Iron-Induced Oxidative Stress in Human Diseases
Abstract
1. Introduction
2. Basic Properties of Iron
3. Redox-Active Iron in Blood
3.1. Serum Transferrin
3.2. A Candidate for Redox-Active Iron in Blood: NTBI
3.3. Estimation of NTBI in the Presence of Ligands
3.4. Experimental Assay of NTBI
4. Redox-Active Iron in Cells: Transit Iron Pool
4.1. Iron Balance in Cells
4.2. Ferritin: A Defence Molecule against Redox-Active Iron
4.3. Indicators for TIP: IRP1/2 and HIF-PHD
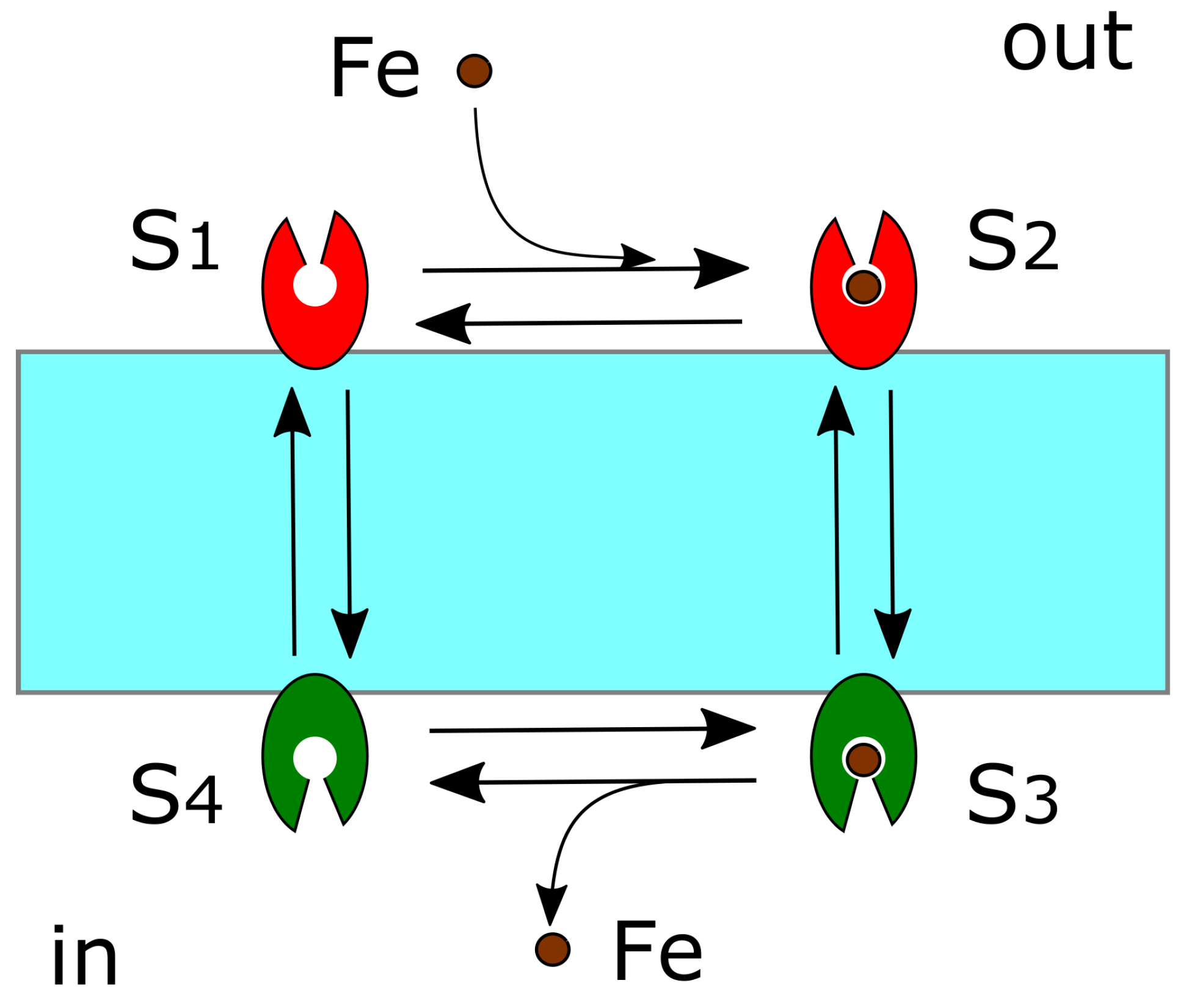
4.4. DMT1 and FPN1
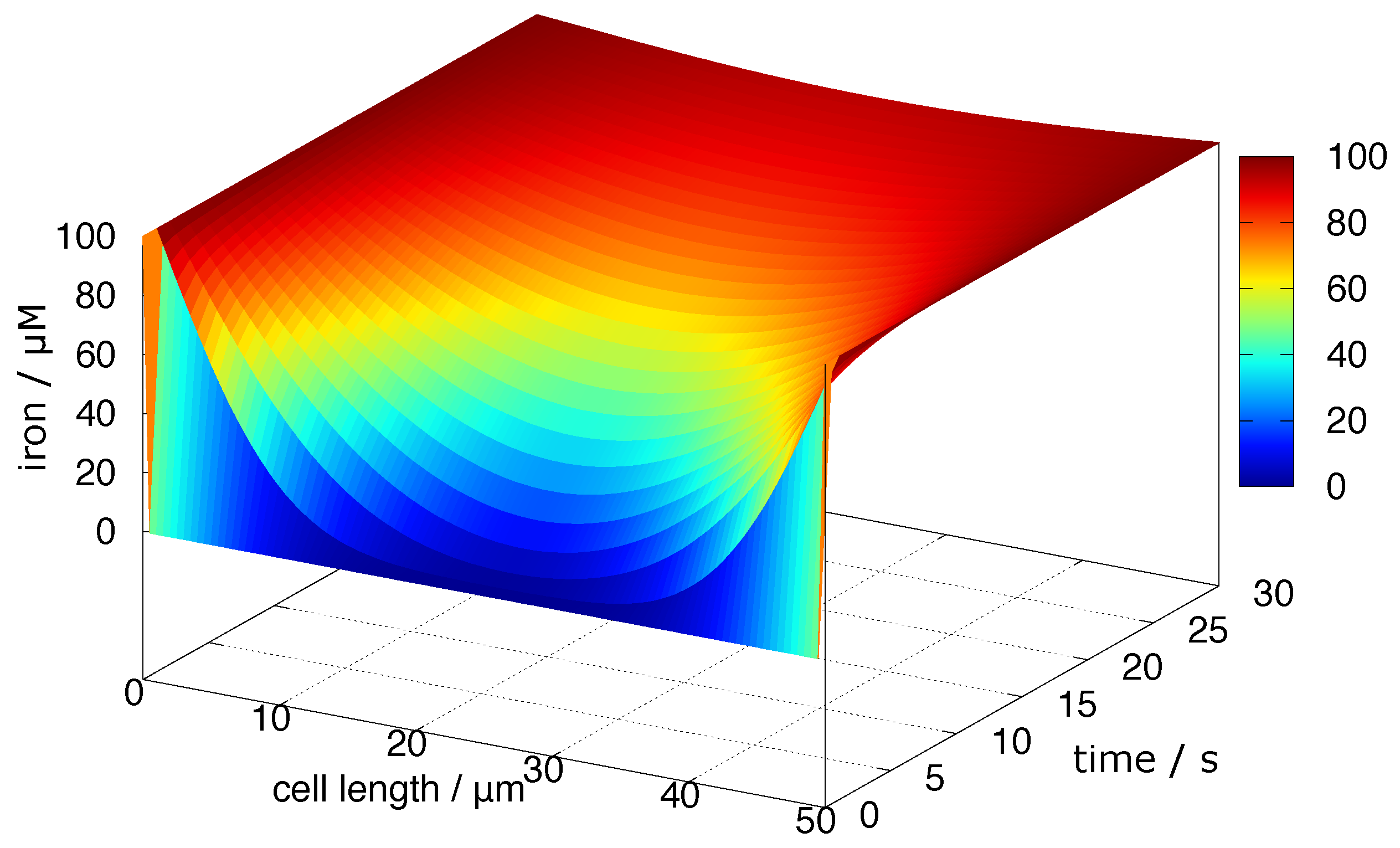
4.5. Diffusion of Redox-Active Iron
4.6. Experimental Assay of the Transit Iron Pool
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ROS | Reactive oxygen species |
| NTBI | Non-transferrin bound iron |
| TIP | Transit iron pool |
| HIF | Hypoxia-inducible factor |
| IRP | Iron regulatory protein |
| IRE | Iron-responsive element |
| bipy | 2,2’-bipyridyl |
| phen | phenanthroline |
| nta | nitrilotriacetic acid |
| dtpa | diethylenetriamine pentaacetic acid |
| edta | ethylenediamine tetraacetic acid |
| DFO | Deferoxamine |
| DMPO | 5,5-Dimethyl-1-pyrroline-N-oxide |
| Tf | Transferrin |
| TFR1/2 | Transferrin receptor 1/2 |
| EPR | Electron paramagnetic resonance |
| TBAR | 2-thiobarbituric acid reactive substance |
| DMT1 | Divalent metal transporter 1 |
| FPN1 | Ferroportin 1 |
| PCBP1/2 | Poly(rC)-binding proteins 1/2 |
| STEAP3 | Six-transmembrane epithelial antigen of prostate 3 |
| ZIP | ZRT/IRT-like protein |
| TRPC6 | Transient receptor potential cation channel subfamily C member 6 |
| PHD | Prolyl hydroxylase domain |
| FRAP | Fluorescence recovery after photobleaching |
| FRET | Fluorescence resonance energy transfer |
References
- Pasricha, S.R.; Tye-Din, J.; Muckenthaler, M.U.; Swinkels, D.W. Iron deficiency. Lancet 2021, 397, 233–248. [Google Scholar] [CrossRef]
- Crichton, R.R. Iron Metabolism: From Molecular Mechanisms to Clinical Consequences, 4th ed.; Wiley: Hoboken, NJ, USA, 2016. [Google Scholar]
- Barton, J.C.; Acton, R.T. Hemochromatosis. J. Diabetes Res. 2017, 2017, 9826930. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, H. The mechanisms of systemic iron homeostasis and etiology, diagnosis, and treatment of hereditary hemochromatosis. Int. J. Hematol. 2018, 107, 31–43. [Google Scholar] [PubMed]
- Brissot, P.; Brissot, E. What’s Important and New in Hemochromatosis? Clin. Hematol. Int. 2020, 2, 143–148. [Google Scholar] [CrossRef]
- Yamada, N.; Karasawa, T.; Wakiya, T.; Sadatomo, A.; Ito, H.; Kamata, R.; Watanabe, S.; Komada, T.; Kimura, H.; Sanada, Y.; et al. Iron overload as a risk factor for hepatic ischemia-reperfusion injury in liver transplantation: Potential role of ferroptosis. Am. J. Transpl. 2020, 20, 1606–1618. [Google Scholar]
- Blomgren, K.; Hagberg, H. Free radicals, mitochondria, and hypoxia-ischemia in the developing brain. Free Radic. Biol. Med. 2006, 40, 388–397. [Google Scholar]
- Lee, N.J.; Ha, S.K.; Sati, P.; Absinta, M.; Nair, G.; Luciano, N.J.; Leibovitch, E.C.; Yen, C.C.; Rouault, T.A.; Silva, A.C.; et al. Potential role of iron in repair of inflammatory demyelinating lesions. J. Clin. Investig. 2019, 129, 4365–4376. [Google Scholar]
- Magid-Bernstein, J.; Girard, R.; Polster, S.; Srinath, A.; Romanos, S.; Awad, I.A.; Sansing, L.H. Cerebral Hemorrhage: Pathophysiology, Treatment, and Future Directions. Circ. Res. 2022, 130, 1204–1229. [Google Scholar]
- Kumar, V.; Abbas, A.K.; Aster, J.C. Robbins and Cotran Pathologic Basis of Disease; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Galaris, D.; Barbouti, A.; Pantopoulos, K. Iron homeostasis and oxidative stress: An intimate relationship. Biochim. Biophys Acta Mol. Cell Res. 2019, 1866, 118535. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Jacob, A.; Worwood, M. Iron in Biochemistry and Medicine, II, 2nd ed.; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Crichton, R.R. Biological Inorganic Chemistry: A New Introduction to Molecular Structure and Function, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Okazaki, Y. The Role of Ferric Nitrilotriacetate in Renal Carcinogenesis and Cell Death: From Animal Models to Clinical Implications. Cancers 2022, 14, 1495. [Google Scholar] [CrossRef]
- Mizuno, R.; Kawabata, T.; Sutoh, Y.; Nishida, Y.; Okada, S. Oxidative renal tubular injuries induced by aminocarboxylate-type iron (III) coordination compounds as candidate renal carcinogens. Biometals 2006, 19, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Fujita, T.; Ohba, S.; Nishida, Y.; Goto, A.; Tokii, T. Tetrapotassium μ-carbonato-μ-oxo-bis[nitrilotriacetatoiron(III)] dimethanol dihydrate. Acta Cryst. 1994, 50, 544–546. [Google Scholar] [CrossRef]
- Nishida, Y.; Ito, Y.; Satoh, T. Origin of renal proximal tubular injuries by Fe(III)-nta chelate. Z Naturforsch. C J. Biosci. 2007, 62, 608–612. [Google Scholar] [CrossRef]
- Nishida, Y.; Ito, S. Comparison on reactivity of Fe(III) and Al(III) compounds in the presence of hydrogen peroxide: Its relevance to possible origin for central nervous system toxicity by aluminum ion. Z Naturforsch. C J. Biosci. 1995, 50, 571–577. [Google Scholar] [CrossRef]
- Hrycay, E.G.; Bandiera, S.M. The monooxygenase, peroxidase, and peroxygenase properties of cytochrome P450. Arch Biochem. Biophys. 2012, 522, 71–89. [Google Scholar] [CrossRef]
- Rumble, J. CRC Handbook of Chemistry and Physics, 100th ed.; CRC Press: New York, NY, USA, 2019. [Google Scholar]
- Wang, Z.; Liu, C.; Wang, X.; Marshall, M.J.; Zachara, J.M.; Rosso, K.M.; Dupuis, M.; Fredrickson, J.K.; Heald, S.; Shi, L. Kinetics of reduction of Fe(III) complexes by outer membrane cytochromes MtrC and OmcA of Shewanella oneidensis MR-1. Appl. Environ. Microbiol. 2008, 74, 6746–6755. [Google Scholar] [CrossRef] [PubMed]
- Filipovic, M.R.; Koppenol, W.H. The Haber-Weiss reaction-The latest revival. Free Radic. Biol. Med. 2019, 145, 221–222. [Google Scholar] [CrossRef]
- Stanbury, D.M. The principle of detailed balancing, the iron-catalyzed disproportionation of hydrogen peroxide, and the Fenton reaction. Dalton Trans. 2022, 51, 2135–2157. [Google Scholar] [CrossRef]
- Koppenol, W.H.; Hider, R.H. Iron and redox cycling. Do’s and don’ts. Free Radic. Biol. Med. 2019, 133, 3–10. [Google Scholar] [CrossRef]
- Stoll, S.; Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 2006, 178, 42–55. [Google Scholar] [CrossRef]
- Ranguelova, K.; Mason, R.P. The fidelity of spin trapping with DMPO in biological systems. Magn. Reson. Chem. 2011, 49, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Makino, K.; Hagiwara, T.; Hagi, A.; Nishi, M.; Murakami, A. Cautionary note for DMPO spin trapping in the presence of iron ion. Biochem. Biophys. Res. Commun. 1990, 172, 1073–1080. [Google Scholar] [CrossRef]
- Makino, K.; Hagiwara, T.; Imaishi, H.; Nishi, M.; Fujii, S.; Ohya, H.; Murakami, A. DMPO spin trapping in the presence of Fe ion. Free Radic. Res. Commun. 1990, 9, 233–240. [Google Scholar] [CrossRef]
- Makino, K.; Imaishi, H.; Morinishi, S.; Hagiwara, T.; Takeuchi, T.; Murakami, A.; Nishi, M. An artifact in the ESR spectrum obtained by spin trapping with DMPO. Free Radic. Res. Commun. 1989, 6, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.N.; Moniza, T.; Castro, B.; Rangel, M. Human transferrin: An inorganic biochemistry perspective. Coord. Chem. Rev. 2021, 449, 214186. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Meng, E.C.; Couch, G.S.; Croll, T.I.; Morris, J.H.; Ferrin, T.E. UCSF ChimeraX: Structure visualization for researchers, educators, and developers. Protein. Sci. 2021, 30, 70–82. [Google Scholar] [CrossRef]
- Cowan, J.A. Inorganic Biochemistry: An Introduction, 2nd ed.; Wiley-VCH: New York, NY, USA, 1997. [Google Scholar]
- Mathies, G.; Gast, P.; Chasteen, N.D.; Luck, A.N.; Mason, A.B.; Groenen, E.J. Exploring the Fe(III) binding sites of human serum transferrin with EPR at 275 GHz. J. Biol. Inorg. Chem. 2015, 20, 487–496. [Google Scholar] [CrossRef][Green Version]
- Azarkh, M.; Peter Gast, P.; Mason, A.B.; Groenen, E.J.J.; Mathies, G. Analysis of the EPR spectra of transferrin: The importance of a zero-field-splitting distribution and 4 th-order terms. Phys. Chem. Chem. Phys. 2019, 21, 16937–16948. [Google Scholar] [CrossRef]
- Abragam, A.; Bleaney, B. Electron Paramagnetic Resonance of Transition Ions; Dover: New York, NY, USA, 1986. [Google Scholar]
- Hershko, C.; Graham, G.; Bates, G.W.; Rachmilewitz, E.A. Non-specific serum iron in thalassaemia: An abnormal serum iron fraction of potential toxicity. Br. J. Haematol. 1978, 40, 255–263. [Google Scholar] [CrossRef]
- Freiser, H. The pointer function—An invaluable calculational tool. J. Chem. Ed. 1994, 71, 297–299. [Google Scholar] [CrossRef]
- Ito, S.; Ikuta, K.; Kato, D.; Shibusa, K.; Niizeki, N.; Tanaka, H.; Addo, L.; Toki, Y.; Hatayama, M.; Inamura, J.; et al. Non-transferrin-bound iron assay system utilizing a conventional automated analyzer. Clin. Chim. Acta 2014, 437, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.J.; Halliwell, B. Measurement of iron and copper in biological systems: Bleomycin and copper-phenanthroline assays. Methods Enzymol. 1994, 233, 82–92. [Google Scholar]
- von Bonsdorff, L.; Lindeberg, E.; Sahlstedt, L.; Lehto, J.; Parkkinen, J. Bleomycin-detectable iron assay for non-transferrin-bound iron in hematologic malignancies. Clin. Chem. 2002, 48, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Angoro, B.; Motshakeri, M.; Hemmaway, C.; Svirskis, D.; Sharma, M. Non-transferrin bound iron. Clin. Chim. Acta 2022, 531, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, H. Transferrin and transferrin receptors update. Free Radic. Biol. Med. 2019, 133, 46–54. [Google Scholar] [CrossRef]
- Yanatori, I.; Kishi, F. DMT1 and iron transport. Free Radic. Biol. Med. 2019, 133, 55–63. [Google Scholar] [CrossRef]
- Knutson, M.D. Non-transferrin-bound iron transporters. Free Radic. Biol. Med. 2019, 133, 101–111. [Google Scholar] [CrossRef]
- Yanatori, I.; Richardson, D.R.; Imada, K.; Kishi, F. Iron Export through the Transporter Ferroportin 1 Is Modulated by the Iron Chaperone PCBP2. J. Biol. Chem. 2016, 291, 17303–17318. [Google Scholar] [CrossRef]
- Yanatori, I.; Richardson, D.R.; Toyokuni, S.; Kishi, F. The iron chaperone poly(rC)-binding protein 2 forms a metabolon with the heme oxygenase 1/cytochrome P450 reductase complex for heme catabolism and iron transfer. J. Biol. Chem. 2017, 292, 13205–13229. [Google Scholar] [CrossRef]
- Yanatori, I.; Richardson, D.R.; Toyokuni, S.; Kishi, F. The new role of poly (rC)-binding proteins as iron transport chaperones: Proteins that could couple with inter-organelle interactions to safely traffic iron. Biochim. Biophys. Acta Gen. Subj. 2020, 1864, 129685. [Google Scholar] [CrossRef]
- Leidgens, S.; Bullough, K.Z.; Shi, H.; Li, F.; Shakoury-Elizeh, M.; Yabe, T.; Subramanian, P.; Hsu, E.; Natarajan, N.; Nandal, A.; et al. Each member of the poly-r(C)-binding protein 1 (PCBP) family exhibits iron chaperone activity toward ferritin. J. Biol. Chem. 2013, 288, 17791–17802. [Google Scholar] [CrossRef] [PubMed]
- Frey, A.G.; Nandal, A.; Park, J.H.; Smith, P.M.; Yabe, T.; Ryu, M.S.; Ghosh, M.C.; Lee, J.; Rouault, T.A.; Park, M.H.; et al. Iron chaperones PCBP1 and PCBP2 mediate the metallation of the dinuclear iron enzyme deoxyhypusine hydroxylase. Proc. Natl. Acad. Sci. USA 2014, 111, 8031–8036. [Google Scholar] [CrossRef]
- Drakesmith, H.; Nemeth, E.; Ganz, T. Ironing out Ferroportin. Cell. Metab. 2015, 22, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, E.; Ganz, T. Hepcidin-Ferroportin Interaction Controls Systemic Iron Homeostasis. Int. J. Mol. Sci. 2021, 22, 6493. [Google Scholar] [CrossRef] [PubMed]
- Petrak, J.; Vyoral, D. Hephaestin—A ferroxidase of cellular iron export. Int. J. Biochem. Cell Biol. 2005, 37, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Arosio, P.; Chasteen, N.D. Iron(II) and hydrogen peroxide detoxification by human H-chain ferritin. An EPR spin-trapping study. Biochemistry 2006, 45, 3429–3436. [Google Scholar] [CrossRef]
- Torti, S.V.; Torti, F.M. CD63 orchestrates ferritin export. Blood 2021, 138, 1387–1389. [Google Scholar] [CrossRef]
- Toyokuni, S.; Kong, Y.; Zheng, H.; Mi, D.; Katabuchi, M.; Motooka, Y.; Ito, F. Double-edged Sword Role of Iron-loaded Ferritin in Extracellular Vesicles. J. Cancer Prev. 2021, 26, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Arosio, P.; Elia, L.; Poli, M. Ferritin, cellular iron storage and regulation. IUBMB Life 2017, 69, 414–422. [Google Scholar] [CrossRef]
- Plays, M.; Müller, S.; Rodriguez, R. Chemistry and biology of ferritin. Metallomics 2021, 13. [Google Scholar] [CrossRef]
- Daru, J.; Colman, K.; Stanworth, S.J.; De La Salle, B.; Wood, E.M.; Pasricha, S.R. Serum ferritin as an indicator of iron status: What do we need to know? Am. J. Clin. Nutr. 2017, 106, 1634S–1639S. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Carmona, W.; Díaz-Fabregat, B.; Yuri Yoshigae, A.; de Aquino, A.M.; Scarano, W.R.; de Souza Castilho, A.C.; Avansini Marsicano, J.; do Prado, R.L.; Pessan, J.P.; de Oliveira Mendes, L. Are Serum Ferritin Levels a Reliable Cancer Biomarker? A Systematic Review and Meta-Analysis. Nutr. Cancer 2021, 74, 1917–1926. [Google Scholar] [CrossRef] [PubMed]
- Truman-Rosentsvit, M.; Berenbaum, D.; Spektor, L.; Cohen, L.A.; Belizowsky-Moshe, S.; Lifshitz, L.; Ma, J.; Li, W.; Kesselman, E.; Abutbul-Ionita, I.; et al. Ferritin is secreted via 2 distinct nonclassical vesicular pathways. Blood 2018, 131, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Holmes-Hampton, G.P.; Ghosh, M.C.; Rouault, T.A. Methods for Studying Iron Regulatory Protein 1: An Important Protein in Human Iron Metabolism. Methods Enzymol. 2018, 599, 139–155. [Google Scholar]
- Zhou, Z.D.; Tan, E.K. Iron regulatory protein (IRP)-iron responsive element (IRE) signaling pathway in human neurodegenerative diseases. Mol. Neurodegener. 2017, 12, 75. [Google Scholar] [CrossRef]
- Hirota, K. An intimate crosstalk between iron homeostasis and oxygen metabolism regulated by the hypoxia-inducible factors (HIFs). Free Radic. Biol. Med. 2019, 133, 118–129. [Google Scholar] [CrossRef]
- Gunshin, H.; Mackenzie, B.; Berger, U.V.; Gunshin, Y.; Romero, M.F.; Boron, W.F.; Nussberger, S.; Gollan, J.L.; Hediger, M.A. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 1997, 388, 482–488. [Google Scholar] [CrossRef]
- Anantram, A.; Janve, M.; Degani, M.; Singhal, R.; Kundaikar, H. Homology modelling of human divalent metal transporter (DMT): Molecular docking and dynamic simulations for duodenal iron transport. J. Mol. Graph. Model. 2018, 85, 145–152. [Google Scholar] [CrossRef]
- Taniguchi, R.; Kato, H.E.; Font, J.; Deshpande, C.N.; Wada, M.; Ito, K.; Ishitani, R.; Jormakka, M.; Nureki, O. Outward- and inward-facing structures of a putative bacterial transition-metal transporter with homology to ferroportin. Nat. Commun. 2015, 6, 8545. [Google Scholar] [CrossRef]
- Billesbølle, C.B.; Azumaya, C.M.; Kretsch, R.C.; Powers, A.S.; Gonen, S.; Schneider, S.; Arvedson, T.; Dror, R.O.; Cheng, Y.; Manglik, A. Structure of hepcidin-bound ferroportin reveals iron homeostatic mechanisms. Nature 2020, 586, 807–811. [Google Scholar] [CrossRef]
- Pan, Y.; Ren, Z.; Gao, S.; Shen, J.; Wang, L.; Xu, Z.; Yu, Y.; Bachina, P.; Zhang, H.; Fan, X.; et al. Structural basis of ion transport and inhibition in ferroportin. Nat. Commun. 2020, 11, 5686. [Google Scholar] [CrossRef] [PubMed]
- Fall, C.P.; Marland, E.S.; Wagner, J.M.; Tyson, J.J. Computational Cell Biology, 1st ed.; Springer: Philadelphia, PA, USA, 2002. [Google Scholar]
- Cegarra, L.; Colins, A.; Gerdtzen, Z.P.; Nuñez, M.T.; Salgado, J.C. Mathematical modeling of the relocation of the divalent metal transporter DMT1 in the intestinal iron absorption process. PLoS ONE 2019, 14, e0218123. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.E.; Sahai, E.; Jenkins, R.P.; O’Dea, R.D.; King, J.R. Parameter estimation in fluorescence recovery after photobleaching: Quantitative analysis of protein binding reactions and diffusion. J. Math. Biol. 2021, 83, 1–38. [Google Scholar] [CrossRef]
- Lippincott-Schwartz, J.; Snapp, E.L.; Phair, R.D. The Development and Enhancement of FRAP as a Key Tool for Investigating Protein Dynamics. Biophys. J. 2018, 115, 1146–1155. [Google Scholar] [CrossRef]
- Needleman, D.; Shelley, M. The stormy fluid dynamics of the living cell. Phys. Today 2019, 72, 32–38. [Google Scholar] [CrossRef]
- Yamamoto, E.; Akimoto, T.; Mitsutake, A.; Metzler, R. Universal Relation between Instantaneous Diffusivity and Radius of Gyration of Proteins in Aqueous Solution. Phys. Rev. Lett. 2021, 126, 128101. [Google Scholar] [CrossRef] [PubMed]
- Kühn, T.; Ihalainen, T.O.; Hyväluoma, J.; Dross, N.; Willman, S.F.; Langowski, J.; Vihinen-Ranta, M.; Timonen, J. Protein diffusion in mammalian cell cytoplasm. PLoS ONE 2011, 6, e22962. [Google Scholar] [CrossRef]
- Espósito, B.P.; Epsztejn, S.; Breuer, W.; Cabantchik, Z.I. A review of fluorescence methods for assessing labile iron in cells and biological fluids. Anal. Biochem. 2002, 304, 1–18. [Google Scholar] [CrossRef]
- Kakhlon, O.; Cabantchik, Z.I. The labile iron pool: Characterization, measurement, and participation in cellular processes(1). Free Radic. Biol. Med. 2002, 33, 1037–1046. [Google Scholar] [CrossRef]
- Ma, Y.; Abbate, V.; Hider, R.C. Iron-sensitive fluorescent probes: Monitoring intracellular iron pools. Metallomics 2015, 7, 212–222. [Google Scholar] [CrossRef]
- Mukaide, T.; Hattori, Y.; Misawa, N.; Funahashi, S.; Jiang, L.; Hirayama, T.; Nagasawa, H.; Toyokuni, S. Histological detection of catalytic ferrous iron with the selective turn-on fluorescent probe RhoNox-1 in a Fenton reaction-based rat renal carcinogenesis model. Free Radic. Res. 2014, 48, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T. Fluorescent probes for the detection of catalytic Fe(II) ion. Free Radic. Biol. Med. 2019, 133, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Ito, F.; Shi, L.; Wang, Y.; Ishida, C.; Hattori, Y.; Niwa, M.; Hirayama, T.; Nagasawa, H.; Iwase, A.; et al. Ovarian endometriosis-associated stromal cells reveal persistently high affinity for iron. Redox. Biol. 2015, 6, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T. Development of Chemical Tools for Imaging of Fe(II) Ions in Living Cells: A Review. Acta Histochem. Cytochem. 2018, 51, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T.; Inden, M.; Tsuboi, H.; Niwa, M.; Uchida, Y.; Naka, Y.; Hozumi, I.; Nagasawa, H. A Golgi-targeting fluorescent probe for labile Fe(ii) to reveal an abnormal cellular iron distribution induced by dysfunction of VPS35. Chem. Sci. 2019, 10, 1514–1521. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T.; Miki, A.; Nagasawa, H. Organelle-specific analysis of labile Fe(ii) during ferroptosis by using a cocktail of various colour organelle-targeted fluorescent probes. Metallomics 2019, 11, 111–117. [Google Scholar] [CrossRef]
- Aron, A.T.; Loehr, M.O.; Bogena, J.; Chang, C.J. An Endoperoxide Reactivity-Based FRET Probe for Ratiometric Fluorescence Imaging of Labile Iron Pools in Living Cells. J. Am. Chem. Soc. 2016, 138, 14338–14346. [Google Scholar] [CrossRef]
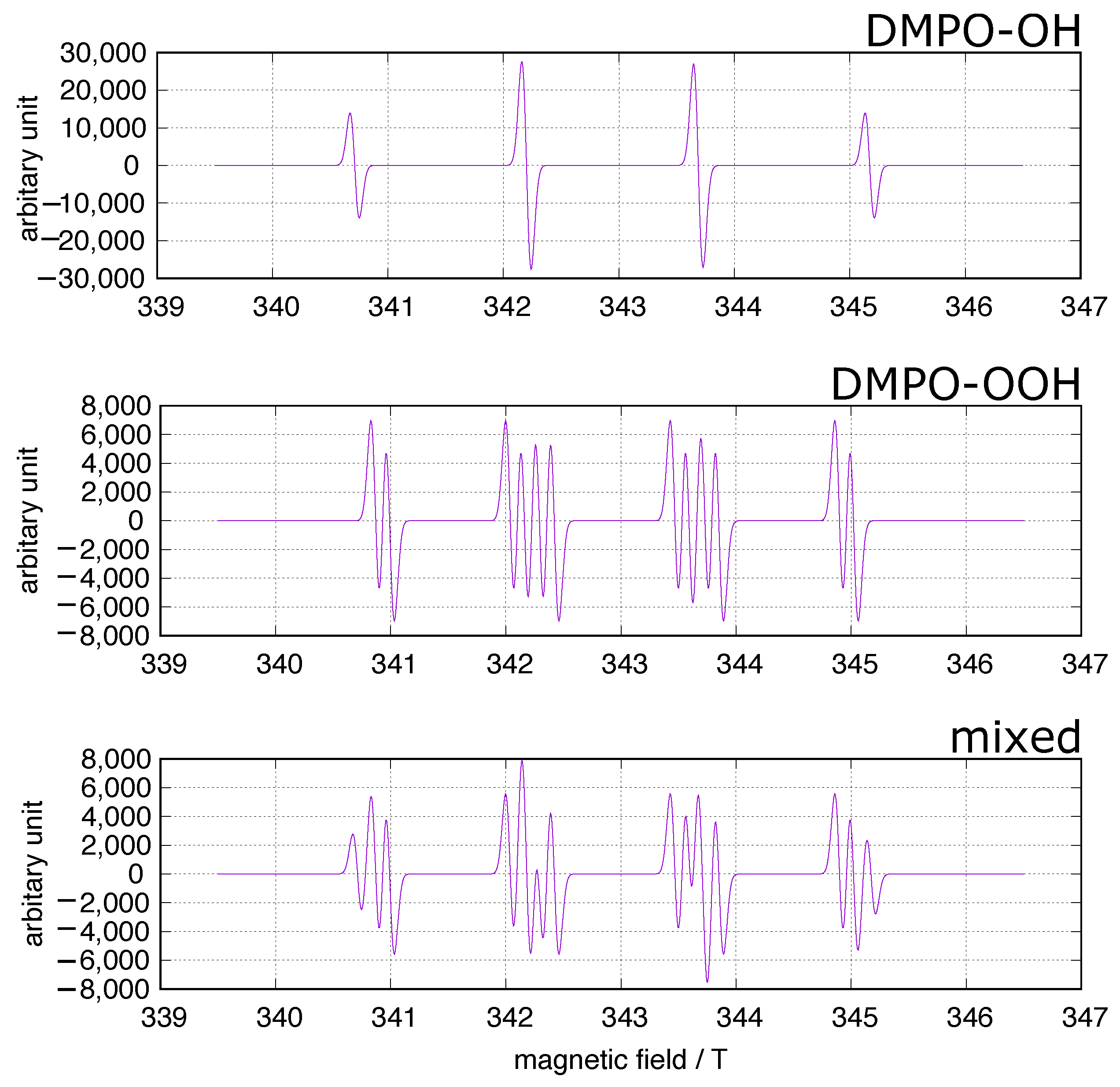
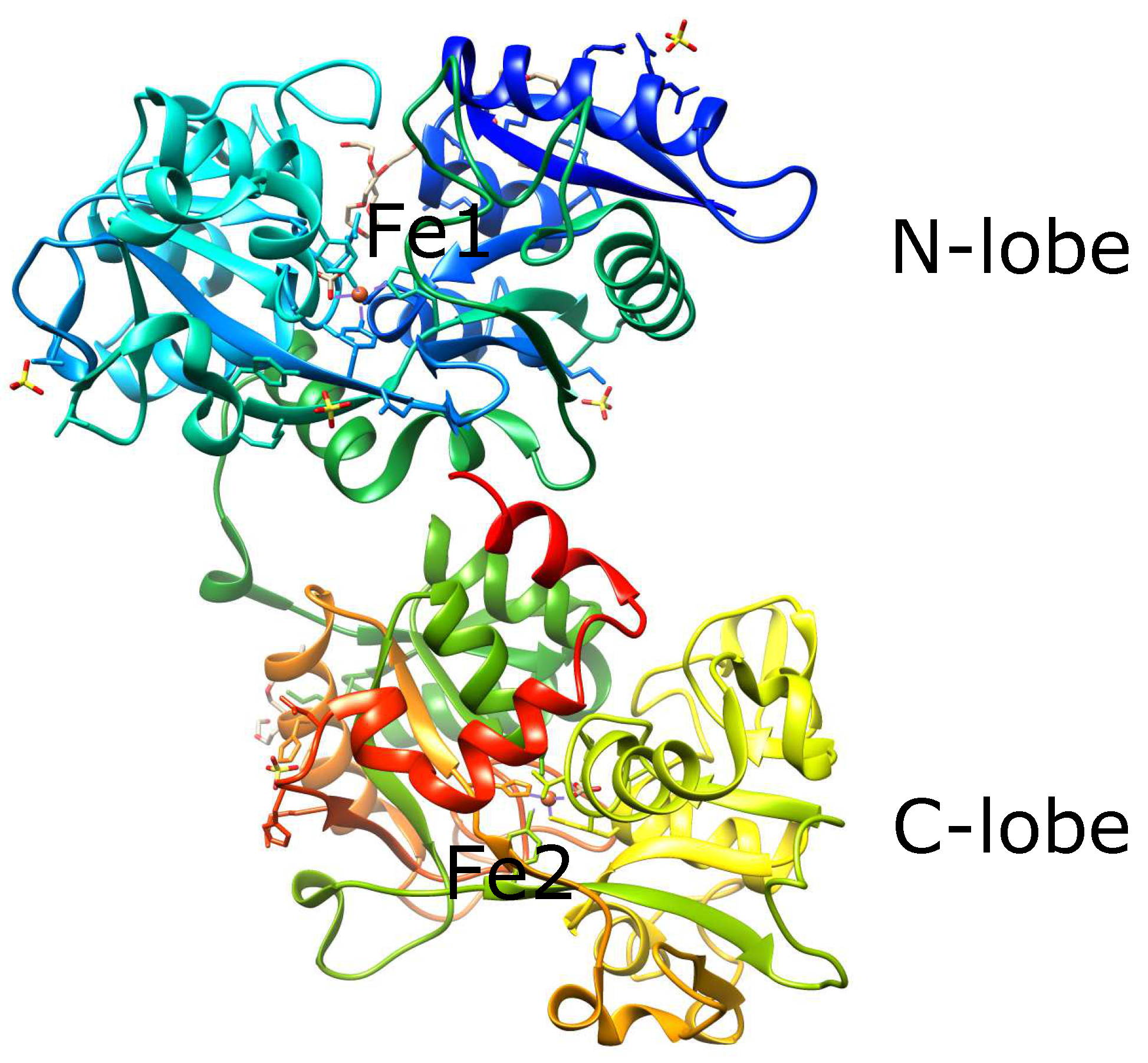
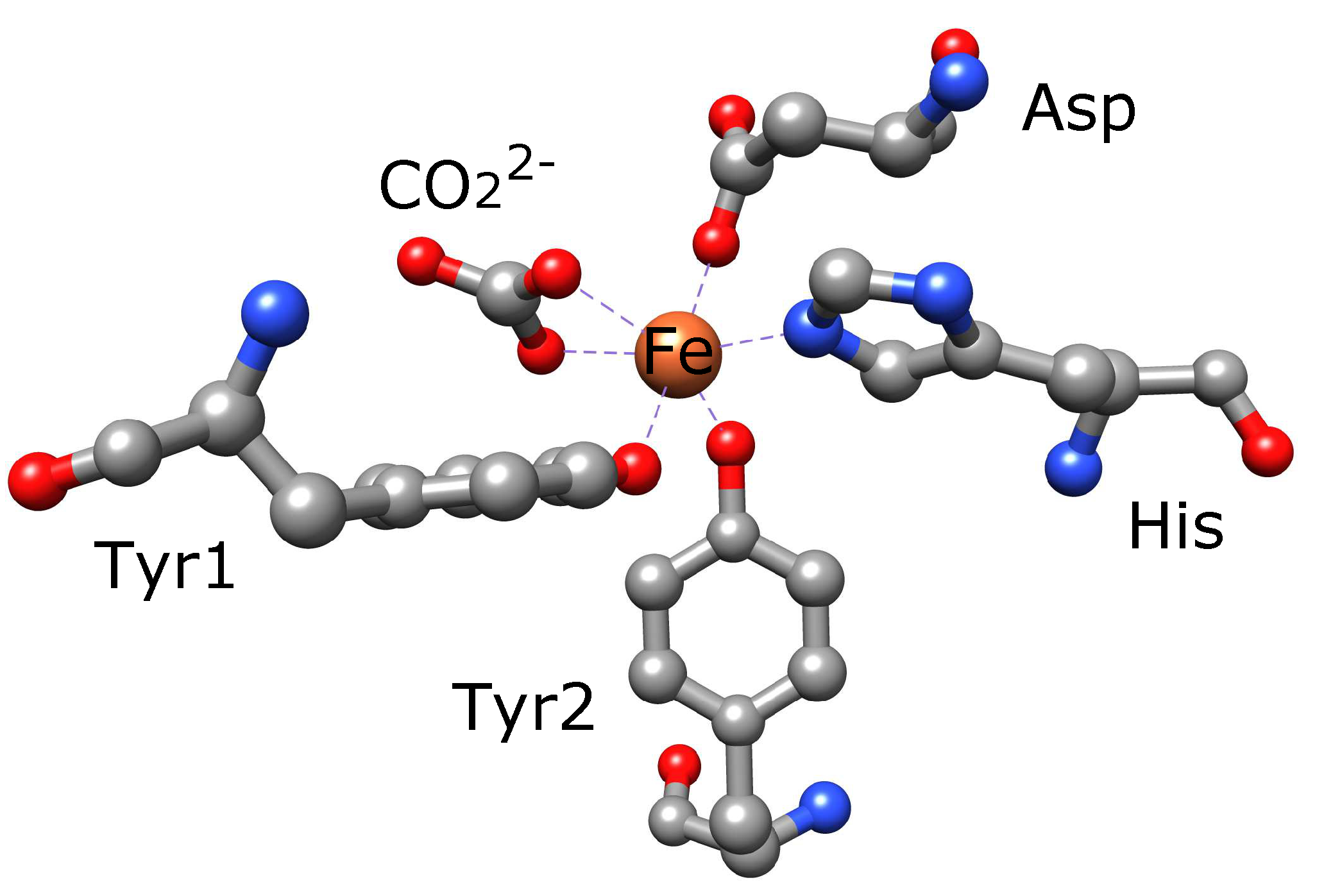
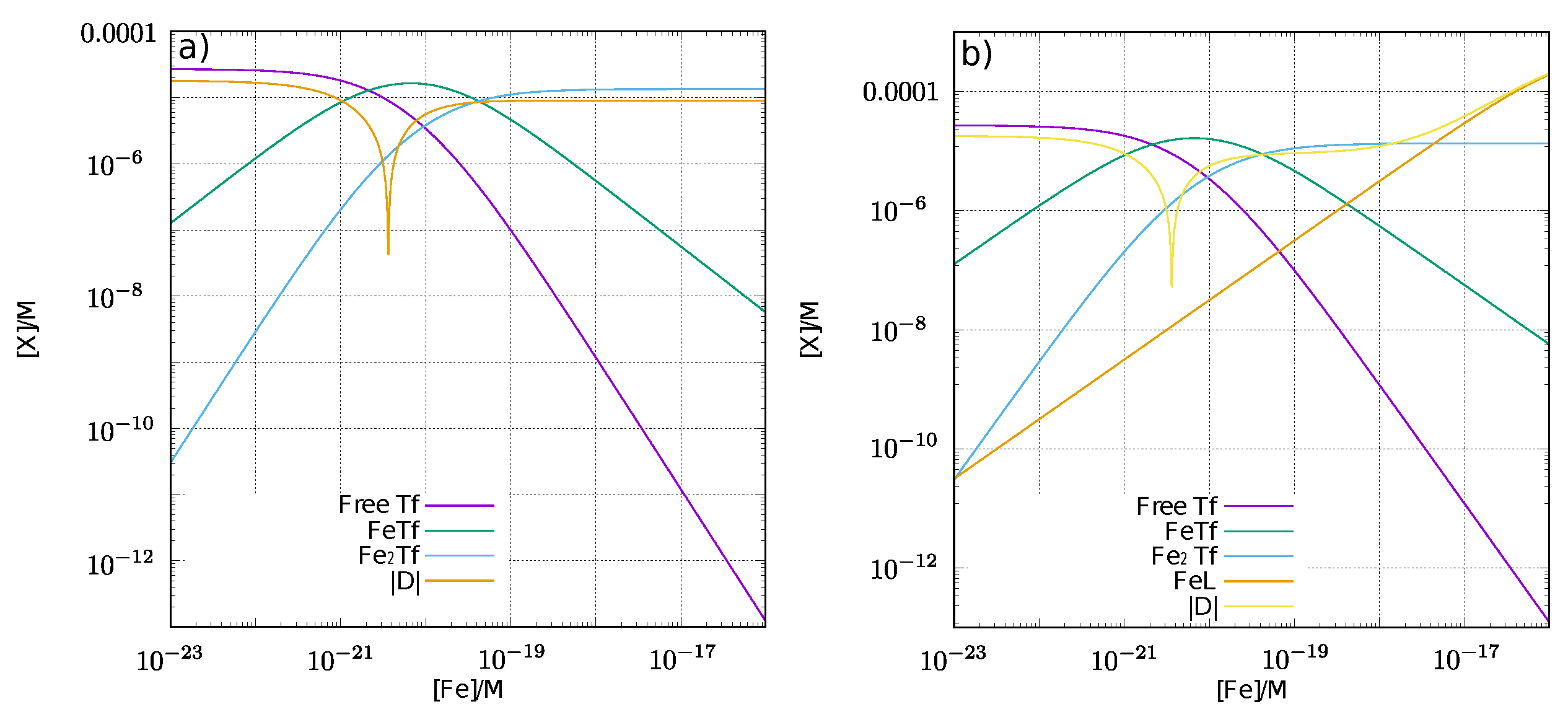
| Redox Couple | Reduction Potential |
|---|---|
| Fe/Fe | 0.771 |
| Fe(OH)/Fe(OH) | −0.56 |
| [Fe(bipy)/[Fe(bipy) | 1.03 |
| [Fe(phen)/[Fe(phen) | 1.147 |
| Fe-nta/Fe-nta | 0.59 |
| Fe-dtpa/Fe-dtpa | 0.03 |
| Fe-edta/Fe-edta | 0.12 |
| Fe-ADP/Fe-ADP | 0.10 |
| Fe-DFO/Fe-DFO | −0.45 |
| Fe-Tf/Fe-Tf | −0.40 |
| pH | K/M | K/M | ||
|---|---|---|---|---|
| 6.7 | 2.3 × 10 | |||
| 7.4 | ||||
| Species | without L/M | with L/M |
|---|---|---|
| free Fe | 3.245 × 10 | 3.242 × 10 |
| free Tf | 10.21 | 10.22 |
| FeTf | 15.57 | 15.57 |
| FeTf | 1.213 | 1.211 |
| Free L | 0 | 500.0 |
| FeL | 0 | 0.006453 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawabata, T. Iron-Induced Oxidative Stress in Human Diseases. Cells 2022, 11, 2152. https://doi.org/10.3390/cells11142152
Kawabata T. Iron-Induced Oxidative Stress in Human Diseases. Cells. 2022; 11(14):2152. https://doi.org/10.3390/cells11142152
Chicago/Turabian StyleKawabata, Teruyuki. 2022. "Iron-Induced Oxidative Stress in Human Diseases" Cells 11, no. 14: 2152. https://doi.org/10.3390/cells11142152
APA StyleKawabata, T. (2022). Iron-Induced Oxidative Stress in Human Diseases. Cells, 11(14), 2152. https://doi.org/10.3390/cells11142152






