PET Imaging Biomarkers of Anti-EGFR Immunotherapy in Esophageal Squamous Cell Carcinoma Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Reverse Transcription Polymerase Chain Reaction
2.3. Immunoblot Analysis
2.4. Flow Cytometry
2.5. Preparation and Characterization of 64Cu-PCTA-Cetuximab
2.6. In Vitro Cell Binding Assay
2.7. Cell Proliferation Inhibition Assay
2.8. Immunotherapy
2.9. Small Animal PET Imaging
2.10. Immunohistochemistry
2.11. Statistical Analysis
3. Results
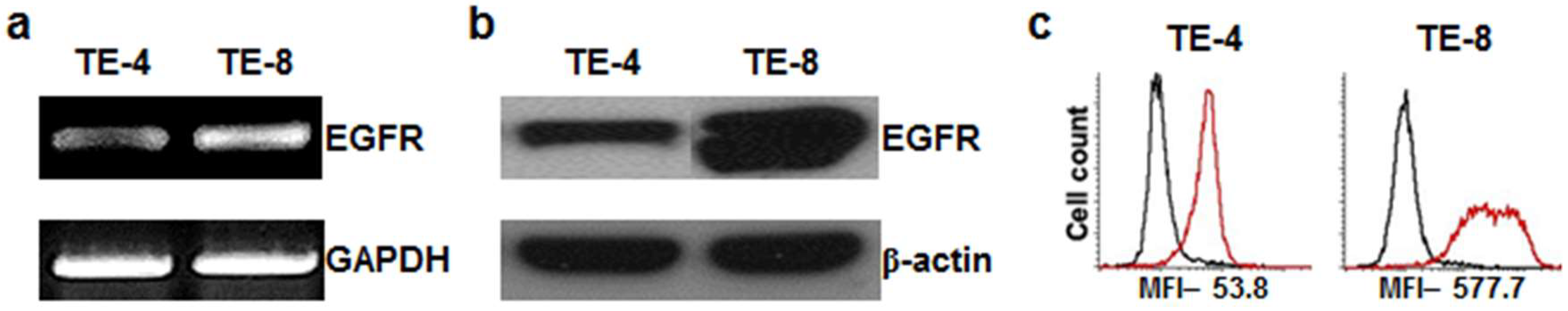
3.1. Characterization of EGFR Expression in Esophageal Squamous Cell Carcinoma
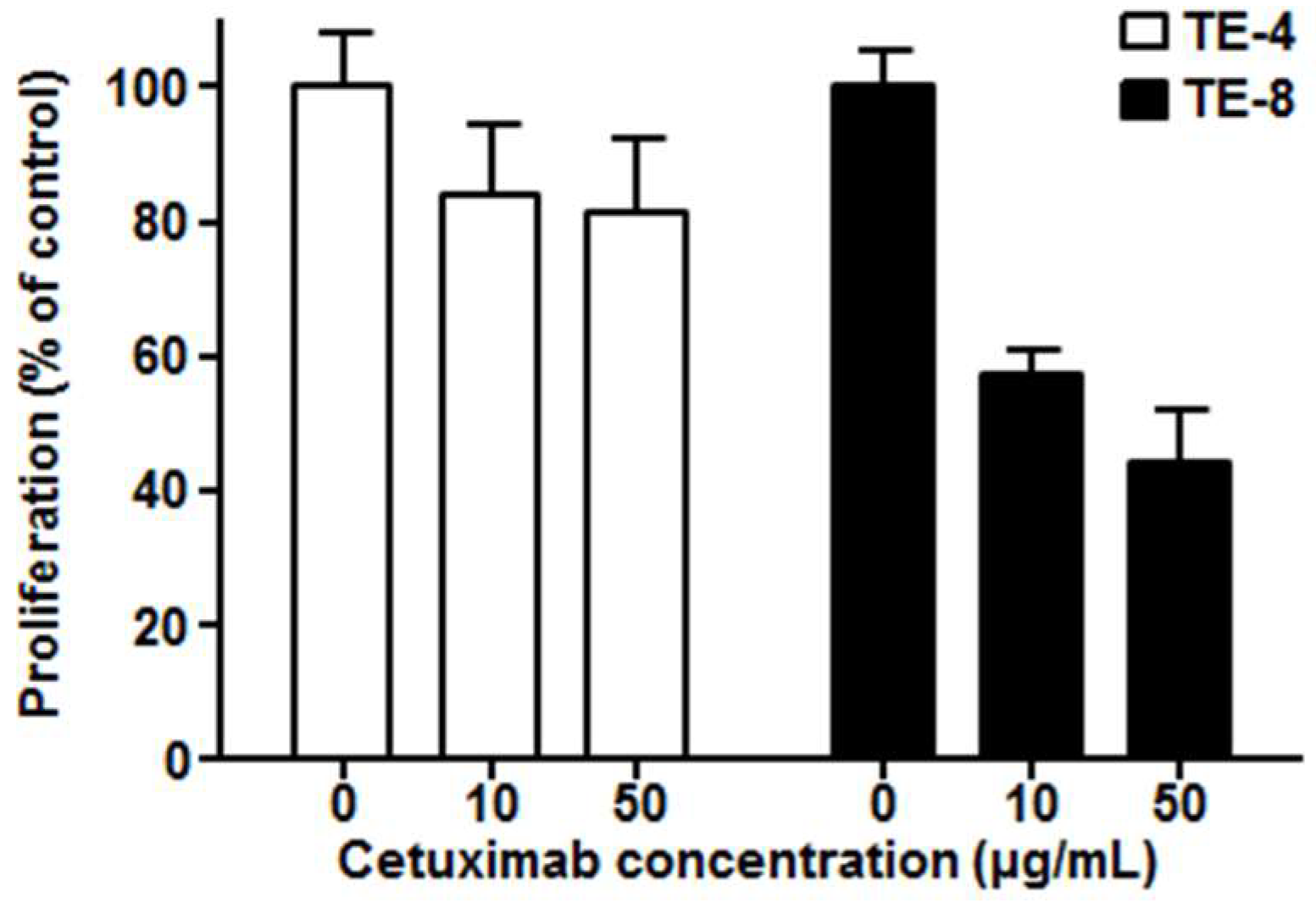
3.2. Cytotoxicity of Cetuximab in ESCC Cells
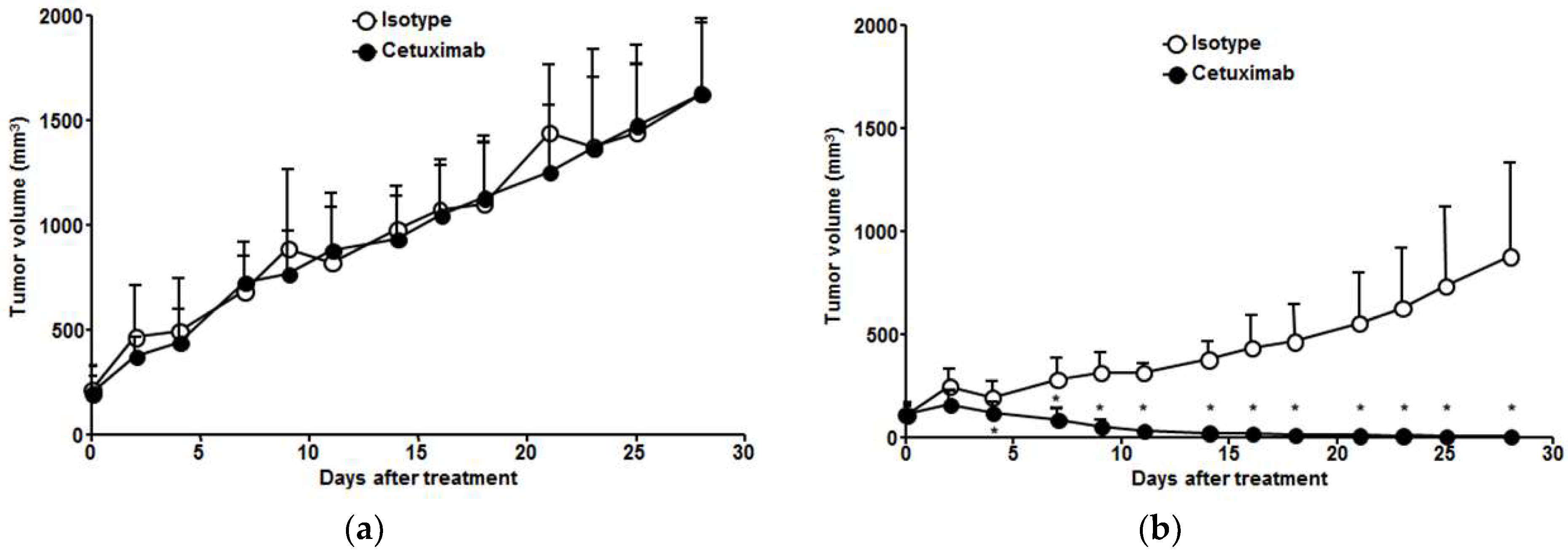
3.3. Therapeutic Effects of Cetuximab in ESCC Tumors
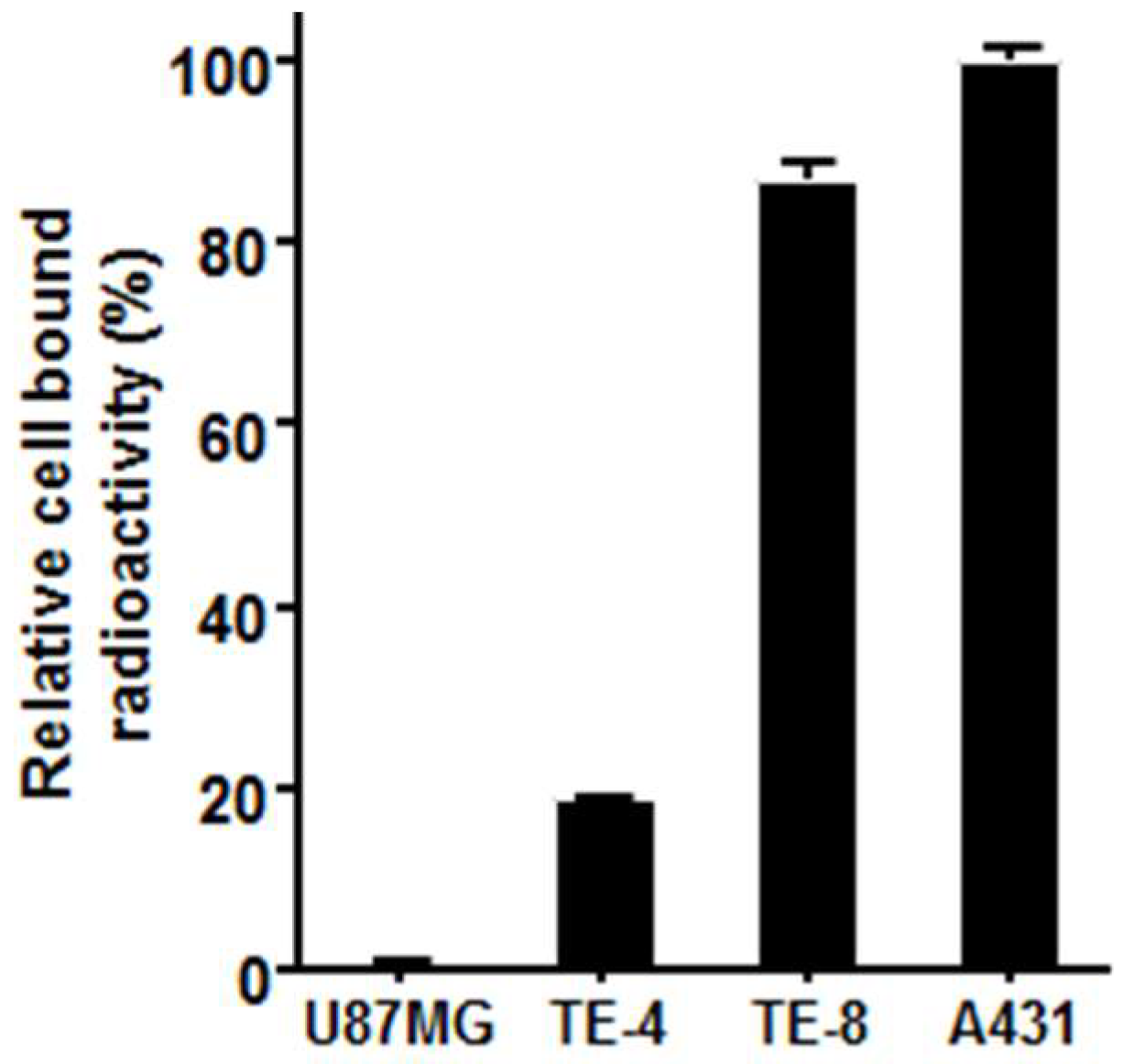
3.4. Characteristics of 64Cu-PCTA-Cetuximab
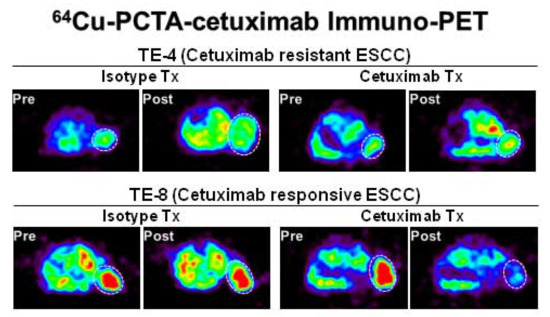
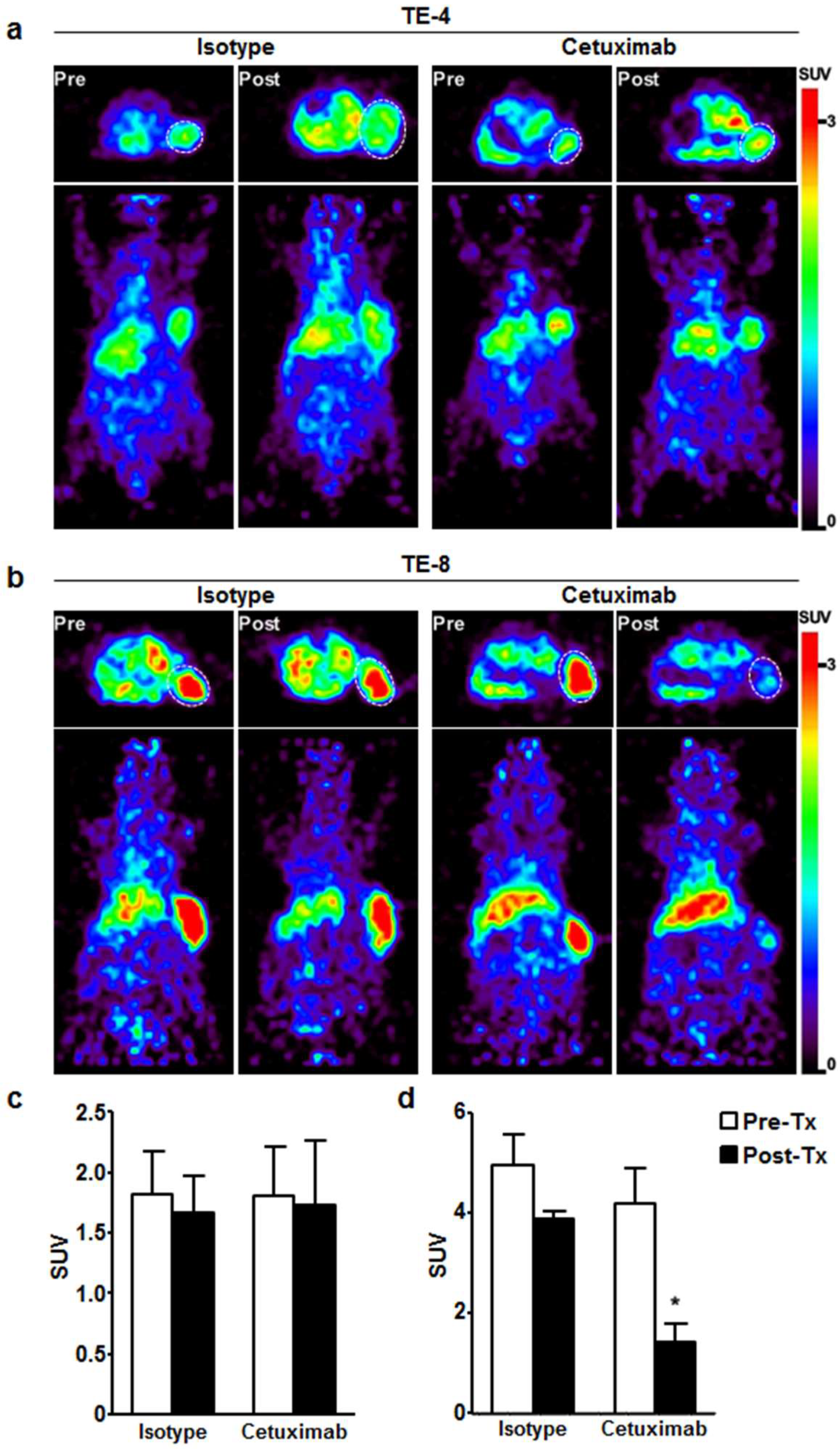
3.5. Immuno-PET Imaging of Cetuximab-Induced Antitumor Activity
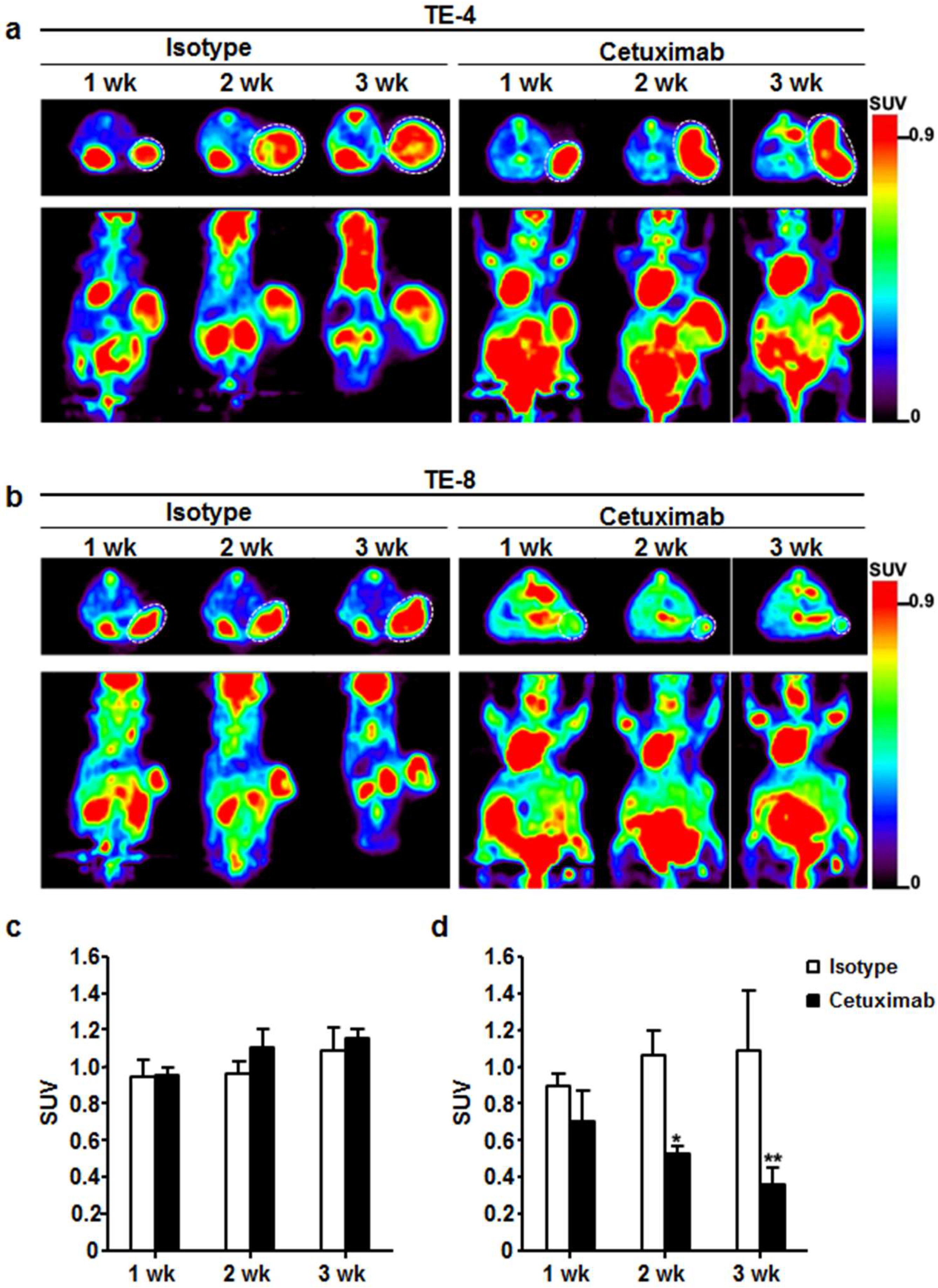
3.6. Monitoring Therapeutic Efficacy Using FDG-PET Imaging
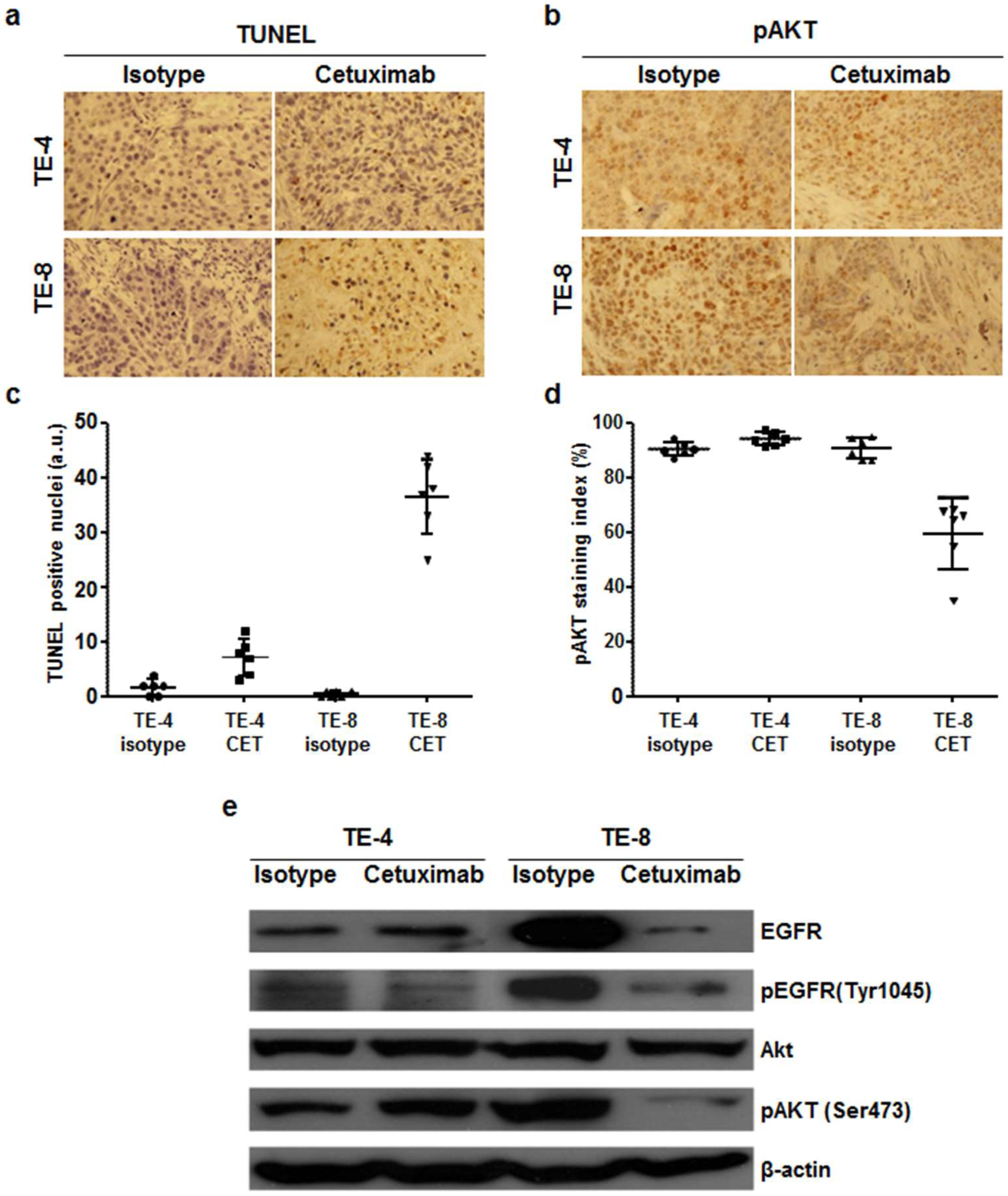
3.7. Cetuximab-Induced Apoptosis and Signaling Blockade
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Totsuka, Y.; He, Y.; Kikuchi, S.; Qiao, Y.; Ueda, J.; Wei, W.; Inoue, M.; Tanaka, H. Epidemiology of esophageal cancer in Japan and China. J. Epidemiol. 2013, 23, 233–242. [Google Scholar] [CrossRef] [PubMed]
- D’Journo, X.B.; Thomas, P.A. Current management of esophageal cancer. J. Thorac. Dis. 2014, 6, S253. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, T.; Kakegawa, T.; Ide, H.; Ando, N.; Watanabe, H.; Tanaka, O.; Takagi, I.; Isono, K.; Ishida, K.; Arimori, M.; et al. Phase II evaluation of cisplatin and 5-fluorouracil in advanced squamous cell carcinoma of the esophagus: A Japanese Esophageal Oncology Group Trial. Jpn. J. Clin. Oncol. 1992, 22, 172–176. [Google Scholar] [PubMed]
- Levard, H.; Pouliquen, X.; Hay, J.M.; Fingerhut, A.; Langlois-Zantain, O.; Huguier, M.; Lozach, P.; Testart, J. 5-Fluorouracil and cisplatin as palliative treatment of advanced oesophageal squamous cell carcinoma. A multicentre randomised controlled trial. The French Associations for Surgical Research. Eur. J. Surg. 1998, 164, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Muro, K.; Hamaguchi, T.; Ohtsu, A.; Boku, N.; Chin, K.; Hyodo, I.; Fujita, H.; Takiyama, W.; Ohtsu, T. A phase II study of single-agent docetaxel in patients with metastatic esophageal cancer. Ann. Oncol. 2004, 15, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Tahara, M.; Hironaka, S.; Muro, K.; Takiuchi, H.; Hamamoto, Y.; Imamoto, H.; Amano, N.; Seriu, T. A phase II study of paclitaxel by weekly 1-h infusion for advanced or recurrent esophageal cancer in patients who had previously received platinum-based chemotherapy. Cancer Chemother. Pharmacol. 2011, 67, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Enzinger, P.C.; Mayer, R.J. Esophageal cancer. N. Engl. J. Med. 2003, 349, 2241–2252. [Google Scholar] [CrossRef] [PubMed]
- Aklilu, M.; Ilson, D.H. Targeted agents and esophageal cancer--the next step? Semin. Radiat. Oncol. 2007, 17, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, R.I.; Gee, J.M.; Harper, M.E. EGFR and cancer prognosis. Eur. J. Cancer 2001, 37 (Suppl. 4), 9–15. [Google Scholar] [CrossRef]
- Hanawa, M.; Suzuki, S.; Dobashi, Y.; Yamane, T.; Kono, K.; Enomoto, N.; Ooi, A. EGFR protein overexpression and gene amplification in squamous cell carcinomas of the esophagus. Int. J. Cancer 2006, 118, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Sunpaweravong, P.; Sunpaweravong, S.; Puttawibul, P.; Mitarnun, W.; Zeng, C.; Baron, A.E.; Franklin, W.; Said, S.; Varella-Garcia, M. Epidermal growth factor receptor and cyclin D1 are independently amplified and overexpressed in esophageal squamous cell carcinoma. J. Cancer Res. Clin. Oncol. 2005, 131, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Boone, J.; van Hillegersberg, R.; Offerhaus, G.J.; van Diest, P.J.; Borel Rinkes, I.H.; Ten Kate, F.J. Targets for molecular therapy in esophageal squamous cell carcinoma: An immunohistochemical analysis. Dis. Esophagus 2009, 22, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhu, H.; Liu, X.; Wang, Q.; Zhang, X.; He, J.; Sun, K.; Liu, X.; Zhou, Z.; Xu, N.; et al. Epidermal growth factor receptor is a prognosis predictor in patients with esophageal squamous cell carcinoma. Ann. Thorac. Surg. 2014, 98, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Li, X.; Wang, H.; Shi, Y.; Xu, C.; Lu, S.; Huang, J.; Xu, Y.; Zeng, H.; Su, J.; et al. The prognostic value of EGFR overexpression and amplification in Esophageal squamous cell Carcinoma. BMC Cancer 2015, 15, 377. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, X.; Bu, S.; He, C.; Wang, W.; Liu, J.; Guo, W.; Tan, B.; Wang, Y.; Wang, J. Promising outcomes of definitive chemoradiation and cetuximab for patients with esophageal squamous cell carcinoma. Cancer Sci. 2012, 103, 1979–1984. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Lin, L.; Liu, J.; Liu, R.; Chen, Y.; Ge, F.; Jia, R.; Jin, Y.; Wang, Y.; Xu, J. A phase II study of concurrent chemoradiotherapy and erlotinib for inoperable esophageal squamous cell carcinoma. Oncotarget 2016, 7, 57310–57316. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zheng, Y.; Sun, X.; Yu, X.; Gu, J.; Wu, W.; Zhang, G.; Hu, J.; Sun, W.; Mao, W. Concurrent radiotherapy with gefitinib in elderly patients with esophageal squamous cell carcinoma: Preliminary results of a phase II study. Oncotarget 2015, 6, 38429–38439. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Niu, H.; Fan, Q.; Lu, P.; Ma, C.; Liu, W.; Liu, Y.; Li, W.; Hu, S.; Ling, Y.; et al. Predictive value of EGFR overexpression and gene amplification on icotinib efficacy in patients with advanced esophageal squamous cell carcinoma. Oncotarget 2016, 7, 24744–24751. [Google Scholar] [CrossRef] [PubMed]
- Waksal, H.W. Role of an anti-epidermal growth factor receptor in treating cancer. Cancer Metastasis Rev. 1999, 18, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, M.; Yamashita, Y.; Kubo, N.; Yashiro, M.; Ohira, M.; Ako, E.; Tanaka, H.; Muguruma, K.; Sawada, T.; Hirakawa, K. Concurrent biological targeting therapy of squamous cell carcinoma of the esophagus with cetuximab and trastuzumab. Oncol. Rep. 2012, 28, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, Y.; Kono, K.; Mimura, K.; Mitsui, F.; Sugai, H.; Akaike, H.; Fujii, H. Targeting EGFR and HER-2 with cetuximab- and trastuzumab-mediated immunotherapy in oesophageal squamous cell carcinoma. Br. J. Cancer 2007, 97, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, Y.; Kono, K.; Mimura, K.; Sugai, H.; Akaike, H.; Fujii, H. Cetuximab induce antibody-dependent cellular cytotoxicity against EGFR-expressing esophageal squamous cell carcinoma. Int. J. Cancer 2007, 120, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Schonnemann, K.R.; Yilmaz, M.; Bjerregaard, J.K.; Nielsen, K.M.; Pfeiffer, P. Phase II study of biweekly cetuximab in combination with irinotecan as second-line treatment in patients with platinum-resistant gastro-oesophageal cancer. Eur. J. Cancer 2012, 48, 510–517. [Google Scholar] [CrossRef] [PubMed]
- De Vita, F.; Orditura, M.; Martinelli, E.; Vecchione, L.; Innocenti, R.; Sileni, V.C.; Pinto, C.; Di Maio, M.; Farella, A.; Troiani, T.; et al. A multicenter phase II study of induction chemotherapy with FOLFOX-4 and cetuximab followed by radiation and cetuximab in locally advanced oesophageal cancer. Br. J. Cancer 2011, 104, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Ruhstaller, T.; Pless, M.; Dietrich, D.; Kranzbuehler, H.; von Moos, R.; Moosmann, P.; Montemurro, M.; Schneider, P.M.; Rauch, D.; Gautschi, O.; et al. Cetuximab in combination with chemoradiotherapy before surgery in patients with resectable, locally advanced esophageal carcinoma: A prospective, multicenter phase IB/II Trial (SAKK 75/06). J. Clin. Oncol. 2011, 29, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Raghu Subramanian, C.; Triadafilopoulos, G. Diagnosis and therapy of esophageal squamous cell dysplasia and early esophageal squamous cell cancer. Gastroenter. Rep. 2017, 5, 247–257. [Google Scholar] [CrossRef]
- Lamberts, L.E.; Williams, S.P.; Terwisscha van Scheltinga, A.G.; Lub-de Hooge, M.N.; Schroder, C.P.; Gietema, J.A.; Brouwers, A.H.; de Vries, E.G. Antibody positron emission tomography imaging in anticancer drug development. J. Clin. Oncol. 2015, 33, 1491–1504. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Chen, K.; He, L.; Cao, Q.; Koong, A.; Chen, X. Quantitative PET of EGFR expression in xenograft-bearing mice using 64Cu-labeled cetuximab, a chimeric anti-EGFR monoclonal antibody. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Eiblmaier, M.; Meyer, L.A.; Watson, M.A.; Fracasso, P.M.; Pike, L.J.; Anderson, C.J. Correlating EGFR expression with receptor-binding properties and internalization of 64Cu-DOTA-cetuximab in 5 cervical cancer cell lines. J. Nucl. Med. 2008, 49, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Rigo, P.; Paulus, P.; Kaschten, B.J.; Hustinx, R.; Bury, T.; Jerusalem, G.; Benoit, T.; Foidart-Willems, J. Oncological applications of positron emission tomography with fluorine-18 fluorodeoxyglucose. Eur. J. Nucl. Med. 1996, 23, 1641–1674. [Google Scholar] [CrossRef] [PubMed]
- Krause, B.J.; Herrmann, K.; Wieder, H.; zum Buschenfelde, C.M. 18F-FDG PET and 18F-FDG PET/CT for assessing response to therapy in esophageal cancer. J. Nucl. Med. 2009, 50 (Suppl. 1), 89S–96S. [Google Scholar] [CrossRef]
- Wieder, H.A.; Brucher, B.L.; Zimmermann, F.; Becker, K.; Lordick, F.; Beer, A.; Schwaiger, M.; Fink, U.; Siewert, J.R.; Stein, H.J.; et al. Time course of tumor metabolic activity during chemoradiotherapy of esophageal squamous cell carcinoma and response to treatment. J. Clin. Oncol. 2004, 22, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Ray, D.; Ahsan, A.; Helman, A.; Chen, G.; Hegde, A.; Gurjar, S.R.; Zhao, L.; Kiyokawa, H.; Beer, D.G.; Lawrence, T.S.; et al. Regulation of EGFR protein stability by the HECT-type ubiquitin ligase SMURF2. Neoplasia 2011, 13, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Song, I.H.; Lee, T.S.; Park, Y.S.; Lee, J.S.; Lee, B.C.; Moon, B.S.; An, G.I.; Lee, H.W.; Kim, K.I.; Lee, Y.J.; et al. Immuno-PET Imaging and Radioimmunotherapy of 64Cu-/177Lu-Labeled Anti-EGFR Antibody in Esophageal Squamous Cell Carcinoma Model. J. Nucl. Med. 2016, 57, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Tseng, J.R.; Dandekar, M.; Subbarayan, M.; Cheng, Z.; Park, J.M.; Louie, S.; Gambhir, S.S. Reproducibility of 3′-deoxy-3′-18F-fluorothymidine microPET studies in tumor xenografts in mice. J. Nucl. Med. 2005, 46, 1851–1857. [Google Scholar] [PubMed]
- Song, I.H.; Noh, Y.; Kwon, J.; Jung, J.H.; Lee, B.C.; Kim, K.I.; Lee, Y.J.; Kang, J.H.; Rhee, C.S.; Lee, C.H.; et al. Immuno-PET imaging based radioimmunotherapy in head and neck squamous cell carcinoma model. Oncotarget 2017, 8, 92090–92105. [Google Scholar] [CrossRef] [PubMed]
- Van Dongen, G.A.; Visser, G.W.; Lub-de Hooge, M.N.; de Vries, E.G.; Perk, L.R. Immuno-PET: A navigator in monoclonal antibody development and applications. Oncologist 2007, 12, 1379–1389. [Google Scholar] [CrossRef] [PubMed]
- Okines, A.; Cunningham, D.; Chau, I. Targeting the human EGFR family in esophagogastric cancer. Nat. Rev. Clin. Oncol. 2011, 8, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Sun, X.; Cao, Q.; Courter, D.; Koong, A.; Le, Q.T.; Gambhir, S.S.; Chen, X. Cetuximab-based immunotherapy and radioimmunotherapy of head and neck squamous cell carcinoma. Clin. Cancer Res. 2010, 16, 2095–2105. [Google Scholar] [CrossRef] [PubMed]
- Berger, A.K.; von Gall, C.; Abel, U.; Delorme, S.; Kloor, M.; Ose, J.; Weber, T.F.; Stange, A.; Haag, G.M.; Haberkorn, U.; et al. A phase II study for metabolic in vivo response monitoring with sequential 18FDG-PET-CT during treatment with the EGFR-monoclonal-antibody cetuximab in metastatic colorectal cancer: The Heidelberg REMOTUX trial. BMC Cancer 2012, 12, 108. [Google Scholar] [CrossRef] [PubMed]
- Smith-Jones, P.M.; Solit, D.; Afroze, F.; Rosen, N.; Larson, S.M. Early tumor response to Hsp90 therapy using HER2 PET: Comparison with 18F-FDG PET. J. Nucl. Med. 2006, 47, 793–796. [Google Scholar] [PubMed]
- Shah, C.; Miller, T.W.; Wyatt, S.K.; McKinley, E.T.; Olivares, M.G.; Sanchez, V.; Nolting, D.D.; Buck, J.R.; Zhao, P.; Ansari, M.S.; et al. Imaging biomarkers predict response to anti-HER2 (ErbB2) therapy in preclinical models of breast cancer. Clin. Cancer Res. 2009, 15, 4712–4721. [Google Scholar] [CrossRef] [PubMed]
- Yonesaka, K.; Zejnullahu, K.; Okamoto, I.; Satoh, T.; Cappuzzo, F.; Souglakos, J.; Ercan, D.; Rogers, A.; Roncalli, M.; Takeda, M.; et al. Activation of ERBB2 signaling causes resistance to the EGFR-directed therapeutic antibody cetuximab. Sci. Transl. Med. 2011, 3, 99ra86. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.L.; Huang, S.; Kruser, T.J.; Nechrebecki, M.M.; Armstrong, E.A.; Benavente, S.; Gondi, V.; Hsu, K.T.; Harari, P.M. Mechanisms of acquired resistance to cetuximab: Role of HER (ErbB) family members. Oncogene 2008, 27, 3944–3956. [Google Scholar] [CrossRef] [PubMed]
- Mimura, K.; Kono, K.; Hanawa, M.; Mitsui, F.; Sugai, H.; Miyagawa, N.; Ooi, A.; Fujii, H. Frequencies of HER-2/neu expression and gene amplification in patients with oesophageal squamous cell carcinoma. Br. J. Cancer 2005, 92, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Mimura, K.; Kono, K.; Maruyama, T.; Watanabe, M.; Izawa, S.; Shiba, S.; Mizukami, Y.; Kawaguchi, Y.; Inoue, M.; Kono, T.; et al. Lapatinib inhibits receptor phosphorylation and cell growth and enhances antibody-dependent cellular cytotoxicity of EGFR- and HER2-overexpressing esophageal cancer cell lines. Int. J. Cancer 2011, 129, 2408–2416. [Google Scholar] [CrossRef] [PubMed]
- Hara, F.; Aoe, M.; Doihara, H.; Taira, N.; Shien, T.; Takahashi, H.; Yoshitomi, S.; Tsukuda, K.; Toyooka, S.; Ohta, T.; et al. Antitumor effect of gefitinib (‘Iressa’) on esophageal squamous cell carcinoma cell lines in vitro and in vivo. Cancer Lett. 2005, 226, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.C.; Hao, J.J.; Nagata, Y.; Xu, L.; Shang, L.; Meng, X.; Sato, Y.; Okuno, Y.; Varela, A.M.; Ding, L.W.; et al. Genomic and molecular characterization of esophageal squamous cell carcinoma. Nat. Genet. 2014, 46, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.R.; Yang, Z.; Dong, H.; Camuso, A.; McGlinchey, K.; Fager, K.; Flefleh, C.; Kan, D.; Inigo, I.; Castaneda, S.; et al. Correlation of pharmacokinetics with the antitumor activity of Cetuximab in nude mice bearing the GEO human colon carcinoma xenograft. Cancer Chemother. Pharmacol. 2005, 56, 455–464. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, T.S.; Song, I.H.; Shin, J.I.; Park, Y.S.; Kim, J.Y.; Kim, K.I.; Lee, Y.J.; Kang, J.H. PET Imaging Biomarkers of Anti-EGFR Immunotherapy in Esophageal Squamous Cell Carcinoma Models. Cells 2018, 7, 187. https://doi.org/10.3390/cells7110187
Lee TS, Song IH, Shin JI, Park YS, Kim JY, Kim KI, Lee YJ, Kang JH. PET Imaging Biomarkers of Anti-EGFR Immunotherapy in Esophageal Squamous Cell Carcinoma Models. Cells. 2018; 7(11):187. https://doi.org/10.3390/cells7110187
Chicago/Turabian StyleLee, Tae Sup, In Ho Song, Jong Il Shin, Yong Serk Park, Jung Young Kim, Kwang Il Kim, Yong Jin Lee, and Joo Hyun Kang. 2018. "PET Imaging Biomarkers of Anti-EGFR Immunotherapy in Esophageal Squamous Cell Carcinoma Models" Cells 7, no. 11: 187. https://doi.org/10.3390/cells7110187
APA StyleLee, T. S., Song, I. H., Shin, J. I., Park, Y. S., Kim, J. Y., Kim, K. I., Lee, Y. J., & Kang, J. H. (2018). PET Imaging Biomarkers of Anti-EGFR Immunotherapy in Esophageal Squamous Cell Carcinoma Models. Cells, 7(11), 187. https://doi.org/10.3390/cells7110187