Natural Killer Cell Therapy Combined with Probiotic Bacteria Supplementation Restores Bone Integrity in Cancer by Promoting IFN-γ Production
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Reagents
2.2. Flow Analysis
2.3. Probiotic Bacteria AJ2 Formulation and Sonication
2.4. Purifying Human NK Cells and Monocytes
2.5. Generation of Osteoclasts and Supercharged NK Cells
2.6. Tumor Implantation, sNK Cells Infusions, and AJ2 Feeding in hu-BLT Mice
2.7. Processing Tissues and Culturing Cells from hu-BLT Mice Tissue Samples
2.8. Isolating NK Cells from the Spleen of hu-BLT Mice
2.9. Enzyme-Linked Immunosorbent Assay (ELISA)
2.10. Bone Analysis
2.11. Histology and Quantitative Histomorphometry
2.12. Statistical Analysis
3. Results
3.1. Successful Reconstitution of Human CD45+ Immune Cells in Tissues of hu-BLT Mice
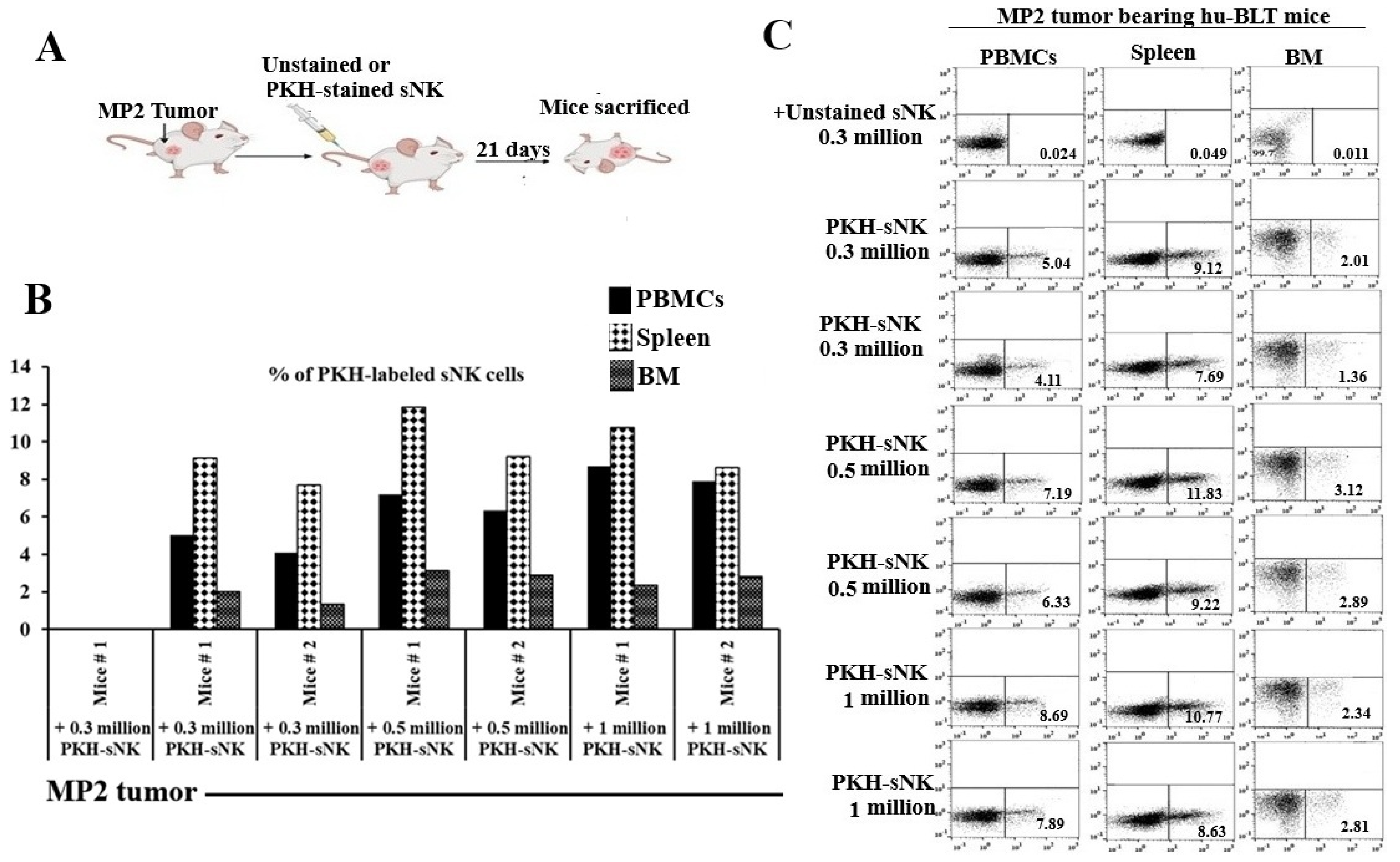
3.2. Adoptively Transferred sNK Cells Migrate to Tissue Compartments of Pancreatic Tumor-Bearing hu-BLT Mice
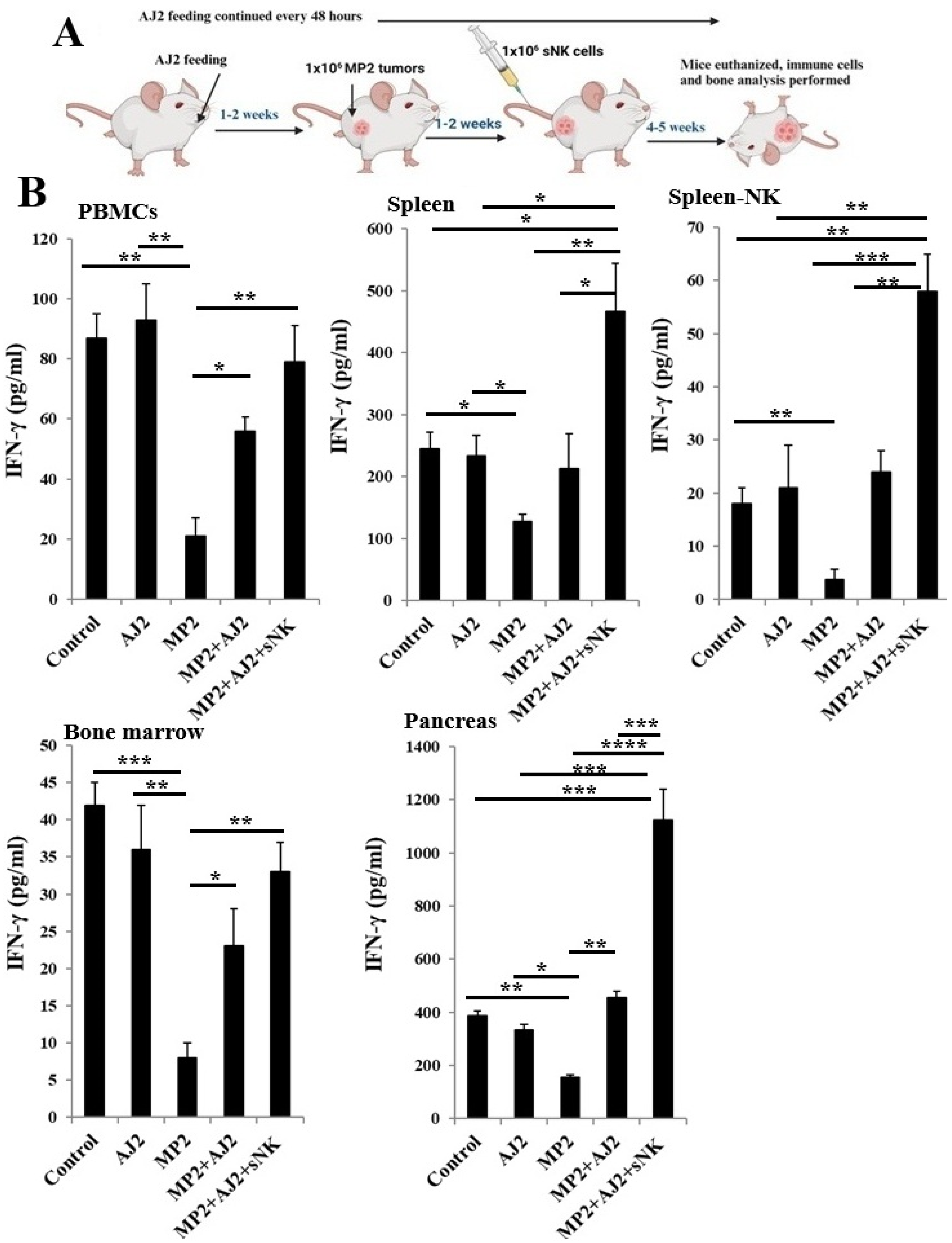
3.3. A Significant Rise in IFN-γ Secretion Was Observed in Mice Tissues When the Mice Were Infused with sNK Cells and Fed Probiotic Bacteria
3.4. Probiotic Bacteria-Fed hu-BLT Mice Showed Increased Bone Formation Compared to the Control Group
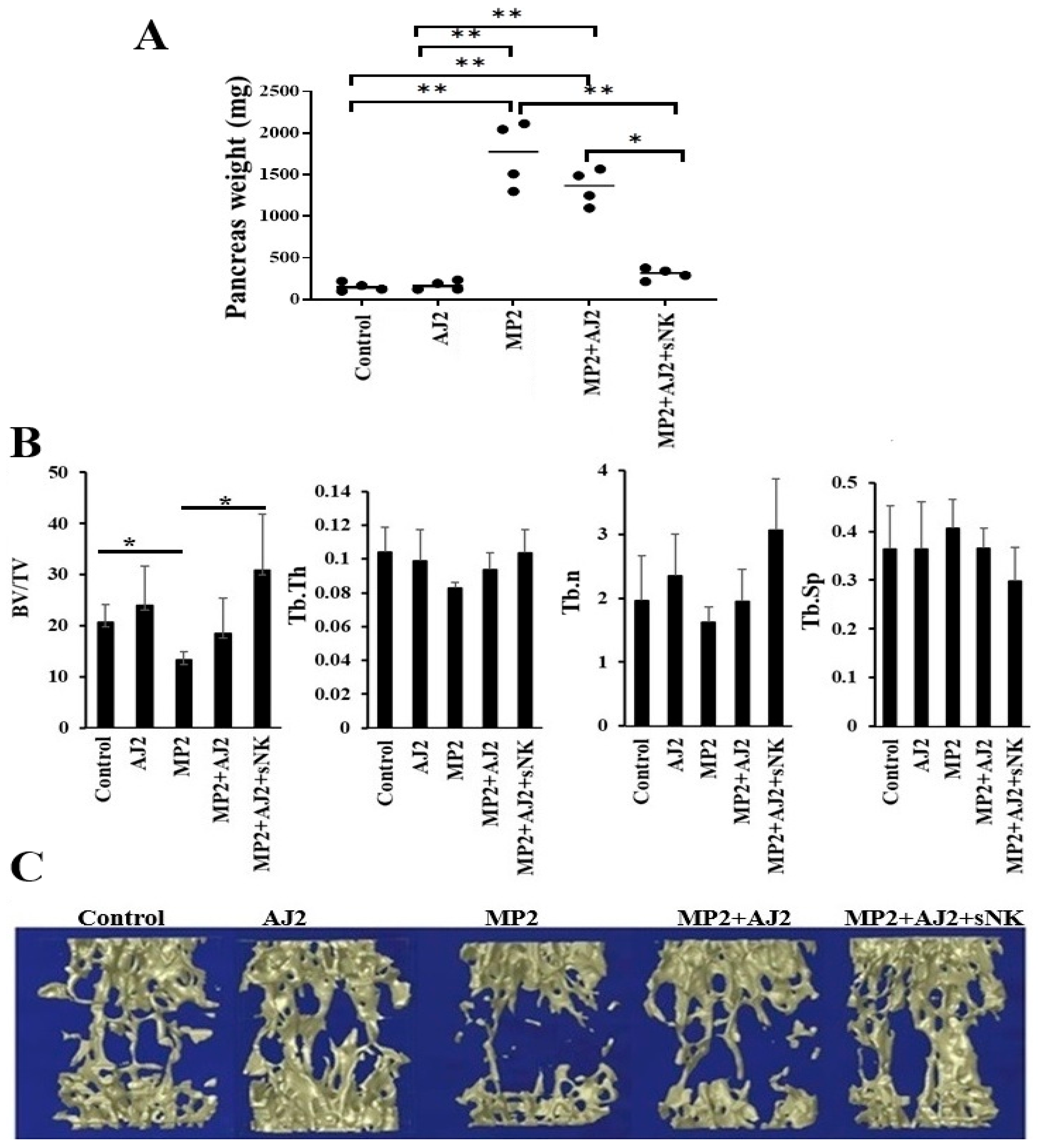
3.5. Using Probiotics as an Adjuvant Alongside sNK Cell Therapy Effectively Suppressed Pancreatic Tumor Growth in hu-BLT Mice
3.6. Bone Formation Increased in Pancreatic Tumor-Bearing hu-BLT Mice Infused with sNK Cells and Given Probiotic Bacteria
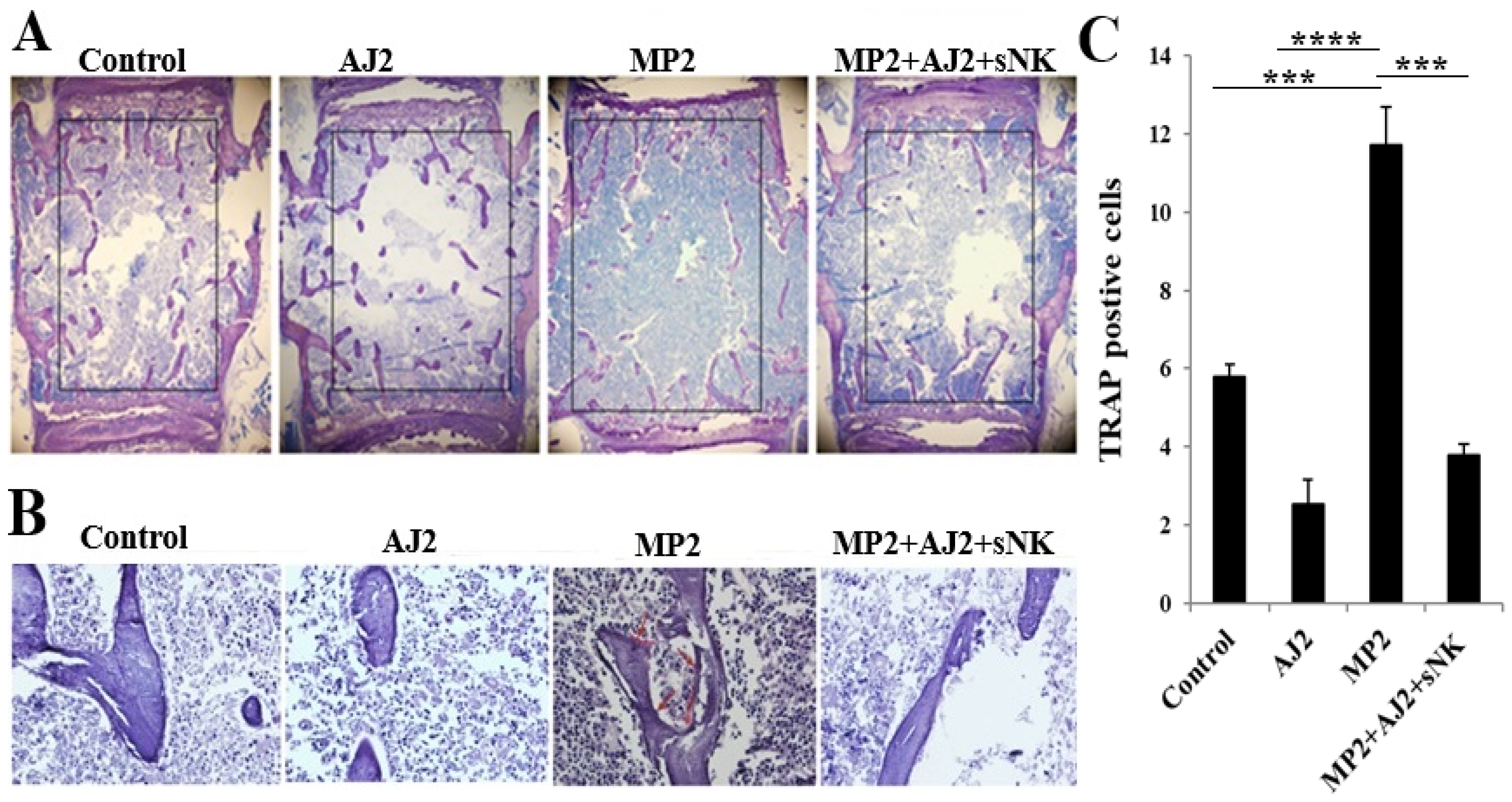
3.7. Increased Trabecular Bone Formation Was Observed in Pancreatic Tumor-Bearing Mice When They Were Fed Probiotic Bacteria, Either Alone or Combined with sNK Cell Infusions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NK cells | Natural killer cells |
| sNK cells | Supercharged NK cells |
| CSCs | Cancer-stem-like-cells |
| Hu-BLT | Humanized-bone marrow/liver/thymus |
| IFN-γ | Interferon-gamma |
| IL-2 | Interleukin 2 |
| OCs | Osteoclasts |
| PBMCs | Peripheral blood-derived mononuclear cells |
| MP2 | MiaPaCa-2 |
| ELISAs | Enzyme-Linked Immunosorbent Assays |
References
- Kopitar-Jerala, N. The Role of Interferons in Inflammation and Inflammasome Activation. Front. Immunol. 2017, 8, 873. [Google Scholar] [CrossRef]
- Zaidi, M.R.; Merlino, G. The Two Faces of Interferon-γ in Cancer. Clin. Cancer Res. 2011, 17, 6118–6124. [Google Scholar] [CrossRef]
- Mendoza, J.L.; Escalante, N.K.; Jude, K.M.; Sotolongo Bellon, J.; Su, L.; Horton, T.M.; Tsutsumi, N.; Berardinelli, S.J.; Haltiwanger, R.S.; Piehler, J.; et al. Structure of the IFNγ receptor complex guides design of biased agonists. Nature 2019, 567, 56–60. [Google Scholar] [CrossRef]
- Vuletić, A.; Jovanić, I.; Jurišić, V.; Milovanović, Z.; Nikolić, S.; Spurnić, I.; Konjević, G. Decreased Interferon γ Production in CD3+ and CD3- CD56+ Lymphocyte Subsets in Metastatic Regional Lymph Nodes of Melanoma Patients. Pathol. Oncol. Res. 2015, 21, 1109–1114. [Google Scholar] [CrossRef]
- Han, J.; Wu, M.; Liu, Z. Dysregulation in IFN-γ signaling and response: The barricade to tumor immunotherapy. Front. Immunol. 2023, 14, 1190333. [Google Scholar] [CrossRef]
- Lee, H.J.; Koh, J.; Kim, J. INF-γ production of NK cells is suppressed in cancer patients. (45.10). J. Immunol. 2012, 188 (Suppl. S1), 45.10. [Google Scholar] [CrossRef]
- Alspach, E.; Lussier, D.M.; Schreiber, R.D. Interferon γ and Its Important Roles in Promoting and Inhibiting Spontaneous and Therapeutic Cancer Immunity. Cold Spring Harb. Perspect. Biol. 2019, 11, 655–669. [Google Scholar] [CrossRef]
- Sica, A.; Dorman, L.; Viggiano, V.; Cippitelli, M.; Ghosh, P.; Rice, N.; Young, H.A. Interaction of NF-kappaB and NFAT with the interferon-gamma promoter. J. Biol. Chem. 1997, 272, 30412–30420. [Google Scholar] [CrossRef]
- Szabo, S.J.; Kim, S.T.; Costa, G.L.; Zhang, X.; Fathman, C.G.; Glimcher, L.H. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 2000, 100, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Schoenborn, J.R.; Wilson, C.B. Regulation of interferon-gamma during innate and adaptive immune responses. Adv. Immunol. 2007, 96, 41–101. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H.S.K.; Sato, K.; Takaoka, A.; Taniguchi, T. Interplay between interferon and other cytokine systems in bone metabolism. Immunol. Rev. 2005, 208, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Duque, G.H.D.; Huang, D.C.; Macoritto, M.; Rivas, D.; Yang, X.F.; Ste-Marie, L.G.; Kremer, R. Autocrine regulation of interferon gamma in mesenchymal stem cells plays a role in early osteoblastogenesis. Stem. Cells 2009, 27, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Akdis, M.; Burgler, S.; Crameri, R.; Eiwegger, T.; Fujita, H.; Gomez, E.; Klunker, S.; Meyer, N.; O’Mahony, L.; Palomares, O.; et al. Interleukins, from 1 to 37, and interferon-[gamma]: Receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 2011, 127, e1. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. Osteoimmunology and the effects of the immune system on bone. Nat. Rev. Rheumatol. 2009, 5, 667–676. [Google Scholar] [CrossRef]
- Ferrari-Lacraz, S.; Ferrari, S. Is IFN-c involved in bone loss or protection? Nothing is simple with cytokines. Bonekey Osteovision 2007, 4, 83–87. [Google Scholar] [CrossRef]
- Takayanagi, H.; Ogasawara, K.; Hida, S.; Chiba, T.; Murata, S.; Sato, K.; Takaoka, A.; Yokochi, T.; Oda, H.; Tanaka, K.; et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 2000, 408, 600–605. [Google Scholar] [CrossRef]
- Ayon Haro, E.R.; Ukai, T.; Yokoyama, M.; Kishimoto, T.; Yoshinaga, Y.; Hara, Y. Locally administered interferon-c accelerates lipopolysaccharide-induced osteoclastogenesis independent of immunohistological RANKL upregulation. J. Periodontal Res. 2011, 46, 361–373. [Google Scholar] [CrossRef]
- Gao, Y.G.F.; Ryan, M.R.; Terauchi, M.; Page, K.; Yang, X.; Weitzmann, M.N.; Pacifici, R. IFN-gamma stimulates osteoclast formation and bone loss in vivo via antigen-driven T cell activation. J. Clin. Investig. 2007, 117, 122–132. [Google Scholar] [CrossRef]
- Takayanagi, H. Mechanistic insight into osteoclast differentiation in osteoimmunology. Mol. Med. 2005, 83, 170–179. [Google Scholar] [CrossRef]
- Delves, P.J.; Roitt, I.M. The immune system. Second of two parts. N. Engl. J. Med. 2000, 343, 108–117. [Google Scholar] [CrossRef]
- Duque, G.H.D.; Huang, D.C.; Dion, N.; Macoritto, M.; Rivas, D.; Li, W.; Yang, X.F.; Li, J.; Lian, J.; Marino, F.T.; et al. Interferon g plays a role in bone formation in vivo and rescues osteoporosis in ovariectomized mice. J. Bone Miner. Res. 2011, 26, 1472–1483. [Google Scholar] [CrossRef]
- Moretta, A.; Marcenaro, E.; Parolini, S.; Ferlazzo, G.; Moretta, L. NK cells at the interface between innate and adaptive immunity. Cell Death Differ. 2008, 15, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Vivier, E.; Raulet, D.H.; Moretta, A.; Caligiuri, M.A.; Zitvogel, L.; Lanier, L.L.; Yokoyama, W.M.; Ugolini, S. Innate or adaptive immunity? The example of natural killer cells. Science 2011, 331, 44–49. [Google Scholar] [CrossRef]
- Jurišić, V. Investigation of NK cell function against two target hematological cell line using radioactive chromium assay. Appl. Radiat. Isot. 2024, 206, 111251. [Google Scholar] [CrossRef]
- Ruggeri, L.; Capanni, M.; Urbani, E.; Perruccio, K.; Shlomchik, W.D.; Tosti, A.; Posati, S.; Rogaia, D.; Frassoni, F.; Aversa, F.; et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science 2002, 295, 2097–2100. [Google Scholar] [CrossRef]
- Venstrom, J.M.; Pittari, G.; Gooley, T.A.; Chewning, J.H.; Spellman, S.; Haagenson, M.; Gallagher, M.M.; Malkki, M.; Petersdorf, E.; Dupont, B.; et al. HLA-C-dependent prevention of leukemia relapse by donor activating KIR2DS1. N. Engl. J. Med. 2012, 367, 805–816. [Google Scholar] [CrossRef]
- Iliopoulou, E.G.; Kountourakis, P.; Karamouzis, M.V.; Doufexis, D.; Ardavanis, A.; Baxevanis, C.N.; Rigatos, G.; Papamichail, M.; Perez, S.A. A phase I trial of adoptive transfer of allogeneic natural killer cells in patients with advanced non-small cell lung cancer. Cancer Immunol. Immunother. 2010, 59, 1781–1789. [Google Scholar] [CrossRef]
- Miller, J.S.; Soignier, Y.; Panoskaltsis-Mortari, A.; McNearney, S.A.; Yun, G.H.; Fautsch, S.K.; McKenna, D.; Le, C.; Defor, T.E.; Burns, L.J.; et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood 2005, 105, 3051–3057. [Google Scholar] [CrossRef]
- Re, F.; Staudacher, C.; Zamai, L.; Vecchio, V.; Bregni, M. Killer cell Ig-like receptors ligand-mismatched, alloreactive natural killer cells lyse primary solid tumors. Cancer 2006, 107, 640–648. [Google Scholar] [CrossRef] [PubMed]
- Geller, M.A.; Cooley, S.; Judson, P.L.; Ghebre, R.; Carson, L.F.; Argenta, P.A.; Jonson, A.L.; Panoskaltsis-Mortari, A.; Curtsinger, J.; McKenna, D.; et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy 2011, 13, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Wolf, N.K.; Kissiov, D.U.; Raulet, D.H. Roles of natural killer cells in immunity to cancer, and applications to immunotherapy. Nat. Rev. Immunol. 2023, 23, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.; Gao, F.; Yan, M.; Zhao, S.; Yan, Z.; Shi, B.; Liu, Y. Natural killer cells: A promising immunotherapy for cancer. J. Transl. Med. 2022, 20, 240. [Google Scholar] [CrossRef]
- Portale, F.; Di Mitri, D. NK Cells in Cancer: Mechanisms of Dysfunction and Therapeutic Potential. Int. J. Mol. Sci. 2023, 24, 9521. [Google Scholar] [CrossRef]
- Fang, F.; Xie, S.; Chen, M.; Li, Y.; Yue, J.; Ma, J.; Shu, X.; He, Y.; Xiao, W.; Tian, Z. Advances in NK cell production. Cell. Mol. Immunol. 2022, 19, 460–481. [Google Scholar] [CrossRef]
- Jurisic, V.; Colovic, N.; Konjevic, G.; Minic, I.; Colovic, M. An aggressive extramedullary cutaneous plasmacytoma associated with extreme alterations in the innate immune system. Onkologie 2010, 33, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Soderstrom, K.; Stein, E.; Colmenero, P.; Purath, U.; Müller-Ladner, U.; Teixeira de Matos, C.; Tarner, I.H.; Robinson, W.H.; Engleman, E.G. Natural killer cells trigger osteoclastogenesis and bone destruction. in arthritis. Proc. Natl. Acad. Sci. USA 2010, 107, 13028–13033. [Google Scholar] [CrossRef]
- van den Berg, W.B. Anti-cytokine therapy in chronic destructive arthritis. Arthritis Res. 2001, 3, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Bemiller, L.S.R.D.; Starko, K.M.; Curnutte, J.T. Safety and effectiveness of long-term interferon gamma therapy in patients with chronic granulomatous disease. Blood Cells Mol. Dis. 1995, 21, 239–247. [Google Scholar] [CrossRef]
- Li, H.; Hong, S.; Qian, J.; Zheng, Y.; Yang, J.; Yi, Q. Cross talk between the bone and immune systems: Osteoclasts function as antigen-presenting cells and activate CD4+ and CD8+ T cells. Blood 2010, 116, 210–217. [Google Scholar] [CrossRef]
- Kaur, K.; Cook, J.; Park, S.H.; Topchyan, P.; Kozlowska, A.; Ohanian, N.; Fang, C.; Nishimura, I.; Jewett, A. Novel Strategy to Expand Super-Charged NK Cells with Significant Potential to Lyse and Differentiate Cancer Stem Cells: Differences in NK Expansion and Function between Healthy and Cancer Patients. Front. Immunol. 2017, 8, 297. [Google Scholar] [CrossRef]
- Ko, M.W.; Mei, A.; Senjor, E.; Nanut, M.P.; Gao, L.W.; Wong, P.; Chen, P.C.; Cohn, W.; Whitelegge, J.P.; Kos, J.; et al. Osteoclast-expanded supercharged NK cells perform superior antitumour effector functions. BMJ Oncol. 2025, 4, e000676. [Google Scholar] [CrossRef] [PubMed]
- Romee, R.; Rosario, M.; Berrien-Elliott, M.M.; Wagner, J.A.; Jewell, B.A.; Schappe, T.; Chen, P.C.; Cohn, W.; Whitelegge, J.P.; Kos, J.; et al. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci. Transl. Med. 2016, 8, 357ra123. [Google Scholar] [CrossRef]
- Terrén, I.; Orrantia, A.; Astarloa-Pando, G.; Amarilla-Irusta, A.; Zenarruzabeitia, O.; Borrego, F. Cytokine-Induced Memory-Like NK Cells: From the Basics to Clinical Applications. Front. Immunol. 2022, 13, 884648. [Google Scholar] [CrossRef]
- Kaur, K.; Safaie, T.; Ko, M.-W.; Wang, Y.; Jewett, A. ADCC against MICA/B Is Mediated against Differentiated Oral and Pancreatic and Not Stem-Like/Poorly Differentiated Tumors by the NK Cells; Loss in Cancer Patients due to Down-Modulation of CD16 Receptor. Cancers 2021, 13, 239. [Google Scholar] [CrossRef]
- Huerta-Yepez, S.; Chen, P.C.; Kaur, K.; Jain, Y.; Singh, T.; Esedebe, F.; Liao, Y.J.; DiBernardo, G.; Moatamed, N.A.; Mei, A.; et al. Supercharged NK cells, unlike primary activated NK cells, effectively target ovarian cancer cells irrespective of MHC-class I expression. BMJ Oncol. 2025, 4, e000618. [Google Scholar] [CrossRef]
- Kaur, K.; Kozlowska, A.K.; Topchyan, P.; Ko, M.W.; Ohanian, N.; Chiang, J.; Cook, J.; Maung, P.O.; Park, S.H.; Cacalano, N.; et al. Probiotic-Treated Super-Charged NK Cells Efficiently Clear Poorly Differentiated Pancreatic Tumors in Hu-BLT Mice. Cancers 2019, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Bui, V.T.; Tseng, H.C.; Kozlowska, A.; Maung, P.O.; Kaur, K.; Topchyan, P.; Jewett, A. Augmented IFN-γ and TNF-α Induced by Probiotic Bacteria in NK Cells Mediate Differentiation of Stem-Like Tumors Leading to Inhibition of Tumor Growth and Reduction in Inflammatory Cytokine Release; Regulation by IL-10. Front. Immunol. 2015, 6, 576. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Topchyan, P.; Kozlowska, A.K.; Ohanian, N.; Chiang, J.; Maung, P.O.; Park, S.H.; Ko, M.W.; Fang, C.; Nishimura, I.; et al. Super-charged NK cells inhibit growth and progression of stem-like/poorly differentiated oral tumors in vivo in humanized BLT mice; effect on tumor differentiation and response to chemotherapeutic drugs. Oncoimmunology 2018, 7, e1426518. [Google Scholar] [CrossRef]
- Dong, H.; Rowland, I.; Yaqoob, P. Comparative effects of six probiotic strains on immune function in vitro. Br. J. Nutr. 2012, 108, 459–470. [Google Scholar] [CrossRef]
- Ko, M.-W.; Kaur, K.; Safaei, T.; Chen, W.; Sutanto, C.; Wong, P.; Jewett, A. Defective Patient NK Function Is Reversed by AJ2 Probiotic Bacteria or Addition of Allogeneic Healthy Monocytes. Cells 2022, 11, 697. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, X.; Zhang, C.; Liu, Z.; Li, C.; Ren, Z. Gut Microbiota and Bone Diseases: A Growing Partnership. Front. Microbiol. 2022, 13, 877776. [Google Scholar] [CrossRef]
- Ticinesi, A.; Siniscalchi, C.; Meschi, T.; Nouvenne, A. Gut microbiome and bone health: Update on mechanisms, clinical correlations, and possible treatment strategies. Osteoporos. Int. 2025, 36, 167–191. [Google Scholar] [CrossRef]
- Han, D.; Wang, W.; Gong, J.; Ma, Y.; Li, Y. Microbiota metabolites in bone: Shaping health and Confronting disease. Heliyon 2024, 10, e28435. [Google Scholar] [CrossRef]
- Cristofori, F.; Dargenio, V.N.; Dargenio, C.; Miniello, V.L.; Barone, M.; Francavilla, R. Anti-Inflammatory and Immunomodulatory Effects of Probiotics in Gut Inflammation: A Door to the Body. Front. Immunol. 2021, 12, 578386. [Google Scholar] [CrossRef]
- Wallimann, A.; Magrath, W.; Thompson, K.; Moriarty, T.; Richards, R.G.; Akdis, C.A.; O'Mahony, L.; Hernandez, C.J. Gut microbial-derived short-chain fatty acids and bone: A potential role in fracture healing. Eur. Cell Mater. 2021, 41, 454–470. [Google Scholar] [CrossRef]
- Lucas, S.; Omata, Y.; Hofmann, J.; Böttcher, M.; Iljazovic, A.; Sarter, K.; Albrecht, O.; Schulz, O.; Krishnacoumar, B.; Krönke, G.; et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat. Commun. 2018, 9, 55. [Google Scholar] [CrossRef]
- Gul, S.; Durante-Mangoni, E. Unraveling the Puzzle: Health Benefits of Probiotics-A Comprehensive Review. J. Clin. Med. 2024, 13, 1436. [Google Scholar] [CrossRef]
- Maftei, N.M.; Raileanu, C.R.; Balta, A.A.; Ambrose, L.; Boev, M.; Marin, D.B.; Lisa, E.L. The Potential Impact of Probiotics on Human Health: An Update on Their Health-Promoting Properties. Microorganisms 2024, 12, 234. [Google Scholar] [CrossRef]
- Ranjha, M.M.A.N.; Shafique, B.; Batool, M.; Kowalczewski, P.Ł.; Shehzad, Q.; Usman, M.; Manzoor, M.F.; Zahra, S.M.; Yaqub, S.; Aadil, R.M. Nutritional and Health Potential of Probiotics: A Review. Appl. Sci. 2021, 11, 11204. [Google Scholar] [CrossRef]
- Chu, X.; Xing, H.; Chao, M.; Xie, P.; Jiang, L. Gut Microbiota Modulation in Osteoporosis: Probiotics, Prebiotics, and Natural Compounds. Metabolites 2025, 15, 301. [Google Scholar] [CrossRef]
- Lyu, Z.; Hu, Y.; Guo, Y.; Liu, D. Modulation of bone remodeling by the gut microbiota: A new therapy for osteoporosis. Bone Res. 2023, 11, 31. [Google Scholar] [CrossRef]
- McCabe, L.R.; Parameswaran, N. Advances in Probiotic Regulation of Bone and Mineral Metabolism. Calcif. Tissue Int. 2018, 102, 480–488. [Google Scholar] [CrossRef]
- Guder, C.; Gravius, S.; Burger, C.; Wirtz, D.C.; Schildberg, F.A. Osteoimmunology: A Current Update of the Interplay Between Bone and the Immune System. Front. Immunol. 2020, 11, 58. [Google Scholar] [CrossRef]
- Walsh, M.C.; Kim, N.; Kadono, Y.; Rho, J.; Lee, S.Y.; Lorenzo, J.; Choi, Y. OSTEOIMMUNOLOGY: Interplay Between the Immune System and Bone Metabolism. Annu. Rev. Immunol. 2006, 24, 33–63. [Google Scholar] [CrossRef]
- dos Santos Freitas, A.; da Silva Fernandes, L.J.; Coelho-Rocha, N.D.; de Jesus, L.C.L.; de Rezende Rodovalho, V.; da Silva, T.F.; de Oliveira Carvalho, R.D.; Azevedo, V. Chapter 16—Immunomodulatory and antiinflammatory mechanisms of probiotics. In Probiotics; Brandelli, A., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 321–341. [Google Scholar]
- Mazziotta, C.; Tognon, M.; Martini, F.; Torreggiani, E.; Rotondo, J.C. Probiotics Mechanism of Action on Immune Cells and Beneficial Effects on Human Health. Cells 2023, 12, 184. [Google Scholar] [CrossRef]
- McCabe, L.; Britton, R.A.; Parameswaran, N. Prebiotic and Probiotic Regulation of Bone Health: Role of the Intestine and its Microbiome. Curr. Osteoporos. Rep. 2015, 13, 363–371. [Google Scholar] [CrossRef]
- Shimizu, S.; Hong, P.; Arumugam, B.; Pokomo, L.; Boyer, J.; Koizumi, N.; Kittipongdaja, P.; Chen, A.; Bristol, G.; Galic, Z.; et al. A highly efficient short hairpin RNA potently down-regulates CCR5 expression in systemic lymphoid organs in the hu-BLT mouse model. Blood 2010, 115, 1534–1544. [Google Scholar] [CrossRef]
- Vatakis, D.N.; Koya, R.C.; Nixon, C.C.; Wei, L.; Kim, S.G.; Avancena, P.; Bristol, G.; Baltimore, D.; Kohn, D.B.; Ribas, A.; et al. Antitumor activity from antigen-specific CD8 T cells generated in vivo from genetically engineered human hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 2011, 108, E1408–E1416. [Google Scholar] [CrossRef]
- Jurisic, V. Multiomic analysis of cytokines in immuno-oncology. Expert. Rev. Proteom. 2020, 17, 663–674. [Google Scholar] [CrossRef]
- Bachmann, M.F.; Oxenius, A. Interleukin 2: From immunostimulation to immunoregulation and back again. EMBO Rep. 2007, 8, 1142–1148. [Google Scholar] [CrossRef]
- Muhammad, S.; Fan, T.; Hai, Y.; Gao, Y.; He, J. Reigniting hope in cancer treatment: The promise and pitfalls of IL-2 and IL-2R targeting strategies. Mol. Cancer 2023, 22, 121. [Google Scholar] [CrossRef]
- Rokade, S.; Damani, A.M.; Oft, M.; Emmerich, J. IL-2 based cancer immunotherapies: An evolving paradigm. Front. Immunol. 2024, 15, 1433989. [Google Scholar] [CrossRef]
- Kaur, K.; Chen, P.-C.; Ko, M.-W.; Mei, A.; Senjor, E.; Malarkannan, S.; Kos, J.; Jewett, A. Sequential therapy with supercharged NK cells with either chemotherapy drug cisplatin or anti-PD-1 antibody decreases the tumor size and significantly enhances the NK function in Hu-BLT mice. Front. Immunol. 2023, 14, 1132807. [Google Scholar] [CrossRef]
- Kaur, K.; Jewett, A. Supercharged NK Cell-Based Immuotherapy in Humanized Bone Marrow Liver and Thymus (Hu-BLT) Mice Model of Oral, Pancreatic, Glioblastoma, Hepatic, Melanoma and Ovarian Cancers. Crit. Rev. Immunol. 2023, 43, 13–25. [Google Scholar] [CrossRef]
- Kaur, K.; Chang, H.H.; Cook, J.; Eibl, G.; Jewett, A. Suppression of Gingival NK Cells in Precancerous and Cancerous Stages of Pancreatic Cancer in KC and BLT-Humanized Mice. Front. Immunol. 2017, 8, 1606. [Google Scholar] [CrossRef]
- Olesen, R.; Wahl, A.; Denton, P.W.; Garcia, J.V. Immune reconstitution of the female reproductive tract of humanized BLT mice and their susceptibility to human immunodeficiency virus infection. J. Reprod. Immunol. 2011, 88, 195–203. [Google Scholar] [CrossRef]
- Denton, P.W.; Olesen, R.; Choudhary, S.K.; Archin, N.M.; Wahl, A.; Swanson, M.D.; Chateau, M.; Nochi, T.; Krisko, J.F.; Spagnuolo, R.A.; et al. Generation of HIV latency in humanized BLT mice. J. Virol. 2012, 86, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Shultz, L.D.; Goodwin, N.; Ishikawa, F.; Hosur, V.; Lyons, B.L.; Greiner, D.L. Human cancer growth and therapy in immunodeficient mouse models. Cold Spring Harb. Protoc. 2014, 2014, 694–708. [Google Scholar] [CrossRef] [PubMed]
- Kozlowska, A.K.; Kaur, K.; Topchyan, P.; Jewett, A. Adoptive transfer of osteoclast-expanded natural killer cells for immunotherapy targeting cancer stem-like cells in humanized mice. Cancer Immunol. Immunother. 2016, 65, 835–845. [Google Scholar] [CrossRef]
- Kaur, K.; Topchyan, P.; Jewett, A. Supercharged Natural Killer (sNK) Cells Inhibit Melanoma Tumor Progression and Restore Endogenous NK Cell Function in Humanized BLT Mice. Cancers 2025, 17, 2430. [Google Scholar] [CrossRef]
- Decombis, S.; Smolander, J.; Bouhlal, J.; Klievink, J.; Lähteenmäki, H.; Jokinen, E.; Lin, L.; Laitinen, H.; Matjusinski, K.; Holappa, E.; et al. Bone Marrow Stromal Cells Hamper Leukemia Cell Induced NK Cell Activation and Cytotoxicity. Blood 2024, 144 (Suppl. S1), 4814. [Google Scholar] [CrossRef]
- Aref, S.; Khaled, N.; Al Gilany, A.H.; Ayed, M.; Abouzeid, T.; Attia, D. Impact of Bone Marrow Natural Killer Cells (NK); Soluble TNF-α and IL-32 Levels in Myelodysplastic Syndrome Patients. Asian Pac. J. Cancer Prev. 2020, 21, 2949–2953. [Google Scholar] [CrossRef]
- Shupp, A.B.; Kolb, A.D.; Mukhopadhyay, D.; Bussard, K.M. Cancer Metastases to Bone: Concepts, Mechanisms, and Interactions with Bone Osteoblasts. Cancers 2018, 10, 182. [Google Scholar] [CrossRef]
- Shi, X.; Wang, X.; Yao, W.; Shi, D.; Shao, X.; Lu, Z.; Chai, Y.; Song, J.; Tang, W.; Wang, X. Mechanism insights and therapeutic intervention of tumor metastasis: Latest developments and perspectives. Signal Transduct. Target. Ther. 2024, 9, 192. [Google Scholar] [CrossRef] [PubMed]
- Martiniakova, M.; Mondockova, V.; Biro, R.; Kovacova, V.; Babikova, M.; Zemanova, N.; Ciernikova, S.; Omelka, R. The link between bone-derived factors osteocalcin, fibroblast growth factor 23, sclerostin, lipocalin 2 and tumor bone metastasis. Front. Endocrinol. 2023, 14, 1113547. [Google Scholar] [CrossRef]
- Satcher, R.L.; Zhang, X.H.F. Evolving cancer–niche interactions and therapeutic targets during bone metastasis. Nat. Rev. Cancer 2022, 22, 85–101. [Google Scholar] [CrossRef]
- Wang, M.; Xia, F.; Wei, Y.; Wei, X. Molecular mechanisms and clinical management of cancer bone metastasis. Bone Res. 2020, 8, 30. [Google Scholar] [CrossRef]
- Clézardin, P.; Coleman, R.; Puppo, M.; Ottewell, P.; Bonnelye, E.; Paycha, F.; Confavreux, C.B.; Holen, I. Bone metastasis: Mechanisms, therapies, and biomarkers. Physiol. Rev. 2021, 101, 797–855. [Google Scholar] [CrossRef]
- Cai, L.; Lv, Y.; Yan, Q.; Guo, W. Cytokines: The links between bone and the immune system. Injury 2024, 55, 111203. [Google Scholar] [CrossRef] [PubMed]
- Di Pompo, G.; Errani, C.; Gillies, R.; Mercatali, L.; Ibrahim, T.; Tamanti, J.; Baldini, N.; Avnet, S. Acid-Induced Inflammatory Cytokines in Osteoblasts: A Guided Path to Osteolysis in Bone Metastasis. Front. Cell Dev. Biol. 2021, 9, 678532. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yu, L.; Liu, F.; Wan, L.; Deng, Z. The effect of cytokines on osteoblasts and osteoclasts in bone remodeling in osteoporosis: A review. Front. Immunol. 2023, 14, 1222129. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Mining genome traits that determine the different gut colonization potential of Lactobacillus and Bifidobacterium species. Microb Genom 2021, 7, 000581. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Sapra, L.; Tiwari, A.; Mishra, P.K.; Srivastava, R.K. Lactobacillus acidophilus ameliorates inflammatory bone loss under post-menopausal osteoporotic conditions via preventing the pathogenic conversion of gut resident pTregs into Th17 cells. bioRxiv 2025, 2025, 03.20.644481. [Google Scholar] [CrossRef]
- Mafe, A.N.; Edo, G.I.; Majeed, O.S.; Gaaz, T.S.; Akpoghelie, P.O.; Isoje, E.F.; Igbuku, U.A.; Owheruo, J.O.; Opiti, R.A.; Garba, Y.; et al. A review on probiotics and dietary bioactives: Insights on metabolic well-being, gut microbiota, and inflammatory responses. Food Chem. Adv. 2025, 6, 100919. [Google Scholar] [CrossRef]
- Collins, F.L.; Rios-Arce, N.D.; Schepper, J.D.; Parameswaran, N.; McCabe, L.R. The Potential of Probiotics as a Therapy for Osteoporosis. Microbiol. Spectr. 2017, 5, 213–233. [Google Scholar] [CrossRef]
- Rodriguez-Arrastia, M.; Martinez-Ortigosa, A.; Rueda-Ruzafa, L.; Folch Ayora, A.; Ropero-Padilla, C. Probiotic Supplements on Oncology Patients’ Treatment-Related Side Effects: A Systematic Review of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2021, 18, 4265. [Google Scholar] [CrossRef]
- Espinoza, I.R.G.; Castro-Ponce, A.; Tapia, A.A.; Juárez-Salazar, G.; Garcia, C.A.C.; Aguilar, C.; Cruz, E.M.; Ramos, F.d.J.V.; Priego, J.M.A.; Chiquillo-Domínguez, M.; et al. Effect of probiotics on quality of life for patients with cancer undergoing immunotherapy. J. Clin. Oncol. 2024, 42 (Suppl. S16), e24148. [Google Scholar] [CrossRef]
- Lei, W.; Zhou, K.; Lei, Y.; Li, Q.; Zhu, H. Gut microbiota shapes cancer immunotherapy responses. Npj Biofilms Microbiomes 2025, 11, 143. [Google Scholar] [CrossRef]
- Kimmelman, J.; Bodilly Kane, P.; Bicer, S.; Carlisle, B.G. Preclinical assessment for translation to humans: The PATH approach for assessing supporting evidence for early-phase trials and innovative care. Med 2024, 5, 1227–1236. [Google Scholar] [CrossRef]
- Indrio, F.; Salatto, A. Gut Microbiota-Bone Axis. Ann. Nutr. Metab. 2025, 81 (Suppl. S1), 47–56. [Google Scholar] [CrossRef]
- Wei, J.; Liu, Q.; Yuen, H.-Y.; Lam, A.C.-H.; Jiang, Y.; Yang, Y.; Liu, Y.; Zhao, X.; Xiao, L. Gut-bone axis perturbation: Mechanisms and interventions via gut microbiota as a primary driver of osteoporosis. J. Orthop. Transl. 2025, 50, 373–387. [Google Scholar] [CrossRef]
- Imširović, V.; Wensveen, F.M.; Polić, B.; Jelenčić, V. Maintaining the Balance: Regulation of NK Cell Activity. Cells 2024, 13, 1464. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, Q.; Zhao, J.; Song, D. The roles of natural killer cells in bone and arthritic disease: A narrative review. Immunol. Med. 2025, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Wang, H.; Yang, Y.; Tang, C.; Sun, X.; Zhou, J.; Liu, S.; Li, Q.; Zhao, L.; Gao, Z. Common mechanisms of Gut microbe-based strategies for the treatment of intestine-related diseases: Based on multi-target interactions with the intestinal barrier. Cell Commun. Signal. 2025, 23, 288. [Google Scholar] [CrossRef]
- Gao, H.; Li, X.; Chen, X.; Hai, D.; Wei, C.; Zhang, L.; Li, P. The Functional Roles of Lactobacillus acidophilus in Different Physiological and Pathological Processes. J. Microbiol. Biotechnol. 2022, 32, 1226–1233. [Google Scholar] [CrossRef]
- Zhao, W.; Liu, Y.; Kwok, L.-Y.; Cai, T.; Zhang, W. The immune regulatory role of Lactobacillus acidophilus: An updated meta-analysis of randomized controlled trials. Food Biosci. 2020, 36, 100656. [Google Scholar] [CrossRef]
- Renukadevi, J.; Helinto, J.S.; Prena, D. Immunotherapeutic Potential of Lactobacillus Species as Immune Checkpoint Inhibitors in Cancer Immunotherapy. J. Bio-X Res. 2025, 8, 0028. [Google Scholar] [CrossRef]
- Qureshi, Z.; Jamil, A.; Altaf, F.; Siddique, R. Efficacy and Safety of Probiotics as Adjunctive Therapy in Cancer Treatment: A Comprehensive Systematic Review and Meta-Analysis. Am. J. Clin. Oncol. 2025, 48, 148–161. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kumar, D.; Guha, D. Insights of probiotics as an alternative medicine for cancer therapy, mechanism, and applications. Med. Microecol. 2024, 22, 100111. [Google Scholar] [CrossRef]
- Aziz, N.; Bonavida, B. Activation of Natural Killer Cells by Probiotics. Onco Ther. 2016, 7, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Gui, Q.; Wang, A.; Zhao, X.; Huang, S.; Tan, Z.; Xiao, C.; Yang, Y. Effects of probiotic supplementation on natural killer cell function in healthy elderly individuals: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2020, 74, 1630–1637. [Google Scholar] [CrossRef]
- Rizzoli, R.; Biver, E. Are Probiotics the New Calcium and Vitamin D for Bone Health? Curr. Osteoporos. Rep. 2020, 18, 273–284. [Google Scholar] [CrossRef]
- Varvara, R.-A.; Vodnar, D.C. Probiotic-driven advancement: Exploring the intricacies of mineral absorption in the human body. Food Chem. X 2024, 21, 101067. [Google Scholar] [CrossRef]
- Parvaneh, K.; Jamaluddin, R.; Karimi, G.; Erfani, R. Effect of probiotics supplementation on bone mineral content and bone mass density. Sci. World J. 2014, 2014, 595962. [Google Scholar] [CrossRef] [PubMed]
- Cenci, S.; Toraldo, G.; Weitzmann, M.N.; Roggia, C.; Gao, Y.; Qian, W.P.; Sierra, O.; Pacifici, R. Estrogen deficiency induces bone loss by increasing T cell proliferation and lifespan through IFN-gamma-induced class II transactivator. Proc. Natl. Acad. Sci. USA 2003, 100, 10405–10410. [Google Scholar] [CrossRef]
- Yang, S.M.P.; Ries, W.; Key, L.L.; Madyastha, P. Characterization of interferon gamma receptors on osteoclasts: Effect of interferon gamma on osteoclastic superoxidegeneration. J. Cell Biochem. 2002, 84, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Grewal, T.S.G.P.; Brabbs, A.C.; Birch, M.; Skerry, M.T. Best5: A novel interferon-inducible gene expressed during bone formation. FASEB J. 2000, 14, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Segal, J.G.L.N.; Tsung, Y.L.; Norton, J.A.; Tsung, K. The role of IFN-gamma in rejection of established tumors by IL-12: Source of production and target. Cancer Res. 2002, 62, 4696–4703. [Google Scholar]
- Steinmuller, C.F.-U.G.; Lohmann-Matthes, M.L.; Emmendorffer, A. Local activation of nonspecific defense against a respiratory model infection by application of interferon- gamma: Comparison between rat alveolar and interstitial lung macrophages. Am. J. Respir. Cell Mol. Biol. 2000, 22, 481–490. [Google Scholar] [CrossRef]
- Xu, Z.; Hurchla, M.A.; Deng, H.; Uluckan, O.; Bu, F.; Berdy, A.; Eaglenton, M.C.; Heller, E.A.; Floyd, D.H.; Dirksen, W.P.; et al. Interferon-gamma targets cancer cells and osteoclasts to prevent tumor-associated bone loss and bone metastases. J. Biol. Chem. 2009, 284, 4658–4666. [Google Scholar] [CrossRef]
- Toh, M.L.; Miossec, P. The role of T cells in rheumatoid arthritis: New subsets and new targets. Curr. Opin. Rheumatol. 2007, 19, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Ji, F.; Wang, Y.; Zhang, Y.; Liu, Q.; Zhang, J.Z.; Matsushima, K.; Cao, Q.; Zhang, Y. Synovial autoreactive T cells in rheumatoid arthritis resist IDO-mediated inhibition. J. Immunol. 2006, 177, 8226–8233. [Google Scholar] [CrossRef] [PubMed]
- Wan, B.; Nie, H.; Liu, A.; Feng, G.; He, D.; Xu, R.; Zhang, Q.; Dong, C.; Zhang, J.Z. Aberrant regulation of synovial T cell activation by soluble costimulatory molecules in rheumatoid arthritis. J. Immunol. 2006, 177, 8844–8850. [Google Scholar] [CrossRef]
- Cochran, A.J.; Wen, D.R.; Farzad, Z.; Stene, M.A.; McBride, W.; Lana, A.M.; Morton, D.L. Immunosuppression by melanoma cells as a factor in the generation of metastatic disease. Anticancer Res. 1989, 9, 859–864. [Google Scholar]
- Kim, R.; Emi, M.; Tanabe, K. Cancer immunoediting from immune surveillance to immune escape. Immunology 2007, 121, 1–14. [Google Scholar] [CrossRef]
- Merchant, M.S.; Melchionda, F.; Sinha, M.; Khanna, C.; Helman, L.; Mackall, C.L. Immune reconstitution prevents metastatic recurrence of murine osteosarcoma. Cancer Immunol. Immunother. 2007, 56, 1037–1046. [Google Scholar] [CrossRef]
- Li, S.; Liu, G.; Hu, S. Osteoporosis: Interferon-gamma-mediated bone remodeling in osteoimmunology. Front. Immunol. 2024, 15, 1396122. [Google Scholar] [CrossRef]
- Tang, M.; Tian, L.; Luo, G.; Yu, X. Interferon-Gamma-Mediated Osteoimmunology. Front. Immunol. 2018, 9, 1508. [Google Scholar] [CrossRef]
- Morales-Mantilla, D.E.; King, K.Y. The Role of Interferon-Gamma in Hematopoietic Stem Cell Development, Homeostasis, and Disease. Curr. Stem Cell Rep. 2018, 4, 264–271. [Google Scholar] [CrossRef]
- Liu, X. The paradoxical role of IFN-γ in cancer: Balancing immune activation and immune evasion. Pathol. Res. Pract. 2025, 272, 156046. [Google Scholar] [CrossRef] [PubMed]
- de Bruin, A.M.; Voermans, C.; Nolte, M.A. Impact of interferon-γ on hematopoiesis. Blood 2014, 124, 2479–2486. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaur, K.; Reese, P.; Chiang, J.; Jewett, A. Natural Killer Cell Therapy Combined with Probiotic Bacteria Supplementation Restores Bone Integrity in Cancer by Promoting IFN-γ Production. Cells 2025, 14, 1347. https://doi.org/10.3390/cells14171347
Kaur K, Reese P, Chiang J, Jewett A. Natural Killer Cell Therapy Combined with Probiotic Bacteria Supplementation Restores Bone Integrity in Cancer by Promoting IFN-γ Production. Cells. 2025; 14(17):1347. https://doi.org/10.3390/cells14171347
Chicago/Turabian StyleKaur, Kawaljit, Patricia Reese, Jason Chiang, and Anahid Jewett. 2025. "Natural Killer Cell Therapy Combined with Probiotic Bacteria Supplementation Restores Bone Integrity in Cancer by Promoting IFN-γ Production" Cells 14, no. 17: 1347. https://doi.org/10.3390/cells14171347
APA StyleKaur, K., Reese, P., Chiang, J., & Jewett, A. (2025). Natural Killer Cell Therapy Combined with Probiotic Bacteria Supplementation Restores Bone Integrity in Cancer by Promoting IFN-γ Production. Cells, 14(17), 1347. https://doi.org/10.3390/cells14171347







