Evidence for Epistatic Interaction between HLA-G and LILRB1 in the Pathogenesis of Nonsegmental Vitiligo
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Laboratory Analysis
2.3. Bioinformatics Analysis
2.4. Statistical Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ezzedine, K.; Eleftheriadou, V.; Whitton, M.; Van Geel, N. Vitiligo. Lancet 2015, 386, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Ezzedine, K.; H.W., L.; Suzuki, T.; Katayama, I.; Hamzavi, I.; Lan, C.C.E.; Goh, B.K.; Anbar, T.; Silva de Castro, C.; Lee, A.Y.; et al. Revised Classification/Nomenclature of Vitiligo and Related Issues: The Vitiligo Global Issues Consensus Confrence. Pigment Cell Melanoma Res. 2011, 25, E1–E13. [Google Scholar] [CrossRef] [PubMed]
- Ezzedine, K.; Silverberg, N. A Practical Approach to the Diagnosis and Treatment of Vitiligo in Children. Pediatrics 2016, 138, e2015412. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cai, Y.; Shi, M.; Jiang, S.; Cui, S.; Wu, Y.; Gao, X.H.; Chen, H.D. The Prevalence of Vitiligo: A Meta-Analysis. PLoS ONE 2016, 11, e0163806. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Ezzedine, K.; Hamzavi, I.; Pandya, A.G.; Harris, J.E. New Discoveries in the Pathogenesis and Classification of Vitiligo. J. Am. Acad. Dermatol. 2017, 77, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Spritz, R.A.; Andersen, G.H.L. Genetics of Vitiligo. Dermatol. Clin. 2017, 35, 245–255. [Google Scholar] [CrossRef]
- Ongenae, K.; Van Geel, N.; Naeyaert, J.M. Evidence for an Autoimmune Pathogenesis of Vitiligo. Pigment. Cell Res. 2003, 16, 90–100. [Google Scholar] [CrossRef]
- Zhang, X.J.; Liu, J.B.; Gui, J.P.; Li, M.; Xiong, Q.G.; Wu, H.B.; Li, J.X.; Yang, S.; Wang, H.Y.; Gao, M.; et al. Characteristics of Genetic Epidemiology and Genetic Models for Vitiligo. J. Am. Acad. Dermatol. 2004, 51, 383–390. [Google Scholar] [CrossRef]
- Picardo, M.; Dell’Anna, M.L.; Ezzedine, K.; Hamzavi, I.; Harris, J.E.; Parsad, D.; Taieb, A. Vitiligo. Nat. Rev. Dis. Prim. 2015, 1, 1–16. [Google Scholar] [CrossRef]
- Manolio, T.A.; Collins, F.S.; Cox, N.J.; Goldstein, D.B.; Hindorff, L.A.; Hunter, D.J.; McCarthy, M.I.; Ramos, E.M.; Cardon, L.R.; Chakravarti, A.; et al. Finding the Missing Heritability of Complex Diseases. Nature 2009, 461, 747–753. [Google Scholar] [CrossRef]
- Zuk, O.; Hechter, E.; Sunyaev, S.R.; Lander, E.S. The Mystery of Missing Heritability: Genetic Interactions Create Phantom Heritability. Proc. Natl. Acad. Sci. USA 2012, 109, 1193–1198. [Google Scholar] [CrossRef] [PubMed]
- Hemani, G.; Knott, S.; Haley, C. An Evolutionary Perspective on Epistasis and the Missing Heritability. PLoS Genet. 2013, 9, e1003295. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.B.; Ehrenreich, I.M. Higher-Order Genetic Interactions and Their Contribution to Complex Traits. Trends Genet. 2015, 31, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Colak, R.; Kim, T.; Kazan, H.; Oh, Y.; Cruz, M.; Valladares-Salgado, A.; Peralta, J.; Escobedo, J.; Parra, E.J.; Kim, P.M.; et al. JBASE: Joint Bayesian Analysis of Subphenotypes and Epistasis. Bioinformatics 2016, 32, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.R.; Miller, K.M.; Maas, N.L.; Roguev, A.; Fillingham, J.; Chu, C.S.; Schuldiner, M.; Gebbia, M.; Recht, J.; Shales, M.; et al. Functional Dissection of Protein Complexes Involved in Yeast Chromosome Biology Using a Genetic Interaction Map. Nature 2007, 446, 806–810. [Google Scholar] [CrossRef]
- Shiroishi, M.; Tsumoto, K.; Amano, K.; Shirakihara, Y.; Colonna, M.; Braud, V.M.; Allan, D.S.J.; Makadzange, A.; Rowland-Jones, S.; Willcox, B.; et al. Human Inhibitory Receptors Ig-like Transcript 2 (ILT2) and ILT4 Compete with CD8 for MHC Class I Binding and Bind Preferentially to HLA-G. Proc. Natl. Acad. Sci. USA 2003, 100, 8856–8861. [Google Scholar] [CrossRef]
- Brown, D.; Trowsdale, J.; Allen, R. The LILR Family: Modulators of Innate and Adaptive Immune Pathways in Health and Disease. Tissue Antigens 2004, 64, 215–225. [Google Scholar] [CrossRef]
- Carosella, E.D.; Favier, B.; Rouas-Freiss, N.; Moreau, P.; Lemaoult, J. Beyond the Increasing Complexity of the Immunomodulatory HLA-G Molecule. Blood 2008, 111, 4862–4870. [Google Scholar] [CrossRef]
- Veiga-Castelli, L.; Oliveira, M.L.d.; Pereira, A.; Debortoli, G.; Marcorin, L.; Fracasso, N.; Silva, G.; Souza, A.; Massaro, L.; Simões, G.; et al. HLA-G Polymorphisms Are Associated with Non-Segmental Vitiligo among Brazilians. Biomolecules 2019, 9, 463. [Google Scholar] [CrossRef]
- Fraser, H.B.; Hirsh, A.E.; Wall, D.P.; Eisen, M.B. Coevolution of Gene Expression among Interacting Proteins. Proc. Natl. Acad. Sci. USA 2004, 101, 9033–9038. [Google Scholar] [CrossRef]
- Miller, S.A.; Dykes, D.D.; Polesky, H.F. A Simple Salting out Procedure for Extracting DNA from Human Nucleated Cells. Nucleic Acids Res. 1988, 16, 1215. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.; Salas, A.; Sánchez, J.J.; Fondevila, M.; Gómez-Tato, A.; Álvarez-Dios, J.; Calaza, M.; de Cal, M.C.; Ballard, D.; Lareu, M.V.; et al. Inferring Ancestral Origin Using a Single Multiplex Assay of Ancestry-Informative Marker SNPs. Forensic Sci. Int. Genet. 2007, 1, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Castelli, E.C.; Gerasimou, P.; Paz, M.A.; Ramalho, J.; Porto, I.O.P.; Lima, T.H.A.; Souza, A.S.; Veiga-Castelli, L.C.; Collares, C.V.A.; Donadi, E.A.; et al. HLA-G Variability and Haplotypes Detected by Massively Parallel Sequencing Procedures in the Geographicaly Distinct Population Samples of Brazil and Cyprus. Mol. Immunol. 2017, 83, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.L.G.; Castelli, E.C.; Veiga-Castelli, L.C.; Pereira, A.E.; Marcorin, L.; Carratto, T.M.T.; Souza, A.S.; Andrade, H.S.; Simões, A.L.; Donadi, E.A.; et al. Genetic Diversity of the LILRB1 and LILRB2 Coding Regions in an Admixed Brazilian Population Sample. HLA 2022, 100, 325–348. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt Removes Adapter Sequences from High-Throughput Sequencing Reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Castelli, E.C.; Paz, M.A.; Souza, A.S.; Ramalho, J.; Mendes-Junior, C.T. Hla-Mapper: An Application to Optimize the Mapping of HLA Sequences Produced by Massively Parallel Sequencing Procedures. Hum. Immunol. 2018, 79, 678–684. [Google Scholar] [CrossRef]
- McKenna, A.; Hanna, M.; Banks, E.; Sivachenko, A.; Cibulskis, K.; Kernytsky, A.; Garimella, K.; Altshuler, D.; Gabriel, S.; Daly, M.; et al. The Genome Analysis Toolkit: A MapReduce Framework for Analyzing next-Generation DNA Sequencing Data. Genome Res. 2010, 20, 1297–1303. [Google Scholar] [CrossRef]
- Stephens, M.; Smith, N.J.; Donnelly, P. A New Statistical Method for Haplotype Reconstruction from Population Data. Am. J. Hum. Genet. 2001, 68, 978–989. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and Accurate Long-Read Alignment with Burrows-Wheeler Transform. Bioinformatics 2010, 26, 589–595. [Google Scholar] [CrossRef]
- Oliveira, M.L.G.d.; Veiga-Castelli, L.C.; Marcorin, L.; Debortoli, G.; Pereira, A.L.E.; Fracasso, N.C.d.A.; Silva, G.d.V.; Souza, A.S.; Massaro, J.D.; Simões, A.L.; et al. Extended HLA-G Genetic Diversity and Ancestry Composition in a Brazilian Admixed Population Sample: Implications for HLA-G Transcriptional Control and for Case-Control Association Studies. Hum. Immunol. 2018, 79, 790–799. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Rosenberg, N.A.; Donnelly, P. Association Mapping in Structured Populations. Am. J. Hum. Genet. 2000, 67, 170–181. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; De Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and Visualization of LD and Haplotype Maps. Bioinformatics 2005, 21, 263–265. [Google Scholar] [CrossRef]
- Cardon, L.R.; Palmer, L.J. Population Stratification and Spurious Allelic Association. Lancet 2003, 361, 598–604. [Google Scholar] [CrossRef]
- Serre, D.; Montpetit, A.; Paré, G.; Engert, J.C.; Yusuf, S.; Keavney, B.; Hudson, T.J.; Anand, S. Correction of Population Stratification in Large Multi-Ethnic Association Studies. PLoS ONE 2008, 3, e1382. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.D.; Hahn, L.W.; Roodi, N.; Bailey, L.R.; Dupont, W.D.; Parl, F.F.; Moore, J.H. Multifactor-Dimensionality Reduction Reveals High-Order Interactions among Estrogen-Metabolism Genes in Sporadic Breast Cancer. Am. J. Hum. Genet. 2001, 69, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.S.; Lucas, A.; Zhang, X.; Veturi, Y.; Dudek, S.; Li, B.; Li, R.; Urbanowicz, R.; Moore, J.H.; Kim, D.; et al. Collective Feature Selection to Identify Crucial Epistatic Variants. BioData Min. 2018, 11, 1–22. [Google Scholar] [CrossRef]
- Ritchie, M.D.; Hahn, L.W.; Moore, J.H. Power of Multifactor Dimensionality Reduction for Detecting Gene-Gene Interactions in the Presence of Genotyping Error, Missing Data, Phenocopy, and Genetic Heterogeneity. Genet. Epidemiol. 2003, 24, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Hahn, L.W.; Ritchie, M.D.; Moore, J.H. Multifactor Dimensionality Reduction Software for Detecting Gene-Gene and Gene-Environment Interactions. Bioinformatics 2003, 19, 376–382. [Google Scholar] [CrossRef]
- Lou, X.Y.; Chen, G.B.; Yan, L.; Ma, J.Z.; Zhu, J.; Elston, R.C.; Li, M.D. A Generalized Combinatorial Approach for Detecting Gene-by-Gene and Gene-by-Environment Interactions with Application to Nicotine Dependence. Am. J. Hum. Genet. 2007, 80, 1125–1137. [Google Scholar] [CrossRef]
- Calle, M.L.; Urrea, V.; Malats, N.; van Steen, K. Mbmdr: An R Package for Exploring Gene-Gene Interactions Associated with Binary or Quantitative Traits. Bioinformatics 2010, 26, 2198–2199. [Google Scholar] [CrossRef] [PubMed]
- Roberts, G.H.L.; Santorico, S.A.; Spritz, R.A. The Genetic Architecture of Vitiligo. Pigment Cell Melanoma Res. 2019, 33, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Iannella, G.; Greco, A.; Didona, D.; Didona, B.; Granata, G.; Manno, A.; Pasquariello, B.; Magliulo, G. Vitiligo: Pathogenesis, Clinical Variants and Treatment Approaches. Autoimmun. Rev. 2016, 15, 335–343. [Google Scholar] [CrossRef]
- Lepe, V.; Moncada, B.; Castanedo-Cazares, J.P.; Torres-Alvarez, M.B.; Ortiz, C.A.; Torres-Rubalcava, A.B. A Double-Blind Randomized Trial of 0.1% Tacrolimus vs 0.05% Clobetasol for the Treatment of Childhood Vitiligo. Arch. Dermatol. 2003, 139, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Speeckaert, R.; van Geel, N. Vitiligo: An Update on Pathophysiology and Treatment Options. Am. J. Clin. Dermatol. 2017, 18, 733–744. [Google Scholar] [CrossRef]
- Li, Z.; Ren, J.; Niu, X.; Xu, Q.; Wang, X.; Liu, Y.; Xiao, S. Meta-Analysis of the Association between Vitiligo and Human Leukocyte Antigen-A. Biomed Res. Int. 2016, 2016, 1–13. [Google Scholar] [CrossRef]
- Kim, S.K.; Hong, M.S.; Shin, M.K.; Uhm, Y.K.; Chung, J.H.; Lee, M.H. Promoter Polymorphisms of the HLA-G Gene, but Not the HLA-E and HLA-F Genes, Is Associated with Non-Segmental Vitiligo Patients in the Korean Population. Arch. Dermatol. Res. 2011, 303, 679–684. [Google Scholar] [CrossRef]
- Jeong, K.H.; Kim, S.K.; Kang, B.K.; Chung, J.H.; Shin, M.K.; Lee, M.H. Association between an HLA-G 14 Bp Insertion/Deletion Polymorphism and Non-Segmental Vitiligo in the Korean Population. Arch. Dermatol. Res. 2014, 306, 577–582. [Google Scholar] [CrossRef]
- Rizzo, R.; Hviid, T.V.F.; Govoni, M.; Padovan, M.; Rubini, M.; Melchiorri, L.; Stignani, M.; Carturan, S.; Grappa, M.T.; Fotinidi, M.; et al. HLA-G Genotype and HLA-G Expression in Systemic Lupus Erythematosus: HLA-G as a Putative Susceptibility Gene in Systemic Lupus Erythematosus. Tissue Antigens 2008, 71, 520–529. [Google Scholar] [CrossRef]
- Veit, T.D.; Cordero, E.A.A.; Mucenic, T.; Monticielo, O.A.; Brenol, J.C.T.; Xavier, R.M.; Delgado-Canedo, A.; Chies, J.A.B. Association of the HLA-G Gene +3142C>G Polymorphism with Systemic Lupus Erythematosus. Lupus 2009, 18, 424–430. [Google Scholar] [CrossRef]
- Glas, J.; Töök, H.P.; Tonenchi, L.; Wetzke, M.; Beynon, V.; Teshome, M.Y.; Cotofana, S.; Schiemann, U.; Griga, T.; Klein, W.; et al. The 14-Bp Deletion Polymorphism in the HLA-G Gene Displays Significant Differences between Ulcerative Colitis and Crohn’s Disease and Is Associated with Ileocecal Resection in Crohn’s Disease. Int. Immunol. 2007, 19, 621–626. [Google Scholar] [CrossRef]
- Zidi, I.; Yahia, H.B.; Bortolotti, D.; Mouelhi, L.; Laaribi, A.B.; Ayadi, S.; Zidi, N.; Houissa, F.; Debbech, R.; Boudabous, A.; et al. Association between SHLA-G and HLA-G 14-Bp Deletion/Insertion Polymorphism in Crohn’s Disease. Int. Immunol. 2015, 27, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Kroner, A.; Grimm, A.; Johannssen, K.; Mäurer, M.; Wiendl, H. The Genetic Influence of the Nonclassical MHC Molecule HLA-G on Multiple Sclerosis. Hum. Immunol. 2007, 68, 422–425. [Google Scholar] [CrossRef] [PubMed]
- Fredj, N.B.; Sakly, K.; Bortolotti, D.; Aissi, M.; Frih-Ayed, M.; Rotola, A.; Caselli, E.; Cura, F.; Sakly, N.; Aouni, M.; et al. The Association between Functional HLA-G 14 Bp Insertion/Deletion and +3142 C > G Polymorphisms and Susceptibility to Multiple Sclerosis. Immunol. Lett. 2016, 180, 24–30. [Google Scholar] [CrossRef]
- Rizzo, R.; Rubini, M.; Govoni, M.; Padovan, M.; Melchiorri, L.; Stignani, M.; Carturan, S.; Ferretti, S.; Trotta, F.; Baricordi, O.R. HLA-G 14-Bp Polymorphism Regulates the Methotrexate Response in Rheumatoid Arthritis. Pharm. Genom. 2006, 16, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Veit, T.D.; Vianna, P.; Scheibel, I.; Brenol, C.; Brenol, J.C.T.; Xavier, R.M.; Delgado-Cañedo, A.; Gutierrez, J.E.; Brandalize, A.P.C.; Schuler-Faccini, L.; et al. Association of the HLA-G 14-Bp Insertion/Deletion Polymorphism with Juvenile Idiopathic Arthritis and Rheumatoid Arthritis. Tissue Antigens 2008, 71, 440–446. [Google Scholar] [CrossRef]
- Gazit, E.; Slomov, Y.; Goldberg, I.; Brenner, S.; Loewenthal, R. HLA-G Is Associated with Pemphigus Vulgaris in Jewish Patients. Hum. Immunol. 2004, 65, 39–46. [Google Scholar] [CrossRef]
- Robert, C.; Kupper, T.S. Inflammatory Skin Diseases, T Cells, and Immune Surveillance. N. Engl. J. Med. 1999, 341, 1817–1828. [Google Scholar] [CrossRef]
- Urosevic, M.; Kempf, W.; Zagrodnik, B.; Panizzon, R.; Burg, G.; Dummer, R. HLA-G Expression in Basal Cell Carcinomas of the Skin Recurring after Radiotherapy. Clin. Exp. Dermatol. 2005, 30, 422–425. [Google Scholar] [CrossRef]
- Shiroishi, M.; Kuroki, K.; Rasubala, L.; Tsumoto, K.; Kumagai, I.; Kurimoto, E.; Kato, K.; Kohda, D.; Maenaka, K. Structural Basis for Recognition of the Nonclassical MHC Molecule HLA-G by the Leukocyte Ig-like Receptor B2 (LILRB2/LIR2/ILT4/CD85d). Proc. Natl. Acad. Sci. USA 2006, 103, 16412–16417. [Google Scholar] [CrossRef]
- Jones, D.C.; Kosmoliaptsis, V.; Apps, R.; Lapaque, N.; Smith, I.; Kono, A.; Chang, C.; Boyle, L.H.; Taylor, C.J.; Trowsdale, J.; et al. HLA Class I Allelic Sequence and Conformation Regulate Leukocyte Ig-Like Receptor Binding. J. Immunol. 2011, 186, 2990–2997. [Google Scholar] [CrossRef] [PubMed]
- Giolo, S.R.; Soler, J.M.P.; Greenway, S.C.; Almeida, M.A.A.; De Andrade, M.; Seidman, J.G.; Seidman, C.E.; Krieger, J.E.; Pereira, A.C. Brazilian Urban Population Genetic Structure Reveals a High Degree of Admixture. Eur. J. Hum. Genet. 2012, 20, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Yie, S.M.; Li, L.H.; Xiao, R.; Librach, C.L. A Single Base-Pair Mutation in the 3′-Untranslated Region of HLA-G MRNA Is Associated with Pre-Eclampsia. Mol. Hum. Reprod. 2008, 14, 649–653. [Google Scholar] [CrossRef]
- Cilião Alves, D.C.; De Oliveira Crispim, J.C.; Castelli, E.C.; Mendes-Junior, C.T.; Deghaide, N.H.S.; Barros Silva, G.E.; Costa, R.S.; Saber, L.T.; Moreau, P.; Donadi, E.A. Human Leukocyte Antigen-G 3’ Untranslated Region Polymorphisms Are Associated with Better Kidney Allograft Acceptance. Hum. Immunol. 2012, 73, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Milet, J.; Courtin, D.; Sabbagh, A.; Massaro, J.D.; Castelli, E.C.; Migot-Nabias, F.; Favier, B.; Rouas-Freiss, N.; Donadi, E.A.; et al. Association of HLA-G 3’UTR Polymorphisms with Response to Malaria Infection: A First Insight. Infect. Genet. Evol. 2013, 16, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Lucena-Silva, N.; Teixeira, M.A.G.; Ramos, A.d.L.; de Albuquerque, R.S.; Diniz, G.T.N.; Mendes-Junior, C.T.; Castelli, E.C.; Donadi, E.A. The +3187A/G HLA-G Polymorphic Site Is Associated with Polar Forms and Reactive Reaction in Leprosy. Mol. Genet. Genom. Med. 2013, 1, 123–130. [Google Scholar] [CrossRef]
- Quach, K.; Grover, S.A.; Kenigsberg, S.; Librach, C.L. A Combination of Single Nucleotide Polymorphisms in the 3’untranslated Region of HLA-G Is Associated with Preeclampsia. Hum. Immunol. 2014, 75, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Catamo, E.; Zupin, L.; Segat, L.; Celsi, F.; Crovella, S. HLA-G and Susceptibility to Develop Celiac Disease. Hum. Immunol. 2015, 76, 36–41. [Google Scholar] [CrossRef]
- Cilião Alves, D.C.; Haddad, R.; Rocha-Júnior, M.C.; De Deus Wagatsuma, V.M.; Martelli-Palomino, G.; Marques, A.A.; Takayanagui, O.M.; Covas, D.T.; Kashima, S.; Donadi, E.A. HLA-G 3′-Untranslated Region Polymorphisms Are Associated with HTLV-1 Infection, Proviral Load and HTLV-Associated Myelopathy/Tropical Spastic Paraparesis Development. J. Gen. Virol. 2016, 97, 2742–2752. [Google Scholar] [CrossRef]
- Mariaselvam, C.M.; Chaaben, A.B.; Salah, S.; Charron, D.; Krishnamoorthy, R.; Tamouza, R.; Negi, V.S. Human Leukocyte Antigen-G Polymorphism Influences the Age of Onset and Autoantibody Status in Rheumatoid Arthritis. Tissue Antigens 2015, 85, 182–189. [Google Scholar] [CrossRef]
- Castelli, E.C.; Veiga-Castelli, L.C.; Yaghi, L.; Moreau, P.; Donadi, E.A. Transcriptional and Posttranscriptional Regulations of the HLA-G Gene. J. Immunol. Res. 2014, 2014, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, J.B.; Kudla, G. Synonymous but Not the Same. Natl. Rev. Genet. 2011, 12, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Komar, A.A. The Yin and Yang of Codon Usage. Hum. Mol. Genet. 2016, 25, R77–R85. [Google Scholar] [CrossRef] [PubMed]
- Stranger, B.E.; Nica, A.C.; Forrest, M.S.; Dimas, A.; Bird, P.; Beazley, C.; Ingle, C.E.; Dunning, M.; Flicek, P.; Montgomery, S.; et al. Population Genomics of Human Gene Expression. Nat. Genet. 2007, 39, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.H. Gene Interaction; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 3, ISBN 9780080961569. [Google Scholar]
- Moore, J.H. Computational Analysis of Gene-Gene Interactions Using Multifactor Dimensionality Reduction. Expert Rev. Mol. Diagn. 2004, 4, 795–803. [Google Scholar] [CrossRef]
- Graebin, P.; Veit, T.D.; Alho, C.S.; Dias, F.S.; Chies, J.A.B. Polymorphic Variants in Exon 8 at the 3’ UTR of the HLA-G Gene Are Associated with Septic Shock in Critically Ill Patients. Crit. Care 2012, 16, R211. [Google Scholar] [CrossRef]
- Rajasekaran, A.; Shivakumar, V.; Kalmady, S.V.; Narayanaswamy, J.C.; Subbana, M.; Venugopal, D.; Amaresha, A.C.; Venkatasubramanian, G.; Berk, M.; Debnath, M. The Impact of HLA-G 3′ UTR Variants and SHLA-G on Risk and Clinical Correlates of Schizophrenia. Hum. Immunol. 2016, 77, 1166–1171. [Google Scholar] [CrossRef]
- Sundaresh, A.; Wu, C.L.; Chinnadurai, R.K.; Rajkumar, R.P.; Mariaselvam, C.M.; LeMaoult, J.; Krishnamoorthy, R.; Leboyer, M.; Negi, V.S.; Tamouza, R. The HLA-G Genetic Contribution to Bipolar Disorder: A Trans-Ethnic Replication. Immunol. Investig. 2018, 47, 593–604. [Google Scholar] [CrossRef]
- Schwich, E.; Rebmann, V.; Michita, R.T.; Rohn, H.; Voncken, J.W.; Horn, P.A.; Kimmig, R.; Kasimir-Bauer, S.; Buderath, P. HLA-G 3′ Untranslated Region Variants +3187G/G, +3196G/G and +3035T Define Diametrical Clinical Status and Disease Outcome in Epithelial Ovarian Cancer. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Jansen, R.; Hottenga, J.J.; Nivard, M.G.; Abdellaoui, A.; Laport, B.; de Geus, E.J.; Wright, F.A.; Penninx, B.W.J.H.; Boomsma, D.I. Conditional EQTL Analysis Reveals Allelic Heterogeneity of Gene Expression. Hum. Mol. Genet. 2017, 26, 1444–1451. [Google Scholar] [CrossRef]
- Györy, I.; Boller, S.; Nechanitzky, R.; Mandel, E.; Pott, S.; Liu, E.; Grosschedl, R. Transcription Factor EBf1 Regulates Differentiation Stage-Specific Signaling, Proliferation, and Survival of B Cells. Genes Dev. 2012, 26, 668–682. [Google Scholar] [CrossRef] [PubMed]
- Noelle, R.J.; Erickson, L.D. Determinations of B Cell Fate in Immunity and Autoimmunity. Curr. Dir. Autoimmun. 2005, 8, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Thompson, N.; Isenberg, D.A.; Jury, E.C.; Ciurtin, C. Exploring BAFF: Its Expression, Receptors and Contribution to the Immunopathogenesis of Sjögren’s Syndrome. Rheumatology 2016, 55, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Harning, R.; Cui, J.; Bystryn, J.-C. Relation between the Incidence and Level of Pigment Cell Antibodies and Disease Activity in Vitiligo. J. Investig. Dermatol. 1991, 97, 1078–1080. [Google Scholar] [CrossRef]
- Castelli, E.C.; Castro, M.V.D.; Naslavsky, M.S.; Scliar, M.O.; Nayane, S. Immunogenetics of Resistance to SARS-CoV-2 Infection in Discordant Couples. medRxiv Prepr. 2021, 1–16. [Google Scholar] [CrossRef]
- Jalel, A.; Ridha, A.; Laurent, D.; Philippe, M.; Hamdaoui, M. Impact of HLA-G in the Outcome of Vitiligo in Tunisian Patients. Indian J. Dermatol. 2010, 55, 25–28. [Google Scholar] [CrossRef]
- Rizzo, R.; Bortolotti, D.; Bolzani, S.; Fainardi, E. HLA-G Molecules in Autoimmune Diseases and Infections. Front. Immunol. 2014, 5, 1–11. [Google Scholar] [CrossRef]
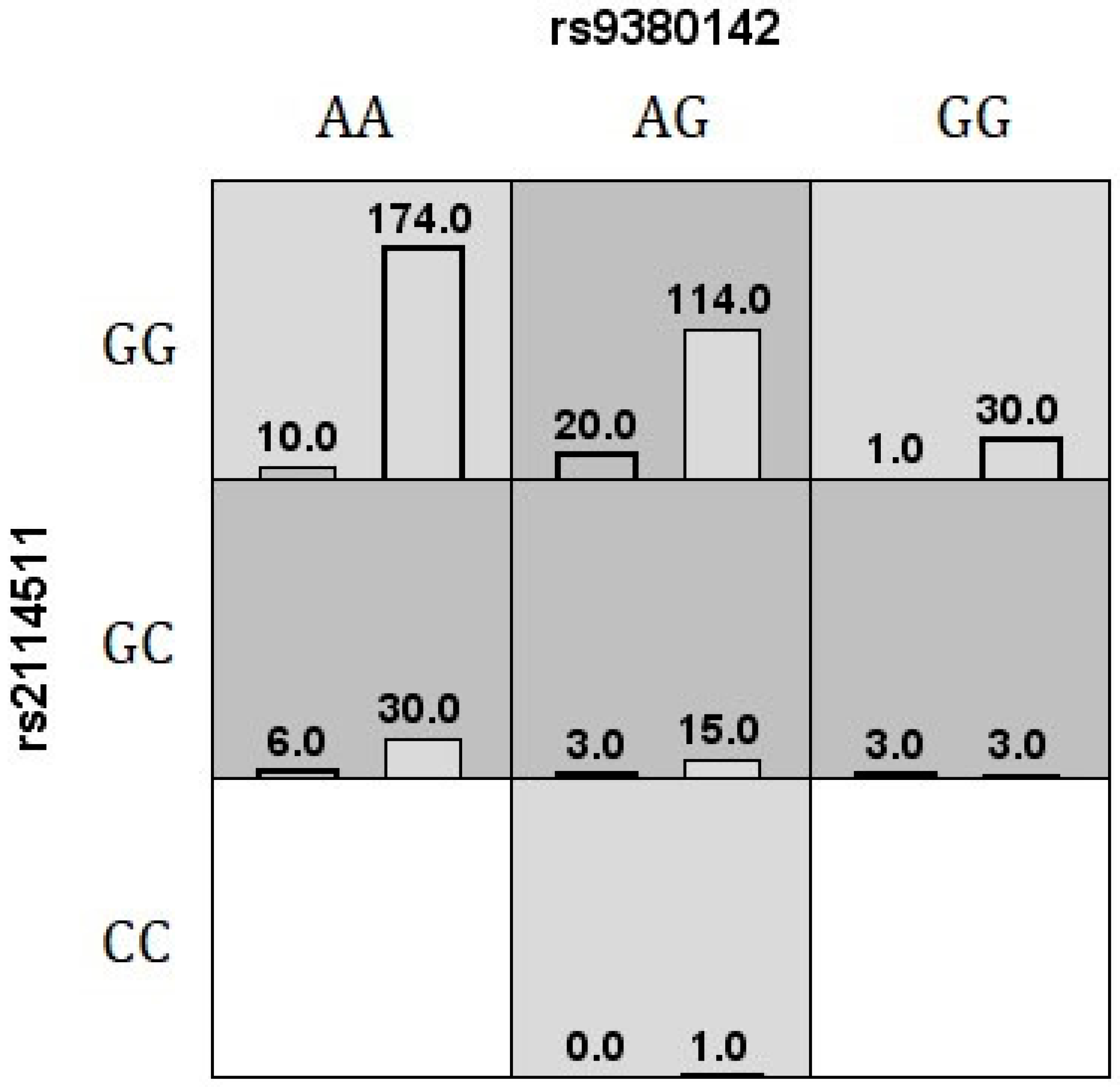
| Additive Model | Before Adjustment | After Adjustment | ||||
|---|---|---|---|---|---|---|
| GENE | SNP | Allele | OR (L95–U95) | p | OR (L95–U95) | p |
| HLA-G | rs17875403 1 | T | 8.902 (1.221–64.890) | 0.0309 | - | - |
| LILRB2 | rs10405713 1 | C | 2.357 (1.091–5.094) | 0.0292 | - | - |
| LILRB2 | rs373032 1 | T | 0.485 (0.251–0.938) | 0.0315 | - | - |
| LILRB1 | rs10427127 | C | 1.957 (1.082–3.538) | 0.0263 | - | - |
| LILRB1 | rs10425827 1 | G | 2.210 (1.160–4.209) | 0.0159 | - | - |
| LILRB1 | rs61739173 | A | 2.173 (1.088–4.337) | 0.0278 | - | - |
| LILRB1 | rs2114511 1 | C | 2.349 (1.156–4.775) | 0.0182 | - | - |
| LILRB1 | rs272423 1 | C | - | - | 0.5611 (0.331–0.950) | 0.0316 |
| Dominant Model | Before Adjustment | After Adjustment | ||||
|---|---|---|---|---|---|---|
| GENE | SNP | Allele | OR (L95–U95) | p | OR (L95–U95) | p |
| HLA-G | rs6932888 | C | 1.914 (1.004–3.649) | 0.0485 | 2.022 (1.052–3.888) | 0.0348 |
| HLA-G | rs6932596 | T | 1.914 (1.004–3.649) | 0.0485 | 2.022 (1.052–3.888) | 0.0348 |
| HLA-G | rs17875403 1 | T | 8.902 (1.221–64.890) | 0.0301 | - | - |
| HLA-G | rs9380142 | G | 2.112 (1.101–4.053) | 0.0246 | 2.225 (1.149–4.308) | 0.0176 |
| LILRB2 | rs10405713 1 | C | 2.660 (1.075–6.580) | 0.0343 | - | - |
| LILRB2 | rs373032 1 | T | 0.434 (0.208–0.906) | 0.0263 | - | - |
| LILRB1 | rs10425827 1 | G | 2.130 (1.010–4.492) | 0.0470 | - | - |
| LILRB1 | rs2114511 1 | C | 2.512 (1.209–5.219) | 0.0135 | - | - |
| LILRB1 | rs272423 1 | C | - | - | 0.502 (0.261–0.965) | 0.0388 |
| MDR Method | Best 2 Order Model | TBA, CVC | p-Value |
|---|---|---|---|
| MDR v3.0.2 | rs9380142, rs2114511 | 0.65, 9/10 | - |
| GMDR v.0.9 | rs9380142, rs2114511 | 0.64, 10/10 | 0.0107 a |
| MB-MDR (before adjustment) | rs373032, rs2114511 | - | 0.0020 b |
| MB-MDR (after adjustment) | rs9380142, rs2114511 | - | 0.0130 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira-Caramez, M.L.d.; Veiga-Castelli, L.; Souza, A.S.; Cardili, R.N.; Courtin, D.; Flória-Santos, M.; Donadi, E.; Giuliatti, S.; Sabbagh, A.; Castelli, E.C.; et al. Evidence for Epistatic Interaction between HLA-G and LILRB1 in the Pathogenesis of Nonsegmental Vitiligo. Cells 2023, 12, 630. https://doi.org/10.3390/cells12040630
Oliveira-Caramez MLd, Veiga-Castelli L, Souza AS, Cardili RN, Courtin D, Flória-Santos M, Donadi E, Giuliatti S, Sabbagh A, Castelli EC, et al. Evidence for Epistatic Interaction between HLA-G and LILRB1 in the Pathogenesis of Nonsegmental Vitiligo. Cells. 2023; 12(4):630. https://doi.org/10.3390/cells12040630
Chicago/Turabian StyleOliveira-Caramez, Maria Luiza de, Luciana Veiga-Castelli, Andreia S. Souza, Renata Nahas Cardili, David Courtin, Milena Flória-Santos, Eduardo Donadi, Silvana Giuliatti, Audrey Sabbagh, Erick C. Castelli, and et al. 2023. "Evidence for Epistatic Interaction between HLA-G and LILRB1 in the Pathogenesis of Nonsegmental Vitiligo" Cells 12, no. 4: 630. https://doi.org/10.3390/cells12040630
APA StyleOliveira-Caramez, M. L. d., Veiga-Castelli, L., Souza, A. S., Cardili, R. N., Courtin, D., Flória-Santos, M., Donadi, E., Giuliatti, S., Sabbagh, A., Castelli, E. C., & Mendes-Junior, C. T. (2023). Evidence for Epistatic Interaction between HLA-G and LILRB1 in the Pathogenesis of Nonsegmental Vitiligo. Cells, 12(4), 630. https://doi.org/10.3390/cells12040630






