Low Dose of IL-2 Normalizes Hypertension and Mitochondrial Function in the RUPP Rat Model of Placental Ischemia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation of Mitochondria
2.2. Mitochondrial Respiration
2.3. Mitochondrial ROS
2.4. Endothelial Mitochondrial ROS
2.5. IL-2 Cytokine Profile
2.6. Statistical Analysis
3. Results
3.1. IL-2 Significantly Lowered Blood Pressure in RUPP Rats
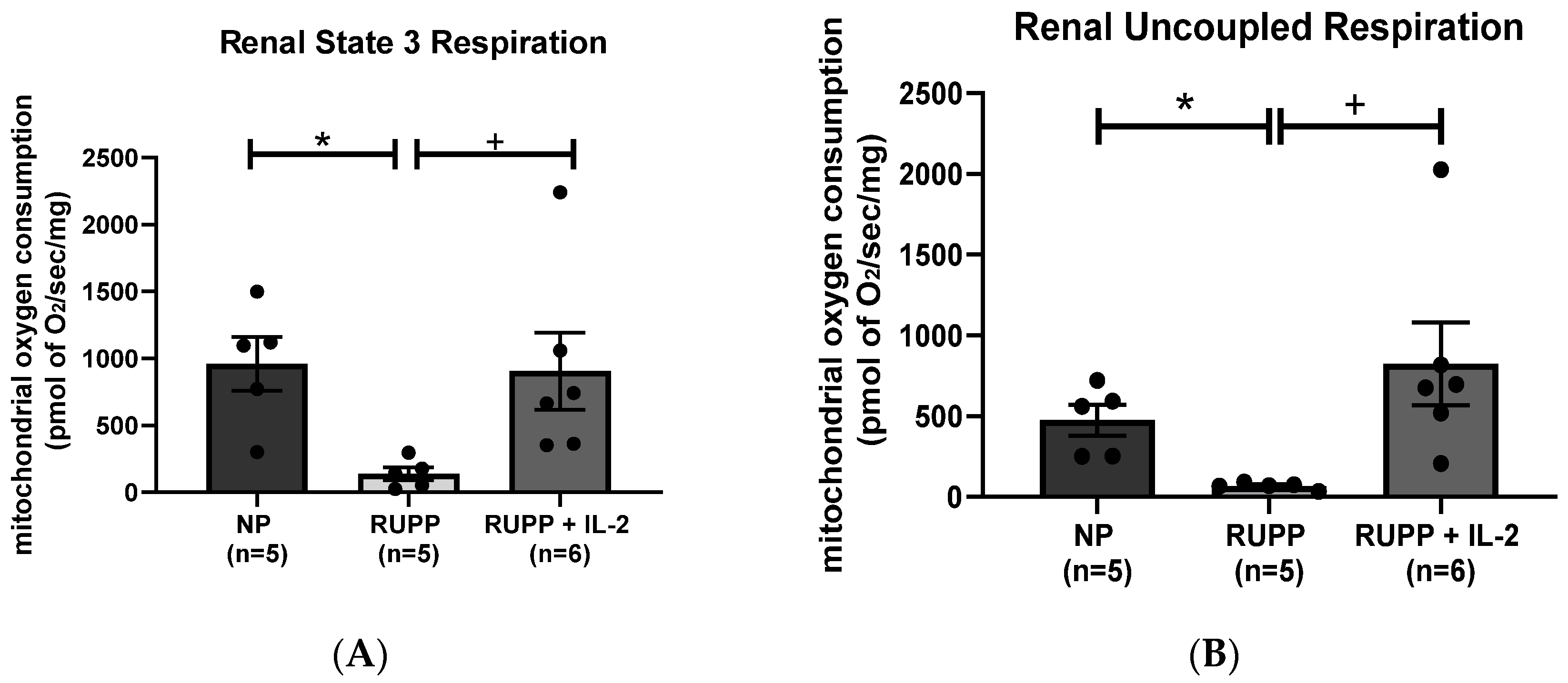
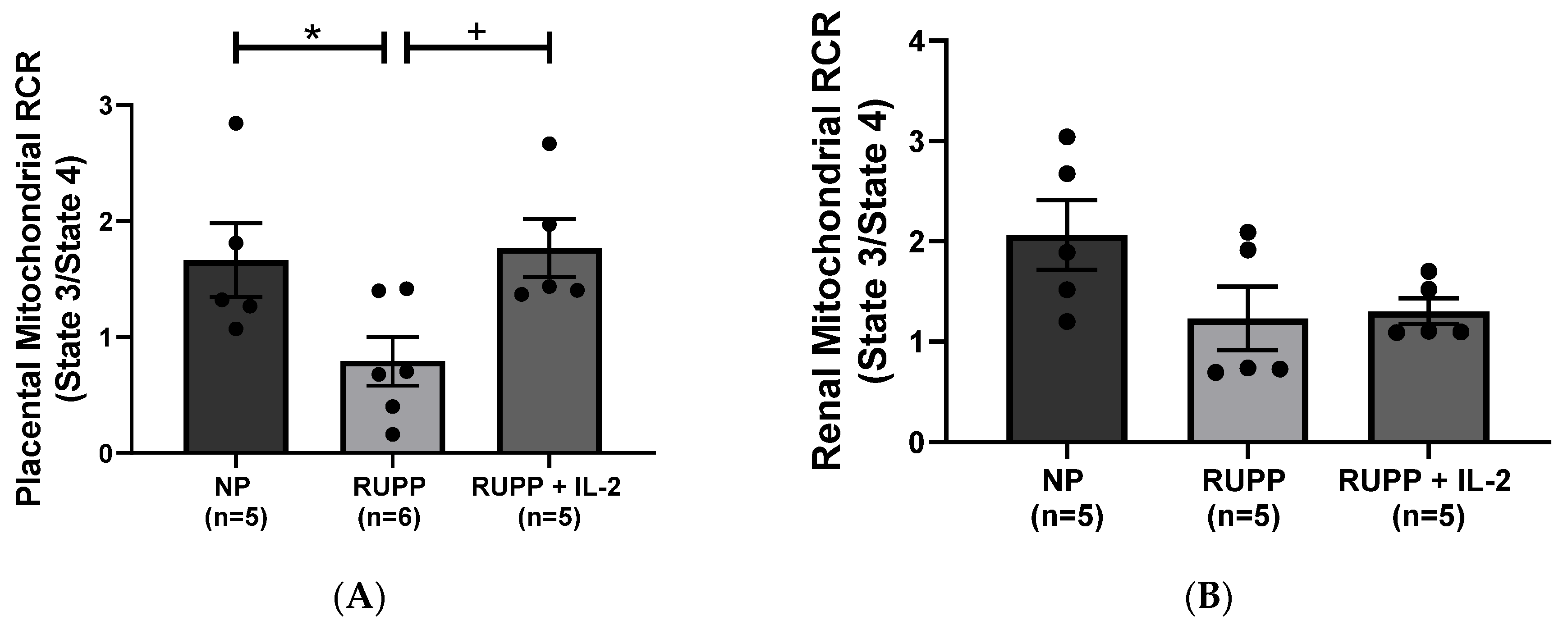
3.2. IL-2 Significantly Improved Multi-Organ mt Function in RUPP Rats
3.3. Serum IL-2 Levels Are Increased in PE
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Herse, F.; LaMarca, B. Angiotensin II Type 1 Receptor Autoantibody (AT1-AA)-Mediated Pregnancy Hypertension. Am. J. Reprod. Immunol. 2013, 69, 413–418. [Google Scholar] [CrossRef] [Green Version]
- Cornelius, D.C.; Cottrell, J.; Amaral, L.M.; Lamarca, B. Inflammatory mediators: A causal link to hypertension during preeclampsia. Br. J. Pharmacol. 2019, 176, 1914–1921. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ye, Y.; Zhang, J.; Ruan, C.-C.; Gao, P.-J. Immune imbalance is associated with the development of preeclampsia. Medicine 2019, 98, e15080. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, M.W., Jr.; Vaka, V.R.; McMaster, K.; Ibrahim, T.; Cornelius, D.C.; Amaral, L.; Campbell, N.; Wallukat, G.; McDuffy, S.; Usry, N.; et al. Renal natural killer cell activation and mitochondrial oxidative stress; new mechanisms in AT1-AA mediated hypertensive pregnancy. Pregnancy Hypertens. 2019, 15, 72–77. [Google Scholar] [CrossRef] [PubMed]
- LaMarca, B.; Parrish, M.R.; Wallace, K. Agonistic Autoantibodies to the Angiotensin II Type I Receptor Cause Pathophysiologic Characteristics of Preeclampsia. Gend. Med. 2012, 9, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaka, V.R.; McMaster, K.M.; Cunningham, M.W., Jr.; Ibrahim, T.; Hazlewood, R.; Usry, N.; Cornelius, D.C.; Amaral, L.M.; LaMarca, B. Role of mitochondrial dysfunction and reactive oxygen species in mediating hypertension in the reduced uterine perfusion pressure rat model of preeclampsia. Hypertension 2018, 72, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Novotny, S.R.; Wallace, K.; Heath, J.; Moseley, J.; Dhillon, P.; Weimer, A.; Wallukat, G.; Herse, F.; Wenzel, K.; Martin, J.N., Jr.; et al. Activating autoantibodies to the angiotensin II type I receptor play an important role in mediating hypertension in response to adoptive transfer of CD4+ T lymphocytes from placental ischemic rats. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2012, 302, R1197–R1201. [Google Scholar] [CrossRef] [Green Version]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Aranguren, L.C.; Prada, C.E.; Riaño-Medina, C.E.; Lopez, M. Endothelial dysfunction and preeclampsia: Role of oxidative stress. Front. Physiol. 2014, 5, 372. [Google Scholar] [CrossRef] [Green Version]
- Jeffery, H.C.; Jeffery, L.E.; Lutz, P.; Corrigan, M.; Webb, G.J.; Hirschfield, G.; Adams, D.; Oo, Y.H. Low-dose interleukin-2 promotes STAT-5 phosphorylation, Treg survival and CTLA-4-dependent function in autoimmune liver diseases. Clin. Exp. Immunol. 2017, 188, 394–411. [Google Scholar] [CrossRef] [Green Version]
- Zhao, T.X.; Kostapanos, M.; Griffiths, C.; Arbon, E.L.; Hubsch, A.; Kaloyirou, F.; Helmy, J.; Hoole, S.P.; Rudd, J.H.F.; Wood, G.; et al. Low-dose interleukin-2 in patients with stable ischaemic heart disease and acute coronary syndromes (LILACS): Protocol and study rationale for a randomised, double-blind, placebo-controlled, phase I/II clinical trial. BMJ Open 2018, 8, e022452. [Google Scholar] [CrossRef] [PubMed]
- Seelig, E.; Howlett, J.; Porter, L.; Truman, L.; Heywood, J.; Kennet, J.; Arbon, E.L.; Anselmiova, K.; Walker, N.M.; Atkar, R.; et al. The DILfrequency study is an adaptive trial to identify optimal IL-2 dosing in patients with type 1 diabetes. JCI Insight 2018, 3, e99306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fishman, M.; Dutcher, J.P.; Clark, J.I.; Alva, A.; Miletello, G.P.; Curti, B.; Agarwal, N.; Hauke, R.; Mahoney, K.M.; Moon, H.; et al. Overall survival by clinical risk category for high dose interleukin-2 (HD IL-2) treated patients with metastatic renal cell cancer (mRCC): Data from the PROCLAIM(SM) registry. J. Immunother. Cancer 2019, 7, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cunningham, M.W.; Amaral, L.M.; Campbell, N.E.; Cornelius, D.C.; Ibrahim, T.; Vaka, V.R.; LaMarca, B. Investigation of interleukin-2-mediated changes in blood pressure, fetal growth restriction, and innate immune activation in normal pregnant rats and in a preclinical rat model of preeclampsia. Biol. Sex Differ. 2021, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Vaka, V.R.; Cunningham, M.W.; Deer, E.; Franks, M.; Ibrahim, T.; Amaral, L.M.; Usry, N.; Cornelius, D.C.; Dechend, R.; Wallukat, G.; et al. Blockade of endogenous angiotensin II type I receptor agonistic autoantibody activity improves mitochondrial reactive oxygen species and hypertension in a rat model of preeclampsia. Am. J. Physiol. Integr. Comp. Physiol. 2020, 318, R256–R262. [Google Scholar] [CrossRef] [PubMed]
- Rebelato, H.J.; Esquisatto, M.A.M.; Moraes, C.; Amaral, M.E.C.; Catisti, R. Gestational protein restriction induces alterations in placental morphology and mitochondrial function in rats during late pregnancy. J. Mol. Histol. 2013, 44, 629–637. [Google Scholar] [CrossRef] [PubMed]
- LaMarca, B.B.D.; Cockrell, K.; Sullivan, E.; Bennett, W.; Granger, J.P. Role of Endothelin in Mediating Tumor Necrosis Factor-Induced Hypertension in Pregnant Rats. Hypertension 2005, 46, 82–86. [Google Scholar] [CrossRef] [Green Version]
- Beyramzadeh, M.; Dikmen, Z.G.; Erturk, N.K.; Tuncer, Z.S.; Akbiyik, F. Placental respiratory chain complex activities in high risk pregnancies. J. Matern. Neonatal Med. 2017, 30, 2911–2917. [Google Scholar] [CrossRef]
- Illsinger, S.; Janzen, N.; Sander, S.; Schmidt, K.-H.; Bednarczyk, J.; Mallunat, L.; Bode, J.; Hagebölling, F.; Hoy, L.; Lücke, T.; et al. Preeclampsia and HELLP Syndrome: Impaired Mitochondrial Function in Umbilical Endothelial Cells. Reprod. Sci. 2010, 17, 219–226. [Google Scholar] [CrossRef]
- Mando’, C.; Marino, M.; Miriam, F.; De Palma, C.; Borelli, M.; Trabattoni, D.; Stampalija, T.; Ferrazzi, E.; Clementi, E.; Cetin, I. OS048 Mitochondrial content and function in placental cells and tissuesof preeclampsia and IUGR. Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health 2012, 2, 203. [Google Scholar] [CrossRef]
- Matsubara, S.; Minakami, H.; Sato, I.; Saito, T. Decrease in cytochrome c oxidase activity detected cytochemically in the placental trophoblast of patients with pre-eclampsia. Placenta 1997, 18, 255–259. [Google Scholar] [CrossRef]
- Rodríguez, J.C.M.; Mendoza, P.Y.; Bernal, M.P.; Acosta, J.L.A. Oxidative stress level and placental histological changes during preeclampsia. Ginecol. Obstet. Mex. 2008, 76, 319–326. [Google Scholar]
- Deer, E.; Vaka, V.R.; McMaster, K.M.; Wallace, K.; Cornelius, D.C.; Amaral, L.M.; Cunningham, M.W.; LaMarca, B. Vascular endothelial mitochondrial oxidative stress in response to preeclampsia: A role for angiotension II type 1 autoantibodies. Am. J. Obstet. Gynecol. MFM 2021, 3, 100275. [Google Scholar] [CrossRef] [PubMed]
- Shibata, E.; Nanri, H.; Ejima, K.; Araki, M.; Fukuda, J.; Yoshimura, K.; Toki, N.; Ikeda, M.; Kashimura, M. Enhancement of Mitochondrial Oxidative Stress and Up-regulation of Antioxidant Protein Peroxiredoxin III/SP-22 in the Mitochondria of Human Pre-eclamptic Placentae. Placenta 2003, 24, 698–705. [Google Scholar] [CrossRef]
- Shi, Z.; Long, W.; Zhao, C.; Guo, X.; Shen, R.; Ding, H. Comparative Proteomics Analysis Suggests that Placental Mitochondria are Involved in the Development of Pre-Eclampsia. PLoS ONE 2013, 8, e64351. [Google Scholar] [CrossRef] [Green Version]
- Shanklin, D.R.; Sibai, B.M. Ultrastructural aspects of preeclampsia: II. Mitochondrial changes. Am. J. Obstet. Gynecol. 1990, 163, 943–953. [Google Scholar] [CrossRef]
- He, L.; Wang, Z.; Sun, Y. Reduced amount of cytochrome c oxidase subunit I messenger RNA in placentas from pregnancies complicated by preeclampsia. Acta Obstet. Gynecol. Scand. 2004, 83, 144–148. [Google Scholar] [CrossRef]
- McCarthy, C.M.; Kenny, L.C. Therapeutically targeting mitochondrial redox signalling alleviates endothelial dysfunction in preeclampsia. Sci. Rep. 2016, 6, 32683. [Google Scholar] [CrossRef] [Green Version]
- Deer, E.; Reeve, K.E.; Amaral, L.M.; Vaka, V.R.; Franks, M.; Campbell, N.; Fitzgerald, S.; Herrock, O.T.; Ibrahim, T.; Cornelius, D.C.; et al. CD4+ T cells cause renal and placental mitochondrial oxidative stress as mechanisms of hypertension in response to placental ischemia. Am. J. Physiol. Physiol. 2021, 320, F47–F54. [Google Scholar] [CrossRef]
- Ye, C.; Brand, D.; Zheng, S.G. Targeting IL-2: An unexpected effect in treating immunological diseases. Signal Transduct. Target. Ther. 2018, 3, 2. [Google Scholar] [CrossRef] [Green Version]
- Szmyrka-Kaczmarek, M.; Kosmaczewska, A.; Ciszak, L.; Szteblich, A.; Wiland, P. Peripheral blood Th17/Treg imbalance in patients with low-active systemic lupus erythematosus. Postęp. Hig. Med. Dośw. 2014, 68, 893–898. [Google Scholar] [CrossRef]
- Goldstein, J.D.; Pérol, L.; Zaragoza, B.; Baeyens, A.; Marodon, G.; Piaggio, E. Role of Cytokines in Thymus- Versus Peripherally Derived-Regulatory T Cell Differentiation and Function. Front. Immunol. 2013, 4, 155. [Google Scholar] [CrossRef] [Green Version]
- Brusko, T.M.; Wasserfall, C.; Clare-Salzler, M.J.; Schatz, D.A.; Atkinson, M.A. Functional Defects and the Influence of Age on the Frequency of CD4+CD25+ T-Cells in Type 1 Diabetes. Diabetes 2005, 54, 1407–1414. [Google Scholar] [CrossRef] [Green Version]
- Xing, Q.; Wang, B.; Su, H.; Cui, J.; Li, J. Elevated Th17 cells are accompanied by FoxP3+ Treg cells decrease in patients with lupus nephritis. Rheumatol. Int. 2012, 32, 949–958. [Google Scholar] [CrossRef]
- Sgouroudis, E.; Kornete, M.; Piccirillo, C.A. IL-2 production by dendritic cells promotes Foxp3+regulatory T-cell expansion in autoimmune-resistant NOD congenic mice. Autoimmunity 2011, 44, 406–414. [Google Scholar] [CrossRef]
- Zhang, H.; Chua, K.S.; Guimond, M.; Kapoor, V.; Brown, M.V.; A Fleisher, T.; Long, L.M.; Bernstein, D.; Hill, B.J.; Douek, D.C.; et al. Lymphopenia and interleukin-2 therapy alter homeostasis of CD4+CD25+ regulatory T cells. Nat. Med. 2005, 11, 1238–1243. [Google Scholar] [CrossRef]
- Matsuoka, K.I.; Koreth, J.; Kim, H.T.; Bascug, G.; McDonough, S.; Kawano, Y.; Murase, K.; Cutler, C.; Ho, V.T.; Alyea, E.P.; et al. Low-dose interleukin-2 therapy restores regulatory T cell homeostasis in patients with chronic graft-versus-host disease. Sci. Transl. Med. 2013, 5, 179ra43. [Google Scholar] [CrossRef] [Green Version]
- Koreth, J.; Matsuoka, K.-I.; Kim, H.T.; McDonough, S.M.; Bindra, B.; Alyea, E.P.; Armand, P.; Cutler, C.; Ho, V.T.; Treister, N.S.; et al. Interleukin-2 and Regulatory T Cells in Graft-versus-Host Disease. N. Engl. J. Med. 2011, 365, 2055–2066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saadoun, D.; Rosenzwajg, M.; Joly, F.; Six, A.; Carrat, F.; Thibault, V.; Sene, D.; Cacoub, P.; Klatzmann, D. Regulatory T-Cell Responses to Low-Dose Interleukin-2 in HCV-Induced Vasculitis. N. Engl. J. Med. 2011, 365, 2067–2077. [Google Scholar] [CrossRef]
- Ito, S.; Bollard, C.M.; Carlsten, M.; Melenhorst, J.J.; Biancotto, A.; Wang, E.; Chen, J.; Kotliarov, Y.; Cheung, F.; Xie, Z.; et al. Ultra-low Dose Interleukin-2 Promotes Immune-modulating Function of Regulatory T Cells and Natural Killer Cells in Healthy Volunteers. Mol. Ther. 2014, 22, 1388–1395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartemann, A.; Bensimon, G.; Payan, C.A.; Jacqueminet, S.; Bourron, O.; Nicolas, N.; Fonfrede, M.; Rosenzwajg, M.; Pharm, C.B.; Klatzmann, D. Low-dose interleukin 2 in patients with type 1 diabetes: A phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2013, 1, 295–305. [Google Scholar] [CrossRef]
- Von Spee-Mayer, C.; Siegert, E.; Abdirama, D.; Rose, A.; Klaus, A.; Alexander, T.; Enghard, P.; Sawitzk, B.; Hiepe, F.; Radbruch, A.; et al. Low-dose interleukin-2 selectively corrects regulatory T cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2016, 75, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Ishimitsu, T.; Uehara, Y.; Numabe, A.; Tsukada, H.; Ogawa, Y.; Yagi, S. Antihypertensive effect of interleukin-2 in salt-sensitive Dahl rats. Hypertension 1994, 23, 68–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majeed, B.; Tawinwung, S.; Eberson, L.S.; Secomb, T.W.; Larmonier, N.; Larson, D.F. Interleukin-2/Anti-Interleukin-2 Immune Complex Expands Regulatory T Cells and Reduces Angiotensin II-Induced Aortic Stiffening. Int. J. Hypertens. 2014, 2014, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Dwyer, C.; Ward, N.C.; Pugliese, A.; Malek, T.R. Promoting Immune Regulation in Type 1 Diabetes Using Low-Dose Interleukin-2. Curr. Diabetes Rep. 2016, 16, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Bluestone, J.A.; Herold, K.; Eisenbarth, G. Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nat. Cell Biol. 2010, 464, 1293–1300. [Google Scholar] [CrossRef] [Green Version]
- Van Belle, T.L.; Coppieters, K.T.; Von Herrath, M.G. Type 1 diabetes: Etiology, immunology, and therapeutic strategies. Physiol. Rev. 2011, 91, 79–118. [Google Scholar] [CrossRef]
- Jørgensen, N.; Persson, G.; Hviid, T.V.F. The Tolerogenic Function of Regulatory T Cells in Pregnancy and Cancer. Front. Immunol. 2019, 10, 911. [Google Scholar] [CrossRef] [Green Version]
- Shields, C.A.; McCalmon, M.; Ibrahim, T.; White, D.L.; Williams, J.M.; Lamarca, B.D.; Cornelius, D.C. Placental ischemia-stimulated T-helper 17 cells induce preeclampsia-associated cytolytic natural killer cells during pregnancy. Am. J. Physiol. Integr. Comp. Physiol. 2018, 315, R336–R343. [Google Scholar] [CrossRef]
- Marzi, M.; Vigano, A.; Trabattoni, D.; Villa, M.L.; Salvaggio, A.; Clerici, M. Characterization of type 1 and type 2 cytokine production profile in physiologic and pathologic human pregnancy. Clin. Exp. Immunol. 1996, 106, 127–133. [Google Scholar] [CrossRef]
- Calleja-Agius, J.; Brincat, M.P. Recurrent miscarriages: What is the role of cytokines? Gynecol. Endocrinol. 2008, 24, 663–668. [Google Scholar] [CrossRef]
- Raghupathy, R. Cytokines as Key Players in the Pathophysiology of Preeclampsia. Med. Princ. Pract. 2013, 22, 8–19. [Google Scholar] [CrossRef]
- Cornish, G.; Sinclair, L.V.; Cantrell, D. Differential regulation of T-cell growth by IL-2 and IL-15. Blood 2006, 108, 600–608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maynard, S.E.; Min, J.-Y.; Merchan, J.; Lim, K.-H.; Li, J.; Mondal, S.; Libermann, T.A.; Morgan, J.P.; Sellke, F.W.; Stillman, I.E.; et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J. Clin. Investig. 2003, 111, 649–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsubara, K.; Matsubara, Y.; Hyodo, S.; Katayama, T.; Ito, M. Role of nitric oxide and reactive oxygen species in the pathogenesis of preeclampsia. J. Obstet. Gynaecol. Res. 2010, 36, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.L.; Brockman, D.; Campos, B.; Myatt, L. Expression of NADPH oxidase isoform 1 (Nox1) in human placenta: Involvement in preeclampsia. Placenta 2006, 27, 422–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Animal Group | Placental Weight (g) | Fetal Weight (g) | % Reabsorptions | % Survived |
|---|---|---|---|---|
| NP | 0.66 ± 0.04 | 2.27 ± 0.05 | 0 ± 0 | 100 ± 0 |
| RUPP | 0.53 ± 0.03 * | 1.99 ± 0.07 * | 19.6 ± 4 * | 80.4 ± 4 * |
| RUPP + LD IL-2 | 0.50 ± 0.02 + | 1.95 ± 0.08 + | 0.43 ± 0.1 # | 99.5 ± 0.1 # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deer, E.; Amaral, L.M.; Campbell, N.; Fitzgerald, S.; Herrock, O.; Ibrahim, T.; LaMarca, B. Low Dose of IL-2 Normalizes Hypertension and Mitochondrial Function in the RUPP Rat Model of Placental Ischemia. Cells 2021, 10, 2797. https://doi.org/10.3390/cells10102797
Deer E, Amaral LM, Campbell N, Fitzgerald S, Herrock O, Ibrahim T, LaMarca B. Low Dose of IL-2 Normalizes Hypertension and Mitochondrial Function in the RUPP Rat Model of Placental Ischemia. Cells. 2021; 10(10):2797. https://doi.org/10.3390/cells10102797
Chicago/Turabian StyleDeer, Evangeline, Lorena M. Amaral, Nathan Campbell, Sarah Fitzgerald, Owen Herrock, Tarek Ibrahim, and Babbette LaMarca. 2021. "Low Dose of IL-2 Normalizes Hypertension and Mitochondrial Function in the RUPP Rat Model of Placental Ischemia" Cells 10, no. 10: 2797. https://doi.org/10.3390/cells10102797
APA StyleDeer, E., Amaral, L. M., Campbell, N., Fitzgerald, S., Herrock, O., Ibrahim, T., & LaMarca, B. (2021). Low Dose of IL-2 Normalizes Hypertension and Mitochondrial Function in the RUPP Rat Model of Placental Ischemia. Cells, 10(10), 2797. https://doi.org/10.3390/cells10102797






