C. ARH is a modular adaptor protein that interacts with the LDL receptor, clathrin, and AP-2. J. Biol. Chem. 2002, 277, 44044–44049. [Google Scholar] [CrossRef]
- Santolini, E.; Puri, C.; Salcini, A.E.; Gagliani, M.C.; Pelicci, P.G.; Tacchetti, C.; Di Fiore, P.P. Numb is an endocytic protein. J. Cell Biol. 2000, 151, 1345–1352. [Google Scholar] [CrossRef]
- Jones, C.; Hammer, R.E.; Li, W.P.; Cohen, J.C.; Hobbs, H.H.; Herz, J. Normal sorting but defective endocytosis of the low density lipoprotein receptor in mice with autosomal recessive hypercholesterolemia. J. Biol. Chem. 2003, 278, 29024–29030. [Google Scholar] [CrossRef]
- Tao, W.; Moore, R.; Meng, Y.; Smith, E.R.; Xu, X.X. Endocytic adaptors Arh and Dab2 control homeostasis of circulatory cholesterol. J. Lipid Res. 2016, 57, 809–817. [Google Scholar] [CrossRef]
- Moore, R.; Cai, K.Q.; Tao, W.; Smith, E.R.; Xu, X.X. Differential requirement for Dab2 in the development of embryonic and extra-embryonic tissues. BMC Dev. Biol. 2013, 13, 39. [Google Scholar] [CrossRef] [PubMed]
- Nagai, M.; Meerloo, T.; Takeda, T.; Farquhar, M.G. The adaptor protein ARH escorts megalin to and through endosomes. Mol. Biol. Cell 2003, 14, 4984–4996. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, G.A.; Chakraborty, S.; Steinhauser, A.L.; Traub, L.M.; Madsen, M. AMN directs endocytosis of the intrinsic factor-vitamin B(12) receptor cubam by engaging ARH or Dab2. Traffic 2010, 11, 706–720. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Garuti, R.; Kim, B.Y.; Wade, J.B.; Welling, P.A. The ARH adaptor protein regulates endocytosis of the ROMK potassium secretory channel in mouse kidney. J. Clin. Investig. 2009, 119, 3278–3289. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, S.; Shah, M.; Nielsen, R.; Iino, N.; Ryan, J.J.; Zhou, H.; Farquhar, M.G. The endocytic adaptor protein ARH associates with motor and centrosomal proteins and is involved in centrosome assembly and cytokinesis. Mol. Biol. Cell 2008, 19, 2949–2961. [Google Scholar] [CrossRef]
- Sun, X.M.; Patel, D.D.; Acosta, J.C.; Gil, J.; Soutar, A.K. Premature senescence in cells from patients with autosomal recessive hypercholesterolemia (ARH): Evidence for a role for ARH in mitosis. Arterioscler Thromb Vasc. Biol. 2011, 31, 2270–2277. [Google Scholar] [CrossRef]
- Teckchandani, A.; Mulkearns, E.E.; Randolph, T.W.; Toida, N.; Cooper, J.A. The clathrin adaptor Dab2 recruits EH domain scaffold proteins to regulate integrin beta1 endocytosis. Mol. Biol. Cell 2012, 23, 2905–2916. [Google Scholar] [CrossRef]
- Morris, S.M.; Arden, S.D.; Roberts, R.C.; Kendrick-Jones, J.; Cooper, J.A.; Luzio, J.P.; Buss, F. Myosin VI binds to and localises with Dab2, potentially linking receptor-mediated endocytosis and the actin cytoskeleton. Traffic 2002, 3, 331–341. [Google Scholar] [CrossRef]
- Mishra, S.K.; Keyel, P.A.; Hawryluk, M.J.; Agostinelli, N.R.; Watkins, S.C.; Traub, L.M. Disabled-2 exhibits the properties of a cargo-selective endocytic clathrin adaptor. EMBO J. 2002, 21, 4915–4926. [Google Scholar] [CrossRef]
- Morris, S.M.; Cooper, J.A. Disabled-2 colocalizes with the LDLR in clathrin-coated pits and interacts with AP-2. Traffic 2001, 2, 111–123. [Google Scholar] [CrossRef]
- Gertler, F.B.; Bennett, R.L.; Clark, M.J.; Hoffmann, F.M. Drosophila abl tyrosine kinase in embryonic CNS axons: A role in axonogenesis is revealed through dosage-sensitive interactions with disabled. Cell 1989, 58, 103–113. [Google Scholar] [CrossRef]
- Fazili, Z.; Sun, W.; Mittelstaedt, S.; Cohen, C.; Xu, X.X. Disabled-2 inactivation is an early step in ovarian tumorigenicity. Oncogene 1999, 18, 3104–3113. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.X.; Yi, T.; Tang, B.; Lambeth, J.D. Disabled-2 (Dab2) is an SH3 domain-binding partner of Grb2. Oncogene 1998, 16, 1561–1569. [Google Scholar] [CrossRef] [PubMed]
- Howell, B.W.; Hawkes, R.; Soriano, P.; Cooper, J.A. Neuronal position in the developing brain is regulated by mouse disabled-1. Nature 1997, 389, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, M.; Rice, D.S.; D’Arcangelo, G.; Yoneshima, H.; Nakajima, K.; Mikoshiba, K.; Howell, B.W.; Cooper, J.A.; Goldowitz, D.; Curran, T. Scrambler and yotari disrupt the disabled gene and produce a reeler-like phenotype in mice. Nature 1997, 389, 730–733. [Google Scholar] [CrossRef]
- Morris, S.M.; Tallquist, M.D.; Rock, C.O.; Cooper, J.A. Dual roles for the Dab2 adaptor protein in embryonic development and kidney transport. EMBO J. 2002, 21, 1555–1564. [Google Scholar] [CrossRef]
- Jiang, Y.; He, X.; Howe, P.H. Disabled-2 (Dab2) inhibits Wnt/beta-catenin signalling by binding LRP6 and promoting its internalization through clathrin. EMBO J. 2012, 31, 2336–2349. [Google Scholar] [CrossRef]
- Cuitino, L.; Matute, R.; Retamal, C.; Bu, G.; Inestrosa, N.C.; Marzolo, M.P. ApoER2 is endocytosed by a clathrin-mediated process involving the adaptor protein Dab2 independent of its Rafts’ association. Traffic 2005, 6, 820–838. [Google Scholar] [CrossRef]
- Lao, Y.; Li, Y.; Hou, Y.; Chen, H.; Qiu, B.; Lin, W.; Sun, A.; Wei, H.; Jiang, Y.; He, F. Proteomic Analysis Reveals Dab2 Mediated Receptor Endocytosis Promotes Liver Sinusoidal Endothelial Cell Dedifferentiation. Sci. Rep. 2017, 7, 13456. [Google Scholar] [CrossRef]
- Hung, W.S.; Huang, C.L.; Fan, J.T.; Huang, D.Y.; Yeh, C.F.; Cheng, J.C.; Tseng, C.P. The endocytic adaptor protein Disabled-2 is required for cellular uptake of fibrinogen. Biochim. Biophys. Acta 2012, 1823, 1778–1788. [Google Scholar] [CrossRef]
- Calderwood, D.A.; Fujioka, Y.; de Pereda, J.M.; Garcia-Alvarez, B.; Nakamoto, T.; Margolis, B.; McGlade, C.J.; Liddington, R.C.; Ginsberg, M.H. Integrin beta cytoplasmic domain interactions with phosphotyrosine-binding domains: A structural prototype for diversity in integrin signaling. Proc. Natl. Acad. Sci. USA 2003, 100, 2272–2277. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Rab, A.; Tang, L.P.; Rowe, S.M.; Bebok, Z.; Collawn, J.F. Dab2 is a key regulator of endocytosis and post-endocytic trafficking of the cystic fibrosis transmembrane conductance regulator. BioChem. J. 2012, 441, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Cihil, K.M.; Ellinger, P.; Fellows, A.; Stolz, D.B.; Madden, D.R.; Swiatecka-Urban, A. Disabled-2 protein facilitates assembly polypeptide-2-independent recruitment of cystic fibrosis transmembrane conductance regulator to endocytic vesicles in polarized human airway epithelial cells. J. Biol. Chem. 2012, 287, 15087–15099. [Google Scholar] [CrossRef] [PubMed]
- Hocevar, B.A.; Mou, F.; Rennolds, J.L.; Morris, S.M.; Cooper, J.A.; Howe, P.H. Regulation of the Wnt signaling pathway by disabled-2 (Dab2). EMBO J. 2003, 22, 3084–3094. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Moore, R.; Smith, E.R.; Xu, X.X. Endocytosis and Physiology: Insights from Disabled-2 Deficient Mice. Front. Cell Dev. Biol. 2016, 4, 129. [Google Scholar] [CrossRef] [PubMed]
- Hocevar, B.A.; Prunier, C.; Howe, P.H. Disabled-2 (Dab2) mediates transforming growth factor beta (TGFbeta)-stimulated fibronectin synthesis through TGFbeta-activated kinase 1 and activation of the JNK pathway. J. Biol. Chem. 2005, 280, 25920–25927. [Google Scholar] [CrossRef]
- Hocevar, B.A.; Smine, A.; Xu, X.X.; Howe, P.H. The adaptor molecule Disabled-2 links the transforming growth factor beta receptors to the Smad pathway. EMBO J. 2001, 20, 2789–2801. [Google Scholar] [CrossRef]
- Finkielstein, C.V.; Capelluto, D.G. Disabled-2: A modular scaffold protein with multifaceted functions in signaling. Bioessays 2016, 38 (Suppl. 1), S45–S55. [Google Scholar] [CrossRef]
- Rula, M.E.; Cai, K.Q.; Moore, R.; Yang, D.H.; Staub, C.M.; Capo-Chichi, C.D.; Jablonski, S.A.; Howe, P.H.; Smith, E.R.; Xu, X.X. Cell autonomous sorting and surface positioning in the formation of primitive endoderm in embryoid bodies. Genesis 2007, 45, 327–338. [Google Scholar] [CrossRef]
- Yang, D.H.; Cai, K.Q.; Roland, I.H.; Smith, E.R.; Xu, X.X. Disabled-2 is an epithelial surface positioning gene. J. Biol. Chem. 2007, 282, 13114–13122. [Google Scholar] [CrossRef]
- Nagai, J.; Christensen, E.I.; Morris, S.M.; Willnow, T.E.; Cooper, J.A.; Nielsen, R. Mutually dependent localization of megalin and Dab2 in the renal proximal tubule. Am. J. Physiol. Renal Physiol. 2005, 289, F569–F576. [Google Scholar] [CrossRef] [PubMed]
- Mok, S.C.; Chan, W.Y.; Wong, K.K.; Cheung, K.K.; Lau, C.C.; Ng, S.W.; Baldini, A.; Colitti, C.V.; Rock, C.O.; Berkowitz, R.S. DOC-2, a candidate tumor suppressor gene in human epithelial ovarian cancer. Oncogene 1998, 16, 2381–2387. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.H.; Fazili, Z.; Smith, E.R.; Cai, K.Q.; Klein-Szanto, A.; Cohen, C.; Horowitz, I.R.; Xu, X.X. Disabled-2 heterozygous mice are predisposed to endometrial and ovarian tumorigenesis and exhibit sex-biased embryonic lethality in a p53-null background. Am. J. Pathol. 2006, 169, 258–267. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rhyu, M.S.; Jan, L.Y.; Jan, Y.N. Asymmetric distribution of numb protein during division of the sensory organ precursor cell confers distinct fates to daughter cells. Cell 1994, 76, 477–491. [Google Scholar] [CrossRef]
- Berdnik, D.; Torok, T.; Gonzalez-Gaitan, M.; Knoblich, J.A. The endocytic protein alpha-Adaptin is required for numb-mediated asymmetric cell division in Drosophila. Dev. Cell 2002, 3, 221–231. [Google Scholar] [CrossRef]
- Guo, M.; Jan, L.Y.; Jan, Y.N. Control of daughter cell fates during asymmetric division: Interaction of Numb and Notch. Neuron 1996, 17, 27–41. [Google Scholar] [CrossRef]
- Tong, X.; Zitserman, D.; Serebriiskii, I.; Andrake, M.; Dunbrack, R.; Roegiers, F. Numb independently antagonizes Sanpodo membrane targeting and Notch signaling in Drosophila sensory organ precursor cells. Mol. Biol. Cell 2010, 21, 802–810. [Google Scholar] [CrossRef]
- McGill, M.A.; Dho, S.E.; Weinmaster, G.; McGlade, C.J. Numb regulates post-endocytic trafficking and degradation of Notch1. J. Biol. Chem. 2009, 284, 26427–26438. [Google Scholar] [CrossRef]
- Shao, X.; Ding, Z.; Zhao, M.; Liu, K.; Sun, H.; Chen, J.; Liu, X.; Zhang, Y.; Hong, Y.; Li, H.; et al. Mammalian Numb protein antagonizes Notch by controlling postendocytic trafficking of the Notch ligand Delta-like 4. J. Biol. Chem. 2017, 292, 20628–20643. [Google Scholar] [CrossRef]
- Zobel, M.; Disanza, A.; Senic-Matuglia, F.; Franco, M.; Colaluca, I.N.; Confalonieri, S.; Bisi, S.; Barbieri, E.; Caldieri, G.; Sigismund, S.; et al. A NUMB-EFA6B-ARF6 recycling route controls apically restricted cell protrusions and mesenchymal motility. J. Cell Biol. 2018, 217, 3161–3182. [Google Scholar] [CrossRef]
- Smith, C.A.; Dho, S.E.; Donaldson, J.; Tepass, U.; McGlade, C.J. The cell fate determinant numb interacts with EHD/Rme-1 family proteins and has a role in endocytic recycling. Mol. Biol. Cell 2004, 15, 3698–3708. [Google Scholar] [CrossRef] [PubMed]
- Miao, L.; Li, J.; Li, J.; Lu, Y.; Shieh, D.; Mazurkiewicz, J.E.; Barroso, M.; Schwarz, J.J.; Xin, H.B.; Singer, H.A.; et al. Cardiomyocyte orientation modulated by the Numb family proteins-N-cadherin axis is essential for ventricular wall morphogenesis. Proc. Natl. Acad. Sci. USA 2019, 116, 15560–15569. [Google Scholar] [CrossRef] [PubMed]
- Kyriazis, G.A.; Wei, Z.; Vandermey, M.; Jo, D.G.; Xin, O.; Mattson, M.P.; Chan, S.L. Numb endocytic adapter proteins regulate the transport and processing of the amyloid precursor protein in an isoform-dependent manner: Implications for Alzheimer disease pathogenesis. J. Biol. Chem. 2008, 283, 25492–25502. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Li, J. Numb family proteins: Novel players in cardiac morphogenesis and cardiac progenitor cell differentiation. BioMol. Concepts 2015, 6, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Kaibuchi, K. Numb controls integrin endocytosis for directional cell migration with aPKC and PAR-3. Dev. Cell 2007, 13, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Watanabe, T.; Wang, S.; Kakeno, M.; Matsuzawa, K.; Matsui, T.; Yokoi, K.; Murase, K.; Sugiyama, I.; Ozawa, M.; et al. Numb controls E-cadherin endocytosis through p120 catenin with aPKC. Mol. Biol. Cell 2011, 22, 3103–3119. [Google Scholar] [CrossRef]
- Li, P.S.; Fu, Z.Y.; Zhang, Y.Y.; Zhang, J.H.; Xu, C.Q.; Ma, Y.T.; Li, B.L.; Song, B.L. The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1. Nat. Med. 2014, 20, 80–86. [Google Scholar] [CrossRef]
- Su, J.F.; Wei, J.; Li, P.S.; Miao, H.H.; Ma, Y.C.; Qu, Y.X.; Xu, J.; Qin, J.; Li, B.L.; Song, B.L.; et al. Numb directs the subcellular localization of EAAT3 through binding the YxNxxF motif. J. Cell Sci. 2016, 129, 3104–3114. [Google Scholar] [CrossRef]
- Zhou, L.; Yang, D.; Wang, D.J.; Xie, Y.J.; Zhou, J.H.; Zhou, L.; Huang, H.; Han, S.; Shao, C.Y.; Li, H.S.; et al. Numb deficiency in cerebellar Purkinje cells impairs synaptic expression of metabotropic glutamate receptor and motor coordination. Proc. Natl. Acad. Sci. USA 2015, 112, 15474–15479. [Google Scholar] [CrossRef]
- Wei, R.; Liu, X.; Voss, C.; Qin, W.; Dagnino, L.; Li, L.; Vigny, M.; Li, S.S. NUMB regulates the endocytosis and activity of the anaplastic lymphoma kinase in an isoform-specific manner. J. Mol. Cell Biol. 2019. [Google Scholar] [CrossRef]
- Ferent, J.; Giguere, F.; Jolicoeur, C.; Morin, S.; Michaud, J.F.; Makihara, S.; Yam, P.T.; Cayouette, M.; Charron, F. Boc Acts via Numb as a Shh-Dependent Endocytic Platform for Ptch1 Internalization and Shh-Mediated Axon Guidance. Neuron 2019, 102, 1157–1171.e1155. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Jiang, M.M.; Schonemann, M.D.; Meneses, J.J.; Pedersen, R.A.; Jan, L.Y.; Jan, Y.N. Mouse numb is an essential gene involved in cortical neurogenesis. Proc. Natl. Acad. Sci. USA 2000, 97, 6844–6849. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.H.; Zou, K.; Hwang, J.K.; Jan, Y.N.; Zhong, W. Progenitor cell maintenance requires numb and numblike during mouse neurogenesis. Nature 2002, 419, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Hong, Y.; Zhu, M.; Zhou, L.; Li, H.; Li, H. Deletion of Numb/Numblike in glutamatergic neurons leads to anxiety-like behavior in mice. Brain Res. 2017, 1665, 36–49. [Google Scholar] [CrossRef]
- Colaluca, I.N.; Tosoni, D.; Nuciforo, P.; Senic-Matuglia, F.; Galimberti, V.; Viale, G.; Pece, S.; Di Fiore, P.P. NUMB controls p53 tumour suppressor activity. Nature 2008, 451, 76–80. [Google Scholar] [CrossRef]
- Colaluca, I.N.; Basile, A.; Freiburger, L.; D’Uva, V.; Disalvatore, D.; Vecchi, M.; Confalonieri, S.; Tosoni, D.; Cecatiello, V.; Malabarba, M.G.; et al. A Numb-Mdm2 fuzzy complex reveals an isoform-specific involvement of Numb in breast cancer. J. Cell Biol. 2018, 217, 745–762. [Google Scholar] [CrossRef]
- Gurevich, V.V.; Gurevich, E.V. Plethora of functions packed into 45 kDa arrestins: Biological implications and possible therapeutic strategies. Cell Mol. Life Sci. 2019. [Google Scholar] [CrossRef]
- Gurevich, V.V.; Gurevich, E.V. Arrestin mutations: Some cause diseases, others promise cure. Prog. Mol. Biol. Transl. Sci. 2019, 161, 29–45. [Google Scholar]
- Goodman, O.B., Jr.; Krupnick, J.G.; Santini, F.; Gurevich, V.V.; Penn, R.B.; Gagnon, A.W.; Keen, J.H.; Benovic, J.L. Beta-arrestin acts as a clathrin adaptor in endocytosis of the beta2-adrenergic receptor. Nature 1996, 383, 447–450. [Google Scholar] [CrossRef]
- Gurevich, V.V.; Gurevich, E.V. The structural basis of the arrestin binding to GPCRs. Mol. Cell Endocrinol. 2019, 484, 34–41. [Google Scholar] [CrossRef]
- Laporte, S.A.; Oakley, R.H.; Holt, J.A.; Barak, L.S.; Caron, M.G. The interaction of beta-arrestin with the AP-2 adaptor is required for the clustering of beta 2-adrenergic receptor into clathrin-coated pits. J. Biol. Chem. 2000, 275, 23120–23126. [Google Scholar] [CrossRef] [PubMed]
- Laporte, S.A.; Oakley, R.H.; Zhang, J.; Holt, J.A.; Ferguson, S.S.; Caron, M.G.; Barak, L.S. The beta2-adrenergic receptor/betaarrestin complex recruits the clathrin adaptor AP-2 during endocytosis. Proc. Natl. Acad. Sci. USA 1999, 96, 3712–3717. [Google Scholar] [CrossRef] [PubMed]
- Krupnick, J.G.; Goodman, O.B., Jr.; Keen, J.H.; Benovic, J.L. Arrestin/clathrin interaction. Localization of the clathrin binding domain of nonvisual arrestins to the carboxy terminus. J. Biol. Chem. 1997, 272, 15011–15016. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Dodd, R.L.; Makino, C.L.; Simon, M.I.; Baylor, D.A.; Chen, J. Prolonged photoresponses in transgenic mouse rods lacking arrestin. Nature 1997, 389, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Bohn, L.M.; Lefkowitz, R.J.; Gainetdinov, R.R.; Peppel, K.; Caron, M.G.; Lin, F.T. Enhanced morphine analgesia in mice lacking beta-arrestin 2. Science 1999, 286, 2495–2498. [Google Scholar] [CrossRef] [PubMed]
- Conner, D.A.; Mathier, M.A.; Mortensen, R.M.; Christe, M.; Vatner, S.F.; Seidman, C.E.; Seidman, J.G. beta-Arrestin1 knockout mice appear normal but demonstrate altered cardiac responses to beta-adrenergic stimulation. Circ. Res. 1997, 81, 1021–1026. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, X.; Zhang, Y.; Zhao, J. Loss of betaarrestin1 and betaarrestin2 contributes to pulmonary hypoplasia and neonatal lethality in mice. Dev. Biol. 2010, 339, 407–417. [Google Scholar] [CrossRef]
- Kohout, T.A.; Lin, F.S.; Perry, S.J.; Conner, D.A.; Lefkowitz, R.J. beta-Arrestin 1 and 2 differentially regulate heptahelical receptor signaling and trafficking. Proc. Natl. Acad. Sci. USA 2001, 98, 1601–1606. [Google Scholar] [CrossRef]
- Fuchs, S.; Nakazawa, M.; Maw, M.; Tamai, M.; Oguchi, Y.; Gal, A. A homozygous 1-base pair deletion in the arrestin gene is a frequent cause of Oguchi disease in Japanese. Nat. Genet. 1995, 10, 360–362. [Google Scholar] [CrossRef]
- Nakazawa, M.; Wada, Y.; Tamai, M. Arrestin gene mutations in autosomal recessive retinitis pigmentosa. Arch. OphthalMol. 1998, 116, 498–501. [Google Scholar] [CrossRef]
- Bogerd, H.P.; Fridell, R.A.; Madore, S.; Cullen, B.R. Identification of a novel cellular cofactor for the Rev/Rex class of retroviral regulatory proteins. Cell 1995, 82, 485–494. [Google Scholar] [CrossRef]
- Fritz, C.C.; Zapp, M.L.; Green, M.R. A human nucleoporin-like protein that specifically interacts with HIV Rev. Nature 1995, 376, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Stutz, F.; Neville, M.; Rosbash, M. Identification of a novel nuclear pore-associated protein as a functional target of the HIV-1 Rev protein in yeast. Cell 1995, 82, 495–506. [Google Scholar] [CrossRef][Green Version]
- Doria, M.; Salcini, A.E.; Colombo, E.; Parslow, T.G.; Pelicci, P.G.; Di Fiore, P.P. The eps15 homology (EH) domain-based interaction between eps15 and hrb connects the molecular machinery of endocytosis to that of nucleocytosolic transport. J. Cell Biol. 1999, 147, 1379–1384. [Google Scholar] [CrossRef]
- Landi, A.; Timermans, C.G.; Naessens, E.; Vanderstraeten, H.; Stove, V.; Verhasselt, B. The human immunodeficiency virus (HIV) Rev-binding protein (HRB) is a co-factor for HIV-1 Nef-mediated CD4 downregulation. J. Gen. Virol. 2016, 97, 778–785. [Google Scholar] [CrossRef]
- Kang-Decker, N.; Mantchev, G.T.; Juneja, S.C.; McNiven, M.A.; van Deursen, J.M. Lack of acrosome formation in Hrb-deficient mice. Science 2001, 294, 1531–1533. [Google Scholar] [CrossRef]
- Dunlevy, J.R.; Berryhill, B.L.; Vergnes, J.P.; SundarRaj, N.; Hassell, J.R. Cloning, chromosomal localization, and characterization of cDNA from a novel gene, SH3BP4, expressed by human corneal fibroblasts. Genomics 1999, 62, 519–524. [Google Scholar] [CrossRef]
- Francavilla, C.; Rigbolt, K.T.; Emdal, K.B.; Carraro, G.; Vernet, E.; Bekker-Jensen, D.B.; Streicher, W.; Wikstrom, M.; Sundstrom, M.; Bellusci, S.; et al. Functional proteomics defines the molecular switch underlying FGF receptor trafficking and cellular outputs. Mol. Cell 2013, 51, 707–722. [Google Scholar] [CrossRef]
- Antas, P.; Novellasdemunt, L.; Kucharska, A.; Massie, I.; Carvalho, J.; Oukrif, D.; Nye, E.; Novelli, M.; Li, V.S.W. SH3BP4 Regulates Intestinal Stem Cells and Tumorigenesis by Modulating beta-Catenin Nuclear Localization. Cell Rep. 2019, 26, 2266–2273.e2264. [Google Scholar] [CrossRef]
- Kim, Y.M.; Stone, M.; Hwang, T.H.; Kim, Y.G.; Dunlevy, J.R.; Griffin, T.J.; Kim, D.H. SH3BP4 is a negative regulator of amino acid-Rag GTPase-mTORC1 signaling. Mol. Cell 2012, 46, 833–846. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, T.R.; Cho, E.G. SH3BP4, a novel pigmentation gene, is inversely regulated by miR-125b and MITF. Exp Mol. Med. 2017, 49, e367. [Google Scholar] [CrossRef] [PubMed]
- Pacault, M.; Nizon, M.; Pichon, O.; Vincent, M.; Le Caignec, C.; Isidor, B. A de novo 2q37.2 deletion encompassing AGAP1 and SH3BP4 in a patient with autism and intellectual disability. Eur. J. Med. Genet. 2018. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, H.; Walther, W.; Zincke, F.; Kobelt, D.; Imbastari, F.; Erdem, M.; Kortum, B.; Dahlmann, M.; Stein, U. MACC1-the first decade of a key metastasis molecule from gene discovery to clinical translation. Cancer Metastasis Rev. 2018, 37, 805–820. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, Y.; Ozawa, M. Increased internalization of p120-uncoupled E-cadherin and a requirement for a dileucine motif in the cytoplasmic domain for endocytosis of the protein. J. Biol. Chem. 2007, 282, 11540–11548. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.E.; Cooper, J.A. The adaptor protein Dab2 sorts LDL receptors into coated pits independently of AP-2 and ARH. J. Cell Sci. 2006, 119, 4235–4246. [Google Scholar] [CrossRef]
- Chu, H.C.; Tseng, W.L.; Lee, H.Y.; Cheng, J.C.; Chang, S.S.; Yung, B.Y.; Tseng, C.P. Distinct effects of Disabled-2 on transferrin uptake in different cell types and culture conditions. Cell Biol. Int. 2014, 38, 1252–1259. [Google Scholar] [CrossRef]
- Chaineau, M.; Danglot, L.; Proux-Gillardeaux, V.; Galli, T. Role of HRB in clathrin-dependent endocytosis. J. Biol. Chem. 2008, 283, 34365–34373. [Google Scholar] [CrossRef]
- De Franceschi, N.; Arjonen, A.; Elkhatib, N.; Denessiouk, K.; Wrobel, A.G.; Wilson, T.A.; Pouwels, J.; Montagnac, G.; Owen, D.J.; Ivaska, J. Selective integrin endocytosis is driven by interactions between the integrin alpha-chain and AP2. Nat. Struct. Mol. Biol. 2016, 23, 172–179. [Google Scholar] [CrossRef]
- Heuser, J. Three-dimensional visualization of coated vesicle formation in fibroblasts. J. Cell Biol. 1980, 84, 560–583. [Google Scholar] [CrossRef]
- Lock, J.G.; Baschieri, F.; Jones, M.C.; Humphries, J.D.; Montagnac, G.; Stromblad, S.; Humphries, M.J. Clathrin-containing adhesion complexes. J. Cell Biol. 2019, 218, 2086–2095. [Google Scholar] [CrossRef]
- Grove, J.; Metcalf, D.J.; Knight, A.E.; Wavre-Shapton, S.T.; Sun, T.; Protonotarios, E.D.; Griffin, L.D.; Lippincott-Schwartz, J.; Marsh, M. Flat clathrin lattices: Stable features of the plasma membrane. Mol. Biol. Cell 2014, 25, 3581–3594. [Google Scholar] [CrossRef] [PubMed]
- Zuidema, A.; Wang, W.; Kreft, M.; Te Molder, L.; Hoekman, L.; Bleijerveld, O.B.; Nahidiazar, L.; Janssen, H.; Sonnenberg, A. Mechanisms of integrin alphaVbeta5 clustering in flat clathrin lattices. J. Cell Sci. 2018, 131. [Google Scholar] [CrossRef] [PubMed]
- Lock, J.G.; Jones, M.C.; Askari, J.A.; Gong, X.; Oddone, A.; Olofsson, H.; Goransson, S.; Lakadamyali, M.; Humphries, M.J.; Stromblad, S. Reticular adhesions are a distinct class of cell-matrix adhesions that mediate attachment during mitosis. Nat. Cell Biol. 2018, 20, 1290–1302. [Google Scholar] [CrossRef] [PubMed]
- Baschieri, F.; Dayot, S.; Elkhatib, N.; Ly, N.; Capmany, A.; Schauer, K.; Betz, T.; Vignjevic, D.M.; Poincloux, R.; Montagnac, G. Frustrated endocytosis controls contractility-independent mechanotransduction at clathrin-coated structures. Nat. Commun. 2018, 9, 3825. [Google Scholar] [CrossRef]
- Leyton-Puig, D.; Isogai, T.; Argenzio, E.; van den Broek, B.; Klarenbeek, J.; Janssen, H.; Jalink, K.; Innocenti, M. Flat clathrin lattices are dynamic actin-controlled hubs for clathrin-mediated endocytosis and signalling of specific receptors. Nat. Commun. 2017, 8, 16068. [Google Scholar] [CrossRef] [PubMed]
- Franck, A.; Laine, J.; Moulay, G.; Lemerle, E.; Trichet, M.; Gentil, C.; Benkhelifa-Ziyyat, S.; Lacene, E.; Bui, M.T.; Brochier, G.; et al. Clathrin plaques and associated actin anchor intermediate filaments in skeletal muscle. Mol. Biol. Cell 2019, 30, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Vassilopoulos, S.; Gentil, C.; Laine, J.; Buclez, P.O.; Franck, A.; Ferry, A.; Precigout, G.; Roth, R.; Heuser, J.E.; Brodsky, F.M.; et al. Actin scaffolding by clathrin heavy chain is required for skeletal muscle sarcomere organization. J. Cell Biol. 2014, 205, 377–393. [Google Scholar] [CrossRef]
- Lane, N.E.; Yao, W.; Nakamura, M.C.; Humphrey, M.B.; Kimmel, D.; Huang, X.; Sheppard, D.; Ross, F.P.; Teitelbaum, S.L. Mice lacking the integrin beta5 subunit have accelerated osteoclast maturation and increased activity in the estrogen-deficient state. J. Bone Miner. Res. 2005, 20, 58–66. [Google Scholar] [CrossRef]
- Gimber, N.; Tadeus, G.; Maritzen, T.; Schmoranzer, J.; Haucke, V. Diffusional spread and confinement of newly exocytosed synaptic vesicle proteins. Nat. Commun. 2015, 6, 8392. [Google Scholar] [CrossRef]
- Kononenko, N.L.; Puchkov, D.; Classen, G.A.; Walter, A.M.; Pechstein, A.; Sawade, L.; Kaempf, N.; Trimbuch, T.; Lorenz, D.; Rosenmund, C.; et al. Clathrin/AP-2 mediate synaptic vesicle reformation from endosome-like vacuoles but are not essential for membrane retrieval at central synapses. Neuron 2014, 82, 981–988. [Google Scholar] [CrossRef]
- Dong, J.; Saunders, D.; Silasi-Mansat, R.; Yu, L.; Zhu, H.; Lupu, F.; Towner, R.; Dong, Y.; Chen, H. Therapeutic efficacy of a synthetic epsin mimetic peptide in glioma tumor model: Uncovering multiple mechanisms beyond the VEGF-associated tumor angiogenesis. J. NeuroOncol. 2018, 138, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wu, H.; Dong, J.; Song, K.; Rahman, H.A.; Towner, R.; Chen, H. Mimetic peptide of ubiquitin-interacting motif of epsin as a cancer therapeutic-perspective in brain tumor therapy through regulating VEGFR2 signaling. Vessel Plus 2017, 1, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wu, H.; Rahman, H.N.; Liu, Y.; Pasula, S.; Tessneer, K.L.; Cai, X.; Liu, X.; Chang, B.; McManus, J.; et al. Motif mimetic of epsin perturbs tumor growth and metastasis. J. Clin. Investig. 2015, 125, 4349–4364. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Chang, J.C.; Fan, E.Y.; Flajolet, M.; Greengard, P. Adaptor complex AP2/PICALM, through interaction with LC3, targets Alzheimer’s APP-CTF for terminal degradation via autophagy. Proc. Natl. Acad. Sci. USA 2013, 110, 17071–17076. [Google Scholar] [CrossRef]
- Bagh, M.B.; Peng, S.; Chandra, G.; Zhang, Z.; Singh, S.P.; Pattabiraman, N.; Liu, A.; Mukherjee, A.B. Misrouting of v-ATPase subunit V0a1 dysregulates lysosomal acidification in a neurodegenerative lysosomal storage disease model. Nat. Commun. 2017, 8, 14612. [Google Scholar] [CrossRef]
- Li, L.; Yang, S.; Zhang, Y.; Ji, D.; Jin, Z.; Duan, X. ATP6V1H regulates the growth and differentiation of bone marrow stromal cells. BioChem. Biophys. Res. Commun. 2018, 502, 84–90. [Google Scholar] [CrossRef]
- Prabhu, Y.; Burgos, P.V.; Schindler, C.; Farias, G.G.; Magadan, J.G.; Bonifacino, J.S. Adaptor protein 2-mediated endocytosis of the beta-secretase BACE1 is dispensable for amyloid precursor protein processing. Mol. Biol. Cell 2012, 23, 2339–2351. [Google Scholar] [CrossRef]
- Gondim, M.V.; Wiltzer-Bach, L.; Maurer, B.; Banning, C.; Arganaraz, E.; Schindler, M. AP-2 Is the Crucial Clathrin Adaptor Protein for CD4 Downmodulation by HIV-1 Nef in Infected Primary CD4+ T Cells. J. Virol. 2015, 89, 12518–12524. [Google Scholar] [CrossRef]
- Janvier, K.; Bonifacino, J.S. Role of the endocytic machinery in the sorting of lysosome-associated membrane proteins. Mol. Biol. Cell 2005, 16, 4231–4242. [Google Scholar] [CrossRef]
- Weixel, K.M.; Bradbury, N.A. Endocytic adaptor complexes bind the C-terminal domain of CFTR. Pflugers Arch. 2001, 443, S70–S74. [Google Scholar]
- Jang, S.; Ohtani, K.; Fukuoh, A.; Mori, K.; Yoshizaki, T.; Kitamoto, N.; Kim, Y.; Suzuki, Y.; Wakamiya, N. Scavenger receptor CL-P1 mediates endocytosis by associating with AP-2mu2. Biochim. Biophys. Acta 2014, 1840, 3226–3237. [Google Scholar] [CrossRef] [PubMed]
- Follows, E.R.; McPheat, J.C.; Minshull, C.; Moore, N.C.; Pauptit, R.A.; Rowsell, S.; Stacey, C.L.; Stanway, J.J.; Taylor, I.W.; Abbott, W.M. Study of the interaction of the medium chain mu 2 subunit of the clathrin-associated adapter protein complex 2 with cytotoxic T-lymphocyte antigen 4 and CD28. BioChem. J. 2001, 359, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Raman, D.; Sai, J.; Hawkins, O.; Richmond, A. Adaptor protein2 (AP2) orchestrates CXCR2-mediated cell migration. Traffic 2014, 15, 451–469. [Google Scholar] [CrossRef] [PubMed]
- Fong, J.T.; Kells, R.M.; Falk, M.M. Two tyrosine-based sorting signals in the Cx43 C-terminus cooperate to mediate gap junction endocytosis. Mol. Biol. Cell 2013, 24, 2834–2848. [Google Scholar] [CrossRef]
- D’Amico, A.; Soragna, A.; Di Cairano, E.; Panzeri, N.; Anzai, N.; Vellea Sacchi, F.; Perego, C. The surface density of the glutamate transporter EAAC1 is controlled by interactions with PDZK1 and AP2 adaptor complexes. Traffic 2010, 11, 1455–1470. [Google Scholar] [CrossRef]
- Rappoport, J.Z.; Simon, S.M. Endocytic trafficking of activated EGFR is AP-2 dependent and occurs through preformed clathrin spots. J. Cell Sci. 2009, 122, 1301–1305. [Google Scholar] [CrossRef]
- Jean, S.; Mikryukov, A.; Tremblay, M.G.; Baril, J.; Guillou, F.; Bellenfant, S.; Moss, T. Extended-synaptotagmin-2 mediates FGF receptor endocytosis and ERK activation in vivo. Dev. Cell 2010, 19, 426–439. [Google Scholar] [CrossRef]
- Yu, A.; Rual, J.F.; Tamai, K.; Harada, Y.; Vidal, M.; He, X.; Kirchhausen, T. Association of Dishevelled with the clathrin AP-2 adaptor is required for Frizzled endocytosis and planar cell polarity signaling. Dev. Cell 2007, 12, 129–141. [Google Scholar] [CrossRef]
- Kittler, J.T.; Delmas, P.; Jovanovic, J.N.; Brown, D.A.; Smart, T.G.; Moss, S.J. Constitutive endocytosis of GABAA receptors by an association with the adaptin AP2 complex modulates inhibitory synaptic currents in hippocampal neurons. J. Neurosci. 2000, 20, 7972–7977. [Google Scholar] [CrossRef]
- Bernhardt, U.; Carlotti, F.; Hoeben, R.C.; Joost, H.G.; Al-Hasani, H. A dual role of the N-terminal FQQI motif in GLUT4 trafficking. Biol. Chem. 2009, 390, 883–892. [Google Scholar] [CrossRef]
- Schmidt, U.; Briese, S.; Leicht, K.; Schurmann, A.; Joost, H.G.; Al-Hasani, H. Endocytosis of the glucose transporter GLUT8 is mediated by interaction of a dileucine motif with the beta2-adaptin subunit of the AP-2 adaptor complex. J. Cell Sci. 2006, 119, 2321–2331. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Liu, L.; Wang, Y.T.; Sheng, M. Clathrin adaptor AP2 and NSF interact with overlapping sites of GluR2 and play distinct roles in AMPA receptor trafficking and hippocampal LTD. Neuron 2002, 36, 661–674. [Google Scholar] [CrossRef]
- Zhao, B.; Wong, A.Y.; Murshid, A.; Bowie, D.; Presley, J.F.; Bedford, F.K. Identification of a novel di-leucine motif mediating K(+)/Cl(−) cotransporter KCC2 constitutive endocytosis. Cell Signal. 2008, 20, 1769–1779. [Google Scholar] [CrossRef] [PubMed]
- Mason, A.K.; Jacobs, B.E.; Welling, P.A. AP-2-dependent internalization of potassium channel Kir2.3 is driven by a novel di-hydrophobic signal. J. Biol. Chem. 2008, 283, 5973–5984. [Google Scholar] [CrossRef]
- Kamiguchi, H.; Long, K.E.; Pendergast, M.; Schaefer, A.W.; Rapoport, I.; Kirchhausen, T.; Lemmon, V. The neural cell adhesion molecule L1 interacts with the AP-2 adaptor and is endocytosed via the clathrin-mediated pathway. J. Neurosci. 1998, 18, 5311–5321. [Google Scholar] [CrossRef]
- Boll, W.; Rapoport, I.; Brunner, C.; Modis, Y.; Prehn, S.; Kirchhausen, T. The mu2 subunit of the clathrin adaptor AP-2 binds to FDNPVY and YppO sorting signals at distinct sites. Traffic 2002, 3, 590–600. [Google Scholar] [CrossRef]
- McCormick, P.J.; Martina, J.A.; Bonifacino, J.S. Involvement of clathrin and AP-2 in the trafficking of MHC class II molecules to antigen-processing compartments. Proc. Natl. Acad. Sci. USA 2005, 102, 7910–7915. [Google Scholar] [CrossRef]
- Chen, Y.T.; Tai, C.Y. mu2-Dependent endocytosis of N-cadherin is regulated by beta-catenin to facilitate neurite outgrowth. Traffic 2017, 18, 287–303. [Google Scholar] [CrossRef]
- Lee, K.H.; Ho, W.K.; Lee, S.H. Endocytosis of somatodendritic NCKX2 is regulated by Src family kinase-dependent tyrosine phosphorylation. Front. Cell. Neurosci. 2013, 7, 14. [Google Scholar] [CrossRef]
- Lavezzari, G.; McCallum, J.; Lee, R.; Roche, K.W. Differential binding of the AP-2 adaptor complex and PSD-95 to the C-terminus of the NMDA receptor subunit NR2B regulates surface expression. Neuropharmacology 2003, 45, 729–737. [Google Scholar] [CrossRef]
- Smith, T.H.; Coronel, L.J.; Li, J.G.; Dores, M.R.; Nieman, M.T.; Trejo, J. Protease-activated Receptor-4 Signaling and Trafficking Is Regulated by the Clathrin Adaptor Protein Complex-2 Independent of beta-Arrestins. J. Biol. Chem. 2016, 291, 18453–18464. [Google Scholar] [CrossRef] [PubMed]
- Royle, S.J.; Qureshi, O.S.; Bobanovic, L.K.; Evans, P.R.; Owen, D.J.; Murrell-Lagnado, R.D. Non-canonical YXXGPhi endocytic motifs: Recognition by AP2 and preferential utilization in P2 × 4 receptors. J. Cell Sci. 2005, 118, 3073–3080. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.S.; Keat, S.J.; Hamati, J.W.; Madsen, P.; Gutzmann, J.J.; Engelsberg, A.; Pedersen, K.M.; Gustafsen, C.; Nykjaer, A.; Gliemann, J.; et al. Different motifs regulate trafficking of SorCS1 isoforms. Traffic 2008, 9, 980–994. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Ehrlich, M.; Henis, Y.I.; Leof, E.B. Transforming growth factor-beta receptors interact with AP2 by direct binding to beta2 subunit. Mol. Biol. Cell 2002, 13, 4001–4012. [Google Scholar] [CrossRef]
- Stephens, D.J.; Crump, C.M.; Clarke, A.R.; Banting, G. Serine 331 and tyrosine 333 are both involved in the interaction between the cytosolic domain of TGN38 and the mu2 subunit of the AP2 clathrin adaptor complex. J. Biol. Chem. 1997, 272, 14104–14109. [Google Scholar] [CrossRef][Green Version]
- Santos, M.S.; Park, C.K.; Foss, S.M.; Li, H.; Voglmaier, S.M. Sorting of the vesicular GABA transporter to functional vesicle pools by an atypical dileucine-like motif. J. Neurosci. 2013, 33, 10634–10646. [Google Scholar] [CrossRef]
- Foss, S.M.; Li, H.; Santos, M.S.; Edwards, R.H.; Voglmaier, S.M. Multiple dileucine-like motifs direct VGLUT1 trafficking. J. Neurosci. 2013, 33, 10647–10660. [Google Scholar] [CrossRef]
- Deming, J.D.; Pak, J.S.; Brown, B.M.; Kim, M.K.; Aung, M.H.; Eom, Y.S.; Shin, J.A.; Lee, E.J.; Pardue, M.T.; Craft, C.M. Visual Cone Arrestin 4 Contributes to Visual Function and Cone Health. Investig. OphthalMol. Vis. Sci. 2015, 56, 5407–5416. [Google Scholar] [CrossRef][Green Version]
- Kan, Z.; Jaiswal, B.S.; Stinson, J.; Janakiraman, V.; Bhatt, D.; Stern, H.M.; Yue, P.; Haverty, P.M.; Bourgon, R.; Zheng, J.; et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 2010, 466, 869–873. [Google Scholar] [CrossRef]
- Anupam, K.; Tusharkant, C.; Gupta, S.D.; Ranju, R. Loss of disabled-2 expression is an early event in esophageal squamous tumorigenesis. World J. Gastroenterol. 2006, 12, 6041–6045. [Google Scholar] [CrossRef]
- Karam, J.A.; Shariat, S.F.; Huang, H.Y.; Pong, R.C.; Ashfaq, R.; Shapiro, E.; Lotan, Y.; Sagalowsky, A.I.; Wu, X.R.; Hsieh, J.T. Decreased DOC-2/DAB2 expression in urothelial carcinoma of the bladder. Clin. Cancer Res. 2007, 13, 4400–4406. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kleeff, J.; Huang, Y.; Mok, S.C.; Zimmermann, A.; Friess, H.; Buchler, M.W. Down-regulation of DOC-2 in colorectal cancer points to its role as a tumor suppressor in this malignancy. Dis. Colon Rectum. 2002, 45, 1242–1248. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.P.; Ely, B.D.; Li, Y.; Pong, R.C.; Hsieh, J.T. Regulation of rat DOC-2 gene during castration-induced rat ventral prostate degeneration and its growth inhibitory function in human prostatic carcinoma cells. Endocrinology 1998, 139, 3542–3553. [Google Scholar] [CrossRef] [PubMed]
- Bernard, O.A.; Mauchauffe, M.; Mecucci, C.; Van den Berghe, H.; Berger, R. A novel gene, AF-1p, fused to HRX in t(1;11)(p32;q23), is not related to AF-4, AF-9 nor ENL. Oncogene 1994, 9, 1039–1045. [Google Scholar]
- Tessneer, K.L.; Cai, X.; Pasula, S.; Dong, Y.; Liu, X.; Chang, B.; McManus, J.; Hahn, S.; Yu, L.; Chen, H. Epsin Family of Endocytic Adaptor Proteins as Oncogenic Regulators of Cancer Progression. J. Can. Res. Updates 2013, 2, 144–150. [Google Scholar]
- Porpaczy, E.; Bilban, M.; Heinze, G.; Gruber, M.; Vanura, K.; Schwarzinger, I.; Stilgenbauer, S.; Streubel, B.; Fonatsch, C.; Jaeger, U. Gene expression signature of chronic lymphocytic leukaemia with Trisomy 12. Eur. J. Clin. Investig. 2009, 39, 568–575. [Google Scholar] [CrossRef]
- Scanlan, M.J.; Welt, S.; Gordon, C.M.; Chen, Y.T.; Gure, A.O.; Stockert, E.; Jungbluth, A.A.; Ritter, G.; Jager, D.; Jager, E.; et al. Cancer-related serological recognition of human colon cancer: Identification of potential diagnostic and immunotherapeutic targets. Cancer Res. 2002, 62, 4041–4047. [Google Scholar]
- Pece, S.; Serresi, M.; Santolini, E.; Capra, M.; Hulleman, E.; Galimberti, V.; Zurrida, S.; Maisonneuve, P.; Viale, G.; Di Fiore, P.P. Loss of negative regulation by Numb over Notch is relevant to human breast carcinogenesis. J. Cell Biol. 2004, 167, 215–221. [Google Scholar] [CrossRef]
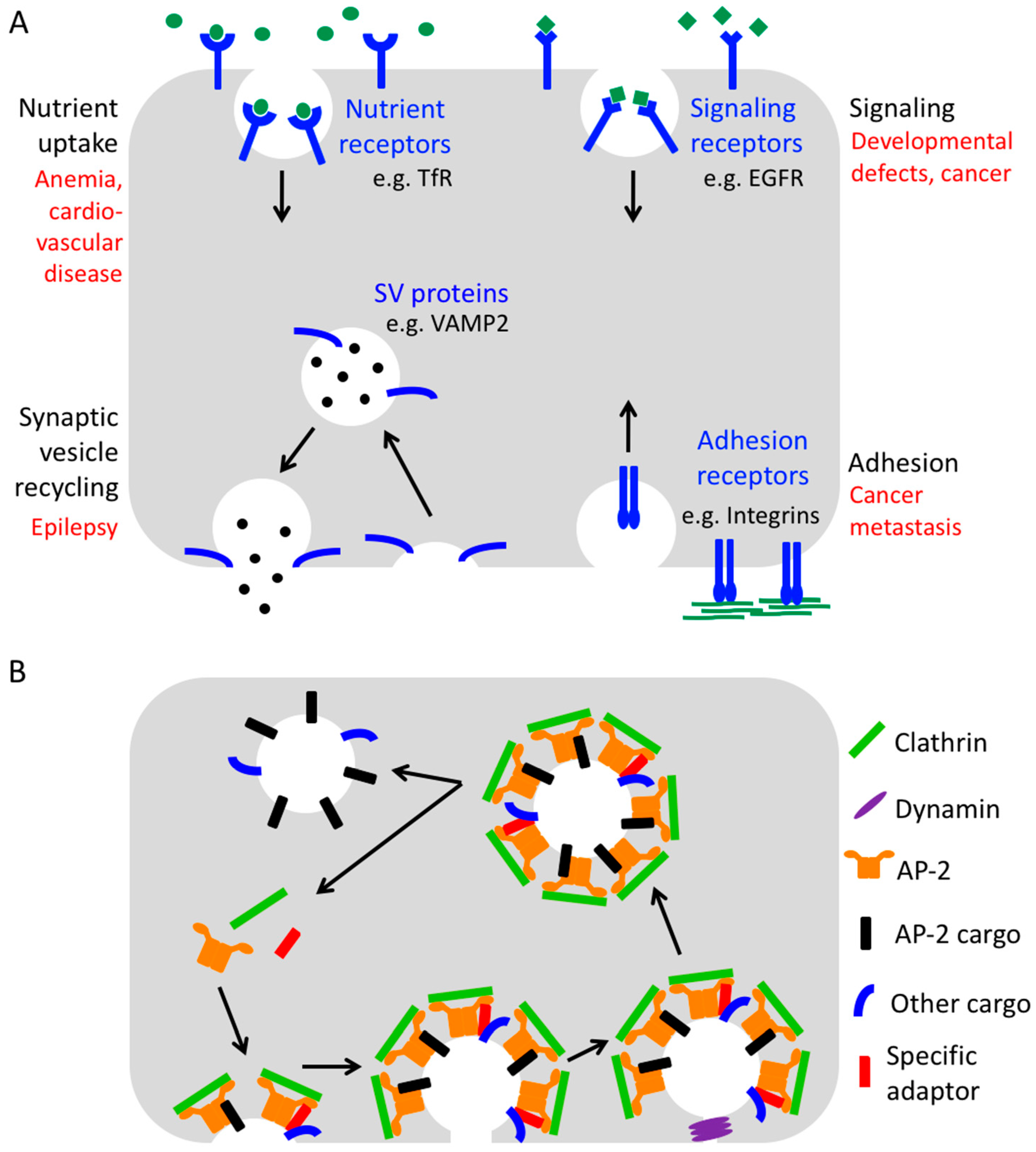
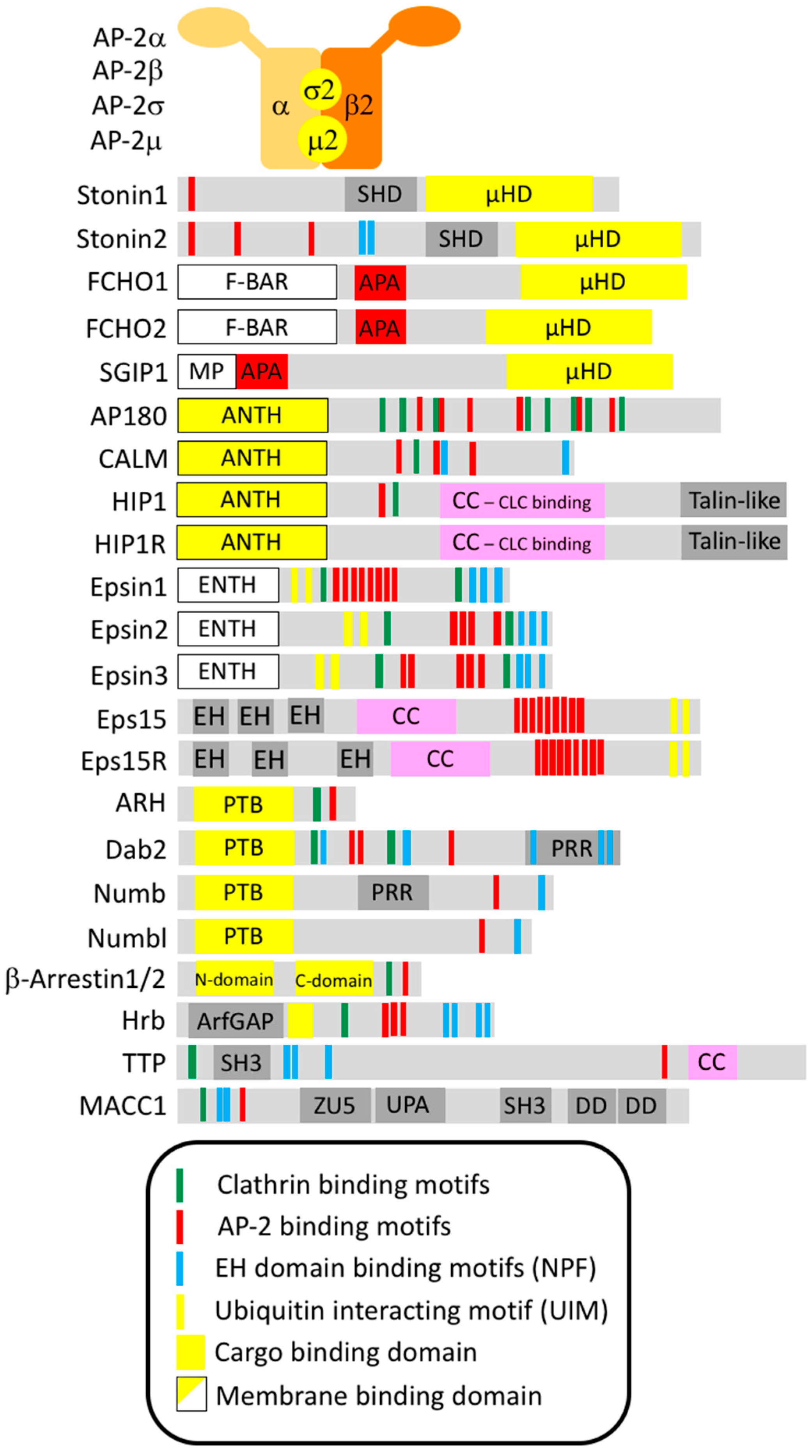
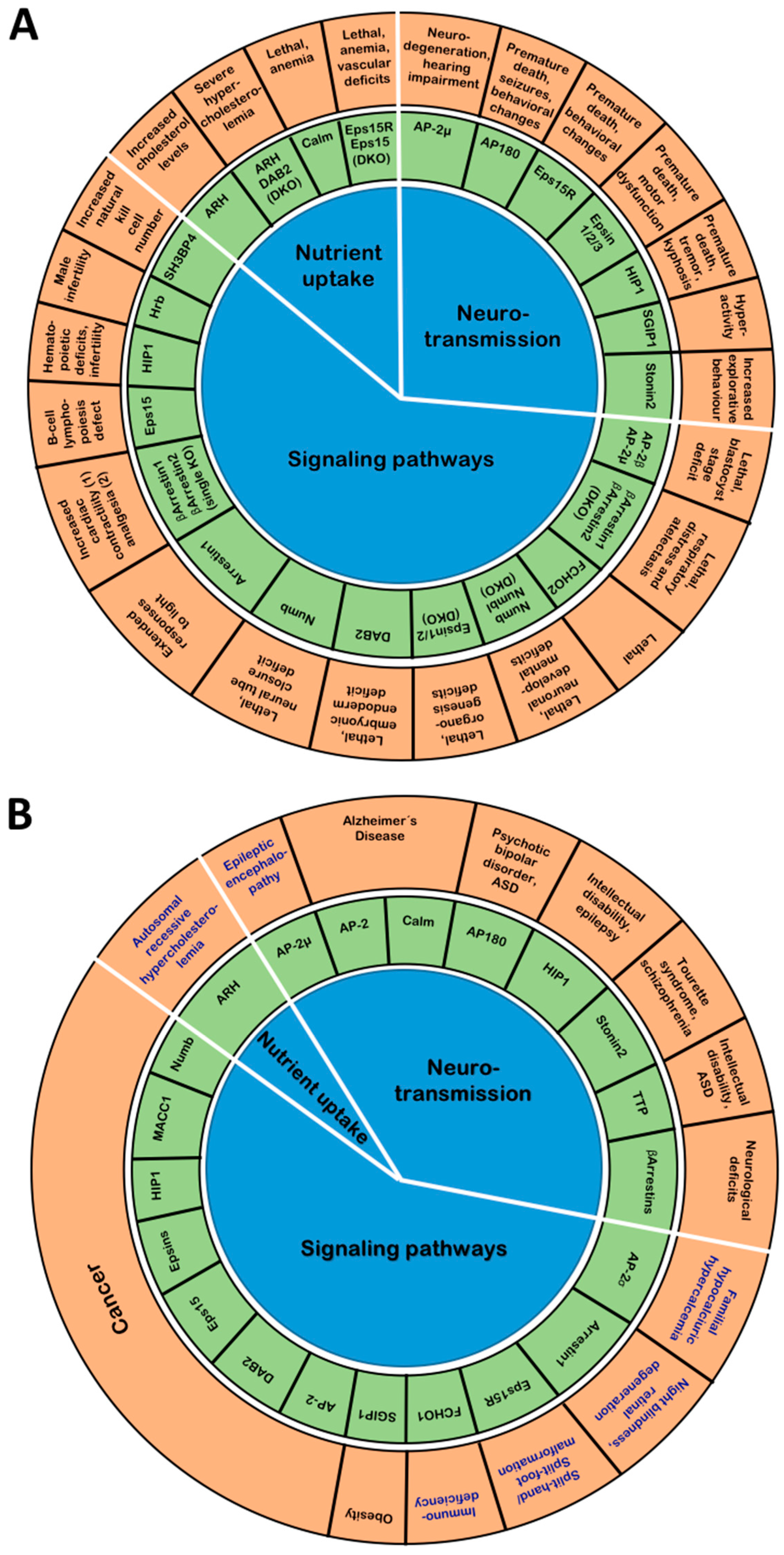
| Adaptor (Gene) | Signal–Adaptor Domain | Cargos | Endocytic Interactors | Mouse Phenotypes | Links to Human Disease |
|---|---|---|---|---|---|
| AP-2 (AP2A1, AP2A2, AP2B1 AP2M1, AP2S1) | YXXΦ-µ2; [DE]XXXL[LI]-α/σ2; C2 domains –µ2 | APP [335], ATP6V0A1 [336], ATP6V1H [337], BACE1 [338], CD4 [339], CD63 [340], CFTR [341], CL-P1 [342], CTLA-4 [343], CXCR2 [344], Cx43 [345], EAAC1 [346], EGFR [347], E-Syt2 [348], Frizzled4 [349], GABAAR [350], GLUT4 [351], GLUT8 [352], GluR2 [353], α2/α4 Integrins [319], KCC2 [354], Kir2.3 [355], L1 [356], Lamp1 [340], Lamp2 [340], LDLR [357], MHC-II [358], N-cadherin [359], NCKX2 [360], NR2B [361], Otoferlin [12], PAR4 [362], P2 × 4 R [363], SorCS1a [364], SorCS1c [364], Synaptotag-min1 [60], TGF-β R [365], TGN38 [366], TfR [9], VGAT [367], VGLUT1 [368] etc. | Amphiphy-sin, Apache, AP180, ARH, β-Arrestins, CALM, Clathrin, DAB2, Epsins, Eps15/R, FCHO1/2, HIP1, Hrb, Intersectin, MACC1, NECAP1, Numb, Numbl, PI(4,5)P2, SGIP1, Stonin1/2, Synaptoja-nin, TTP etc. | Constitutive AP2M1 KO: embryonic lethality [39]; constitutive AP2B1 KO: perinatal lethality and cleft palate [40]; AP2S1 del17 mouse: embryonic lethality; neuron-specific AP2M1 KO: neurodegeneration and premature death [38]; IHC-specific AP2M1 KO: hearing deficit [12] | AP2M1 mutation: epileptic encephalopathy [3]; AP2S1 mutation: familial hypocalciuric hypercalcemia type 3 [43]; AP-2α, AP-2β, AP-2σ downregulated in gliomas [48]; AP2A1, AP2A2 association with AD [47] |
| AP180 (SNAP91) | SNARE motif-ANTH | VAMP2 [121,122] | AP-2, Clathrin, PI(4,5)P2 | Constitutive KO: behavioral alterations, epileptic seizures, premature death [105] | Link to psychotic bipolar disorder [125] and to ASDs [126]; downregulated in gliomas [48] |
| ARH (LDLRAP1) | [FY]XNPX[YF]-PTB | Amnionless [234], β5 Integrin [323], LDLR [8], LRP2/Mega-lin [233], ROMK [235] | AP-2, Clathrin, PI(4,5)P2 | Constitutive KO: increased cholesterol levels [230,231], altered ROMK response [235]; ARH/Dab2 DKO: pronounced hypercholesterolemia [256] | Mutated in Autosomal recessive hypercholestero-lemia (ARH) [1] |
| Arrestin1 (SAG) | pSer/pThr in GPCR-NT | Rhodopsin [295] | Constitutive KO: extended response to light [295] | Mutated in Oguchi syndrome: night blindness and sometimes retinal degeneration [300] | |
| Arrestin4 (ARR3) | pSer/ pThr in GPCR-NT | Cone opsins [369] | Constitutive KO: diminished visual acuity and contrast sensitivity [369] | ||
| β-Arres-tin1 = Arrestin2 (ARRB1) | pSer/ pThr in GPCR-NT | Hundreds of GPCRs, e.g. β2-adre-nergic receptor [290] | AP-2, Clathrin | Constitutive KO: altered µ-opioid receptor signaling causing enhanced morphine analgesia [297]; DKO with β-arrestin2: embryonic lethality due to developmental defects [298] | Polymorphisms of unclear significance linked to neurological diseases [289]; somatic mutations in breast cancer [370] |
| β-Arres-tin2 = Arrestin3 (ARRB2) | pSer/ pThr in GPCR-NT | Constitutive KO: altered cardiac β-adrenergic receptor signaling causing increased cardiac contractility [296] | |||
| CALM (PICALM) | SNARE motif-ANTH | APP [129], Aβ-bound LRP1 [128], Nicastrin [131], TfR [113,124], VAMP2/3/4/7/8 [110,112,122] | AP-2, Clathrin, PI(4,5)P2 | Constitutive KO: anemia, embryonal or perinatal lethality [113]; adult onset KO: viable [113,124] | SNPs in AD [127]; somatic mutation/gene fusion in ALL and AML [104] |
| Dab2 (DAB2) | [FY]XNPX[YF]-PTB | Amnionless [234], ApoER [249], CFTR [253], E-Cadherin [260], EGFR [256], FGFR [256], β1/β5 Integrin [276], LDLR [81], LRP1 [256], LRP2/Mega-lin [233], LRP6 [248], VEGFR [250] | AP-2, Clathrin, Eps15, FCHO2, Intersectin, MyosinVI, PI(4,5)P2 | Constitutive KO: lethal prior to gastrulation [247]; conditional ubiquitous KO: mild proteinuria, mild increase in serum cholesterol, reduced tumor incidence [231,232,247,256,264] (see also [256] for complete list) | Downregulated in cancers (bladder, breast, colorectal, oesophageal, ovarian, prostate) [243,256,263,371,372,373,374] |
| Eps15 (EPS15) | Ubiqui-tin-UIM; CC-Met | Met [187], TfR [170], Ub-Cx43 [185], Ub-EGFR [184], Ub-GluA1 [186] | AP-2, CHC, Dynamin, Epsins, Intersectin, Numb, Stonin2, Synaptoja-nin | Constitutive KO: altered B cell lymphopoiesis [195] | Somatic mutations/gene fusion in AML and lung cancer [370,375] |
| Eps15R (EPS15L1) | Ubiqui-tin-UIM | EphB2/ ephrinB [180], TfR [170] | AP-2, CHC, Dynamin, Epsins, Intersectin, Numb, Stonin2, Synaptoja-nin | Constitutive KO: pre-/postnatal lethality, problems with respiration and feeding, growth deficits, behavioral alterations [170]; Eps15/Eps15R DKO: embryonic lethality, anemia, vascular defects [170] | Gene deletion causing SHFM |
| Epsin-1 (EPN1) | Ubiqui-tin-UIM | Ub-EGFR [207], ErbB3 [211], Notch ligands [212,213,214,215,216], LRP1 [219], PAR1 [206], VEGFR2/3 [217,218] | AP-2, Clathrin, Eps15, HIP1R, PI(4,5)P2 | Single KOs: no effect; DKO: embryonic lethality [213]; Vascular endothelium- specific DKO: disorganized tumor vasculature; brain- specific DKO: less animals born, progressive motor dysfunction, premature death [217] | Upregulated in cancer [2,224,376] |
| Epsin-2 (EPN2) | |||||
| Epsin-3 (EPN3) | Ubiqui-tin-UIM | AP-2, Clathrin, Eps15, PI(4,5)P2 | Constitutive KO: no effect | ||
| FCHO1 (FCHO1) | Not known-µHD | Alk8 (zebrafish) [87] Mid2 (yeast) [26] | AP-2, Dab2, Eps15, PIs | No KO mouse reported | Mutated in combined immuno-deficiency [94]; downregulated in gliomas [48] |
| FCHO2 (FCHO2) | Not known-µHD | KO mouse at IMPC: preweaning lethality | |||
| HIP1 (HIP1) | AMPAR [143] | AP-2, CHC, CLC, F-Actin | Constitutive KO (multiple lines): progressive tremor, ataxia, kyphosis culminating in premature death [138], decreased LTD [143], testicular degeneration [151], haematopoietic alterations, ophthalmic defects [153], partial protection against arthritis [156] and prostate tumorigenesis [157]; effects aggravated in HIP1/HIP1R DKO [139] | Chromosomal microdeletion causing neurological deficits [167]; overexpression/ somatic mutations/gene fusion in diverse cancers [157,158,159,160,166,370]; potential involvement in HD [134,168] | |
| HIP1R (HIP1R) | CLC, Cortactin, F-Aktin | Constitutive KO: loss of gastric epithelial cells, epithelial abnormalities [154] | Overexpressed in colon cancer and CLL [377,378] | ||
| Hrb (AGFG1) | VAMP7 longin domain-CT un-struc-tured domain | VAMP7 [35] | Eps15 | Constitutive KO: infertile due to defective acrosome formation [307] | |
| Hrbl (AFGF2) | Eps15 | KO mouse at IMPC: no alterations | |||
| MACC1 (MACC1) | AP-2, Clathrin | Upregulated in metastatic tissue, cancer-related SNPs [314] | |||
| Numb (NUMB) | [FY]XNPX[YF]-PTB | Alk [281], Boc [282], EAAT3 [279], E-Cadherin [277], β5/β1 Integrin [276], NPC1L1 [278], mGlu1 [280], Sanpodo/ Notch [267] | AP-2, Clathrin, PI(4,5)P2 | Constitutive KO: embryonal lethality at E11.5 due to defect in neural tube closure [283]; different conditional KOs: reduced cholesterol upake [278], alterations in behaviour [285] and motor coordination [280] | Downregulated in breast cancer [379] |
| Numbl (NUMBL) | [FY]XNPX[YF] –PTB | ? | AP-2, Clathrin, PI(4,5)P2 | Constitutive KO: no severe defects [33]; Numb/Numbl DKO: embryonic lethality [284] | |
| SGIP1 (SGIP1) | C2A-µHD | Synaptotag-min1 [92] | AP-2, Dab2, Endophilin, Eps15, Intersectin, PIs, PS | KO mouse at IMPC: abnormal behaviour, cardiovascular phenotype etc. | Associations with obesity, EEG and ECG abnormalities to be confirmed [98,99,100,101] |
| Stonin1 (Ston1) | Not known-µHD | No directly interacting cargo known | AP-2 | Constitutive KO: no obvious phenotype [77] | Upregulated in gliomas [48] |
| Stonin2 (Ston2) | C2A –µHD | Synaptotag-min1 [63,66] | AP-2, Eps15, Intersectin | Constitutive KO: behavioral and electrophysiological changes; no aggravation of defects in Stonin1/2 DKO [73] | Association with schizophrenia [79], lies within region mapped for Tourette disorder spectrum [78] |
| TTP (SH3BP4) | Not known-SH3 domain | Lamp1 [36], TfR [36] | AP-2, Clathrin, Dynamin, Eps15 | KO mouse at IMPC: increased NK cell number; intestine-specific KO: increase in intestinal stem cells [310] | Deleted together with AGAP1 in ASD/intellectual disability patient [313] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azarnia Tehran, D.; López-Hernández, T.; Maritzen, T. Endocytic Adaptor Proteins in Health and Disease: Lessons from Model Organisms and Human Mutations. Cells 2019, 8, 1345. https://doi.org/10.3390/cells8111345
Azarnia Tehran D, López-Hernández T, Maritzen T. Endocytic Adaptor Proteins in Health and Disease: Lessons from Model Organisms and Human Mutations. Cells. 2019; 8(11):1345. https://doi.org/10.3390/cells8111345
Chicago/Turabian StyleAzarnia Tehran, Domenico, Tania López-Hernández, and Tanja Maritzen. 2019. "Endocytic Adaptor Proteins in Health and Disease: Lessons from Model Organisms and Human Mutations" Cells 8, no. 11: 1345. https://doi.org/10.3390/cells8111345
APA StyleAzarnia Tehran, D., López-Hernández, T., & Maritzen, T. (2019). Endocytic Adaptor Proteins in Health and Disease: Lessons from Model Organisms and Human Mutations. Cells, 8(11), 1345. https://doi.org/10.3390/cells8111345