Drought Tolerance Strategies and Autophagy in Resilient Wheat Genotypes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drought Stress Treatment
2.2. Measuring the Impact of Drought on Plant Development
2.3. Measuring the Impact of Drought on Photosynthetic Parameters Using Phenomics Platform
2.4. Measuring the Impact of Drought on Photosystem I
2.5. Measuring Peroxisome Abundance
2.6. Microscopy
2.7. Measuring Activity of ROS Scavenging Enzymes
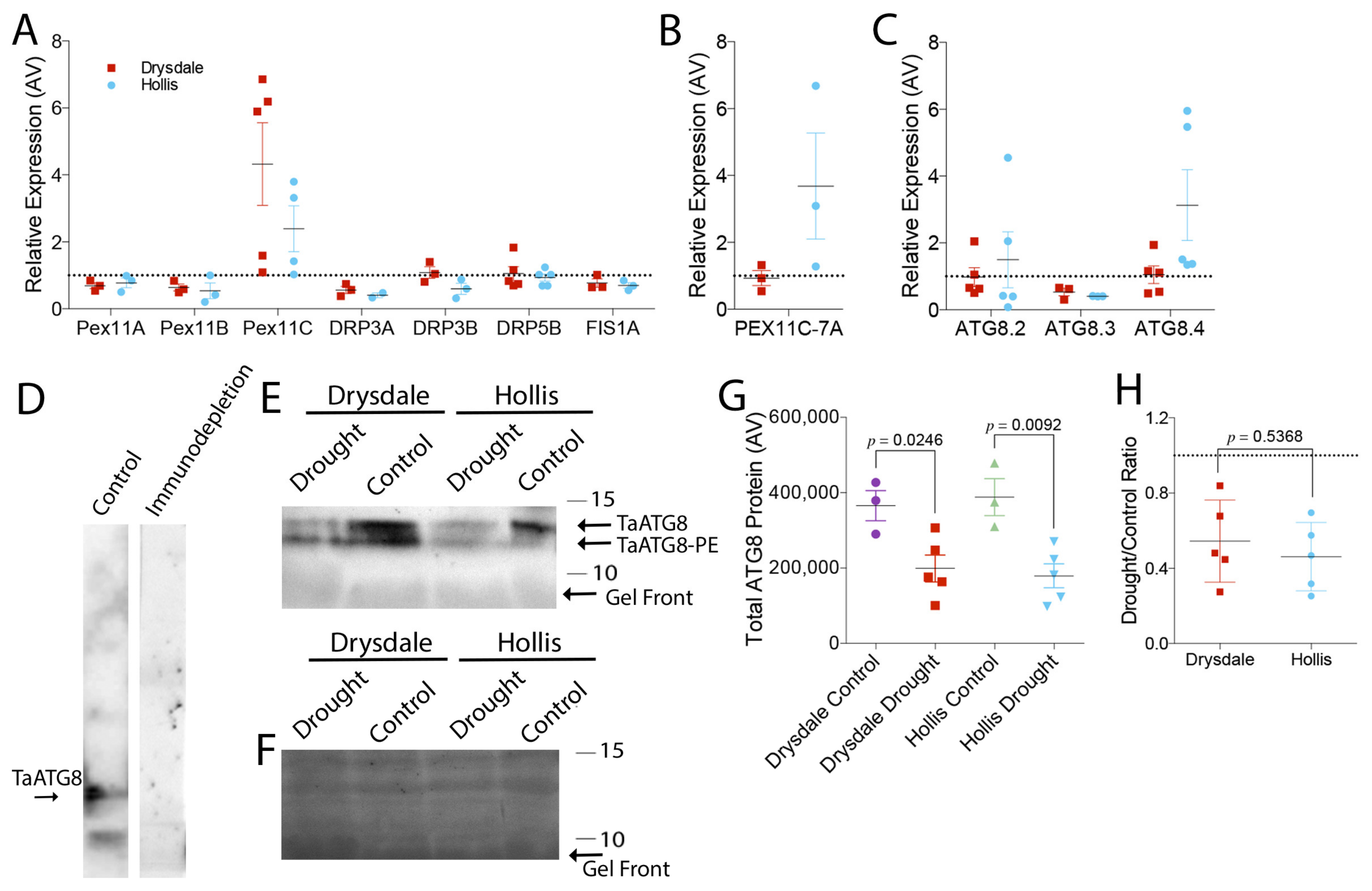
2.8. Analysis of Gene Expression
2.9. RNA-Seq Analysis
2.10. Preparation of Antibodies and Western Blotting
3. Results
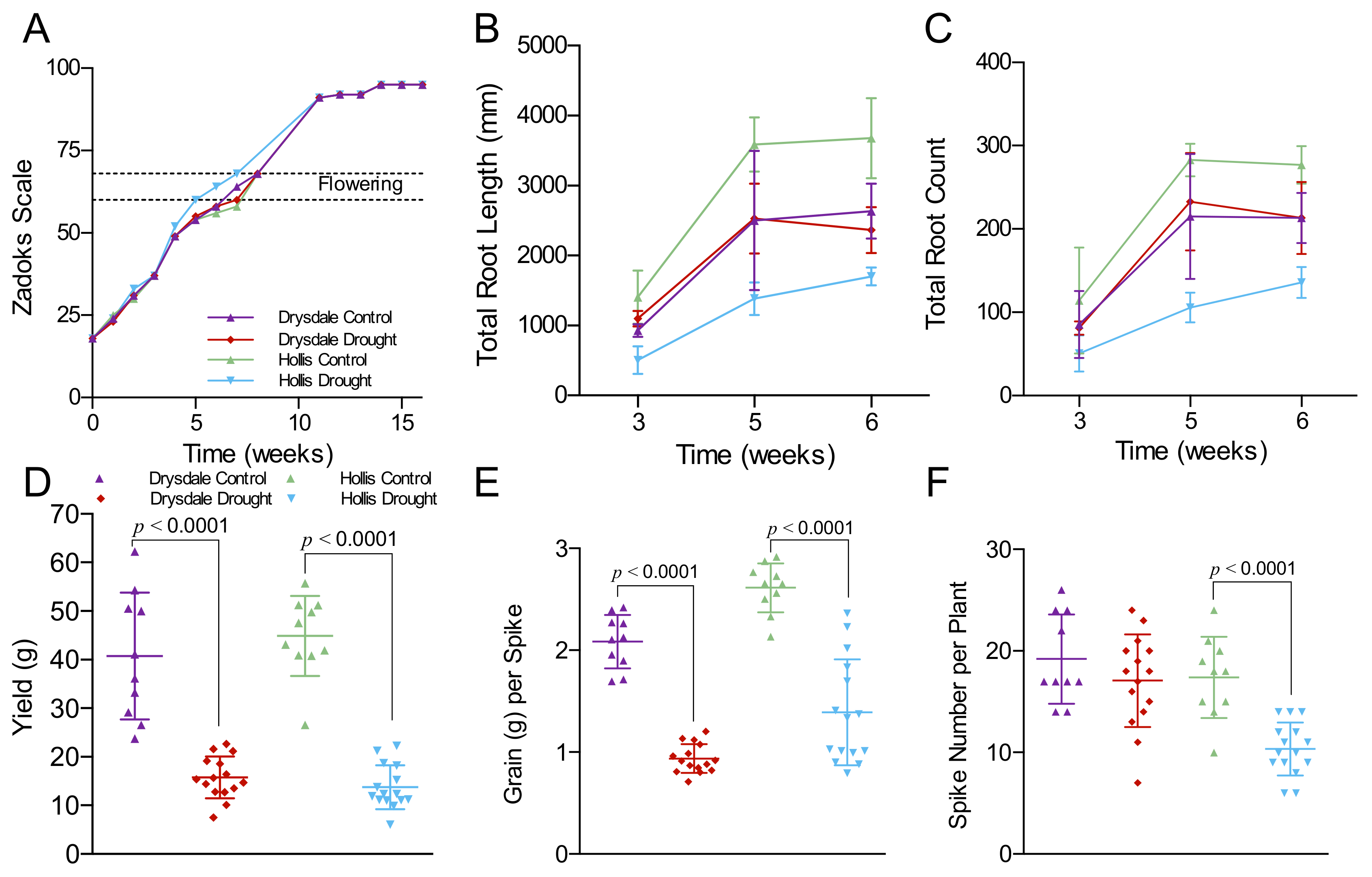
3.1. Impact of Drought on Root Architecture and Yield
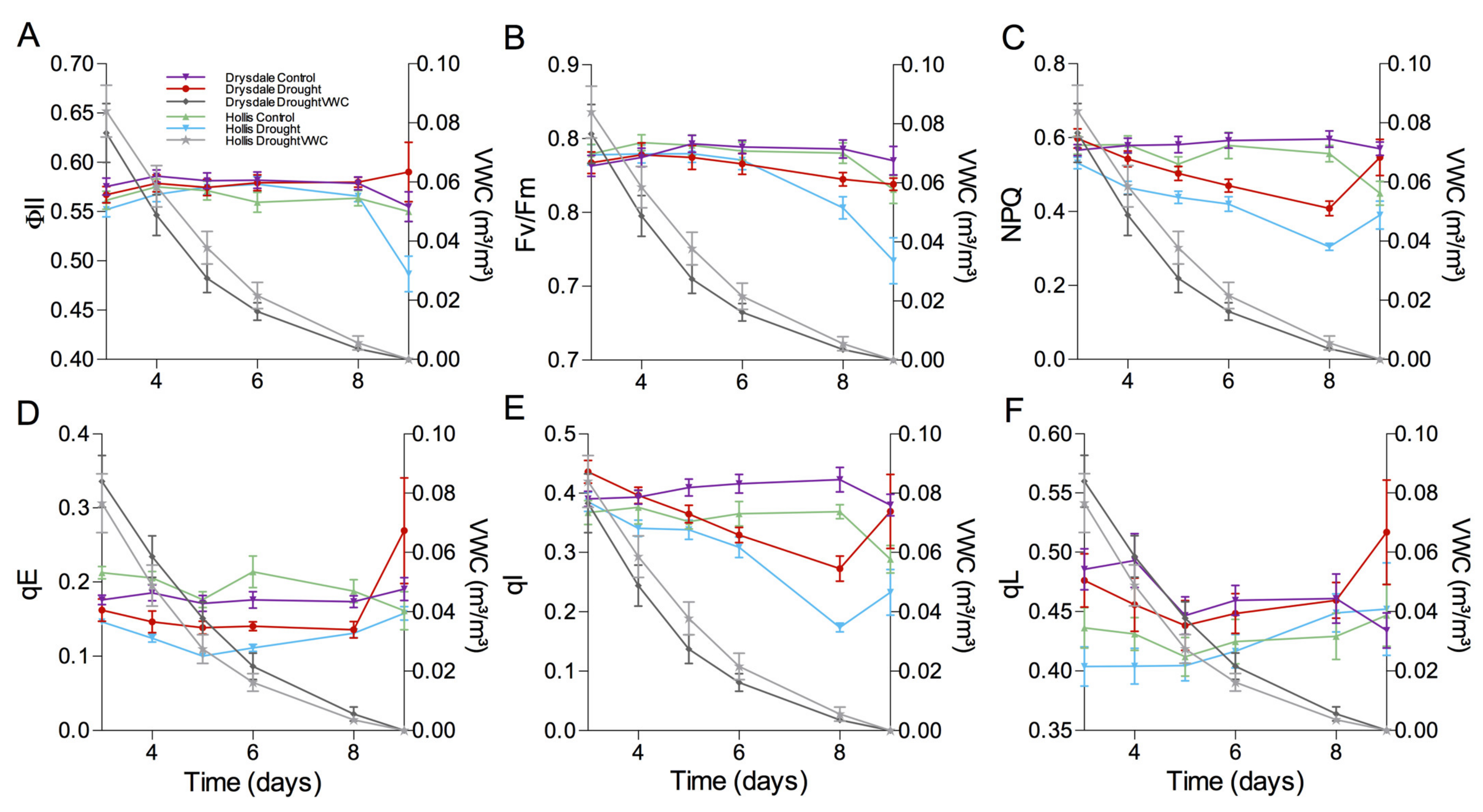
3.2. Impact of Drought on the Photosynthetic Parameters
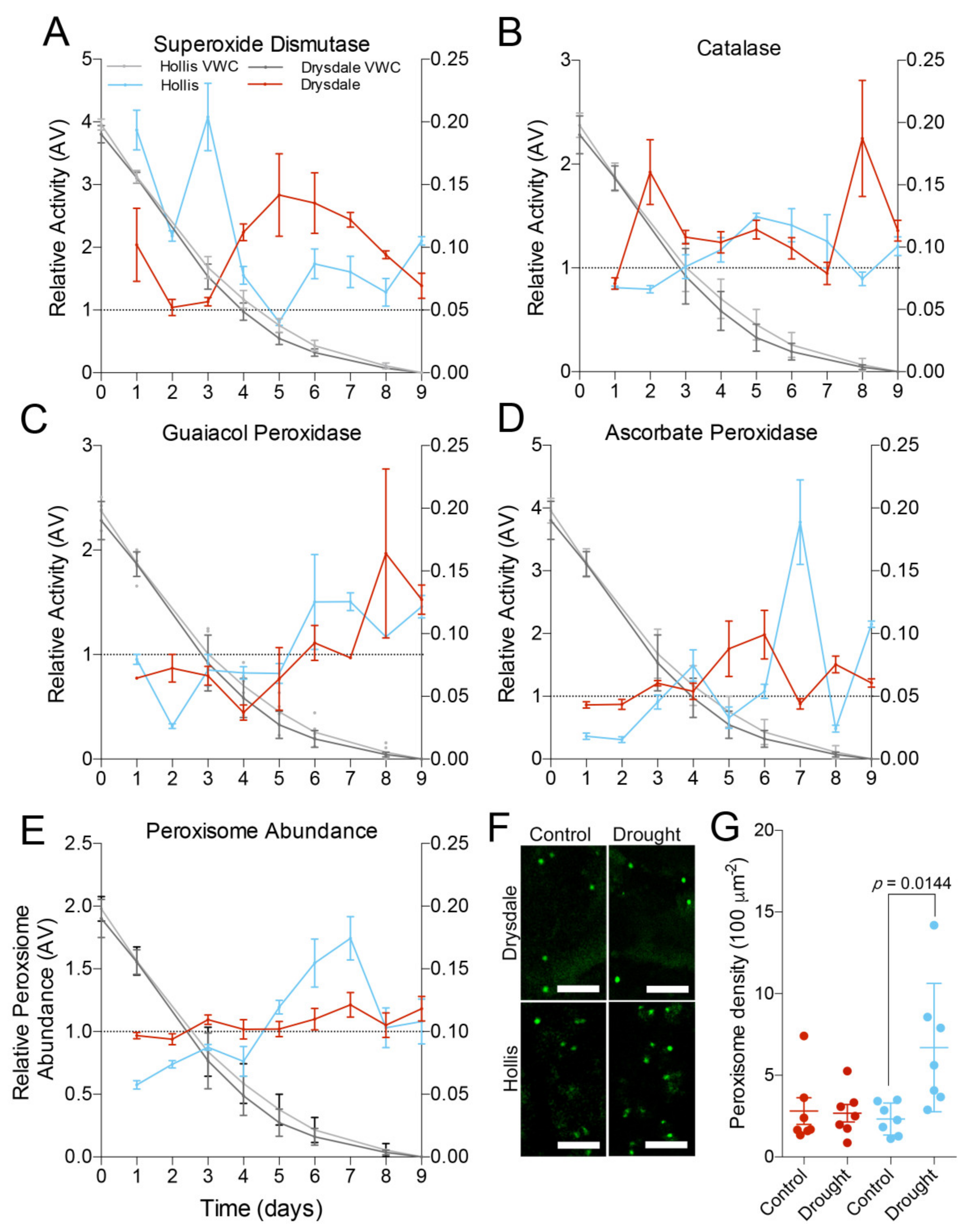
3.3. Activity of the ROS-Scavenging System
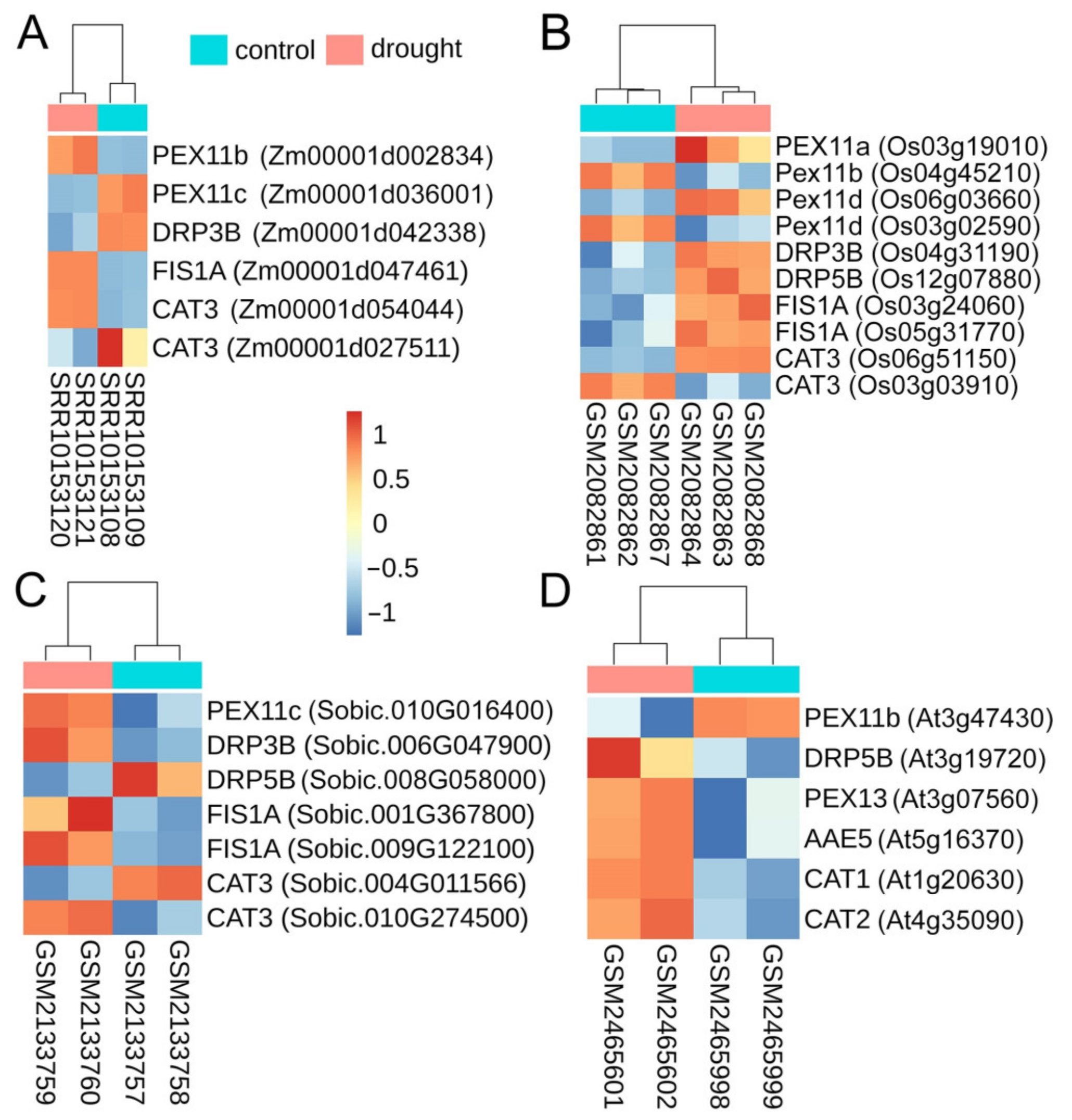
3.4. Impact of Drought on Peroxisomes
4. Discussion
4.1. Drought Escape and Avoidance Mechanisms in Hollis and Drysdale
4.2. Impact of Drought on Photosynthesis
4.3. ROS Scavenging System under Drought
4.4. Role and Regulation of Peroxisomal Homeostasis in Stress Tolerance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wheat Initiative. An International Vision for Wheat Improvement. 2013. Available online: https://www.affrc.maff.go.jp/kokusaikenkyu/attach/pdf/network_detail-1.pdf (accessed on 8 February 2019).
- Araus, J.; Ferrio, J.; Buxo, R.; Voltas, J. The Historical Perspective of Dryland Agriculture: Lessons Learned from 10 000 Years of Wheat Cultivation. J. Exp. Bot. 2006, 58, 131–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NASA Global Climate Change. Effects|Facts—Climate Change: Vital Signs of the Planet. Available online: https://climate.nasa.gov/effects/ (accessed on 8 February 2019).
- Leng, G.; Hall, J. Crop Yield Sensitivity of Global Major Agricultural Countries to Droughts and the Projected Changes in the Future. Sci. Total Environ. 2019, 654, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Zandalinas, S.I.; Fritschi, F.B.; Mittler, R. Global Warming, Climate Change, and Environmental Pollution: Recipe for a Multifactorial Stress Combination Disaster. Trends Plant Sci. 2021, 26, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Liu, B.; Piao, S.; Wang, X.; Lobell, D.B.; Huang, Y.; Huang, M.; Yao, Y.; Bassu, S.; Ciais, P.; et al. Temperature Increase Reduces Global Yields of Major Crops in Four Independent Estimates. Proc. Natl. Acad. Sci. USA 2017, 114, 9326–9331. [Google Scholar] [CrossRef] [Green Version]
- Tricker, P.J.; ElHabti, A.; Schmidt, J.; Fleury, D. The Physiological and Genetic Basis of Combined Drought and Heat Tolerance in Wheat. J. Exp. Bot. 2018, 69, 3195–3210. [Google Scholar] [CrossRef] [Green Version]
- Sallam, A.; Alqudah, A.M.; Dawood, M.F.A.; Baenziger, P.S.; Börner, A. Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research. Int. J. Mol. Sci. 2019, 20, 3137. [Google Scholar] [CrossRef] [Green Version]
- International Wheat Genome Sequencing Consortium (IWGSC). A Chromosome-Based Draft Sequence of the Hexaploid Bread Wheat (Triticum aestivum) Genome. Science 2014, 345, 1251788. [Google Scholar] [CrossRef]
- Juliana, P.; Singh, R.P.; Poland, J.; Shrestha, S.; Huerta-Espino, J.; Govindan, V.; Mondal, S.; Crespo-Herrera, L.A.; Kumar, U.; Joshi, A.K.; et al. Elucidating the Genetics of Grain Yield and Stress-Resilience in Bread Wheat Using a Large-Scale Genome-Wide Association Mapping Study with 55,568 Lines. Sci. Rep. 2021, 11, 5254. [Google Scholar] [CrossRef]
- Verma, A.; Niranjana, M.; Jha, S.K.; Mallick, N.; Agarwal, P.; Vinod. QTL Detection and Putative Candidate Gene Prediction for Leaf Rolling under Moisture Stress Condition in Wheat. Sci. Rep. 2020, 10, 18696. [Google Scholar] [CrossRef]
- Bilgrami, S.S.; Ramandi, H.D.; Shariati, V.; Razavi, K.; Tavakol, E.; Fakheri, B.A.; Mahdi Nezhad, N.; Ghaderian, M. Detection of Genomic Regions Associated with Tiller Number in Iranian Bread Wheat under Different Water Regimes Using Genome-Wide Association Study. Sci. Rep. 2020, 10, 14034. [Google Scholar] [CrossRef]
- Basu, S.; Ramegowda, V.; Kumar, A.; Pereira, A. Plant Adaptation to Drought Stress. F1000Research 2016, 5, 1554. [Google Scholar] [CrossRef]
- Shavrukov, Y.; Kurishbayev, A.; Jatayev, S.; Shvidchenko, V.; Zotova, L.; Koekemoer, F.; de Groot, S.; Soole, K.; Langridge, P. Early Flowering as a Drought Escape Mechanism in Plants: How Can It Aid Wheat Production? Front. Plant Sci. 2017, 8, 1950. [Google Scholar] [CrossRef]
- Fang, Y.; Xiong, L. General Mechanisms of Drought Response and Their Application in Drought Resistance Improvement in Plants. Cell. Mol. Life Sci. 2015, 72, 673–689. [Google Scholar] [CrossRef]
- Voss, I.; Sunil, B.; Scheibe, R.; Raghavendra, A.S. Emerging Concept for the Role of Photorespiration as an Important Part of Abiotic Stress Response. Plant Biol. 2013, 15, 713–722. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Stress-Triggered Redox Signalling: What’s in PROSpect? Plant Cell Environ. 2016, 39, 951–964. [Google Scholar] [CrossRef]
- Waszczak, C.; Carmody, M.; Kangasjärvi, J. Reactive Oxygen Species in Plant Signaling. Annu. Rev. Plant Biol. 2018, 69, 209–236. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Ullah, F.; Zhou, D.X.; Yi, M.; Zhao, Y. Mechanisms of ROS Regulation of Plant Development and Stress Responses. Front. Plant Sci. 2019, 10, 800. [Google Scholar] [CrossRef]
- Held, P. An Introduction to Reactive Oxygen Species Measurement of ROS in Cells; BioTek Instruments Inc.: Winooski, VT, USA, 2012. [Google Scholar]
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive Oxygen Species Homeostasis and Signalling during Drought and Salinity Stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef]
- Caverzan, A.; Casassola, A.; Brammer, S.P. Antioxidant Responses of Wheat Plants under Stress. Genet. Mol. Biol. 2016, 39, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Mittler, R. ROS Are Good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Demidchik, V. Mechanisms of Oxidative Stress in Plants: From Classical Chemistry to Cell Biology. Environ. Exp. Bot. 2015, 109, 212–228. [Google Scholar] [CrossRef]
- Møller, I.M.; Jensen, P.E.; Hansson, A. Oxidative Modifications to Cellular Components in Plants. Annu. Rev. Plant Biol. 2007, 58, 459–481. [Google Scholar] [CrossRef] [Green Version]
- Mittler, R. Oxidative Stress, Antioxidants and Stress Tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- You, J.; Chan, Z. ROS Regulation During Abiotic Stress Responses in Crop Plants. Front. Plant Sci. 2015, 6, 1092. [Google Scholar] [CrossRef] [Green Version]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive Oxygen Species, Abiotic Stress and Stress Combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop Production under Drought and Heat Stress: Plant Responses and Management Options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef] [Green Version]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef] [Green Version]
- Op den Camp, R.G.L. Rapid Induction of Distinct Stress Responses after the Release of Singlet Oxygen in Arabidopsis. Plant Cell Online 2003, 15, 2320–2332. [Google Scholar] [CrossRef] [Green Version]
- Wagner, D. The Genetic Basis of Singlet Oxygen-Induced Stress Responses of Arabidopsis Thaliana. Science 2004, 306, 1183–1185. [Google Scholar] [CrossRef]
- Foyer, C.H.; Shigeoka, S. Understanding Oxidative Stress and Antioxidant Functions to Enhance Photosynthesis. Plant Physiol. 2011, 155, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Smirnoff, N.; Arnaud, D. Hydrogen Peroxide Metabolism and Functions in Plants. New Phytol. 2019, 221, 1197–1214. [Google Scholar] [CrossRef] [PubMed]
- Ślesak, I.; Libik, M.; Karpinska, B.; Karpinski, S.; Miszalski, Z. The Role of Hydrogen Peroxide in Regulation of Plant Metabolism and Cellular Signalling in Response to Environmental Stresses. Acta Biochim. Pol. 2007, 54, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Redox Sensing and Signalling Associated with Reactive Oxygen in Chloroplasts, Peroxisomes and Mitochondria. Physiol. Plant. 2003, 119, 355–364. [Google Scholar] [CrossRef] [Green Version]
- Das, K.; Roychoudhury, A. Reactive Oxygen Species (ROS) and Response of Antioxidants as ROS-Scavengers during Environmental Stress in Plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef] [Green Version]
- Mignolet-Spruyt, L.; Xu, E.; Idänheimo, N.; Hoeberichts, F.A.; Mühlenbock, P.; Brosché, M.; Van Breusegem, F.; Kangasjärvi, J. Spreading the News: Subcellular and Organellar Reactive Oxygen Species Production and Signalling. J. Exp. Bot. 2016, 67, 3831–3844. [Google Scholar] [CrossRef] [Green Version]
- Mhamdi, A.; Noctor, G.; Baker, A. Plant Catalases: Peroxisomal Redox Guardians. Arch. Biochem. Biophys. 2012, 525, 181–194. [Google Scholar] [CrossRef]
- Sofo, A.; Scopa, A.; Nuzzaci, M.; Vitti, A. Ascorbate Peroxidase and Catalase Activities and Their Genetic Regulation in Plants Subjected to Drought and Salinity Stresses. Int. J. Mol. Sci. 2015, 16, 13561–13578. [Google Scholar] [CrossRef] [Green Version]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as Antioxidants in Plants: Location and Functional Significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Anee, T.I.; Parvin, K.; Nahar, K.; Mahmud, J.A.; Fujita, M. Regulation of Ascorbate-Glutathione Pathway in Mitigating Oxidative Damage in Plants under Abiotic Stress. Antioxidants 2019, 8, 384. [Google Scholar] [CrossRef] [Green Version]
- Luna, C.M.; Pastori, G.M.; Driscoll, S.; Groten, K.; Bernard, S.; Foyer, C.H. Drought Controls on H2O2 Accumulation, Catalase (CAT) Activity and CAT Gene Expression in Wheat; Oxford University Press: Oxford, UK, 2005; Volume 56, pp. 417–423. [Google Scholar]
- Nikolaeva, M.K.; Maevskaya, S.N.; Shugaev, A.G.; Bukhov, N.G. Effect of Drought on Chlorophyll Content and Antioxidant Enzyme Activities in Leaves of Three Wheat Cultivars Varying in Productivity. Russ. J. Plant Physiol. 2010, 57, 87–95. [Google Scholar] [CrossRef]
- Tyagi, S.; Shumayla; Madhu; Singh, K.; Upadhyay, S.K. Molecular Characterization Revealed the Role of Catalases under Abiotic and Arsenic Stress in Bread Wheat (Triticum aestivum L.). J. Hazard. Mater. 2021, 403, 123585. [Google Scholar] [CrossRef]
- Wang, Z.-Y.; Li, F.-M.; Xiong, Y.-C.; Xu, B.-C. Soil-Water Threshold Range of Chemical Signals and Drought Tolerance Was Mediated by ROS Homeostasis in Winter Wheat during Progressive Soil Drying. J. Plant Growth Regul. 2008, 27, 309–319. [Google Scholar] [CrossRef]
- Lou, L.; Li, X.; Chen, J.; Li, Y.; Tang, Y.; Lv, J. Photosynthetic and Ascorbate-Glutathione Metabolism in the Flag Leaves as Compared to Spikes under Drought Stress of Winter Wheat (Triticum aestivum L.). PLoS ONE 2018, 13, e0194625. [Google Scholar] [CrossRef] [Green Version]
- Sairam, R.K.; Rao, K.V.; Srivastava, G.C. Differential Response of Wheat Genotypes to Long Term Salinity Stress in Relation to Oxidative Stress, Antioxidant Activity and Osmolyte Concentration. Plant Sci. 2002, 163, 1037–1046. [Google Scholar] [CrossRef]
- Ford, K.L.; Cassin, A.; Bacic, A. Quantitative Proteomic Analysis of Wheat Cultivars with Differing Drought Stress Tolerance. Front. Plant Sci. 2011, 2, 44. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.; Gupta, A.K.; Kaur, N. Differential Responses of Antioxidative Defence System to Long-Term Field Drought in Wheat (Triticum aestivum L.) Genotypes Differing in Drought Tolerance. J. Agron. Crop Sci. 2012, 198, 185–195. [Google Scholar] [CrossRef]
- Ebeed, H.T.; Stevenson, S.R.; Cuming, A.C.; Baker, A. Conserved and Differential Transcriptional Responses of Peroxisome Associated Pathways to Drought, Dehydration and ABA. J. Exp. Bot. 2018, 69, 4971–4985. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, M.; Zhao, Z.; Ren, Y.; Li, Q.; Wang, W. Wheat TaPUB1 Modulates Plant Drought Stress Resistance by Improving Antioxidant Capability. Sci. Rep. 2017, 7, 7549. [Google Scholar] [CrossRef] [Green Version]
- Bowne, J.B.; Erwin, T.A.; Juttner, J.; Schnurbusch, T.; Langridge, P.; Bacic, A.; Roessner, U. Drought Responses of Leaf Tissues from Wheat Cultivars of Differing Drought Tolerance at the Metabolite Level. Mol. Plant 2012, 5, 418–429. [Google Scholar] [CrossRef] [Green Version]
- Nxele, X.; Klein, A.; Ndimba, B.K. Drought and Salinity Stress Alters ROS Accumulation, Water Retention, and Osmolyte Content in Sorghum Plants. S. Afr. J. Bot. 2017, 108, 261–266. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Tian, S.S.; Liu, S.S.; Wang, W.Q.; Sui, N. Energy Dissipation and Antioxidant Enzyme System Protect Photosystem II of Sweet Sorghum under Drought Stress. Photosynthetica 2018, 56, 861–872. [Google Scholar] [CrossRef]
- Varoquaux, N.; Cole, B.; Gao, C.; Pierroz, G.; Baker, C.R.; Patel, D.; Madera, M.; Jeffers, T.; Hollingsworth, J.; Sievert, J.; et al. Transcriptomic Analysis of Field-Droughted Sorghum from Seedling to Maturity Reveals Biotic and Metabolic Responses. Proc. Natl. Acad. Sci. USA 2019, 116, 27124–27132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, H.; Yu, J.; Miao, J.; Li, J.; Zhang, H.; Wang, X.; Liu, P.; Zhao, Y.; Jiang, C.; Yin, Z.; et al. Natural Variation in OsLG3 Increases Drought Tolerance in Rice by Inducing ROS Scavenging. Plant Physiol. 2018, 178, 451–467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, X.M.; Huang, L.F.; Zhang, X.; Wang, M.L.; Xu, G.Y.; Xia, X.J. OsCML4 Improves Drought Tolerance through Scavenging of Reactive Oxygen Species in Rice. J. Plant Biol. 2015, 58, 68–73. [Google Scholar] [CrossRef]
- Duan, J.; Zhang, M.; Zhang, H.; Xiong, H.; Liu, P.; Ali, J.; Li, J.; Li, Z. OsMIOX, a Myo-Inositol Oxygenase Gene, Improves Drought Tolerance through Scavenging of Reactive Oxygen Species in Rice (Oryza sativa L.). Plant Sci. 2012, 196, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Pieters, A.J.; El Souki, S. Effects of Drought during Grain Filling on PS II Activity in Rice. J. Plant Physiol. 2005, 162, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Ijaz, R.; Ejaz, J.; Gao, S.; Liu, T.; Imtiaz, M.; Ye, Z.; Wang, T. Overexpression of Annexin Gene AnnSp2, Enhances Drought and Salt Tolerance through Modulation of ABA Synthesis and Scavenging ROS in Tomato. Sci. Rep. 2017, 7, 12087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, R.; Kong, L.; Yu, X.; Ottosen, C.O.; Zhao, T.; Jiang, F.; Wu, Z. Oxidative Damage and Antioxidant Mechanism in Tomatoes Responding to Drought and Heat Stress. Acta Physiol. Plant. 2019, 41, 1–11. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I.; et al. Drought Induced Changes in Growth, Osmolyte Accumulation and Antioxidant Metabolism of Three Maize Hybrids. Front. Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef]
- Zheng, H.; Yang, Z.; Wang, W.; Guo, S.; Li, Z.; Liu, K.; Sui, N. Transcriptome Analysis of Maize Inbred Lines Differing in Drought Tolerance Provides Novel Insights into the Molecular Mechanisms of Drought Responses in Roots. Plant Physiol. Biochem. 2020, 149, 11–26. [Google Scholar] [CrossRef]
- Chugh, V.; Kaur, N.; Gupta, A.K. Evaluation of Oxidative Stress Tolerance in Maize (Zea mays L.) Seedlings in Response to Drought. Indian J. Biochem. Biophys 2011, 48, 47–53. [Google Scholar]
- Li, B.; Fan, R.; Sun, G.; Sun, T.; Fan, Y.; Bai, S.; Guo, S.; Huang, S.; Liu, J.; Zhang, H.; et al. Flavonoids Improve Drought Tolerance of Maize Seedlings by Regulating the Homeostasis of Reactive Oxygen Species. Plant Soil 2021, 461, 389–405. [Google Scholar] [CrossRef]
- Smertenko, A. Can Peroxisomes Inform Cellular Response to Drought? Trends Plant Sci. 2017, 22, 1005–1007. [Google Scholar] [CrossRef]
- Del Río, L.A.; López-Huertas, E. ROS Generation in Peroxisomes and Its Role in Cell Signaling. Plant Cell Physiol. 2016, 57, pcw076. [Google Scholar] [CrossRef]
- Schrader, M.; Bonekamp, N.A.; Islinger, M. Fission and Proliferation of Peroxisomes. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2012, 1822, 1343–1357. [Google Scholar] [CrossRef] [Green Version]
- Desai, M.; Hu, J. Light Induces Peroxisome Proliferation in Arabidopsis Seedlings through the Photoreceptor Phytochrome A, the Transcription Factor HY5 HOMOLOG, and the Peroxisomal Protein PEROXIN11b. Plant Physiol. 2008, 146, 1117–1127. [Google Scholar] [CrossRef] [Green Version]
- Goto-Yamada, S.; Mano, S.; Yamada, K.; Oikawa, K.; Hosokawa, Y.; Hara-Nishimura, I.; Nishimura, M. Dynamics of the Light-Dependent Transition of Plant Peroxisomes. Plant Cell Physiol. 2015, 56, 1264–1271. [Google Scholar] [CrossRef] [Green Version]
- Oksanen, E.; Häikiö, E.; Sober, J.; Karnosky, D.F. Ozone-Induced H2O2 Accumulation in Field-Grown Aspen and Birch Is Linked to Foliar Ultrastructure and Peroxisomal Activity. New Phytol. 2004, 161, 791–799. [Google Scholar] [CrossRef]
- Fahy, D.; Sanad, M.N.M.E.; Duscha, K.; Lyons, M.; Liu, F.; Bozhkov, P.; Kunz, H.-H.; Hu, J.; Neuhaus, H.E.; Steel, P.G.; et al. Impact of Salt Stress, Cell Death, and Autophagy on Peroxisomes: Quantitative and Morphological Analyses Using Small Fluorescent Probe N-BODIPY. Sci. Rep. 2017, 7, 39069. [Google Scholar] [CrossRef]
- Mitsuya, S.; El-Shami, M.; Sparkes, I.A.; Charlton, W.L.; De Marcos Lousa, C.; Johnson, B.; Baker, A. Salt Stress Causes Peroxisome Proliferation, but Inducing Peroxisome Proliferation Does Not Improve NaCl Tolerance in Arabidopsis Thaliana. PLoS ONE 2010, 5, e9408. [Google Scholar] [CrossRef]
- Castillo, M.C.; Sandalio, L.M.; Del Río, L.A.; León, J. Peroxisome Proliferation, Wound-Activated Responses and Expression of Peroxisome-Associated Genes Are Cross-Regulated but Uncoupled in Arabidopsis Thaliana. Plant Cell Environ. 2008, 31, 492–505. [Google Scholar] [CrossRef]
- Ulloa, R.M.; Raices, M.; MacIntosh, G.C.; Maldonado, S.; Tellez-Inon, M.T. Jasmonic Acid Affects Plant Morphology and Calcium-Dependent Protein Kinase Expression and Activity in Solanum Tuberosum. Physiol. Plant. 2002, 115, 417–427. [Google Scholar] [CrossRef]
- Hinojosa, L.; Sanad, M.N.M.E.; Jarvis, D.E.; Steel, P.; Murphy, K.; Smertenko, A. Impact of Heat and Drought Stress on Peroxisome Proliferation in Quinoa. Plant J. 2019, 99, 1144–1158. [Google Scholar] [CrossRef]
- Sanad, M.N.M.E.; Smertenko, A.; Garland-Campbell, K.A. Differential Dynamic Changes of Reduced Trait Model for Analyzing the Plastic Response to Drought Phases: A Case Study in Spring Wheat. Front. Plant Sci. 2019, 10, 504. [Google Scholar] [CrossRef]
- McCarthy, I.; Romero-Puertas, M.C.; Palma, J.M.; Sandalio, L.M.; Corpas, F.J.; Gómez, M.; Del Río, L.A. Cadmium Induces Senescence Symptoms in Leaf Peroxisomes of Pea Plants. Plant Cell Environ. 2001, 24, 1065–1073. [Google Scholar] [CrossRef]
- Hu, J. Molecular Basis of Peroxisome Division and Proliferation in Plants; Elsevier Inc.: Amsterdam, The Netherlands, 2010; ISSN 1937-6448. [Google Scholar]
- Koch, A.; Schneider, G.; Lüers, G.H.; Schrader, M. Peroxisome Elongation and Constriction but Not Fission Can Occur Independently of Dynamin-like Protein 1. J. Cell Sci. 2004, 117, 3995–4006. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Baker, A.; Bartel, B.; Linka, N.; Mullen, R.T.; Reumann, S.; Zolman, B.K. Plant Peroxisomes: Biogenesis and Function. Plant Cell 2012, 24, 2279–2303. [Google Scholar] [CrossRef] [Green Version]
- Koch, J.; Pranjic, K.; Huber, A.; Ellinger, A.; Hartig, A.; Kragler, F.; Brocard, C. PEX11 Family Members Are Membrane Elongation Factors That Coordinate Peroxisome Proliferation and Maintenance. J. Cell Sci. 2010, 123, 3389–3400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lingard, M.J.; Trelease, R.N. Five Arabidopsis Peroxin 11 Homologs Individually Promote Peroxisome Elongation, Duplication or Aggregation. J. Cell Sci. 2006, 119, 1961–1972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orth, T.; Reumann, S.; Zhang, X.; Fan, J.; Wenzel, D.; Quan, S.; Hu, J. The PEROXIN11 Protein Family Controls Peroxisome Proliferation in Arabidopsis. Plant Cell 2007, 19, 333–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Hu, J. The Arabidopsis Chloroplast Division Protein DYNAMIN-RELATED PROTEIN5B Also Mediates Peroxisome Division. Plant Cell 2010, 22, 431–442. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Hu, J. Two Small Protein Families, DYNAMIN-RELATED PROTEIN3 and FISSION1, Are Required for Peroxisome Fission in Arabidopsis. Plant J. 2009, 57, 146–159. [Google Scholar] [CrossRef]
- Zhang, X.-C.; Hu, J.-P. FISSION1A and FISSION1B Proteins Mediate the Fission of Peroxisomes and Mitochondria in Arabidopsis. Mol. Plant 2008, 1, 1036–1047. [Google Scholar] [CrossRef]
- Cui, P.; Liu, H.; Islam, F.; Li, L.; Farooq, M.A.; Ruan, S.; Zhou, W. OsPEX11, a Peroxisomal Biogenesis Factor 11, Contributes to Salt Stress Tolerance in Oryza sativa. Front. Plant Sci. 2016, 7, 1357. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Hu, J. Using Co-Expression Analysis and Stress-Based Screens to Uncover Arabidopsis Peroxisomal Proteins Involved in Drought Response. PLoS ONE 2015, 10, e0137762. [Google Scholar] [CrossRef]
- Lopez-Huertas, E.; Charlton, W.L.; Johnson, B.; Graham, I.A.; Baker, A. Stress Induces Peroxisome Biogenesis Genes. EMBO J. 2000, 19, 6770–6777. [Google Scholar] [CrossRef]
- Shibata, M.; Oikawa, K.; Yoshimoto, K.; Kondo, M.; Mano, S.; Yamada, K.; Hayashi, M.; Sakamoto, W.; Ohsumi, Y.; Nishimura, M. Highly Oxidized Peroxisomes Are Selectively Degraded via Autophagy in Arabidopsis. Plant Cell 2013, 25, 4967–4983. [Google Scholar] [CrossRef] [Green Version]
- Young, P.G.; Bartel, B. Pexophagy and Peroxisomal Protein Turnover in Plants. Biochim. Biophys. Acta 2016, 1863, 999–1005. [Google Scholar] [CrossRef]
- Farmer, L.M.; Rinaldi, M.A.; Young, P.G.; Danan, C.H.; Burkhart, S.E.; Bartel, B. Disrupting Autophagy Restores Peroxisome Function to an Arabidopsis Lon2 Mutant and Reveals a Role for the LON2 Protease in Peroxisomal Matrix Protein Degradation. Plant Cell 2013, 25, 4085–4100. [Google Scholar] [CrossRef] [Green Version]
- Calero-Muñoz, N.; Exposito-Rodriguez, M.; Collado-Arenal, A.M.; Rodríguez-Serrano, M.; Laureano-Marín, A.M.; Santamaría, M.E.; Gotor, C.; Díaz, I.; Mullineaux, P.M.; Romero-Puertas, M.C.; et al. Cadmium Induces Reactive Oxygen Species-Dependent Pexophagy in Arabidopsis Leaves. Plant Cell Environ. 2019, 42, 2696–2714. [Google Scholar] [CrossRef]
- Sun, X.; Wang, P.; Jia, X.; Huo, L.; Che, R.; Ma, F. Improvement of Drought Tolerance by Overexpressing MdATG18a Is Mediated by Modified Antioxidant System and Activated Autophagy in Transgenic Apple. Plant Biotechnol. J. 2018, 16, 545–557. [Google Scholar] [CrossRef] [Green Version]
- Bao, Y.; Song, W.-M.; Wang, P.; Yu, X.; Li, B.; Jiang, C.; Shiu, S.-H.; Zhang, H.; Bassham, D.C. COST1 Regulates Autophagy to Control Plant Drought Tolerance. Proc. Natl. Acad. Sci. USA 2020, 117, 7482–7493. [Google Scholar] [CrossRef]
- Liu, Y.; Xiong, Y.; Bassham, D.C. Autophagy Is Required for Tolerance of Drought and Salt Stress in Plants. Autophagy 2009, 5, 954–963. [Google Scholar] [CrossRef] [Green Version]
- Condon, A.G.; Richards, R.A.; Rebetzke, G.J.; Farquhar, G.D. Breeding for High Water-Use Efficiency. J. Exp. Bot. 2004, 55, 2447–2460. [Google Scholar] [CrossRef] [Green Version]
- Kidwell, K.K.; Shelton, G.B.; DeMacon, V.L.; Burns, J.W.; Carter, B.P.; Morris, C.F.; Chen, X.M.; Bosque-Perez, N.A. Registration of ‘Hollis’ Wheat. Crop Sci. 2004, 44, 1871–1872. [Google Scholar] [CrossRef]
- Zadoks, J.C.; Chang, T.T.; Konzak, C.F. A Decimal Code for the Growth Stages of Cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Nilkens, M.; Kress, E.; Lambrev, P.; Miloslavina, Y.; Müller, M.; Holzwarth, A.R.; Jahns, P. Identification of a Slowly Inducible Zeaxanthin-Dependent Component of Non-Photochemical Quenching of Chlorophyll Fluorescence Generated under Steady-State Conditions in Arabidopsis. Biochim. Biophys. Acta BBA Bioenerg. 2010, 1797, 466–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef] [Green Version]
- Tietz, S.; Puthiyaveetil, S.; Enlow, H.M.; Yarbrough, R.; Wood, M.; Semchonok, D.A.; Lowry, T.; Li, Z.; Jahns, P.; Boekema, E.J.; et al. Functional Implications of Photosystem II Crystal Formation in Photosynthetic Membranes. J. Biol. Chem. 2015, 290, 14091–14106. [Google Scholar] [CrossRef] [Green Version]
- Klughammer, C.; Schreiber, U. Deconvolution of Ferredoxin, Plastocyanin, and P700 Transmittance Changes in Intact Leaves with a New Type of Kinetic LED Array Spectrophotometer. Photosynth. Res. 2016, 128, 195–214. [Google Scholar] [CrossRef] [Green Version]
- Bradford, M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in Vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen Peroxide Is Scavenged by Ascorbate-Specific Peroxidase in Spinach Chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Dias, M.A.; Costa, M.M. Effect of Low Salt Concentrations on Nitrate Reductase and Peroxidase of Sugar Beet Leaves. J. Exp. Bot. 1983, 34, 537–543. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide Dismutases. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef]
- Giménez, M.J.; Pistón, F.; Atienza, S.G. Identification of Suitable Reference Genes for Normalization of QPCR Data in Comparative Transcriptomics Analyses in the Triticeae. Planta 2011, 233, 163–173. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Pertea, M.; Kim, D.; Pertea, G.M.; Leek, J.T.; Salzberg, S.L. Transcript-Level Expression Analysis of RNA-Seq Experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 2016, 11, 1650–1667. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. The Sequence Alignment/Map Format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Kaur, N.; Hu, J. Defining the Plant Peroxisomal Proteome: From Arabidopsis to Rice. Front. Plant Sci. 2011, 2, 103. [Google Scholar] [CrossRef] [Green Version]
- Emms, D.M.; Kelly, S. OrthoFinder: Phylogenetic Orthology Inference for Comparative Genomics. Genome Biol. 2019, 20, 238. [Google Scholar] [CrossRef] [Green Version]
- Walter, W.; Sánchez-Cabo, F.; Ricote, M. GOplot: An R Package for Visually Combining Expression Data with Functional Analysis. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef]
- Luo, W.; Brouwer, C. Pathview: An R/Bioconductor Package for Pathway-Based Data Integration and Visualization. Bioinformatics 2013, 29, 1830–1831. [Google Scholar] [CrossRef] [Green Version]
- Smertenko, A.P.; Chang, H.Y.; Wagner, V.; Kaloriti, D.; Fenyk, S.; Sonobe, S.; Lloyd, C.; Hauser, M.T.; Hussey, P.J. The Arabidopsis Microtubule-Associated Protein AtMAP65-1: Molecular Analysis of Its Microtubule Bundling Activity. Plant Cell 2004, 16, 2035–2047. [Google Scholar] [CrossRef] [Green Version]
- Smertenko, A.P.; Kaloriti, D.; Chang, H.Y.; Fiserova, J.; Opatrny, Z.; Hussey, P.J. The C-Terminal Variable Region Specifies the Dynamic Properties of Arabidopsis Microtubule-Associated Protein MAP65 Isotypes. Plant Cell 2008, 20, 3346–3358. [Google Scholar] [CrossRef] [Green Version]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [Green Version]
- Manschadi, A.M.; Christopher, J.; de Voil, P.; Hammer, G.L. The Role of Root Architectural Traits in Adaptation of Wheat to Water-Limited Environments. Funct. Plant Biol. 2006, 33, 823. [Google Scholar] [CrossRef] [Green Version]
- Ghimire, B.; Hulbert, S.H.; Steber, C.M.; Garland-Campbell, K.; Sanguinet, K.A. Characterization of Root Traits for Improvement of Spring Wheat in the Pacific Northwest. Agron. J. 2020, 112, 228–240. [Google Scholar] [CrossRef]
- Ma, J.; Stiller, J.; Wei, Y.; Zheng, Y.-L.; Devos, K.M.; Doležel, J.; Liu, C. Extensive Pericentric Rearrangements in the Bread Wheat (Triticum aestivum L.) Genotype “Chinese Spring” Revealed from Chromosome Shotgun Sequence Data. Genome Biol. Evol. 2014, 6, 3039–3048. [Google Scholar] [CrossRef]
- Zhou, C.; Dong, Z.; Zhang, T.; Wu, J.; Yu, S.; Zeng, Q.; Han, D.; Tong, W. Genome-Scale Analysis of Homologous Genes among Subgenomes of Bread Wheat (Triticum aestivum L.). Int. J. Mol. Sci. 2020, 21, 3015. [Google Scholar] [CrossRef] [PubMed]
- Üstün, S.; Hafrén, A.; Liu, Q.; Marshall, R.S.; Minina, E.A.; Bozhkov, P.V.; Vierstra, R.D.; Hofius, D. Bacteria Exploit Autophagy for Proteasome Degradation and Enhanced Virulence in Plants. Plant Cell 2018, 30, 668–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yue, W.; Nie, X.; Cui, L.; Zhi, Y.; Zhang, T.; Du, X.; Song, W. Genome-Wide Sequence and Expressional Analysis of Autophagy Gene Family in Bread Wheat (Triticum aestivum L.). J. Plant Physiol. 2018, 229, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Reumann, S.; Babujee, L.; Changle, M.; Wienkoop, S.; Siemsen, T.; Antonicelli, G.E.; Rasche, N.; Lüder, F.; Weckwerth, W.; Jahnd, O. Proteome Analysis of Arabidopsis Leaf Peroxisomes Reveals Novel Targeting Peptides, Metabolic Pathways, and Defense Mechanisms. Plant Cell 2007, 19, 3170–3193. [Google Scholar] [CrossRef] [Green Version]
- Bussell, J.D.; Behrens, C.; Ecke, W.; Eubel, H. Arabidopsis Peroxisome Proteomics. Front. Plant Sci. 2013, 4, 101. [Google Scholar] [CrossRef] [Green Version]
- Condon, A.G.; Farquhar, G.D.; Rebetzke, G.J.; Richards, R.A. The Application of Carbon Isotope Discrimination in Cereal Improvement for Water-Limited Environments. In Drought Adaptation in Cereals; Ribaut, J.M., Ed.; Food Products Press: Binghamton, NY, USA, 2006; pp. 171–222. [Google Scholar]
- Ji, X.; Shiran, B.; Wan, J.; Lewis, D.C.; Jenkins, C.L.D.; Condon, A.G.; Richards, R.A.; Dolferus, R. Importance of Pre-Anthesis Anther Sink Strength for Maintenance of Grain Number during Reproductive Stage Water Stress in Wheat. Plant Cell Environ. 2010, 33, 926–942. [Google Scholar] [CrossRef]
- Rivera-Amado, C.; Molero, G.; Trujillo-Negrellos, E.; Reynolds, M.; Foulkes, J. Estimating Organ Contribution to Grain Filling and Potential for Source Upregulation in Wheat Cultivars with a Contrasting Source-Sink Balance. Agronomy 2020, 10, 1527. [Google Scholar] [CrossRef]
- Asseng, S.; Kassie, B.T.; Labra, M.H.; Amador, C.; Calderini, D.F. Simulating the Impact of Source-Sink Manipulations in Wheat. Field Crops Res. 2017, 202, 47–56. [Google Scholar] [CrossRef]
- Schapendonk, A.H.C.M.; Xu, H.Y.; Van Der Putten, P.E.L.; Spiertz, J.H.J. Heat-Shock Effects on Photosynthesis and Sink-Source Dynamics in Wheat (Triticum aestivum L.). NJAS Wagening. J. Life Sci. 2007, 55, 37–54. [Google Scholar] [CrossRef] [Green Version]
- Uga, Y.; Sugimoto, K.; Ogawa, S.; Rane, J.; Ishitani, M.; Hara, N.; Kitomi, Y.; Inukai, Y.; Ono, K.; Kanno, N.; et al. Control of Root System Architecture by DEEPER ROOTING 1 Increases Rice Yield under Drought Conditions. Nat. Genet. 2013, 45, 1097–1102. [Google Scholar] [CrossRef]
- Henry, A.; Gowda, V.R.P.; Torres, R.O.; McNally, K.L.; Serraj, R. Variation in Root System Architecture and Drought Response in Rice (Oryza sativa): Phenotyping of the OryzaSNP Panel in Rainfed Lowland Fields. Field Crops Res. 2011, 120, 205–214. [Google Scholar] [CrossRef]
- Lynch, J.P. Steep, Cheap and Deep: An Ideotype to Optimize Water and N Acquisition by Maize Root Systems. Ann. Bot. 2013, 112, 347–357. [Google Scholar] [CrossRef] [Green Version]
- Sebastian, J.; Yee, M.-C.; Goudinho Viana, W.; Rellán-Álvarez, R.; Feldman, M.; Priest, H.D.; Trontin, C.; Lee, T.; Jiang, H.; Baxter, I.; et al. Grasses Suppress Shoot-Borne Roots to Conserve Water during Drought. Proc. Natl. Acad. Sci. USA 2016, 113, 8861–8866. [Google Scholar] [CrossRef] [Green Version]
- Burton, A.L.; Johnson, J.M.; Foerster, J.M.; Hirsch, C.N.; Buell, C.R.; Hanlon, M.T.; Kaeppler, S.M.; Brown, K.M.; Lynch, J.P. QTL Mapping and Phenotypic Variation for Root Architectural Traits in Maize (Zea mays L.). Theor. Appl. Genet. 2014, 127, 2293–2311. [Google Scholar] [CrossRef]
- Al-Naggar, A.M.M.; Shafik, M.M.; Elsheikh, M.O.A. Putative Mechanisms of Drought Tolerance in Maize (Zea mays L.) via Root System Architecture Traits. Annu. Res. Rev. Biol. 2019, 32, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Saradadevi, R.; Bramley, H.; Palta, J.A.; Edwards, E.; Siddique, K.H.M. Root Biomass in the Upper Layer of the Soil Profile Is Related to the Stomatal Response of Wheat as the Soil Dries. Funct. Plant Biol. 2015, 43, 62–74. [Google Scholar] [CrossRef]
- Manschadi, A.M.; Hammer, G.L.; Christopher, J.T.; DeVoil, P. Genotypic Variation in Seedling Root Architectural Traits and Implications for Drought Adaptation in Wheat (Triticum aestivum L.). Plant Soil 2008, 303, 115–129. [Google Scholar] [CrossRef]
- Dalal, M.; Sahu, S.; Tiwari, S.; Rao, A.R.; Gaikwad, K. Transcriptome Analysis Reveals Interplay between Hormones, ROS Metabolism and Cell Wall Biosynthesis for Drought-Induced Root Growth in Wheat. Plant Physiol. Biochem. 2018, 130, 482–492. [Google Scholar] [CrossRef]
- Van der Weele, C.M.; Spollen, W.G.; Sharp, R.E.; Baskin, T.I. Growth of Arabidopsis Thaliana Seedlings under Water Deficit Studied by Control of Water Potential in Nutrient-agar Media. J. Exp. Bot. 2000, 51, 1555–1562. [Google Scholar] [CrossRef] [Green Version]
- Yamaguchi, M.; Valliyodan, B.; Zhang, J.; Lenoble, M.E.; YU, O.; Rogers, E.E.; Nguyen, H.T.; Sharp, R.E. Regulation of Growth Response to Water Stress in the Soybean Primary Root. I. Proteomic Analysis Reveals Region-Specific Regulation of Phenylpropanoid Metabolism and Control of Free Iron in the Elongation Zone. Plant Cell Environ. 2010, 33, 223–243. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Sharp, R.E. Complexity and Coordination of Root Growth at Low Water Potentials: Recent Advances from Transcriptomic and Proteomic Analyses. Plant Cell Environ. 2010, 33, 590–603. [Google Scholar] [CrossRef]
- Song, L.; Prince, S.; Valliyodan, B.; Joshi, T.; Maldonado dos Santos, J.V.; Wang, J.; Lin, L.; Wan, J.; Wang, Y.; Xu, D.; et al. Genome-Wide Transcriptome Analysis of Soybean Primary Root under Varying Water-Deficit Conditions. BMC Genom. 2016, 17, 57. [Google Scholar] [CrossRef] [Green Version]
- Sharp, R.E.; Silk, W.K.; Hsiao, T.C. Growth of the Maize Primary Root at Low Water Potentials. Plant Physiol. 1988, 87, 50–57. [Google Scholar] [CrossRef] [Green Version]
- Schoppach, R.; Sadok, W. Differential Sensitivities of Transpiration to Evaporative Demand and Soil Water Deficit among Wheat Elite Cultivars Indicate Different Strategies for Drought Tolerance. Environ. Exp. Bot. 2012, 84, 1–10. [Google Scholar] [CrossRef]
- Murchie, E.H.; Lawson, T. Chlorophyll Fluorescence Analysis: A Guide to Good Practice and Understanding Some New Applications. J. Exp. Bot. 2013, 64, 3983–3998. [Google Scholar] [CrossRef] [Green Version]
- Eisenhut, M.; Bräutigam, A.; Timm, S.; Florian, A.; Tohge, T.; Fernie, A.R.; Bauwe, H.; Weber, A.P.M. Photorespiration Is Crucial for Dynamic Response of Photosynthetic Metabolism and Stomatal Movement to Altered CO2 Availability. Mol. Plant 2017, 10, 47–61. [Google Scholar] [CrossRef] [Green Version]
- Guo, P.; Baum, M.; Varshney, R.K.; Graner, A.; Grando, S.; Ceccarelli, S. QTLs for Chlorophyll and Chlorophyll Fluorescence Parameters in Barley under Post-Flowering Drought. Euphytica 2008, 163, 203–214. [Google Scholar] [CrossRef]
- Long, S.P.; Humphries, S.; Falkowski, P.G. Photoinhibition of Photosynthesis in Nature. Annu. Rev. Plant Biol. 1994, 45, 633–662. [Google Scholar] [CrossRef]
- Murata, N.; Takahashi, S.; Nishiyama, Y.; Allakhverdiev, S.I. Photoinhibition of Photosystem II under Environmental Stress. Biochim. Biophys. Acta BBA Bioenerg. 2007, 1767, 414–421. [Google Scholar] [CrossRef] [Green Version]
- Mishra, K.B.; Iannacone, R.; Petrozza, A.; Mishra, A.; Armentano, N.; La Vecchia, G.; Trtílek, M.; Cellini, F.; Nedbal, L. Engineered Drought Tolerance in Tomato Plants Is Reflected in Chlorophyll Fluorescence Emission. Plant Sci. 2012, 182, 79–86. [Google Scholar] [CrossRef]
- Efeoǧlu, B.; Ekmekçi, Y.; Çiçek, N. Physiological Responses of Three Maize Cultivars to Drought Stress and Recovery. S. Afr. J. Bot. 2009, 75, 34–42. [Google Scholar] [CrossRef] [Green Version]
- Faseela, P.; Sinisha, A.K.; Brestič, M.; Puthur, J.T. Chlorophyll a Fluorescence Parameters as Indicators of a Particular Abiotistress in Rice. Photosynthetica 2020, 58, 293–300. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Guo, P.; Michael, B.; Stefania, G.; Salvatore, C. Evaluation of Chlorophyll Content and Fluorescence Parameters as Indicators of Drought Tolerance in Barley. Agric. Sci. China 2006, 5, 751–757. [Google Scholar] [CrossRef]
- Kohzuma, K.; Cruz, J.A.; Akashi, K.; Hoshiyasu, S.; Munekage, Y.N.; Yokota, A.; Kramer, D.M. The Long-Term Responses of the Photosynthetic Proton Circuit to Drought. Plant Cell Environ. 2009, 32, 209–219. [Google Scholar] [CrossRef]
- Hura, T.; Hura, K.; Ostrowska, A.; Gadzinowska, J.; Grzesiak, M.T.; Dziurka, K.; Dubas, E. Rieske Iron-Sulfur Protein of Cytochrome-B6f Is Involved in Plant Recovery after Drought Stress. Environ. Exp. Bot. 2018, 156, 228–239. [Google Scholar] [CrossRef]
- Wada, S.; Takagi, D.; Miyake, C.; Makino, A.; Suzuki, Y. Responses of the Photosynthetic Electron Transport Reactions Stimulate the Oxidation of the Reaction Center Chlorophyll of Photosystem I, P700, under Drought and High Temperatures in Rice. Int. J. Mol. Sci. 2019, 20, 2068. [Google Scholar] [CrossRef] [Green Version]
- Baniulis, D.; Hasan, S.S.; Stofleth, J.T.; Cramer, W.A. Mechanism of Enhanced Superoxide Production in the Cytochrome b 6 f Complex of Oxygenic Photosynthesis. Biochemistry 2013, 52, 8975–8983. [Google Scholar] [CrossRef] [Green Version]
- Hou, J.; Huang, X.; Sun, W.; Du, C.; Wang, C.; Xie, Y.; Ma, Y.; Ma, D. Accumulation of Water-Soluble Carbohydrates and Gene Expression in Wheat Stems Correlates with Drought Resistance. J. Plant Physiol. 2018, 231, 182–191. [Google Scholar] [CrossRef]
- Marček, T.; Hamow, K.Á.; Végh, B.; Janda, T.; Darko, E. Metabolic Response to Drought in Six Winter Wheat Genotypes; Public Library of Science: San Francisco, CA, USA, 2019; Volume 14, p. e0212411. [Google Scholar]
- Goggin, D.E.; Setter, T.L. Fructosyltransferase Activity and Fructan Accumulation during Development in Wheat Exposed to Terminal Drought. Funct. Plant Biol. 2004, 31, 11–21. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Oxygen Processing in Photosynthesis: Regulation and Signalling. New Phytol. 2000, 146, 359–388. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Serrano, M.; Romero-Puertas, M.C.; Sanz-Fernández, M.; Hu, J.; Sandalio, L.M. Peroxisomes Extend Peroxules in a Fast Response to Stress via a Reactive Oxygen Species-Mediated Induction of the Peroxin PEX11a. Plant Physiol. 2016, 171, 1665–1674. [Google Scholar] [CrossRef] [Green Version]
- Voitsekhovskaja, O.V.; Schiermeyer, A.; Reumann, S. Plant Peroxisomes Are Degraded by Starvation-Induced and Constitutive Autophagy in Tobacco BY-2 Suspension-Cultured Cells. Front. Plant Sci. 2014, 5, 629. [Google Scholar] [CrossRef] [Green Version]
- Bassham, D.C. Methods for Analysis of Autophagy in Plants. Methods 2015, 75, 181–188. [Google Scholar] [CrossRef]
- Klionsky, D.J.; Abdel-Aziz, A.K.; Abdelfatah, S.; Abdellatif, M.; Abdoli, A.; Abel, S.; Abeliovich, H.; Abildgaard, M.H.; Abudu, Y.P.; Acevedo-Arozena, A.; et al. Guidelines for the Use and Interpretation of Assays for Monitoring Autophagy (4th Edition). Autophagy 2021, 17, 1–382. [Google Scholar] [CrossRef]
- Xiong, Y.; Contento, A.L.; Nguyen, P.Q.; Bassham, D.C. Degradation of Oxidized Proteins by Autophagy during Oxidative Stress in Arabidopsis. Plant Physiol. 2007, 143, 291–299. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Wang, J.; Yu, J.-Q.; Chen, Z. Role and Regulation of Autophagy in Heat Stress Responses of Tomato Plants. Front. Plant Sci. 2014, 5, 174. [Google Scholar] [CrossRef] [Green Version]
- Signorelli, S.; Tarkowski, Ł.P.; Van den Ende, W.; Bassham, D.C. Linking Autophagy to Abiotic and Biotic Stress Responses. Trends Plant Sci. 2019, 24, 413–430. [Google Scholar] [CrossRef] [Green Version]
- Su, T.; Li, X.; Yang, M.; Shao, Q.; Zhao, Y.; Ma, C.; Wang, P. Autophagy: An Intracellular Degradation Pathway Regulating Plant Survival and Stress Response. Front. Plant Sci. 2020, 11, 164. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Wu, R.; Feng, J.; Feng, T.; Wang, C.; Hu, J.; Zhan, N.; Li, Y.; Ma, X.; Ren, B.; et al. Transnitrosylation Mediated by the Non-Canonical Catalase ROG1 Regulates Nitric Oxide Signaling in Plants. Dev. Cell 2020, 53, 444–457.e5. [Google Scholar] [CrossRef]
- Sandalio, L.M.; Gotor, C.; Romero, L.C.; Romero-Puertas, M.C. Multilevel Regulation of Peroxisomal Proteome by Post-Translational Modifications. Int. J. Mol. Sci. 2019, 20, 4881. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Wang, J.; Cheng, Y.; Chi, Y.J.; Fan, B.; Yu, J.Q.; Chen, Z. NBR1-Mediated Selective Autophagy Targets Insoluble Ubiquitinated Protein Aggregates in Plant Stress Responses. PLoS Genet. 2013, 9, e1003196. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Chen, Z. Broad and Complex Roles of NBR1-Mediated Selective Autophagy in Plant Stress Responses. Cells 2020, 9, 2562. [Google Scholar] [CrossRef]
- Quan, W.; Liu, X.; Wang, L.; Yin, M.; Yang, Z.; Chan, Z. Ectopic expression of Medicago truncatula homeodomain finger pro-tein, MtPHD6, enhances drought tolerance in Arabidopsis. BMC Genom. 2019, 20, 982. [Google Scholar] [CrossRef] [Green Version]
- Yin, M.; Wang, Y.; Zhang, L.; Li, J.; Quan, W.; Yang, L.; Wang, Q.; Chan, Z. The Arabidopsis Cys2/His2 zinc finger transcription factor ZAT18 is a positive regulator of plant tolerance to drought stress. J. Exp. Bot. 2017, 68, 2991–3005. [Google Scholar] [CrossRef]
- Galbiati, F.; Chiozzotto, R.; Locatelli, F.; Spada, A.; Genga, A.; Fornara, F. Hd3a, RFT1 and Ehd1 integrate photoperiodic and drought stress signals to delay the floral transition in rice. Plant Cell Environ. 2016, 39, 1982–1993. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, F.; Zhang, F.; Wang, W.; Zhou, Y.; Fu, B.; Li, Z. Comparative transcriptome sequencing of tolerant rice introgression line and its parents in response to drought stress. BMC Genom. 2014, 15, 1026. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Liu, C.; Zhang, D.; He, C.; Zhang, J.; Li, Z. Effects of maize organ-specific drought stress response on yields from transcriptome analysis. BMC Plant Biol. 2019, 19, 335. [Google Scholar] [CrossRef] [Green Version]
- Ding, Y.; Virlouvet, L.; Liu, N.; Riethoven, J.J.; Fromm, M.; Avramova, Z. Dehydration stress memory genes of Zea mays; comparison with Arabidopsis thaliana. BMC Plant Biol. 2014, 14, 141. [Google Scholar] [CrossRef] [Green Version]
- Fracasso, A.; Trindade, L.M.; Amaducci, S. Drought stress tolerance strategies revealed by RNA-Seq in two sorghum geno-types with contrasting WUE. BMC Plant Biol. 2016, 16, 115. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hickey, K.; Wood, M.; Sexton, T.; Sahin, Y.; Nazarov, T.; Fisher, J.; Sanguinet, K.A.; Cousins, A.; Kirchhoff, H.; Smertenko, A. Drought Tolerance Strategies and Autophagy in Resilient Wheat Genotypes. Cells 2022, 11, 1765. https://doi.org/10.3390/cells11111765
Hickey K, Wood M, Sexton T, Sahin Y, Nazarov T, Fisher J, Sanguinet KA, Cousins A, Kirchhoff H, Smertenko A. Drought Tolerance Strategies and Autophagy in Resilient Wheat Genotypes. Cells. 2022; 11(11):1765. https://doi.org/10.3390/cells11111765
Chicago/Turabian StyleHickey, Kahleen, Magnus Wood, Tom Sexton, Yunus Sahin, Taras Nazarov, Jessica Fisher, Karen A. Sanguinet, Asaph Cousins, Helmut Kirchhoff, and Andrei Smertenko. 2022. "Drought Tolerance Strategies and Autophagy in Resilient Wheat Genotypes" Cells 11, no. 11: 1765. https://doi.org/10.3390/cells11111765
APA StyleHickey, K., Wood, M., Sexton, T., Sahin, Y., Nazarov, T., Fisher, J., Sanguinet, K. A., Cousins, A., Kirchhoff, H., & Smertenko, A. (2022). Drought Tolerance Strategies and Autophagy in Resilient Wheat Genotypes. Cells, 11(11), 1765. https://doi.org/10.3390/cells11111765






