The Use of Nanomedicine to Target Signaling by the PAK Kinases for Disease Treatment
Abstract
1. Introduction
1.1. The PAK Kinases
1.2. PAK Kinases and the Regulation of Cell Survival and Apoptosis
1.3. The PAKs as Potential Therapeutic Targets
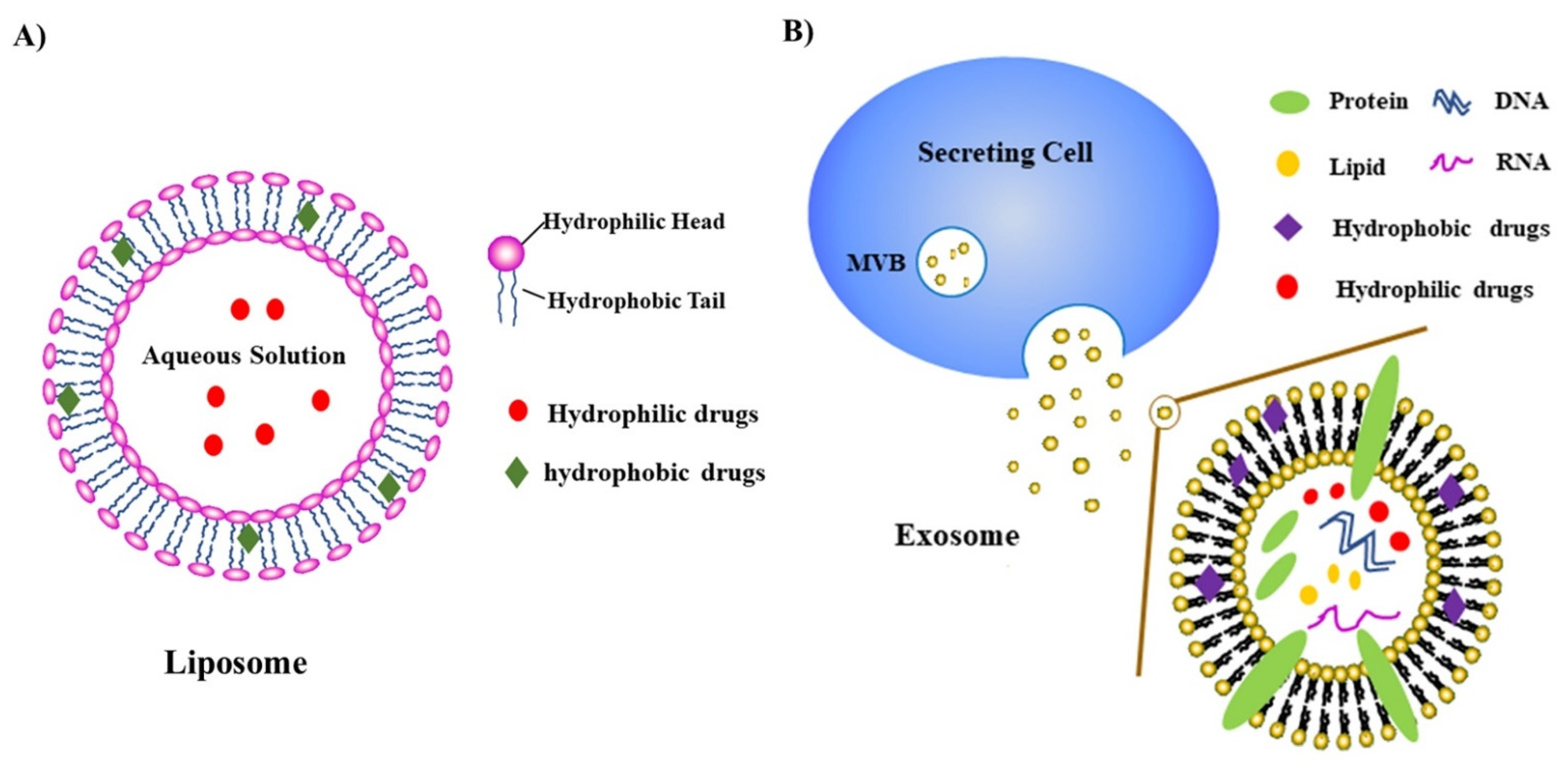
1.4. Nanoparticles/Nanomedicines
1.5. Use of Nanomedicine to Target PAK Kinases
2. Inhibition of Pak1 for the Treatment of Prostate Cancer
3. Inhibition of Pak1 for the Treatment of Triple Negative Breast Cancer
4. Inhibition of PAK4 Prolongs Survival in a Pancreatic Cancer Mouse Model
5. Cocktail Therapy (Combinational Treatment) Delivered by Nanoparticles to Treat Hepatocellular Carcinoma (HCC)
6. Inhibition of Pak4 for the Treatment of Atherosclerosis
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Linet, M.S.; Ries, L.A.G.; Smith, M.A.; Tarone, R.E.; Devesa, S.S. Cancer Surveillance Series: Recent Trends in Childhood Cancer Incidence and Mortality in the United States. J. Natl. Cancer Inst. 1999, 91, 1051–1058. [Google Scholar] [CrossRef]
- Singh, A.; Settleman, J. EMT, cancer stem cells and drug resistance: An emerging axis of evil in the war on cancer. Oncogene 2010, 29, 4741–4751. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Schwind, S.; Yu, B.; Santhanam, R.; Wang, H.; Hoellerbauer, P.; Mims, A.; Klisovic, R.; Walker, A.R.; Chan, K.K.; et al. Targeted Delivery of microRNA-29b by Transferrin-Conjugated Anionic Lipopolyplex Nanoparticles: A Novel Therapeutic Strategy in Acute Myeloid Leukemia. Clin. Cancer Res. 2013, 19, 2355–2367. [Google Scholar] [CrossRef] [PubMed]
- Najahi-Missaoui, W.; Quach, N.D.; Jenkins, A.; Dabke, I.; Somanath, P.R.; Cummings, B.S. Effect of P21-activated kinase 1 (PAK-1) inhibition on cancer cell growth, migration, and invasion. Pharmacol. Res. Perspect. 2019, 7, e00518. [Google Scholar] [CrossRef]
- Rane, C.K.; Minden, A. P21 activated kinase signaling in cancer. Semin. Cancer Biol. 2019, 54, 40–49. [Google Scholar] [CrossRef]
- Senapedis, W.; Crochiere, M.; Baloglu, E.; Landesman, Y. Therapeutic potential of targeting PAK signaling. Anti-Cancer Agents Med. Chem. (Former. Curr. Med. Chem.-Anti-Cancer Agents) 2016, 16, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Yépez, E.A.; Saldívar-Cerón, H.I.; Villamar-Cruz, O.; Pérez-Plasencia, C.; Arias-Romero, L.E. p21 Activated kinase 1: Nuclear activity and its role during DNA damage repair. DNA Repair 2018, 65, 42–46. [Google Scholar] [CrossRef]
- Nekrasova, T.; Minden, A. Role for p21-activated kinase PAK4 in development of the mammalian heart. Transgenic Res. 2011, 21, 797–811. [Google Scholar] [CrossRef]
- Abo, A.; Qu, J.; Cammarano, M.S.; Dan, C.; Fritsch, A.; Baud, V.; Belisle, B.; Minden, A. PAK4, a novel effector for Cdc42Hs, is implicated in the reorganization of the actin cytoskeleton and in the formation of filopodia. EMBO J. 1998, 17, 6527–6540. [Google Scholar] [CrossRef] [PubMed]
- Cordover, E.; Wei, J.; Patel, C.; Shan, N.L.; Gionco, J.; Sargsyan, D.; Wu, R.; Cai, L.; Kong, A.-N.T.; Jacinto, E.; et al. KPT-9274, an Inhibitor of PAK4 and NAMPT, Leads to Downregulation of mTORC2 in Triple Negative Breast Cancer Cells. Chem. Res. Toxicol. 2020, 33, 482–491. [Google Scholar] [CrossRef] [PubMed]
- Gnesutta, N.; Qu, J.; Minden, A. The Serine/Threonine Kinase PAK4 Prevents Caspase Activation and Protects Cells from Apoptosis. J. Biol. Chem. 2001, 276, 14414–14419. [Google Scholar] [CrossRef] [PubMed]
- Cotteret, S.; Jaffer, Z.M.; Beeser, A.; Chernoff, J. p21-Activated Kinase 5 (Pak5) Localizes to Mitochondria and Inhibits Apoptosis by Phosphorylating BAD. Mol. Cell. Biol. 2003, 23, 5526–5539. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, X.; Carr, H.S.; Dan, I.; Ruvolo, P.P.; Frost, J.A. p21 activated kinase 5 activates Raf-1 and targets it to mitochondria. J. Cell. Biochem. 2008, 105, 167–175. [Google Scholar] [CrossRef]
- Gnesutta, N.; Minden, A. Death Receptor-Induced Activation of Initiator Caspase 8 Is Antagonized by Serine/Threonine Kinase PAK4. Mol. Cell. Biol. 2003, 23, 7838–7848. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.H.; Frost, J.A. Phosphorylation of Raf-1 by p21-activated Kinase 1 and Src Regulates Raf-1 Autoinhibition. J. Biol. Chem. 2003, 278, 11221–11226. [Google Scholar] [CrossRef]
- King, A.J.; Sun, H.; Diaz, B.; Barnard, D.; Miao, W.; Bagrodia, S.; Marshall, M.S. The protein kinase Pak3 positively regulates Raf-1 activity through phosphorylation of serine 338. Nature 1998, 396, 180–183. [Google Scholar] [CrossRef]
- Campbell, H.; Salvi, A.M.; O’Brien, T.; Superfine, R.; DeMali, K.A. PAK2 links cell survival to mechanotransduction and metabolism. J. Cell Biol. 2019, 218, 1958–1971. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Huang, A.; Poplawski, A.; DiPino, F.; Traugh, J.A.; Ling, J. PAK2 activated by Cdc42 and caspase 3 mediates different cellular responses to oxidative stress-induced apoptosis. Biochim. Biophys. Acta (BBA) Bioenerg. 2020, 1867, 118645. [Google Scholar] [CrossRef]
- Khare, V.; Dammann, K.; Asboth, M.; Krnjic, A.; Jambrich, M.; Gasche, C. Overexpression of PAK1 Promotes Cell Survival in Inflammatory Bowel Diseases and Colitis-associated Cancer. Inflamm. Bowel Dis. 2015, 21, 287–296. [Google Scholar] [CrossRef]
- Furnari, M.A.; Jobes, M.L.; Nekrasova, T.; Minden, A.; Wagner, G.C. Functional Deficits in Pak5, Pak6 and Pak5/Pak6 Knockout Mice. PLoS ONE 2013, 8, e61321. [Google Scholar] [CrossRef]
- Siu, M.K.Y.; Wong, E.S.Y.; Chan, H.Y.; Kong, D.S.H.; Woo, N.W.S.; Tam, K.F.; Ngan, H.Y.S.; Chan, Q.K.Y.; Chan, D.C.W.; Chan, K.Y.K.; et al. Differential expression and phosphorylation of Pak1 and Pak2 in ovarian cancer: Effects on prognosis and cell invasion. Int. J. Cancer 2009, 127, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Luo, C.; Abukiwan, A.; Wang, G.; He, J.; Huang, L.; Weber, C.E.M.; Lv, N.; Xiao, X.; Eichmüller, S.B.; et al. miR-137 inhibits proliferation of melanoma cells by targeting PAK2. Exp. Dermatol. 2015, 24, 947–952. [Google Scholar] [CrossRef]
- Li, Q.; Wu, X.; Guo, L.; Shi, J.; Li, J. MicroRNA-7-5p induces cell growth inhibition, cell cycle arrest and apoptosis by targeting PAK2 in non-small cell lung cancer. FEBS Open Bio 2019, 9, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Byrne, K.M.; Monsefi, N.; Dawson, J.; Degasperi, A.; Wills, J.; Volinsky, N.; Dobrzyński, M.; Birtwistle, M.; Tsyganov, M.A.; Kiyatkin, A.; et al. Bistability in the Rac1, PAK, and RhoA Signaling Network Drives Actin Cytoskeleton Dynamics and Cell Motility Switches. Cell Syst. 2016, 2, 38–48. [Google Scholar] [CrossRef]
- Dan, C.; Nath, N.; Liberto, M.; Minden, A. PAK5, a New Brain-Specific Kinase, Promotes Neurite Outgrowth in N1E-115 Cells. Mol. Cell. Biol. 2002, 22, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, J.; Crawford, J.J.; Hoeflich, K.P.; Wang, W. Inhibitors of p21-Activated Kinases (PAKs). J. Med. Chem. 2015, 58, 111–129. [Google Scholar] [CrossRef]
- Feng, Q.; Albeck, J.G.; Cerione, R.A.; Yang, W. Regulation of the Cool/Pix Proteins: Key binding partners of the Cdc42/Rac targets, the p21-activated kinases. J. Biol. Chem. 2002, 277, 5644–5650. [Google Scholar] [CrossRef]
- Kamai, T.; Shirataki, H.; Nakanishi, K.; Furuya, N.; Kambara, T.; Abe, H.; Oyama, T.; Yoshida, K.-I. Increased Rac1 activity and Pak1 overexpression are associated with lymphovascular invasion and lymph node metastasis of upper urinary tract cancer. BMC Cancer 2010, 10, 164. [Google Scholar] [CrossRef]
- Aboukameel, A.; Muqbil, I.; Senapedis, W.; Baloglu, E.; Landesman, Y.; Shacham, S.; Kauffman, M.; Philip, P.A.; Mohammad, R.M.; Azmi, A.S. Novel p21-Activated Kinase 4 (PAK4) Allosteric Modulators Overcome Drug Resistance and Stemness in Pancreatic Ductal Adenocarcinoma. Mol. Cancer Ther. 2017, 16, 76–87. [Google Scholar] [CrossRef]
- Mpilla, G.B.; Uddin, H.; Al-Hallak, M.N.; Aboukameel, A.; Li, Y.; Kim, S.H.; Beydoun, R.; Dyson, G.; Baloglu, E.; Senapedis, W.T.; et al. PAK4-NAMPT Dual Inhibition Sensitizes Pancreatic Neuroendocrine Tumors to Everolimus. Mol. Cancer Ther. 2021, 20, 1836–1845. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.C.; Gierke, S.; Pitt, C.; Sagolla, M.; Cheng, C.K.; Zhou, W.; Jubb, A.M.; Strickland, L.; Schmidt, M.; Duron, S.G.; et al. Small molecule inhibition of group I p21-activated kinases in breast cancer induces apoptosis and potentiates the activity of microtubule stabilizing agents. Breast Cancer Res. 2015, 17, 59. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, Y.; Chen, J.; Yang, Y.; Zhu, L.; Zhao, J.; Yang, Y.; Cai, X.; Hu, C.; Rosell, R.; et al. Identification of a novel PAK1 inhibitor to treat pancreatic cancer. Acta Pharm. Sin. B 2020, 10, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Licciulli, S.; Maksimoska, J.; Zhou, C.; Troutman, S.; Kota, S.; Liu, Q.; Duron, S.; Campbell, D.; Chernoff, J.; Field, J.; et al. FRAX597, a Small Molecule Inhibitor of the p21-activated Kinases, Inhibits Tumorigenesis of Neurofibromatosis Type 2 (NF2)-associated Schwannomas. J. Biol. Chem. 2013, 288, 29105–29114. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, X.; Xu, L.; Tao, Y.; Yang, C.; Chen, X.; Fang, F.; Wu, Y.; Ding, X.; Zhao, H.; et al. Inhibition of neuroblastoma proliferation by PF-3758309, a small-molecule inhibitor that targets p21-activated kinase 4. Oncol. Rep. 2017, 38, 2705–2716. [Google Scholar] [CrossRef]
- Fu, X.; Feng, J.; Zeng, D.; Ding, Y.; Yu, C.; Yang, B. PAK4 confers cisplatin resistance in gastric cancer cells via PI3K/Akt- and MEK/ERK-dependent pathways. Biosci. Rep. 2014, 34, e00094. [Google Scholar] [CrossRef]
- Mohammad, R.M.; Li, Y.; Muqbil, I.; Aboukameel, A.; Senapedis, W.; Baloglu, E.; Landesman, Y.; Philip, P.A.; Azmi, A.S. Targeting Rho GTPase effector p21 activated kinase 4 (PAK4) suppresses p-Bad-microRNA drug resistance axis leading to inhibition of pancreatic ductal adenocarcinoma proliferation. Small GTPases 2017, 10, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, T.; Zhu, J.; Li, Z.; Lorent, J.; Zhao, C.; Dahlman-Wright, K.; Strömblad, S. p21-activated kinase group II small compound inhibitor GNE-2861 perturbs estrogen receptor alpha signaling and restores tamoxifen-sensitivity in breast cancer cells. Oncotarget 2015, 6, 43853–43868. [Google Scholar] [CrossRef] [PubMed]
- Najahi-Missaoui, W.; Quach, N.D.; Somanath, P.R.; Cummings, B.S. Liposomes Targeting P21 Activated Kinase-1 (PAK-1) and Selective for Secretory Phospholipase A2 (sPLA2) Decrease Cell Viability and Induce Apoptosis in Metastatic Triple-Negative Breast Cancer Cells. Int. J. Mol. Sci. 2020, 21, 9396. [Google Scholar] [CrossRef]
- Kuželová, K.; Grebeňová, D.; Holoubek, A.; Röselová, P.; Obr, A. Group I PAK Inhibitor IPA-3 Induces Cell Death and Affects Cell Adhesivity to Fibronectin in Human Hematopoietic Cells. PLoS ONE 2014, 9, e92560. [Google Scholar] [CrossRef]
- Lin, H.; Rothe, K.; Chen, M.; Wu, A.; Babaian, A.; Yen, R.; Zeng, J.; Ruschmann, J.; Petriv, O.I.; O’Neill, K.; et al. The miR-185/PAK6 axis predicts therapy response and regulates survival of drug-resistant leukemic stem cells in CML. Blood 2020, 136, 596–609. [Google Scholar] [CrossRef]
- Knippler, C.M.; Saji, M.; Rajan, N.; Porter, K.; La Perle, K.M.D.; Ringel, M.D. MAPK- and AKT-activated thyroid cancers are sensitive to group I PAK inhibition. Endocr.-Relat. Cancer 2019, 26, 699–712. [Google Scholar] [CrossRef]
- Abu Aboud, O.; Chen, C.-H.; Senapedis, W.; Baloglu, E.; Argueta, C.; Weiss, R.H. Dual and Specific Inhibition of NAMPT and PAK4 by KPT-9274 Decreases Kidney Cancer Growth. Mol. Cancer Ther. 2016, 15, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Rane, C.; Senapedis, W.; Baloglu, E.; Landesman, Y.; Crochiere, M.; Das-Gupta, S.; Minden, A. A novel orally bioavailable compound KPT-9274 inhibits PAK4, and blocks triple negative breast cancer tumor growth. Sci. Rep. 2017, 7, srep42555. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Duan, X.; Dong, J.; Lu, Q.; Zhou, J.; Zhao, Z.; Bao, J.; Jing, Z. p21-Activated Kinase 4 Promotes Intimal Hyperplasia and Vascular Smooth Muscle Cells Proliferation during Superficial Femoral Artery Restenosis after Angioplasty. BioMed Res. Int. 2017, 2017, 5296516. [Google Scholar] [CrossRef]
- Knaus, U.G.; Bokoch, G.M. The p21Rac/Cdc42-activated kinases (PAKs). Int. J. Biochem. Cell Biol. 1998, 30, 857–862. [Google Scholar] [CrossRef]
- Pallesen, L.T.; Gustafsen, C.; Cramer, J.F.; Petersen, S.V.; Thirup, S.S.; Madsen, P.; Petersen, C.M. PAK Kinases Target Sortilin and Modulate Its Sorting. Mol. Cell. Biol. 2020, 40, e00411-19. [Google Scholar] [CrossRef]
- Taglieri, D.M.; Ushio-Fukai, M.; Monasky, M.M. P21-activated kinase in inflammatory and cardiovascular disease. Cell. Signal. 2014, 26, 2060–2069. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.D.; Jaffer, Z.M.; Park, S.-J.; Burgin, S.; Hofmann, C.; Sells, M.A.; Chen, S.; Derr-Yellin, E.; Michels, E.G.; McDaniel, A.; et al. p21-activated kinase regulates mast cell degranulation via effects on calcium mobilization and cytoskeletal dynamics. Blood 2009, 113, 2695–2705. [Google Scholar] [CrossRef]
- McFawn, P.K.; Shen, L.; Vincent, S.G.; Mak, A.; Van Eyk, J.E.; Fisher, J.T. Calcium-independent contraction and sensitization of airway smooth muscle by p21-activated protein kinase. Am. J. Physiol. Lung Cell. Mol. Physiol. 2003, 284, 863–870. [Google Scholar] [CrossRef]
- Civiero, L.; Cirnaru, M.D.; Beilina, A.; Rodella, U.; Russo, I.; Belluzzi, E.; Lobbestael, E.; Reyniers, L.; Hondhamuni, G.; Lewis, P.; et al. Leucine-rich repeat kinase 2 interacts with p21-activated kinase 6 to control neurite complexity in mammalian brain. J. Neurochem. 2015, 135, 1242–1256. [Google Scholar] [CrossRef]
- Luo, S.; Mizuta, H.; Rubinsztein, D.C. p21-activated kinase 1 promotes soluble mutant huntingtin self-interaction and enhances toxicity. Hum. Mol. Genet. 2007, 17, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wang, Y.; Fan, T.; Zeng, C.; Sun, Z.S. The p21-activated kinases in neural cytoskeletal remodeling and related neurological disorders. Protein Cell 2020. [Google Scholar] [CrossRef]
- Ma, Q.-L.; Yang, F.; Calon, F.; Ubeda, O.J.; Hansen, J.E.; Weisbart, R.H.; Beech, W.; Frautschy, S.A.; Cole, G.M. p21-activated Kinase-aberrant Activation and Translocation in Alzheimer Disease Pathogenesis. J. Biol. Chem. 2008, 283, 14132–14143. [Google Scholar] [CrossRef] [PubMed]
- Lauterborn, J.C.; Cox, C.D.; Chan, S.W.; Vanderklish, P.W.; Lynch, G.; Gall, C.M. Synaptic actin stabilization protein loss in Down syndrome and Alzheimer disease. Brain Pathol. 2019, 30, 319–331. [Google Scholar] [CrossRef]
- Ding, I.; Ostrowska-Podhorodecka, Z.; Lee, W.; Liu, R.S.; Carneiro, K.; Janmey, P.A.; McCulloch, C.A. Cooperative roles of PAK1 and filamin A in regulation of vimentin assembly and cell extension formation. Biochim. Biophys. Acta (BBA) Bioenerg. 2020, 1867, 118739. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.C.; Sanders, L.C.; Bokoch, G.M.; Gill, G.N. Activation of LIM-kinase by Pak1 couples Rac/Cdc42 GTPase signalling to actin cytoskeletal dynamics. Nature 1999, 1, 253–259. [Google Scholar] [CrossRef]
- Xu, L.; Faruqu, F.N.; Lim, Y.M.; Lim, K.Y.; Liam-Or, R.; Walters, A.A.; Lavender, P.; Fear, D.; Wells, C.; Wang, J.T.-W.; et al. Exosome-mediated RNAi of PAK4 prolongs survival of pancreatic cancer mouse model after loco-regional treatment. Biomaterials 2020, 264, 120369. [Google Scholar] [CrossRef]
- Means, N.; Elechalawar, C.K.; Chen, W.R.; Bhattacharya, R.; Mukherjee, P. Revealing macropinocytosis using nanoparticles. Mol. Asp. Med. 2021, 100993. [Google Scholar] [CrossRef]
- Zhu, J.; Huang, Q. Nanoencapsulation of functional food ingredients. Adv. Food Nutr. Res. 2019, 88, 129–165. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-K. Liposomes for enhanced bioavailability of water-insoluble drugs: In vivo evidence and recent approaches. Pharmaceutics 2020, 12, 264. [Google Scholar] [CrossRef]
- Leung, A.W.Y.; Amador, C.; Wang, L.C.; Mody, U.V.; Bally, M.B. What Drives Innovation: The Canadian Touch on Liposomal Therapeutics. Pharmaceutics 2019, 11, 124. [Google Scholar] [CrossRef]
- Patel, M.M.; Patel, B.M. Crossing the Blood–Brain Barrier: Recent Advances in Drug Delivery to the Brain. CNS Drugs 2017, 31, 109–133. [Google Scholar] [CrossRef] [PubMed]
- Lucchinetti, E.; Lou, P.; Wawrzyniak, P.; Wawrzyniak, M.; Scharl, M.; Holtzhauer, G.; Krämer, S.D.; Hersberger, M.; Rogler, G.; Zaugg, M. Novel Strategies to Prevent Total Parenteral Nutrition-Induced Gut and Liver Inflammation, and Adverse Metabolic Outcomes. Mol. Nutr. Food Res. 2021, 65, e1901270. [Google Scholar] [CrossRef]
- McClements, D.J. Nanoemulsion-based oral delivery systems for lipophilic bioactive components: Nutraceuticals and pharmaceuticals. Ther. Deliv. 2013, 4, 841–857. [Google Scholar] [CrossRef] [PubMed]
- Ladefoged, K.; Jarnum, S. Long-term parenteral nutrition. BMJ 1978, 2, 262–266. [Google Scholar] [CrossRef][Green Version]
- Hörmann, K.; Zimmer, A. Drug delivery and drug targeting with parenteral lipid nanoemulsions—A review. J. Control. Release 2016, 223, 85–98. [Google Scholar] [CrossRef]
- Joshi, M.D.; Patravale, V.; Prabhu, R. Polymeric nanoparticles for targeted treatment in oncology: Current insights. Int. J. Nanomed. 2015, 10, 1001–1018. [Google Scholar] [CrossRef]
- Czajkowska-Kośnik, A.; Szekalska, M.; Winnicka, K. Nanostructured lipid carriers: A potential use for skin drug delivery systems. Pharmacol. Rep. 2018, 71, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.; Cai, G.; Li, Q. Exosomes as Actively Targeted Nanocarriers for Cancer Therapy. Int. J. Nanomed. 2020, 15, 4257–4273. [Google Scholar] [CrossRef]
- Xie, M. Phospholipids. Encycl. Food Chem. 2019, 214–217. [Google Scholar] [CrossRef]
- Patil, Y.P.; Jadhav, S. Novel methods for liposome preparation. Chem. Phys. Lipids 2014, 177, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Akbarzadeh, A.; Rezaei-Sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N.; Hanifehpour, Y.; Samiei, M.; Kouhi, M.; Nejati-Koshki, K. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 102. [Google Scholar] [CrossRef]
- Yong, T.; Zhang, X.; Bie, N.; Zhang, H.; Zhang, X.; Li, F.; Hakeem, A.; Hu, J.; Gan, L.; Santos, H.A.; et al. Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy. Nat. Commun. 2019, 10, 3838. [Google Scholar] [CrossRef]
- Bunggulawa, E.J.; Wang, W.; Yin, T.; Wang, N.; Durkan, C.; Wang, Y.; Wang, G. Recent advancements in the use of exosomes as drug delivery systems. J. Nanobiotechnol. 2018, 16, 81. [Google Scholar] [CrossRef]
- Fu, S.; Wang, Y.; Xia, X.; Zheng, J.C. Exosome engineering: Current progress in cargo loading and targeted delivery. NanoImpact 2020, 20, 100261. [Google Scholar] [CrossRef]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR effect: Unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv. Drug Deliv. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef]
- Bahrami, B.; Farsangi, M.H.; Mohammadi, H.; Anvari, E.; Ghalamfarsa, G.; Yousefi, M.; Jadidi-Niaragh, F. Nanoparticles and targeted drug delivery in cancer therapy. Immunol. Lett. 2017, 190, 64–83. [Google Scholar] [CrossRef]
- Ferreira, D.D.S.; Lopes, S.C.D.A.; Franco, M.S.; Oliveira, M.C. pH-sensitive liposomes for drug delivery in cancer treatment. Ther. Deliv. 2013, 4, 1099–1123. [Google Scholar] [CrossRef] [PubMed]
- Jose, A.; Ninave, K.M.; Karnam, S.; Venuganti, V.V.K. Temperature-sensitive liposomes for co-delivery of tamoxifen and imatinib for synergistic breast cancer treatment. J. Liposome Res. 2019, 29, 153–162. [Google Scholar] [CrossRef]
- Hu, Y.; Gong, X.; Zhang, J.; Chen, F.; Fu, C.; Li, P.; Zou, L.; Zhao, G. Activated Charge-Reversal Polymeric Nano-System: The Promising Strategy in Drug Delivery for Cancer Therapy. Polymers 2016, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Xu, Y.; Yang, W.; Niu, P.; Li, X.; Chen, Y.; Li, Z.; Liu, Y.; An, Y.; Liu, Y.; et al. Investigating the EPR effect of nanomedicines in human renal tumors via ex vivo perfusion strategy. Nano Today 2020, 35, 100970. [Google Scholar] [CrossRef]
- Lewinski, N.; Colvin, V.; Drezek, R. Cytotoxicity of Nanoparticles. Small 2008, 4, 26–49. [Google Scholar] [CrossRef]
- Wells, C.M.; Jones, G.E. The emerging importance of group II PAKs. Biochem. J. 2010, 425, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Hu, Q.; Wu, W.; Wang, M.; Huang, J.; Zhao, X.; Tang, G.; Liang, T. Tumor-triggered personalized microRNA cocktail therapy for hepatocellular carcinoma. Biomater. Sci. 2020, 8, 6579–6591. [Google Scholar] [CrossRef] [PubMed]
- Radu, M.; Semenova, G.; Kosoff, R.; Chernoff, J. PAK signalling during the development and progression of cancer. Nat. Rev. Cancer 2014, 14, 13–25. [Google Scholar] [CrossRef]
- Al-Azayzih, A.; Missaoui, W.N.; Cummings, B.S.; Somanath, P.R. Liposome-mediated delivery of the p21 activated kinase-1 (PAK-1) inhibitor IPA-3 limits prostate tumor growth in vivo. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 1231–1239. [Google Scholar] [CrossRef] [PubMed]
- Salvi, V.R.; Pawar, P. Nanostructured lipid carriers (NLC) system: A novel drug targeting carrier. J. Drug Deliv. Sci. Technol. 2019, 51, 255–267. [Google Scholar] [CrossRef]
- Tang, W.; Fan, W.; Lau, J.; Deng, L.; Shen, Z.; Chen, X. Emerging blood–brain-barrier-crossing nanotechnology for brain cancer theranostics. Chem. Soc. Rev. 2019, 48, 2967–3014. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Q.; Peng, S.; Yao, K.; Chen, J.; Tao, Y.; Gao, Z.; Wang, F.; Li, H.; Cai, W.; et al. The metastatic promoter DEPDC1B induces epithelial-mesenchymal transition and promotes prostate cancer cell proliferation via Rac1-PAK1 signaling. Clin. Transl. Med. 2020, 10, e191. [Google Scholar] [CrossRef]
- Verma, A.; Najahi-Missaoui, W.; Cummings, B.S.; Somanath, P.R. Sterically stabilized liposomes targeting P21 (RAC1) activated kinase-1 and secreted phospholipase A2 suppress prostate cancer growth and metastasis. Oncol. Lett. 2020, 20, 179. [Google Scholar] [CrossRef]
- Verma, A.; Artham, S.; Alwhaibi, A.; Adil, M.S.; Cummings, B.S.; Somanath, P.R. PAK1 inhibitor IPA-3 mitigates metastatic prostate cancer-induced bone remodeling. Biochem. Pharmacol. 2020, 177, 113943. [Google Scholar] [CrossRef] [PubMed]
- Al-Azayzih, A.; Gao, F.; Somanath, P.R. P21 activated kinase-1 mediates transforming growth factor β1-induced prostate cancer cell epithelial to mesenchymal transition. Biochim. Biophys. Acta (BBA) Bioenerg. 2015, 1853, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.J.; Kim, E.H.; Roy, A.; Kim, J.-H. Evidence for a Novel Mechanism of the PAK1 Interaction with the Rho-GTPases Cdc42 and Rac. PLoS ONE 2013, 8, e71495. [Google Scholar] [CrossRef]
- Mock, J.N.; Costyn, L.J.; Wilding, S.L.; Arnold, R.; Cummings, B.S. Evidence for distinct mechanisms of uptake and antitumor activity of secretory phospholipase A2 responsive liposome in prostate cancer. Integr. Biol. 2012, 5, 172–182. [Google Scholar] [CrossRef]
- Quach, N.D.; Mock, J.N.; Scholpa, N.E.; Eggert, M.W.; Payré, C.; Lambeau, G.; Arnold, R.D.; Cummings, B.S. Role of the Phospholipase A2 Receptor in Liposome Drug Delivery in Prostate Cancer Cells. Mol. Pharm. 2014, 11, 3443–3451. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.-I.; Ogawa, M. Overexpression of group II phospholipase A2 in human breast cancer tissues is closely associated with their malignant potency. Br. J. Cancer 1994, 69, 1166–1170. [Google Scholar] [CrossRef]
- He, L.-F.; Xu, H.-W.; Chen, M.; Xian, Z.-R.; Wen, X.-F.; Du, C.-W.; Huang, W.-H.; Wu, J.-D.; Zhang, G.-J.; Chen, M.-N. Activated-PAK4 predicts worse prognosis in breast cancer and promotes tumorigenesis through activation of PI3K/AKT signaling. Oncotarget 2017, 8, 17573–17585. [Google Scholar] [CrossRef]
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell. Mol. Neurobiol. 2016, 36, 301–312. [Google Scholar] [CrossRef]
- Callegari, E.; D’Abundo, L.; Guerriero, P.; Simioni, C.; Elamin, B.K.; Russo, M.; Cani, A.; Bassi, C.; Zagatti, B.; Giacomelli, L.; et al. miR-199a-3p Modulates MTOR and PAK4 Pathways and Inhibits Tumor Growth in a Hepatocellular Carcinoma Transgenic Mouse Model. Mol. Ther. Nucleic Acids 2018, 11, 485–493. [Google Scholar] [CrossRef]
- Lu, S.-X.; Zhang, C.Z.; Luo, R.-Z.; Wang, C.-H.; Liu, L.-L.; Fu, J.; Zhang, L.; Wang, H.; Xie, D.; Yun, J.-P. Zic2 promotes tumor growth and metastasis via PAK4 in hepatocellular carcinoma. Cancer Lett. 2017, 402, 71–80. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, C.; Liu, Y.; Wang, S. Bacterial adhesion on the metal-polymer composite coatings. Int. J. Adhes. Adhes. 2007, 27, 85–91. [Google Scholar] [CrossRef]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef]
- Zhang, B.; Pan, X.; Cobb, G.; Anderson, T. microRNAs as oncogenes and tumor suppressors. Dev. Biol. 2007, 302, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Lin, L.; Zhou, W.; Wang, Z.; Ding, G.; Dong, Q.; Qin, L.; Wu, X.; Zheng, Y.; Yang, Y.; et al. Identification of miRNomes in Human Liver and Hepatocellular Carcinoma Reveals miR-199a/b-3p as Therapeutic Target for Hepatocellular Carcinoma. Cancer Cell 2011, 19, 232–243. [Google Scholar] [CrossRef]
- Li, S.-Q.; Wang, Z.-H.; Mi, X.-G.; Liu, L.; Tan, Y. MiR-199a/b-3p suppresses migration and invasion of breast cancer cells by downregulating PAK4/MEK/ERK signaling pathway. IUBMB Life 2015, 67, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Fornari, F.; Milazzo, M.; Chieco, P.; Negrini, M.; Calin, G.A.; Grazi, G.L.; Pollutri, D.; Croce, C.M.; Bolondi, L.; Gramantieri, L.; et al. MiR-199a-3p Regulates mTOR and c-Met to Influence the Doxorubicin Sensitivity of Human Hepatocarcinoma Cells. Cancer Res. 2010, 70, 5184–5193. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, G.N.; Bella, L.; Zona, S.; Burton, M.J.; Lam, E.W.-F. Insights into a Critical Role of the FOXO3a-FOXM1 Axis in DNA Damage Response and Genotoxic Drug Resistance. Curr. Drug Targets 2016, 17, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.-P.; Wang, C.-Y.; Jin, X.-H.; Li, M.; Wang, F.-W.; Huang, W.-J.; Yun, J.-P.; Xu, R.-H.; Cai, Q.-Q.; Xie, D. Acidic microenvironment up-regulates exosomal miR-21 and miR-10b in early-stage hepatocellular carcinoma to promote cancer cell proliferation and metastasis. Theranostics 2019, 9, 1965–1979. [Google Scholar] [CrossRef]
- Ladeiro, Y.; Couchy, G.; Balabaud, C.; Bioulac-Sage, P.; Pelletier, L.; Rebouissou, S.; Zucman-Rossi, J. MicroRNA profiling in hepatocellular tumors is associated with clinical features and oncogene/tumor suppressor gene mutations. Hepatology 2008, 47, 1955–1963. [Google Scholar] [CrossRef]
- Chen, X.; Zhao, W.; Yuan, Y.; Bai, Y.; Sun, Y.; Zhu, W.; Du, Z. MicroRNAs tend to synergistically control expression of genes encoding extensively-expressed proteins in humans. PeerJ 2017, 5, e3682. [Google Scholar] [CrossRef]
- Hu, S.; Huang, M.; Nguyen, P.K.; Gong, Y.; Li, Z.; Jia, F.; Lan, F.; Liu, J.; Nag, D.; Robbins, R.C.; et al. Novel MicroRNA Prosurvival Cocktail for Improving Engraftment and Function of Cardiac Progenitor Cell Transplantation. Circulation 2011, 124, S27–S34. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Xiao, H.; Lu, L.; Chen, Y.; Wang, Y.; Xia, W.; Long, M.; Tao, J.; Shen, J.; Shuai, X. Polymeric Vector-Mediated Targeted Delivery of Anti-PAK1 siRNA to Macrophages for Efficient Atherosclerosis Treatment. ACS Biomater. Sci. Eng. 2019, 5, 4455–4462. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Minden, A. The Use of Nanomedicine to Target Signaling by the PAK Kinases for Disease Treatment. Cells 2021, 10, 3565. https://doi.org/10.3390/cells10123565
Wang Y, Minden A. The Use of Nanomedicine to Target Signaling by the PAK Kinases for Disease Treatment. Cells. 2021; 10(12):3565. https://doi.org/10.3390/cells10123565
Chicago/Turabian StyleWang, Yiling, and Audrey Minden. 2021. "The Use of Nanomedicine to Target Signaling by the PAK Kinases for Disease Treatment" Cells 10, no. 12: 3565. https://doi.org/10.3390/cells10123565
APA StyleWang, Y., & Minden, A. (2021). The Use of Nanomedicine to Target Signaling by the PAK Kinases for Disease Treatment. Cells, 10(12), 3565. https://doi.org/10.3390/cells10123565





