Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: A Connecting Hypothesis
Abstract
1. Introduction
2. Relationship between Hormones and the Pathogenesis of Acne
2.1. Hormones and the Sebaceous Gland
2.1.1. Androgens
2.1.2. IGF-1
2.1.3. Estrogens
2.1.4. CRH and Cortisol
2.1.5. Cytokines and Inflammation
3. Evidence of Human Exposures to EDCs
3.1. Classes of EDCs
3.2. Routes of Exposure
3.3. Evidence of Exposure
4. Effects of EDCs on Endogenous Hormone Pathways
4.1. EDC-Mediated Alteration of Endogenous Hormone Levels
4.1.1. Androgens
4.1.2. Estrogens and IGF-1
4.1.3. CRH and Cortisol
5. EDC-Mediated Interference with Hormone Receptor Function
5.1. EDCs, Hormones and their Receptors
5.2. Molecular Mechanisms of EDC Action on Nuclear Hormone Receptors
5.2.1. Effects on EDCs on Classical Nuclear Hormone Receptor Pathways
5.2.2. Ligand Binding
5.2.3. Agonist/Antagonist Activity of EDCs
5.2.4. Recruitment of Transcriptional Coregulators
5.2.5. DNA Binding and Gene Expression
5.2.6. EDCs and Nongenomic NHR Signaling
5.2.7. Epigenetic Effects of EDCS
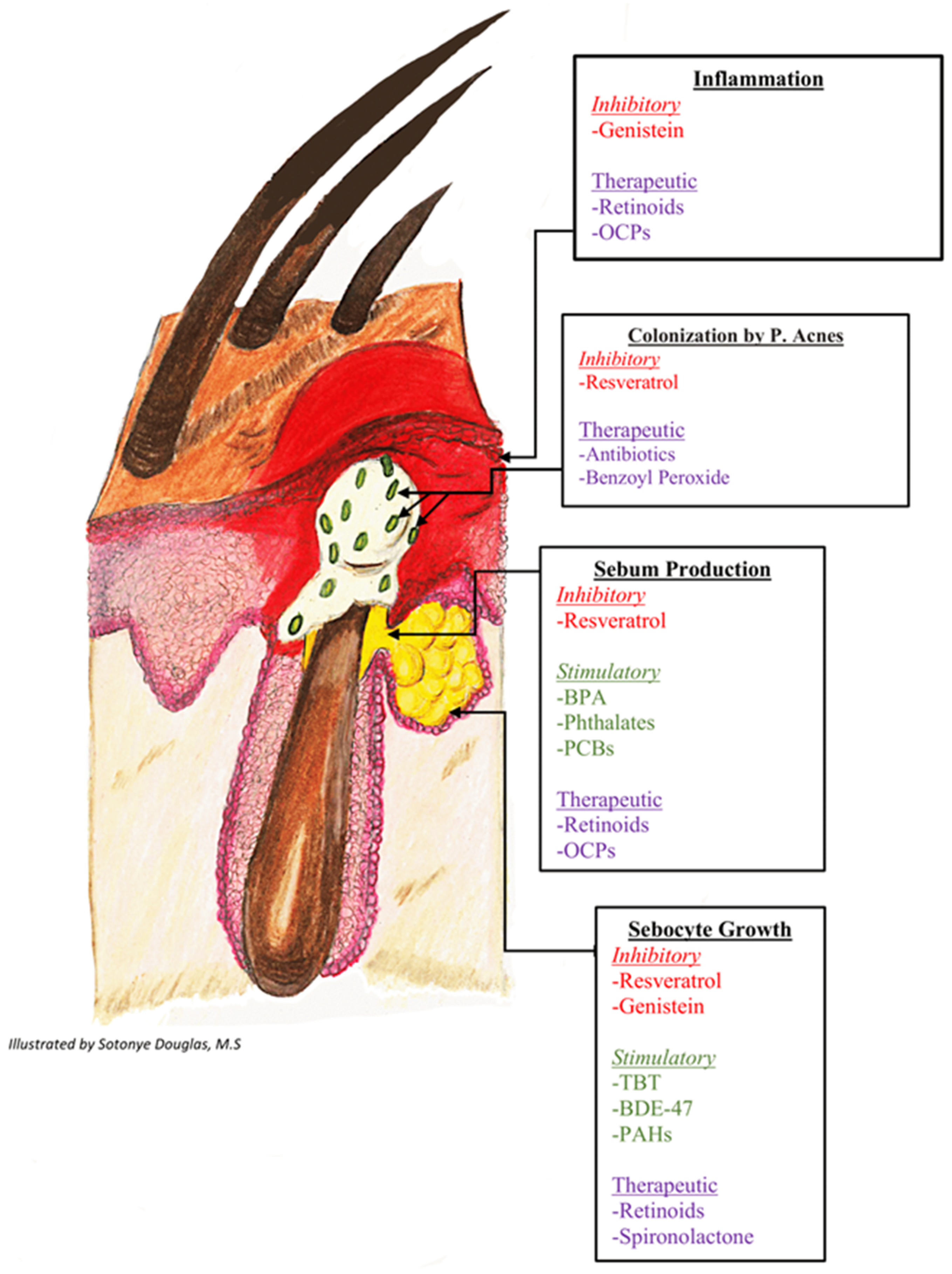
6. Potential Mechanistic Links Between EDC Exposure and Acne
6.1. Androgens
6.2. Estrogens
6.3. CRH and Cortisol
6.4. PPARy
7. EDCs and Acne Therapeutics
7.1. EDCs and Retinoids
7.2. EDCs and Hormonal Therapies
8. Discussion
- BPA exposure can be reduced with the use of products labeled BPA free, and by replacing plastics with glassware for food storage and cooking.
- Phthalate consumption can be minimized with by avoiding plastics in food prep and storage, consumption of filtered water, and the use of fragrance-free cleaning products and cosmetics.
- Exposure to hormones in dairy products can be minimized by the consumption of organic products. Likewise, pesticide consumption can be avoided with organic produce.
- Mineral sunscreens containing zinc oxide or titanium dioxide as active ingredients are much safer alternatives to those containing oxybenzone (Benzophenone-3). Oxybenzone is an established EDC with estrogenic and androgenic activities, and the agent is prevalent in the body fluids of humans [93].
- Minimize exposure to air-born EDCs (Dioxins) with smoking cessation and by wearing facial masks with air filters when outside on poor air quality days or in cities with high levels of air pollution: https://www.iqair.com/us/world-air-quality-ranking; accessed on 5 May 2021.
- Topical products containing tea tree and lavender oils should be used with caution in acne patients given that these agents also possess estrogenic and androgenic properties.
- Based on existing evidence, acne patients may be counseled to follow a low glycemic, plant-rich diet.
9. Future Areas of Research
Funding
Acknowledgments
Conflicts of Interest
References
- Williams, H.C.; Dellavalle, R.P.; Garner, S. Acne vulgaris. Lancet 2012, 379, 361–372. [Google Scholar] [CrossRef]
- Hay, R.J.; Johns, N.E.; Williams, H.C.; Bolliger, I.W.; Dellavalle, R.P.; Margolis, D.J.; Marks, R.; Naldi, L.; Weinstock, M.A.; Wulf, S.K.; et al. The global burden of skin disease in 2010: An analysis of the prevalence and impact of skin conditions. J. Investig. Derm. 2014, 134, 1527–1534. [Google Scholar] [CrossRef]
- Collier, C.N.; Harper, J.C.; Cafardi, J.A.; Cantrell, W.C.; Wang, W.; Foster, K.W.; Elewski, B.E. The prevalence of acne in adults 20 years and older. J. Am. Acad. Derm. 2008, 58, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Dreno, B.; Bagatin, E.; Blume-Peytavi, U.; Rocha, M.; Gollnick, H. Female type of adult acne: Physiological and psychological considerations and management. J. Dtsch. Derm. Ges. 2018, 16, 1185–1194. [Google Scholar] [CrossRef]
- Altunay, I.K.; Ozkur, E.; Dalgard, F.J.; Gieler, U.; Tomas Aragones, L.; Lien, L.; Poot, F.; Jemec, G.B.; Misery, L.; Szabo, C.; et al. Psychosocial Aspects of Adult Acne: Data from 13 European Countries. Acta Derm. Venereol. 2020, 100, adv00051. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Chen, W.C.; Thornton, M.J.; Qin, K.; Rosenfield, R. Sexual hormones in human skin. Horm. Metab. Res. 2007, 39, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Zaenglein, A.L. Acne Vulgaris. N. Engl. J. Med. 2018, 379, 1343–1352. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef] [PubMed]
- Moosa, A.; Shu, H.; Sarachana, T.; Hu, V.W. Are endocrine disrupting compounds environmental risk factors for autism spectrum disorder? Horm. Behav. 2018, 101, 13–21. [Google Scholar] [CrossRef]
- Makrantonaki, E.; Ganceviciene, R.; Zouboulis, C. An update on the role of the sebaceous gland in the pathogenesis of acne. Derm. 2011, 3, 41–49. [Google Scholar] [CrossRef]
- Ju, Q.; Tao, T.; Hu, T.; Karadag, A.S.; Al-Khuzaei, S.; Chen, W. Sex hormones and acne. Clin. Derm. 2017, 35, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Youn, S.W.; Park, E.S.; Lee, D.H.; Huh, C.H.; Park, K.C. Does facial sebum excretion really affect the development of acne? Br. J. Derm. 2005, 153, 919–924. [Google Scholar] [CrossRef]
- Sertznig, P.; Reichrath, J. Peroxisome proliferator-activated receptors (PPARs) in dermatology: Challenge and promise. Derm. 2011, 3, 130–135. [Google Scholar] [CrossRef]
- Saric-Bosanac, S.; Clark, A.K.; Sivamani, R.K.; Shi, V.Y. The role of hypothalamus-pituitary-adrenal (HPA)-like axis in inflammatory pilosebaceous disorders. Derm. Online J. 2020, 26. Available online: https://escholarship.org/uc/item/8949296f (accessed on 8 June 2021).
- Arora, M.K.; Yadav, A.; Saini, V. Role of hormones in acne vulgaris. Clin. Biochem. 2011, 44, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Ganceviciene, R.; Bohm, M.; Fimmel, S.; Zouboulis, C.C. The role of neuropeptides in the multifactorial pathogenesis of acne vulgaris. Derm. Endocrinol. 2009, 1, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Ganceviciene, R.; Graziene, V.; Fimmel, S.; Zouboulis, C.C. Involvement of the corticotropin-releasing hormone system in the pathogenesis of acne vulgaris. Br. J. Derm. 2009, 160, 345–352. [Google Scholar] [CrossRef]
- Yosipovitch, G.; Tang, M.; Dawn, A.G.; Chen, M.; Goh, C.L.; Huak, Y.; Seng, L.F. Study of psychological stress, sebum production and acne vulgaris in adolescents. Acta Derm. Venereol. 2007, 87, 135–139. [Google Scholar] [CrossRef]
- Chiu, A.; Chon, S.Y.; Kimball, A.B. The response of skin disease to stress: Changes in the severity of acne vulgaris as affected by examination stress. Arch. Derm. 2003, 139, 897–900. [Google Scholar] [CrossRef]
- Tanghetti, E.A. The role of inflammation in the pathology of acne. J. Clin. Aesthet. Derm. 2013, 6, 27–35. [Google Scholar]
- Tochio, T.; Tanaka, H.; Nakata, S.; Ikeno, H. Accumulation of lipid peroxide in the content of comedones may be involved in the progression of comedogenesis and inflammatory changes in comedones. J. Cosmet. Derm. 2009, 8, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, M.; Alestas, T.; Flori, E.; Mastrofrancesco, A.; Zouboulis, C.C.; Picardo, M. Peroxidated squalene induces the production of inflammatory mediators in HaCaT keratinocytes: A possible role in acne vulgaris. J. Investig. Derm. 2006, 126, 2430–2437. [Google Scholar] [CrossRef]
- Kang, S.; Cho, S.; Chung, J.H.; Hammerberg, C.; Fisher, G.J.; Voorhees, J.J. Inflammation and extracellular matrix degradation mediated by activated transcription factors nuclear factor-kappaB and activator protein-1 in inflammatory acne lesions in vivo. Am. J. Pathol. 2005, 166, 1691–1699. [Google Scholar] [CrossRef]
- Lee, W.J.; Jung, H.D.; Chi, S.G.; Kim, B.S.; Lee, S.J.; Kim, D.W.; Kim, M.K.; Kim, J.C. Effect of dihydrotestosterone on the upregulation of inflammatory cytokines in cultured sebocytes. Arch. Derm. Res. 2010, 302, 429–433. [Google Scholar] [CrossRef]
- La Merrill, M.A.; Vandenberg, L.N.; Smith, M.T.; Goodson, W.; Browne, P.; Patisaul, H.B.; Guyton, K.Z.; Kortenkamp, A.; Cogliano, V.J.; Woodruff, T.J.; et al. Consensus on the key characteristics of endocrine-disrupting chemicals as a basis for hazard identification. Nat. Rev. Endocrinol. 2020, 16, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Welshons, W.V.; Nagel, S.C.; vom Saal, F.S. Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 2006, 147, S56–S69. [Google Scholar] [CrossRef]
- Swedenborg, E.; Ruegg, J.; Makela, S.; Pongratz, I. Endocrine disruptive chemicals: Mechanisms of action and involvement in metabolic disorders. J. Mol. Endocrinol. 2009, 43, 1–10. [Google Scholar] [CrossRef]
- Hall, J.M.; Korach, K.S. Analysis of the molecular mechanisms of human estrogen receptors alpha and beta reveals differential specificity in target promoter regulation by xenoestrogens. J. Biol. Chem. 2002, 277, 44455–44461. [Google Scholar] [CrossRef]
- Walker, C.L. Minireview: Epigenomic Plasticity and Vulnerability to EDC Exposures. Mol. Endocrinol. 2016, 30, 848–855. [Google Scholar] [CrossRef]
- Janesick, A.S.; Shioda, T.; Blumberg, B. Transgenerational inheritance of prenatal obesogen exposure. Mol. Cell. Endocrinol. 2014, 398, 31–35. [Google Scholar] [CrossRef]
- Li, G.; Chang, H.; Xia, W.; Mao, Z.; Li, Y.; Xu, S. F0 maternal BPA exposure induced glucose intolerance of F2 generation through DNA methylation change in Gck. Toxicol. Lett. 2014, 228, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Stel, J.; Legler, J. The Role of Epigenetics in the Latent Effects of Early Life Exposure to Obesogenic Endocrine Disrupting Chemicals. Endocrinology 2015, 156, 3466–3472. [Google Scholar] [CrossRef]
- Nelson, A.M.; Zhao, W.; Gilliland, K.L.; Zaenglein, A.L.; Liu, W.; Thiboutot, D.M. Temporal changes in gene expression in the skin of patients treated with isotretinoin provide insight into its mechanism of action. Derm. Endocrinol. 2009, 1, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. Executive Summary to EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, 593–602. [Google Scholar] [CrossRef]
- Azzouz, A.; Rascón, A.J.; Ballesteros, E. Simultaneous determination of parabens, alkylphenols, phenylphenols, bisphenol A and triclosan in human urine, blood and breast milk by continuous solid-phase extraction and gas chromatography–mass spectrometry. J. Pharm. Biomed. Anal. 2016, 119, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Genuis, S.J.; Beesoon, S.; Lobo, R.A.; Birkholz, D. Human Elimination of Phthalate Compounds: Blood, Urine, and Sweat (BUS) Study. Sci. World, J. 2012, 2012. [Google Scholar] [CrossRef]
- WHO. Human Biomonitoring: Facts and Figures; WHO: Copenhagen, Denmark, 2015; p. 88. [Google Scholar]
- Lehmler, H.-J.; Liu, B.; Gadogbe, M.; Bao, W. Exposure to Bisphenol A, Bisphenol F, and Bisphenol S in U.S. Adults and Children: The National Health and Nutrition Examination Survey 2013–2014. ACS Omega 2018, 3, 6523–6532. [Google Scholar] [CrossRef]
- DiGangi, J.; Schettler, T.; Cobbing, M.; Rossi, M. Aggregate Exposures to Phthlates in Humans; Health Care Without Harm: Washington, DC, USA, 2002; p. 50. [Google Scholar]
- Bunay, J.; Larriba, E.; Moreno, R.D.; Del Mazo, J. Chronic low-dose exposure to a mixture of environmental endocrine disruptors induces microRNAs/isomiRs deregulation in mouse concomitant with intratesticular estradiol reduction. Sci. Rep. 2017, 7, 3373. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Jacobs, D.R., Jr. New approaches to cope with possible harms of low-dose environmental chemicals. J. Epidemiol. Commun. Health 2019, 73, 193–197. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R., Jr.; Lee, D.H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and endocrine-disrupting chemicals: Low-dose effects and nonmonotonic dose responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-Disrupting Chemicals: An Endocrine Society Scientific Statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef]
- Cao, Y.; Calafat, A.M.; Doerge, D.R.; Umbach, D.M.; Bernbaum, J.C.; Twaddle, N.C.; Ye, X.; Rogan, W.J. Isoflavones in urine, saliva, and blood of infants: Data from a pilot study on the estrogenic activity of soy formula. J. Expo. Sci. Environ. Epidemiol. 2009, 19, 223–234. [Google Scholar] [CrossRef]
- Rizzati, V.; Briand, O.; Guillou, H.; Gamet-Payrastre, L. Effects of pesticide mixtures in human and animal models: An update of the recent literature. Chem. Biol. Interact. 2016, 254, 231–246. [Google Scholar] [CrossRef]
- Mazioti, M. Chemical environmental factors: Can they affect acne? Indian J. Derm. Venereol. Leprol. 2017, 83, 522–524. [Google Scholar] [CrossRef]
- Akgül, S.; Sur, Ü.; Düzçeker, Y.; Balcı, A.; Kızılkan, M.P.; Kanbur, N.; Bozdağ, G.; Erkekoğlu, P.; Gümüş, E.; Kocer-Gumusel, B.; et al. Bisphenol A and phthalate levels in adolescents with polycystic ovary syndrome. Gynecol. Endocrinol. 2019, 35, 1084–1087. [Google Scholar] [CrossRef]
- Zhou, W.; Liu, J.; Liao, L.; Han, S.; Liu, J. Effect of bisphenol A on steroid hormone production in rat ovarian theca-interstitial and granulosa cells. Mol. Cell. Endocrinol. 2008, 283, 12–18. [Google Scholar] [CrossRef]
- Reilly, M.P.; Weeks, C.D.; Topper, V.Y.; Thompson, L.M.; Crews, D.; Gore, A.C. The effects of prenatal PCBs on adult social behavior in rats. Horm. Behav. 2015, 73, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.B.; Cho, E.; Kang, Y.S. Changes in 5-hydroxytryptamine and cortisol plasma levels in menopausal women after inhalation of clary sage oil. Phytother. Res. 2014, 28, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.M.; Couse, J.F.; Korach, K.S. The multifaceted mechanisms of estradiol and estrogen receptor signaling. J. Biol. Chem. 2001, 276, 36869–36872. [Google Scholar] [CrossRef] [PubMed]
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schutz, G.; Umesono, K.; Blumberg, B.; Kastner, P.; Mark, M.; Chambon, P.; et al. The nuclear receptor superfamily: The second decade. Cell 1995, 83, 835–839. [Google Scholar] [CrossRef]
- Balaguer, P.; Delfosse, V.; Grimaldi, M.; Bourguet, W. Structural and functional evidences for the interactions between nuclear hormone receptors and endocrine disruptors at low doses. C. R. Biol. 2017, 340, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, Y.; Liu, W.; Schlenk, D.; Liu, J. Glucocorticoid and mineralocorticoid receptors and corticosteroid homeostasis are potential targets for endocrine-disrupting chemicals. Env. Int. 2019, 133, 105133. [Google Scholar] [CrossRef]
- McDonnell, D.P.; Clemm, D.L.; Hermann, T.; Goldman, M.E.; Pike, J.W. Analysis of estrogen receptor function in vitro reveals three distinct classes of antiestrogens. Mol. Endocrinol. 1995, 9, 659–669. [Google Scholar] [CrossRef][Green Version]
- Paige, L.A.; Christensen, D.J.; Gron, H.; Norris, J.D.; Gottlin, E.B.; Padilla, K.M.; Chang, C.Y.; Ballas, L.M.; Hamilton, P.T.; McDonnell, D.P.; et al. Estrogen receptor (ER) modulators each induce distinct conformational changes in ER alpha and ER beta. Proc. Natl. Acad. Sci. USA 1999, 96, 3999–4004. [Google Scholar] [CrossRef]
- Katzenellenbogen, J.A.; O’Malley, B.W.; Katzenellenbogen, B.S. Tripartite steroid hormone receptor pharmacology: Interaction with multiple effector sites as a basis for the cell- and promoter-specific action of these hormones. Mol. Endocrinol. 1996, 10, 119–131. [Google Scholar] [CrossRef]
- Shibata, H.; Spencer, T.E.; Onate, S.A.; Jenster, G.; Tsai, S.Y.; Tsai, M.J.; O’Malley, B.W. Role of co-activators and co-repressors in the mechanism of steroid/thyroid receptor action. Recent Prog. Horm. Res. 1997, 52, 141–164, discussion 164–165. [Google Scholar] [PubMed]
- Delfosse, V.; Grimaldi, M.; Pons, J.-L.; Boulahtouf, A.; le Maire, A.; Cavailles, V.; Labesse, G.; Bourguet, W.; Balaguer, P. Structural and mechanistic insights into bisphenols action provide guidelines for risk assessment and discovery of bisphenol A substitutes. Proc. Natl. Acad. Sci. USA 2012, 109, 14930. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.M.; McDonnell, D.P.; Korach, K.S. Allosteric regulation of estrogen receptor structure, function, and coactivator recruitment by different estrogen response elements. Mol. Endocrinol. 2002, 16, 469–486. [Google Scholar] [CrossRef]
- Bai, J.; He, Z.; Li, Y.; Jiang, X.; Yu, H.; Tan, Q. Mono-2-ethylhexyl phthalate induces the expression of genes involved in fatty acid synthesis in HepG2 cells. Env. Toxicol. Pharm. 2019, 69, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, R.L.; Kentsis, A.; Deplewski, D.; Ciletti, N. Rat preputial sebocyte differentiation involves peroxisome proliferator-activated receptors. J. Investig. Derm. 1999, 112, 226–232. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Barton, M. Estrogen biology: New insights into GPER function and clinical opportunities. Mol. Cell. Endocrinol. 2014, 389, 71–83. [Google Scholar] [CrossRef]
- Filardo, E.J.; Thomas, P. Minireview: G Protein-Coupled Estrogen Receptor-1, GPER-1: Its Mechanism of Action and Role in Female Reproductive Cancer, Renal and Vascular Physiology. Endocrinology 2012, 153, 2953–2962. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Hathaway, H.J. What have we learned about GPER function in physiology and disease from knockout mice? J. Steroid Biochem. Mol. Biol. 2015, 153, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Nadal, A.; Ropero, A.B.; Laribi, O.; Maillet, M.; Fuentes, E.; Soria, B. Nongenomic actions of estrogens and xenoestrogens by binding at a plasma membrane receptor unrelated to estrogen receptor α and estrogen receptor β. Proc. Natl. Acad. Sci. USA 2000, 97, 11603–11608. [Google Scholar] [CrossRef] [PubMed]
- Henley, D.V.; Lipson, N.; Korach, K.S.; Bloch, C.A. Prepubertal gynecomastia linked to lavender and tea tree oils. N. Engl. J. Med. 2007, 356, 479–485. [Google Scholar] [CrossRef]
- Ramsey, J.T.; Li, Y.; Arao, Y.; Naidu, A.; Coons, L.A.; Diaz, A.; Korach, K.S. Lavender Products Associated With Premature Thelarche and Prepubertal Gynecomastia: Case Reports and Endocrine-Disrupting Chemical Activities. J. Clin. Endocrinol. Metab. 2019, 104, 5393–5405. [Google Scholar] [CrossRef]
- Banaszewska, B.; Wrotyńska-Barczyńska, J.; Spaczynski, R.Z.; Pawelczyk, L.; Duleba, A.J. Effects of Resveratrol on Polycystic Ovary Syndrome: A Double-blind, Randomized, Placebo-controlled Trial. J. Clin. Endocrinol. Metab. 2016, 101, 4322–4328. [Google Scholar] [CrossRef] [PubMed]
- Karantanos, T.; Karanika, S.; Wang, J.; Yang, G.; Dobashi, M.; Park, S.; Ren, C.; Li, L.; Basourakos, S.P.; Hoang, A.; et al. Caveolin-1 regulates hormone resistance through lipid synthesis, creating novel therapeutic opportunities for castration-resistant prostate cancer. Oncotarget 2016, 7, 46321–46334. [Google Scholar] [CrossRef]
- Lai, J.-J.; Chang, P.; Lai, K.-P.; Chen, L.; Chang, C. The role of androgen and androgen receptor in skin-related disorders. Arch. Derm. Res. 2012, 304, 499–510. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, H.; Zou, J.; Feng, X.; Feng, D. Bisphenol A induces cholesterol biosynthesis in HepG2 cells via SREBP-2/HMGCR signaling pathway. J. Toxicol. Sci. 2019, 44, 481–491. [Google Scholar] [CrossRef]
- Pihlajamaa, P.; Zhang, F.-P.; Saarinen, L.; Mikkonen, L.; Hautaniemi, S.; Jänne, O.A. The Phytoestrogen Genistein Is a Tissue-Specific Androgen Receptor Modulator. Endocrinology 2011, 152, 4395–4405. [Google Scholar] [CrossRef] [PubMed]
- Ji, G.; Zhang, Y.; Yang, Q.; Cheng, S.; Hao, J.; Zhao, X.; Jiang, Z. Genistein suppresses LPS-induced inflammatory response through inhibiting NF-κB following AMP kinase activation in RAW 264.7 macrophages. PLoS ONE 2012, 7, e53101. [Google Scholar] [CrossRef] [PubMed]
- Law, M.P.; Chuh, A.A.; Molinari, N.; Lee, A. An investigation of the association between diet and occurrence of acne: A rational approach from a traditional Chinese medicine perspective. Clin. Exp. Derm. 2010, 35, 31–35. [Google Scholar] [CrossRef]
- Todorovic-Rakovic, N.; Ivanovic, V.; Demajo, M.; Neskovic, B.; Neskovic-Konstantinovic, Z.; Kanjer, K.; Markićević, M.; Nikolic-Vukosavljevic, D. TGF-beta1 in breast cancer-estrogen regulation. Arch. Oncol. 2002, 10, 164–165. [Google Scholar] [CrossRef]
- McNairn, A.J.; Doucet, Y.; Demaude, J.; Brusadelli, M.; Gordon, C.B.; Uribe-Rivera, A.; Lambert, P.F.; Bouez, C.; Breton, L.; Guasch, G. TGFβ signaling regulates lipogenesis in human sebaceous glands cells. BMC Derm. 2013, 13, 2. [Google Scholar] [CrossRef]
- Polito, F.; Marini, H.; Bitto, A.; Irrera, N.; Vaccaro, M.; Adamo, E.B.; Micali, A.; Squadrito, F.; Minutoli, L.; Altavilla, D. Genistein aglycone, a soy-derived isoflavone, improves skin changes induced by ovariectomy in rats. Br. J. Pharm. 2012, 165, 994–1005. [Google Scholar] [CrossRef]
- Sargis, R.M.; Johnson, D.N.; Choudhury, R.A.; Brady, M.J. Environmental endocrine disruptors promote adipogenesis in the 3T3-L1 cell line through glucocorticoid receptor activation. Obesity 2010, 18, 1283–1288. [Google Scholar] [CrossRef]
- Trivedi, N.R.; Cong, Z.; Nelson, A.M.; Albert, A.J.; Rosamilia, L.L.; Sivarajah, S.; Gilliland, K.L.; Liu, W.; Mauger, D.T.; Gabbay, R.A.; et al. Peroxisome proliferator-activated receptors increase human sebum production. J. Investig. Derm. 2006, 126, 2002–2009. [Google Scholar] [CrossRef]
- Kim, S.Y.; Hyun, M.Y.; Go, K.C.; Zouboulis, C.C.; Kim, B.J. Resveratrol exerts growth inhibitory effects on human SZ95 sebocytes through the inactivation of the PI3-K/Akt pathway. Int. J. Mol. Med. 2015, 35, 1042–1050. [Google Scholar] [CrossRef]
- Hall, J.M.; Powell, H.A.; Rajic, L.; Korach, K.S. The Role of Dietary Phytoestrogens and the Nuclear Receptor PPARγ in Adipogenesis: An in Vitro Study. Environ. Health Perspect. 2019, 127, 37007. [Google Scholar] [CrossRef]
- Oge, L.K.; Broussard, A.; Marshall, M.D. Acne Vulgaris: Diagnosis and Treatment. Am. Fam. Physician 2019, 100, 475–484. [Google Scholar]
- Titus, S.; Hodge, J. Diagnosis and treatment of acne. Am. Fam Physician 2012, 86, 734–740. [Google Scholar]
- Hauk, L. Acne Vulgaris: Treatment Guidelines from the AAD. Am. Fam. Physician 2017, 95, 740–741. [Google Scholar] [PubMed]
- Zaenglein, A.L.; Pathy, A.L.; Schlosser, B.J.; Alikhan, A.; Baldwin, H.E.; Berson, D.S.; Bowe, W.P.; Graber, E.M.; Harper, J.C.; Kang, S.; et al. Guidelines of care for the management of acne vulgaris. J. Am. Acad. Derm. 2016, 74, 945–973.e33. [Google Scholar] [CrossRef]
- Kassir, M.; Karagaiah, P.; Sonthalia, S.; Katsambas, A.; Galadari, H.; Gupta, M.; Lotti, T.; Wollina, U.; Abdelmaksoud, A.; Grabbe, S.; et al. Selective RAR agonists for acne vulgaris: A narrative review. J. Cosmet. Derm. 2020. [Google Scholar] [CrossRef] [PubMed]
- Leyden, J.; Stein-Gold, L.; Weiss, J. Why Topical Retinoids Are Mainstay of Therapy for Acne. Derm. Ther. 2017, 7, 293–304. [Google Scholar] [CrossRef]
- Melnik, B. Dietary intervention in acne: Attenuation of increased mTORC1 signaling promoted by Western diet. Derm. Endocrinol. 2012, 4, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Linking diet to acne metabolomics, inflammation, and comedogenesis: An update. Clin. Cosmet. Investig. Derm. 2015, 8, 371–388. [Google Scholar] [CrossRef]
- Clark, A.K.; Haas, K.N.; Sivamani, R.K. Edible Plants and Their Influence on the Gut Microbiome and Acne. Int. J. Mol. Sci. 2017, 18, 1070. [Google Scholar] [CrossRef]
- Tan, A.U.; Schlosser, B.J.; Paller, A.S. A review of diagnosis and treatment of acne in adult female patients. Int. J. Womens Derm. 2017, 4, 56–71. [Google Scholar] [CrossRef]
- Ghazipura, M.; McGowan, R.; Arslan, A.; Hossain, T. Exposure to benzophenone-3 and reproductive toxicity: A systematic review of human and animal studies. Reprod. Toxicol. 2017, 73, 175–183. [Google Scholar] [CrossRef] [PubMed]
| Class | Sources | Examples | Comments |
|---|---|---|---|
| Phytoestrogens | Breads, cereals, nuts, soy, legumes, fruits, vegetables |
|
|
| Industrial Chemicals | Pesticides, flame retardants, combustion products |
|
|
| Household and Consumer Items | Food and beverage packaging materials, contaminated foods, well-water, toys, cosmetics, sunscreens, other topical formulations |
|
|
| Medical Devices | disposable gloves, plastic devices, intravenous tubing |
|
|
| Pharmaceuticals | Leakage into drinking water and soil |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, A.; Douglas, S.C.; Hall, J.M. Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: A Connecting Hypothesis. Cells 2021, 10, 1439. https://doi.org/10.3390/cells10061439
Rao A, Douglas SC, Hall JM. Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: A Connecting Hypothesis. Cells. 2021; 10(6):1439. https://doi.org/10.3390/cells10061439
Chicago/Turabian StyleRao, Akshatha, Sotonye C. Douglas, and Julianne M. Hall. 2021. "Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: A Connecting Hypothesis" Cells 10, no. 6: 1439. https://doi.org/10.3390/cells10061439
APA StyleRao, A., Douglas, S. C., & Hall, J. M. (2021). Endocrine Disrupting Chemicals, Hormone Receptors, and Acne Vulgaris: A Connecting Hypothesis. Cells, 10(6), 1439. https://doi.org/10.3390/cells10061439






