Rho-Proteins and Downstream Pathways as Potential Targets in Sepsis and Septic Shock: What Have We Learned from Basic Research
Abstract
1. Introduction
2. Experimental Models Used to Study Sepsis-Associated Dysfunctions
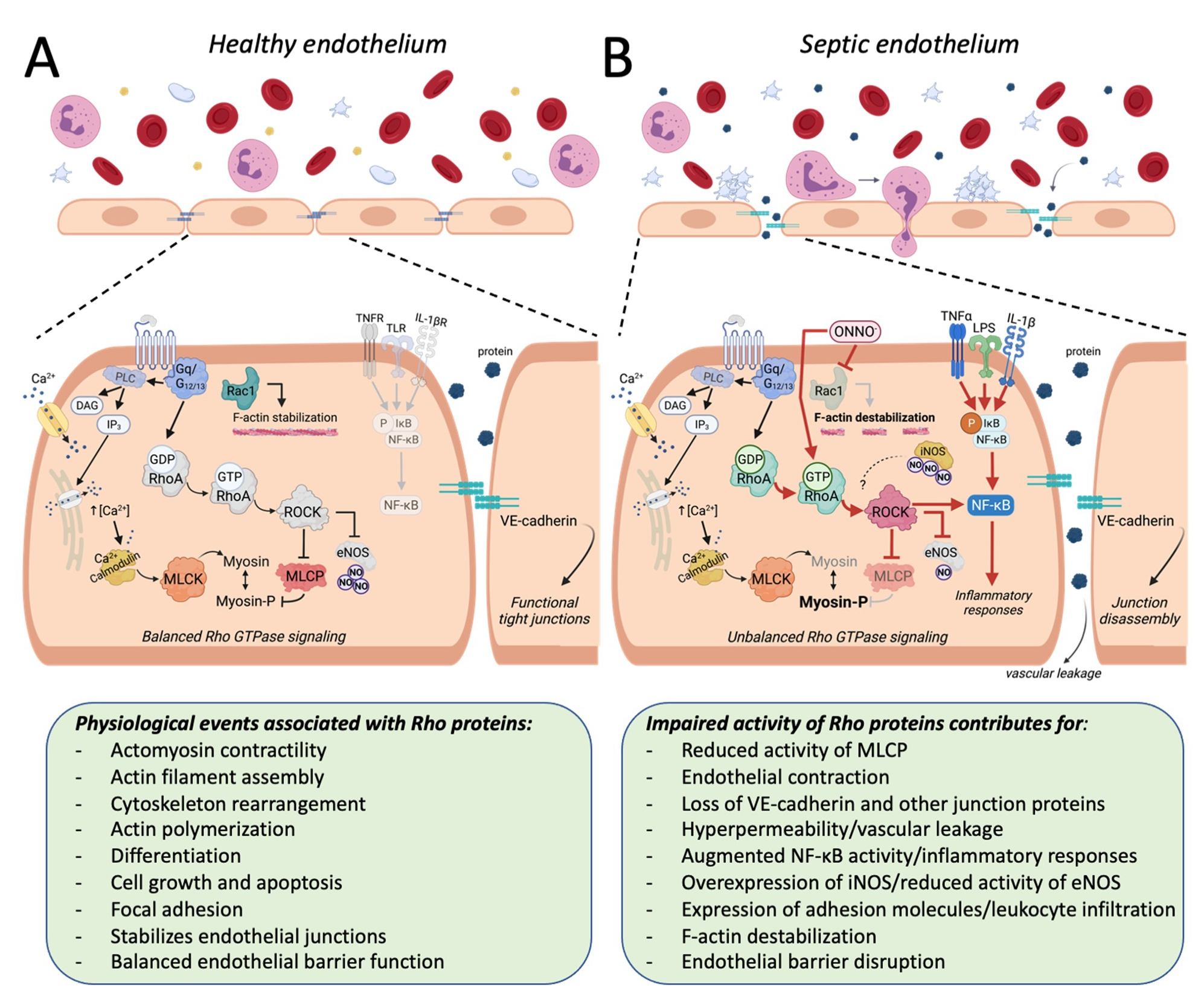
3. Rho Proteins and Their Impact on Endothelial Function in Sepsis-Related Experimental Approaches
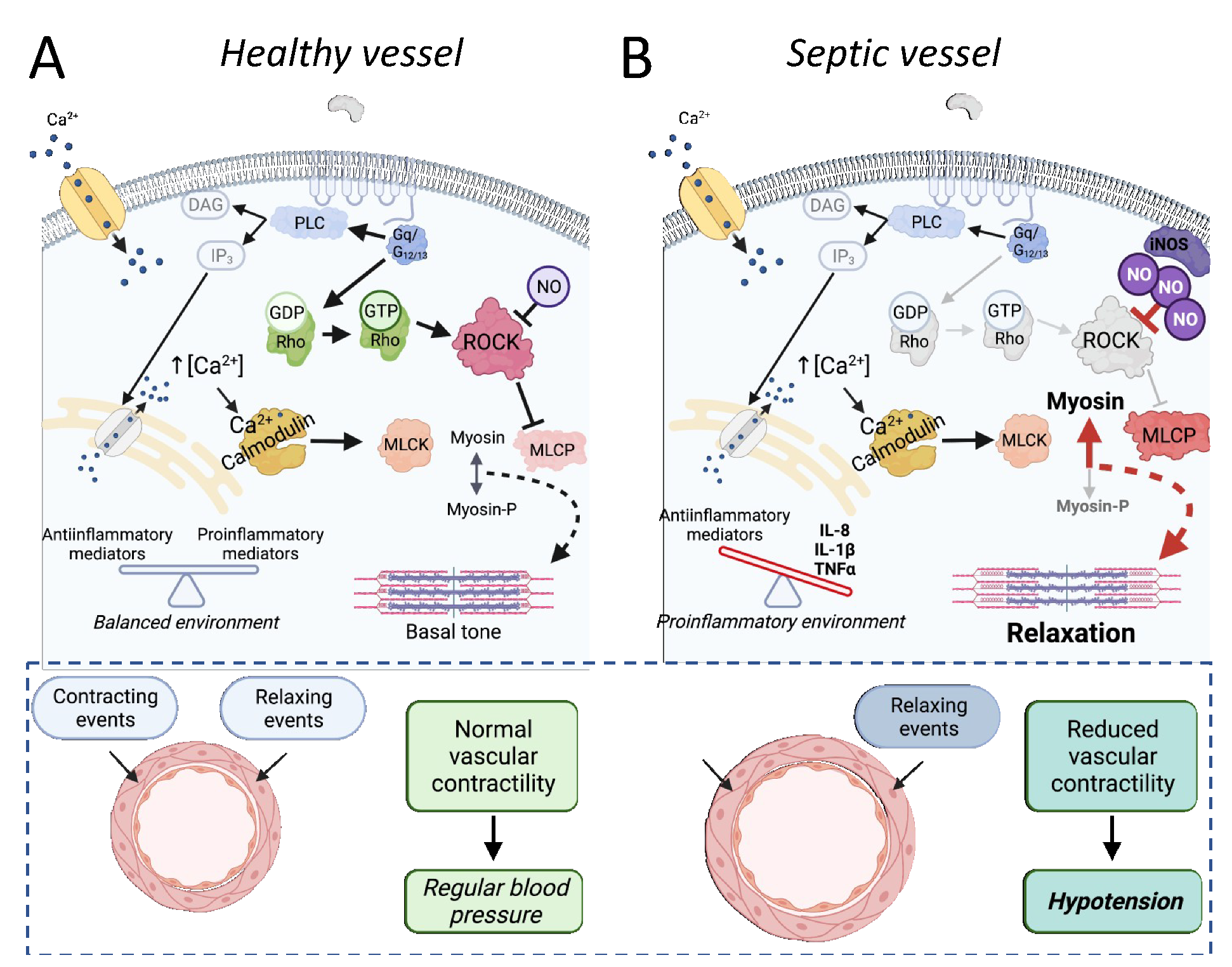
4. Rho Proteins and Their Impact on the Vascular Function in Sepsis-Related Experimental Approaches
5. Rho Proteins and Their Impact on the Heart Function in Sepsis-Related Experimental Approaches
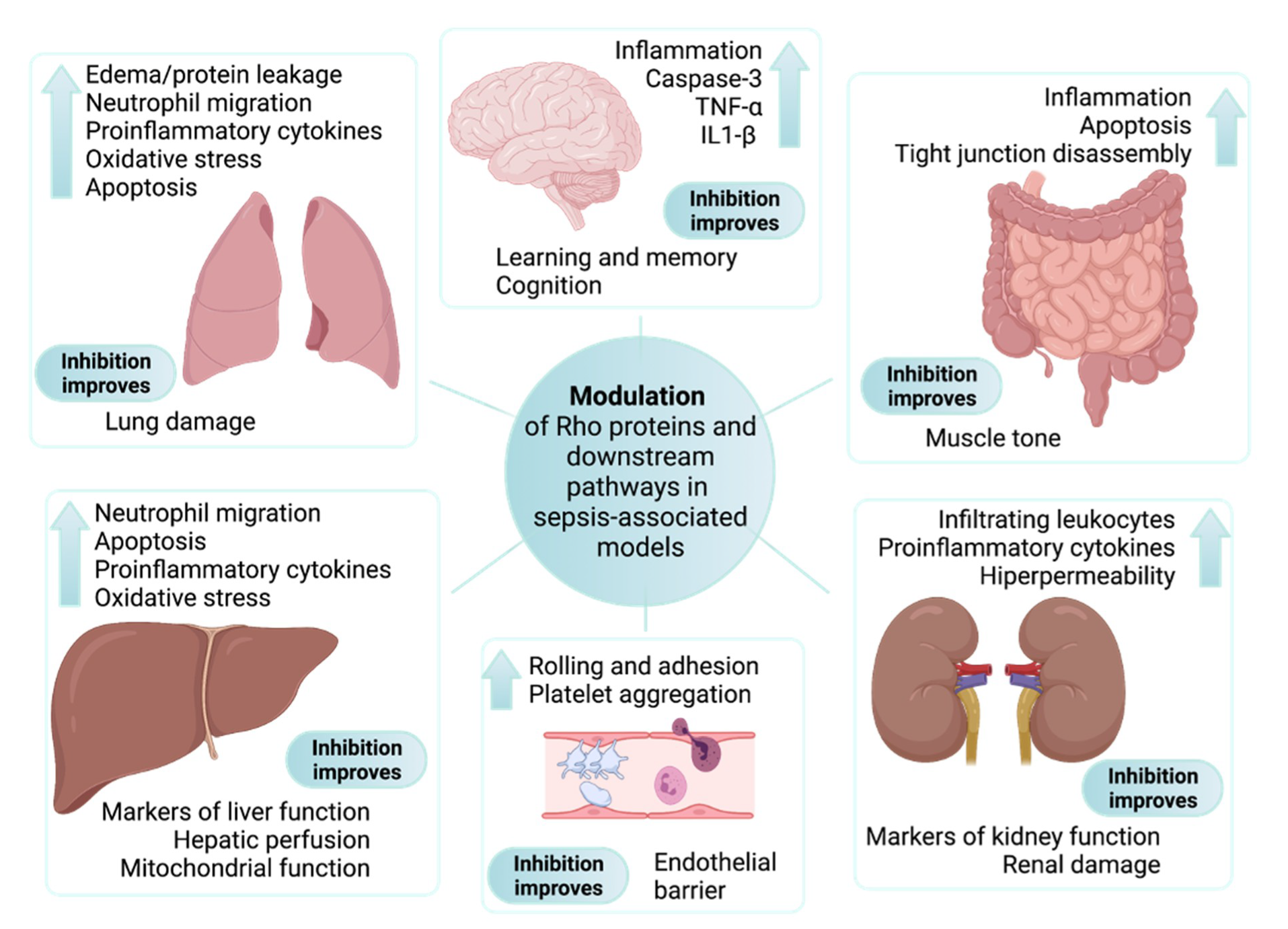
6. Rho Proteins and Their Impact on Sepsis Outside the Cardiovascular System
7. Final Remarks and Conclusions
- (i)
- Drugs with pleiotropic or indirect effects, and unknown molecular mechanisms regarding the effects on Rho proteins and downstream targets (i.e., statins);
- (ii)
- A lack of information regarding which Rho protein is affected in different organs during the ongoing sepsis;
- (iii)
- A single point of evaluation;
- (iv)
- The usage of LPS instead of more reliable experimental models of sepsis (i.e., CLP);
- (iv)
- A lack of dose–response evaluation;
- (vi)
- The use of acute treatments only, often as a pretreatment, missing details regarding the benefices of a post-treatment and continuous therapy, and;
- (vii)
- The absence of toxicological and safety evaluation.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- E Rudd, K.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: Analysis for the global burden of disease study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef]
- Gogos, C.A.; Drosou, E.; Bassaris, H.P.; Skoutelis, A. Pro-versus anti-inflammatory cytokine profile in patients with severe sepsis: A marker for prognosis and future therapeutic options. J. Infect. Dis. 2000, 181, 176–180. [Google Scholar] [CrossRef]
- Rovas, A.; Sackarnd, J.; Rossaint, J.; Kampmeier, S.; Pavenstädt, H.; Vink, H.; Kümpers, P. Identification of novel sublingual parameters to analyze and diagnose microvascular dysfunction in sepsis: The Nostradamus study. Crit. Care 2021, 25, 1–14. [Google Scholar] [CrossRef]
- Ridley, A.J. Rho GTPases and cell migration. J. Cell Sci. 2001, 114, 2713–2722. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.M.; Ohanian, J.; Heagerty, A.M. Calcium Sensitivity and Agonist-Induced Calcium Sensitization in Small Arteries of Young and Adult Spontaneously Hypertensive Rats. Hypertension 1997, 30, 442–448. [Google Scholar] [CrossRef]
- Uehata, M.; Ishizaki, T.; Satoh, H.; Ono, T.; Kawahara, T.; Morishita, T.; Tamakawa, H.; Yamagami, K.; Inui, J.; Maekawa, M.; et al. Calcium sensitization of smooth muscle mediated by a Rho-associated protein kinase in hypertension. Nat. Cell Biol. 1997, 389, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Birchmeier, C.; Broek, D.; Wigler, M. RAS proteins can induce meiosis in xenopus oocytes. Cell 1985, 43, 615–621. [Google Scholar] [CrossRef]
- Aznar, S.; Lacal, J.C. Rho signals to cell growth and apoptosis. Cancer Lett. 2001, 165, 1–10. [Google Scholar] [CrossRef]
- Bertrand, J.; Winton, M.J.; Rodriguez-Hernandez, N.; Campenot, R.B.; McKerracher, L. Application of Rho antagonist to neuronal cell bodies promotes neurite growth in compartmented cultures and regeneration of retinal ganglion cell axons in the optic nerve of adult rats. J. Neurosci. 2005, 25, 1113–1121. [Google Scholar] [CrossRef]
- Chrzanowska, M.; Burridge, K. Rho-stimulated contractility drives the formation of stress fibers and focal adhesions. J. Cell Biol. 1996, 133, 1403–1415. [Google Scholar] [CrossRef]
- Ridley, A.; Hall, A. The small GTP-binding protein Rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 1992, 70, 389–399. [Google Scholar] [CrossRef]
- Amano, M.; Nakayama, M.; Kaibuchi, K. Rho-kinase/ROCK: A key regulator of the cytoskeleton and cell polarity. Cytoskeleton 2010, 67, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Nobes, C.D.; Hall, A. Rho GTPases control polarity, protrusion, and adhesion during cell movement. J. Cell Biol. 1999, 144, 1235–1244. [Google Scholar] [CrossRef] [PubMed]
- Tatsumoto, T.; Xie, X.; Blumenthal, R.; Okamoto, I.; Miki, T. Human Ect2 is an exchange factor for Rho GTPases, phosphorylated in G2/M phases, and involved in cytokinesis. J. Cell Biol. 1999, 147, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Jantsch-Plunger, V.; Gonczy, P.; Romano, A.; Schnabel, H.; Hamill, D.; Schnabel, R.; Hyman, A.A.; Glotzer, M. Cyk-4: A Rho family GTPase activating protein (GAP) required for central spindle formation and cytokinesis. J. Cell Biol. 2000, 149, 1391–1404. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-A.; Carpenter, C.L.; Abrams, C.S. Rho and Rho-kinase mediate thrombin-induced phosphatidylinositol 4-phosphate 5-kinase trafficking in platelets. J. Biol. Chem. 2004, 279, 42331–42336. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, K.; Matsuda, M. Localized RhoA activation as a requirement for the induction of membrane ruffling. Mol. Biol. Cell 2005, 16, 4294–4303. [Google Scholar] [CrossRef] [PubMed]
- McCusker, D. Cellular self-organization: Generating order from the abyss. Mol. Biol. Cell 2020, 31, 143–148. [Google Scholar] [CrossRef]
- Nguyen, L.K.; Kholodenko, B.N.; Von Kriegsheim, A. Rac1 and RhoA: Networks, loops and bistability. Small GTPases 2016, 9, 316–321. [Google Scholar] [CrossRef]
- Saha, S.; Nagy, T.L.; Weiner, O.D. Joining forces: Crosstalk between biochemical signalling and physical forces orchestrates cellular polarity and dynamics. Philos. Trans. R Soc. B Biol. Sci. 2018, 373, 145. [Google Scholar] [CrossRef]
- Sah, V.P.; Hoshijima, M.; Chien, K.R.; Brown, J.H. Rho is required for Gαq and α1-adrenergic receptor signaling in cardiomyocytes. J. Biol. Chem. 1996, 271, 31185–31190. [Google Scholar] [CrossRef]
- Le Page, S.L.; Bi, Y.; Williams, J.A. CCK-A receptor activates RhoA through Gα12/13 in NIH3T3 cells. Am. J. Physiol. Physiol. 2003, 285, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Clark, E.A.; King, W.G.; Brugge, J.S.; Symons, M.; Hynes, R.O. Integrin-mediated signals regulated by members of the Rho family of GTPases. J. Cell Biol. 1998, 142, 573–586. [Google Scholar] [CrossRef]
- Wakino, S.; Hayashi, K.; Kanda, T.; Tatematsu, S.; Homma, K.; Yoshioka, K.; Takamatsu, I.; Saruta, T. Peroxisome proliferator-activated receptor γ ligands inhibit Rho/Rho kinase pathway by inducing protein tyrosine phosphatase SHP-2. Circ. Res. 2004, 95, 45–55. [Google Scholar] [CrossRef]
- Osuchowski, M.F.; Ayala, A.; Bahrami, S.; Bauer, M.; Boros, M.; Cavaillon, J.-M.; Chaudry, I.H.; Coopersmith, C.M.; Deutschman, C.; Drechsler, S.; et al. Minimum quality threshold in pre-clinical sepsis studies (MQTiPSS): An international expert consensus initiative for improvement of animal modeling in sepsis. Infection 2018, 46, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Sordi, R.; Menezes-De-Lima, O.; Della-Justina, A.M.; Rezende, E.; Assreuy, J. Pneumonia-induced sepsis in mice: Temporal study of inflammatory and cardiovascular parameters. Int. J. Exp. Pathol. 2013, 94, 144–155. [Google Scholar] [CrossRef]
- Rittirsch, D.; Huber-Lang, M.S.; Flierl, M.A.; Ward, P.A. Immunodesign of experimental sepsis by cecal ligation and puncture. Nat. Protoc. 2008, 4, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Bosscha, V.B.N.K. A standardised and reproducible model of intra-abdominal infection and abscess formation in rats. Eur. J. Surg. 2000, 166, 963–967. [Google Scholar] [CrossRef]
- Weinstein, W.M.; Onderdonk, A.B.; Bartlett, J.G.; Gorbach, S.L. Experimental intra-abdominal abscesses in rats: Development of an experimental model. Infect. Immun. 1974, 10, 1250–1255. [Google Scholar] [CrossRef]
- Essler, M.; Staddon, J.M.; Weber, P.C.; Aepfelbacher, M. Cyclic AMP blocks bacterial lipopolysaccharide-induced myosin light chain phosphorylation in endothelial cells through inhibition of Rho/Rho kinase signaling. J. Immunol. 2000, 164, 6543–6549. [Google Scholar] [CrossRef] [PubMed]
- Slotta, J.E.; Braun, O.O.; Menger, M.D.; Thorlacius, H. Fasudil, a Rho-kinase inhibitor, inhibits leukocyte adhesion in inflamed large blood vessels in vivo. Inflamm. Res. 2006, 55, 364–367. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, G.-M.; Zhu, Y.; Peng, X.-Y.; Li, T.; Liu, L.-M. Role of connexin 43 in vascular hyperpermeability and relationship to Rock1-MLC20 pathway in septic rats. Am. J. Physiol. Cell. Mol. Physiol. 2015, 309, 1323. [Google Scholar] [CrossRef]
- Han, J.; Ding, R.; Zhao, D.; Zhang, Z.; Ma, X. Unfractionated heparin attenuates lung vascular leak in a mouse model of sepsis:Role of RhoA/Rho kinase pathway. Thromb. Res. 2013, 132, 42–47. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Liu, W.; Zhang, L. Role of the Rho/ROCK signaling pathway in the protective effects of fasudil against acute lung injury in septic rats. Mol. Med. Rep. 2018, 18, 4486–4498. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Nemoto, K.; Ninomiya, N.; Kuno, M.; Kubota, M.; Yokota, H. Fasudil, a Rho-kinase inhibitor, attenuates lipopolysaccharide-induced vascular hyperpermeability and colonic muscle relaxation in guinea pigs. J. Surg. Res. 2012, 178, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Thorlacius, K.; Slotta, J.E.; Laschke, M.W.; Wang, Y.; Menger, M.D.; Jeppsson, B.; Thorlacius, H. Protective effect of fasudil, a Rho-kinase inhibitor, on chemokine expression, leukocyte recruitment, and hepatocellular apoptosis in septic liver injury. J. Leukoc. Biol. 2006, 79, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Tan, Q.; Chen, R.; Cao, B.; Li, W. Sevoflurane prevents lipopolysaccharide-induced barrier dysfunction in human lung microvascular endothelial cells: Rho-mediated alterations of VE-cadherin. Biochem. Biophys. Res. Commun. 2015, 468, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Haidari, M.; Zhang, W.; Chen, Z.; Ganjehei, L.; Mortazavi, A.; Warier, N.; Vanderslice, P.; Dixon, R.A. Atorvastatin preserves the integrity of endothelial adherens junctions by inhibiting vascular endothelial cadherin tyrosine phosphorylation. Exp. Cell Res. 2012, 318, 1673–1684. [Google Scholar] [CrossRef]
- Xie, K.; Wang, W.; Chen, H.; Han, H.; Liu, D.; Wang, G.; Yu, Y. Hydrogen-rich medium attenuated lipopolysaccharide-induced monocyte-endothelial cell adhesion and vascular endothelial permeability via Rho-associated coiled-coil protein kinase. Shock 2015, 44, 58–64. [Google Scholar] [CrossRef]
- Wang, J.; Xu, J.; Zhao, X.; Xie, W.; Wang, H.; Kong, H. Fasudil inhibits neutrophil-endothelial cell interactions by regulating the expressions of GRP78 and BMPR. Exp. Cell Res. 2018, 365, 97–105. [Google Scholar] [CrossRef]
- Sander, E.E.; Klooster, J.P.T.; Van Delft, S.; Van Der Kammen, R.A.; Collard, J.G. Rac downregulates Rho activity. J. Cell Biol. 1999, 147, 1009–1022. [Google Scholar] [CrossRef]
- Mammoto, T.; Parikh, S.M.; Mammoto, A.; Gallagher, D.; Chan, B.; Mostoslavsky, G.; Ingber, D.E.; Sukhatme, V.P. Angiopoietin-1 requires p190 RhoGAP to protect against vascular leakage in vivo. J. Biol. Chem. 2007, 282, 23910–23918. [Google Scholar] [CrossRef]
- Bogatcheva, N.; Zemskova, M.A.; Poirier, C.; Mirzapoiazova, T.; Kolosova, I.; Bresnick, A.R.; Verin, A.D. The suppression of myosin light chain (MLC) phosphorylation during the response to lipopolysaccharide (LPS): Beneficial or detrimental to endothelial barrier? J. Cell. Physiol. 2011, 226, 3132–3146. [Google Scholar] [CrossRef]
- Rahman, F.A.; D’Almeida, S.; Zhang, T.; Asadi, M.; Bozoglu, T.; Bongiovanni, D.; von Scheidt, M.; Dietzel, S.; Schwedhelm, E.; Hinkel, R.; et al. Sphingosine-1-phosphate attenuates lipopolysaccharide-induced pericyte loss via activation of Rho-A and MRTF-A. Thromb. Haemost. 2021, 121, 341–350. [Google Scholar] [CrossRef]
- Strey, A.; Janning, A.; Barth, H.; Gerke, V. Endothelial Rho signaling is required for monocyte transendothelial migration. FEBS Lett. 2002, 517, 261–266. [Google Scholar] [CrossRef]
- Adamson, R.H.; Curry, F.E.; Adamson, G.; Liu, B.; Jiang, Y.; Aktories, K.; Barth, H.; Daigeler, A.; Golenhofen, N.; Ness, W.; et al. Rho and rho kinase modulation of barrier properties: Cultured endothelial cells and intact microvessels of rats and mice. J. Physiol. 2002, 539, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Waschke, J.; Burger, S.; Curry, F.-R.E.; Drenckhahn, D.; Adamson, R.H. Activation of Rac-1 and Cdc42 stabilizes the microvascular endothelial barrier. Histochem. Cell Biol. 2005, 125, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Gavard, J.; Gutkind, J.S. Protein Kinase C-related Kinase and ROCK Are required for thrombin-induced endothelial cell permeability downstream from Gα12/13 and Gα11/q. J. Biol. Chem. 2008, 283, 29888–29896. [Google Scholar] [CrossRef]
- Bentzer, P.; Fisher, J.; Kong, H.J.; Mörgelin, M.; Boyd, J.H.; Walley, K.R.; Russell, J.A.; Linder, A. Heparin-binding protein is important for vascular leak in sepsis. Intensive Care Med. Exp. 2016, 4, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.D.; Dimitropoulou, C.; Thangjam, G.; Snead, C.; Feldman, S.; Barabutis, N.; Fulton, D.; Hou, Y.; Kumar, S.; Patel, V.; et al. Hsp90 inhibitors prevent LPS-induced endothelial barrier dysfunction by disrupting RhoA signaling. Am. J. Respir. Cell Mol. Biol. 2013, 50, 170–179. [Google Scholar] [CrossRef]
- Baumer, Y.; Bürger, S.; Curry, F.E.; Golenhofen, N.; Drenckhahn, D.; Waschke, J. Differential role of Rho GTPases in endothelial barrier regulation dependent on endothelial cell origin. Histochem. Cell Biol. 2007, 129, 179–191. [Google Scholar] [CrossRef]
- Wu, T.; Xing, J.; Birukova, A.A. Cell-type-specific crosstalk between p38 MAPK and Rho signaling in lung micro- and macrovascular barrier dysfunction induced by Staphylococcus aureus-derived pathogens. Transl. Res. 2013, 162, 45–55. [Google Scholar] [CrossRef][Green Version]
- Zheng, S.; Lin, Z.; Liu, Z.; Liu, Y.; Wu, W. Lipopolysaccharide mediates the destruction of intercellular tight junction among renal tubular epithelial cells via RhoT1/SMAD-4/JAM-3 pathway. Int. J. Med. Sci. 2018, 15, 595–602. [Google Scholar] [CrossRef]
- Nwariaku, F.E.; Rothenbach, P.; Liu, Z.; Zhu, X.; Turnage, R.H.; Terada, L.S. Rho inhibition decreases TNF-induced endothelial MAPK activation and monolayer permeability. J. Appl. Physiol. 2003, 95, 1889–1895. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Liu, S.Y.; Luo, L.; Na Gu, N.; Zeng, Y.; Chen, X.Y.; Xu, S.; Zhang, D. Anti-inflammatory mechanism of ulinastatin: Inhibiting the hyperpermeability of vascular endothelial cells induced by TNF-α via the RhoA/ROCK signal pathway. Int. Immunopharmacol. 2017, 46, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, N.; Waschke, J. Impaired cAMP and Rac 1 signaling contribute to TNF-α-induced endothelial barrier breakdown in microvascular endothelium. Microcirculation 2009, 16, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Birukova, A.A.; Tian, Y.; Meliton, A.Y.; Leff, A.R.; Wu, T.; Birukov, K.G. Stimulation of Rho signaling by pathologic mechanical stretch is a “second hit” to Rho-independent lung injury induced by IL-6. Am. J. Physiol. Cell. Mol. Physiol. 2012, 302, 965–975. [Google Scholar] [CrossRef]
- Clark, P.R.; Kim, R.K.; Pober, J.S.; Kluger, M.S. Tumor necrosis factor disrupts Claudin-5 endothelial tight junction barriers in two distinct NF-κB-dependent phases. PLoS ONE 2015, 10, e0120075. [Google Scholar] [CrossRef]
- Hippenstiel, S.; Soeth, S.; Kellas, B.; Fuhrmann, O.; Seybold, J.; Krüll, M.; Eichel-Streiber, C.; Goebeler, M.; Ludwig, S.; Suttorp, N. Rho proteins and the p38-MAPK pathway are important mediators for LPS-induced interleukin-8 expression in human endothelial cells. Blood 2000, 95, 3044–3051. [Google Scholar] [CrossRef]
- Schmeck, B.; Beermann, W.; van Laak, V.; Opitz, B.; Hocke, A.C.; Meixenberger, K.; Eitel, J.; Chakraborty, T.; Schmidt, G.; Barth, H.; et al. Listeria monocytogenes induced Rac1-dependent signal transduction in endothelial cells. Biochem. Pharmacol. 2006, 72, 1367–1374. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Tang, J.; Zhou, Z.; Dou, Y.; Van Lonkhuyzen, D.R.; Gao, C.; Huan, J. GEF-H1-RhoA signaling pathway mediates LPS-induced NF-κB transactivation and IL-8 synthesis in endothelial cells. Mol. Immunol. 2012, 50, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Karki, P.; Ke, Y.; Tian, Y.; Ohmura, T.; Sitikov, A.; Sarich, N.; Montgomery, C.; Birukova, A.A. Staphylococcus aureus-induced endothelial permeability and inflammation are mediated by microtubule destabilization. J. Biol. Chem. 2019, 294, 3369–3384. [Google Scholar] [CrossRef] [PubMed]
- Ding, R.; Zhao, D.; Li, X.; Liu, B.; Ma, X. Rho-kinase inhibitor treatment prevents pulmonary inflammation and coagulation in lipopolysaccharide-induced lung injury. Thromb. Res. 2017, 150, 59–64. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Peng, J.; Deng, X.-L.; Yang, L.-F.; Wu, L.-W.; Zhang, C.-L.; Yin, F. RhoA and NF-κB are involved in lipopolysaccharide-induced brain microvascular cell line hyperpermeability. Neuroscience 2011, 188, 35–47. [Google Scholar] [CrossRef]
- Dong, R.; Hu, D.; Yang, Y.; Chen, Z.; Fu, M.; Wang, D.W.; Xu, X.; Tu, L. EETs reduces LPS-induced hyperpermeability by targeting GRP78 mediated Src activation and subsequent Rho/ROCK signaling pathway. Oncotarget 2017, 8, 50958–50971. [Google Scholar] [CrossRef][Green Version]
- McGown, C.C.; Brown, N.; Hellewell, P.G.; Brookes, Z.L. ROCK induced inflammation of the microcirculation during endotoxemia mediated by nitric oxide synthase. Microvasc. Res. 2011, 81, 281–288. [Google Scholar] [CrossRef]
- Yang, T.; Wang, L.; Sun, R.; Chen, H.; Zhang, H.; Yu, Y.; Wang, Y.; Wang, G.; Yu, Y.; Xie, K. Hydrogen-rich medium ameliorates lipopolysaccharide-induced barrier dysfunction via Rhoa-Mdia1 signaling in Caco-2 cells. Shock 2016, 45, 228–237. [Google Scholar] [CrossRef]
- Higuchi, H.; Satoh, T. Endothelin-1 induces vasoconstriction and nitric oxide release via endothelin ETB receptors in isolated perfused rat liver. Eur. J. Pharmacol. 1997, 328, 175–182. [Google Scholar] [CrossRef]
- Whittle, B.; Lopez-Belmonte, J.; Rees, D. Modulation of the vasodepressor actions of acetylcholine, bradykinin, substance P and endothelin in the rat by a specific inhibitor of nitric oxide formation. Br. J. Pharmacol. 1989, 98, 646–652. [Google Scholar] [CrossRef]
- Kwok, W.; Clemens, M.G. Rho-kinase activation contributes to Lps-induced impairment of endothelial nitric oxide synthase activation by endothelin-1 in cultured hepatic sinusoidal endothelial cells. Shock 2014, 42, 554–561. [Google Scholar] [CrossRef]
- Rafikov, R.; Dimitropoulou, C.; Aggarwal, S.; Kangath, A.; Gross, C.; Pardo, D.; Sharma, S.; Jezierska-Drutel, A.; Patel, V.; Snead, C.; et al. Lipopolysaccharide-induced lung injury involves the nitration-mediated activation of RhoA. J. Biol. Chem. 2014, 289, 4710–4722. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yegambaram, M.; Gross, C.; Sun, X.; Lu, Q.; Wang, H.; Wu, X.; Kangath, A.; Tang, H.; Aggarwal, S.; et al. RAC1 nitration at Y32 is involved in the endothelial barrier disruption associated with lipopolysaccharide-mediated acute lung injury. Redox Biol. 2021, 38, 101794. [Google Scholar] [CrossRef] [PubMed]
- Alblas, J.; Ulfman, L.; Hordijk, P.; Koenderman, L. Activation of RhoA and ROCK are essential for detachment of migrating leukocytes. Mol. Biol. Cell 2001, 12, 2137–2145. [Google Scholar] [CrossRef]
- Breslin, J.W.; Sun, H.; Xu, W.; Rodarte, C.; Moy, A.B.; Wu, M.H.; Yuan, S.Y. Involvement of ROCK-mediated endothelial tension development in neutrophil-stimulated microvascular leakage. Am. J. Physiol. Circ. Physiol. 2006, 290, 741–750. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Breslin, J.W.; Yuan, S.Y. Involvement of RhoA and Rho kinase in neutrophil-stimulated endothelial hyperpermeability. Am. J. Physiol. Circ. Physiol. 2004, 286, 1057–1062. [Google Scholar] [CrossRef]
- Dorard, C.; Cseh, B.; Ehrenreiter, K.; Wimmer, R.; Varga, A.; Hirschmugl, T.; Maier, B.; Kramer, K.; Fürlinger, S.; Doma, E.; et al. RAF dimers control vascular permeability and cytoskeletal rearrangements at endothelial cell-cell junctions. FEBS J. 2019, 286, 2277–2294. [Google Scholar] [CrossRef]
- Eriksson, A.; Cao, R.; Roy, J.; Tritsaris, K.; Wahlestedt, C.; Dissing, S.; Thyberg, J.; Cao, Y. Small GTP-binding protein Rac is an essential mediator of vascular endothelial growth factor-induced endothelial fenestrations and vascular permeability. Circulation 2003, 107, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Gavard, J.; Gutkind, J.S. VEGF controls endothelial-cell permeability by promoting the β-arrestin-dependent endocytosis of VE-cadherin. Nat. Cell Biol. 2006, 8, 1223–1234. [Google Scholar] [CrossRef] [PubMed]
- Herrera, J.L.; Komatsu, M. R-Ras deficiency in pericytes causes frequent microphthalmia and perturbs retinal vascular development. J. Vasc. Res. 2021, 58, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Perrot, C.Y.; Sawada, J.; Komatsu, M. Prolonged activation of cAMP signaling leads to endothelial barrier disruption via transcriptional repression of RRAS. FASEB J. 2018, 32, 5793–5812. [Google Scholar] [CrossRef] [PubMed]
- Vähätupa, M.; Prince, S.; Vataja, S.; Mertimo, T.; Kataja, M.; Kinnunen, K.; Marjomäki, V.; Uusitalo, H.; Komatsu, M.; Järvinen, T.A.; et al. Lack of R-Ras leads to increased vascular permeability in ischemic retinopathy. Investig. Opthalmol. Vis. Sci. 2016, 57, 4898–4909. [Google Scholar] [CrossRef] [PubMed]
- Amerongen, G.P.V.N.; van Hinsbergh, V.W. Cytoskeletal effects of Rho-like small guanine nucleotide-binding proteins in the vascular system. Arter. Thromb. Vasc. Biol. 2001, 21, 300–311. [Google Scholar] [CrossRef]
- Worthylake, R.A.; Lemoine, S.; Watson, J.M.; Burridge, K. RhoA is required for monocyte tail retraction during transendothelial migration. J. Cell Biol. 2001, 154, 147–160. [Google Scholar] [CrossRef]
- Xu, W.; Wittchen, E.S.; Hoopes, S.L.; Stefanini, L.; Burridge, K.; Caron, K.M. Small GTPase Rap1A/B is required for lymphatic development and adrenomedullin-induced stabilization of lymphatic endothelial junctions. Arter. Thromb. Vasc. Biol. 2018, 38, 2410–2422. [Google Scholar] [CrossRef]
- Yan, Z.; Wang, Z.-G.; Segev, N.; Hu, S.; Minshall, R.D.; Dull, R.O.; Zhang, M.; Malik, A.B.; Hu, G. Rab11a mediates vascular endothelial-cadherin recycling and controls endothelial barrier function. Arter. Thromb. Vasc. Biol. 2016, 36, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Vouret-Craviari, V.; Boquet, P.; Pouysségur, J.; Van Obberghen-Schilling, E. Regulation of the actin cytoskeleton by thrombin in human endothelial cells: Role of Rho proteins in endothelial barrier function. Mol. Biol. Cell 1998, 9, 2639–2653. [Google Scholar] [CrossRef] [PubMed]
- Xiaolu, D.; Jing, P.; Fang, H.; Lifen, Y.; Liwen, W.; Ciliu, Z.; Fei, Y. Role of p115RhoGEF in lipopolysaccharide-induced mouse brain microvascular endothelial barrier dysfunction. Brain Res. 2011, 1387, 1–7. [Google Scholar] [CrossRef]
- Zhou, Z.; Guo, F.; Dou, Y.; Tang, J.; Huan, J. Guanine nucleotide exchange factor-H1 signaling is involved in lipopolysaccharide-induced endothelial barrier dysfunction. Surgery 2013, 154, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, H.; Gao, C.; Liu, D.; Fan, Y.; Li, W.; Chen, Y.; Pan, S. Tetramethylpyrazine alleviates LPS-induced inflammatory injury in HUVECs by inhibiting Rho/ROCK pathway. Biochem. Biophys. Res. Commun. 2019, 514, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Liang, X.; Hu, X.; He, H.; Tang, L.; Yao, W. Tetrahydroxystilbene glucoside protects against LPS-induced endothelial dysfunction via inhibiting RhoA/ROCK signaling and F-actin remodeling. Gen. Physiol. Biophys. 2020, 39, 407–417. [Google Scholar] [CrossRef]
- Xiao, H.; Qin, X.; Ping, D.; Zuo, K. Inhibition of Rho and Rac geranylgeranylation by atorvastatin is critical for preservation of endothelial junction integrity. PLoS ONE 2013, 8, e59233. [Google Scholar] [CrossRef]
- Xing, J.; Wang, Q.; Coughlan, K.; Viollet, B.; Moriasi, C.; Zou, M.-H. Inhibition of AMP-activated protein kinase accentuates lipopolysaccharide-induced lung endothelial barrier dysfunction and lung injury in vivo. Am. J. Pathol. 2013, 182, 1021–1030. [Google Scholar] [CrossRef]
- Xu, J.; Wei, G.; Wang, J.; Zhu, J.; Yu, M.; Zeng, X.; Wang, H.; Xie, W.; Kong, H. Glucagon-like peptide-1 receptor activation alleviates lipopolysaccharide-induced acute lung injury in mice via maintenance of endothelial barrier function. Lab. Investig. 2019, 99, 577–587. [Google Scholar] [CrossRef]
- Meng, L.; Cao, H.; Wan, C.; Jiang, L. MiR-539-5p alleviates sepsis-induced acute lung injury by targeting ROCK. Folia Histochem. Cytobiol. 2020, 57, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Boer, C.; Amerongen, G.P.V.N.; Groeneveld, A.B.J.; Scheffer, G.J.; De Lange, J.J.; Westerhof, N.; Van Hinsbergh, V.W.M.; Sipkema, P. Smooth muscle F-actin disassembly and RhoA/Rho-kinase signaling during endotoxin-induced alterations in pulmonary arterial compliance. Am. J. Physiol. Cell. Mol. Physiol. 2004, 287, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.-Y.; Huang, K.-C.; Chou, Y.-H.; Hsieh, P.-F.; Lin, K.-H.; Lin, W.-W. The Role of Rho-associated kinase in differential regulation by statins of interleukin-1β- and lipopolysaccharide-mediated nuclear factor κB activation and inducible nitric-oxide synthase gene expression in vascular smooth muscle cells. Mol. Pharmacol. 2005, 69, 960–967. [Google Scholar] [CrossRef]
- Büyükafşar, K.; Arikan, O.; Ark, M.; Kubat, H.; Ozveren, E.; Arıkan, O. Upregulation of Rho-kinase (ROCK-2) expression and enhanced contraction to endothelin-1 in the mesenteric artery from lipopolysaccharide-treated rats. Eur. J. Pharmacol. 2004, 498, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Bernardelli, A.K.; Da Silva, R.D.C.D.A.; Corrêa, T.; Da Silva-Santos, J.E. Vasoplegia in sepsis depends on the vascular system, vasopressor, and time-point: A comparative evaluation in vessels from rats subjected to the cecal ligation puncture model. Can. J. Physiol. Pharmacol. 2016, 94, 1227–1236. [Google Scholar] [CrossRef]
- El-Awady, M.S.H.; Smirnov, S.V.; Watson, M.L. Voltage-independent calcium channels mediate lipopolysaccharide-induced hyporeactivity to endothelin-1 in the rat aorta. Am. J. Physiol. Circ. Physiol. 2009, 296, 1408–1415. [Google Scholar] [CrossRef]
- Da Silva-Santos, J.E.; Chiao, C.-W.; Leite, R.; Webb, R.C. The Rho-A/Rho-kinase pathway is up-regulated but remains inhibited by cyclic guanosine monophosphate–dependent mechanisms during endotoxemia in small mesenteric arteries. Crit. Care Med. 2009, 37, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Liao, M.-H.; Shih, C.-C.; Tsao, C.-M.; Chen, S.-J.; Wu, C.-C. RhoA/Rho-kinase and nitric oxide in vascular reactivity in rats with endotoxaemia. PLoS ONE 2013, 8, e56331. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.-L.; Yang, G.-M.; Li, T.; Liu, L.-M. Effects of interleukin-1β on vascular reactivity after lipopolysaccharide-induced endotoxic shock in rabbits and its relationship with PKC and Rho kinase. J. Cardiovasc. Pharmacol. 2013, 62, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Kuang, L.; He, J.; Zhang, Z.; Zheng, D.; Duan, C.; Zhu, Y.; Wu, Y.; Zhang, J.; Peng, X.; et al. Role of tumor necrosis factor-α in vascular hyporeactivity following endotoxic shock and its mechanism. J. Trauma Acute Care Surg. 2019, 87, 1346–1353. [Google Scholar] [CrossRef]
- Bivalacqua, T.J.; Champion, H.C.; Usta, M.F.; Cellek, S.; Chitaley, K.; Webb, R.C.; Lewis, R.L.; Mills, T.M.; Hellstrom, W.J.G.; Kadowitz, P.J. RhoA/Rho-kinase suppresses endothelial nitric oxide synthase in the penis: A mechanism for diabetes-associated erectile dysfunction. Proc. Natl. Acad. Sci. USA 2004, 101, 9121–9126. [Google Scholar] [CrossRef]
- Chitaley, K.; Webb, R.C. Nitric oxide induces dilation of rat aorta via inhibition of Rho-kinase signaling. Hypertension 2002, 39, 438–442. [Google Scholar] [CrossRef]
- Reddi, B.A.; Beltrame, J.F.; Young, R.L.; Wilson, D.P. Calcium desensitisation in late polymicrobial sepsis is associated with loss of vasopressor sensitivity in a murine model. Intensive Care Med. Exp. 2015, 3, 36. [Google Scholar] [CrossRef]
- de Souza, P.; Guarido, K.L.; Scheschowitsch, K.; Da Silva, L.M.; Werner, M.F.; Assreuy, J.; Da Silva-Santos, J.E. Impaired vascular function in sepsis-surviving rats mediated by oxidative stress and Rho-kinase pathway. Redox Biol. 2016, 10, 140–147. [Google Scholar] [CrossRef]
- Guarido, K.L.; Gonçalves, R.P.M.; Júnior, A.G.; da Silva-Santos, J.E. Increased activation of the Rho-A/Rho-kinase pathway in the renal vascular system is responsible for the enhanced reactivity to exogenous vasopressin in endotoxemic rats. Crit. Care Med. 2014, 42, 461–471. [Google Scholar] [CrossRef]
- Tseng, T.; Chen, M.; Liu, C.-H.; Pang, C.; Hsu, Y.; Lee, T.J.F. Induction of endothelium-dependent constriction of mesenteric arteries in endotoxemic hypotensive shock. Br. J. Pharmacol. 2016, 173, 1179–1195. [Google Scholar] [CrossRef]
- Soliman, H.; Craig, G.P.; Nagareddy, P.; Yuen, V.G.; Lin, G.; Kumar, U.; McNeill, J.H.; MacLeod, K. Role of inducible nitric oxide synthase in induction of RhoA expression in hearts from diabetic rats. Cardiovasc. Res. 2008, 79, 322–330. [Google Scholar] [CrossRef][Green Version]
- Zhu, H.; Shan, L.; Peng, T. Rac1 mediates sex difference in cardiac tumor necrosis factor-α expression via NADPH oxidase–ERK1/2/p38 MAPK pathway in endotoxemia. J. Mol. Cell. Cardiol. 2009, 47, 264–274. [Google Scholar] [CrossRef]
- Preau, S.; Delguste, F.; Yu, Y.; Remy-Jouet, I.; Richard, V.; Saulnier, F.; Boulanger, E.; Neviere, R. Endotoxemia engages the RhoA kinase pathway to impair cardiac function by altering cytoskeleton, mitochondrial fission, and autophagy. Antioxid. Redox Signal. 2016, 24, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Lu, X.; Beier, F.; Feng, Q. Rac1 activation induces tumour necrosis factor-α expression and cardiac dysfunction in endotoxemia. J. Cell. Mol. Med. 2010, 15, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Shi, Y.; Chen, G.; Wang, L.; Zheng, M.; Jin, H.; Chen, Y.-Y. TNF-α induces Drp1-mediated mitochondrial fragmentation during inflammatory cardiomyocyte injury. Int. J. Mol. Med. 2018, 41, 2317–2327. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.; Cheng, Y.; Zhou, F.; Wang, L.; Zhong, L.; Li, H.T.; Wang, X.; Dang, S. Neuregulin-1 protects cardiac function in septic rats through multiple targets based on endothelial cells. Int. J. Mol. Med. 2019, 44, 1255–1266. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wu, H.; Wu, Y.; Zhang, J.; Peng, X.; Zang, J.; Xiang, X.; Liu, L.; Li, T. Beneficial effect of Intermedin1-53 in septic shock rats. Shock 2016, 46, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Jafri, S.M.; LaVine, S.; Field, B.E.; Ahorozian, M.T.; Carlson, R.W. Left ventricular diastolic function in sepsis. Crit. Care Med. 1990, 18, 709–714. [Google Scholar] [CrossRef]
- Poelaert, J.; Declerck, C.; Vogelaers, D.; Colardyn, F.; A Visser, C. Left ventricular systolic and diastolic function in septic shock. Intensive Care Med. 1997, 23, 553–560. [Google Scholar] [CrossRef]
- Natanson, C.; Fink, M.P.; Ballantyne, H.K.; MacVittie, T.J.; Conklin, J.J.; E Parrillo, J. Gram-negative bacteremia produces both severe systolic and diastolic cardiac dysfunction in a canine model that simulates human septic shock. J. Clin. Investig. 1986, 78, 259–270. [Google Scholar] [CrossRef]
- Gonçalves, R.P.; Guarido, K.L.; Assreuy, J.; da Silva-Santos, J.E. Gender-specific differences in the in situ cardiac function of endotoxemic rats detected by pressure-volume catheter. Shock 2014, 42, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Habimana, R.; Choi, I.; Cho, H.J.; Kim, D.; Lee, K.; Jeong, I. Sepsis-induced cardiac dysfunction: A review of pathophysiology. Acute Crit. Care 2020, 35, 57–66. [Google Scholar] [CrossRef]
- Chang, J.; Xie, M.; Shah, V.R.; Schneider, M.; Entman, M.L.; Wei, L.; Schwartz, R.J. Activation of Rho-associated coiled-coil protein kinase 1 (ROCK-1) by caspase-3 cleavage plays an essential role in cardiac myocyte apoptosis. Proc. Natl. Acad. Sci. USA 2006, 103, 14495–14500. [Google Scholar] [CrossRef]
- Talukder, M.A.H.; Elnakish, M.T.; Yang, F.; Nishijima, Y.; Alhaj, M.A.; Velayutham, M.; Hassanain, H.H.; Zweier, J.L. Cardiomyocyte-specific overexpression of an active form of Rac predisposes the heart to increased myocardial stunning and ischemia-reperfusion injury. Am. J. Physiol. Circ. Physiol. 2013, 304, 294–302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- He, Q.; Lapointe, M.C. Interleukin-1β regulation of the human brain natriuretic peptide promoter involves Ras-, Rac-, and p38 kinase-dependent pathways in cardiac myocytes. Hypertension 1999, 33, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Shimokawa, H.; Higashi, M.; Hiroki, J.; Mukai, Y.; Tsutsui, H.; Kaibuchi, K.; Takeshita, A. Long-term inhibition of Rho-kinase suppresses left ventricular remodeling after myocardial infarction in mice. Circulation 2004, 109, 2234–2239. [Google Scholar] [CrossRef] [PubMed]
- Thorburn, J.; Xu, S.; Thorburn, A. MAP kinase- and Rho-dependent signals interact to regulate gene expression but not actin morphology in cardiac muscle cells. EMBO J. 1997, 16, 1888–1900. [Google Scholar] [CrossRef]
- Hines, W.A.; Thorburn, A. Ras and Rho are required for Gαq-induced hypertrophic gene expression in neonatal rat cardiac myocytes. J. Mol. Cell. Cardiol. 1998, 30, 485–494. [Google Scholar] [CrossRef]
- Satoh, M.; Ogita, H.; Takeshita, K.; Mukai, Y.; Kwiatkowski, D.J.; Liao, J.K. Requirement of Rac1 in the development of cardiac hypertrophy. Proc. Natl. Acad. Sci. USA 2006, 103, 7432–7437. [Google Scholar] [CrossRef]
- Aikawa, R.; Komuro, I.; Yamazaki, T.; Zou, Y.; Kudoh, S.; Zhu, W.; Kadowaki, T.; Yazaki, Y. Rho family small G proteins play critical roles in mechanical stress-induced hypertrophic responses in cardiac myocytes. Circ. Res. 1999, 84, 458–466. [Google Scholar] [CrossRef]
- Derangeon, M.; Bourmeyster, N.; Plaisance, I.; Pinet-Charvet, C.; Chen, Q.; Duthe, F.; Popoff, M.R.; Sarrouilhe, D.; Hervé, J.-C. RhoA GTPase and F-actin dynamically regulate the permeability of Cx43-made channels in rat cardiac myocytes. J. Biol. Chem. 2008, 283, 30754–30765. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Ha, T.; Zhang, X.; Wang, X.; Liu, L.; Kalbfleisch, J.; Singh, K.; Williams, D.; Li, C. The Toll-like receptor 9 ligand, CpG oligodeoxynucleotide, attenuates cardiac dysfunction in polymicrobial sepsis, involving activation of both phosphoinositide 3 kinase/Akt and extracellular-signal-related kinase signaling. J. Infect. Dis. 2013, 207, 1471–1479. [Google Scholar] [CrossRef]
- Baliga, R.R.; Pimental, D.R.; Zhao, Y.-Y.; Simmons, W.W.; Marchionni, M.A.; Sawyer, D.B.; Kelly, R.A. NRG-1-induced cardiomyocyte hypertrophy. Role of PI-3-kinase, p70S6K, and MEK-MAPK-RSK. Am. J. Physiol. Circ. Physiol. 1999, 277, 2026–2037. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Gu, X.; Li, Z.; Li, X.; Li, H.; Chang, J.; Chen, P.; Jin, J.; Xi, B.; Chen, D.; et al. Neuregulin-1/erbB-activation improves cardiac function and survival in models of ischemic, dilated, and viral cardiomyopathy. J. Am. Coll. Cardiol. 2006, 48, 1438–1447. [Google Scholar] [CrossRef] [PubMed]
- Rentschler, S.; Zander, J.; Meyers, K.; France, D.; Levine, R.; Porter, G.; Rivkees, S.A.; Morley, G.E.; Fishman, G.I. Neuregulin-1 promotes formation of the murine cardiac conduction system. Proc. Natl. Acad. Sci. USA 2002, 99, 10464–10469. [Google Scholar] [CrossRef]
- Bell, D.; McDermott, B. Intermedin (adrenomedullin-2): A novel counter-regulatory peptide in the cardiovascular and renal systems. Br. J. Pharmacol. 2008, 153, 247–262. [Google Scholar] [CrossRef]
- Lagu, T.; Rothberg, M.B.; Shieh, M.-S.; Pekow, P.S.; Steingrub, J.S.; Lindenauer, P.K. Hospitalizations, costs, and outcomes of severe sepsis in the United States 2003 to 2007. Crit. Care Med. 2012, 40, 754–761. [Google Scholar] [CrossRef]
- Pfister, D.; Siegemund, M.; Dell-Kuster, S.; Smielewski, P.; Rüegg, S.; Strebel, S.P.; Marsch, S.C.; Pargger, H.; Steiner, L.A. Cerebral perfusion in sepsis-associated delirium. Crit. Care 2008, 12, 63. [Google Scholar] [CrossRef]
- Jianjun, Z.; Baochun, Z.; Limei, M.; Lijun, L. Exploring the beneficial role of ROCK inhibitors in sepsis-induced cerebral and cognitive injury in rats. Fundam. Clin. Pharmacol. 2021, in press. [Google Scholar] [CrossRef]
- Barichello, T.; Martins, M.; Reinke, A.; Constantino, L.; Machado, R.; Valvassori, S.; Moreira, J.; Quevedo, J.; Dal-Pizzol, F. Behavioral deficits in sepsis-surviving rats induced by cecal ligation and perforation. Braz. J. Med Biol. Res. 2007, 40, 831–837. [Google Scholar] [CrossRef]
- Leite, F.B.; Prediger, R.D.; Silva, M.V.; De Sousa, J.B.; Carneiro, F.P.; Gasbarri, A.; Tomaz, C.; Queiroz, A.J.; Martins, N.T.; Ferreira, V.M. Role of nicotine on cognitive and behavioral deficits in sepsis-surviving rats. Brain Res. 2013, 1507, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Si, Y.; Meng, Q.; Jing, L.; Chen, L.; Zhang, Y.; Bao, H. Suppression of the RAC1/MLK3/p38 signaling pathway by β-elemene alleviates sepsis-associated encephalopathy in mice. Front. Neurosci. 2019, 13, 358. [Google Scholar] [CrossRef] [PubMed]
- Angus, D.C.; Linde-Zwirble, W.T.; Lidicker, J.; Clermont, G.; Carcillo, J.; Pinsky, M.R. Epidemiology of severe sepsis in the United States: Analysis of incidence, outcome, and associated costs of care. Crit. Care Med. 2001, 29, 1303–1310. [Google Scholar] [CrossRef]
- Hasan, Z.; Palani, K.S.H.; Zhang, S.; Lepsenyi, M.; Hwaiz, R.; Rahman, M.; Syk, I.; Jeppsson, B.; Thorlacius, H. Rho kinase regulates induction of T-cell immune dysfunction in abdominal sepsis. Infect. Immun. 2013, 81, 2499–2506. [Google Scholar] [CrossRef]
- Palani, K.S.H.; Rahman, M.; Hasan, Z.; Zhang, S.; Qi, Z.; Jeppsson, B.; Thorlacius, H. Rho-kinase regulates adhesive and mechanical mechanisms of pulmonary recruitment of neutrophils in abdominal sepsis. Eur. J. Pharmacol. 2012, 682, 181–187. [Google Scholar] [CrossRef]
- Tasaka, S.; Koh, H.; Yamada, W.; Shimizu, M.; Ogawa, Y.; Hasegawa, N.; Yamaguchi, K.; Ishii, Y.; Richer, S.E.; Doerschuk, C.M.; et al. Attenuation of endotoxin-induced acute lung injury by the Rho-associated kinase inhibitor, Y-27632. Am. J. Respir. Cell Mol. Biol. 2005, 32, 504–510. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.; Wang, Z.; Zhang, X.-H.; Wu, W.-K. Fasudil attenuates lipopolysaccharide-induced acute lung injury in mice through the Rho/Rho kinase pathway. Med. Sci. Monit. 2010, 16, 112–118. [Google Scholar]
- Cinel, I.; Ark, M.; Dellinger, P.; Karabacak, T.; Tamer, L.; Cinel, L.; Michael, P.; Hussein, S.; Parrillo, J.E.; Kumar, A.; et al. Involvement of Rho kinase (ROCK) in sepsis-induced acute lung injury. J. Thorac. Dis. 2012, 4, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Chen, X.; Han, Y.; Li, C.; Chen, P.; Su, S.; Zhang, Y.; Pan, Z. Rho kinase inhibition by fasudil suppresses lipopolysaccharide-induced apoptosis of rat pulmonary microvascular endothelial cells via JNK and p38 MAPK pathway. Biomed. Pharmacother. 2014, 68, 267–275. [Google Scholar] [CrossRef]
- Chen, T.; Guo, Q.; Wang, H.; Zhang, H.; Wang, C.; Zhang, P.; Meng, S.; Li, Y.; Ji, H.; Yan, T. Effects of esculetin on lipopolysaccharide (LPS)-induced acute lung injury via regulation of RhoA/Rho Kinase/NF-кB pathways in vivo and in vitro. Free Radic. Res. 2015, 49, 1459–1468. [Google Scholar] [CrossRef]
- Siddiqui, M.R.; Akhtar, S.; Shahid, M.; Tauseef, M.; McDonough, K.; Shanley, T.P. miR-144-mediated inhibition of ROCK1 protects against LPS-induced lung endothelial hyperpermeability. Am. J. Respir. Cell Mol. Biol. 2019, 61, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Hu, X.; Li, R.; Liu, B.; Zheng, X.; Fang, Z.; Chen, L.; Chen, W.; Min, L.; Hu, S. LncRNA THRIL aggravates sepsis-induced acute lung injury by regulating miR-424/ROCK2 axis. Mol. Immunol. 2020, 126, 111–119. [Google Scholar] [CrossRef]
- Iba, T.; Levy, J.H.; Warkentin, T.E.; Thachil, J.; Van Der Poll, T.; Levi, M. Diagnosis and management of sepsis-induced coagulopathy and disseminated intravascular coagulation. J. Thromb. Haemost. 2019, 17, 1989–1994. [Google Scholar] [CrossRef] [PubMed]
- Vardon-Bounes, F.; Ruiz, S.; Gratacap, M.-P.; Garcia, C.; Payrastre, B.; Minville, V. Platelets are critical key players in sepsis. Int. J. Mol. Sci. 2019, 20, 3494. [Google Scholar] [CrossRef] [PubMed]
- Aburima, A.; Walladbegi, K.; Wake, J.D.; Naseem, K.M. cGMP signaling inhibits platelet shape change through regulation of the RhoA-Rho kinase-MLC phosphatase signaling pathway. J. Thromb. Haemost. 2017, 15, 1668–1678. [Google Scholar] [CrossRef]
- Nishioka, H.; Horiuchi, H.; Tabuchi, A.; Yoshioka, A.; Shirakawa, R.; Kita, T. Small GTPase Rho regulates thrombin-induced platelet aggregation. Biochem. Biophys. Res. Commun. 2001, 280, 970–975. [Google Scholar] [CrossRef]
- Slotta, J.E.; Braun, O.Ö.; Menger, M.D.; Thorlacius, H. Central role of Rho kinase in lipopolysaccharide-induced platelet capture on venous endothelium. J. Investig. Med. 2008, 56, 720–725. [Google Scholar] [CrossRef]
- Reddi, B.A.; Iannella, S.M.; O’Connor, S.N.; Deane, A.M.; Willoughby, S.R.; Wilson, D.P. Attenuated platelet aggregation in patients with septic shock is independent from the activity state of myosin light chain phosphorylation or a reduction in Rho kinase-dependent inhibition of myosin light chain phosphatase. Intensive Care Med. Exp. 2015, 3, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Spates, S.T.; Cullen, J.J.; Ephgrave, K.S.; Hinkhouse, M.M. Effect of endotoxin on canine colonic motility and transit. J. Gastrointest. Surg. 1998, 2, 391–398. [Google Scholar] [CrossRef]
- Yoseph, B.P.; Klingensmith, N.J.; Liang, Z.; Breed, E.; Burd, E.M.; Mittal, R.; Dominguez, J.A.; Petrie, B.; Ford, M.L.; Coopersmith, C.M. Mechanisms of intestinal barrier dysfunction in sepsis. Shock 2016, 46, 52–59. [Google Scholar] [CrossRef]
- Du, L.; Kim, J.J.; Shen, J.; Dai, N. Crosstalk between inflammation and ROCK/MLCK signaling pathways in gastrointestinal disorders with intestinal hyperpermeability. Gastroenterol. Res. Pract. 2016, 2016, 7374197. [Google Scholar] [CrossRef] [PubMed]
- Haydont, V.; Bourgier, C.; Vozenin-Brotons, M.-C. Rho/ROCK pathway as a molecular target for modulation of intestinal radiation-induced toxicity. Br. J. Radiol. 2007, 80, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Blikslager, A.T. The regulation of intestinal mucosal barrier by myosin light chain kinase/Rho kinases. Int. J. Mol. Sci. 2020, 21, 3550. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.A.; Chen, X.; Sabbatini, M.E. Small G proteins as key regulators of pancreatic digestive enzyme secretion. Am. J. Physiol. Metab. 2009, 296, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Ares, G.; Buonpane, C.; Sincavage, J.; Yuan, C.; Wood, D.R.; Hunter, C.J. Caveolin 1 is associated with upregulated claudin 2 in necrotizing enterocolitis. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Gong, M.C.; Iizuka, K.; Nixon, G.; Browne, J.P.; Hall, A.; Eccleston, J.F.; Sugai, M.; Kobayashi, S.; Somlyo, A.V. Role of guanine nucleotide-binding proteins--ras-family or trimeric proteins or both—In Ca2+ sensitization of smooth muscle. Proc. Natl. Acad. Sci. USA 1996, 93, 1340–1345. [Google Scholar] [CrossRef]
- Buonpane, C.; Yuan, C.Y.; Wood, D.R.; Ares, G.; Klonoski, S.C.; Hunter, C.J. ROCK1 inhibitor stabilizes E-cadherin and improves barrier function in experimental necrotizing enterocolitis. Am. J. Physiol. Liver Physiol. 2020, 318, 781–792. [Google Scholar] [CrossRef]
- Wu, F.; Li, H.; Zhang, H.; Liao, Y.; Ren, H.; Wu, J.; Zheng, D. Phosphorylated-myosin light chain mediates the destruction of small intestinal epithelial tight junctions in mice with acute liver failure. Mol. Med. Rep. 2021, 23, 1–7. [Google Scholar] [CrossRef]
- Ozdemir, D.; Cilaker, S.; Tugyan, K.; Dagdelen, M.K.; Derinoz, O.; Guneli, E. The effect of Rho kinase inhibitor Y-27632 on endotoxemia-induced intestinal apoptosis in infant rats. J. Mol. Histol. 2011, 43, 81–87. [Google Scholar] [CrossRef]
- Li, Z.; Gao, M.; Yang, B.; Zhang, H.; Wang, K.; Liu, Z.; Xiao, X.; Yang, M. Naringin attenuates MLC phosphorylation and NF-κB activation to protect sepsis-induced intestinal injury via RhoA/ROCK pathway. Biomed. Pharmacother. 2018, 103, 50–58. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Yang, W.; Zhao, X.; Zhang, R. Effect of simvastatin on the intestinal Rho/ROCK signaling pathway in rats with sepsis. J. Surg. Res. 2018, 232, 531–538. [Google Scholar] [CrossRef]
- Yan, J.; Li, S.; Li, S. The role of the liver in sepsis. Int. Rev. Immunol. 2014, 33, 498–510. [Google Scholar] [CrossRef]
- Pizzino, G.; Bitto, A.; Pallio, G.; Irrera, N.; Galfo, F.; Interdonato, M.; Mecchio, A.; De Luca, F.; Minutoli, L.; Squadrito, F.; et al. Blockade of the JNK signalling as a rational therapeutic approach to modulate the early and late steps of the inflammatory cascade in polymicrobial sepsis. Mediat. Inflamm. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Slotta, J.E.; Laschke, M.W.; Menger, M.D.; Thorlacius, H. Rho-kinase signalling mediates endotoxin hypersensitivity after partial hepatectomy. BJS 2008, 95, 976–984. [Google Scholar] [CrossRef]
- Ding, R.; Han, J.; Zhao, D.; Hu, Z.; Ma, X. Pretreatment with Rho-kinase inhibitor ameliorates lethal endotoxemia-induced liver injury by improving mitochondrial function. Int. Immunopharmacol. 2016, 40, 125–130. [Google Scholar] [CrossRef]
- Pohlmann, S.; Scheu, S.; Ziegler, V.; Schupp, N.; Henninger, C.; Fritz, G. Hepatic Rac1 GTPase contributes to liver-mediated basal immune homeostasis and LPS-induced endotoxemia. Biochim. Biophys. Acta Bioenerg. 2018, 1865, 1277–1292. [Google Scholar] [CrossRef]
- Zarjou, A.; Agarwal, A. Sepsis and acute kidney injury. J. Am. Soc. Nephrol. 2011, 22, 999–1006. [Google Scholar] [CrossRef]
- Meyer-Schwesinger, C.; Dehde, S.; Von Ruffer, C.; Gatzemeier, S.; Klug, P.; Wenzel, U.O.; Stahl, R.A.K.; Thaiss, F.; Meyer, T.N. Rho kinase inhibition attenuates LPS-induced renal failure in mice in part by attenuation of NF-κB p65 signaling. Am. J. Physiol. Physiol. 2009, 296, 1088–1099. [Google Scholar] [CrossRef]
- Wu, X.; Guo, R.; Chen, P.; Wang, Q.; Cunningham, P.N. TNF induces caspase-dependent inflammation in renal endothelial cells through a Rho- and myosin light chain kinase-dependent mechanism. Am. J. Physiol. Physiol. 2009, 297, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Wu, X.; Hack, B.K.; Bao, L.; Cunningham, P.N. TNF causes changes in glomerular endothelial permeability and morphology through a Rho and myosin light chain kinase-dependent mechanism. Physiol. Rep. 2015, 3, 12636. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, L.; Chen, Y.; Zhang, H.; Yu, C.; Zhou, F.; Zhang, Z.; Jiang, L.; Li, R.; Ma, J.; et al. RhoA deficiency disrupts podocyte cytoskeleton and induces podocyte apoptosis by inhibiting YAP/dendrin signal. BMC Nephrol. 2016, 17, 1–12. [Google Scholar] [CrossRef]
- Mashmoushi, A.K.; Oates, J.C. Lipopolysaccharide induces inducible nitric oxide synthase-dependent podocyte dysfunction via a hypoxia-inducible factor 1α and cell division control protein 42 and Ras-related C3 botulinum toxin substrate 1 pathway. Free Radic. Biol. Med. 2015, 84, 185–195. [Google Scholar] [CrossRef]
- Hoeppner, L.H.; Sinha, S.; Wang, Y.; Bhattacharya, R.; Dutta, S.; Gong, X.; Bedell, V.; Suresh, S.; Chun, C.Z.; Ramchandran, R.; et al. RhoC maintains vascular homeostasis by regulating VEGF-induced signaling in endothelial cells. J. Cell Sci. 2015, 128, 3556–3568. [Google Scholar] [CrossRef]
- Vega, F.M.; Ridley, A.J. The RhoB small GTPase in physiology and disease. Small GTPases 2018, 9, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Wojciak-Stothard, B.; Zhao, L.; Oliver, E.; Dubois, O.; Wu, Y.; Kardassis, D.; Vasilaki, E.; Huang, M.; Mitchell, J.A.; Harrington, L.S.; et al. Role of RhoB in the regulation of pulmonary endothelial and smooth muscle cell responses to hypoxia. Circ. Res. 2012, 110, 1423–1434. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, K.; Tian, X.; Sun, M.; Wei, N.; Zhu, X.; Yang, H.; Wang, T.; Jiang, G.; Chen, K. Effects of RhoC downregulation on the angiogenesis characteristics of myeloma vascular endothelial cells. Cancer Med. 2019, 8, 3502–3510. [Google Scholar] [CrossRef]
- Wang, X.H.; Wang, Y.; Diao, F.; Lu, J. RhoB is involved in lipopolysaccharide-induced inflammation in mouse in vivo and in vitro. J. Physiol. Biochem. 2012, 69, 189–197. [Google Scholar] [CrossRef]
- Kim, J.; Islam, R.; Cho, J.Y.; Jeong, H.; Cap, K.; Park, Y.; Hossain, A.J.; Park, J. Regulation of RhoA GTPase and various transcription factors in the RhoA pathway. J. Cell. Physiol. 2018, 233, 6381–6392. [Google Scholar] [CrossRef]
- Lu, Q.; Lu, L.; Chen, W.; Chen, H.; Xu, X.; Zheng, Z. RhoA/mDia-1/profilin-1 signaling targets microvascular endothelial dysfunction in diabetic retinopathy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 253, 669–680. [Google Scholar] [CrossRef]
- Weernink, P.A.O.; Meletiadis, K.; Hommeltenberg, S.; Hinz, M.; Ishihara, H.; Schmidt, M.; Jakobs, K.H. Activation of type I phosphatidylinositol 4-phosphate 5-kinase isoforms by the Rho GTPases, RhoA, Rac1, and Cdc42. J. Biol. Chem. 2004, 279, 7840–7849. [Google Scholar] [CrossRef]
- Dasso, L.; Al-Khaled, T.; Sonty, S.; Aref, A.A. Profile of netarsudil ophthalmic solution and its potential in the treatment of open-angle glaucoma: Evidence to date. Clin. Ophthalmol. 2018, 12, 1939–1944. [Google Scholar] [CrossRef]
- Tanihara, H.; Kakuda, T.; Sano, T.; Kanno, T.; Gunji, R. Safety and efficacy of ripasudil in Japanese patients with glaucoma or ocular hypertension: 12-month interim analysis of ROCK-J, a post-marketing surveillance study. BMC Ophthalmol. 2020, 20, 1–11. [Google Scholar] [CrossRef]
- Tanaka, K.; Minami, H.; Kota, M.; Kuwamura, K.; Kohmura, E. Treatment of cerebral vasospasm with intra-arterial fasudil hydrochloride. Neurosurgery 2005, 56, 214–223. [Google Scholar] [CrossRef]
- Clinical Trials. gov. Inhibition of Rho Kinase (ROCK) with Fasudil as Disease-Modifying Treatment for ALS. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03792490?term=Rho-kinase&draw=5&rank=7 (accessed on 25 May 2021).
- Koch, J.C.; Kuttler, J.; Maass, F.; Lengenfeld, T.; Zielke, E.; Bähr, M.; Lingor, P. Compassionate use of the ROCK inhibitor fasudil in three patients with amyotrophic lateral sclerosis. Front. Neurol. 2020, 11, 173. [Google Scholar] [CrossRef]
- Nakagawa, O.; Fujisawa, K.; Ishizaki, T.; Saito, Y.; Nakao, K.; Narumiya, S. ROCK-I and ROCK-II, two isoforms of Rho-associated coiled-coil forming protein serine/threonine kinase in mice. FEBS Lett. 1996, 392, 189–193. [Google Scholar] [CrossRef]
- Feng, Y.; Lograsso, P.V.; Defert, O.; Li, R. Rho kinase (ROCK) inhibitors and their therapeutic potential. J. Med. Chem. 2015, 59, 2269–2300. [Google Scholar] [CrossRef]
| System and Model | Component(s) Evaluated | Experimental Setup | Impact on the System and Main Findings a | Ref. |
|---|---|---|---|---|
| HLMVEC; LPS | Rac1, RhoA | Indirect modulation + siRNA (includes in vivo evaluation) | Activation of Rac1 and inhibition of RhoA prevents vascular leakage; stabilizes VE-cadherin. | [43] |
| ROCK | Direct inhibition + siRNA | ROCK can prevent or enhance vascular leakage. | [44] | |
| RhoA | Direct inhibition + siRNA (includes in vivo evaluation) | Inhibition of RhoA nitration prevents vascular leakage. | [72] | |
| RhoA, ROCK | Indirect modulation + direct inhibition | Inhibition of RhoA/ROCK prevents vascular leakage. | [51] | |
| Rac1 | Direct modulation (includes in vivo evaluation) | Prevention of Rac1 nitration limits vascular leakage. | [73] | |
| HLMVEC; LTA + PepG | MCLP, MLC | Direct inhibition (includes in vivo evaluation) | Inhibition of ROCK prevents vascular leakage. | [53] |
| HLMVEC; TNF-α | RhoA, ROCK | Direct inhibition | Inhibition of RhoA/ROCK pathway prevents vascular leakage. | [55] |
| HUVEC; LPS | MLC, pMLC | Direct inhibition | Inhibition of ROCK prevents vascular leakage. | [31] |
| RhoA, GEF-H1 | iRNA | Reduction in RhoA activation disfavor inflammatory pathway. | [62] | |
| RhoA, MLCP | siRNA | RhoA inhibition prevents vascular leakage and stabilizes VE-cadherin. | [39] | |
| ROCK, RhoA, MLCP | Direct inhibition (includes in vivo evaluation) | Inhibition of ROCK prevents vascular leakage. | [66] | |
| ROCK, GEF-H1 | Direct inhibition + siRNA | Inhibition of ROCK and GEF-H1 prevents vascular leakage, stabilizes adherens and tight junctions. | [89] | |
| RhoA, Rac, Cdc42 | Indirect modulation + siRNA | Inhibition of RhoA and Rac prevents vascular leakage, stabilizes junctions, and disfavors inflammation. | [92] | |
| RhoA | Direct inhibition | Inhibition of ROCK prevents vascular leakage and stabilizes VE-cadherin. | [40] | |
| ROCK | Indirect modulation + direct inhibition | Downregulation of ROCK disfavor inflammatory pathway. | [90] | |
| RhoA, ROCK | Direct inhibition + siRNA | Inhibition of ROCK reduces stress fiber formation. | [91] | |
| HUVEC; TNF-α | RhoA, ROCK, MLCP | Indirect modulation | Inhibition of ROCK prevents vascular leakage and stabilizes VE-cadherin. | [56] |
| HUVEC; HBP | ROCK | Indirect modulation + direct inhibition | Inhibition of ROCK prevents vascular leakage. | [50] |
| HVEC; thrombin | RhoA, ROCK | Indirect modulation + siRNA | Inhibition of ROCK prevents vascular leakage, stabilizes VE-cadherin, reduces stress fiber formation. | [49] |
| HPMEC; | ROCK | Direct inhibition | Inhibition of ROCK disfavor inflammatory and coagulation pathways. | [64] |
| LPS | ROCK | Direct inhibition | Inhibition of ROCK reduces vascular leakage and apoptosis. | [35] |
| Rho-GTP | Indirect modulation | Downregulation of ROCK prevents vascular leakage, stabilizes protein junctions (i.e., VE-cadherin). | [94] | |
| HPAEC; LPS | Rac, Cdc42, MLC | Indirect modulation | Rac1 and Cdc42 activation prevents vascular leakage, stabilizes VE-cadherin. | [93] |
| HPAEC; IL-6 | ROCK | Direct inhibition + siRNA (includes in vivo evaluation) | Inhibition of ROCK prevents vascular leakage, stabilizes VE-cadherin, avoids leucocyte adhesion. | [58] |
| HPAEC; Sthaphyloccocus aureus | Rho-GEF-H1 | siRNA | Inhibition of GEF-H1 prevents vascular leakage, disfavor inflammatory pathway. | [63] |
| HDMEC; LPS | RhoA, Rac1 | Direct inhibition | Downregulation of Rac1 worse vascular leakage. | [57] |
| HDMEC; | RhoA | Indirect modulation | Inhibition of RhoA reduces leukocyte migration. | [46] |
| TNF-α | ROCK, MLC | Direct inhibition + siRNA | Inhibition of ROCK prevents vascular leakage. | [59] |
| HDMEC; CNF-1 | RhoA, Rac1, Cdc42 | Direct inhibition or activation | ROCK enhances and Rac1/Cdc42 reduce vascular leakage; Rac1/Cdc42 inactivation worse junction stability. | [52] |
| MPVEC; LPS | ROCK | miRNA | Inhibition of ROCK prevents apoptosis and inflammation. | [95] |
| PAEC; CNF-1 | RhoA, Rac1, Cdc42, ROCK | Direct inhibition or activation | ROCK enhances and Rac1/Cdc42 reduce vascular leakage; Rac1/Cdc42 inactivation worse junction stability. | [52] |
| ROCK, pMLC | RNAi (includes in vivo evaluation) | Inhibition of ROCK prevents vascular leakage, stabilizes connexin 43. | [33] | |
| MyEnd; CNF-1 | Rac1, Cdc42 | Indirect modulation (includes in vivo evaluation) | Upregulation of Rac1 and Cdc42 improves the endothelial barrier. | [48] |
| RhoA, Rac1, Cdc42, ROCK | Direct inhibition or activation | ROCK enhances and Rac1/Cdc42 reduce vascular leakage; Rac1/Cdc42 inactivation worse junction stability. | [52] | |
| MesEnd; CNF-1 | RhoA, Rac1, Cdc42, ROCK | Direct inhibition or activation | ROCK enhances and Rac1/Cdc42 reduce vascular leakage; Rac1/Cdc42 inactivation worse junction stability. | [52] |
| bEnd.3; LPS | RhoA; GEF | Direct inhibition + siRNA | Inhibition of RhoA and GEF prevents vascular leakage, stabilizes zonnula occludent 1 and reduces stress fiber formation. | [88] |
| Pericytes; LPS | RhoA | Indirect modulation | Activation of RhoA prevents vascular leakage. | [45] |
| LSEC; LPS | ROCK | Direct inhibition | Inhibition of ROCK and its nitration prevents vascular leakage. | [71] |
| Mice lung; LPS | Rho-GTP, ROCK, MLCP | Indirect modulation | Downregulation of Rho-GTP, ROCK, and MLCP prevents vascular leakage. | [34] |
| Rat mesenteric artery; LPS | ROCK | Direct inhibition | Inhibition of ROCK prevents vascular leakage and avoids leucocyte adhesion. | [67] |
| Guinea pig skin; LPS | ROCK | Direct inhibition | Inhibition of ROCK prevents vascular leakage. | [36] |
| System and Model | Component(s) Evaluated | Experimental Setup | Impact on the System and Main Findings a | Ref. |
|---|---|---|---|---|
| SM artery; Rats; LPS b | ROCK | Direct inhibition + functional + molecular approaches | Upregulation of ROCK enhances contractile responses. | [98] |
| RhoA, ROCK | Indirect modulation + direct inhibition + functional + molecular approaches | RhoA is reduced; activation of the pathway improves contractile responses. | [110] | |
| SM artery; Rabbits; IL-1β c/TNF-α c | ROCK, MLCP | Direct inhibition + functional + molecular approaches | Inhibition of ROCK contributes to IL-1β-induced vascular hyporeactivity. | [103] |
| ROCK, MLCP | Functional + molecular approaches | Downregulation of ROCK contributes to TNF-α-induced vascular hyporeactivity. | [104] | |
| RM artery; Rats; LPSb | RhoA, ROCK, MLCP | Direct inhibition + functional + molecular approaches | Upregulation of Rho components fails to trigger contractile responses; RhoA/ROCK is inhibited by the nitric oxide/guanylate cyclase pathway. | [101] |
| Aorta; Rats; LPS b | MLC, ROCK | Functional + molecular approaches | Hyporeactivity to ET-1 does not involve the RhoA/ROCK pathway. | [100] |
| RhoA, ROCK, MLCP | Direct inhibition + functional + molecular approaches | The activity of RhoA increases increases (1–2 h) and reduces (4–6 h) after LPS. Norepinephrine-induced vasoconstriction is more sensitive to ROCK inhibition. | [102] | |
| Aorta; Rats; CLP | RhoA, ROCK, MLCP | Direct inhibition + functional + molecular approaches | Upregulation of RhoA and ROCK at 60 days after CLP; augmented activation of RhoA/ROCK pathway accounts for enhanced contractile responses to angiotensin II. | [108] |
| Renal vascular bed and blood pressure; Rats; LPS b | RhoA, ROCK, MLCP | Direct inhibition + functional + molecular approaches (includes in vivo treatment/evaluation) | Increased RhoA/ROCK in the renal vascular bed accounts for enhanced pressor responses to vasopressin. | [109] |
| Femoral artery; Mice; CLP | MLCP | Functional + molecular approaches | Thromboxane A2-induced vasoconstriction and phosphorylation of MLCP were reduced 5 days after CLP. | [107] |
| Pulmonary artery; Rats; LPS c | RhoA | Direct activation + molecular approaches | RhoA activation prevents vascular damage/F-actin rearrangement. | [96] |
| VSMC; Rats; LPS/IL-1β c | RhoA, ROCK, Rac1, MLCP | Direct inhibition + molecular approaches | LPS reduces RhoA activity. IL-1β increases RhoA activity. ROCK negatively modulates NF-κB. | [97] |
| Blood pressure; Rats; CLP | RhoA, ROCK | Direct inhibition + systemic effects + molecular approaches (includes in vivo treatment/evaluation) | RhoA/ROCK pathway is up-regulated; inhibition of ROCK improves blood pressure. | [35] |
| Cardiomyocytes; Rats; LPS c | RhoA | Molecular approaches | RhoA expression and activity are further increased by LPS and nitric oxide in tissues from diabetic animals. | [111] |
| Heart; Mice; LPS b | Rac1 | Molecular approaches | Lack of Rac1 reduces inflammatory markers, including TNF. | [112] |
| ROCK | Direct inhibition + functional + molecular approaches (includes in vivo treatment) | Inhibition of ROCK improved contractile function and mitochondrial respiration. | [113] | |
| Cardiomyocytes and heart; Mice; LPS b,c | Rac1 | Functional + molecular approaches | Rac1 expression and activity are increased; lack of Rac1 reduces TNF and improves cardiac function. | [114] |
| H9C2; TNF-α c | RhoA, Cdc42, Rac1 | Direct inhibition + molecular approaches | TNF-α increases RhoA, and ROCK inhibition attenuates mitochondrial fragmentation. | [115] |
| Heart and H9C2; Rats; LPS | RhoA, ROCK | Indirect modulation + molecular approaches (includes in vivo treatment) | RhoA/ROCK proteins are increased, and the reduction is associated with improved cardiac function and reduced apoptosis. | [116] |
| Papillary muscle; Rats; CLP | ROCK | Indirect modulation + functional + molecular approaches (includes in vivo treatment) | Block of ROCK avoids intermedin 1-53-mediated cardiac troponin I phosphorylation. | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hahmeyer, M.L.d.S.; da Silva-Santos, J.E. Rho-Proteins and Downstream Pathways as Potential Targets in Sepsis and Septic Shock: What Have We Learned from Basic Research. Cells 2021, 10, 1844. https://doi.org/10.3390/cells10081844
Hahmeyer MLdS, da Silva-Santos JE. Rho-Proteins and Downstream Pathways as Potential Targets in Sepsis and Septic Shock: What Have We Learned from Basic Research. Cells. 2021; 10(8):1844. https://doi.org/10.3390/cells10081844
Chicago/Turabian StyleHahmeyer, Maria Luísa da Silveira, and José Eduardo da Silva-Santos. 2021. "Rho-Proteins and Downstream Pathways as Potential Targets in Sepsis and Septic Shock: What Have We Learned from Basic Research" Cells 10, no. 8: 1844. https://doi.org/10.3390/cells10081844
APA StyleHahmeyer, M. L. d. S., & da Silva-Santos, J. E. (2021). Rho-Proteins and Downstream Pathways as Potential Targets in Sepsis and Septic Shock: What Have We Learned from Basic Research. Cells, 10(8), 1844. https://doi.org/10.3390/cells10081844






