Grassland Management Influences the Response of Soil Respiration to Drought
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Design
2.3. Measurements of Soil Respiration and δ13C of Respired CO2
2.4. Statistical Analyses
3. Results
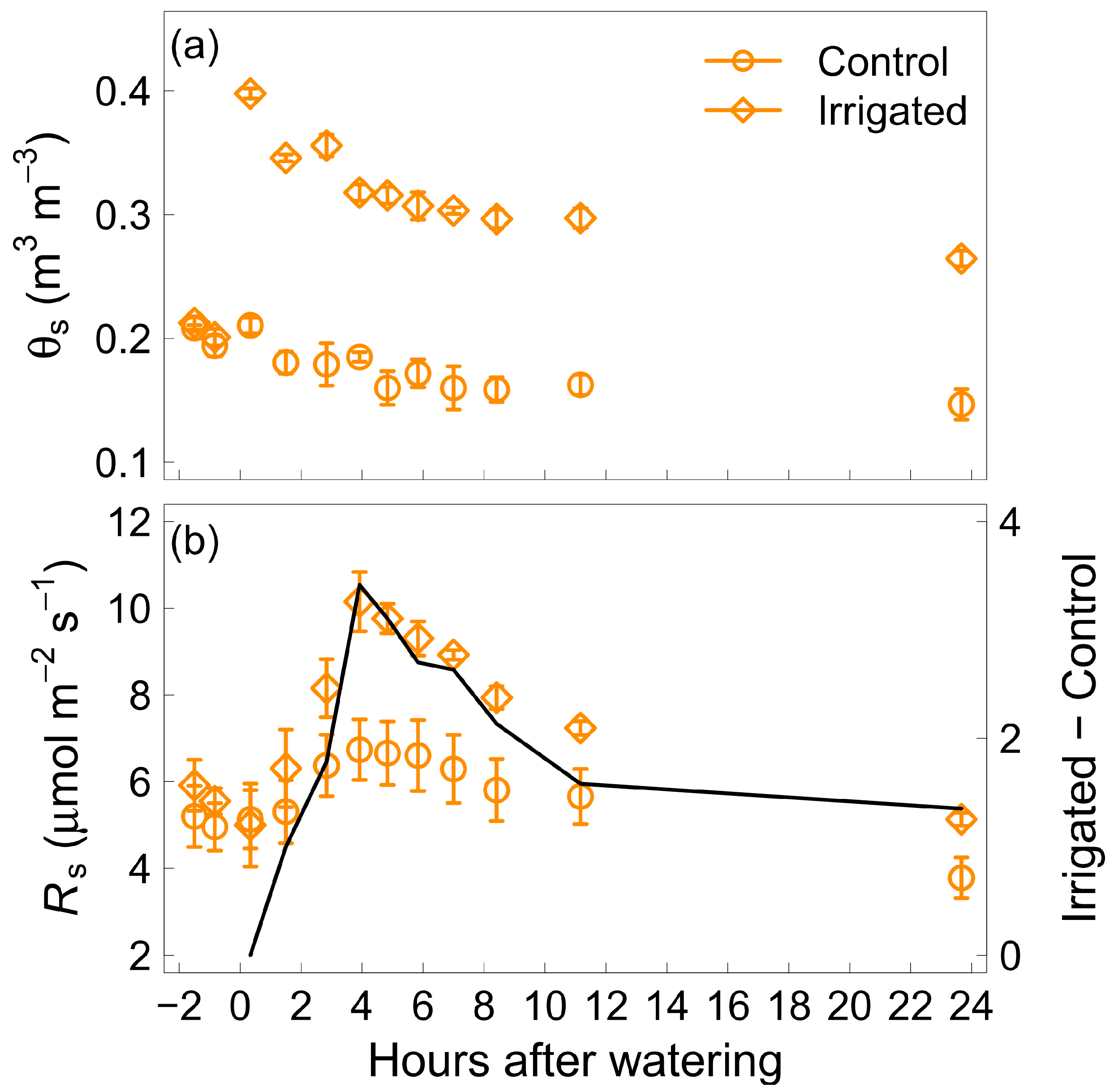
3.1. Hourly Measurements: Short-Term Response of Soil Respiration to Water Addition
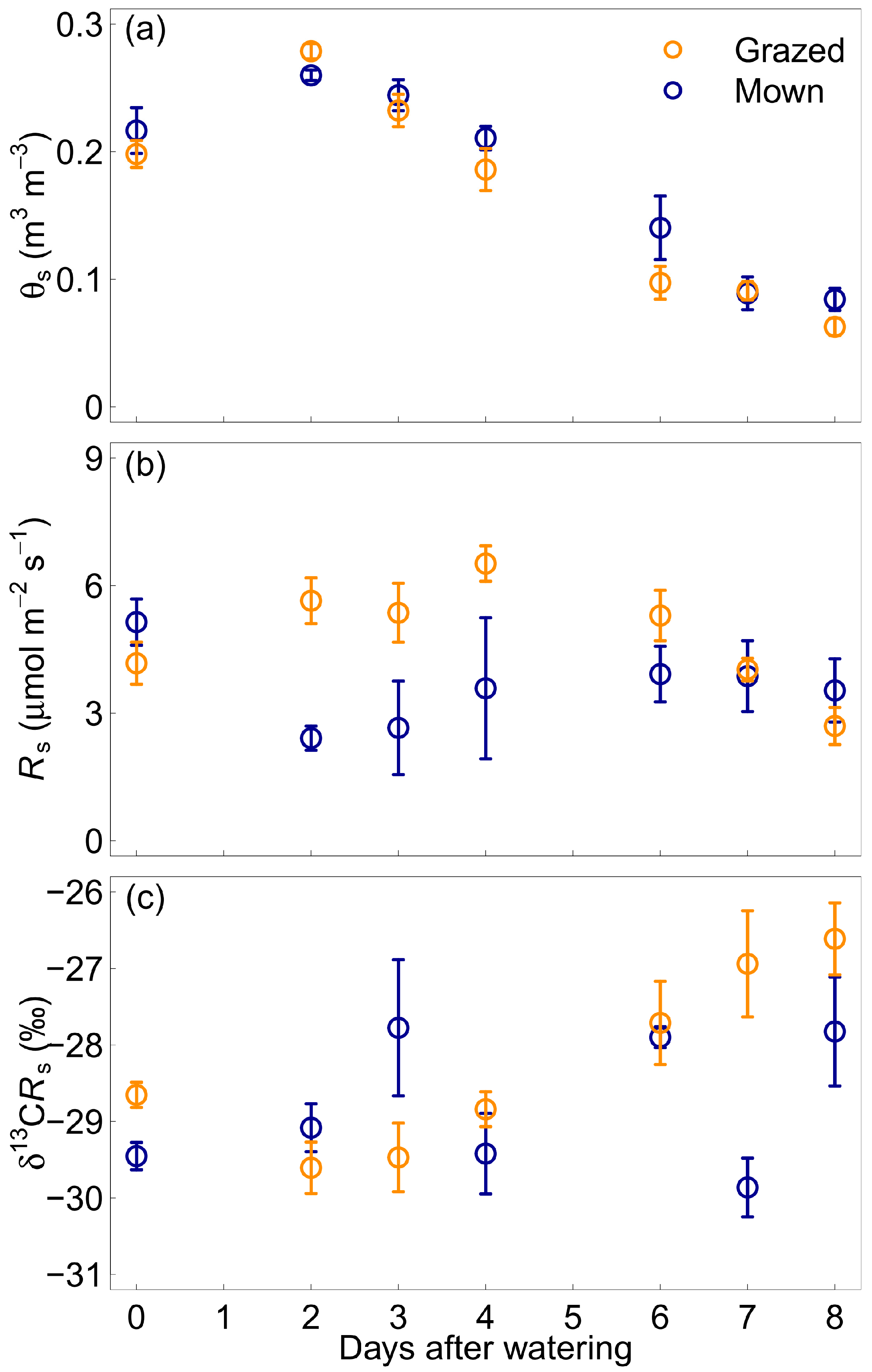
3.2. Daily Changes after Water Addition
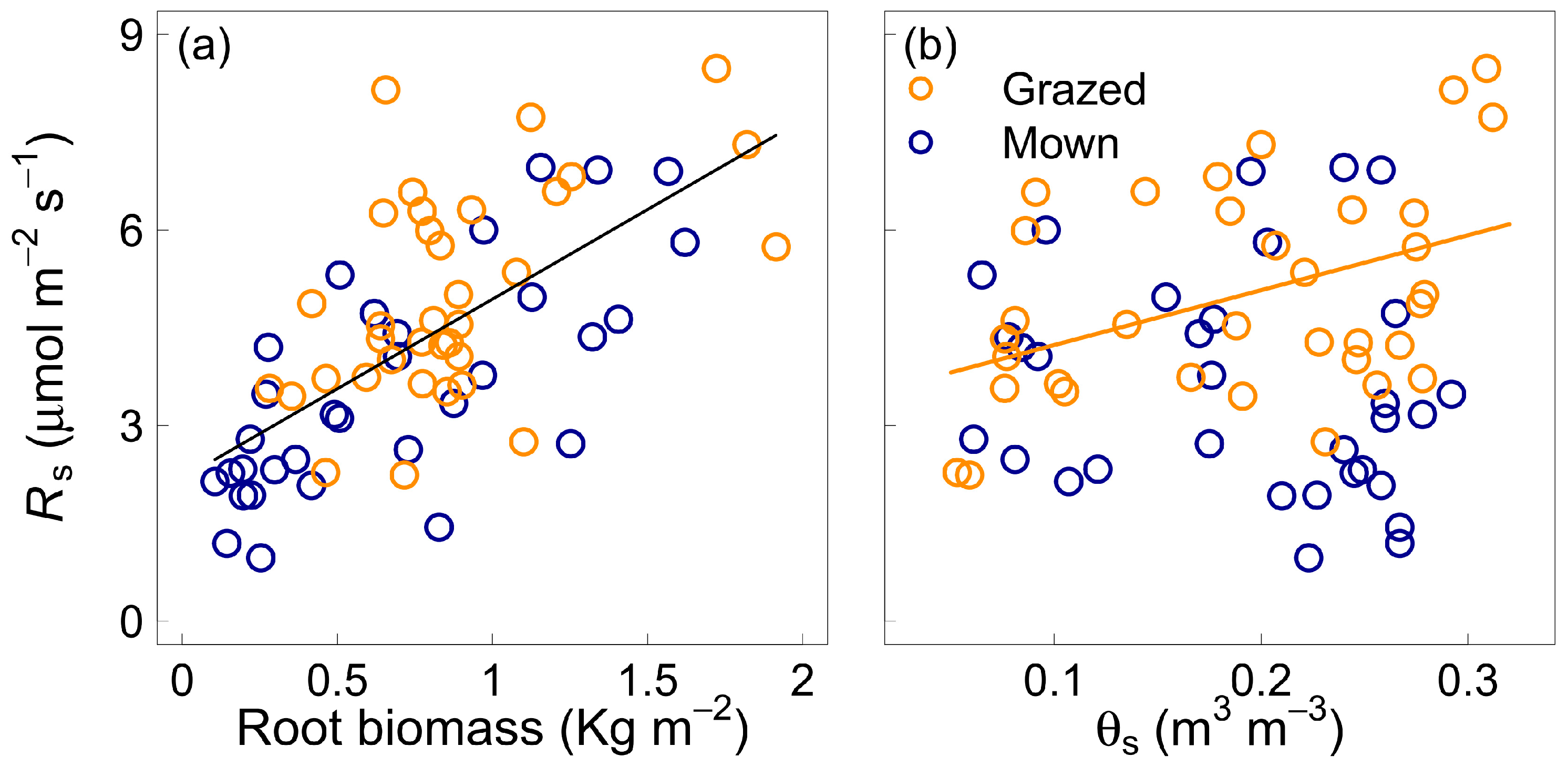
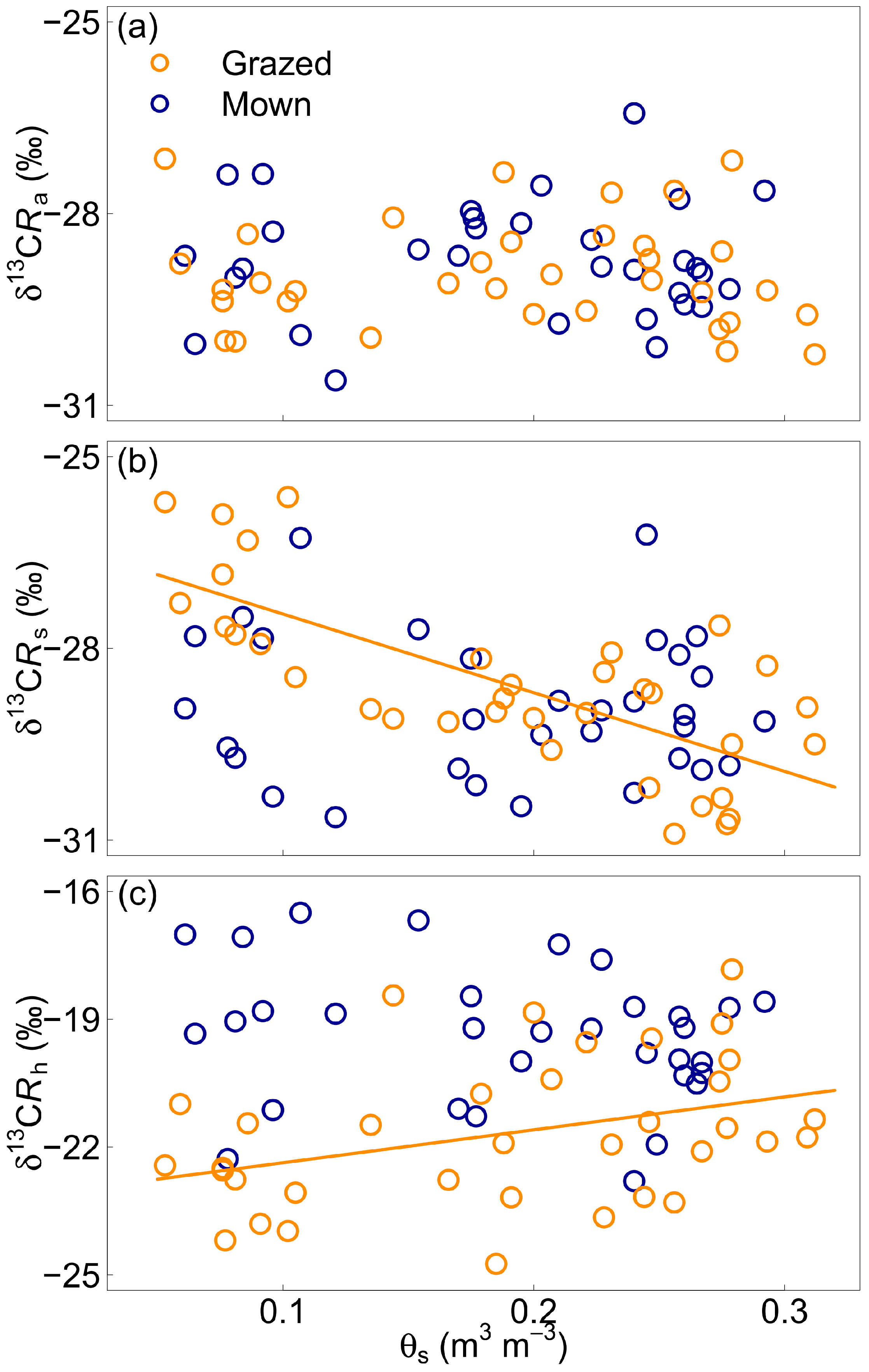
3.3. Effect of Soil Volumetric Water Content
4. Discussion
4.1. Birch Effect
4.2. Soil Water Content and Isotopic Discrimination
4.3. Soil Water Content and Components of Soil Respiration
4.4. Consequences for Soil Carbon Sequestration
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Rs | Soil respiration |
| δ13CRs | 13C isotopic signature of CO2 respired from the whole soil |
| δ13CRa | 13C isotopic signature of CO2 respired from roots |
| δ13CRh | 13C isotopic signature of CO2 respired from root and rhizosphere free soil |
| θs | Soil volumetric soil water content |
| Ts | Soil temperature at 100 mm depth |
References
- Steinfeld, H.; Gerber, P.; Wassenaar, T.; Castel, V.; Rosales, M.; Haan, C. Livestock’s Long Shadow: Environmental Issues and Options; Food & Agriculture Organization: Rome, Italy, 2006. [Google Scholar]
- Stockmann, U.; Adams, M.A.; Crawford, J.W.; Field, D.J.; Henakaarchchi, N.; Jenkins, M.; Minasny, B.; McBratney, A.B.; de Courcelles, V.D.R.; Singh, K.; et al. The knowns, known unknowns and unknowns of sequestration of soil organic carbon. Agric. Ecosyst. Environ. 2013, 164, 80–99. [Google Scholar] [CrossRef]
- De Coninck, H.; Revi, A.; Babiker, M.; Bertoldi, P.; Buckeridge, M.; Cartwright, A.; Dong, W.; Ford, J.; Fuss, S.; Hourcade, J.C.; et al. Strengthening and implementing the global response. In Global Warming of 1.5 °C, An IPCC Special Report; Masson-Delmotte, V., Zhai, P., Pörtner, H.O., Roberts, D., Skea, J., Shukla, P.R., Pirani, A., Eds.; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2018. [Google Scholar]
- Paustian, K.; Lehmann, J.; Ogle, S.; Reay, D.; Robertson, G.P.; Smith, P. Climate-smart soils. Nature 2016, 532, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Chabbi, A.; Lehmann, L.; Ciais, P.; Loescher, H.L.; Cotrufo, M.F.; Don, A.; San-Clements, M.; Schipper, L.; Six, J.; Smith, P.; et al. Aligning agriculture and climate policy. Nat. Clim. Chang. 2017, 7, 307–309. [Google Scholar] [CrossRef]
- Rumpel, C.; Crème, A.; Ngo, P.T.; Velásquez, G.; Mora, M.L.; Chabbi, A. The impact of grassland management on biogeochemical cycles involving carbon, nitrogen and phosphorus. J. Soil Sci. Plant Nutr. 2015, 15, 353–371. [Google Scholar] [CrossRef]
- Dignac, M.-F.; Derrien, D.; Barré, P.; Barot, S.; Cécillon, L.; Chenu, C.; Chevallier, T.; Freschet, G.T.; Garnier, P.; Guenet, B.; et al. Increasing soil carbon storage: Mechanisms, effects of agricultural practices and proxies. A review. Agron. Sustain. Dev. 2017, 37, 14. [Google Scholar] [CrossRef]
- Whitehead, D.; Schipper, L.A.; Pronger, J.; Moinet, G.Y.K.; Mudge, P.L.; Calvelo Pereira, R.; Kirschbaum, M.U.F.; McNally, S.R.; Beare, M.H.; Camps-Arbestain, M. Management practices to reduce losses or increase soil carbon stocks in temperate grazed grasslands: New Zealand as a case study. Agric. Ecosyst. Environ. 2018, 265, 432–443. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Andrews, J.A. Soil respiration and the global carbon cycle. Biogeochemistry 2000, 48, 7–20. [Google Scholar] [CrossRef]
- Field, C.B.; Intergovernmental Panel on Climate Change; Working Group II. Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2014. [Google Scholar]
- Borken, W.; Matzner, E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob. Chang. Biol. 2009, 15, 808–824. [Google Scholar] [CrossRef]
- Tezara, W.; Mitchell, V.J.; Driscoll, S.D.; Lawlor, D.W. Water stress inhibits plant photosynthesis by decreasing coupling factor and ATP. Nature 1999, 401, 914–917. [Google Scholar] [CrossRef]
- Moyano, F.E.; Manzoni, S.; Chenu, C. Responses of soil heterotrophic respiration to moisture availability: An exploration of processes and models. Soil Biol. Biochem. 2013, 59, 72–85. [Google Scholar] [CrossRef]
- Davidson, E.A.; Verchot, L.V.; Cattânio, J.H.; Ackerman, I.L.; Carvalho, J.E.M. Effects of soil water content on soil respiration. Biogeochemistry 2000, 48, 53–69. [Google Scholar] [CrossRef]
- Moinet, G.Y.K.; Cieraad, E.; Turnbull, M.H.; Whitehead, D. Effects of irrigation and addition of nitrogen fertiliser on net ecosystem carbon balance for a grassland. Sci. Total Environ. 2017, 579, 1715–1725. [Google Scholar] [CrossRef] [PubMed]
- Canarini, A.; Dijkstra, F.A. Dry-rewetting cycles regulate wheat carbon rhizodeposition, stabilization and nitrogen cycling. Soil Biol. Biochem. 2015, 81, 195–203. [Google Scholar] [CrossRef]
- Moinet, G.Y.K.; Cieraad, E.; Hunt, J.E.; Fraser, A.; Turnbull, M.H.; Whitehead, D. Soil heterotrophic respiration is insensitive to changes in soil water content but related to microbial access to organic matter. Geoderma 2016, 274, 68–78. [Google Scholar] [CrossRef]
- Huang, S.; Ye, G.; Lin, J.; Chen, K.; Xu, X.; Ruan, H.; Tan, F.; Chen, H.Y.H. Autotrophic and heterotrophic soil respiration responds asymmetrically to drought in a subtropical forest in the Southeast China. Soil Biol. Biochem. 2018, 123, 242–249. [Google Scholar] [CrossRef]
- Millard, P.; Midwood, A.J.; Hunt, J.E.; Barbour, M.M.; Whitehead, D. Quantifying the contribution of soil organic matter turnover to forest soil respiration, using natural abundance δ13C. Soil Biol. Biochem. 2010, 42, 935–943. [Google Scholar] [CrossRef]
- Moinet, G.Y.K.; Hunt, J.E.; Kirschbaum, M.U.F.; Morcom, C.P.; Midwood, A.J.; Millard, P. The temperature sensitivity of soil organic matter decomposition is constrained by microbial access to substrates. Soil Biol. Biochem. 2018, 116, 333–339. [Google Scholar] [CrossRef]
- Moinet, G.Y.K.; Midwood, A.J.; Hunt, J.E.; Whitehead, D.; Hannam, K.D.; Jenkins, M.; Brewer, M.J.; Adams, M.A.; Millard, P. Estimates of rhizosphere priming effects are affected by soil disturbance. Geoderma 2018, 313, 1–6. [Google Scholar] [CrossRef]
- Boström, B.; Comstedt, D.; Ekblad, A. Isotope fractionation and 13C enrichment in soil profiles during the decomposition of soil organic matter. Oecologia 2007, 153, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Snell, H.S.K.; Robinson, D.; Midwood, A.J. Minimising methodological biases to improve the accuracy of partitioning soil respiration using natural abundance 13C. Rapid Commun. Mass Spectrom. 2014, 28, 2341–2351. [Google Scholar] [CrossRef] [PubMed]
- Soussana, J.F.; Allard, V.; Pilegaard, K.; Ambus, P.; Amman, C.; Campbell, C.; Ceschia, E.; Clifton-Brown, J.; Czobel, S.; Domingues, R.; et al. Full accounting of the greenhouse gas (CO2, N2O, CH4) budget of nine European grassland sites. Agric. Ecosyst. Environ. 2007, 121, 121–134. [Google Scholar] [CrossRef]
- Soussana, J.F.; Tallec, T.; Blanfort, V. Mitigating the greenhouse gas balance of ruminant production systems through carbon sequestration in grasslands. Animal 2010, 4, 334–350. [Google Scholar] [CrossRef] [PubMed]
- FAO-ISRIC. Guidelines for Soil Description, revised 3rd ed.; Food and Agricultural Organisation: Rome, Italy, 1990. [Google Scholar]
- Chabbi, A.; Kögel-Knabner, I.; Rumpel, C. Stabilised carbon in subsoil horizons is located in spatially distinct parts of the soil profile. Soil Biol. Biochem 2009, 41, 256–261. [Google Scholar] [CrossRef]
- Senapati, N.; Chabbi, A.; Giostri, A.F.; Yeluripati, J.B.; Smith, P. Modelling nitrous oxide emissions from mown-grass and grain-cropping systems: Testing and sensitivity analysis of DailyDayCent using high frequency measurements. Sci. Total Environ. 2016, 572, 955–977. [Google Scholar] [CrossRef] [PubMed]
- Midwood, A.J.; Thornton, B.; Millard, P. Measuring the 13C content of soil-respired CO2 using a novel open chamber system. Rapid Commun. Mass Spectrom. 2008, 22, 2073–2081. [Google Scholar] [CrossRef] [PubMed]
- Midwood, A.J.; Millard, P. Challenges in measuring the δ13C of the soil surface CO2 efflux. Rapid Commun. Mass Spectrom. 2011, 25, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Zuur, A.F.; Ieno, E.N.; Walker, N.J.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Gail, M., Krickeberg, K., Samet, J.M., Tsiatis, A., Wong, W., Eds.; Spring Science and Business Media: New York, NY, USA, 2009. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach; Springer: New York, NY, USA, 2003. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Powers, H.H.; Hunt, J.E.; Hanson, D.T.; McDowell, N.G. A dynamic soil chamber system coupled with a tunable diode laser for online measurements of δ13C, δ18O, and efflux rate of soil-respired CO2: Measurements of δ13C, δ18O, and efflux rate of soil-respired CO2. Rapid Commun. Mass Spectrom. 2010, 24, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Fierer, N.; Schimel, J.P. Effects of drying–rewetting frequency on soil carbon and nitrogen transformations. Soil Biol. Biochem. 2002, 34, 777–787. [Google Scholar] [CrossRef]
- Read, J.; Farquhar, G. Comparative studies in Nothofagus (Fagaceae). I. Leaf carbon isotope discrimination. Funct. Ecol. 1991, 5, 684–695. [Google Scholar] [CrossRef]
- Schulze, E.-D.; Williams, R.J.; Farquhar, G.D.; Schulze, W.; Langridge, J.; Miller, J.M.; Walker, B.H. Carbon and nitrogen isotope discrimination and nitrogen nutrition of trees along a rainfall gradient in northern Australia. Funct. Plant Biol. 1998, 25, 413–425. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Gavrichkova, O. Review: Time lag between photosynthesis and carbon dioxide efflux from soil: A review of mechanisms and controls. Glob. Chang. Biol. 2010, 16, 3386–3406. [Google Scholar] [CrossRef]
- Barthel, M.; Cieraad, E.; Zakharova, A.; Hunt, J.E. Sudden cold temperature delays plant carbon transport and shifts allocation from growth to respiratory demand. Biogeosciences 2014, 11, 1425–1433. [Google Scholar] [CrossRef]
- Raich, J.W.; Schlesinger, W.H. The global carbon dioxide flux in soil respiration and its relationship to vegetation and climate. Tellus B 1992, 44, 81–99. [Google Scholar] [CrossRef]
- Hanson, P.J.; Edwards, N.T.; Garten, C.T.; Andrews, J.A. Separating root and soil microbial contributions to soil respiration: A review of methods and observations. Biogeochemistry 2000, 48, 115–146. [Google Scholar] [CrossRef]
- Subke, J.-A.; Inglima, I.; Francesca Cotrufo, M. Trends and methodological impacts in soil CO2 efflux partitioning: A metaanalytical review. Glob. Chang. Biol. 2006, 12, 921–943. [Google Scholar] [CrossRef]
- Flanagan, L.B.; Wever, L.A.; Carlson, P.J. Seasonal and interannual variation in carbon dioxide exchange and carbon balance in a northern temperate grassland. Glob. Chang. Biol. 2002, 8, 599–615. [Google Scholar] [CrossRef]
- Hunt, J.E.; Kelliher, F.M.; McSeveny, T.M.; Ross, D.J.; Whitehead, D. Long-term carbon exchange in a sparse, seasonally dry tussock grassland. Glob. Chang. Biol. 2004, 10, 1785–1800. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, H.; Jia, G.; Wylie, B.; Gilmanov, T.; Howard, D.; Ji, L.; Xiao, J.; Li, J.; Yuan, W.; et al. Net ecosystem productivity of temperate grasslands in northern China: An upscaling study. Agric. For. Meteorol. 2014, 184, 71–81. [Google Scholar] [CrossRef]
- Sanaullah, M.; Chabbi, A.; Rumpel, C.; Kuzyakov, Y. Carbon allocation in grassland communities under drought stress followed by 14C pulse labeling. Soil Biol. Biochem. 2012, 55, 132–139. [Google Scholar] [CrossRef]
- White, L.M. Carbohydrate reserves of grasses: A review. J. Range Manag. 1973, 26, 13–18. [Google Scholar] [CrossRef]
- Steen, E.; Larsson, K. Carbohydrates in roots and rhizomes of perennial grasses. New Phytol. 1986, 104, 339–346. [Google Scholar] [CrossRef]
- Donaghy, D.J.; Fulkerson, W.J. The importance of water-soluble carbohydrate reserves on regrowth and root growth of Lolium perenne (L.). Grass Forage Sci. 1997, 52, 401–407. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moinet, G.Y.K.; Midwood, A.J.; Hunt, J.E.; Rumpel, C.; Millard, P.; Chabbi, A. Grassland Management Influences the Response of Soil Respiration to Drought. Agronomy 2019, 9, 124. https://doi.org/10.3390/agronomy9030124
Moinet GYK, Midwood AJ, Hunt JE, Rumpel C, Millard P, Chabbi A. Grassland Management Influences the Response of Soil Respiration to Drought. Agronomy. 2019; 9(3):124. https://doi.org/10.3390/agronomy9030124
Chicago/Turabian StyleMoinet, Gabriel Y. K., Andrew J. Midwood, John E. Hunt, Cornelia Rumpel, Peter Millard, and Abad Chabbi. 2019. "Grassland Management Influences the Response of Soil Respiration to Drought" Agronomy 9, no. 3: 124. https://doi.org/10.3390/agronomy9030124
APA StyleMoinet, G. Y. K., Midwood, A. J., Hunt, J. E., Rumpel, C., Millard, P., & Chabbi, A. (2019). Grassland Management Influences the Response of Soil Respiration to Drought. Agronomy, 9(3), 124. https://doi.org/10.3390/agronomy9030124






