Radiation Without Borders: Unraveling Bystander and Non-Targeted Effects in Oncology
Highlights
- Radiation-induced bystander effects (BEs) and non-targeted effects (NTEs) extend biological consequences beyond irradiated tissues, involving complex intercellular signaling mechanisms such as gap junctions, cytokines, extracellular vesicles, and oxidative stress.
- Clinically, BEs contributes to systemic toxicities and long-term complications, including neurocognitive decline, cardiovascular disease, pulmonary fibrosis, gastrointestinal injury, and secondary malignancies, especially in pediatric and long-term cancer survivors.
- Radiotherapy planning must evolve from a localized to a systemic perspective, incorporating strategies to recognize and mitigate BEs to improve therapeutic efficacy and survivorship outcomes.
- Targeted countermeasures such as antioxidants, COX-2 inhibitors, exosome blockers, and FLASH RT offer promising avenues to reduce off-target damage, suggesting that BEs is a modifiable axis of RT biology with translational potential.
Abstract
1. Radiation in Cancer Treatment
| Risk Category | Examples | Typical Onset | Ref. |
|---|---|---|---|
| Acute toxicities | Dermatitis, mucositis, diarrhea, nausea, fatigue, skin desquamation, oral ulcers, hair loss, low blood counts | During/soon after RT | [45,46,47] |
| Delayed toxicities | Pulmonary fibrosis, enteritis, xerostomia, chronic diarrhea, lymphedema, fibrosis, osteoradionecrosis, hypothyroidism, telangiectasia | Month-years post-RT | [46,48,49] |
| Neurocognitive effects | Memory loss, impaired processing, attention deficits, executive dysfunction | Late | [46,47,49] |
| Secondary cancers | Sarcomas, leukemias, carcinomas | Years/decades later | [47] |
| Normal tissue toxicities | Cardiotoxicity, pneumonitis, hypothyroidism, chronic cystitis, infertility, bowel/bladder dysfunction | Acute/Late | [46,47,48,49] |
| Secondary cellular responses | Genomic instability, chronic inflammation | Acute/Late | [49,50] |
| Radio resistance | Increased recurrence, molecular mayhem | Any time post-RT | [49,50] |
| Immunosuppression | Lymphopenia, increased infection risk, impaired T-cell function | Acute/Late | [47,49,50] |
| Musculoskeletal effects | Muscle fibrosis, joint stiffness, limited mobility, and bone fractures | Late | [46,47,49] |
| Lymphedema | Swelling of neck, arms, or legs; heavy/achy limbs | Early/Late | [47] |
| Skin changes | Redness, dryness, pigmentation changes, ulceration, chronic scarring | Acute/Late | [47] |
| Gastrointestinal effects | Nausea, diarrhea, proctitis, malabsorption, rectal bleeding, pain | Acute/Late | [45,46,47] |
| Genitourinary/reproductive effects | Bladder irritation, cystitis, incontinence, infertility, erectile dysfunction, menstrual changes | Acute/Late | [46,47,49] |
| Fatigue | Daily tiredness, weakness, reduced activity levels | Acute/Late | [47] |
| Psychosocial effects | Depression, anxiety, and body image disturbance | Any phase | [47,48,51] |
| Rare, serious complications | Tissue necrosis, catastrophic bleeding, and organ failure | Late | [46,48] |
2. Unintended and Non-Targeted Biological Effects of RT: Radiation Bystander Effects (BEs)
3. Molecular Signaling Mechanisms That Coordinate Radiation BEs
3.1. Immediate Early, Stress and Survival Signaling
3.2. Redox Imbalance and Metabolic Rewiring
3.3. DNA Damage, Epigenetics, and Chromatin Remodeling
3.4. Immune Response and Inflammation
3.5. Stem Cell Niche, TME, and Systemic Effects
3.6. Unique and Emerging Mechanisms
3.7. Mechanistic Diversity in Radiation BEs
3.8. Rationale and Impact of In Vitro Findings
3.9. Prioritization and Translational Value of Animal and Human Studies
3.10. Radiation Quality and Radiation BEs
3.11. Dose-Rate Modulation of Radiation BEs
4. Clinical Implications and Significance of Radiation BEs
5. Countermeasures for Radiation BEs
5.1. Cyclooxygenase-2 (COX-2) Inhibitors
5.2. Antioxidant Therapies
5.3. Melatonin and Related Compounds
5.4. Statins, Metformin, and Herbal Extracts
5.5. Cell-Free Chromatin (cfCh) Degradation Strategies
5.6. Targeting microRNAs and Exosome-Mediated Signaling
5.7. Mitochondrial and Metabolic Regulators
5.8. Inflammasome and Immune Targets
5.9. Senescence and SASP-Mediated Bystander Signaling
5.10. Precision and Personalized Countermeasures
5.11. Emerging Translational Approaches
5.12. Radiopharmaceutical Therapy: High-LET Microdosimetry, Systemic Biodistribution, and BEs/Abscopal Biology
5.13. Survivorship and Long-Term Monitoring
5.14. FLASH-RT Minimize Radiation BEs
5.15. Advanced Particle Therapies
5.16. Spatially Fractionated and Lattice Radiotherapy
5.17. Fractionation Schedules and Dose Rate Modulation
5.18. Shielding Non-Targeted Tissues
5.19. Clinical Imperative and Technological Innovations
5.20. Converging on Exosome Biogenesis and Uptake
5.21. Degradation of cfCh and DAMPs
5.22. Dual Inhibition—Crosstalk Between Pathways
5.23. Population-Level and Occupational Exposure Considerations
5.24. Radiation BEs Is Not Always Bad
6. Experts’ Opinion
6.1. Radiation BEs Are Defined and Not Stochastic
6.2. Radiation BEs Inflicts Systemic and Lasting Consequences
6.3. Radiation BEs Are Radiation-, System- and Disease-Specific
6.4. Molecular-Targeted Radioprotectors Are Superior over General/Global Agents for Radiation BEs
7. Conclusions
8. Methodological Transparency: Criteria for the Selection of Studies
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| 3D-CRT | Three-dimensional Conformal Radiotherapy |
| ADAR1 | Adenosine Deaminase Acting on RNA 1 |
| AI | Artificial Intelligence |
| AMPK | AMP-activated Protein Kinase |
| ASC | Apoptosis-associated Speck-like protein containing a CARD |
| α-SMA | Alpha-smooth Muscle Actin |
| ATM | Ataxia-telangiectasia Mutated |
| ATR | Ataxia Telangiectasia and Rad3-related |
| BCCSS | British Childhood Cancer Survivor Study |
| BEs | Bystander Effects |
| BRAC1 | Breast Cancer Type 1 |
| Caspase-1 | Cysteine-aspartic protease 1 |
| CCSS | Childhood Cancer Survivor Study |
| cfCh | Cell-free Chromatin |
| cGAS | Cyclic GMP-AMP Synthase |
| Chk1 | Checkpoint Kinase 1 |
| circRNA | Circular RNA |
| CNPs | Chromatin-Neutralizing Nanoparticles |
| CNS | Central Nervous System |
| CT | Computed Tomography |
| CTLA-4 | Cytotoxic T-Lymphocyte-Associated Protein 4 |
| DAMP | Damage-associated Molecular Pattern |
| DDR | DNA Damage Response |
| DFS | Disease-free Survival |
| DNA-PK | DNA-dependent Protein Kinase |
| DR5 | Death Receptor 5 |
| DVH | Dose-volume Histograms |
| ECM | Extracellular Matrix |
| EGFR | Epidermal Growth Factor Receptor |
| ERK | Extracellular signal-regulated kinase |
| EV | Extracellular vesicles |
| FasL | Fas Ligand |
| FGFR | Fibroblast Growth Factor Receptor |
| FMT | Fecal Microbiota Transplantation |
| GI | Gastrointestinal |
| GJIC | Gap junction intercellular communication |
| GPX1 | Glutathione Peroxidase 1 |
| GRP75 | Glucose-Regulated Protein 75 |
| GST | Glutathione S-transferase |
| H2AX | Histone H2A variant X |
| HIF-1a | Hypoxia-Inducible Factor 1-alpha |
| HMGB1 | High Mobility Group Box 1 |
| HOTAIR | HOX Transcript Antisense RNA |
| HSP | Heat Shock Proteins (e.g., HSP70, HSP90) |
| ICIs | Immune Checkpoint Inhibitors |
| IGRT | Image-guided Radiotherapy |
| IL-18 | Interleukin-18 |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IMRT | Intensity-modulated Radiotherapy |
| iNOS | Inducible Nitric Oxide Synthase |
| IORT | Intraoperative Radiotherapy |
| IP3R | Inositol 1,4,5-trisphosphate receptor |
| IR | Ionizing Radiation |
| IRF3 | Interferon Regulatory Factor 3 |
| JNK | c-Jun N-terminal Kinase |
| Ku70/Ku80 | DNA repair proteins |
| LC3B | Microtubule-associated protein 1A/1B-light chain 3 |
| LLPS | Liquid–Liquid Phase Separation |
| lncRNA | Long Non-Coding RNA |
| MAM | Mitochondria-Associated ER Membrane |
| MAPK | Mitogen-Activated Protein Kinase |
| MCT1 | Monocarboxylate Transporter 1 |
| MFN2 | Mitofusin 2 |
| miRNA | MicroRNA |
| MMP | Matrix Metalloproteinases |
| MRI | Magnetic Resonance Imaging |
| mTOR | Mechanistic Target of Rapamycin |
| NCS | Neural Stem Cells |
| NFκB | Nuclear Factor kappa-light-chain-enhancer of activated B cells |
| NK | Natural Killer |
| NLRP3 | NOD-, LRR- and pyrin domain-containing protein 3 |
| NO | Nitric Oxide |
| NOX | NADPH Oxidase |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| NTEs | Non-targeted Effects |
| NT-proBNP | N-terminal pro-B-type Natriuretic Peptide |
| PARKIN | E3 ubiquitin-protein ligase Parkin |
| PARP | Poly (ADP-ribose) Polymerase |
| PBT | Proton Beam Therapy |
| PD-1/PD-L1 | Programmed Death-1/Programmed Death Ligand-1 |
| PDGFR | Platelet-Derived Growth Factor Receptor |
| PINK1 | PTEN-induced kinase 1 |
| PKR | Protein Kinase R |
| Rad51 | DNA repair protein RAD51 homolog 1 |
| RBE | Relative Biological Effectiveness |
| REDD1 | Regulated in Development and DNA Damage Responses 1 |
| RIGI | Radiation Induced Genomic Instability |
| RNS | Reactive Nitrogen Species |
| ROS | Reactive Oxygen Species |
| RPT | Radiopharmaceutical Therapy |
| RT | Radiotherapy |
| S1P | Sphingosine-1-Phosphate |
| SBRT | Stereotactic Body Radiotherapy |
| SNP | Single Nucleotide Polymorphisms |
| SOD2 | Superoxide Dismutase 2 |
| SRS | Stereotactic Radiosurgery |
| STING | Stimulator of Interferon Genes |
| TAMs | Tumor-Associated Macrophages |
| TBK1 | TANK-binding Kinase 1 |
| TGF-β | Transforming Growth Factor Beta |
| TIMP | Tissue Inhibitor of Metalloproteinases |
| TLR | Toll-like Receptors |
| TME | Tumor microenvironment |
| TNF-α | Tumor Necrosis Factor |
| TNTs | Tunneling Nanotubes |
| TRAIL | TNF-related apoptosis-inducing ligand |
| UPR | Unfolded Protein Response |
| VDAC1 | Voltage-dependent anion-selective channel protein 1 |
| VEGF | Vascular Endothelial Growth Factor |
| VMAT | Volumetric Modulated Arc Therapy |
| XRCC1 | X-ray Repair Cross-Complementing 1 |
| YAP/TAZ | Yes-associated protein/Transcriptional coactivator with PDZ-binding motif |
References
- Siegel, R.L.; Kratzer, T.B.; Giaquinto, A.N.; Sung, H.; Jemal, A. Cancer statistics, 2025. CA Cancer J. Clin. Flagship J. Am. Cancer Soc. 2025, 75, 10–45. [Google Scholar] [CrossRef]
- Wang, M.; Yu, F.; Zhang, Y. Present and future of cancer nano-immunotherapy: Opportunities, obstacles and challenges. Mol. Cancer 2025, 24, 26. [Google Scholar] [CrossRef]
- Baskar, R.; Yap, S.P.; Chua, K.L.M.; Itahana, K. The diverse and complex roles of radiation on cancer treatment: Therapeutic target and genome maintenance. Am. J. Cancer Res. 2012, 2, 372. [Google Scholar] [PubMed]
- Wang, J.-s.; Wang, H.-j.; Qian, H.-l. Biological effects of radiation on cancer cells. Mil. Med. Res. 2018, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Lee, K.A.; Yeo, R.; Yeoh, K.-W. Cancer and radiation therapy: Current advances and future directions. Int. J. Med. Sci. 2012, 9, 193. [Google Scholar] [CrossRef] [PubMed]
- Grégoire, V.; Guckenberger, M.; Haustermans, K.; Lagendijk, J.J.; Ménard, C.; Pötter, R.; Slotman, B.J.; Tanderup, K.; Thorwarth, D.; Van Herk, M. Image guidance in radiation therapy for better cure of cancer. Mol. Oncol. 2020, 14, 1470–1491. [Google Scholar] [CrossRef]
- Chen, H.H.; Kuo, M.T. Improving radiotherapy in cancer treatment: Promises and challenges. Oncotarget 2017, 8, 62742. [Google Scholar] [CrossRef]
- Hanna, T.; Shafiq, J.; Delaney, G.; Vinod, S.; Thompson, S.; Barton, M. The population benefit of evidence-based radiotherapy: 5-Year local control and overall survival benefits. Radiother. Oncol. 2018, 126, 191–197. [Google Scholar] [CrossRef]
- Bentzen, S.M. Preventing or reducing late side effects of radiation therapy: Radiobiology meets molecular pathology. Nat. Rev. Cancer 2006, 6, 702–713. [Google Scholar] [CrossRef]
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment: Estimating optimal utilization from a review of evidence-based clinical guidelines. Cancer Interdiscip. Int. J. Am. Cancer Soc. 2005, 104, 1129–1137. [Google Scholar] [CrossRef]
- Al-Absi, E.; Farrokhyar, F.; Sharma, R.; Whelan, K.; Corbett, T.; Patel, M.; Ghert, M. A systematic review and meta-analysis of oncologic outcomes of pre-versus postoperative radiation in localized resectable soft-tissue sarcoma. Ann. Surg. Oncol. 2010, 17, 1367–1374. [Google Scholar] [CrossRef]
- Baskar, R.; Dai, J.; Wenlong, N.; Yeo, R.; Yeoh, K.-W. Biological response of cancer cells to radiation treatment. Front. Mol. Biosci. 2014, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, X.; Chen, D.; Yu, J. Radiotherapy combined with immunotherapy: The dawn of cancer treatment. Signal Transduct. Target. Ther. 2022, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Formenti, S.C. Radiotherapy effects on anti-tumor immunity: Implications for cancer treatment. Front. Oncol. 2013, 3, 128. [Google Scholar] [CrossRef]
- Lutz, S.; Balboni, T.; Jones, J.; Lo, S.; Petit, J.; Rich, S.E.; Wong, R.; Hahn, C. Palliative radiation therapy for bone metastases: Update of an ASTRO evidence-based guideline. Pract. Radiat. Oncol. 2017, 7, 4–12. [Google Scholar] [CrossRef]
- Chow, E.; Zeng, L.; Salvo, N.; Dennis, K.; Tsao, M.; Lutz, S. Update on the systematic review of palliative radiotherapy trials for bone metastases. Clin. Oncol. 2012, 24, 112–124. [Google Scholar] [CrossRef]
- Lutz, S.T.; Jones, J.; Chow, E. Role of radiation therapy in palliative care of the patient with cancer. J. Clin. Oncol. 2014, 32, 2913–2919. [Google Scholar] [CrossRef] [PubMed]
- Fiorino, C.; Guckenberger, M.; Schwarz, M.; van der Heide, U.A.; Heijmen, B. Technology-driven research for radiotherapy innovation. Mol. Oncol. 2020, 14, 1500–1513. [Google Scholar] [CrossRef]
- Vollmering, K. What Are the Types of Radiation Therapy Used for Cancer Treatment; The University of Texas, MD Anderson Cancer Centre: Houston, TX, USA, 2021. [Google Scholar]
- Runham, J.; McDowall, W.; Bryant, D.; Martin, J. A 3D conformal radiation therapy class solution for dose escalated prostate irradiation. Radiographer 2008, 55, 13–17. [Google Scholar] [CrossRef]
- Vaarkamp, J.; Malde, R.; Dixit, S.; Hamilton, C. A comparison of conformal and intensity modulated treatment planning techniques for early prostate cancer. J. Med. Imaging Radiat. Oncol. 2009, 53, 310–317. [Google Scholar] [CrossRef]
- García-Figueiras, R.; Baleato-González, S.; Luna, A.; Padhani, A.R.; Vilanova, J.C.; Carballo-Castro, A.M.; Oleaga-Zufiria, L.; Vallejo-Casas, J.A.; Marhuenda, A.; Gómez-Caamaño, A. How imaging advances are defining the future of precision radiation therapy. Radiographics 2024, 44, e230152. [Google Scholar] [CrossRef]
- Alvarez-Moret, J.; Pohl, F.; Koelbl, O.; Dobler, B. Evaluation of volumetric modulated arc therapy (VMAT) with Oncentra MasterPlan® for the treatment of head and neck cancer. Radiat. Oncol. 2010, 5, 110. [Google Scholar] [CrossRef]
- Dietrich, A.; Koi, L.; Zöphel, K.; Sihver, W.; Kotzerke, J.; Baumann, M.; Krause, M. Improving external beam radiotherapy by combination with internal irradiation. Br. J. Radiol. 2015, 88, 20150042. [Google Scholar] [CrossRef]
- Nitipir, C.; Niculae, D.; Orlov, C.; Barbu, M.A.; Popescu, B.; Popa, A.M.; Stoian Pantea, A.M.; Stanciu, A.E.; Galateanu, B.; Ginghina, O. Update on radionuclide therapy in oncology. Oncol. Lett. 2017, 14, 7011–7015. [Google Scholar] [CrossRef]
- Chargari, C.; Deutsch, E.; Blanchard, P.; Gouy, S.; Martelli, H.; Guérin, F.; Dumas, I.; Bossi, A.; Morice, P.; Viswanathan, A.N. Brachytherapy: An overview for clinicians. CA A Cancer J. Clin. 2019, 69, 386–401. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, C. Brachytherapy: Basic concepts, current clinical indications and future perspectives. Rev. Oncol. 2002, 4, 512–516. [Google Scholar] [CrossRef]
- Guo, S.; Reddy, C.A.; Kolar, M.; Woody, N.; Mahadevan, A.; Deibel, F.C.; Dietz, D.W.; Remzi, F.H.; Suh, J.H. Intraoperative radiation therapy with the photon radiosurgery system in locally advanced and recurrent rectal cancer: Retrospective review of the Cleveland clinic experience. Radiat. Oncol. 2012, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Krengli, M.; Pisani, C.; Deantonio, L.; Surico, D.; Volpe, A.; Surico, N.; Terrone, C. Intraoperative radiotherapy in gynaecological and genito-urinary malignancies: Focus on endometrial, cervical, renal, bladder and prostate cancers. Radiat. Oncol. 2017, 12, 18. [Google Scholar] [CrossRef]
- Biswas, T.; Okunieff, P.; Schell, M.C.; Smudzin, T.; Pilcher, W.H.; Bakos, R.S.; Vates, G.E.; Walter, K.A.; Wensel, A.; Korones, D.N. Stereotactic radiosurgery for glioblastoma: Retrospective analysis. Radiat. Oncol. 2009, 4, 11. [Google Scholar] [CrossRef]
- Nieder, C.; Grosu, A.L.; Gaspar, L.E. Stereotactic radiosurgery (SRS) for brain metastases: A systematic review. Radiat. Oncol. 2014, 9, 155. [Google Scholar] [CrossRef]
- Hu, M.; Jiang, L.; Cui, X.; Zhang, J.; Yu, J. Proton beam therapy for cancer in the era of precision medicine. J. Hematol. Oncol. 2018, 11, 136. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.-Z.; Zhan, Z.-J.; Qian, C.-N. New frontiers in proton therapy: Applications in cancers. Cancer Commun. 2019, 39, 61. [Google Scholar] [CrossRef] [PubMed]
- Sardaro, A.; Carbonara, R.; Petruzzelli, M.F.; Turi, B.; Moschetta, M.; Scardapane, A.; Stabile Ianora, A.A. Proton therapy in the most common pediatric non-central nervous system malignancies: An overview of clinical and dosimetric outcomes. Ital. J. Pediatr. 2019, 45, 170. [Google Scholar] [CrossRef]
- Corradini, S.; Alongi, F.; Andratschke, N.; Belka, C.; Boldrini, L.; Cellini, F.; Debus, J.; Guckenberger, M.; Hörner-Rieber, J.; Lagerwaard, F. MR-guidance in clinical reality: Current treatment challenges and future perspectives. Radiat. Oncol. 2019, 14, 92. [Google Scholar] [CrossRef]
- Timmerman, R.D.; Herman, J.; Cho, L.C. Emergence of stereotactic body radiation therapy and its impact on current and future clinical practice. J. Clin. Oncol. 2014, 32, 2847–2854. [Google Scholar] [CrossRef]
- Boucher, S.; Esarey, E.; Geddes, C.; Johnstone, C.; Kutsaev, S.; Loo, B.W., Jr.; Méot, F.; Mustapha, B.; Nakamura, K.; Nanni, E. Transformative technology for FLASH radiation therapy: A snowmass 2021 white paper. arXiv 2022, arXiv:2203.11047. [Google Scholar] [CrossRef]
- Favaudon, V.; Caplier, L.; Monceau, V.; Pouzoulet, F.; Sayarath, M.; Fouillade, C.; Poupon, M.-F.; Brito, I.; Hupé, P.; Bourhis, J. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci. Transl. Med. 2014, 6, ra245–ra293. [Google Scholar] [CrossRef] [PubMed]
- Pouget, J.-P.; Georgakilas, A.G.; Ravanat, J.-L. Targeted and off-target (bystander and abscopal) effects of radiation therapy: Redox mechanisms and risk/benefit analysis. Antioxid. Redox Signal. 2018, 29, 1447–1487. [Google Scholar] [CrossRef]
- Emran, A.A.; Marzese, D.M.; Menon, D.R.; Hammerlindl, H.; Ahmed, F.; Richtig, E.; Duijf, P.; Hoon, D.S.; Schaider, H. Commonly integrated epigenetic modifications of differentially expressed genes lead to adaptive resistance in cancer. Epigenomics 2019, 11, 732–737. [Google Scholar] [CrossRef]
- Travis, L.B.; Ng, A.K.; Allan, J.M.; Pui, C.-H.; Kennedy, A.R.; Xu, X.G.; Purdy, J.A.; Applegate, K.; Yahalom, J.; Constine, L.S. Second malignant neoplasms and cardiovascular disease following radiotherapy. J. Natl. Cancer Inst. 2012, 104, 357–370. [Google Scholar] [CrossRef]
- Wang, C.; Kishan, A.U.; James, B.Y.; Raldow, A.; King, C.R.; Iwamoto, K.S.; Chu, F.-I.; Steinberg, M.L.; Kupelian, P.A. Association between long-term second malignancy risk and radiation: A comprehensive analysis of the entire Surveillance, Epidemiology, and End Results Database (1973–2014). Adv. Radiat. Oncol. 2019, 4, 738–747. [Google Scholar] [CrossRef] [PubMed]
- Daguenet, E.; Louati, S.; Wozny, A.-S.; Vial, N.; Gras, M.; Guy, J.-B.; Vallard, A.; Rodriguez-Lafrasse, C.; Magné, N. Radiation-induced bystander and abscopal effects: Important lessons from preclinical models. Br. J. Cancer 2020, 123, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Cai, L.; He, X.; Niu, Z.; Huang, H.; Hu, W.; Bian, H.; Huang, H. Radiation-induced bystander effect and its clinical implications. Front. Oncol. 2023, 13, 1124412. [Google Scholar] [CrossRef] [PubMed]
- Berkey, F.J. Managing the Adverse Effects of Radiation Therapy. Am. Fam. Physician 2010, 82, 381–388. [Google Scholar]
- Fendler, W.; Tomasik, B.; Atkins, K.; Stawiski, K.; Chałubińska-Fendler, J.; Kozono, D. The clinician’s guide to radiotherapy complications. Pol. Arch. Intern. Med. 2022, 132, 16190. [Google Scholar] [CrossRef]
- Chaput, G.; Regnier, L. Radiotherapy: Clinical pearls for primary care. Can. Fam. Physician 2021, 67, 753–757. [Google Scholar] [CrossRef]
- Dilalla, V.; Chaput, G.; Williams, T.; Sultanem, K. Radiotherapy side effects: Integrating a survivorship clinical lens to better serve patients. Curr. Oncol. 2020, 27, 107–112. [Google Scholar] [CrossRef]
- Wang, K.; Tepper, J.E. Radiation therapy-associated toxicity: Etiology, management, and prevention. CA A Cancer J. Clin. 2021, 71, 437–454. [Google Scholar] [CrossRef]
- Verginadis, I.I.; Citrin, D.E.; Ky, B.; Feigenberg, S.J.; Georgakilas, A.G.; Hill-Kayser, C.E.; Koumenis, C.; Maity, A.; Bradley, J.D.; Lin, A. Radiotherapy toxicities: Mechanisms, management, and future directions. Lancet 2025, 405, 338–352. [Google Scholar] [CrossRef]
- Atia Elasrag, G.A.E.; Alshammari, M.S.S.; Ahmed Ouda, M.M.; Alqadi, R.A.; Hendy, A.; Hendy, A.; Gawad Sallam, S.A.E. Impact of Educational Guidelines on Radiotherapy Side Effects and Lifestyle in Patients With Advanced Head and Neck Cancer. SAGE Open Nurs. 2025, 11, 23779608251317809. [Google Scholar] [CrossRef]
- Morgan, W.F. Non-targeted and delayed effects of exposure to ionizing radiation: I. Radiation-induced genomic instability and bystander effects in vitro. Radiat. Res. 2003, 159, 567–580. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects: Past history and future directions. Radiat. Res. 2001, 155, 759–767. [Google Scholar] [CrossRef]
- Nagasawa, H.; Little, J.B. Induction of sister chromatid exchanges by extremely low doses of α-particles. Cancer Res. 1992, 52, 6394–6396. [Google Scholar] [PubMed]
- Mothersill, C.; Seymour, C. Radiation-induced bystander and other non-targeted effects: Novel intervention points in cancer therapy? Curr. Cancer Drug Targets 2006, 6, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.; Hei, T.K. Radiation induced bystander effect in vivo. Acta Medica Nagasaki. 2009, 53, S65–S69. [Google Scholar]
- Seymour, C.B.; Mothersill, C. Radiation-induced bystander effects—Implications for cancer. Nat. Rev. Cancer 2004, 4, 158–164. [Google Scholar] [CrossRef]
- Prise, K.M.; O’sullivan, J.M. Radiation-induced bystander signalling in cancer therapy. Nat. Rev. Cancer 2009, 9, 351–360. [Google Scholar] [CrossRef]
- Azzam, E.I.; De Toledo, S.M.; Little, J.B. Direct evidence for the participation of gap junction-mediated intercellular communication in the transmission of damage signals from α-particle irradiated to nonirradiated cells. Proc. Natl. Acad. Sci. USA 2001, 98, 473–478. [Google Scholar] [CrossRef]
- Azzam, E.I.; De Toledo, S.M.; Gooding, T.; Little, J.B. Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat. Res. 1998, 150, 497–504. [Google Scholar] [CrossRef]
- Azzam, E.I.; Jay-Gerin, J.-P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett. 2012, 327, 48–60. [Google Scholar] [CrossRef]
- Kadhim, M.; Salomaa, S.; Wright, E.; Hildebrandt, G.; Belyakov, O.V.; Prise, K.M.; Little, M.P. Non-targeted effects of ionising radiation—Implications for low dose risk. Mutat. Res. Rev. Mutat. Res. 2013, 752, 84–98. [Google Scholar] [CrossRef]
- Smolarz, M.; Skoczylas, Ł.; Gawin, M.; Krzyżowska, M.; Pietrowska, M.; Widłak, P. Radiation-induced bystander effect mediated by exosomes involves the replication stress in recipient cells. Int. J. Mol. Sci. 2022, 23, 4169. [Google Scholar] [CrossRef]
- Hei, T.K.; Zhou, H.; Ivanov, V.N.; Hong, M.; Lieberman, H.B.; Brenner, D.J.; Amundson, S.A.; Geard, C.R. Mechanism of radiation-induced bystander effects: A unifying model. J. Pharm. Pharmacol. 2008, 60, 943–950. [Google Scholar] [CrossRef]
- Aravindan, S.; Natarajan, M.; Awasthi, V.; Herman, T.S.; Aravindan, N. Novel synthetic monoketone transmute radiation-triggered NFκB-dependent TNFα cross-signaling feedback maintained NFκB and favors neuroblastoma regression. PLoS ONE 2013, 8, e72464. [Google Scholar] [CrossRef][Green Version]
- Spray, D.C.; Hanstein, R.; Lopez-Quintero, S.V.; Stout Jr, R.F.; Suadicani, S.O.; Thi, M.M. Gap junctions and bystander effects: Good Samaritans and executioners. Wiley Interdiscip. Rev. Membr. Transp. Signal. 2013, 2, 1–15. [Google Scholar] [CrossRef]
- Ameziane-El-Hassani, R.; Talbot, M.; de Souza Dos Santos, M.C.; Al Ghuzlan, A.; Hartl, D.; Bidart, J.-M.; De Deken, X.; Miot, F.; Diallo, I.; de Vathaire, F. NADPH oxidase DUOX1 promotes long-term persistence of oxidative stress after an exposure to irradiation. Proc. Natl. Acad. Sci. USA 2015, 112, 5051–5056. [Google Scholar] [CrossRef] [PubMed]
- Lorimore, S.; Wright, E. Radiation-induced genomic instability and bystander effects: Related inflammatory-type responses to radiation-induced stress and injury? A review. Int. J. Radiat. Biol. 2003, 79, 15–25. [Google Scholar] [CrossRef]
- Ilnytskyy, Y.; Koturbash, I.; Kovalchuk, O. Radiation-induced bystander effects in vivo are epigenetically regulated in a tissue-specific manner. Environ. Mol. Mutagen. 2009, 50, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Koturbash, I.; Rugo, R.E.; Hendricks, C.A.; Loree, J.; Thibault, B.; Kutanzi, K.; Pogribny, I.; Yanch, J.C.; Engelward, B.P.; Kovalchuk, O. Irradiation induces DNA damage and modulates epigenetic effectors in distant bystander tissue in vivo. Oncogene 2006, 25, 4267–4275. [Google Scholar] [CrossRef]
- Iyer, R.; Lehnert, B.E. Factors underlying the cell growth-related bystander responses to α particles. Cancer Res. 2000, 60, 1290–1298. [Google Scholar] [PubMed]
- Herberg, M.; Siebert, S.; Quaas, M.; Thalheim, T.; Rother, K.; Hussong, M.; Altmüller, J.; Kerner, C.; Galle, J.; Schweiger, M.R. Loss of Msh2 and a single-radiation hit induce common, genome-wide, and persistent epigenetic changes in the intestine. Clin. Epigenetics 2019, 11, 65. [Google Scholar] [CrossRef]
- He, X.; Cai, L.; Tang, H.; Chen, W.; Hu, W. Epigenetic modifications in radiation-induced non-targeted effects and their clinical significance. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2023, 1867, 130386. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Ding, N.; Pei, H.; Hu, W.; Wei, W.; Zhang, X.; Zhou, G.; Wang, J. MiR-21 is involved in radiation-induced bystander effects. RNA Biol. 2014, 11, 1161–1170. [Google Scholar] [CrossRef]
- Apetoh, L.; Ghiringhelli, F.; Tesniere, A.; Obeid, M.; Ortiz, C.; Criollo, A.; Mignot, G.; Maiuri, M.C.; Ullrich, E.; Saulnier, P. Toll-like receptor 4–dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat. Med. 2007, 13, 1050–1059. [Google Scholar] [CrossRef]
- Wang, L.; Lynch, C.; Pitroda, S.P.; Piffkó, A.; Yang, K.; Huser, A.K.; Liang, H.L.; Weichselbaum, R.R. Radiotherapy and immunology. J. Exp. Med. 2024, 221, e20232101. [Google Scholar] [CrossRef]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti–CTLA-4 antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar] [CrossRef] [PubMed]
- Schaue, D.; Ratikan, J.A.; Iwamoto, K.S.; McBride, W.H. Maximizing tumor immunity with fractionated radiation. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 1306–1310. [Google Scholar] [CrossRef]
- Fernandez-Palomo, C.; Schültke, E.; Bräuer-Krisch, E.; Laissue, J.A.; Blattmann, H.; Seymour, C.; Mothersill, C. Investigation of abscopal and bystander effects in immunocompromised mice after exposure to pencilbeam and microbeam synchrotron radiation. Health Phys. 2016, 111, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhou, T.; Liu, W.; Zuo, L. Molecular mechanism of bystander effects and related abscopal/cohort effects in cancer therapy. Oncotarget 2018, 9, 18637. [Google Scholar] [CrossRef]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar] [CrossRef]
- Hamada, N.; Matsumoto, H.; Hara, T.; Kobayashi, Y. Intercellular and intracellular signaling pathways mediating ionizing radiation-induced bystander effects. J. Radiat. Res. 2007, 48, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Seymour, C. Radiation-induced non-targeted effects: Some open questions. Radiat. Prot. Dosim. 2015, 166, 125–130. [Google Scholar] [CrossRef]
- Seymour, C.M. Medium from irradiated human epithelial cells but not human fibroblasts reduces the clonogenic survival of unirradiated cells. Int. J. Radiat. Biol. 1997, 71, 421–427. [Google Scholar] [CrossRef]
- Zhou, H.; Ivanov, V.N.; Gillespie, J.; Geard, C.R.; Amundson, S.A.; Brenner, D.J.; Yu, Z.; Lieberman, H.B.; Hei, T.K. Mechanism of radiation-induced bystander effect: Role of the cyclooxygenase-2 signaling pathway. Proc. Natl. Acad. Sci. USA 2005, 102, 14641–14646. [Google Scholar] [CrossRef]
- Najafi, M.; Fardid, R.; Hadadi, G.; Fardid, M. The mechanisms of radiation-induced bystander effect. J. Biomed. Phys. Eng. 2014, 4, 163. [Google Scholar]
- Redon, C.E.; Dickey, J.S.; Bonner, W.M.; Sedelnikova, O.A. γ-H2AX as a biomarker of DNA damage induced by ionizing radiation in human peripheral blood lymphocytes and artificial skin. Adv. Space Res. 2009, 43, 1171–1178. [Google Scholar] [CrossRef]
- Hu, B.; Wu, L.; Han, W.; Zhang, L.; Chen, S.; Xu, A.; Hei, T.K.; Yu, Z. The time and spatial effects of bystander response in mammalian cells induced by low dose radiation. Carcinogenesis 2006, 27, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Motevaseli, E.; Shirazi, A.; Geraily, G.; Rezaeyan, A.; Norouzi, F.; Rezapoor, S.; Abdollahi, H. Mechanisms of inflammatory responses to radiation and normal tissues toxicity: Clinical implications. Int. J. Radiat. Biol. 2018, 94, 335–356. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, J.; Baldwin, J.; Held, K.D.; Prise, K.M.; Redmond, R.W.; Liber, H.L. Ionizing radiation-induced bystander mutagenesis and adaptation: Quantitative and temporal aspects. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2009, 671, 20–25. [Google Scholar] [CrossRef]
- Abdel-Aziz, N.; Elkady, A.A.; Elgazzar, E.M. Effect of low-dose gamma radiation and lipoic acid on high-radiation-dose induced rat brain injuries. Dose-Response 2021, 19, 15593258211044845. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Wang, B.; Wang, H.; Meng, L.; Zhao, Q.; Li, X.; Xin, Y.; Jiang, X. Radiation-induced normal tissue damage: Oxidative stress and epigenetic mechanisms. Oxidative Med. Cell. Longev. 2019, 2019, 3010342. [Google Scholar] [CrossRef]
- Sharma, N.K.; Stone, S.; Kumar, V.P.; Biswas, S.; Aghdam, S.Y.; Holmes-Hampton, G.P.; Fam, C.M.; Cox, G.N.; Ghosh, S.P. Mitochondrial Degeneration and Autophagy Associated With Delayed Effects of Radiation in the Mouse Brain. Front. Aging Neurosci. 2019, 11, 357. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, J.; Fu, J.; Wang, J.; Ye, S.; Liu, W.; Shao, C. Role of ROS-mediated autophagy in radiation-induced bystander effect of hepatoma cells. Int. J. Radiat. Biol. 2015, 91, 452–458. [Google Scholar] [CrossRef]
- Liao, E.C.; Hsu, Y.T.; Chuah, Q.Y.; Lee, Y.J.; Hu, J.Y.; Huang, T.C.; Yang, P.M.; Chiu, S.J. Radiation induces senescence and a bystander effect through metabolic alterations. Cell Death Dis. 2014, 5, e1255. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, M.V.; Neumann, R.D. Radiation-induced bystander effects in cultured human stem cells. PLoS ONE 2010, 5, e14195. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Tiku, A.B. Role of mTOR pathway in modulation of radiation induced bystander effects. Int. J. Radiat. Biol. 2022, 98, 173–182. [Google Scholar] [CrossRef]
- Marín, A.; Martín, M.; Liñán, O.; Alvarenga, F.; López, M.; Fernández, L.; Büchser, D.; Cerezo, L. Bystander effects and radiotherapy. Rep. Pract. Oncol. Radiother. 2014, 20, 12–21. [Google Scholar] [CrossRef]
- Yu, H.; Aravindan, N.; Xu, J.; Natarajan, M. Inter- and intra-cellular mechanism of NF-kB-dependent survival advantage and clonal expansion of radio-resistant cancer cells. Cell. Signal. 2017, 31, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Veeraraghavan, J.; Natarajan, M.; Aravindan, S.; Herman, T.S.; Aravindan, N. Radiation-triggered tumor necrosis factor (TNF) α-NFκB cross-signaling favors survival advantage in human neuroblastoma cells. J. Biol. Chem. 2011, 286, 21588–21600. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, N.; Veeraraghavan, J.; Madhusoodhanan, R.; Herman, T.S.; Natarajan, M. Curcumin regulates low-linear energy transfer γ-radiation-induced NFκB-dependent telomerase activity in human neuroblastoma cells. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1206–1215. [Google Scholar] [CrossRef]
- Somasundaram, D.B.; Aravindan, S.; Major, R.; Natarajan, M.; Aravindan, N. MMP-9 reinforces radiation-induced delayed invasion and metastasis of neuroblastoma cells through second-signaling positive feedback with NFκB via both ERK and IKK activation. Cell Biol. Toxicol. 2023, 39, 1053–1076. [Google Scholar] [CrossRef]
- Aravindan, S.; Natarajan, M.; Ramraj, S.K.; Pandian, V.; Khan, F.H.; Herman, T.S.; Aravindan, N. Abscopal effect of low-LET γ-radiation mediated through Rel protein signal transduction in a mouse model of nontargeted radiation response. Cancer Gene Ther. 2014, 21, 54–59. [Google Scholar] [CrossRef]
- Aravindan, N.; Madhusoodhanan, R.; Natarajan, M.; Herman, T.S. Alteration of apoptotic signaling molecules as a function of time after radiation in human neuroblastoma cells. Mol. Cell. Biochem. 2008, 310, 167–179. [Google Scholar] [CrossRef]
- Elbakrawy, E.; Kaur Bains, S.; Bright, S.; Al-Abedi, R.; Mayah, A.; Goodwin, E.; Kadhim, M. Radiation-Induced Senescence Bystander Effect: The Role of Exosomes. Biology 2020, 9, 191. [Google Scholar] [CrossRef]
- Al-Hawary, S.I.S.; Abdalkareem Jasim, S.; Altalbawy, F.M.A.; Kumar, A.; Kaur, H.; Pramanik, A.; Jawad, M.A.; Alsaad, S.B.; Mohmmed, K.H.; Zwamel, A.H. miRNAs in radiotherapy resistance of cancer; a comprehensive review. Cell Biochem. Biophys. 2024, 82, 1665–1679. [Google Scholar] [CrossRef]
- Al-Mayah, A.; Bright, S.; Chapman, K.; Irons, S.; Luo, P.; Carter, D.; Goodwin, E.; Kadhim, M. The non-targeted effects of radiation are perpetuated by exosomes. Mutat. Res. 2015, 772, 38–45. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, Y.; Han, W.; Zhao, G.; Zhu, L.; Wang, J.; Bao, L.; Jiang, E.; Xu, A.; Hei, T.K.; et al. Mitochondria-dependent signalling pathway are involved in the early process of radiation-induced bystander effects. Br. J. Cancer 2008, 98, 1839–1844. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D. Low-dose non-targeted effects and mitochondrial control. Int. J. Mol. Sci. 2023, 24, 11460. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-H.; Yang, P.-M.; Chuah, Q.-Y.; Lee, Y.-J.; Hsieh, Y.-F.; Peng, C.-W.; Chiu, S.-J. Autophagy promotes radiation-induced senescence but inhibits bystander effects in human breast cancer cells. Autophagy 2014, 10, 1212–1228. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, M.; Neumann, R. Changes in gene expression as one of the key mechanisms involved in radiation-induced bystander effect. Biomed. Rep. 2018, 9, 99–111. [Google Scholar] [CrossRef]
- Hedden, L.; Benes, C.H.; Soltoff, S.P. P2X(7) receptor antagonists display agonist-like effects on cell signaling proteins. Biochim. Biophys. Acta 2011, 1810, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Kam, W.W.; Banati, R.B. Effects of ionizing radiation on mitochondria. Free Radic. Biol. Med. 2013, 65, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, V.N.; Ghandhi, S.A.; Zhou, H.; Huang, S.X.; Chai, Y.; Amundson, S.A.; Hei, T.K. Radiation response and regulation of apoptosis induced by a combination of TRAIL and CHX in cells lacking mitochondrial DNA: A role for NF-κB–STAT3-directed gene expression. Exp. Cell Res. 2011, 317, 1548–1566. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Davidson, M.M.; Hei, T.K. Mitochondria regulate DNA damage and genomic instability induced by high LET radiation. Life Sci. Space Res. 2014, 1, 80–88. [Google Scholar] [CrossRef]
- Zheng, X.; Jin, X.; Ye, F.; Liu, X.; Yu, B.; Li, Z.; Zhao, T.; Chen, W.; Liu, X.; Di, C.; et al. Ferroptosis: A novel regulated cell death participating in cellular stress response, radiotherapy, and immunotherapy. Exp. Hematol. Oncol. 2023, 12, 65. [Google Scholar] [CrossRef]
- Mittra, I.; Samant, U.; Sharma, S.; Raghuram, G.V.; Saha, T.; Tidke, P.; Pancholi, N.; Gupta, D.; Prasannan, P.; Gaikwad, A.; et al. Cell-free chromatin from dying cancer cells integrate into genomes of bystander healthy cells to induce DNA damage and inflammation. Cell Death Discov. 2017, 3, 17015. [Google Scholar] [CrossRef]
- Li, L.; Wang, L.; Prise, K.M.; Yu, K.N.; Chen, G.; Chen, L.; Mei, Y.; Han, W. Akt/mTOR mediated induction of bystander effect signaling in a nucleus independent manner in irradiated human lung adenocarcinoma epithelial cells. Oncotarget 2017, 8, 18010. [Google Scholar] [CrossRef][Green Version]
- Laiakis, E.C.; Strassburg, K.; Bogumil, R.; Lai, S.; Vreeken, R.J.; Hankemeier, T.; Langridge, J.; Plumb, R.S.; Fornace, A.J.J.; Astarita, G. Metabolic Phenotyping Reveals a Lipid Mediator Response to Ionizing Radiation. J. Proteome Res. 2014, 13, 4143–4154. [Google Scholar] [CrossRef]
- Burdak-Rothkamm, S.; Short, S.C.; Folkard, M.; Rothkamm, K.; Prise, K.M. ATR-dependent radiation-induced γH2AX foci in bystander primary human astrocytes and glioma cells. Oncogene 2007, 26, 993–1002. [Google Scholar] [CrossRef]
- Hei, T.K.; Zhou, H.; Chai, Y.; Ponnaiya, B.; Ivanov, V.N. Radiation induced non-targeted response: Mechanism and potential clinical implications. Curr. Mol. Pharmacol. 2011, 4, 96–105. [Google Scholar] [CrossRef]
- Xu, S.; Wang, J.; Ding, N.; Hu, W.; Zhang, X.; Wang, B.; Hua, J.; Wei, W.; Zhu, Q. Exosome-mediated microRNA transfer plays a role in radiation-induced bystander effect. RNA Biol. 2015, 12, 1355–1363. [Google Scholar] [CrossRef]
- Yang, H.; Asaad, N.; Held, K.D. Medium-mediated intercellular communication is involved in bystander responses of X-ray-irradiated normal human fibroblasts. Oncogene 2005, 24, 2096–2103. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Xu, S.; Yao, B.; Hong, M.; Wu, X.; Pei, H.; Chang, L.; Ding, N.; Gao, X.; Ye, C.; et al. MiR-663 inhibits radiation-induced bystander effects by targeting TGFB1 in a feedback mode. RNA Biol. 2014, 11, 1189–1198. [Google Scholar] [CrossRef]
- Wang, B.; Tanaka, K.; Katsube, T.; Maruyama, K.; Ninomiya, Y.; Vares, G.; Liu, C.; Hirakawa, H.; Murakami, M.; Fardous, Z. Reduced high-dose radiation-induced residual genotoxic damage by induction of radioadaptive response and prophylactic mild dietary restriction in mice. Dose-Response 2021, 19, 1559325820982166. [Google Scholar] [CrossRef] [PubMed]
- Baulch, J.E.; Aypar, U.; Waters, K.M.; Yang, A.J.; Morgan, W.F. Genetic and Epigenetic Changes in Chromosomally Stable and Unstable Progeny of Irradiated Cells. PLoS ONE 2014, 9, e107722. [Google Scholar] [CrossRef] [PubMed]
- Streffer, C. An update on the mechanisms and pathophysiological consequences of genomic instability with a focus on ionizing radiation. Rep. Radiother. Biol. 2015, 2015, 225–233. [Google Scholar] [CrossRef]
- Kavanagh, J.; Waring, E.; Prise, K. Radiation responses of stem cells: Targeted and non-targeted effects. Radiat. Prot. Dosim. 2015, 166, 110–117. [Google Scholar] [CrossRef]
- Yahyapour, R.; Salajegheh, A.; Safari, A.; Amini, P.; Rezaeyan, A.; Amraee, A.; Najafi, M. Radiation-induced non-targeted effect and carcinogenesis; implications in clinical radiotherapy. J. Biomed. Phys. Eng. 2018, 8, 435. [Google Scholar] [CrossRef]
- Farhood, B.; Hoseini-Ghahfarokhi, M.; Motevaseli, E.; Mirtavoos-Mahyari, H.; Musa, A.E.; Najafi, M. TGF-β in radiotherapy: Mechanisms of tumor resistance and normal tissues injury. Pharmacol. Res. 2020, 155, 104745. [Google Scholar] [CrossRef]
- Chaudhry, M.A. Small nucleolar RNA host genes and long non-coding RNA responses in directly irradiated and bystander cells. Cancer Biother. Radiopharm. 2014, 29, 135–141. [Google Scholar] [CrossRef]
- Jassi, C.; Kuo, W.-W.; Kuo, C.-H.; Chang, C.-M.; Chen, M.-C.; Shih, T.-C.; Li, C.-C.; Huang, C.-Y. Mediation of radiation-induced bystander effect and epigenetic modification: The role of exosomes in cancer radioresistance. Heliyon 2024, 10, e34460. [Google Scholar] [CrossRef]
- Sokolov, M.; Neumann, R. Global gene expression alterations as a crucial constituent of human cell response to low doses of ionizing radiation exposure. Int. J. Mol. Sci. 2015, 17, 55. [Google Scholar] [CrossRef]
- Barcellos-Hoff, M.H.; Mao, J.H. HZE radiation non-targeted effects on the microenvironment that mediate mammary carcinogenesis. Front. Oncol. 2016, 6, 57. [Google Scholar] [CrossRef] [PubMed]
- Heeran, A.B.; Berrigan, H.P.; O’Sullivan, J. The radiation-induced bystander effect (RIBE) and its connections with the hallmarks of cancer. Radiat. Res. 2019, 192, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, S.; Natarajan, M.; Veeraraghavan, J.; Herman, T.S.; Aravindan, N. Inflammatory Signature after Low Dose γ-Radiation in Mice Brain and Gut: Switch from Therapeutic Benefit to Inflammation. Eur. J. Inflamm. 2013, 11, 405–418. [Google Scholar] [CrossRef]
- Shareef, M.M.; Cui, N.; Burikhanov, R.; Gupta, S.; Satishkumar, S.; Shajahan, S.; Mohiuddin, M.; Rangnekar, V.M.; Ahmed, M.M. Role of tumor necrosis factor-α and TRAIL in high-dose radiation–induced bystander signaling in lung adenocarcinoma. Cancer Res. 2007, 67, 11811–11820. [Google Scholar] [CrossRef]
- Nikitaki, Z.; Mavragani, I.V.; Laskaratou, D.A.; Gika, V.; Moskvin, V.P.; Theofilatos, K.; Vougas, K.; Stewart, R.D.; Georgakilas, A.G. Systemic mechanisms and effects of ionizing radiation: A new old paradigm of how the bystanders and distant can become the players. Semin. Cancer Biol. 2016, 37, 77–95. [Google Scholar] [CrossRef]
- Hussien, S.M.; Rashed, E.R. Immune system modulation by low-dose ionizing radiation-induced adaptive response. Int. J. Immunopathol. Pharmacol. 2023, 37, 03946320231172080. [Google Scholar] [CrossRef]
- Szatmári, T.; Kis, D.; Bogdándi, E.N.; Benedek, A.; Bright, S.; Bowler, D.; Persa, E.; Kis, E.; Balogh, A.; Naszályi, L.N.; et al. Extracellular vesicles mediate radiation-induced systemic bystander signals in the bone marrow and spleen. Front. Immunol. 2017, 8, 347. [Google Scholar] [CrossRef]
- Rodina, A.V.; Semochkina, Y.P.; Vysotskaya, O.V.; Romantsova, A.N.; Strepetov, A.N.; Moskaleva, E.Y. Low dose gamma irradiation pretreatment modulates the sensitivity of CNS to subsequent mixed gamma and neutron irradiation of the mouse head. Int. J. Radiat. Biol. 2021, 97, 926–942. [Google Scholar] [CrossRef]
- Peng, X.; Wu, Y.; Brouwer, U.; van Vliet, T.; Wang, B.; Demaria, M.; Barazzuol, L.; Coppes, R.P. Cellular senescence contributes to radiation-induced hyposalivation by affecting the stem/progenitor cell niche. Cell Death Dis. 2020, 11, 854. [Google Scholar] [CrossRef]
- Minafra, L.; Bravatà, V.; Cammarata, F.P.; Di Maggio, F.M.; Forte, G.I. SASPects of Radiation Induced Senescence. Ann. Radiat. Ther. Oncol. 2017, 1, 1006. [Google Scholar]
- Shireman, J.M.; White, Q.; Agrawal, N.; Ni, Z.; Chen, G.; Zhao, L.; Gonugunta, N.; Wang, X.; McCarthy, L.; Kasulabada, V.; et al. Genomic Analysis of Human Brain Metastases Treated with Stereotactic Radiosurgery Under the Phase-II Clinical Trial (NCT03398694) Reveals DNA Damage Repair at the Peripheral Tumor Edge. medRxiv 2023. [Google Scholar] [CrossRef]
- Venkatesulu, B.P.; Mahadevan, L.S.; Aliru, M.L.; Yang, X.; Bodd, M.H.; Singh, P.K.; Yusuf, S.W.; Abe, J.-i.; Krishnan, S. Radiation-Induced Endothelial Vascular Injury: A Review of Possible Mechanisms. JACC Basic. Transl. Sci. 2018, 3, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Wijerathne, H.; Langston, J.C.; Yang, Q.; Sun, S.; Miyamoto, C.; Kilpatrick, L.E.; Kiani, M.F. Mechanisms of radiation-induced endothelium damage: Emerging models and technologies. Radiother. Oncol. 2021, 158, 21–32. [Google Scholar] [CrossRef]
- Ma, L.; Ye, Z.; Zhang, Y.; Shi, W.; Wang, J.; Yang, H. Irradiated microvascular endothelial cells may induce bystander effects in neural stem cells leading to neurogenesis inhibition. J. Radiat. Res. 2022, 63, 192–201. [Google Scholar] [CrossRef]
- Zhang, L.-Y.; Yong, W.-X.; Wang, L.; Zhang, L.-X.; Zhang, Y.-M.; Gong, H.-X.; He, J.-P.; Liu, Y.-Q. Astragalus polysaccharide eases G1 phase-correlative bystander effects through mediation of TGF-β R/MAPK/ROS signal pathway after carbon ion irradiation in BMSCs. Am. J. Chin. Med. 2019, 47, 595–612. [Google Scholar] [CrossRef]
- Jiang, Y.; Chen, X.; Tian, W.; Yin, X.; Wang, J.; Yang, H. The role of TGF-β1–miR-21–ROS pathway in bystander responses induced by irradiated non-small-cell lung cancer cells. Br. J. Cancer 2014, 111, 772–780. [Google Scholar] [CrossRef]
- Mancuso, M.; Pasquali, E.; Leonardi, S.; Tanori, M.; Rebessi, S.; Di Majo, V.; Pazzaglia, S.; Toni, M.P.; Pimpinella, M.; Covelli, V. Oncogenic bystander radiation effects in Patched heterozygous mouse cerebellum. Proc. Natl. Acad. Sci. USA 2008, 105, 12445–12450. [Google Scholar] [CrossRef]
- Ivanov, V.N.; Hei, T.K. Radiation-induced glioblastoma signaling cascade regulates viability, apoptosis and differentiation of neural stem cells (NSC). Apoptosis 2014, 19, 1736–1754. [Google Scholar] [CrossRef][Green Version]
- Ivanov, V.N.; Hei, T.K. A role for TRAIL/TRAIL-R2 in radiation-induced apoptosis and radiation-induced bystander response of human neural stem cells. Apoptosis 2014, 19, 399–413. [Google Scholar] [CrossRef] [PubMed]
- Grigorieva, E.V. Radiation Effects on Brain Extracellular Matrix. Front. Oncol. 2020, 10, 576701. [Google Scholar] [CrossRef]
- Lee, W.H.; Warrington, J.P.; Sonntag, W.E.; Lee, Y.W. Irradiation alters mmp-2/timp-2 system and collagen type iv degradation in brain. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1559–1566. [Google Scholar] [CrossRef]
- Gupta, K.; Burns, T.C. Radiation-Induced Alterations in the Recurrent Glioblastoma Microenvironment: Therapeutic Implications. Front. Oncol. 2018, 8, 503. [Google Scholar] [CrossRef]
- La Verde, G.; Artiola, V.; Pugliese, M.; La Commara, M.; Arrichiello, C.; Muto, P.; Netti, P.A.; Fusco, S.; Panzetta, V. Radiation therapy affects YAP expression and intracellular localization by modulating lamin A/C levels in breast cancer. Front. Bioeng. Biotechnol. 2022, 10, 969004. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aziz, N.; Haroun, R.A.; Mohamed, H.E. Low-dose gamma radiation modulates liver and testis tissues response to acute whole body irradiation. Dose-Response 2022, 20, 15593258221092365. [Google Scholar] [CrossRef] [PubMed]
- Xin, J.-Y.; Wang, J.; Ding, Q.-Q.; Chen, W.; Xu, X.-K.; Wei, X.-T.; Lv, Y.-H.; Wei, Y.-P.; Feng, Y.; Zu, X.-P. Potential role of gut microbiota and its metabolites in radiation-induced intestinal damage. Int. J. Radiat. Biol. 2022, 248, 114341. [Google Scholar] [CrossRef]
- Wang, W.; Cui, B.; Nie, Y.; Sun, L.; Zhang, F. Radiation injury and gut microbiota-based treatment. Protein Cell 2024, 15, 83–97. [Google Scholar] [CrossRef]
- Dakup, P.P.; Porter, K.I.; Gajula, R.P.; Goel, P.N.; Cheng, Z.; Gaddameedhi, S. The circadian clock protects against ionizing radiation-induced cardiotoxicity. FASEB J. 2020, 34, 3347–3358. [Google Scholar] [CrossRef]
- Chevalier, F.; Hamdi, D.H.; Saintigny, Y.; Lefaix, J.-L. Proteomic overview and perspectives of the radiation-induced bystander effects. Mutat. Res./Rev. Mutat. Res. 2015, 763, 280–293. [Google Scholar] [CrossRef]
- Amoroso, F.; Glass, K.; Singh, R.; Liberal, F.; Steele, R.E.; Maguire, S.; Tarapore, R.; Allen, J.E.; Van Schaeybroeck, S.; Butterworth, K.T. Modulating the unfolded protein response with ONC201 to impact on radiation response in prostate cancer cells. Sci. Rep. 2021, 11, 4252. [Google Scholar] [CrossRef]
- Shaler, T.; Lin, H.; Bakke, J.; Chen, S.; Grover, A.; Chang, P. Particle radiation-induced dysregulation of protein homeostasis in primary human and mouse neuronal cells. Life Sci. Space Res. 2020, 25, 9–17. [Google Scholar] [CrossRef]
- Harada, K.; Nonaka, T.; Hamada, N.; Sakurai, H.; Hasegawa, M.; Funayama, T.; Kakizaki, T.; Kobayashi, Y.; Nakano, T. Heavy-ion-induced bystander killing of human lung cancer cells: Role of gap junctional intercellular communication. Cancer Sci. 2009, 100, 684–688. [Google Scholar] [CrossRef] [PubMed]
- McBride, W.H.; Iwamoto, K.S.; Syljuasen, R.; Pervan, M.; Pajonk, F. The role of the ubiquitin/proteasome system in cellular responses to radiation. Oncogene 2003, 22, 5755–5773. [Google Scholar] [CrossRef]
- Tong, J.X.; Vogelbaum, M.A.; Drzymala, R.E.; Rich, K.M. Radiation-induced apoptosis in dorsal root ganglion neurons. J. Neurocytol. 1997, 26, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Baljinnyam, E.; Venkatesh, S.; Gordan, R.; Mareedu, S.; Zhang, J.; Xie, L.H.; Azzam, E.I.; Suzuki, C.K.; Fraidenraich, D. Effect of densely ionizing radiation on cardiomyocyte differentiation from human-induced pluripotent stem cells. Physiol. Rep. 2017, 5, e13308. [Google Scholar] [CrossRef]
- Nagasawa, H.; Cremesti, A.; Kolesnick, R.; Fuks, Z.; Little, J.B. Involvement of membrane signaling in the bystander effect in irradiated cells. Cancer Res. 2002, 62, 2531–2534. [Google Scholar] [PubMed]
- Hehlgans, S.; Eke, I.; Storch, K.; Haase, M.; Baretton, G.B.; Cordes, N. Caveolin-1 mediated radioresistance of 3D grown pancreatic cancer cells. Radiother. Oncol. 2009, 92, 362–370. [Google Scholar] [CrossRef]
- Cordes, N.; Frick, S.; Brunner, T.B.; Pilarsky, C.; Grützmann, R.; Sipos, B.; Klöppel, G.; McKenna, W.G.; Bernhard, E.J. Human pancreatic tumor cells are sensitized to ionizing radiation by knockdown of caveolin-1. Oncogene 2007, 26, 6851–6862. [Google Scholar] [CrossRef]
- Suzuki, K.; Yamashita, S. Radiation-Induced Bystander Response: Mechanism and Clinical Implications. Adv. Wound Care 2014, 3, 16–24. [Google Scholar] [CrossRef]
- Shuryak, I.; Brenner, D.J. Review of quantitative mechanistic models of radiation-induced non-targeted effects (NTE). Radiat. Prot. Dosim. 2020, 192, 236–252. [Google Scholar] [CrossRef] [PubMed]
- Shabrish, S.; Pal, K.; Khare, N.K.; Satsangi, D.; Pilankar, A.; Jadhav, V.; Shinde, S.; Raphael, N.; Sriram, G.; Lopes, R.; et al. Cell-free chromatin particles released from dying cancer cells activate immune checkpoints in human lymphocytes: Implications for cancer therapy. Front. Immunol. 2024, 14, 1331491. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.Y.; Butler, N.J.; Makrigiorgos, G.M.; Adelstein, S.J.; Kassis, A.I. Bystander effect produced by radiolabeled tumor cells in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 13765–13770. [Google Scholar] [CrossRef] [PubMed]
- Mitchel, R.E. The bystander effect: Recent developments and implications for understanding the dose response. Nonlinearity Biol. Toxicol. Med. 2004, 2, 173–183. [Google Scholar] [CrossRef]
- Bahreyni Toossi, M.T.; Khademi, S.; Azimian, H.; Mohebbi, S.; Soleymanifard, S. Assessment of The Dose-Response Relationship of Radiation-Induced Bystander Effect in Two Cell Lines Exposed to High Doses of Ionizing Radiation (6 and 8 Gy). Cell J. 2017, 19, 434–442. [Google Scholar] [CrossRef]
- Merrifield, M.; Kovalchuk, O. Epigenetics in radiation biology: A new research frontier. Front. Genet. 2013, 4, 40. [Google Scholar] [CrossRef]
- Bonner, W.M. Low-dose radiation: Thresholds, bystander effects, and adaptive responses. Proc. Natl. Acad. Sci. USA 2003, 100, 4973–4975. [Google Scholar] [CrossRef]
- Pinto, M.; Azzam, E.I.; Howell, R.W. Investigation of adaptive responses in bystander cells in 3D cultures containing tritium-labeled and unlabeled normal human fibroblasts. Radiat. Res. 2010, 174, 216–227. [Google Scholar] [CrossRef]
- Rezaee, M.; Adhikary, A. The Effects of Particle LET and Fluence on the Complexity and Frequency of Clustered DNA Damage. DNA 2024, 4, 34–51. [Google Scholar] [CrossRef]
- Sedelnikova, O.A.; Nakamura, A.; Kovalchuk, O.; Koturbash, I.; Mitchell, S.A.; Marino, S.A.; Brenner, D.J.; Bonner, W.M. DNA double-strand breaks form in bystander cells after microbeam irradiation of three-dimensional human tissue models. Cancer Res. 2007, 67, 4295–4302. [Google Scholar] [CrossRef]
- Konopacka, M.; Rogoliński, J.; Slosarek, K. Bystander effects induced by direct and scattered radiation generated during penetration of medium inside a water phantom. Rep. Pract. Oncol. Radiother. 2011, 16, 256–261. [Google Scholar] [CrossRef]
- Kirolikar, S.; Prasannan, P.; Raghuram, G.V.; Pancholi, N.; Saha, T.; Tidke, P.; Chaudhari, P.; Shaikh, A.; Rane, B.; Pandey, R.; et al. Prevention of radiation-induced bystander effects by agents that inactivate cell-free chromatin released from irradiated dying cells. Cell Death Dis. 2018, 9, 1142. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Shao, C. Research progress of radiation induced bystander and abscopal effects in normal tissue. Radiat. Med. Prot. 2020, 1, 69–74. [Google Scholar] [CrossRef]
- da Silva, P.F.L.; Ogrodnik, M.; Kucheryavenko, O.; Glibert, J.; Miwa, S.; Cameron, K.; Ishaq, A.; Saretzki, G.; Nagaraja-Grellscheid, S.; Nelson, G.; et al. The bystander effect contributes to the accumulation of senescent cells in vivo. Aging Cell 2019, 18, e12848. [Google Scholar] [CrossRef]
- Belli, M.; Tabocchini, M.A. Ionizing Radiation-Induced Epigenetic Modifications and Their Relevance to Radiation Protection. Int. J. Mol. Sci. 2020, 21, 5993. [Google Scholar] [CrossRef] [PubMed]
- Kovalchuk, A.; Mychasiuk, R.; Muhammad, A.; Hossain, S.; Ilnytskyy, Y.; Ghose, A.; Kirkby, C.; Ghasroddashti, E.; Kolb, B.; Kovalchuk, O. Profound and Sexually Dimorphic Effects of Clinically-Relevant Low Dose Scatter Irradiation on the Brain and Behavior. Front. Behav. Neurosci. 2016, 10, 84. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Tian, W.; Wang, L.; Wang, J.; Zhang, S.; Cao, J.; Yang, H. Radiation quality-dependence of bystander effect in unirradiated fibroblasts is associated with TGF-β1-Smad2 pathway and miR-21 in irradiated keratinocytes. Sci. Rep. 2015, 5, 11373. [Google Scholar] [CrossRef]
- Datta, K.; Suman, S.; Kallakury, B.V.S.; Fornace, A.J., Jr. Exposure to Heavy Ion Radiation Induces Persistent Oxidative Stress in Mouse Intestine. PLoS ONE 2012, 7, e42224. [Google Scholar] [CrossRef]
- Buonanno, M.; de Toledo, S.M.; Pain, D.; Azzam, E.I. Long-term consequences of radiation-induced bystander effects depend on radiation quality and dose and correlate with oxidative stress. Radiat. Res. 2011, 175, 405–415. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects and adaptive responses—The Yin and Yang of low dose radiobiology? Mutat. Res. 2004, 568, 121–128. [Google Scholar] [CrossRef]
- Zhang, D.; Zhou, T.; He, F.; Rong, Y.; Lee, S.H.; Wu, S.; Zuo, L. Reactive oxygen species formation and bystander effects in gradient irradiation on human breast cancer cells. Oncotarget 2016, 7, 41622–41636. [Google Scholar] [CrossRef] [PubMed]
- Rosini, G.; Ciarrocchi, E.; D’Orsi, B. Mechanisms of the FLASH effect: Current insights and advances. Front. Cell Dev. Biol. 2025, 13, 1575678. [Google Scholar] [CrossRef]
- Brady, D.; O’Sullivan, J.M.; Prise, K.M. What is the Role of the Bystander Response in Radionuclide Therapies? Front. Oncol. 2013, 3, 215. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, N.; Aravindan, S.; Pandian, V.; Khan, F.H.; Ramraj, S.K.; Natt, P.; Natarajan, M. Acquired tumor cell radiation resistance at the treatment site is mediated through radiation-orchestrated intercellular communication. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Hargitai, R.; Kis, D.; Persa, E.; Szatmári, T.; Sáfrány, G.; Lumniczky, K. Oxidative stress and gene expression modifications mediated by extracellular vesicles: An in vivo study of the radiation-induced bystander effect. Antioxidants 2021, 10, 156. [Google Scholar] [CrossRef]
- Tian, W.; Yin, X.; Wang, L.; Wang, J.; Zhu, W.; Cao, J.; Yang, H. The key role of miR-21-regulated SOD2 in the medium-mediated bystander responses in human fibroblasts induced by α-irradiated keratinocytes. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2015, 780, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Veeraraghavan, J.; Natarajan, M.; Herman, T.S.; Aravindan, N. Low-dose γ-radiation-induced oxidative stress response in mouse brain and gut: Regulation by NFκB–MnSOD cross-signaling. Mutat. Res./Genet. Toxicol. Environ. Mutagen. 2011, 718, 44–55. [Google Scholar] [CrossRef]
- Yahyapour, R.; Motevaseli, E.; Rezaeyan, A.; Abdollahi, H.; Farhood, B.; Cheki, M.; Rezapoor, S.; Shabeeb, D.; Musa, A.E.; Najafi, M.; et al. Reduction–oxidation (redox) system in radiation-induced normal tissue injury: Molecular mechanisms and implications in radiation therapeutics. Clin. Transl. Oncol. 2018, 20, 975–988. [Google Scholar] [CrossRef]
- Kong, Y.; Cheng, S.H.; Yu, K.N. Induction of autophagy and interleukin 6 secretion in bystander cells: Metabolic cooperation for radiation-induced rescue effect? J. Radiat. Res. 2018, 59, 129–140. [Google Scholar] [CrossRef]
- Aravindan, S.; Natarajan, M.; Herman, T.S.; Awasthi, V.; Aravindan, N. Molecular basis of ‘hypoxic’ breast cancer cell radio-sensitization: Phytochemicals converge on radiation induced Rel signaling. Radiat. Oncol. 2013, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Criswell, T.; Leskov, K.; Miyamoto, S.; Luo, G.; Boothman, D.A. Transcription factors activated in mammalian cells after clinically relevant doses of ionizing radiation. Oncogene 2003, 22, 5813–5827. [Google Scholar] [CrossRef]
- Cagatay, S.T.; Mayah, A.; Mancuso, M.; Giardullo, P.; Pazzaglia, S.; Saran, A.; Daniel, A.; Traynor, D.; Meade, A.D.; Lyng, F.; et al. Phenotypic and functional characteristics of exosomes derived from irradiated mouse organs and their role in the mechanisms driving non-targeted effects. Int. J. Mol. Sci. 2020, 21, 8389. [Google Scholar] [CrossRef] [PubMed]
- Simone, N.L.; Soule, B.P.; Ly, D.; Saleh, A.D.; Savage, J.E.; DeGraff, W.; Cook, J.; Harris, C.C.; Gius, D.; Mitchell, J.B. Ionizing radiation-induced oxidative stress alters miRNA expression. PLoS ONE 2009, 4, e6377. [Google Scholar] [CrossRef]
- Aisling, B.; Heeran, A.; Helen, P.; Berrigan, A.; Croí, E.; Buckley, A.; Heleena Moni Bottu, B.; Orla Prendiville, B.; Amy, M.; Buckley, A.; et al. Radiation-induced Bystander Effect (RIBE) alters mitochondrial metabolism using a human rectal cancer ex vivo explant model. Transl. Oncol. 2021, 14, 100882. [Google Scholar] [CrossRef]
- Jella, K.K.; Moriarty, R.; McClean, B.; Byrne, H.J.; Lyng, F.M. Reactive oxygen species and nitric oxide signaling in bystander cells. PLoS ONE 2018, 13, e0195371. [Google Scholar] [CrossRef]
- Lorimore, S.A.; McIlrath, J.M.; Coates, P.J.; Wright, E.G. Chromosomal instability in unirradiated hemopoietic cells resulting from a delayed in vivo bystander effect of γ radiation. Cancer Res. 2005, 65, 5668–5673. [Google Scholar] [CrossRef]
- Koturbash, I.; Boyko, A.; Rodriguez-Juarez, R.; McDonald, R.J.; Tryndyak, V.P.; Kovalchuk, I.; Pogribny, I.P.; Kovalchuk, O. Role of epigenetic effectors in maintenance of the long-term persistent bystander effect in spleen in vivo. Carcinogenesis 2007, 28, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Calì, B.; Ceolin, S.; Ceriani, F.; Bortolozzi, M.; Agnellini, A.H.R.; Zorzi, V.; Predonzani, A.; Bronte, V.; Molon, B.; Mammano, F. Critical role of gap junction communication, calcium and nitric oxide signaling in bystander responses to focal photodynamic injury. Oncotarget 2015, 6, 10161–10174. [Google Scholar] [CrossRef] [PubMed]
- Tsukimoto, M. Purinergic Signaling Is a Novel Mechanism of the Cellular Response to Ionizing Radiation. Biol. Pharm. Bull. 2015, 38, 951–959. [Google Scholar] [CrossRef]
- Bao, X.; Xie, L. Targeting purinergic pathway to enhance radiotherapy-induced immunogenic cancer cell death. J. Exp. Clin. Cancer Res. 2022, 41, 222. [Google Scholar] [CrossRef]
- Surace, L.; Lysenko, V.; Fontana, A.O.; Cecconi, V.; Janssen, H.; Bicvic, A.; Okoniewski, M.; Pruschy, M.; Dummer, R.; Neefjes, J.; et al. Complement Is a Central Mediator of Radiotherapy-Induced Tumor-Specific Immunity and Clinical Response. Immunity 2015, 42, 767–777. [Google Scholar] [CrossRef]
- Beauford, S.S.; Kumari, A.; Garnett-Benson, C. Ionizing radiation modulates the phenotype and function of human CD4+ induced regulatory T cells. BMC Immunol. 2020, 21, 18. [Google Scholar] [CrossRef]
- Koturbash, I.; Kutanzi, K.; Hendrickson, K.; Rodriguez-Juarez, R.; Kogosov, D.; Kovalchuk, O. Radiation-induced bystander effects in vivo are sex specific. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2008, 642, 28–36. [Google Scholar] [CrossRef]
- Kalamida, D.; Karagounis, I.V.; Giatromanolaki, A.; Koukourakis, M.I. Important Role of Autophagy in Endothelial Cell Response to Ionizing Radiation. PLoS ONE 2014, 9, e102408. [Google Scholar] [CrossRef]
- Song, M.; Wang, Y.; Shang, Z.-F.; Liu, X.-D.; Xie, D.-F.; Wang, Q.; Guan, H.; Zhou, P.-K. Bystander autophagy mediated by radiation-induced exosomal miR-7-5p in non-targeted human bronchial epithelial cells. Sci. Rep. 2016, 6, 30165. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Martin, J.; Devic, C.; Bräuer-Krisch, E.; Diserbo, M.; Thariat, J. Impact of dose-rate on the low-dose hyper-radiosensitivity and induced radioresistance (HRS/IRR) response. Int. J. Radiat. Biol. 2013, 89, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Narendran, N.; Luzhna, L.; Kovalchuk, O. Sex Difference of Radiation Response in Occupational and Accidental Exposure. Front. Genet. 2019, 10, 260. [Google Scholar] [CrossRef] [PubMed]
- Constanzo, J.; Faget, J.; Ursino, C.; Badie, C.; Pouget, J.-P. Radiation-Induced Immunity and Toxicities: The Versatility of the cGAS-STING Pathway. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Deng, L.; Liang, H.; Xu, M.; Yang, X.; Burnette, B.; Arina, A.; Li, X.-D.; Mauceri, H.; Beckett, M.; Darga, T.; et al. STING-Dependent Cytosolic DNA Sensing Promotes Radiation-Induced Type I Interferon-Dependent Antitumor Immunity in Immunogenic Tumors. Immunity 2014, 41, 843–852. [Google Scholar] [CrossRef]
- Liu, Y.-g.; Chen, J.-k.; Zhang, Z.-t.; Ma, X.-j.; Chen, Y.-c.; Du, X.-m.; Liu, H.; Zong, Y.; Lu, G.-c. NLRP3 inflammasome activation mediates radiation-induced pyroptosis in bone marrow-derived macrophages. Cell Death Dis. 2017, 8, e2579. [Google Scholar] [CrossRef]
- Testard, I.; Garcia-Chartier, E.; Issa, A.; Collin-Faure, V.; Aude-Garcia, C.; Candéias, S.M. Bystander signals from low- and high-dose irradiated human primary fibroblasts and keratinocytes modulate the inflammatory response of peripheral blood mononuclear cells. J. Radiat. Res. 2023, 64, 304–316. [Google Scholar] [CrossRef]
- Dong, C.; Tu, W.; He, M.; Fu, J.; Kobayashi, A.; Konishi, T.; Shao, C. Role of Endoplasmic Reticulum and Mitochondrion in Proton Microbeam Radiation-Induced Bystander Effect. Radiat. Res. 2019, 193, 63–72. [Google Scholar] [CrossRef]
- Rajendran, S.; Harrison, S.H.; Thomas, R.A.; Tucker, J.D. The role of mitochondria in the radiation-induced bystander effect in human lymphoblastoid cells. Radiat. Res. 2011, 175, 159–171. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, J.; Zhang, Y.; Mo, F.; Patel, G.; Butterworth, K.; Shao, C.; Prise, K.M. The Roles of HIF-1α in Radiosensitivity and Radiation-Induced Bystander Effects Under Hypoxia. Front. Cell Dev. Biol. 2021, 9, 637454. [Google Scholar] [CrossRef] [PubMed]
- Borges dos Reis, R.; Rodrigues Junior, A.A.; Feres, R.N.; Muglia, V.F. Editorial Comment: Evaluation of HIF-1α and VEGF-A expression in radiation-induced cystitis: A case-control study. Int. Braz. J. Urol. 2021, 47, 306–307. [Google Scholar] [CrossRef]
- Xu, Y.; Hu, W.; Zhou, G. Role of heat shock protein in radiation-induced effect and related potential clinical application. Radiat. Med. Prot. 2024, 6, 51–60. [Google Scholar] [CrossRef]
- Schmid, T.E.; Multhoff, G. Radiation-induced stress proteins - the role of heat shock proteins (HSP) in anti- tumor responses. Curr. Med. Chem. 2012, 19, 1765–1770. [Google Scholar] [CrossRef] [PubMed]
- Yakovlev, V.A. Role of nitric oxide in the radiation-induced bystander effect. Redox Biol. 2015, 6, 396–400. [Google Scholar] [CrossRef]
- Xu, Y.; Hu, W.; Liu, Y.; Xu, P.; Li, Z.; Wu, R.; Shi, X.; Tang, Y. P2Y6 Receptor-Mediated Microglial Phagocytosis in Radiation-Induced Brain Injury. Mol. Neurobiol. 2016, 53, 3552–3564. [Google Scholar] [CrossRef]
- Yamaga, S.; Aziz, M.; Murao, A.; Brenner, M.; Wang, P. DAMPs and radiation injury. Front. Immunol. 2024, 15, 1353990. [Google Scholar] [CrossRef]
- Crittenden, M.R.; Baird, J.; Friedman, D.; Savage, T.; Uhde, L.; Alice, A.; Cottam, B.; Young, K.; Newell, P.; Nguyen, C.; et al. Mertk on tumor macrophages is a therapeutic target to prevent tumor recurrence following radiation therapy. Oncotarget 2016, 7, 78653–78666. [Google Scholar] [CrossRef] [PubMed]
- Ladjohounlou, R.; Louati, S.; Lauret, A.; Gauthier, A.; Ardail, D.; Magne, N.; Alphonse, G.; Rodriguez-Lafrasse, C. Ceramide-Enriched Membrane Domains Contribute to Targeted and Nontargeted Effects of Radiation through Modulation of PI3K/AKT Signaling in HNSCC Cells. Int. J. Mol. Sci. 2020, 21, 7200. [Google Scholar] [CrossRef] [PubMed]
- Greene-Schloesser, D.; Robbins, M.E. Radiation-induced cognitive impairment-from bench to bedside. Neuro-Oncology 2012, 14, iv37–iv44. [Google Scholar] [CrossRef]
- Merchant, T.E.; Goloubeva, O.; Pritchard, D.L.; Gaber, M.W.; Xiong, X.; Danish, R.K.; Lustig, R.H. Radiation dose-volume effects on growth hormone secretion. Int. J. Radiat. Oncol. Biol. Phys. 2002, 52, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.M.; Grimm, J.; McIntyre, R.; Anderson-Keightly, H.; Kleinberg, L.R.; Hales, R.K.; Moore, J.; Vannorsdall, T.; Redmond, K.J. A prospective evaluation of hippocampal radiation dose volume effects and memory deficits following cranial irradiation. Radiother. Oncol. 2017, 125, 234–240. [Google Scholar] [CrossRef]
- Gondi, V.; Hermann, B.P.; Mehta, M.P.; Tomé, W.A. Hippocampal dosimetry predicts neurocognitive function impairment after fractionated stereotactic radiotherapy for benign or low-grade adult brain tumors. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, e487–e493. [Google Scholar] [CrossRef]
- Gan, C.; Li, W.; Xu, J.; Pang, L.; Tang, L.; Yu, S.; Li, A.; Ge, H.; Huang, R.; Cheng, H. Advances in the study of the molecular biological mechanisms of radiation-induced brain injury. Am. J. Cancer Res. 2023, 13, 3275. [Google Scholar]
- Szatmári, T.; Hargitai, R.; Sáfrány, G.; Lumniczky, K. Extracellular vesicles in modifying the effects of ionizing radiation. Int. J. Mol. Sci. 2019, 20, 5527. [Google Scholar] [CrossRef]
- Rueckriegel, S.M.; Bruhn, H.; Thomale, U.W.; Hernáiz Driever, P. Cerebral white matter fractional anisotropy and tract volume as measured by MR imaging are associated with impaired cognitive and motor function in pediatric posterior fossa tumor survivors. Pediatr. Blood Cancer 2015, 62, 1252–1258. [Google Scholar] [CrossRef]
- Robinson, K.E.; Fraley, C.E.; Pearson, M.M.; Kuttesch, J.F.J.; Compas, B.E. Neurocognitive late effects of pediatric brain tumors of the posterior fossa: A quantitative review. J. Int. Neuropsychol. Soc. 2013, 19, 44–53. [Google Scholar] [CrossRef]
- Simmons, D.A.; Lartey, F.M.; Schüler, E.; Rafat, M.; King, G.; Kim, A.; Ko, R.; Semaan, S.; Gonzalez, S.; Jenkins, M.; et al. Reduced cognitive deficits after FLASH irradiation of whole mouse brain are associated with less hippocampal dendritic spine loss and neuroinflammation. Radiother. Oncol. 2019, 139, 4–10. [Google Scholar] [CrossRef]
- Wang, H.; Wei, J.; Zheng, Q.; Meng, L.; Xin, Y.; Yin, X.; Jiang, X. Radiation-induced heart disease: A review of classification, mechanism and prevention. Int. J. Biol. Sci. 2019, 15, 2128. [Google Scholar] [CrossRef]
- Darby, S.C.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Brønnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N. Engl. J. Med. 2013, 368, 987–998. [Google Scholar] [CrossRef]
- Ng, A.K.; van Leeuwen, F.E. Hodgkin lymphoma: Late effects of treatment and guidelines for surveillance. Semin. Hematol. 2016, 53, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Lipshultz, S.E.; Adams, M.J.; Colan, S.D.; Constine, L.S.; Herman, E.H.; Hsu, D.T.; Hudson, M.M.; Kremer, L.C.; Landy, D.C.; Miller, T.L.; et al. Long-term cardiovascular toxicity in children, adolescents, and young adults who receive cancer therapy: Pathophysiology, course, monitoring, management, prevention, and research directions: A scientific statement from the American Heart Association. Circulation 2013, 128, 1927–1995. [Google Scholar] [CrossRef] [PubMed]
- Zou, B.; Schuster, J.P.; Niu, K.; Huang, Q.; Rühle, A.; Huber, P.E. Radiotherapy-induced heart disease: A review of the literature. Precis. Clin. Med. 2019, 2, 270–282. [Google Scholar] [CrossRef]
- Adams, M.J.; Lipsitz, S.R.; Colan, S.D.; Tarbell, N.J.; Treves, S.T.; Diller, L.; Greenbaum, N.; Mauch, P.; Lipshultz, S.E. Cardiovascular status in long-term survivors of Hodgkin’s disease treated with chest radiotherapy. J. Clin. Oncol. 2004, 22, 3139–3148. [Google Scholar] [CrossRef] [PubMed]
- Azizova, T.V.; Muirhead, C.R.; Druzhinina, M.B.; Grigoryeva, E.S.; Vlasenko, E.V.; Sumina, M.V.; O’Hagan, J.A.; Zhang, W.; Haylock, R.G.; Hunter, N. Cardiovascular diseases in the cohort of workers first employed at Mayak PA in 1948–1958. Radiat. Res 2010, 174, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Sanzari, J.K.; Billings, P.C.; Wilson, J.M.; Diffenderfer, E.S.; Arce-Esquivel, A.A.; Thorne, P.K.; Laughlin, M.H.; Kennedy, A.R. Effect of electron radiation on vasomotor function of the left anterior descending coronary artery. Life Sci. Space Res. 2015, 4, 6–10. [Google Scholar] [CrossRef]
- Boero, I.J.; Paravati, A.J.; Triplett, D.P.; Hwang, L.; Matsuno, R.K.; Gillespie, E.F.; Yashar, C.M.; Moiseenko, V.; Einck, J.P.; Mell, L.K.; et al. Modern Radiation Therapy and Cardiac Outcomes in Breast Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 700–708. [Google Scholar] [CrossRef]
- Liu, X.C.; Zhou, P.K. Tissue reactions and mechanism in cardiovascular diseases induced by radiation. Int. J. Mol. Sci. 2022, 23, 14786. [Google Scholar] [CrossRef]
- Lenarczyk, M.; Lam, V.; Jensen, E.; Fish, B.L.; Su, J.; Koprowski, S.; Komorowski, R.A.; Harmann, L.; Migrino, R.Q.; Li, X.A.; et al. Cardiac injury after 10 Gy total body irradiation: Indirect role of effects on abdominal organs. Radiat. Res. 2013, 180, 247–258. [Google Scholar] [CrossRef]
- Skyttä, T.; Tuohinen, S.; Boman, E.; Virtanen, V.; Raatikainen, P.; Kellokumpu-Lehtinen, P.-L. Troponin T-release associates with cardiac radiation doses during adjuvant left-sided breast cancer radiotherapy. Radiat. Oncol. 2015, 10, 141. [Google Scholar] [CrossRef]
- Shah, K.S.; Yang, E.H.; Maisel, A.S.; Fonarow, G.C. The role of biomarkers in detection of cardio-toxicity. Curr. Oncol. Rep. 2017, 19, 42. [Google Scholar] [CrossRef] [PubMed]
- Lierova, A.; Jelicova, M.; Nemcova, M.; Proksova, M.; Pejchal, J.; Zarybnicka, L.; Sinkorova, Z. Cytokines and radiation-induced pulmonary injuries. J. Radiat. Res. 2018, 59, 709–753. [Google Scholar] [CrossRef]
- Fuks, Z.; Kolesnick, R. Engaging the vascular component of the tumor response. Cancer Cell 2005, 8, 89–91. [Google Scholar] [CrossRef]
- Feng, M.; Moran, J.M.; Koelling, T.; Chughtai, A.; Chan, J.L.; Freedman, L.; Hayman, J.A.; Jagsi, R.; Jolly, S.; Larouere, J.; et al. Development and validation of a heart atlas to study cardiac exposure to radiation following treatment for breast cancer. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Eblan, M.J.; Deal, A.M.; Lipner, M.; Zagar, T.M.; Wang, Y.; Mavroidis, P.; Lee, C.B.; Jensen, B.C.; Rosenman, J.G.; et al. Cardiac Toxicity After Radiotherapy for Stage III Non-Small-Cell Lung Cancer: Pooled Analysis of Dose-Escalation Trials Delivering 70 to 90 Gy. J. Clin. Oncol. 2017, 35, 1387–1394. [Google Scholar] [CrossRef]
- Zhang, X.J.; Sun, J.G.; Sun, J.; Ming, H.; Wang, X.X.; Wu, L.; Chen, Z.T. Prediction of radiation pneumonitis in lung cancer patients: A systematic review. J. Cancer Res. Clin. Oncol. 2012, 138, 2103–2116. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Williams, J.; Ding, I.; Hernady, E.; Liu, W.; Smudzin, T.; Finkelstein, J.N.; Rubin, P.; Okunieff, P. Radiation pneumonitis and early circulatory cytokine markers. Semin. Radiat. Oncol. 2002, 12, 26–33. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, C.; Liu, L.; A, X.; Chen, B.; Li, Y.; Du, J. Macrophage-Derived mir-155-Containing Exosomes Suppress Fibroblast Proliferation and Promote Fibroblast Inflammation during Cardiac Injury. Mol. Ther. 2017, 25, 192–204. [Google Scholar] [CrossRef]
- Waldo, C.; Denisse, M.; Quintero-Millán, X.; Negrete-García, M.C.; Ruiz, V.; Sommer, B.; Romero-Rodríguez, D.P.; Montes-Martínez, E. Circulating MicroRNAs in Idiopathic Pulmonary Fibrosis: A Narrative Review. Curr. Issues Mol. Biol. 2024, 46, 13746–13766. [Google Scholar] [CrossRef]
- Bonella, F.; Spagnolo, P.; Ryerson, C. Current and Future Treatment Landscape for Idiopathic Pulmonary Fibrosis. Drugs 2023, 83, 1581–1593. [Google Scholar] [CrossRef]
- Ruysscher, D.D.; Wauters, E.; Jendrossek, V.; Filippi, A.R.; Revel, M.-P.; Faivre-Finn, C.; Naidoo, J.; Ramella, S.; Guckenberger, M.; Ricardi, U.; et al. Diagnosis and treatment of radiation induced pneumonitis in patients with lung cancer: An ESTRO clinical practice guideline. Radiother. Oncol. 2025, 207, 110837. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, G.; Yan, O.; Wang, S.; Wang, X. Radiation-induced injury and the gut microbiota: Insights from a microbial perspective. Ther. Adv. Gastroenterol. 2025, 18, 17562848251347347. [Google Scholar] [CrossRef]
- Andreyev, H.; Davidson, S.; Gillespie, C.; Allum, W.; Swarbrick, E. Practice guidance on the management of acute and chronic gastrointestinal problems arising as a result of treatment for cancer. Gut 2012, 61, 179–192. [Google Scholar] [CrossRef]
- Wang, A.; Ling, Z.; Yang, Z.; Kiela, P.; Wang, T.; Wang, C.; Cao, L.; Geng, F.; Shen, M.; Ran, X.; et al. Gut microbial dysbiosis may predict diarrhea and fatigue in patients undergoing pelvic cancer radiotherapy: A pilot study. PLoS ONE 2015, 10, e0126312. [Google Scholar] [CrossRef] [PubMed]
- Paris, F.; Fuks, Z.; Kang, A.; Capodieci, P.; Juan, G.; Ehleiter, D.; Haimovitz-Friedman, A.; Cordon-Cardo, C.; Kolesnick, R. Endothelial apoptosis as the primary lesion initiating intestinal radiation damage in mice. Science 2001, 293, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Ivanov, V.N.; Lien, Y.-C.; Davidson, M.; Hei, T.K. Mitochondrial function and nuclear factor-κB–mediated signaling in radiation-induced bystander effects. Cancer Res. 2008, 68, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Xiao, H.; Li, Y.; Zhou, L.; Zhao, S.; Luo, D.; Zheng, Q.; Dong, J.; Zhao, Y.; Zhang, X.; et al. Faecal microbiota transplantation protects against radiation-induced toxicity. EMBO Mol. Med. 2017, 9, 448–461. [Google Scholar] [CrossRef]
- Ghosh, G. Radiation-induced Bystander Effect and Its Possible Countermeasures. J. Cell Signal. 2023, 4, 13–20. [Google Scholar] [CrossRef]
- Mohd Zainudin, N.H.; Talik Sisin, N.N.; Rashid, R.A.; Jamil, A.; Khairil Anuar, M.A.; Razak, K.A.; Abdullah, R.; Rahman, W.N. Cellular analysis on the radiation induced bystander effects due to bismuth oxide nanoparticles with 6 MV photon beam radiotherapy. J. Radiat. Res. Appl. Sci. 2022, 15, 318–325. [Google Scholar] [CrossRef]
- Spałek, M. Chronic radiation-induced dermatitis: Challenges and solutions. Clin. Cosmet. Investig. Dermatol. 2016, 9, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Xu, C.; Song, B.; Zhang, S.; Chen, C.; Li, C.; Zhang, S. Tissue fibrosis induced by radiotherapy: Current understanding of the molecular mechanisms, diagnosis and therapeutic advances. J. Transl. Med. 2023, 21, 708. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.; Bao, S.; Yao, L.; Wen, Z.; Xu, L.; Chen, X.; Guo, S.; Pang, H.; Zhou, Y.; et al. Deciphering the fibrotic process: Mechanism of chronic radiation skin injury fibrosis. Front. Immunol. 2024, 15, 1338922. [Google Scholar] [CrossRef]
- Otranto, M.; Sarrazy, V.; Bonté, F.; Hinz, B.; Gabbiani, G.; Desmoulière, A. The role of the myofibroblast in tumor stroma remodeling. Cell Adhes. Migr. 2012, 6, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Bukowska, J.; Kopcewicz, M.; Kur-Piotrowska, A.; Szostek-Mioduchowska, A.Z.; Walendzik, K.; Gawronska-Kozak, B. Effect of TGFβ1, TGFβ3 and keratinocyte conditioned media on functional characteristics of dermal fibroblasts derived from reparative (Balb/c) and regenerative (Foxn1 deficient; nude) mouse models. Cell Tissue Res. 2018, 374, 149–163. [Google Scholar] [CrossRef]
- Shephard, P.; Martin, G.; Smola-Hess, S.; Brunner, G.; Krieg, T.; Smola, H. Myofibroblast differentiation is induced in keratinocyte-fibroblast co-cultures and is antagonistically regulated by endogenous transforming growth factor-beta and interleukin-1. Am. J. Pathol. 2004, 164, 2055–2066. [Google Scholar] [CrossRef] [PubMed]
- Famoso, J.; Laughlin, B.; McBride, A.; Gonzalez, V. Pentoxifylline and vitamin E drug compliance after adjuvant breast radiation therapy. Adv. Radiat. Oncol. 2017, 3, 19–24. [Google Scholar] [CrossRef]
- Jacobson, G.; Bhatia, S.; Smith, B.; Button, A.; Bodeker, K.; Buatti, J. Randomized trial of pentoxifylline and vitamin E vs standard follow-up after breast irradiation to prevent breast fibrosis, evaluated by tissue compliance meter. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, 604–608. [Google Scholar] [CrossRef]
- Singh, G.; Yadav, V.; Singh, P.; Bhowmik, K. Radiation-Induced Malignancies Making Radiotherapy a “Two-Edged Sword”: A Review of Literature. World J. Oncol. 2017, 8, 1–6. [Google Scholar] [CrossRef]
- Robison, L. Treatment-associated subsequent neoplasms among long-term survivors of childhood cancer: The experience of the Childhood Cancer Survivor Study. Pediatr. Radiol. 2009, 39, S32–S37. [Google Scholar] [CrossRef]
- Demoor-Goldschmidt, C.; de Vathaire, F. Review of risk factors of secondary cancers among cancer survivors. Br. J. Radiol. 2019, 92, 20180390. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects, carcinogenesis and models. Oncogene 2003, 22, 7028–7033. [Google Scholar] [CrossRef]
- Gopinathan, L.; Gopinathan, C. Ionizing radiation-induced cancer: Perplexities of the bystander effect. Ecancermedicalscience 2023, 17, 1579. [Google Scholar] [CrossRef]
- Shemetun, O.; Pilinska, M. Radiation-induced bystander effect-modeling, manifestation, mechanisms, persistence, cancer risks (literature review). Probl. Radiac. Med. Radiobiol. 2019, 24, 65–92. [Google Scholar] [CrossRef] [PubMed]
- Dracham, C.B.; Shankar, A.; Madan, R. Radiation induced secondary malignancies: A review article. Radiat. Oncol. J. 2018, 36, 85–94. [Google Scholar] [CrossRef]
- Morton, L.M.; Onel, K.; Curtis, R.E.; Hungate, E.A.; Armstrong, G.T. The Rising Incidence of Second Cancers: Patterns of Occurrence and Identification of Risk Factors for Children and Adults. Am. Soc. Clin. Oncol. Educ. Book. 2014, 34, e57–e67. [Google Scholar] [CrossRef]
- Berrington de Gonzalez, A.; Gilbert, E.; Curtis, R.; Inskip, P.; Kleinerman, R.; Morton, L.; Rajaraman, P.; Little, M.P. Second solid cancers after radiation therapy: A systematic review of the epidemiologic studies of the radiation dose-response relationship. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 224–233. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.M.; Donaldson, S.S.; Balise, R.R.; Whittemore, A.S.; Link, M.P. Second malignant neoplasms in survivors of pediatric Hodgkin’s lymphoma treated with low-dose radiation and chemotherapy. J. Clin. Oncol. 2010, 28, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Wolden, S.L.; Lamborn, K.R.; Cleary, S.F.; Tate, D.J.; Donaldson, S.S. Second cancers following pediatric Hodgkin’s disease. J. Clin. Oncol. 1998, 16, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Chadha, V.D. Role of epigenetic mechanisms in propagating off-targeted effects following radiation based therapies–A review. Mutat. Res./Rev. Mutat. Res. 2021, 787, 108370. [Google Scholar] [CrossRef]
- Armstrong, G.T.; Liu, Q.; Yasui, Y.; Huang, S.; Ness, K.K.; Leisenring, W.; Hudson, M.M.; Donaldson, S.S.; King, A.A.; Stovall, M.; et al. Long-term outcomes among adult survivors of childhood central nervous system malignancies in the Childhood Cancer Survivor Study. JNCI J. Natl. Cancer Inst. 2009, 101, 946–958. [Google Scholar] [CrossRef]
- Oeffinger, K.C.; Mertens, A.C.; Sklar, C.A.; Kawashima, T.; Hudson, M.M.; Meadows, A.T.; Friedman, D.L.; Neyssa Marina, M.D.; Wendy Hobbie, M.D.; Wendy Leisenring, M.D.; et al. Chronic health conditions in adult survivors of childhood cancer. N. Engl. J. Med. 2006, 355, 1572–1582. [Google Scholar] [CrossRef]
- Constine, L.S.; Woolf, P.D.; Cann, D.; Mick, G.; McCormick, K.; Raubertas, R.F.; Rubin, P. Hypothalamic-pituitary dysfunction after radiation for brain tumors. N. Engl. J. Med. 1993, 328, 87–94. [Google Scholar] [CrossRef]
- Tamminga, J.; Kovalchuk, O. Role of DNA damage and epigenetic DNA methylation changes in radiation-induced genomic instability and bystander effects in germline in vivo. Curr. Mol. Pharmacol. 2011, 4, 115–125. [Google Scholar] [CrossRef]
- Monje, M.; Dietrich, J. Cognitive side effects of cancer therapy demonstrate a functional role for adult neurogenesis. Behav. Brain Res. 2012, 227, 376–379. [Google Scholar] [CrossRef]
- Rola, R.; Raber, J.; Rizk, A.; Otsuka, S.; VandenBerg, S.; Morhardt, D.; Fike, J. Radiation-induced impairment of hippocampal neurogenesis is associated with cognitive deficits in young mice. Exp. Neurol. 2004, 188, 316–330. [Google Scholar] [CrossRef] [PubMed]
- Zeller, B.; Tamnes, C.K.; Kanellopoulos, A.; Amlien, I.K.; Andersson, S.; Due-Tønnessen, P.; Fjell, A.M.; Walhovd, K.B.; Westlye, L.T.; Ruud, E. Reduced neuroanatomic volumes in long-term survivors of childhood acute lymphoblastic leukemia. J. Clin. Oncol. 2013, 31, 2078–2085. [Google Scholar] [CrossRef]
- Reddick, W.E.; Glass, J.O.; Palmer, S.L.; Wu, S.; Gajjar, A.; Langston, J.W.; Kun, L.E.; Xiong, X.; Mulhern, R.K. Atypical white matter volume development in children following craniospinal irradiation. Neuro-Oncology 2005, 7, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Morganti, J.M.; Jopson, T.D.; Liu, S.; Gupta, N.; Rosi, S. Cranial irradiation alters the brain’s microenvironment and permits CCR2+ macrophage infiltration. PLoS ONE 2014, 9, e93650. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tian, J.; Liu, D.; Li, T.; Mao, Y.; Zhu, C. Microglia in radiation-induced brain injury: Cellular and molecular mechanisms and therapeutic potential. CNS Neurosci. Ther. 2024, 30, e14794. [Google Scholar] [CrossRef]
- King, T.Z.; Wang, L.; Mao, H. Disruption of white matter integrity in adult survivors of childhood brain tumors: Correlates with long-term intellectual outcomes. PLoS ONE 2015, 10, e0131744. [Google Scholar] [CrossRef]
- Palmer, S.L.; Glass, J.O.; Li, Y.; Ogg, R.; Qaddoumi, I.; Armstrong, G.T.; Wright, K. White matter integrity is associated with cognitive processing in patients treated for a posterior fossa brain tumor. Neuro-Oncology 2012, 14, 1185–1193. [Google Scholar] [CrossRef]
- Morgan, W.F.; Sowa, M.B. Non-targeted effects induced by ionizing radiation: Mechanisms and potential impact on radiation induced health effects. Cancer Lett. 2015, 356, 17–21. [Google Scholar] [CrossRef]
- Citrin, D.; Cotrim, A.P.; Hyodo, F.; Baum, B.J.; Krishna, M.C.; Mitchell, J.B. Radioprotectors and mitigators of radiation-induced normal tissue injury. Oncologist 2010, 15, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Lorimore, S.A.; Coates, P.J.; Scobie, G.E.; Milne, G.; Wright, E.G. Inflammatory-type responses after exposure to ionizing radiation in vivo: A mechanism for radiation-induced bystander effects? Oncogene 2001, 20, 7085–7095. [Google Scholar] [CrossRef] [PubMed]
- Oeffinger, K.C.; Argenbright, K.E.; Levitt, G.A.; McCabe, M.S.; Anderson, P.R.; Berry, E.; Maher, J.; Merrill, J.; Wollins, D.S. Models of cancer survivorship health care: Moving forward. Am. Soc. Clin. Oncol. Educ. Book 2014, 34, 205–213. [Google Scholar] [CrossRef]
- Sawal, H.; Asghar, K.; Bureik, M.; Jalal, N. Bystander signaling via oxidative metabolism. Onco Targets Ther. 2017, 10, 3925–3940. [Google Scholar] [CrossRef]
- Schaue, D.; Kachikwu, E.L.; McBride, W.H. Cytokines in radiobiological responses: A review. Radiat. Res. 2012, 178, 505–523. [Google Scholar] [CrossRef]
- Azzam, E.I.; de Toledo, S.M.; Little, J.B. Oxidative metabolism, gap junctions and the ionizing radiation-induced bystander effect. Oncogene 2003, 22, 7050–7057. [Google Scholar] [CrossRef]
- Gilman, K.E.; Camden, J.M.; Woods, L.T.; Weisman, G.A.; Limesand, K.H. Indomethacin Treatment Post-irradiation Improves Mouse Parotid Salivary Gland Function via Modulation of Prostaglandin E(2) Signaling. Front. Bioeng. Biotechnol. 2021, 9, 697671. [Google Scholar] [CrossRef]
- Shao, C.; Folkard, M.; Prise, K.M. Role of TGF-β1 and nitric oxide in the bystander response of irradiated glioma cells. Oncogene 2008, 27, 434–440. [Google Scholar] [CrossRef]
- Laube, M.; Kniess, T.; Pietzsch, J. Development of Antioxidant COX-2 Inhibitors as Radioprotective Agents for Radiation Therapy—A Hypothesis-Driven Review. Antioxidants 2016, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Shedid, S.M.; Abdel-Aziz, N.; Algeda, F.R.; Saada, H.N. The Mitigating Effect of Melatonin Against Radiation-Induced Inflammation and Disturbance of Reproductive Hormones in Female Albino Rats. Dose-Response 2025, 23, 15593258251323796. [Google Scholar] [CrossRef]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin: A Versatile Protector against Oxidative DNA Damage. Molecules 2018, 23, 530. [Google Scholar] [CrossRef]
- Shimura, T.; Shiga, R.; Sasatani, M.; Kamiya, K.; Ushiyama, A. Melatonin and MitoEbselen-2 Are Radioprotective Agents to Mitochondria. Genes 2022, 14, 45. [Google Scholar] [CrossRef]
- Esmaely, F.; Mahmoudzadeh, A.; Cheki, M.; Shirazi, A. The radioprotective effect of melatonin against radiation-induced DNA double-strand breaks in radiology. J. Cancer Res. Ther. 2020, 16, S59–S63. [Google Scholar]
- Abdullaev, S.A.; Glukhov, S.I.; Gaziev, A.I. Radioprotective and Radiomitigative Effects of Melatonin in Tissues with Different Proliferative Activity. Antioxidants 2021, 10, 1885. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Bernatoniene, J. Molecular Mechanisms of Melatonin-Mediated Cell Protection and Signaling in Health and Disease. Pharmaceutics 2021, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Ait-Aissa, K.; Leng, L.N.; Lindsey, N.R.; Guo, X.; Juhr, D.; Koval, O.M.; Grumbach, I.M. Mechanisms by which statins protect endothelial cells from radiation-induced injury in the carotid artery. Front. Cardiovasc. Med. 2023, 10, 1133315. [Google Scholar] [CrossRef]
- Camara Planek, M.I.; Silver, A.J.; Volgman, A.S.; Okwuosa, T.M. Exploratory Review of the Role of Statins, Colchicine, and Aspirin for the Prevention of Radiation-Associated Cardiovascular Disease and Mortality. J. Am. Heart Assoc. 2020, 9, e014668. [Google Scholar] [CrossRef]
- Ziegler, V.; Henninger, C.; Simiantonakis, I.; Buchholzer, M.; Ahmadian, M.R.; Budach, W.; Fritz, G. Rho inhibition by lovastatin affects apoptosis and DSB repair of primary human lung cells in vitro and lung tissue in vivo following fractionated irradiation. Cell Death Dis. 2017, 8, e2978. [Google Scholar] [CrossRef]
- Algire, C.; Moiseeva, O.; Deschênes-Simard, X.; Amrein, L.; Petruccelli, L.; Birman, E.; Viollet, B.; Ferbeyre, G.; Pollak, M.N. Metformin reduces endogenous reactive oxygen species and associated DNA damage. Cancer Prev. Res. 2012, 5, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Song, C.W.; Lee, H.; Dings, R.P.M.; Williams, B.; Powers, J.; Santos, T.D.; Choi, B.-H.; Park, H.J. Metformin kills and radiosensitizes cancer cells and preferentially kills cancer stem cells. Sci. Rep. 2012, 2, 362. [Google Scholar] [CrossRef]
- Minafra, L.; Porcino, N.; Bravatà, V.; Gaglio, D.; Bonanomi, M.; Amore, E.; Cammarata, F.P.; Russo, G.; Militello, C.; Savoca, G.; et al. Radiosensitizing effect of curcumin-loaded lipid nanoparticles in breast cancer cells. Sci. Rep. 2019, 9, 11134. [Google Scholar] [CrossRef]
- Mader, S.; Pantel, K. Liquid biopsy: Current status and future perspectives. Oncol. Res. Treat. 2017, 40, 404–408. [Google Scholar] [CrossRef]
- McLaren, D.B.; Aitman, T.J. Redefining precision radiotherapy through liquid biopsy. Br. J. Cancer 2023, 129, 900–903. [Google Scholar] [CrossRef]
- Khanvilkar, S.; Mittra, I. Copper Imparts a New Therapeutic Property to Resveratrol by Generating ROS to Deactivate Cell-Free Chromatin. Pharmaceuticals 2025, 18, 132. [Google Scholar] [CrossRef] [PubMed]
- Mittra, I. Exploiting the damaging effects of ROS for therapeutic use by deactivating cell-free chromatin: The alchemy of resveratrol and copper. Front. Pharmacol. 2024, 15, 1345786. [Google Scholar] [CrossRef] [PubMed]
- Pal, K.; Raghuram, G.V.; Dsouza, J.; Shinde, S.; Jadhav, V.; Shaikh, A.; Rane, B.; Tandel, H.; Kondhalkar, D.; Chaudhary, S.; et al. A pro-oxidant combination of resveratrol and copper down-regulates multiple biological hallmarks of ageing and neurodegeneration in mice. Sci. Rep. 2022, 12, 17209. [Google Scholar] [CrossRef]
- Alekseeva, L.; Sen’kova, A.; Savin, I.; Zenkova, M.; Mironova, N. Human Recombinant DNase I (Pulmozyme®) Inhibits Lung Metastases in Murine Metastatic B16 Melanoma Model That Correlates with Restoration of the DNase Activity and the Decrease SINE/LINE and c-Myc Fragments in Blood Cell-Free DNA. Int. J. Mol. Sci. 2021, 22, 12074. [Google Scholar] [CrossRef]
- Ray, P.; Haideri, N.; Haque, I.; Mohammed, O.; Chakraborty, S.; Banerjee, S.; Quadir, M.; Brinker, A.E.; Banerjee, S.K. The Impact of Nanoparticles on the Immune System: A Gray Zone of Nanomedicine. J. Immunol. Sci. 2021, 5, 19–33. [Google Scholar] [CrossRef]
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K.; et al. Potential Adverse Effects of Resveratrol: A Literature Review. Int. J. Mol. Sci. 2020, 21, 2084. [Google Scholar] [CrossRef]
- Sahini, S.S.; Pazouki, N.; Rodrigues, K.S. Resveratrol and Copper Nutraceutical Combination: Protection against Chemotherapy-Induced Toxicity. Med. Res. J. 2024. Available online: https://www.medicalandresearch.com/current_issue/2104 (accessed on 3 November 2025).
- Du, Y.; Du, S.; Liu, L.; Gan, F.; Jiang, X.; Wangrao, K.; Lyu, P.; Gong, P.; Yao, Y. Radiation-Induced Bystander Effect can be Transmitted Through Exosomes Using miRNAs as Effector Molecules. Radiat. Res. 2020, 194, 89–100. [Google Scholar] [CrossRef]
- Mo, L.J.; Song, M.; Huang, Q.H.; Guan, H.; Liu, X.D.; Xie, D.F.; Huang, B.; Huang, R.X.; Zhou, P.K. Exosome-packaged miR-1246 contributes to bystander DNA damage by targeting LIG4. Br. J. Cancer 2018, 119, 492–502. [Google Scholar] [CrossRef] [PubMed]
- Mann, M.; Mehta, A.; Zhao, J.L.; Lee, K.; Marinov, G.K.; Garcia-Flores, Y.; Lu, L.-F.; Rudensky, A.Y.; Baltimore, D. An NF-κB-microRNA regulatory network tunes macrophage inflammatory responses. Nat. Commun. 2017, 8, 851. [Google Scholar] [CrossRef] [PubMed]
- Kovalchuk, O.; Zemp, F.J.; Filkowski, J.N.; Altamirano, A.M.; Dickey, J.S.; Jenkins-Baker, G.; Marino, S.A.; Brenner, D.J.; Bonner, W.M.; Sedelnikova, O.A. microRNAome changes in bystander three-dimensional human tissue models suggest priming of apoptotic pathways. Carcinogenesis 2010, 31, 1882–1888. [Google Scholar] [CrossRef]
- Akkaya-Ulum, Y.Z.; Sen, B.; Akbaba, T.H.; Balci-Peynircioglu, B. InflammamiRs in focus: Delivery strategies and therapeutic approaches. FASEB J. 2024, 38, e23528. [Google Scholar] [CrossRef]
- Yuan, X.; Berg, N.; Lee, J.W.; Le, T.T.; Neudecker, V.; Jing, N.; Eltzschig, H. MicroRNA miR-223 as regulator of innate immunity. J. Leukoc. Biol. 2018, 104, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Irep, N.; Inci, K.; Tokgun, P.E.; Tokgun, O. Exosome inhibition improves response to first-line therapy in small cell lung cancer. J. Cell Mol. Med. 2024, 28, e18138. [Google Scholar] [CrossRef]
- Lee, Y.-C.; Chien, P.-J.; Chang, Y.-T.; Huang, Y.-H.; Chang, C.-F.; Li, S.-T.; Chang, W.-W. Radioresistant triple-negative breast cancer cells release β-catenin containing extracellular vesicles to promote cancer stem cell activity of bystanders. J. Cancer 2025, 16, 2890–2902. [Google Scholar] [CrossRef] [PubMed]
- Mrowczynski, O.D.; Madhankumar, A.B.; Sundstrom, J.M.; Zhao, Y.; Kawasawa, Y.I.; Slagle-Webb, B.; Mau, C.; Payne, R.A.; Rizk, E.B.; Zacharia, B.E.; et al. Exosomes impact survival to radiation exposure in cell line models of nervous system cancer. Oncotarget 2018, 9, 36083–36101. [Google Scholar] [CrossRef] [PubMed]
- Neudecker, V.; Haneklaus, M.; Jensen, O.; Khailova, L.; Masterson, J.C.; Tye, H.; Biette, K.; Jedlicka, P.; Brodsky, K.S.; Gerich, M.E.; et al. Myeloid-derived miR-223 regulates intestinal inflammation via repression of the NLRP3 inflammasome. J. Exp. Med. 2017, 214, 1737–1752. [Google Scholar] [CrossRef]
- Ohshima, Y.; Tsukimoto, M.; Harada, H.; Kojima, S. Involvement of connexin43 hemichannel in ATP release after γ-irradiation. J. Radiat. Res. 2012, 53, 551–557. [Google Scholar] [CrossRef]
- Tsukimoto, M.; Homma, T.; Ohshima, Y.; Kojima, S. Involvement of purinergic signaling in cellular response to gamma radiation. Radiat. Res. 2010, 173, 298–309. [Google Scholar] [CrossRef]
- Klumpp, D.; Misovic, M.; Szteyn, K.; Shumilina, E.; Rudner, J.; Huber, S.M. Targeting TRPM2 Channels Impairs Radiation-Induced Cell Cycle Arrest and Fosters Cell Death of T Cell Leukemia Cells in a Bcl-2-Dependent Manner. Oxidative Med. Cell. Longev. 2016, 2016, 8026702. [Google Scholar] [CrossRef]
- Nishimaki, N.; Tsukimoto, M.; Kitami, A.; Kojima, S. Autocrine regulation of γ-irradiation-induced DNA damage response via extracellular nucleotides-mediated activation of P2Y6 and P2Y12 receptors. DNA Repair 2012, 11, 657–665. [Google Scholar] [CrossRef]
- Ramadan, R.; Vromans, E.; Anang, D.C.; Decrock, E.; Mysara, M.; Monsieurs, P.; Baatout, S.; Leybaert, L.; Aerts, A. Single and fractionated ionizing radiation induce alterations in endothelial connexin expression and channel function. Sci. Rep. 2019, 9, 4643. [Google Scholar] [CrossRef]
- Kim, H.J.; Kang, S.U.; Lee, Y.S.; Jang, J.Y.; Kang, H.; Kim, C.H. Protective Effects of N-Acetylcysteine against Radiation-Induced Oral Mucositis In Vitro and In Vivo. Cancer Res. Treat. 2020, 52, 1019–1030. [Google Scholar] [CrossRef]
- Bao, X.; Liu, X.; Wu, Q.; Ye, F.; Shi, Z.; Xu, D.; Zhang, J.; Dou, Z.; Huang, G.; Zhang, H.; et al. Mitochondrial-Targeted Antioxidant MitoQ-Mediated Autophagy: A Novel Strategy for Precise Radiation Protection. Antioxidants 2023, 12, 453. [Google Scholar] [CrossRef]
- Lewis, J.E.; Singh, N.; Holmila, R.J.; Sumer, B.D.; Williams, N.S.; Furdui, C.M.; Kemp, M.L.; Boothman, D.A. Targeting NAD(+) Metabolism to Enhance Radiation Therapy Responses. Semin. Radiat. Oncol. 2019, 29, 6–15. [Google Scholar] [CrossRef]
- Judge, J.L.; Owens, K.M.; Pollock, S.J.; Woeller, C.F.; Thatcher, T.H.; Williams, J.P.; Phipps, R.P.; Sime, P.J.; Kottmann, R.M. Ionizing radiation induces myofibroblast differentiation via lactate dehydrogenase. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L879–L887. [Google Scholar] [CrossRef]
- Dwarkanath, B.S.; Zolzer, F.; Chandana, S.; Bauch, T.; Adhikari, J.S.; Muller, W.U.; Streffer, C.; Jain, V. Heterogeneity in 2-deoxy-D-glucose-induced modifications in energetics and radiation responses of human tumor cell lines. Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 1051–1061. [Google Scholar] [CrossRef]
- Siteni, S.; Barron, S.; Luitel, K.; Shay, J.W. Radioprotective effect of the anti-diabetic drug metformin. PLoS ONE 2024, 19, e0307598. [Google Scholar] [CrossRef] [PubMed]
- Muaddi, H.; Chowdhury, S.; Vellanki, R.; Zamiara, P.; Koritzinsky, M. Contributions of AMPK and p53 dependent signaling to radiation response in the presence of metformin. Radiother. Oncol. 2013, 108, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Kuefner, M.A.; Brand, M.; Ehrlich, J.; Braga, L.; Uder, M.; Semelka, R.C. Effect of antioxidants on X-ray-induced γ-H2AX foci in human blood lymphocytes: Preliminary observations. Radiology 2012, 264, 59–67. [Google Scholar] [CrossRef]
- Li, H.; Yang, T.; Zhang, J.; Xue, K.; Ma, X.; Yu, B.; Jin, X. Pyroptotic cell death: An emerging therapeutic opportunity for radiotherapy. Cell Death Discov. 2024, 10, 32. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, Q.; Chu, Y.; Xu, B.; Song, Q. The Influence of Radiotherapy on AIM2 Inflammasome in Radiation Pneumonitis. Inflammation 2016, 39, 1827–1834. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Jin, C.; Li, H.-B.; Tong, J.; Ouyang, X.; Cetinbas, N.M.; Zhu, S.; Strowig, T.; Lam, F.C.; Zhao, C.; et al. The DNA-sensing AIM2 inflammasome controls radiation-induced cell death and tissue injury. Science 2016, 354, 765–768. [Google Scholar] [CrossRef]
- Liu, Z.; Luo, S.; Wang, Z.; Xu, Y.; He, J.; Wang, W.; He, S.; Ai, X.; Li, C.; Cao, W. MCC950, a NLRP3 inflammasome inhibitor, prevents radiation-induced cognitive impairment in mice. Chin. J. Radiol. Med. Prot. 2020, 733–739. [Google Scholar]
- Cheng, H.; Chen, L.; Huang, M.; Hou, J.; Chen, Z.; Yang, X. Hunting down NLRP3 inflammasome: An executioner of radiation-induced injury. Front. Immunol. 2022, 13, 967989. [Google Scholar] [CrossRef]
- Christersdottir, T.; Pirault, J.; Gisterå, A.; Bergman, O.; Gallina, A.L.; Baumgartner, R.; Lundberg, A.M.; Eriksson, P.; Yan, Z.Q.; Paulsson-Berne, G.; et al. Prevention of radiotherapy-induced arterial inflammation by interleukin-1 blockade. Eur. Heart J. 2019, 40, 2495–2503. [Google Scholar] [CrossRef]
- Jia, Y.; Huang, T. Overview of Antabuse® (Disulfiram) in Radiation and Cancer Biology. Cancer Manag. Res. 2021, 13, 4095–4101. [Google Scholar] [CrossRef]
- Grujicic, J.; Allen, A.R. MnSOD Mimetics in Therapy: Exploring Their Role in Combating Oxidative Stress-Related Diseases. Antioxidants 2024, 13, 1444. [Google Scholar] [CrossRef]
- Liu, W.; Ding, I.; Chen, K.; Olschowka, J.; Xu, J.; Hu, D.; Morrow, G.R.; Okunieff, P. Interleukin 1beta (IL1B) signaling is a critical component of radiation-induced skin fibrosis. Radiat. Res. 2006, 165, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Brown, S.L.; Gordon, M.N. Radiation-induced senescence: Therapeutic opportunities. Radiat. Oncol. 2023, 18, 10. [Google Scholar] [CrossRef] [PubMed]
- Hickson, L.J.; Langhi Prata, L.G.P.; Bobart, S.A.; Evans, T.K.; Giorgadze, N.; Hashmi, S.K.; Herrmann, S.M.; Jensen, M.D.; Jia, Q.; Jordan, K.L.; et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine 2019, 47, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Yousefzadeh, M.J.; Zhu, Y.; McGowan, S.J.; Angelini, L.; Fuhrmann-Stroissnigg, H.; Xu, M.; Ling, Y.Y.; Melos, K.I.; Pirtskhalava, T.; Inman, C.L.; et al. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine 2018, 36, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Li, Y.; Wan, C.; Sun, Y.; Dai, X.; Huang, J.; Hu, Y.; Gao, Y.; Wu, B.; Zhang, Z.; et al. Targeting senescence-like fibroblasts radiosensitizes non-small cell lung cancer and reduces radiation-induced pulmonary fibrosis. JCI Insight 2021, 6, e146334. [Google Scholar] [CrossRef]
- Fielder, E.; Wan, T.; Alimohammadiha, G.; Ishaq, A.; Low, E.; Weigand, B.M.; Kelly, G.; Parker, C.; Griffin, B.; Jurk, D. Short senolytic or senostatic interventions rescue progression of radiation-induced frailty and premature ageing in mice. eLife 2022, 11, e75492. [Google Scholar] [CrossRef]
- Iglesias-Bartolome, R.; Patel, V.; Cotrim, A.; Leelahavanichkul, K.; Molinolo, A.A.; Mitchell, J.B.; Gutkind, J.S. mTOR inhibition prevents epithelial stem cell senescence and protects from radiation-induced mucositis. Cell Stem Cell 2012, 11, 401–414. [Google Scholar] [CrossRef]
- de Vos, S.; Leonard, J.P.; Friedberg, J.W.; Zain, J.; Dunleavy, K.; Humerickhouse, R.; Hayslip, J.; Pesko, J.; Wilson, W.H. Safety and efficacy of navitoclax, a BCL-2 and BCL-X(L) inhibitor, in patients with relapsed or refractory lymphoid malignancies: Results from a phase 2a study. Leuk. Lymphoma 2021, 62, 810–818. [Google Scholar] [CrossRef]
- Ganz, P.A. Survivorship: Adult cancer survivors. Prim. Care Clin. Off. Pract. 2009, 36, 721–741. [Google Scholar] [CrossRef]
- Baumann, M.; Krause, M.; Overgaard, J.; Debus, J.; Bentzen, S.M.; Daartz, J.; Richter, C.; Zips, D.; Bortfeld, T. Radiation oncology in the era of precision medicine. Nat. Rev. Cancer 2016, 16, 234–249. [Google Scholar] [CrossRef] [PubMed]
- Barnett, G.C.; West, C.M.; Dunning, A.M.; Elliott, R.M.; Coles, C.E.; Pharoah, P.D.; Burnet, N.G. Normal tissue reactions to radiotherapy: Towards tailoring treatment dose by genotype. Nat. Rev. Cancer 2009, 9, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Shu, Y.; Zhou, H.; Zhang, W.; Wang, H. Radiogenomics helps to achieve personalized therapy by evaluating patient responses to radiation treatment. Carcinogenesis 2015, 36, 307–317. [Google Scholar] [CrossRef][Green Version]
- Chang-Claude, J.; Ambrosone, C.B.; Lilla, C.; Kropp, S.; Helmbold, I.; Von Fournier, D.; Haase, W. Genetic polymorphisms in DNA repair and damage response genes and late normal tissue complications of radiotherapy for breast cancer. Br. J. Cancer 2009, 100, 1680–1686. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gong, Z.; Platek, M.E.; Till, C.; Goodman, P.J.; Tangen, C.M.; Platz, E.A.; Neuhouser, M.L.; Thompson, I.M.; Santella, R.M.; Ambrosone, C.B. Associations between polymorphisms in genes related to oxidative stress and DNA repair, interactions with serum antioxidants, and prostate cancer risk: Results from the prostate cancer prevention trial. Front. Oncol. 2022, 11, 808715. [Google Scholar] [CrossRef]
- Yan, Z.; Tong, X.; Ma, Y.; Liu, S.; Yang, L.; Yang, X.; Yang, X.; Bai, M.; Fan, H. Association between ATM gene polymorphisms, lung cancer susceptibility and radiation-induced pneumonitis: A meta-analysis. BMC Pulm. Med. 2017, 17, 205. [Google Scholar] [CrossRef]
- McDuff, S.G.; Bellon, J.R.; Shannon, K.M.; Gadd, M.A.; Dunn, S.; Rosenstein, B.S.; Ho, A.Y. ATM variants in breast cancer: Implications for breast radiation therapy treatment recommendations. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 1373–1382. [Google Scholar] [CrossRef]
- Fernet, M.; Hall, J. Genetic biomarkers of therapeutic radiation sensitivity. DNA Repair 2004, 3, 1237–1243. [Google Scholar] [CrossRef]
- Singh, V.K.; Seed, T.M. Pharmacological management of ionizing radiation injuries: Current and prospective agents and targeted organ systems. Expert Opin. Pharmacother. 2020, 21, 317–337. [Google Scholar] [CrossRef] [PubMed]
- Obrador, E.; Salvador, R.; Villaescusa, J.I.; Soriano, J.M.; Estrela, J.M.; Montoro, A. Radioprotection and radiomitigation: From the bench to clinical practice. Biomedicines 2020, 8, 461. [Google Scholar] [CrossRef] [PubMed]
- Kouvaris, J.R.; Kouloulias, V.E.; Vlahos, L.J. Amifostine: The first selective-target and broad-spectrum radioprotector. Oncologist 2007, 12, 738–747. [Google Scholar] [CrossRef]
- Holmlund, J.; Brill, J.M.; Lee, C.M.; Saunders, D.; Sonis, S.T.; Downs, M.; Anderson, C.M. Roman: Reduction in oral mucositis with avasopasem manganese (GC4419)—Phase 3 trial in patients receiving chemoradiotherapy for locally-advanced, non-metastatic head and neck cancer. J. Clin. Oncol. 2019, 37, 2019. [Google Scholar] [CrossRef]
- Mapuskar, K.A.; Anderson, C.M.; Spitz, D.R.; Batinic-Haberle, I.; Allen, B.G.; Oberley-Deegan, R.E. Utilizing superoxide dismutase mimetics to enhance radiation therapy response while protecting normal tissues. Semin. Radiat. Oncol. 2019, 29, 72–80. [Google Scholar] [CrossRef]
- Sonis, S.T. Superoxide dismutase as an intervention for radiation therapy-associated toxicities: Review and profile of avasopasem manganese as a treatment option for radiation-induced mucositis. Drug Des. Dev. Ther. 2021, 1021–1029. [Google Scholar] [CrossRef]
- Yamanaka, G.; Ishida, Y.; Kanou, K.; Suzuki, S.; Watanabe, Y.; Takamatsu, T.; Morichi, S.; Go, S.; Oana, S.; Yamazaki, T. Towards a Treatment for Neuroinflammation in Epilepsy: Interleukin-1 Receptor Antagonist, Anakinra, as a Potential Treatment in Intractable Epilepsy. Int. J. Mol. Sci. 2021, 22, 6282. [Google Scholar] [CrossRef]
- Weichselbaum, R.R.; Liang, H.; Deng, L.; Fu, Y.-X. Radiotherapy and immunotherapy: A beneficial liaison? Nat. Rev. Clin. Oncol. 2017, 14, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Mole, R.H. Whole body irradiation—Radiobiology or medicine? Br. J. Radiol. 1953, 26, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Reynders, K.; Illidge, T.; Siva, S.; Chang, J.Y.; De Ruysscher, D. The abscopal effect of local radiotherapy: Using immunotherapy to make a rare event clinically relevant. Cancer Treat. Rev. 2015, 41, 503–510. [Google Scholar] [CrossRef]
- Theelen, W.S.; Chen, D.; Verma, V.; Hobbs, B.P.; Peulen, H.M.; Aerts, J.G.; Bahce, I. Pembrolizumab with or without radiotherapy for metastatic non-small-cell lung cancer: A pooled analysis of two randomised trials. Lancet Respir. Med. 2021, 9, 467–475. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N. Engl. J. Med. 2018, 379, 2342–2350. [Google Scholar] [CrossRef]
- Santin, A.D.; Deng, W.; Frumovitz, M.; Buza, N.; Bellone, S.; Huh, W.; Khleif, S. Phase II evaluation of nivolumab in the treatment of persistent or recurrent cervical cancer (NCT02257528/NRG-GY002). Gynecol. Oncol. 2020, 157, 161–166. [Google Scholar] [CrossRef]
- Golden, E.B.; Frances, D.; Pellicciotta, I.; Demaria, S.; Barcellos-Hoff, M.H.; Formenti, S.C. Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death. Oncoimmunology 2014, 3, e28518. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, M.J. Targeting the DNA damage response in cancer. Mol. Cell 2015, 60, 547–560. [Google Scholar] [CrossRef]
- Mahdi, H.; Hafez, N.; Doroshow, D.; Sohal, D.; Keedy, V.; Do, K.T.; LoRusso, P. Ceralasertib-mediated ATR inhibition combined with olaparib in advanced cancers harboring DNA damage response and repair alterations (olaparib combinations). JCO Precis. Oncol. 2021, 5, 1432–1442. [Google Scholar] [CrossRef]
- Shea, A.G.; Idrissou, M.B.; Torres, A.I.; Chen, T.; Hernandez, R.; Morris, Z.S.; Sodji, Q.H. Immunological effects of radiopharmaceutical therapy. Front. Nucl. Med. 2024, 4, 1331364. [Google Scholar] [CrossRef]
- Marcu, L.; Bezak, E.; Allen, B.J. Global comparison of targeted alpha vs targeted beta therapy for cancer: In vitro, in vivo and clinical trials. Crit. Rev. Oncol./Hematol. 2018, 123, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Dekempeneer, Y.; Keyaerts, M.; Krasniqi, A.; Puttemans, J.; Muyldermans, S.; Lahoutte, T.; D’huyvetter, M.; Devoogdt, N. Targeted alpha therapy using short-lived alpha-particles and the promise of nanobodies as targeting vehicle. Expert Opin. Biol. Ther. 2016, 16, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Pouget, J.-P.; Navarro-Teulon, I.; Bardiès, M.; Chouin, N.; Cartron, G.; Pèlegrin, A.; Azria, D. Clinical radioimmunotherapy—The role of radiobiology. Nat. Rev. Clin. Oncol. 2011, 8, 720–734. [Google Scholar] [CrossRef]
- Tranel, J.; Feng, F.Y.; St James, S.; Hope, T.A. Effect of microdistribution of alpha and beta-emitters in targeted radionuclide therapies on delivered absorbed dose in a GATE model of bone marrow. Phys. Med. Biol. 2021, 66, 035016. [Google Scholar] [CrossRef]
- Lim, J.Y.H.; Gerber, S.A.; Murphy, S.P.; Lord, E.M. Type I interferons induced by radiation therapy mediate recruitment and effector function of CD8+ T cells. Cancer Immunol. Immunother. 2014, 63, 259–271. [Google Scholar] [CrossRef]
- Ferreira, C.A.; Potluri, H.K.; Massey, C.; Grudzinski, J.J.; Carston, A.; Clemons, N.; Thickens, A.; Rosenkrans, Z.; Choi, C.; Pinchuk, A. Profound immunomodulatory effects of 225Ac-NM600 drive enhanced anti-tumor response in prostate cancer. bioRxiv 2022. [Google Scholar] [CrossRef]
- Sanli, Y.; Simsek, D.H.; Sanli, O.; Subramaniam, R.M.; Kendi, A.T. (177)Lu-PSMA Therapy in Metastatic Castration-Resistant Prostate Cancer. Biomedicines 2021, 9, 430. [Google Scholar] [CrossRef]
- Kim, J.W.; Shin, M.S.; Kang, Y.; Kang, I.; Petrylak, D.P. Immune Analysis of Radium-223 in Patients With Metastatic Prostate Cancer. Clin. Genitourin. Cancer 2018, 16, e469–e476. [Google Scholar] [CrossRef]
- Mulrooney, D.A.; Yeazel, M.W.; Kawashima, T.; Mertens, A.C.; Mitby, P.; Stovall, M.; Donaldson, S.S. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: Retrospective analysis of the Childhood Cancer Survivor Study cohort. BMJ 2009, 339, b4606. [Google Scholar] [CrossRef]
- Sak, A.; Stuschke, M. Use of γH2AX and other biomarkers of double-strand breaks during radiotherapy. Semin. Radiat. Oncol. 2010, 20, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.J.; Urbanic, J.J.; Case, L.D.; Takita, C.; Wright, J.L.; Brown, D.R.; Langefeld, C.D.; Lively, M.O.; Mitchell, S.E.; Thakrar, A.; et al. Association Between Inflammatory Biomarker C-Reactive Protein and Radiotherapy-Induced Early Adverse Skin Reactions in a Multiracial/Ethnic Breast Cancer Population. J. Clin. Oncol. 2018, 36, 2473–2482. [Google Scholar] [CrossRef]
- Gales, L.; Forsea, L.; Mitrea, D.; Stefanica, I.; Stanculescu, I.; Mitrica, R.; Georgescu, M.; Trifanescu, O.; Anghel, R.; Serbanescu, L. Antidiabetics, anthelmintics, statins, and beta-blockers as co-adjuvant drugs in cancer therapy. Medicina 2022, 58, 1239. [Google Scholar] [CrossRef]
- Olson, K.; Sands, S.A. Cognitive training programs for childhood cancer patients and survivors: A critical review and future directions. Child. Neuropsychol. 2016, 22, 509–536. [Google Scholar] [CrossRef]
- Alhaddad, L.; Osipov, A.N.; Leonov, S. Flash radiotherapy: Benefits, mechanisms, and obstacles to its clinical application. Int. J. Mol. Sci. 2024, 25, 12506. [Google Scholar] [CrossRef]
- Bourhis, J.; Montay-Gruel, P.; Jorge, P.G.; Bailat, C.; Petit, B.; Ollivier, J.; Jeanneret-Sozzi, W.; Ozsahin, M.; Bochud, F.; Moeckli, R.; et al. Clinical translation of FLASH radiotherapy: Why and how? Radiother. Oncol. 2019, 139, 11–17. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Petersson, K.; Jaccard, M.; Boivin, G.; Germond, J.F.; Petit, B.; Doenlen, R.; Favaudon, V.; Bochud, F.; Bailat, C.; et al. Irradiation in a flash: Unique sparing of memory in mice after whole brain irradiation with dose rates above 100 Gy/s. Radiother. Oncol. 2017, 124, 365–369. [Google Scholar] [CrossRef]
- Diffenderfer, E.S.; Verginadis, I.I.; Kim, M.M.; Shoniyozov, K.; Velalopoulou, A.; Goia, D.; Putt, M.; Hagan, S.; Avery, S.; Teo, K.; et al. Design, implementation, and in vivo validation of a novel proton FLASH radiation therapy system. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Vozenin, M.-C.; De Fornel, P.; Petersson, K.; Favaudon, V.; Jaccard, M.; Germond, J.-F.; Petit, B. The advantage of FLASH radiotherapy confirmed in mini-pig and cat-cancer patients. Clin. Cancer Res. 2019, 25, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Geirnaert, F.; Kerkhove, L.; Montay-Gruel, P.; Gevaert, T.; Dufait, I.; De Ridder, M. Exploring the Metabolic Impact of FLASH Radiotherapy. Cancers 2025, 17, 133. [Google Scholar] [CrossRef] [PubMed]
- Pakniyat, F.; Mozdarani, H.; Nedaie, H.A.; Mahmoudzadeh, A.; Salimi, M.; Gholami, S. Bystander Response Following High-Dose X-irradiation; Time-dependent Nature of GammaH2AX Foci and Cell Death Consequences. J. Biomed. Phys. Eng. 2023, 13, 17–28. [Google Scholar] [CrossRef]
- Buonanno, M.; Gonon, G.; Pandey, B.N.; Azzam, E.I. The intercellular communications mediating radiation-induced bystander effects and their relevance to environmental, occupational, and therapeutic exposures. Int. J. Radiat. Biol. 2023, 99, 964–982. [Google Scholar] [CrossRef]
- Pratx, G.; Kapp, D.S. A computational model of radiolytic oxygen depletion during FLASH irradiation and its effect on the oxygen enhancement ratio. Phys. Med. Biol. 2019, 64, 185005. [Google Scholar] [CrossRef]
- Newhauser, W.; Durante, M. Assessing the risk of second malignancies after modern radiotherapy. Nat. Rev. Cancer 2011, 11, 438–448. [Google Scholar] [CrossRef]
- Mu, X.; Björk-Eriksson, T.; Nill, S.; Oelfke, U.; Johansson, K.-A.; Gagliardi, G.; Johansson, L.; Karlsson, M.; Zackrisson, D.B. Does electron and proton therapy reduce the risk of radiation induced cancer after spinal irradiation for childhood medulloblastoma? A comparative treatment planning study. Acta Oncol. 2005, 44, 554–562. [Google Scholar] [CrossRef]
- Eaton, B.R.; Esiashvili, N.; Kim, S.; Patterson, B.; Weyman, E.A.; Thornton, L.T.; Mazewski, C.; MacDonald, T.J.; Ebb, D.; MacDonald, S.M.; et al. Endocrine outcomes with proton and photon radiotherapy for standard risk medulloblastoma. Neuro-Oncology 2016, 18, 881–887. [Google Scholar] [CrossRef]
- Yock, T.I.; Yeap, B.Y.; Ebb, D.H.; Weyman, E.; Eaton, B.R.; Sherry, N.A.; Jones, R.M.; MacDonald, S.M.; Pulsifer, M.B.; Lavally, B.; et al. Long-term toxic effects of proton radiotherapy for paediatric medulloblastoma: A phase 2 single-arm study. Lancet Oncol. 2016, 17, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.S.; Yock, T.I.; Nelson, K.; Xu, Y.; Keating, N.L.; Tarbell, N.J. Incidence of second malignancies among patients treated with proton versus photon radiation. Int. J. Radiat. Oncol. Biol. Phys. 2013, 87, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Indelicato, D.J.; Bates, J.E.; Mailhot Vega, R.B.; Rotondo, R.L.; Hoppe, B.S.; Morris, C.G.; Looi, W.S.; Sandler, E.S.; Aldana, P.R.; Bradley, J.A. Second tumor risk in children treated with proton therapy. Pediatr. Blood Cancer 2021, 68, e28941. [Google Scholar] [CrossRef] [PubMed]
- Vitti, E.T.; Parsons, J.L. The radiobiological effects of proton beam therapy: Impact on DNA damage and repair. Cancers 2019, 11, 946. [Google Scholar] [CrossRef]
- Durante, M.; Loeffler, J.S. Charged particles in radiation oncology. Nat. Rev. Clin. Oncol. 2010, 7, 37–43. [Google Scholar] [CrossRef]
- Tsujii, H.; Kamada, T. A review of update clinical results of carbon ion radiotherapy. Jpn. J. Clin. Oncol. 2012, 42, 670–685. [Google Scholar] [CrossRef]
- Held, K.D.; Kawamura, H.; Kaminuma, T.; Paz, A.E.S.; Yoshida, Y.; Liu, Q.; Willers, H.; Takahashi, A. Effects of charged particles on human tumor cells. Front. Oncol. 2016, 6, 23. [Google Scholar] [CrossRef]
- Ran, J.; Wang, J.; Dai, Z.; Miao, Y.; Gan, J.; Zhao, C.; Guan, Q. Irradiation-induced changes in the immunogenicity of lung cancer cell lines: Based on comparison of X-rays and carbon ions. Front. Public. Health 2021, 9, 666282. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Yang, M.; Zhang, J.; Yin, Y.; Fan, X.; Zhang, Y.; Qin, S.; Zhang, H.; Yu, F. Immunogenic cell death induction by ionizing radiation. Front. Immunol. 2021, 12, 705361. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, S.; Araki, N.; Outani, H.; Hamada, K.i.; Yoshikawa, H.; Kamada, T.; Imai, R. Complication rate, functional outcomes, and risk factors associated with carbon ion radiotherapy for patients with unresectable pelvic bone sarcoma. Cancer 2020, 126, 4188–4196. [Google Scholar] [CrossRef] [PubMed]
- Pennicooke, B.; Laufer, I.; Sahgal, A.; Varga, P.P.; Gokaslan, Z.L.; Bilsky, M.H.; Yamada, Y.J. Safety and local control of radiation therapy for chordoma of the spine and sacrum: A systematic review. Spine 2016, 41, S186–S192. [Google Scholar] [CrossRef]
- Duriseti, S.; Kavanaugh, J.; Goddu, S.; Price, A.; Knutson, N.; Reynoso, F.; Michalski, J.; Mutic, S.; Robinson, C.; Spraker, M.B. Spatially fractionated stereotactic body radiation therapy (Lattice) for large tumors. Adv. Radiat. Oncol. 2021, 6, 100639. [Google Scholar] [CrossRef]
- Yan, W.; Khan, M.K.; Wu, X.; Simone, C.B., 2nd; Fan, J.; Gressen, E.; Zhang, X.; Limoli, C.L.; Bahig, H.; Tubin, S.; et al. Spatially fractionated radiation therapy: History, present and the future. Clin. Transl. Radiat. Oncol. 2020, 20, 30–38. [Google Scholar] [CrossRef]
- Van der Meeren, A.; Monti, P.; Vandamme, M.; Squiban, C.; Wysocki, J.; Griffiths, N. Abdominal radiation exposure elicits inflammatory responses and abscopal effects in the lungs of mice. Radiat. Res. 2005, 163, 144–152. [Google Scholar] [CrossRef]
- Stoecklein, V.M.; Osuka, A.; Ishikawa, S.; Lederer, M.R.; Wanke-Jellinek, L.; Lederer, J.A. Radiation exposure induces inflammasome pathway activation in immune cells. J. Immunol. 2015, 194, 1178–1189. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C. Bystander and delayed effects after fractionated radiation exposure. Radiat. Res. 2002, 158, 626–633. [Google Scholar] [CrossRef]
- Cao, Q.; Tan, J.; Ren, Y.; Xiong, W.; Wei, X.; Cheng, W. Evaluation of Radiation Shielding Requirements and Self-shielding Characteristics for a Novel Radiosurgery System. Health Phys. 2021, 121, 506–512. [Google Scholar] [CrossRef]
- Najafi, M.; Fardid, R.; Takhshid, M.A.; Mosleh-Shirazi, M.A.; Rezaeyan, A.H.; Salajegheh, A. Radiation-Induced Oxidative Stress at Out-of-Field Lung Tissues after Pelvis Irradiation in Rats. Cell J. 2016, 18, 340–345. [Google Scholar] [CrossRef]
- Sun, L.; Inaba, Y.; Sogo, Y.; Ito, A.; Bekal, M.; Chida, K.; Moritake, T. Total body irradiation causes a chronic decrease in antioxidant levels. Sci. Rep. 2021, 11, 6716. [Google Scholar] [CrossRef] [PubMed]
- Dona Lemus, O.M.; Cao, M.; Cai, B.; Cummings, M.; Zheng, D. Adaptive Radiotherapy: Next-Generation Radiotherapy. Cancers 2024, 16, 1206. [Google Scholar] [CrossRef]
- Saw, C.B.; Brandner, E.; Selvaraj, R.; Chen, H.; Saiful Huq, M.; Heron, D.E. A review on the clinical implementation of respiratory-gated radiation therapy. Biomed. Imaging Interv. J. 2007, 3, e40. [Google Scholar] [CrossRef] [PubMed]
- Molitoris, J.K.; Diwanji, T.; Snider, J.W., 3rd; Mossahebi, S.; Samanta, S.; Badiyan, S.N.; Simone, C.B., 2nd; Mohindra, P. Advances in the use of motion management and image guidance in radiation therapy treatment for lung cancer. J. Thorac. Dis. 2018, 10, S2437–S2450. [Google Scholar] [CrossRef] [PubMed]
- West, C.M.; Barnett, G.C. Genetics and genomics of radiotherapy toxicity: Towards prediction. Genome Med. 2011, 3, 52. [Google Scholar] [CrossRef]
- Alsner, J.; Andreassen, C.N.; Overgaard, J. Genetic markers for prediction of normal tissue toxicity after radiotherapy. Semin. Radiat. Oncol. 2008, 18, 126–135. [Google Scholar] [CrossRef]
- Hosny, A.; Parmar, C.; Quackenbush, J.; Schwartz, L.H.; Aerts, H.J. Artificial intelligence in radiology. Nat. Rev. Cancer 2018, 18, 500–510. [Google Scholar] [CrossRef]
- Wu, J.; Tha, K.K.; Xing, L.; Li, R. Radiomics and radiogenomics for precision radiotherapy. J. Radiat. Res. 2018, 59, i25–i31. [Google Scholar] [CrossRef]
- Carbonara, R.; Bonomo, P.; Di Rito, A.; Didonna, V.; Gregucci, F.; Ciliberti, M.P.; Surgo, A.; Bonaparte, I.; Fiorentino, A.; Sardaro, A. Investigation of radiation-induced toxicity in head and neck cancer patients through radiomics and machine learning: A systematic review. J. Oncol. 2021, 2021, 5566508. [Google Scholar] [CrossRef]
- Kniep, H.C.; Madesta, F.; Schneider, T.; Hanning, U.; Schönfeld, M.H.; Schön, G.; Fiehler, J.; Gauer, T.; Werner, R.; Gellissen, S. Radiomics of brain MRI: Utility in prediction of metastatic tumor type. Radiology 2019, 290, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Tabatadze, N.; Savonenko, A.; Song, H.; Bandaru, V.V.; Chu, M.; Haughey, N.J. Inhibition of neutral sphingomyelinase-2 perturbs brain sphingolipid balance and spatial memory in mice. J. Neurosci. Res. 2010, 88, 2940–2951. [Google Scholar] [CrossRef] [PubMed]
- Tallon, C.; Hollinger, K.R.; Pal, A.; Bell, B.J.; Rais, R.; Tsukamoto, T.; Witwer, K.W.; Haughey, N.J.; Slusher, B.S. Nipping disease in the bud: nSMase2 inhibitors as therapeutics in extracellular vesicle-mediated diseases. Drug Discov. Today 2021, 26, 1656–1668. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.S.; Harris, E.N.; Weigel, P.H. HARE-Mediated Endocytosis of Hyaluronan and Heparin Is Targeted by Different Subsets of Three Endocytic Motifs. Int. J. Cell Biol. 2015, 2015, 524707. [Google Scholar] [CrossRef]
- Basagiannis, D.; Zografou, S.; Galanopoulou, K.; Christoforidis, S. Dynasore impairs VEGFR2 signalling in an endocytosis-independent manner. Sci. Rep. 2017, 7, 45035. [Google Scholar] [CrossRef]
- Victoir, B.; Croix, C.; Gouilleux, F.; Prié, G. Targeted Therapeutic Strategies for the Treatment of Cancer. Cancers 2024, 16, 461. [Google Scholar] [CrossRef]
- Vandevoorde, C.; Franck, C.; Bacher, K.; Breysem, L.; Smet, M.H.; Ernst, C.; De Backer, A.; Van De Moortele, K.; Smeets, P.; Thierens, H. γ-H2AX foci as in vivo effect biomarker in children emphasize the importance to minimize x-ray doses in paediatric CT imaging. Eur. Radiol. 2015, 25, 800–811. [Google Scholar] [CrossRef]
- Tang, D.; Kang, R.; Livesey, K.M.; Cheh, C.-W.; Farkas, A.; Loughran, P.; Hoppe, G.; Bianchi, M.E.; Tracey, K.J.; Zeh, H.J., III. Endogenous HMGB1 regulates autophagy. J. Cell Biol. 2010, 190, 881–892. [Google Scholar] [CrossRef]
- Apostolov, E.O.; Soultanova, I.; Savenka, A.; Bagandov, O.O.; Yin, X.; Stewart, A.G.; Walker, R.B.; Basnakian, A.G. Deoxyribonuclease I is essential for DNA fragmentation induced by gamma radiation in mice. Radiat. Res. 2009, 172, 481–492. [Google Scholar] [CrossRef]
- Turner, H.C.; Shuryak, I.; Taveras, M.; Bertucci, A.; Perrier, J.R.; Chen, C.; Elliston, C.D.; Johnson, G.W.; Smilenov, L.B.; Amundson, S.A.; et al. Effect of dose rate on residual γ-H2AX levels and frequency of micronuclei in X-irradiated mouse lymphocytes. Radiat. Res. 2015, 183, 315–324. [Google Scholar] [CrossRef]
- Medvedeva, N.G.; Panyutin, I.V.; Panyutin, I.G.; Neumann, R.D. Phosphorylation of Histone H2AX in Radiation-Induced Micronuclei. Radiat. Res. 2007, 168, 493–498. [Google Scholar] [CrossRef]
- Dauer, L.T.; Brooks, A.L.; Hoel, D.G.; Morgan, W.F.; Stram, D.; Tran, P. Review and evaluation of updated research on the health effects associated with low-dose ionising radiation. Radiat. Prot. Dosim. 2010, 140, 103–136. [Google Scholar] [CrossRef] [PubMed]
- Chancellor, J.C.; Blue, R.S.; Cengel, K.A.; Auñón-Chancellor, S.M.; Rubins, K.H.; Katzgraber, H.G.; Kennedy, A.R. Limitations in predicting the space radiation health risk for exploration astronauts. npj Microgravity 2018, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Souza, E.d.; Soares, J.P.d.M. Occupational and technical correlations of interventional radiology. J. Vasc. Bras. 2008, 7, 341–350. [Google Scholar] [CrossRef]
- Morgan, W.F.; Sowa, M.B. Non-targeted effects of ionizing radiation: Implications for risk assessment and the radiation dose response profile. Health Phys. 2009, 97, 426–432. [Google Scholar] [CrossRef]
- Hu, L.; Yin, X.; Zhang, Y.; Pang, A.; Xie, X.; Yang, S.; Zhu, C.; Li, Y.; Zhang, B.; Huang, Y.; et al. Radiation-induced bystander effects impair transplanted human hematopoietic stem cells via oxidative DNA damage. Blood 2021, 137, 3339–3350. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Formenti, S. Radiation as an immunological adjuvant: Current evidence on dose and fractionation. Front. Oncol. 2012, 2, 153. [Google Scholar] [CrossRef]
- Yang, K.-L.; Wang, Y.-S.; Chang, C.-C.; Huang, S.-C.; Huang, Y.-C.; Chi, M.-S.; Chi, K.-H. Reciprocal complementation of the tumoricidal effects of radiation and natural killer cells. PLoS ONE 2013, 8, e61797. [Google Scholar] [CrossRef]
- Yang, N.; Lode, K.; Berzaghi, R.; Islam, A.; Martinez-Zubiaurre, I.; Hellevik, T. Irradiated tumor fibroblasts avoid immune recognition and retain immunosuppressive functions over natural killer cells. Front. Immunol. 2021, 11, 602530. [Google Scholar] [CrossRef]
- Burnette, B.C.; Liang, H.; Lee, Y.; Chlewicki, L.; Khodarev, N.N.; Weichselbaum, R.R.; Fu, Y.-X.; Auh, S.L. The efficacy of radiotherapy relies upon induction of type I interferon–dependent innate and adaptive immunity. Cancer Res. 2011, 71, 2488–2496. [Google Scholar] [CrossRef]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. Lancet Oncol. 2009, 10, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Schaue, D.; McBride, W.H. Opportunities and challenges of radiotherapy for treating cancer. Nat. Rev. Clin. Oncol. 2015, 12, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Koturbash, I.; Loree, J.; Kutanzi, K.; Koganow, C.; Pogribny, I.; Kovalchuk, O. In vivo bystander effect: Cranial X-irradiation leads to elevated DNA damage, altered cellular proliferation and apoptosis, and increased p53 levels in shielded spleen. Int. J. Radiat. Oncol. Biol. Phys. 2008, 70, 554–562. [Google Scholar] [CrossRef]
- Pogribny, I.; Raiche, J.; Slovack, M.; Kovalchuk, O. Dose-dependence, sex- and tissue-specificity, and persistence of radiation-induced genomic DNA methylation changes. Biochem. Biophys. Res. Commun. 2004, 320, 1253–1261. [Google Scholar] [CrossRef]
- Cheki, M.; Yahyapour, R.; Farhood, B.; Rezaeyan, A.; Shabeeb, D.; Amini, P.; Rezapoor, S.; Najafi, M. COX-2 in radiotherapy: A potential target for radioprotection and radiosensitization. Curr. Mol. Pharmacol. 2018, 11, 173–183. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects and stress-induced mutagenesis. In Stress-Induced Mutagenesis; Springer: New York, NY, USA, 2013; pp. 199–222. [Google Scholar]
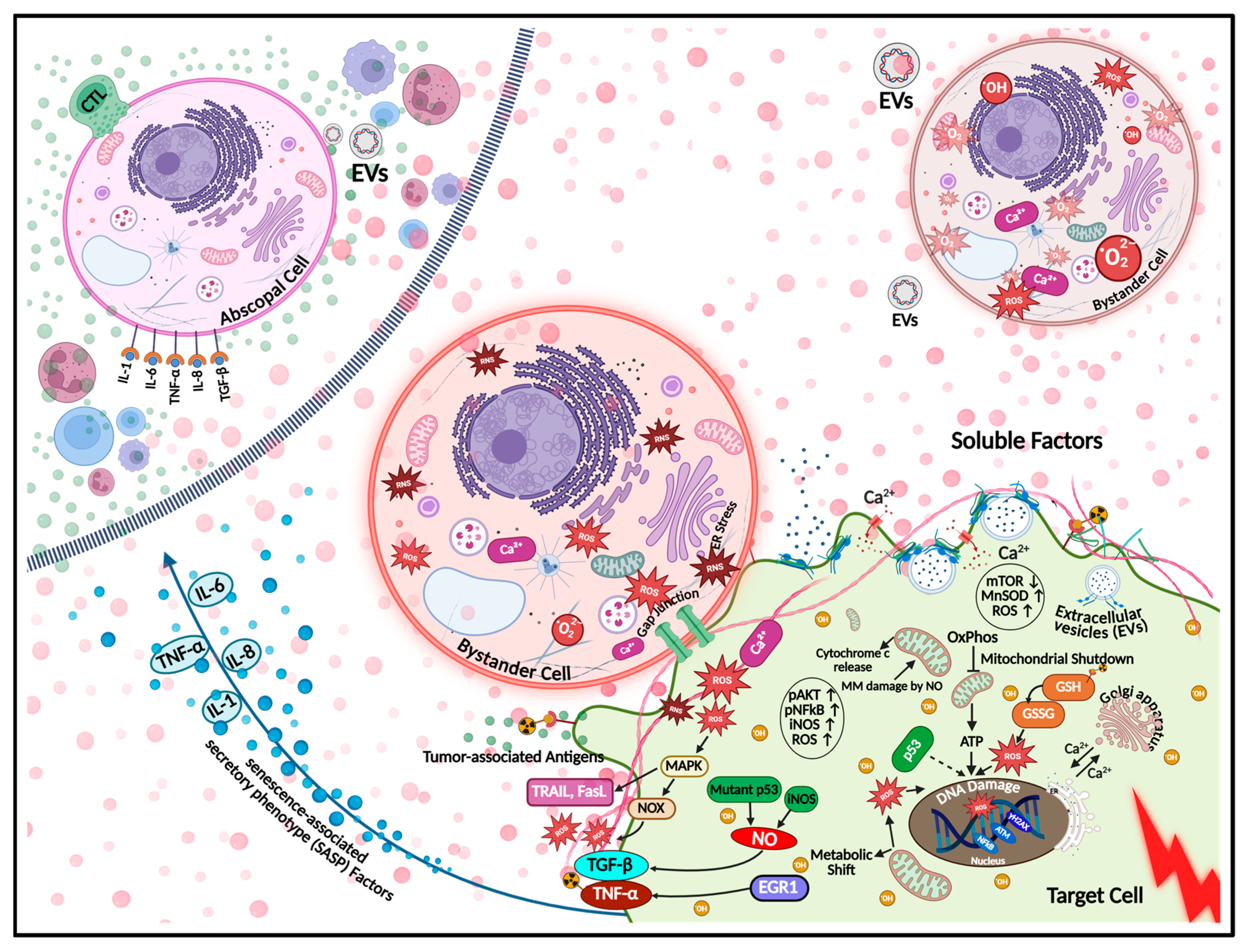
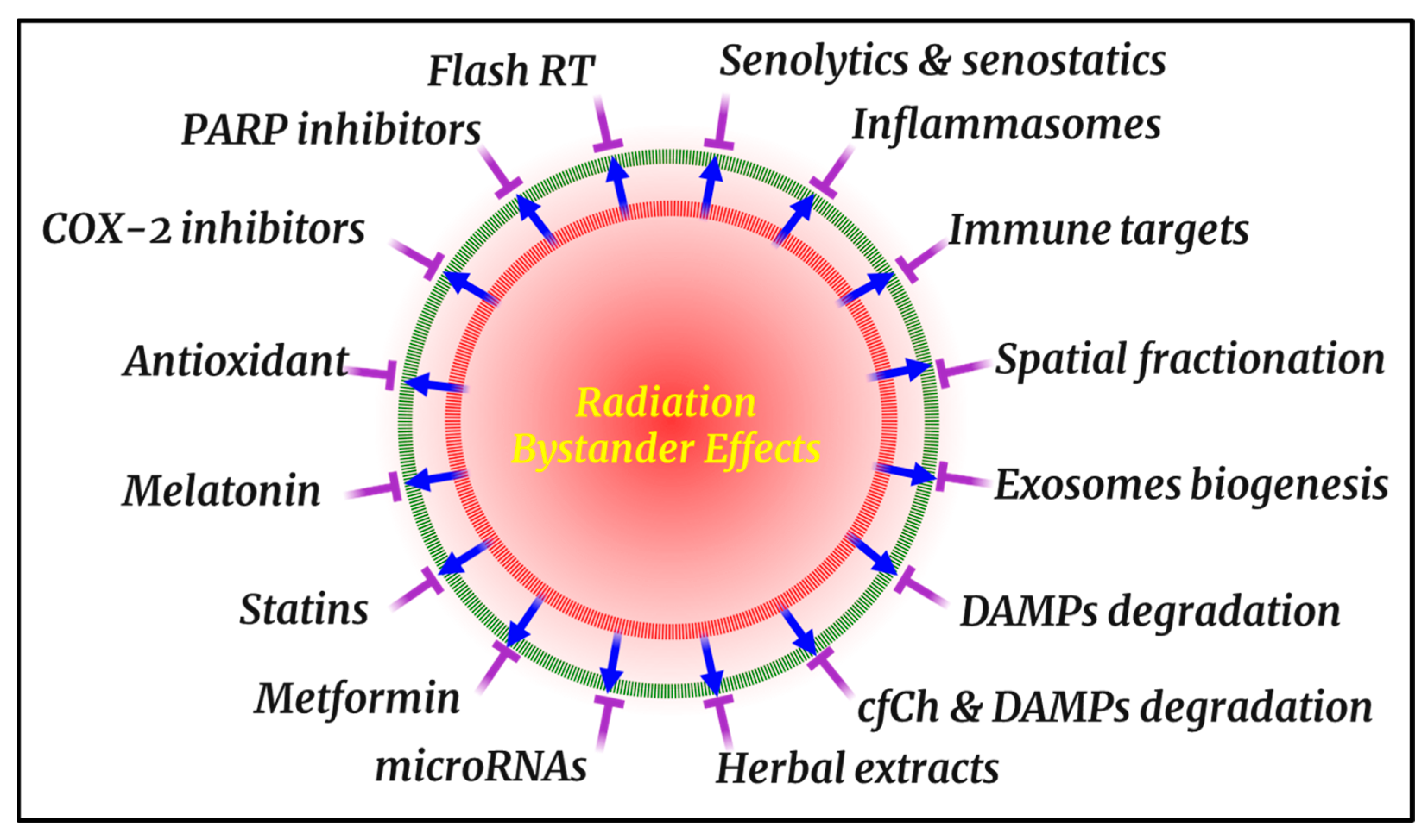
| Category | Mechanism of Action | Mediators | Ref. |
|---|---|---|---|
| Gap junction-mediated communication | Intercellular communication through gap junctions enables the transfer of signals between irradiated and bystander cells. | Connexins (Cx43), calcium ions, ROS, cytokines (TNF-α, IL-1α) | [88,122,128,135,138,164,195] |
| Soluble factor/cytokine signaling | Irradiated cells release cytokines and soluble factors that activate signaling pathways in bystander cells | Pro-inflammatory cytokines (TNF-α, IL-6, IL-8, IL-1, IL-33, TGF-β), Anti-inflammatory (IL-10), Growth factors (CSF2, VEGF), Signaling pathways (NFκB, STAT3, JAK-STAT, MAPK) | [85,100,110,128,134,135,136,137,138] |
| Oxidative stress and ROS/RNS signaling | Radiation-induced ROS and RNS propagate damage to bystander cells through direct diffusion or signaling cascades | ROS generators (NADPH oxidases NOX1-5, COX-2, mitochondrial dysfunction), RNS (NO, peroxynitrite via iNOS), Antioxidant suppression (SOD2, catalase, GST), Signaling (NFκB, MAPK, NLRP3 inflammasome) | [85,88,90,109,113,115,128,138,164,196,197,198,199] |
| NFκB-mediated signaling networks | NFκB activation serves as a central hub for multiple bystander signaling pathways, regulating survival, inflammation, and stress responses | NFκB components (RelA/p65, p50, IκBα), Downstream targets (Survivin, cIAP1/2, Bcl-2, MnSOD, hTERT), Feedback loops (TNF-α, MMP9, telomerase activation) | [89,91,92,95,100,102,104,114,122,138,198,199,200,201,202] |
| Extracellular vesicle/exosome-mediated transfer | Irradiated cells release EVs containing miRNAs, proteins, and other cargo that transfer signals to bystander cells | Exosomal miRNAs (miR-744-3p, miR-152, miR-21), Signaling (TLR4, DNA damage response), Cargo (ROS, nitric oxide, calcium signaling molecules) | [68,122,135,140,196,203] |
| MicroRNA regulation | Radiation alters miRNA expression in irradiated cells, with specific miRNAs propagating bystander effects | miR-21 (suppresses SOD2, promotes ROS and DNA damage), miR-663 (regulates TGF-β1 in feedback loops), let-7 family (stress response regulation), miR-30c, miR-144, miR-34a (epigenetic regulation) | [12,74,92,121,122,124,130,138,149,197,199,203,204] |
| TGF-β signaling pathways | TGF-β serves as a key mediator of BEs through both Smad-dependent and independent pathways | TGF-β/TGF-βRII signaling, Downstream (COX-2, iNOS, NADPH oxidases), Interactions (miR-21, miR-663, MAPK JNK/ERK), Tissue remodeling (Wnt/β-catenin, Notch signaling) | [95,109,120,124,125,128,130,134,148,149] |
| Mitochondrial dysfunction and signaling | Radiation disrupts mitochondrial function, leading to persistent ROS production and release of damage signals | Mitochondrial ROS, mtDNA release, Electron transport chain (ETC) dysfunction, Biophoton emission, exosome signaling, Crosstalk with nuclear DNA damage response | [89,90,91,92,109,113,114,115] |
| DNA damage response and repair signaling | DNA damage sensors and repair pathways in irradiated cells generate signals that propagate to bystander cells | DNA damage sensors (ATM, ATR, DNA-PK), Repair proteins (BRCA1, FANCD2, Chk1), Damage markers (γH2AX, 53BP1 foci), Checkpoint proteins (p53, p21) | [68,74,90,120,121,122,123,124,125] |
| Death receptor/apoptosis signaling | Irradiated cells release death ligands that induce apoptosis in bystander cells through receptor-mediated pathways | Death ligands (TRAIL, FasL, TNF-α), Receptors (TRAIL-R2 DR5, Fas, TNFR1), Apoptosis machinery (Caspases, PAR-4, Bcl-2 family), Survival signals (AKT, NFκB) | [114,137,138,151,163,205,206] |
| Autophagy modulation | Radiation affects autophagy pathways, influencing cell survival and bystander signaling | Autophagy markers (LC3-II, p62), Regulation (CSF2 signaling, rapamycin), Dual roles (Protective vs. pro-invasive effects) | [110,200] |
| Epigenetic modifications | Radiation induces epigenetic changes that can propagate BEs and genomic instability | DNA methylation (DNMTs, hypomethylation), Histone modifications (HDACs), miRNA regulation (miR-21, miR-30c, miR-144, miR-34a), Chromatin remodeling | [92,121,128,129,130] |
| Immune system modulation | Radiation alters immune cell function and signaling, affecting bystander responses through immune-mediated pathways | Immune cells (TAMs, T-cells, microglia), Immune signaling (TLR2/4/9, NLRs, DAMPs), Cytokines (IL-2, IL-10, TGF-β, TNF-α, IFN-γ), Adaptive responses (M1/M2 polarization) | [89,125,134,138,139,140,141] |
| Metabolic reprogramming | Radiation induces metabolic changes that contribute to BEs through altered energy metabolism and metabolite signaling | Glycolysis (GAPDH, LDHA, lactate production), Energy sensing (AMPK activation), Metabolite transport (MCT1), Microenvironment acidification | [95] |
| Stem cell and tissue microenvironment effects | Radiation affects stem cell niches and tissue microenvironments, influencing bystander cell behavior | Stem cell expansion (TGF-β, Notch pathway), Microenvironment remodeling (Stromal cells, ECM), Neural stem cells (Neurogenesis modulation), Hematopoietic effects (HSC dysfunction) | [128,134,141,144,151,163,207] |
| Systemic/abscopal effects | Localized radiation induces systemic signals that affect distant, non-irradiated tissues | Circulating factors (Cytokines, clastogenic factors), Systemic stress responses (Antioxidant pathways, Nrf2), Organ-specific effects (Heart, brain, bone marrow), Adaptive responses (Radioprotection, preconditioning) | [91,103,125,136,139,157] |
| Dose–response and temporal dynamics | BEs show specific dose–response relationships and temporal patterns | Low-dose specificity (Adaptive responses), Temporal waves (Biphasic responses, delayed effects), Feedback mechanisms (Adaptive inhibition), Spatial constraints (Distance-dependent effects) | [90,104,125,138,139,157,164] |
| Tissue-specific and cell-type-specific effects | Different cell types and tissues exhibit distinct bystander response patterns and mechanisms | Neural tissues (Glioblastoma, NSCs, astrocytes), Lung (Fibroblasts, epithelial cells), Breast (Mammary epithelial cells), Hematopoietic (HSCs, lymphoblasts) | [74,120,122,123,141,144,148,150,151,152,164] |
| Senescence-associated secretory phenotype (SASP) | Radiation-induced senescent cells secrete pro-inflammatory and growth-promoting factors, inducing bystander senescence through metabolic alterations. | SASP factors, AMPK/NFκB pathway activation, Glycolysis inhibition, Senolytic resistance pathways, Metabolic alterations | [142,143] |
| Ferroptosis-mediated BEs | Iron-dependent lipid peroxidation-driven cell death pathway with CD8+ T cell regulation and lipid peroxide transfer to bystander cells | Iron metabolism, Lipid peroxidation, CD8+ T cells, CCL5-SCD4 axis, B2M-TFRC axis, Lipid peroxide transfer | [116] |
| Enhanced metabolic reprogramming and bioenergetic alterations | Specific metabolite signaling and metabolic pathway disruption propagating to bystander cells through bioenergetic alterations | Lactate, Succinate, α-ketoglutarate, Itaconate, Glycolysis modulation, AMPK activation, Metabolic pathway disruption | [95,205,207] |
| Chromatin remodeling and histone modifications | Epigenetic modifications and chromatin remodeling complexes affecting gene expression and propagating bystander signals | Chromatin remodeling complexes, Histone modifications, Epigenetic modifications, Gene expression alterations | [44,208] |
| Intercellular calcium signaling networks | Gap-junction-mediated calcium ion cascades and small molecule transfer propagating bystander signals | Gap junctions, Calcium ion cascades, Nitric oxide, small molecule transfer, Calcium signaling pathways | [66,209] |
| Radiation-induced genomic instability | NTEs causing genomic instability in cells not directly irradiated through DNA damage and repair pathway disruption | Genomic instability, DNA damage, DNA repair pathway disruption, NTEs | [126,127,209] |
| Purinergic signaling networks | ATP and adenosine release from radiation-damaged cells activate purinergic receptors in bystander cells | ATP, Adenosine, P2X/P2Y receptors, A2A/A2B receptors, Calcium signaling cascades, Inflammatory responses | [210,211] |
| Complement system activation | Radiation-induced complement cascade activation, with complement proteins serving as bystander signals | Complement proteins (C3a, C5a), Complement cascade, Complement receptors, Inflammatory response propagation | [212] |
| Radiation-induced trained immunity | Epigenetic reprogramming in immune cells creates “trained immunity” states affecting bystander immune responses | Macrophages, Monocytes, Trained immunity, Epigenetic reprogramming, Metabolic reprogramming, Chromatin modifications | [213] |
| Circadian rhythm disruption | Radiation-induced circadian clock mechanism disruption with temporal signaling disruptions propagating to bystander cells | Circadian clock mechanisms, Temporal signaling, Circadian factors, Cell cycle regulation, DNA repair timing | [160] |
| Extracellular matrix (ECM) remodeling | Changes in ECM composition and matricellular proteins propagate mechanical and biochemical signals through integrin-mediated pathways | Thrombospondin, Osteopontin, Tenascin-C, Integrin-mediated pathways, Matricellular proteins | [153] |
| Cellular reprogramming factor release | Radiation-induced activation of pluripotency factors and developmental pathway molecules affecting bystander cell differentiation | Pluripotency factors, Developmental pathway molecules, Partial cellular reprogramming, Cell differentiation | [214] |
| Autophagy-lysosome pathway crosstalk | Dysregulated autophagy-lysosome pathways with secretion of lysosomal contents and autophagy-related proteins | Lysosomal contents, Autophagy-related proteins, Autophagy-lysosome pathway dysregulation | [110,215,216] |
| Non-coding RNA-based communication | In addition to miRNAs and extracellular vesicles, other non-coding RNAs (ncRNAs) such as long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs) can modulate BEs by regulating DNA damage response, apoptosis, and intercellular signaling. | lncRNA HOTAIR, circRNAs, exosomal lncRNAs. | [111,131,132] |
| Hormetic and adaptive responses post low-dose RT | Hyper-radiosensitivity and induced radioresistance (HRS/IRR) show that not only low but also moderate doses can induce distinct adaptive BEs, both protective and detrimental, depending on the timing and microenvironment. | DNA repair pathways (Rad51, Ku70/80) and cell cycle checkpoint proteins. | [84,217] |
| Sex-specific and age-dependent BEs | BEs can vary by sex and age due to differences in hormonal environment, immune modulation, and antioxidant defenses. | Estrogen/testosterone, age-related senescence pathways, and altered cytokine profiles. | [214,218] |
| Microbiome and BEs interactions | The microbiome can influence and be influenced by BEs Via immune modulation, metabolite production, and gut–brain axis signaling after radiation exposure. | Microbial metabolites (short-chain fatty acids), microbial-derived ROS/NO, and TLR signaling. | [158,159] |
| Proteostasis and unfolded protein response (UPR) | Radiation-induced ER stress can trigger UPR in both irradiated and bystander cells, promoting inflammation, apoptosis, and potentially chronic BEs phenotypes. | PERK, ATF6, IRE1, chaperone proteins (GRP78/BiP). | [161,162] |
| Receptor tyrosine kinase signaling | Ligand-based activation of mitogenic/inflammatory pathways in bystander cells | EGFR, PDGFR, VEGFR, FGFR | [64,85] |
| cGAS–STING signaling | Cytosolic DNA detection leads to IFN and immune gene expression | cGAS, STING, IRF3, TBK1 | [219,220] |
| Mitophagy dysregulation | Impaired mitochondrial turnover enhances ROS spread | PINK1, PARKIN, LC3B | [93,94] |
| Inflammasome activation | mtDNA or ROS triggers inflammasome-driven cytokine release | NLRP3, ASC, Caspase-1, IL-1β, IL-18 | [221,222] |
| Mitochondria-associated endoplasmic reticulum membrane (MAM) disruption | Altered ER-mitochondria junctions affect Ca2+, ROS, apoptosis | IP3R, VDAC1, GRP75, MFN2 | [223,224] |
| HIF-mediated hypoxic responses | Radiation-induced hypoxia drives pro-survival and angiogenic BEs | HIF-1α, VEGF, REDD1 | [201,225,226] |
| RNA editing and dsRNA signaling | A-to-I RNA editing alters immune and stress signaling in bystander cells | ADAR1, PKR, RIG-I, IFN genes | [111,133] |
| Lipid mediator signaling | Radiation induces lipid remodeling that influences inflammation or apoptosis signaling. | Ceramide, S1P, eicosanoids (PGE2, LTB4) | [85,119] |
| Ion channel remodeling | Radiation alters voltage-gated ion channel activity | Kv (Voltage-gated potassium channels), Nav, Cl− channels, Ca2+ channels | [166,167] |
| Endothelial bystander signaling | Angiocrine signals from irradiated endothelium affect nearby cells | Angiopoietin-2, E-selectin, ICAM-1 | [145,146,147] |
| Protease/ECM enzyme activation | ECM degradation propagates pro-inflammatory and proinvasive signals | MMP2, MMP9, TIMP, matrikines | [154,155] |
| Heat shock protein signaling | Extracellular HSPs activate DAMP and immune sensor pathways | HSP70, HSP90, TLR4 | [227,228] |
| YAP/TAZ mechanotransduction | Mechanical stress alters transcription and cellular adaptation | YAP, TAZ, RhoA, actin, integrins | [156] |
| Nitrosative stress | RNS signaling damage in bystander cells | ONOO−, iNOS, nitrotyrosine | [206,229] |
| Adenylate kinase signaling | Extracellular ATP/AMP affects purinergic and metabolic sensors | Adenylate kinase, CD73, AMPK | [59,95,96] |
| mTOR pathway signaling | Translational and nutrient signaling in bystander stress responses | mTORC1, S6K, eIF4E, TSC2 | [97,118] |
| Ubiquitin-proteasome dysfunction | Altered protein degradation affects bystander signaling | RNF ligases, proteasomal subunits | [163,165] |
| Phagocytosis of irradiated debris | Engulfment of damaged cells triggers inflammatory cascades | DAMPs, phosphatidylserine, MerTK | [230,231,232] |
| Lipid rafts and membrane reorganization | Disruption affects receptor clustering, vesicle formation, and signal dissemination. | Caveolin-1, GM1, flotillin | [168,169,170,233] |
| Countermeasure | * Evidence Level | Efficacy (Quantitative) | Safety Profile/Cautionary Note |
|---|---|---|---|
| Predictive modeling and SNP-guided planning (Precision/Personalized Countermeasures) | Emerging | Predictive modeling of BEs susceptibility | Requires validation; ethical considerations |
| mitochondrial antioxidants with gap junction blockers (Dual Inhibition Strategies) | Emerging | Synergistic suppression of BEs | Complexity in dosing, toxicity |
| NS-398, celecoxib (COX-2 Inhibitors) | High | ~40% reduction in micronuclei formation | GI and cardiovascular toxicity with prolonged use |
| Avasopasem Manganese (GC4419) [superoxide dismutase (SOD) mimetic] | High | ↓ oral mucositis severity | Favorable safety; FDA fast-tracked |
| Proton Therapy (Modality selection for reduced integral dose) | High | ↓ secondary malignancies, normal tissue toxicity | Cost, access limitations |
| Shielding/Motion Gating (Physical exclusion of non-targeted tissues) | High | ↓ oxidative stress, DNA damage | Standard practice; enhanced by adaptive RT |
| DNase I, CNPs, R-Cu (cfCh Degradation) | Low | ↓ γ-H2AX, IL-6, chromosomal aberrations | DNase I: off-target risk; CNPs: immunogenicity; R-Cu: untested in humans |
| GW4869, miR-21 antagomirs (Exosome/miRNA Targeting) | Low to Moderate | ↓ DNA damage, apoptosis in bystanders | GW4869: experimental; antagomirs: delivery challenges |
| Spatial Fractionation/Lattice RT | Low to Moderate | ↓ systemic inflammation, γ-H2AX | Not widely implemented |
| Exosome Biogenesis/Uptake Inhibitors | Low to Moderate | ↓ γ-H2AX, senescence in bystanders | Experimental; specificity concerns |
| DNase I, anti-HMGB1 (cfCh/DAMP Clearance) | Low to Moderate | ↓ γ-H2AX, inflammation | DNase I: systemic effects; antibodies: immunogenicity |
| Melatonin (Pre-treatment Radioprotectants]) | Moderate | ↓ IL-6 and TNF-α in shielded organs | Favorable safety; crosses blood–brain barrier |
| Statins, Metformin, Herbal Extracts (Traditional radioprotectors) | Moderate | ↓ ROS, DNA damage, cytokine release | Statins: myopathy risk; Metformin: GI upset; Herbal: variable |
| MitoQ, metformin (Mitochondrial/Metabolic Modulators) | Moderate | ↓ γ-H2AX, apoptosis, oxidative stress | Mitochondrial inhibitors: cytotoxicity; timing critical |
| MCC950, Anakinra (Inflammasome/Immune Modulators) | Moderate | ↓ IL-1β, pyroptosis, fibrosis | Risk of immunosuppression; timing essential |
| D+Q, navitoclax, rapamycin (Senolytics/Senostatics) | Moderate | ↓ fibrosis, senescence markers | Navitoclax: thrombocytopenia; rapamycin: metabolic effects |
| Anakinra (IL-1 Receptor Antagonists) | Moderate | ↓ pneumonitis, fibrosis | Potential immunosuppression |
| PARP, ATR inhibitors (DDR Inhibitors) | Moderate | ↑ tumor response; possible BEs amplification | Tumor-specific targeting needed |
| FLASH RT | Moderate | ↓ fibrosis, neuroinflammation, BEs markers | Delivery challenges; under investigation |
| Carbon Ions (Heavy Ion Therapy) | Moderate | ↑ tumor control; ↓ normal tissue dose | LET-specific toxicity; limited availability |
| Hyperfractionation and dose Rate Modulation (Temporal control of radiation delivery) | Moderate | ↓ BEs markers with hyperfractionation | Requires precise planning |
| NAC, Vitamin E (Antioxidants) | Moderate to High | Reduced γ-H2AX foci and lipid peroxidation | Generally safe; tumor protection risk debated |
| Alpha-emitter (225Ac-NM600, 223Ra dichloride), Beta-emitter (177Lu-PSMA-617, 131I), combination therapy (RPT + anti-PD-1 antibodies) (Radiopharmaceuticals therapy) | Moderate | α-emitter RPT ↓ Tregs and ↑ activated CD8+ T cells; clinical trials report immune activation and survival benefits | Complex systemic biodistribution; antioxidants may interfere with immune priming; LET-tailored testing required |
| 2-Deoxyglucose (Glycolysis Inhibitor) | Low | Disrupts metabolic coupling | Cytotoxicity concerns |
| Apyrase, Suramin (Purinergic Signaling Inhibitors) | Experimental | ↓ ATP-mediated BEs signaling | Limited data |
| Disulfiram (Gasdermin D inhibitor) | Emerging | Blocks pyroptosis and cytokine release | Known drug; repurposing potential |
| Amifostine (Thiol-based free radical scavenger) | Moderate | ↓ oxidative stress; radioprotection in preclinical models | Limited clinical uptake due to toxicity and inconsistent efficacy |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramamurthy, M.O.; Subramanian, P.; Aravindan, S.; Periyasamy, L.; Aravindan, N. Radiation Without Borders: Unraveling Bystander and Non-Targeted Effects in Oncology. Cells 2025, 14, 1761. https://doi.org/10.3390/cells14221761
Ramamurthy MO, Subramanian P, Aravindan S, Periyasamy L, Aravindan N. Radiation Without Borders: Unraveling Bystander and Non-Targeted Effects in Oncology. Cells. 2025; 14(22):1761. https://doi.org/10.3390/cells14221761
Chicago/Turabian StyleRamamurthy, Madhi Oli, Poorvi Subramanian, Sivaroopan Aravindan, Loganayaki Periyasamy, and Natarajan Aravindan. 2025. "Radiation Without Borders: Unraveling Bystander and Non-Targeted Effects in Oncology" Cells 14, no. 22: 1761. https://doi.org/10.3390/cells14221761
APA StyleRamamurthy, M. O., Subramanian, P., Aravindan, S., Periyasamy, L., & Aravindan, N. (2025). Radiation Without Borders: Unraveling Bystander and Non-Targeted Effects in Oncology. Cells, 14(22), 1761. https://doi.org/10.3390/cells14221761








