Modeling Carbon and Water Fluxes of Managed Grasslands: Comparing Flux Variability and Net Carbon Budgets between Grazed and Mowed Systems
Abstract
:1. Introduction
- test the ability of the CenW model to simulate water and CO2 flux dynamics of two temperate grassland ecosystems under mowing and grazing management, respectively;
- evaluate the model’s ability to capture the seasonal and interannual dynamics of CO2 and water fluxes in response to climate variability (five years) in interaction with two contrasting management practices (mowing and grazing); and
- determine the effects of mowing and grazing on eddy covariance fluxes and on the CO2 budget of managed grasslands.
2. Materials and Methods
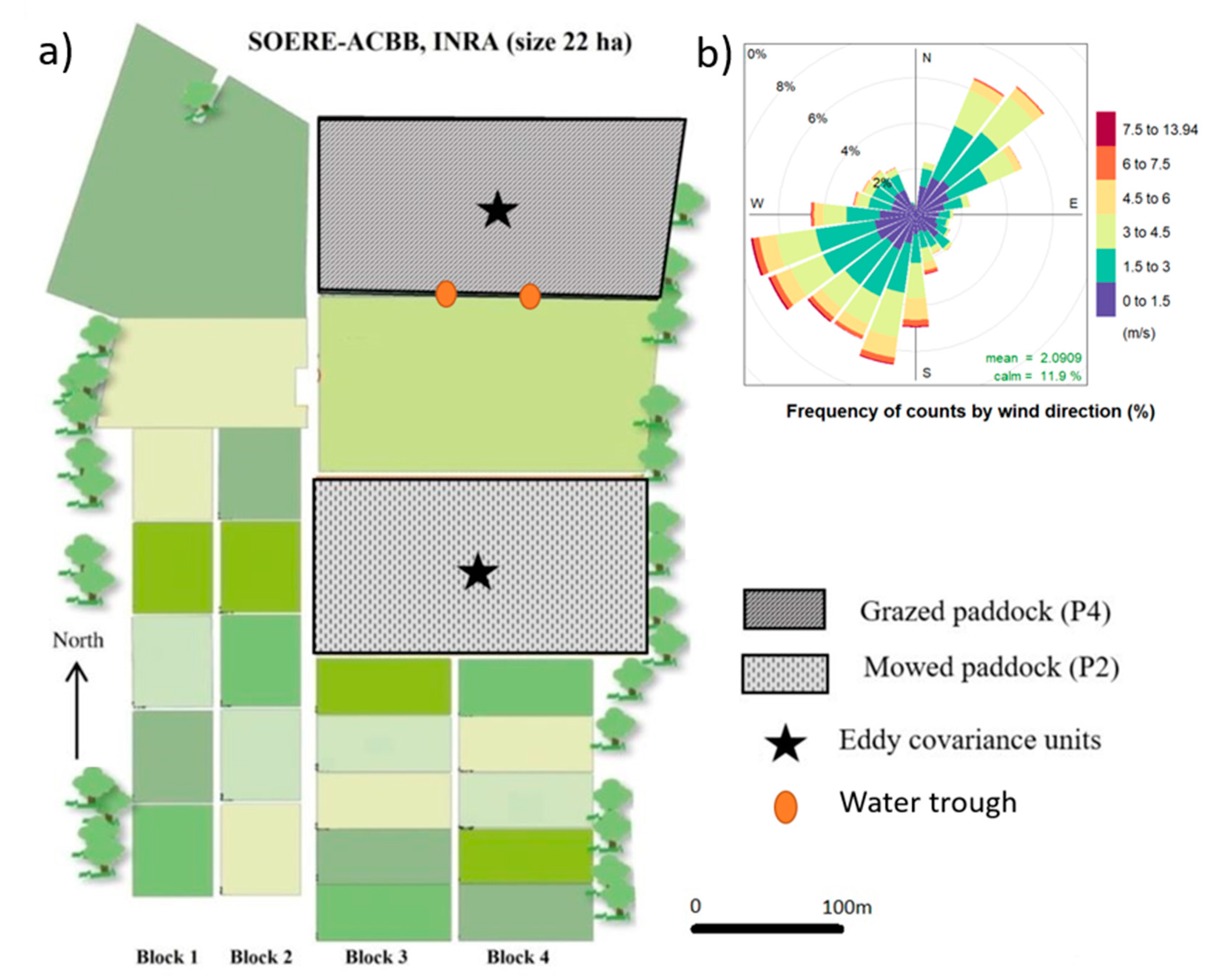
2.1. Experimental Details
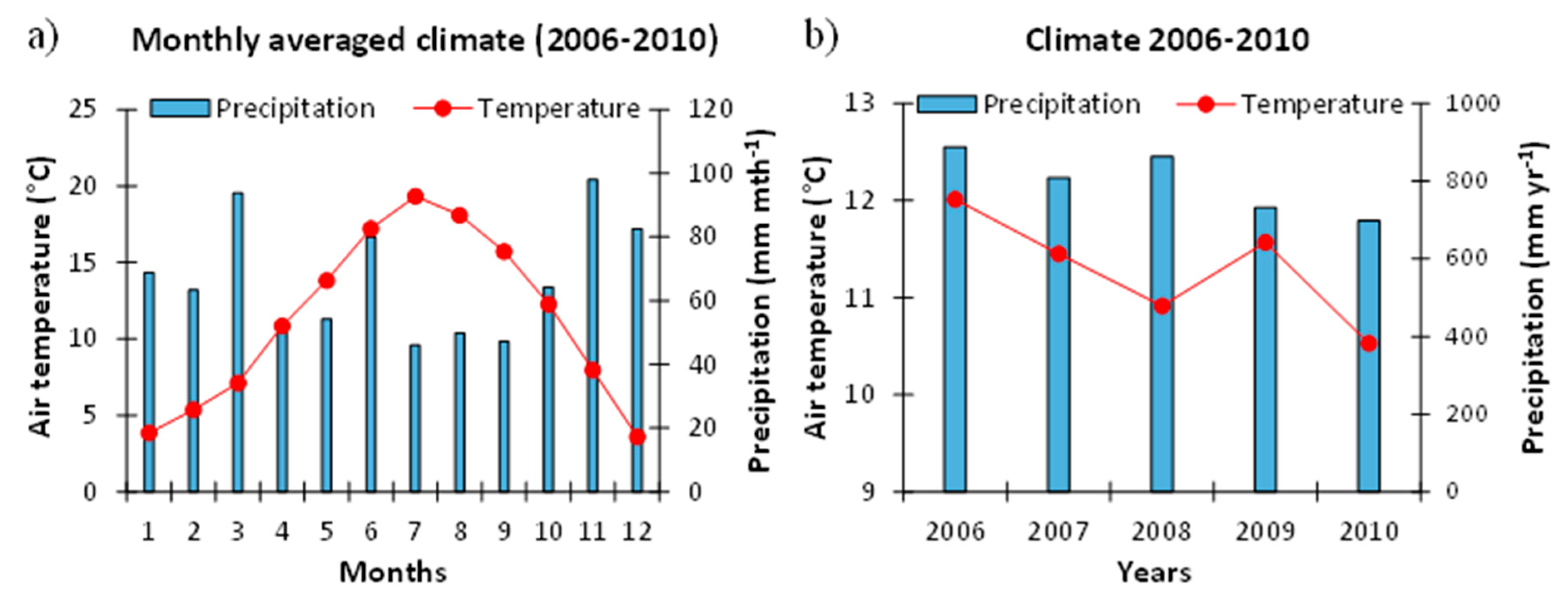
2.1.1. Meteorological Conditions at the Study Site
2.1.2. Eddy Covariance (EC) Measurements and Processing
- NEE values lower than −35 or higher than 25 µmol m−2 s−1
- NEE values higher than 3.5 µmol m−2 s−1 when PAR was above 400 µmol m−2 s−1
- NEE values lower than −2 µmol m−2 s−1 when PAR was below 25 µmol m−2 s−1
- Rn > 300 W m−2 and LE < 0 W m−2
- If precipitation > 0 mm
- If u* < 0.1 m s−1
- λE values higher than 750 or lower than −100 W m−2
- H values higher than 750 or lower than −100 W m−2
- Atmospheric CO2 concentration higher than 650 or lower than 320 ppm, respectively.
2.1.3. Vegetation and Soil Organic Carbon Measurements
2.2. Modeling Details
2.2.1. CenW 4.2 Overview
2.2.2. Model Parameterization and Statistical Analysis
3. Results
3.1. CenW Performances to Simulate Carbon Dioxide and Water Fluxes of Mown and Grazed Grasslands
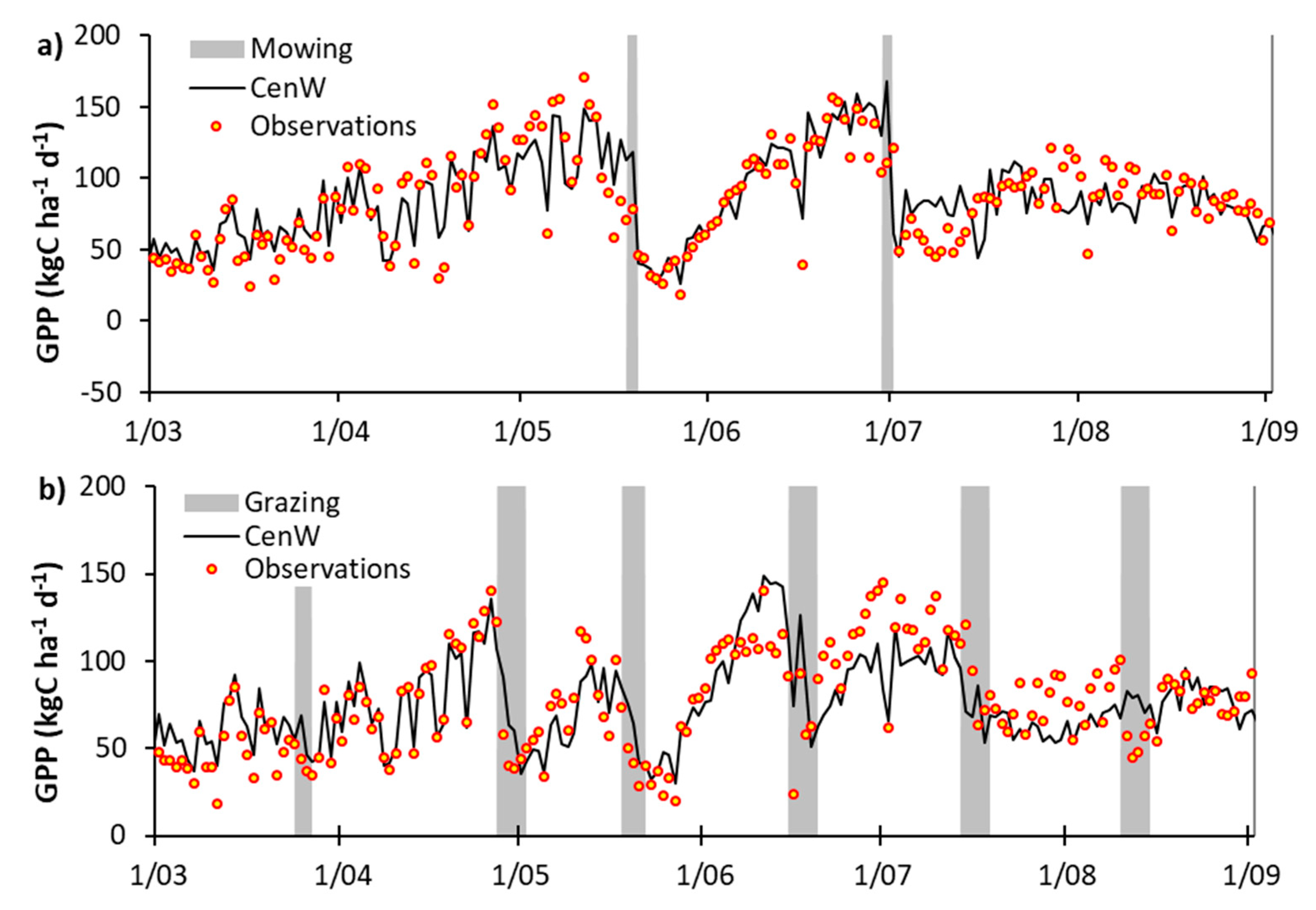
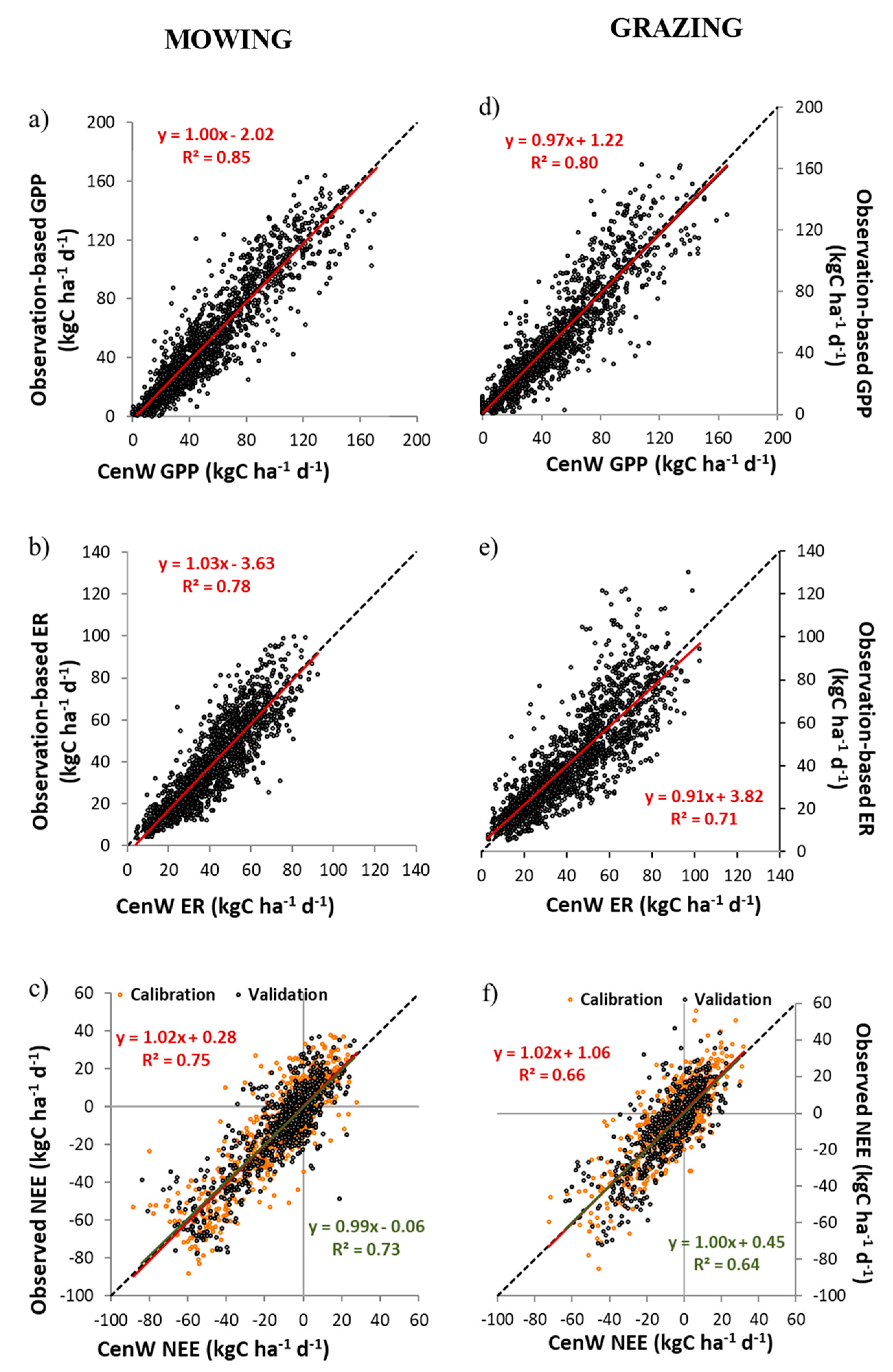
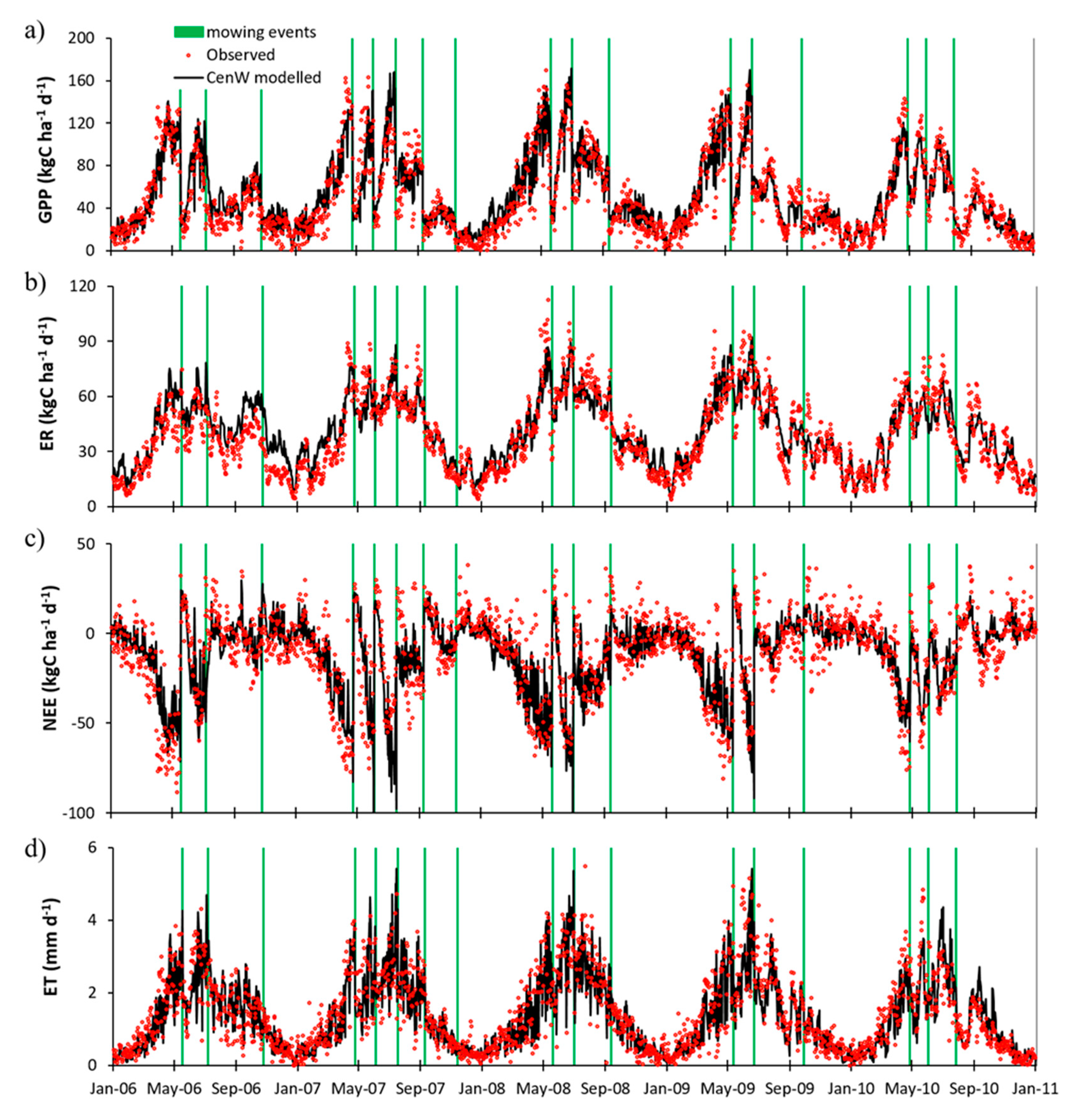
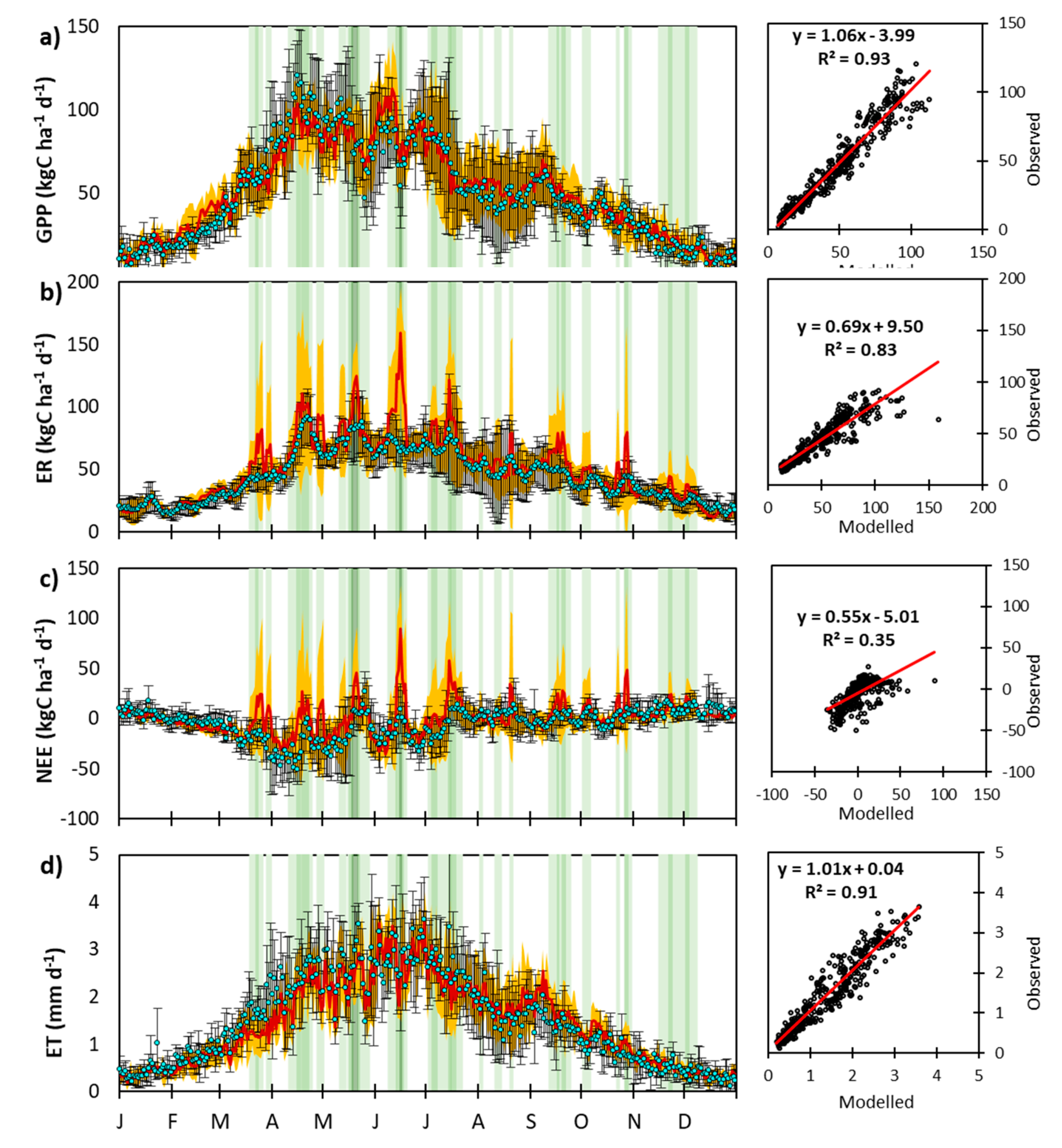
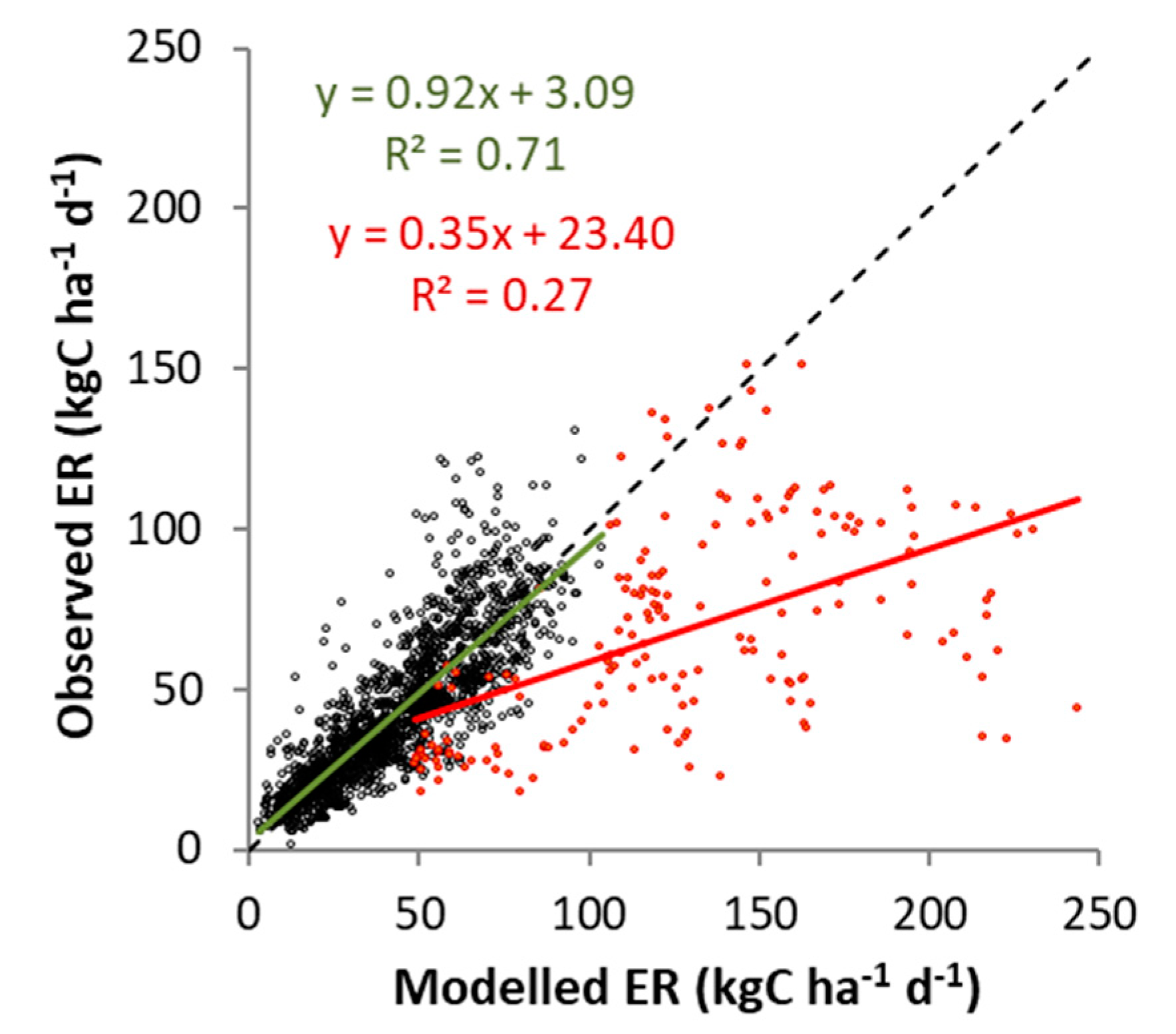
3.1.1. Carbon Dioxide Fluxes
- the calibration of the model with data that strongly depended on another simpler model (i.e., the Reichstein gap-filling and partitioning tool) and
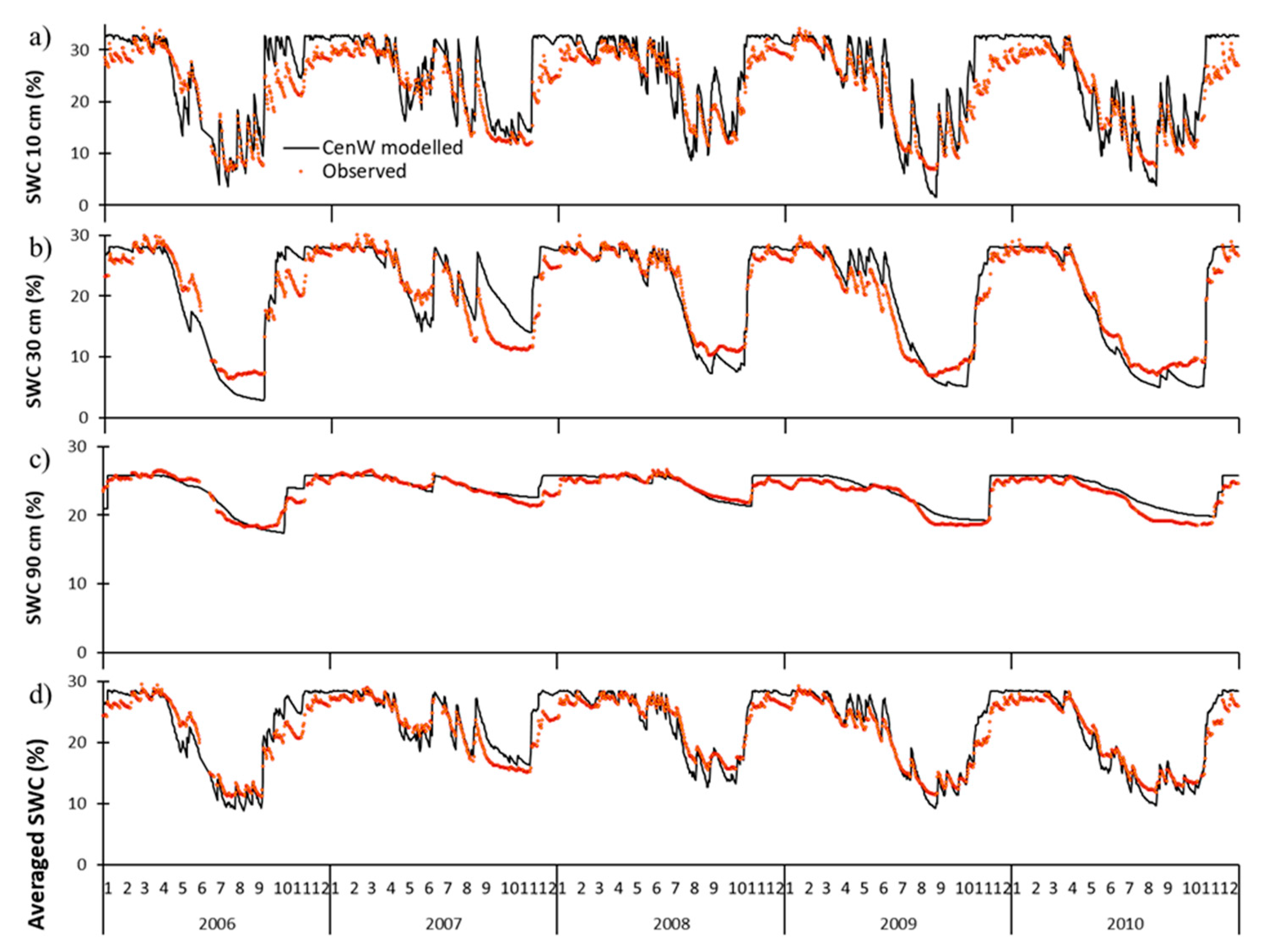
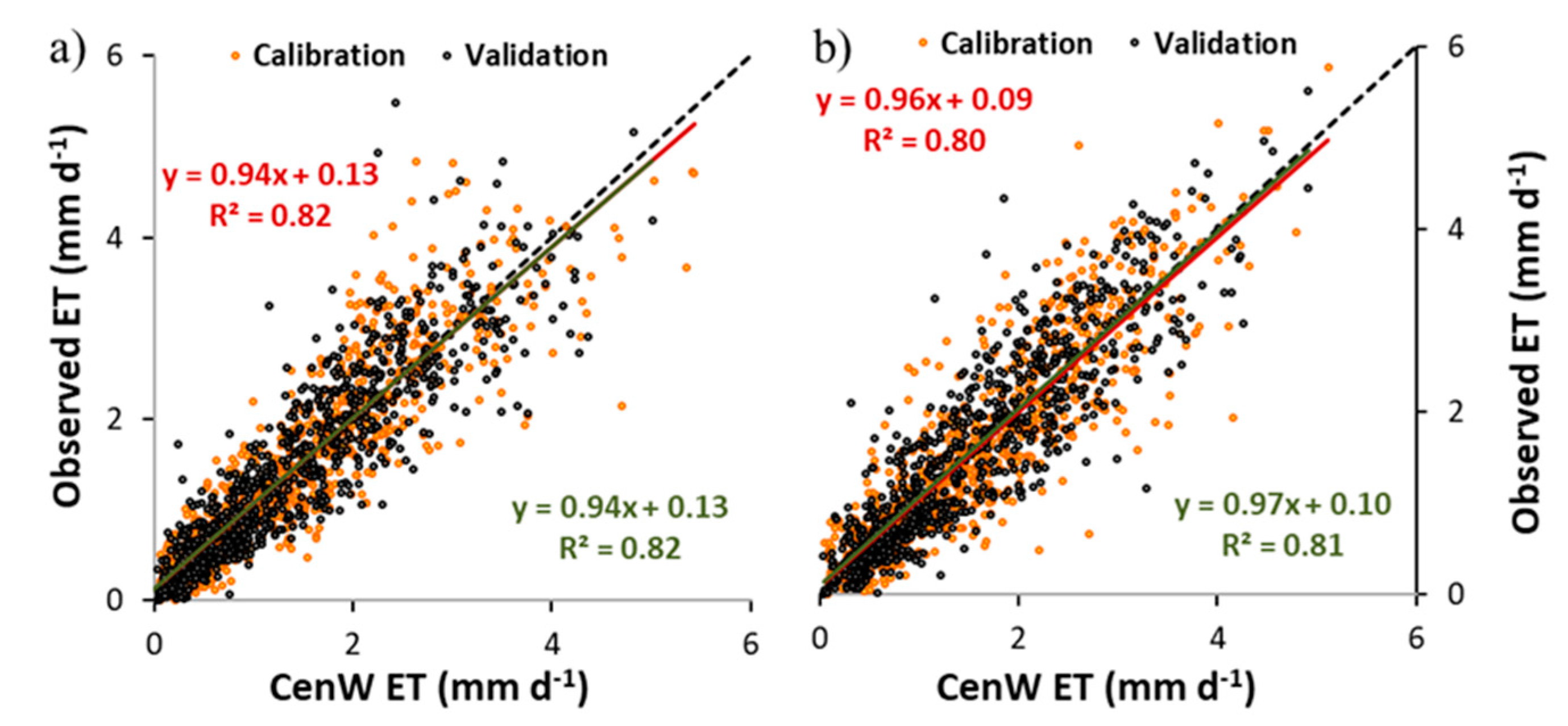
3.1.2. Soil Water Content and Evapotranspiration
3.2. Seasonal and Interannual Variabilities of Modeled and Observed Carbon Dioxide and Water Fluxes
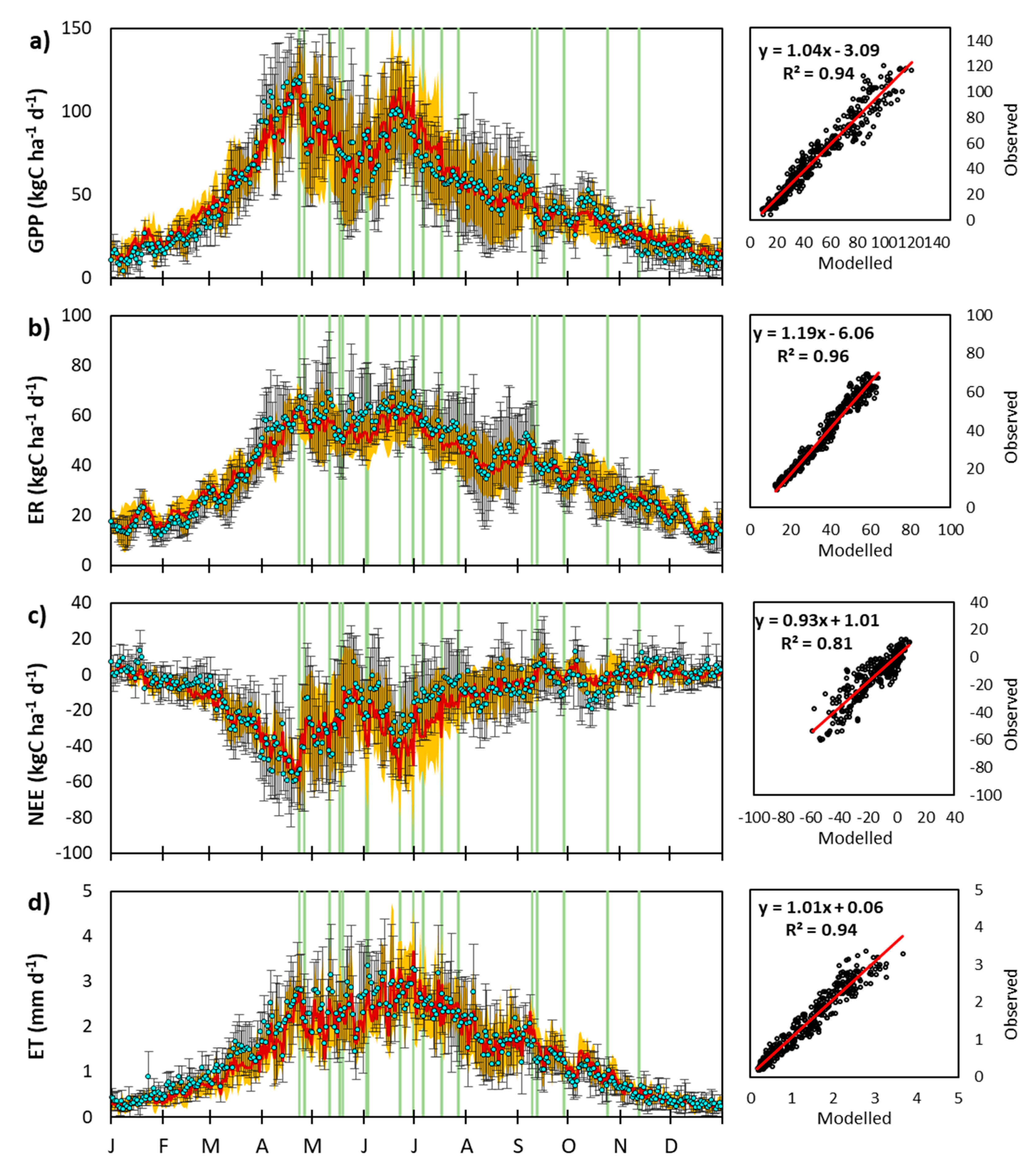
3.2.1. Day-to-Day and Seasonal CO2 and Water Fluxes Variability
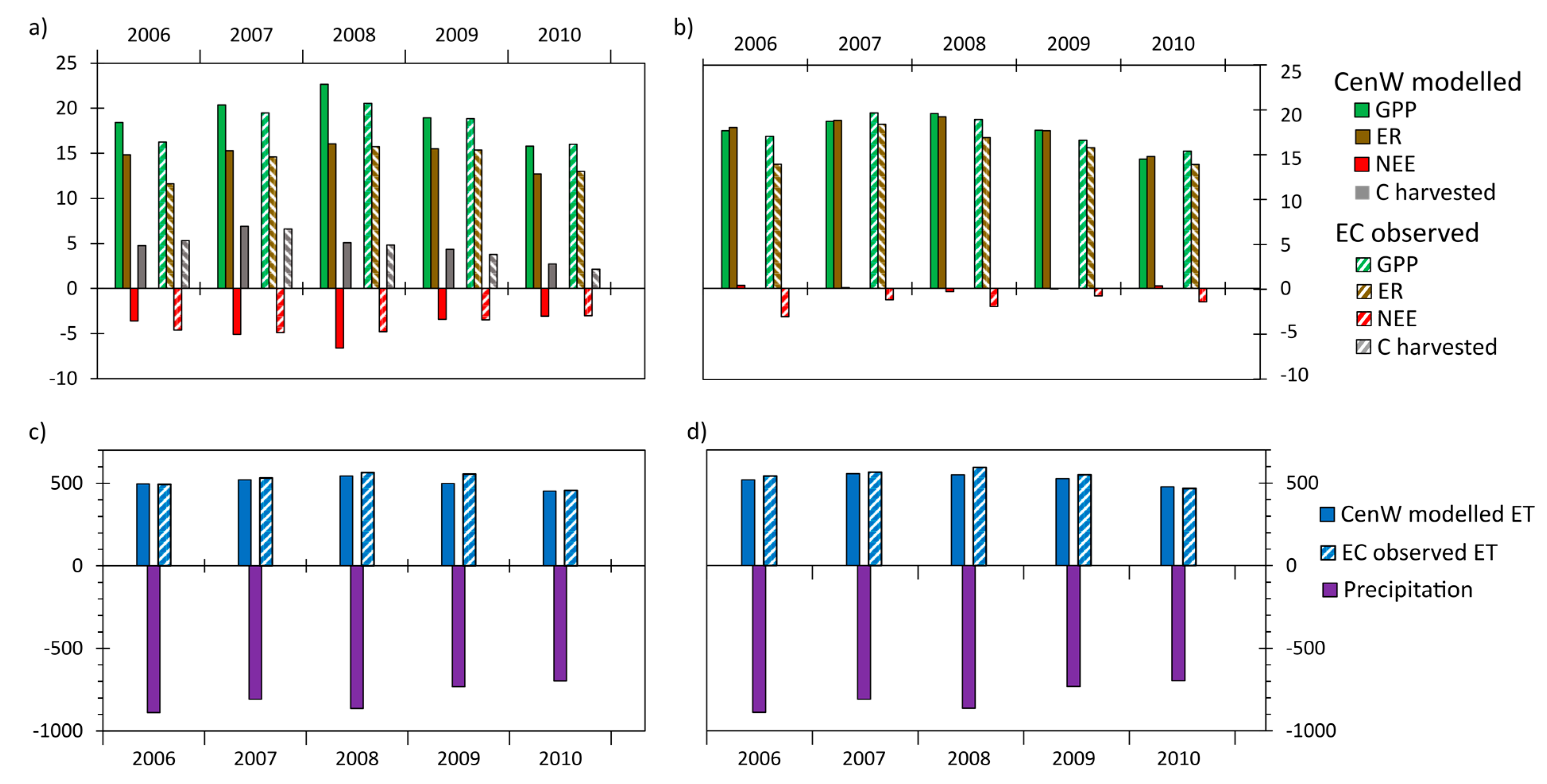
3.2.2. Interannual Variability of CenW Modeled and EC Measurements of CO2 and H2O Fluxes
Interannual Variations in Mean Daily Fluxes
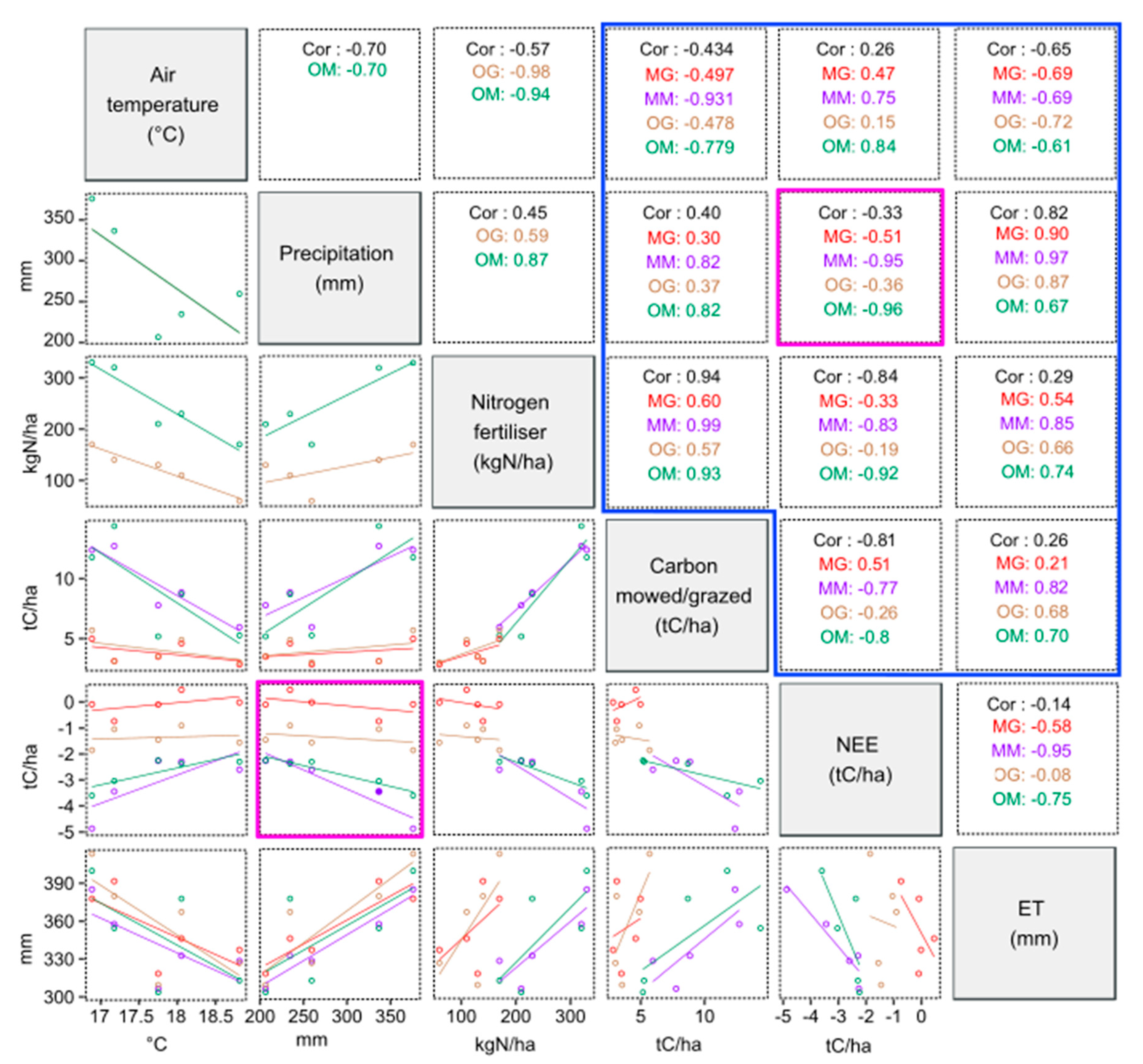
Variability of Annual CO2 and Water Fluxes
4. Discussion
4.1. Performances of the CenW Model to Simulate Gas Exchanges of Mowed and Grazed Pastures
4.2. Cow Respiration in Observed and Modeled CO2 Fluxes
4.3. Seasonal Variability of Observed and Modeled CO2 and Water Fluxes
5. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Management Records
| Mowed Paddock (P2) | Grazed Paddock (P4) | |||||||
|---|---|---|---|---|---|---|---|---|
| Year | Date of Mowing | Date of N Fertilizer Application | Amount (kgN ha−1) | Starting Date of Grazing Event | Length of Grazing Period (Day) | Stocking Rate (Head ha−1) | Date of N Fertilizer Application | Amount (kgN ha−1) |
| 2006 | 17-May | 26-Feb | 60 | 11-Apr | 7 | 16.8 | 5-Apr | 30 |
| 6-Jun | 24-May | 60 | 19-May | 10 | 16.8 | 24-May | 30 | |
| 24-Oct | 28-Sep | 50 | 3-Jul | 10 | 3.9 | |||
| 2-Oct | 4.5 | 17.1 | ||||||
| 16-Nov | 18 | 12.9 | ||||||
| 2007 | 23-Apr | 22-Feb | 80 | 19-Mar | 5 | 13.5 | 28-Mar | 50 |
| 5-Jun | 27-Apr | 60 | 16-Apr | 4 | 21.3 | 19-Jun | 30 | |
| 17-Jul | 12-Jun | 60 | 16-May | 7 | 19.4 | 19-Sep | 30 | |
| 10-Sep | 26-Jul | 60 | 14-Jun | 3 | 16.1 | 20-Sep | 30 | |
| 12-Nov | 19-Sep | 60 | 15-Jul | 8 | 10.6 | |||
| 20-Sep | 60 | 20-Aug | 2 | 11 | ||||
| 17-Sep | 4 | 17.4 | ||||||
| 22-Oct | 2 | 19.7 | ||||||
| 2008 | 19-May | 29-Jan | 120 | 25-Mar | 2 | 24.8 | 29-Jan | 30 |
| 30-Jun | 22-May | 90 | 28-Apr | 4 | 22.9 | 22-May | 30 | |
| 15-Sep | 15-Jul | 60 | 19-May | 2.4 | 20.6 | 28-Jul | 60 | |
| 17-Sep | 60 | 16-Jun | 4 | 20.6 | 17-Sep | 50 | ||
| 15-Jul | 4 | 16.8 | ||||||
| 11-Aug | 3.4 | 15 | ||||||
| 12-Sep | 6 | 18.7 | ||||||
| 27-Oct | 2 | 22.3 | ||||||
| 1-Dec | 8.25 | 4.5 | ||||||
| 2009 | 11-May | 17-Feb | 110 | 23-Mar | 2 | 25.2 | 17-Feb | 50 |
| 22-Jun | 19-May | 60 | 20-Apr | 4.5 | 24.5 | 19-May | 60 | |
| 28-Sep | 7-Oct | 60 | 11-May | 3.5 | 24.5 | |||
| 9-Jun | 8 | 19 | ||||||
| 13-Jul | 2.5 | 12.9 | ||||||
| 21-Sep | 4 | 17 | ||||||
| 27-Oct | 4 | 19.5 | ||||||
| 2010 | 26-Apr | 16-Mar | 90 | 29-Mar | 2.5 | 19.4 | 16-Mar | 60 |
| 2-Jun | 29-Apr | 70 | 19-Apr | 3.5 | 19.4 | 29-Apr | 50 | |
| 26-Jul | 8-Jun | 50 | 17-May | 5.5 | 19 | |||
| 14-Jun | 3.5 | 19 | ||||||
| 5-Jul | 2.5 | 14.8 | ||||||
| 2-Aug | 2 | 16.5 | ||||||
| 20-Sep | 1.5 | 15.2 | ||||||
| 22-Nov | 1.5 | 12.6 | ||||||
Appendix B. CenW Model Calibrated Parameters
| Parameter Description | Lusignan Mowed | Lusignan Grazed | Units | |
|---|---|---|---|---|
| Stand | Minimum foliage turn-over | 0.022 | 0.022 | yr−1 |
| Fine-root turn-over | 2.49 | 2.49 | yr−1 | |
| Low-light senescence limit | 0.056 | 0.08 | MJ m−2 d−1 | |
| Max daily low-light senescence | 0.015 | 0.017 | % d−1 | |
| Max drought foliage death rate | 6.08 | 6.76 | % d−1 | |
| Drought death of roots relative to foliage | 0.062 | 0.066 | – | |
| Mycorrhizal uptake | 0.01 | 0.01 | g kg−1 d−1 | |
| Soil water stress threshold (Wcrit) | 0.60 | 0.60 | – | |
| Respiration ratio per unit N | 0.18 | 0.44 | – | |
| beta parameter in T response of respiration | 1.98 | 1.96 | – | |
| Temperature for maximum respiration | 47 | 47 | °C | |
| Growth respiration | 0.29 | 0.32 | – | |
| Time constant for acclimation response of respiration | 364 | 247 | d | |
| Water-logging threshold (Llog) | 0.999 | 0.994 | – | |
| Water-logging sensitivity (sL) | 8.3 | 7.33 | – | |
| Ratio of [N] in senescing and live foliage | 0.99 | 0.99 | – | |
| Ratio of [N] in average foliage to leaves at the top | 0.83 | 0.78 | – | |
| Biological N fixation | 1.71 | 7.9 | gN kgC−1 | |
| Growth Km for carbon | 0.97 | 1.8 | % | |
| Growth Km for nitrogen | 1.94 | 3.7 | % | |
| Drop of standing dead leaves | 2.11 | 2.11 | % d−1 | |
| Decomposability of standing dead relative to metabolic litter | 0.7 | 0.7 | – | |
| photosynthesis | Specific leaf area | 17.5 | 19.3 | m2 (kg DW)−1 |
| Foliage albedo | 6.77 | 6.75 | % | |
| Transmissivity | 1.57 | 1.56 | % | |
| Loss as volatile organic carbon | 0 | 0 | % | |
| Threshold N concentrations (No) | 6.33 | 5.76 | gN (kg DW)−1 | |
| Non-limiting N concentration (Nsat) | 41.6 | 42.4 | gN (kg DW)−1 | |
| Light-saturated maximum photosynthetic rate (Amax) | 45.7 | 47.2 | µmol m−2 s−1 | |
| Maximum quantum yield | 0.06 | 0.06 | mol mol−1 | |
| Curvature in light response function | 0.412 | 0.412 | – | |
| Light extinction coefficient | 0.86 | 0.86 | – | |
| Ball–Berry stomatal parameter (unstressed) bb1 | 10.1 | 11.9 | – | |
| Ball–Berry stomatal parameter (stressed) bb2 | 8 | 8 | – | |
| Minimum temperature for photosynthesis (Tn) | -4.1 | -4.1 | °C | |
| Lower optimum temperature for photosynthesis (Topt, lower) | 25.8 | 25.8 | °C | |
| Upper optimum temperature for photosynthesis (Topt, upper) | 30.06 | 30.06 | °C | |
| Maximum temperature for photosynthesis (Tx) | 38.8 | 38.8 | °C | |
| Temperature damage sensitivity (sT) | 0.04 | 0.04 | – | |
| Threshold for frost damage | 0.19 | 0.19 | °C | |
| allocation | Allocation to reproductive organs | None | None | – |
| Fine root: foliage target ratio (nitrogen-unstressed) | 0.98 | 0.90 | – | |
| Fine root: foliage target ratio (nitrogen-stressed) | 3.6 | 4.6 | – | |
| Used target-oriented dynamic root-shoot allocation | Yes | Yes | – | |
| Fine root:foliage [N] ratio | 0.82 | 0.82 | – | |
| decomposition | Relative temperature dependence of heterotrophic respn | 0.49 | 0.75 | – |
| Foliar lignin concentration | 11.9 | 12 | % | |
| Root lignin concentration | 14.6 | 14.6 | % | |
| Organic matter transfer from surface to soil | 90 | 90 | % yr−1 | |
| Critical C:N ratio | 8.03 | 8 | – | |
| Ratio of C:N ratios in structural and metabolic pools | 4.83 | 4.09 | – | |
| Exponential term in lignin inhibition | 5 | 5 | – | |
| Water stress sens. of decomp. relative to plant processes | 0.68 | 1.03 | – | |
| Residual decomposition under dry conditions | 0.05 | 0.05 | – | |
| Mineral N immobilized | 5.32 | 5.38 | % d−1 | |
| site | Atmospheric N deposition | 2 | 2 | kgN ha−1 yr−1 |
| Volatilization fraction | 10.1 | 10.1 | % | |
| Leaching fraction | 0.46 | 0.46 | – | |
| Litter water-holding capacity | 2 | 2 | g gDW−1 | |
| Mulching effect of litter | 2.8 | 2.8 | % tDW−1 | |
| Canopy aerodynamic resistance | 83 | 78.7 | s m−1 | |
| Canopy rainfall interception | 0.044 | 0.044 | mm LAI−1 | |
| Maximum rate of soil evaporation | 1.55 | 1.25 | mm d−1 |
Appendix C. EC-Derived and Modeled GPP Time Series
References
- FAO and ITPS. Status of the World’s Soil Resources—Main Report; FAO and ITPS: Rome, Italy, 2015; ISBN 978-92-5-109004-6. [Google Scholar]
- FAOSTAT. Database Collection of the Food and Agriculture Organization of the United Nation. 2019. Available online: http://www.fao.org/faostat/en/#home (accessed on 4 January 2019).
- Scurlock, J.M.O.; Hall, D.O. The global carbon sink: A grassland perspective. Glob. Chang. Biol. 1998, 4, 229–233. [Google Scholar] [CrossRef]
- White, R.P.; Murray, S.; Rohweder, M. Pilot Analysis of Global Ecosystems: Grassland Ecosystems; World Resources Institute: Washington, DC, USA, 2000; ISBN 1-56973-461-5. [Google Scholar]
- Conant, R.T.; Paustian, K.; Elliott, E.T. Grassland management and conversion into grassland: Effects on soil carbon. Ecol. Appl. 2001, 11, 343–355. [Google Scholar] [CrossRef]
- Wang, W.; Fang, J. Soil respiration and human effects on global grasslands. Glob. Planet. Chang. 2009, 67, 20–28. [Google Scholar] [CrossRef]
- Herrero, M.; Henderson, B.; Havlík, P.; Thornton, P.K.; Conant, R.T.; Smith, P.; Wirsenius, S.; Hristov, A.N.; Gerber, P.; Gill, M.; et al. Greenhouse gas mitigation potentials in the livestock sector. Nat. Clim. Chang. 2016, 6, 452–461. [Google Scholar] [CrossRef]
- Soussana, J.F.; Allard, V.; Pilegaard, K.; Ambus, P.; Amman, C.; Campbell, C.; Ceschia, E.; Clifton-Brown, J.; Czobel, S.; Domingues, R.; et al. Full accounting of the greenhouse gas (CO2, N2O, CH4) budget of nine European grassland sites. Agric. Ecosyst. Environ. 2007, 121, 121–134. [Google Scholar] [CrossRef]
- Abberton, M.; Conant, R.; Batello, C. Grassland Carbon Sequestration: Management, Policy and Economics; Proceedings of the Workshop on the Role of Grassland Carbon Sequestration in the Mitigation of Climate Change; FAO: Rome, Italy, 2010; ISBN 978-92-5-106695-9. [Google Scholar]
- Reid, R.S.; Thornton, P.K.; McCrabb, G.J.; Kruska, R.L.; Atieno, F.; Jones, P.G. Is it possible to mitigate greenhouse gas emissions in pastoral ecosystems of the tropics? Environ. Dev. Sustain. 2004, 6, 91–109. [Google Scholar] [CrossRef]
- Allard, V.; Soussana, J.-F.; Falcimagne, R.; Berbigier, P.; Bonnefond, J.M.; Ceschia, E.; D’hour, P.; Hénault, C.; Laville, P.; Martin, C.; et al. The role of grazing management for the net biome productivity and greenhouse gas budget (CO2, N2O and CH4) of semi-natural grassland. Agric. Ecosyst. Environ. 2007, 121, 47–58. [Google Scholar] [CrossRef]
- Smith, P. Do grasslands act as a perpetual sink for carbon? Glob. Chang. Biol. 2014, 20, 2708–2711. [Google Scholar] [CrossRef] [PubMed]
- Olff, H.; Ritchie, M.E.; Prins, H.H.T. Global environmental controls of diversity in large herbivores. Nature 2002, 415, 901–904. [Google Scholar] [CrossRef]
- Jones, M.B.; Donnelly, A. Carbon sequestration in temperate grassland ecosystems and the influence of management, climate and elevated CO2. New Phytol. 2004, 164, 423–439. [Google Scholar] [CrossRef]
- Soussana, J.F.; Tallec, T.; Blanfort, V. Mitigating the greenhouse gas balance of ruminant production systems through carbon sequestration in grasslands. Animal 2010, 4, 334–350. [Google Scholar] [CrossRef]
- McSherry, M.E.; Ritchie, M.E. Effects of grazing on grassland soil carbon: A global review. Glob. Chang. Biol. 2013, 19, 1347–1357. [Google Scholar] [CrossRef]
- Senapati, N.; Jansson, P.-E.; Smith, P.; Chabbi, A. Modelling heat, water and carbon fluxes in mown grassland under multi-objective and multi-criteria constraints. Environ. Model. Softw. 2016, 80, 201–224. [Google Scholar] [CrossRef]
- Ammann, C.; Flechard, C.R.; Leifeld, J.; Neftel, A.; Fuhrer, J. The carbon budget of newly established temperate grassland depends on management intensity. Agric. Ecosyst. Environ. 2007, 121, 5–20. [Google Scholar] [CrossRef]
- Rumpel, C.; Crème, A.; Ngo, P.T.; Velásquez, G.; Mora, M.L.; Chabbi, A. The impact of grassland management on biogeochemical cycles involving carbon, nitrogen and phosphorus. J. Soil Sci. Plant Nutr. 2015, 15, 353–371. [Google Scholar] [CrossRef]
- Fetzel, T.; Havlik, P.; Herrero, M.; Erb, K.-H. Seasonality constraints to livestock grazing intensity. Glob. Chang. Biol. 2017, 23, 1636–1647. [Google Scholar] [CrossRef] [PubMed]
- Soussana, J.-F.; Loiseau, P.; Vuichard, N.; Ceschia, E.; Balesdent, J.; Chevallier, T.; Arrouays, D. Carbon cycling and sequestration opportunities in temperate grasslands. Soil Use Manag. 2004, 20, 219–230. [Google Scholar] [CrossRef]
- Jérôme, E.; Beckers, Y.; Bodson, B.; Heinesch, B.; Moureaux, C.; Aubinet, M. Impact of grazing on carbon dioxide exchanges in an intensively managed Belgian grassland. Agric. Ecosyst. Environ. 2014, 194, 7–16. [Google Scholar] [CrossRef]
- Oates, L.G.; Jackson, R.D. Livestock Management Strategy Affects Net Ecosystem Carbon Balance of Subhumid Pasture. Rangel. Ecol. Manag. 2014, 67, 19–29. [Google Scholar] [CrossRef]
- Dlamini, P.; Chivenge, P.; Chaplot, V. Overgrazing decreases soil organic carbon stocks the most under dry climates and low soil pH: A meta-analysis shows. Agric. Ecosyst. Environ. 2016, 221, 258–269. [Google Scholar] [CrossRef]
- Poeplau, C.; Marstorp, H.; Thored, K.; Kätterer, T. Effect of grassland cutting frequency on soil carbon storage—A case study on public lawns in three Swedish cities. SOIL Discuss. 2016, 2, 175–184. [Google Scholar] [CrossRef]
- Senapati, N.; Chabbi, A.; Gastal, F.; Smith, P.; Mascher, N.; Loubet, B.; Cellier, P.; Naisse, C. Net carbon storage measured in a mowed and grazed temperate sown grassland shows potential for carbon sequestration under grazed system. Carbon Manag. 2014, 5, 131–144. [Google Scholar] [CrossRef]
- Smith, P. How long before a change in soil organic carbon can be detected? Glob. Chang. Biol. 2004, 10, 1878–1883. [Google Scholar] [CrossRef]
- Allen, D.E.; Pringle, M.J.; Page, K.L.; Dalal, R.C. A review of sampling designs for the measurement of soil organic carbon in Australian grazing lands. Rangel. J. 2010, 32, 227–246. [Google Scholar] [CrossRef]
- Arrouays, D.; Marchant, B.P.; Saby, N.P.A.; Meersmans, J.; Orton, T.G.; Martin, M.P.; Bellamy, P.H.; Lark, R.M.; Kibblewhite, M. Generic Issues on Broad-Scale Soil Monitoring Schemes: A Review. Pedosphere 2012, 22, 456–469. [Google Scholar] [CrossRef]
- Osborne, B.; Saunders, M.; Walmsley, D.; Jones, M.; Smith, P. Key questions and uncertainties associated with the assessment of the cropland greenhouse gas balance. Agric. Ecosyst. Environ. 2010, 139, 293–301. [Google Scholar] [CrossRef]
- Mudge, P.L.; Wallace, D.F.; Rutledge, S.; Campbell, D.I.; Schipper, L.A.; Hosking, C.L. Carbon balance of an intensively grazed temperate pasture in two climatically contrasting years. Agric. Ecosyst. Environ. 2011, 144, 271–280. [Google Scholar] [CrossRef]
- Rutledge, S.; Mudge, P.L.; Campbell, D.I.; Woodward, S.L.; Goodrich, J.P.; Wall, A.M.; Kirschbaum, M.U.F.; Schipper, L.A. Carbon balance of an intensively grazed temperate dairy pasture over four years. Agric. Ecosyst. Environ. 2015, 206, 10–20. [Google Scholar] [CrossRef]
- Law, B.E.; Falge, E.; Gu, L.; Baldocchi, D.D.; Bakwin, P.; Berbigier, P.; Davis, K.; Dolman, A.J.; Falk, M.; Fuentes, J.D.; et al. Environmental controls over carbon dioxide and water vapor exchange of terrestrial vegetation. Agric. For. Meteorol. 2002, 113, 97–120. [Google Scholar] [CrossRef]
- Chen, Z.; Yu, G.; Ge, J.; Wang, Q.; Zhu, X.; Xu, Z. Roles of Climate, Vegetation and Soil in Regulating the Spatial Variations in Ecosystem Carbon Dioxide Fluxes in the Northern Hemisphere. PLoS ONE 2015, 10, e0125265. [Google Scholar] [CrossRef]
- Tian, H.; Lu, C.; Ciais, P.; Michalak, A.M.; Canadell, J.G.; Saikawa, E.; Huntzinger, D.N.; Gurney, K.R.; Sitch, S.; Zhang, B.; et al. The terrestrial biosphere as a net source of greenhouse gases to the atmosphere. Nature 2016, 531, 225–228. [Google Scholar] [CrossRef]
- Wohlfahrt, G. Modelling Fluxes and Concentrations of CO2, H2O and Sensible Heat Within and Above a Mountain Meadow Canopy: A Comparison of Three Lagrangian Models and Three Parameterisation Options for the Lagrangian Time Scale. Bound.-Layer Meteorol. 2004, 113, 43–80. [Google Scholar] [CrossRef]
- Jaksic, V.; Kiely, G.; Albertson, J.; Oren, R.; Katul, G.; Leahy, P.; Byrne, K.A. Net ecosystem exchange of grassland in contrasting wet and dry years. Agric. For. Meteorol. 2006, 139, 323–334. [Google Scholar] [CrossRef]
- Keenan, T.F.; Baker, I.; Barr, A.; Ciais, P.; Davis, K.; Dietze, M.; Dragoni, D.; Gough, C.M.; Grant, R.; Hollinger, D.; et al. Terrestrial biosphere model performance for inter-annual variability of land-atmosphere CO2 exchange. Glob. Chang. Biol. 2012, 18, 1971–1987. [Google Scholar] [CrossRef]
- Fischer, E.M.; Sedláček, J.; Hawkins, E.; Knutti, R. Models agree on forced response pattern of precipitation and temperature extremes. Geophys. Res. Lett. 2014, 41, 8554–8562. [Google Scholar] [CrossRef]
- Reyer, C. Forest Productivity Under Environmental Change—A Review of Stand-Scale Modeling Studies. Curr. For. Rep. 2015, 1, 53–68. [Google Scholar] [CrossRef]
- Baldocchi, D.D.; Wilson, K.B. Modeling CO2 and water vapor exchange of a temperate broadleaved forest across hourly to decadal time scales. Ecol. Model. 2001, 142, 155–184. [Google Scholar] [CrossRef]
- Baldocchi, D.D. Assessing the eddy covariance technique for evaluating carbon dioxide exchange rates of ecosystems: Past, present and future. Glob. Chang. Biol. 2003, 9, 479–492. [Google Scholar] [CrossRef]
- Baldocchi, D. Measuring and modelling carbon dioxide and water vapour exchange over a temperate broad-leaved forest during the 1995 summer drought. Plant Cell Environ. 1997, 20, 1108–1122. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhuang, Q. Parameterization and sensitivity analysis of a process-based terrestrial ecosystem model using adjoint method. J. Adv. Model. Earth Syst. 2014, 6, 315–331. [Google Scholar] [CrossRef]
- Walker, W.E.; Harremoës, P.; Rotmans, J.; van der Sluijs, J.P.; van Asselt, M.B.A.; Janssen, P.; Krayer von Kraus, M.P. Defining Uncertainty: A Conceptual Basis for Uncertainty Management in Model-Based Decision Support. Integr. Assess. 2003, 4. [Google Scholar] [CrossRef]
- Braswell, B.H.; Sacks, W.J.; Linder, E.; Schimel, D.S. Estimating diurnal to annual ecosystem parameters by synthesis of a carbon flux model with eddy covariance net ecosystem exchange observations. Glob. Chang. Biol. 2005, 11, 335–355. [Google Scholar] [CrossRef]
- Stoy, P.C.; Katul, G.G.; Siqueira, M.B.S.; Juang, J.-Y.; McCarthy, H.R.; Kim, H.-S.; Oishi, A.C.; Oren, R. Variability in net ecosystem exchange from hourly to inter-annual time scales at adjacent pine and hardwood forests: A wavelet analysis. Tree Physiol. 2005, 25, 887–902. [Google Scholar] [CrossRef]
- Siqueira, M.B.; Katul, G.G.; Sampson, D.A.; Stoy, P.C.; Juang, J.-Y.; Mccarthy, H.R.; Oren, R. Multiscale model intercomparisons of CO2 and H2O exchange rates in a maturing southeastern US pine forest. Glob. Chang. Biol. 2006, 12, 1189–1207. [Google Scholar] [CrossRef]
- Hollinger, D.; Richardson, A. Uncertainty in eddy covariance measurements and its application to physiological models. Tree Physiol. 2005, 25, 873–885. [Google Scholar] [CrossRef]
- Parsons, A.J.; Schwinning, S.; Carrère, P. Plant growth functions and possible spatial and temporal scaling errors in models of herbivory. Grass Forage Sci. 2001, 56, 21–34. [Google Scholar] [CrossRef]
- Johnson, I.R.; Chapman, D.F.; Snow, V.; Eckard, R.; Parsons, A.; Lambert, M.G.; Cullen, B. DairyMod and EcoMod: Biophysical pasture-simulation models for Australia and New Zealand. Aust. J. Exp. Agric. 2008, 48, 621–631. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F. CenW, a forest growth model with linked carbon, energy, nutrient and water cycles. Ecol. Model. 1999, 118, 17–59. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F.; Keith, H.; Leuning, R.; Cleugh, H.A.; Jacobsen, K.L.; van Gorsel, E.; Raison, R.J. Modelling net ecosystem carbon and water exchange of a temperate Eucalyptus delegatensis forest using multiple constraints. Agric. For. Meteorol. 2007, 145, 48–68. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F.; Watt, M.S. Use of a process-based model to describe spatial variation in Pinus radiata productivity in New Zealand. For. Ecol. Manag. 2011, 262, 1008–1019. [Google Scholar] [CrossRef]
- Parton, W.J.; Schimel, D.S.; Cole, C.V.; Ojima, D.S. Analysis of Factors Controlling Soil Organic Matter Levels in Great Plains Grasslands1. Soil Sci. Soc. Am. J. 1987, 51, 1173–1179. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F.; Rutledge, S.; Kuijper, I.A.; Mudge, P.L.; Puche, N.; Wall, A.M.; Roach, C.G.; Schipper, L.A.; Campbell, D.I. Modelling carbon and water exchange of a grazed pasture in New Zealand constrained by eddy covariance measurements. Sci. Total Environ. 2015, 512–513, 273–286. [Google Scholar] [CrossRef]
- Kirschbaum, M.; Schipper, L.; Mudge, P.; Rutledge-Jonker, S.; Puche, N.J.; Campbell, D. The trade-offs between milk production and soil organic carbon storage in dairy systems under different management and environmental factors. Sci. Total Environ. 2016, 577, 61–72. [Google Scholar] [CrossRef]
- Moni, C.; Chabbi, A.; Nunan, N.; Rumpel, C.; Chenu, C. Spatial dependance of organic carbon–metal relationships: A multi-scale statistical analysis, from horizon to field. Geoderma 2010, 158, 120–127. [Google Scholar] [CrossRef]
- Chabbi, A.; Kögel-Knabner, I.; Rumpel, C. Stabilised carbon in subsoil horizons is located in spatially distinct parts of the soil profile. Soil Biol. Biochem. 2009, 41, 256–261. [Google Scholar] [CrossRef]
- Kunrath, T.R.; de Berranger, C.; Charrier, X.; Gastal, F.; de Faccio Carvalho, P.C.; Lemaire, G.; Emile, J.-C.; Durand, J.-L. How much do sod-based rotations reduce nitrate leaching in a cereal cropping system? Agric. Water Manag. 2015, 150, 46–56. [Google Scholar] [CrossRef]
- Senapati, N.; Chabbi, A.; Smith, P. Modelling daily to seasonal carbon fluxes and annual net ecosystem carbon balance of cereal grain-cropland using DailyDayCent: A model data comparison. Agric. Ecosyst. Environ. 2018, 252, 159–177. [Google Scholar] [CrossRef]
- Fuchs, M.; Tanner, C.B. Calibration and Field Test of Soil Heat Flux Plates 1. Soil Sci. Soc. Am. J. 1968, 32, 326–328. [Google Scholar] [CrossRef]
- Aubinet, M.; Grelle, A.; Ibrom, A.; Rannik, Ü.; Moncrieff, J.; Foken, T.; Kowalski, A.S.; Martin, P.; Berbigier, P.; Bernhofer, C.; et al. Estimates of the Annual Net Carbon and Water Exchange of Forests: The EUROFLUX Methodology. Adv. Ecol. Res. 2000, 30, 113–175. [Google Scholar]
- Ferrara, R.M.; Loubet, B.; Di Tommasi, P.; Bertolini, T.; Magliulo, V.; Cellier, P.; Eugster, W.; Rana, G. Eddy covariance measurement of ammonia fluxes: Comparison of high frequency correction methodologies. Agric. For. Meteorol. 2012, 158–159, 30–42. [Google Scholar] [CrossRef]
- Webb, E.K.; Pearman, G.I.; Leuning, L. Correction of Flux Measurements for Density Effects Due to Heat and Water-Vapor Transfer. Quart. J. R. Meteorol. Soc. 1980, 106, 85–100. [Google Scholar] [CrossRef]
- Burba, G. Eddy Covariance Method for Scientific, Industrial, Agricultural and Regulatory Applications: A Field Book on Measuring Ecosystem Gas Exchange and Areal Emission Rates; LI-COR Biosciences: Lincoln, NE, USA, 2013; ISBN 978-0-615-76827-4. [Google Scholar]
- Falge, E.; Baldocchi, D.; Olson, R.; Anthoni, P.; Aubinet, M.; Bernhofer, C.; Burba, G.; Ceulemans, R.; Clement, R.; Dolman, H.; et al. Gap filling strategies for long term energy flux data sets. Agric. For. Meteorol. 2001, 107, 71–77. [Google Scholar] [CrossRef]
- Falge, E.; Baldocchi, D.; Olson, R.; Anthoni, P.; Aubinet, M.; Bernhofer, C.; Burba, G.; Ceulemans, R.; Clement, R.; Dolman, H.; et al. Gap filling strategies for defensible annual sums of net ecosystem exchange. Agric. For. Meteorol. 2001, 107, 43–69. [Google Scholar] [CrossRef]
- Reichstein, M.; Falge, E.; Baldocchi, D.; Papale, D.; Aubinet, M.; Berbigier, P.; Bernhofer, C.; Buchmann, N.; Gilmanov, T.; Granier, A.; et al. On the separation of net ecosystem exchange into assimilation and ecosystem respiration: Review and improved algorithm. Glob. Chang. Biol. 2005, 11, 1424–1439. [Google Scholar] [CrossRef]
- Moffat, A.M.; Papale, D.; Reichstein, M.; Hollinger, D.Y.; Richardson, A.D.; Barr, A.G.; Beckstein, C.; Braswell, B.H.; Churkina, G.; Desai, A.R.; et al. Comprehensive comparison of gap-filling techniques for eddy covariance net carbon fluxes. Agric. For. Meteorol. 2007, 147, 209–232. [Google Scholar] [CrossRef]
- Lloyd, J.; Taylor, J.A. On the Temperature Dependence of Soil Respiration. Funct. Ecol. 1994, 8, 315–323. [Google Scholar] [CrossRef]
- Sands, P. Modelling Canopy Production. I. Optimal Distribution of Photosynthetic Resources. Funct. Plant Biol. 1995, 22, 593–601. [Google Scholar] [CrossRef]
- Baisden, W.T.; Amundson, R.; Brenner, D.L.; Cook, A.C.; Kendall, C.; Harden, J.W. A multiisotope C and N modeling analysis of soil organic matter turnover and transport as a function of soil depth in a California annual grassland soil chronosequence. Glob. Biogeochem. Cycles 2002, 16, 82-1–82-26. [Google Scholar] [CrossRef]
- Pal, P.; Clough, T.; Kelliher, F.; van Koten, C.; Sherlock, R. Intensive Cattle Grazing Affects Pasture Litter-Fall: An Unrecognized Nitrous Oxide Source. J. Environ. Qual. 2012, 41, 444–448. [Google Scholar] [CrossRef]
- Zeeman, M.J.; Hiller, R.; Gilgen, A.K.; Michna, P.; Plüss, P.; Buchmann, N.; Eugster, W. Management and climate impacts on net CO2 fluxes and carbon budgets of three grasslands along an elevational gradient in Switzerland. Agric. For. Meteorol. 2010, 150, 519–530. [Google Scholar] [CrossRef]
- Kelliher, F.M.; Clark, H. Chapter 9: Ruminants. In Methane and Climate Change; Reay, D., Smith, P., van Amstel, A., Eds.; Earthscan: London, UK, 2010; pp. 136–150. [Google Scholar]
- Crush, J.R.; Waghorn, G.C.; Rolston, M.P. Greenhouse gas emissions from pasture and arable crops grown on a Kairanga soil in the Manawatu, North Island, New Zealand. N. Z. J. Agric. Res. 1992, 35, 253–257. [Google Scholar] [CrossRef]
- Nash, J.E.; Sutcliffe, J.V. River flow forecasting through conceptual models part I—A discussion of principles. J. Hydrol. 1970, 10, 282–290. [Google Scholar] [CrossRef]
- Felber, R.; Neftel, A.; Ammann, C. Discerning the cows from the pasture: Quantifying and partitioning the NEE of a grazed pasture using animal position data. Agric. For. Meteorol. 2016, 216, 37–47. [Google Scholar] [CrossRef]
- Huntzinger, D.N.; Post, W.M.; Wei, Y.; Michalak, A.M.; West, T.O.; Jacobson, A.R.; Baker, I.T.; Chen, J.M.; Davis, K.J.; Hayes, D.J.; et al. North American Carbon Program (NACP) regional interim synthesis: Terrestrial biospheric model intercomparison. Ecol. Model. 2012, 232, 144–157. [Google Scholar] [CrossRef]
- Warszawski, L.; Friend, A.; Ostberg, S.; Frieler, K.; Lucht, W.; Schaphoff, S.; Beerling, D.; Cadule, P.; Ciais, P.; Clark, D.B.; et al. A multi-model analysis of risk of ecosystem shifts under climate change. Environ. Res. Lett. 2013, 8, 044018. [Google Scholar] [CrossRef]
- Chang, X.; Bao, X.; Wang, S.; Wilkes, A.; Erdenetsetseg, B.; Baival, B.; Avaadorj, D.; Maisaikhan, T.; Damdinsuren, B. Simulating effects of grazing on soil organic carbon stocks in Mongolian grasslands. Agric. Ecosyst. Environ. 2015, 212, 278–284. [Google Scholar] [CrossRef]
- White, T.A.; Johnson, I.R.; Snow, V.O. Comparison of outputs of a biophysical simulation model for pasture growth and composition with measured data under dryland and irrigated conditions in New Zealand. Grass Forage Sci. 2008, 63, 339–349. [Google Scholar] [CrossRef]
- Graux, A.-I.; Bellocchi, G.; Lardy, R.; Soussana, J.-F. Ensemble modelling of climate change risks and opportunities for managed grasslands in France. Agric. For. Meteorol. 2013, 170, 114–131. [Google Scholar] [CrossRef]
- Ben Touhami, H.; Bellocchi, G. Bayesian calibration of the Pasture Simulation model (PaSim) to simulate European grasslands under water stress. Ecol. Inform. 2015, 30, 356–364. [Google Scholar] [CrossRef]
- Ehrhardt, F.; Soussana, J.-F.; Bellocchi, G.; Grace, P.; McAuliffe, R.; Recous, S.; Sándor, R.; Smith, P.; Snow, V.; de Antoni Migliorati, M.; et al. Assessing uncertainties in crop and pasture ensemble model simulations of productivity and N2O emissions. Glob. Chang. Biol. 2018, 24, 603–616. [Google Scholar] [CrossRef]
- Baldocchi, D. “Breathing” of the terrestrial biosphere: Lessons learned from a global network of carbon dioxide flux measurement systems. Aust. J. Bot. 2008, 56, 1–26. [Google Scholar] [CrossRef]
- Wohlfahrt, G.; Hammerle, A.; Haslwanter, A.; Bahn, M.; Tappeiner, U.; Cernusca, A. Seasonal and inter-annual variability of the net ecosystem CO2 exchange of a temperate mountain grassland: Effects of weather and management. J. Geophys. Res. 2008, 113, 1–14. [Google Scholar] [CrossRef]
- Yan, L.; Chen, S.; Huang, J.; Lin, G. Water regulated effects of photosynthetic substrate supply on soil respiration in a semiarid steppe. Glob. Chang. Biol. 2011, 17, 1990–2001. [Google Scholar] [CrossRef]
- Cleverly, J.; Boulain, N.; Villalobos-Vega, R.; Grant, N.; Faux, R.; Wood, C.; Cook, P.G.; Yu, Q.; Leigh, A.; Eamus, D. Dynamics of component carbon fluxes in a semi-arid Acacia woodland, central Australia. J. Geophys. Res. Biogeosci. 2013, 118, 1168–1185. [Google Scholar] [CrossRef]
- Eamus, D.; Cleverly, J.; Boulain, N.; Grant, N.; Faux, R.; Villalobos-Vega, R. Carbon and water fluxes in an arid-zone Acacia savanna woodland: An analyses of seasonal patterns and responses to rainfall events. Agric. For. Meteorol. 2013, 182–183, 225–238. [Google Scholar] [CrossRef]
- Rogiers, N.; Eugster, W.; Furger, M.; Siegwolf, R. Effect of land management on ecosystem carbon fluxes at a subalpine grassland site in the Swiss Alps. Theor. Appl. Climatol. 2005, 80, 187–203. [Google Scholar] [CrossRef]
- Peichl, M.; Carton, O.; Kiely, G. Management and climate effects on carbon dioxide and energy exchanges in a maritime grassland. Agric. Ecosyst. Environ. 2012, 158, 132–146. [Google Scholar] [CrossRef]
- Felber, R.; Bretscher, D.; Münger, A.; Neftel, A.; Ammann, C. Determination of the carbon budget of a pasture: Effect of system boundaries and flux uncertainties. Biogeosciences 2016, 13, 2959–2969. [Google Scholar] [CrossRef]
- Gourlez de la Motte, L.; Mamadou, O.; Beckers, Y.; Bodson, B.; Heinesch, B.; Aubinet, M. Rotational and continuous grazing does not affect the total net ecosystem exchange of a pasture grazed by cattle but modifies CO2 exchange dynamics. Agric. Ecosyst. Environ. 2018, 253, 157–165. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, Z.; Wang, S.; Hu, Y.; Xu, G.; Luo, C.; Chang, X.; Duan, J.; Lin, Q.; Xu, B.; et al. Response of ecosystem respiration to warming and grazing during the growing seasons in the alpine meadow on the Tibetan plateau. Agric. For. Meteorol. 2011, 151, 792–802. [Google Scholar] [CrossRef]
- Skinner, R.H. High Biomass Removal Limits Carbon Sequestration Potential of Mature Temperate Pastures. J. Environ. Qual. 2008, 37, 1319–1326. [Google Scholar] [CrossRef]
- Falge, E.; Baldocchi, D.; Tenhunen, J.; Aubinet, M.; Bakwin, P.; Berbigier, P.; Bernhofer, C.; Burba, G.; Clement, R.; Davis, K.J.; et al. Seasonality of ecosystem respiration and gross primary production as derived from FLUXNET measurements. Agric. For. Meteorol. 2002, 113, 53–74. [Google Scholar] [CrossRef]
- Falge, E.; Tenhunen, J.; Baldocchi, D.; Aubinet, M.; Bakwin, P.; Berbigier, P.; Bernhofer, C.; Bonnefond, J.-M.; Burba, G.; Clement, R.; et al. Phase and amplitude of ecosystem carbon release and uptake potentials as derived from FLUXNET measurements. Agric. For. Meteorol. 2002, 113, 75–95. [Google Scholar] [CrossRef]
- Nieveen, J.P.; Campbell, D.I.; Schipper, L.A.; Blair, I.J. Carbon exchange of grazed pasture on a drained peat soil. Glob. Chang. Biol. 2005, 11, 607–618. [Google Scholar] [CrossRef]
- Richardson, A.D.; Hollinger, D.Y.; Burba, G.G.; Davis, K.J.; Flanagan, L.B.; Katul, G.G.; William Munger, J.; Ricciuto, D.M.; Stoy, P.C.; Suyker, A.E.; et al. A multi-site analysis of random error in tower-based measurements of carbon and energy fluxes. Agric. For. Meteorol. 2006, 136, 1–18. [Google Scholar] [CrossRef]

| Mowed Paddock | Grazed Paddock | |||||||
|---|---|---|---|---|---|---|---|---|
| Daily | Weekly | Daily | Weekly | |||||
| Calibration | Validation | Total | Total | Calibration | Validation | Total | Total | |
| GPP | - | 0.85 | 0.85 | 0.87 | - | 0.80 | 0.80 | 0.79 |
| ER | - | 0.77 | 0.77 | 0.78 | - | 0.72 | 0.72 | 0.67 |
| NEE | 0.75 | 0.73 | 0.74 | 0.72 | 0.64 | 0.66 | 0.65 | 0.64 |
| ET | 0.82 | 0.81 | 0.82 | 0.87 | 0.81 | 0.80 | 0.80 | 0.85 |
| Averaged SWC | - | 0.85 | 0.85 | 0.87 | NA | NA | NA | NA |
| Harvested biomass | - | 0.80 | - | NA | NA | NA | NA | NA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puche, N.; Senapati, N.; Flechard, C.R.; Klumpp, K.; Kirschbaum, M.U.F.; Chabbi, A. Modeling Carbon and Water Fluxes of Managed Grasslands: Comparing Flux Variability and Net Carbon Budgets between Grazed and Mowed Systems. Agronomy 2019, 9, 183. https://doi.org/10.3390/agronomy9040183
Puche N, Senapati N, Flechard CR, Klumpp K, Kirschbaum MUF, Chabbi A. Modeling Carbon and Water Fluxes of Managed Grasslands: Comparing Flux Variability and Net Carbon Budgets between Grazed and Mowed Systems. Agronomy. 2019; 9(4):183. https://doi.org/10.3390/agronomy9040183
Chicago/Turabian StylePuche, Nicolas, Nimai Senapati, Christophe R. Flechard, Katia Klumpp, Miko U.F. Kirschbaum, and Abad Chabbi. 2019. "Modeling Carbon and Water Fluxes of Managed Grasslands: Comparing Flux Variability and Net Carbon Budgets between Grazed and Mowed Systems" Agronomy 9, no. 4: 183. https://doi.org/10.3390/agronomy9040183
APA StylePuche, N., Senapati, N., Flechard, C. R., Klumpp, K., Kirschbaum, M. U. F., & Chabbi, A. (2019). Modeling Carbon and Water Fluxes of Managed Grasslands: Comparing Flux Variability and Net Carbon Budgets between Grazed and Mowed Systems. Agronomy, 9(4), 183. https://doi.org/10.3390/agronomy9040183







