Chronic Inflammation and Radiation-Induced Cystitis: Molecular Background and Therapeutic Perspectives
Abstract
1. Introduction
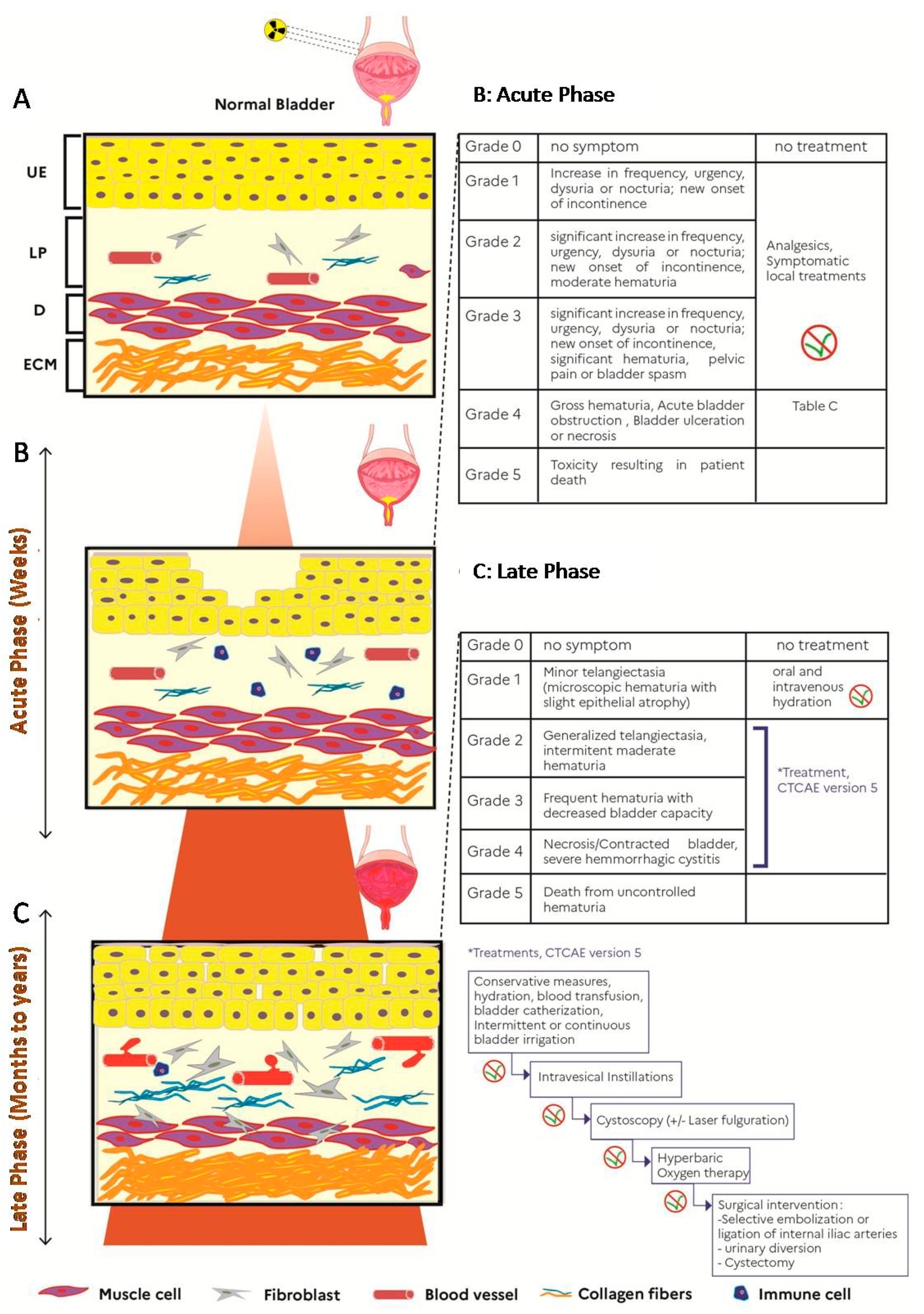
2. Background Information
2.1. Acute Radiation Cystitis
2.2. Late Radiation Cystitis
3. Current Treatments and Clinical Trials
3.1. Acute and Late Radiation Cystitis with Storage, Voiding Symptoms or Occasional Bleeding
3.2. Late Radiation Cystitis with Persistent or Recurrent Hematuria
3.2.1. Intravesical Instillations
3.2.2. Hyperbaric Oxygen Therapy (HBOT)
3.3. Late Radiation Cystitis with Refractory or Life-Threatening Hematuria
3.3.1. Arterial Embolization
3.3.2. Cystectomy and Urinary Diversion
4. Clinical Trials: Other Therapeutic Avenues, Antifibrotics
4.1. Antifibrotic or Antioxidant Pharmacological Agents
4.2. Angiotensin-Converting-Enzyme Inhibitors
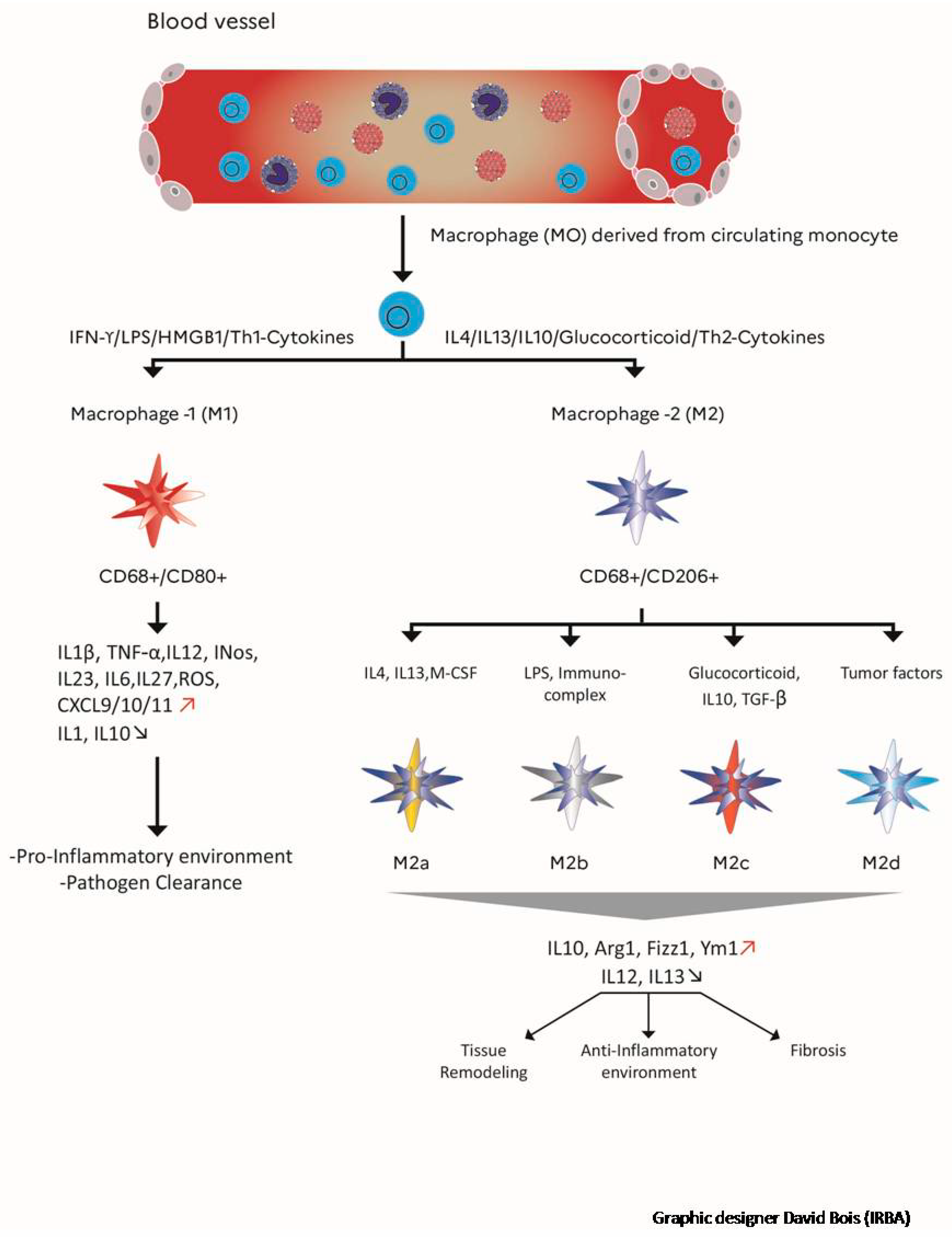
5. Impact of Macrophages in the Development of Radiation Fibrosis
6. Preclinical Studies of Radiation Cystitis and Cell Therapy: A New Therapeutic Avenue
6.1. Preclinical Studies of Radiation-Induced Cystitis
6.2. Stem Cell Therapy: A New Therapeutic Avenue
7. Discussion and Conclusions
Funding
Conflicts of Interest
References
- Rehailia-Blanchard, A.; He, M.Y.; Rancoule, C.; Guillaume, É.; Guy, J.-B.; Vial, N.; Nivet, A.; Orliac, H.; Chargari, C.; Magné, N. Medical prevention and treatment of radiation-induced urological and nephrological complications. Cancer Radiother. J. Soc. Fr. Radiother. Oncol. 2019, 23, 151–160. [Google Scholar]
- Martin, S.E.; Begun, E.M.; Samir, E.; Azaiza, M.T.; Allegro, S.; Abdelhady, M. Incidence and Morbidity of Radiation-Induced Hemorrhagic Cystitis in Prostate Cancer. Urology 2019, 131, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Freites-Martinez, A.; Santana, N.; Arias-Santiago, S.; Viera, A. CTCAE versión 5.0. Evaluación de la gravedad de los eventos adversos dermatológicos de las terapias antineoplásicas. Actas Dermo-Sifiliográficas 2020. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, J.; Hetet, J.-F.; Bouchot, O. Management of radiation cystitis. Progres. En. Urol. J. Assoc. Fr. Urol. Soc. Fr. Urol. 2004, 14, 568–572. [Google Scholar]
- Smit, S.G.; Heyns, C.F. Management of radiation cystitis. Nat. Rev. Urol. 2010, 7, 206–214. [Google Scholar] [CrossRef]
- Mazeron, R.; Castelnau-Marchand, P.; Dumas, I.; Del Campo, E.R.; Kom, L.K.; Martinetti, F.; Farha, G.; Tailleur, A.; Morice, P.; Chargari, C.; et al. Impact of treatment time and dose escalation on local control in locally advanced cervical cancer treated by chemoradiation and image-guided pulsed-dose rate adaptive brachytherapy. Radiother. Oncol. 2015, 114, 257–263. [Google Scholar] [CrossRef]
- Zwaans, B.M.M.; Chancellor, M.B.; Lamb, L.E. Modeling and Treatment of Radiation Cystitis. Urology 2016, 88, 14–21. [Google Scholar] [CrossRef]
- Cox, J.D.; Stetz, J.; Pajak, T.F. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1341–1346. [Google Scholar] [CrossRef]
- Denton, A.S.; Clarke, N.W.; Maher, E.J. Non-surgical interventions for late radiation cystitis in patients who have received radical radiotherapy to the pelvis. Cochrane Database Syst. Rev. 2002, CD001773. [Google Scholar] [CrossRef]
- Pavlidakey, P.G.; MacLennan, G.T. Radiation cystitis. J. Urol. 2009, 182, 1172–1173. [Google Scholar] [CrossRef]
- Sun, R.; Koubaa, I.; Limkin, E.J.; Dumas, I.; Bentivegna, E.; Castanon, E.; Gouy, S.; Baratiny, C.; Monnot, F.; Maroun, P.; et al. Locally advanced cervical cancer with bladder invasion: Clinical outcomes and predictive factors for vesicovaginal fistulae. Oncotarget 2018, 9, 9299–9310. [Google Scholar] [CrossRef] [PubMed]
- Marks, L.B.; Carroll, P.R.; Dugan, T.C.; Anscher, M.S. The response of the urinary bladder, urethra, and ureter to radiation and chemotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1257–1280. [Google Scholar] [CrossRef]
- Manea, E.; Escande, A.; Bockel, S.; Khettab, M.; Dumas, I.; Lazarescu, I.; Fumagalli, I.; Morice, P.; Deutsch, E.; Haie-Meder, C.; et al. Risk of Late Urinary Complications Following Image Guided Adaptive Brachytherapy for Locally Advanced Cervical Cancer: Refining Bladder Dose-Volume Parameters. Int. J. Radiat. Oncol. 2018, 101, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Mendenhall, W.M.; Henderson, R.H.; Costa, J.A.; Hoppe, B.S.; Dagan, R.; Bryant, C.M.; Nichols, R.C.; Williams, C.R.; Harris, S.E.; Mendenhall, N.P. Hemorrhagic Radiation Cystitis. Am. J. Clin. Oncol. 2015, 38, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Muruve, N.A. Radiation Cystitis. 2017. Available online: https://emedicine.medscape.com/article/2055124-overview (accessed on 23 December 2020).
- D’Ancona, C.; Haylen, B.; Oelke, M.; Abranches-Monteiro, L.; Arnold, E.; Goldman, H.; Hamid, R.; Homma, Y.; Marcelissen, T.; Rademakers, K.; et al. The International Continence Society (ICS) report on the terminology for adult male lower urinary tract and pelvic floor symptoms and dysfunction. Neurourol. Urodynam. 2019, 38, 433–477. [Google Scholar] [CrossRef]
- Pascoe, C.; Duncan, C.; Lamb, B.W.; Davis, N.F.; Lynch, T.H.; Murphy, D.G.; Lawrentschuk, N. Current management of radiation cystitis: A review and practical guide to clinical management. BJU Int. 2019, 123, 585–594. [Google Scholar] [CrossRef]
- Martinez, D.R.; Ercole, C.E.; Lopez, J.G.; Parker, J.; Hall, M.K. A Novel Approach for the Treatment of Radiation-Induced Hemorrhagic Cystitis with the GreenLightTM XPS Laser. Int. Braz. J. Urol. 2015, 41, 584–587. [Google Scholar] [CrossRef]
- Arrizabalaga, M.; Extramina, J.; Parra, J.L.; Ramos, C.; Gonzàlez, R.D.; Leiva, O. Treatment of Massive Haematuria with Aluminous Salts. BJU Int. 1987, 60, 223–226. [Google Scholar] [CrossRef]
- Westerman, M.E.; Boorjian, S.A.; Linder, B.J. Safety and efficacy of intravesical alum for intractable hemorrhagic cystitis: A contemporary evaluation. Int. Braz. J. Urol. 2016, 42, 1144–1149. [Google Scholar] [CrossRef]
- Donahue, L.A.; Frank, I.N. Intravesical formalin for hemorrhagic cystitis: Analysis of therapy. J. Urol. 1989, 141, 809–812. [Google Scholar] [CrossRef]
- Shao, Y.; Lu, G.; Shen, Z. Comparison of intravesical hyaluronic acid instillation and hyperbaric oxygen in the treatment of radiation-induced hemorrhagic cystitis. BJU Int. 2012, 109, 691–694. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.-C.; Kim, D.K.; Chiang, P.-H.; Chancellor, M.B. Bladder botulinum toxin A injection can benefit patients with radiation and chemical cystitis. BJU Int. 2008, 102, 704–706. [Google Scholar] [CrossRef] [PubMed]
- Hazewinkel, M.H.; Stalpers, L.J.A.; Dijkgraaf, M.G.; Roovers, J.-P.W.R. Prophylactic vesical instillations with 0.2% chondroitin sulfate may reduce symptoms of acute radiation cystitis in patients undergoing radiotherapy for gynecological malignancies. Int. Urogynecol. J. 2011, 22, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Bonfili, P.; Franzese, P.; Marampon, F.; La Verghetta, M.E.; Parente, S.; Cerasani, M.; Di Genova, D.; Mancini, M.; Vittorini, F.; Gravina, G.L.; et al. Intravesical instillations with polydeoxyribonucleotides reduce symptoms of radiation-induced cystitis in patients treated with radiotherapy for pelvic cancer: A pilot study. Support. Care Cancer 2013, 22, 1155–1159. [Google Scholar] [CrossRef]
- Mićić, S.; Genbacev, O. Post-irradiation cystitis improved by instillation of early placental extract in saline. Eur. Urol. 1988, 14, 291–293. [Google Scholar]
- Villeirs, L.; Tailly, T.; Ost, P.; Waterloos, M.; Decaestecker, K.; Fonteyne, V.; Van Praet, C.; Lumen, N. Hyperbaric oxygen therapy for radiation cystitis after pelvic radiotherapy: Systematic review of the recent literature. Int. J. Urol. 2019, 27, 98–107. [Google Scholar] [CrossRef]
- Capelli-Schellpfeffer, M.; Gerber, G.S. The use of hyperbaric oxygen in urology. J. Urol. 1999, 162, 647–654. [Google Scholar] [CrossRef]
- Dellis, A.; Deliveliotis, C.; Kalentzos, V.; Vavasis, P.; Skolarikos, A. Is there a role for hyberbaric oxygen as primary treatment for grade IV radiation-induced haemorrhagic cystitis? A prospective pilot-feasibility study and review of literature. Int. Braz J. Urol. 2014, 40, 296–305. [Google Scholar] [CrossRef][Green Version]
- Oscarsson, N.; Müller, B.; Rosén, A.; Lodding, P.; Mölne, J.; Giglio, D.; Hjelle, K.M.; Vaagbø, G.; Hyldegaard, O.; Vangedal, M.; et al. Radiation-induced cystitis treated with hyperbaric oxygen therapy (RICH-ART): A randomised, controlled, phase 2–3 trial. Lancet Oncol. 2019, 20, 1602–1614. [Google Scholar] [CrossRef]
- Pereira, D.; Ferreira, C.; Catarino, R.; Correia, T.; Cardoso, A.; Reis, F.; Cerqueira, M.; Prisco, R.; Camacho, O. Hyperbaric oxygen for radiation-induced cystitis: A long-term follow-up. Actas Urológicas Españolas (English Edition) 2020, 44, 561–567. [Google Scholar] [CrossRef]
- Cardinal, J.; Slade, A.; McFarland, M.; Keihani, S.; Hotaling, J.N.; Myers, J.B. Scoping Review and Meta-analysis of Hyperbaric Oxygen Therapy for Radiation-Induced Hemorrhagic Cystitis. Curr. Urol. Rep. 2018, 19, 38. [Google Scholar] [CrossRef]
- Chong, K.T.; Hampson, N.B.; Corman, J.M. Early hyperbaric oxygen therapy improves outcome for radiation-induced hemorrhagic cystitis. Urology 2005, 65, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Nakada, T.; Nakada, H.; Yoshida, Y.; Nakashima, Y.; Banya, Y.; Fujihira, T.; Karasawa, K. Hyperbaric Oxygen Therapy for Radiation Cystitis in Patients with Prostate Cancer: A Long-Term Follow-Up Study. Urol. Int. 2012, 89, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Choong, S.K.; Walkden, M.; Kirby, R. The management of intractable haematuria. BJU Int. 2000, 86, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Loffroy, R.; Pottecher, P.; Cherblanc, V.; Favelier, S.; Estivalet, L.; Koutlidis, N.; Moulin, M.; Cercueil, J.; Cormier, L.; Krausé, D. Current role of transcatheter arterial embolization for bladder and prostate hemorrhage. Diagn. Interv. Imaging 2014, 95, 1027–1034. [Google Scholar] [CrossRef]
- Gowda, G.G.; Vijayakumar, R.; Tigga, M.P. Endovascular Management of Radiation-Induced Hemorrhagic Cystitis. Indian J. Palliat. Care 2019, 25, 471–473. [Google Scholar]
- Linder, B.J.; Tarrell, R.F.; Boorjian, S.A. Cystectomy for refractory hemorrhagic cystitis: Contemporary etiology, presentation and outcomes. J. Urol. 2014, 192, 1687–1692. [Google Scholar] [CrossRef]
- Marquardt, D.; Williams, J.A.; Kučerka, N.; Atkinson, J.; Wassall, S.R.; Katsaras, J.; Harroun, T.A. Tocopherol Activity Correlates with Its Location in a Membrane: A New Perspective on the Antioxidant Vitamin E. J. Am. Chem. Soc. 2013, 135, 7523–7533. [Google Scholar] [CrossRef]
- Jacobson, G.M.; Bhatia, S.; Smith, B.J.; Button, A.M.; Bodeker, K.; Buatti, J. Randomized Trial of Pentoxifylline and Vitamin E vs Standard Follow-up After Breast Irradiation to Prevent Breast Fibrosis, Evaluated by Tissue Compliance Meter. Int. J. Radiat. Oncol. 2013, 85, 604–608. [Google Scholar] [CrossRef]
- Younus, H. Therapeutic potentials of superoxide dismutase. Int. J. Health Sci. 2018, 12, 88–93. [Google Scholar]
- Campana, F.; Zervoudis, S.; Perdereau, B.; Gez, E.; Fourquet, A.; Badiu, C.; Tsakiris, G.; Koulaloglou, S. Topical superoxide dismutase reduces post-irradiation breast cancer fibrosis. J. Cell. Mol. Med. 2007, 8, 109–116. [Google Scholar] [CrossRef]
- Kharofa, J.; Cohen, E.P.; Tomic, R.; Xiang, Q.; Gore, E. Decreased risk of radiation pneumonitis with incidental concurrent use of angiotensin-converting enzyme inhibitors and thoracic radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Gensel, J.C.; Zhang, B. Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res. 2015, 1619, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bourgier, C.; Auperin, A.; Rivera, S.; Boisselier, P.; Petit, B.; Lang, P.; Lassau, N.; Taourel, P.; Tetreau, R.; Azria, D.; et al. Pravastatin Reverses Established Radiation-Induced Cutaneous and Subcutaneous Fibrosis in Patients With Head and Neck Cancer: Results of the Biology-Driven Phase 2 Clinical Trial Pravacur. Int. J. Radiat. Oncol. 2019, 104, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Chargari, C.; Supiot, S.; Hennequin, C.; Chapel, A.; Simon, J.-M. Treatment of radiation-induced late effects: What’s new? Cancer Radiother. J. Soc. Francaise Radiother. Oncol. 2020, 24, 602–611. [Google Scholar]
- Bentzen, S.M. Preventing or reducing late side effects of radiation therapy: Radiobiology meets molecular pathology. Nat. Rev. Cancer 2006, 6, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Wu, G.R.; Xiong, W.N.; Gu, W.K.; Wang, C.Y. Macrophages: Friend or foe in idiopathic pulmonary fibrosis? Respir. Res. 2018, 19, 170. [Google Scholar] [CrossRef]
- Malyshev, I.; Malyshev, Y. Current Concept and Update of the Macrophage Plasticity Concept: Intracellular Mechanisms of Reprogramming and M3 Macrophage ‘Switch’ Phenotype. BioMed Res. Int. 2015, 2015, 341308. [Google Scholar] [CrossRef]
- Zhou, D.; Huang, C.; Lin, Z.; Zhan, S.; Kong, L.; Fang, C.; Li, J. Macrophage polarization and function with emphasis on the evolving roles of coordinated regulation of cellular signaling pathways. Cell. Signal. 2014, 26, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Lech, M.; Anders, H.-J. Macrophages and fibrosis: How resident and infiltrating mononuclear phagocytes orchestrate all phases of tissue injury and repair. Biochim. Biophys. Acta 2013, 1832, 989–997. [Google Scholar] [CrossRef] [PubMed]
- Braga, T.T.; Agudelo, J.S.H.; Camara, N.O.S. Macrophages During the Fibrotic Process: M2 as Friend and Foe. Front. Immunol. 2015, 6, 602. [Google Scholar] [CrossRef] [PubMed]
- Chou, J.; Chan, M.F.; Werb, Z. Metalloproteinases: A Functional Pathway for Myeloid Cells. Microbiol. Spectr. 2016, 4, 1–15. [Google Scholar]
- Pakshir, P.; Hinz, B. The big five in fibrosis: Macrophages, myofibroblasts, matrix, mechanics, and miscommunication. Matrix Biol. J. Int. Soc. Matrix Biol. 2018, 68–69, 81–93. [Google Scholar] [CrossRef]
- Wynn, T.A.; Barron, L. Macrophages: Master regulators of inflammation and fibrosis. Semin. Liver Dis. 2010, 30, 245–257. [Google Scholar] [CrossRef]
- Nikolic-Paterson, D.J.; Wang, S.; Lan, H.Y. Macrophages promote renal fibrosis through direct and indirect mechanisms. Kidney Int. Suppl. 2014, 4, 34–38. [Google Scholar] [CrossRef]
- Sunderkötter, C.; Steinbrink, K.; Goebeler, M.; Bhardwaj, R.; Sorg, C. Macrophages and angiogenesis. J. Leukoc. Biol. 1994, 55, 410–422. [Google Scholar] [CrossRef]
- Vernon, M.A.; Mylonas, K.J.; Hughes, J. Macrophages and renal fibrosis. Semin. Nephrol. 2010, 30, 302–317. [Google Scholar] [CrossRef]
- Park, H.-R.; Jo, S.-K.; Jung, U. Ionizing Radiation Promotes Epithelial-to-Mesenchymal Transition in Lung Epithelial Cells by TGF-β-producing M2 Macrophages. Vivo Athens Greece 2019, 33, 1773–1784. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, W.; Yu, F.; Gao, F. The Cellular and Molecular Mechanism of Radiation-Induced Lung Injury. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 3446–3450. [Google Scholar] [CrossRef] [PubMed]
- Duru, N.; Wolfson, B.; Zhou, Q. Mechanisms of the alternative activation of macrophages and non-coding RNAs in the development of radiation-induced lung fibrosis. World J. Biol. Chem. 2016, 7, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Noureddine, L. MicroRNAs and fibrosis. Curr. Opin. Nephrol. Hypertens. 2012, 21, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Luo, H.; Li, Y.; Zhou, Y.; Jiang, Y.; Chai, J.; Xiao, X.; You, Y.; Zuo, X. MicroRNA-21 in Scleroderma Fibrosis and its Function in TGF-β- Regulated Fibrosis-Related Genes Expression. J. Clin. Immunol. 2013, 33, 1100–1109. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Coates, P.J.; Lorimore, S.A.; Wright, E.G. Responses to ionizing radiation mediated by inflammatory mechanisms. J. Pathol. 2014, 232, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Meziani, L.; Deutsch, E.; Mondini, M. Macrophages in radiation injury: A new therapeutic target. Oncoimmunology 2018, 7, e1494488. [Google Scholar] [CrossRef]
- Ghita, M.; McMahon, S.J.; Thompson, H.F.; McGarry, C.K.; King, R.B.; Osman, S.O.S.; Kane, J.L.; Tulk, A.; Schettino, G.; Butterworth, K.T.; et al. Small field dosimetry for the small animal radiotherapy research platform (SARRP). Radiat. Oncol. 2017, 12, 204. [Google Scholar] [CrossRef]
- Gieseck, R.L.; Wilson, M.S.; Wynn, T.A. Type 2 immunity in tissue repair and fibrosis. Nat. Rev. Immunol. 2018, 18, 62–76. [Google Scholar] [CrossRef]
- Zwaans, B.M.; Krueger, S.; Bartolone, S.N.; Chancellor, M.B.; Marples, B.; Lamb, L.E. Modeling of chronic radiation-induced cystitis in mice. Adv. Radiat. Oncol. 2016, 1, 333–343. [Google Scholar] [CrossRef][Green Version]
- Oscarsson, N.; Ny, L.; Mölne, J.; Lind, F.; Ricksten, S.-E.; Seeman-Lodding, H.; Giglio, D. Hyperbaric oxygen treatment reverses radiation induced pro-fibrotic and oxidative stress responses in a rat model. Free. Radic. Biol. Med. 2017, 103, 248–255. [Google Scholar] [CrossRef]
- Rajaganapathy, B.R.; Jayabalan, N.; Tyagi, P.; Kaufman, J.; Chancellor, M.B. Advances in Therapeutic Development for Radiation Cystitis. Low. Urin. Tract Symptoms 2014, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Soler, R.; Vianello, A.; Füllhase, C.; Wang, Z.; Atala, A.; Soker, S.; Yoo, J.J.; Williams, J.K. Vascular therapy for radiation cystitis. Neurourol. Urodynam. 2010, 30, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Role of vascular endothelial growth factor in physiologic and pathologic angiogenesis: Therapeutic implications. Semin. Oncol. 2002, 29, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Palchesko, R.N.; Lathrop, K.L.; Funderburgh, J.L.; Feinberg, A.W. In vitro expansion of corneal endothelial cells on biomimetic substrates. Sci. Rep. 2015, 5, 7955. [Google Scholar] [CrossRef]
- Magné, N.; Toillon, R.-A.; Bottero, V.; Didelot, C.; Van Houtte, P.; Gérard, J.-P.; Peyron, J.-F. NF-kappaB modulation and ionizing radiation: Mechanisms and future directions for cancer treatment. Cancer Lett. 2006, 231, 158–168. [Google Scholar] [CrossRef]
- Hellweg, C.E. The Nuclear Factor κB pathway: A link to the immune system in the radiation response. Cancer Lett. 2015, 368, 275–289. [Google Scholar] [CrossRef]
- Kowaliuk, J.; Sarsarshahi, S.; Hlawatsch, J.; Kastsova, A.; Kowaliuk, M.; Krischak, A.; Kuess, P.; Duong, L.; Dörr, W. Translational Aspects of Nuclear Factor-Kappa B and Its Modulation by Thalidomide on Early and Late Radiation Sequelae in Urinary Bladder Dysfunction. Int. J. Radiat. Oncol. 2020, 107, 377–385. [Google Scholar] [CrossRef]
- Bauditz, J. Effective treatment of gastrointestinal bleeding with thalidomide--Chances and limitations. World J. Gastroenterol. 2016, 22, 3158–3164. [Google Scholar] [CrossRef]
- Lee, S.J.; Yi, C.O.; Song, D.H.; Cho, Y.J.; Jeong, Y.Y.; Kang, K.M.; Roh, G.S.; Lee, J.D. Clarithromycin Attenuates Radiation-Induced Lung Injury in Mice. PLoS ONE 2015, 10, e0131671. [Google Scholar] [CrossRef]
- Abernathy, L.M.; Fountain, M.D.; Rothstein, S.E.; David, J.M.; Yunker, C.K.; Rakowski, J.; Lonardo, F.; Joiner, M.C.; Hillman, G.G. Soy Isoflavones Promote Radioprotection of Normal Lung Tissue by Inhibition of Radiation-Induced Activation of Macrophages and Neutrophils. J. Thorac. Oncol. 2015, 10, 1703–1712. [Google Scholar] [CrossRef]
- Aliasgharzadeh, A.; Farhood, B.; Amini, P.; Saffar, H.; Motevaseli, E.; Rezapoor, S.; Nouruzi, F.; Shabeeb, D.H.; Musa, A.E.; Mohseni, M.; et al. Melatonin Attenuates Upregulation of Duox1 and Duox2 and Protects against Lung Injury following Chest Irradiation in Rats. Cell J 2019, 21, 236–242. [Google Scholar] [PubMed]
- Chung, S.I.; Horton, J.A.; Ramalingam, T.R.; White, A.O.; Chung, E.J.; Hudak, K.E.; Scroggins, B.T.; Arron, J.R.; Wynn, T.A.; Citrin, D.E. IL-13 is a therapeutic target in radiation lung injury. Sci. Rep. 2016, 6, 39714. [Google Scholar] [CrossRef] [PubMed]
- Van de Putte, D.; Demarquay, C.; Van Daele, E.; Moussa, L.; Vanhove, C.; Benderitter, M.; Ceelen, W.; Pattyn, P.; Mathieu, N. Adipose-Derived Mesenchymal Stromal Cells Improve the Healing of Colonic Anastomoses Following High Dose of Irradiation Through Anti-Inflammatory and Angiogenic Processes. Cell Transplant. 2017, 26, 1919–1930. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.B.; Moncivais, K.; Caplan, A.I. Mesenchymal stem cells: Environmentally responsive therapeutics for regenerative medicine. Exp. Mol. Med. 2013, 45, e54. [Google Scholar] [CrossRef] [PubMed]
- François, S.; Bensidhoum, M.; Mouiseddine, M.; Mazurier, C.; Allenet, B.; Semont, A.; Frick, J.; Saché, A.; Bouchet, S.; Thierry, D.; et al. Local Irradiation Not Only Induces Homing of Human Mesenchymal Stem Cells at Exposed Sites but Promotes Their Widespread Engraftment to Multiple Organs: A Study of Their Quantitative Distribution After Irradiation Damage. Stem Cells 2006, 24, 1020–1029. [Google Scholar] [CrossRef]
- Sensebé, L.; Bourin, P. Mesenchymal stem cells for therapeutic purposes. Transplantation 2009, 87, S49–S53. [Google Scholar] [CrossRef]
- Charbord, P. Bone marrow mesenchymal stem cells: Historical overview and concepts. Hum. Gene Ther. 2010, 21, 1045–1056. [Google Scholar] [CrossRef]
- Khalifa, J.; François, S.; Rancoule, C.; Riccobono, D.; Magné, N.; Drouet, M.; Chargari, C. Gene therapy and cell therapy for the management of radiation damages to healthy tissues: Rationale and early results. Cancer/Radiothérapie 2019, 23, 449–465. [Google Scholar] [CrossRef]
- Voswinkel, J.; François, S.; Simon, J.-M.; Benderitter, M.; Gorin, N.-C.; Mohty, M.; Fouillard, L.; Chapel, A.; Chapel, A. Use of Mesenchymal Stem Cells (MSC) in Chronic Inflammatory Fistulizing and Fibrotic Diseases: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2013, 45, 180–192. [Google Scholar] [CrossRef]
- Zanoni, M.; Cortesi, M.; Zamagni, A.; Tesei, A. The Role of Mesenchymal Stem Cells in Radiation-Induced Lung Fibrosis. Int. J. Mol. Sci. 2019, 20, 3876. [Google Scholar] [CrossRef]
- François, S.; Usunier, B.; Forgue-Lafitte, M.-E.; L’Homme, B.; Benderitter, M.; Douay, L.; Gorin, N.-C.; Larsen, A.K.; Chapel, A. Mesenchymal Stem Cell Administration Attenuates Colon Cancer Progression by Modulating the Immune Component within the Colorectal Tumor Microenvironment. Stem Cells Transl. Med. 2019, 8, 285–300. [Google Scholar] [CrossRef]
- Linard, C.; Busson, E.; Holler, V.; Strup-Perrot, C.; Lacave-Lapalun, J.-V.; Lhomme, B.; Prat, M.; Devauchelle, P.; Sabourin, J.-C.; Simon, J.-M.; et al. Repeated Autologous Bone Marrow-Derived Mesenchymal Stem Cell Injections Improve Radiation-Induced Proctitis in Pigs. Stem Cells Transl. Med. 2013, 2, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Bessout, R.; Semont, A.; Demarquay, C.; Charcosset, A.; Benderitter, M.; Mathieu, N. Mesenchymal stem cell therapy induces glucocorticoid synthesis in colonic mucosa and suppresses radiation-activated T cells: New insights into MSC immunomodulation. Mucosal Immunol. 2014, 7, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Bessout, R.; Demarquay, C.; Moussa, L.; René, A.; Doix, B.; Benderitter, M.; Sémont, A.; Mathieu, N. TH17 predominant T-cell responses in radiation-induced bowel disease are modulated by treatment with adipose-derived mesenchymal stromal cells. J. Pathol. 2015, 237, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, B.; Tian, J.; Hong, H.; Du, Y.; Li, K.; Li, X.; Wang, N.; Yu, X.; Wei, X. Dental Follicle Stem Cells Ameliorate Lipopolysaccharide-Induced Inflammation by Secreting TGF-β3 and TSP-1 to Elicit Macrophage M2 Polarization. Cell. Physiol. Biochem. 2018, 51, 2290–2308. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.S.; Choi, Y.; Kim, H.O. Adipose-Derived Mesenchymal Stem Cells Promote M2 Macrophage Phenotype through Exosomes. Stem Cells Int. 2019, 2019, 7921760. [Google Scholar] [CrossRef]
- He, X.; Dong, Z.; Cao, Y.; Wang, H.; Liu, S.; Liao, L.; Jin, Y.; Yuan, L.; Li, B. MSC-Derived Exosome Promotes M2 Polarization and Enhances Cutaneous Wound Healing. Stem Cells Int. 2019, 2019, 1–16. [Google Scholar] [CrossRef]
- Ti, D.; Hao, H.; Tong, C.; Liu, J.; Dong, L.; Zheng, J.; Zhao, Y.; Liu, H.; Fu, X.; Han, W. LPS-preconditioned mesenchymal stromal cells modify macrophage polarization for resolution of chronic inflammation via exosome-shuttled let-7b. J. Transl. Med. 2015, 13, 1–14. [Google Scholar] [CrossRef]
- Qi, Y.; Jiang, D.; Sindrilaru, A.; Stegemann, A.; Schatz, S.; Treiber, N.; Rojewski, M.; Schrezenmeier, H.; Beken, S.V.; Wlaschek, M.; et al. TSG-6 Released from Intradermally Injected Mesenchymal Stem Cells Accelerates Wound Healing and Reduces Tissue Fibrosis in Murine Full-Thickness Skin Wounds. J. Investig. Dermatol. 2014, 134, 526–537. [Google Scholar] [CrossRef]
- Wu, Y.; Huang, S.; Enhe, J.; Ma, K.; Yang, S.; Sun, T.; Fu, X. Bone marrow-derived mesenchymal stem cell attenuates skin fibrosis development in mice. Int. Wound J. 2013, 11, 701–710. [Google Scholar] [CrossRef]
- Wang, B.; Yao, K.; Huuskes, B.M.; Shen, H.H.; Zhuang, J.L.; Godson, C.; Brennan, E.P.; Wilkinson-Berka, J.L.; Wise, A.F.; Ricardo, S.D. Mesenchymal Stem Cells Deliver Exogenous MicroRNA-let7c via Exosomes to Attenuate Renal Fibrosis. Mol. Ther. J. Am. Soc. Gene Ther. 2016, 24, 1290–1301. [Google Scholar] [CrossRef] [PubMed]
- Zwaans, B.M.M.; Bartolone, S.N.; Chancellor, M.B.; Nicolai, H.E.; Lamb, L.E. Altered Angiogenic Growth Factors in Urine of Prostate Cancer Survivors With Radiation History and Radiation Cystitis. Urology 2018, 120, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Zwaans, B.M.M.; Nicolai, H.E.; Chancellor, M.B.; Lamb, L.E. Prostate cancer survivors with symptoms of radiation cystitis have elevated fibrotic and vascular proteins in urine. PLoS ONE 2020, 15, e0241388. [Google Scholar] [CrossRef]
- Wojtan, P.; Mierzejewski, M.; Osińska, I.; Domagała-Kulawik, J. Macrophage polarization in interstitial lung diseases. Cent. Eur. J. Immunol. 2016, 41, 159–164. [Google Scholar] [CrossRef]
- Song, W.-J.; Li, Q.; Ryu, M.-O.; Ahn, J.-O.; Bhang, D.H.; Jung, Y.C.; Youn, H.-Y. TSG-6 Secreted by Human Adipose Tissue-derived Mesenchymal Stem Cells Ameliorates DSS-induced colitis by Inducing M2 Macrophage Polarization in Mice. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Hong, I.-S.; Lee, H.-Y.; Kang, K.-S. Mesenchymal stem cells and cancer: Friends or enemies? Mutat. Res. 2014, 768, 98–106. [Google Scholar] [CrossRef]
- Cortes-Dericks, L.; Galetta, D. The therapeutic potential of mesenchymal stem cells in lung cancer: Benefits, risks and challenges. Cell. Oncol. Dordr. 2019, 42, 727–738. [Google Scholar] [CrossRef]
- Galland, S.; Stamenkovic, I. Mesenchymal stromal cells in cancer: A review of their immunomodulatory functions and dual effects on tumor progression. J. Pathol. 2020, 250, 555–572. [Google Scholar] [CrossRef]
- Karlsson, H.; Erkers, T.; Nava, S.; Ruhm, S.; Westgren, M.; Ringdén, O. Stromal cells from term fetal membrane are highly suppressive in allogeneic settings in vitro. Clin. Exp. Immunol. 2012, 167, 543–555. [Google Scholar] [CrossRef]
- Moll, G.; Ignatowicz, L.; Catar, R.; Luecht, C.; Sadeghi, B.; Hamad, O.; Jungebluth, P.; Dragun, D.; Schmidtchen, A.; Ringden, O. Different Procoagulant Activity of Therapeutic Mesenchymal Stromal Cells Derived from Bone Marrow and Placental Decidua. Stem Cells Dev. 2015, 24, 2269–2279. [Google Scholar] [CrossRef]
- Aronsson-Kurttila, W.; Baygan, A.; Moretti, G.; Remberger, M.; Khoein, B.; Moll, G.; Sadeghi, B.; Ringdén, O. Placenta-Derived Decidua Stromal Cells for Hemorrhagic Cystitis after Stem Cell Transplantation. Acta Haematol. 2018, 139, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, B.; Moretti, G.; Arnberg, F.; Samén, E.; Kohein, B.; Catar, R.; Kamhieh-Milz, J.; Geissler, S.; Moll, G.; Holmin, S.; et al. Preclinical Toxicity Evaluation of Clinical Grade Placenta-Derived Decidua Stromal Cells. Front. Immunol. 2019, 10, 2685. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I.; Dennis, J.E. Mesenchymal stem cells as trophic mediators. J. Cell. Biochem. 2006, 98, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Gnecchi, M.; Danieli, P.; Malpasso, G.; Ciuffreda, M.C. Paracrine Mechanisms of Mesenchymal Stem Cells in Tissue Repair. Methods Mol. Biol. 2016, 1416, 123–146. [Google Scholar] [PubMed]
- Xu, S.; Liu, C.; Ji, H.-L. Concise Review: Therapeutic Potential of the Mesenchymal Stem Cell Derived Secretome and Extracellular Vesicles for Radiation-Induced Lung Injury: Progress and Hypotheses. Stem Cells Transl. Med. 2019, 8, 344–354. [Google Scholar] [CrossRef]
- Jafarinia, M.; Alsahebfosoul, F.; Salehi, H.; Eskandari, N.; Ganjalikhani-Hakemi, M. Mesenchymal Stem Cell-Derived Extracellular Vesicles: A Novel Cell-Free Therapy. Immunol. Invest. 2020, 49, 758–780. [Google Scholar] [CrossRef]
- Cavallero, S.; Riccobono, D.; Drouet, M.; François, S. MSC-Derived Extracellular Vesicles: New Emergency Treatment to Limit the Development of Radiation-Induced Hematopoietic Syndrome? Health Phys. 2020, 119, 21–36. [Google Scholar] [CrossRef]
- McGrath, M.S.; Kahn, J.O.; Herndier, B.G. Development of WF10, a novel macrophage-regulating agent. Curr. Opin. Investig. Drugs Lond. Engl. 2002, 3, 365–373. [Google Scholar]
- Giese, T.; McGrath, M.S.; Stumm, S.; Schempp, H.; Elstner, E.; Meuer, S.C. Differential effects on innate versus adaptive immune responses by WF10. Cell. Immunol. 2004, 229, 149–158. [Google Scholar] [CrossRef]
- Srisupundit, S.; Kraiphibul, P.; Sangruchi, S.; Linasmita, V.; Chingskol, K.; Veerasarn, V. The efficacy of chemically-stabilized chlorite-matrix (TCDO) in the management of late postradiation cystitis. J. Med Assoc. Thail. 1999, 82, 798–802. [Google Scholar]
- Veerasarn, V.; Khorprasert, C.; Lorvidhaya, V.; Sangruchi, S.; Tantivatana, T.; Narkwong, L.; Kongthanarat, Y.; Chitapanarux, I.; Tesavibul, C.; Panichevaluk, A. Reduced recurrence of late hemorrhagic radiation cystitis by WF10 therapy in cervical cancer patientsA multicenter, randomized, two-arm, open-label trial. Radiother. Oncol. 2004, 73, 179–185. [Google Scholar] [CrossRef]
- Veerasarn, V.; Boonnuch, W.; Kakanaporn, C. A phase II study to evaluate WF10 in patients with late hemorrhagic radiation cystitis and proctitis. Gynecol. Oncol. 2006, 100, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Fountain, M.D.; Abernathy, L.M.; Lonardo, F.; Rothstein, S.E.; Dominello, M.M.; Yunker, C.K.; Chen, W.; Gadgeel, S.; Joiner, M.C.; Hillman, G.G. Radiation-Induced Esophagitis is Mitigated by Soy Isoflavones. Front. Oncol. 2015, 5, 238. [Google Scholar] [CrossRef] [PubMed]
- Chung, E.J.; Sowers, A.; Thetford, A.; McKay-Corkum, G.; Chung, S.I.; Mitchell, J.B.; Citrin, D.E. Mammalian Target of Rapamycin Inhibition With Rapamycin Mitigates Radiation-Induced Pulmonary Fibrosis in a Murine Model. Int. J. Radiat. Oncol. 2016, 96, 857–866. [Google Scholar] [CrossRef]
- Farhood, B.; Aliasgharzadeh, A.; Amini, P.; Saffar, H.; Motevaseli, E.; Rezapour, S.; Nouruzi, F.; Shabeeb, D.; Musa, A.E.; Ashabi, G.; et al. Radiation-Induced Dual Oxidase Upregulation in Rat Heart Tissues: Protective Effect of Melatonin. Medicina 2019, 55, 317. [Google Scholar] [CrossRef]
- Xu, G.; Wu, H.; Zhang, J.; Li, D.; Wang, Y.; Wang, Y.; Zhang, H.; Lu, L.; Li, C.; Huang, S.; et al. Metformin ameliorates ionizing irradiation-induced long-term hematopoietic stem cell injury in mice. Free. Radic. Biol. Med. 2015, 87, 15–25. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.-K. Brown seaweed fucoidan: Biological activity and apoptosis, growth signaling mechanism in cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef]
- Yu, H.-H.; Ko, E.C.; Chang, C.-L.; Yuan, K.S.-P.; Wu, A.T.; Shan, Y.; Wu, S.-Y. Fucoidan Inhibits Radiation-Induced Pneumonitis and Lung Fibrosis by Reducing Inflammatory Cytokine Expression in Lung Tissues. Mar. Drugs 2018, 16, 392. [Google Scholar] [CrossRef]
- Chargari, C.; Levy, A.; Paoletti, X.; Soria, J.-C.; Massard, C.; Weichselbaum, R.R.; Deutsch, E. Methodological Development of Combination Drug and Radiotherapy in Basic and Clinical Research. Clin. Cancer Res. 2020, 26, 4723–4736. [Google Scholar] [CrossRef]
| Animals | Method of Radiation Cystitis Induction | Treatments | Effect(s) | References | |
|---|---|---|---|---|---|
| Administration Type | Time Post-Radiation Exposure (PE) | ||||
| Adult female Sprague–Dawley rats | Single 20 Gy dose by a linear accelerator (6MV) | 20 sessions of HBOT over a fortnight | 14 days PE | Reduction of oxidative stress and proinflammatory factors | Oscarsson N et al., 2017 |
| Adult female Sprague–Dawley rats | SARRP, singledose 40 Gy 6–8 weeks PE: histological tissue damage to the bladder | Liposomal tacrolimus instillation | 6 weeks PE | Increase in inter-micturition intervals | Rajaganapathy BR et al., 2015 |
| Adult female Lewis rats | A single 20 Gy dose of using a cesium isotope-based irradiator. | Injection into the bladder wall of a solution containing VEGF +/− endothelial cells | 30 days PE | Revascularization of radiation-damaged urinary bladders | Soler R et al., 2011 |
| Female BALB/c mice | Single 10 Gy dose by Siemens Stabilipan X-ray to the whole lung, Thickened alveolar septa, reflective of pneumonitis at 18 weeks PE | Isoflavone mixture gavage | Before and after radiation exposure | M1 subtype switched to an anti-inflammatory M2 subtype with increased levels of Arg-1 and decreased NOS2 | Abernathy LM et al., 2015 |
| Female C57BL/6J mice | Single 18 Gy dose by linear accelerator (21EX 3153 VARIAN) to the whole lung Interstitial edema and fibrosis sections at 16 weeks PE, | Oral clarithromycin | Before and after radiation exposure, and continuing until the day of sacrifice | Inhibition of fibrosis scoring, influx of macrophages and interstitial edema | Lee SJ et al., 2015 |
| C57BL/6 female mice | 5 × 6 Gy thoracic irradiation by X-RAD 320, Macrophage accumulation in the irradiated lung at 10 weeks PE | Purified murine anti-IL-13 IgG antibody by intraperitoneal (ip) injection | Weekly ip injection over 8 weeks, starting 3 weeksPE | Inhibition of recruitment and polarization of alternatively activated YM-1 positive macrophages | Chung SI et al., 2016 |
| Adult male Wistar rats | 60 Co source Single dose Gy 15 to the whole lung
|
| 30 min before irradiation | Increased levels of IL-4, DuoX1, Duox-2 and decreased lymphocyte and macrophage infiltration | Aliasgharzadeh A et al., 2019 |
| Sprague-Dawley rats | Single 27 Gy dose by 60 Co irradiator inthe colorectal region, Anastomosis in the colon at 4 weeks PE | Iterative IV infusion 5 × 10 adipose-MSCs/infusion | 3 weeks PE | The proportion of anti-inflammatory M2 macrophages grew, favoring the M2 phenotype and promoting wound healing | Van de Putte D et al., 2017 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helissey, C.; Cavallero, S.; Brossard, C.; Dusaud, M.; Chargari, C.; François, S. Chronic Inflammation and Radiation-Induced Cystitis: Molecular Background and Therapeutic Perspectives. Cells 2021, 10, 21. https://doi.org/10.3390/cells10010021
Helissey C, Cavallero S, Brossard C, Dusaud M, Chargari C, François S. Chronic Inflammation and Radiation-Induced Cystitis: Molecular Background and Therapeutic Perspectives. Cells. 2021; 10(1):21. https://doi.org/10.3390/cells10010021
Chicago/Turabian StyleHelissey, Carole, Sophie Cavallero, Clément Brossard, Marie Dusaud, Cyrus Chargari, and Sabine François. 2021. "Chronic Inflammation and Radiation-Induced Cystitis: Molecular Background and Therapeutic Perspectives" Cells 10, no. 1: 21. https://doi.org/10.3390/cells10010021
APA StyleHelissey, C., Cavallero, S., Brossard, C., Dusaud, M., Chargari, C., & François, S. (2021). Chronic Inflammation and Radiation-Induced Cystitis: Molecular Background and Therapeutic Perspectives. Cells, 10(1), 21. https://doi.org/10.3390/cells10010021





