“Hierarchy” and “Holacracy”; A Paradigm of the Hematopoietic System
Abstract
1. Introduction
2. Discussion
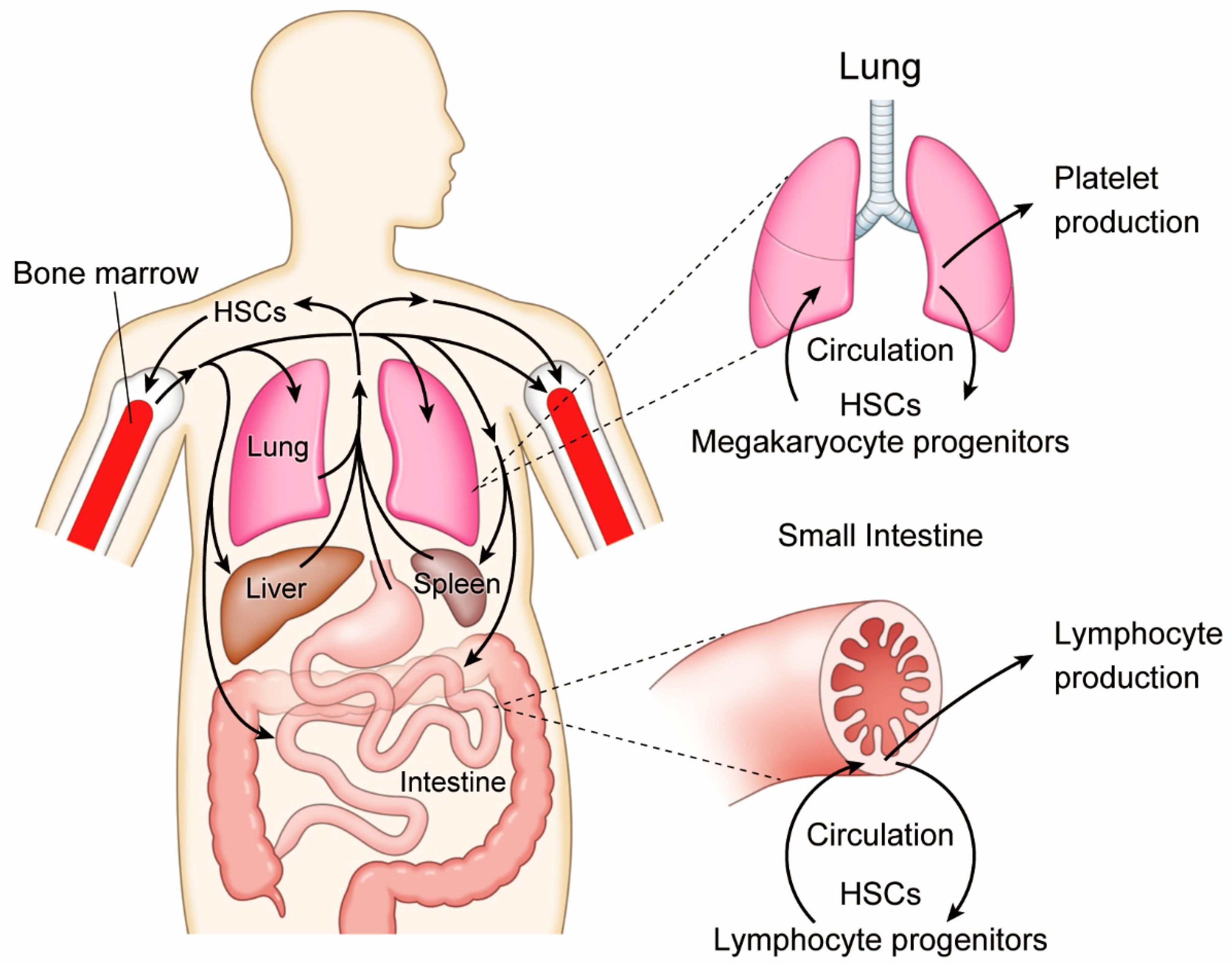
2.1. Active Hematopoiesis Outside of Bone Marrow
2.2. Lung Megakaryopoiesis
2.3. Intestinal Lymphopoiesis
2.4. Contribution of Circulating HSCs to Myeloid Lineage in the Periphery
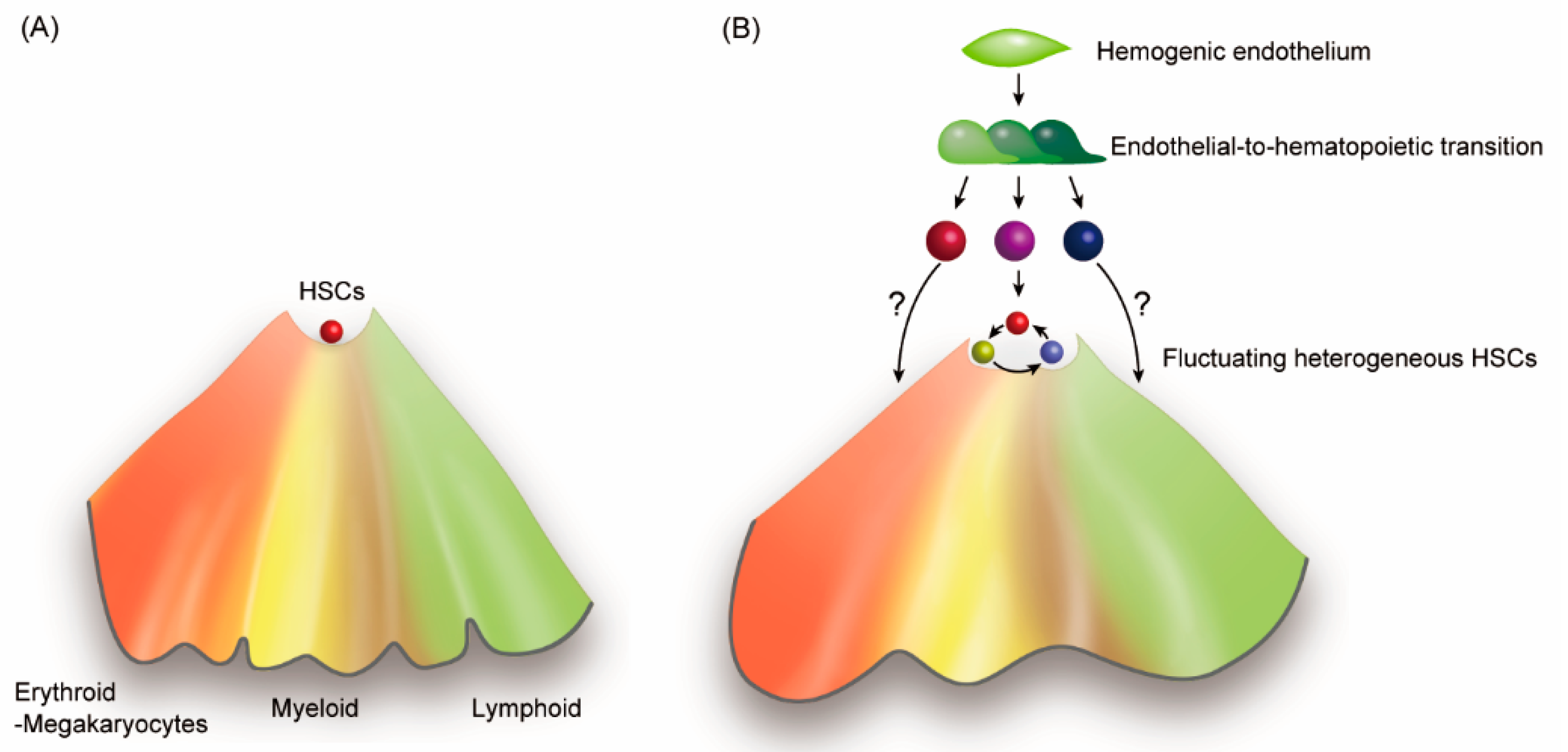
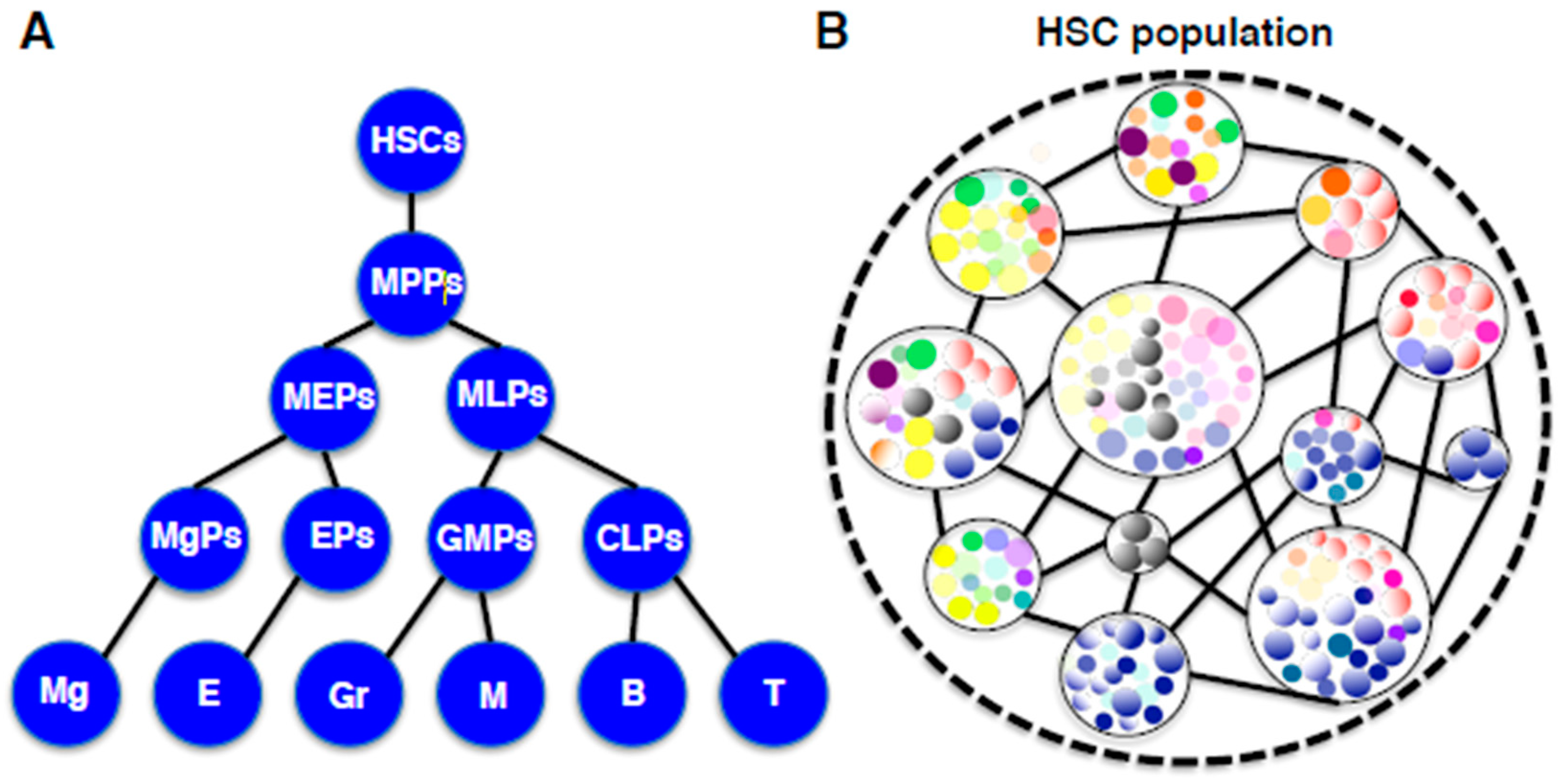
2.5. Diversity and Fluctuation in the HSC Compartment
2.6. Diversity of the “HSC Niche”
2.7. Hierarchy and Holacracy
3. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Mikkola, H.K.A.; Orkin, S.H. The Journey of Developing Hematopoietic Stem Cells. Development 2006, 133, 3733–3744. [Google Scholar] [CrossRef] [PubMed]
- Dzierzak, E.; Speck, N.A. Of Lineage and Legacy: The Development of Mammalian Hematopoietic Stem Cells. Nat. Immunol. 2008, 9, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Tavian, M.; Péault, B. The Changing Cellular Environments of Hematopoiesis in Human Development in Utero. Exp. Hematol. 2005, 33, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, M.; Yoder, M.C. Developmental Biology: Birth of the Blood Cell. Nature 2009, 457, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Yokomizo, T.; Yamada-Inagawa, T.; Yzaguirre, A.D.; Chen, M.J.; Speck, N.A.; Dzierzak, E. Whole-Mount Three-Dimensional Imaging of Internally Localized Immunostained Cells within Mouse Embryos. Nat. Protoc. 2012, 7, 421–431. [Google Scholar] [CrossRef] [PubMed]
- North, T.E.; Goessling, W.; Peeters, M.; Li, P.; Ceol, C.; Lord, A.M.; Weber, G.J.; Harris, J.; Cutting, C.C.; Huang, P.; et al. Hematopoietic Stem Cell Development Is Dependent on Blood Flow. Cell 2009, 137, 736–748. [Google Scholar] [CrossRef]
- Levine, R.F.; Eldor, A.; Shoff, P.K.; Kirwin, S.; Tenza, D.; Cramer, E.M. Circulating Megakaryocytes: Delivery of Large Numbers of Intact, Mature Megakaryocytes to the Lungs. Eur. J. Haematol. 1993, 51, 233–246. [Google Scholar] [CrossRef]
- Zucker-Franklin, D.; Philipp, C.S. Platelet Production in the Pulmonary Capillary Bed: New Ultrastructural Evidence for an Old Concept. Am. J. Pathol. 2000, 157, 69–74. [Google Scholar] [CrossRef]
- Lefrançais, E.; Ortiz-Muñoz, G.; Caudrillier, A.; Mallavia, B.; Liu, F.; Sayah, D.M.; Thornton, E.E.; Headley, M.B.; David, T.; Coughlin, S.R.; et al. The Lung Is a Site of Platelet Biogenesis and a Reservoir for Haematopoietic Progenitors. Nature 2017, 544, 105–109. [Google Scholar] [CrossRef]
- Ito, Y.; Nakamura, S.; Sugimoto, N.; Shigemori, T.; Kato, Y.; Ohno, M.; Sakuma, S.; Ito, K.; Kumon, H.; Hirose, H.; et al. Turbulence Activates Platelet Biogenesis to Enable Clinical Scale Ex Vivo Production. Cell 2018, 174, 636–648.e18. [Google Scholar] [CrossRef]
- Laky, K.; Lefrançois, L.; Lingenheld, E.G.; Ishikawa, H.; Lewis, J.M.; Olson, S.; Suzuki, K.; Tigelaar, R.E.; Puddington, L. Enterocyte Expression of Interleukin 7 Induces Development of Gammadelta T Cells and Peyer’s Patches. J. Exp. Med. 2000, 191, 1569–1580. [Google Scholar] [CrossRef]
- Nonaka, S.; Naito, T.; Chen, H.; Yamamoto, M.; Moro, K.; Kiyono, H.; Hamada, H.; Ishikawa, H. Intestinal Gamma Delta T Cells Develop in Mice Lacking Thymus, All Lymph Nodes, Peyer’s Patches, and Isolated Lymphoid Follicles. J. Immunol. 2005, 174, 1906–1912. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Hamada, H.; Nonaka, S.; Yamamoto, H.; Nanno, M.; Katayama, Y.; Takahashi, H.; Ishikawa, H. Homeostatic Regulation of Intestinal Villous Epithelia by B Lymphocytes. J. Immunol. 2002, 168, 2626–2633. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Ishimaru, K.; Nanno, M.; Maki, K.; Ikuta, K.; Nariuchi, H.; Ishikawa, H. Identification of Novel Lymphoid Tissues in Murine Intestinal Mucosa Where Clusters of c-kit⁺ IL-7R⁺ Thy1⁺ Lympho-Hemopoietic Progenitors Develop. J. Exp. Med. 1996, 184, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Kanamori, Y.; Takemori, T.; Nariuchi, H.; Kubota, E.; Takahashi-Iwanaga, H.; Iwanaga, T.; Ishikawa, H. Generation of Intestinal T Cells from Progenitors Residing in Gut Cryptopatches. Science 1998, 280, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Lynch, L.; O’Donoghue, D.; Dean, J.; O’Sullivan, J.; O’Farrelly, C.; Golden-Mason, L. Detection and Characterization of Hemopoietic Stem Cells in the Adult Human Small Intestine. J. Immunol. 2006, 176, 5199–5204. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Zuber, J.; Martinez, M.; Shonts, B.; Obradovic, A.; Wang, H.; Lau, S.P.; Xia, A.; Waffarn, E.E.; Frangaj, K.; et al. Human Intestinal Allografts Contain Functional Hematopoietic Stem and Progenitor Cells That Are Maintained by a Circulating Pool. Cell Stem Cell 2019, 24, 227–239.e8. [Google Scholar] [CrossRef]
- Watanabe, M.; Ueno, Y.; Yajima, T.; Iwao, Y.; Tsuchiya, M.; Ishikawa, H.; Aiso, S.; Hibi, T.; Ishii, H. Interleukin 7 Is Produced by Human Intestinal Epithelial Cells and Regulates the Proliferation of Intestinal Mucosal Lymphocytes. J. Clin. Investig. 1995, 95, 2945–2953. [Google Scholar] [CrossRef]
- Reinecker, H.C.; MacDermott, R.P.; Mirau, S.; Dignass, A.; Podolsky, D.K. Intestinal Epithelial Cells Both Express and Respond to Interleukin 15. Gastroenterology 1996, 111, 1706–1713. [Google Scholar] [CrossRef]
- Winkler, I.G.; Lévesque, J.P. Mechanisms of Hematopoietic Stem Cell Mobilization: When Innate Immunity Assails the Cells That Make Blood and Bone. Exp. Hematol. 2006, 34, 996–1009. [Google Scholar] [CrossRef]
- de Kruijf, E.F.M.; Fibbe, W.E.; van Pel, M. Cytokine-Induced Hematopoietic Stem and Progenitor Cell Mobilization: Unraveling Interactions between Stem Cells and Their Niche. Ann. N. Y. Acad. Sci. 2019. [Google Scholar] [CrossRef]
- Wright, D.E.; Wagers, A.J.; Gulati, A.P.; Johnson, F.L.; Weissman, I.L. Physiological Migration of Hematopoietic Stem and Progenitor Cells. Science 2001, 294, 1933–1936. [Google Scholar] [CrossRef]
- Massberg, S.; Schaerli, P.; Knezevic-Maramica, I.; Köllnberger, M.; Tubo, N.; Moseman, E.A.; Huff, I.V.; Junt, T.; Wagers, A.J.; Mazo, I.B.; et al. Immunosurveillance by Hematopoietic Progenitor Cells Trafficking through Blood, Lymph, and Peripheral Tissues. Cell 2007, 131, 994–1008. [Google Scholar] [CrossRef]
- Nagai, Y.; Garrett, K.P.; Ohta, S.; Bahrun, U.; Kouro, T.; Akira, S.; Takatsu, K.; Kincade, P.W. Toll-Like Receptors on Hematopoietic Progenitor Cells Stimulate Innate Immune System Replenishment. Immunity 2006, 24, 801–812. [Google Scholar] [CrossRef]
- Takizawa, H.; Fritsch, K.; Kovtonyuk, L.V.; Saito, Y.; Yakkala, C.; Jacobs, K.; Ahuja, A.K.; Lopes, M.; Hausmann, A.; Hardt, W.D.; et al. Pathogen-Induced TLR4-TRIF Innate Immune Signaling in Hematopoietic Stem Cells Promotes Proliferation but Reduces Competitive Fitness. Cell Stem Cell 2017, 21, 225–240.e5. [Google Scholar] [CrossRef] [PubMed]
- Dykstra, B.; Kent, D.; Bowie, M.; McCaffrey, L.; Hamilton, M.; Lyons, K.; Lee, S.J.; Brinkman, R.; Eaves, C. Long-Term Propagation of Distinct Hematopoietic Differentiation Programs In Vivo. Cell Stem Cell 2007, 1, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Morita, Y.; Ema, H.; Nakauchi, H. Heterogeneity and Hierarchy within the Most Primitive Hematopoietic Stem Cell Compartment. J. Exp. Med. 2010, 207, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Challen, G.A.; Boles, N.C.; Chambers, S.M.; Goodell, M.A. Distinct Hematopoietic Stem Cell Subtypes Are Differentially Regulated by TGF-beta1. Cell Stem Cell 2010, 6, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Benz, C.; Copley, M.R.; Kent, D.G.; Wohrer, S.; Cortes, A.; Aghaeepour, N.; Ma, E.; Mader, H.; Rowe, K.; Day, C.; et al. Hematopoietic Stem Cell Subtypes Expand Differentially during Development and Display Distinct Lymphopoietic Programs. Cell Stem Cell 2012, 10, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, R.; Morita, Y.; Ooehara, J.; Hamanaka, S.; Onodera, M.; Rudolph, K.L.; Ema, H.; Nakauchi, H. Clonal Analysis Unveils Self-Renewing Lineage-Restricted Progenitors Generated Directly from Hematopoietic Stem Cells. Cell 2013, 154, 1112–1126. [Google Scholar] [CrossRef]
- Quesenberry, P.J.; Goldberg, L.R.; Dooner, M.S. Concise Reviews: A Stem Cell Apostasy: A Tale of Four H Words. Stem Cells 2015, 33, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Ramos, A.; Chapman, B.; Johnnidis, J.B.; Le, L.; Ho, Y.J.; Klein, A.; Hofmann, O.; Camargo, F.D. Clonal Dynamics of Native Haematopoiesis. Nature 2014, 514, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Busch, K.; Klapproth, K.; Barile, M.; Flossdorf, M.; Holland-Letz, T.; Schlenner, S.M.; Reth, M.; Höfer, T.; Rodewald, H.R. Fundamental Properties of Unperturbed Haematopoiesis from Stem Cells In Vivo. Nature 2015, 518, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Kiel, M.J.; Yilmaz, O.H.; Iwashita, T.; Yilmaz, O.H.; Terhorst, C.; Morrison, S.J. SLAM Family Receptors Distinguish Hematopoietic Stem and Progenitor Cells and Reveal Endothelial Niches for Stem Cells. Cell 2005, 121, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Balazs, A.B.; Fabian, A.J.; Esmon, C.T.; Mulligan, R.C. Endothelial Protein C Receptor (CD201) Explicitly Identifies Hematopoietic Stem Cells in Murine Bone Marrow. Blood 2006, 107, 2317–2321. [Google Scholar] [CrossRef] [PubMed]
- Yokota, T.; Oritani, K.; Butz, S.; Kokame, K.; Kincade, P.W.; Miyata, T.; Vestweber, D.; Kanakura, Y. The Endothelial Antigen ESAM Marks Primitive Hematopoietic Progenitors throughout Life in Mice. Blood 2009, 113, 2914–2923. [Google Scholar] [CrossRef] [PubMed]
- Gentles, A.J.; Plevritis, S.K.; Majeti, R.; Alizadeh, A.A. Association of a Leukemic Stem Cell Gene Expression Signature with Clinical Outcomes in Acute Myeloid Leukemia. JAMA 2010, 304, 2706–2715. [Google Scholar] [CrossRef] [PubMed]
- Notta, F.; Doulatov, S.; Laurenti, E.; Poeppl, A.; Jurisica, I.; Dick, J.E. Isolation of Single Human Hematopoietic Stem Cells Capable of Long-Term Multilineage Engraftment. Science 2011, 333, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, T.; Yokota, T.; Tanaka, H.; Ichii, M.; Sudo, T.; Satoh, Y.; Doi, Y.; Ueda, T.; Tanimura, A.; Hamanaka, Y.; et al. ESAM Is a Novel Human Hematopoietic Stem Cell Marker Associated with a Subset of Human Leukemias. Exp. Hematol. 2016, 44, 269–281. [Google Scholar] [CrossRef]
- Yokota, T.; Oritani, K.; Butz, S.; Ewers, S.; Vestweber, D.; Kanakura, Y. Markers for Hematopoietic Stem Cells: Histories and Recent Achievements. In Advances in Hematopoietic Stem Cell Research; IntechOpen: London, UK, 2012; pp. 77–88. [Google Scholar]
- Wilson, A.; Laurenti, E.; Oser, G.; van der Wath, R.C.; Blanco-Bose, W.; Jaworski, M.; Offner, S.; Dunant, C.F.; Eshkind, L.; Bockamp, E.; et al. Hematopoietic Stem Cells Reversibly Switch from Dormancy to Self-Renewal during Homeostasis and Repair. Cell 2008, 135, 1118–1129. [Google Scholar] [CrossRef]
- Sudo, T.; Yokota, T.; Oritani, K.; Satoh, Y.; Sugiyama, T.; Ishida, T.; Shibayama, H.; Ezoe, S.; Fujita, N.; Tanaka, H.; et al. The Endothelial Antigen ESAM Monitors Hematopoietic Stem Cell Status between Quiescence and Self-Renewal. J. Immunol. 2012, 189, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, C.; Kaneko, K. Chaotic Expression Dynamics Implies Pluripotency: When Theory and Experiment Meet. Biol. Direct 2009, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Furusawa, C.; Kaneko, K. Oscillatory Protein Expression Dynamics Endows Stem Cells with Robust Differentiation Potential. PLoS ONE 2011, 6, e27232. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, C.; Kaneko, K. A Dynamical-Systems View of Stem Cell Biology. Science 2012, 338, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Nestorowa, S.; Hamey, F.K.; Pijuan Sala, B.; Diamanti, E.; Shepherd, M.; Laurenti, E.; Wilson, N.K.; Kent, D.G.; Göttgens, B. A Single-Cell Resolution Map of Mouse Hematopoietic Stem and Progenitor Cell Differentiation. Blood 2016, 128, e20–e31. [Google Scholar] [CrossRef]
- Yu, V.W.C.; Yusuf, R.Z.; Oki, T.; Wu, J.; Saez, B.; Wang, X.; Cook, C.; Baryawno, N.; Ziller, M.J.; Lee, E.; et al. Epigenetic Memory Underlies Cell-Autonomous Heterogeneous Behavior of Hematopoietic Stem Cells. Cell 2016, 167, 1310–1322.e17. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Yokota, T.; Satoh, Y.; Okuzaki, D.; Tokunaga, M.; Ishibashi, T.; Sudo, T.; Ueda, T.; Shingai, Y.; Ichii, M.; et al. Variable SATB1 Levels Regulate Hematopoietic Stem Cell Heterogeneity with Distinct Lineage Fate. Cell Rep. 2018, 23, 3223–3235. [Google Scholar] [CrossRef]
- Satoh, Y.; Yokota, T.; Sudo, T.; Kondo, M.; Lai, A.; Kincade, P.W.; Kouro, T.; Iida, R.; Kokame, K.; Miyata, T.; et al. The Satb1 Protein Directs Hematopoietic Stem Cell Differentiation Toward Lymphoid Lineages. Immunity 2013, 38, 1105–1115. [Google Scholar] [CrossRef]
- Alvarez, J.D.; Yasui, D.H.; Niida, H.; Joh, T.; Loh, D.Y.; Kohwi-Shigematsu, T. The MAR-Binding Protein SATB1 Orchestrates Temporal and Spatial Expression of Multiple Genes during T-Cell Development. Genes Dev. 2000, 14, 521–535. [Google Scholar]
- Yasui, D.; Miyano, M.; Cai, S.; Varga-Weisz, P.; Kohwi-Shigematsu, T. SATB1 Targets Chromatin Remodelling to Regulate Genes over Long Distances. Nature 2002, 419, 641–645. [Google Scholar] [CrossRef]
- Cui, K.; Zang, C.; Roh, T.Y.; Schones, D.E.; Childs, R.W.; Peng, W.; Zhao, K. Chromatin Signatures in Multipotent Human Hematopoietic Stem Cells Indicate the Fate of Bivalent Genes during Differentiation. Cell Stem Cell 2009, 4, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Oguro, H.; Yuan, J.; Ichikawa, H.; Ikawa, T.; Yamazaki, S.; Kawamoto, H.; Nakauchi, H.; Iwama, A. Poised Lineage Specification in Multipotential Hematopoietic Stem and Progenitor Cells by the Polycomb Protein Bmi1. Cell Stem Cell 2010, 6, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Waddington, C.H. The Strategy of the Genes; a Discussion of Some Aspects of Theoretical Biology; Allen and Unwin: London, UK, 1957. [Google Scholar]
- Yokota, T.; Huang, J.; Tavian, M.; Nagai, Y.; Hirose, J.; Zúñiga-Pflücker, J.C.; Péault, B.; Kincade, P.W. Tracing the first waves of lymphopoiesis in mice. Development 2006, 133, 2041–2051. [Google Scholar] [CrossRef] [PubMed]
- Guibentif, C.; Rönn, R.E.; Böiers, C.; Lang, S.; Saxena, S.; Soneji, S.; Enver, T.; Karlsson, G.; Woods, N.-B. Single-Cell Analysis Identifies Distinct Stages of Human Endothelial-to-Hematopoietic Transition. Cell Rep. 2017, 19, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Yokota, T.; Oritani, K.; Mitsui, H.; Aoyama, K.; Ishikawa, J.; Sugahara, H.; Matsumura, I.; Tsai, S.; Tomiyama, Y.; Kanakura, Y.; et al. Growth-Supporting Activities of Fibronectin on Hematopoietic Stem/Progenitor Cells In Vitro and In Vivo: Structural Requirement for Fibronectin Activities of CS1 and Cell-Binding Domains. Blood 1998, 91, 3263–3272. [Google Scholar] [PubMed]
- Coutu, D.L.; Kokkaliaris, K.D.; Kunz, L.; Schroeder, T. Three-Dimensional Map of Nonhematopoietic Bone and Bone-Marrow Cells and Molecules. Nat. Biotechnol. 2017, 35, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, A.C.; Ishida, R.; Kikuchi, M.; Sudo, K.; Morita, M.; Crisostomo, R.V.; Yamamoto, R.; Loh, K.M.; Nakamura, Y.; Watanabe, M.; et al. Long-Term Ex Vivo Haematopoietic-Stem-Cell Expansion Allows Nonconditioned Transplantation. Nature 2019, 571, 117–121. [Google Scholar] [CrossRef]
- Lok, S.; Kaushansky, K.; Holly, R.D.; Kuijper, J.L.; Lofton-Day, C.E.; Oort, P.J.; Grant, F.J.; Heipel, M.D.; Burkhead, S.K.; Kramer, J.M.; et al. Cloning and Expression of Murine Thrombopoietin cDNA and Stimulation of Platelet Production In Vivo. Nature 1994, 369, 565–568. [Google Scholar] [CrossRef]
- Kaushansky, K.; Lok, S.; Holly, R.D.; Broudy, V.C.; Lin, N.; Bailey, M.C.; Forstrom, J.W.; Buddle, M.M.; Oort, P.J.; Hagen, F.S.; et al. Promotion of Megakaryocyte Progenitor Expansion and Differentiation by the c-Mpl Ligand Thrombopoietin. Nature 1994, 369, 568–571. [Google Scholar] [CrossRef]
- Wendling, F.; Maraskovsky, E.; Debili, N.; Florindo, C.; Teepe, M.; Titeux, M.; Methia, N.; Breton-Gorius, J.; Cosman, D.; Vainchenker, W. cMpl Ligand Is a Humoral Regulator of Megakaryocytopoiesis. Nature 1994, 369, 571–574. [Google Scholar] [CrossRef]
- Fox, N.; Priestley, G.; Papayannopoulou, T.; Kaushansky, K. Thrombopoietin Expands Hematopoietic Stem Cells after Transplantation. J. Clin. Investig. 2002, 110, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Morrison, S.J.; Scadden, D.T. The Bone Marrow Niche for Haematopoietic Stem Cells. Nature 2014, 505, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Birbrair, A.; Frenette, P.S. Niche Heterogeneity in the Bone Marrow. Ann. N. Y. Acad. Sci. 2016, 1370, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Tajer, P.; Pike-Overzet, K.; Arias, S.; Havenga, M.; Staal, F.J.T. Ex Vivo Expansion of Hematopoietic Stem Cells for Therapeutic Purposes: Lessons from Development and the Niche. Cells 2019, 8, 169. [Google Scholar] [CrossRef]
- Velten, L.; Haas, S.F.; Raffel, S.; Blaszkiewicz, S.; Islam, S.; Hennig, B.P.; Hirche, C.; Lutz, C.; Buss, E.C.; Nowak, D.; et al. Human Haematopoietic Stem Cell Lineage Commitment Is a Continuous Process. Nat. Cell Biol. 2017, 19, 271–281. [Google Scholar] [CrossRef]
- Robertson, B. Organization at the Leading Edge: Introducing Holacracy™ Evolving Organization. Integr. Leadersh. Rev. 2007, 7, 1–13. [Google Scholar]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokota, T. “Hierarchy” and “Holacracy”; A Paradigm of the Hematopoietic System. Cells 2019, 8, 1138. https://doi.org/10.3390/cells8101138
Yokota T. “Hierarchy” and “Holacracy”; A Paradigm of the Hematopoietic System. Cells. 2019; 8(10):1138. https://doi.org/10.3390/cells8101138
Chicago/Turabian StyleYokota, Takafumi. 2019. "“Hierarchy” and “Holacracy”; A Paradigm of the Hematopoietic System" Cells 8, no. 10: 1138. https://doi.org/10.3390/cells8101138
APA StyleYokota, T. (2019). “Hierarchy” and “Holacracy”; A Paradigm of the Hematopoietic System. Cells, 8(10), 1138. https://doi.org/10.3390/cells8101138



