The Role of Calcium-Independent Phospholipase A2 in the Molecular Mechanisms of Schizophrenia
Abstract
1. Introduction
2. Cortical Atrophy and Negative/Cognitive Symptoms in Schizophrenia
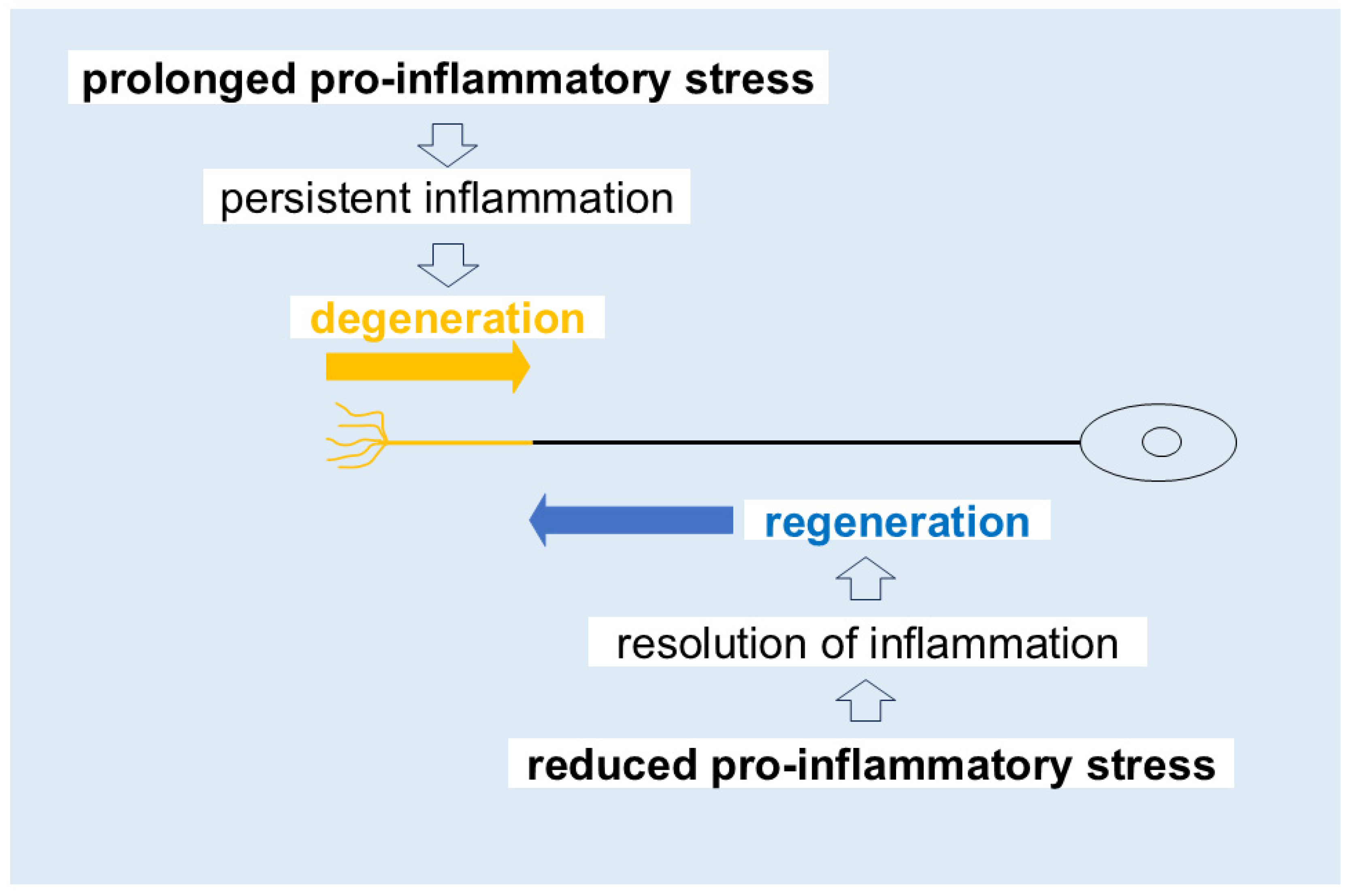
3. Hyper-Regeneration/Sprouting and Positive Symptoms: Involvement of Calcium-Independent PLA2
4. Hyper-Regeneration/Sprouting of Monoamine Axons in Peripheral Tissues
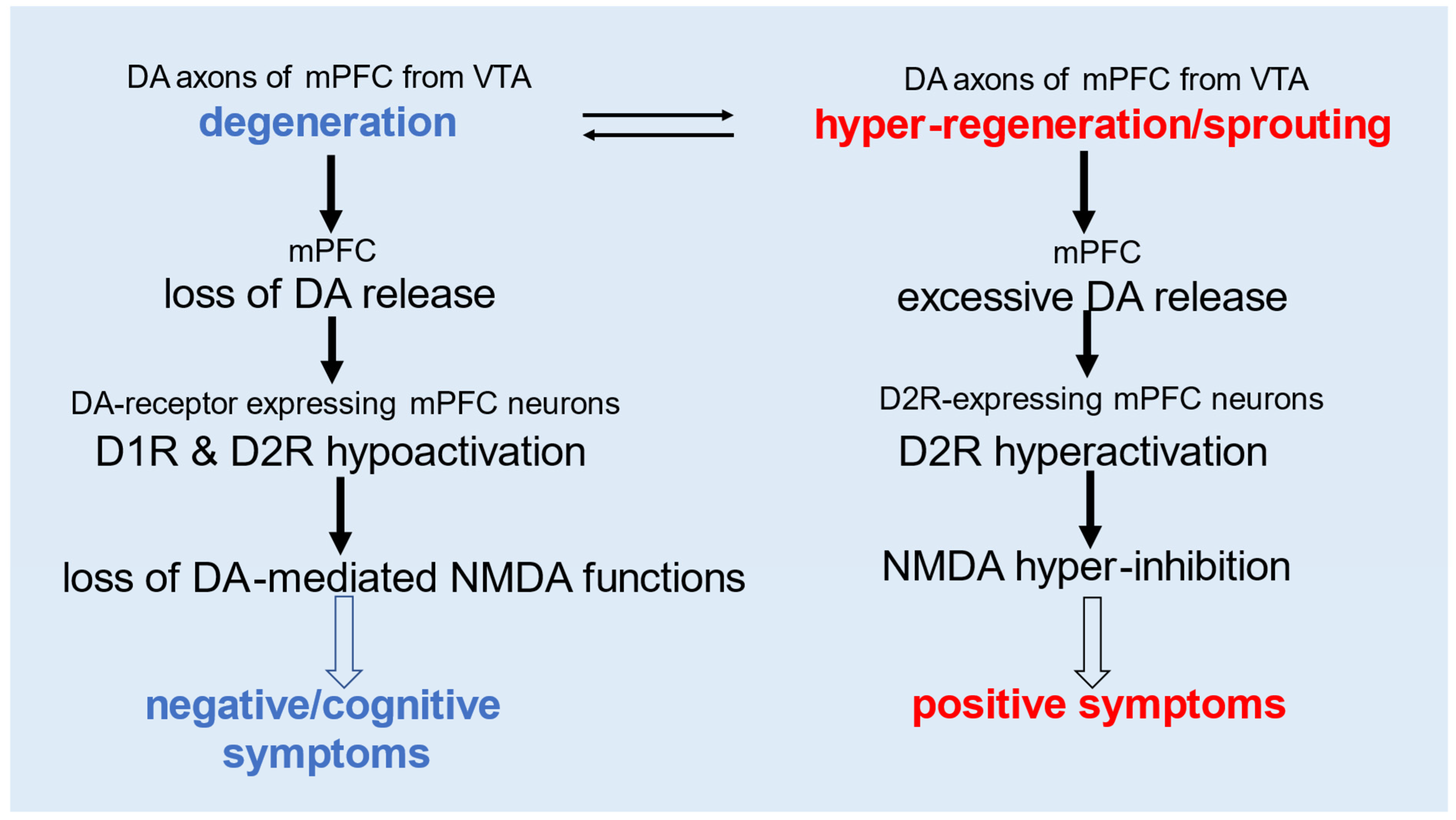
5. The Involvement of mPFC Neurons in the Occurrence of Positive and Negative/Cognitive Symptoms via Inactivation of DA-Mediated NMDA Responses
6. Diagnosis and Treatment of Schizophrenia, Depression, and Bipolar Disorder as Monoamine Axon Disorders
7. Conclusions and Perspective
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | Arachidonic acid |
| cPLA2 | Cytosolic phospholipase A2 |
| DA | Dopamine |
| D1R | Dopamine D1 receptor |
| D2R | Dopamine D2 receptor |
| DHA | Docosahexaenoic acid |
| EPA | Eicosapentaenoic acid |
| GSK3 | Glycogen synthase kinase-3 |
| 5-HT | Serotonin |
| iPLA2 | Calcium-independent phospholipase A2 |
| Li | Lithium |
| mPFC | Medial prefrontal cortex |
| NA | Noradrenaline |
| NGF | Nerve growth factor |
| NMDA | N-methyl-D-aspartate |
| NPD1 | Neuroprotectin D1 |
| NRG1 | Neuregulin-1 |
| PET | Positron emission tomography |
| PLA2 | Phospholipase A2 |
| VTA | Ventral tegmental area |
References
- Frankfurt, M.; Azmitia, E. Regeneration of serotonergic fibers in the rat hypothalamus following unilateral 5,7-dihydroxytryptamine injection. Brain Res. 1984, 298, 273–282. [Google Scholar] [CrossRef]
- Fritschy, J.; Grzanna, R. Restoration of ascending noradrenergic projections by residual locus coeruleus neurons: Compensatory response to neurotoxin-induced cell death in the adult rat brain. J. Comp. Neurol. 1992, 321, 421–441. [Google Scholar] [CrossRef]
- Ueda, S.; Kawata, M. Regeneration of serotonergic immunoreactive fibers in the brain of 5,6-dihydroxytryptamine treated rat. J. Hirnforsch. 1994, 35, 159–180. [Google Scholar]
- Zhou, D.; Grecksch, G.; Becker, A.; Frank, C.; Huether, G.; Pilz, J. Serotonergic hyperinnervation of the frontal cortex in an animal model of depression, the bulbectomized rat. J. Neurosci. Res. 1998, 54, 109–116. [Google Scholar] [CrossRef]
- Taylor, J.B.; Cunningham, M.C.; Benes, F.M. Neonatal raphe lesions increase dopamine fibers in prefrontal cortex of adult rats. NeuroReport 1998, 9, 1811–1815. [Google Scholar] [CrossRef]
- Gagnon, D.; Eid, L.; Coudé, D.; Whissel, C.; Di Paolo, T.; Parent, A.; Parent, M. Evidence for Sprouting of Dopamine and Serotonin Axons in the Pallidum of Parkinsonian Monkeys. Front. Neuroanat. 2018, 12, 38. [Google Scholar] [CrossRef]
- Nakamura, S. Integrated pathophysiology of schizophrenia, major depression, and bipolar disorder as monoamine axon disorder. Front. Biosci. 2022, 14, 4. [Google Scholar] [CrossRef]
- Nakamura, S. Perspective Chapter: Depression as a Disorder of Monoamine Axon Degeneration May Hold an Answer to Two Antidepressant Questions-Delayed Clinical Efficacy and Treatment-Resistant Depression. In COVID-19 Pandemic, Mental Health and Neuroscience-New Scenarios for Understanding and Treatment; IntechOpen: London, UK, 2023. [Google Scholar] [CrossRef]
- Farooqui, A.A.; Ong, W.-Y.; Horrocks, L.A. Inhibitors of Brain Phospholipase A2 Activity: Their Neuropharmacological Effects and Therapeutic Importance for the Treatment of Neurologic Disorders. Pharmacol. Rev. 2006, 58, 591–620. [Google Scholar] [CrossRef]
- Layé, S.; Nadjar, A.; Joffre, C.; Bazinet, R.P. Anti-Inflammatory Effects of Omega-3 Fatty Acids in the Brain: Physiological Mechanisms and Relevance to Pharmacology. Pharmacol. Rev. 2018, 70, 12–38. [Google Scholar] [CrossRef]
- Nakamura, S. Involvement of phospholipase A2 in axonal regeneration of brain noradrenergic neurones. NeuroReport 1993, 4, 371–374. [Google Scholar] [CrossRef]
- Shinzawa, K.; Sumi, H.; Ikawa, M.; Matsuoka, Y.; Okabe, M.; Sakoda, S.; Tsujimoto, Y. Neuroaxonal Dystrophy Caused by Group VIA Phospholipase A2 Deficiency in Mice: A Model of Human Neurodegenerative Disease. J. Neurosci. 2008, 28, 2212–2220. [Google Scholar] [CrossRef]
- Beck, G.; Shinzawa, K.; Hayakawa, H.; Baba, K.; Sumi-Akamaru, H.; Tsujimoto, Y.; Mochizuki, H.; Lee, J. Progressive Axonal Degeneration of Nigrostriatal Dopaminergic Neurons in Calcium-Independent Phospholipase A2β Knockout Mice. PLoS ONE 2016, 11, e0153789. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.L.; He, J.; Kakazu, A.H.; Jun, B.; Bazan, N.G.; Bazan, H.E. Defining a mechanistic link between pigment epithelium–derived factor, docosahexaenoic acid, and corneal nerve regeneration. J. Biol. Chem. 2017, 292, 18486–18499. [Google Scholar] [CrossRef] [PubMed]
- Plum, F. Prospects for research on schizophrenia. 3. Neurophysiology. Neuropathological findings. Neurosci. Res. Program Bull. 1972, 10, 384–388. [Google Scholar] [PubMed]
- Chaves, C.; Dursun, S.M.; Tusconi, M.; Hallak, J.E.C. Neuroinflammation and schizophrenia—Is there a link? Front. Psychiatry 2024, 15, 1356975. [Google Scholar] [CrossRef]
- Konopaske, G.T.; Lange, N.; Coyle, J.T.; Benes, F.M. Prefrontal Cortical Dendritic Spine Pathology in Schizophrenia and Bipolar Disorder. JAMA Psychiatry 2014, 71, 1323–1331. [Google Scholar] [CrossRef]
- Pierri, J.N.; Volk, C.L.E.; Auh, S.; Sampson, A.; Lewis, D.A. Decreased Somal Size of Deep Layer 3 Pyramidal Neurons in the Prefrontal Cortex of Subjects With Schizophrenia. Arch. Gen. Psychiatry 2001, 58, 466–473. [Google Scholar] [CrossRef]
- Akil, M.; Pierri, J.N.; Whitehead, R.E.; Edgar, C.L.; Mohila, C.; Sampson, A.R.; Lewis, D.A. Lamina-Specific Alterations in the Dopamine Innervation of the Prefrontal Cortex in Schizophrenic Subjects. Am. J. Psychiatry 1999, 156, 1580–1589. [Google Scholar] [CrossRef]
- Miller, A.H.; Raison, C.L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2015, 16, 22–34. [Google Scholar] [CrossRef]
- Pfau, M.L.; Ménard, C.; Russo, S.J. Inflammatory Mediators in Mood Disorders: Therapeutic Opportunities. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 411–428. [Google Scholar] [CrossRef]
- Bierhaus, A.; Wolf, J.; Andrassy, M.; Rohleder, N.; Humpert, P.M.; Petrov, D.; Ferstl, R.; von Eynatten, M.; Wendt, T.; Rudofsky, G.; et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc. Natl. Acad. Sci. USA 2003, 100, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Dunn, A.J.; Swiergiel, A.H.; de Beaurepaire, R. Cytokines as mediators of depression: What can we learn from animal studies? Neurosci. Biobehav. Rev. 2005, 29, 891–909. [Google Scholar] [CrossRef] [PubMed]
- Pace, T.W.; Mletzko, T.C.; Alagbe, O.; Musselman, D.L.; Nemeroff, C.B.; Miller, A.H.; Heim, C.M. Increased Stress-Induced Inflammatory Responses in Male Patients With Major Depression and Increased Early Life Stress. Am. J. Psychiatry 2006, 163, 1630–1633. [Google Scholar] [CrossRef]
- Darios, F.; Davletov, B. Omega-3 and omega-6 fatty acids stimulate cell membrane expansion by acting on syntaxin 3. Nature 2006, 440, 813–817. [Google Scholar] [CrossRef]
- Robson, L.G.; Dyall, S.C.; Sidloff, D.; Michael-Titus, A.T. Omega-3 polyunsaturated fatty acids increase the neurite outgrowth of rat sensory neurones throughout development and in aged animals. Neurobiol. Aging 2010, 31, 678–687. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Yip, P.K.; Adams, L.; Davies, M.; Lee, J.W.; Michael, G.J.; Priestley, J.V.; Michael-Titus, A.T. A Single Bolus of Docosahexaenoic Acid Promotes Neuroplastic Changes in the Innervation of Spinal Cord Interneurons and Motor Neurons and Improves Functional Recovery after Spinal Cord Injury. J. Neurosci. 2015, 35, 12733–12752. [Google Scholar] [CrossRef]
- Ross, B.M.; Hudson, C.; Erlich, J.; Warsh, J.J.; Kish, S.J. Increased Phospholipid Breakdown in Schizophrenia. Arch. Gen. Psychiatry 1997, 54, 487–494. [Google Scholar] [CrossRef]
- Xu, C.; Yang, X.; Sun, L.; Yang, T.; Cai, C.; Wang, P.; Jiang, J.; Qing, Y.; Hu, X.; Wang, D.; et al. An investigation of calcium-independent phospholipase A2 (iPLA2) and cytosolic phospholipase A2 (cPLA2) in schizophrenia. Psychiatry Res. 2019, 273, 782–787. [Google Scholar] [CrossRef]
- Lin, S.-H.; Liu, C.-M.; Chang, S.-S.; Hwu, H.-G.; Liu, S.K.; Hwang, T.J.; Hsieh, M.-H.; Guo, S.-C.; Chen, W.J. Familial Aggregation in Skin Flush Response to Niacin Patch Among Schizophrenic Patients and Their Nonpsychotic Relatives. Schizophr. Bull. 2006, 33, 174–182. [Google Scholar] [CrossRef]
- Crayton, J.W.; Meltzer, H.Y. Degeneration and regeneration of motor neurons in psychotic patients. Biol. Psychiatry 1979, 14, 803–819. [Google Scholar]
- Ross-Stanton, J. Motor Neuron Branching Patterns in Psychotic Patients. Arch. Gen. Psychiatry 1981, 38, 1097–1103. [Google Scholar] [CrossRef]
- Hasan, W.; Jama, A.; Donohue, T.; Wernli, G.; Onyszchuk, G.; Al-Hafez, B.; Bilgen, M.; Smith, P.G. Sympathetic hyperinnervation and inflammatory cell NGF synthesis following myocardial infarction in rats. Brain Res. 2006, 1124, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-M.; Lin, S.-Z.; Chang, N.-C. Inhibition of glycogen synthase kinase-3β prevents sympathetic hyperinnervation in infarcted rats. Exp. Biol. Med. 2015, 240, 979–992. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.L.; Fox, S.; Abell, K.M. Catecholamine Modulation of Lymphocyte Homing to Lymphoid Tissues. Brain Behav. Immun. 1997, 11, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.J.; Hudson, C.N.; Powley, T.L. Sympathetic axonopathies and hyperinnervation in the small intestine smooth muscle of aged Fischer 344 rats. Auton. Neurosci. 2013, 179, 108–121. [Google Scholar] [CrossRef]
- Rook, W.; Johnson, C.D.; Coney, A.M.; Marshall, J.M. Prenatal Hypoxia Leads to Increased Muscle Sympathetic Nerve Activity, Sympathetic Hyperinnervation, Premature Blunting of Neuropeptide Y Signaling, and Hypertension in Adult Life. Hypertension 2014, 64, 1321–1327. [Google Scholar] [CrossRef]
- Swissa, M.; Zhou, S.; Tan, A.Y.; Fishbein, M.C.; Chen, P.-S.; Chen, L.S. Atrial sympathetic and parasympathetic nerve sprouting and hyperinnervation induced by subthreshold electrical stimulation of the left stellate ganglion in normal dogs. Cardiovasc. Pathol. 2008, 17, 303–308. [Google Scholar] [CrossRef]
- Muñoz-Montaño, J.R.; Lim, F.; Moreno, F.J.; Avila, J.; Díaz-Nido, J. Glycogen Synthase Kinase-3 Modulates Neurite Outgrowth in Cultured Neurons: Possible Implications for Neurite Pathology in Alzheimer’s Disease. J. Alzheimer’s Dis. 1999, 1, 361–378. [Google Scholar] [CrossRef]
- Shah, S.; Patel, C.; Feng, A.; Kollmar, R. Lithium alters the morphology of neurites regenerating from cultured adult spiral ganglion neurons. Hear. Res. 2013, 304, 137–144. [Google Scholar] [CrossRef][Green Version]
- Nadri, C.; Belmaker, R.; Agam, G. Oxygen restriction of neonate rats elevates neuregulin-1α isoform levels: Possible relationship to schizophrenia. Neurochem. Int. 2007, 51, 447–450. [Google Scholar] [CrossRef]
- Kato, T.; Abe, Y.; Sotoyama, H.; Kakita, A.; Kominami, R.; Hirokawa, S.; Ozaki, M.; Takahashi, H.; Nawa, H. Transient exposure of neonatal mice to neuregulin-1 results in hyperdopaminergic states in adulthood: Implication in neurodevelopmental hypothesis for schizophrenia. Mol. Psychiatry 2010, 16, 307–320. [Google Scholar] [CrossRef]
- Vincent, S.; Khan, Y.; Benes, F. Cellular distribution of dopamine D1 and D2 receptors in rat medial prefrontal cortex. J. Neurosci. 1993, 13, 2551–2564. [Google Scholar] [CrossRef]
- Santana, N.; Mengod, G.; Artigas, F. Quantitative Analysis of the Expression of Dopamine D1 and D2 Receptors in Pyramidal and GABAergic Neurons of the Rat Prefrontal Cortex. Cereb. Cortex 2008, 19, 849–860. [Google Scholar] [CrossRef]
- Zheng, P.; Zhang, X.-X.; Bunney, B.; Shi, W.-X. Opposite modulation of cortical N-methyl-d-aspartate receptor-mediated responses by low and high concentrations of dopamine. Neuroscience 1999, 91, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Rolland, B.; Jardri, R.; Amad, A.; Thomas, P.; Cottencin, O.; Bordet, R. Pharmacology of Hallucinations: Several Mechanisms for One Single Symptom? BioMed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Balu, D.T. The NMDA receptor and schizophrenia: From pathophysiology to treatment. Adv. Pharmacol. 2016, 76, 351–382. [Google Scholar] [CrossRef] [PubMed]
- Tagliaferro, P.; Burke, R.E. Retrograde Axonal Degeneration in Parkinson Disease. J. Park. Dis. 2016, 6, 1–15. [Google Scholar] [CrossRef]
- Salvadores, N.; Sanhueza, M.; Manque, P.; Court, F.A. Axonal Degeneration during Aging and Its Functional Role in Neurodegenerative Disorders. Front. Neurosci. 2017, 11, 451. [Google Scholar] [CrossRef]
- Kneynsberg, A.; Combs, B.; Christensen, K.; Morfini, G.; Kanaan, N.M. Axonal Degeneration in Tauopathies: Disease Relevance and Underlying Mechanisms. Front. Neurosci. 2017, 11, 572. [Google Scholar] [CrossRef]
- Aron, L.; Ngian, Z.K.; Qiu, C.; Choi, J.; Liang, M.; Drake, D.M.; Hamplova, S.E.; Lacey, E.K.; Roche, P.; Yuan, M.; et al. Lithium deficiency and the onset of Alzheimer’s disease. Nature 2025, 1–10. [Google Scholar] [CrossRef]
- Nakamura, S. Antidepressants induce regeneration of catecholaminergic axon terminals in the rat cerebral cortex. Neurosci. Lett. 1990, 111, 64–68. [Google Scholar] [CrossRef]
- Nakamura, S. Effects of mianserin and fluoxetine on axonal re generation of brain catecholamine neurons. Neuroreport 1991, 2, 525–528. [Google Scholar] [CrossRef]
- Kitayama, I.; Yaga, T.; Kayahara, T.; Nakano, K.; Murase, S.; Otani, M.; Nomura, J. Long-Term Stress Degenerates, But Imipramine Regenerates, Noradrenergic Axons in the Rat Cerebral Cortex. Biol. Psychiatry 1997, 42, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Huang, K.; Kecojevic, A.; Welsh, A.M.; Koliatsos, V.E. Evidence that serotonin reuptake modulators increase the density of serotonin innervation in the forebrain. J. Neurochem. 2005, 96, 396–406. [Google Scholar] [CrossRef]
- Gonzalez, M.M.C.; Aston-Jones, G. Light deprivation damages monoamine neurons and produces a depressive behavioral phenotype in rats. Proc. Natl. Acad. Sci. USA 2008, 105, 4898–4903. [Google Scholar] [CrossRef]
- Zahrai, A.; Vahid-Ansari, F.; Daigle, M.; Albert, P.R. Fluoxetine-induced recovery of serotonin and norepinephrine projections in a mouse model of post-stroke depression. Transl. Psychiatry 2020, 10, 334. [Google Scholar] [CrossRef]
- Vahid-Ansari, F.; Zahrai, A.; Daigle, M.; Albert, P.R. Chronic Desipramine Reverses Deficits in Cell Activity, Norepinephrine Innervation, and Anxiety–Depression Phenotypes in Fluoxetine-Resistant cF1ko Mice. J. Neurosci. 2023, 44. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakamura, S. The Role of Calcium-Independent Phospholipase A2 in the Molecular Mechanisms of Schizophrenia. Cells 2025, 14, 1348. https://doi.org/10.3390/cells14171348
Nakamura S. The Role of Calcium-Independent Phospholipase A2 in the Molecular Mechanisms of Schizophrenia. Cells. 2025; 14(17):1348. https://doi.org/10.3390/cells14171348
Chicago/Turabian StyleNakamura, Shoji. 2025. "The Role of Calcium-Independent Phospholipase A2 in the Molecular Mechanisms of Schizophrenia" Cells 14, no. 17: 1348. https://doi.org/10.3390/cells14171348
APA StyleNakamura, S. (2025). The Role of Calcium-Independent Phospholipase A2 in the Molecular Mechanisms of Schizophrenia. Cells, 14(17), 1348. https://doi.org/10.3390/cells14171348





