Sperm Selection for ICSI: Do We Have a Winner?
Abstract
1. Introduction
2. Natural Sperm Selection
3. Classic Methods
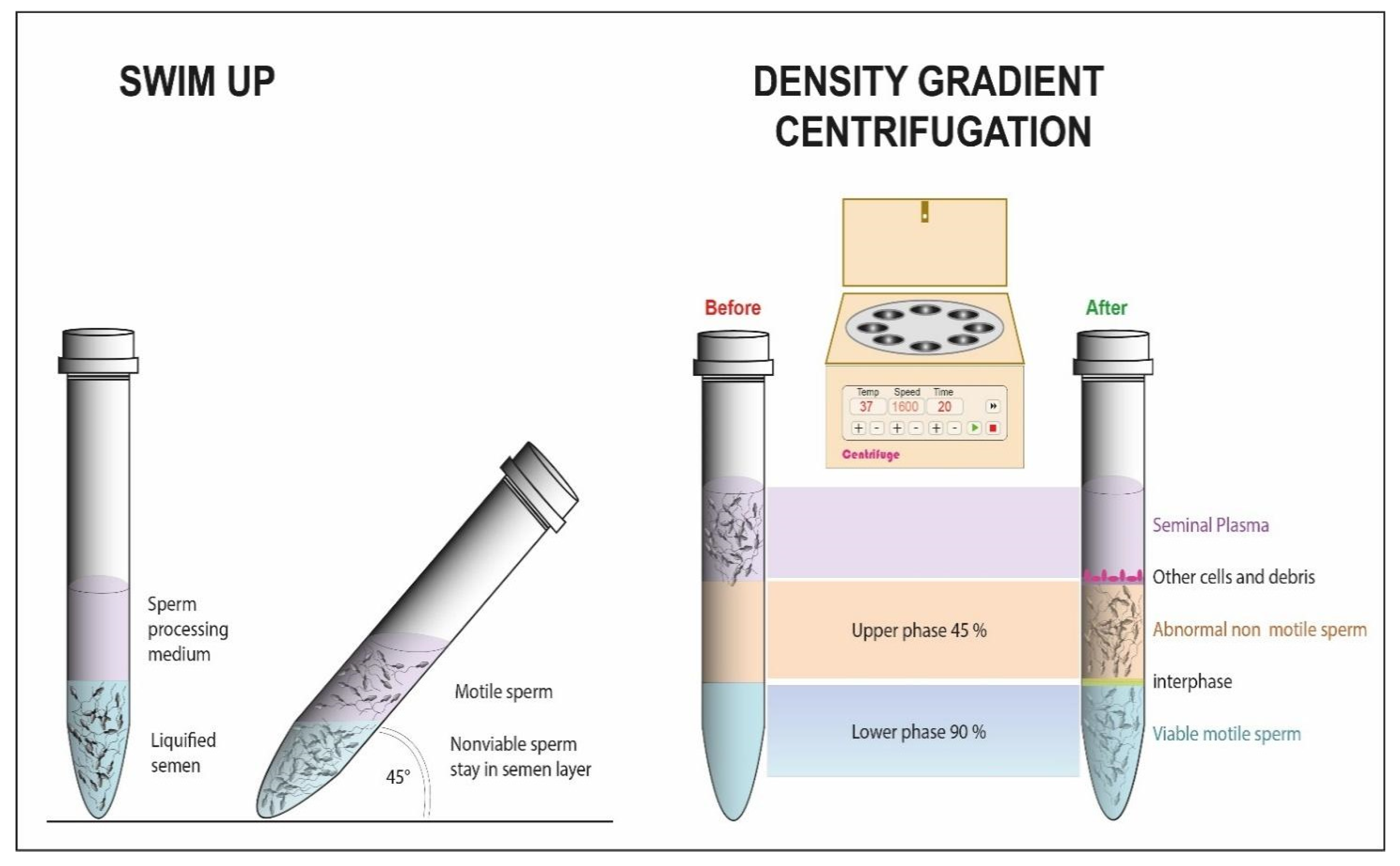
3.1. Swim-Up
3.2. DGC
4. Advanced Methods
4.1. Selection Methods for Sperm Cells with Reduced Motility
4.2. Sperm Cells Selection by Membrane Characteristics
4.3. Selection Based on Morphology–IMSI
4.4. Dynamic Selection of Spermatozoa
- Human spermatozoa orient their motion, by rheotaxis, against the fluid stream directed towards them [129]. Rheotaxis is defined as the tendency of certain living beings to move in response to the mechanical stimulus of a current of water. Some studies have confirmed the presence of a flow-directed towards the uterus in the oviductal region, capable of increasing its intensity after intercourse, which is able to attract the sperm cells [130]. In the technique developed by Nagata et al. [131], using bull sperm as a mammalian model, a flow is created through a series of microchannels towards a well where the sperm sample is delivered. In response to the flow, the spermatozoa swim towards it passing through the microchannels and finally reaching a receptive well where they can be collected for ART applications. Another study was conducted on normospermic patients [132] to select spermatozoa using rheotaxis: compared to an untreated sample and a sample subjected to density gradient centrifugation, the recovered spermatozoa showed higher chromatin compactness (99% vs. 71% vs. 83%). In recent years, attempts have been made to include the concept of rheotaxis in microfluidics [133], but despite the promising data, an analysis on the quality of selected spermatozoa has not been conducted and the passive nature of rheotaxis correlates to the inability to discern the different types of motility.
- Progesterone plays the main role as a chemo-attractor for the navigation of spermatozoa through the environment surrounding the cumulus-oocyte complex [134]. Only capacitated spermatozoa possess the receptors to recognize and bind this type of molecule [134]. Some sperm cell selection techniques exploit this feature to distinguish capacitated spermatozoa from non-capacitated ones. Several studies have been conducted on different types of sperm populations, using a device where a progesterone concentration gradient is constituted (sperm selection assay) [135]. In this method, two wells are connected by a 2 mm length per 2.5 mm diameter tube. One of the wells is filled with a media containing the chemoattractant molecule in solution (progesterone for instance) and the other well is filled with the human sperm sample. The chemoattractant diffuses through the tube generating a gradient and the spermatozoa respond by moving towards the higher concentration and accumulating in the initial well free of cells where they can be used for ART applications. The recovered spermatozoa from this technique exhibit better morphology, less DNA fragmentation and a reduced rate of apoptosis as compared to those selected with density gradient centrifugation [136]. Although the results are also promising in this case, further studies need to be conducted to confirm the improvements in the clinical field.
- The spermatozoa can direct their motion according to the variation in temperature, moving from cold areas to warmer areas [137]. Several studies show that this mechanism underlies the movement of sperm cells from the fallopian tubes to the ampulla [138,139]. Even in this case, however, only the capacitated spermatozoa can respond to the temperature gradient, making this motion a mechanism for the selection of sperm cells with better fertilizing characteristics. The method of sperm selection by thermotaxis was developed by Pérez-Cerezales et al. [140]. Spermatozoa are placed in a drop of medium, connected by a capillary to a second drop free of cells. A temperature gradient is generated between both drops, as the highest temperature is maintained in the drop free of cells. Spermatozoa respond by thermotaxis migrating towards the warmer temperature and accumulating in the second drop where they can be collected for downstream applications.
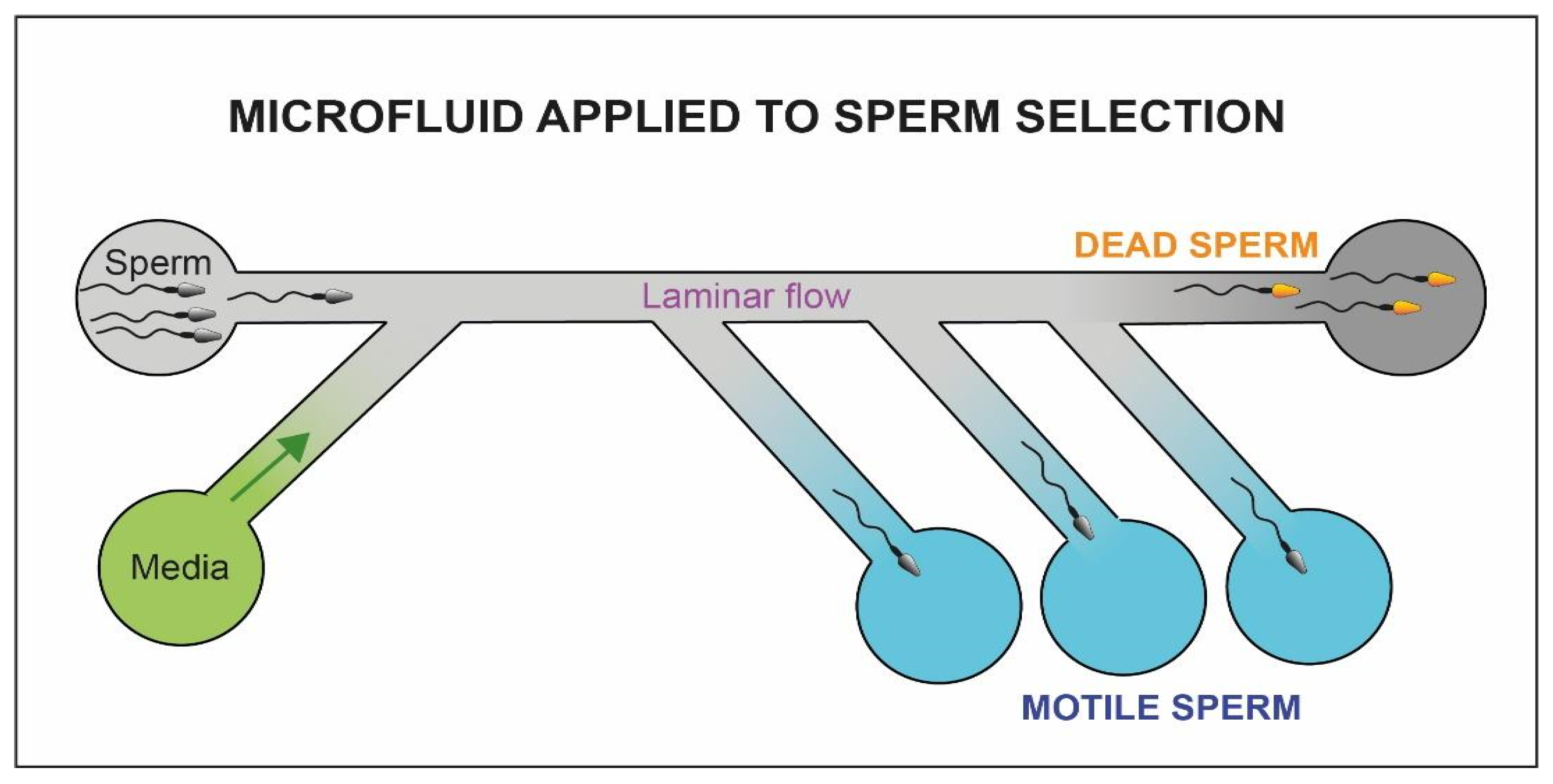
4.5. Microfluidics Applied to Sperm Selection
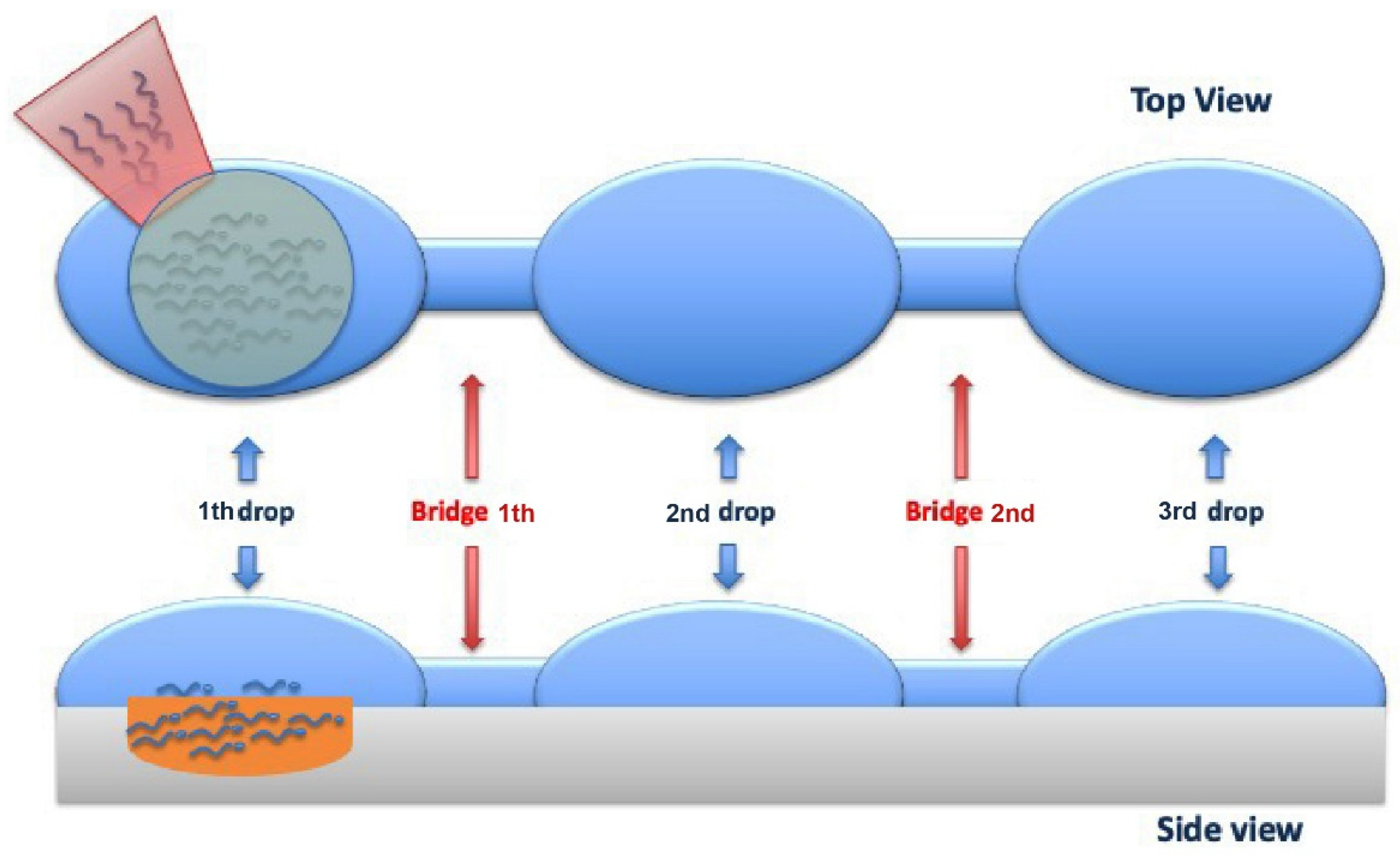
4.6. Horizontal Sperm Migration
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schwarze, J.E.; Jeria, R.; Crosby, J.; Villa, S.; Ortega, C.; Pommer, R. Is there a reason to perform ICSI in the absence of male factor? Lessons from the Latin American Registry of ART. Hum. Reprod. Open 2017, 2017, hox013. [Google Scholar] [CrossRef]
- CDC. 2016—National Summary Report—Assisted Reproductive Technology; US Department of Health. Human Service: Washington, DC, USA, 2018. [Google Scholar]
- De Geyter, C.; Calhaz-Jorge, C.; Kupka, M.S.; Wyns, C.; Mocanu, E.; Motrenko, T.; Scaravelli, G.; Smeenk, J.; Vidakovic, S.; Goossens, V. European IVF-monitoring Consortium (EIM) for the European Society of Human Reproduction and Embryology (ESHRE). ART in Europe, 2014: Results generated from European registries by ESHRE: The European IVF-monitoring Consortium (EIM) for the European Society of Human Reproduction and Embryology (ESHRE). Hum. Reprod. 2018, 33, 1586–1601. [Google Scholar] [CrossRef]
- Henningsen, A.A.; Gissler, M.; Rasmussen, S.; Opdahl, S.; Wennerholm, U.B.; Spangsmose, A.L.; Tiitinen, A.; Bergh, C.; Romundstad, L.B.; Laivuori, H.; et al. Imprinting disorders in children born after ART: A nordic study from the CoNARTaS group. Hum. Reprod. 2020, 35, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Vander Borght, M.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jorgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Mindlis, I.; Pinotti, R.; Swan, S.H. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum. Reprod. Update 2017, 23, 646–659. [Google Scholar] [CrossRef]
- Nilsson, E.E.; Sadler-Riggleman, I.; Skinner, M.K. Environmentally induced epigenetic transgenerational inheritance of disease. Environ. Epigenet. 2018, 4, dvy016. [Google Scholar] [CrossRef]
- Janecka, M.; Mill, J.; Basson, M.A.; Goriely, A.; Spiers, H.; Reichenberg, A.; Schalkwyk, L.; Fernandes, C. Advanced paternal age effects in neurodevelopmental disorders—review of potential underlying mechanisms. Transl. Psychiatry 2017, 7, e1019. [Google Scholar] [CrossRef] [PubMed]
- D’Onofrio, B.M.; Rickert, M.E.; Frans, E.; Kuja-Halkola, R.; Almqvist, C.; Sjolander, A.; Larsson, H.; Lichtenstein, P. Paternal age at childbearing and offspring psychiatric and academic morbidity. JAMA Psychiatry 2014, 71, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Belva, F.; Bonduelle, M.; Roelants, M.; Michielsen, D.; Van Steirteghem, A.; Verheyen, G.; Tournaye, H. Semen quality of young adult ICSI offspring: The first results. Hum. Reprod. 2016, 31, 2811–2820. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Calabuig, M.J.; López-Cardona, A.P.; Fernández-González, R.; Ramos-Ibeas, P.; Fonseca Balvís, N.; Laguna-Barraza, R.; Pericuesta, E.; Gutiérrez-Adán, A.; Bermejo-Álvarez, P. Potential Health Risks Associated to ICSI: Insights from Animal Models and Strategies for a Safe Procedure. Front. Public Health 2014, 2, 241. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, D.; Ramalingam, M.; Garrido, N.; Barratt, C.L. Sperm selection in natural conception: What can we learn from Mother Nature to improve assisted reproduction outcomes? Hum. Reprod. Update 2015, 21, 711–726. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cerezales, S.; Ramos-Ibeas, P.; Acuña, O.S.; Avilés, M.; Coy, P.; Rizos, D.; Gutiérrez-Adán, A. The oviduct: From sperm selection to the epigenetic landscape of the embryo. Biol. Reprod. 2018, 98, 262–276. [Google Scholar] [CrossRef]
- Suarez, S.S.; Pacey, A.A. Sperm transport in the female reproductive tract. Hum. Reprod. Update 2006, 12, 23–27. [Google Scholar] [CrossRef]
- Puga Molina, L.C.; Luque, G.M.; Balestrini, P.A.; Marín-Briggiler, C.I.; Romarowski, A.; Buffone, M.G. Molecular Basis of Human Sperm Capacitation. Front. Cell Dev. Biol. 2018, 6, 72. [Google Scholar] [CrossRef]
- Hernández-Silva, G.; Chirinos, M. Proteins from male and female reproductive tracts involved in sperm function regulation. Zygote 2019, 27, 5–16. [Google Scholar] [CrossRef]
- ESHRE Guideline Group on Good Practice in IVF Labs; De los Santos, M.J.; Apter, S.; Coticchio, G.; Debrock, S.; Lundin, K.; Vermeulen, N. Revised guidelines for good practice in IVF laboratories. Hum. Reprod. 2015, 31, 685–686. [Google Scholar]
- Vaughan, D.A.; Sakkas, D. Sperm selection methods in the 21st century. Biol. Reprod. 2019, 101, 1076–1082. [Google Scholar] [CrossRef]
- Lopata, A.; Patullo, M.J.; Chang, A.; James, B. A method collecting motile spermatozoa from human semen. Fertil. Steril. 1976, 27, 677. [Google Scholar] [CrossRef]
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012. [Google Scholar] [CrossRef]
- Ghumman, S.; Adiga, S.K.; Upadhya, D.; Kalthur, G.; Jayaraman, V.; Rao, S.B.; Kumar, P. Combination of swim-up and density gradient separation methods effectively eliminate DNA damaged sperm. J. Turk. Ger. Gynecol. Assoc. 2011, 12, 148–152. [Google Scholar] [CrossRef]
- Jayaraman, V.; Upadhya, D.; Narayan, P.K.; Adiga, S.K. Sperm processing by swim-up and density gradient is effective in elimination of sperm with DNA damage. J. Assist. Reprod. Genet. 2012, 29, 557–563. [Google Scholar] [CrossRef]
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th ed.; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Henkel, R.R.; Schill, W.B. Sperm preparation for ART. Reprod. Biol. Endocrinol. 2003, 1, 108. [Google Scholar] [CrossRef]
- Harrison, R.A. A highly efficient method for washing mammalian spermatozoa. J. Reprod. Fertil. 1976, 48, 347–353. [Google Scholar] [CrossRef]
- Lee, D.; Jee, B.C. Evaluation of normal morphology, DNA fragmentation, and hyaluronic acid binding ability of human spermatozoa after using four different commercial media for density gradient centrifugation. Clin. Exp. Reprod. Med. 2019, 46, 8–13. [Google Scholar] [CrossRef]
- Strehler, E.; Baccetti, B.; Sterzik, K.; Capitani, S.; Collodel, G.; De Santo, M.; Gambera, L.; Piomboni, P. Detrimental effects of polyvinylpyrrolidone on the ultrastructure of spermatozoa (Notulae seminologicae 13). Hum. Reprod. 1998, 13, 120–123. [Google Scholar] [CrossRef]
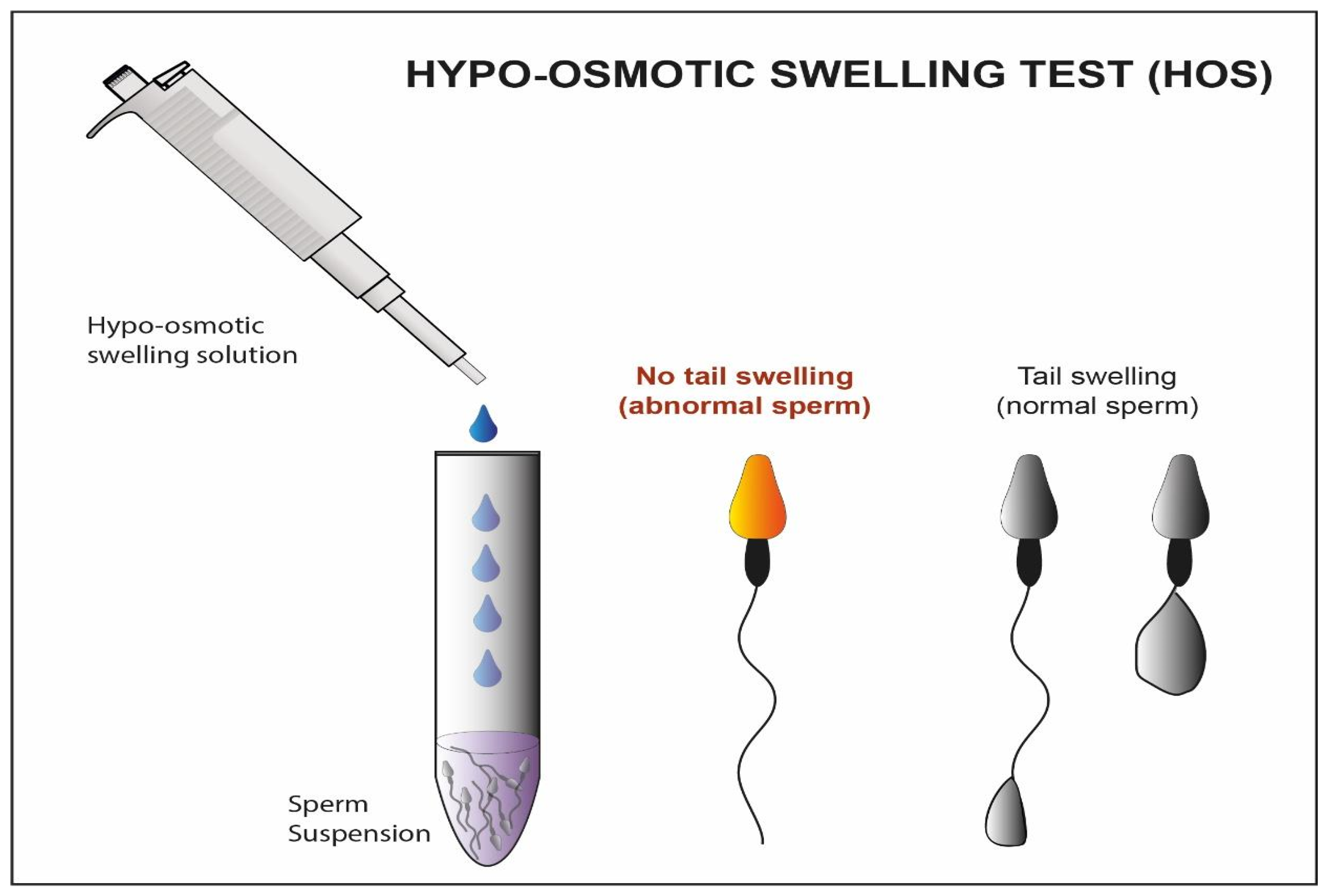
- Jeyendran, R.S.; Van der Ven, H.H.; Perez-Pelaez, M.; Crabo, B.G.; Zaneveld, L.J. Development of an assay to assess the functional integrity of the human sperm membrane and its relationship to other semen characteristics. J. Reprod. Fertil. 1984, 70, 219–228. [Google Scholar] [CrossRef]
- Bassiri, F.; Tavalaee, M.; Shiravi, A.H.; Mansouri, S.; Nasr-Esfahani, M.H. Is there an association between HOST grades and sperm quality? Hum Reprod. 2012, 27, 2277–2284. [Google Scholar] [CrossRef]
- Bloch, A.; Rogers, E.J.; Nicolas, C.; Martin-Denavit, T.; Monteiro, M.; Thomas, D.; Morel, H.; Lévy, R.; Siffroi, J.P.; Dupont, C.; et al. Detailed cell-level analysis of sperm nuclear quality among the different hypo-osmotic swelling test (HOST) classes. J. Assist. Reprod. Genet. 2021, 38, 2491–2499. [Google Scholar] [CrossRef]
- Casper, R.F.; Meriano, J.S.; Jarvi, K.A.; Cowan, L.; Lucato, M.L. The hypo-osmotic swelling test for selection of viable sperm for intracytoplasmic injection in men with complete asthenozoospermia. Fertil. Steril. 1996, 65, 972–976. [Google Scholar] [CrossRef]
- Tsai, Y.L.; Liu, J.; Garcia, J.E.; Katz, E.; Compton, G.; Baramki, T.A. Establishment of optimal hypo-osmotic swelling test by examining single spermatozoa from different hypo-osmotic solutions. Hum. Reprod. 1997, 12, 11–113. [Google Scholar] [CrossRef]
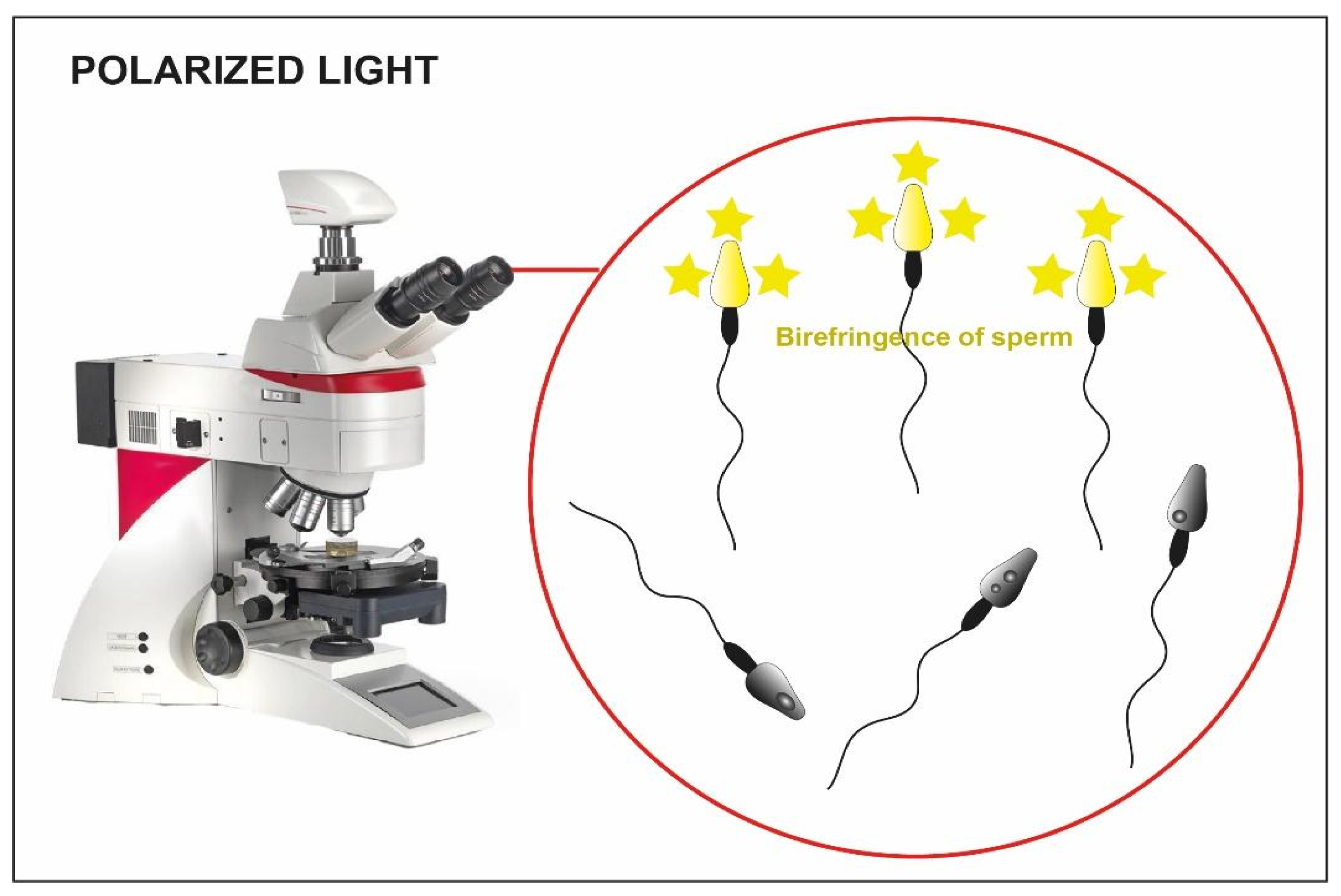
- Vermey, B.G.; Chapman, M.G.; Cooke, S.; Kilani, S. The relationship between sperm head retardance using polarized light microscopy and clinical outcomes. Reprod. Biomed. Online 2015, 30, 67–73. [Google Scholar] [CrossRef]
- Baccetti, B. Microscopical advances in assisted reproduction. J. Submicrosc. Cytol. Pathol. 2004, 36, 333–339. [Google Scholar]
- Aktan, T.M.; Montag, M.; Duman, S.; Gorkemli, H.; Rink, K.; Yurdakul, T. Use of a laser to detect viable but immotile spermatozoa. Andrologia 2004, 36, 366–369. [Google Scholar] [CrossRef]
- Birowo, P.; Tendi, W.; Rasyid, N.; Turek, P.J.; Sini, I.R.; Rizal, M. Successful Targeted Testicular Sperm Extraction Using Microsurgical Technique (microTESE) Following Fine Needle Aspiration (FNA) Mapping in a Non-Obstructive Azoospermia (NOA) Patient: A Case Report. J. Reprod. Infertil. 2021, 22, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Gerber, P.A.; Kruse, R.; Hirchenhain, J.; Krüssel, J.S.; Neumann, N.J. Pregnancy after laser-assisted selection of viable spermatozoa before intracytoplasmatic sperm injection in a couple with male primary cilia dyskinesia. Fertil. Steril. 2008, 89, e9–e12. [Google Scholar] [CrossRef]
- Ozkavukcu, S.; Celik-Ozenci, C.; Konuk, E.; Atabekoglu, C. Live birth after Laser Assisted Viability Assessment (LAVA) to detect pentoxifylline resistant ejaculated immotile spermatozoa during ICSI in a couple with male Kartagener’s syndrome. Reprod. Biol. Endocrinol. 2018, 16, 10. [Google Scholar] [CrossRef]
- Salman Yazdi, R.; Bakhshi, S.; Jannat Alipoor, F.; Akhoond, M.R.; Borhani, S.; Farrahi, F.; Lotfi Panah, M.; Sadighi Gilani, M.A. Effect of 830-nm diode laser irradiation on human sperm motility. Lasers Med. Sci. 2014, 29, 97–104. [Google Scholar] [CrossRef]
- Montag, M.; Rink, K.; Delacrétaz, G.; van der Ven, H. Laser-induced immobilization and plasma membrane permeabilization in human spermatozoa. Hum. Reprod. 2000, 15, 846–852. [Google Scholar] [CrossRef]
- Hoogendijk, C.F.; Kruger, T.F.; Bouic, P.J.; Henkel, R.R. A novel approach for the selection of human sperm using annexin V-binding and flow cytometry. Fertil. Steril. 2009, 91, 1285–1292. [Google Scholar] [CrossRef]
- Grunewald, S.; Paasch, U.; Glander, H.J. Enrichment of non-apoptotic human spermatozoa after cryopreservation by immunomagnetic cell sorting. Cell Tissue Bank. 2001, 2, 127–133. [Google Scholar] [CrossRef]
- Lee, T.H.; Liu, C.H.; Shih, Y.T.; Tsao, H.M.; Huang, C.C.; Chen, H.H.; Lee, M.S. Magnetic-activated cell sorting for sperm preparation reduces spermatozoa with apoptotic markers and improves the acrosome reaction in couples with unexplained infertility. Hum. Reprod. 2010, 25, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Degheidy, T.; Abdelfattah, H.; Seif, A.; Albuz, F.K.; Gazi, S.; Abbas, S. Magnetic activated cell sorting: An effective method for reduction of sperm DNA fragmentation in varicocele men prior to assisted reproductive techniques. Andrologia 2015, 47, 892–896. [Google Scholar] [CrossRef]
- Nadalini, M.; Tarozzi, N.; Di Santo, M.; Borini, A. Annexin V magnetic-activated cell sorting versus swim-up for the selection of human sperm in ART: Is the new approach better than the traditional one? J. Assist. Reprod. Genet. 2014, 31, 1045–1051. [Google Scholar] [CrossRef]
- Cayli, S.; Jakab, A.; Ovari, L.; Delpiano, E.; Celik-Ozenci, C.; Sakkas, D.; Ward, D.; Huszar, G. Biochemical markers of sperm function: Male fertility and sperm selection for ICSI. Reprod. Biomed. Online. 2003, 7, 462–468. [Google Scholar] [CrossRef]
- Huszar, G.; Ozkavukcu, S.; Jakab, A.; Celik-Ozenci, C.; Sati, G.L.; Cayli, S. Hyaluronic acid binding ability of human sperm reflects cellular maturity and fertilizing potential: Selection of sperm for intracytoplasmic sperm injection. Curr. Opin. Obstet. Gynecol. 2006, 18, 260–267. [Google Scholar] [CrossRef]
- Jakab, A.; Sakkas, D.; Delpiano, E.; Cayli, S.; Kovanci, E.; Ward, D.; Revelli, A.; Huszar, G. Intracytoplasmic sperm injection: A novel selection method for sperm with normal frequency of chromosomal aneuploidies. Fertil. Steril. 2005, 84, 1665–1673. [Google Scholar] [CrossRef]
- Van Den Bergh, M.J.; Fahy-Deshe, M.; Hohl, M.K. Pronuclear zygote score following intracytoplasmic injection of hyaluronan-bound spermatozoa: A prospective randomized study. Reprod. Biomed. Online 2009, 19, 796–801. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, U.; Krassnigg, F.; Schatz, H.; Schill, W.B. Separation of human X and Y spermatozoa by free-flow electrophoresis. Gamete Res. 1988, 19, 151–160. [Google Scholar] [CrossRef]
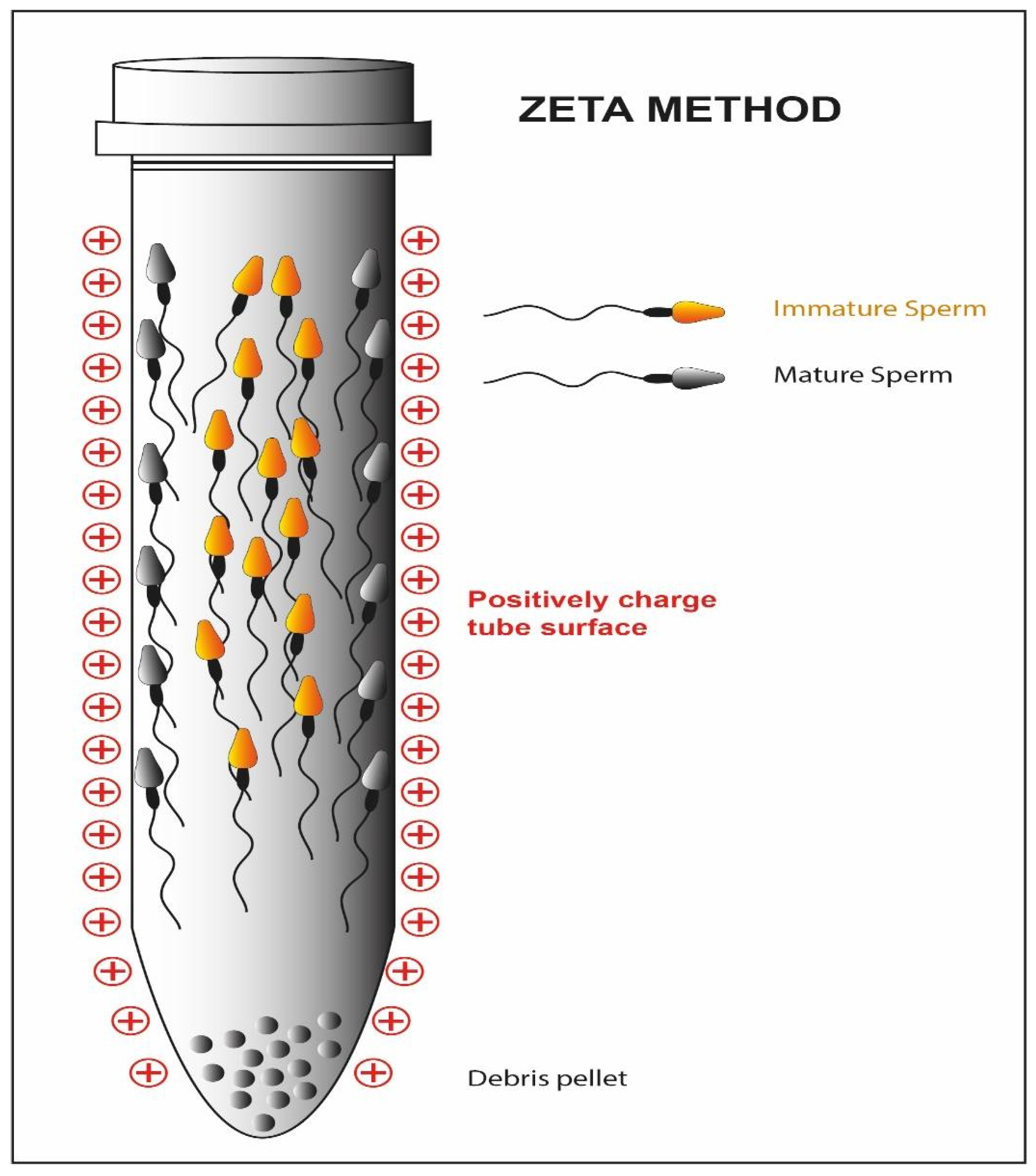
- Kheirollahi-Kouhestani, M.; Razavi, S.; Tavalaee, M.; Deemeh, M.R.; Mardani, M.; Moshtaghian, J.; Nasr-Esfahani, M.H. Selection of sperm based on combined density gradient and Zeta method may improve ICSI outcome. Hum. Reprod. 2009, 24, 2409–2416. [Google Scholar] [CrossRef]
- Razavi, S.H.; Nasr-Esfahani, M.H.; Deemeh, M.R.; Shayesteh, M.; Tavalaee, M. Evaluation of zeta and HA-binding methods for selection of spermatozoa with normal morphology, protamine content and DNA integrity. Andrologia 2010, 42, 13–19. [Google Scholar] [CrossRef]
- Nasr Esfahani, M.H.; Deemeh, M.R.; Tavalaee, M.; Sekhavati, M.H.; Gourabi, H. Zeta Sperm Selection Improves Pregnancy Rate and Alters Sex Ratio in Male Factor Infertility Patients: A Double-Blind, Randomized Clinical Trial. Int. J. Fertil. Steril. 2016, 10, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.J.; Jacobson, J.D.; Corselli, J.U.; Patton, W.C. A simple zeta method for sperm selection based on membrane charge. Fertil. Steril. 2006, 85, 481–486. [Google Scholar] [CrossRef]
- Ainsworth, C.; Nixon, B.; Aitken, R.J. Development of a novel electrophoretic system for the isolation of human spermatozoa. Hum. Reprod. 2005, 20, 2261–2270. [Google Scholar] [CrossRef]
- Shalom-Paz, E.; Anabusi, S.; Michaeli, M.; Karchovsky-Shoshan, E.; Rothfarb, N.; Shavit, T.; Ellenbogen, A. Can intra cytoplasmatic morphologically selected sperm injection (IMSI) technique improve outcome in patients with repeated IVF-ICSI failure? a comparative study. Gynecol. Endocrinol. 2015, 31, 247–251. [Google Scholar] [CrossRef]
- Leandri, R.D.; Gachet, A.; Pfeffer, J.; Celebi, C.; Rives, N.; Carre-Pigeon, F.; Kulski, O.; Mitchell, V.; Parinaud, J. Is intracytoplasmic morphologically selected sperm injection (IMSI) beneficial in the first ART cycle? a multicentric randomized controlled trial. Andrology 2013, 1, 692–697. [Google Scholar] [CrossRef]
- Teixeira, D.M.; Hadyme Miyague, A.; Barbosa, M.A.; Navarro, P.A.; Raine-Fenning, N.; Nastri, C.O.; Martins, W.P. Regular (ICSI) versus ultra-high magnification (IMSI) sperm selection for assisted reproduction. Cochrane Database Syst. Rev. 2020, 2, CD010167. [Google Scholar] [CrossRef]
- Fortunato, A.; Boni, R.; Leo, R.; Nacchia, G.; Liguori, F.; Casale, S.; Bonassisa, P.; Tosti, E. Vacuoles in sperm head are not associated with head morphology, DNA damage and reproductive success. Reprod. Biomed. Online 2016, 32, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.D.; Takayama, S. Application of microfluidic technologies to human assisted reproduction. Mol. Hum. Reprod. 2017, 23, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Parrella, A.C.D.; Keating, D.; Rosenwaks, Z.; Palermo, G.D. A microfluidic devide for selecting the most progressively motile spermatozoa yields a higher rate of euploid embryos. Fertil. Steril. 2018, 110, e342. [Google Scholar] [CrossRef]
- Baldini, D.; Baldini, A.; Silvestris, E.; Vizziello, G.; Ferri, D.; Vizziello, D. A fast and safe technique for sperm preparation in ICSI treatments within a randomized controlled trial (RCT). Reprod. Biol. Endocrinol. 2020, 18, 88. [Google Scholar] [CrossRef] [PubMed]
- Palini, S.; Stefani, S.; Primiterra, M.; Benedetti, S.; Barone, S.; Carli, L.; Vaccari, E.; Murat, U.; Feichtinger, W. Comparison of in vitro fertilization outcomes in ICSI cycles after human sperm preparation by density gradient centrifugation and direct micro swim-up without centrifugation. JBRA Assist. Reprod. 2017, 21, 89–93. [Google Scholar] [CrossRef]
- Mahadevan, M.; Baker, G. Assessment and Preparation of Semen for in Vitro Fertilization Clinical in Vitro Fertilization, and A. Trounson; Springer: London, UK, 1984; pp. 83–97. [Google Scholar]
- Sanchez, R.; Stalf, T.; Khanaga, O.; Turley, H.; Gips, H.; Schill, W.B. Sperm selection methods for intracytoplasmic sperm injection (ICSI) in andrological patients. J. Assist. Reprod. Genet. 1996, 13, 228–233. [Google Scholar] [CrossRef]
- Zini, A.; Finelli, A.; Phang, D.; Jarvi, K. Influence of semen processing technique on human sperm DNA integrity. Urology 2000, 56, 1081–1084. [Google Scholar] [CrossRef]
- Raad, G.; Bakos, H.W.; Bazzi, M.; Mourad, Y.; Fakih, F.; Shayya, S.; Mchantaf, L.; Fakih, C. Differential impact of four sperm preparation techniques on sperm motility, morphology, DNA fragmentation, acrosome status, oxidative stress, and mitochondrial activity: A prospective study. Andrology 2021, 9, 1549–1559. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Silva, G.; López-Torres, A.S.; Maldonado-Rosas, I.; Mata-Martínez, E.; Larrea, F.; Torres-Flores, V.; Treviño, C.L.; Chirinos, M. Effects of Semen Processing on Sperm Function: Differences between Swim-Up and Density Gradient Centrifugation. World J. Mens. Health 2021, 39, 740–749. [Google Scholar] [CrossRef]
- Xue, X.; Wang, W.S.; Shi, J.Z.; Zhang, S.L.; Zhao, W.Q.; Shi, W.H.; Guo, B.Z.; Qin, Z. Efficacy of swim-up versus density gradient centrifugation in improving sperm deformity rate and DNA fragmentation index in semen samples from teratozoospermic patients. J. Assist. Reprod. Genet. 2014, 31, 1161–1166. [Google Scholar] [CrossRef]
- Muratori, M.; Tarozzi, N.; Carpentiero, F.; Danti, S.; Perrone, F.M.; Cambi, M.; Casini, A.; Azzari, C.; Boni, L.; Maggi, M.; et al. Sperm selection with density gradient centrifugation and swim up: Effect on DNA fragmentation in viable spermatozoa. Sci. Rep. 2019, 9, 7492. [Google Scholar] [CrossRef]
- De Martin, H.; Miranda, E.P.; Cocuzza, M.S.; Monteleone, P.A.A. Density gradient centrifugation and swim-up for ICSI: Useful, unsafe, or just unsuitable? J. Assist. Reprod. Genet. 2019, 36, 2421–2423. [Google Scholar] [CrossRef] [PubMed]
- Avendaño, C.; Oehninger, S. DNA fragmentation in morphologically normal spermatozoa: How much should we be concerned in the ICSI era? J. Androl. 2011, 32, 356–363. [Google Scholar] [CrossRef]
- Ezeh, U.I.; Moore, H.D.; Cooke, I.D. A prospective study of multiple needle biopsies versus a single open biopsy for testicular sperm extraction in men with non-obstructive azoospermia. Hum. Reprod. 1998, 13, 3075–3080. [Google Scholar] [CrossRef]
- Girardi, S.K.; Schlegel, P.N. Microsurgical epididymal sperm aspiration: Review of techniques, preoperative considerations, and results. J. Androl. 1996, 17, 5–9. [Google Scholar]
- Ozkocer, S.E.; Konac, E. The current perspective on genetic and epigenetic factors in sperm maturation in the epididymis. Andrologia 2021, 53, e13989. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, N.M.; Vaca Sánchez, R.; Rodriguez Fiesta, S.; Lopez Salgado, T.; Rodríguez, R.; Bethencourt, J.C.; Blanes Zamora, R. Pregnancy with frozen-thawed and fresh testicular biopsy after motile and immotile sperm microinjection, using the mechanical touch technique to assess viability. Hum. Reprod. 2004, 19, 262–265. [Google Scholar] [CrossRef]
- Soares, J.B.; Glina, S.; Galuppo, A.G. Pregnancy with frozen-thawed and fresh testicular biopsy after motile and immotile sperm microinjection, using the mechanical touch technique to assess viability. Hum. Reprod. 2005, 20, 569. [Google Scholar] [CrossRef][Green Version]
- Rossato, M.; Di Virgilio, F.; Foresta, C. Involvement of osmo-sensitive calcium influx in human sperm activation. Mol. Hum. Reprod. 1996, 2, 903–909. [Google Scholar] [CrossRef]
- Nowicka-Bauer, K.; Szymczak-Cendlak, M. Structure and Function of Ion Channels Regulating Sperm Motility—An Overview. Int. J. Mol. Sci. 2021, 22, 3259. [Google Scholar] [CrossRef] [PubMed]
- Aydos, K.; Aydos, O.S. Sperm Selection Procedures for Optimizing the Outcome of ICSI in Patients with NOA. J. Clin. Med. 2021, 10, 2687. [Google Scholar] [CrossRef]
- Gianaroli, L.; Magli, M.C.; Collodel, G.; Moretti, E.; Ferraretti, A.P.; Baccetti, B. Sperm head’s birefringence: A new criterion for sperm selection. Fertil. Steril. 2008, 90, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Chattopadhyay, R.; Bose, G.; Ganesh, A.; Das, S.; Chakravarty, B.N. Selection of birefringent spermatozoa under Polscope: Effect on intracytoplasmic sperm injection outcome. Andrologia 2012, 44 (Suppl. 1), 734–738. [Google Scholar] [CrossRef]
- Agarwal, A.; Said, T.M. Role of sperm chromatin abnormalities and DNA damage in male infertility. Hum. Reprod. Update 2003, 9, 331–345. [Google Scholar] [CrossRef]
- González-Marín, C.; Gosálvez, J.; Roy, R. Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int. J. Mol. Sci. 2012, 13, 14026–14052. [Google Scholar] [CrossRef]
- Rappa, K.L.; Rodriguez, H.F.; Hakkarainen, G.C.; Anchan, R.M.; Mutter, G.L.; Asghar, W. Sperm processing for advanced reproductive technologies: Where are we today? Biotechnol. Adv. 2016, 34, 578–587. [Google Scholar] [CrossRef]
- Majzoub, A.; Agarwal, A.; Esteves, S.C. Clinical utility of sperm DNA damage in male infertility. Panminerva Med. 2019, 61, 118–127. [Google Scholar] [CrossRef]
- Oleszczuk, K.; Augustinsson, L.; Bayat, N.; Giwercman, A.; Bungum, M. Prevalence of high DNA fragmentation index in male partners of unexplained infertile couples. Andrology 2013, 1, 357–360. [Google Scholar] [CrossRef]
- Robinson, L.; Gallos, I.D.; Conner, S.J.; Rajkhowa, M.; Miller, D.; Lewis, S.; Kirkman-Brown, J.; Coomarasamy, A. The effect of sperm DNA fragmentation on miscarriage rates: A systematic review and meta-analysis. Hum. Reprod. 2012, 27, 2908–2917. [Google Scholar] [CrossRef]
- Pinto, S.; Carrageta, D.F.; Alves, M.G.; Rocha, A.; Agarwal, A.; Barros, A.; Oliveira, P.F. Sperm selection strategies and their impact on assisted reproductive technology outcomes. Andrologia 2021, 53, e13725. [Google Scholar] [CrossRef] [PubMed]
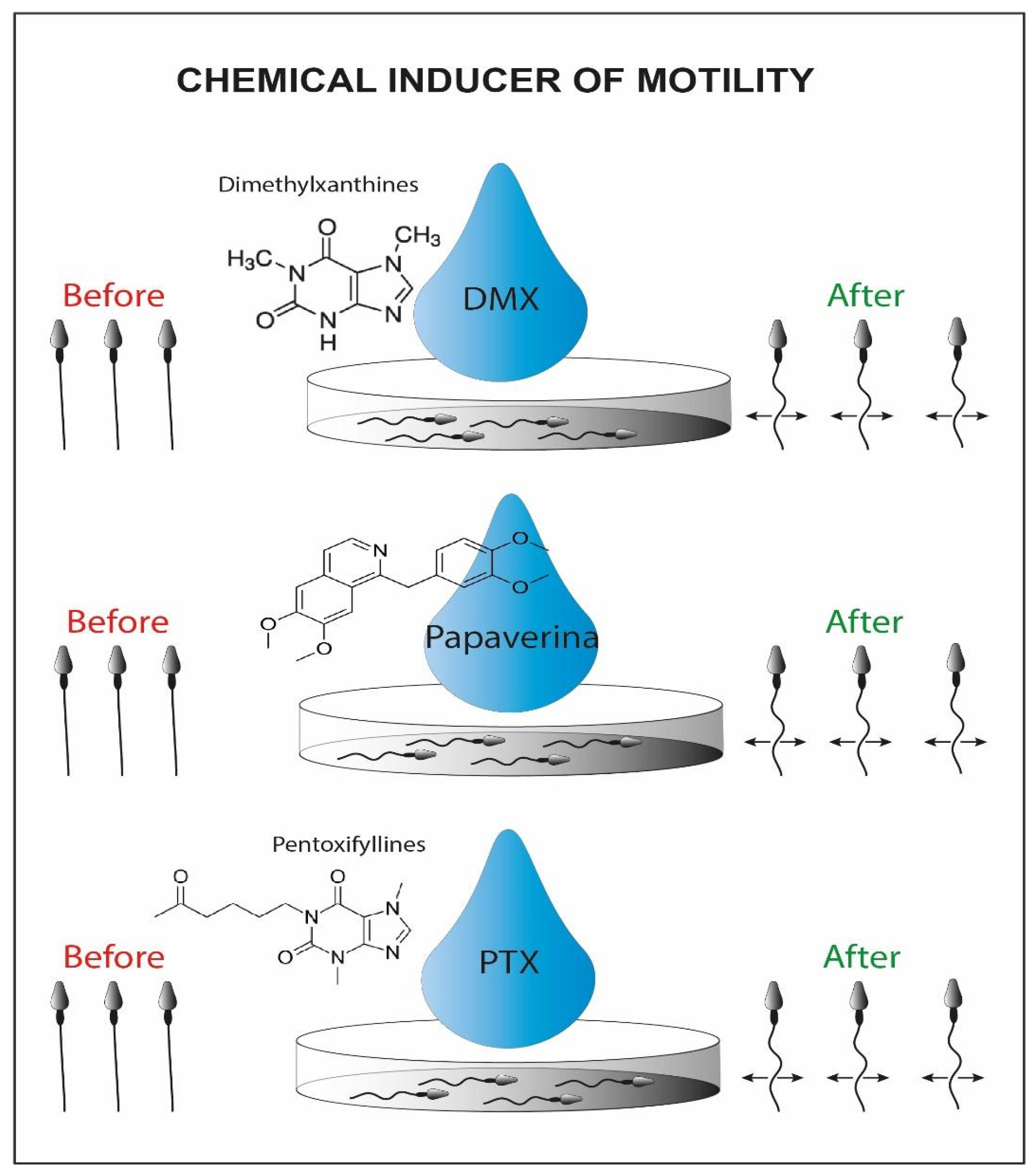
- Morales, P.; Llanos, M.; Yovich, J.L.; Cummins, J.M.; Vigil, P. Pentoxifylline increases sperm penetration into zona-free hamster oocytes without increasing the acrosome reaction. Andrologia 1993, 25, 359–362. [Google Scholar] [CrossRef]
- Mahaldashtian, M.; Khalili, M.A.; Nottola, S.A.; Woodward, B.; Macchiarelli, G.; Miglietta, S. Does in vitro application of pentoxifylline have beneficial effects in assisted male reproduction? Andrologia 2021, 53, e13722. [Google Scholar] [CrossRef] [PubMed]
- Satish, M.; Kumari, S.; Deeksha, W.; Abhishek, S.; Nitin, K.; Adiga, S.K.; Hegde, P.; Dasappa, J.P.; Kalthur, G.; Rajakumara, E. Structure-based redesigning of pentoxifylline analogs against selective phosphodiesterases to modulate sperm functional competence for assisted reproductive technologies. Sci. Rep. 2021, 11, 12293. [Google Scholar] [CrossRef] [PubMed]
- Navas, P.; Paffoni, A.; Intra, G.; González-Utor, A.; Clavero, A.; Gonzalvo, M.C.; Díaz, R.; Peña, R.; Restelli, L.; Somigliana, E.; et al. Obstetric and neo-natal outcomes of ICSI cycles using pentoxifylline to identify viable spermatozoa in patients with immotile spermatozoa. Reprod. Biomed. Online 2017, 34, 414–421. [Google Scholar] [CrossRef][Green Version]
- Gu, Y.F.; Zhou, Q.W.; Zhang, S.P.; Lu, C.F.; Gong, F.; Shi, Y.; Lu, G.X.; Lin, G. The clinical and neonatal outcomes after stimulation of immotile spermatozoa using SperMagic medium. Andrologia 2018, 50, e13056. [Google Scholar] [CrossRef]
- Ibis, E.; Hayme, S.; Baysal, E.; Gul, N.; Ozkavukcu, S. Efficacy and safety of papaverine as an in vitro motility enhancer on human spermatozoa. J. Assist. Reprod. Genet. 2021, 38, 1523–1537. [Google Scholar] [CrossRef]
- Sandi-Monroy, N.L.; Musanovic, S.; Zhu, D.; Szabó, Z.; Vogl, A.; Reeka, N.; Eibner, K.; Bundschu, K.; Gagsteiger, F. Use of dimethylxanthine theophylline (SpermMobil®) does not affect clinical, obstetric or perinatal outcomes. Arch. Gynecol. Obstet. 2019, 300, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Mangoli, V.; Mangoli, R.; Dandekar, S.; Suri, K.; Desai, S. Selection of viable spermatozoa from testicular biopsies: A comparative study between pentoxifylline and hypoosmotic swelling test. Fertil. Steril. 2011, 95, 631–634. [Google Scholar] [CrossRef]
- Park, Y.J.; Pang, M.G. Mitochondrial Functionality in Male Fertility: From Spermatogenesis to Fertilization. Antioxidants 2021, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Ebner, T.; Moser, M.; Tews, G. Possible applications of a non-contact 1.48 microm wavelength diode laser in assisted reproduction technologies. Hum. Reprod. Update 2005, 11, 425–435. [Google Scholar] [CrossRef]
- Tarlatzis, B.C.; Kolibianakis, E.M.; Bontis, J.; Tousiou, M.; Lagos, S.; Mantalenakis, S. Effect of pentoxifylline on human sperm motility and fertilizing capacity. Arch. Androl. 1995, 34, 33–42. [Google Scholar] [CrossRef]
- Nordhoff, V.; Schüring, A.N.; Krallmann, C.; Zitzmann, M.; Schlatt, S.; Kiesel, L.; Kliesch, S. Optimizing TESE-ICSI by laser-assisted selection of immotile spermatozoa and polarization microscopy for selection of oocytes. Andrology 2013, 1, 67–74. [Google Scholar] [CrossRef]
- Neri, Q.V.; Lee, B.; Rosenwaks, Z.; Machaca, K.; Palermo, G.D. Understanding fertilization through intracytoplasmic sperm injection (ICSI). Cell Calcium. 2014, 55, 24–37. [Google Scholar] [CrossRef]
- Guse, A.H.; Gil Montoya, D.C.; Diercks, B.P. Mechanisms and functions of calcium microdomains produced by ORAI channels, d-myo-inositol 1,4,5-trisphosphate receptors, or ryanodine receptors. Pharmacol. Ther. 2021, 223, 107804. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. The Inositol Trisphosphate/Calcium Signaling Pathway in Health and Disease. Physiol. Rev. 2016, 96, 1261–1296. [Google Scholar] [CrossRef]
- Ceyhan, Y.; Zhang, M.; Guo, J.; Sandoval, C.G.; Vacher, J.; Kaftanovskaya, E.M.; Agoulnik, A.I.; Agoulnik, I.U. Deletion of inositol polyphosphate 4-phosphatase type-II B affects spermatogenesis in mice. PLoS ONE 2020, 15, e0233163. [Google Scholar] [CrossRef]
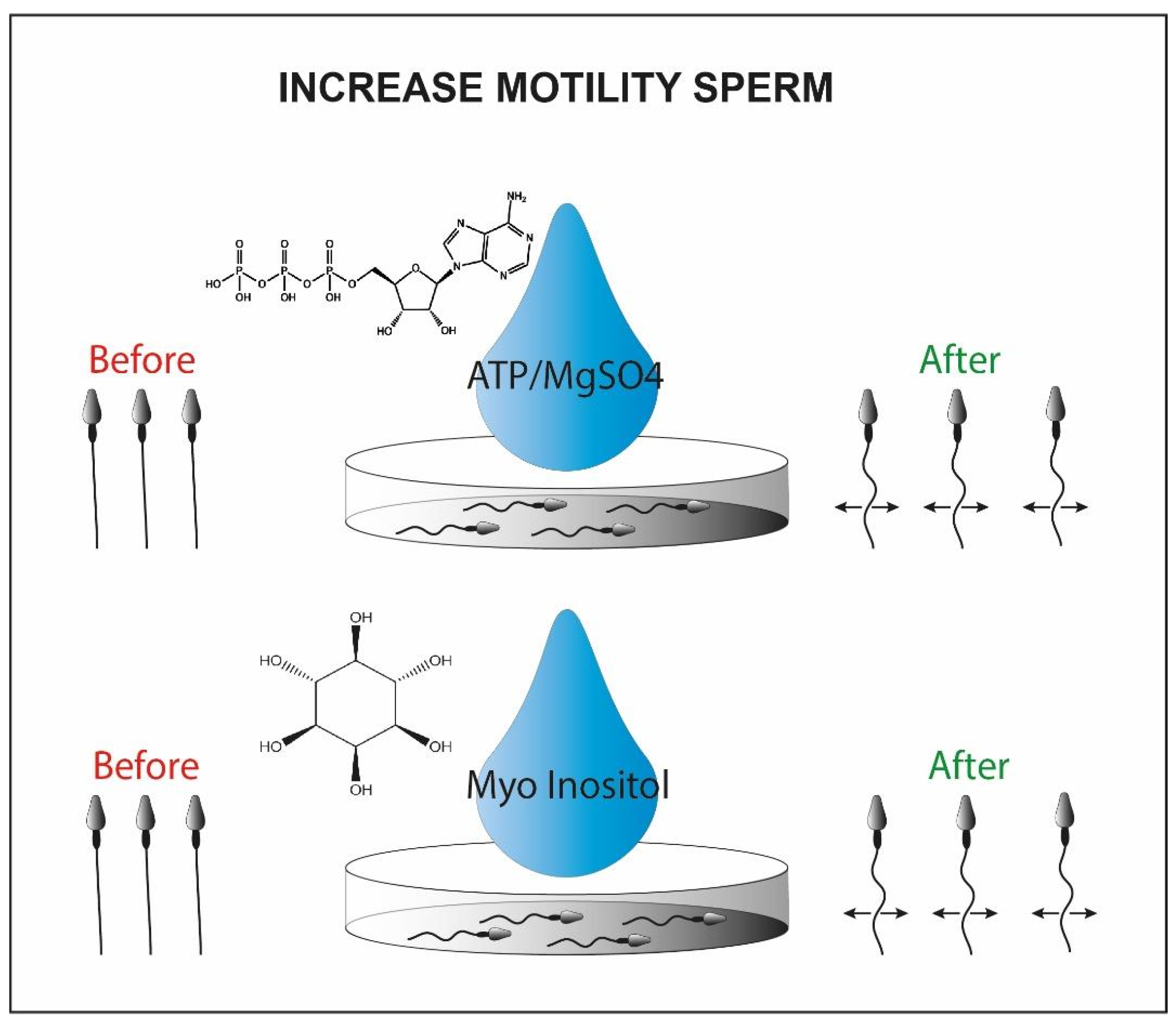
- Abdolsamadi, M.; Mohammadi, F.; Nashtaei, M.S.; Teimouri, M.; Sardar, R.; Dayani, M.; Haghighi, M.; Ghasemi, S.; Vatannejad, A.; Zandieh, Z. Does myoinositol supplement improve sperm parameters and DNA integrity in patients with oligoasthenoteratozoospermia after the freezing-thawing process? Cell Tissue Bank. 2020, 21, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, R.A.; Barbagallo, F.; Calogero, A.E.; Cannarella, R.; Crafa, A.; La Vignera, S. D-Chiro-Inositol Improves Sperm Mitochondrial Membrane Potential: In Vitro Evidence. J. Clin. Med. 2020, 9, 1373. [Google Scholar] [CrossRef] [PubMed]
- Rubino, P.; Palini, S.; Chigioni, S.; Carlomagno, G.; Quagliariello, A.; De Stefani, S.; Baglioni, A.; Bulletti, C. Improving fertilization rate in ICSI cycles by adding myoinositol to the semen preparation procedures: A prospective, bicentric, randomized trial on sibling oocytes. J. Assist. Reprod. Genet. 2015, 32, 387–394. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shan, S.; Xu, F.; Hirschfeld, M.; Brenig, B. Sperm Lipid Markers of Male Fertility in Mammals. Int. J. Mol. Sci. 2021, 22, 8767. [Google Scholar] [CrossRef]
- Hernández-Silva, G.; Fabián López-Araiza, J.E.; López-Torres, A.S.; Larrea, F.; Torres-Flores, V.; Chirinos, M. Proteomic characterization of human sperm plasma membrane-associated proteins and their role in capacitation. Andrology 2020, 8, 171–180. [Google Scholar] [CrossRef]
- Tumova, L.; Zigo, M.; Sutovsky, P.; Sedmikova, M.; Postlerova, P. Ligands and Receptors Involved in the Sperm-Zona Pellucida Interactions in Mammals. Cells 2021, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Nikkhoo, B.; Moradveisi, B.; Allahveisi, A. Effect of sperm selection methods on ICSI outcomes in patients with oligoteratozoospermia. Am. J. Clin. Exp. Urol. 2021, 9, 170–176. [Google Scholar]
- Izadi, M.; Khalili, M.A.; Salehi-Abargouei, A.; Rezvani, M.E.; Aflatoonian, B. Use of zona pellucida-bound spermatozoa as a natural selection in improvement of ICSI outcomes: A systematic review and meta-analysis. Andrologia 2021, 53, e14022. [Google Scholar] [CrossRef]
- Plouffe, B.D.; Murthy, S.K.; Lewis, L.H. Fundamentals and application of magnetic particles in cell isolation and enrichment: A review. Rep. Prog. Phys. 2015, 78, 016601. [Google Scholar] [CrossRef] [PubMed]
- Vermes, I.; Haanen, C.; Steffens-Nakken, H.; Reutelingsperger, C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin, V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef]
- Arends, M.J.; Wyllie, A.H. Apoptosis: Mechanisms and roles in pathology. Int. Rev. Exp. Pathol. 1991, 32, 223–254. [Google Scholar] [CrossRef] [PubMed]
- Tavalaee, M.; Deemeh, M.R.; Arbabian, M.; Nasr-Esfahani, M.H. Density gradient centrifugation before or after magnetic-activated cell sorting: Which technique is more useful for clinical sperm selection? J. Assist. Reprod. Genet. 2012, 29, 31–38. [Google Scholar] [CrossRef]
- Dirican, E.K.; Ozgün, O.D.; Akarsu, S.; Akin, K.O.; Ercan, O.; Uğurlu, M.; Camsari, C.; Kanyilmaz, O.; Kaya, A.; Unsal, A. Clinical outcome of magnetic activated cell sorting of non-apoptotic spermatozoa before density gradient centrifugation for assisted reproduction. J. Assist. Reprod. Genet. 2008, 25, 375–381. [Google Scholar] [CrossRef]
- Dandekar, P.; Aggeler, J.; Talbot, P. Structure, distribution and composition of the extracellular matrix of human oocytes and cumulus masses. Hum. Reprod. 1992, 7, 391–398. [Google Scholar] [CrossRef]
- Parmegiani, L.; Cognigni, G.E.; Ciampaglia, W.; Pocognoli, P.; Marchi, F.; Filicori, M. Efficiency of hyaluronic acid (HA) sperm selection. J. Assist. Reprod. Genet. 2010, 27, 13–16. [Google Scholar] [CrossRef]
- Barak, Y.; Menezo, Y.; Veiga, A.; Elder, K. A physiological replacement for polyvinylpyrrolidone (PVP) in assisted reproductive technology. Hum. Fertil. 2001, 4, 99–103. [Google Scholar] [CrossRef]
- Parmegiani, L.; Cognigni, G.E.; Bernardi, S.; Troilo, E.; Ciampaglia, W.; Filicori, M. “Physiologic ICSI”: Hyaluronic acid (HA) favors selection of spermatozoa without DNA fragmentation and with normal nucleus, resulting in improvement of embryo quality. Fertil. Steril. 2010, 93, 598–604. [Google Scholar] [CrossRef]
- Yanagimachi, R.; Noda, Y.D.; Fujimoto, M.; Nicolson, G.L. The distribution of negative surface charges on mammalian spermatozoa. Am. J. Anat. 1972, 135, 497–519. [Google Scholar] [CrossRef] [PubMed]
- Zarei-Kheirabadi, M.; Shayegan Nia, E.; Tavalaee, M.; Deemeh, M.R.; Arabi, M.; Forouzanfar, M.; Javadi, G.R.; Nasr-Esfahani, M.H. Evaluation of ubiquitin and annexin V in sperm population selected based on density gradient centrifugation and zeta potential (DGC-Zeta). J. Assist. Reprod. Genet. 2012, 29, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, A.; Tavalaee, M.; Deemeh, M.R.; Azadi, L.; Fazilati, M.; Nasr-Esfahani, M.H. Zeta potential vs apoptotic marker: Which is more suitable for ICSI sperm selection? J. Assist. Reprod. Genet. 2013, 30, 1181–1186. [Google Scholar] [CrossRef]
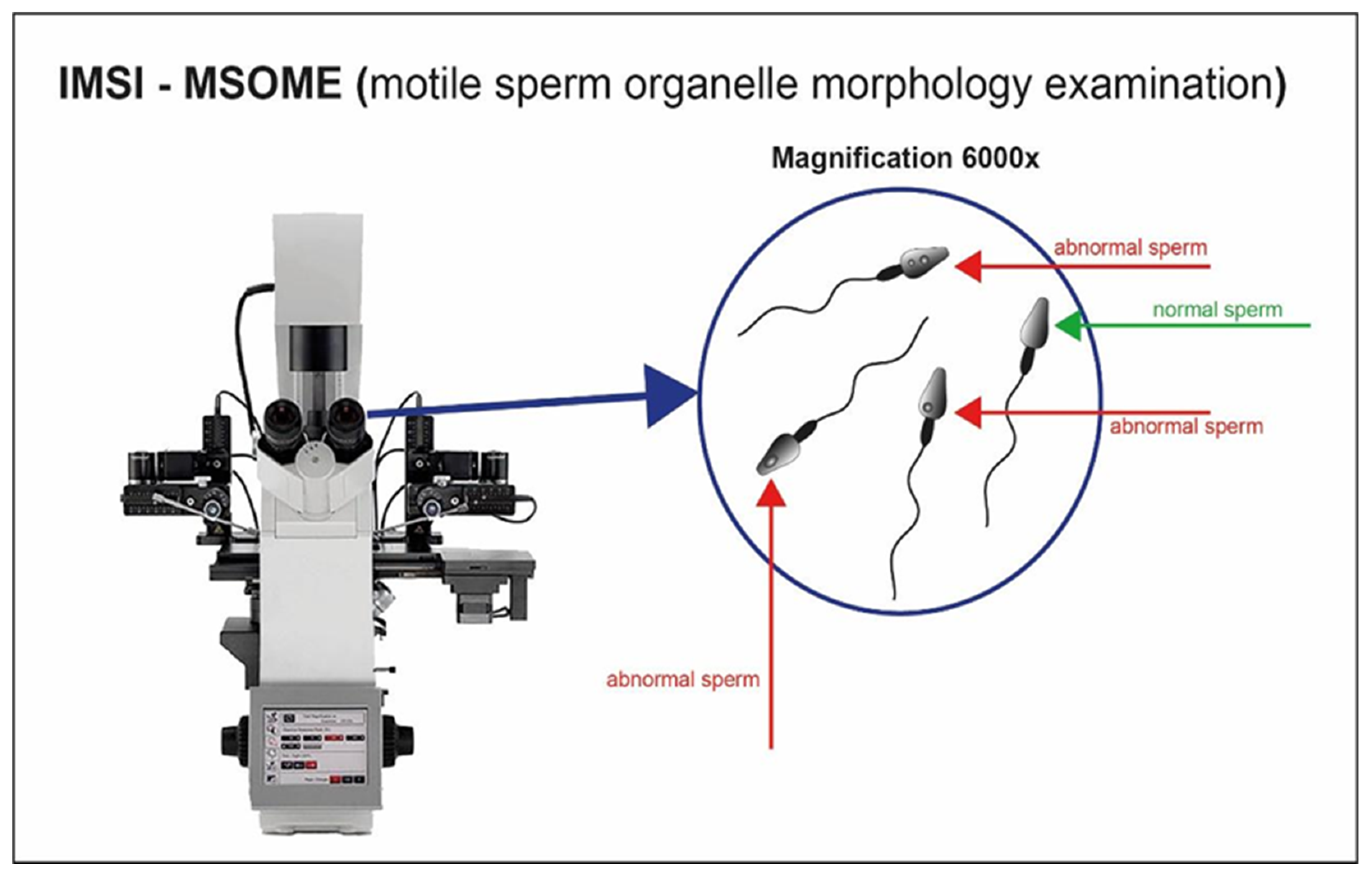
- Bartoov, B.; Berkovitz, A.; Eltes, F. Selection of spermatozoa with normal nuclei to improve the pregnancy rate with intracytoplasmic sperm injection. N. Engl. J. Med. 2001, 345, 1067–1068. [Google Scholar] [CrossRef]
- Lo Monte, G.; Murisier, F.; Piva, I.; Germond, M.; Marci, R. Focus on intracytoplasmic morphologically selected sperm injection (IMSI): A mini-review. Asian J. Androl. 2013, 15, 608–615. [Google Scholar] [CrossRef]
- Franco, J.G., Jr.; Baruffi, R.L.; Mauri, A.L.; Petersen, C.G.; Oliveira, J.B.; Vagnini, L. Significance of large nuclear vacuoles in human spermatozoa: Implications for ICSI. Reprod. Biomed. Online 2008, 17, 42–45. [Google Scholar] [CrossRef]
- Miki, K.; Clapham, D.E. Rheotaxis guides mammalian sperm. Curr. Biol. 2013, 23, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Hino, T.; Yanagimachi, R. Active peristaltic movements and fluid production of the mouse oviduct: Their roles in fluid and sperm transport and fertilization. Biol. Reprod. 2019, 101, 40–49. [Google Scholar] [CrossRef]
- Nagata, M.P.B.; Endo, K.; Ogata, K.; Yamanaka, K.; Egashira, J.; Katafuchi, N.; Yamanouchi, T.; Matsuda, H.; Goto, Y.; Sakatani, M.; et al. Live births from artificial insemination of microfluidic-sorted bovine spermatozoa characterized by trajectories correlated with fertility. Proc. Natl. Acad. Sci. USA 2018, 115, E3087–E3096. [Google Scholar] [CrossRef]
- De Martin, H.; Cocuzza, M.S.; Tiseo, B.C.; Wood, G.J.A.; Miranda, E.P.; Monteleone, P.A.A.; Soares, J.M., Jr.; Serafini, P.C.; Srougi, M.; Baracat, E.C. Positive rheotaxis extended drop: A one-step procedure to select and recover sperm with mature chromatin for intracytoplasmic sperm injection. J. Assist. Reprod. Genet. 2017, 34, 1699–1708. [Google Scholar] [CrossRef] [PubMed]
- Zaferani, M.; Cheong, S.H.; Abbaspourrad, A. Rheotaxis-based separation of sperm with progressive motility using a microfluidic corral system. Proc. Natl. Acad. Sci. USA 2018, 115, 8272–8277. [Google Scholar] [CrossRef]
- Oren-Benaroya, R.; Orvieto, R.; Gakamsky, A.; Pinchasov, M.; Eisenbach, M. The sperm chemoattractant secreted from human cumulus cells is progesterone. Hum. Reprod. 2008, 23, 2339–2345. [Google Scholar] [CrossRef]
- Gatica, L.V.; Guidobaldi, H.A.; Montesinos, M.M.; Teves, M.E.; Moreno, A.I.; Uñates, D.R.; Molina, R.I.; Giojalas, L.C. Picomolar gradients of progesterone select functional human sperm even in subfertile samples. Mol. Hum. Reprod. 2013, 19, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Li, R.; Ni, Y.; Sun, P.; Liu, Y.; Zhang, D.; Huang, H. Novel distance-progesterone-combined selection approach improves human sperm quality. J. Transl. Med. 2018, 16, 203. [Google Scholar] [CrossRef]
- Bahat, A.; Eisenbach, M. Sperm thermotaxis. Mol. Cell Endocrinol. 2006, 252, 115–119. [Google Scholar] [CrossRef]
- Bahat, A.; Tur-Kaspa, I.; Gakamsky, A.; Giojalas, L.C.; Breitbart, H.; Eisenbach, M. Thermotaxis of mammalian sperm cells: A potential navigation mechanism in the female genital tract. Nat Med. 2003, 9, 149–150. [Google Scholar] [CrossRef]
- Bahat, A.; Caplan, S.R.; Eisenbach, M. Thermotaxis of human sperm cells in extraordinarily shallow temperature gradients over a wide range. PLoS ONE 2012, 7, e41915. [Google Scholar] [CrossRef]
- Pérez-Cerezales, S.; Laguna-Barraza, R.; de Castro, A.C.; Sánchez-Calabuig, M.J.; Cano-Oliva, E.; de Castro-Pita, F.J.; Montoro-Buils, L.; Pericuesta, E.; Fernández-González, R.; Gutiérrez-Adán, A. Sperm selection by thermotaxis improves ICSI outcome in mice. Sci. Rep. 2018, 8, 2902. [Google Scholar] [CrossRef]
- Sackmann, E.K.; Fulton, A.L.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Shirota, K.; Yotsumoto, F.; Itoh, H.; Obama, H.; Hidaka, N.; Nakajima, K.; Miyamoto, S. Separation efficiency of a microfluidic sperm sorter to minimize sperm DNA damage. Fertil. Steril. 2016, 105, 315–321.e1. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Xie, L.; Han, C.; Su, K.; Qiu, T.; Wang, L.; Huang, G.; Xing, W.; Qiao, J.; Wang, J.; et al. In vitro fertilization on a single-oocyte positioning system integrated with motile sperm selection and early embryo development. Anal. Chem. 2011, 83, 2964–2970. [Google Scholar] [CrossRef]
- Simon, L.; Murphy, K.; Aston, K.I.; Emery, B.R.; Hotaling, J.M.; Carrell, D.T. Optimization of microelectrophoresis to select highly negatively charged sperm. J. Assist. Reprod. Genet. 2016, 33, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.J.; Maeng, J.H.; Hwang, S.Y.; Ahn, Y. Design, Fabrication, and Testing of a Microfluidic Device for Thermotaxis and Chemotaxis Assays of Sperm. SLAS Technol. 2018, 23, 507–515. [Google Scholar] [CrossRef]
- Bhagwat, S.; Sontakke, S.K.D.; Parte, P.; Jadhav, S. Chemotactic behavior of spermatozoa captured using a microfluidic chip. Biomicrofluidics 2018, 12, 024112. [Google Scholar] [CrossRef]
- Ferraz, M.A.M.M.; Rho, H.S.; Hemerich, D.; Henning, H.H.W.; van Tol, H.T.A.; Hölker, M.; Besenfelder, U.; Mokry, M.; Vos, P.L.A.M.; Stout, T.A.E.; et al. An oviduct-on-a-chip provides an enhanced in vitro environment for zygote genome reprogramming. Nat. Commun. 2018, 9, 4934. [Google Scholar] [CrossRef]
- Eravuchira, P.J.; Mirsky, S.K.; Barnea, I.; Levi, M.; Balberg, M.; Shaked, N.T. Individual sperm selection by microfluidics integrated with interferometric phase microscopy. Methods 2018, 136, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Amaral, S.; Da Costa, R.; Wübbeling, F.; Redmann, K.; Schlatt, S. Raman micro-spectroscopy analysis of different sperm regions: A species comparison. Mol. Hum. Reprod. 2018, 24, 185–202. [Google Scholar] [CrossRef]
- Di Caprio, G.; Ferrara, M.A.; Miccio, L.; Merola, F.; Memmolo, P.; Ferraro, P.; Coppola, G. Holographic imaging of unlabelled sperm cells for semen analysis: A review. J. Biophotonics 2015, 8, 779–789. [Google Scholar] [CrossRef]
- de Wagenaar, B.; Geijs, D.J.; de Boer, H.; Bomer, J.G.; Olthuis, W.; van den Berg, A.; Segerink, L.I. Spermometer: Electrical characterization of single boar sperm motility. Fertil. Steril. 2016, 106, 773–780.e6. [Google Scholar] [CrossRef]

| Procedures | Advantages | Disadvantages |
|---|---|---|
| Swim-up | ||
| Centrifugation on density gradient | ||
| HOST | ||
| Polarization microscopy |
| |
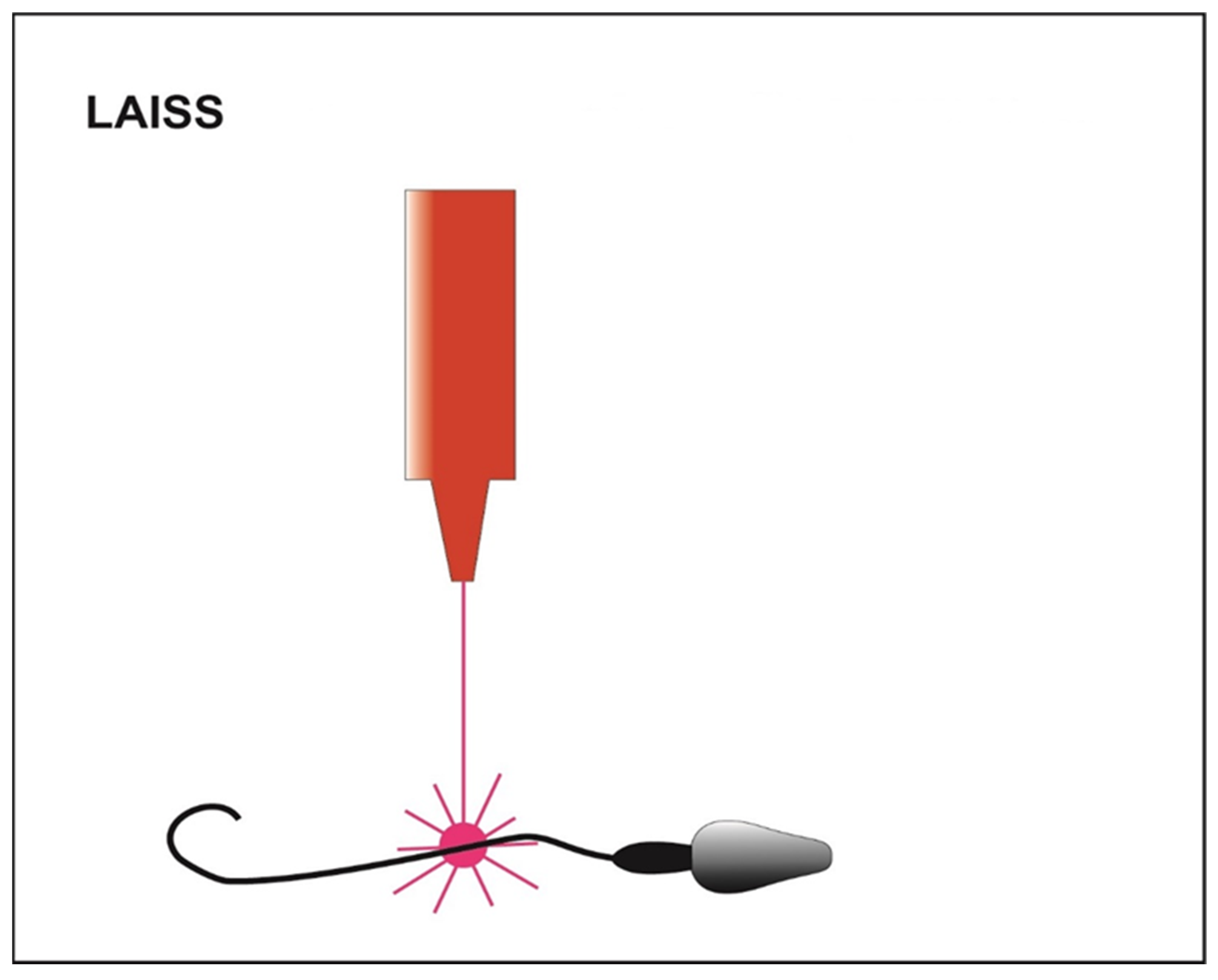
| LAISS | ||
| MACS |
|
|
| PICSI and selection with hyaluronic acid |
|
|
| Zeta potential | ||
| IMSI |
| |
| Microfluidic separation |
| |
| Horizontal Sperm migration |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldini, D.; Ferri, D.; Baldini, G.M.; Lot, D.; Catino, A.; Vizziello, D.; Vizziello, G. Sperm Selection for ICSI: Do We Have a Winner? Cells 2021, 10, 3566. https://doi.org/10.3390/cells10123566
Baldini D, Ferri D, Baldini GM, Lot D, Catino A, Vizziello D, Vizziello G. Sperm Selection for ICSI: Do We Have a Winner? Cells. 2021; 10(12):3566. https://doi.org/10.3390/cells10123566
Chicago/Turabian StyleBaldini, Domenico, Daniele Ferri, Giorgio Maria Baldini, Dario Lot, Assunta Catino, Damiano Vizziello, and Giovanni Vizziello. 2021. "Sperm Selection for ICSI: Do We Have a Winner?" Cells 10, no. 12: 3566. https://doi.org/10.3390/cells10123566
APA StyleBaldini, D., Ferri, D., Baldini, G. M., Lot, D., Catino, A., Vizziello, D., & Vizziello, G. (2021). Sperm Selection for ICSI: Do We Have a Winner? Cells, 10(12), 3566. https://doi.org/10.3390/cells10123566






