Extracellular Vesicle Surface Signatures in IPF Patients: A Multiplex Bead-Based Flow Cytometry Approach
Abstract
1. Introduction
2. Materials and Methods
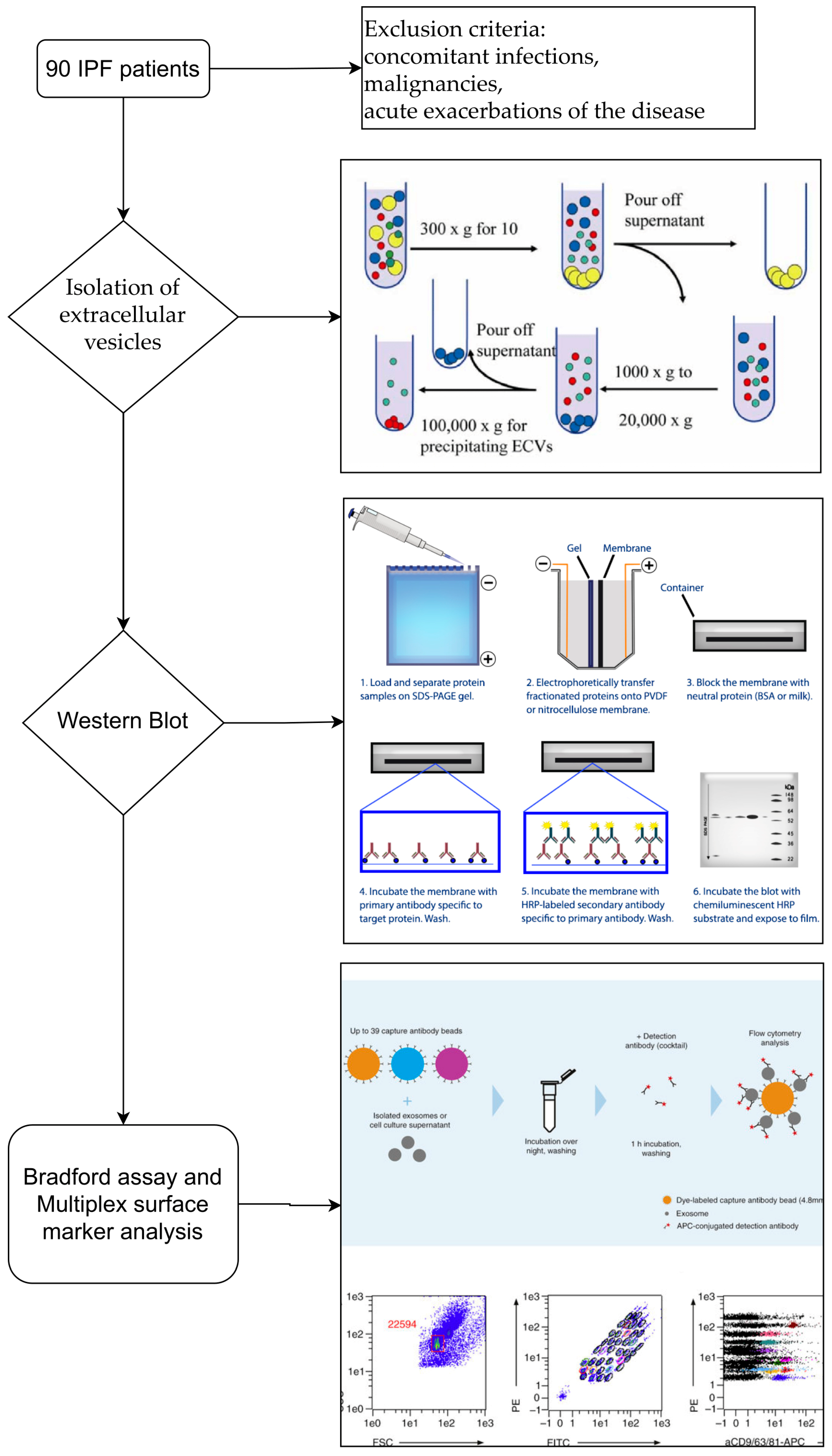
2.1. Study Population
2.2. Human Blood Samples
2.3. Isolation of Extracellular Vesicles
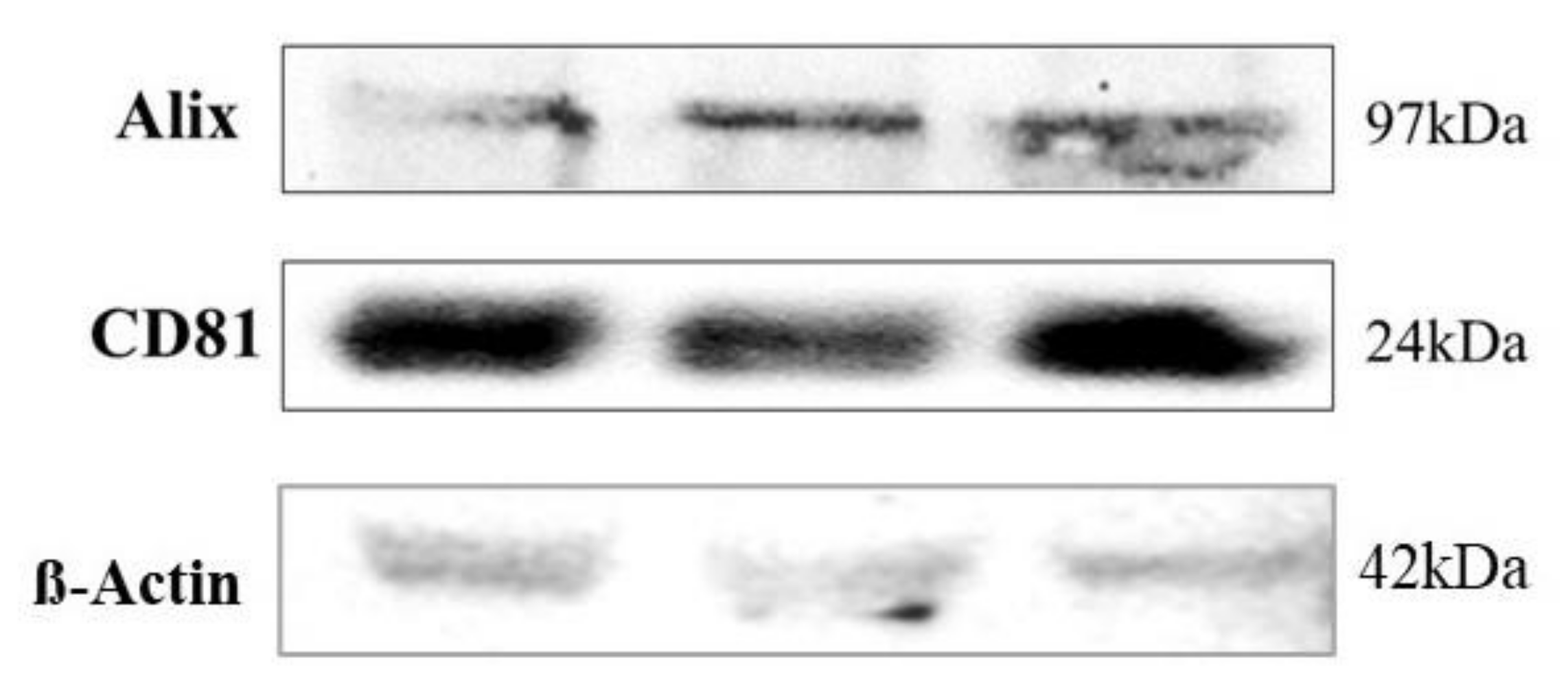
2.4. Western Blot
2.5. Bradford Assay
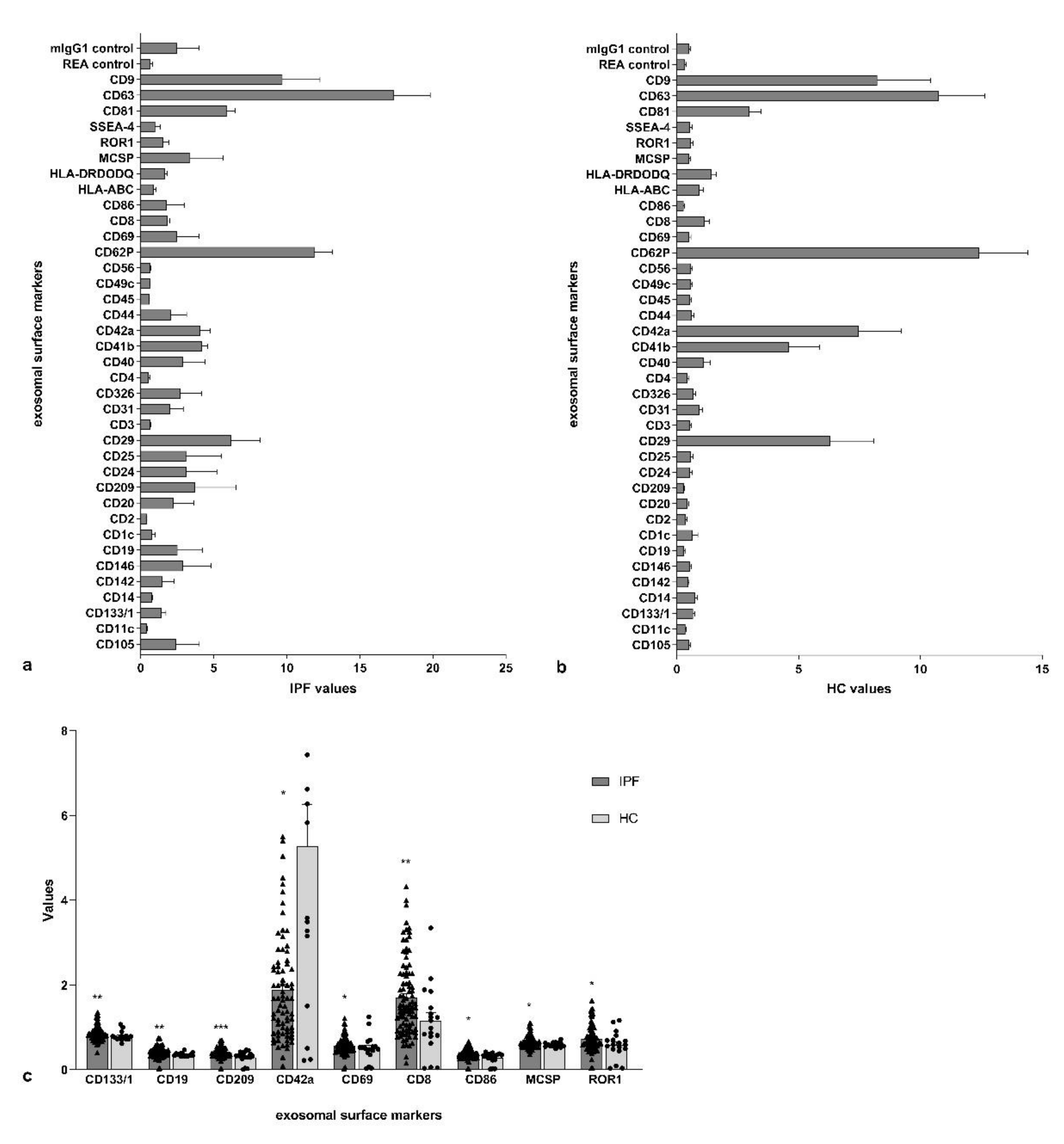
2.6. Multiplex Surface Marker Analysis
2.7. Statistical Analysis
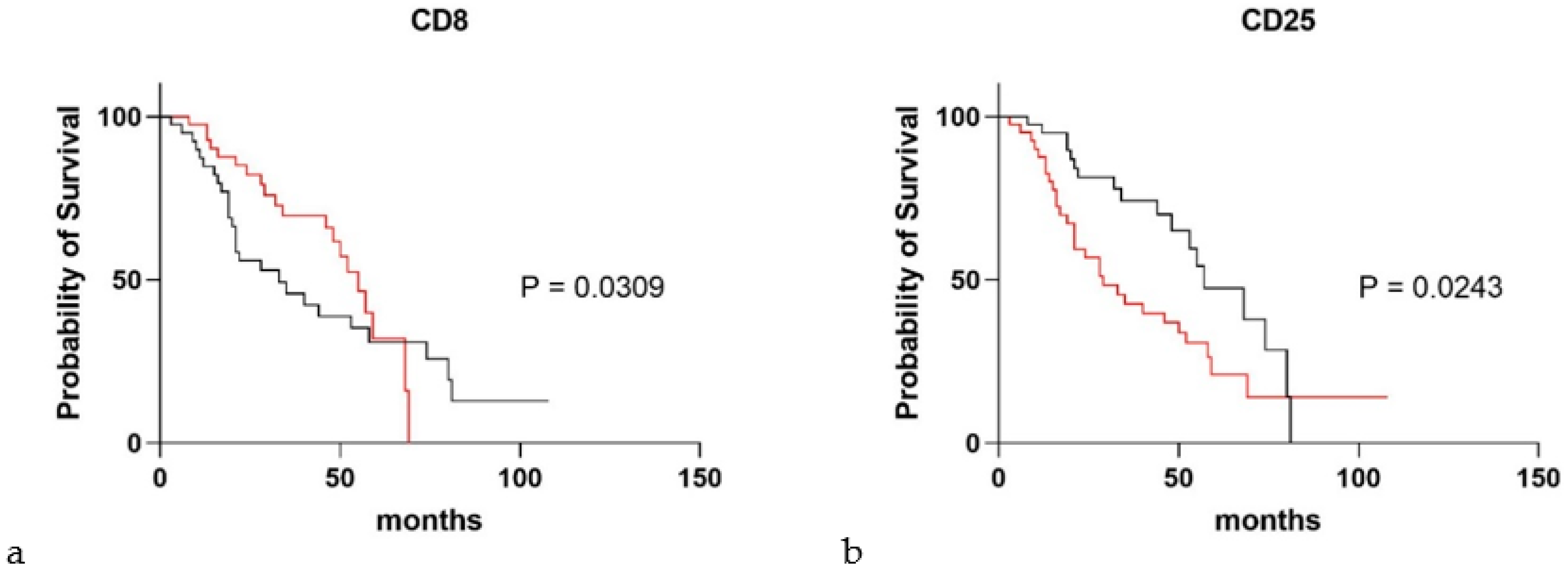
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mittelbrunn, M.; Sánchez-Madrid, F. Intercellular Communication: Diverse Structures for Exchange of Genetic Information. Nat. Rev. Mol. Cell Biol. 2012, 13, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Cocucci, E.; Meldolesi, J. Ectosomes and Exosomes: Shedding the Confusion between Extracellular Vesicles. Trends Cell Biol. 2015, 25, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Lötvall, J.; Hill, A.F.; Hochberg, F.; Buzás, E.I.; Di Vizio, D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal Experimental Requirements for Definition of Extracellular Vesicles and Their Functions: A Position Statement from the International Society for Extracellular Vesicles. J. Extracell Vesicles 2014, 3, 26913. [Google Scholar] [CrossRef] [PubMed]
- Makiguchi, T.; Yamada, M.; Yoshioka, Y.; Sugiura, H.; Koarai, A.; Chiba, S.; Fujino, N.; Tojo, Y.; Ota, C.; Kubo, H.; et al. Serum Extracellular Vesicular MiR-21-5p Is a Predictor of the Prognosis in Idiopathic Pulmonary Fibrosis. Respir. Res. 2016, 17, 110. [Google Scholar] [CrossRef]
- Tan, D.B.A.; Armitage, J.; Teo, T.-H.; Ong, N.E.; Shin, H.; Moodley, Y.P. Elevated Levels of Circulating Exosome in COPD Patients Are Associated with Systemic Inflammation. Respir. Med. 2017, 132, 261–264. [Google Scholar] [CrossRef]
- Kadota, T.; Yoshioka, Y.; Fujita, Y.; Kuwano, K.; Ochiya, T. Extracellular Vesicles in Lung Cancer-From Bench to Bedside. Semin. Cell Dev. Biol. 2017, 67, 39–47. [Google Scholar] [CrossRef]
- Taverna, S.; Giallombardo, M.; Gil-Bazo, I.; Carreca, A.P.; Castiglia, M.; Chacártegui, J.; Araujo, A.; Alessandro, R.; Pauwels, P.; Peeters, M.; et al. Exosomes Isolation and Characterization in Serum Is Feasible in Non-Small Cell Lung Cancer Patients: Critical Analysis of Evidence and Potential Role in Clinical Practice. Oncotarget 2016, 7, 28748–28760. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Lanzarone, N.; Antonietta Mazzei, M.; Alonzi, V.; Sestini, P.; Bargagli, E. Serum KL-6 Levels in Pulmonary Langerhans’ Cell Histiocytosis. Eur. J. Clin. Investig. 2020, e13242. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Carleo, A.; Cameli, P.; Bergantini, L.; Perrone, A.; Vietri, L.; Lanzarone, N.; Vagaggini, C.; Sestini, P.; Bargagli, E. BAL Biomarkers’ Panel for Differential Diagnosis of Interstitial Lung Diseases. Clin. Exp. Med. 2020, 20, 207–216. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Vietri, L.; Lanzarone, N.; Alonzi, V.; Pieroni, M.; Refini, R.M.; Sestini, P.; Bonella, F.; et al. Krebs von Den Lungen-6 as a Biomarker for Disease Severity Assessment in Interstitial Lung Disease: A Comprehensive Review. Biomark Med. 2020, 14, 665–674. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Pieroni, M.; Refini, R.M.; Sestini, P.; Bargagli, E. Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy. Cancers 2021, 13. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Torricelli, E.; Cameli, P.; Lavorini, F.; Pieroni, M.; Refini, R.M.; Sestini, P.; Bargagli, E. Systematic Review and Metanalysis of Oncomarkers in IPF Patients and Serial Changes of Oncomarkers in a Prospective Italian Real-Life Case Series. Cancers 2021, 13. [Google Scholar] [CrossRef]
- Bergantini, L.; d’Alessandro, M.; Vietri, L.; Rana, G.D.; Cameli, P.; Acerra, S.; Sestini, P.; Bargagli, E. Utility of Serological Biomarker’ Panels for Diagnostic Accuracy of Interstitial Lung Diseases. Immunol. Res. 2020, 68, 414–421. [Google Scholar] [CrossRef]
- Raghu, G.; Remy-Jardin, M.; Myers, J.L.; Richeldi, L.; Ryerson, C.J.; Lederer, D.J.; Behr, J.; Cottin, V.; Danoff, S.K.; Morell, F.; et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2018, 198, e44–e68. [Google Scholar] [CrossRef]
- Rottoli, P.; Bargagli, E. Is Bronchoalveolar Lavage Obsolete in the Diagnosis of Interstitial Lung Disease? Curr. Opin. Pulm. Med. 2003, 9, 418–425. [Google Scholar] [CrossRef]
- Vietri, L.; Cameli, P.; Perruzza, M.; Cekorja, B.; Bergantini, L.; d’Alessandro, M.; Refini, R.M.; Pieroni, M.; Fossi, A.; Bennett, D.; et al. Pirfenidone in Idiopathic Pulmonary Fibrosis: Real-Life Experience in the Referral Centre of Siena. Adv. Respir. Dis. 2020, 14, 1753466620906326. [Google Scholar] [CrossRef]
- Cameli, P.; Refini, R.M.; Bergantini, L.; d’Alessandro, M.; Alonzi, V.; Magnoni, C.; Rottoli, P.; Sestini, P.; Bargagli, E. Long-Term Follow-Up of Patients With Idiopathic Pulmonary Fibrosis Treated With Pirfenidone or Nintedanib: A Real-Life Comparison Study. Front. Mol. Biosci. 2020, 7, 581828. [Google Scholar] [CrossRef]
- Culver, B.H.; Graham, B.L.; Coates, A.L.; Wanger, J.; Berry, C.E.; Clarke, P.K.; Hallstrand, T.S.; Hankinson, J.L.; Kaminsky, D.A.; MacIntyre, N.R.; et al. Recommendations for a Standardized Pulmonary Function Report. An Official American Thoracic Society Technical Statement. Am. J. Respir. Crit. Care Med. 2017, 196, 1463–1472. [Google Scholar] [CrossRef] [PubMed]
- MACSPlex Exosome Kit, Human | Multiplex Assays | Kits and Support Reagents | MACS Flow Cytometry | Products | Miltenyi Biotec | Suomi. Available online: https://www.miltenyibiotec.com/FI-en/products/macsplex-exosome-kit-human.html#gref (accessed on 22 March 2021).
- Carleo, A.; Landi, C.; Prasse, A.; Bergantini, L.; d’Alessandro, M.; Cameli, P.; Janciauskiene, S.; Rottoli, P.; Bini, L.; Bargagli, E. Proteomic Characterization of Idiopathic Pulmonary Fibrosis Patients: Stable versus Acute Exacerbation. Monaldi Arch. Chest Dis. 2020, 90. [Google Scholar] [CrossRef]
- Bargagli, E.; Cameli, P.; Carleo, A.; Refini, R.M.; Bergantini, L.; d’Alessandro, M.; Vietri, L.; Perillo, F.; Volterrani, L.; Rottoli, P.; et al. The Effect of Cigarette Smoking on Bronchoalveolar Lavage Protein Profiles from Patients with Different Interstitial Lung Diseases. Panminerva Med. 2019. [Google Scholar] [CrossRef]
- Landi, C.; Bargagli, E.; Carleo, A.; Bianchi, L.; Gagliardi, A.; Prasse, A.; Perari, M.G.; Refini, R.M.; Bini, L.; Rottoli, P. A System Biology Study of BALF from Patients Affected by Idiopathic Pulmonary Fibrosis (IPF) and Healthy Controls. Proteom. Clin. Appl. 2014, 8, 932–950. [Google Scholar] [CrossRef] [PubMed]
- Andreu, Z.; Yáñez-Mó, M. Tetraspanins in Extracellular Vesicle Formation and Function. Front. Immunol. 2014, 5, 442. [Google Scholar] [CrossRef] [PubMed]
- Komura, K.; Yanaba, K.; Horikawa, M.; Ogawa, F.; Fujimoto, M.; Tedder, T.F.; Sato, S. CD19 Regulates the Development of Bleomycin-Induced Pulmonary Fibrosis in a Mouse Model. Arthritis Rheum. 2008, 58, 3574–3584. [Google Scholar] [CrossRef] [PubMed]
- Todd, N.W.; Scheraga, R.G.; Galvin, J.R.; Iacono, A.T.; Britt, E.J.; Luzina, I.G.; Burke, A.P.; Atamas, S.P. Lymphocyte Aggregates Persist and Accumulate in the Lungs of Patients with Idiopathic Pulmonary Fibrosis. J. Inflamm. Res. 2013, 6, 63–70. [Google Scholar] [CrossRef]
- Asai, Y.; Chiba, H.; Nishikiori, H.; Kamekura, R.; Yabe, H.; Kondo, S.; Miyajima, S.; Shigehara, K.; Ichimiya, S.; Takahashi, H. Aberrant Populations of Circulating T Follicular Helper Cells and Regulatory B Cells Underlying Idiopathic Pulmonary Fibrosis. Respir Res. 2019, 20, 244. [Google Scholar] [CrossRef]
- Cebrián, M.; Yagüe, E.; Rincón, M.; López-Botet, M.; de Landázuri, M.O.; Sánchez-Madrid, F. Triggering of T Cell Proliferation through AIM, an Activation Inducer Molecule Expressed on Activated Human Lymphocytes. J. Exp. Med. 1988, 168, 1621–1637. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Fanetti, M.; Alderighi, L.; Armati, M.; Refini, R.M.; Alonzi, V.; Sestini, P.; Bargagli, E. Immunologic Responses to Antifibrotic Treatment in IPF Patients. Int. Immunopharmacol. 2021, 95, 107525. [Google Scholar] [CrossRef]
- Yamauchi, K.; Kasuya, Y.; Kuroda, F.; Tanaka, K.; Tsuyusaki, J.; Ishizaki, S.; Matsunaga, H.; Iwamura, C.; Nakayama, T.; Tatsumi, K. Attenuation of Lung Inflammation and Fibrosis in CD69-Deficient Mice after Intratracheal Bleomycin. Respir. Res. 2011, 12, 131. [Google Scholar] [CrossRef]
- De Maria, R.; Cifone, M.G.; Trotta, R.; Rippo, M.R.; Festuccia, C.; Santoni, A.; Testi, R. Triggering of Human Monocyte Activation through CD69, a Member of the Natural Killer Cell Gene Complex Family of Signal Transducing Receptors. J. Exp. Med. 1994, 180, 1999–2004. [Google Scholar] [CrossRef]
- Testi, R.; Phillips, J.H.; Lanier, L.L. T Cell Activation via Leu-23 (CD69). J. Immunol. 1989, 143, 1123–1128. [Google Scholar]
- Moretta, A.; Poggi, A.; Pende, D.; Tripodi, G.; Orengo, A.M.; Pella, N.; Augugliaro, R.; Bottino, C.; Ciccone, E.; Moretta, L. CD69-Mediated Pathway of Lymphocyte Activation: Anti-CD69 Monoclonal Antibodies Trigger the Cytolytic Activity of Different Lymphoid Effector Cells with the Exception of Cytolytic T Lymphocytes Expressing T Cell Receptor Alpha/Beta. J. Exp. Med. 1991, 174, 1393–1398. [Google Scholar] [CrossRef]
- McKinney, E.F.; Lee, J.C.; Jayne, D.R.W.; Lyons, P.A.; Smith, K.G.C. T-Cell Exhaustion, Co-Stimulation and Clinical Outcome in Autoimmunity and Infection. Nature 2015, 523, 612–616. [Google Scholar] [CrossRef]
- Desai, B.; Mattson, J.; Paintal, H.; Nathan, M.; Shen, F.; Beaumont, M.; Malinao, M.-C.; Li, Y.; Canfield, J.; Basham, B.; et al. Differential Expression of Monocyte/Macrophage- Selective Markers in Human Idiopathic Pulmonary Fibrosis. Exp. Lung Res. 2011, 37, 227–238. [Google Scholar] [CrossRef]
- Birjandi, S.Z.; Palchevskiy, V.; Xue, Y.Y.; Nunez, S.; Kern, R.; Weigt, S.S.; Lynch, J.P.; Chatila, T.A.; Belperio, J.A. CD4(+)CD25(Hi)Foxp3(+) Cells Exacerbate Bleomycin-Induced Pulmonary Fibrosis. Am. J. Pathol. 2016, 186, 2008–2020. [Google Scholar] [CrossRef]
- Chakraborty, K.; Chatterjee, S.; Bhattacharyya, A. Impact of Treg on Other T Cell Subsets in Progression of Fibrosis in Experimental Lung Fibrosis. Tissue Cell 2018, 53, 87–92. [Google Scholar] [CrossRef]
- Esposito, I.; Perna, F.; Ponticiello, A.; Perrella, M.; Gilli, M.; Sanduzzi, A. Natural Killer Cells in Bal and Peripheral Blood of Patients with Idiopathic Pulmonary Fibrosis (IPF). Int. J. Immunopathol. Pharm. 2005, 18, 541–545. [Google Scholar] [CrossRef]
- Linsley, P.S.; Brady, W.; Grosmaire, L.; Aruffo, A.; Damle, N.K.; Ledbetter, J.A. Binding of the B Cell Activation Antigen B7 to CD28 Costimulates T Cell Proliferation and Interleukin 2 MRNA Accumulation. J. Exp. Med. 1991, 173, 721–730. [Google Scholar] [CrossRef]
- Kaneko, Y.; Kuwano, K.; Kunitake, R.; Kawasaki, M.; Hagimoto, N.; Hara, N. B7-1, B7-2 and Class II MHC Molecules in Idiopathic Pulmonary Fibrosis and Bronchiolitis Obliterans-Organizing Pneumonia. Eur. Respir. J. 2000, 15, 49–55. [Google Scholar]
- Crooks, M.G.; Fahim, A.; Naseem, K.M.; Morice, A.H.; Hart, S.P. Increased Platelet Reactivity in Idiopathic Pulmonary Fibrosis Is Mediated by a Plasma Factor. PLoS ONE 2014, 9, e111347. [Google Scholar] [CrossRef]
- Bozza, F.A.; Shah, A.M.; Weyrich, A.S.; Zimmerman, G.A. Amicus or Adversary: Platelets in Lung Biology, Acute Injury, and Inflammation. Am. J. Respir. Cell Mol. Biol. 2009, 40, 123–134. [Google Scholar] [CrossRef]
- Fahim, A.; Crooks, M.G.; Morice, A.H.; Hart, S.P. Increased Platelet Binding to Circulating Monocytes in Idiopathic Pulmonary Fibrosis. Lung 2014, 192, 277–284. [Google Scholar] [CrossRef]
- Bargagli, E.; Madioni, C.; Bianchi, N.; Refini, R.M.; Cappelli, R.; Rottoli, P. Serum Analysis of Coagulation Factors in IPF and NSIP. Inflammation 2014, 37, 10–16. [Google Scholar] [CrossRef]
- Bergantini, L.; d’Alessandro, M.; Cameli, P.; Carleo, A.; Landi, C.; Vietri, L.; Lanzarone, N.; Pieroni, M.; Sestini, P.; Bargagli, E. Antithrombin III as Predictive Indicator of Survival in IPF Patients Treated with Nintedanib: A Preliminary Study. Intern. Med. J. 2020. [Google Scholar] [CrossRef]
- Bargagli, E.; Olivieri, C.; Nikiforakis, N.; Cintorino, M.; Magi, B.; Perari, M.G.; Vagaggini, C.; Spina, D.; Prasse, A.; Rottoli, P. Analysis of Macrophage Migration Inhibitory Factor (MIF) in Patients with Idiopathic Pulmonary Fibrosis. Respir. Physiol. Neurobiol. 2009, 167, 261–267. [Google Scholar] [CrossRef]
- Tsoutsou, P.G.; Gourgoulianis, K.I.; Petinaki, E.; Mpaka, M.; Efremidou, S.; Maniatis, A.; Molyvdas, P.A. ICAM-1, ICAM-2 and ICAM-3 in the Sera of Patients with Idiopathic Pulmonary Fibrosis. Inflammation 2004, 28, 359–364. [Google Scholar] [CrossRef]
- Bellamri, N.; Morzadec, C.; Joannes, A.; Lecureur, V.; Wollin, L.; Jouneau, S.; Vernhet, L. Alteration of Human Macrophage Phenotypes by the Anti-Fibrotic Drug Nintedanib. Int. Immunopharmacol. 2019, 72, 112–123. [Google Scholar] [CrossRef]
- Yamada, M.; Kubo, H.; Kobayashi, S.; Ishizawa, K.; Numasaki, M.; Ueda, S.; Suzuki, T.; Sasaki, H. Bone Marrow-Derived Progenitor Cells Are Important for Lung Repair after Lipopolysaccharide-Induced Lung Injury. J. Immunol. 2004, 172, 1266–1272. [Google Scholar] [CrossRef]
- Shmelkov, S.V.; Butler, J.M.; Hooper, A.T.; Hormigo, A.; Kushner, J.; Milde, T.; St Clair, R.; Baljevic, M.; White, I.; Jin, D.K.; et al. CD133 Expression Is Not Restricted to Stem Cells, and Both CD133+ and CD133- Metastatic Colon Cancer Cells Initiate Tumors. J. Clin. Investig. 2008, 118, 2111–2120. [Google Scholar] [CrossRef]
- Germano, D.; Blyszczuk, P.; Valaperti, A.; Kania, G.; Dirnhofer, S.; Landmesser, U.; Lüscher, T.F.; Hunziker, L.; Zulewski, H.; Eriksson, U. Prominin-1/CD133+ Lung Epithelial Progenitors Protect from Bleomycin-Induced Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2009, 179, 939–949. [Google Scholar] [CrossRef]
- Cameli, P.; Bergantini, L.; D’Alessandro, M.; Vietri, L.; Refini, R.M.; Pieroni, M.; Sestini, P.; Bargagli, E. Alveolar Nitric Oxide Is Related to Periostin Levels in Idiopathic Pulmonary Fibrosis. Minerva Med. 2020, 111, 324–329. [Google Scholar] [CrossRef]
- Cameli, P.; Bergantini, L.; D’alessandro, M.; Vietri, L.; Cameli, M.; Sestini, P.; Bargagli, E. Serum CD59: A Novel Biomarker of Idiopathic Pulmonary Fibrosis? Panminerva Med. 2020. [Google Scholar] [CrossRef]
- Vietri, L.; d’Alessandro, M.; Bergantini, L.; Carleo, A.; Cameli, P.; Mazzei, M.A.; Sestini, P.; Bargagli, E. Specificity of Serum Amyloid A as a Biomarker of Idiopathic Pulmonary Fibrosis. Intern. Med. J. 2020, 50, 1571–1574. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Lanzarone, N.; Perillo, F.; Perrone, A.; Bargagli, E. BAL and Serum Multiplex Lipid Profiling in Idiopathic Pulmonary Fibrosis and Fibrotic Hypersensitivity Pneumonitis. Life Sci. 2020, 256, 117995. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, M.; Bergantini, L.; Refini, R.M.; Cameli, P.; Perillo, F.; Landi, C.; Icorne, F.; Perrone, A.; Sestini, P.; Bonella, F.; et al. Adiponectin and Leptin Levels in Idiopathic Pulmonary Fibrosis: A New Method for BAL and Serum Assessment. Immunobiology 2020, 225, 151997. [Google Scholar] [CrossRef]
- D’Alessandro, M.; De Vita, E.; Bergantini, L.; Mazzei, M.A.; di Valvasone, S.; Bonizzoli, M.; Peris, A.; Sestini, P.; Bargagli, E.; Bennett, D. Galactin-1, 3 and 9: Potential Biomarkers in Idiopathic Pulmonary Fibrosis and Other Interstitial Lung Diseases. Respir. Physiol. Neurobiol. 2020, 282, 103546. [Google Scholar] [CrossRef] [PubMed]
- Bargagli, E.; Refini, R.M.; d’Alessandro, M.; Bergantini, L.; Cameli, P.; Vantaggiato, L.; Bini, L.; Landi, C. Metabolic Dysregulation in Idiopathic Pulmonary Fibrosis. Int. J. Mol. Sci. 2020, 21. [Google Scholar] [CrossRef]
- Ampofo, E.; Schmitt, B.M.; Menger, M.D.; Laschke, M.W. The Regulatory Mechanisms of NG2/CSPG4 Expression. Cell Mol. Biol. Lett. 2017, 22, 4. [Google Scholar] [CrossRef]
- Hanumegowda, C.; Farkas, L.; Kolb, M. Angiogenesis in Pulmonary Fibrosis: Too Much or Not Enough? Chest 2012, 142, 200–207. [Google Scholar] [CrossRef]
- Landi, C.; Bergantini, L.; Cameli, P.; d’Alessandro, M.; Carleo, A.; Shaba, E.; Rottoli, P.; Bini, L.; Bargagli, E. Idiopathic Pulmonary Fibrosis Serum Proteomic Analysis before and after Nintedanib Therapy. Sci. Rep. 2020, 10, 9378. [Google Scholar] [CrossRef]
- Li, C.; Smith, S.M.; Peinado, N.; Gao, F.; Li, W.; Lee, M.K.; Zhou, B.; Bellusci, S.; Pryhuber, G.S.; Ho, H.-Y.H.; et al. WNT5a-ROR Signaling Is Essential for Alveologenesis. Cells 2020, 9. [Google Scholar] [CrossRef]
- Flaherty, K.R.; Travis, W.D.; Colby, T.V.; Toews, G.B.; Kazerooni, E.A.; Gross, B.H.; Jain, A.; Strawderman, R.L.; Flint, A.; Lynch, J.P.; et al. Histopathologic Variability in Usual and Nonspecific Interstitial Pneumonias. Am. J. Respir. Crit. Care Med. 2001, 164, 1722–1727. [Google Scholar] [CrossRef]
- Serini, G.; Gabbiani, G. Mechanisms of Myofibroblast Activity and Phenotypic Modulation. Exp. Cell Res. 1999, 250, 273–283. [Google Scholar] [CrossRef]
- Vuga, L.J.; Ben-Yehudah, A.; Kovkarova-Naumovski, E.; Oriss, T.; Gibson, K.F.; Feghali-Bostwick, C.; Kaminski, N. WNT5A Is a Regulator of Fibroblast Proliferation and Resistance to Apoptosis. Am. J. Respir. Cell Mol. Biol. 2009, 41, 583–589. [Google Scholar] [CrossRef]
- Monsel, A.; Zhu, Y.-G.; Gudapati, V.; Lim, H.; Lee, J.W. Mesenchymal Stem Cell Derived Secretome and Extracellular Vesicles for Acute Lung Injury and Other Inflammatory Lung Diseases. Expert Opin. Biol. 2016, 16, 859–871. [Google Scholar] [CrossRef]
- Mack, G.S. Trial Set to Test How Stem Cells Heal a Broken Heart. Nat. Med. 2006, 12, 483. [Google Scholar] [CrossRef]
| Parameters | HC (n = 19) | IPF (n = 90) | p Value |
|---|---|---|---|
| Age (median IQR) | 63 (52–65) | 71 (66–75) | 0.3111 |
| Gender, M/F | 12/7 | 69/21 | 0.2911 |
| Smoking habit (never/former) | 10/9 | 42/48 | 0.3102 |
| BMI (kg/m2) | 26 (24–27) | 25 (23–29) | 0.0892 |
| Pulmonary function parameters | |||
| FVC % | 101.1(88.3–107.2) | 68 (59–91) | 0.0238 |
| FEV1 % | 98 (89.1–110.5) | 75 (62–92) | 0.0319 |
| DLCO % | 89.9 (82.3–98.4) | 47 (39–58) | 0.0127 |
| EVs total protein concentrations (mg/mL) | 2.2 (2–2.3) | 2.9 (2.5–3.3) | 0.285 |
| Pirfenidone/Nintedanib | 53/37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
d’Alessandro, M.; Soccio, P.; Bergantini, L.; Cameli, P.; Scioscia, G.; Foschino Barbaro, M.P.; Lacedonia, D.; Bargagli, E. Extracellular Vesicle Surface Signatures in IPF Patients: A Multiplex Bead-Based Flow Cytometry Approach. Cells 2021, 10, 1045. https://doi.org/10.3390/cells10051045
d’Alessandro M, Soccio P, Bergantini L, Cameli P, Scioscia G, Foschino Barbaro MP, Lacedonia D, Bargagli E. Extracellular Vesicle Surface Signatures in IPF Patients: A Multiplex Bead-Based Flow Cytometry Approach. Cells. 2021; 10(5):1045. https://doi.org/10.3390/cells10051045
Chicago/Turabian Styled’Alessandro, Miriana, Piera Soccio, Laura Bergantini, Paolo Cameli, Giulia Scioscia, Maria Pia Foschino Barbaro, Donato Lacedonia, and Elena Bargagli. 2021. "Extracellular Vesicle Surface Signatures in IPF Patients: A Multiplex Bead-Based Flow Cytometry Approach" Cells 10, no. 5: 1045. https://doi.org/10.3390/cells10051045
APA Styled’Alessandro, M., Soccio, P., Bergantini, L., Cameli, P., Scioscia, G., Foschino Barbaro, M. P., Lacedonia, D., & Bargagli, E. (2021). Extracellular Vesicle Surface Signatures in IPF Patients: A Multiplex Bead-Based Flow Cytometry Approach. Cells, 10(5), 1045. https://doi.org/10.3390/cells10051045









