Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil
Abstract
1. Introduction
2. Materials and Methods
2.1. Greenhouse Conditions, Plant Material, and Experimental Design
2.2. Nutrient Solution Management, NO3−: Cl− Treatments, and Omperazole Application
2.3. Biometric and Leaf Color Measurements
2.4. Soil Plant Analysis Development Index and Leaf Gas Exchange
2.5. Ion Analysis of Leaf Tissue
2.6. Statistics
3. Results
3.1. Morphological Responses Induced by OMP under Different NO3−:Cl− Ratios
3.2. Physiological Responses Induced by OMP under Different NO3−:Cl− Ratios
3.3. Ion Content Induced by OMP under Different NO3−:Cl− Ratios
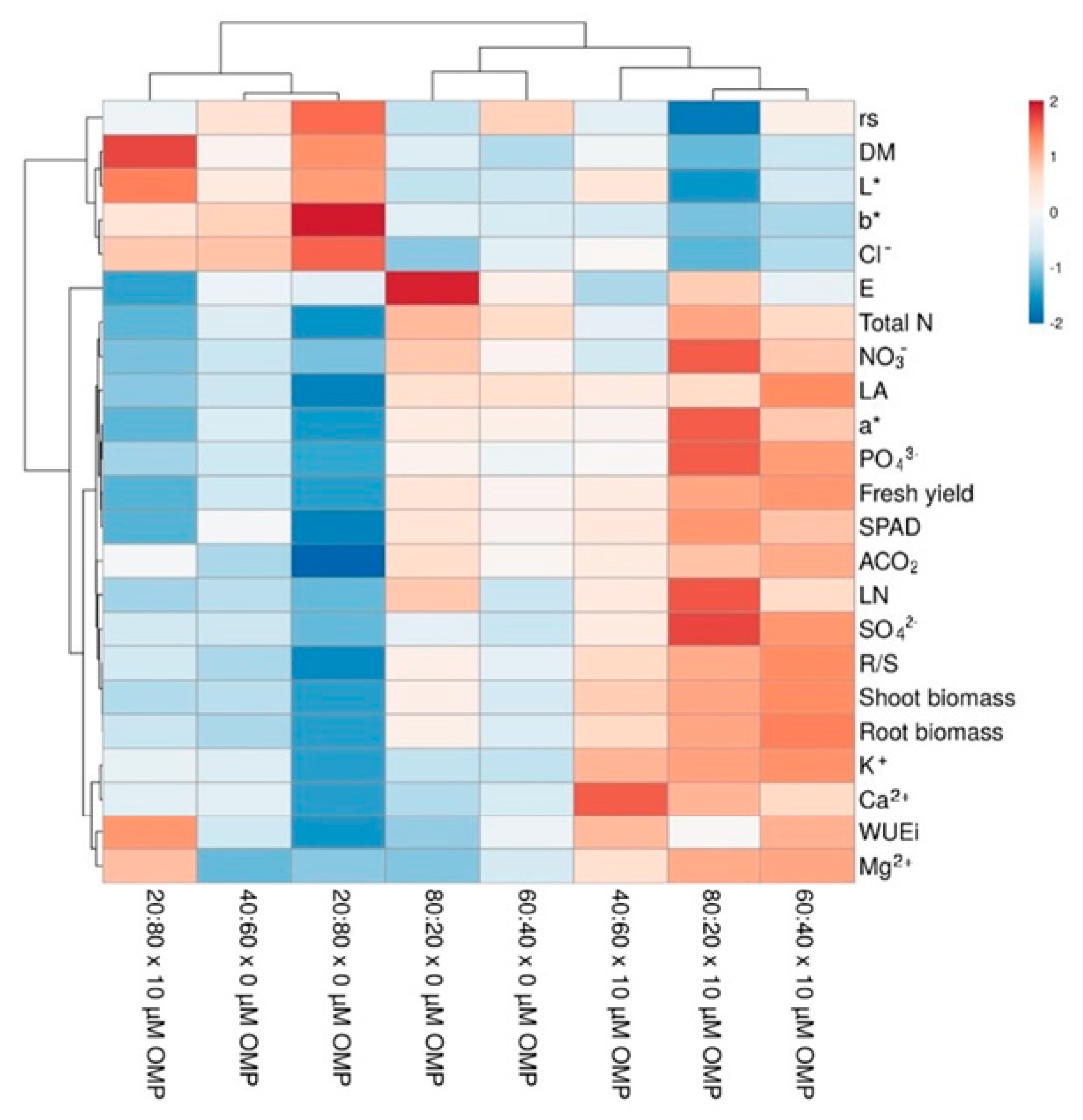
3.4. Heat Map Analysis
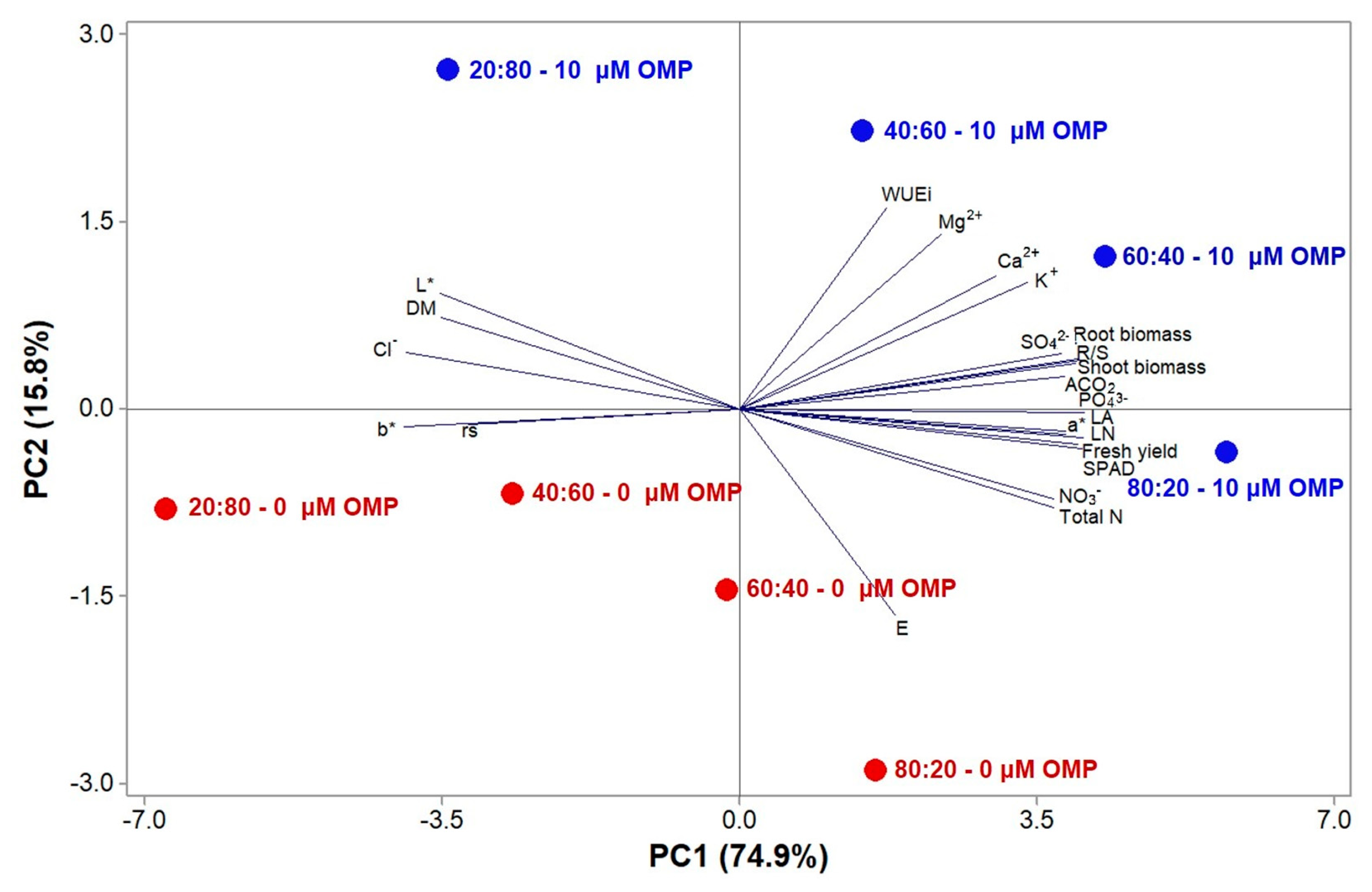
3.5. Principal Component Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Ann. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S.; Munns, R. Salinity stress: Physiological constraints and adaptive mechanisms. Plant Stress Physiol. 2012, 1, 59–63. [Google Scholar] [CrossRef]
- Carillo, P.; Cirillo, C.; De Micco, V.; Arena, C.; De Pascale, S.; Rouphael, Y. Morpho-anatomical; physiological and biochemical adaptive responses to saline water of Bougainvillea spectabilis Willd. trained to different canopy shapes. Agric. Water Manag. 2019, 212, 12–22. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; d Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol Plant. Mol. Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [PubMed]
- Flowers, T.J.; Munns, R.; Colmer, T.D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann. Bot. 2015, 115, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Scagel, C.F.; Bryla, D.R.; Lee, J. Salt Exclusion and mycorrhizal symbiosis increase tolerance to NaCl and CaCl2 salinity in ‘Siam Queen’ basil. HortScience 2017, 52, 278–287. [Google Scholar] [CrossRef]
- Ferchichi, S.; Hessini, K.; Dell’Aversana, E.; D’Amelia, L.; Woodrow, P.; Ciarmiello, L.F.; Fuggi, A.; Carillo, P. Hordeum vulgare and Hordeum maritimum respond to extended salinity stress displaying different temporal accumulation pattern of metabolites. Funct. Plant Biol. 2018, 45, 1096–1109. [Google Scholar] [CrossRef]
- Gorham, J.; Läuchli, A.; Leidi, E.O. Plant Responses to Salinity. In Physiology of Cotton; Stewart, J.M., Oosterhuis, D.M., Heitholt, J.J., Mauney, J.R., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 129–141. [Google Scholar]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Dell’Aversana, E.; Carillo, P. Spatial and temporal profile of glycine betaine accumulation in plants under abiotic stresses. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- FAO. Small-Scale Irrigation for Arid Zones. Principles and Options; FAO, Development Series 2; Food and Agriculture Organisation of the United Nations, FAO: Rome, Italy, 1997; p. 51. ISBN 97892 51038 963.
- Chinnusamy, V.; Jagendorf, A.; Zhu, J.K. Understanding and improving salt tolerance in plants. Crop Sci. 2005, 45, 437–448. [Google Scholar] [CrossRef]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef]
- Puniran-Hartley, N.; Hartley, J.; Shabala, L.; Shabala, S. Salinity-induced accumulation of organic osmolytes in barley and wheat leaves correlates with increased oxidative stress tolerance: In planta evidence for cross-tolerance. Plant Physiol. Biochem. 2014, 83, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Al Hassan, M.; Naranjo, M.A.; Agrawal, V.; Boscaiu, M.; Vicente, O. Effects of salinity and drought on growth; ionic relations; compatible solutes and activation of antioxidant systems in oleander (Nerium oleander L.). PLoS ONE 2017, 12, e0185017. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.M.F. Nitrogen containing compounds and adaptation of plants to salinity stress. Biol. Plant. 2000, 43, 491–500. [Google Scholar] [CrossRef]
- Yancey, P.H. Organic osmolytes as compatible; metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J. Exp. Biol. 2005, 208, 2819–2830. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P. GABA shunt in durum wheat. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Kaschani, F.; van der Hoorn, R. Small molecule approaches in plants. Curr. Opin. Chem. Biol. 2007, 11, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Hicks, G.R.; Raikhel, N.V. Small molecules present large opportunities in plant biology. Ann. Rev. Plant Biol. 2012, 63, 261–282. [Google Scholar] [CrossRef]
- Chan, Z.; Shi, H. Improved abiotic stress tolerance of bermudagrass by exogenous small molecules. Plant Signal. Behav. 2015, 10, e991577. [Google Scholar] [CrossRef]
- Lace, B.; Prandi, C. Shaping small bioactive molecules to untangle their biological function: A focus on fluorescent plant hormones. Mol. Plant 2016, 9, 1099–1118. [Google Scholar] [CrossRef]
- Van Oosten, M.J.; Silletti, S.; Guida, G.; Cirillo, V.; Di Stasio, E.; Carillo, P.; Woodrow, P.; Maggio, A.; Raimondi, G. A benzimidazole proton pump inhibitor increases growth and tolerance to salt stress in tomato. Front. Plant Sci. 2017, 8, 1220. [Google Scholar] [CrossRef]
- Rouphael, Y.; Raimondi, G.; Lucini, L.; Carillo, P.; Kyriacou, M.C.; Colla, G.; Cirillo, V.; Pannico, A.; El-Nakhel, C.; De Pascale, S. Physiological and metabolic responses triggered by omeprazole improve tomato plant tolerance to NaCl stress. Front. Plant Sci. 2018, 9, 249. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P.; Raimondi, G.; Kyriacou, M.C.; Pannico, A.; El-Nakhel, C.; Cirillo, V.; Colla, G.; De Pascale, S.; Rouphael, Y. Morpho-physiological and homeostatic adaptive responses triggered by omeprazole enhance lettuce tolerance to salt stress. Sci. Hortic. 2019, 249, 22–30. [Google Scholar] [CrossRef]
- Cirillo, V.; Van Oosten, M.J.; Izzo, M.; Maggio, A. Omeprazole treatment elicits contrasting responses to salt stress in two basil genotypes. Ann. Appl. Biol. 2019, 1–10. [Google Scholar] [CrossRef]
- Okamoto, T.; Takatani, S.; Noutoshi, Y.; Motose, H.; Takahashi, T. Omeprazole enhances mechanical stress-induced root growth reduction in Arabidopsis thaliana. Plant Cell Physiol. 2018, 59, 1581–1591. [Google Scholar] [CrossRef] [PubMed]
- Fellenius, E.; Berglindh, T.; Sachs, G.; Olbe, L.; Elander, B.; Sjöstrand, S.E.; Wallmark, B. Substituted benzimidazoles inhibit gastric acid secretion by blocking (H+ + K+) ATPase. Nature 1981, 290, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Clissold, S.; Campoli-Richards, D. Omeprazole. A preliminary review of its pharmacodynamic and pharmacokinetic properties; and therapeutic potential in peptic ulcer disease and Zollinger-Ellison syndrome. Drugs 1986, 32, 15–47. [Google Scholar] [CrossRef] [PubMed]
- Massoomi, F.; Savage, J.; Destache, C.J. Omeprazole: A comprehensive review. Pharmacother. J. Hum. Pharmacol. Drug Ther. 1993, 13, 46–59. [Google Scholar] [CrossRef]
- Blumwald, E. Sodium transport and salt tolerance in plants. Curr. Opin. Cell Biol. 2000, 12. [Google Scholar] [CrossRef]
- Pardo, J.M.; Quintero, F.J. Plants and sodium ions: Keeping company with the enemy. Genome Biol. 2002, 3, REVIEWS1017. [Google Scholar] [CrossRef]
- Munns, R.; Rebetzke, G.J.; Husain, S.; James, R.A.; Hare, R.A. Genetic control of sodium exclusion in durum wheat. Aust. J. Agric. Res. 2003, 54, 627–635. [Google Scholar] [CrossRef]
- Davenport, R.; James, R.; Zakrisson-Plogander, A.; Tester, M.; Munns, R. Control of Sodium Transport in Durum Wheat. Plant Physiol. 2005, 137, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Cassaniti, C.; Leonardi, C.; Flowers, T.J. The effects of sodium chloride on ornamental shrubs. Sci. Hortic. 2009, 122, 586–593. [Google Scholar] [CrossRef]
- Cuin, T.A.; Zhou, M.; Parsons, D.; Shabala, S. Genetic behaviour of physiological traits conferring cytosolic K+/Na+ homeostasis in wheat. Plant Biol. 2012, 14, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P.; Mastrolonardo, G.; Nacca, F.; Parisi, D.; Verlotta, A.; Fuggi, A. Nitrogen metabolism in durum wheat under salinity: Accumulation of proline and glycine betaine. Funct. Plant Biol. 2008, 35, 412–426. [Google Scholar] [CrossRef]
- Fraire-Velázquez, S.L.; Balderas-Hernández, V.E. Abiotic Stress in Plants and Metabolic Responses. In Abiotic Stress—Plant Responses and Applications in Agriculture; Vahdati, K., Leslie, C., Eds.; InTech: Rijeka, Croatia, 2013. [Google Scholar] [CrossRef]
- Goodrich, B.; Koski, R.R.; Jacobi, W. Roadside vegetation health condition and magnesium chloride (MgCl2) dust suppressant use in two Colorado, U.S. Counties. Arboric. Urban. For. 2008, 34, 252. [Google Scholar]
- Geilfus, C.M. Chloride: From nutrient to toxicant. Plant Cell Physiol. 2018, 59, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Britto, D.T.; Kronzucker, H.J. Futile cycling at the plasma membrane: A hallmark of low-affinity nutrient transport. Trends Plant Sci. 2006, 11, 529–534. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition in Higher Plants, 2nd ed.; Academic Press: London, UK, 1995. [Google Scholar] [CrossRef]
- Xu, G.; Magen, H.; Tarchitzky, J.; Kafkafi, U.; Donald, L.S. Advances in chloride nutrition of plants. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: San Diego, CA, USA, 1999; pp. 96–150. [Google Scholar]
- Broadley, M.; Brown, P.; Cakmak, I.; Rengel, Z.; Zhao, F. Chapter 7—Function of Nutrients: Micronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Academic Press: San Diego, CA, USA, 2012; pp. 191–248. [Google Scholar]
- Franco-Navarro, J.D.; Brumós, J.; Rosales, M.A.; Cubero-Font, P.; Talón, M.; Colmenero-Flores, J.M. Chloride regulates leaf cell size and water relations in tobacco plants. J. Exp. Bot. 2016, 67, 873–891. [Google Scholar] [CrossRef]
- Tavakkoli, E.; Rengasamy, P.; McDonald, G.K. High concentrations of Na+ and Cl- ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J. Exp. Bot. 2010, 61, 4449–4459. [Google Scholar] [CrossRef]
- Tavakkoli, E.; Fatehi, F.; Coventry, S.; Rengasamy, P.; McDonald, G.K. Additive effects of Na+ and Cl- ions on barley growth under salinity stress. J. Exp. Bot. 2011, 62, 2189–2203. [Google Scholar] [CrossRef]
- Borgognone, D.; Rouphael, Y.; Cardarelli, M.; Lucini, L.; Colla, G. Changes in biomass, mineral composition, and quality of cardoon in response to NO3-:Cl- ratio and nitrate deprivation from the nutrient solution. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Lucini, L.; Rouphael, Y.; Cardarelli, M.; Kalunke, R.M.; Colla, G. Insight into the role of grafting and arbuscular mycorrhiza on cadmium stress tolerance in tomato. Front. Plant Sci. 2015, 6, 477. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Colla, G.; Giordano, M.; El-Nakhel, C.; Kyriacou, M.C.; De Pascale, S. Foliar applications of a legume-derived protein hydrolysate elicit dose-dependent increases of growth, leaf mineral composition, yield and fruit quality in two greenhouse tomato cultivars. Sci. Hortic. 2017, 226, 353–360. [Google Scholar] [CrossRef]
- Bremner, J.M. Total nitrogen. In Methods of Soil Analysis-Part 2; Black, C.A., Ed.; American Society of Agronomy, Soil Science Society of America: Madison, WI, USA, 1965. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Graziani, G.; Pannico, A.; Soteriou, G.; Giordano, M.; Giordano, M.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Functional qualityin novel food sources: Genotypic variation in the nutritive and phytochemical composition of thirteen microgreens species. Food Chem. 2019, 277, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Ciarmiello, L.F.; Piccirillo, P.; Carillo, P.; De Luca, A.; Woodrow, P. Determination of the genetic relatedness of fig (Ficus carica L.) accessions using RAPD fingerprint and their agro-morphological characterization. South Afr. J. Bot. 2015, 97, 40–47. [Google Scholar] [CrossRef]
- Carillo, P.; Cacace, D.; De Pascale, S.; Rapacciuolo, M.; Fuggi, A. Organic vs. traditional potato powder. Food Chem. 2012, 133, 1264–1273. [Google Scholar] [CrossRef]
- Ju, J.H.; Yeum, K.J.; Son, H.M.; and Yoon, Y.H. Morphological and physiological responses of purple chrysanthemum (Aster sphathulifolius) under long-term stress of calcium chloride as deicing salt. Appl. Ecol. Environ. Res. 2018, 16, 605–616. [Google Scholar] [CrossRef]
- Borghesi, E.; Carmassi, G.; Uguccioni, M.C.; Vernieri, P.; Malorgio, F. Effects of calcium and salinity stress on quality of lettuce in soilless culture. J. Plant Nutr. 2013, 36, 677–690. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Jawad, R.; Kumar, P.; Rea, E.; Cardarelli, M. The effectiveness of grafting to improve NaCl and CaCl2 tolerance in cucumber. Sci. Hortic. 2013, 164, 380–391. [Google Scholar] [CrossRef]
- Borgognone, D.; Cardarelli, M.; Lucini, L.; Colla, G. Does CaCl2 play a role in improving biomass yield and quality of cardoon grown in a floating system under saline conditions? HortScience 2014, 49, 1523–1528. [Google Scholar] [CrossRef]
- Diatloff, E.; Roberts, M.; Sanders, D.; Roberts, S.K. Characterization of anion channels in the plasma membrane of Arabidopsis epidermal root cells and the identification of a citrate-permeable channel induced by phosphate starvation. Plant Physiol. 2004, 136, 4136–4149. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P.; Mastrolonardo, G.; Nacca, F.; Fuggi, A. Nitrate reductase in durum wheat seedlings as affected by nitrate nutrition and salinity. Funct. Plant Biol. 2005, 32, 209–219. [Google Scholar] [CrossRef]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Touraine, B.; Clarkson, D.T.; Muller, B. Regulation of nitrate uptake at the whole plant level. In A Whole Plant Perspective on Carbon-Nitrogen Interactions; Roy, J., Garnier, E., Eds.; SPB Academic Publishing: Hague, The Netherlands, 1994; pp. 11–30. [Google Scholar]
- Galangau, F.; Daniel-Vedele, F.; Moureaux, T.; Dorbe, M.F.; Leydecker, M.T.; Caboche, M. Expression of Leaf Nitrate Reductase Genes from Tomato and Tobacco in Relation to Light-Dark Regimes and Nitrate Supply. Plant Physiol. 1998, 88, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Valadier, M.H.; Migge, A.; Becker, T.W. Drought-induced effects on nitrate reductase activity and mRNA and on the coordination of nitrogen and carbon metabolism in maize leaves. Plant Physiol. 1998, 117, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, F.; Crawford, N.M.; Wang, Y. Molecular regulation of nitrate responses in plants. Intern. J. Mol. Sci. 2018, 19, 2039. [Google Scholar] [CrossRef] [PubMed]
- Soltabayeva, A.; Srivastava, S.; Kurmanbayeva, A.; Bekturova, A.; Fluhr, R.; Sagi, M. Early senescence in older leaves of low nitrate-grown atxdh1 uncovers a role for purine catabolism in N supply. Plant Physiol. 2018, 178, 1027–1044. [Google Scholar] [CrossRef]
- Suhayda, C.G.; Redmann, R.E.; Harvey, B.L.; Cipywnyk, A.L. Comparative response of cultivated and wild barley species to salinity stress and calcium supply. Crop. Sci. 1992, 32, 154–163. [Google Scholar] [CrossRef]
- Abel, G.H. Inheritance of the capacity for chloride inclusion and chloride exclusion by soybeans. Crop. Sci. 1969, 9, 697–698. [Google Scholar] [CrossRef]
- Li, B.; Tester, M.; Gilliham, M. Chloride on the Move. Trends Plant Sci. 2017, 22, 236–248. [Google Scholar] [CrossRef]
- Henderson, S.W.; Gilliham, M. The Gatekeeper Concept: Cell-Type Specific Molecular Mechanisms of Plant Adaptation to Abiotic Stress. In Molecular Mechanisms in Plant Adaptation; Laitinen, R.A., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2015; pp. 83–115. [Google Scholar] [CrossRef]
- Li, B.; Qiu, J.; Jayakannan, M.; Xu, B.; Li, Y.; Mayo, G.M.; Tester, M.; Gilliham, M.; Roy, S.J. AtNPF2.5 modulates chloride (Cl−) efflux from roots of Arabidopsis thaliana. Front. Plant Sci. 2017, 7, 2013. [Google Scholar] [CrossRef] [PubMed]
- Wege, S.; Jossier, M.; Filleur, S.; Thomine, S.; Barbier-Brygoo, H.; Gambale, F.; De Angeli, A. The proline 160 in the selectivity filter of the Arabidopsis NO3−/H+ exchanger AtCLCa is essential for nitrate accumulation in planta. Plant J. 2010, 63, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Wegner, L.H.; Raschke, K. Ion channels in the xylem parenchyma of barley roots—A procedure to isolate protoplasts from this tissue and a patch-clamp exploration of salt passageways into the xylem vessels. Plant Physiol. 1994, 105, 799–813. [Google Scholar] [CrossRef] [PubMed]
- Teakle, N.L.; Tyerman, S.D. Mechanisms of Cl- transport contributing to salt tolerance. Plant Cell Environ. 2010, 33, 566–589. [Google Scholar] [CrossRef] [PubMed]


| Source of variance. | Leaf Number | Leaf Area | Root-to-Shoot Ratio | Leaf Dry Matter | L* | a* | b* |
|---|---|---|---|---|---|---|---|
| (no. plant−1) | (cm2 plant−1) | (%) | |||||
| NO3−:Cl− (N) | |||||||
| 80:20 | 103 a | 1130 a | 0.11 a | 8.73 c | 41.08 c | −15.73 a | 21.11 c |
| 60:40 | 87 b | 1196 a | 0.11 a | 8.78 c | 42.34 c | −16.20 ab | 21.20 c |
| 40:60 | 84 b | 983 b | 0.09 b | 9.56 b | 44.40 b | −16.92 b | 23.14 b |
| 20:80 | 73 c | 751 c | 0.07 c | 11.18 a | 46.49 a | −18.19 c | 25.87 a |
| *** | *** | *** | *** | *** | *** | *** | |
| Omeprazole (μM) (OMP) | |||||||
| 0 | 81 | 955 | 0.08 | 9.62 | 43.66 | −17.13 | 24.03 |
| 10 | 93 | 1076 | 0.11 | 9.51 | 43.49 | −16.39 | 21.62 |
| t-test | 0.030 | 0.150 | 0.001 | 0.832 | 0.876 | 0.125 | 0.021 |
| N × OMP | |||||||
| 80:20 × 0 μM OMP | 98 | 1123 | 0.10 | 9.16 | 41.98 | −16.42 | 22.04 |
| 60:40 × 0 μM OMP | 78 | 1124 | 0.09 | 8.71 | 42.19 | −16.55 | 21.64 |
| 40:60 × 0 μM OMP | 77 | 895 | 0.08 | 9.66 | 44.22 | −17.20 | 24.72 |
| 20:80 × 0 μM OMP | 71 | 678 | 0.06 | 10.93 | 46.25 | −18.35 | 27.74 |
| 80:20 × 10 μM OMP | 109 | 1138 | 0.12 | 8.31 | 40.19 | −15.03 | 20.18 |
| 60:40 × 10 μM OMP | 95 | 1268 | 0.13 | 8.86 | 42.48 | −15.85 | 20.76 |
| 40:60 × 10 μM OMP | 91 | 1072 | 0.11 | 9.46 | 44.57 | −16.63 | 21.55 |
| 20:80 × 10 μM OMP | 75 | 825 | 0.08 | 11.42 | 46.73 | −18.03 | 24.01 |
| ns | ns | ns | ns | ns | ns | ns |
| Source of Variance | SPAD Index | ACO2 | rs | E | WUEi |
|---|---|---|---|---|---|
| (μmol CO2 m−2 s−1) | (m−2 s−1 mol−1) | (mol H2O m−2 s−1) | (μmol CO2 mol−1 H2O) | ||
| NO3−:Cl− (N) | |||||
| 80:20 | 39.59 a | 11.89 a | 5.56 b | 3.81 a | 3.16 |
| 60:40 | 38.35 ab | 11.67 ab | 8.01 a | 3.29 ab | 3.60 |
| 40:60 | 37.25 b | 10.60 bc | 7.52 ab | 3.09 b | 3.48 |
| 20:80 | 31.59 c | 9.57 c | 8.35 a | 2.96 b | 3.32 |
| *** | ** | * | * | ns | |
| Omeprazole (μM) (OMP) | |||||
| 0 | 35.64 | 10.22 | 8.10 | 3.45 | 2.98 |
| 10 | 37.74 | 11.64 | 6.61 | 3.13 | 3.79 |
| t-test | 0.145 | 0.014 | 0.050 | 0.126 | 0.001 |
| N × OMP | |||||
| 80:20 × 0 μM OMP | 38.27 | 11.71 | 6.34 | 4.03 | 2.92 |
| 60:40 × 0 μM OMP | 37.04 | 11.02 | 8.41 | 3.38 | 3.29 |
| 40:60 × 0 μM OMP | 36.47 | 9.87 | 8.10 | 3.22 | 3.09 |
| 20:80 × 0 μM OMP | 30.79 | 8.29 | 9.56 | 3.16 | 2.63 |
| 80:20 × 10 μM OMP | 40.90 | 12.06 | 4.78 | 3.59 | 3.41 |
| 60:40 × 10 μM OMP | 39.65 | 12.32 | 7.60 | 3.20 | 3.90 |
| 40:60 × 10 μM OMP | 38.04 | 11.33 | 6.93 | 2.97 | 3.87 |
| 20:80 × 10 μM OMP | 32.38 | 10.86 | 7.14 | 2.75 | 4.00 |
| ns | ns | ns | ns | ns |
| Source of Variance | Total N | NO3− | PO43− | SO42− | K+ | Ca2+ | Mg2+ | Cl− |
|---|---|---|---|---|---|---|---|---|
| (g 100 g−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | |
| NO3−:Cl− (N) | ||||||||
| 80:20 | 4.40 a | 29.09 a | 33.13 a | 3.68 a | 54.16 a | 11.55 a | 2.41 ab | 5.31 d |
| 60:40 | 4.05 b | 19.82 b | 29.78 b | 3.30 ab | 54.53 a | 11.57 a | 2.52 a | 13.21 c |
| 40:60 | 3.16 c | 6.69 c | 21.77 c | 2.93 b | 54.92 a | 12.37 a | 2.29 b | 26.58 b |
| 20:80 | 2.19 d | 0.92 d | 13.66 d | 2.35 c | 43.26 b | 10.15 b | 2.39 ab | 36.80 a |
| *** | *** | *** | *** | *** | ** | ns | *** | |
| Omeprazole (μM) (OMP) | ||||||||
| 0 | 3.37 | 11.76 | 19.86 | 2.50 | 43.50 | 10.34 | 2.07 | 24.44 |
| 10 | 3.53 | 16.51 | 29.31 | 3.63 | 59.93 | 12.48 | 2.74 | 16.51 |
| t-test | 0.674 | 0.339 | 0.012 | 0.001 | 0.001 | 0.001 | 0.001 | 0.048 |
| N × O | ||||||||
| 80:20 × 0 μM OMP | 4.34 | 24.34 b | 26.03 b | 2.86 bc | 44.59 | 10.28 | 2.02 | 6.73 de |
| 60:40 × 0 μM OMP | 4.03 | 15.56 c | 23.25 bc | 2.51 cd | 44.37 | 10.76 | 2.22 | 16.57 c |
| 40:60 × 0 μM OMP | 3.12 | 6.13 d | 18.85 cd | 2.53 cd | 47.95 | 11.00 | 1.98 | 32.61 b |
| 20:80 × 0 μM OMP | 2.02 | 0.99 e | 11.31 e | 2.10 d | 37.10 | 9.32 | 2.04 | 41.85 a |
| 80:20 × 10 μM OMP | 4.47 | 33.84 a | 40.23 a | 4.50 a | 63.74 | 12.82 | 2.80 | 3.90 e |
| 60:40 × 10 μM OMP | 4.08 | 24.08 b | 36.31 a | 4.08 a | 64.68 | 12.38 | 2.81 | 9.85 d |
| 40:60 × 10 μM OMP | 3.21 | 7.26 d | 24.70 b | 3.33 b | 61.88 | 13.75 | 2.60 | 20.55 c |
| 20:80 × 10 μM OMP | 2.37 | 0.85 e | 16.00 d | 2.60 cd | 49.42 | 10.97 | 2.74 | 31.75 b |
| ns | ** | * | * | ns | ns | ns | * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carillo, P.; Woodrow, P.; Raimondi, G.; El-Nakhel, C.; Pannico, A.; Kyriacou, M.C.; Colla, G.; Mori, M.; Giordano, M.; De Pascale, S.; et al. Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil. Agronomy 2019, 9, 355. https://doi.org/10.3390/agronomy9070355
Carillo P, Woodrow P, Raimondi G, El-Nakhel C, Pannico A, Kyriacou MC, Colla G, Mori M, Giordano M, De Pascale S, et al. Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil. Agronomy. 2019; 9(7):355. https://doi.org/10.3390/agronomy9070355
Chicago/Turabian StyleCarillo, Petronia, Pasqualina Woodrow, Giampaolo Raimondi, Christophe El-Nakhel, Antonio Pannico, Marios C. Kyriacou, Giuseppe Colla, Mauro Mori, Maria Giordano, Stefania De Pascale, and et al. 2019. "Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil" Agronomy 9, no. 7: 355. https://doi.org/10.3390/agronomy9070355
APA StyleCarillo, P., Woodrow, P., Raimondi, G., El-Nakhel, C., Pannico, A., Kyriacou, M. C., Colla, G., Mori, M., Giordano, M., De Pascale, S., & Rouphael, Y. (2019). Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil. Agronomy, 9(7), 355. https://doi.org/10.3390/agronomy9070355















