Effects of Natural Hail on the Growth, Physiological Characteristics, Yield, and Quality of Vitis vinifera L. cv. Thompson Seedless under Mediterranean Growing Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Layout
- (a)
- leaves still attached to grapevines with holes punched through them, creating hail injury;
- (b)
- partial defoliation to primary and lateral shoots;
- (c)
- partial injury or total cracking of berries;
- (d)
- partial stem and bark bruising;
- (e)
- primary and lateral shoots with partial hail injury on the epidermis.
2.2. Leaf Gas-Exchange Measurements
2.3. Vegetative Data, Productive Traits, and Fruit Composition
2.4. Carbohydrate and Nitrogen Storage in Above-Ground Permanent Vine Organs
2.5. Shoot Fertility and Blind Buds
2.6. Statistical Analysis
3. Results and Discussion
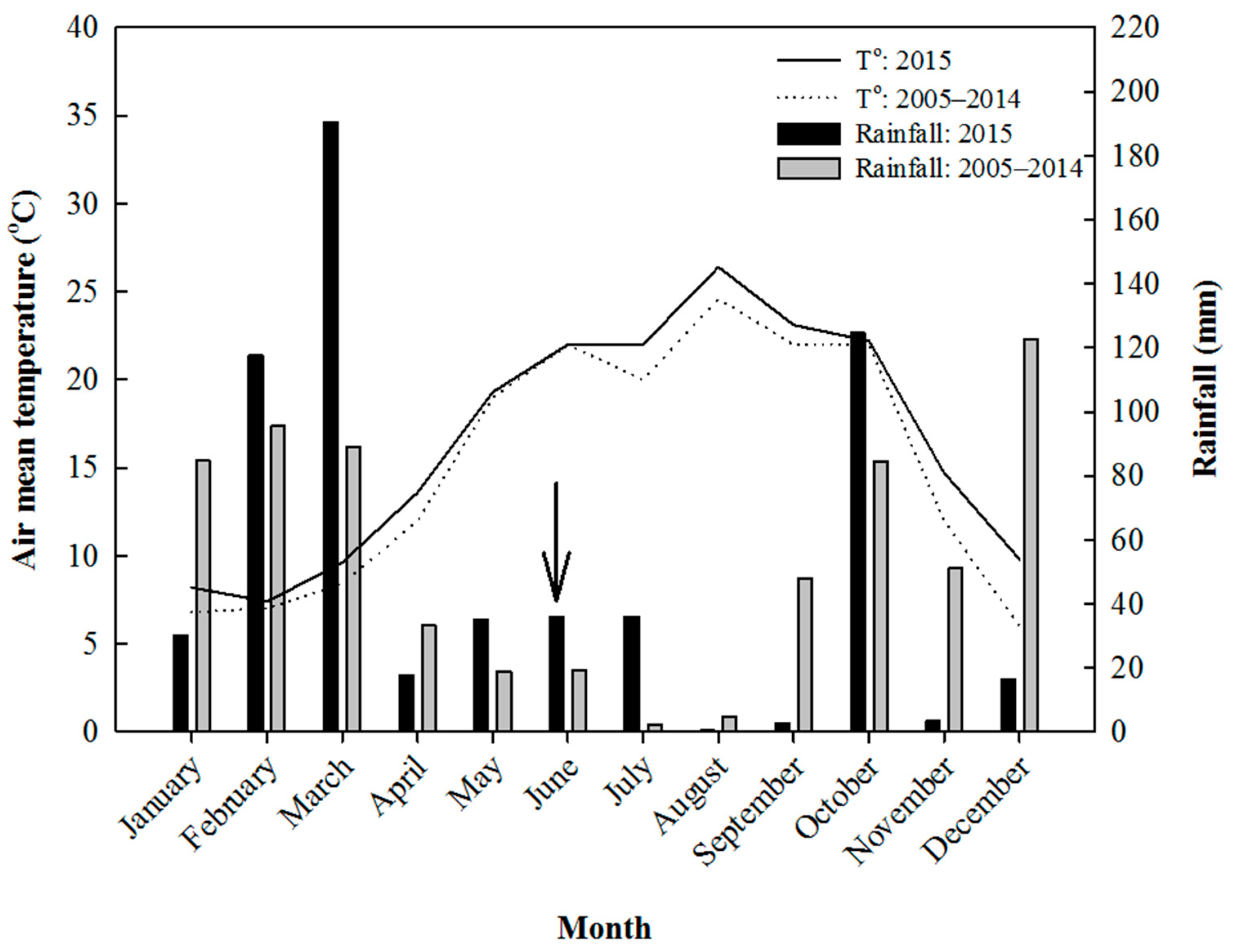
3.1. Meteorological Data
3.2. Effect of Natural Hail on Vegetative Growth
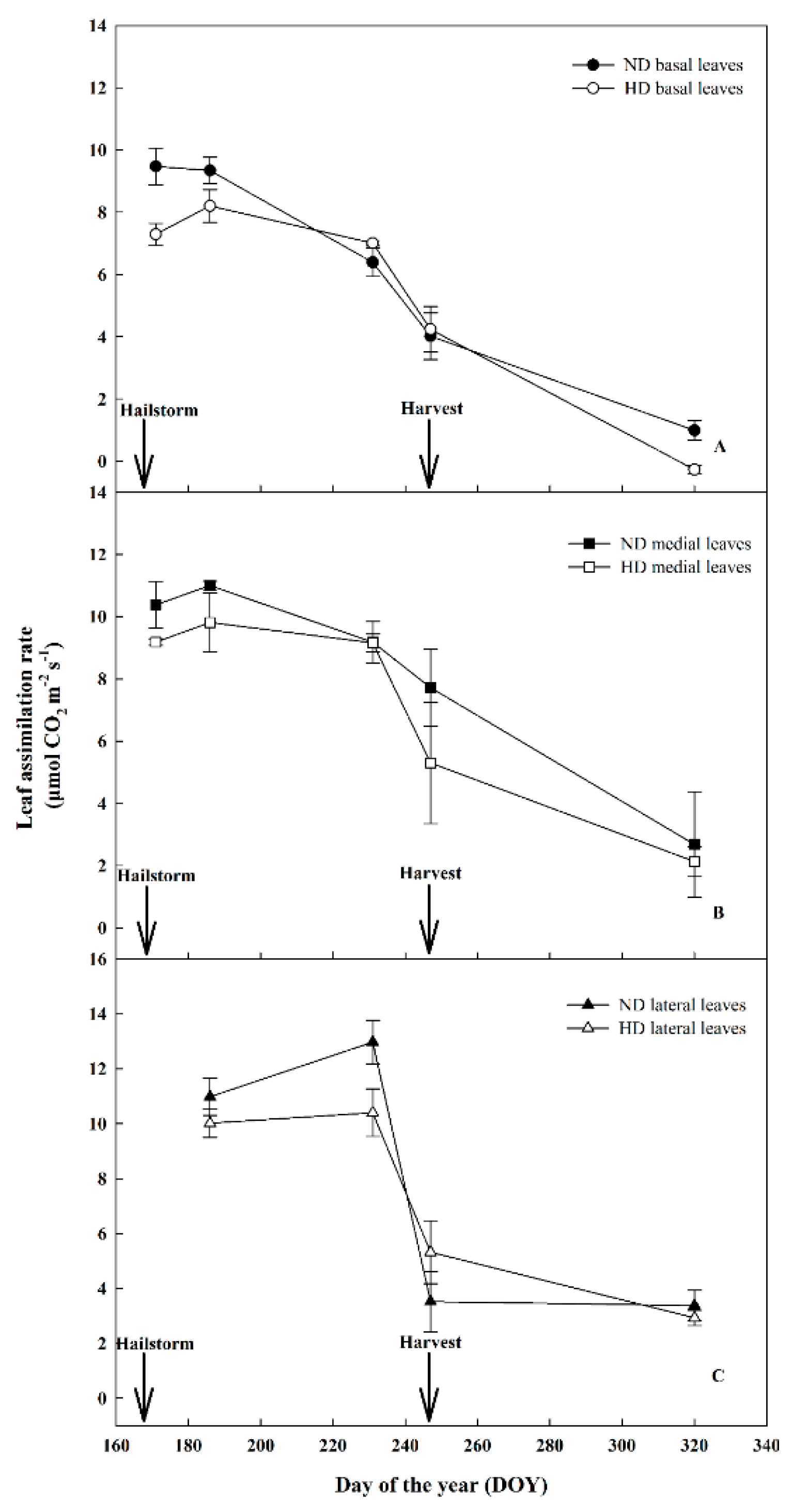
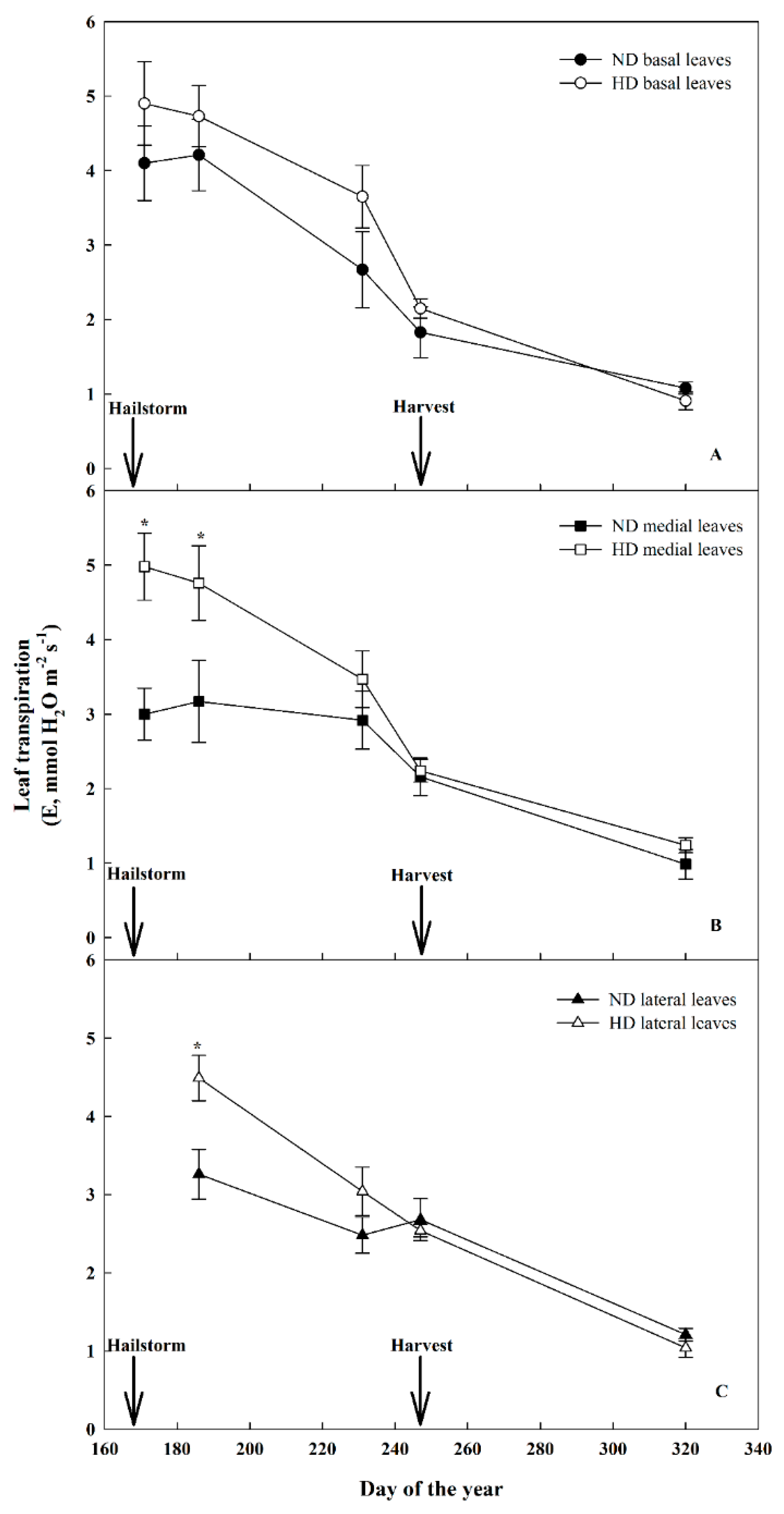
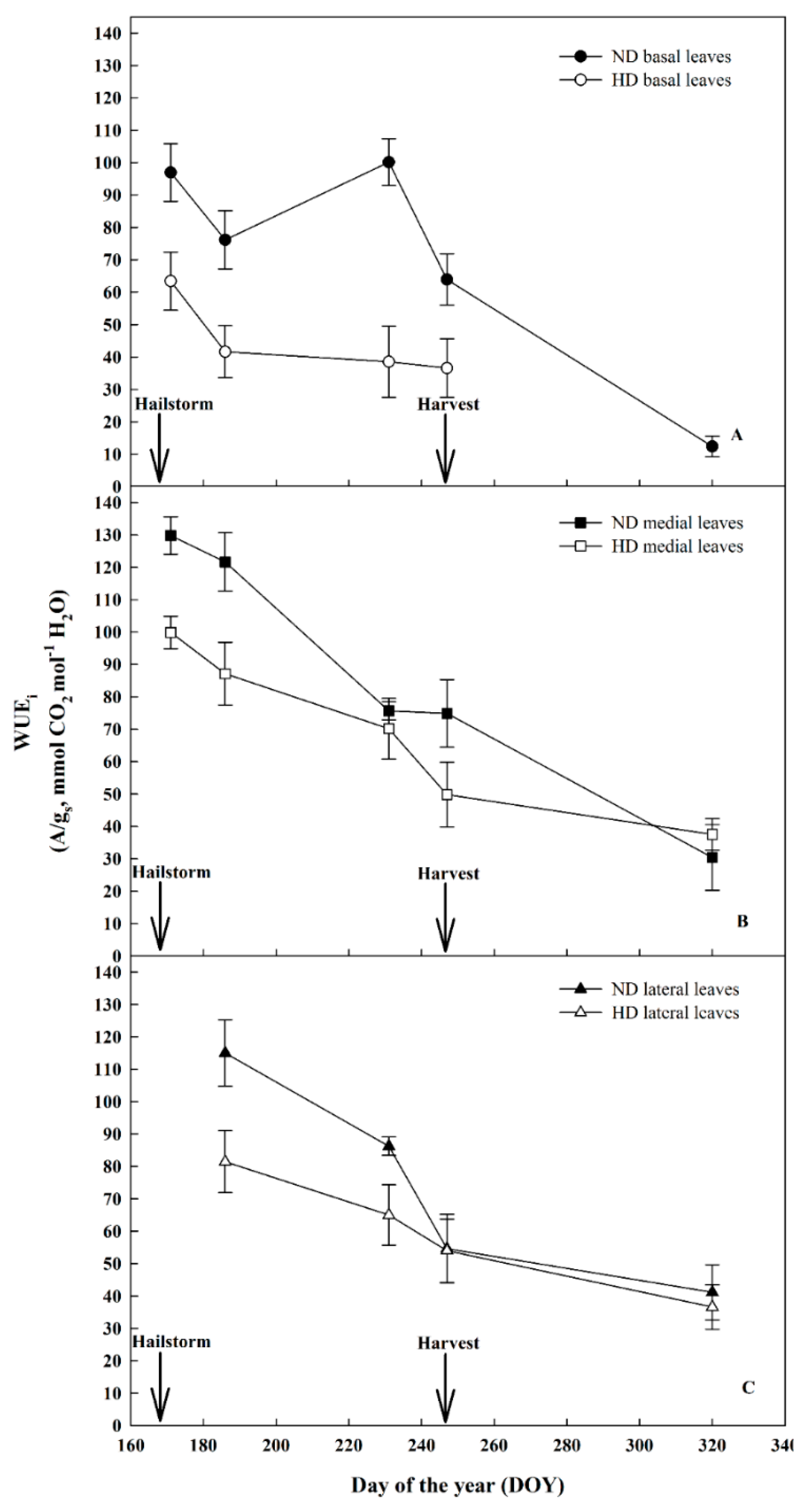
3.3. Effect of Natural Hail on Leaf Gas Exchange
3.4. Effect of Natural Hail on Productive Traits
3.5. Effect of Natural Hail on Fruit Composition
3.6. Effect of Natural Hail on Pruning Data, Vine Balance Indices, and Carbohydrate and Nitrogen Storage in Permanent Vine Organs and Shoot Fertility
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Logothetis, B. The development of the vine and of viticulture in Greece based on archaeological findings in the area. In Epistimoniki Epetiris tis Geoponikis kai Dasologikis Scholis; University of Thessaloniki: Thessaloniki, Greece, 1970; Volume 13, pp. 167–249, (In Greek with English summary). [Google Scholar]
- Staurakas, D.E. Ampelography, 2nd ed.; Ziti Press: Thessaloniki, Greece, 2015; pp. 588–594. ISBN 978-960-456-241-1. [Google Scholar]
- Sioutas, M.; Meaden, G.; Webb, J. Hail frequency distribution and intensity in Northern Greece. Atmos. Res. 2007, 93, 526–533. [Google Scholar] [CrossRef]
- Papagiannaki, K.; Lagouvardos, K.; Kotroni, V. A database of high-impact weather events in Greece: A descriptive impact analysis for the period 2001–2011. Nat. Hazards Earth Syst. Sci. 2013, 13, 727–736. [Google Scholar] [CrossRef]
- Papagiannaki, K.; Lagouvardos, K.; Kotroni, V.; Papagiannakis, G. Agricultural losses related to frost events: Use of the 850 hPa level temperature as an explanatory variable of the damage cost. Nat. Hazards Earth Syst. Sci. 2014, 14, 2375–2386. [Google Scholar] [CrossRef]
- Tartachnyk, I.; Blanke, M.M. Effect of mechanically-hail simulated on photosynthesis, dark respiration and transpiration of apple leaves. Environ. Exp. Bot. 2002, 48, 169–175. [Google Scholar] [CrossRef]
- Wells, L.D.; MacManus, P.S. Effects of simulated hail events and subsequent fungicide applications on cranberry fruit rot incidence and yield. Plant Dis. 2013, 97, 1207–1211. [Google Scholar] [CrossRef] [PubMed]
- Jalali, A.H. Potato (Solanum tuberosum L.) yield response to simulated hail damage. Arch. Agron. Soil Sci. 2013, 59, 981–987. [Google Scholar] [CrossRef]
- Miya, S.P.; Modi, A.T.; Mabhaudhi, T. Interactive effects of simulated hail damage and plant density on maize seed quality. Seed Sci. Technol. 2017, 45, 100–111. [Google Scholar] [CrossRef]
- Anda, A.; Burucs, Z.; Lõke, Z.; Decsi, É.K. Effects of Hail on Evapotranspiration and Plant Temperature of Maize. J. Agron. Crop Sci. 2002, 188, 335–341. [Google Scholar] [CrossRef]
- Robertson, A.E.; Munkwold, G.P.; Hurburg, C.R.; Ensley, S. Effects of Natural Hail Damage on Ear Rots, Mycotoxins, and Grain Quality Characteristics of Corn. Environ. Entomol. 2011, 103, 1193–1199. [Google Scholar] [CrossRef]
- Yue, Y.; Zhou, L.; Zhu, A.-X.; Ye, X. Vulnerability of cotton subjected to hail damage. PLoS ONE 2019, 14, e0210787. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, J.G.; Bleasdale, G.E.; Cadman, R.S.; Keens, I.L. Comparison of trellises and pruning levels for Sultana vines in the Murray Valley. J. Exp. Agric. Anim. Husb. 1979, 19, 634–640. [Google Scholar] [CrossRef]
- Williams, L.E.; Ayars, J.E. Water use of Thompson Seedless grapevines as affected by the application of gibberellic acid (GA3) and trunk girdling-Practices to increase berry size. Agric. For. Meteorol. 2005, 129, 85–94. [Google Scholar] [CrossRef]
- Warusavitharana, A.J.; Tambe, T.B.; Kshirsagar, D.B. Effect of cytokinins and brassinosteroid with gibberellic acid on yield and quality of Thompson Seedless grapes. Acta Hortic. 2008, 785, 217–224. [Google Scholar] [CrossRef]
- Abu-Zahra, T.R. Berry size of Thompson seedless as influenced by the application of Gibberellic acid and cane girdling. Pak. J. Bot. 2010, 42, 1755–1760. [Google Scholar]
- Webb, L.; Whiting, J.; Watt, A.; Hill, T.; Wigg, F.; Dunn, G.; Needs, S.; Barlow, E.W.R. Managing grapevines through severe heat: A survey of growers after the 2009 summer heatwave in south-eastern. Aust. J. Wine Res. 2010, 21, 147–165. [Google Scholar] [CrossRef]
- Flexas, J.; Bota, J.; Escalona, J.M.; Sampol, B.; Medrano, H. Effects of drought on photosynthesis in grapevines under field conditions: An evaluation of stomatal and mesophyll limitations. Funct. Plant Biol. 2002, 29, 461–471. [Google Scholar] [CrossRef]
- Mosedale, J.R.; Wilson, R.J.; Maclean, I.M.D. Climate Change and Crop Exposure to Adverse Weather: Changes to Frost Risk and Grapevine Flowering Conditions. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Morales, M.; Toro, G.; Riquelme, A.; Sellés, G.; Pinto, M.; Ferreyra, R. Effect of different rootstocks on photosynthesis and nutritional response of grapevines cultivar ‘Sultanina’ under flooding stress. Acta Hortic. 2014, 1045, 123–131. [Google Scholar] [CrossRef]
- Takahashi, K.; Kuranaka, M.; Miyagawa, A. The effects of wind on grapevine growth: Windbreaks for vineyards. Bull. Shimane Agric. Exp. Stn. 1976, 14, 39–83. [Google Scholar]
- Manja, K.; Aoun, M. The use of nets for tree fruit crops and their impact on the production: A review. Sci. Hortic. 2019, 246, 110–122. [Google Scholar] [CrossRef]
- Keller, M.; Romero, P.; Gohil, H.; Smithyman, R.P.; Riley, W.R.; Casassa, L.F.; Harbertson, J.F. Deficit irrigation alters grapevine growth, physiology, and fruit microclimate. Am. J. Enol. Vitic. 2016, 67, 426–435. [Google Scholar] [CrossRef]
- Hendrickson, L.; Ball, M.C.; Wood, J.T.; Chow, W.S.; Furbank, R.T. Low temperature effects on photosynthesis and growth of grapevine. Plant Cell Environ. 2004, 27, 795–809. [Google Scholar] [CrossRef]
- Petoumenou, G.D. Growth Rate, Morpho-Physiological and Ultrastructural Behaviour in Vitis Vinifera L. under Limited and Unlimited Conditions. Ph.D. Thesis, Università degli Studi di Perugia, Perugia, Italy, 2007. [Google Scholar]
- Greer, D.H.; Weedon, M.M. The impact of high temperatures on Vitis vinifera cv. Semillon grapevine performance and berry ripening. Front. Plant Sci. 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Orduña, R. Climate change associated effects on grape and wine quality and production. Food Res. Int. 2010, 43, 1844–1855. [Google Scholar] [CrossRef]
- Mori, K.; Goto-Yamamoto, N.; Kitayama, M.; Hashizume, K. Loss of anthocyanins in red-wine grape under high temperature. J. Exp. Bot. 2007, 58, 1935–1945. [Google Scholar] [CrossRef]
- Sweetman, C.; Sadras, V.O.; Hancock, R.D.; Soole, K.L.; Ford, C.M. Metabolic effects of elevated temperature on organic acid degradation in ripening Vitis vinifera fruit. J. Exp. Bot. 2014, 65, 5975–5988. [Google Scholar] [CrossRef]
- Lorentz, D.H.; Eichhorn, K.W.; Bleiholder, H.; Klose, R.; Meier, U.; Weber, E. Growth Stages of the Grapevine: Phenological growth stages of the grapevine (Vitis vinifera L. ssp. vinifera)-Codes and descriptions according to the extended BBCH scale. Aust. J. Grape Wine Res. 1995, 1, 100–103. [Google Scholar] [CrossRef]
- Tello, J.; Ibáñez, J. Evaluation of indexes for the quantitative and objective estimation of grapevine bunch compactness. Vitis 2014, 53, 9–16. [Google Scholar]
- Slinkard, K.; Singleton, V.L. Total Phenol Analysis: Automation and Comparison with Manual Methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar]
- Morris, D.L. Quantitative determination of carbohydrates with Dreywood’s anthrone reagent. Science 1948, 107, 254–255. [Google Scholar] [CrossRef]
- Loewus, F.A. Improvement in anthrone method for determination of carbohydrates. Anal. Chem. 1952, 24, 219. [Google Scholar] [CrossRef]
- Fernandes, G.W.; Oki, Y.; Sales, N.M.; Quintini, A.V.; Freitas, C.; Caires, T.B. Hailstorm impact across plant taxa: Leaf fall in a mountain environment. Neotrop. Biol. Conserv. 2012, 7, 8–15. [Google Scholar]
- Vescovo, L.; Gianelle, D.; Dalponte, M.; Miglietta, F.; Carotenuto, F.; Torresan, C. Hail defoliation assessment in corn (Zea mays L.) using airborne LiDAR. Field Crop. Res. 2016, 196, 426–437. [Google Scholar] [CrossRef]
- Candolfi-Vasconcelos, M.C.; Koblet, W. Yield, fruit-quality, bud fertility and starch reserves of the wood as a function of leaf removal in Vitis vinifera-Evidence of compensation and stress recovering. Vitis 1990, 29, 199–221. [Google Scholar]
- Caspari, H.W.; Lang, A.; Alspach, P. Effects of girdling and leaf removal on fruit set and vegetative growth in grape. Am. J. Enol. Vitic. 1998, 49, 359–366. [Google Scholar]
- Tartachnyk, I.; Blanke, M.M. Temperature, evapotranspiration and primary photochemical responses of apple leaves to hail. J. Plant Physiol. 2008, 165, 1847–1852. [Google Scholar] [CrossRef]
- Aldea, M.; Hamilton, J.G.; Resti, J.P.; Zangelr, A.R.; Byerenbaur, M.R.; Delucia, E.H. Indirect effects of insect herbivory on leaf gas exchange in soybean. Plant Cell Environ. 2005, 28, 402–411. [Google Scholar] [CrossRef]
- Kerchev, P.I.; Fenton, B.; Foyer, C.H.; Hancock, R.D. Plant responses to insect herbivory: Interactions between photosynthesis, reactive oxygen species and hormonal signalling pathways. Plant Cell Environ. 2012, 35, 441–453. [Google Scholar] [CrossRef]
- Vasconcelos, M.C.; Castagnoli, S. Leaf canopy structure and wine performance. Am. J. Enol. Vitic. 2000, 51, 390–396. [Google Scholar]
- Tello, J.; Ibáñez, J. What we know about grapevine bunch compactness? A state-of-the-art review. Aust. J. Wine Res. 2018, 24, 6–23. [Google Scholar] [CrossRef]
- Sabbatini, P.; Howell, G.S. Effects of early defoliation on yield, fruit composition, and harvest season cluster rot complex of grapevines. HortScience 2010, 45, 1804–1808. [Google Scholar] [CrossRef]
- Pastore, C.; Zenoni, S.; Fasoli, M.; Pezzotti, M.; Tornielli, G.B.; Filippetti, I. Selective defoliation affects plant growth, fruit transcriptional ripening program and flavonoid metabolism in grapevine. BMC Plant Biol. 2013, 13, 30. [Google Scholar] [CrossRef]
- Poni, S.; Casalini, L.; Bernizzoni, F.; Civardi, S.; Intrieri, C. Effects of early defoliation on shoot photosynthesis, yield components, and grape composition. Am J. Enol. Vitic. 2006, 57, 397–407. [Google Scholar]
- Poni, S.; Bernizzoni, F.; Civardi, S. The effect of early leaf removal on whole-canopy gas exchange and vine performance of Vitis vinifera L. “Sangiovese”. Vitis 2008, 47, 1–6. [Google Scholar]
- Pastore, C.; Zenoni, S.; Tornielli, G.B.; Allegro, G.; Dal Santo, S.; Valentini, G.; Intrieri, C.; Pezzotti, M.; Filippetti, I. Increasing the source/sink ratio in Vitis vinifera (cv. Sangiovese) induces extensive transcriptome reprogramming and modifies berry ripening. BMC Genomics 2011, 12, 631. [Google Scholar] [CrossRef]
- Ranganna, S. Maturity Indeces and Quality Criteria. Grapes in Handbook of Analysis and Quality Control for Fruit and Vegetable Products, 2nd ed.; Tata McGraw-Hill Publishing Company Ltd.: New Delhi, India, 1986; p. 171. ISBN 007-451-851-8. [Google Scholar]
- Poni, S.; Bernizzoni, F.; Civardi, S.; Libelli, N. Effects of pre-bloom leaf removal on growth of berry tissues and must composition in two red Vitis vinifera L. cultivars. Aust. J. Grape Wine Res. 2009, 15, 185–193. [Google Scholar] [CrossRef]
- Mirás-Avalos, J.M.; Intrigliolo, D.S. Grape Composition under Abiotic Constrains: Water Stress and Salinity. Front. Plant Sci. 2017, 8, 851. [Google Scholar] [CrossRef]
- Sadras, V.O.; Moran, M.A. Elevated temperature decouples anthocyanins and sugars in berries of Shiraz and Cabernet Franc. Aust. J. Grape Wine Res. 2012, 18, 115–122. [Google Scholar] [CrossRef]
- Wu, B.H.; Niu, N.; Li, J.H.; Li, S.H. Leaf:fruit ratio affects the proteomic profile of grape berry skins. J. Am. Soc. Hortic. Sci. 2013, 138, 416–427. [Google Scholar] [CrossRef]
- May, P. The value of an estimate of fruiting potential in the ‘Sultana’. Vitis 1961, 3, 15–26. [Google Scholar]
- Palliotti, A.; Gardi, T.; Berrios, J.G.; Civardi, S.; Poni, S. Early source limitation as a tool for yield control and wine quality improvement in a high-yielding red Vitis vinifera L. cultivar. Sci. Hortic. 2012, 145, 10–16. [Google Scholar] [CrossRef]
- Lohitnavy, N.; Bastian, S.; Collins, C. Early leaf removal increases flower abscission in Vitis vinifera‚ Semillon. Vitis 2010, 49, 51–53. [Google Scholar]
 northeastern Peloponnese, Greece). (B) The red and yellow dotted lines indicate the approximate areas of the vineyard that were affected or unaffected by the hailstorm, respectively. Black arrowheads indicate the approximate direction of the hailstorm on 18 June 2015.
northeastern Peloponnese, Greece). (B) The red and yellow dotted lines indicate the approximate areas of the vineyard that were affected or unaffected by the hailstorm, respectively. Black arrowheads indicate the approximate direction of the hailstorm on 18 June 2015.
 northeastern Peloponnese, Greece). (B) The red and yellow dotted lines indicate the approximate areas of the vineyard that were affected or unaffected by the hailstorm, respectively. Black arrowheads indicate the approximate direction of the hailstorm on 18 June 2015.
northeastern Peloponnese, Greece). (B) The red and yellow dotted lines indicate the approximate areas of the vineyard that were affected or unaffected by the hailstorm, respectively. Black arrowheads indicate the approximate direction of the hailstorm on 18 June 2015.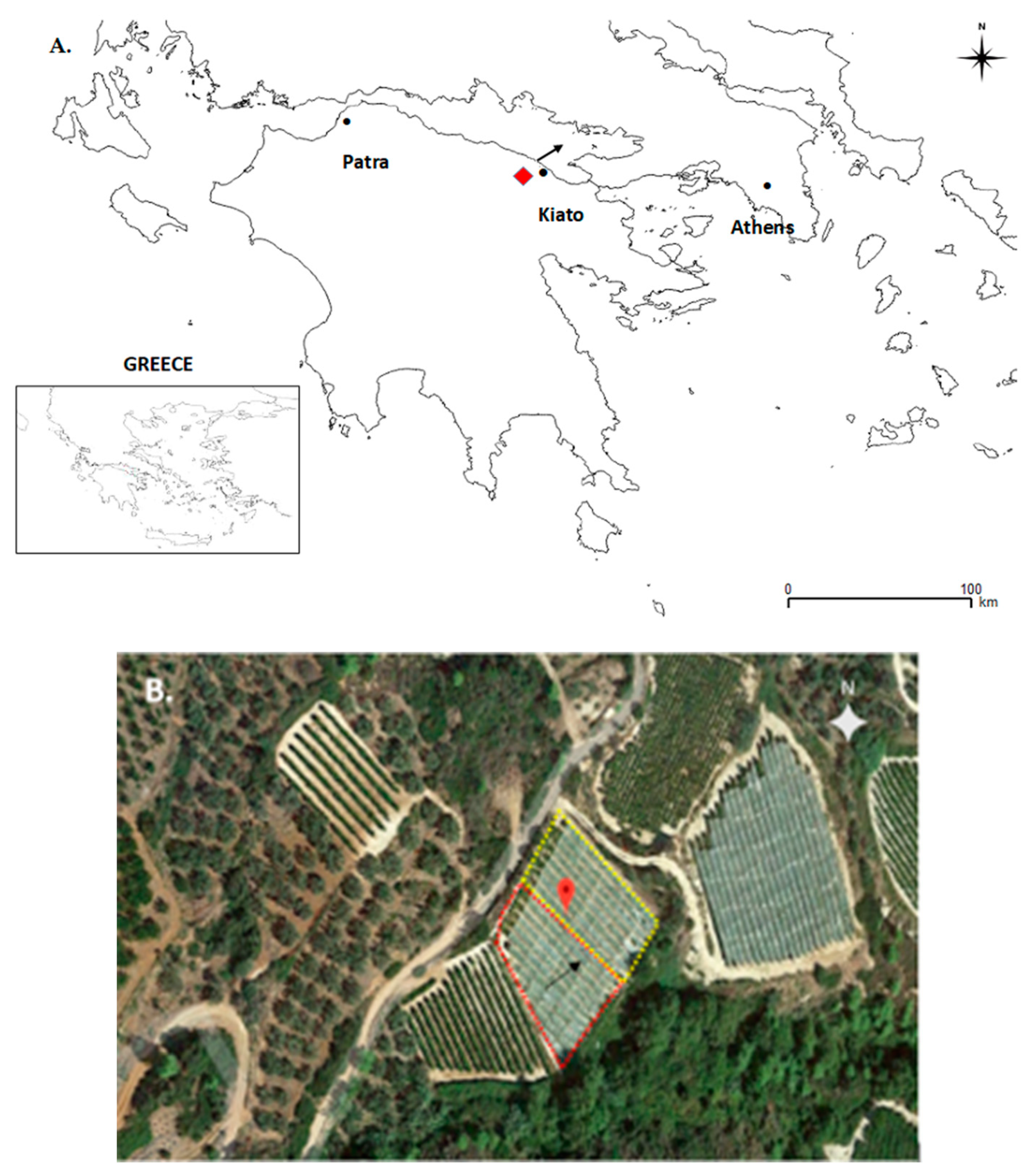



| Parameters | ND | HD | Signif. a |
|---|---|---|---|
| Just after hail (DOY 169) | |||
| Total leaf area from primary canes (m2/vine) | 8.90 ± 0.33 | 7.50 ± 0.18 | * |
| Total leaf area from lateral canes (m2/vine) | 2.11 ± 0.12 | 1.70 ± 0.10 | * |
| Total leaf area (m2/vine) | 11.01 ± 0.28 | 9.20 ± 0.17 | * |
| At harvest (DOY 248) | |||
| Shoots/vine | 12.7 ± 0.05 | 13.0 ± 0.08 | ns |
| Lateral shoots/vine | 9.43 ± 0.04 | 12.34 ± 0.06 | * |
| Cane diameter at 3rd internode (mm) | 10.37 ± 0.29 | 10.73 ± 0.13 | ns |
| Cane diameter at 10th internode (mm) | 7.02 ± 0.39 | 7.15 ± 0.40 | ns |
| Lateral cane diameter at 3rd internode (mm) | 5.70 ± 0.22 | 5.65 ± 0.13 | ns |
| Total leaf area from primary canes (m2/vine) | 10.97 ± 0.21 | 10.02 ± 0.28 | * |
| Total leaf area from lateral shoots (m2/vine) | 3.13 ± 0.09 | 3.40 ± 0.07 | * |
| Total leaf area (m2/vine) | 14.38 ± 0.28 | 13.16 ± 0.37 | * |
| Parameters | ND | HD | Signif. a |
|---|---|---|---|
| Bunches (number/vine) | 15.0 ± 1.33 | 13.2 ± 2.65 | ns |
| Yield (kg/vine) | 13.10 ± 0.33 | 8.20 ± 0.65 | * |
| Bunch weight (g) | 874.10 ± 107.61 | 620.20 ± 117.79 | * |
| Bunch length (cm) | 21.8 ± 1.6 | 20.9 ± 1.2 | ns |
| Bunch width (cm) | 17.2 ± 0.7 | 14.6 ± 0.8 | * |
| Bunch compactness index (g/(cm)2) | 1.89 ± 0.13 | 1.40 ± 0.19 | * |
| Undamaged berries per bunch (n) | 167.0 ± 3.2 a | 93.0 ± 3.1 | ** |
| Undamaged berry weight (g) | 5.07 ± 0.51 | 5.19 ± 0.61 | ns |
| Hail-damaged berries per bunch (n) | - † | 45.0 ± 1.8 | - |
| Hail-damaged berry fresh weight (g) | - | 3.3 ± 0.02 | - |
| Undamaged berry length (mm) | 27.11 ± 0.44 | 26.2 ± 0.66 | ns |
| Undamaged berry diameter (mm) | 17.88 ± 0.31 | 17.57 ± 0.42 | ns |
| Force pedicel detachment (N) | 1.6 ± 0.22 | 1.1 ± 0.12 | ns |
| Parameters | ND | HD | Signif. a |
|---|---|---|---|
| Total soluble solids (TSS, °Bx) | 20.0 ± 1.13 | 21.0 ± 0.85 | * |
| Total acidity (TA, %) | 0.39 ± 0.01 | 0.37 ± 0.02 | ns |
| Must pH | 4.27 ± 0.08 | 4.37 ± 0.10 | ns |
| Maturity index (TSS/TA) | 51.1 ± 0.05 | 57.0 ± 0.19 | ns |
| Total phenolics (mg/kg) | 1900.7 ± 39.2 | 975.5 ± 15.4 | * |
| Parameters | ND | HD | Signif. a |
|---|---|---|---|
| Pruning weight (kg/vine) | 2.99 ± 1.64 | 3.03 ± 0.13 | ns |
| Yield-to-pruning-weight ratio (kg/kg) | 4.38 ± 0.04 | 2.71 ± 0.13 | ** |
| Leaf-to-fruit ratio (m2/kg) | 1.10 ± 0.15 | 1.60 ± 0.09 | * |
| Soluble solids of primary canes at 3rd internode (mg/g DW) | 167.88 ± 16.98 | 155.79 ± 9.04 | ns |
| Starch of primary canes at 3rd internode (mg/g DW) | 40.62 ± 14.09 | 50.82 ± 11.58 | ns |
| Soluble solids of primary canes at 10th internode (mg/g DW) | 147.06 ± 3.63 | 151.66 ± 7.12 | ns |
| Starch of primary canes at 10th internode (mg/g DW) | 35.96 ± 2.49 | 35.58 ± 7.61 | ns |
| Soluble solids of lateral canes at 3rd internode (mg/g DW) | 119.19 ± 7.66 | 146.36 ± 17.95 | ns |
| Starch of canes of lateral canes at 3rd internode (mg/g DW) | 22.48 ± 10.25 | 39.67 ± 5.47 | ns |
| ǂ Bud fertility index | 1.55 ± 0.20 | 2.08 ± 0.40 | ns |
| ǂ Blind buds/vine | 4.00 ± 1.73 | 5.67 ± 2.40 | ns |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petoumenou, D.G.; Biniari, K.; Xyrafis, E.; Mavronasios, D.; Daskalakis, I.; Palliotti, A. Effects of Natural Hail on the Growth, Physiological Characteristics, Yield, and Quality of Vitis vinifera L. cv. Thompson Seedless under Mediterranean Growing Conditions. Agronomy 2019, 9, 197. https://doi.org/10.3390/agronomy9040197
Petoumenou DG, Biniari K, Xyrafis E, Mavronasios D, Daskalakis I, Palliotti A. Effects of Natural Hail on the Growth, Physiological Characteristics, Yield, and Quality of Vitis vinifera L. cv. Thompson Seedless under Mediterranean Growing Conditions. Agronomy. 2019; 9(4):197. https://doi.org/10.3390/agronomy9040197
Chicago/Turabian StylePetoumenou, Despoina G., Katerina Biniari, Efstratios Xyrafis, Dimitrios Mavronasios, Ioannis Daskalakis, and Alberto Palliotti. 2019. "Effects of Natural Hail on the Growth, Physiological Characteristics, Yield, and Quality of Vitis vinifera L. cv. Thompson Seedless under Mediterranean Growing Conditions" Agronomy 9, no. 4: 197. https://doi.org/10.3390/agronomy9040197
APA StylePetoumenou, D. G., Biniari, K., Xyrafis, E., Mavronasios, D., Daskalakis, I., & Palliotti, A. (2019). Effects of Natural Hail on the Growth, Physiological Characteristics, Yield, and Quality of Vitis vinifera L. cv. Thompson Seedless under Mediterranean Growing Conditions. Agronomy, 9(4), 197. https://doi.org/10.3390/agronomy9040197




