Effect of High-Fat Diet on Cardiac Metabolites and Implications for Vulnerability to Ischemia and Reperfusion Injury
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
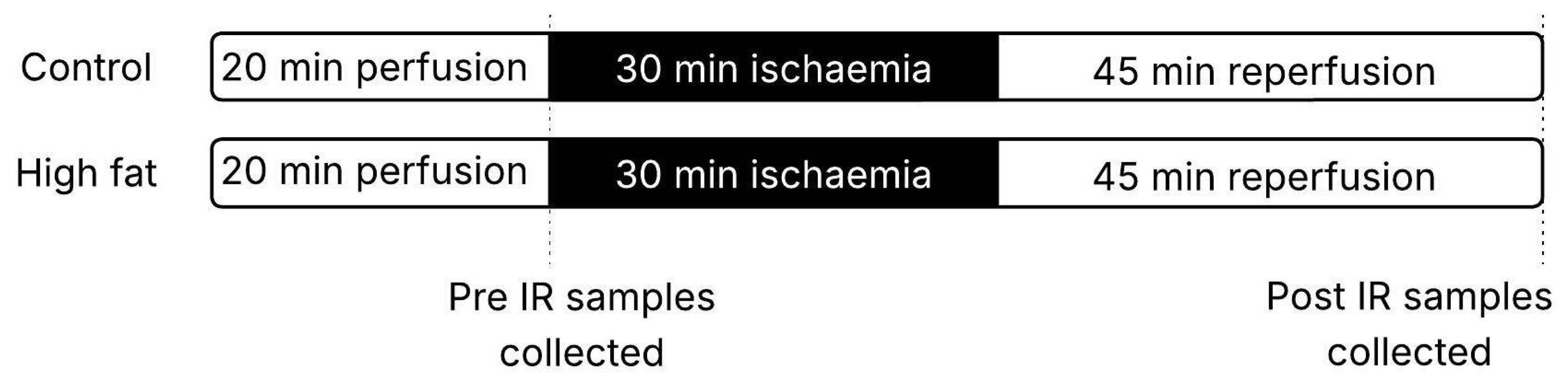
2.2. Functional Measurements of Isolated Hearts
2.3. Collection of Myocardial Tissue
2.4. Extraction and Measurement of Cardiac Metabolites
2.5. Extraction and Measurement of Cardiac Glycogen
2.6. Statistical Analysis
3. Results
3.1. The Effect of a High-Fat Diet on Body Weight and Heart Weight
3.2. Effect of High-Fat Diet on Basal Ventricular Metabolite Concentrations
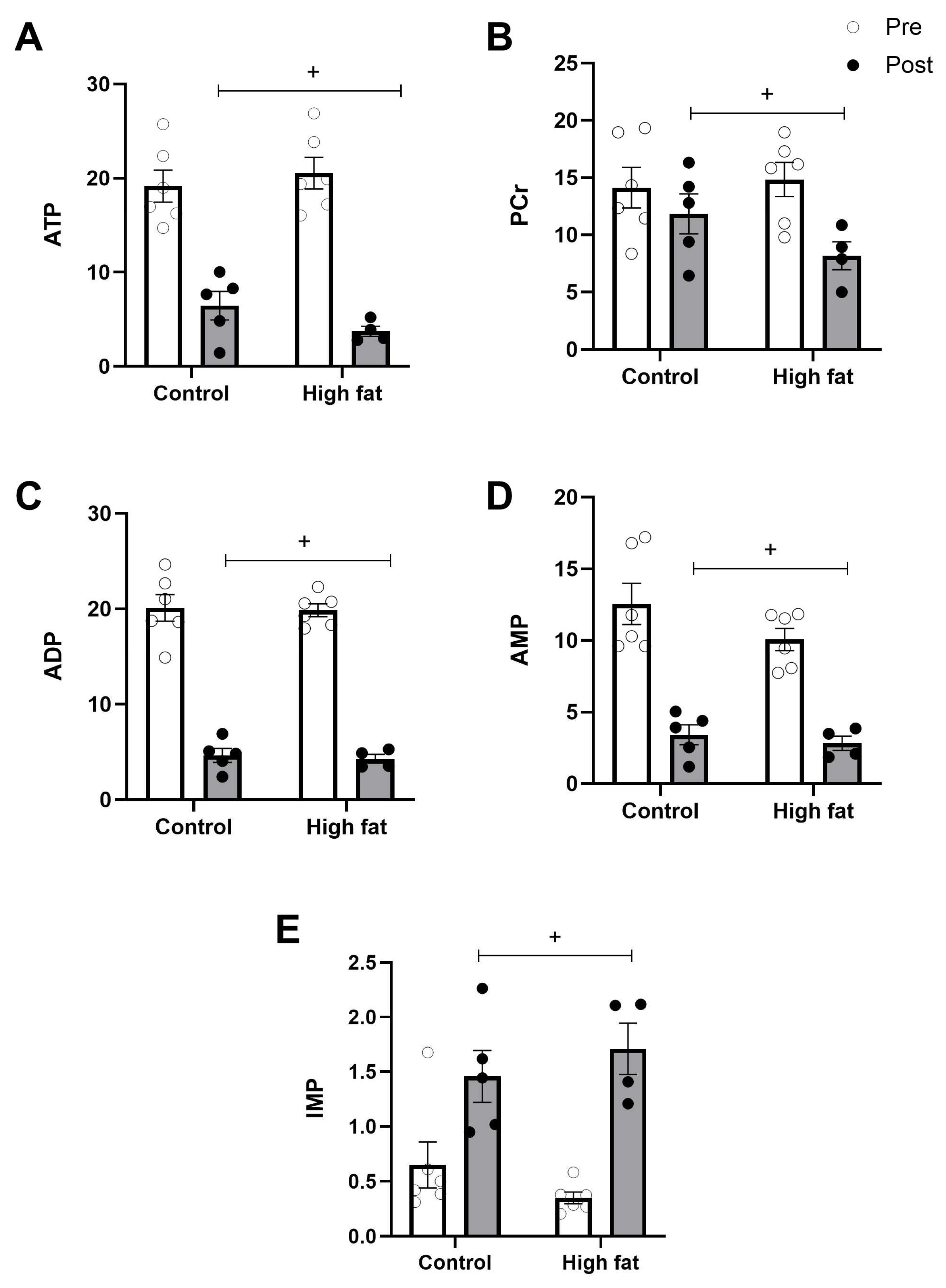
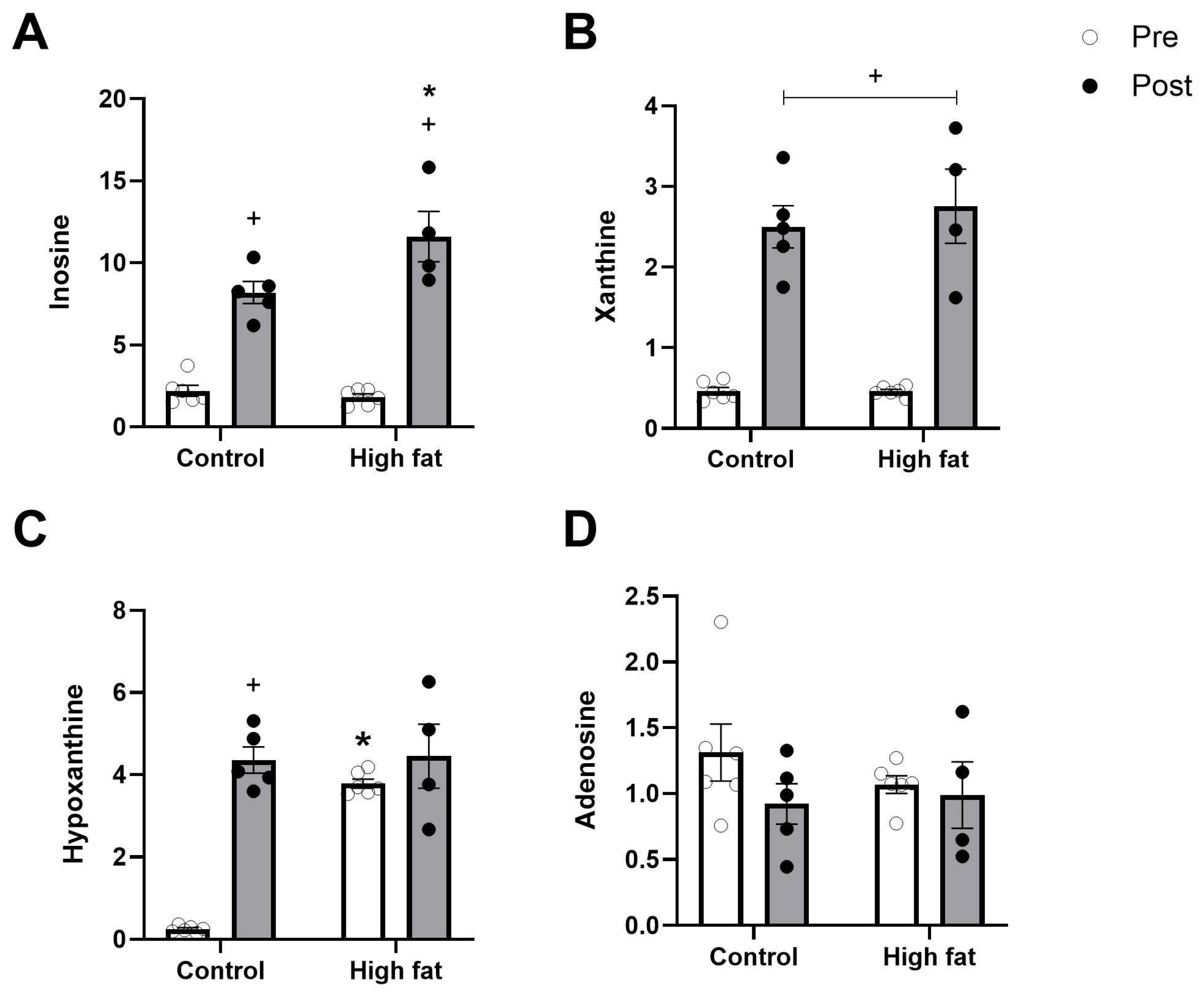
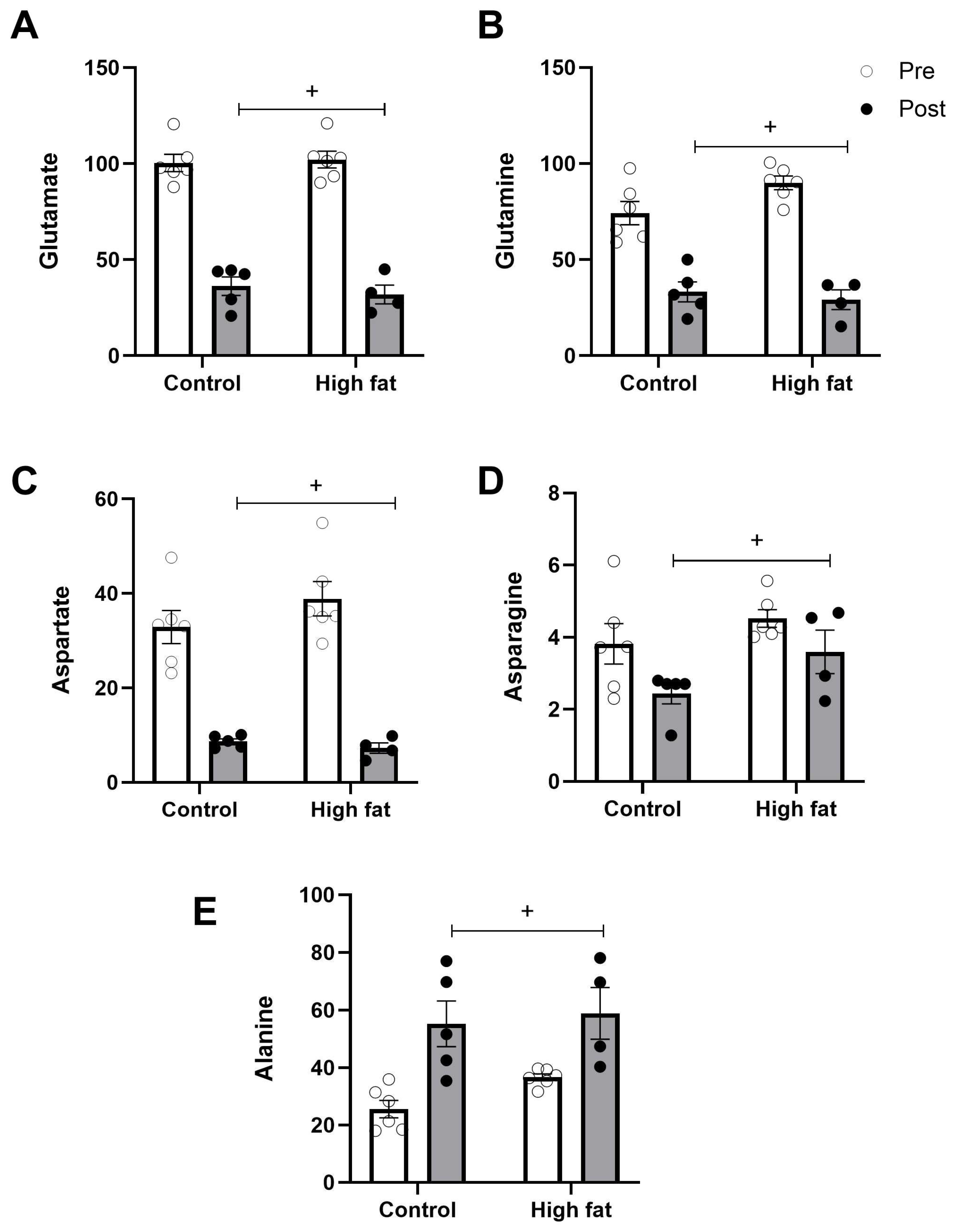
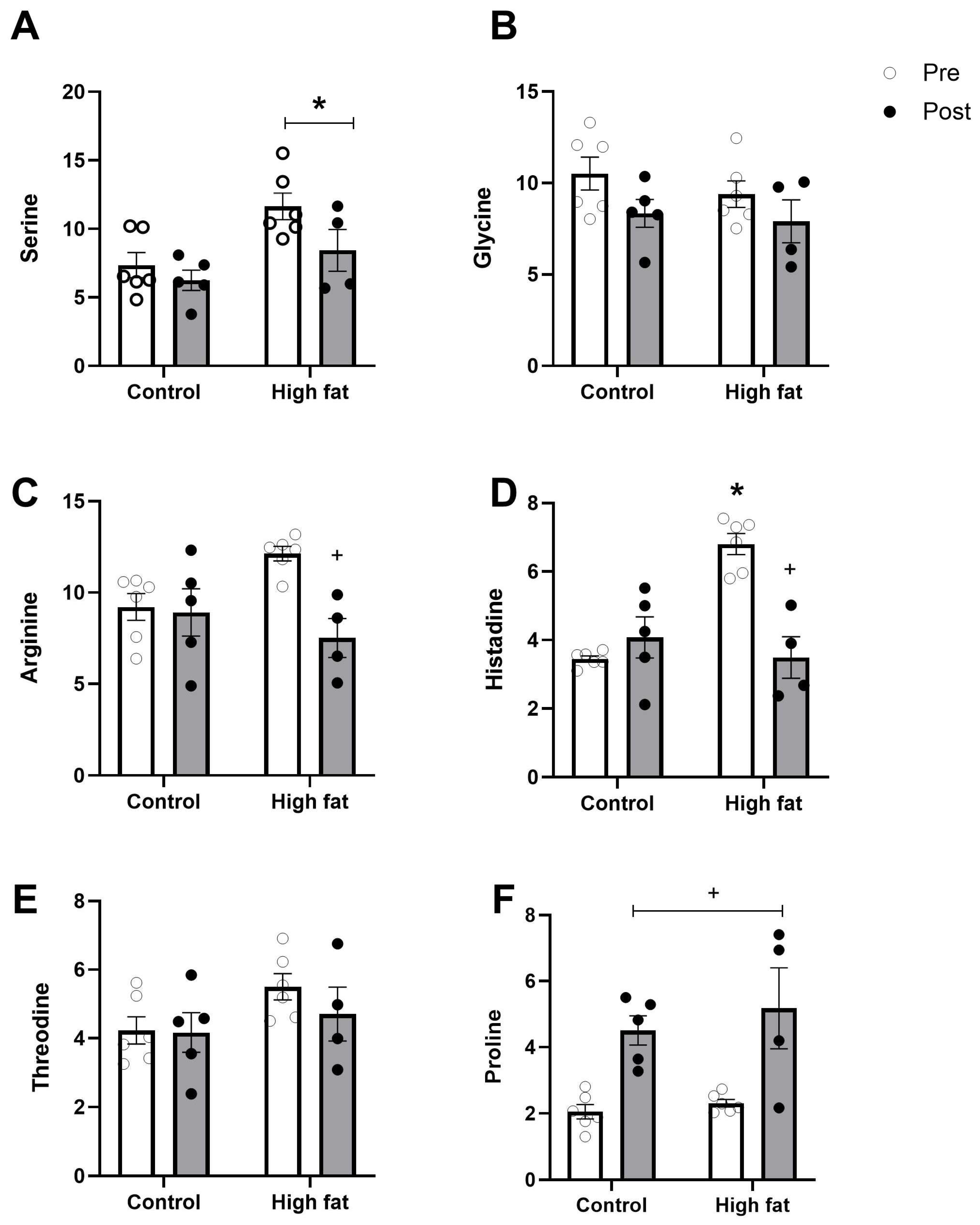
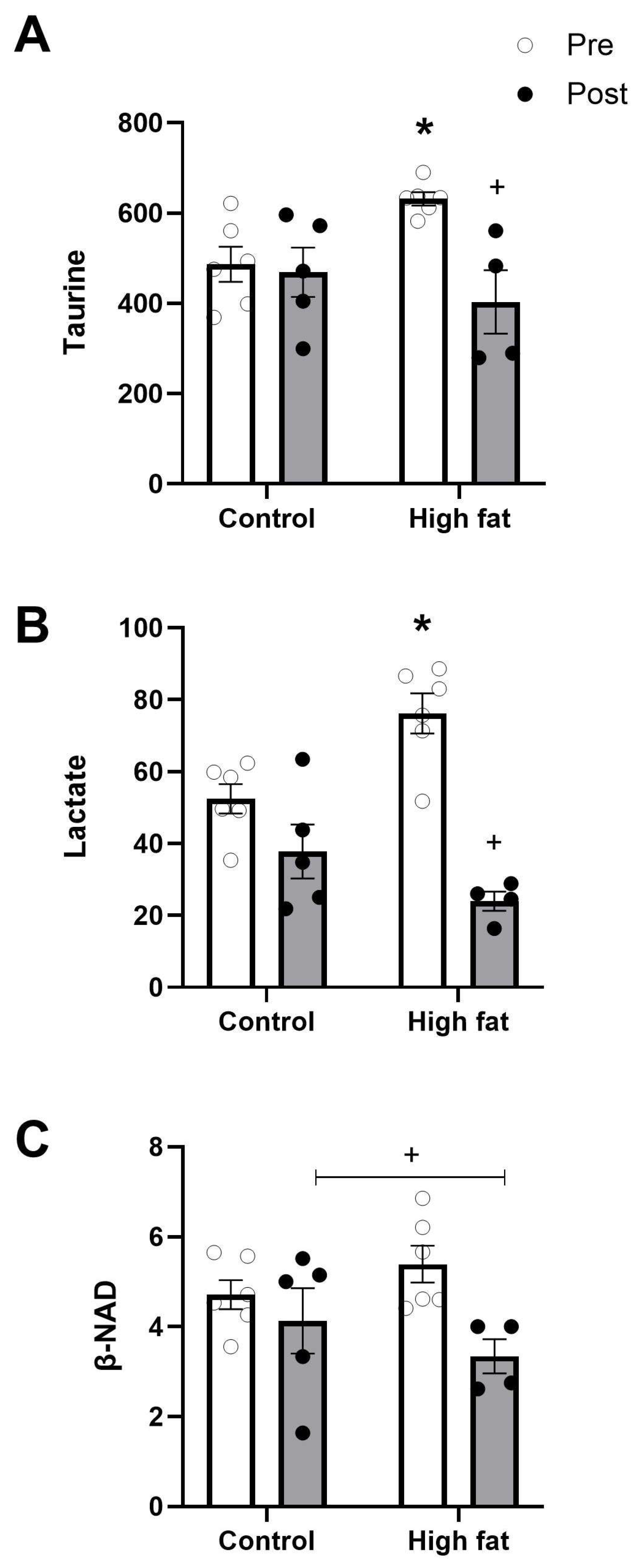
3.3. Effect of High-Fat Diet on Changes in Ventricular Metabolites During I/R
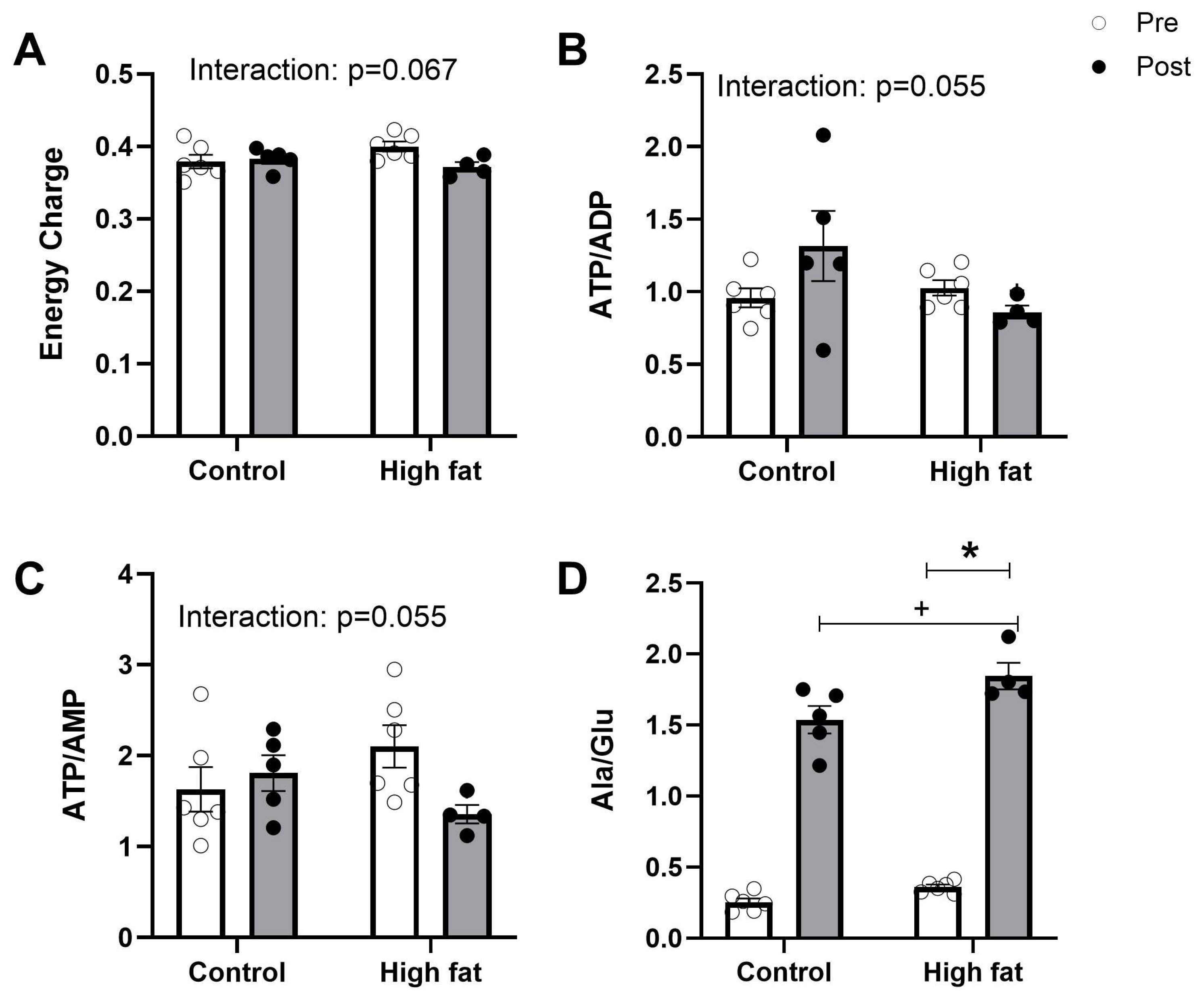
3.4. Effect of High-Fat Diet on Cardiac Ischemic/Metabolic Stress During I/R
3.5. Effect of High-Fat Diet on Cardiac Pump Function Before and During Ischemia
4. Discussion
4.1. Non-Obesogenic, High-Fat Feeding Mouse Model
4.2. Chronic Changes in Myocardial Glycogen, Lactate, and Amino Acids, but Not in Energy-Rich Phosphates Due to Feeding a Non-Obesogenic High-Fat Diet
4.3. Acute Changes in Cardiac Metabolites During I/R Are More Evident in High-Fat-Diet Hearts and Were Associated with Significant Myocardial Ischemic/Metabolic Stress
4.3.1. Cardiac Energetics and Mitochondria
4.3.2. Lactate and Alanine/Glutamate Ratio
4.3.3. Taurine and Glutamine Loss
4.4. The Changes in Metabolites Due to High-Fat Feeding Alter the Changes in Hemodynamic Parameters During Index Ischemia
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HPLC | High-performance liquid chromatography |
| I/R | Ischemic reperfusion |
References
- Calligaris, S.D.; Lecanda, M.; Solis, F.; Ezquer, M.; Gutierrez, J.; Brandan, E.; Leiva, A.; Sobrevia, L.; Conget, P. Mice long-term high-fat diet feeding recapitulates human cardiovascular alterations: An animal model to study the early phases of diabetic cardiomyopathy. PLoS ONE 2013, 8, e60931. [Google Scholar] [CrossRef]
- Noyan-Ashraf, M.H.; Shikatani, E.A.; Schuiki, I.; Mukovozov, I.; Wu, J.; Li, R.K.; Volchuk, A.; Robinson, L.A.; Billia, F.; Drucker, D.J.; et al. A glucagon-like peptide-1 analog reverses the molecular pathology and cardiac dysfunction of a mouse model of obesity. Circulation 2013, 127, 74–85. [Google Scholar] [CrossRef]
- Hallfrisch, J.; Cohen, L.; Reiser, S. Effects of feeding rats sucrose in a high fat diet. J. Nutr. 1981, 111, 531–536. [Google Scholar] [CrossRef]
- Littlejohns, B.; Pasdois, P.; Duggan, S.; Bond, A.R.; Heesom, K.; Jackson, C.L.; Angelini, G.D.; Halestrap, A.P.; Suleiman, M.S. Hearts from Mice Fed a Non-Obesogenic High-Fat Diet Exhibit Changes in Their Oxidative State, Calcium and Mitochondria in Parallel with Increased Susceptibility to Reperfusion Injury. PLoS ONE 2014, 9, e100579. [Google Scholar] [CrossRef]
- Surwit, R.S.; Kuhn, C.M.; Cochrane, C.; McCubbin, J.A.; Feinglos, M.N. Diet-induced type II diabetes in C57BL/6J mice. Diabetes 1988, 37, 1163–1167. [Google Scholar] [CrossRef]
- Toida, S.; Takahashi, M.; Shimizu, H.; Sato, N.; Shimomura, Y.; Kobayashi, I. Effect of high sucrose feeding on fat accumulation in the male Wistar rat. Obes. Res. 1996, 4, 561–568. [Google Scholar] [CrossRef]
- Gallou-Kabani, C.; Vige, A.; Gross, M.S.; Rabes, J.P.; Boileau, C.; Larue-Achagiotis, C.; Tome, D.; Jais, J.P.; Junien, C. C57BL/6J and A/J mice fed a high-fat diet delineate components of metabolic syndrome. Obesity (Silver Spring) 2007, 15, 1996–2005. [Google Scholar] [CrossRef]
- Williams, T.D.; Chambers, J.B.; Roberts, L.M.; Henderson, R.P.; Overton, J.M. Diet-induced obesity and cardiovascular regulation in C57BL/6J mice. Clin. Exp. Pharmacol. Physiol. 2003, 30, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Mills, E.; Kuhn, C.M.; Feinglos, M.N.; Surwit, R. Hypertension in CB57BL/6J mouse model of non-insulin-dependent diabetes mellitus. Am. J. Physiol. 1993, 264, R73–R78. [Google Scholar] [CrossRef] [PubMed]
- Chase, A.; Jackson, C.L.; Angelini, G.L.; Suleiman, M.S. Coronary artery disease progression is associated with increased resistance of hearts and myocytes to cardiac insults. Crit. Care Med. 2007, 35, 2344–2351. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, P.; Douglas, S.L.; Tate, J.M.; Sham, S.; Lloyd, S.G. Impact of high-fat, low-carbohydrate diet on myocardial substrate oxidation, insulin sensitivity, and cardiac function after ischemia-reperfusion. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H1–H10. [Google Scholar] [CrossRef]
- Liu, J.; Wang, P.; Zou, L.; Qu, J.; Litovsky, S.; Umeda, P.; Zhou, L.; Chatham, J.; Marsh, S.A.; Dell’Italia, L.J.; et al. High-fat, low-carbohydrate diet promotes arrhythmic death and increases myocardial ischemia-reperfusion injury in rats. Am. J. Physiol. Heart Circ. Physiol. 2014, 307, H598–H608. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lloyd, S.G. High-fat, low-carbohydrate diet alters myocardial oxidative stress and impairs recovery of cardiac function after ischemia and reperfusion in obese rats. Nutr. Res. 2013, 33, 311–321. [Google Scholar] [CrossRef]
- Panagia, M.; Gibbons, G.F.; Radda, G.K.; Clarke, K. PPAR-alpha activation required for decreased glucose uptake and increased susceptibility to injury during ischemia. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H2677–H2683. [Google Scholar] [CrossRef]
- Lloyd-Jones, D.M.; Evans, J.C.; Larson, M.G.; O’Donnell, C.J.; Wilson, P.W.; Levy, D. Cross-classification of JNC VI blood pressure stages and risk groups in the Framingham Heart Study. Arch. Intern. Med. 1999, 159, 2206–2212. [Google Scholar] [CrossRef]
- Wang, P.; Tate, J.M.; Lloyd, S.G. Low carbohydrate diet decreases myocardial insulin signaling and increases susceptibility to myocardial ischemia. Life Sci. 2008, 83, 836–844. [Google Scholar] [CrossRef]
- Imura, H.; Caputo, M.; Parry, A.; Pawade, A.; Angelini, G.; Suleiman, M. Age-dependent and hypoxia-related differences in myocardial protection during pediatric open heart surgery. Circulation 2001, 103, 1551–1556. [Google Scholar] [CrossRef]
- Di Lisa, F.; Menabo, R.; Canton, M.; Barile, M.; Bernardi, P. Opening of the mitochondrial permeability transition pore causes depletion of mitochondrial and cytosolic NAD+ and is a causative event in the death of myocytes in postischemic reperfusion of the heart. J. Biol. Chem. 2001, 276, 2571–2575. [Google Scholar] [CrossRef]
- Abdul-Ghani, S.; Fleishman, A.N.; Khaliulin, I.; Meloni, M.; Angelini, G.D.; Suleiman, M.S. Remote ischemic preconditioning triggers changes in autonomic nervous system activity: Implications for cardioprotection. Physiol. Rep. 2017, 5, e13085. [Google Scholar] [CrossRef] [PubMed]
- Paigen, B.; Morrow, A.; Holmes, P.A.; Mitchell, D.; Williams, R.A. Quantitative assessment of atherosclerotic lesions in mice. Atherosclerosis 1987, 68, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Paigen, B.; Morrow, A.; Brandon, C.; Mitchell, D.; Holmes, P. Variation in susceptibility to atherosclerosis among inbred strains of mice. Atherosclerosis 1985, 57, 65–73. [Google Scholar] [CrossRef]
- Breslow, J.L. Mouse models of atherosclerosis. Science 1996, 272, 685–688. [Google Scholar] [CrossRef]
- Wong, C.; Marwick, T.H. Alterations in myocardial characteristics associated with obesity: Detection, mechanisms, and implications. Trends Cardiovasc. Med. 2007, 17, 1–5. [Google Scholar] [CrossRef]
- Diniz, Y.S.; Cicogna, A.C.; Padovani, C.R.; Santana, L.S.; Faine, L.A.; Novelli, E.L. Diets rich in saturated and polyunsaturated fatty acids: Metabolic shifting and cardiac health. Nutrition 2004, 20, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.L.; Wambolt, R.B.; Schonekess, B.O.; Lopaschuk, G.D.; Allard, M.F. Contribution of glycogen to aerobic myocardial glucose utilization. Circulation 1996, 93, 1549–1555. [Google Scholar] [CrossRef]
- Khaliulin, I.; Parker, J.E.; Halestrap, A.P. Consecutive pharmacological activation of PKA and PKC mimics the potent cardioprotection of temperature preconditioning. Cardiovasc. Res. 2010, 88, 324–333. [Google Scholar] [CrossRef]
- Pasdois, P.; Parker, J.E.; Halestrap, A.P. Extent of mitochondrial hexokinase II dissociation during ischemia correlates with mitochondrial cytochrome c release, reactive oxygen species production, and infarct size on reperfusion. J. Am. Heart Assoc. 2013, 2, e005645. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.; Szobi, A.; Balaska, D.; Khaliulin, I.; Adameova, A.; Griffiths, E.; Orchard, C.H.; Suleiman, M.S. Consecutive Isoproterenol and Adenosine Treatment Confers Marked Protection against Reperfusion Injury in Adult but Not in Immature Heart: A Role for Glycogen. Int. J. Mol. Sci. 2018, 19, 494. [Google Scholar] [CrossRef]
- Lopez-Schenk, R.; Collins, N.L.; Schenk, N.A.; Beard, D.A. Integrated Functions of Cardiac Energetics, Mechanics, and Purine Nucleotide Metabolism. Compr. Physiol. 2023, 14, 5345–5369. [Google Scholar] [CrossRef] [PubMed]
- Skeffington, K.L.; Moscarelli, M.; Abdul-Ghani, S.; Fiorentino, F.; Emanueli, C.; Reeves, B.C.; Punjabi, P.P.; Angelini, G.D.; Suleiman, M.S. Pathology-related changes in cardiac energy metabolites, inflammatory response and reperfusion injury following cardioplegic arrest in patients undergoing open-heart surgery. Front. Cardiovasc. Med. 2022, 9, 911557. [Google Scholar] [CrossRef]
- Suleiman, M.S.; Moffatt, A.C.; Dihmis, W.C.; Caputo, M.; Hutter, J.A.; Angelini, G.D.; Bryan, A.J. Effect of ischaemia and reperfusion on the intracellular concentration of taurine and glutamine in the hearts of patients undergoing coronary artery surgery. Biochim. Biophys. Acta 1997, 1324, 223–231. [Google Scholar] [CrossRef]
- Baliou, S.; Adamaki, M.; Ioannou, P.; Pappa, A.; Panayiotidis, M.I.; Spandidos, D.A.; Christodoulou, I.; Kyriakopoulos, A.M.; Zoumpourlis, V. Protective role of taurine against oxidative stress (Review). Mol. Med. Rep. 2021, 24, 1–19. [Google Scholar] [CrossRef]
- Ramila, K.C.; Jong, C.J.; Pastukh, V.; Ito, T.; Azuma, J.; Schaffer, S.W. Role of protein phosphorylation in excitation-contraction coupling in taurine deficient hearts. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H232–H239. [Google Scholar] [CrossRef]
- Stanley, W.C.; Lopaschuk, G.D.; Hall, J.L.; McCormack, J.G. Regulation of myocardial carbohydrate metabolism under normal and ischaemic conditions. Potential for pharmacological interventions. Cardiovasc. Res. 1997, 33, 243–257. [Google Scholar] [CrossRef]
- Lopaschuk, G.D. Metabolic abnormalities in the diabetic heart. Heart Fail. Rev. 2002, 7, 149–159. [Google Scholar] [CrossRef]
- Pepe, S.; McLennan, P.L. Cardiac membrane fatty acid composition modulates myocardial oxygen consumption and postischemic recovery of contractile function. Circulation 2002, 105, 2303–2308. [Google Scholar] [CrossRef]
- Harper, C.R.; Jacobson, T.A. The fats of life: The role of omega-3 fatty acids in the prevention of coronary heart disease. Arch. Intern. Med. 2001, 161, 2185–2192. [Google Scholar] [CrossRef] [PubMed]
- Adameova, A.; Kuzelova, M.; Faberova, V.; Svec, P. Protective effect of simvastatin and VULM 1457 in ischaemic-reperfused myocardium of the diabetic-hypercholesterolemic rats. Pharmazie 2006, 61, 807–808. [Google Scholar]
- Dong, S.; Qian, L.; Cheng, Z.; Chen, C.; Wang, K.; Hu, S.; Zhang, X.; Wu, T. Lactate and Myocardiac Energy Metabolism. Front. Physiol. 2021, 12, 715081. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, S.W.; Solodushko, V.; Kakhniashvili, D. Beneficial effect of taurine depletion on osmotic sodium and calcium loading during chemical hypoxia. Am. J. Physiol. Cell Physiol. 2002, 282, C1113–C1120. [Google Scholar] [CrossRef] [PubMed]
- Herzog, N.; Laager, R.; Thommen, E.; Widmer, M.; Vincent, A.M.; Keller, A.; Becker, C.; Beck, K.; Perrig, S.; Bernasconi, L.; et al. Association of Taurine with In-Hospital Mortality in Patients after Out-of-Hospital Cardiac Arrest: Results from the Prospective, Observational COMMUNICATE Study. J. Clin. Med. 2020, 9, 1405. [Google Scholar] [CrossRef] [PubMed]
- Justice, C.N.; Zhu, X.; Li, J.; O’Donnell, J.M.; Vanden Hoek, T.L. Intra-ischemic hypothermia cardioprotection involves modulation of PTEN/Akt/ERK signaling and fatty acid oxidation. Physiol. Rep. 2023, 11, e15611. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, S.W.; Jong, C.J.; Ito, T.; Azuma, J. Effect of taurine on ischemia-reperfusion injury. Amino Acids 2014, 46, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Rennie, M.J.; Tadros, L.; Khogali, S.; Ahmed, A.; Taylor, P.M. Glutamine transport and its metabolic effects. J. Nutr. 1994, 124, 1503S–1508S. [Google Scholar] [CrossRef]
- Howarth, F.C.; Qureshi, M.A.; Gbewonyo, A.J.; Tariq, S.; Adeghate, E. The progressive effects of a fat enriched diet on ventricular myocyte contraction and intracellular Ca2+ in the C57BL/6J mouse. Mol. Cell. Biochem. 2005, 273, 87–95. [Google Scholar] [CrossRef]
| Control Diet | High-Fat Diet | |
|---|---|---|
| (n = 10) | (n = 11) | |
| Body weight (g) | 35.7 ± 1.2 | 42.2 ± 1.2 * |
| Heart | ||
| Dry heart weight (mg) | 39 ± 2 | 35 ± 1 |
| Wet heart weight (mg) | 209 ± 13 | 176 ± 8 * |
| % water content | 81.1 ± 0.2 | 79.8 ± 0.4 * |
| Wet heart weight/body weight (%) | 0.58 ± 0.02 | 0.42 ± 0.02 * |
| Cardiac protein content (% wet weight) | 10.1 ± 0.7 | 10.3 ± 0.9 |
| Control Diet (n = 6) | High-Fat Diet (n = 6) | ||
|---|---|---|---|
| Energy-rich and related metabolites | Phosphocreatine | 14.1 ± 1.8 | 14.9 ± 1.5 |
| ATP | 19.2 ± 1.7 | 20.5 ± 1.7 | |
| ADP | 20.1 ± 1.4 | 19.9 ± 0.7 | |
| AMP | 12.5 ± 1.4 | 10.1 ± 0.8 | |
| IMP | 0.65 ± 0.21 | 0.35 ± 0.05 | |
| Inosine | 2.2 ± 0.3 | 1.8 ± 0.2 | |
| Xanthine | 0.46 ± 0.05 | 0.46 ± 0.02 | |
| Hypoxanthine | 0.25 ± 0.03 | 3.78 ± 0.11 * | |
| Adenosine | 1.3 ± 0.2 | 1.1 ± 0.1 | |
| B-NAD | 4.7 ± 0.3 | 5.4 ± 0.4 | |
| Protein amino acids | Aspartate | 32.9 ± 3.5 | 38.9 ± 3.6 |
| Glutamate | 100.4 ± 4.5 | 102.2 ± 4.4 | |
| Serine | 7.4 ± 0.9 | 11.6 ± 1.0 * | |
| Asparagine | 3.8 ± 0.6 | 4.5 ± 0.2 | |
| Glutamine | 74.2 ± 6.1 | 90.0 ± 3.5 * | |
| Glycine | 10.5 ± 0.9 | 9.4 ± 0.7 | |
| Histidine | 3.4 ± 0.1 | 6.8 ± 0.3 * | |
| Arginine | 9.2 ± 0.7 | 12.1 ± 0.4 * | |
| Threonine | 4.2 ± 0.4 | 5.5 ± 0.4 * | |
| Alanine | 25.6 ± 3.0 | 36.7 ± 1.2 * | |
| Proline | 2.1 ± 0.2 | 2.3 ± 0.1 | |
| Non-protein amino acid | Taurine | 487 ± 39 | 632 ± 15 * |
| Other metabolites | Lactate | 52.5 ± 4.1 | 76.2 ± 5.6 * |
| Glycogen (µmol glycosyl units/g wet weight) | 126 ± 7 | 87 ± 12 * |
| Parameter/Activity | Control Diet (n = 16) | High-Fat Diet (n = 13) | |
|---|---|---|---|
| Haemodynamic parameters pre-ischemia | LVDP (mmHg) | 31.5 ± 3.2 | 31.8 ± 3.6 |
| Heart rate (bpm) | 394 ± 20 | 417 ± 19 | |
| Changes during ischemia | Time to arrest (min) | 3.5 ± 0.3 | 3.8 ± 0.3 |
| Time to onset of rigor contracture (min) | 15.5 ± 1.0 | 13.0 ± 0.8 (p = 0.050) | |
| Time to max ischemic contracture (mins) | 22.1 ± 0.8 | 17.5 ± 0.9 * | |
| Maximum end diastolic pressure (mmHg) | 42.0 ± 4.1 | 45.7 ± 5.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hawi, J.S.; Skeffington, K.L.; Young, M.; Caputo, M.; Ascione, R.; Suleiman, M.-S. Effect of High-Fat Diet on Cardiac Metabolites and Implications for Vulnerability to Ischemia and Reperfusion Injury. Cells 2025, 14, 1329. https://doi.org/10.3390/cells14171329
Hawi JS, Skeffington KL, Young M, Caputo M, Ascione R, Suleiman M-S. Effect of High-Fat Diet on Cardiac Metabolites and Implications for Vulnerability to Ischemia and Reperfusion Injury. Cells. 2025; 14(17):1329. https://doi.org/10.3390/cells14171329
Chicago/Turabian StyleHawi, Jihad S., Katie L. Skeffington, Megan Young, Massimo Caputo, Raimondo Ascione, and M-Saadeh Suleiman. 2025. "Effect of High-Fat Diet on Cardiac Metabolites and Implications for Vulnerability to Ischemia and Reperfusion Injury" Cells 14, no. 17: 1329. https://doi.org/10.3390/cells14171329
APA StyleHawi, J. S., Skeffington, K. L., Young, M., Caputo, M., Ascione, R., & Suleiman, M.-S. (2025). Effect of High-Fat Diet on Cardiac Metabolites and Implications for Vulnerability to Ischemia and Reperfusion Injury. Cells, 14(17), 1329. https://doi.org/10.3390/cells14171329








