The Multiple Functions of Rho GTPases in Fission Yeasts
Abstract
1. Introduction
2. Rho GTPase Regulation
3. Rho GTPases and Their Regulators in Schizosaccharomyces pombe
3.1. Rho1
3.2. Rho2
3.3. Rho3
3.4. Rho4
3.5. Rho5
| GEF | GTPase | Localization | Regulated/Involved Processes |
|---|---|---|---|
| Scd1 | Rho4 Cdc42 | Nucleus and mitotic spindle [108] and active growing sites: on the membrane at cell poles (interphase), division area (cytokinesis) [57,72,80,109,110] | Morphology and cell polarity [57,65]: oscillates between the two cell poles [67] and restricts Gef1 localization to sites of polarization to prevent ectopic Cdc42 activation (cytokinesis) and to maintain cell shape (interphase) [74] Involved in endocytic trafficking with Nrf1 [111] Mitosis: Involved in spindle formation with Moe1 [108,112] Cytokinesis: localizes later than Gef1 to the ingressing furrow and promotes septum formation [86] Sexual differentiation: mating [57,93] |
| Gef1 | Cdc42 | Active growing sites: cell poles (cytosol, active during interphase) and division area (cytokinesis) [75,80,113] | Cell polarity: enables NETO [59,67] promoting Scd1 recruitment at the new end to allow the transition from monopolar to bipolar growth [74] Cytokinesis: localizes before Scd1 to the actomyosin ring and promotes timely constriction [86], interacts with Hob3, which promotes cytokinesis [80] and promotes Scd1 localization to the division site through recruitment of Scd2 [74] |
| Gef2 | Rho1 Rho4 | Contractile ring Precursor cortical nodes [102,114] | Cytokinesis: involved in division-site and contractile-ring positioning by interacting with Mid1 [101] and contributes to the positioning of the division-site and contractile-ring stability together with Nod1 [102] |
| Gef3 | Rho3 Rho4 | Septin ring [45] | Cytokinesis: possible scaffold for septin-mediated Rho3-directed polarized secretion [45] and interacts with the septin complex and Mid2 and activates Rho4 [107] |
| Rgf1 | Rho1 Rho2? | Active growing sites: cell poles and division area [19,27] Fully formed contractile rings [31] Cell nucleus during stalled replication by HU [115] | Cell polarity: regulates Rho1-mediated cell wall deposition during polarized growth [19] Cell wall integrity: activates CIP, via Rho1 and Pck2 [24] Cytokinesis: promotes Rho1 activation during septum formation [19,27], and participates in a cytokinesis checkpoint during cell wall damage, via Rho1 and Pck2-Pmk1 [31] Other functions: Its nuclear accumulation promotes tolerance and survival during replication stress [115], and is required for double-strand break repair via Rho1 [116] |
| Rgf2 | Rho1 | Periphery of the spore after meiosis I and II [117] Cell tips and septum during vegetative growth (upon mild overexpression) [26,27,117] | Cell polarity: secondary function (redundant to Rgf1): regulates Rho1-mediated cell wall deposition during polarized growth at least through Bgs2, but not exclusively [117] Cytokinesis: Rho1 activation during septum formation [25,26,27] Sexual differentiation: main function during assembly of the spore cell wall by activation of GS subunit Bgs2 [117] |
| Rgf3 | Rho1 | Ring/membrane interphase [27,114,118], depends on Art1 [119] | Cell wall integrity: essential for maintaining cell integrity during cell separation [25,26] Cytokinesis: activates Rho1 specifically during cytokinesis [25,26,27]. Regulated by transcription factor Ace2, which promotes its maximal expression during septation [25,26,120], and arrestin Art1 [119] |
| GAP | GTPase | Localization | Regulated/Involved Processes |
|---|---|---|---|
| Rga1 | Rho1 | Cell poles (interphase) and the division site (cytokinesis) [103] | Negative regulator of Rho1 and is involved in actin-patch localization, cell morphogenesis, septation, and cell wall synthesis [103] |
| Rga2 | Rho2 Rho1 (+) | Cell poles (interphase) and the division site at the ring/membrane interphase (cytokinesis). Localization depends on polarity markers and actin polymerization [36,114] | Cell polarity and morphogenesis: Positive effect, direct or indirect, in Cdc42 activation playing an antagonistic role to Rga4 to maintain cell dimensions [36] Cell wall integrity: Rga2 negatively regulates Rho2-Pck2 interaction and Pck2 stability. Rga2 acts as a negative regulator of the Rho2-Pck2 interaction with the CIP. Lack of Rga2 suppress the lysis of mok1-664 at 32 °C [36] |
| Rga3 | Rho1 (+) Cdc42 | Cell poles and the division site with Cdc42-GTP during mitotic growth [73]. During sexual differentiation, Cdc42 patches and sites of polarity [73] | Cell polarity: paralogue of Rga4 and synergizes with Rga4 and Rga6 to restrict Cdc42-GTP zone sizes during mitotic growth [73] Sexual differentiation: limits the lifetime of unstable Cdc42-GFP patches important for the wandering motion that favors mating and confers a competitive advantage during sexual reproduction [73] |
| Rga4 | Rho2 Cdc42 | To the plasma membrane at the cell sides (interphase) and the division site (at the end of cytokinesis). Forms clusters at cell sides and nongrowing cell pole [121,122] | Cell morphogenesis and polarity: restricts Cdc42 activation at the cell sides controlling cell diameter and symmetry breaking [121,122]. Pom1 regulates its localization and phosphorylation [105,122]. Its exclusion from cell poles to allow bipolar Cdc42 activation also depends on Dis2 [123] Cell wall integrity: Rho2 GAP negatively regulates the activity of the CIP, which is not involved in the Rga4-dependent control of cell shape [105]. Not involved in Pom1 negative regulation of the CIP. Rga4 positively regulates cell wall integrity and cell separation independently of the Pmk1 pathway, acting as a Cdc42 GAP [105] |
| Rga5 | Rho1 | Cell poles (interphase) and the division site (cytokinesis) upon mild overexpression [124] | Cell morphology: participates in the regulation of cell morphology and cell wall biosynthesis at high temperature [124] Cell wall integrity: Specific Rho1 GAP that negatively modulates the Rho1-Pck1, and to a lesser extent Pck2 interaction, decreasing their stability. Negative regulation of 1,3-β-GS activity [124] Cytokinesis: cell separation defect and/or delay [124] Sporulation: rga1∆ rga5∆ spores are unable to germinate [124] |
| Rga6 | Rho2 Cdc42 | Plasma membrane at the cell sides, forming clusters different from those made by Rga4 and growing cell poles [125] | Cell morphology and polarity: Cdc42 GAP and collaborates with Rga4, although its role could be higher than Rga4 in the negative regulation of Cdc42 at the growing cell pole [125] Cell wall integrity: Rho2 GAP [36,105] |
| Rga7 | Rho2 | Cell poles (interphase) and the division site (cytokinesis) [126] | Cell wall integrity: Rho2 GAP involved in the negative regulation of the CIP [105] Cytokinesis: The GAP activity is dispensable for Rga7 function in cytokinesis [126]. Cooperates with Cdc15 and Imp2 in actomyosin ring stability and proper disassembly, and successful septum formation and separation to ensure cell integrity [126]. Participates with Rng10 in Bgs4 trafficking from the Golgi to plasma membrane adjacent to the contractile ring [127] |
| Rga8 | Rho1 | Cell poles (interphase) and the division site (cytokinesis) [128], with a monopolar pattern when Pak1 activity is abolished [128] | Polarity: Rho1 GAP and is a downstream target of Pak1 [128], and participates in the crosstalk between Rho1 and Cdc42 [5,128] Overexpression causes morphological defects, a cytokinesis delay and cell lysis (like rho1+ overexpression) [128] |
| Rga9 | Rho4 (+) Cdc42 (+) | Rho4 GAP and may function cooperatively with Rdi1 to negatively control the cellular localization of Rho4 [47] and Cdc42 (GAP) [47] |
3.6. Cdc42
4. On the Role of Rho GTPases during Polarized Growth
4.1. Schizosaccharomyces pombe
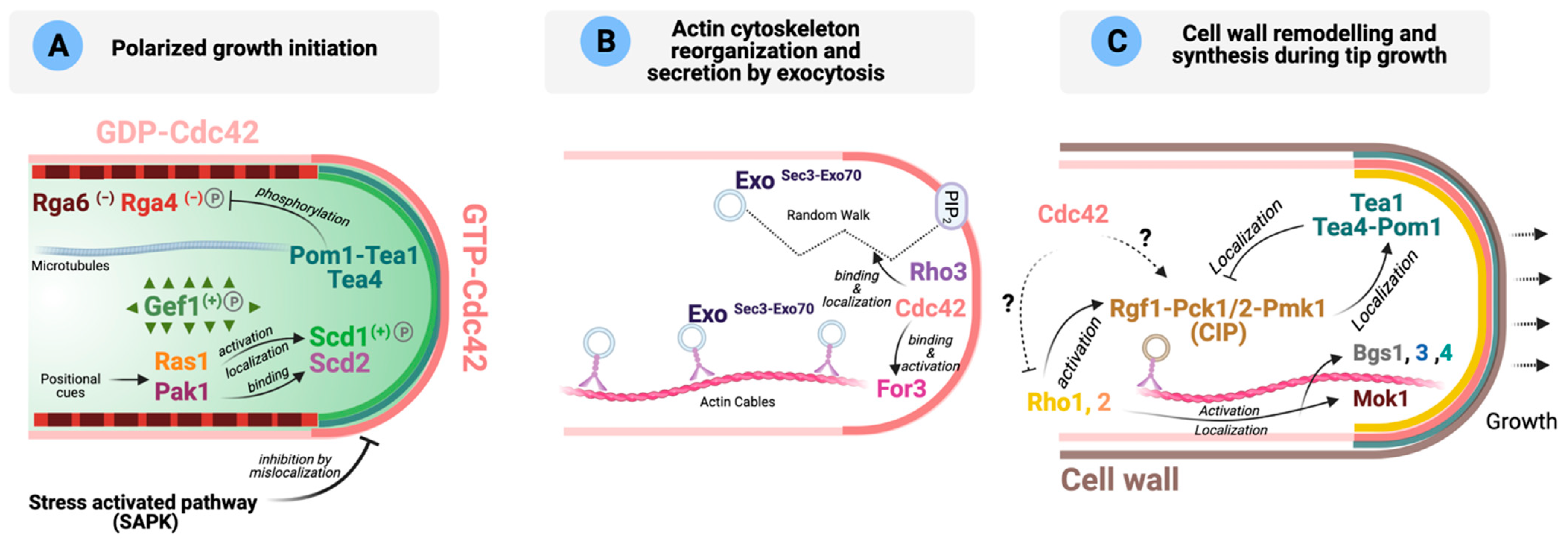
4.2. Schizosaccharomyces japonicus
5. Regulation of Cell Integrity by Rho GTPases
 ,
, ); PS: primary septum; SS: secondary septum. (B) Schematic representation of S. pombe CIP activation. Wsc1 and Mtl2 activate Rho1 through the GEF Rgf1. Rho1 activates the β-GS and stabilizes Pck1 and Pck2. After activation by the phospholipid-dependent kinase Ksg1, both kinases activate the GS. Rho2 regulates Mok1 via Pck2. Rho2-Pck2 are the main activators of the CIP. Please see main text for more details.
); PS: primary septum; SS: secondary septum. (B) Schematic representation of S. pombe CIP activation. Wsc1 and Mtl2 activate Rho1 through the GEF Rgf1. Rho1 activates the β-GS and stabilizes Pck1 and Pck2. After activation by the phospholipid-dependent kinase Ksg1, both kinases activate the GS. Rho2 regulates Mok1 via Pck2. Rho2-Pck2 are the main activators of the CIP. Please see main text for more details.
 ,
, ); PS: primary septum; SS: secondary septum. (B) Schematic representation of S. pombe CIP activation. Wsc1 and Mtl2 activate Rho1 through the GEF Rgf1. Rho1 activates the β-GS and stabilizes Pck1 and Pck2. After activation by the phospholipid-dependent kinase Ksg1, both kinases activate the GS. Rho2 regulates Mok1 via Pck2. Rho2-Pck2 are the main activators of the CIP. Please see main text for more details.
); PS: primary septum; SS: secondary septum. (B) Schematic representation of S. pombe CIP activation. Wsc1 and Mtl2 activate Rho1 through the GEF Rgf1. Rho1 activates the β-GS and stabilizes Pck1 and Pck2. After activation by the phospholipid-dependent kinase Ksg1, both kinases activate the GS. Rho2 regulates Mok1 via Pck2. Rho2-Pck2 are the main activators of the CIP. Please see main text for more details.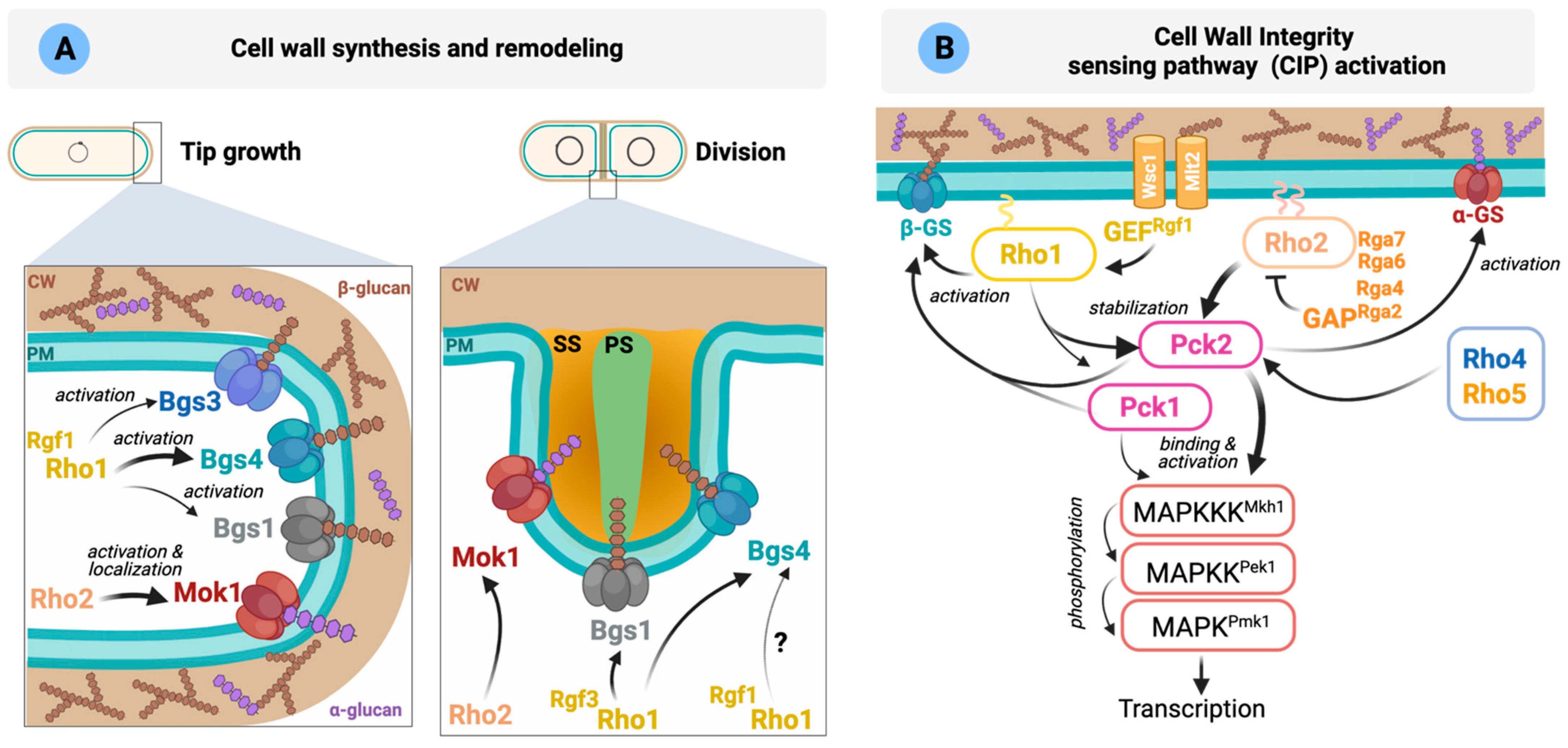
6. Rho Signaling in Cytokinesis
6.1. Schizosaccharomyces pombe
6.2. Schizosaccharomyces japonicus
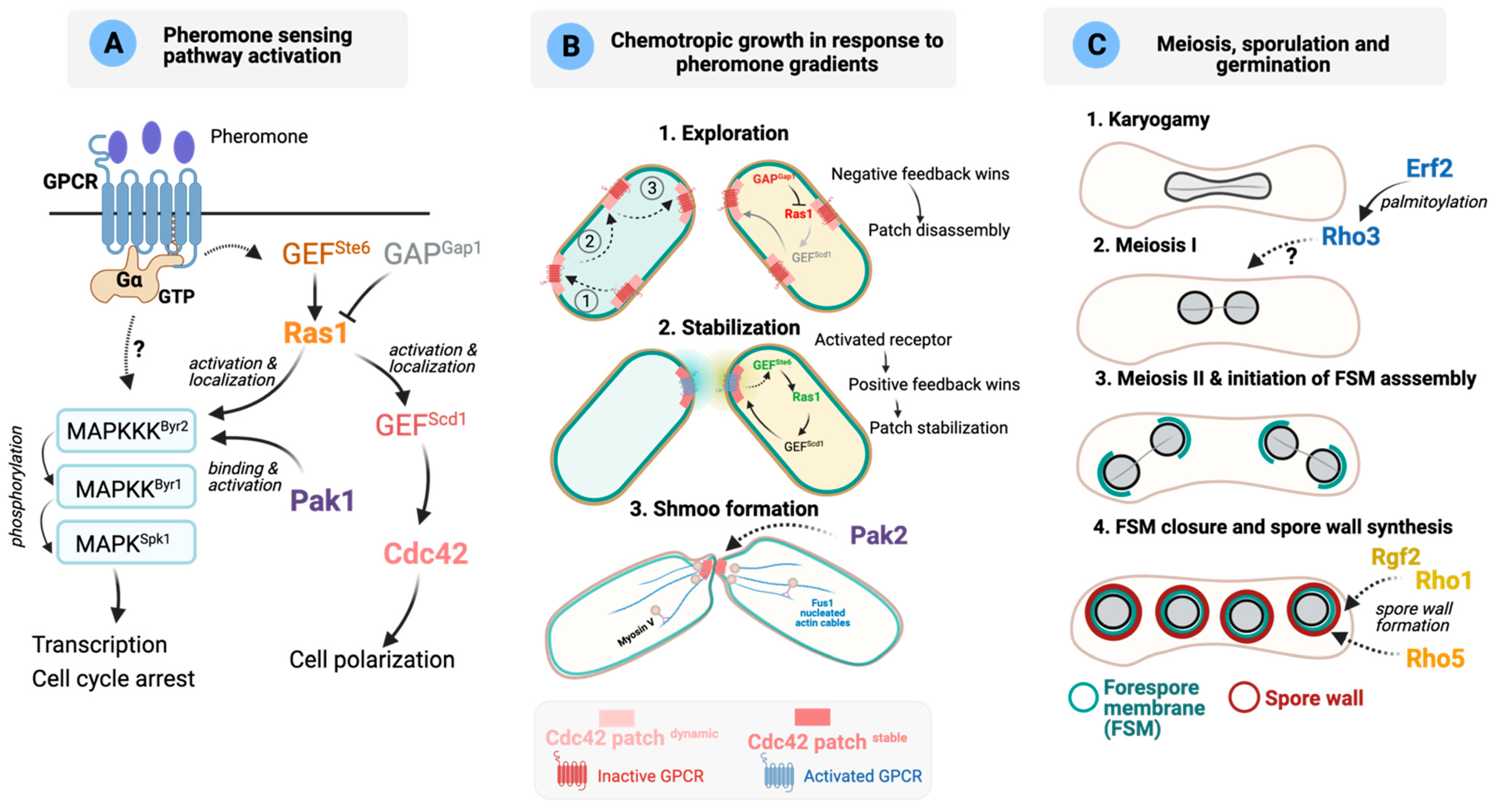
7. Functions of Rho GTPases during Sexual Differentiation
8. Future Avenues
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hodge, R.G.; Ridley, A.J. Regulating Rho GTPases and their regulators. Nat. Rev. Mol. Cell. Biol. 2016, 17, 496–510. [Google Scholar] [CrossRef]
- Phuyal, S.; Farhan, H. Multifaceted Rho GTPase signaling at the endomembranes. Front. Cell. Cev. Biol. 2019, 7, 127. [Google Scholar] [CrossRef] [PubMed]
- Arellano, M.; Duran, A.; Perez, P. Rho1 GTPase activates the (1-3)β-D-glucan synthase and is involved in Schizosaccharomyces pombe morphogenesis. EMBO J. 1996, 15, 4584–4591. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.J.; Johnson, D.I. Cdc42p GTPase is involved in controlling polarized cell growth in Schizosaccharomyces pombe. Mol. Cell. Biol. 1994, 14, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Pérez, P.; Cortés, J.C.G.; Cansado, J.; Ribas, J.C. Fission yeast cell wall biosynthesis and cell integrity signalling. Cell Surf. 2018, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hercyk, B.; Das, M. Rho family GTPases in fission yeast cytokinesis. Commun. Integr. Biol. 2019, 12, 171–180. [Google Scholar] [CrossRef]
- Martin, S.G. Molecular mechanisms of chemotropism and cell fusion in unicellular fungi. J. Cell. Sci. 2019, 132. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.J.S. Schizosaccharomyces japonicus yeast poised to become a favorite experimental organism for eukaryotic research. G3 (Bethesda) 2013, 3, 1869–1873. [Google Scholar] [CrossRef]
- Niki, H. Schizosaccharomyces japonicus: The fission yeast is a fusion of yeast and hyphae. Yeast 2014, 31, 83–90. [Google Scholar] [CrossRef]
- Wickerham, L.J.; Duprat, E. A remarkable fission yeast, Schizosaccharomyces versatilis nov. sp. J. Bacteriol. 1945, 50, 597–607. [Google Scholar] [CrossRef]
- Kinnaer, C.; Dudin, O.; Martin, S.G. Yeast-to-hypha transition of Schizosaccharomyces japonicus in response to environmental stimuli. Mol. Biol. Cell 2019, 30, 975–991. [Google Scholar] [CrossRef]
- Nozaki, S.; Furuya, K.; Niki, H. The Ras1-Cdc42 pathway is involved in hyphal development of Schizosaccharomyces japonicus. FEMS Yeast Res. 2018, 18. [Google Scholar] [CrossRef]
- Cherfils, J.; Zeghouf, M. Regulation of small GTPases by GEFs, GAPs, and GDIs. Physiol. Rev. 2013, 93, 269–309. [Google Scholar] [CrossRef]
- Bos, J.L.; Rehmann, H.; Wittinghofer, A. GEFs and GAPs: Critical elements in the control of small G proteins. Cell 2007, 129, 865–877. [Google Scholar] [CrossRef]
- Liu, M.; Bi, F.; Zhou, X.; Zheng, Y. Rho GTPase regulation by miRNAs and covalent modifications. Trends Cell. Biol. 2012, 22, 365–373. [Google Scholar] [CrossRef]
- Croft, D.R.; Olson, M.F. Transcriptional regulation of Rho GTPase signaling. Transcription 2011, 2, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Arellano, M.; Duran, A.; Perez, P. Localisation of the Schizosaccharomyces pombe Rho1p GTPase and its involvement in the organisation of the actin cytoskeleton. J. Cell Sci. 1997, 110, 2547–2555. [Google Scholar] [CrossRef] [PubMed]
- Arellano, M.; Valdivieso, M.H.; Calonge, T.M.; Coll, P.M.; Duran, A.; Perez, P. Schizosaccharomyces pombe protein kinase C homologues, Pck1p and Pck2p, are targets of Rho1p and Rho2p and differentially regulate cell integrity. J. Cell Sci. 1999, 112, 3569–3578. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Tajadura, V.; Garcia, I.; Sanchez, Y. Rgf1p is a specific Rho1-GEF that coordinates cell polarization with cell wall biogenesis in fission yeast. Mol. Biol. Cell 2006, 17, 1620–1631. [Google Scholar] [CrossRef]
- Sayers, L.G.; Katayama, S.; Nakano, K.; Mellor, H.; Mabuchi, I.; Toda, T.; Parker, P.J. Rho-dependence of Schizosaccharomyces pombe Pck2. Genes Cells 2000, 5, 17–27. [Google Scholar] [CrossRef]
- Sanchez-Mir, L.; Soto, T.; Franco, A.; Madrid, M.; Viana, R.A.; Vicente, J.; Gacto, M.; Perez, P.; Cansado, J. Rho1 GTPase and PKC ortholog Pck1 are upstream activators of the cell integrity MAPK pathway in fission yeast. PLoS ONE 2014, 9, e88020. [Google Scholar] [CrossRef]
- Madrid, M.; Jiménez, R.; Sánchez-Mir, L.; Soto, T.; Franco, A.; Vicente-Soler, J.; Gacto, M.; Pérez, P.; Cansado, J. Multiple layers of regulation influence cell integrity control by the PKC ortholog Pck2 in fission yeast. J. Cell Sci. 2015, 128, 266–280. [Google Scholar] [CrossRef]
- Madrid, M.; Vázquez-Marín, B.; Soto, T.; Franco, A.; Gómez-Gil, E.; Vicente-Soler, J.; Gacto, M.; Pérez, P.; Cansado, J. Differential functional regulation of protein kinase C (PKC) orthologs in fission yeast. J. Biol. Chem. 2017, 292, 11374–11387. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Tajadura, V.; Sanchez, Y. The Rho1p exchange factor Rgf1p signals upstream from the Pmk1 mitogen-activated protein kinase pathway in fission yeast. Mol. Biol. Cell 2009, 20, 721–731. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tajadura, V.; Garcia, B.; Garcia, I.; Garcia, P.; Sanchez, Y. Schizosaccharomyces pombe Rgf3p is a specific Rho1 GEF that regulates cell wall β-glucan biosynthesis through the GTPase Rho1p. J. Cell Sci. 2004, 117, 6163–6174. [Google Scholar] [CrossRef] [PubMed]
- Morrell-Falvey, J.L.; Ren, L.; Feoktistova, A.; Haese, G.D.; Gould, K.L. Cell wall remodeling at the fission yeast cell division site requires the Rho-GEF Rgf3p. J. Cell Sci. 2005, 118, 5563–5573. [Google Scholar] [CrossRef]
- Mutoh, T.; Nakano, K.; Mabuchi, I. Rho1-GEFs Rgf1 and Rgf2 are involved in formation of cell wall and septum, while Rgf3 is involved in cytokinesis in fission yeast. Genes Cells 2005, 10, 1189–1202. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.-W.; Zhou, M.; Bimbo, A.; Balasubramanian, M.K.; McCollum, D. A role for the septation initiation network in septum assembly revealed by genetic analysis of sid2-250 suppressors. Genetics 2006, 172, 2101–2112. [Google Scholar] [CrossRef]
- Pinar, M.; Coll, P.M.; Rincon, S.A.; Perez, P. Schizosaccharomyces pombe Pxl1 is a paxillin homologue that modulates Rho1 activity and participates in cytokinesis. Mol. Biol. Cell 2008, 19, 1727–1738. [Google Scholar] [CrossRef]
- Alcaide-Gavilan, M.; Lahoz, A.; Daga, R.R.; Jimenez, J. Feedback regulation of SIN by Etd1 and Rho1 in fission yeast. Genetics 2014, 196, 455–470. [Google Scholar] [CrossRef]
- Edreira, T.; Celador, R.; Manjón, E.; Sánchez, Y. A novel checkpoint pathway controls actomyosin ring constriction trigger in fission yeast. eLife 2020, 9, e59333. [Google Scholar] [CrossRef]
- Hirata, D.; Nakano, K.; Fukui, M.; Takenaka, H.; Miyakawa, T.; Mabuchi, I. Genes that cause aberrant cell morphology by overexpression in fission yeast: A role of a small GTP-binding protein Rho2 in cell morphogenesis. J. Cell Sci. 1998, 111, 149–159. [Google Scholar] [CrossRef]
- Sanchez-Mir, L.; Franco, A.; Martin-Garcia, R.; Madrid, M.; Vicente-Soler, J.; Soto, T.; Gacto, M.; Perez, P.; Cansado, J. Rho2 palmitoylation is required for plasma membrane localization and proper signaling to the fission yeast cell integrity mitogen- activated protein kinase pathway. Mol. Cell. Biol. 2014, 34, 2745–2759. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Kuno, T.; Kita, A.; Asayama, Y.; Sugiura, R. Rho2 is a target of the farnesyltransferase Cpp1 and acts upstream of Pmk1 mitogen-activated protein kinase signaling in fission yeast. Mol. Biol. Cell 2006, 17, 5028–5037. [Google Scholar] [CrossRef]
- Katayama, S.; Hirata, D.; Arellano, M.; Pérez, P.; Toda, T. Fission yeast α-glucan synthase Mok1 requires the actin cytoskeleton to localize the sites of growth and plays an essential role in cell morphogenesis downstream of protein kinase C function. J. Cell Biol. 1999, 144, 1173–1186. [Google Scholar] [CrossRef] [PubMed]
- Villar-Tajadura, M.A.; Coll, P.M.; Madrid, M.; Cansado, J.; Santos, B.; Perez, P. Rga2 is a Rho2 GAP that regulates morphogenesis and cell integrity in S. pombe. Mol. Microbiol. 2008, 70, 867–881. [Google Scholar] [CrossRef]
- Calonge, T.M.; Nakano, K.; Arellano, M.; Arai, R.; Katayama, S.; Toda, T.; Mabuchi, I.; Perez, P. Schizosaccharomyces pombe Rho2p GTPase regulates cell wall α-glucan biosynthesis through the protein kinase Pck2p. Mol. Biol. Cell 2000, 11, 4393–4401. [Google Scholar] [CrossRef] [PubMed]
- Barba, G.; Soto, T.; Madrid, M.; Nunez, A.; Vicente, J.; Gacto, M.; Cansado, J. Activation of the cell integrity pathway is channelled through diverse signalling elements in fission yeast. Cell Signal 2008, 20, 748–757. [Google Scholar] [CrossRef]
- Cortés, J.C.G.; Sato, M.; Muñoz, J.; Moreno, M.B.; Clemente-Ramos, J.A.; Ramos, M.; Okada, H.; Osumi, M.; Durán, A.; Ribas, J.C. Fission yeast Ags1 confers the essential septum strength needed for safe gradual cell abscission. J. Cell Biol. 2012, 198, 637–656. [Google Scholar] [CrossRef]
- Nakano, K.; Imai, J.; Arai, R.; Toh, E.A.; Matsui, Y.; Mabuchi, I. The small GTPase Rho3 and the diaphanous/formin For3 function in polarized cell growth in fission yeast. J. Cell Sci. 2002, 115, 4629–4639. [Google Scholar] [CrossRef]
- Nakano, K.; Toya, M.; Yoneda, A.; Asami, Y.; Yamashita, A.; Kamasawa, N.; Osumi, M.; Yamamoto, M. Pob1 ensures cylindrical cell shape by coupling two distinct Rho signaling events during secretory vesicle targeting. Traffic 2011, 12, 726–739. [Google Scholar] [CrossRef]
- Wang, H.; Tang, X.; Balasubramanian, M.K. Rho3p regulates cell separation by modulating exocyst function in Schizosaccharomyces pombe. Genetics 2003, 164, 1323–1331. [Google Scholar] [CrossRef]
- Kita, A.; Li, C.; Yu, Y.; Umeda, N.; Doi, A.; Yasuda, M.; Ishiwata, S.; Taga, A.; Horiuchi, Y.; Sugiura, R. Role of the Small GTPase Rho3 in Golgi/Endosome trafficking through functional interaction with adaptin in fission yeast. PLoS ONE 2011, 6, e16842. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Li, C.; Kita, A.; Katayama, Y.; Kubouchi, K.; Udo, M.; Imanaka, Y.; Ueda, S.; Masuko, T.; Sugiura, R. Sip1, an AP-1 accessory protein in fission yeast, is required for localization of Rho3 GTPase. PLoS ONE 2013, 8, e68488. [Google Scholar] [CrossRef]
- Munoz, S.; Manjon, E.; Sanchez, Y. The putative exchange factor Gef3p interacts with Rho3p GTPase and the septin ring during cytokinesis in fission yeast. J. Biol. Chem. 2014, 289, 21995–22007. [Google Scholar] [CrossRef] [PubMed]
- Sedwick, C. Palmitoylation: A new regulatory role in meiosis. PLoS Biol. 2013, 11, e1001598. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Mutoh, T.; Arai, R.; Mabuchi, I. The small GTPase Rho4 is involved in controlling cell morphology and septation in fission yeast. Genes Cells 2003, 8, 357–370. [Google Scholar] [CrossRef]
- Santos, B.; Gutierrez, J.; Calonge, T.M.; Perez, P. Novel Rho GTPase involved in cytokinesis and cell wall integrity in the fission yeast Schizosaccharomyces pombe. Eukaryot. Cell 2003, 2, 521–533. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Santos, B.; Martin-Cuadrado, A.B.; Vazquez de Aldana, C.R.; del Rey, F.; Perez, P. Rho4 GTPase is involved in secretion of glucanases during fission yeast cytokinesis. Eukaryot. Cell 2005, 4, 1639–1645. [Google Scholar] [CrossRef]
- Perez, P.; Portales, E.; Santos, B. Rho4 interaction with exocyst and septins regulates cell separation in fission yeast. Microbiology 2015, 161, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Doi, A.; Kita, A.; Kanda, Y.; Uno, T.; Asami, K.; Satoh, R.; Nakano, K.; Sugiura, R. Geranylgeranyltransferase Cwg2-Rho4/Rho5 module is implicated in the Pmk1 MAP kinase-mediated cell wall integrity pathway in fission yeast. Genes Cells 2015, 20, 310–323. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Arai, R.; Mabuchi, I. Small GTPase Rho5 is a functional homologue of Rho1, which controls cell shape and septation in fission yeast. FEBS Lett. 2005, 579, 5181–5186. [Google Scholar] [CrossRef]
- Rincon, S.A.; Santos, B.; Perez, P. Fission yeast Rho5p GTPase is a functional paralogue of Rho1p that plays a role in survival of spores and stationary-phase cells. Eukaryot. Cell 2006, 5, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.I. Cdc42: An essential Rho-type GTPase controlling eukaryotic cell polarity. Microbiol. Mol. Biol. Rev. 1999, 63, 54–105. [Google Scholar] [CrossRef] [PubMed]
- Merla, A.; Johnson, D.I. The Cdc42p GTPase is targeted to the site of cell division in the fission yeast Schizosaccharomyces pombe. Eur J. Cell Biol. 2000, 79, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Bendezu, F.O.; Vincenzetti, V.; Vavylonis, D.; Wyss, R.; Vogel, H.; Martin, S.G. Spontaneous Cdc42 polarization independent of GDI-mediated extraction and actin-based trafficking. PLoS Biol. 2015, 13, e1002097. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.C.; Barr, M.; Wang, Y.; Jung, V.; Xu, H.P.; Wigler, M.H. Cooperative interaction of S. pombe proteins required for mating and morphogenesis. Cell 1994, 79, 131–141. [Google Scholar] [CrossRef]
- Feierbach, B.; Chang, F. Roles of the fission yeast formin For3p in cell polarity, actin cable formation and symmetric cell division. Curr. Biol. 2001, 11, 1656–1665. [Google Scholar] [CrossRef]
- Coll, P.M.; Trillo, Y.; Ametzazurra, A.; Perez, P. Gef1p, a new guanine nucleotide exchange factor for Cdc42p, regulates polarity in Schizosaccharomyces pombe. Mol. Biol. Cell 2003, 14, 313–323. [Google Scholar] [CrossRef]
- Martin, S.G.; Rincon, S.A.; Basu, R.; Perez, P.; Chang, F. Regulation of the formin For3p by Cdc42p and Bud6p. Mol. Biol. Cell 2007, 18, 4155–4167. [Google Scholar] [CrossRef]
- Das, M.; Wiley, D.J.; Chen, X.; Shah, K.; Verde, F. The conserved NDR kinase Orb6 controls polarized cell growth by spatial regulation of the small GTPase Cdc42. Curr. Biol. 2009, 19, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Rincon, S.A.; Ye, Y.; Villar-Tajadura, M.A.; Santos, B.; Martin, S.G.; Perez, P. Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. Mol. Biol. Cell 2009, 20, 4390–4399. [Google Scholar] [CrossRef]
- Perez, P.; Rincón, S.A. Rho GTPases: Regulation of cell polarity and growth in yeasts. Biochem. J. 2010, 426, 243–253. [Google Scholar] [CrossRef]
- Bendezu, F.O.; Martin, S.G. Actin cables and the exocyst form two independent morphogenesis pathways in the fission yeast. Mol. Biol. Cell 2011, 22, 44–53. [Google Scholar] [CrossRef]
- Kelly, F.D.; Nurse, P. Spatial control of Cdc42 activation determines cell width in fission yeast. Mol. Biol. Cell 2011, 22, 3801–3811. [Google Scholar] [CrossRef]
- Kelly, F.D.; Nurse, P. De novo growth zone formation from fission yeast spheroplasts. PLoS ONE 2011, 6, e27977. [Google Scholar] [CrossRef]
- Das, M.; Drake, T.; Wiley, D.J.; Buchwald, P.; Vavylonis, D.; Verde, F. Oscillatory dynamics of Cdc42 GTPase in the control of polarized growth. Science 2012, 337, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.G.; Arkowitz, R.A. Cell polarization in budding and fission yeasts. FEMS Microbiol. Rev. 2014, 38, 228–253. [Google Scholar] [CrossRef] [PubMed]
- Rincon, S.A.; Estravis, M.; Perez, P. Cdc42 regulates polarized growth and cell integrity in fission yeast. Biochem. Soc. Trans. 2014, 42, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Nunez, I.; Rodriguez, M.; Wiley, D.J.; Rodriguez, J.; Sarkeshik, A.; Yates, J.R., 3rd; Buchwald, P.; Verde, F. Phosphorylation-dependent inhibition of Cdc42 GEF Gef1 by 14-3-3 protein Rad24 spatially regulates Cdc42 GTPase activity and oscillatory dynamics during cell morphogenesis. Mol. Biol. Cell 2015, 26, 3520–3534. [Google Scholar] [CrossRef]
- Martin, S.G. Spontaneous cell polarization: Feedback control of Cdc42 GTPase breaks cellular symmetry. Bioessays 2015, 37, 1193–1201. [Google Scholar] [CrossRef]
- Chiou, J.G.; Balasubramanian, M.K.; Lew, D.J. Cell Polarity in Yeast. Annu. Rev. Cell Dev. Biol. 2017, 33, 77–101. [Google Scholar] [CrossRef] [PubMed]
- Gallo Castro, D.; Martin, S.G. Differential GAP requirement for Cdc42-GTP polarization during proliferation and sexual reproduction. J. Cell Biol. 2018, 217, 4215–4229. [Google Scholar] [CrossRef] [PubMed]
- Hercyk, B.S.; Rich-Robinson, J.; Mitoubsi, A.S.; Harrell, M.A.; Das, M.E. A novel interplay between GEFs orchestrates Cdc42 activity during cell polarity and cytokinesis. J. Cell Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hercyk, B.S.; Das, M.E. F-BAR Cdc15 promotes Cdc42 activation during cytokinesis and cell polarization in Schizosaccharomyces pombe. Genetics 2019. [Google Scholar] [CrossRef] [PubMed]
- Lamas, I.; Merlini, L.; Vještica, A.; Vincenzetti, V.; Martin, S.G. Optogenetics reveals Cdc42 local activation by scaffold-mediated positive feedback and Ras GTPase. PLoS Biol. 2020, 18, e3000600. [Google Scholar] [CrossRef]
- Perez, P.; Soto, T.; Gomez-Gil, E.; Cansado, J. Functional interaction between Cdc42 and the stress MAPK signaling pathway during the regulation of fission yeast polarized growth. Int. Microbiol. 2019. [Google Scholar] [CrossRef]
- Merla, A.; Johnson, D.I. The Schizosaccharomyces pombe Cdc42p GTPase signals through Pak2p and the Mkh1p-Pek1p-Spm1p MAP kinase pathway. Curr. Genet. 2001, 39, 205–209. [Google Scholar] [CrossRef]
- Coll, P.M.; Rincon, S.A.; Izquierdo, R.A.; Perez, P. Hob3p, the fission yeast ortholog of human BIN3, localizes Cdc42p to the division site and regulates cytokinesis. EMBO J. 2007, 26, 1865–1877. [Google Scholar] [CrossRef]
- Rincon, S.; Coll, P.M.; Perez, P. Spatial regulation of Cdc42 during cytokinesis. Cell Cycle 2007, 6, 1687–1691. [Google Scholar] [CrossRef]
- Loo, T.H.; Balasubramanian, M. Schizosaccharomyces pombe Pak-related protein, Pak1p/Orb2p, phosphorylates myosin regulatory light chain to inhibit cytokinesis. J. Cell Biol. 2008, 183, 785–793. [Google Scholar] [CrossRef]
- Lo Presti, L.; Chang, F.; Martin, S.G. Myosin Vs organize actin cables in fission yeast. Mol. Biol. Cell 2012, 23, 4579–4591. [Google Scholar] [CrossRef]
- Coffman, V.C.; Sees, J.A.; Kovar, D.R.; Wu, J.Q. The formins Cdc12 and For3 cooperate during contractile ring assembly in cytokinesis. J. Cell Biol. 2013, 203, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Oliferenko, S. Comparative biology of cell division in the fission yeast clade. Curr. Opin. Microbiol. 2015, 28, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Lee, I.J.; Rask, G.; Wu, J.Q. Roles of the TRAPP-II complex and the exocyst in membrane deposition during fission yeast cytokinesis. PLoS Biol. 2016, 14, e1002437. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Hercyk, B.S.; Mattson, N.; Mohammadi, A.; Rich, J.; DeBruyne, E.; Clark, M.M.; Das, M. Unique spatiotemporal activation pattern of Cdc42 by Gef1 and Scd1 promotes different events during cytokinesis. Mol. Biol. Cell 2016, 27, 1235–1245. [Google Scholar] [CrossRef] [PubMed]
- Pollard, L.W.; Bookwalter, C.S.; Tang, Q.; Krementsova, E.B.; Trybus, K.M.; Lowey, S. Fission yeast myosin Myo2 is down-regulated in actin affinity by light chain phosphorylation. Proc. Natl. Acad. Sci. USA 2017, 114, E7236–E7244. [Google Scholar] [CrossRef] [PubMed]
- Hercyk, B.S.; Onwubiko, U.N.; Das, M.E. Coordinating septum formation and the actomyosin ring during cytokinesis in Schizosaccharomyces pombe. Mol. Microbiol. 2019. [Google Scholar] [CrossRef]
- Magliozzi, J.O.; Sears, J.; Brady, M.; Opalko, H.E.; Kettenbach, A.N.; Moseley, J.B. Defining how Pak1 regulates cell polarity and cell division in fission yeast. bioRxiv 2019, 722900. [Google Scholar] [CrossRef]
- Onwubiko, U.N.; Mlynarczyk, P.J.; Wei, B.; Habiyaremye, J.; Clack, A.; Abel, S.M.; Das, M.E. A Cdc42 GEF, Gef1, through endocytosis organizes F-BAR Cdc15 along the actomyosin ring and promotes concentric furrowing. J. Cell Sci. 2019, 132. [Google Scholar] [CrossRef]
- Gómez-Gil, E.; Martín-García, R.; Vicente-Soler, J.; Franco, A.; Vázquez-Marín, B.; Prieto-Ruiz, F.; Soto, T.; Pérez, P.; Madrid, M.; Cansado, J. Stress-activated MAPK signaling controls fission yeast actomyosin ring integrity by modulating formin For3 levels. eLife 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Magliozzi, J.O.; Sears, J.; Cressey, L.; Brady, M.; Opalko, H.E.; Kettenbach, A.N.; Moseley, J.B. Fission yeast Pak1 phosphorylates anillin-like Mid1 for spatial control of cytokinesis. J. Cell Biol. 2020, 219. [Google Scholar] [CrossRef] [PubMed]
- Bendezu, F.O.; Martin, S.G. Cdc42 explores the cell periphery for mate selection in fission yeast. Curr. Biol. 2013, 23, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Merlini, L.; Khalili, B.; Bendezu, F.O.; Hurwitz, D.; Vincenzetti, V.; Vavylonis, D.; Martin, S.G. Local pheromone release from dynamic polarity sites underlies cell-cell pairing during yeast mating. Curr. Biol. 2016, 26, 1117–1125. [Google Scholar] [CrossRef]
- Merlini, L.; Khalili, B.; Dudin, O.; Michon, L.; Vincenzetti, V.; Martin, S.G. Inhibition of Ras activity coordinates cell fusion with cell-cell contact during yeast mating. J. Cell Biol. 2018, 217, 1467–1483. [Google Scholar] [CrossRef]
- Khalili, B.; Merlini, L.; Vincenzetti, V.; Martin, S.G.; Vavylonis, D. Exploration and stabilization of Ras1 mating zone: A mechanism with positive and negative feedbacks. PLoS Comput. Biol. 2018, 14, e1006317. [Google Scholar] [CrossRef]
- Wang, H.; Tang, X.; Liu, J.; Trautmann, S.; Balasundaram, D.; McCollum, D.; Balasubramanian, M.K. The multiprotein exocyst complex is essential for cell separation in Schizosaccharomyces pombe. Mol. Biol. Cell 2002, 13, 515–529. [Google Scholar] [CrossRef]
- Nakano, K.; Arai, R.; Mabuchi, I. The small GTP-binding protein Rho1 is a multifunctional protein that regulates actin localization, cell polarity, and septum formation in the fission yeast Schizosaccharomyces pombe. Genes Cells 1997, 2, 679–694. [Google Scholar] [CrossRef]
- Ozaki, K.; Tanaka, K.; Imamura, H.; Hihara, T.; Kameyama, T.; Nonaka, H.; Hirano, H.; Matsuura, Y.; Takai, Y. Rom1p and Rom2p are GDP/GTP exchange proteins (GEPs) for the Rho1p small GTP binding protein in Saccharomyces cerevisiae. EMBO J. 1996, 15, 2196–2207. [Google Scholar] [CrossRef]
- Schmidt, A.; Bickle, M.; Beck, T.; Hall, M.N. The yeast phosphatidylinositol kinase homolog TOR2 activates RHO1 and RHO2 via the exchange factor ROM2. Cell 1997, 88, 531–542. [Google Scholar] [CrossRef]
- Ye, Y.; Lee, I.J.; Runge, K.W.; Wu, J.Q. Roles of putative Rho-GEF Gef2 in division-site positioning and contractile-ring function in fission yeast cytokinesis. Mol. Biol. Cell 2012, 23, 1181–1195. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.H.; Ye, Y.; Wu, Z.; Wu, J.Q. Cooperation between Rho-GEF Gef2 and its binding partner Nod1 in the regulation of fission yeast cytokinesis. Mol. Biol. Cell 2013, 24, 3187–3204. [Google Scholar] [CrossRef]
- Nakano, K.; Mutoh, T.; Mabuchi, I. Characterization of GTPase-activating proteins for the function of the Rho-family small GTPases in the fission yeast Schizosaccharomyces pombe. Genes Cells 2001, 6, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Mabuchi, I. Isolation and sequencing of two cDNA clones encoding Rho proteins from the fission yeast Schizosaccharomyces pombe. Gene 1995, 155, 119–122. [Google Scholar] [CrossRef]
- Soto, T.; Villar-Tajadura, M.A.; Madrid, M.; Vicente, J.; Gacto, M.; Perez, P.; Cansado, J. Rga4 modulates the activity of the fission yeast cell integrity MAPK pathway by acting as a Rho2 GTPase-activating protein. J. Biol. Chem. 2010, 285, 11516–11525. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, N.; Karatsu, K.; Miyamoto, M. Role of guanine nucleotide exchange factors for Rho family GTPases in the regulation of cell morphology and actin cytoskeleton in fission yeast. Biochem. Biophys. Res. Commun. 2003, 312, 414–420. [Google Scholar] [CrossRef]
- Wang, N.; Wang, M.; Zhu, Y.H.; Grosel, T.W.; Sun, D.; Kudryashov, D.S.; Wu, J.Q. The Rho-GEF Gef3 interacts with the septin complex and activates the GTPase Rho4 during fission yeast cytokinesis. Mol. Biol. Cell 2015, 26, 238–255. [Google Scholar] [CrossRef]
- Chen, C.R.; Li, Y.C.; Chen, J.; Hou, M.C.; Papadaki, P.; Chang, E.C. Moe1, a conserved protein in Schizosaccharomyces pombe, interacts with a Ras effector, Scd1, to affect proper spindle formation. Proc. Natl. Acad. Sci. USA 1999, 96, 517–522. [Google Scholar] [CrossRef]
- Chang, E.; Bartholomeusz, G.; Pimental, R.; Chen, J.; Lai, H.; Wang, L.; Yang, P.; Marcus, S. Direct binding and in vivo regulation of the fission yeast p21-activated kinase Shk1 by the SH3 domain protein Scd2. Mol. Cell. Biol. 1999, 19, 8066–8074. [Google Scholar] [CrossRef]
- Endo, M.; Shirouzu, M.; Yokoyama, S. The Cdc42 binding and scaffolding activities of the fission yeast adaptor protein Scd2. J. Biol. Chem. 2003, 278, 843–852. [Google Scholar] [CrossRef]
- Murray, J.M.; Johnson, D.I. The Cdc42p GTPase and its regulators Nrf1p and Scd1p are involved in endocytic trafficking in the fission yeast Schizosaccharomyces pombe. J. Biol. Chem. 2001, 276, 3004–3009. [Google Scholar] [CrossRef]
- Li, Y.C.; Chen, C.R.; Chang, E.C. Fission yeast Ras1 effector Scd1 interacts with the spindle and affects its proper formation. Genetics 2000, 156, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Tay, Y.D.; Leda, M.; Goryachev, A.B.; Sawin, K.E. Local and global Cdc42 guanine nucleotide exchange factors for fission yeast cell polarity are coordinated by microtubules and the Tea1-Tea4-Pom1 axis. J. Cell Sci. 2018, 131. [Google Scholar] [CrossRef] [PubMed]
- McDonald, N.A.; Lind, A.L.; Smith, S.E.; Li, R.; Gould, K.L. Nanoscale architecture of the Schizosaccharomyces pombe contractile ring. eLife 2017, 6, e28865. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, S.; Manjón, E.; García, P.; Sunnerhagen, P.; Sánchez, Y. The checkpoint-dependent nuclear accumulation of Rho1p exchange factor Rgf1p is important for tolerance to chronic replication stress. Mol. Biol. Cell 2014, 25, 1137–1150. [Google Scholar] [CrossRef] [PubMed]
- Manjón, E.; Edreira, T.; Muñoz, S.; Sánchez, Y. Rgf1p (Rho1p GEF) is required for double-strand break repair in fission yeast. Nucleic Acids Res. 2017, 45, 5269–5284. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garcia, P.; Garcia, I.; Marcos, F.; de Garibay, G.R.; Sanchez, Y. Fission yeast Rgf2p is a Rho1p guanine nucleotide exchange factor required for spore wall maturation and for the maintenance of cell integrity in the absence of Rgf1p. Genetics 2009, 181, 1321–1334. [Google Scholar] [CrossRef][Green Version]
- Ren, L.; Willet, A.H.; Roberts-Galbraith, R.H.; McDonald, N.A.; Feoktistova, A.; Chen, J.S.; Huang, H.; Guillen, R.; Boone, C.; Sidhu, S.S.; et al. The Cdc15 and Imp2 SH3 domains cooperatively scaffold a network of proteins that redundantly ensure efficient cell division in fission yeast. Mol. Biol. Cell 2015, 26, 256–269. [Google Scholar] [CrossRef]
- Davidson, R.; Laporte, D.; Wu, J.-Q. Regulation of Rho-GEF Rgf3 by the arrestin Art1 in fission yeast cytokinesis. Mol. Biol. Cell 2014, 26, 453–466. [Google Scholar] [CrossRef]
- Rustici, G.; Mata, J.; Kivinen, K.; Lió, P.; Penkett, C.J.; Burns, G.; Hayles, J.; Brazma, A.; Nurse, P.; Bähler, J. Periodic gene expression program of the fission yeast cell cycle. Nat. Genet. 2004, 36, 809–817. [Google Scholar] [CrossRef]
- Das, M.; Wiley, D.J.; Medina, S.; Vincent, H.A.; Larrea, M.; Oriolo, A.; Verde, F. Regulation of cell diameter, For3p localization, and cell symmetry by fission yeast Rho-GAP Rga4p. Mol. Biol. Cell 2007, 18, 2090–2101. [Google Scholar] [CrossRef]
- Tatebe, H.; Nakano, K.; Maximo, R.; Shiozaki, K. Pom1 DYRK regulates localization of the Rga4 GAP to ensure bipolar activation of Cdc42 in fission yeast. Curr. Biol. 2008, 18, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Kokkoris, K.; Gallo Castro, D.; Martin, S.G. The Tea4-PP1 landmark promotes local growth by dual Cdc42 GEF recruitment and GAP exclusion. J. Cell Sci. 2014, 127, 2005–2016. [Google Scholar] [CrossRef]
- Calonge, T.M.; Arellano, M.; Coll, P.M.; Perez, P. Rga5p is a specific Rho1p GTPase-activating protein that regulates cell integrity in Schizosaccharomyces pombe. Mol. Microbiol. 2003, 47, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Revilla-Guarinos, M.T.; Martin-Garcia, R.; Villar-Tajadura, M.A.; Estravis, M.; Coll, P.M.; Perez, P. Rga6 is a fission yeast Rho GAP involved in Cdc42 regulation of polarized growth. Mol. Biol. Cell 2016, 27, 1524–1535. [Google Scholar] [CrossRef] [PubMed]
- Martín-García, R.; Coll, P.M.; Pérez, P. F-BAR domain protein Rga7 collaborates with Cdc15 and Imp2 to ensure proper cytokinesis in fission yeast. J. Cell Sci. 2014, 127, 4146–4158. [Google Scholar] [CrossRef] [PubMed]
- Arasada, R.; Pollard, T.D. A role for F-BAR protein Rga7p during cytokinesis in S. pombe. J. Cell Sci. 2015, 128, 2259–2268. [Google Scholar] [CrossRef]
- Yang, P.; Qyang, Y.; Bartholomeusz, G.; Zhou, X.; Marcus, S. The novel Rho GTPase-activating protein family protein, Rga8, provides a potential link between Cdc42/p21-activated kinase and Rho signaling pathways in the fission yeast, Schizosaccharomyces pombe. J. Biol. Chem. 2003, 278, 48821–48830. [Google Scholar] [CrossRef]
- Murray, J.M.; Johnson, D.I. Isolation and characterization of Nrf1p, a novel negative regulator of the Cdc42p GTPase in Schizosaccharomyces pombe. Genetics 2000, 154, 155–165. [Google Scholar] [CrossRef]
- Ottilie, S.; Miller, P.J.; Johnson, D.I.; Creasy, C.L.; Sells, M.A.; Bagrodia, S.; Forsburg, S.L.; Chernoff, J. Fission yeast pak1+ encodes a protein kinase that interacts with Cdc42p and is involved in the control of cell polarity and mating. EMBO J. 1995, 14, 5908–5919. [Google Scholar] [CrossRef]
- Fukui, Y.; Yamamoto, M. Isolation and characterization of Schizosaccharomyces pombe mutants phenotypically similar to ras1. Mol. Gen. Genet. 1988, 215, 26–31. [Google Scholar] [CrossRef]
- Hirota, K.; Tanaka, K.; Ohta, K.; Yamamoto, M. Gef1p and Scd1p, the Two GDP-GTP exchange factors for Cdc42p, form a ring structure that shrinks during cytokinesis in Schizosaccharomyces pombe. Mol. Biol. Cell 2003, 14, 3617–3627. [Google Scholar] [CrossRef]
- Davì, V.; Tanimoto, H.; Ershov, D.; Haupt, A.; De Belly, H.; Le Borgne, R.; Couturier, E.; Boudaoud, A.; Minc, N. Mechanosensation dynamically coordinates polar growth and cell wall assembly to promote cell survival. Dev. Cell 2018, 45, 170–182. [Google Scholar] [CrossRef]
- Bohnert, K.A.; Gould, K.L. Cytokinesis-Based constraints on polarized cell growth in fission yeast. PLoS Genet. 2012, 8, e1003004. [Google Scholar] [CrossRef] [PubMed]
- Makushok, T.; Alves, P.; Huisman, S.M.; Kijowski, A.R.; Brunner, D. Sterol-Rich membrane domains define fission yeast cell polarity. Cell 2016, 165, 1182–1196. [Google Scholar] [CrossRef]
- Estravis, M.; Rincon, S.A.; Santos, B.; Perez, P. Cdc42 regulates multiple membrane traffic events in fission yeast. Traffic 2011, 12, 1744–1758. [Google Scholar] [CrossRef] [PubMed]
- Estravis, M.; Rincon, S.A.; Portales, E.; Perez, P.; Santos, B. Cdc42 activation state affects its localization and protein levels in fission yeast. Microbiology 2017, 163, 1156–1166. [Google Scholar] [CrossRef] [PubMed]
- Sohrmann, M.; Peter, M. Polarizing without a c(l)ue. Trends Cell Biol. 2003, 13, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Wedlich-Soldner, R.; Altschuler, S.; Wu, L.; Li, R. Spontaneous cell polarization through actomyosin-based delivery of the Cdc42 GTPase. Science 2003, 299, 1231–1235. [Google Scholar] [CrossRef] [PubMed]
- Witte, K.; Strickland, D.; Glotzer, M. Cell cycle entry triggers a switch between two modes of Cdc42 activation during yeast polarization. eLife 2017, 6, e26722. [Google Scholar] [CrossRef]
- Woods, B.; Kuo, C.C.; Wu, C.F.; Zyla, T.R.; Lew, D.J. Polarity establishment requires localized activation of Cdc42. J. Cell Biol. 2015, 211, 19–26. [Google Scholar] [CrossRef]
- Rapali, P.; Mitteau, R.; Braun, C.; Massoni-Laporte, A.; Ünlü, C.; Bataille, L.; Arramon, F.S.; Gygi, S.P.; McCusker, D. Scaffold-mediated gating of Cdc42 signalling flux. eLife 2017, 6, e25257. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.C.; Savage, N.S.; Chen, H.; Wu, C.F.; Zyla, T.R.; Lew, D.J. Inhibitory GEF phosphorylation provides negative feedback in the yeast polarity circuit. Curr. Biol. 2014, 24, 753–759. [Google Scholar] [CrossRef]
- Okada, S.; Leda, M.; Hanna, J.; Savage, N.S.; Bi, E.; Goryachev, A.B. Daughter cell identity emerges from the Interplay of Cdc42, Septins, and Exocytosis. Dev. Cell 2013, 26, 148–161. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.M.; Lambert, Q.T.; Reuther, G.W.; Malliri, A.; Siderovski, D.P.; Sondek, J.; Collard, J.G.; Der, C.J. Tiam1 mediates Ras activation of Rac by a PI(3)K-independent mechanism. Nat. Cell Biol. 2002, 4, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Bendezu, F.O.; Vincenzetti, V.; Martin, S.G. Fission yeast Sec3 and Exo70 are transported on actin cables and localize the exocyst complex to cell poles. PLoS ONE 2012, 7, e40248. [Google Scholar] [CrossRef]
- Dodgson, J.; Chessel, A.; Vaggi, F.; Giordan, M.; Yamamoto, M.; Arai, K.; Madrid, M.; Geymonat, M.; Abenza, J.F.; Cansado, J.; et al. Reconstructing regulatory pathways by systematically mapping protein localization interdependency networks. bioRxiv 2017, 116749. [Google Scholar] [CrossRef]
- Gulli, M.P.; Jaquenoud, M.; Shimada, Y.; Niederhäuser, G.; Wiget, P.; Peter, M. Phosphorylation of the Cdc42 exchange factor Cdc24 by the PAK-like kinase Cla4 may regulate polarized growth in yeast. Mol. Cell 2000, 6, 1155–1167. [Google Scholar] [CrossRef]
- Martin, S.G.; McDonald, W.H.; Yates, J.R., 3rd; Chang, F. Tea4p links microtubule plus ends with the formin For3p in the establishment of cell polarity. Dev. Cell 2005, 8, 479–491. [Google Scholar] [CrossRef]
- Tatebe, H.; Shimada, K.; Uzawa, S.; Morigasaki, S.; Shiozaki, K. Wsh3/Tea4 is a novel cell-end factor essential for bipolar distribution of Tea1 and protects cell polarity under environmental stress in S. pombe. Curr. Biol. 2005, 15, 1006–1015. [Google Scholar] [CrossRef]
- Martin, S.G.; Berthelot-Grosjean, M. Polar gradients of the DYRK-family kinase Pom1 couple cell length with the cell cycle. Nature 2009, 459, 852–856. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Tabares, I.; Grallert, A.; Ortiz, J.M.; Hagan, I.M. Schizosaccharomyces pombe Protein Phosphatase 1 in mitosis, endocytosis and a partnership with Wsh3/Tea4 to control polarised growth. J. Cell Sci. 2007, 120, 3589–3601. [Google Scholar] [CrossRef] [PubMed]
- Hachet, O.; Berthelot-Grosjean, M.; Kokkoris, K.; Vincenzetti, V.; Moosbrugger, J.; Martin, S.G. A phosphorylation cycle shapes gradients of the DYRK family kinase Pom1 at the plasma membrane. Cell 2011, 145, 1116–1128. [Google Scholar] [CrossRef]
- Bi, E.; Park, H.-O. Cell polarization and cytokinesis in budding yeast. Genetics 2012, 191, 347–387. [Google Scholar] [CrossRef]
- Mata, J.; Nurse, P. tea1 and the microtubular cytoskeleton are important for generating global spatial order within the fission yeast cell. Cell 1997, 89, 939–949. [Google Scholar] [CrossRef]
- Bahler, J.; Pringle, J.R. Pom1p, a fission yeast protein kinase that provides positional information for both polarized growth and cytokinesis. Genes Dev. 1998, 12, 1356–1370. [Google Scholar] [CrossRef] [PubMed]
- Rich-Robinson, J.; Russell, A.; Mancini, E.; Das, M. Cdc42 reactivation at growth sites is regulated by cell-cycle-dependent removal of its GAP Rga4 in fission yeast. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zheng, S.; Zheng, B.; Liu, Z.; Wei, W.; Fu, C. The Cdc42 GTPase activating protein Rga6 promotes the cortical localization of Septin. bioRxiv 2021. [Google Scholar] [CrossRef]
- Iwase, M.; Luo, J.; Nagaraj, S.; Longtine, M.; Kim, H.B.; Haarer, B.K.; Caruso, C.; Tong, Z.; Pringle, J.R.; Bi, E. Role of a Cdc42p effector pathway in recruitment of the yeast septins to the presumptive bud site. Mol. Biol. Cell 2006, 17, 1110–1125. [Google Scholar] [CrossRef]
- Pino, M.R.; Nuñez, I.; Chen, C.; Das, M.E.; Wiley, D.J.; D’Urso, G.; Buchwald, P.; Vavylonis, D.; Verde, F. Cdc42 GTPase activating proteins (GAPs) maintain generational inheritance of cell polarity and cell shape in fission yeast. bioRxiv 2020. [Google Scholar] [CrossRef]
- Bugaj, L.J.; Choksi, A.T.; Mesuda, C.K.; Kane, R.S.; Schaffer, D.V. Optogenetic protein clustering and signaling activation in mammalian cells. Nat. Methods 2013, 10, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yu, X.; Li, K.; Klejnot, J.; Yang, H.; Lisiero, D.; Lin, C. Photoexcited CRY2 interacts with CIB1 to regulate transcription and floral initiation in Arabidopsis. Science 2008, 322, 1535–1539. [Google Scholar] [CrossRef]
- Lamas, I.; Weber, N.; Martin, S.G. Activation of Cdc42 GTPase upon CRY2-induced cortical recruitment is antagonized by GAPs in fission yeast. Cells 2020, 9, 2089. [Google Scholar] [CrossRef] [PubMed]
- Sells, M.A.; Barratt, J.T.; Caviston, J.; Ottilie, S.; Leberer, E.; Chernoff, J. Characterization of Pak2p, a pleckstrin homology domain-containing, p21-activated protein kinase from fission yeast. J. Biol. Chem. 1998, 273, 18490–18498. [Google Scholar] [CrossRef]
- Marcus, S.; Polverino, A.; Chang, E.; Robbins, D.; Cobb, M.H.; Wigler, M.H. Shk1, a homolog of the Saccharomyces cerevisiae Ste20 and mammalian p65PAK protein kinases, is a component of a Ras/Cdc42 signaling module in the fission yeast Schizosaccharomyces pombe. Proc. Natl. Acad. Sci. USA 1995, 92, 6180–6184. [Google Scholar] [CrossRef]
- Verde, F.; Wiley, D.J.; Nurse, P. Fission yeast Orb6, a Ser/Thr protein kinase related to mammalian Rho kinase and myotonic dystrophy kinase, is required for maintenance of cell polarity and coordinates cell morphogenesis with the cell cycle. Proc. Natl. Acad. Sci. USA 1998, 95, 7526–7531. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Kansra, S.; Pimental, R.A.; Gilbreth, M.; Marcus, S. Cloning and characterization of shk2, a gene encoding a novel p21-activated protein kinase from fission yeast. J. Biol. Chem. 1998, 273, 18481–18489. [Google Scholar] [CrossRef] [PubMed]
- Qyang, Y.; Yang, P.; Du, H.; Lai, H.; Kim, H.; Marcus, S. The p21-activated kinase, Shk1, is required for proper regulation of microtubule dynamics in the fission yeast Schizosaccharomyces pombe. Mol. Microbiol. 2002, 44, 325–334. [Google Scholar] [CrossRef]
- Tu, H.; Wigler, M. Genetic evidence for Pak1 autoinhibition and its release by Cdc42. Mol. Cell. Biol. 1999, 19, 602–611. [Google Scholar] [CrossRef]
- Lei, M.; Lu, W.; Meng, W.; Parrini, M.C.; Eck, M.J.; Mayer, B.J.; Harrison, S.C. Structure of PAK1 in an autoinhibited conformation reveals a multistage activation switch. Cell 2000, 102, 387–397. [Google Scholar] [CrossRef]
- Morreale, A.; Venkatesan, M.; Mott, H.R.; Owen, D.; Nietlispach, D.; Lowe, P.N.; Laue, E.D. Structure of Cdc42 bound to the GTPase binding domain of PAK. Nat. Struct. Biol. 2000, 7, 384–388. [Google Scholar] [CrossRef]
- Kim, H.; Yang, P.; Catanuto, P.; Verde, F.; Lai, H.; Du, H.; Chang, F.; Marcus, S. The kelch repeat protein, Tea1, is a potential substrate target of the p21-activated kinase, Shk1, in the fission yeast Schizosaccharomyces pombe. J. Biol. Chem. 2003, 278, 30074–30082. [Google Scholar] [CrossRef] [PubMed]
- Geymonat, M.; Chessel, A.; Dodgson, J.; Punter, H.; Horns, F.; Nagy, A.C.; Salas, R.E.C. Activation of polarized cell growth by inhibition of cell polarity. bioRxiv 2018, 402990. [Google Scholar] [CrossRef]
- Martin, S.G.; Chang, F. Dynamics of the formin For3p in actin cable assembly. Curr. Biol. 2006, 16, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.J.; Neidt, E.M.; Kovar, D.R. The functionally distinct fission yeast formins have specific actin-assembly properties. Mol. Biol. Cell 2011, 22, 3826–3839. [Google Scholar] [CrossRef] [PubMed]
- Motegi, F.; Arai, R.; Mabuchi, I. Identification of two type V myosins in fission yeast, one of which functions in polarized cell growth and moves rapidly in the cell. Mol. Biol. Cell 2001, 12, 1367–1380. [Google Scholar] [CrossRef]
- Cortes, J.C.; Carnero, E.; Ishiguro, J.; Sanchez, Y.; Duran, A.; Ribas, J.C. The novel fission yeast (1,3)β-D-glucan synthase catalytic subunit Bgs4p is essential during both cytokinesis and polarized growth. J. Cell Sci. 2005, 118, 157–174. [Google Scholar] [CrossRef]
- Mulvihill, D.P.; Edwards, S.R.; Hyams, J.S. A critical role for the type V myosin, Myo52, in septum deposition and cell fission during cytokinesis in Schizosaccharomyces pombe. Cell. Motil. Cytoskelet. 2006, 63, 149–161. [Google Scholar] [CrossRef]
- Glynn, J.M.; Lustig, R.J.; Berlin, A.; Chang, F. Role of Bud6p and Tea1p in the interaction between actin and microtubules for the establishment of cell polarity in fission yeast. Curr. Biol. 2001, 11, 836–845. [Google Scholar] [CrossRef]
- Dong, Y.; Pruyne, D.; Bretscher, A. Formin-dependent actin assembly is regulated by distinct modes of Rho signaling in yeast. J. Cell Biol. 2003, 161, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- TerBush, D.R.; Maurice, T.; Roth, D.; Novick, P. The Exocyst is a multiprotein complex required for exocytosis in Saccharomyces cerevisiae. EMBO J. 1996, 15, 6483–6494. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Guo, W. The exocyst complex in polarized exocytosis. Curr. Opin. Cell Biol. 2009, 21, 537–542. [Google Scholar] [CrossRef]
- Finger, F.P.; Hughes, T.E.; Novick, P. Sec3p is a spatial landmark for polarized secretion in budding yeast. Cell 1998, 92, 559–571. [Google Scholar] [CrossRef]
- Mutavchiev, D.R.; Leda, M.; Sawin, K.E. Remodeling of the fission yeast Cdc42 cell-polarity module via the Sty1 p38 Stress-Activated Protein Kinase pathway. Curr. Biol. 2016, 26, 2921–2928. [Google Scholar] [CrossRef]
- Haupt, A.; Ershov, D.; Minc, N. A positive feedback between growth and polarity provides directional persistency and flexibility to the process of tip growth. Curr. Biol. 2018, 28, 3342–3351. [Google Scholar] [CrossRef] [PubMed]
- Brewster, J.L.; Gustin, M.C. Hog1: 20 years of discovery and impact. Sci. Signal. 2014, 7, re7. [Google Scholar] [CrossRef]
- Koyano, T.; Kume, K.; Konishi, M.; Toda, T.; Hirata, D. Search for kinases related to transition of growth polarity in fission yeast. Biosci. Biotechnol. Biochem. 2010, 74, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Papp, L.; Sipiczki, M.; Holb, I.J.; Miklós, I. Optimal conditions for mycelial growth of Schizosaccharomyces japonicus cells in liquid medium: It enables the molecular investigation of dimorphism. Yeast 2014, 31, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Furuya, K.; Niki, H. The DNA damage checkpoint regulates a transition between yeast and hyphal growth in Schizosaccharomyces japonicus. Mol. Cell. Biol. 2010, 30, 2909–2917. [Google Scholar] [CrossRef]
- Furuya, K.; Niki, H. Hyphal differentiation induced via a DNA damage checkpoint-dependent pathway engaged in crosstalk with nutrient stress signaling in Schizosaccharomyces japonicus. Curr. Genet. 2012, 58, 291–303. [Google Scholar] [CrossRef][Green Version]
- Chen, H.; Zhou, X.; Ren, B.; Cheng, L. The regulation of hyphae growth in Candida albicans. Virulence 2020, 11, 337–348. [Google Scholar] [CrossRef]
- Gómez-Gil, E.; Franco, A.; Madrid, M.; Vázquez-Marín, B.; Gacto, M.; Fernández-Breis, J.; Vicente-Soler, J.; Soto, T.; Cansado, J. Quorum sensing and stress-activated MAPK signaling repress yeast to hypha transition in the fission yeast Schizosaccharomyces japonicus. PLoS Genet. 2019, 15, e1008192. [Google Scholar] [CrossRef]
- Mazur, P.; Baginsky, W. In vitro activity of 1,3-β-D-glucan synthase requires the GTP-binding protein Rho1. J. Biol. Chem. 1996, 271, 14604–14609. [Google Scholar] [CrossRef]
- Mukai, H. The structure and function of PKN, a protein kinase having a catalytic domain homologous to that of PKC. J. Biochem. 2003, 133, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Estravis, M.; Rincon, S.; Perez, P. Cdc42 regulation of polarized traffic in fission yeast. Commun. Integr. Biol. 2012, 5, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Sugiura, R.; Ohta, K.; Tada, K.; Suzuki, M.; Hirata, M.; Nakamura, S.-I.; Shuntoh, H.; Kuno, T. Phosphatidylinositol-4-phosphate 5-kinase regulates fission yeast cell integrity through a phospholipase c-mediated protein kinase C-independent pathway. J. Biol. Chem. 2005, 280, 27561–27568. [Google Scholar] [CrossRef] [PubMed]
- Martín, V.; García, B.; Carnero, E.; Durán, A.; Sánchez, Y. Bgs3p, a putative 1,3-β-glucan synthase subunit, is required for cell wall assembly in Schizosaccharomyces pombe. Eukaryot. Cell 2003, 2, 159–169. [Google Scholar] [CrossRef]
- Cruz, S.; Munoz, S.; Manjon, E.; Garcia, P.; Sanchez, Y. The fission yeast cell wall stress sensor-like proteins Mtl2 and Wsc1 act by turning on the GTPase Rho1p but act independently of the cell wall integrity pathway. Microbiologyopen 2013, 2, 778–794. [Google Scholar] [CrossRef]
- Neeli-Venkata, R.; Celador, R.; Sanchez, Y.; Minc, N. Detection of surface forces by a cell wall mechanosensor. bioRxiv 2020. [Google Scholar] [CrossRef]
- Davi, V.; Minc, N. Mechanics and morphogenesis of fission yeast cells. Curr. Opin. Microbiol. 2015, 28, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.E. Regulation of cell wall biogenesis in Saccharomyces cerevisiae: The cell wall integrity signaling pathway. Genetics 2011, 189, 1145–1175. [Google Scholar] [CrossRef]
- Perez, P.; Cansado, J. Cell integrity signaling and response to stress in fission yeast. Curr. Protein Pept. Sci. 2010, 11, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Shen, X.; Yan, G.; Ma, D.; Bai, X.; Li, S.; Jiang, Y. A Map kinase dependent feedback mechanism controls Rho1 GTPase and actin distribution in yeast. PLoS ONE 2009, 4, e6089. [Google Scholar] [CrossRef] [PubMed]
- Kabeche, R.; Madrid, M.; Cansado, J.; Moseley, J.B. Eisosomes regulate phosphatidylinositol 4,5-Bisphosphate (PI(4,5)P2) Cortical Clusters and Mitogen-activated Protein (MAP) kinase signaling upon osmotic stress. J. Biol. Chem. 2015, 290, 25960–25973. [Google Scholar] [CrossRef] [PubMed]
- Franco, A.; Soto, T.; Martín-García, R.; Madrid, M.; Vázquez-Marín, B.; Vicente-Soler, J.; Coll, P.M.; Gacto, M.; Pérez, P.; Cansado, J. Distinct functional relevance of dynamic GTPase cysteine methylation in fission yeast. Sci. Rep. 2017, 7, 6057. [Google Scholar] [CrossRef]
- Wang, M.; Casey, P.J. Protein prenylation: Unique fats make their mark on biology. Nat. Rev. Mol. Cell. Biol. 2016, 17, 110–122. [Google Scholar] [CrossRef]
- Cansado, J. To finish things well: Cysteine methylation ensures selective GTPase membrane localization and signalling. Curr. Genet. 2018, 64, 341–344. [Google Scholar] [CrossRef]
- Viana, R.A.; Pinar, M.; Soto, T.; Coll, P.M.; Cansado, J.; Perez, P. Negative functional interaction between cell integrity MAPK pathway and Rho1 GTPase in fission yeast. Genetics 2013, 195, 421–432. [Google Scholar] [CrossRef]
- Kampmeyer, C.; Johansen, J.V.; Holmberg, C.; Karlson, M.; Gersing, S.K.; Bordallo, H.N.; Kragelund, B.B.; Lerche, M.H.; Jourdain, I.; Winther, J.R.; et al. Mutations in a single signaling pathway allow cell growth in heavy water. ACS Synth. Biol. 2020, 9, 733–748. [Google Scholar] [CrossRef] [PubMed]
- Madrid, M.; Soto, T.; Khong, H.K.; Franco, A.; Vicente, J.; Perez, P.; Gacto, M.; Cansado, J. Stress-induced response, localization, and regulation of the Pmk1 cell integrity pathway in Schizosaccharomyces pombe. J. Biol. Chem. 2006, 281, 2033–2043. [Google Scholar] [CrossRef] [PubMed]
- Kono, K.; Saeki, Y.; Yoshida, S.; Tanaka, K.; Pellman, D. Proteasomal degradation resolves competition between cell polarization and cellular wound healing. Cell 2012, 150, 151–164. [Google Scholar] [CrossRef]
- Rincon, S.A.; Paoletti, A. Molecular control of fission yeast cytokinesis. Semin. Cell Dev. Biol. 2016, 53, 28–38. [Google Scholar] [CrossRef]
- Pollard, T.D.; Wu, J.Q. Understanding cytokinesis: Lessons from fission yeast. Nat. Rev. Mol. Cell. Biol. 2010, 11, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Garcia Cortes, J.C.; Ramos, M.; Osumi, M.; Perez, P.; Ribas, J.C. The Cell biology of fission yeast septation. Microbiol. Mol. Biol. Rev. 2016, 80, 779–791. [Google Scholar] [CrossRef]
- Onwubiko, U.N.; Rich-Robinson, J.; Mustaf, R.A.; Das, M.E. Cdc42 promotes Bgs1 recruitment for septum synthesis and glucanase localization for cell separation during cytokinesis in fission yeast. Small GTPases 2020, 1–8. [Google Scholar] [CrossRef]
- Meitinger, F.; Boehm, M.E.; Hofmann, A.; Hub, B.; Zentgraf, H.; Lehmann, W.D.; Pereira, G. Phosphorylation-dependent regulation of the F-BAR protein Hof1 during cytokinesis. Genes Dev. 2011, 25, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Bose, I.; Irazoqui, J.E.; Moskow, J.J.; Bardes, E.S.; Zyla, T.R.; Lew, D.J. Assembly of scaffold-mediated complexes containing Cdc42p, the exchange factor Cdc24p, and the effector Cla4p required for cell cycle-regulated phosphorylation of Cdc24p. J. Biol. Chem. 2001, 276, 7176–7186. [Google Scholar] [CrossRef] [PubMed]
- Versele, M.; Thorner, J. Septin collar formation in budding yeast requires GTP binding and direct phosphorylation by the PAK, Cla4. J. Cell Biol. 2004, 164, 701–715. [Google Scholar] [CrossRef]
- Muñoz, J.; Cortés, J.C.G.; Sipiczki, M.; Ramos, M.; Clemente-Ramos, J.A.; Moreno, M.B.; Martins, I.M.; Pérez, P.; Ribas, J.C. Extracellular cell wall β(1,3)glucan is required to couple septation to actomyosin ring contraction. J. Cell Biol. 2013, 203, 265–282. [Google Scholar] [CrossRef]
- Arasada, R.; Pollard, T.D. Contractile ring stability in S. pombe depends on F-BAR protein Cdc15p and Bgs1p transport from the Golgi Complex. Cell Rep. 2014, 8, 1533–1544. [Google Scholar] [CrossRef]
- Watanabe, S.; Okawa, K.; Miki, T.; Sakamoto, S.; Morinaga, T.; Segawa, K.; Arakawa, T.; Kinoshita, M.; Ishizaki, T.; Narumiya, S. Rho and anillin-dependent control of mDia2 localization and function in cytokinesis. Mol. Biol. Cell 2010, 21, 3193–3204. [Google Scholar] [CrossRef]
- Yoshida, S.; Kono, K.; Lowery, D.M.; Bartolini, S.; Yaffe, M.B.; Ohya, Y.; Pellman, D. Polo-Like Kinase Cdc5 controls the local activation of Rho1 to promote cytokinesis. Science 2006, 313, 108. [Google Scholar] [CrossRef] [PubMed]
- Cortés, J.C.; Pujol, N.; Sato, M.; Pinar, M.; Ramos, M.; Moreno, B.; Osumi, M.; Ribas, J.C.; Pérez, P. Cooperation between Paxillin-like Protein Pxl1 and glucan synthase Bgs1 is essential for actomyosin ring stability and septum formation in fission yeast. PLoS Genet 2015, 11, e1005358. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.H.; Hyun, J.; Pan, Y.Z.; Hopper, J.E.; Rizo, J.; Wu, J.Q. Roles of the fission yeast UNC-13/Munc13 protein Ync13 in late stages of cytokinesis. Mol. Biol. Cell 2018, 29, 2259–2279. [Google Scholar] [CrossRef]
- Dundon, S.E.R.; Pollard, T.D. Microtubule nucleation promoters Mto1 and Mto2 regulate cytokinesis in fission yeast. Mol. Biol. Cell 2020, 31, 1846–1856. [Google Scholar] [CrossRef] [PubMed]
- Madrid, M.; Núñez, A.; Soto, T.; Vicente-Soler, J.; Gacto, M.; Cansado, J. Stress-activated protein kinase-mediated down-regulation of the cell integrity pathway mitogen-activated protein kinase Pmk1p by protein phosphatases. Mol. Biol. Cell 2007, 18, 4405–4419. [Google Scholar] [CrossRef]
- Satoh, R.; Morita, T.; Takada, H.; Kita, A.; Ishiwata, S.; Doi, A.; Hagihara, K.; Taga, A.; Matsumura, Y.; Tohda, H.; et al. Role of the RNA-binding protein Nrd1 and Pmk1 mitogen-activated protein kinase in the regulation of myosin mRNA stability in fission yeast. Mol. Biol. Cell 2009, 20, 2473–2485. [Google Scholar] [CrossRef]
- Martín-García, R.; Arribas, V.; Coll, P.M.; Pinar, M.; Viana, R.A.; Rincón, S.A.; Correa-Bordes, J.; Ribas, J.C.; Pérez, P. Paxillin-mediated recruitment of calcineurin to the contractile ring is required for the correct progression of cytokinesis in fission yeast. Cell Rep. 2018, 25, 772–783.e774. [Google Scholar] [CrossRef]
- Simanis, V. Pombe’s thirteen—Control of fission yeast cell division by the septation initiation network. J. Cell Sci. 2015, 128, 1465–1474. [Google Scholar] [CrossRef]
- Martin-Garcia, R.; Santos, B. The price of independence: Cell separation in fission yeast. World J. Microbiol. Biotechnol. 2016, 32, 65. [Google Scholar] [CrossRef]
- Gu, Y.; Yam, C.; Oliferenko, S. Rewiring of cellular division site selection in evolution of fission yeasts. Curr. Biol. 2015, 25, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Oliferenko, S. Cellular geometry scaling ensures robust division site positioning. Nat. Commun. 2019, 10, 268. [Google Scholar] [CrossRef]
- Yonetani, A.; Lustig, R.J.; Moseley, J.B.; Takeda, T.; Goode, B.L.; Chang, F. Regulation and targeting of the fission yeast formin cdc12p in cytokinesis. Mol. Biol. Cell 2008, 19, 2208–2219. [Google Scholar] [CrossRef] [PubMed]
- Park, H.O.; Bi, E. Central roles of small GTPases in the development of cell polarity in yeast and beyond. Microbiol. Mol. Biol. Rev. 2007, 71, 48–96. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Yamamoto, M. The fission yeast mating pheromone P-factor: Its molecular structure, gene structure, and ability to induce gene expression and G1 arrest in the mating partner. Genes Dev. 1994, 8, 328–338. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Davey, J. Mating pheromones of the fission yeast Schizosaccharomyces pombe: Purification and structural characterization of M-factor and isolation and analysis of two genes encoding the pheromone. EMBO J. 1992, 11, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Davey, J.; Imai, Y.; Yamamoto, M. Schizosaccharomyces pombe map3+ encodes the putative M-factor receptor. Mol. Cell. Biol. 1993, 13, 80–88. [Google Scholar] [CrossRef]
- Kitamura, K.; Shimoda, C. The Schizosaccharomyces pombe mam2 gene encodes a putative pheromone receptor which has a significant homology with the Saccharomyces cerevisiae Ste2 protein. EMBO J. 1991, 10, 3743–3751. [Google Scholar] [CrossRef]
- Nadin-Davis, S.A.; Nasim, A. Schizosaccharomyces pombe ras1 and byr1 are functionally related genes of the ste family that affect starvation-induced transcription of mating-type genes. Mol. Cell. Biol. 1990, 10, 549–560. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, H.P.; Riggs, M.; Rodgers, L.; Wigler, M. byr2, a Schizosaccharomyces pombe gene encoding a protein kinase capable of partial suppression of the Ras1 mutant phenotype. Mol. Cell. Biol. 1991, 11, 3554–3563. [Google Scholar] [CrossRef]
- Hughes, D.A.; Ashworth, A.; Marshall, C.J. Complementation of byr1 in fission yeast by mammalian MAP kinase kinase requires coexpression of Raf kinase. Nature 1993, 364, 349–352. [Google Scholar] [CrossRef]
- Bar, E.E.; Ellicott, A.T.; Stone, D.E. Gβγ recruits Rho1 to the site of polarized growth during mating in budding yeast. J. Biol. Chem. 2003, 278, 21798–21804. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.A.; Yabana, N.; Yamamoto, M. Transcriptional regulation of a Ras nucleotide-exchange factor gene by extracellular signals in fission yeast. J. Cell Sci. 1994, 107, 3635–3642. [Google Scholar] [CrossRef] [PubMed]
- Nadin-Davis, S.A.; Nasim, A.; Beach, D. Involvement of ras in sexual differentiation but not in growth control in fission yeast. EMBO J. 1986, 5, 2963–2971. [Google Scholar] [CrossRef]
- Davey, J. Fusion of a fission yeast. Yeast 1998, 14, 1529–1566. [Google Scholar] [CrossRef]
- Merlini, L.; Dudin, O.; Martin, S.G. Mate and fuse: How yeast cells do it. Open Biol. 2013, 3, 130008. [Google Scholar] [CrossRef]
- Tu, H.; Barr, M.; Dong, D.L.; Wigler, M. Multiple regulatory domains on the Byr2 protein kinase. Mol. Cell. Biol. 1997, 17, 5876–5887. [Google Scholar] [CrossRef]
- Leeuw, T.; Fourest-Lieuvin, A.; Wu, C.; Chenevert, J.; Clark, K.; Whiteway, M.; Thomas, D.Y.; Leberer, E. Pheromone response in yeast: Association of Bem1p with proteins of the MAP kinase cascade and actin. Science 1995, 270, 1210–1213. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.; Bahler, J. Global roles of Ste11p, cell type, and pheromone in the control of gene expression during early sexual differentiation in fission yeast. Proc. Natl. Acad. Sci. USA 2006, 103, 15517–15522. [Google Scholar] [CrossRef]
- Vještica, A.; Merlini, L.; Nkosi, P.J.; Martin, S.G. Gamete fusion triggers bipartite transcription factor assembly to block re-fertilization. Nature 2018, 560, 397–400. [Google Scholar] [CrossRef]
- Zhang, M.M.; Wu, P.Y.; Kelly, F.D.; Nurse, P.; Hang, H.C. Quantitative control of protein S-palmitoylation regulates meiotic entry in fission yeast. PLoS Biol. 2013, 11, e1001597. [Google Scholar] [CrossRef] [PubMed]
- Yoo, B.Y.; Calleja, G.B.; Johnson, B.F. Ultrastructural changes of the fission yeast (Schizosaccharomyces pombe) during ascospore formation. Arch. Mikrobiol. 1973, 91, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Tajadura, V.; Garcia, I.; Sanchez, Y. Role of Rho GTPases and Rho-GEFs in the regulation of cell shape and integrity in fission yeast. Yeast 2006, 23, 1031–1043. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, M.; Shimoda, C. The cyclic AMP/PKA signal pathway is required for initiation of spore germination in Schizosaccharomyces pombe. Yeast 2001, 18, 207–217. [Google Scholar] [CrossRef]
- Bonazzi, D.; Julien, J.D.; Romao, M.; Seddiki, R.; Piel, M.; Boudaoud, A.; Minc, N. Symmetry breaking in spore germination relies on an interplay between polar cap stability and spore wall mechanics. Dev. Cell 2014, 28, 534–546. [Google Scholar] [CrossRef] [PubMed]
- Ellerbroek, S.M.; Wennerberg, K.; Burridge, K. Serine phosphorylation negatively regulates RhoA in vivo. J. Biol. Chem. 2003, 278, 19023–19031. [Google Scholar] [CrossRef]
- Forget, M.A.; Desrosiers, R.R.; Gingras, D.; Béliveau, R. Phosphorylation states of Cdc42 and RhoA regulate their interactions with Rho GDP dissociation inhibitor and their extraction from biological membranes. Biochem. J. 2002, 361, 243–254. [Google Scholar] [CrossRef]
- Tay, Y.D.; Leda, M.; Spanos, C.; Rappsilber, J.; Goryachev, A.B.; Sawin, K.E. Fission yeast NDR/LATS kinase Orb6 regulates exocytosis via phosphorylation of the exocyst complex. Cell Rep. 2019, 26, 1654–1667. [Google Scholar] [CrossRef]

 ) composed of multiple proteins localized into the cell middle by anillin-like protein Mid1. After synthesis of the F-actin network by formins Cdc12 and For3, nodes condense into the CAR through actomyosin interactions. The fully formed ring matures by recruitment of additional cytokinesis proteins (mature car is indicated in magenta). Cdc42 effector Pak1 promotes phosphorylation of CAR components Mid1, Cdc15 and Rlc1. Cdc42 mediates Bgs1 recruitment at the ring during anaphase. Rho1 GTPase participates in a cytokinetic checkpoint that delays CAR maturation after cell wall damage. (B) CAR maturation is followed by constriction and cell wall deposition (primary septum, in green, and secondary septum, in brown), which facilitates proper ring closure. Rho1 and Rho2 bind and activate the enzymes involved in septum synthesis. Question mark denotes that the molecular mechanism by which SIN might activate Rho1 remains unsolved. (C) Cells separation occurs by digestion of the primary septum and the surrounding cell wall by Agn1 (endo-α-1,3 glucanase) and Eng1 (endo-β-1,3 glucanase). Secretion of Agn1 and Eng1 depends on the activity of Rho4 (activated by Gef3). Both enzymes are targeted to the septum by the exocyst subunits Sec8 and Exo70 regulated by Rho4 and Rho3. Agn1 and Eng1 distribution at the ring is influenced by Cdc42. Please see main text for more details.
) composed of multiple proteins localized into the cell middle by anillin-like protein Mid1. After synthesis of the F-actin network by formins Cdc12 and For3, nodes condense into the CAR through actomyosin interactions. The fully formed ring matures by recruitment of additional cytokinesis proteins (mature car is indicated in magenta). Cdc42 effector Pak1 promotes phosphorylation of CAR components Mid1, Cdc15 and Rlc1. Cdc42 mediates Bgs1 recruitment at the ring during anaphase. Rho1 GTPase participates in a cytokinetic checkpoint that delays CAR maturation after cell wall damage. (B) CAR maturation is followed by constriction and cell wall deposition (primary septum, in green, and secondary septum, in brown), which facilitates proper ring closure. Rho1 and Rho2 bind and activate the enzymes involved in septum synthesis. Question mark denotes that the molecular mechanism by which SIN might activate Rho1 remains unsolved. (C) Cells separation occurs by digestion of the primary septum and the surrounding cell wall by Agn1 (endo-α-1,3 glucanase) and Eng1 (endo-β-1,3 glucanase). Secretion of Agn1 and Eng1 depends on the activity of Rho4 (activated by Gef3). Both enzymes are targeted to the septum by the exocyst subunits Sec8 and Exo70 regulated by Rho4 and Rho3. Agn1 and Eng1 distribution at the ring is influenced by Cdc42. Please see main text for more details.
 ) composed of multiple proteins localized into the cell middle by anillin-like protein Mid1. After synthesis of the F-actin network by formins Cdc12 and For3, nodes condense into the CAR through actomyosin interactions. The fully formed ring matures by recruitment of additional cytokinesis proteins (mature car is indicated in magenta). Cdc42 effector Pak1 promotes phosphorylation of CAR components Mid1, Cdc15 and Rlc1. Cdc42 mediates Bgs1 recruitment at the ring during anaphase. Rho1 GTPase participates in a cytokinetic checkpoint that delays CAR maturation after cell wall damage. (B) CAR maturation is followed by constriction and cell wall deposition (primary septum, in green, and secondary septum, in brown), which facilitates proper ring closure. Rho1 and Rho2 bind and activate the enzymes involved in septum synthesis. Question mark denotes that the molecular mechanism by which SIN might activate Rho1 remains unsolved. (C) Cells separation occurs by digestion of the primary septum and the surrounding cell wall by Agn1 (endo-α-1,3 glucanase) and Eng1 (endo-β-1,3 glucanase). Secretion of Agn1 and Eng1 depends on the activity of Rho4 (activated by Gef3). Both enzymes are targeted to the septum by the exocyst subunits Sec8 and Exo70 regulated by Rho4 and Rho3. Agn1 and Eng1 distribution at the ring is influenced by Cdc42. Please see main text for more details.
) composed of multiple proteins localized into the cell middle by anillin-like protein Mid1. After synthesis of the F-actin network by formins Cdc12 and For3, nodes condense into the CAR through actomyosin interactions. The fully formed ring matures by recruitment of additional cytokinesis proteins (mature car is indicated in magenta). Cdc42 effector Pak1 promotes phosphorylation of CAR components Mid1, Cdc15 and Rlc1. Cdc42 mediates Bgs1 recruitment at the ring during anaphase. Rho1 GTPase participates in a cytokinetic checkpoint that delays CAR maturation after cell wall damage. (B) CAR maturation is followed by constriction and cell wall deposition (primary septum, in green, and secondary septum, in brown), which facilitates proper ring closure. Rho1 and Rho2 bind and activate the enzymes involved in septum synthesis. Question mark denotes that the molecular mechanism by which SIN might activate Rho1 remains unsolved. (C) Cells separation occurs by digestion of the primary septum and the surrounding cell wall by Agn1 (endo-α-1,3 glucanase) and Eng1 (endo-β-1,3 glucanase). Secretion of Agn1 and Eng1 depends on the activity of Rho4 (activated by Gef3). Both enzymes are targeted to the septum by the exocyst subunits Sec8 and Exo70 regulated by Rho4 and Rho3. Agn1 and Eng1 distribution at the ring is influenced by Cdc42. Please see main text for more details.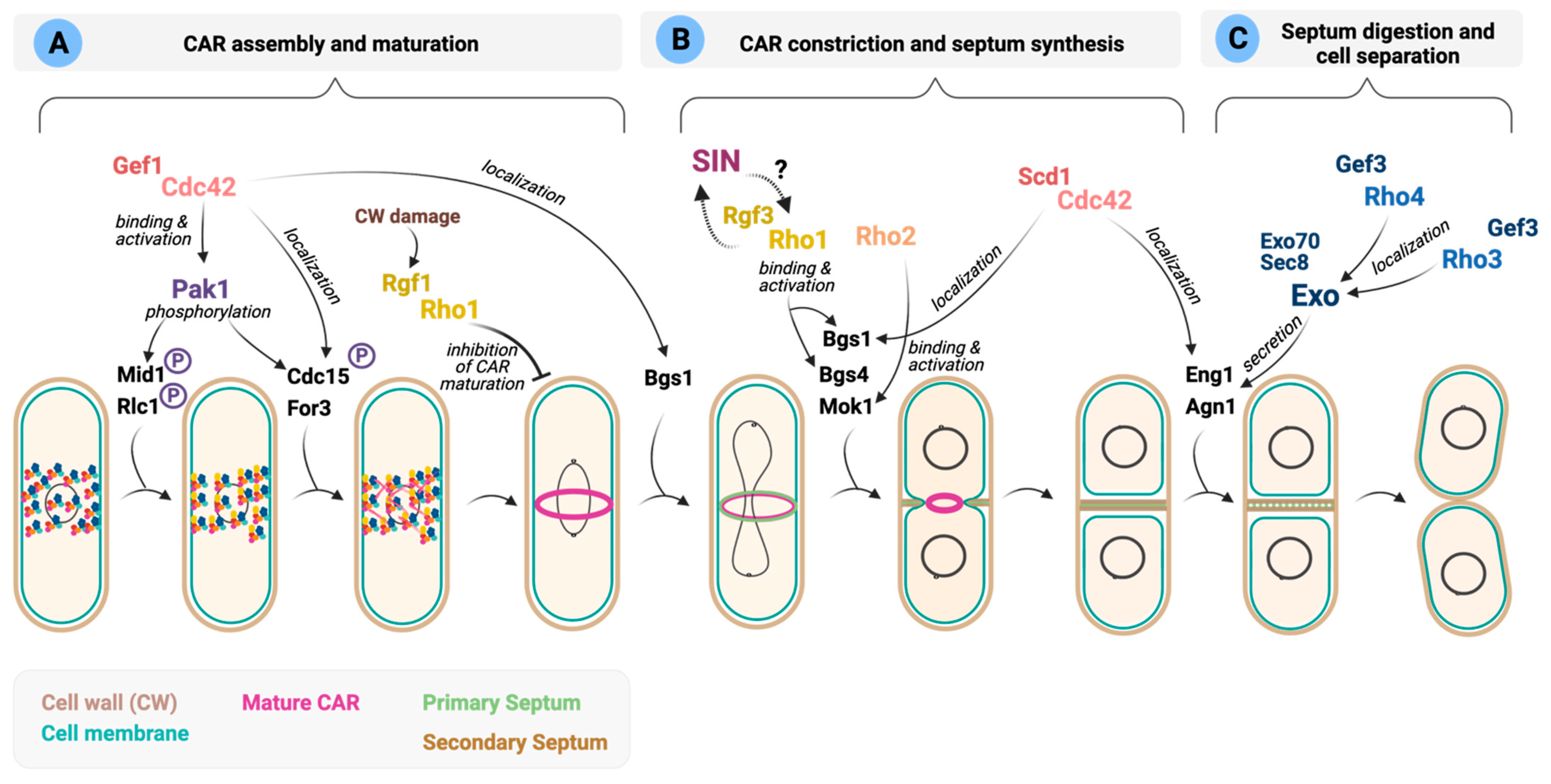
| GTPase | GEF | GAP | GDI | Localization | Regulated/Involved Processes |
|---|---|---|---|---|---|
| Rho1 (●) | Gef2 (+) Rgf1 Rgf2 Rgf3 (●) | Rga1 Rga5 Rga8 | Rdi1 | Active growing sites (cell poles, division area) [17] |
|
| Rho2 | Rgf1? | Rga2 Rga4 Rga6 Rga7 | Active growing sites (cell poles, division area) [32] Modified by palmitoylation [33] and target of farnesyltransferase Cpp1 [34] |
| |
| Rho3 (●) | Gef3 (+) | Division area [40] | |||
| Rho4 | Scd1 Gef2 (+) Gef3 | Rga9 | Rdi1 | Cell periphery (interphase) and division area (M-phase) [47], septum [48] | |
| Rho5 | Gef2 (+) | Cell poles (interphase) and division area (M-phase) [52] | |||
| Cdc42 (●) | Scd1 Gef1 | Rga3 Rga4 Rga6 Rga9 (+) | Rdi1 | Endomembranes and active growing sites (cell poles, division area) [4,54,55,56] |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vicente-Soler, J.; Soto, T.; Franco, A.; Cansado, J.; Madrid, M. The Multiple Functions of Rho GTPases in Fission Yeasts. Cells 2021, 10, 1422. https://doi.org/10.3390/cells10061422
Vicente-Soler J, Soto T, Franco A, Cansado J, Madrid M. The Multiple Functions of Rho GTPases in Fission Yeasts. Cells. 2021; 10(6):1422. https://doi.org/10.3390/cells10061422
Chicago/Turabian StyleVicente-Soler, Jero, Teresa Soto, Alejandro Franco, José Cansado, and Marisa Madrid. 2021. "The Multiple Functions of Rho GTPases in Fission Yeasts" Cells 10, no. 6: 1422. https://doi.org/10.3390/cells10061422
APA StyleVicente-Soler, J., Soto, T., Franco, A., Cansado, J., & Madrid, M. (2021). The Multiple Functions of Rho GTPases in Fission Yeasts. Cells, 10(6), 1422. https://doi.org/10.3390/cells10061422






